Stem Cells from a Female Rat Model of Type 2 Diabetes/Obesity and Stress Urinary Incontinence Are Damaged by In Vitro Exposure to its Dyslipidemic Serum, Predicting Inadequate Repair Capacity In Vivo
Abstract
1. Introduction
2. Results
2.1. miR-GTS showed Key Differences Between the Female and Male MDSC, and Between the Female MDSC from T2D/O and Normal Rats, Suggesting that miR-GTS might be MDSC Identity Biomarkers
2.2. The Female ZF4-SC miR-GTS for Myostatin-Related miRs and some Unrelated miRs were Affected by in vitro Exposure to Dyslipidemic Serum Similarly to the Reported Male ED-MDSC miRs, but the ZL4-SC Were Less Sensitive.
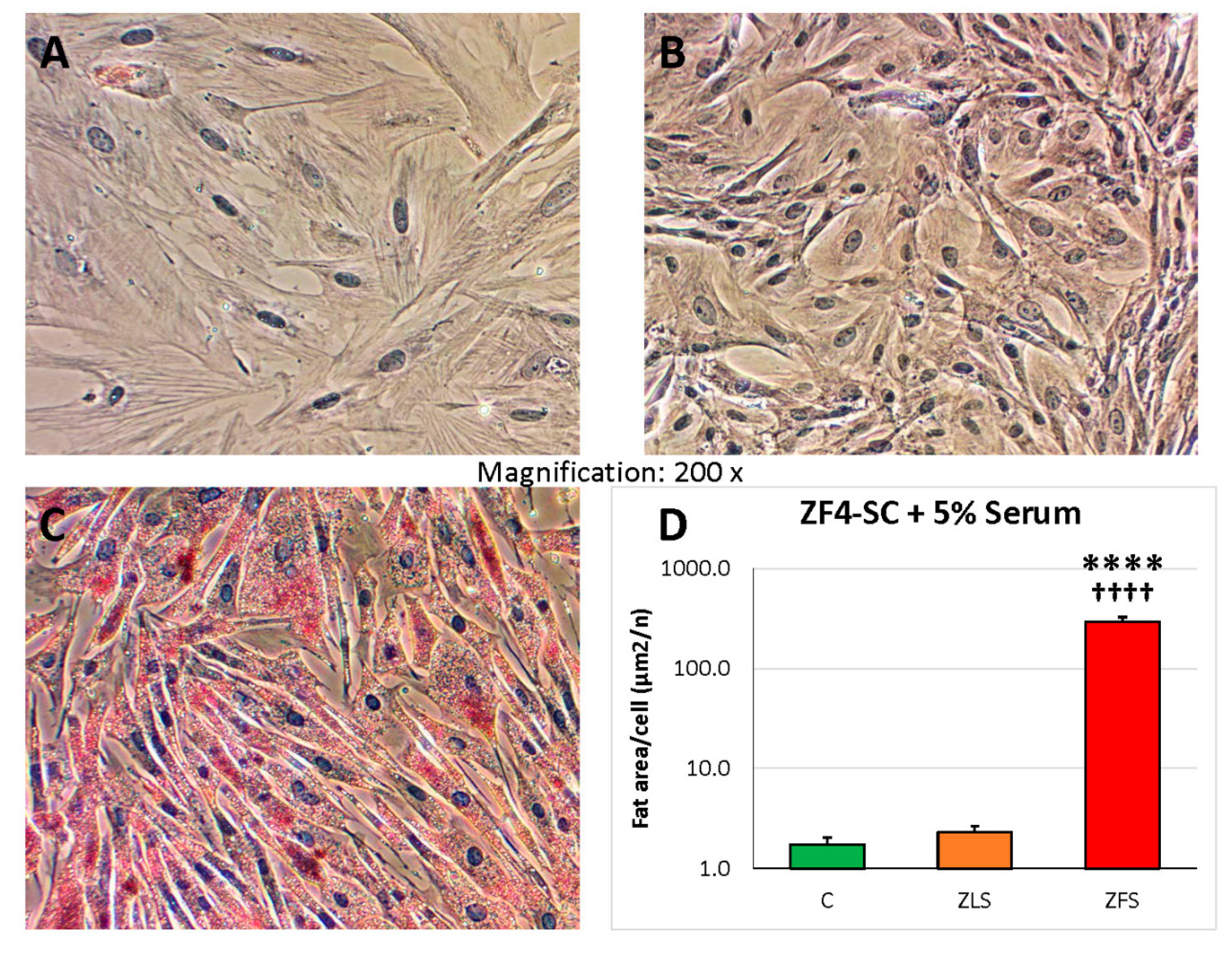
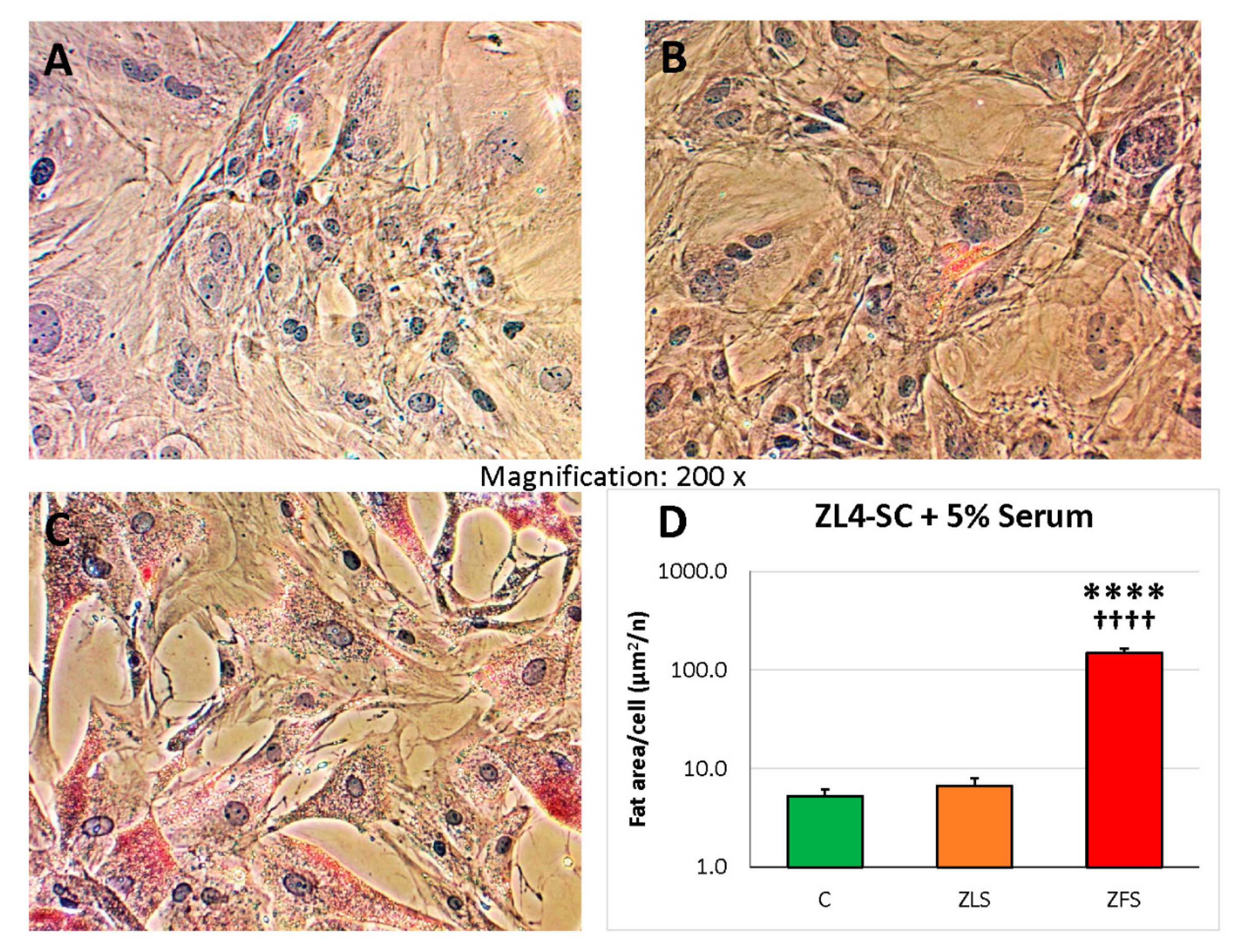
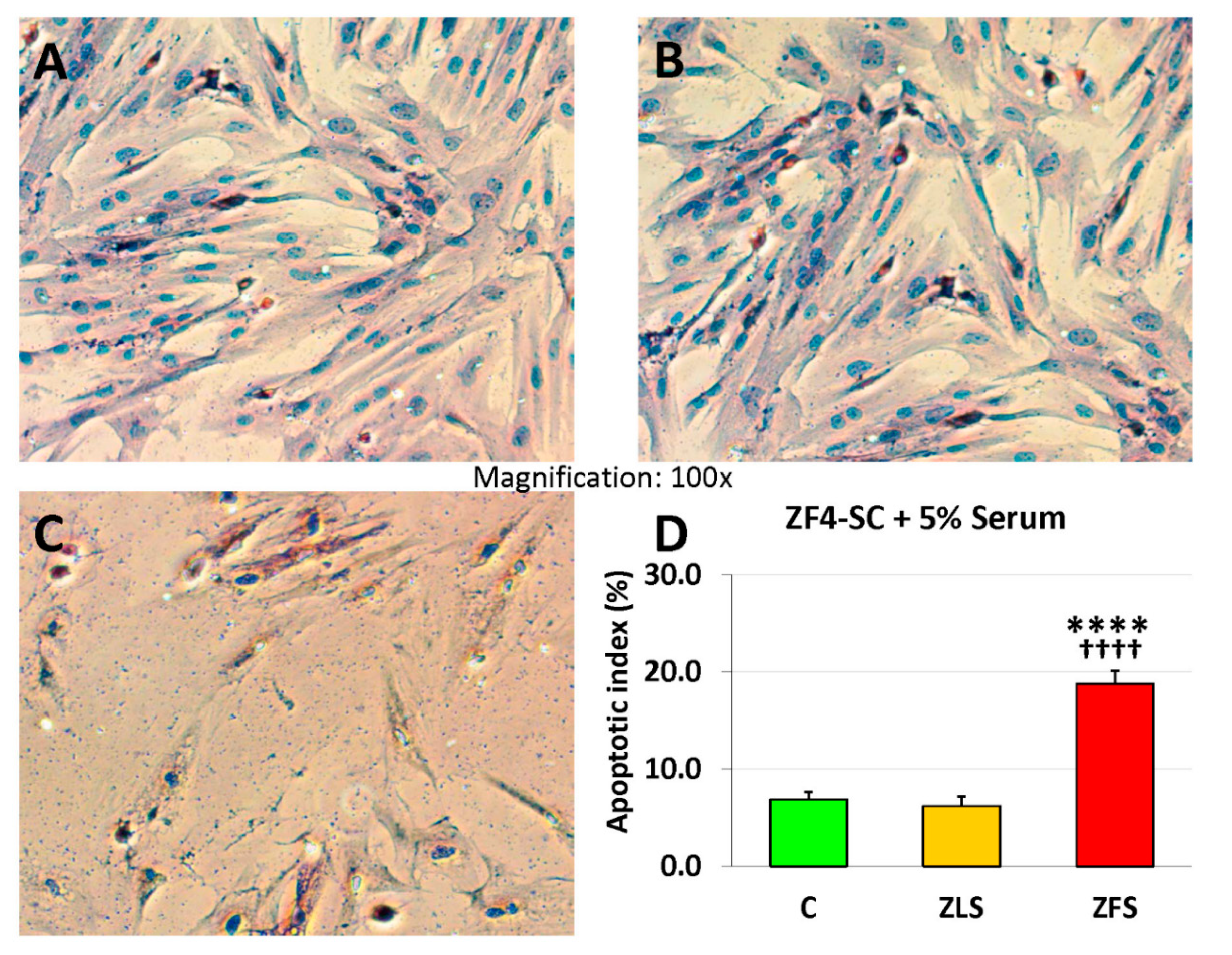
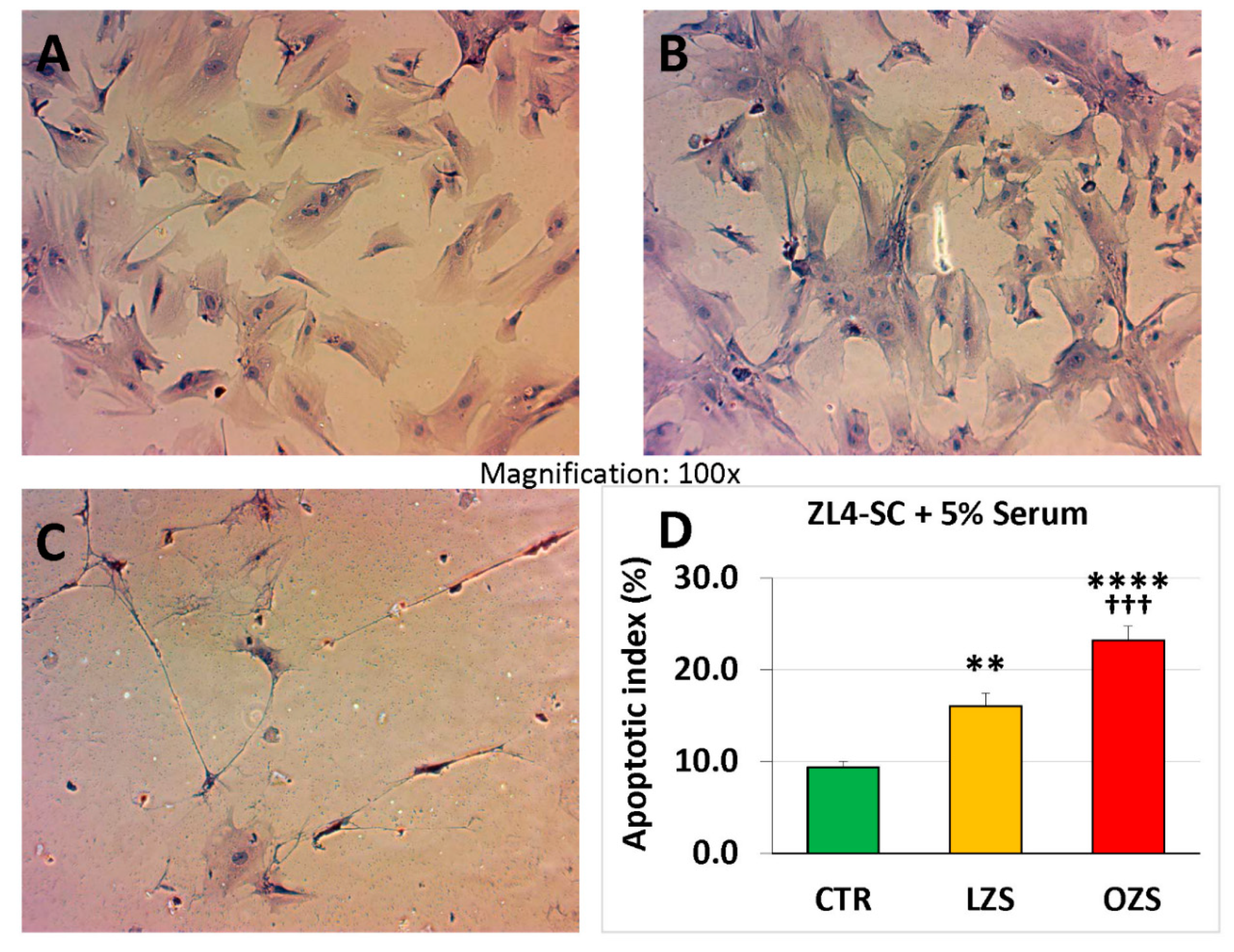
2.3. miR-GTS Changes in the ZF4-SC by ZFS were Accompanied by Intracellular Fat Deposits and Apoptosis that in the ZL4-SC Occurred Similarly to ZF4-SC for Fat Deposits, and less for Apoptosis
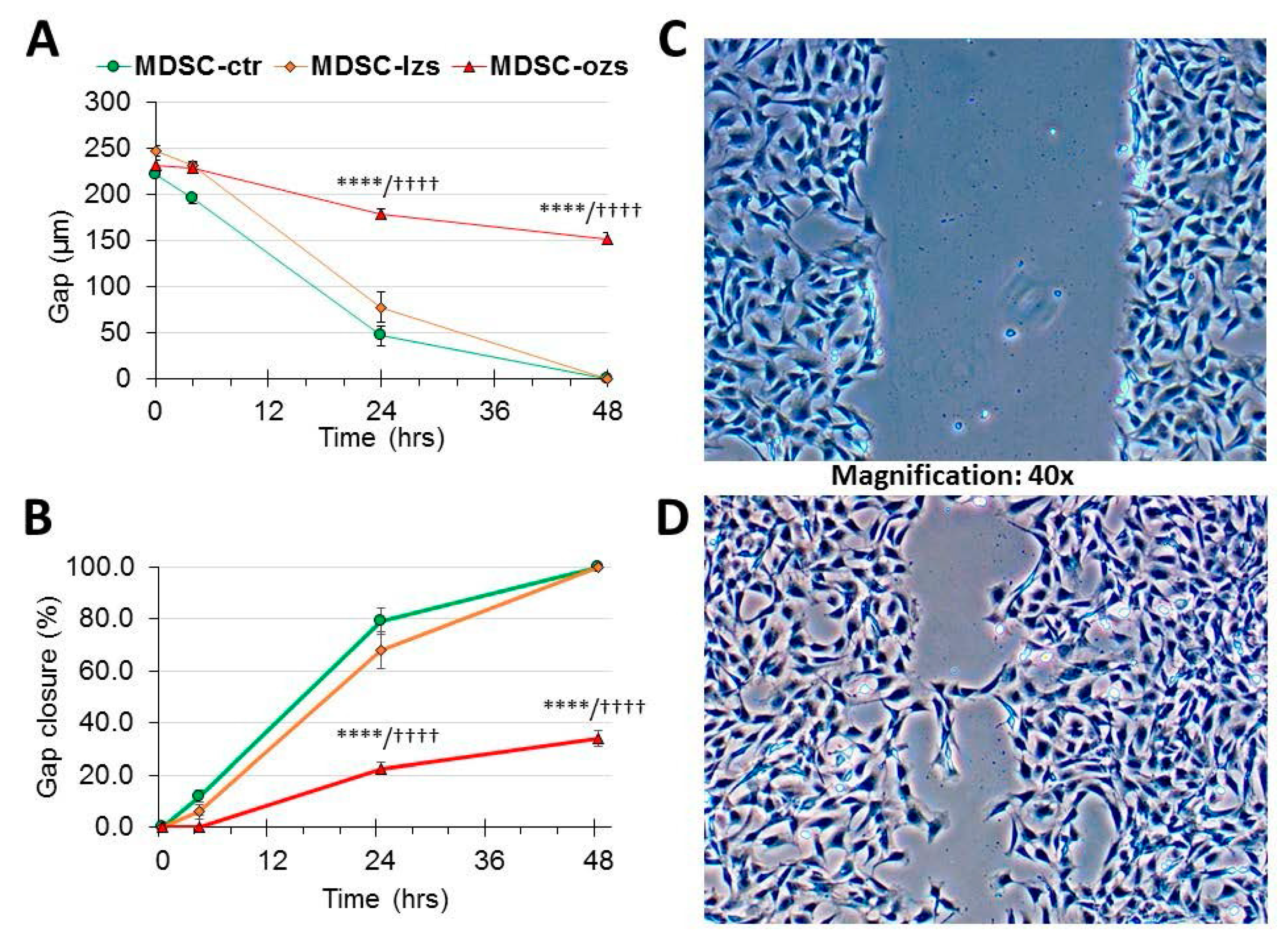
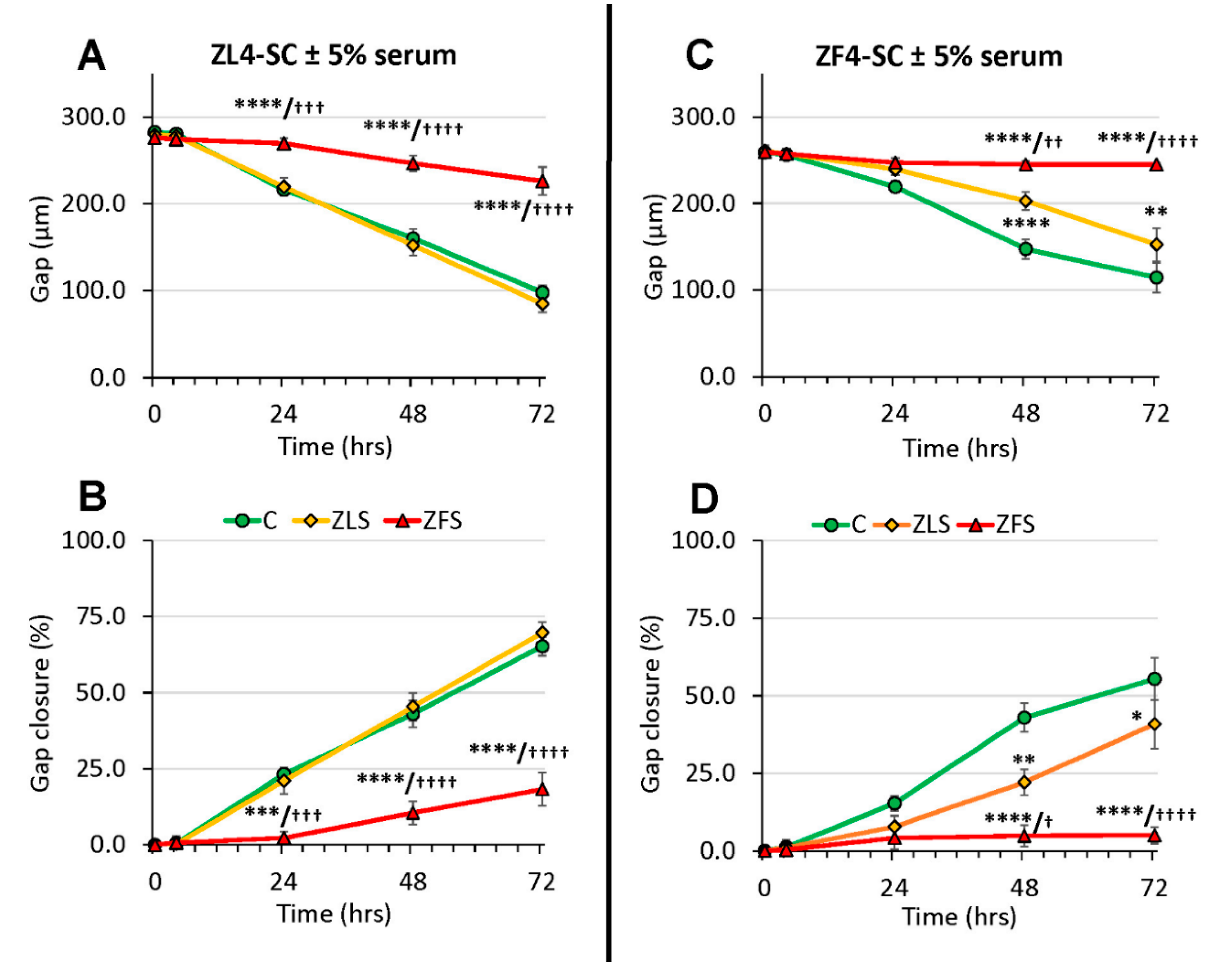
2.4. The Female Stem Cell Damage Exerted in vitro by ZFS was also Accompanied by Inhibition of in Vitro Scratch Healing Repair in both the Male and Female MDSC, with the ZF4-SC being the Most Affected and the Male MDSC the Most Resistant
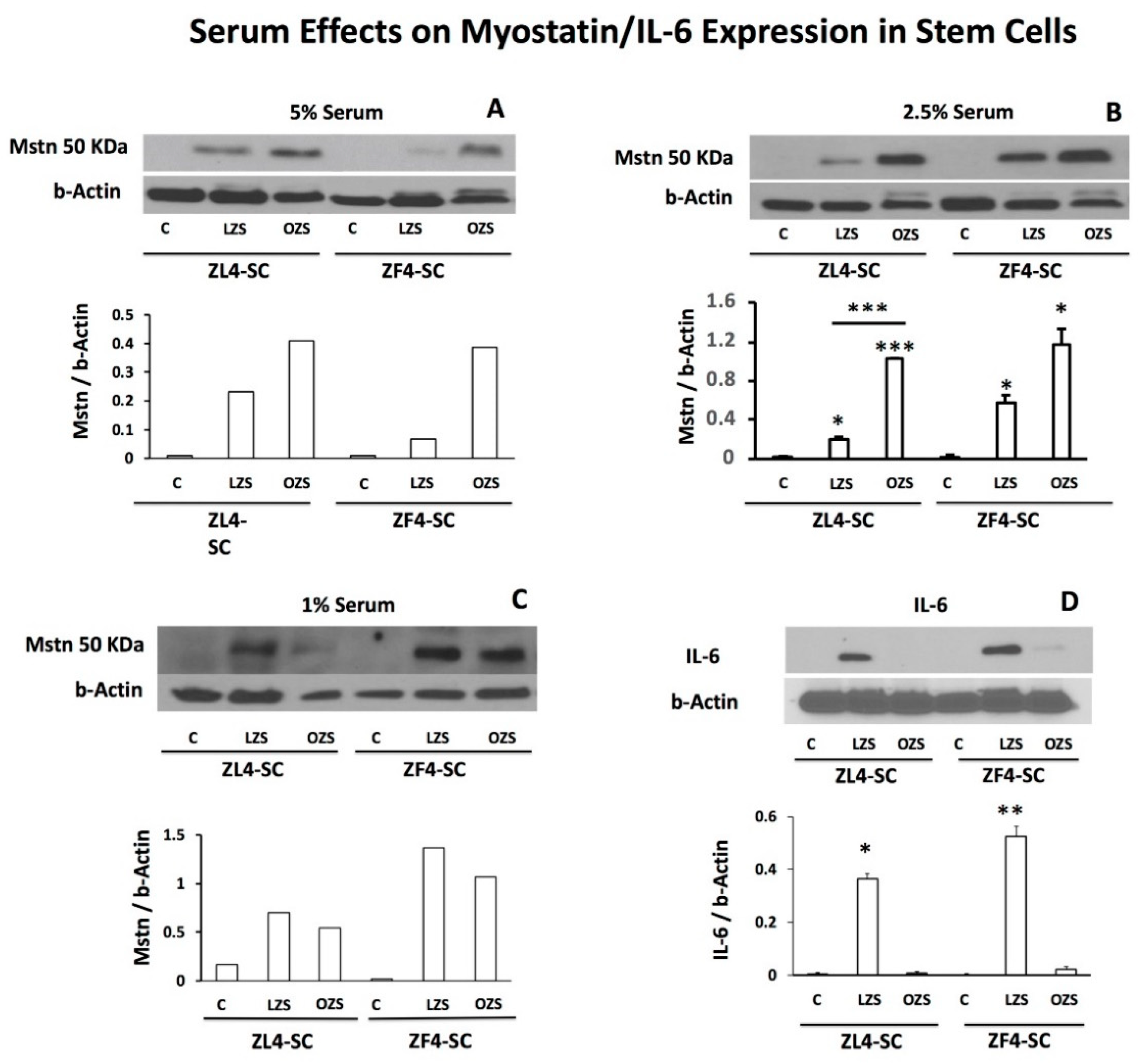
2.5. ZFS Induced in vitro Myostatin Over-Expression in both the ZF4-SC and ZL4-SC, as Previously Reported for the Male ED-MDSC, and this was Accompanied by the Inhibition of Interleukin-6, a Myostatin Counteractive Agent
3. Discussion
4. Materials and Methods
4.1. Stem Cells and Serum Isolation
4.2. MDSC Incubations and Scratch Wound Assay
4.3. Quantitative Histochemistry
4.4. Western Blots
4.5. Global miR-GTS
4.6. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| C or ctr | incubation controls without any rat serum added |
| ED-MDSC | early diabetes MDSC from male 12 weeks old Zucker obese fatty (ZF) rats (Crl:ZUC Leprfa) with mild hyperglycemia/dyslipidemia and moderate overweight |
| Gene-GTS | gene global transcriptional signature |
| lzs | serum from late T2D/O 32 weeks old male LZ rats |
| LD-MDSC | late diabetes MDSC from male 32 weeks old ZF rats, with moderate hyperglycemia, high dyslipidemia, and morbid obesity |
| MDSC | muscle derived stem cells |
| miR | microRNA |
| miR-GTS | miR global transcriptional signature |
| ozs | serum from late T2D/O 32 weeks old male OZ rats |
| QIA | quantitative image analysis |
| T2D/O | type 2 diabetes mellitus/obesity |
| ZF | Zucker obese fatty rats |
| ZFS | serum from late T2D/O 24 weeks old (female) ZF rats |
| ZF4-SC | stem cells from 12 weeks old female ZF rats |
| ZL | Zucker lean rats |
| ZLS | serum from 24 weeks old female ZL rats |
| ZL4-SC | stem cells from 12 weeks old female ZL rats |
References
- Aoki, Y.; Brown, H.W.; Brubaker, L.; Cornu, J.N.; Daly, J.O.; Cartwright, R. Correction: Urinary incontinence in women. Nat. Rev. Dis. Primers 2017, 3, 17097. [Google Scholar] [CrossRef]
- Capobianco, G.; Madonia, M.; Morelli, S.; Dessole, F.; De Vita, D.; Cherchi, P.L.; Dessole, S. Management of female stress urinary incontinence: A care pathway and update. Maturitas 2018, 109, 32–38. [Google Scholar] [CrossRef]
- Lukacz, E.S.; Santiago-Lastra, Y.; Albo, M.E.; Brubaker, L. Urinary Incontinence in Women. JAMA 2017, 318, 1592–1604. [Google Scholar] [CrossRef]
- Bennington, J.; Williams, J.K.; Andersson, K.-E. New concepts in regenerative medicine approaches to the treatment of female stress urinary incontinence. Curr. Opin. Urol. 2019, 29, 380–384. [Google Scholar] [CrossRef]
- Thomaz, R.P.; Colla, C.; Darski, C.; Paiva, L.L. Influence of pelvic floor muscle fatigue on stress urinary incontinence: A systematic review. Int. Urogynecol. J. 2018, 29, 197–204. [Google Scholar] [CrossRef]
- Phelan, S.; Kanaya, A.M.; Subak, L.L.; Hogan, P.E.; Espeland, M.A.; Wing, R.R.; Burgio, K.L.; DiLillo, V.; Gorin, A.A.; West, D.S.; et al. Prevalence and Risk Factors for Urinary Incontinence in Overweight and Obese Diabetic Women: Action for Health in Diabetes (Look AHEAD) study. Diabetes Care 2009, 32, 1391–1397. [Google Scholar] [CrossRef]
- Fuselier, A.; Hanberry, J.; Margaret Lovin, J.; Gomelsky, A. Obesity and Stress Urinary Incontinence: Impact on Pathophysiology and Treatment. Curr. Urol. Rep. 2018, 19, 10. [Google Scholar] [CrossRef]
- Kobashi, K.C.; Albo, M.E.; Dmochowski, R.R.; Ginsberg, D.A.; Goldman, H.B.; Gomelsky, A.; Kraus, S.R.; Sandhu, J.S.; Shepler, T.; Treadwell, J.R.; et al. Surgical Treatment of Female Stress Urinary Incontinence: AUA/SUFU Guideline. J. Urol. 2017, 198, 875–883. [Google Scholar] [CrossRef]
- Amend, B.; Vaegler, M.; Fuchs, K.; Mannheim, J.G.; Will, S.; Kramer, U.; Hart, M.L.; Feitz, W.; Chapple, C.; Stenzl, A.; et al. Regeneration of Degenerated Urinary Sphincter Muscles: Improved Stem Cell-Based Therapies and Novel Imaging Technologies. Cell Transplant. 2015, 24, 2171–2183. [Google Scholar] [CrossRef]
- Aragón, I.M.; Imbroda, B.H.; Lara, M.F. Cell Therapy Clinical Trials for Stress Urinary Incontinence: Current Status and Perspectives. Int. J. Med. Sci. 2018, 15, 195–204. [Google Scholar] [CrossRef]
- Sharifiaghdas, F.; Tajalli, F.; Taheri, M.; Naji, M.; Moghadasali, R.; Aghdami, N.; Baharvand, H.; Azimian, V.; Jaroughi, N. Effect of autologous muscle-derived cells in the treatment of urinary incontinence in female patients with intrinsic sphincter deficiency and epispadias: A prospective study. Int. J. Urol. 2016, 23, 581–586. [Google Scholar] [CrossRef]
- Kuismanen, K.; Sartoneva, R.; Haimi, S.; Mannerström, B.; Tomás, E.; Miettinen, S.; Nieminen, K. Autologous Adipose Stem Cells in Treatment of Female Stress Urinary Incontinence: Results of a Pilot Study. STEM CELLS Transl. Med. 2014, 3, 936–941. [Google Scholar] [CrossRef]
- Rania, H.M. Stem Cell Therapy for Treatment of Female Stress Urinary Incontinence. Available online: https://www.clinicaltrials.gov/ (accessed on 30 June 2019).
- Tran, C.; Damaser, M.S. The potential role of stem cells in the treatment of urinary incontinence. Ther. Adv. Urol. 2015, 7, 22–40. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, H.; Liu, X.; Liu, L.; Liang, Z. Inhibition of Fibroblast Differentiation of Muscle-Derived Stem Cells in Cell Implantation Treatment of Stress Urinary Incontinence. Cell. Reprogram. 2011, 13, 459–464. [Google Scholar] [CrossRef]
- Xu, Y.; Song, Y.F.; Lin, Z.X. Transplantation of muscle-derived stem cells plus biodegradable fibrin glue restores the urethral sphincter in a pudendal nerve-transected rat model. Braz. J. Med. Biol. Res 2010, 43, 1076–1083. [Google Scholar] [CrossRef]
- Kwon, D.; Kim, Y.; Pruchnic, R.; Jankowski, R.; Usiene, I.; de Miguel, F.; Huard, J.; Chancellor, M.B. Periurethral cellular injection: Comparison of muscle-derived progenitor cells and fibroblasts with regard to efficacy and tissue contractility in an animal model of stress urinary incontinence. Urology 2006, 68, 449–454. [Google Scholar] [CrossRef]
- Masouminia, M.; Gelfand, R.; Kovanecz, I.; Vernet, D.; Tsao, J.; Salas, R.; Castro, K.; Loni, L.; Rajfer, J.; Gonzalez-Cadavid, N.F. Dyslipidemia Is a Major Factor in Stem Cell Damage Induced by Uncontrolled Long-Term Type 2 Diabetes and Obesity in the Rat, as Suggested by the Effects on Stem Cell Culture. J. Sex. Med. 2018, 15, 1678–1697. [Google Scholar] [CrossRef]
- Kovanecz, I.; Vernet, D.; Masouminia, M.; Gelfand, R.; Loni, L.; Aboagye, J.; Tsao, J.; Rajfer, J.; Gonzalez-Cadavid, N.F. Implanted Muscle-Derived Stem Cells Ameliorate Erectile Dysfunction in a Rat Model of Type 2 Diabetes, but Their Repair Capacity Is Impaired by Their Prior Exposure to the Diabetic Milieu. J. Sex. Med. 2016, 13, 786–797. [Google Scholar] [CrossRef]
- Kovanecz, I.; Masouminia, M.; Gelfand, R.; Vernet, D.; Rajfer, J.; Gonzalez-Cadavid, N.F. Myostatin, a profibrotic factor and the main inhibitor of striated muscle mass, is present in the penile and vascular smooth muscle. Int. J. Impot. Res. 2017, 29, 194–201. [Google Scholar] [CrossRef]
- Wang, L.; Lin, G.; Lee, Y.-C.; Reed-Maldonado, A.B.; Sanford, M.T.; Wang, G.; Li, H.; Banie, L.; Xin, Z.; Lue, T.F. Transgenic animal model for studying the mechanism of obesity-associated stress urinary incontinence. BJU Int. 2017, 119, 317–324. [Google Scholar] [CrossRef]
- Toblli, J.E.; Cao, G.; Giani, J.F.; Angerosa, M.; Dominici, F.P.; Gonzalez-Cadavid, N.F. Antifibrotic Effects of Pioglitazone at Low Doses on the Diabetic Rat Kidney Are Associated with the Improvement of Markers of Cell Turnover, Tubular and Endothelial Integrity, and Angiogenesis. Kidney Blood Press. Res. 2011, 34, 20–33. [Google Scholar] [CrossRef]
- Toblli, J.E.; Ferrini, M.G.; Cao, G.; Vernet, D.; Angerosa, M.; Gonzalez-Cadavid, N.F. Antifibrotic effects of pioglitazone on the kidney in a rat model of type 2 diabetes mellitus. Nephrol. Dial. Transplant. 2009, 24, 2384–2391. [Google Scholar] [CrossRef][Green Version]
- Kovanecz, I.; Nolazco, G.; Ferrini, M.G.; Toblli, J.E.; Heydarkhan, S.; Vernet, D.; Rajfer, J.; Gonzalez-Cadavid, N.F. Early onset of fibrosis within the arterial media in a rat model of type 2 diabetes mellitus with erectile dysfunction. BJU Int. 2009, 103, 1396–1404. [Google Scholar] [CrossRef]
- Lee, Y.-C.; Lin, G.; Wang, G.; Reed-Maldonado, A.; Lu, Z.; Wang, L.; Banie, L.; Lue, T.F. Impaired contractility of the circular striated urethral sphincter muscle may contribute to stress urinary incontinence in female zucker fatty rats. Neurourol. Urodyn. 2017, 36, 1503–1510. [Google Scholar] [CrossRef]
- Tsao, J.; Vernet, D.A.; Gelfand, R.; Kovanecz, I.; Nolazco, G.; Bruhn, K.W.; Gonzalez-Cadavid, N.F. Myostatin genetic inactivation inhibits myogenesis by muscle-derived stem cells in vitro but not when implanted in the mdx mouse muscle. Stem Cell Res. Ther. 2013, 4, 4. [Google Scholar] [CrossRef]
- Justus, C.R.; Leffler, N.; Ruiz-Echevarria, M.; Yang, L.V. In vitro Cell Migration and Invasion Assays. J. Vis. Exp. 2014, 8. [Google Scholar] [CrossRef]
- Pinto, B.I.; Cruz, N.D.; Lujan, O.R.; Propper, C.R.; Kellar, R.S. In Vitro Scratch Assay to Demonstrate Effects of Arsenic on Skin Cell Migration. J. Vis. Exp. 2019, 144. [Google Scholar] [CrossRef]
- Walter, M.N.M.; Wright, K.T.; Fuller, H.R.; MacNeil, S.; Johnson, W.E.B. Mesenchymal stem cell-conditioned medium accelerates skin wound healing: An in vitro study of fibroblast and keratinocyte scratch assays. Exp. Cell Res. 2010, 316, 1271–1281. [Google Scholar] [CrossRef]
- Huh, J.Y. The role of exercise-induced myokines in regulating metabolism. Arch. Pharm. Res. 2018, 41, 14–29. [Google Scholar] [CrossRef]
- Li, F.; Li, Y.; Duan, Y.; Hu, C.-A.A.; Tang, Y.; Yin, Y. Myokines and adipokines: Involvement in the crosstalk between skeletal muscle and adipose tissue. Cytokine Growth Factor Rev. 2017, 33, 73–82. [Google Scholar] [CrossRef]
- Lightfoot, A.P.; Cooper, R.G. The role of myokines in muscle health and disease. Curr. Opin. Rheumatol. 2016, 28, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.-L.; Chang, A.-C.; Huang, C.-C.; Tsai, C.-H.; Lin, C.-C.; Tang, C.-H. Myostatin Promotes Interleukin-1β Expression in Rheumatoid Arthritis Synovial Fibroblasts through Inhibition of miR-21-5p. Front. Immunol. 2017, 8, 1747. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Yu, C.; Shi, L.; Li, D.; Chen, X.; Xia, D.; Zhou, J.; Xu, W.; Ma, C.; Gu, L.; et al. MicroRNA-199a-5p aggravates primary hypertension by damaging vascular endothelial cells through inhibition of autophagy and promotion of apoptosis. Exp. Ther. Med. 2018, 16, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.J.; Kelly, G.; Sengupta, A.; Heydendael, W.; Nicholas, B.; Beltrami, S.; Luz, S.; Peixoto, L.; Abel, T.; Bhatnagar, S. MicroRNAs as biomarkers of resilience or vulnerability to stress. Neuroscience 2015, 305, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Ge, G.; Yang, D.; Tan, Y.; Chen, Y.; Jiang, D.; Jiang, A.; Li, Q.; Liu, Y.; Zhong, Z.; Li, X.; et al. miR-10b-5p Regulates C2C12 Myoblasts Proliferation and Differentiation. Biosci. Biotech. Biochem. 2019, 83, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Dissaranan, C.; Cruz, M.A.; Kiedrowski, M.J.; Balog, B.M.; Gill, B.C.; Penn, M.S.; Goldman, H.B.; Damaser, M.S. Rat Mesenchymal Stem Cell Secretome Promotes Elastogenesis and Facilitates Recovery from Simulated Childbirth Injury. Cell Transplant. 2014, 23, 1395–1406. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, N.; Tamaki, T.; Hirata, M.; Soeda, S.; Nitta, M.; Hoshi, A.; Terachi, T. Purified Human Skeletal Muscle-Derived Stem Cells Enhance the Repair and Regeneration in the Damaged Urethra. Transplantation 2017, 101, 2312–2320. [Google Scholar] [CrossRef]
- Payab, M.; Goodarzi, P.; Foroughi Heravani, N.; Hadavandkhani, M.; Zarei, Z.; Falahzadeh, K.; Larijani, B.; Rahim, F.; Arjmand, B. Stem Cell and Obesity: Current State and Future Perspective. Adv. Exp. Med. Biol. 2018, 1089, 1–22. [Google Scholar]
- Solanas, G.; Peixoto, F.O.; Perdiguero, E.; Jardí, M.; Ruiz-Bonilla, V.; Datta, D.; Symeonidi, A.; Castellanos, A.; Welz, P.-S.; Caballero, J.M.; et al. Aged Stem Cells Reprogram Their Daily Rhythmic Functions to Adapt to Stress. Cell 2017, 170, 678–692.e20. [Google Scholar] [CrossRef]
- Zhang, X.; Bowles, A.C.; Semon, J.A.; Scruggs, B.A.; Zhang, S.; Strong, A.L.; Gimble, J.M.; Bunnell, B.A. Transplantation of Autologous Adipose Stem Cells Lacks Therapeutic Efficacy in the Experimental Autoimmune Encephalomyelitis Model. PLoS ONE 2014, 9, e85007. [Google Scholar] [CrossRef]
- Penton, C.M.; Thomas-Ahner, J.M.; Johnson, E.K.; McAllister, C.; Montanaro, F. Muscle Side Population Cells from Dystrophic or Injured Muscle Adopt a Fibro-Adipogenic Fate. PLoS ONE 2013, 8, e54553. [Google Scholar] [CrossRef]
- Magee, T.R.; Artaza, J.N.; Ferrini, M.G.; Vernet, D.; Zuniga, F.I.; Cantini, L.; Reisz-Porszasz, S.; Rajfer, J.; Gonzalez-Cadavid, N.F. Myostatin short interfering hairpin RNA gene transfer increases skeletal muscle mass. J. Gene Med. 2006, 8, 1171–1181. [Google Scholar] [CrossRef]
- Wang, X.H.; Mitch, W.E. Mechanisms of muscle wasting in chronic kidney disease. Nat. Rev. Nephrol. 2014, 10, 504–516. [Google Scholar] [CrossRef]
- Artaza, J.N.; Bhasin, S.; Magee, T.R.; Reisz-Porszasz, S.; Shen, R.; Groome, N.P.; Fareez, M.M.; Gonzalez-Cadavid, N.F. Myostatin Inhibits Myogenesis and Promotes Adipogenesis in C3H 10T(1/2) Mesenchymal Multipotent Cells. Endocrinology 2006, 146, 3547–3557, Erratum in: Endocrinology 2006, 147, 4679. [Google Scholar] [CrossRef]
- Deng, B.; Zhang, F.; Wen, J.; Ye, S.; Wang, L.; Yang, Y.; Gong, P.; Jiang, S. The function of myostatin in the regulation of fat mass in mammals. Nutr. Metab. 2017, 14, 29. [Google Scholar] [CrossRef]
- Zhou, X.; Yi, D.; Wu, Y.; Pei, X.; Yu, H.; Chen, Y.; Jiang, Y.; Li, W. Expression of diaphragmatic myostatin and correlation with apoptosis in rats with chronic obstructive pulmonary disease. Exp. Ther. Med. 2018. [Google Scholar] [CrossRef]
- Nozaki, M.; Ota, S.; Terada, S.; Li, Y.; Uehara, K.; Gharaibeh, B.; Fu, F.H.; Huard, J. Timing of the administration of suramin treatment after muscle injury. Muscle Nerve 2012, 46, 70–79. [Google Scholar] [CrossRef]
- Elkasrawy, M.; Immel, D.; Wen, X.; Liu, X.; Liang, L.-F.; Hamrick, M.W. Immunolocalization of Myostatin (GDF-8) Following Musculoskeletal Injury and the Effects of Exogenous Myostatin on Muscle and Bone Healing. J. Histochem. Cytochem. 2012, 60, 22–30. [Google Scholar] [CrossRef]
- Tsao, J.; Kovanecz, I.; Avadalla, N.; Gelfand, R.; Sinha-Hikim, I.; White, R.A.; Gonzalez-Cadavid, N.F. Muscle Derived Stem Cells Stimulate Muscle Myofiber Repair and Counteract Fat Infiltration in a Diabetic Mouse Model of Critical Limb Ischemia. J. Stem Cell Res. Ther. 2016, 6, 370. [Google Scholar]
- Valenti, M.; Dalle Carbonare, L.; Mottes, M. Osteogenic Differentiation in Healthy and Pathological Conditions. Int. J. Mol. Sci. 2016, 18, 41. [Google Scholar] [CrossRef]
- Magne, D.; Bougault, C. What understanding tendon cell differentiation can teach us about pathological tendon ossification. Histol. Histopathol. 2015, 30, 901–910. [Google Scholar]
- Van Linthout, S.; Spillmann, F.; Schultheiss, H.-P.; Tschöpe, C. Effects of mesenchymal stromal cells on diabetic cardiomyopathy. Curr. Pharm. Des. 2011, 17, 3341–3347. [Google Scholar] [CrossRef]
- Escacena, N.; Quesada-Hernández, E.; Capilla-Gonzalez, V.; Soria, B.; Hmadcha, A. Bottlenecks in the Efficient Use of Advanced Therapy Medicinal Products Based on Mesenchymal Stromal Cells. Stem Cells Int. 2015, 2015, 895714. [Google Scholar] [CrossRef]
- Albiero, M.; Ciciliot, S.; Tedesco, S.; Menegazzo, L.; D’Anna, M.; Scattolini, V.; Cappellari, R.; Zuccolotto, G.; Rosato, A.; Cignarella, A.; et al. Diabetes Associated Myelopoiesis Drives Stem Cell Mobilopathy Through an OSM-p66Shc Signaling Pathway. Diabetes 2019, 68, 1303–1314. [Google Scholar] [CrossRef]
- Fadini, G.; Ciciliot, S.; Albiero, M. Concise Review: Perspectives and Clinical Implications of Bone Marrow and Circulating Stem Cell Defects in Diabetes. Stem Cells 2017, 35, 106–116. [Google Scholar] [CrossRef]
- Sacco, A.; Doyonnas, R.; Kraft, P.; Vitorovic, S.; Blau, H.M. Self-renewal and expansion of single transplanted muscle stem cells. Nature 2008, 456, 502–506. [Google Scholar] [CrossRef]
- Kovanecz, I.; Gelfand, R.; Masouminia, M.; Gharib, S.; Segura, D.; Vernet, D.; Rajfer, J.; Li, D.K.; Kannan, K.; Gonzalez-Cadavid, N.F. Oral Bisphenol A (BPA) given to rats at moderate doses is associated with erectile dysfunction, cavernosal lipofibrosis and alterations of global gene transcription. Int. J. Impot. Res. 2014, 26, 67–75. [Google Scholar] [CrossRef]
| miR or Let | ZF4-SC-C | ZF4-SC-C/ ED-MDSC-C | ZL4-SC-C/ ED-MDSC-C | ZF4-SC-C/ ZL4-SC-C |
|---|---|---|---|---|
| ID | per 1000 | Ratios | ||
| miR-143-3p | 273.0 | 2.25 | 2.60 | 0.88 |
| miR-99b-5p | 204.0 | 1.49 | 0.88 | 1.69 |
| miR-10a-5p | 128.0 | 0.80 | 1.64 | 0.49 |
| miR-21-5p | 53.0 | 0.71 | 0.78 | 0.91 |
| miR-100-5p | 53.0 | 0.37 | 0.41 | 0.91 |
| miR-99a-5p | 25.0 | 0.52 | 0.21 | 2.50 |
| miR-10b-5p | 22.0 | 1.83 | 1.89 | 0.97 |
| miR-125a-5p | 18.0 | 2.65 | 0.67 | 3.95 |
| miR-191a-5p | 17.0 | 1.18 | 0.61 | 1.92 |
| miR-26a-5p | 17.0 | 0.67 | 0.32 | 2.12 |
| let-7f-5p | 17.0 | 0.53 | 0.32 | 1.67 |
| miR-199a-5p | 13.0 | 0.65 | 0.48 | 1.35 |
| miR-221-5p | 12.0 | 3.24 | 2.02 | 1.61 |
| miR-222-3p | 11.0 | 10.77 | 2.18 | 4.94 |
| miR-27b-3p | 9.6 | 0.80 | 0.21 | 3.87 |
| miR-24-3p | 9.3 | 1.03 | 0.57 | 1.81 |
| miR-145-3p | 8.2 | 1.91 | 1.50 | 1.27 |
| miR-145-5p | 8.0 | 2.53 | 4.07 | 0.62 |
| miR-30a-5p | 7.3 | 1.41 | 1.51 | 0.93 |
| let-7i-5p | 7.2 | 0.41 | 0.24 | 1.70 |
| miR-148a-3p | 6.2 | 1.17 | 2.06 | 0.57 |
| miR-23a-3p | 5.8 | 1.04 | 0.92 | 1.13 |
| miR-30d-5p | 5.7 | 1.10 | 0.63 | 1.74 |
| miR-146b-5p | 4.4 | 0.08 | 0.04 | 2.10 |
| miR-151-3p | 4.2 | 1.55 | 0.83 | 1.88 |
| miR-199a-3p | 4.0 | 0.36 | 0.31 | 1.17 |
| miR-152-3p | 3.8 | 0.38 | 0.39 | 0.97 |
| miR-351-5p | 3.2 | 2.11 | 0.55 | 3.83 |
| let-7c-5p | 3.0 | 0.57 | 0.24 | 2.41 |
| miR-148b-3p | 2.9 | 0.94 | 0.52 | 1.82 |
| miR-22-3p | 2.8 | 1.01 | 0.44 | 2.28 |
| miR-221-3p | 2.2 | 2.28 | 0.91 | 2.52 |
| miR-27a-3p | 2.1 | 1.08 | 0.76 | 1.42 |
| rno-let-7g-5p | 2.0 | 0.48 | 0.39 | 1.22 |
| miR-181a-5p | 1.8 | 1.64 | 1.56 | 1.04 |
| miR-92a-3p | 1.8 | 2.42 | 0.91 | 2.67 |
| miR-342-3p | 1.7 | 2.48 | 2.00 | 1.24 |
| let-7b-5p | 1.5 | 0.68 | 0.27 | 2.47 |
| miR-23b-3p | 1.5 | 0.61 | 0.24 | 2.51 |
| miR-30e-5p | 1.4 | 0.88 | 0.70 | 1.26 |
| miR-7a-5p | 1.4 | 2.21 | 4.94 | 0.45 |
| miR-143-5p | 1.2 | 2.71 | 2.18 | 1.24 |
| miR-186-5p | 1.1 | 1.07 | 0.70 | 1.54 |
| miR-192-5p | 1.1 | 1.45 | 0.62 | 2.35 |
| miR-28-3p | 1.1 | 1.27 | 1.01 | 1.26 |
| miR-365-3p | 1.0 | 1.44 | 1.17 | 1.23 |
| ID for miR or Let | In Vivo | C Value per 106 Reads | In Vitro Added 5% Serum (S) | |||
|---|---|---|---|---|---|---|
| MDSC | ZF4-SC | ZL4-SC | ||||
| LD/ED Ratio | Ratios to C | |||||
| ZFS/C | ZLS/C | ZFS/C | ZLS/C | |||
| miR-21-5p | 0.16 | 364070 | 0.52 | 1.17 | 0.69 | 1.13 |
| miR-199a-5p | 0.29 | 88010 | 0.41 | 0.86 | 0.86 | 0.97 |
| rmiR-23a-3p | 0.50 | 39134 | 1.11 | 1.09 | 1.14 | 0.86 |
| miR-199a-3p | 0.25 | 27501 | 0.41 | 0.86 | 0.86 | 0.97 |
| miR-27a-3p | 0.41 | 14228 | 0.99 | 1.08 | 1.02 | 1.16 |
| miR-181a-5p | 0.39 | 12538 | 0.54 | 0.94 | 1.66 | 0.89 |
| miR-30e-5p | 0.42 | 9387 | 0.44 | 1.14 | 0.63 | 0.94 |
| miR-101a-3p | 0.38 | 4728 | 1.06 | 0.97 | 1.05 | 0.98 |
| miR-214-3p | 0.22 | 4525 | 0.46 | 0.98 | 0.79 | 0.86 |
| miR-29a-3p | 0.25 | 2401 | 0.59 | 1.26 | 1.81 | 1.21 |
| miR-101b-3p | 0.09 | 804 | 0.49 | 1.13 | 0.86 | 1.17 |
| miR-132-3p | 0.02 | 340 | 0.68 | 0.99 | 1.46 | 0.90 |
| ID for miR or Let | In Vivo | C Value per 107Reads | In Vitro Added 5% Serum (S) | |||
|---|---|---|---|---|---|---|
| MDSC | ZF4-SC | ZL4-SC | ||||
| LD/ED Ratio | Ratios to C | |||||
| ZFS/C | ZLS/C | ZFS/C | ZLS/C | |||
| miR-99b-5p | 0.25 | 1393619 | 0.53 | 0.89 | 0.83 | 0.94 |
| miR-10a-5p | 0.28 | 871279 | 0.66 | 0.95 | 0.49 | 0.90 |
| miR-100-5p | 0.22 | 361258 | 0.63 | 0.78 | 0.64 | 0.84 |
| miR-99a-5p | 2.65 | 167053 | 0.37 | 0.95 | 0.83 | 0.85 |
| miR-10b-5p | 0.45 | 149991 | 0.33 | 0.92 | 0.46 | 0.84 |
| miR-26a-5p | 0.32 | 115483 | 0.99 | 1.12 | 1.89 | 1.04 |
| let-7f-5p | 0.32 | 115006 | 0.89 | 1.14 | 1.45 | 1.23 |
| miR-221-5p | 0.34 | 82561 | 0.59 | 1.26 | 0.96 | 1.19 |
| let-7i-5p | 0.12 | 49022 | 0.86 | 1.11 | 1.32 | 1.28 |
| miR-148a-3p | 0.25 | 42455 | 0.27 | 0.86 | 0.65 | 0.80 |
| miR-152-3p | 0.33 | 25783 | 0.42 | 0.97 | 0.88 | 1.01 |
| miR-148b-3p | 0.16 | 19779 | 0.51 | 1.06 | 0.98 | 1.26 |
| let-7g-5p | 0.22 | 13892 | 1.10 | 1.08 | 1.86 | 1.68 |
| miR-92a-3p | 0.17 | 12460 | 0.59 | 1.19 | 1.35 | 1.30 |
| miR-342-3p | 0.26 | 11421 | 0.22 | 0.99 | 0.48 | 0.84 |
| miR-212-5p | 0.08 | 4903 | 0.87 | 1.12 | 1.98 | 1.11 |
| miR-10b-3p | 0.10 | 4130 | 0.69 | 1.08 | 1.03 | 1.70 |
| miR-25-3p | 2.20 | 3984 | 0.67 | 0.94 | 1.10 | 1.34 |
| miR-362-5p | 0.20 | 1234 | 0.36 | 1.56 | 0.60 | 1.18 |
| miR-31a-5p | 0.29 | 1020 | 1.40 | 1.24 | 2.10 | 1.31 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovanecz, I.; Gelfand, R.; Lin, G.; Sharifzad, S.; Ohanian, A.; Ricks, R.; Lue, T.; Gonzalez-Cadavid, N.F. Stem Cells from a Female Rat Model of Type 2 Diabetes/Obesity and Stress Urinary Incontinence Are Damaged by In Vitro Exposure to its Dyslipidemic Serum, Predicting Inadequate Repair Capacity In Vivo. Int. J. Mol. Sci. 2019, 20, 4044. https://doi.org/10.3390/ijms20164044
Kovanecz I, Gelfand R, Lin G, Sharifzad S, Ohanian A, Ricks R, Lue T, Gonzalez-Cadavid NF. Stem Cells from a Female Rat Model of Type 2 Diabetes/Obesity and Stress Urinary Incontinence Are Damaged by In Vitro Exposure to its Dyslipidemic Serum, Predicting Inadequate Repair Capacity In Vivo. International Journal of Molecular Sciences. 2019; 20(16):4044. https://doi.org/10.3390/ijms20164044
Chicago/Turabian StyleKovanecz, Istvan, Robert Gelfand, Guiting Lin, Sheila Sharifzad, Alec Ohanian, Randy Ricks, Tom Lue, and Nestor F. Gonzalez-Cadavid. 2019. "Stem Cells from a Female Rat Model of Type 2 Diabetes/Obesity and Stress Urinary Incontinence Are Damaged by In Vitro Exposure to its Dyslipidemic Serum, Predicting Inadequate Repair Capacity In Vivo" International Journal of Molecular Sciences 20, no. 16: 4044. https://doi.org/10.3390/ijms20164044
APA StyleKovanecz, I., Gelfand, R., Lin, G., Sharifzad, S., Ohanian, A., Ricks, R., Lue, T., & Gonzalez-Cadavid, N. F. (2019). Stem Cells from a Female Rat Model of Type 2 Diabetes/Obesity and Stress Urinary Incontinence Are Damaged by In Vitro Exposure to its Dyslipidemic Serum, Predicting Inadequate Repair Capacity In Vivo. International Journal of Molecular Sciences, 20(16), 4044. https://doi.org/10.3390/ijms20164044






