An Integrated Transcriptome and Proteome Analysis Reveals New Insights into Russeting of Bagging and Non-Bagging “Golden Delicious” Apple
Abstract
1. Introduction
2. Results
2.1. Histological Analysis of Russeting Apple
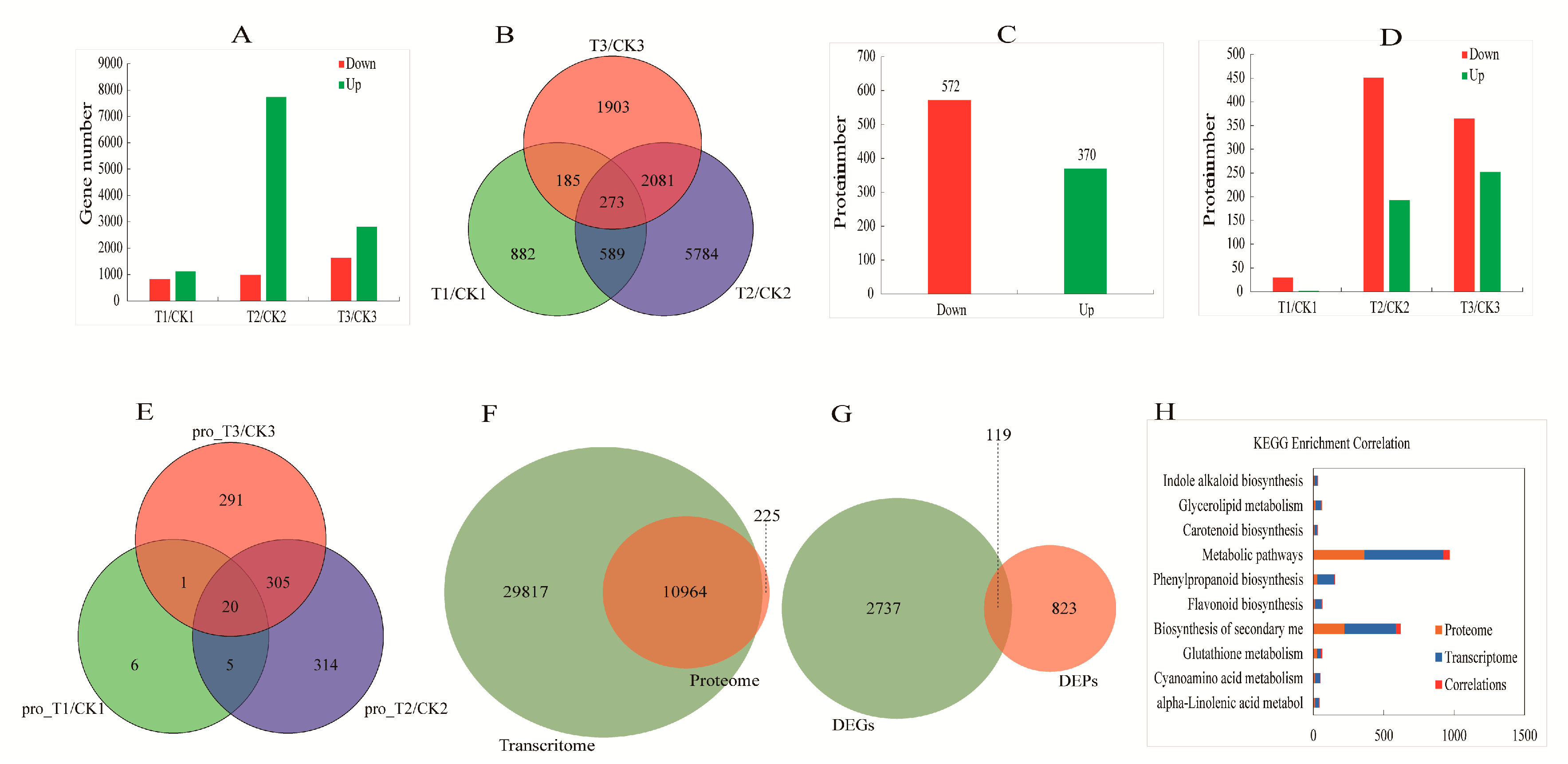
2.2. General Characterization of Transcriptome Data
2.3. General Characterization of Proteome Data
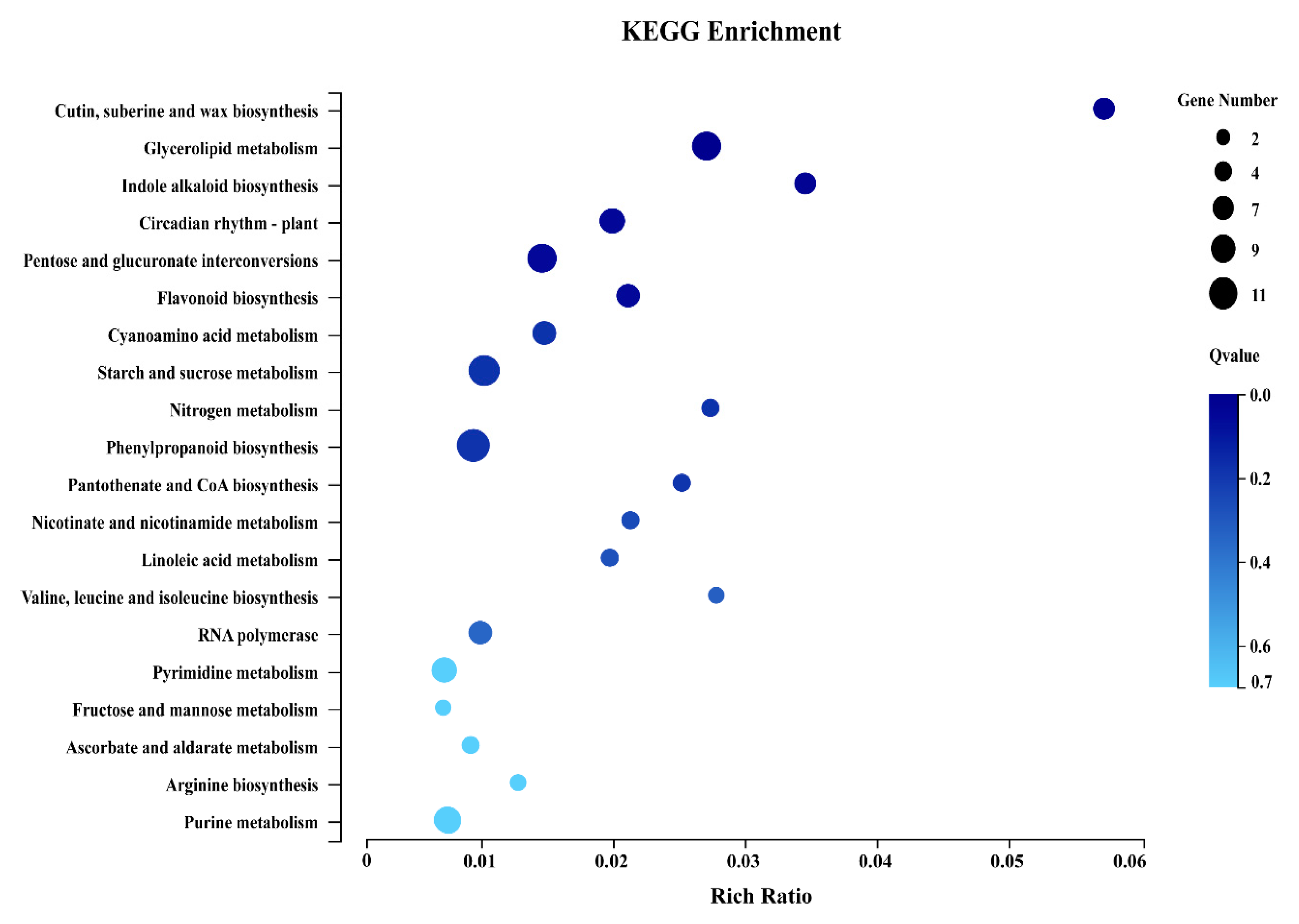
2.4. Proteome and Transcriptome Correlation Analysis
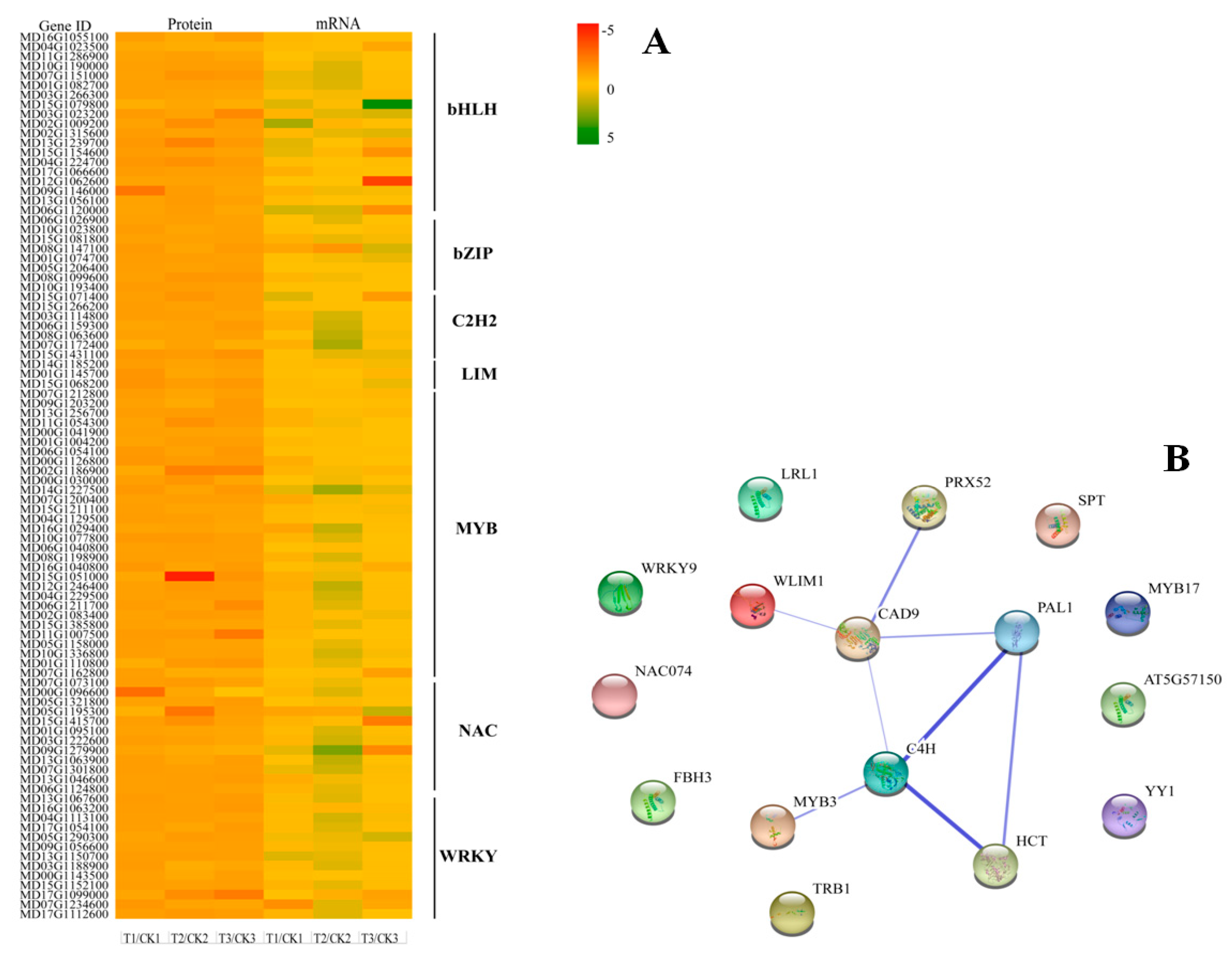
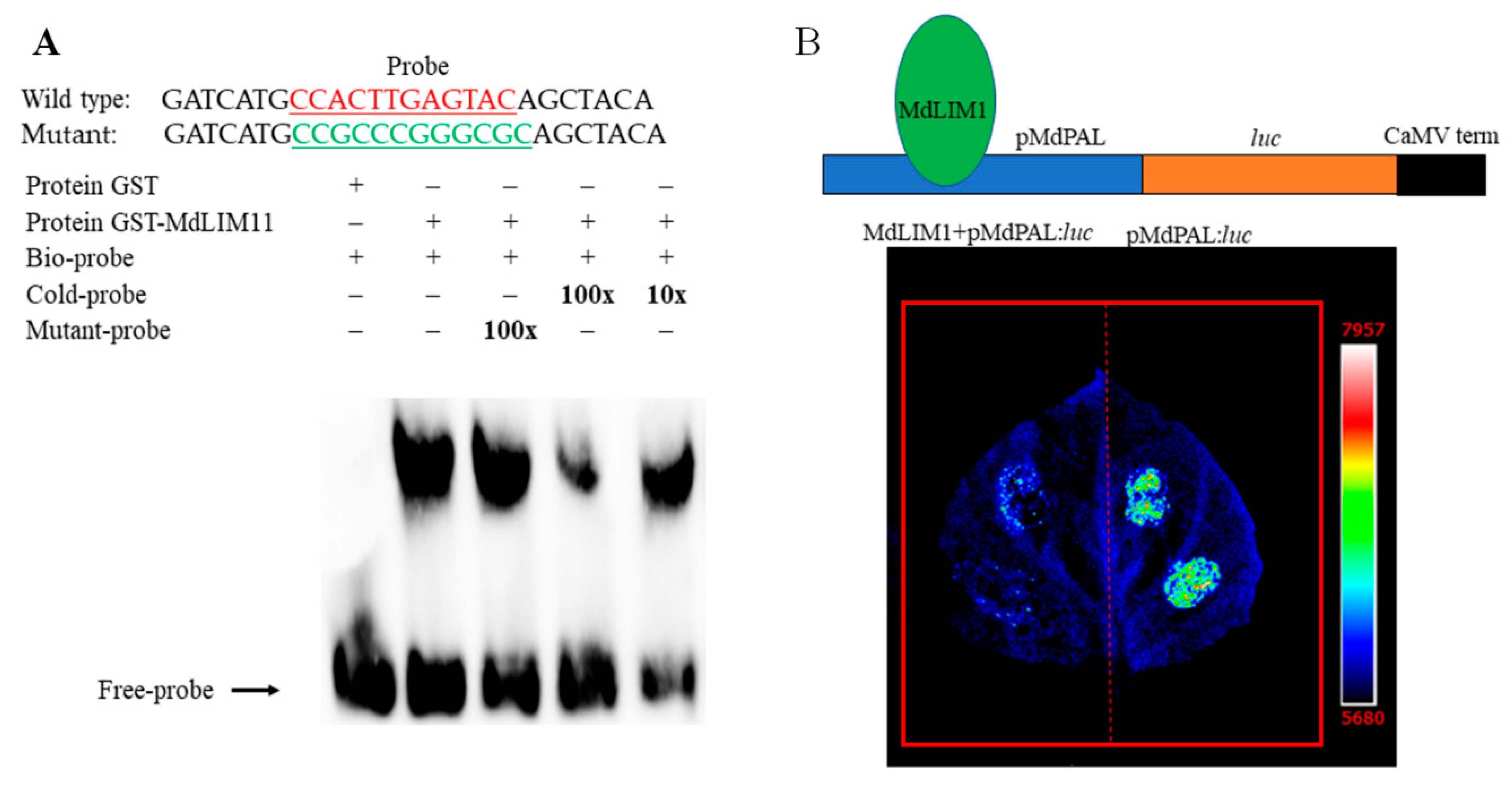
2.5. Identification of Transcription Factors Involved in Lignin Biosynthesis
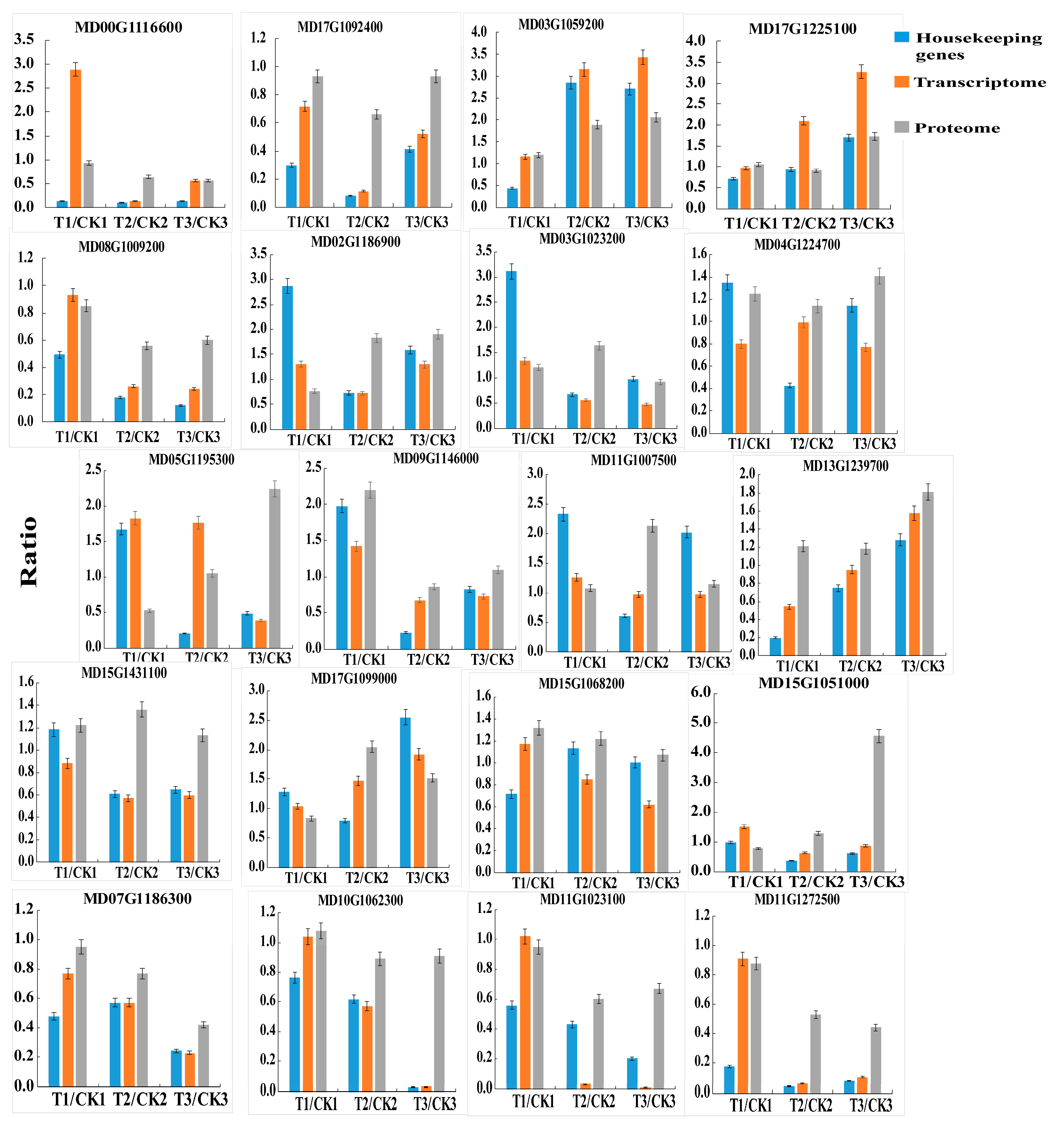
2.6. Quantitative Real-Time PCR Confirmation of Selected Genes
2.7. Functional Identification of MD15G1068200
3. Discussion
3.1. Histologic Change of Russeted Skin
3.2. Changes in Related Genes of Lignin Biosynthesis
3.3. Proteome and Transcriptome Correlation Analysis
3.4. Regulatory Factors of Lignin Biosynthesis Related Genes
4. Materials and Methods
4.1. Plant Materials
4.2. Scanning Electron Microscope for Epidermis Structure
4.3. Light Microscopy for Epidermis Cell
4.4. RNA Extraction and Sequencing
4.5. Protein Extraction, iTRAQ Labeling, and Mass Spectrometry Analysis
4.6. Protein and RNA Correlation Analysis
4.7. Quantitative Real-time PCR Validation
4.8. Functional Association Network
4.9. Phylogenetic Analysis
4.10. Electrophoretic Mobility Shift Assay
4.11. Luciferase Reporter Assays in Nicotiana Benthamiana Leaves
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DAF | days after flowering |
| SEM | scanning electron microscopy |
| LM | light microscope |
| PAL | phenylalanine ammonia-lyase |
| CCR | cinnamyl CoA reductase |
| CAD | cinnamyl-alcohol dehydrogenase |
| POD | peroxidase |
| PRX | peroxidase |
| C4H | cinnamate hydroxylase |
| C3H | coumaric acid 3-hydroxylase |
| 4CL | 4-coumaric acid- CoA ligase |
| PDC | pyruvate decarboxylase |
| HCT | shikimate O-hydroxycinnamoyl transferase |
| PDA | phospholipid: diacylglycerol acyltransferase |
| DEGs | differentially expressed genes |
| DEPs | differentially expressed proteins |
| qPCR | Quantitative real-time PCR |
| iTRAQ | isobaric tags for relative and absolute quantitation |
| RNA-Seq | RNA sequencing |
| NR | NCBI non-redundant protein sequences |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| Ko | KEGG ortholog |
| GO | Gene ontology |
| TF | transcription factor |
| HPLC | high-performance liquid chromatography |
| MS/MS | tandem mass spectrometry |
| NJ | Neighbor-joining |
References and Note
- Bannier, H.J. Moderne Apfelzüchtung: Genetische Verarmung und Tendenzen zur Inzucht. Erwerbs-Obstbau 2011, 52, 85–110. [Google Scholar] [CrossRef]
- Faust, M.; Shear, C.B. Russeting of apples, an interpretive review. Hort. Sci. 1972, 7, 233–235. [Google Scholar]
- Curry, E. Increase in Epidermal Planar Cell Density Accompanies Decreased Russeting of “Golden Delicious” Apples Treated with Gibberellin A4+7. Hort. Sci. 2012, 47, 232–237. [Google Scholar] [CrossRef]
- Khanal, B.P.; Grimm, E.; Knoche, M. Russeting in apple and pear: A plastic periderm replaces a stiff cuticle. AoB PLANTS 2013, 5, pls048. [Google Scholar] [CrossRef] [PubMed]
- Knoche, M.; Khanal, B.P.; Stopar, M. Russeting and microcrackingof “Golden Delicious” apple fruit concomitantly decline due to gibberellin A4+7 application. J. Am. Soc. Hort. Sci. 2011, 136, 159–164. [Google Scholar] [CrossRef]
- Skene, D.S. Cracking and russeting in apple fruits. Report of East Mailing Research Station for 1964. 1965, 99–101.
- Knoche, M.; Grimm, E. Surface moisture induces microcracks in the cuticle of “Golden Delicious” apple. Hort. Sci. 2008, 43, 1929–1931. [Google Scholar] [CrossRef]
- Simons, R.K.; Aubertin, M.P. Development of epidermal, hypodermal and cortical tissues in the Golden Delicious apple as influenced by induced mechanical injury. J. Am. Soc. Hort. Sci. 1959, 74, 1–9. [Google Scholar]
- Duso, C.; Castagnoli, M.; Simoni, S.; Angeli, G. The impact of eriophyoids on crops: Recent issues on aculus schlechtendali, calepitrimerus vitis and aculops lycopersici. Exp. Appl. Acarol. 2010, 51, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.; Soto, J.M.; Uvalle, J.X.; Hernandez, A.P.; Ruiz, J.M.; Romero, L. Chemical treatments in ‘Golden Delicious Spur’ fruits in relation to russeting and nutritional status. J. Plant Nutr. 2001, 24, 191–202. [Google Scholar] [CrossRef]
- Gildemacher, P.; Heijne, B.; Silvestri, M.; Houbraken, J.; Hoekstra, E.; Theelen, B.; Boekhout, T. Interactions between yeasts, fungicides and apple fruit russeting. Fems Yeast Res. 2006, 6, 1149–1156. [Google Scholar] [CrossRef]
- Barandoozi, F.N.; Talaie, A. The effect of gibberellins on russeting in Golden Delicious apples. J. Hortic. 2009, 1, 61–64. [Google Scholar]
- Taylor, B.K. Effects of gibberellin sprays on fruit russet and tree performance of Golden Delicious apple. J. Hortic. Sci. 1978, 55, 167–169. [Google Scholar] [CrossRef]
- Sharma, R.R.; Pal, R.K.; Asrey, R.; Sagar, V.R.; Rana, M.R. Pre-harvest fruit bagging influences fruit color and quality of apple cv. Delicious. Agric. Sci. 2013, 4, 443–448. [Google Scholar] [CrossRef]
- Jing, C. Effect of Bagging on Apple Fruit Coloring and the Expression of Related Genes. Ph.D. Thesis, Northwest Agricultural & Forestry University, Yangling, China, 2017. [Google Scholar]
- Lashbrooke, J.; Aharoni, A.; Costa, F. Genome investigation suggests mdshn3, an apetala2-domain transcription factor gene, to be a positive regulator of apple fruit cuticle formation and an inhibitor of russet development. J. Exp. Bot. 2015, 66, 6579–6589. [Google Scholar] [CrossRef] [PubMed]
- Falginella, L.; Cipriani, G.; Monte, C.; Gregori, R.; Tartarini, S. A major QTL controlling apple skin russeting maps on the linkage group 12 of ‘Renetta Grigia di Torriana’. BMC Plant Biol. 2015, 15, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Simons, R.K. Developmental changes in russet sports of Golden Delicious apples-morphological and anatomical comparison with normal fruit. J. Am. Soc. Hort. Sci. 1960, 76, 41–51. [Google Scholar]
- Alston, F.H.; Watkins, R. Apple breeding at East Malling. In Proceedings of the Eucarpia Fruit Section Symposium V: Top Fruit Breeding, Canterbury; Brown, A.G., Watkins, R., Alston, F.H., Eds.; 1973; pp. 14–29. Available online: https://www.researchgate.net/publication/227041152_Breeding_Apple_Malus_x_Domestica_Borkh (accessed on 9 September 2019).
- Legay, S.; Guerriero, G.; Deleruelle, A.; Lateur, M.; Evers, D.; André, C.M.; Hausman, J.F. Apple russetingas seen through the RNA-seq lens: Strong alterations in the exocarp cell wall. Plant Mol. Biol. 2015, 88, 21–40. [Google Scholar] [CrossRef] [PubMed]
- Legay, S.; Guerriero, G.; André, C.; Guignard, C.; Cocco, E.; Charton, S.; Boutry, M.; Rowland, O.; Hausman, J.F. MdMyb93 is a regulator of suberin deposition in russeted apple fruit skins. New Phytol. 2016, 212, 977–991. [Google Scholar] [CrossRef] [PubMed]
- Lashbrooke, J.; Cohen, H.; Levy-Samocha, D.; Tzfadia, O.; Panizel, I.; Zeisler, V.; Massalha, H.; Stern, A.; Trainotti, L.; Schreiber, L.; et al. Myb107 and myb9 homologs regulate suberin deposition in angiosperms. Plant Cell 2016, 28, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Heng, W.; Liu, L.; Wang, M.D.; Jia, B.; Liu, P.; Ye, Z.F.; Zhu, L.W. Differentially expressed genes related to the formation of russet fruit skin in a mutant of ‘dangshansuli’ pear (pyrus bretchnederirehd.) determined by suppression subtractive hybridization. Euphytica 2014, 196, 285–297. [Google Scholar] [CrossRef]
- Gutierrez, B.L.; Zhong, G.; Brown, S.K. Increased phloridzin content associated with russeting in apple (Malus domestica (Suckow) Borkh.) fruit. Genet. Resour. Crop Evol. 2018, 65, 2135–2149. [Google Scholar] [CrossRef]
- Kawaoka, A.; Kaothien, P.; Yoshida, K.; Endo, S.; Yamada, K.; Ebinuma, H. Functional analysis of tobacco LIM protein Ntliml involved in lignin biosynthesis. Plant J. 2000, 22, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.P.D.; Wamser, A.F. Fruit bagging effect on the damage caused by biotics and abiotics factors in an organic apple orchard. Rev. Bras. Frutic. 2006, 28, 168–171. [Google Scholar] [CrossRef]
- Ma, K.Y.; Cheng, F.H.; Fu, Y.H.; Jia, Y.X.; Chen, J.Y. The Development of Fruit Dot and Russeting in ‘Yali’ Pear. Acta Hortic. Sin. 1995, 22, 295–296. [Google Scholar]
- Zhang, H.Y.; Wang, S.G.; Mou, Q.Y.; Jiang, M.X.; Sun, F.L. Effect of Bagging on the Peel Structure and PPO, POD activity of Pyrus bretschneideri. Acta Hortic. Sin. 1996, 23, 23–26. [Google Scholar]
- Ashizawa, M.; Horigome, Y.; Chujo, T. Histological studies on the cause of russet in “Golden Delicious” apple. Acta Hortic. 2005, 30, 47–52. [Google Scholar]
- Xie, S. Effects of Boron Deficiency on Lignin Content and the Key Gene Expression of Lignin Biosynthesis in Two Kinds Citrus Rootstocks. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2014. [Google Scholar]
- Baucher, M.; Monties, B.; Montagu, M.V.; Boerjan, W. Biosynthesis and genetic engineering of lignin. Crit. Rev. Plant Sci. 1998, 17, 125–197. [Google Scholar] [CrossRef]
- Shi, C.H.; Wang, X.Q.; Zhang, X.Y.; Luo, J. Effects of Bagging on Rust Formation and Physiology of Fruit Exocarp in Pear Variety “Cuiguan”. Acta Agric. Jiangxi 2018, 30, 1–7. [Google Scholar]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Raes, J.; Rohde, A.; Christensen, J.H.; Van de Peer, Y.; Boerjan, W. Genome-wide characterization of the lignification toolbox in Arabidopsis. Plant Physiol. 2003, 133, 1051–1071. [Google Scholar] [CrossRef]
- Rohde, A.; Morreel, K.; Ralph, J.; Goeminne, G.; Hostyn, V.; De Rycke, R.; Kushnir, S.; Van Doorsselaere, J.; Joseleau, J.P.; Vuylsteke, M.; et al. Molecular phenotyping of the pa11and pa12 mutants ofArabidopsis thaliana reveals far-reaching consequences on Phenylpropanoid, Amino Acid and Carbonhydrate Metabolism. Plant Cell 2004, 16, 2479–2771. [Google Scholar] [CrossRef] [PubMed]
- Hoffrnann, L.; Besseau, S.; Geoffroy, P.; Ritzenthaler, C.; Meyer, D.; Lapierre, C.; Pollet, B.; Legrand, M. Silencing of hydroxycinnamoyl-coenzyme A shikimate/quinate hydroxycinnamoyltransferase affects phenylpropanoid biosynthesis. Plant Cell 2004, 16, 1446–1465. [Google Scholar] [CrossRef] [PubMed]
- Fraser, A.C.M.; Chapple, C. The phenylpropanoid pathway in arabidopsis. Arab. Book 2011, 9, e0152. [Google Scholar] [CrossRef] [PubMed]
- Sibout, R. Cinnamyl alcohol dehydrogenase-c and -d are the primary genes involved in lignin biosynthesis in the floral stem of arabidopsis. Plant Cell Online 2005, 17, 2059–2076. [Google Scholar] [CrossRef] [PubMed]
- Cosio, C.; Ranocha, P.; Francoz, E.; Burlat, V.; Zheng, Y.; Perry, S.E.; Ripoll, J.J.; Yanofshy, M.; Dunand, C. The class III peroxidase PRX 17 is a direct target of themMADS-box transcription factor AGAMOUS-L1KE15 (AGL15) and participates in lignified tissue formation. New Phytol. 2017, 213, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kajita, S.; Kawai, S.; Katayama, Y.; Morohoshi, N. Down-regulation of an anionic peroxidase in transgenic aspen and its effect on lignin characteristics. J. Plant Res. 2003, 116, 175–182. [Google Scholar] [CrossRef]
- Ring, L.; Yeh, S.Y.; Hncherig, S.; Hoffmann, T.; Blanco-Portales, R.; Fouche, M.; Villatoro, C.; Denoyes, B.; Monfort, A.; Cabatlero, J.L.; et al. Metabolic interaction between anthocyanin and lignin biosynthesis is associated with peroxidase FaPRX27 in strawberry fruit. Plant Physiol. 2013, 163, 43–60. [Google Scholar] [CrossRef]
- Yeh, S.Y.; Huang, F.C.; Hoffmann, T.; Mayershofer, M.; Schwab, W. FaPOD27 functions in the metabolism of polyphenols in strawberry fruit (Fragaria sp.). Front Plant Sci. 2014, 5, 518. [Google Scholar] [CrossRef]
- Haura, E.B.; Müller, A.; Breitwieser, F.P.; Li, J.; Grebien, F.; Colinge, J.; Bennett, K.L. Using iTRAQ combined with tandem affinity purification to enhance low abundance proteins associated with somatically mutated EGFR core complexes in lung cancer. J. Proteome Res. 2011, 10, 182–190. [Google Scholar] [CrossRef]
- Dowle, A.A.; Wilson, J.; Thomas, J.R. Comparing the diagnostic classification accuracy of iTRAQ, Peak-Area, Spectral- Counting, and emPAI methods for relative quantification in expression proteomics. J. Proteome Res. 2016, 15, 3550–3562. [Google Scholar] [CrossRef]
- Muers, M. Gene expression: Transcriptome to proteome and back to genome. Nat. Rev. Genet. 2011, 12, 518. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, S.S.; Kohler, A.; Yan, B.; Luo, H.M.; Chen, X.M. Itraq and rna-seq analyses provide new insights into regulation mechanism of symbiotic germination of dendrobium officinale seeds (orchidaceae). J. Proteome Res. 2017, 16, 2174. [Google Scholar] [CrossRef] [PubMed]
- Naoumkina, M.A.; Zhao, Q.; Gallego-Giraldo, L.; Dai, X.; Zhao, P.X.; Dixon, R.A. Genome-wide analysis of phenylpropanoid defence pathways. Mol. Plant Pathol. 2010, 11, 829–846. [Google Scholar] [CrossRef]
- Patzlaff, A.; Newman, L.J.; Dubos, C.; Whetten, R.W.; Smith, C.; McInnis, S.; Bevan, M.W.; Sederoff, R.R.; Campbell, M.M. Characterisation of PtMYB1, an R2R3-MYB from pine xylem. Plant Mol. Biol. 2003, 53, 597–608. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, R.L.; Zhong, R.; Fowler, S.; Lyskowski, D.; Piyasena, H.; Carleton, K.; Spicer, C.; Ye, Z.H. The poplar MYB transcription factors, PtrMYB3 and PtrMYB20, are involved in the regulation of secondary wall biosynthesis. Plant Cell Physiol. 2010, 51, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Choi, H.; An, G. Roles of lignin biosynthesis and regulatory genes in plant development. J Integr. Plant Biol. 2015, 57, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Lee, C.; Zhong, R.; Ye, Z.H. Myb58 and myb63 are transcriptional activators of the lignin biosynthetic pathway during secondary cell wall formation in arabidopsis. Plant Cell 2009, 21, 248–266. [Google Scholar] [CrossRef] [PubMed]
- Hatton, D.; Sablowski, R.; Yung, M.H.; Smith, C.; Schuch, W.; Bevan, M. Two classes of cis sequences contribute to tissue-specific expression of a pal2 promoter in transgenic tobacco. Plant J. 1995, 7, 859–876. [Google Scholar] [CrossRef]
- Bell-Lelong, D.A. Cinnamate-4-hydroxylase expression in arabidopsis. regulation in response to development and the environment. Plant Physiol. 1997, 113, 729–738. [Google Scholar] [CrossRef]
- Li, T. The Study on the Transcriptional Regulation of Lignin Biosynthesis and the Recognition of Interaction Protein by Sorghum SbLIM1. Master’s Thesis, Shandong University, Jinan, China, 2017. [Google Scholar]
- Zhou, J.P. Phenotypic Changes Caused by Overexpressing a LIM Domain Gene in Cotton. Master’s Thesis, Southwest University, Chongqing, China, 2006. [Google Scholar]
- Sun, S.; Xin, L.; Gao, H.; Wang, J.; Li, P. Response of phenolic compounds in “Golden Delicious” and ‘Red Delicious’ apples peel to fruit bagging and subsequent sunlight re-exposure. Sci. Hortic-Amst. 2014, 168, 161–167. [Google Scholar] [CrossRef]
- Min, T.; Bao, Y.Q.; Zhou, B.X.; Yi, Y.; Wang, L.M.; Hou, W.F.; Ai, Y.W.; Wang, H.X. RTranscription Profiles Reveal the Regulatory Synthesis of Phenols during the Development of Lotus Rhizome (Nelumbo nucifera Gaertn). Int. J. Mol. Sci. 2019, 20, 2735. [Google Scholar] [CrossRef] [PubMed]
- Wen, B.; Zhou, R.; Feng, Q.; Wang, Q.; Wang, J.; Liu, S. IQuant: An automated pipeline for quantitative proteomics based upon isobaric tags. Proteomics 2014, 14, 2280–2285. [Google Scholar] [CrossRef] [PubMed]
- Savitski, M.M.; Wilhelm, M.; Hahne, H.; Kuster, B.; Bantscheff, M. A scalable approach for protein false discovery rate estimation in large proteomic data sets. Mol. Cell Proteom. 2015, 14, 2394–2404. [Google Scholar] [CrossRef] [PubMed]
- Perez-Riverol, Y.; Csordas, A.; Bai, J.; Bernal-Llinares, M.; Hewapathirana, S.; Kundu, D.J.; Inuganti, A.; Griss, J.; Mayer, G.; Eisenacher, M.; et al. The PRIDE database and related tools and resources in 2019: Improving support for quantification data. Nucleic Acids Res. 2019, 47, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.; Higgins, D.G. Multiple sequence alignment using ClustalW and ClustalX. Curr. Protoc Bioinform. 2002, 00, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]

| Gene ID | Gene Length (bp) | Log2(T/CK) | Description | Ko Number |
|---|---|---|---|---|
| MD01G1042500 | 2011 | −2.41 | CAD, cinnamyl-alcohol dehydrogenase [EC:1.1.1.195] | K00083 |
| MD01G1042800 | 1779 | −2.66 | CAD, cinnamyl-alcohol dehydrogenase [EC:1.1.1.195] | K00083 |
| MD02G1161000 | 2472 | −2.02 | PDA, phospholipid: diacylglycerol acyltransferase [EC:2.3.1.158] | K00679 |
| MD03G1126700 | 5941 | −1.39 | PDC, pyruvate decarboxylase [EC:4.1.1.1] | K01568 |
| MD04G1096200 | 3458 | −1.81 | PAL, phenylalanine ammonia-lyase [EC:4.3.1.24] | K10775 |
| MD06G1194000 | 1697 | −2.24 | UBR4, E3 ubiquitin-protein ligase [EC:2.3.2.27] | K10691 |
| MD11G1153800 | 3440 | −1.56 | PDA, phospholipid: diacylglycerol acyltransferase [EC:2.3.1.158] | K00679 |
| MD11G1210200 | 1645 | −2.72 | UBR4, E3 ubiquitin-protein ligase UBR4 [EC:2.3.2.27] | K10691 |
| MD11G1214800 | 2056 | −1.86 | PDA, phospholipid: diacylglycerol acyltransferase [EC:2.3.1.158] | K00679 |
| MD12G1252500 | 2161 | 1.32 | PDA, phospholipid: diacylglycerol acyltransferase [EC:2.3.1.158] | K00679 |
| MD17G1157600 | 2990 | −2.25 | HCT, shikimate O-hydroxycinnamoyl transferase [EC:2.3.1.133] | K13065 |
| Gene ID | Gene Length (bp) | Log2(T/CK) | Log2(RAE/WAE) | Description | Ko Number |
|---|---|---|---|---|---|
| MD03G1073600 | 1904 | −2.79 | 3.97 | CYP86A1, long-chain fatty acid omega-monooxygenase [EC:1.14.14.80] | K15401 |
| MD08G1058900 | 1808 | 1.67 | - | HHT1, omega-hydroxypalmitate O-feruloyl transferase [EC:2.3.1.188] | K15400 |
| MD09G1007600 | 1797 | −2.23 | - | HHT1, omega-hydroxypalmitate O-feruloyl transferase [EC:2.3.1.188] | K15400 |
| MD13G1004700 | 2106 | −2.84 | 4.75 | CYP86B1, fatty acid omega-hydroxylase [EC:1.14.-.-] | K15402 |
| MD17G1011500 | 1790 | −2.27 | - | HHT1, omega-hydroxypalmitate O-feruloyl transferase [EC:2.3.1.188] | K15400 |
| Protein ID | Mass (kD) | Mean Ratio (T/CK) | Description | Ko ID |
|---|---|---|---|---|
| MD01G1205400 | 33.79 | 0.60 | At4g11410, WW domain-containing oxidoreductase | K19329 |
| MD02G1009300 | 53.71 | 0.67 | At2g24270, glyceraldehyde-3-phosphate dehydrogenase (NADP+) [EC:1.2.1.9] | K00131 |
| MD02G1157800 | 35.27 | 0.58 | -, photosystem II oxygen-evolving enhancer protein 1 | K02716 |
| MD05G1122100 | 42.35 | 0.65 | At1g68010, hydroxypyruvate reductase 1 [EC:1.1.1.29] | K15893 |
| MD05G1209300 | 40.59 | 0.63 | At3g14420, (S)-2-hydroxy-acid oxidase [EC:1.1.3.15] | K11517 |
| MD08G1007200 | 16.92 | 0.67 | -, plastocyanin | K02638 |
| MD08G1168600 | 48.75 | 0.60 | At5g08640, flavonol synthase [EC:1.14.20.6] | K05278 |
| MD09G1050500 | 49.97 | 0.74 | At5g39830, HtrA serine peptidase 2 [EC:3.4.21.108] | K08669 |
| MD09G1252100 | 20.72 | 0.58 | -, ribulose-bisphosphate carboxylase [EC:4.1.1.39] | K01602 |
| MD09G1262800 | 41.17 | 0.65 | At1g66430, fructokinase [EC:2.7.1.4] | K00847 |
| MD10G1011900 | 19.93 | 0.59 | At4g03520, thioredoxin 1 | K03671 |
| MD10G1195700 | 52.49 | 0.55 | At3g14420, (S)-2-hydroxy-acid oxidase [EC:1.1.3.15] | K11517 |
| MD14G1219000 | 41.45 | 0.69 | -, chitinase domain-containing protein 1 | K17525 |
| MD15G1006300 | 16.86 | 0.72 | -, plastocyanin | K02638 |
| MD15G1222600 | 23.68 | 0.64 | At3g26060, peroxiredoxin Q/BCP [EC:1.11.1.15] | K03564 |
| MD15G1272500 | 35.24 | 0.53 | -, photosystem II oxygen-evolving enhancer | K02716 |
| MD15G1307200 | 56.88 | 0.53 | At4g37930, glycine hydroxymethyl transferase | K00600 |
| MD15G1421500 | 27.39 | 0.69 | -, exocyst complex component 2 | - |
| MD17G1020900 | 32.92 | 0.67 | At4g24770, nucleolin | K11294 |
| MD17G1132900 | 28.77 | 0.56 | -, photosystem II oxygen-evolving enhancer | K02717 |
| ID | Protein | Gene | Description | Ko ID |
|---|---|---|---|---|
| T/CK | Log2(T/CK) | |||
| MD00G1116600 | 1.72 | −1.77 | UBR4, E3 ubiquitin-protein ligase | K00679 |
| MD03G1059200 | 0.67 | 1.31 | PRX, Peroxidase | K00430 |
| MD08G1009200 | 0.84 | −1.21 | CAD, Cinnamyl-alcohol dehydrogenase | K00083 |
| MD17G1092400 | 1.42 | −1.85 | PRX, Peroxidase | K00430 |
| MD17G1225100 | 1.00 | 1.12 | HCT, Shikimate O-hydroxycinnamoyl transferase | K13065 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, G.; Bian, S.; Han, X.; He, S.; Liu, K.; Zhang, C.; Cong, P. An Integrated Transcriptome and Proteome Analysis Reveals New Insights into Russeting of Bagging and Non-Bagging “Golden Delicious” Apple. Int. J. Mol. Sci. 2019, 20, 4462. https://doi.org/10.3390/ijms20184462
Yuan G, Bian S, Han X, He S, Liu K, Zhang C, Cong P. An Integrated Transcriptome and Proteome Analysis Reveals New Insights into Russeting of Bagging and Non-Bagging “Golden Delicious” Apple. International Journal of Molecular Sciences. 2019; 20(18):4462. https://doi.org/10.3390/ijms20184462
Chicago/Turabian StyleYuan, Gaopeng, Shuxun Bian, Xiaolei Han, Shanshan He, Kai Liu, Caixia Zhang, and Peihua Cong. 2019. "An Integrated Transcriptome and Proteome Analysis Reveals New Insights into Russeting of Bagging and Non-Bagging “Golden Delicious” Apple" International Journal of Molecular Sciences 20, no. 18: 4462. https://doi.org/10.3390/ijms20184462
APA StyleYuan, G., Bian, S., Han, X., He, S., Liu, K., Zhang, C., & Cong, P. (2019). An Integrated Transcriptome and Proteome Analysis Reveals New Insights into Russeting of Bagging and Non-Bagging “Golden Delicious” Apple. International Journal of Molecular Sciences, 20(18), 4462. https://doi.org/10.3390/ijms20184462




