A Narrative Role of Vitamin D and Its Receptor: With Current Evidence on the Gastric Tissues
Abstract
1. Introduction
2. Vitamin D Ligands
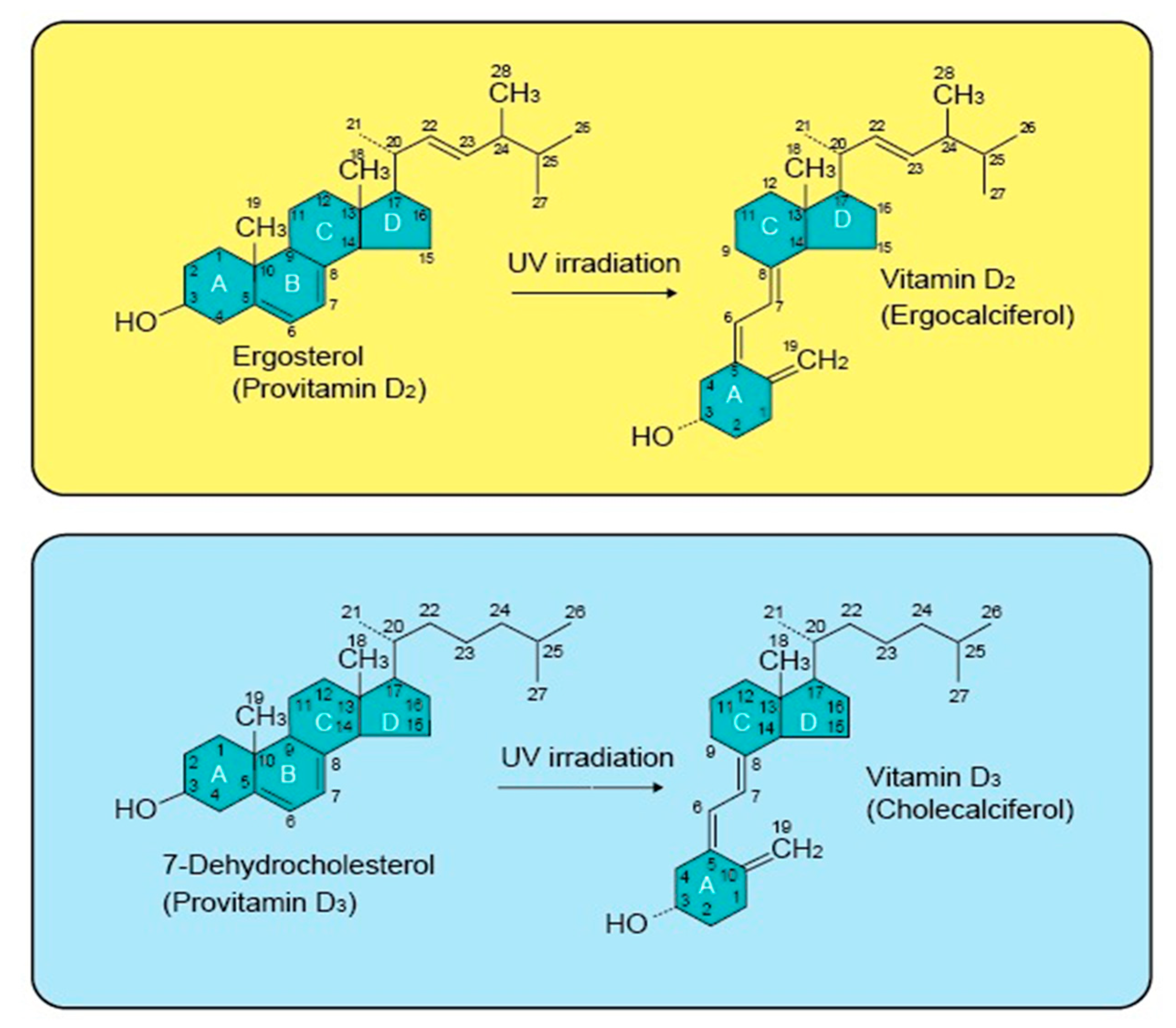
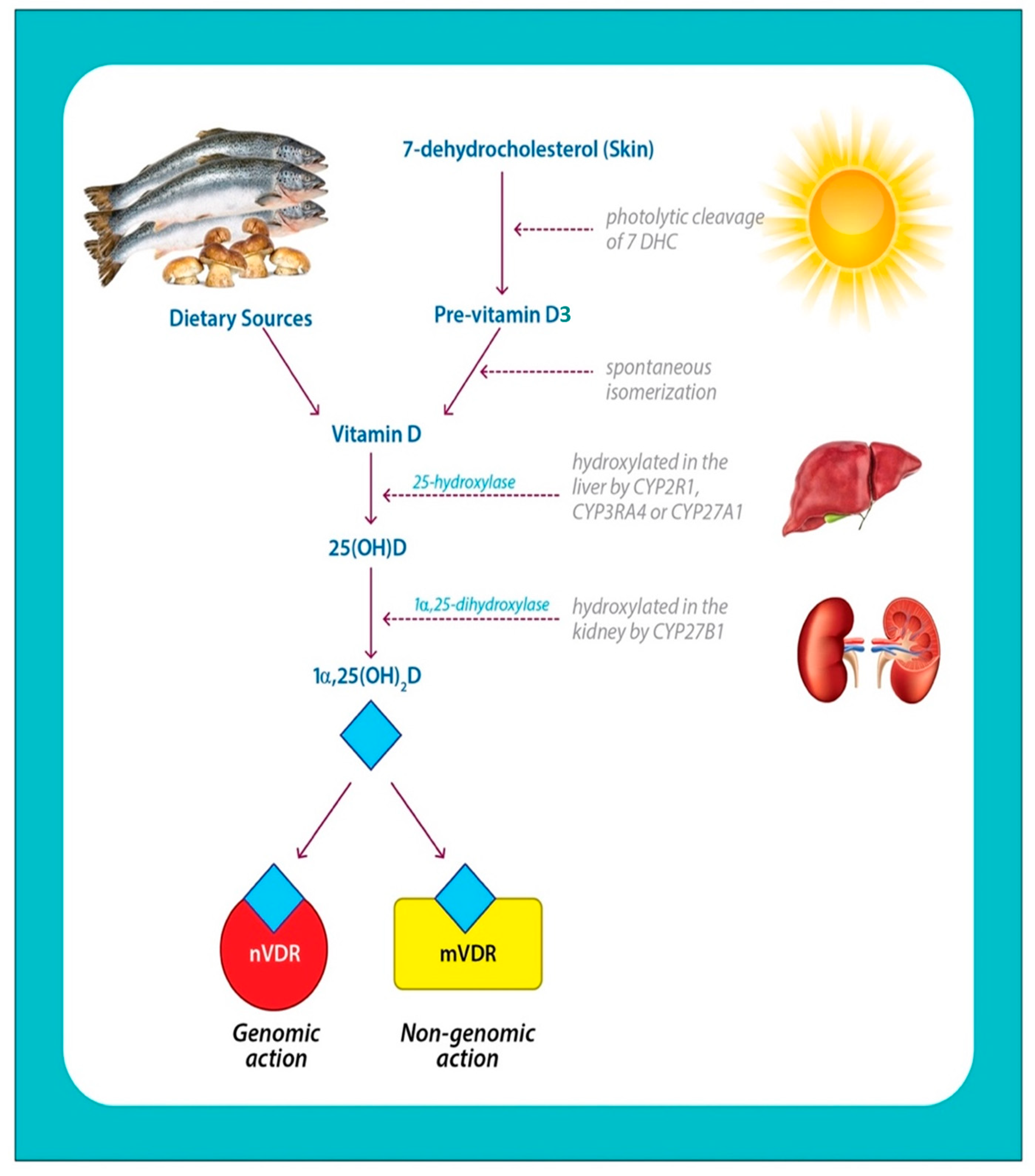
2.1. Vitamin D Structure and Biosynthesis
2.2. Vitamin D Metabolites
2.2.1. 25-hydroxyvitamin-D2 (25(OH)D2)
2.2.2. 25-hydroxyvitamin-D3 (25(OH)D3)
2.2.3. 24, 25-dihydroxyvitamin-D (24,25(OH)2D)
2.2.4. 1-α, 25-dihydroxyvitamin-D3 (1α,25(OH)2D3 or 1,25D3)
2.2.5. 1-α, 25-dihydroxy vitamin-D2 (1α,25(OH)2D2 or 1,25D2)
2.2.6. 3-epi-vitamin D
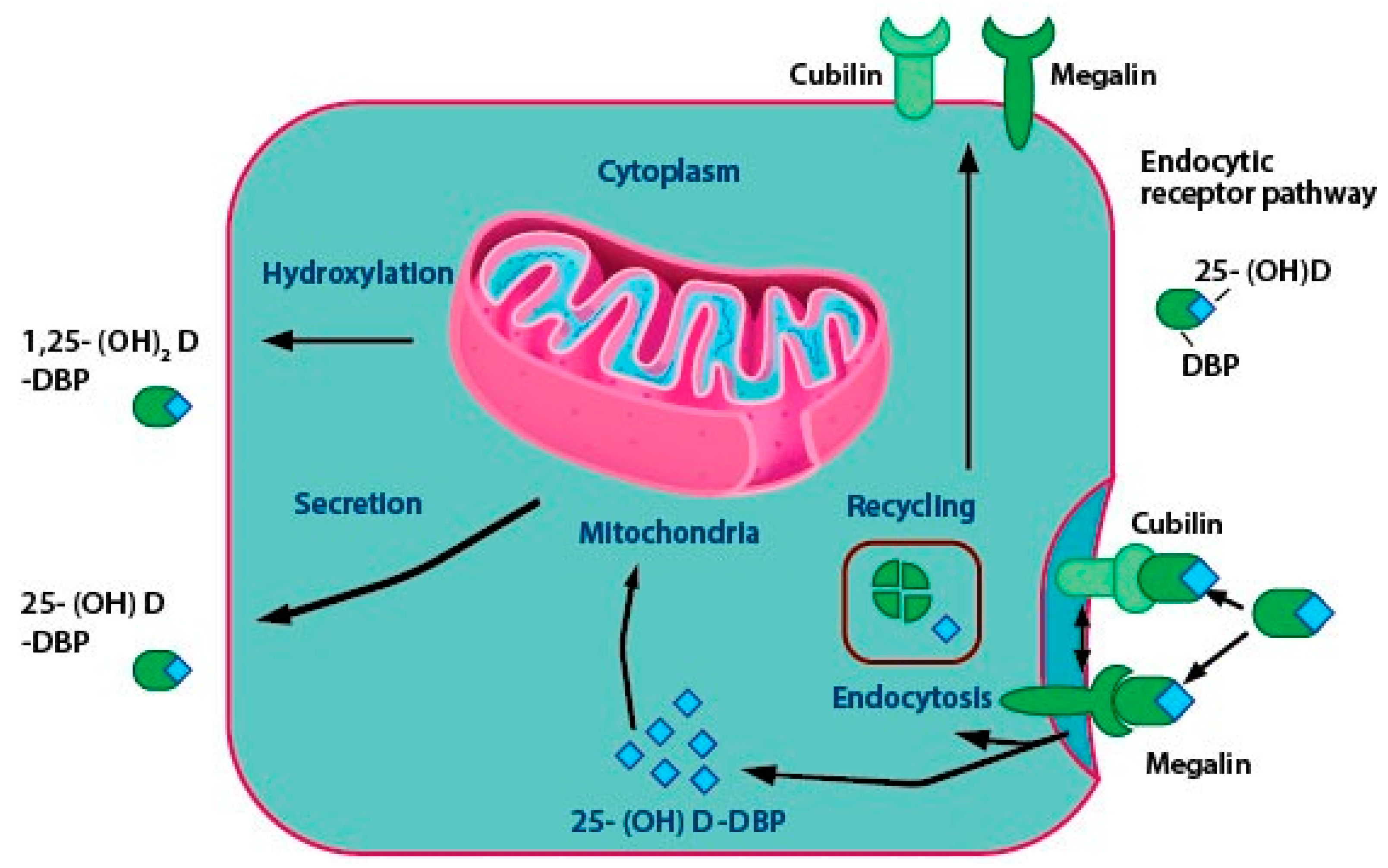
2.3. Vitamin D Transport
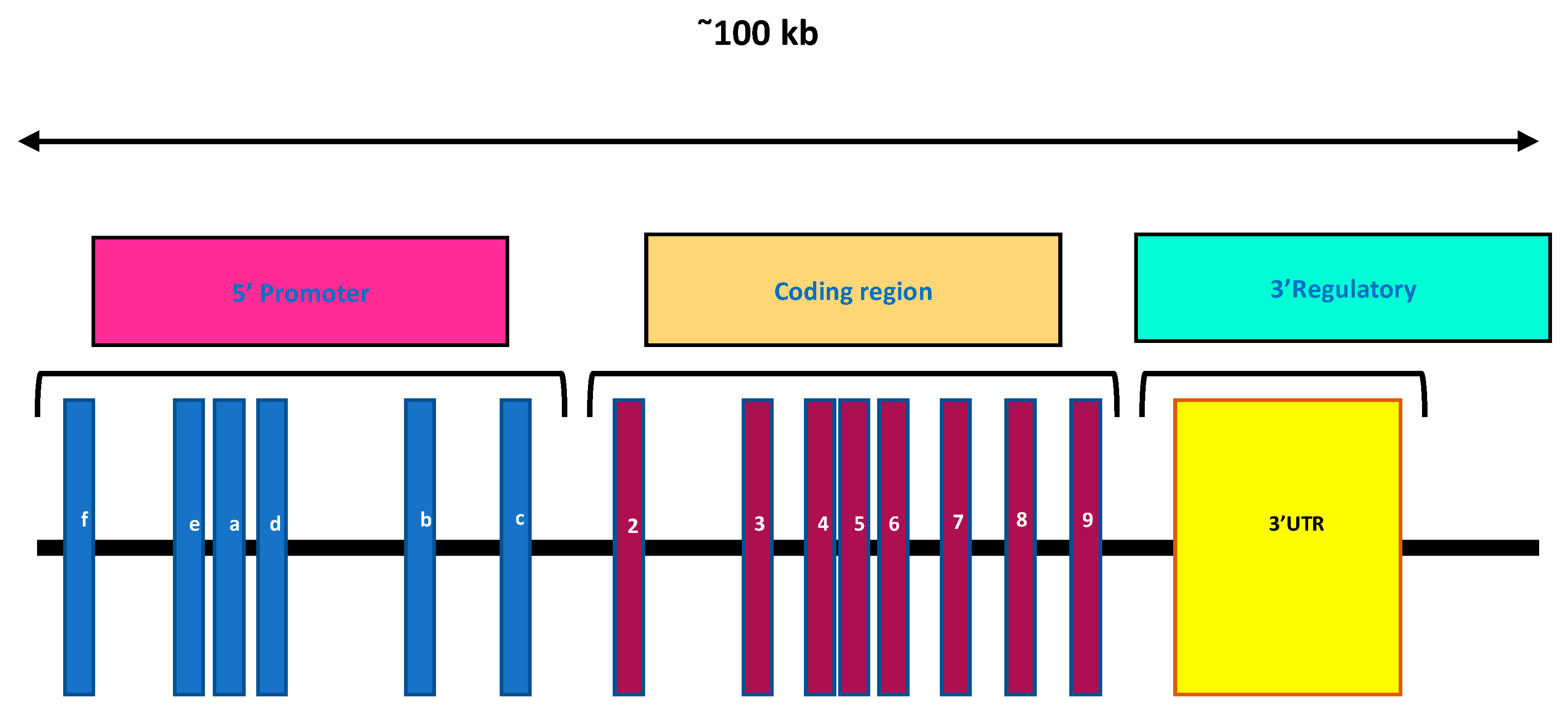
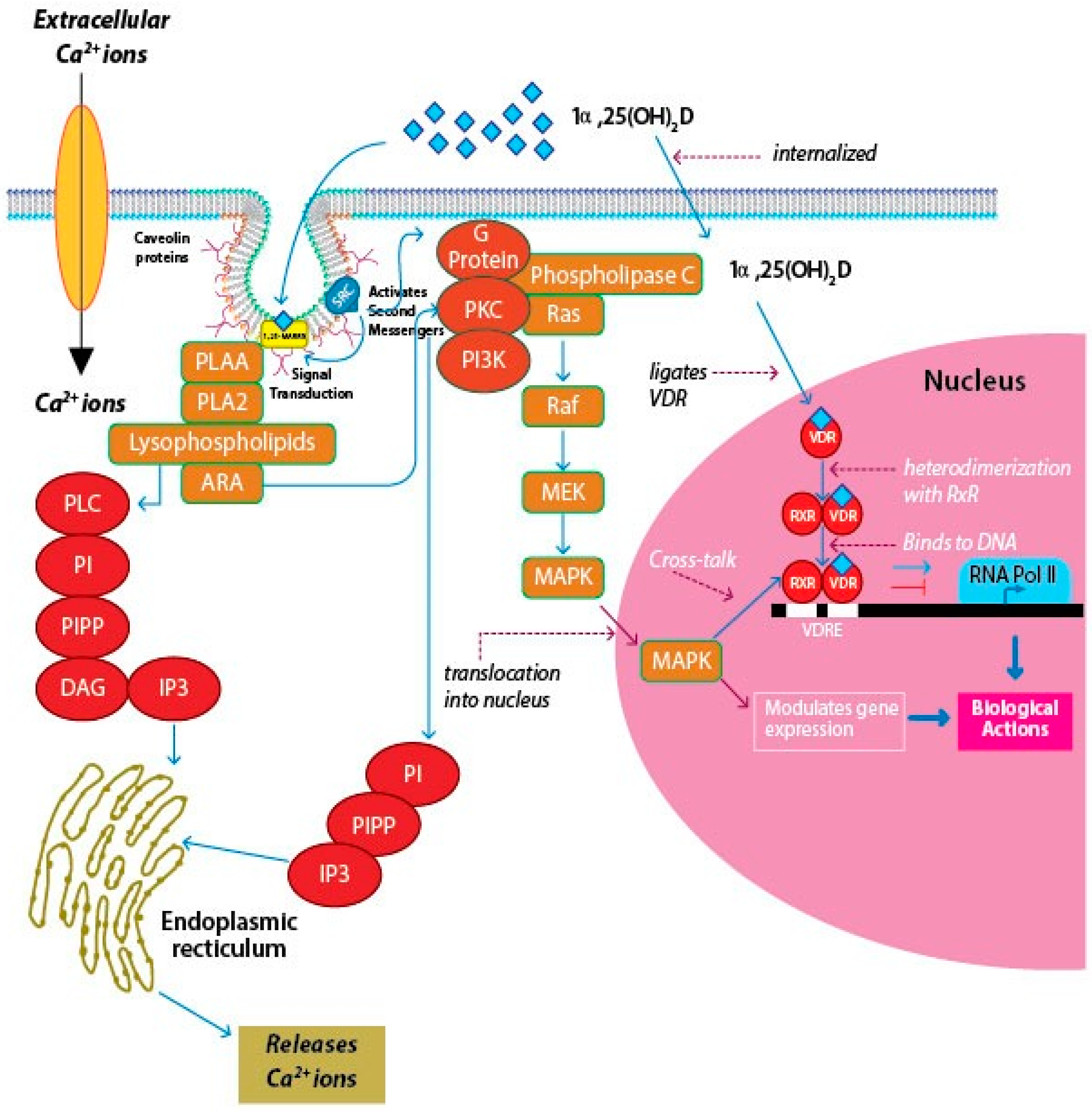
3. Vitamin D Receptors (VDRs)
3.1. Nuclear VDR (nVDR)
3.2. Membrane VDR (mVDR)
4. Signaling Pathways of Vitamin D
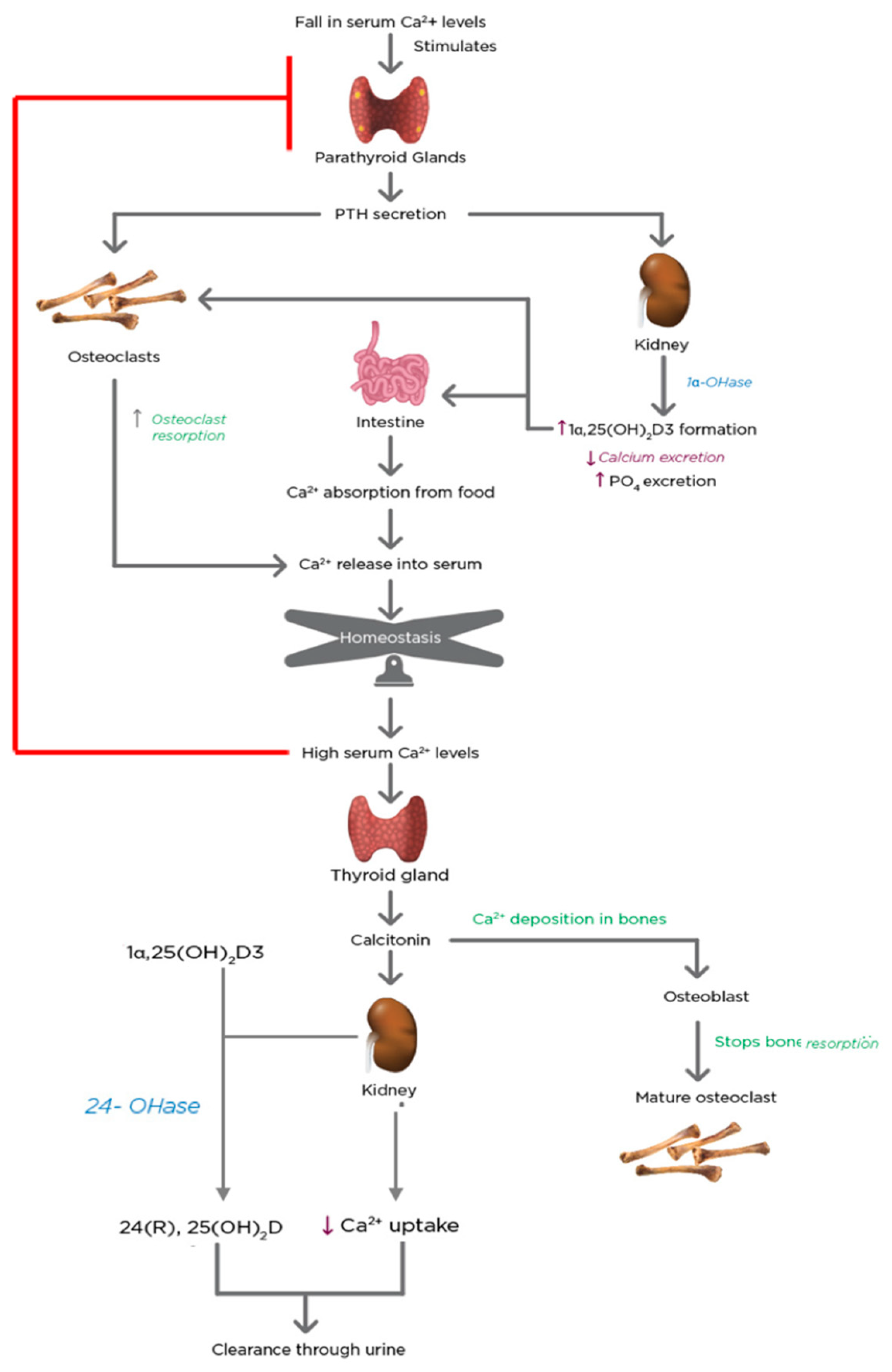
5. Regulation of Calcium and Phosphate Homeostasis by 1α,25(OH)2D3 and PTH
6. Vitamin D and the Gastric Tissue
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 1α,25(OH)2D2 | 1α,25-dihydroxyvitamin D2 |
| 1α,25(OH)2D3 | 1α,25-dihydroxyvitamin D3 |
| 1,25-MARRS | 1,25 membrane-associated rapid response steroid-binding proteins |
| 24(R),25-(OH)2D | 24R, 25-dihydroxyvitamin D |
| 25(OH)D | 25-hydroxyvitamin D |
| 25(OH)D3 | 25-hydroxyvitamin-D3 |
| 5azadC | 5-Aza-2′-deoxycytidine |
| ASAP2 | ArfGAP with SH3 domain, ankyrin repeat and PH domain 2 |
| cAMP | Cyclic AMP |
| CaSR | Calcium-sensing receptor |
| CAMP | Cathelicidin antimicrobial protein |
| CLIA | Chemiluminescent immunoassay |
| CYP | Cytochrome P450 |
| DAG | Diacylglycerol |
| DBP | Vitamin D binding protein |
| DNMT | DNA methyltransferase |
| ECL | Enterochromaffin-like cells |
| ELISA | Enzyme-linked immunoassay |
| ER | Endoplasmic recticulum |
| ERp57 | Endoplasmic reticulum proteins 57 |
| ERp60 | Endoplasmic reticulum proteins 60 |
| FGF23 | Fibroblast growth factor-23 |
| FOXP3 | Fork head box P3 |
| GBP | Gastric bypass surgery |
| GC | Gastric cancer |
| GIT | Gastrointestinal tract |
| GRP58 | Glucose-regulated protein 58 |
| HDAC | Histone deacetylases |
| HPLC | High-performance liquid chromatography |
| IL-8 | Interleukin-8 |
| IP3 | Inositol triphosphate |
| LC-MS/MS | Liquid Chromatography-Tandem Mass Spectrometry |
| LBD | Ligand binding domains |
| mVDR | Membrane VDR |
| MAH | Malignancy-associated hypercalcemia |
| MAPK | Mitogen-Activated Protein Kinase |
| MBD | Metabolic bone disease |
| NINJ1 | Ninjurin 1 |
| nVDR | Nuclear VDR |
| Pdia3 | Protein disulfide isomerase family A member 3 |
| PI | Phosphatidyl inositol |
| PIP3 | Phosphatidylinositol triphosphate |
| PIPP | Polyisoprenyl phosphate |
| PKA | Protein kinase A |
| PKC | Protein kinase C |
| PLA2 | Phospholipase A2 |
| PLAA | Phospholipase A2-activating protein |
| PTEN | Phosphatase and tensin homolog deleted on chromosome 10 |
| PTH | Parathyroid hormone |
| PTHLH | Parathyroid hormone like hormone |
| RAF/MAPK | Rapidly Accelerated Fibrosarcoma/Mitogen-Activated Protein Kinase |
| RIA | Radioimmunoassay |
| SRC 1-3 | Steroid receptor coactivator family 1-3 |
| STM | Soft-tissue mineralization |
| TME | Tumor microenvironment |
| Trpv6 | Transient Receptor Potential Vanilloid 6 |
| TSA | Trichostatin A |
| UVB | Ultraviolet B |
| VDD | Vitamin D deficiency |
| VDR | Vitamin D receptor |
| VDRE | Vitamin D response element |
| VDT | Vitamin D Toxicity |
References
- Sahota, O. Understanding vitamin D deficiency. Age Ageing 2014, 43, 589–591. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.J.; Murray, B.F.; O’Keane, M.; Kilbane, M.T. Rising trend in vitamin D status from 1993 to 2013: Dual concerns for the future. Endocr. Connect. 2015, 4, 163–171. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kuchuk, N.O.; Pluijm, S.M.F.; Van Schoor, N.M.; Looman, C.W.N.; Smit, J.H.; Lips, P. Relationships of Serum 25-Hydroxyvitamin D to Bone Mineral Density and Serum Parathyroid Hormone and Markers of Bone Turnover in Older Persons. J. Clin. Endocrinol. Metab. 2009, 94, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.; Jaceldo-Siegl, K.; Fraser, G.E. Serum 25-hydroxyvitamin D status of vegetarians, partial vegetarians, and nonvegetarians: The Adventist Health Study-21234. Am. J. Clin. Nutr. 2009, 89, 1686S–1692S. [Google Scholar] [CrossRef] [PubMed]
- Looker, A.C.; Bowman, B.A.; Hollis, B.W.; Gillespie, C.; Allen, C.; Doughertly, C.; Gunter, E.W.; Cogswell, M.E.; Nesby-O’Dell, S.; Scanlon, K.S. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: Third National Health and Nutrition Examination Survey, 1988–1994. Am. J. Clin. Nutr. 2002, 76, 187–192. [Google Scholar]
- Vitamin D Deficiency: The Ultimate Guide. Available online: https://www.your.md/blog/vitamin-d-deficiency/ (accessed on 21 March 2019).
- Van Schoor, N.; Lips, P. Global Overview of Vitamin D Status. Endocrinol. Metab. Clin. N. Am. 2017, 46, 845–870. [Google Scholar] [CrossRef] [PubMed]
- 90% of UAE Population Vitamin D Deficient, Says DHA Official. Available online: https://gulfnews.com/going-out/society/90-of-uae-population-vitamin-d-deficient-says-dha-official-1.2113556 (accessed on 22 March 2019).
- Buyukuslu, N.; Esin, K.; Hizli, H.; Sunal, N.; Yiğit, P.; Garipağaoğlu, M. Clothing preference affects vitamin D status of young women. Nutr. Res. 2014, 34, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Vitamin D Deficiency Could Cause Deadly Diseases, Warn UAE Doctors—Khaleej Times. Available online: https://www.khaleejtimes.com/nation/abu-dhabi/vitamin-d-deficiency-could-cause-deadly-diseases-warn-uae-doctors (accessed on 22 March 2019).
- Dunn, P.M. Francis Glisson (1597–1677) and the “Discovery” of Rickets. Arch. Dis. Child. Fetal Neonatal Ed. 1998, 78, F154–F155. [Google Scholar] [CrossRef]
- O’Riordan, J.L.H.; Bijvoet, O.L.M. Rickets before the discovery of vitamin D. BoneKEy Rep. 2014, 3, 3. [Google Scholar] [CrossRef][Green Version]
- The History of Vitamin D|Ddrops® Blog. Available online: http://blog.vitaminddrops.com/the-history-of-vitamin-d/ (accessed on 22 March 2019).
- McCollum, E.V.; Simmonds, N.; Becker, J.E.; Shipley, P.G. Studies on experimental rickets. Nutr. Rev. 1975, 33, 48–50. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Hayes, C.E.; DeLuca, H.F. 1,25-Dihydroxycholecalciferol Inhibits the Progression of Arthritis in Murine Models of Human Arthritis. J. Nutr. 1998, 128, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Chun, R.F.; Peercy, B.E.; Orwoll, E.S.; Nielson, C.M.; Adams, J.S.; Hewison, M. Vitamin D and DBP: The free hormone hypothesis revisited. J. Steroid Biochem. Mol. Biol. 2014, 144, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Christakos, S.; Hewison, M.; Gardner, D.G.; Wagner, C.L.; Sergeev, I.N.; Rutten, E.; Pittas, A.G.; Boland, R.; Ferrucci, L.; Bikle, D.D. Vitamin D: Beyond bone: Vitamin D: Beyond bone. Ann. N. Y. Acad. Sci. 2013, 1287, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the immune system: New perspectives on an old theme. Endocrinol. Metab. Clin. N. Am. 2010, 39, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Welsh, J. Cellular and molecular effects of vitamin D on carcinogenesis. Arch. Biochem. Biophys. 2012, 523, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Mondul, A.M.; Weinstein, S.J.; Layne, T.M.; Albanes, D. Vitamin D and Cancer Risk and Mortality: State of the Science, Gaps, and Challenges. Epidemiol. Rev. 2017, 39, 28–48. [Google Scholar] [CrossRef]
- Guillot, X.; Semerano, L.; Saidenberg-Kermanac’h, N.; Falgarone, G.; Boissier, M.-C. Vitamin D and inflammation. Joint Bone Spine 2010, 77, 552–557. [Google Scholar] [CrossRef]
- Wolf, M.; Shah, A.; Gutierrez, O.; Ankers, E.; Monroy, M.; Tamez, H.; Steele, D.; Chang, Y.; Camargo, C.; Tonelli, M.; et al. Vitamin D levels and early mortality among incident hemodialysis patients. Kidney Int. 2007, 72, 1004–1013. [Google Scholar] [CrossRef]
- Vimaleswaran, K.S.; Cavadino, A.; Berry, D.J.; Jorde, R.; Dieffenbach, A.K.; Lu, C.; Alves, A.C.; Heerspink, H.J.L.; Tikkanen, E.; Eriksson, J.; et al. Association of vitamin D status with arterial blood pressure and hypertension risk: A mendelian randomisation study. Lancet Diabetes Endocrinol. 2014, 2, 719–729. [Google Scholar] [CrossRef]
- Vanlint, S. Vitamin D and Obesity. Nutrients 2013, 5, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Grübler, M.; Gaksch, M.; Schwetz, V.; Trummer, C.; Hartaigh, B.Ó.; Verheyen, N.; Tomaschitz, A.; März, W. Vitamin D and Mortality. Anticancer Res. 2016, 36, 1379–1387. [Google Scholar] [PubMed]
- Penckofer, S.; Kouba, J.; Wallis, D.E.; Emanuele, M.A. Vitamin D and Diabetes Let the Sunshine in. Diabetes Educ. 2008, 34, 939–954. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.Y.; Park, K.M.; Lee, M.J.; Yang, D.H.; Kim, S.H.; Lee, S.-Y. Vitamin D and Hypertension. Electrolyte Blood Press 2017, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Low Vitamin D Levels in Adults|Empoweryourhealth.org. Available online: https://www.empoweryourhealth.org/issue-1/Low-Vitamin-D-LEVELS-IN-ADULTS (accessed on 31 March 2019).
- Schmidt, J.A. Measurement of 25-Hydroxyvitamin D Revisited. Clin. Chem. 2006, 52, 2304–2305. [Google Scholar] [CrossRef][Green Version]
- Terry, A.H.; Sandrock, T.; Meikle, A.W. Measurement of 25-Hydroxyvitamin D by the Nichols ADVANTAGE, DiaSorin LIAISON, DiaSorin RIA, and Liquid Chromatography-Tandem Mass Spectrometry. Clin. Chem. 2005, 51, 1565–1566. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shah, I.; James, R.; Barker, J.; Petroczi, A.; Naughton, D.P. Misleading measures in Vitamin D analysis: A novel LC-MS/MS assay to account for epimers and isobars. Nutr. J. 2011, 10, 46. [Google Scholar] [CrossRef]
- De Koning, L.; Al-Turkmani, M.R.; Berg, A.H.; Shkreta, A.; Law, T.; Kellogg, M.D. Variation in clinical vitamin D status by DiaSorin Liaison and LC-MS/MS in the presence of elevated 25-OH vitamin D2. Clin. Chim. Acta 2013, 415, 54–58. [Google Scholar] [CrossRef]
- Vitamin D Deficiency: How Common Is It and What Can I Do about It?|Ddrops® Blog. Available online: http://blog.vitaminddrops.com/vitamin-d-deficiency-common-can/ (accessed on 22 March 2019).
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118. [Google Scholar]
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium Dietary Reference Intakes for Calcium and Vitamin D; Ross, A.C., Taylor, C.L., Yaktine, A.L., Del Valle, H.B., Eds.; The National Academies Collection: Reports Funded by National Institutes of Health; National Academies Press (US): Washington, DC, USA, 2011. [Google Scholar]
- Jäpelt, R.B.; Jakobsen, J. Vitamin D in plants: A review of occurrence, analysis, and biosynthesis. Front. Plant Sci. 2013, 4, 136. [Google Scholar] [CrossRef]
- Wierzbicka, J.; Piotrowska, A.; Żmijewski, M.A. The renaissance of vitamin D. Acta Biochim. Pol. 2014, 61, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Prohormones—A Comprehensive Guide to Prohormones—Updated|Predator Nutrition. Available online: https://www.predatornutrition.com/articlesdetail?cid=prohormones-a-comprehensive-guide-to-prohormones-updated (accessed on 26 March 2019).
- Vitamin D|Hormone Health Network. Available online: https://www.hormone.org/hormones-and-health/hormones/vitamin-d (accessed on 26 March 2019).
- Maestro, M.A.; Molnár, F.; Mouriño, A.; Carlberg, C. Vitamin D receptor 2016: Novel ligands and structural insights. Expert Opin. Ther. Patents 2016, 26, 1291–1306. [Google Scholar] [CrossRef] [PubMed]
- Seuter, S.; Ryynänen, J.; Carlberg, C. The ASAP2 gene is a primary target of 1,25-dihydroxyvitamin D3 in human monocytes and macrophages. J. Steroid Biochem. Mol. Boil. 2014, 144, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.W.; Roth, J.; Orci, L. The Vitamin D Endocrine System: Steroid Metabolism, Hormone Receptors, and Biological Response (Calcium Binding Proteins). Endocr. Rev. 1982, 3, 331–366. [Google Scholar] [CrossRef] [PubMed]
- Menegaz, D.; Mizwicki, M.T.; Barrientos-Duran, A.; Chen, N.; Henry, H.L.; Norman, A.W. Vitamin D Receptor (VDR) Regulation of Voltage-Gated Chloride Channels by Ligands Preferring a VDR-Alternative Pocket (VDR-AP). Mol. Endocrinol. 2011, 25, 1289–1300. [Google Scholar] [CrossRef]
- Jovičić, S.; Ignjatovic, S.; Majkić-Singh, N. Biochemistry and metabolism of vitamin D/Biohemija i metabolizam vitamina D. J. Med. Biochem. 2012, 31, 309–315. [Google Scholar] [CrossRef]
- Wikvall, K. Cytochrome P450 enzymes in the bioactivation of vitamin D to its hormonal form (Review). Int. J. Mol. Med. 2001, 7, 201–209. [Google Scholar] [CrossRef]
- Jones, G.; Prosser, D.E.; Kaufmann, M. Cytochrome P450-mediated metabolism of vitamin D. J. Lipid Res. 2014, 55, 13–31. [Google Scholar] [CrossRef]
- Hollis, B.W. Comparison of equilibrium and disequilibrium assay conditions for ergocalciferol, cholecalciferol and their major metabolites. J. Steroid Biochem. 1984, 21, 81–86. [Google Scholar] [CrossRef]
- Hewison, M. Antibacterial effects of vitamin D. Nat. Rev. Endocrinol. 2011, 7, 337–345. [Google Scholar] [CrossRef]
- Bikle, D. Vitamin D: Production, Metabolism, and Mechanisms of Action. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., Dungan, K., Grossman, A., Hershman, J.M., Kaltsas, G., Koch, C., Kopp, P., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Kamao, M.; Tatematsu, S.; Hatakeyama, S.; Sakaki, T.; Sawada, N.; Inouye, K.; Ozono, K.; Kubodera, N.; Reddy, G.S.; Okano, T. C-3 Epimerization of Vitamin D3 Metabolites and Further Metabolism of C-3 Epimers 25-hydroxyvitamin D3 is metabolized to 3-epi-25-hydroxyvitamin D3 and subsequently metabolized through C-1α or C-24 hydroxylation. J. Biol. Chem. 2004, 279, 15897–15907. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.; Veljkovic, K.; Yazdanpanah, M.; Adeli, K. Analytical measurement and clinical relevance of vitamin D3 C3-epimer. Clin. Biochem. 2013, 46, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Hayes, A.; Galvin, K.; Merkel, J.; Jones, G.; Kaufmann, M.; Hoofnagle, A.N.; Carter, G.D.; Durazo-Arvizu, R.A.; Sempos, C.T. Significance of Serum 24,25-Dihydroxyvitamin D in the Assessment of Vitamin D Status: A Double-edged Sword? Clin. Chem. 2015, 61, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Michio, I.; Kunihisa, K.; Toru, Y.; Toshiaki, O. A simple and sensitive assay for 25-hydroxyvitamin D, 24,25-dihydroxyvitamin D and 1,25-dihydroxyvitamin D in human serum. Clin. Chim. Acta 1982, 124, 63–73. [Google Scholar] [CrossRef]
- Somjen, D.; Somjen, G.J.; Weisman, Y.; Binderman, I.; Dokoh, S.; Haussler, M.R.; Pike, J.W.; Danan, J.L.; Mathieu, H.; Lawson, D.E.M.; et al. Evidence for 24,25-dihydroxycholecalciferol receptors in long bones of newborn rats. Biochem. J. 1982, 204, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Knutson, J.C.; Hollis, B.W.; LeVan, L.W.; Valliere, C.; Gould, K.G.; Bishop, C.W. Metabolism of 1 alpha-hydroxyvitamin D2 to activated dihydroxyvitamin D 2 metabolites decreases endogenous 1 alpha, 25-dihydroxyvitamin D 3 in rats and monkeys. Endocrinology 1995, 136, 4749–4753. [Google Scholar] [CrossRef]
- Reddy, G.S.; Muralidharan, K.R.; Okamura, W.H.; Tserng, K.Y.; McLane, J.A. Metabolism of 1alpha,25-dihydroxyvitamin D(3) and its C-3 epimer 1alpha,25-dihydroxy-3-epi-vitamin D(3) in neonatal human keratinocytes. Steroids 2001, 66, 441–450. [Google Scholar] [CrossRef]
- Brown, A.J.; Ritter, C.; Slatopolsky, E.; Muralidharan, K.R.; Okamura, W.H.; Reddy, G.S. 1Alpha,25-dihydroxy-3-epi-vitamin D3, a natural metabolite of 1alpha,25-dihydroxyvitamin D3, is a potent suppressor of parathyroid hormone secretion. J. Cell. Biochem. 1999, 73, 106–113. [Google Scholar] [CrossRef]
- Kaseda, R.; Hosojima, M.; Sato, H.; Saito, A. Role of Megalin and Cubilin in the Metabolism of Vitamin D3. Ther. Apher. Dial. 2011, 15, 14–17. [Google Scholar] [CrossRef]
- Romagnoli, E.; Pepe, J.; Piemonte, S.; Cipriani, C.; Minisola, S. Value and limitations of assessing vitamin D nutritional status and advised levels of vitamin D supplementation. Eur. J. Endocrinol. 2013, 169, R59–R69. [Google Scholar] [CrossRef]
- Chun, R.F.; Peercy, B.E.; Adams, J.S.; Hewison, M. Vitamin D Binding Protein and Monocyte Response to 25-Hydroxy Vitamin D and 1,25-Dihydroxy Vitamin D: Analysis by Mathematical Modeling. PLoS ONE 2012, 7, E30773. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Armas, L.A.G.; Shary, J.R.; Bell, N.H.; Binkley, N.; Hollis, B.W. 25-Hydroxylation of vitamin D3: Relation to circulating vitamin D3 under various input conditions. Am. J. Clin. Nutr. 2008, 87, 1738–1742. [Google Scholar] [CrossRef] [PubMed]
- Nussey, S.; Whitehead, S. Endocrinology: An Integrated Approach; BIOS Scientific Publishers: Oxford, UK, 2001; ISBN 978-1-85996-252-7. [Google Scholar]
- Rowling, M.J.; Taffany, D.A.; Welsh, J.; Kemmis, C.M. Megalin-Mediated Endocytosis of Vitamin D Binding Protein Correlates with 25-Hydroxycholecalciferol Actions in Human Mammary Cells. J. Nutr. 2006, 136, 2754–2759. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Karpf, A.R.; Deeb, K.K.; Muindi, J.R.; Morrison, C.D.; Johnson, C.S.; Trump, D.L. Epigenetic Regulation of Vitamin D 24-Hydroxylase/CYP24A1 in Human Prostate Cancer. Cancer Res. 2010, 70, 5953–5962. [Google Scholar] [CrossRef] [PubMed]
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schütz, G.; Umesono, K.; Blumberg, B.; Kastner, P.; Mark, M.; Chambon, P.; et al. The nuclear receptor superfamily: The second decade. Cell 1995, 83, 835–839. [Google Scholar] [CrossRef]
- Rastinejad, F.; Huang, P.; Chandra, V.; Khorasanizadeh, S. Understanding nuclear receptor form and function using structural biology. J. Mol. Endocrinol. 2013, 51, T1–T21. [Google Scholar] [CrossRef]
- Luong, K.V.Q.; Nguyen, L.T.H.; Lương, K.V.Q.; Nguyễn, L.T.H. The beneficial role of vitamin D and its analogs in cancer treatment and prevention. Crit. Rev. Oncol. 2010, 73, 192–201. [Google Scholar] [CrossRef]
- Schwartz, Z.; Sylvia, V.L.; Larsson, D.; Nemere, I.; Casasola, D.; Dean, D.D.; Boyan, B.D. 1α,25(OH) 2 D 3 Regulates Chondrocyte Matrix Vesicle Protein Kinase C (PKC) Directly via G-protein-dependent Mechanisms and Indirectly via Incorporation of PKC during Matrix Vesicle Biogenesis. J. Boil. Chem. 2002, 277, 11828–11837. [Google Scholar] [CrossRef]
- Zhang, C.; Tang, W.; Li, Y.; Yang, F.; Dowd, D.R.; Macdonald, P.N. Osteoblast-Specific Transcription Factor Osterix Increases Vitamin D Receptor Gene Expression in Osteoblasts. PLoS ONE 2011, 6, e26504. [Google Scholar] [CrossRef]
- Valdivielso, J.M.; Fernández, E. Vitamin D receptor polymorphisms and diseases. Clin. Chim. Acta 2006, 371, 1–12. [Google Scholar] [CrossRef]
- Vdr MGI Mouse Gene Detail—MGI:103076—Vitamin D (1,25-Dihydroxyvitamin D3) Receptor. Available online: http://www.informatics.jax.org/marker/MGI:103076 (accessed on 16 April 2019).
- Garcia-Vallvé, S.; Palau, J. Nuclear receptors, nuclear-receptor factors, and nuclear-receptor-like orphans form a large paralog cluster in Homo sapiens. Mol. Boil. Evol. 1998, 15, 665–682. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kouzmenko, A.; Ohtake, F.; Fujiki, R.; Kato, S. Chapter 12—Epigenetic Modifications in Vitamin D Receptor-mediated Transrepression. In Vitamin D, 3rd ed.; Feldman, D., Pike, J.W., Adams, J.S., Eds.; Academic Press: San Diego, CA, USA, 2011; pp. 227–234. ISBN 978-0-12-381978-9. [Google Scholar]
- Wei, P.; Inamdar, N.; Vedeckis, W.V. Transrepression of c- jun Gene Expression by the Glucocorticoid Receptor Requires Both AP-1 Sites in the c- jun Promoter. Mol. Endocrinol. 1998, 12, 1322–1333. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wu, J.; Hsieh, J.C.; Whitfield, G.K.; Jurutka, P.W.; Haussler, M.R.; Gardner, D.G. Suppression of ANP Gene Transcription by Liganded Vitamin D Receptor. Hypertension 1998, 31, 1338–1342. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, S.; Väisänen, S.; Pehkonen, P.; Seuter, S.; Benes, V.; Carlberg, C. Nuclear hormone 1α,25-dihydroxyvitamin D3 elicits a genome-wide shift in the locations of VDR chromatin occupancy. Nucleic Acids Res. 2011, 39, 9181–9193. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M.B.; Goetsch, P.D.; Pike, J.W. VDR/RXR and TCF4/β-catenin cistromes in colonic cells of colorectal tumor origin: Impact on c-FOS and c-MYC gene expression. Mol. Endocrinol. 2012, 26, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Meyer, M.B. Regulation of Mouse Cyp24a1 Expression via Promoter-Proximal and Downstream-Distal Enhancers Highlights New Concepts of 1,25-Dihydroxyvitamin D3 Action. Arch. Biochem. Biophys. 2012, 523, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C.; Seuter, S.; De Mello, V.D.F.; Schwab, U.; Voutilainen, S.; Pulkki, K.; Nurmi, T.; Virtanen, J.; Tuomainen, T.-P.; Uusitupa, M. Primary Vitamin D Target Genes Allow a Categorization of Possible Benefits of Vitamin D3 Supplementation. PLoS ONE 2013, 8, e71042. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.W.; Mizwicki, M.T.; Norman, D.P.G. Steroid-hormone rapid actions, membrane receptors and a conformational ensemble model. Nat. Rev. Drug Discov. 2004, 3, 27–41. [Google Scholar] [CrossRef]
- Rochel, N.; Wurtz, J.-M.; Mitschler, A.; Klaholz, B.; Moras, D. The Crystal Structure of the Nuclear Receptor for Vitamin D Bound to Its Natural Ligand. Mol. Cell 2000, 5, 173–179. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Renganaath, K.; Mehrotra, R.; Mehrotra, S. Combinatorial Control of Gene Expression. BioMed Res. Int. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Uitterlinden, A.G.; Fang, Y.; Van Meurs, J.B.; Pols, H.A.; Van Leeuwen, J.P. Genetics and biology of vitamin D receptor polymorphisms. Gene 2004, 338, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Xie, Z.-F. Polymorphisms in the vitamin D receptor gene and multiple sclerosis risk: A meta-analysis of case-control studies. J. Neurol. Sci. 2012, 313, 79–85. [Google Scholar] [CrossRef]
- Hii, C.S.; Ferrante, A. The Non-Genomic Actions of Vitamin D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Doroudi, M.; Cheung, J.; Grozier, A.L.; Schwartz, Z.; Boyan, B.D. Plasma membrane Pdia3 and VDR interact to elicit rapid responses to 1α,25(OH)2D3. Cell Signal. 2013, 25, 2362–2373. [Google Scholar] [CrossRef] [PubMed]
- Nemere, I.; Farach-Carson, M.C.; Rohe, B.; Sterling, T.M.; Norman, A.W.; Boyan, B.D.; Safford, S.E. Ribozyme knockdown functionally links a 1,25(OH)2D3 membrane binding protein (1,25D3-MARRS) and phosphate uptake in intestinal cells. Proc. Natl. Acad. Sci. USA 2004, 101, 7392–7397. [Google Scholar] [CrossRef]
- Nemere, I. Immunochemical studies on the putative plasmalemmal receptor for 1,25-dihydroxyvitamin D3. III. Vitamin D status. Steroids 2000, 65, 451–457. [Google Scholar] [CrossRef]
- Pike, J.W.; Meyer, M.B. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D3. Endocrinol. Metab. Clin. N. Am. 2010, 39, 255–269. [Google Scholar] [CrossRef]
- Larriba, M.J.; González-Sancho, J.M.; Bonilla, F.; Muñoz, A. Interaction of vitamin D with membrane-based signaling pathways. Front. Physiol. 2014, 5, 60. [Google Scholar] [CrossRef]
- Zhao, G.; Simpson, R.U. Interaction between vitamin D receptor with caveolin-3 and regulation by 1,25-dihydroxyvitamin D3 in adult rat cardiomyocytes. J. Steroid Biochem. Mol. Biol. 2010, 121, 159–163. [Google Scholar] [CrossRef]
- Garbi, N.; Tanaka, S.; Momburg, F.; Hämmerling, G.J. Impaired assembly of the major histocompatibility complex class I peptide-loading complex in mice deficient in the oxidoreductase ERp57. Nat. Immunol. 2006, 7, 93–102. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, J.; Lee, C.S.; Nizkorodov, A.; Riemenschneider, K.; Martin, D.; Hyzy, S.; Schwartz, Z.; Boyan, B.D. Disruption of Pdia3 gene results in bone abnormality and affects 1alpha,25-dihydroxy-vitamin D3-induced rapid activation of PKC. J. Steroid Biochem. Mol. Biol. 2010, 121, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Nemere, I.; Garbi, N.; Hämmerling, G.J.; Khanal, R.C. Intestinal Cell Calcium Uptake and the Targeted Knockout of the 1,25D3-MARRS (Membrane-associated, Rapid Response Steroid-binding) Receptor/PDIA3/Erp57. J. Boil. Chem. 2010, 285, 31859–31866. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Sylvia, V.L.; McKinney, N.; Schwartz, Z. Membrane actions of vitamin D metabolites 1α,25(OH)2D3 and 24R,25(OH)2D3 are retained in growth plate cartilage cells from vitamin D receptor knockout mice. J. Cell. Biochem. 2003, 90, 1207–1223. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.H.; Murray, F.; Insel, P.A. Caveolae as Organizers of Pharmacologically Relevant Signal Transduction Molecules. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 359–391. [Google Scholar] [CrossRef] [PubMed]
- Bosch, M.; Marí, M.; Herms, A.; Fernández, A.; Fajardo, A.; Kassan, A.; Giralt, A.; Colell, A.; Balgoma, D.; Barbero, E.; et al. Caveolin-1 deficiency causes cholesterol dependent mitochondrial dysfunction and apoptotic susceptibility. Curr. Boil. 2011, 21, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Mesbah, M.; Nemere, I.; Papagerakis, P.; Nefussi, J.-R.; Nessmann, C.; Berdal, A.; Orestes-Cardoso, S.; Orestes-Cardoso, S. Expression of a 1,25-Dihydroxyvitamin D3 Membrane-Associated Rapid-Response Steroid Binding Protein During Human Tooth and Bone Development and Biomineralization. J. Bone Miner. Res. 2002, 17, 1588–1596. [Google Scholar] [CrossRef]
- Norman, A.W. Receptors for 1α,25(OH)2D3: Past, Present, and Future. J. Bone Miner. Res. 1998, 13, 1360–1369. [Google Scholar] [CrossRef]
- Richard, C.L.; Farach-Carson, M.C.; Rohe, B.; Nemere, I.; Meckling, K.A. Involvement of 1,25D3-MARRS (membrane associated, rapid response steroid-binding), a novel vitamin D receptor, in growth inhibition of breast cancer cells. Exp. Cell Res. 2010, 316, 695–703. [Google Scholar] [CrossRef]
- Farach-Carson, M.; Nemere, I. Membrane Receptors for Vitamin D Steroid Hormones: Potential NewDrug Targets. Curr. Drug Targets 2003, 4, 67–76. [Google Scholar] [CrossRef]
- Sitrin, M. Rapid effects of 1,25(OH)2 vitamin D3 on signal transduction systems in colonic cells. Steroids 1999, 64, 137–142. [Google Scholar] [CrossRef]
- Chen, A.; Davis, B.H.; Bissonnette, M.; Scaglione-Sewell, B.; Brasitus, T.A. 1,25-Dihydroxyvitamin D3 Stimulates Activator Protein-1-dependent Caco-2 Cell Differentiation. J. Boil. Chem. 1999, 274, 35505–35513. [Google Scholar] [CrossRef]
- Doroudi, M.; Boyan, B.D.; Schwartz, Z. Rapid 1α,25(OH)2D3 membrane-mediated activation of Ca2+/calmodulin-dependent protein kinase II in growth plate chondrocytes requires Pdia3, PLAA and caveolae. Connect. Tissue Res. 2014, 55, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Rohe, B.; Safford, S.E.; Nemere, I.; Farach-Carson, M.C. Identification and characterization of 1,25D3-membrane-associated rapid response, steroid (1,25D3-MARRS)-binding protein in rat IEC-6 cells. Steroids 2005, 70, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Nemere, I.; Safford, S.E.; Rohe, B.; DeSouza, M.M.; Farach-Carson, M.C. Identification and characterization of 1,25D3-membrane-associated rapid response, steroid (1,25D3-MARRS) binding protein. J. Steroid Biochem. Mol. Boil. 2004, 89, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.; Bikle, D.D.; Su, M.J.; Ratnam, A.; Abe, J. 1,25-Dihydroxyvitamin D3 upregulates the phosphatidylinositol signaling pathway in human keratinocytes by increasing phospholipase C levels. J. Clin. Investig. 1995, 96, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Alkon, D.; Rasmussen, H. A spatial-temporal model of cell activation. Science 1988, 239, 998–1005. [Google Scholar] [CrossRef]
- Sato, K.; Imaki, T.; Toraya, S.; Demura, H.; Tanaka, M.; Kasajima, T.; Takeuchi, A.; Kobayashi, T. Increased 1,25-(OH)2D2 Concentration in a Patient with Malignancy-Associated Hypercalcemia Receiving Intravenous Hyperalimentation Inadvertently Supplemented with Vitamin D2. Intern. Med. 1993, 32, 886–890. [Google Scholar] [CrossRef][Green Version]
- Boyan, B.D.; Wang, L.; Wong, K.L.; Jo, H.; Schwartz, Z. Plasma membrane requirements for 1alpha,25(OH)2D3 dependent PKC signaling in chondrocytes and osteoblasts. Steroids 2006, 71, 286–290. [Google Scholar] [CrossRef]
- Schwartz, Z.; Shaked, D.; Hardin, R.; Gruwell, S.; Dean, D.; Sylvia, V.; Boyan, B.; Dean, D. 1α,25(OH)2D3 causes a rapid increase in phosphatidylinositol-specific PLC-β activity via phospholipase A2-dependent production of lysophospholipid. Steroids 2003, 68, 423–437. [Google Scholar] [CrossRef]
- Peehl, D.M.; Skowronski, R.J.; Leung, G.K.; Wong, S.T.; Stamey, T.A.; Feldman, D. Antiproliferative effects of 1,25-dihydroxyvitamin D3 on primary cultures of human prostatic cells. Cancer Res. 1994, 54, 805–810. [Google Scholar]
- Lin, R.; Nagai, Y.; Sladek, R.; Bastien, Y.; Ho, J.; Petrecca, K.; Sotiropoulou, G.; Diamandis, E.P.; Hudson, T.J.; White, J.H. Expression Profiling in Squamous Carcinoma Cells Reveals Pleiotropic Effects of Vitamin D3 Analog EB1089 Signaling on Cell Proliferation, Differentiation, and Immune System Regulation. Mol. Endocrinol. 2002, 16, 1243–1256. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.-Y.; Yao, J.; Lee, Y.-F. 1alpha, 25-dihydroxyvitamin D3 suppresses interleukin-8-mediated prostate cancer cell angiogenesis. Carcinogenesis 2006, 27, 1883–1893. [Google Scholar] [CrossRef] [PubMed]
- Hilliard, G.M.; Cook, R.G.; Weigel, N.L.; Pike, J.W. 1,25-Dihydroxyvitamin D3 modulates phosphorylation of serine 205 in the human vitamin D receptor: Site-directed mutagenesis of this residue promotes alternative phosphorylation. Biochemistry 1994, 33, 4300–4311. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Beilhartz, G.; Roy, Y.; Richard, C.L.; Curtin, M.; Brown, L.; Cadieux, D.; Coppolino, M.; Farach-Carson, M.C.; Nemere, I.; et al. Nuclear translocation of the 1,25D3-MARRS (membrane associated rapid response to steroids) receptor protein and NFkappaB in differentiating NB4 leukemia cells. Exp. Cell Res. 2010, 316, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Barletta, F.; Freedman, L.P.; Christakos, S. Enhancement of VDR-Mediated Transcription by Phosphorylation: Correlation with Increased Interaction between the VDR and DRIP205, a Subunit of the VDR-Interacting Protein Coactivator Complex. Mol. Endocrinol. 2002, 16, 301–314. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Norman, A.W. Vitamin D Receptor: New Assignments for an Already Busy Receptor. Endocrinology 2006, 147, 5542–5548. [Google Scholar] [CrossRef] [PubMed]
- Deeb, K.K.; Trump, D.L.; Johnson, C.S. Vitamin D signalling pathways in cancer: Potential for anticancer therapeutics. Nat. Rev. Cancer 2007, 7, 684–700. [Google Scholar] [CrossRef]
- Nussey, S.; Whitehead, S. The Parathyroid Glands and Vitamin D; BIOS Scientific Publishers: Didcott, UK, 2001. [Google Scholar]
- Tanaka, Y.; DeLuca, H. Bone mineral mobilization activity of 1,25-dihydroxycholecalciferol, a metabolite of vitamin D. Arch. Biochem. Biophys. 1971, 146, 574–578. [Google Scholar] [CrossRef]
- Parathyroid Hormone|You and Your Hormones from the Society for Endocrinology. Available online: http://www.yourhormones.info/hormones/parathyroid-hormone/ (accessed on 8 April 2019).
- Garabedian, M.; Holick, M.; DeLuca, H.F.; Boyle, I.T. Control of 25-Hydroxycholecalciferol Metabolism by Parathyroid Glands. Proc. Natl. Acad. Sci. USA 1972, 69, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Brenza, H.L.; DeLuca, H.F. Regulation of 25-Hydroxyvitamin D3 1α-Hydroxylase Gene Expression by Parathyroid Hormone and 1,25-Dihydroxyvitamin D3. Arch. Biochem. Biophys. 2000, 381, 143–152. [Google Scholar] [CrossRef]
- Chambers, T.J.; Magnus, C.J. Calcitonin alters behaviour of isolated osteoclasts. J. Pathol. 1982, 136, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Shinki, T.; Ueno, Y.; DeLuca, H.F.; Suda, T. Calcitonin is a major regulator for the expression of renal 25-hydroxyvitamin D3-1α-hydroxylase gene in normocalcemic rats. Proc. Natl. Acad. Sci. USA 1999, 96, 8253–8258. [Google Scholar] [CrossRef] [PubMed]
- Genetics Home Reference. CYP24A1 Gene. Available online: https://ghr.nlm.nih.gov/gene/CYP24A1 (accessed on 8 April 2019).
- Vuolo, L.; Di Somma, C.; Faggiano, A.; Colao, A. Vitamin D and cancer. Front. Endocrinol. 2012, 3, 58. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.; Kost, S.B.; Ennis, B.; Stumpf, W.; Kumar, R. Effect of 1,25-dihydroxyvitamin D3 on mouse mammary tumor (GR) cells: Evidence for receptors, cellular uptake, inhibition of growth and alteration in morphology at physiologic concentrations of hormone. J. Bone Miner. Res. 1986, 1, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Narvaez, C.J.; Matthews, D.; LaPorta, E.; Simmons, K.M.; Beaudin, S.; Welsh, J. The impact of vitamin D in breast cancer: Genomics, pathways, metabolism. Front. Physiol. 2014, 5, 5. [Google Scholar] [CrossRef]
- Fleet, J.C. Molecular Actions of Vitamin D Contributing to Cancer Prevention. Mol. Asp. Med. 2008, 29, 388–396. [Google Scholar] [CrossRef]
- Skowronski, R.J.; Peehl, D.M.; Feldman, D. Vitamin D and prostate cancer: 1,25 dihydroxyvitamin D3 receptors and actions in human prostate cancer cell lines. Endocrinology 1993, 132, 1952–1960. [Google Scholar] [CrossRef] [PubMed]
- Basit, S. Vitamin D in health and disease: A literature review. Br. J. Biomed. Sci. 2013, 70, 161–172. [Google Scholar] [CrossRef]
- Mahendra, A.; Karishma; Choudhury, B.K.; Sharma, T.; Bansal, N.; Bansal, R.; Gupta, S. Vitamin D and gastrointestinal cancer. J. Lab. Physicians 2018, 10, 1–5. [Google Scholar]
- Leigh-Clare, J.L. A note on the vitamin D content of the stomach oil of the australasian petrel (aestralata lessoni). Biochem. J. 1927, 21, 725–727. [Google Scholar] [CrossRef]
- Selye, H.; Bois, P. On the role of corticoids in conditioning the gastric mucosa to certain toxic actions of ergocalciferol. Br. J. Nutr. 1957, 11, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, W.; Sar, M.; Reid, F.; Tanaka, Y.; DeLuca, H. Target cells for 1,25-dihydroxyvitamin D3 in intestinal tract, stomach, kidney, skin, pituitary, and parathyroid. Science 1979, 206, 1188–1190. [Google Scholar] [CrossRef] [PubMed]
- Kirui, N.A.; Weisbrode, S.E.; Kindig, O.R. The role of dietary calcium on the development of soft tissue mineralization due to 1,25-dihydroxyvitamin D3 intoxication in rats. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1981, 37, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Selking, Ö.; Borch, K.; Johansson, H.; Ljunghall, S.; Wide, L. Evaluation of Parathyroid Function in Patients with Hypergastrinaemia and Pernicious Anaemia. Upsala J. Med Sci. 1982, 87, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Kurose, T.; Seino, Y.; Ishida, H.; Tsuji, K.; Fukumoto, H.; Koh, G.; Takeda, J.; Kitano, N.; Inagaki, N.; Tsuda, K.; et al. Effect of vitamin D on gastrin and gastric somatostatin secretion from the isolated perfused rat stomach. Life Sci. 1988, 42, 1995–2001. [Google Scholar] [CrossRef]
- Axelson, J.; Persson, P.; Gagnemo-Persson, R.; Hakanson, R. Importance of the stomach in maintaining calcium homoeostasis in the rat. Gut 1991, 32, 1298–1302. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.; Holick, M. Noncalcemic actions of 1,25-dihydroxyvitamin D3 and clinical applications. Bone 1995, 17, S107–S111. [Google Scholar] [CrossRef]
- Stumpf, W.E. Vitamin D sites and mechanisms of action: A histochemical perspective. Reflections on the utility of autoradiography and cytopharmacology for drug targeting. Histochem. Cell Boil. 1995, 104, 417–427. [Google Scholar] [CrossRef]
- Ikezaki, S.; Nishikawa, A.; Furukawa, F.; Tanakamura, Z.; Kim, H.C.; Mori, H.; Takahashi, M. Chemopreventive effects of 24R,25-dihydroxyvitamin D3, a vitamin D3 derivative, on glandular stomach carcinogenesis induced in rats by N-methyl-N’-nitro-N-nitrosoguanidine and sodium chloride. Cancer Res. 1996, 56, 2767–2770. [Google Scholar]
- Gagnemo-Persson, R.; Persson, P.; Bryngelsson, T.; Green, B.; Håkanson, R. Rat stomach ECL-cell histidine decarboxylase activity is suppressed by ergocalciferol but unaffected by parathyroid hormone and calcitonin. Regul. Pept. 1999, 79, 131–139. [Google Scholar] [CrossRef]
- Stumpf, W.E. Vitamin D and the digestive system. Eur. J. Drug Metab. Pharmacokinet. 2008, 33, 85–100. [Google Scholar] [CrossRef] [PubMed]
- Häkkinen, I.; Lindgren, I. The localization of tetracycline in the metastatic calcifications in the stomach of rat induced by overdosage of dihydrotachysterol and vitamin D3. Acta Pathol. Microbiol. Scand. 2009, 59, 428–434. [Google Scholar] [CrossRef]
- Sahin, H.H.; Cumbul, A.; Uslu, Ü.; Yilmaz, Z.; Ercan, F.; Alican, I. The effect of 1,25 dihydroxyvitamin D3 on HCl/Ethanol-induced gastric injury in rats. Tissue Cell 2018, 51, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Paterson, C.R.; Woods, C.G. Search for osteomalacia in 1228 patients after gastrectomy and other operations on the stomach. Lancet 1965, 2, 1085–1088. [Google Scholar]
- Kimura, K.; Nozawa, Y.; Kitamura, S.; Takahashi, H.; Ota, M.; Norimatsu, H.; Wada, H. An autopsy case of hypervitaminosis D. Pathol. Int. 1967, 17, 377–386. [Google Scholar] [CrossRef]
- Lawrence, W. Nutritional consequences of surgical resection of the gastrointestinal tract for cancer. Cancer Res. 1977, 37, 2379–2386. [Google Scholar] [PubMed]
- Giovannucci, E.; Liu, Y.; Willett, W.C. Cancer Incidence and Mortality and Vitamin D in Black and White Male Health Professionals. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2467–2472. [Google Scholar] [CrossRef]
- Chen, W.; Dawsey, S.M.; Qiao, Y.-L.; Mark, S.D.; Dong, Z.-W.; Taylor, P.R.; Zhao, P.; Abnet, C.C. Prospective study of serum 25(OH)-vitamin D concentration and risk of oesophageal and gastric cancers. Br. J. Cancer 2007, 97, 123–128. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pan, L.; Matloob, A.F.; Du, J.; Pan, H.; Dong, Z.; Zhao, J.; Feng, Y.; Zhong, Y.; Huang, B.; Lu, J. Vitamin D stimulates apoptosis in gastric cancer cells in synergy with trichostatin A/sodium butyrate-induced and 5-aza-2′-deoxycytidine-induced PTEN upregulation. FEBS J. 2010, 277, 989–999. [Google Scholar] [CrossRef] [PubMed]
- Antico, A.; Tozzoli, R.; Giavarina, D.; Tonutti, E.; Bizzaro, N. Hypovitaminosis D as predisposing factor for atrophic type A gastritis: A case-control study and review of the literature on the interaction of Vitamin D with the immune system. Clin. Rev. Allergy Immunol. 2012, 42, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Park, M.R.; Lee, J.H.; Park, M.S.; Hwang, J.E.; Shim, H.J.; Cho, S.H.; Chung, I.-J.; Bae, W.K. Suppressive Effect of 19-nor-1α-25-Dihydroxyvitamin D2 on Gastric Cancer Cells and Peritoneal Metastasis Model. J. Korean Med. Sci. 2012, 27, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Kopic, S.; Geibel, J.P. Gastric Acid, Calcium Absorption, and Their Impact on Bone Health. Physiol. Rev. 2013, 93, 189–268. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Chen, W.; Zhu, H.; Chen, Y.; Wan, X.; Yang, N.; Xu, S.; Yu, C.; Chen, L. Helicobacter pylori induces increased expression of the vitamin D receptor in immune responses. Helicobacter 2014, 19, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Da, M.; Zhang, Y.; Peng, L.; Yao, J.; Duan, Y. Alterations in vitamin D signaling pathway in gastric cancer progression: A study of vitamin D receptor expression in human normal, premalignant, and malignant gastric tissue. Int. J. Clin. Exp. Pathol. 2015, 8, 13176–13184. [Google Scholar]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Högenauer, C.; Pieber, T.R. Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur. J. Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef]
- Vyas, N.; Companioni, R.C.; Tiba, M.; AlKhawam, H.; Catalano, C.; Sogomonian, R.; Baum, J.; Walfish, A. Association between serum vitamin D levels and gastric cancer: A retrospective chart analysis. World J. Gastrointest. Oncol. 2016, 8, 688–694. [Google Scholar] [CrossRef]
- Du, C.; Yang, S.; Zhao, X.; Dong, H. Pathogenic roles of alterations in vitamin D and vitamin D receptor in gastric tumorigenesis. Oncotarget 2017, 8, 29474–29486. [Google Scholar] [CrossRef]
- Yıldırım, O.; Yildirim, T.; Seçkin, Y.; Osanmaz, P.; Bilgic, Y.; Mete, R. The influence of vitamin D deficiency on eradication rates of Helicobacter pylori. Adv. Clin. Exp. Med. 2017, 26, 1377–1381. [Google Scholar] [CrossRef]
- El Shahawy, M.S.; Hemida, M.H.; El Metwaly, I.; Shady, Z.M. The effect of vitamin D deficiency on eradication rates of Helicobacter pylori infection. JGH Open 2018, 2, 270–275. [Google Scholar] [CrossRef]
- Fletcher, J.; Cooper, S.C.; Ghosh, S.; Hewison, M. The Role of Vitamin D in Inflammatory Bowel Disease: Mechanism to Management. Nutrients 2019, 11, 1019. [Google Scholar] [CrossRef]
- Clark, I.; Smith, M.R. Effects of hypervitaminosis A and D on skeletal metabolism. J. Boil. Chem. 1964, 239, 1266–1271. [Google Scholar]
- DeWind, L.T. Hypervitaminosis D with Osteosclerosis. Arch. Dis. Child. 1961, 36, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Compston, J. Vitamin D. Molecular Biology, Physiology and Clinical Applications. Gut 2000, 46, 584. [Google Scholar] [CrossRef][Green Version]
- Albrechtsson, E.; Jonsson, T.; Möller, S.; Höglund, M.; Ohlsson, B.; Axelson, J. Vitamin D receptor is expressed in pancreatic cancer cells and a vitamin D3 analogue decreases cell number. Pancreatology 2003, 3, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the intracrinology of innate immunity. Mol. Cell. Endocrinol. 2010, 321, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Sandgren, M.E.; Brönnegård, M.; DeLuca, H.F. Tissue distribution of the 1,25-dihydroxyvitamin D3 receptor in the male rat. Biochem. Biophys. Res. Commun. 1991, 181, 611–616. [Google Scholar] [CrossRef]
- Nilas, L.; Christiansen, C.; Christiansen, J. Regulation of vitamin D and calcium metabolism after gastrectomy. Gut 1985, 26, 252–257. [Google Scholar] [CrossRef]
- Chakhtoura, M.T.; Nakhoul, N.F.; Akl, E.A.; Safadi, B.Y.; Mantzoros, C.S.; Fuleihan, G.E.-H. Vitamin D supplementation for obese adults undergoing bariatric surgery. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Carrasco, F.; Basfi-Fer, K.; Rojas, P.; Valencia, A.; Csendes, A.; Codoceo, J.; Inostroza, J.; Ruz, M. Changes in Bone Mineral Density After Sleeve Gastrectomy or Gastric Bypass: Relationships with Variations in Vitamin D, Ghrelin, and Adiponectin Levels. Obes. Surg. 2014, 24, 877–884. [Google Scholar] [CrossRef]
- Eddy, R.L. Metabolic bone disease after gastrectomy. Am. J. Med. 1971, 50, 442–449. [Google Scholar] [CrossRef]
- Zittel, T.T.; Zeeb, B.; Maier, G.W.; Kaiser, G.W.; Zwirner, M.; Liebich, H.; Starlinger, M.; Becker, H.D. High prevalence of bone disorders after gastrectomy. Am. J. Surg. 1997, 174, 431–438. [Google Scholar] [CrossRef]
- Rino, Y.; Oshima, T.; Yoshikawa, T. Changes in fat-soluble vitamin levels after gastrectomy for gastric cancer. Surg. Today 2017, 47, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.L.; Peura, D.A. Control of Gastric Acid Secretion in Health and Disease. Gastroenterology 2008, 134, 1842–1860. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Petersen, C.D.; Coy, D.H.; Jiang, J.-K.; Thomas, C.J.; Pollak, M.R.; Wank, S.A. Calcium-sensing receptor is a physiologic multimodal chemosensor regulating gastric G-cell growth and gastrin secretion. Proc. Natl. Acad. Sci. USA 2010, 107, 17791–17796. [Google Scholar] [CrossRef]
- Kantham, L.; Quinn, S.J.; Egbuna, O.I.; Baxi, K.; Butters, R.; Pang, J.L.; Pollak, M.R.; Goltzman, D.; Brown, E.M. The calcium-sensing receptor (CaSR) defends against hypercalcemia independently of its regulation of parathyroid hormone secretion. Am. J. Physiol. Metab. 2009, 297, E915–E923. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, J.; Rehfeld, J.F.; Stadil, F. The effect of calcium on gastric acid and gastrin secretion in antrectomized subjects. Gut 1974, 15, 622–625. [Google Scholar] [CrossRef][Green Version]
- Harris, S.S. Vitamin D and African Americans. J. Nutr. 2006, 136, 1126–1129. [Google Scholar] [CrossRef]
- Dixon, M.F. Pathology of Gastritis and Peptic Ulceration. In Helicobacter Pylori: Physiology and Genetics; Mobley, H.L., Mendz, G.L., Hazell, S.L., Eds.; ASM Press: Washington, DC, USA, 2001; ISBN 978-1-55581-213-3. [Google Scholar]
- Waterhouse, M.; Hope, B.; Krause, L.; Morrison, M.; Protani, M.M.; Zakrzewski, M.; Neale, R.E. Vitamin D and the gut microbiome: A systematic review of in vivo studies. Eur. J. Nutr. 2018, 1–16. [Google Scholar] [CrossRef]
- Gigek, C.O.; Chen, E.S.; Calcagno, D.Q.; Wisnieski, F.; Burbano, R.R.; Smith, M.A.C. Epigenetic mechanisms in gastric cancer. Epigenomics 2012, 4, 279–294. [Google Scholar] [CrossRef]
- De Ruijter, A.J.; Van Gennip, A.H.; Caron, H.N.; Kemp, S.; Van Kuilenburg, A.B. Histone deacetylases (HDACs): Characterization of the classical HDAC family. Biochem. J. 2003, 370, 737–749. [Google Scholar] [CrossRef]
- Meshorer, E.; Misteli, T. Chromatin in pluripotent embryonic stem cells and differentiation. Nat. Rev. Mol. Cell Boil. 2006, 7, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Dokmanovic, M.; Clarke, C.; Marks, P.A. Histone Deacetylase Inhibitors: Overview and Perspectives. Mol. Cancer Res. 2007, 5, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A. The Role of DNA Methylation in Mammalian Epigenetics. Science 2001, 293, 1068–1070. [Google Scholar] [CrossRef] [PubMed]
- Gowher, H.; Jeltsch, A. Mechanism of inhibition of DNA methyltransferases by cytidine analogs in cancer therapy. Cancer Boil. Ther. 2004, 3, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
| 25(OH)D Levels | Condition |
|---|---|
| <30 ng/mL | Severe deficiency |
| 30–50 ng/mL | Minor deficiency |
| 50–70 ng/mL | Adequate levels |
| >80 ng/mL | Excess |
| Target Gene | Cell Type | Function |
|---|---|---|
| ASAP2 (ArfGAP with SH3 domain, ankyrin repeat and PH domain 2) | Human THP-1 cells (monocytes) | Regulates autophagy, cellular migration and vesicular transport [44] |
| CYP24A1 (Cytochrome P450 24A1) | Kidney | Vitamin-D3 catabolizing enzyme [82] |
| PTHLH (Parathyroid hormone-like hormone) | Cytoplasm, golgi complex and nucleus of most cells | Activates PLC signaling pathways, proliferation of chondrocytes, regulation of bone formation by promoting recruitment and survival of osteoblasts, and plays a role in the physiological regulation of bone resorption [83] |
| CAMP (Cathelicidin antimicrobial protein) | Primary keratinocytes, monocytes, phagocytes, B cells and neutrophils | Antibacterial, antifungal and antiviral activities, the encoded protein functions in cell chemotaxis, immune mediator induction and inflammatory response regulation [84] |
| Trpv6 (Transient Receptor Potential Vanilloid 6) | Brush border membranes of the intestinal epithelia | Potential mediator of calcium uptake into the enterocyte [85] |
| CYP27B1(Cytochrome P450 27B1) | Kidneys, epithelial cells, lungs, breast, intestine, stomach, endocrine glands, cells of the immune system, osteoblasts chondrocytes | Expression of 1a-hydroxylase [44] |
| CALB1 | Avian intestine and kidney and mammalian intestine, respectively | Codes for Calbindin-D28K and Calbindin-D9K proteins, upregulated by 1α,25(OH)2D3 [86,87] |
| Osteocalcin | Osteocytes, cartilages | Mineral deposition, bone resorption [86] |
| FOXP3 (fork head box P3) | Immune system | Maturity and performance of T regulatory cells [88] |
| CD14 (Cluster of differentiation) | Monocytes and most tissue macrophages and to a minor extent in monoblasts and promonocytes | Interacts with soluble lipopolysaccharide (LPS) released from gram-negative bacteria in combination with a plasma protein, LPS-binding protein [44] |
| NINJ1 (Ninjurin 1) | Hepatic stellate cells | Promotes axonal growth, may play a role in nerve regeneration and in the formation and function of other tissues [89] |
| No. | References | Organism Studied | Form of vitamin D Used in the Study | Key Findings | Ref. No. |
|---|---|---|---|---|---|
| 1 | Sato K. (1993) | Rats | 1α,25(OH)2D3 | Elevated levels of 1α,25(OH)2D3 when administered to hypercalcemic patient with PTHrP-producing gastric carcinoma, aggravated the severity of malignancy-associated hypercalcemia (MAH). | [110] |
| 2 | Leigh-Clare J. (1927) | Australasian Petrel | Vitamin D | One of the first articles to describe the presence of Vitamin D in stomach oil. The study was an attempt to elucidate the source of vitamin D in the oil. | [136] |
| 3 | Selye H. and Bois (1957) | Sprague-Dawley rats | Ergocalciferol | VDT lead to calcium deposition in the muscularis of rat stomachs | [137] |
| 4 | Stumpf W. (1979) | Rats | 1α,25(OH)2D3 and its metabolites | Vitamin D receptors for 1α,25(OH)2D3 or its metabolites target tissues of the GIT including the nuclei of some of the cells of the stomach | [138] |
| 5 | Kirui N. (1981) | Rats | 1α,25(OH)2D3 | 1α,25(OH)2D3 has a direct role on hypercalcemia and STM with lesions forming in most of the soft tissues, including the gastric glandular mucosa and muscularis | [139] |
| 6 | Selking O. (1982) | Rats | Ergocalciferol | Vitamin D induced hypercalcemia in parathyroidectomized rats is associated with a thickened gastric mucosa, but the serum gastrin, number of gastrin (G) cells or antral gastrin remained unchanged | [140] |
| 7 | Kurose, T. (1988) | Rats | 1α,25(OH)2D3 | Calcium and 1α,25(OH)2D3 deficiency impair gastrin and somatostain secretion in the perfused rat stomach | [141] |
| 8 | Axelson J. (1991) | Rats | 1α,25(OH)2D3 | Gastrectomy may lead to an increase in the levels of serum 1α,25(OH)2D3, resulting in enhanced absorption of calcium by the small intestine. This may be the cause for diseases like osteomalacia in the gastrectomized patients. | [142] |
| 9 | Holick M. (1995) | Rats, mice, humans | 1α,25(OH)2D3 | Non-calcemic tissues including the gonads, pituitary gland, thymus, pancreas, stomach, breast, and skin possess the nuclear receptors for 1α,25(OH)2D3, targeting these receptors with analogs of 1α,25(OH)2D3 provides treatment against different diseases. | [143] |
| 10 | Stumpf W. (1995) | Two- month old mice | 1α,25(OH)2D3 and its analogue 22-Oxacalcitriol (OCT) | Autoradiographic studies showed nuclear concentration and retention of 1α,25(OH)2D3 and its analogue 22-Oxacalcitriol (OCT) in neck mucus cells of gastric and pyloric glands and in dispersed endocrine cells in the antrum | [144] |
| 11 | Ikezaki S. (1996) | Male Wistar rats | 24R,25(OH)2D3 | The development of atypical hyperplasias and adenocarcinomas in the glandular stomachs was decreased by exposure to 24R,25(OH)2D3, which shows that 24R,25(OH)2D3 has chemopreventive effects. | [145] |
| 12 | Gagnemo- Persson R. (1999) | Male Sprague-Dawley rats | Ergocalciferol/Vitamin D2 | Gastrin–ECL-cell axis can be suppressed by vitamin D or by vitamin D-dependent mechanisms. Also, vitamin D receptor gene expression was seen in the rat oxyntic mucosa. | [146] |
| 13 | Stumpf W. (2008) | Rats, mice, hamsters and zebra finch | 1α,25(OH)2D3 | Autoradiography studies confirmed the binding of 1α,25(OH)2D3 and its oxygen analog OCT in numerous regions of the digestive tract | [147] |
| 14 | Häkkinen I. and Lindgren I. (2009) | Albino rats | 1α,25(OH)2D3 | Excess of 1α,25(OH)2D3 leads to calcification of gastric tissues | [148] |
| 15 | Sahin H. (2018) | Rats | 1α,25(OH)2D3 | 1α,25(OH)2D3 protects the gastric mucosa via attenuation of inflammatory reaction, oxidative stress and apoptosis. | [149] |
| No. | First Author, Year | Form of Vitamin D Used in the Study | Key Findings | Ref. No. |
|---|---|---|---|---|
| 1 | Paterson C. (1965) | 1α,25(OH)2D3 | Gastrectomy and other surgeries of the stomach can trigger osteomalacia due to the increased levels of serum vitamin D which results in increased absorption of calcium by the small intestine. | [150] |
| 2 | Kimura K. (1967) | 1α,25(OH)2D3 | Calcium deposits were found in the soft tissues of a patient who died of uremia after administration of toxic levels of Vitamin D for 6 months | [151] |
| 3 | Lawrence, W. (1977) | 1α,25(OH)2D3 | Inadequate absorption of Vitamin D in patients who underwent gastrectomy is due to steatorrhea which may contribute to osteomalacia later on. | [152] |
| 4 | Giovannucci E. (2006) | 1α,25(OH)2D3 | Black men with poor vitamin D status are highly susceptible for digestive system cancer and have a higher mortality rate. | [153] |
| 5 | Chen W. (2007) | 25(OH)D | A direct association between higher serum 25(OH)D concentration and increased risk of oesophageal squamous cell carcinomas (ESCC) in men but not women but no association with risk of gastric cardia or noncardia adenocarcinoma in either sex. | [154] |
| 6 | Pan L. (2010) | 1α,25(OH)2D3 | Studies in HGC-27 adenocarcinoma cells showed that Vitamin D can be used in gastric cancer therapies in association with trichostatin A ⁄ sodium butyrate and 5-aza-2-deoxycytidine | [155] |
| 7 | Antico A. (2012) | 1,25(OH)2D | Low vitamin D concentration in H. pylori gastritis patients might lead to a more severe Th1-type aggression to the stomach epithelium | [156] |
| 8 | Park M.R. (2012) | Paricalcitol (19-nor-1,25-(OH)2D2) | Paricalcitol, has anticancer activity on GC cells by regulating cell cycle, apoptosis and inflammation | [157] |
| 9 | Kopic S. (2013) | 1α,25(OH)2D3 | Gastric acid, gastrin secretion is increased in humans under conditions of hyperparathyroidism with the combined actions of both PTH and vitamin D | [158] |
| 10 | Guo L. (2014) | 1α,25(OH)2D3 | VDR and CAMP expression is up-regulated in the gastric epithelium during H. pylori infection; thus, VDR is important in maintaining the homeostasis of gastric mucosa and protection from H. pylori infection. | [159] |
| 11 | Wen Y (2015) | 1,25(OH)2D3 | VDR expression was seen to decline from the premalignant stage, to low expression in gastric cancer tissues. VDR could be a potential prognostic factor for patients with gastric cancer. | [160] |
| 12 | Bashir M. (2016) | 1α,25(OH)2D3 | Vitamin D influences the composition of gut microbiome. | [161] |
| 13 | Vyas N. (2016) | 25(OH)D | There is a positive relationship between VDD and gastric adenocarcinoma | [162] |
| 14 | Du C. (2017) | 1α,25(OH)2D3 | Vitamin D supplement might be a safe and economical way to prevent or treat gastric cancer | [163] |
| 15 | Yildirim O. (2017) | 25(OH)D | Deficiency of 25(OH)D may be a risk factor related to eradication failure of H. pylori, and supplementation of 25(OH)D before eradication of H. pylori may provide better results. | [164] |
| 16 | El Shahawy M.S. (2018) | 25(OH)D3 | 25(OH)D deficiency can be a risk factor in eradication failure of H. pylori infection. | [165] |
| 17 | Fletcher J. (2019) | 1α,25(OH)2D | 1α,25(OH)2D protects the gastrointestinal barrier and evokes immune responses | [166] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sirajudeen, S.; Shah, I.; Al Menhali, A. A Narrative Role of Vitamin D and Its Receptor: With Current Evidence on the Gastric Tissues. Int. J. Mol. Sci. 2019, 20, 3832. https://doi.org/10.3390/ijms20153832
Sirajudeen S, Shah I, Al Menhali A. A Narrative Role of Vitamin D and Its Receptor: With Current Evidence on the Gastric Tissues. International Journal of Molecular Sciences. 2019; 20(15):3832. https://doi.org/10.3390/ijms20153832
Chicago/Turabian StyleSirajudeen, Shaima, Iltaf Shah, and Asma Al Menhali. 2019. "A Narrative Role of Vitamin D and Its Receptor: With Current Evidence on the Gastric Tissues" International Journal of Molecular Sciences 20, no. 15: 3832. https://doi.org/10.3390/ijms20153832
APA StyleSirajudeen, S., Shah, I., & Al Menhali, A. (2019). A Narrative Role of Vitamin D and Its Receptor: With Current Evidence on the Gastric Tissues. International Journal of Molecular Sciences, 20(15), 3832. https://doi.org/10.3390/ijms20153832






