Use Microfluidic Chips to Study the Phototaxis of Lung Cancer Cells
Abstract
1. Introduction
2. Results and Discussion
2.1. Calculation and Simulation of Concentrations and Absorbance
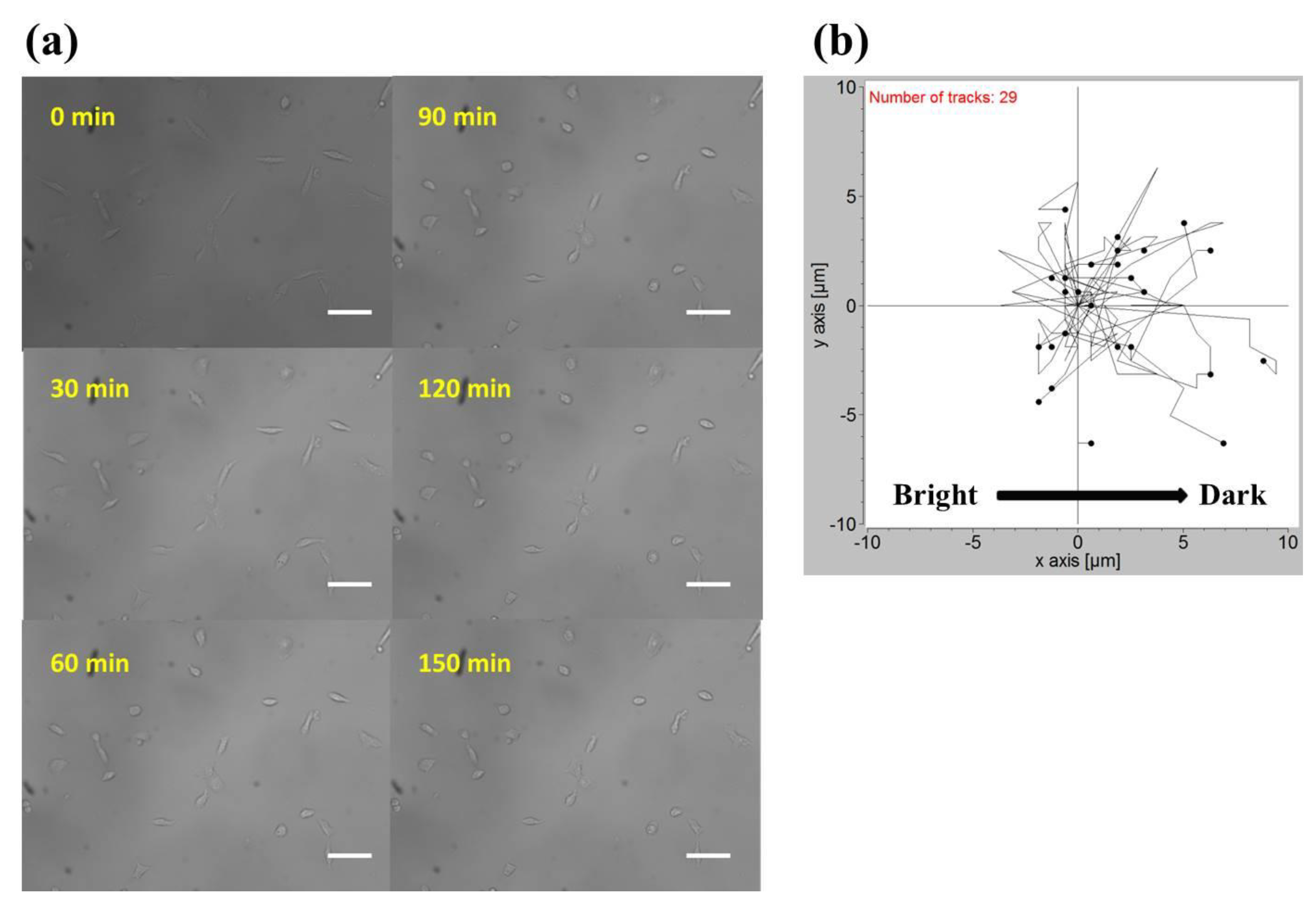
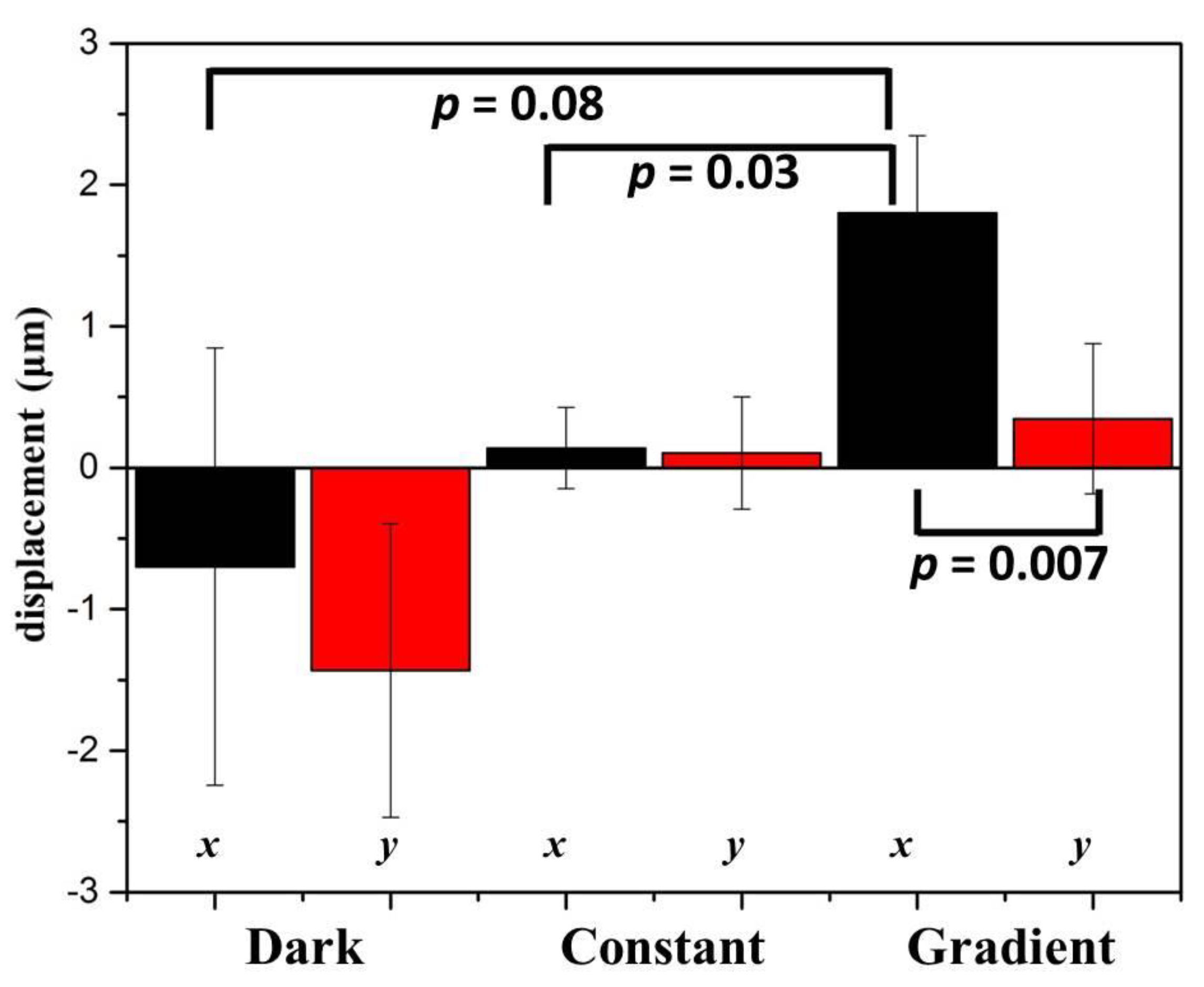
2.2. Phototaxis of A549 Cells Induced by a Blue Light Gradient
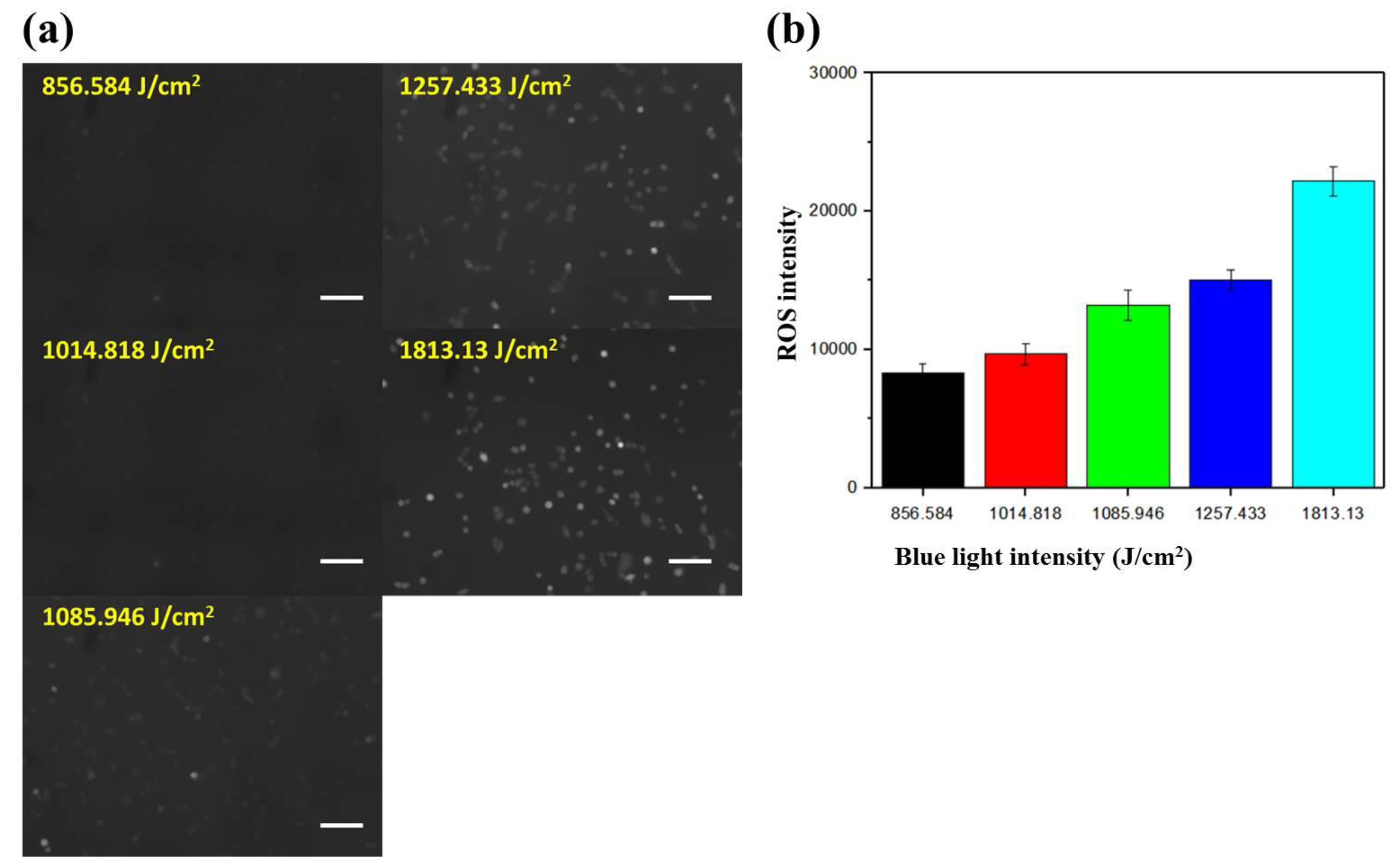
2.3. Dependence of the Intracellular ROS Level on the Intensity of Blue Light
2.4. Response of NIH/3T3 Cells to a Blue Light Gradient
3. Materials and Methods
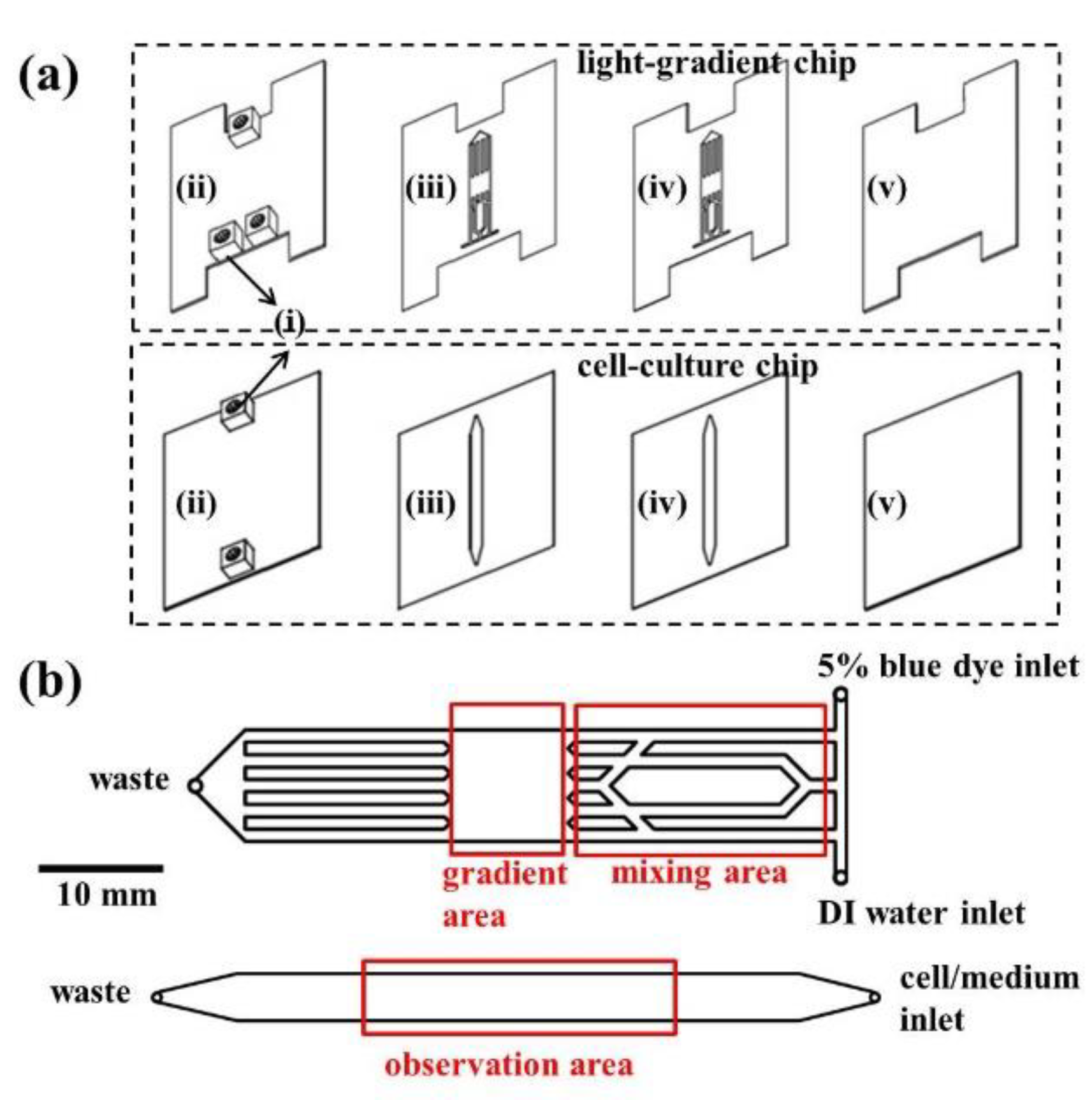
3.1. Chip Design and Fabrication
3.2. Calculation and Simulation of Concentrations and Absorbance
3.3. Cell Preparation
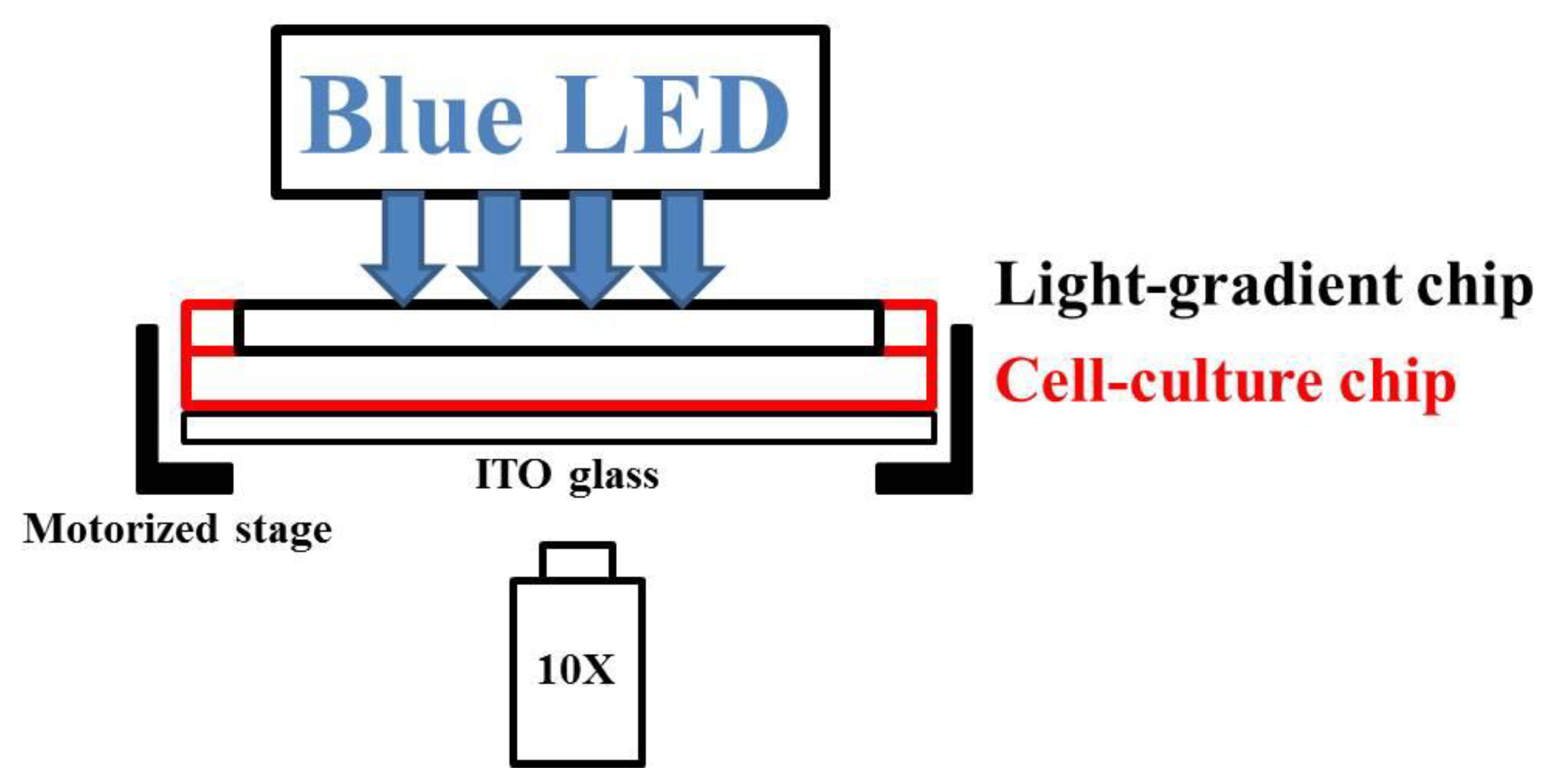
3.4. Experimental System and Procedure
3.4.1. Chip Assembly and Cell Injection
3.4.2. Blue Light Treatments
3.4.3. Cell Staining
3.5. Data Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ratheesh, A.; Belyaeva, V.; Siekhaus, D.E. Drosophila immune cell migration and adhesion during embryonic development and larval immune responses. Curr. Opin. Cell Biol. 2015, 36, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.H.; Kim, C.T.; Jung, Y.H.; Lee, N.S.; Jeong, Y.G. Early cerebellar granule cell migration in the mouse embryonic development. Anat. Cell Biol. 2010, 43, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.; Harrington, K.I.; Lovegrove, H.E.; Page, D.J.; Chakravartula, S.; Bentley, K.; Herbert, S.P. Asymmetric division coordinates collective cell migration in angiogenesis. Nat. Cell Biol. 2016, 18, 1292–1301. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, Y.; Zhao, M.; Jiang, J. Collective cell migration: Implications for wound healing and cancer invasion. Burns Trauma 2013, 1, 21–26. [Google Scholar] [PubMed]
- Arciero, J.C.; Mi, Q.; Branca, M.F.; Hackam, D.J.; Swigon, D. Continuum model of collective cell migration in wound healing and colony expansion. Biophys. J. 2011, 100, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Rivero, E.M.; Pinero, C.P.; Gargiulo, L.; Entschladen, F.; Zanker, K.; Bruzzone, A.; Luthy, I.A. The beta 2-Adrenergic Agonist Salbutamol Inhibits Migration, Invasion and Metastasis of the Human Breast Cancer MDA-MB-231 Cell Line. Curr. Cancer Drug Targets 2017, 17, 756–766. [Google Scholar] [CrossRef]
- Cooper, C.R.; Pienta, K.J. Cell adhesion and chemotaxis in prostate cancer metastasis to bone: A minireview. Prostate Cancer Prostatic Dis. 2000, 3, 6–12. [Google Scholar] [CrossRef]
- Li, J.; Ley, K. Lymphocyte Migration into Atherosclerotic Plaque. Arterioscl. Throm. Vas. 2015, 35, 40–49. [Google Scholar] [CrossRef]
- Gelfand, E.W. Importance of the leukotriene B4-BLT1 and LTB4-BLT2 pathways in asthma. Semin. Immunol. 2017, 33, 44–51. [Google Scholar] [CrossRef]
- Sun, Y.S.; Peng, S.W.; Cheng, J.Y. In vitro electrical-stimulated wound-healing chip for studying electric field-assisted wound-healing process. Biomicrofluidics 2012, 6, 34117. [Google Scholar] [CrossRef]
- Levin, M. Bioelectric mechanisms in regeneration: Unique aspects and future perspectives. Semin. Cell Dev. Biol. 2009, 20, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.N.; Yang, Z.; Law, J.B.K.; He, A.Y.; Abbas, A.A.; Denslin, V.; Kamarul, T.; Hui, J.H.P.; Lee, E.H. The Combined Effect of Substrate Stiffness and Surface Topography on Chondrogenic Differentiation of Mesenchymal Stem Cells. Tissue Eng. Part A 2017, 23, 43–54. [Google Scholar] [CrossRef]
- Wormer, D.B.; Davis, K.A.; Henderson, J.H.; Turner, C.E. The focal adhesion-localized CdGAP regulates matrix rigidity sensing and durotaxis. PLoS ONE 2014, 9, e91815. [Google Scholar] [CrossRef] [PubMed]
- Basan, M.; Elgeti, J.; Hannezo, E.; Rappel, W.J.; Levine, H. Alignment of cellular motility forces with tissue flow as a mechanism for efficient wound healing. Proc. Natl. Acad. Sci. USA 2013, 110, 2452–2459. [Google Scholar] [CrossRef]
- Pattanaik, B.; Whitaker, M.J.; Montgomery, B.L. Light Quantity Affects the Regulation of Cell Shape in Fremyella diplosiphon. Front. Microbiol. 2012, 3, 170. [Google Scholar] [CrossRef]
- Liebel, F.; Kaur, S.; Ruvolo, E.; Kollias, N.; Southall, M.D. Irradiation of skin with visible light induces reactive oxygen species and matrix-degrading enzymes. J. Investig. Dermatol. 2012, 132, 1901–1907. [Google Scholar] [CrossRef] [PubMed]
- de Jager, T.L.; Cockrell, A.E.; Du Plessis, S.S. Ultraviolet Light Induced Generation of Reactive Oxygen Species. Adv. Exp. Med. Biol. 2017, 996, 15–23. [Google Scholar]
- Yao, J.; Liu, Y.; Wang, X.; Shen, Y.; Yuan, S.; Wan, Y.; Jiang, Q. UVB radiation induces human lens epithelial cell migration via NADPH oxidase-mediated generation of reactive oxygen species and up-regulation of matrix metalloproteinases. Int. J. Mol. Med. 2009, 24, 153–159. [Google Scholar]
- Lan, C.C.; Lu, E.Y.; Pan, H.J.; Lee, C.H. Directional migration of cancer cells induced by a blue light intensity gradient. Biomed. Opt. Express 2015, 6, 2624–2632. [Google Scholar] [CrossRef]
- Xiao, J.L.; Lu, D.H.; Lee, C.H. Guiding the migration of adherent cells by using optical micropatterns. Appl. Phys. Lett. 2013, 102, 123703. [Google Scholar] [CrossRef]
- Xiao, J.L.; Pan, H.J.; Lee, C.H. Optically micropatterned culture of adherent cells. J. Biomed. Opt. 2012, 17, 075004. [Google Scholar] [CrossRef]
- El-Ali, J.; Sorger, P.K.; Jensen, K.F. Cells on chips. Nature 2006, 442, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Lo, K.Y.; Wu, S.Y.; Sun, Y.S. A microfluidic device for studying the production of reactive oxygen species and the migration in lung cancer cells under single or coexisting chemical/electrical stimulation. Microfluid. Nanofluid. 2016, 20, 15. [Google Scholar] [CrossRef]
- Sun, Y.S. Studying Electrotaxis in Microfluidic Devices. Sensors 2017, 17, 2048. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Vazquez, M. A Gal-MmicroS Device to Evaluate Cell Migratory Response to Combined Galvano-Chemotactic Fields. Biosensors 2017, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.C.; Hsieh, M.H.; Liu, C.C.; Pan, H.J.; Liao, W.Y.; Cheng, J.Y.; Kuo, P.L.; Lee, C.H. Modulating chemotaxis of lung cancer cells by using electric fields in a microfluidic device. Biomicrofluidics 2014, 8, 024107. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Hou, H.S.; Lo, K.Y.; Cheng, J.Y.; Sun, Y.S. Use microfluidic chips to study the effects of ultraviolet lights on human fibroblasts. Microfluid. Nanofluid. 2017, 21, 79. [Google Scholar] [CrossRef]
- Oh, P.S.; Kim, H.S.; Kim, E.M.; Hwang, H.; Ryu, H.H.; Lim, S.; Sohn, M.H.; Jeong, H.J. Inhibitory effect of blue light emitting diode on migration and invasion of cancer cells. J. Cell. Physiol. 2017, 232, 3444–3453. [Google Scholar] [CrossRef] [PubMed]
- Ushio-Fukai, M.; Nakamura, Y. Reactive oxygen species and angiogenesis: NADPH oxidase as target for cancer therapy. Cancer Lett. 2008, 266, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Thannickal, V.J.; Fanburg, B.L. Reactive oxygen species in cell signaling. American journal of physiology. Lung Cell. Mol. Physiol. 2000, 279, L1005–L1028. [Google Scholar] [CrossRef]
- Lockwood, D.B.; Wataha, J.C.; Lewis, J.B.; Tseng, W.Y.; Messer, R.L.; Hsu, S.D. Blue light generates reactive oxygen species (ROS) differentially in tumor vs. normal epithelial cells. Dent. Mater. 2005, 21, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Zhang, L.; Feng, C.; Gong, R.; Idiiatullina, E.; Huang, Q.; He, M.; Guo, S.; Yang, F.; Li, Y.; et al. Blue light emitting diodes irradiation causes cell death in colorectal cancer by inducing ROS production and DNA damage. Int. J. Biochem. Cell Biol. 2018, 103, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.W.; Cheng, J.Y.; Yen, M.H.; Young, T.H. Electrotaxis of lung cancer cells in a multiple-electric-field chip. Biosens. Bioelectron. 2009, 24, 3510–3516. [Google Scholar] [CrossRef]
- Ishida, T.; Shimamoto, T.; Ozaki, N.; Takaki, S.; Kuchimaru, T.; Kizaka-Kondoh, S.; Omata, T. Investigation of the Influence of Glucose Concentration on Cancer Cells by Using a Microfluidic Gradient Generator without the Induction of Large Shear Stress. Micromachines-Basel 2016, 7, 155. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Butcher, E.C. T cell chemotaxis in a simple microfluidic device. Lab Chip 2006, 6, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Choi, S.C.; Oh, J. Sheath Flow-Controlled Molecular Diffusion in Three Dimensional Multiple Micro-Mixer. Dig. J. Nanomater. Bios. 2015, 10, 497–503. [Google Scholar]
- Sun, Y.S.; Peng, S.W.; Lin, K.H.; Cheng, J.Y. Electrotaxis of lung cancer cells in ordered three-dimensional scaffolds. Biomicrofluidics 2012, 6, 014102. [Google Scholar] [CrossRef] [PubMed]
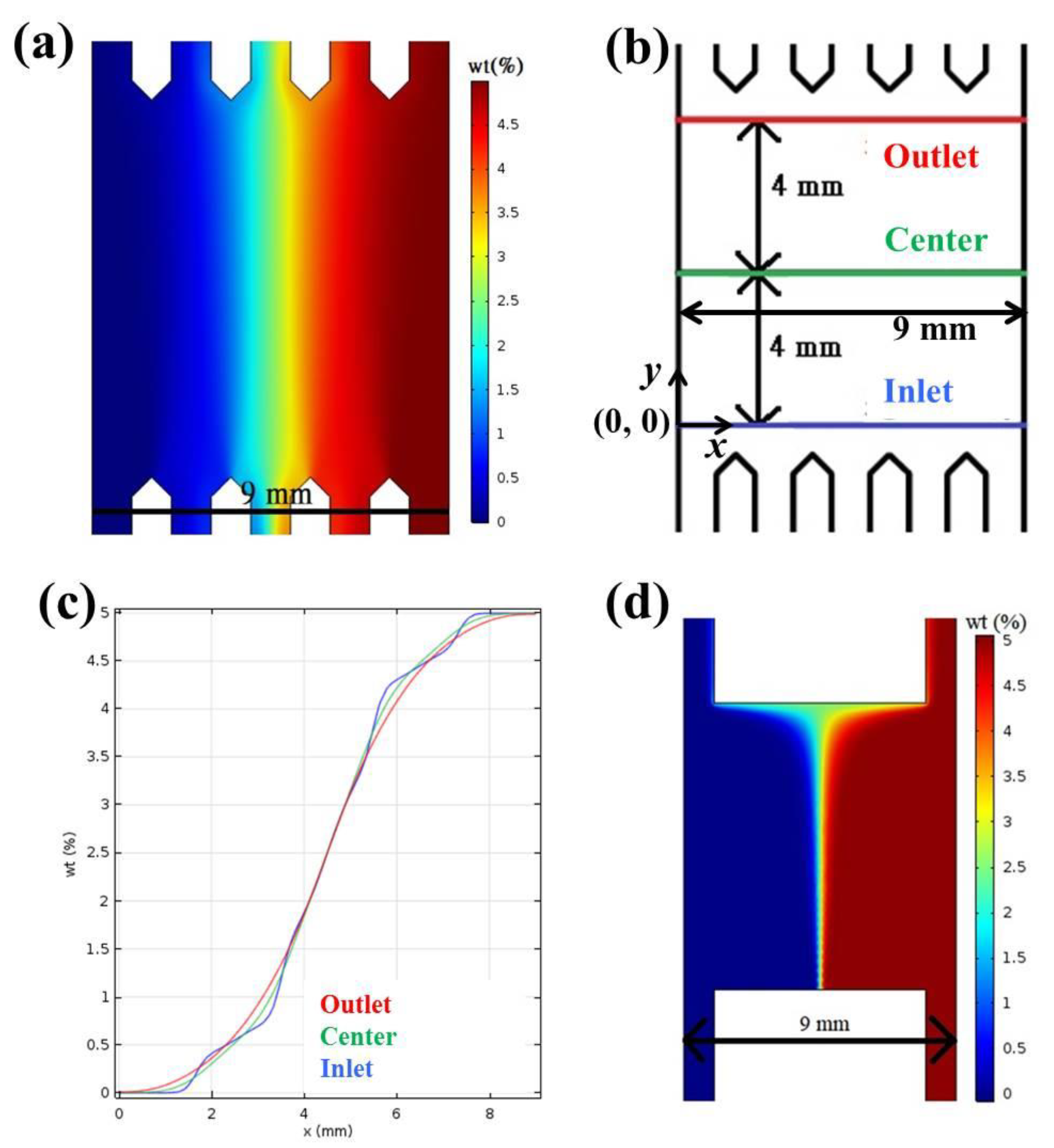
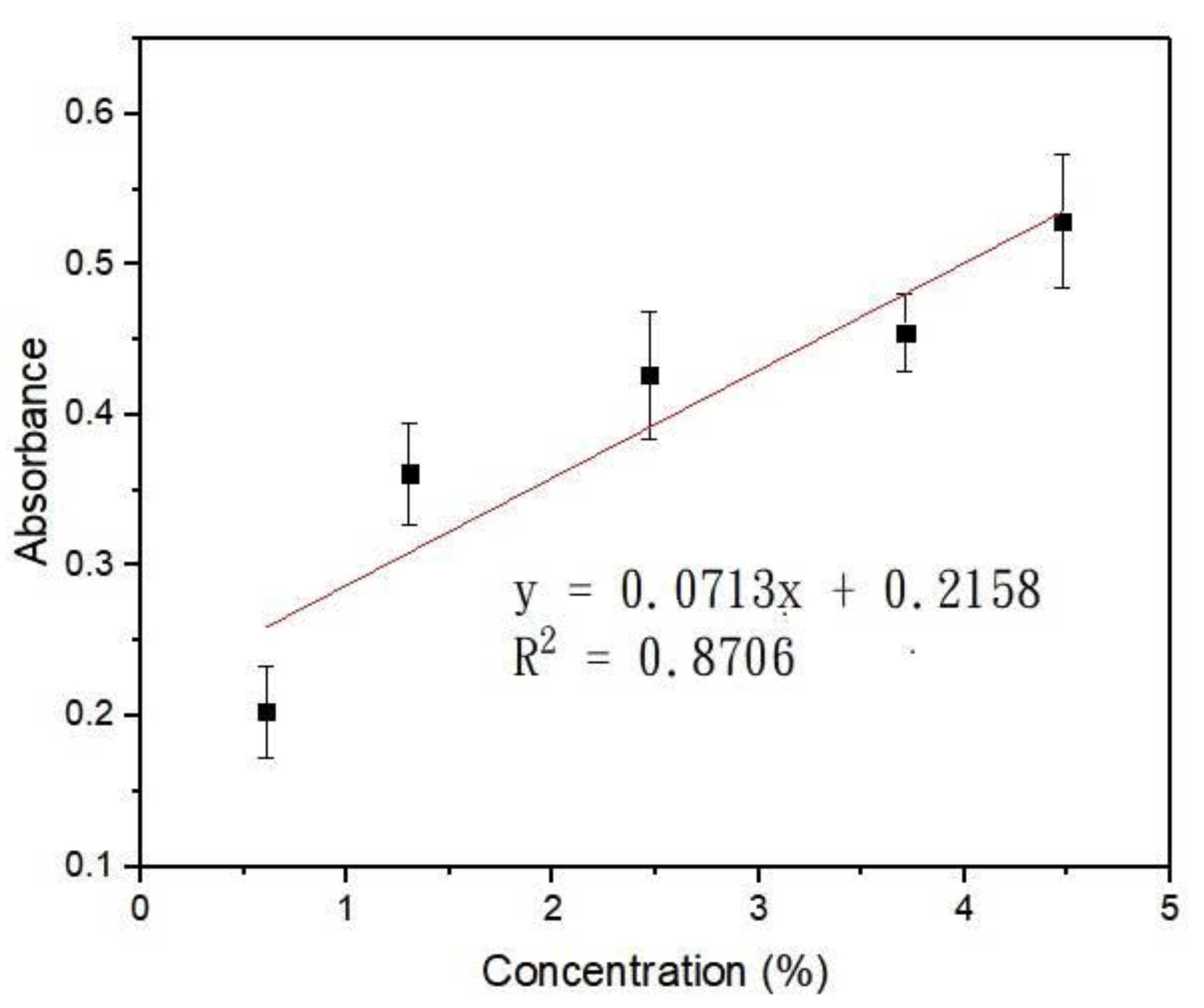

| x position (nm) | 0.5 | 2.5 | 3.5 | 4.5 | 5.5 | 6.5 |
|---|---|---|---|---|---|---|
| Power (W/cm2) | 0.320 ± 0.031 | 0.201 ± 0.0198 | 0.140 ± 0.0141 | 0.121 ± 0.0164 | 0.113 ± 0.0096 | 0.095 ± 0.0128 |
| Transmittance | 1 ± 0.097 | 0.630 ± 0.062 | 0.437 ± 0.044 | 0.377 ± 0.051 | 0.353 ± 0.030 | 0.298 ± 0.040 |
| Absorbance | 0.203 ± 0.053 | 0.361 ± 0.058 | 0.427 ± 0.073 | 0.455 ± 0.045 | 0.529 ± 0.076 | |
| Concentration | 0.61% | 1.30% | 2.47% | 3.71% | 4.47% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, F.-Y.; Lin, J.-Y.; Lo, K.-Y.; Sun, Y.-S. Use Microfluidic Chips to Study the Phototaxis of Lung Cancer Cells. Int. J. Mol. Sci. 2019, 20, 4515. https://doi.org/10.3390/ijms20184515
Lin F-Y, Lin J-Y, Lo K-Y, Sun Y-S. Use Microfluidic Chips to Study the Phototaxis of Lung Cancer Cells. International Journal of Molecular Sciences. 2019; 20(18):4515. https://doi.org/10.3390/ijms20184515
Chicago/Turabian StyleLin, Fong-Yi, Jin-Young Lin, Kai-Yin Lo, and Yung-Shin Sun. 2019. "Use Microfluidic Chips to Study the Phototaxis of Lung Cancer Cells" International Journal of Molecular Sciences 20, no. 18: 4515. https://doi.org/10.3390/ijms20184515
APA StyleLin, F.-Y., Lin, J.-Y., Lo, K.-Y., & Sun, Y.-S. (2019). Use Microfluidic Chips to Study the Phototaxis of Lung Cancer Cells. International Journal of Molecular Sciences, 20(18), 4515. https://doi.org/10.3390/ijms20184515






