Endogenous n-3 Polyunsaturated Fatty Acids Are Beneficial to Dampen CD8+ T Cell-Mediated Inflammatory Response upon the Viral Infection in Mice
Abstract
1. Introduction
2. Results
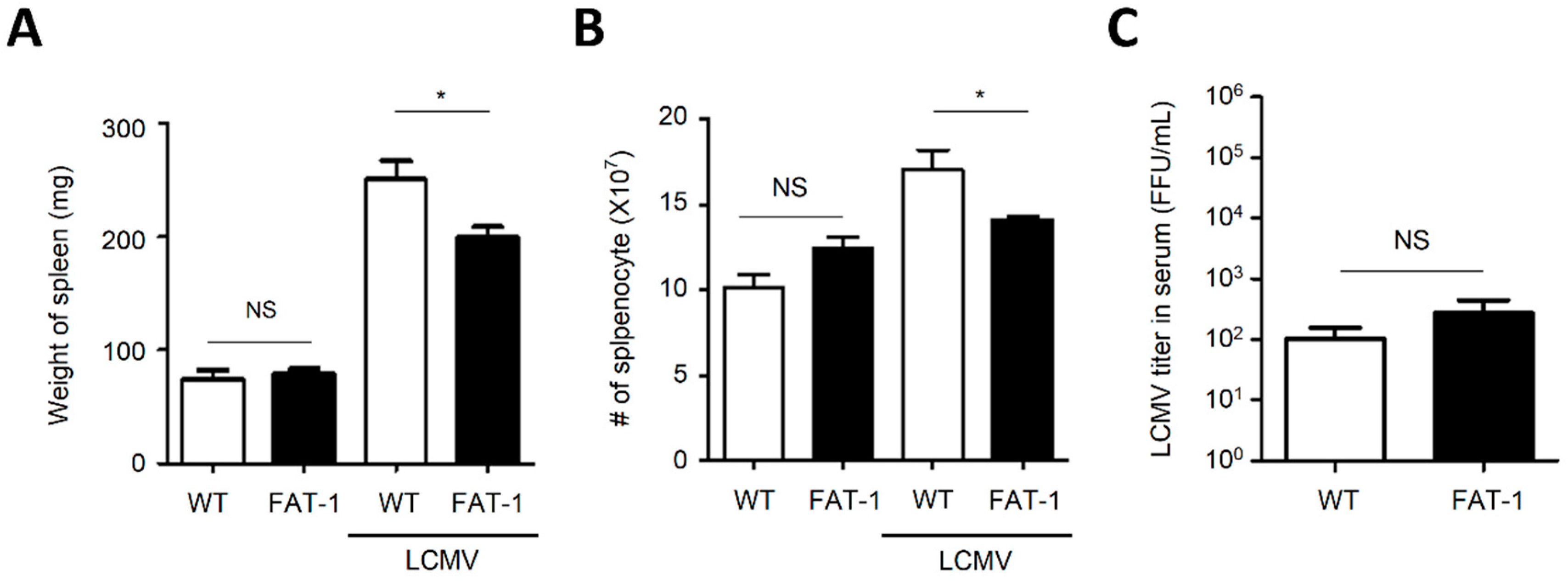
2.1. The Expansion of Splenocytes in FAT-1 Mice Was Limited upon Acute LCMV Infection
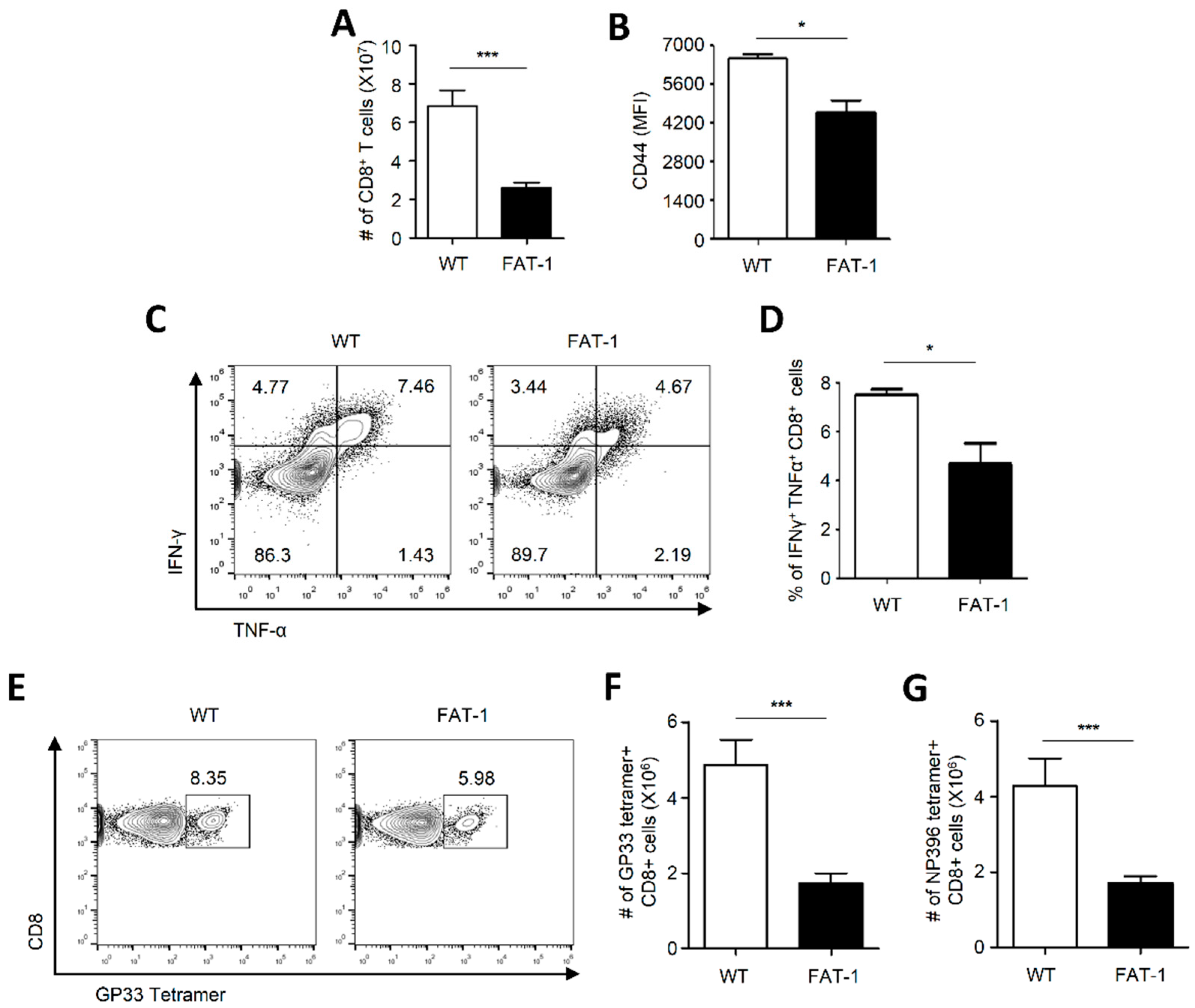
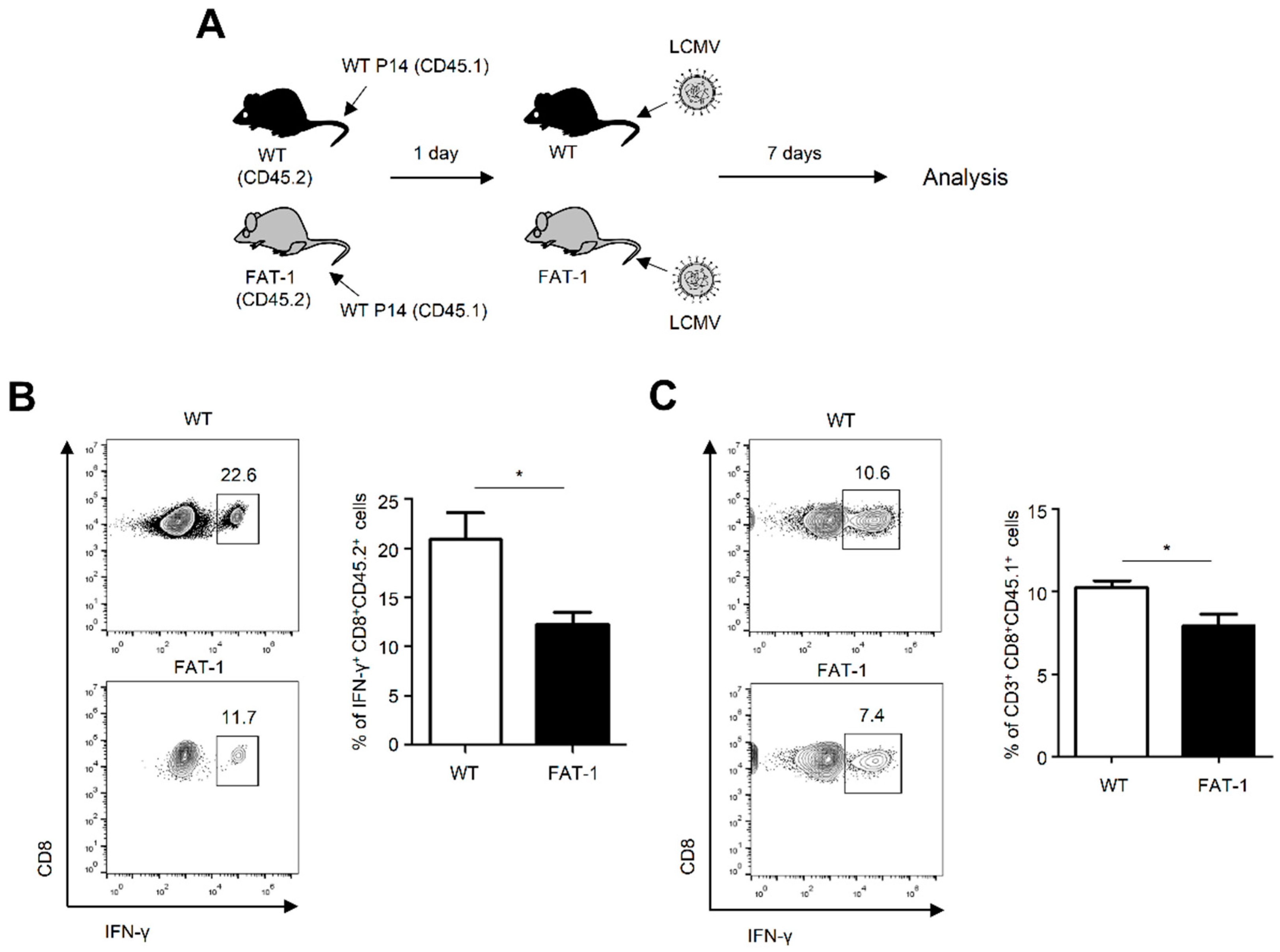
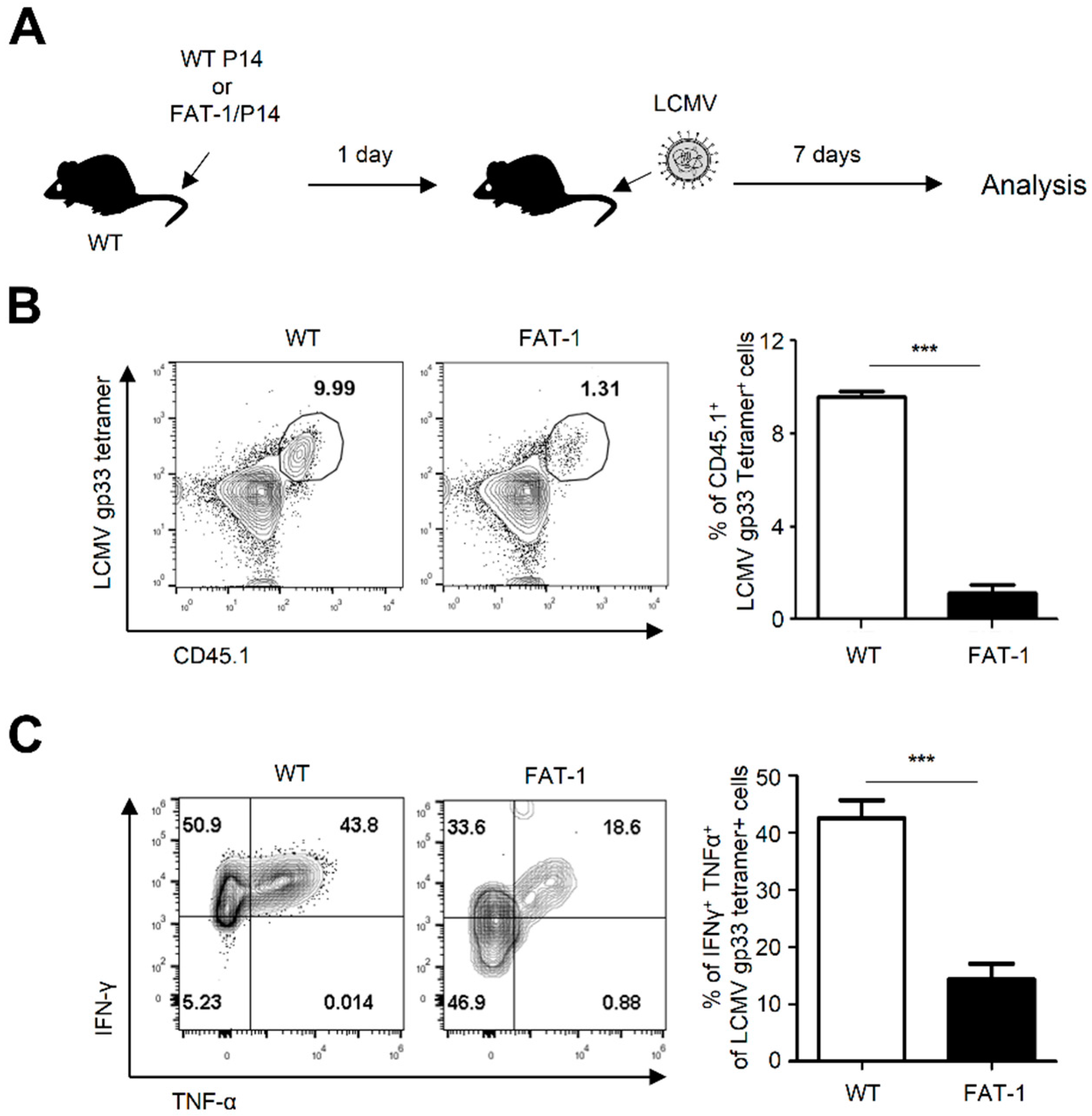
2.2. Anti-Viral CD8+ T Cell Responses Were Suppressed in FAT-1 Mice
2.3. Anti-Viral CD8+ T Cell Responses in Peripheral Blood of FAT-1 Mice Were Suppressed
2.4. Anti-Viral CD8+ T Cell Responses in FAT-1 Mice Were Affected by Extracellular Factors
2.5. Anti-Viral CD4+ T Cell Responses in FAT-1 Mice Were Impaired
2.6. Anti-Viral CD8+ T Cells Carrying the Fat-1 Gene Exhibited a Decreased Capability to Proliferate and Produce Inflammatory Cytokines against LCMV Infection
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Virus and Infection
4.3. Reagents and Antibodies
4.4. Determination of LCMV Titers
4.5. Isolation of Splenocytes
4.6. Intracellular Cytokine Staining
4.7. Flow Cytometric Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saini, R.K.; Keum, Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.A.; Norris, P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015, 15, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X.; Wang, J.; Wu, L.; Kang, Z.B. Transgenic mice: Fat-1 mice convert n-6 to n-3 fatty acids. Nature 2004, 427, 504. [Google Scholar] [CrossRef] [PubMed]
- Bellenger, J.; Bellenger, S.; Bataille, A.; Massey, K.A.; Nicolaou, A.; Rialland, M.; Tessier, C.; Kang, J.X.; Narce, M. High pancreatic n-3 fatty acids prevent STZ-induced diabetes in fat-1 mice: Inflammatory pathway inhibition. Diabetes 2011, 60, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Monk, J.M.; Jia, Q.; Callaway, E.; Weeks, B.; Alaniz, R.C.; McMurray, D.N.; Chapkin, R.S. Th17 cell accumulation is decreased during chronic experimental colitis by (n-3) PUFA in Fat-1 mice. J. Nutr. 2012, 142, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.Y.; Lim, K.; Lee, S.M.; Park, B.H. Effects of n-3 PUFA on the CD4(+) type 2 helper T-cell-mediated immune responses in Fat-1 mice. Mol. Nutr. Food Res. 2014, 58, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Mickleborough, T.D.; Ionescu, A.A.; Rundell, K.W. Omega-3 Fatty acids and airway hyperresponsiveness in asthma. J. Altern. Complement. Med. 2004, 10, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Mozaffari, H.; Daneshzad, E.; Larijani, B.; Bellissimo, N.; Azadbakht, L. Dietary intake of fish, n-3 polyunsaturated fatty acids, and risk of inflammatory bowel disease: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.D.; Abreu, M.T. Diet as a Trigger or Therapy for Inflammatory Bowel Diseases. Gastroenterology 2017, 152, 398–414.e6. [Google Scholar] [CrossRef]
- Ruggiero, C.; Lattanzio, F.; Lauretani, F.; Gasperini, B.; Andres-Lacueva, C.; Cherubini, A. Omega-3 polyunsaturated fatty acids and immune-mediated diseases: Inflammatory bowel disease and rheumatoid arthritis. Curr. Pharm. Des. 2009, 15, 4135–4148. [Google Scholar] [CrossRef]
- Philippou, E.; Nikiphorou, E. Are we really what we eat? Nutrition and its role in the onset of rheumatoid arthritis. Autoimmun. Rev. 2018, 17, 1074–1077. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, D.L.; Fan, Y.Y.; Chapkin, R.S.; McMurray, D.N. Transgenic Mice Enriched in Omega-3 Fatty Acids Are More Susceptible to Pulmonary Tuberculosis: Impaired Resistance to Tuberculosis in fat-1 Mice. J. Infect. Dis. 2010, 201, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Tiesset, H.; Pierre, M.; Desseyn, J.L.; Guery, B.; Beermann, C.; Galabert, C.; Gottrand, F.; Husson, M.O. Dietary (n-3) Polyunsaturated Fatty Acids Affect the Kinetics of Pro- and Antiinflammatory Responses in Mice with Pseudomonas aeruginosa Lung Infection. J. Nutr. 2009, 139, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Pierre, M.; Husson, M.O.; Le Berre, R.; Desseyn, J.L.; Galabert, C.; Beghin, L.; Beermann, C.; Dagenais, A.; Berthiaume, Y.; Cardinaud, B.; et al. Omega-3 polyunsaturated fatty acids improve host response in chronic Pseudomonas aeruginosa lung infection in mice. Am. J. Physiol.-Lung C 2007, 292, L1422–L1431. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fritsche, K.L.; Shahbazian, L.M.; Feng, C.; Berg, J.N. Dietary fish oil reduces survival and impairs bacterial clearance in C3H/Hen mice challenged with Listeria monocytogenes. Clin. Sci 1997, 92, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Schwerbrock, N.M.J.; Karlsson, E.A.; Shi, Q.; Sheridan, P.A.; Beck, M.A. Fish Oil-Fed Mice Have Impaired Resistance to Influenza Infection. J. Nutr. 2009, 139, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.J.B.; Roper, R.L. The effects of diets enriched in omega-3 polyunsaturated fatty acids on systemic vaccinia virus infection. Sci. Rep. 2017, 7, 15999. [Google Scholar] [CrossRef] [PubMed]
- Halle, S.; Halle, O.; Forster, R. Mechanisms and Dynamics of T Cell-Mediated Cytotoxicity In Vivo. Trends Immunol. 2017, 38, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Wong, P.; Pamer, E.G. CD8 T cell responses to infectious pathogens. Annu. Rev. Immunol. 2003, 21, 29–70. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Chang, J. Recombinant baculovirus-based vaccine expressing M2 protein induces protective CD8(+) T-cell immunity against respiratory syncytial virus infection. J. Microbiol. 2017, 55, 900–908. [Google Scholar] [CrossRef]
- Trapani, J.A.; Smyth, M.J. Functional significance of the perforin/granzyme cell death pathway. Nat. Rev. Immunol. 2002, 2, 735–747. [Google Scholar] [CrossRef] [PubMed]
- van Dommelen, S.L.; Sumaria, N.; Schreiber, R.D.; Scalzo, A.A.; Smyth, M.J.; Degli-Esposti, M.A. Perforin and granzymes have distinct roles in defensive immunity and immunopathology. Immunity 2006, 25, 835–848. [Google Scholar] [CrossRef] [PubMed]
- Ramana, C.V.; Cheng, G.S.; Kumar, A.; Kwon, H.J.; Enelow, R.I. Role of alveolar epithelial early growth response-1 (Egr-1) in CD8+ T cell-mediated lung injury. Mol. Immunol. 2009, 47, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Bradfute, S.B.; Warfield, K.L.; Bavari, S. Functional CD8+ T cell responses in lethal Ebola virus infection. J. Immunol. 2008, 180, 4058–4066. [Google Scholar] [CrossRef] [PubMed]
- Tagliapietra, V.; Rosa, R.; Hauffe, H.C.; Laakkonen, J.; Voutilainen, L.; Vapalahti, O.; Vaheri, A.; Henttonen, H.; Rizzoli, A. Spatial and temporal dynamics of lymphocytic choriomeningitis virus in wild rodents, northern Italy. Emerg. Infect. Dis. 2009, 15, 1019–1025. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oldstone, M.B.A.; Ware, B.C.; Horton, L.E.; Welch, M.J.; Aiolfi, R.; Zarpellon, A.; Ruggeri, Z.M.; Sullivan, B.M. Lymphocytic choriomeningitis virus Clone 13 infection causes either persistence or acute death dependent on IFN-1, cytotoxic T lymphocytes (CTLs), and host genetics. Proc. Natl. Acad. Sci. USA 2018, 115, E7814–E7823. [Google Scholar] [CrossRef]
- Wherry, E.J.; Blattman, J.N.; Murali-Krishna, K.; van der Most, R.; Ahmed, R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 2003, 77, 4911–4927. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.M.; Teijaro, J.R.; de la Torre, J.C.; Oldstone, M.B. Early virus-host interactions dictate the course of a persistent infection. PLoS Pathog. 2015, 11, e1004588. [Google Scholar] [CrossRef]
- Seo, Y.J.; Jothikumar, P.; Suthar, M.S.; Zhu, C.; Grakoui, A. Local Cellular and Cytokine Cues in the Spleen Regulate In Situ T Cell Receptor Affinity, Function, and Fate of CD8(+) T Cells. Immunity 2016, 45, 988–998. [Google Scholar] [CrossRef]
- Althaus, C.L.; Ganusov, V.V.; De Boer, R.J. Dynamics of CD8+ T cell responses during acute and chronic lymphocytic choriomeningitis virus infection. J. Immunol. 2007, 179, 2944–2951. [Google Scholar] [CrossRef]
- Abdulrazaq, M.; Innes, J.K.; Calder, P.C. Effect of omega-3 polyunsaturated fatty acids on arthritic pain: A systematic review. Nutrition 2017, 39–40, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Valentine, R.C.; Valentine, D.L. Omega-3 fatty acids in cellular membranes: A unified concept. Prog. Lipid Res. 2004, 43, 383–402. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Fan, Y.Y.; Barhoumi, R.; Smith, R.; McMurray, D.N.; Chapkin, R.S. n-3 polyunsaturated fatty acids suppress the localization and activation of signaling proteins at the immunological synapse in murine CD4+ T cells by affecting lipid raft formation. J. Immunol. 2008, 181, 6236–6243. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.Y.; McMurray, D.N.; Chapkin, R.S. Omega-3 fatty acids, lipid rafts, and T cell signaling. Eur. J. Pharm. 2016, 785, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Stulnig, T.M.; Huber, J.; Leitinger, N.; Imre, E.M.; Angelisova, P.; Nowotny, P.; Waldhausl, W. Polyunsaturated eicosapentaenoic acid displaces proteins from membrane rafts by altering raft lipid composition. J. Biol. Chem. 2001, 276, 37335–37340. [Google Scholar] [CrossRef] [PubMed]
- Stulnig, T.M.; Berger, M.; Sigmund, T.; Raederstorff, D.; Stockinger, H.; Waldhausl, W. Polyunsaturated fatty acids inhibit T cell signal transduction by modification of detergent-insoluble membrane domains. J. Cell Biol. 1998, 143, 637–644. [Google Scholar] [CrossRef]
- Wherry, E.J. T cell exhaustion. Nat. Immunol. 2011, 12, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Moskophidis, D.; Lechner, F.; Pircher, H.; Zinkernagel, R.M. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature 1993, 362, 758–761. [Google Scholar] [CrossRef]
- Wherry, E.J.; Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 2015, 15, 486–499. [Google Scholar] [CrossRef]
- Welsh, R.M.; Seedhom, M.O. Lymphocytic choriomeningitis virus (LCMV): Propagation, quantitation, and storage. Curr. Protoc. Microbiol. 2008, 8, 15A.1.1–15A.1.11. [Google Scholar] [CrossRef]
- Battegay, M.; Cooper, S.; Althage, A.; Banziger, J.; Hengartner, H.; Zinkernagel, R.M. Quantification of lymphocytic choriomeningitis virus with an immunological focus assay in 24- or 96-well plates. J. Virol. Methods 1991, 33, 191–198. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, K.W.; Kim, S.; Cho, Y.-B.; Ryu, S.R.; Seo, Y.-J.; Lee, S.-M. Endogenous n-3 Polyunsaturated Fatty Acids Are Beneficial to Dampen CD8+ T Cell-Mediated Inflammatory Response upon the Viral Infection in Mice. Int. J. Mol. Sci. 2019, 20, 4510. https://doi.org/10.3390/ijms20184510
Kang KW, Kim S, Cho Y-B, Ryu SR, Seo Y-J, Lee S-M. Endogenous n-3 Polyunsaturated Fatty Acids Are Beneficial to Dampen CD8+ T Cell-Mediated Inflammatory Response upon the Viral Infection in Mice. International Journal of Molecular Sciences. 2019; 20(18):4510. https://doi.org/10.3390/ijms20184510
Chicago/Turabian StyleKang, Kyung Won, Seyoung Kim, Yong-Bin Cho, Seung Rok Ryu, Young-Jin Seo, and Sang-Myeong Lee. 2019. "Endogenous n-3 Polyunsaturated Fatty Acids Are Beneficial to Dampen CD8+ T Cell-Mediated Inflammatory Response upon the Viral Infection in Mice" International Journal of Molecular Sciences 20, no. 18: 4510. https://doi.org/10.3390/ijms20184510
APA StyleKang, K. W., Kim, S., Cho, Y.-B., Ryu, S. R., Seo, Y.-J., & Lee, S.-M. (2019). Endogenous n-3 Polyunsaturated Fatty Acids Are Beneficial to Dampen CD8+ T Cell-Mediated Inflammatory Response upon the Viral Infection in Mice. International Journal of Molecular Sciences, 20(18), 4510. https://doi.org/10.3390/ijms20184510




