Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities
Abstract
1. Introduction
2. Interplay between ASCs and Cancer Cells: Mechanisms Underlying Tumor Progression in Experimental Models
2.1. The Role of ASCs in the Tumor Microenvironment and Cancer Progression
2.2. Exosome Releasing
2.3. Functional Changes in ASCs Induced by Cancer Cells
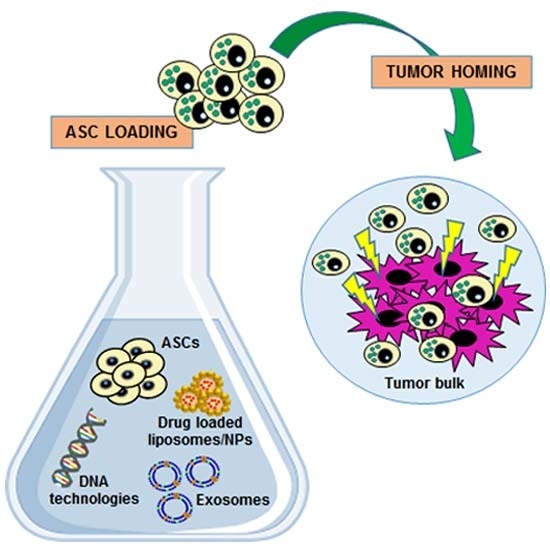
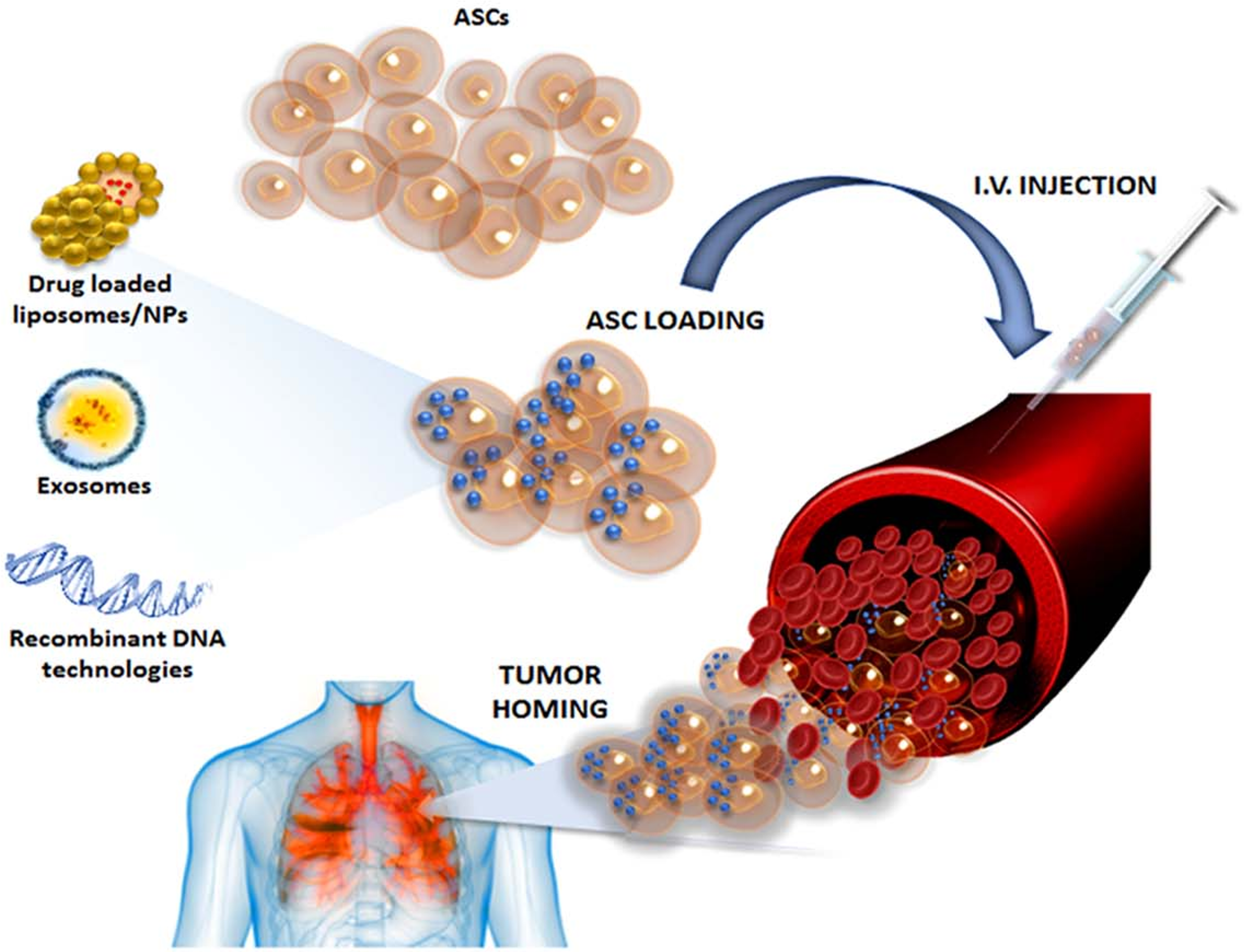
3. ASCs as Anti-Tumor Agent Carriers: A “Trojan Horse” Against Cancer Progression
4. Oncological Safety of ASC Clinical Applications
5. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Whiteside, T.L. The tumor microenvironment and its role in promoting tumor growth. Oncogene 2008, 27, 5904. [Google Scholar] [CrossRef] [PubMed]
- O’Halloran, N.; Courtney, D.; Kerin, M.J.; Lowery, A.J. Adipose-Derived Stem Cells in Novel Approaches to Breast Reconstruction: Their Suitability for Tissue Engineering and Oncological Safety. Breast Cancer Basic Clin. Res. 2017, 11, 1178223417726777. [Google Scholar] [CrossRef] [PubMed]
- Khamis, Z.I.; Sahab, Z.J.; Sang, Q.X. Active roles of tumor stroma in breast cancer metastasis. Int. J. Breast Cancer 2012, 2012, 574025. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, F.; Lohsiriwat, V.; Petit, J.Y.; Kolonin, M.G. Adipose tissue cells, lipotransfer and cancer: A challenge for scientists, oncologists and surgeons. Biochim. Biophys. Acta 2012, 1826, 209. [Google Scholar] [CrossRef] [PubMed]
- Friedenstein, A.J.; Piatetzky, S., II; Petrakova, K.V. Osteogenesis in transplants of bone marrow cells. J. Embryol. Exp. Morphol. 1966, 16, 381. [Google Scholar] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. Int. Soc. Cell. Ther. Position Statement Cytother. 2006, 8, 315. [Google Scholar]
- Mizuno, H.; Tobita, M.; Uysal, A.C. Concise review: Adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells (Dayt. Ohio) 201, 30, 804. [Google Scholar] [CrossRef] [PubMed]
- Vellasamy, S.; Sandrasaigaran, P.; Vidyadaran, S.; Abdullah, M.; George, E.; Ramasamy, R. Mesenchymal stem cells of human placenta and umbilical cord suppress T-cell proliferation at G0 phase of cell cycle. Cell Biol. Int. 2013, 37, 250. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Huang, K.J.; Wu, J.C.; Hu, M.S.; Sanyal, M.; Hu, M.; Longaker, M.T.; Lorenz, H.P. Peripheral blood-derived mesenchymal stem cells: Candidate cells responsible for healing critical-sized calvarial bone defects. Stem Cells Transl. Med. 2015, 4, 359. [Google Scholar] [CrossRef] [PubMed]
- Ceusters, J.; Lejeune, J.P.; Sandersen, C.; Niesten, A.; Lagneaux, L.; Serteyn, D. From skeletal muscle to stem cells: An innovative and minimally-invasive process for multiple species. Sci. Rep. 2017, 7, 696. [Google Scholar] [CrossRef] [PubMed]
- Young, H.E.; Steele, T.A.; Bray, R.A.; Hudson, J.; Floyd, J.A.; Hawkins, K.; Thomas, K.; Austin, T.; Edwards, C.; Cuzzourt, J.; et al. Human reserve pluripotent mesenchymal stem cells are present in the connective tissues of skeletal muscle and dermis derived from fetal, adult, and geriatric donors. Anat. Rec. 2001, 264, 51. [Google Scholar] [CrossRef] [PubMed]
- De Bari, C.; Dell’Accio, F.; Vandenabeele, F.; Vermeesch, J.R.; Raymackers, J.M.; Luyten, F.P. Skeletal muscle repair by adult human mesenchymal stem cells from synovial membrane. J. Cell Biol. 2003, 160, 909. [Google Scholar] [CrossRef] [PubMed]
- Hass, R.; Kasper, C.; Bohm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun. Signal. CCS 2011, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Koren, S.; Bentires-Alj, M. Breast Tumor Heterogeneity: Source of Fitness, Hurdle for Therapy. Mol. Cell 2015, 60, 537. [Google Scholar] [CrossRef] [PubMed]
- Le Nail, L.R.; Brennan, M.; Rosset, P.; Deschaseaux, F.; Piloquet, P.; Pichon, O.; Le Caignec, C.; Crenn, V.; Layrolle, P.; Herault, O.; et al. Comparison of Tumor-and Bone Marrow-Derived Mesenchymal Stromal/Stem Cells from Patients with High-Grade Osteosarcoma. Int. J. Mol. Sci. 2018, 19, 707. [Google Scholar] [CrossRef] [PubMed]
- Abarrategi, A.; Tornin, J.; Martinez-Cruzado, L.; Hamilton, A.; Martinez-Campos, E.; Rodrigo, J.P.; Gonzalez, M.V.; Baldini, N.; Garcia-Castro, J.; Rodriguez, R. Osteosarcoma: Cells-of-Origin, Cancer Stem Cells, and Targeted Therapies. Stem Cells Int. 2016, 2016, 3631764. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, G.; Fox, J.; Ashton, B.; Middleton, J. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells (Dayt. Ohio) 2007, 25, 2739. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science (N. Y.) 1999, 284, 143. [Google Scholar] [CrossRef]
- Fraser, J.K.; Wulur, I.; Alfonso, Z.; Hedrick, M.H. Fat tissue: An underappreciated source of stem cells for biotechnology. Trends Biotechnol. 2006, 24, 150. [Google Scholar] [CrossRef]
- Locke, M.; Windsor, J.; Dunbar, P.R. Human adipose-derived stem cells: Isolation, characterization and applications in surgery. ANZ J. Surg. 2009, 79, 235. [Google Scholar] [CrossRef]
- Martin-Padura, I.; Gregato, G.; Marighetti, P.; Mancuso, P.; Calleri, A.; Corsini, C.; Pruneri, G.; Manzotti, M.; Lohsiriwat, V.; Rietjens, M.; et al. The white adipose tissue used in lipotransfer procedures is a rich reservoir of CD34+ progenitors able to promote cancer progression. Cancer Res. 2012, 72, 325. [Google Scholar] [CrossRef] [PubMed]
- Hass, R.; Otte, A. Mesenchymal stem cells as all-round supporters in a normal and neoplastic microenvironment. Cell Commun. Signal. CCS 2012, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Shigeura, T.; Matsumoto, D.; Sato, T.; Takaki, Y.; Aiba-Kojima, E.; Sato, K.; Inoue, K.; Nagase, T.; Koshima, I.; et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J. Cell. Physiol. 2006, 208, 64. [Google Scholar] [CrossRef] [PubMed]
- Corselli, M.; Crisan, M.; Murray, I.R.; West, C.C.; Scholes, J.; Codrea, F.; Khan, N.; Peault, B. Identification of perivascular mesenchymal stromal/stem cells by flow cytometry. Cytometry. Part A J. Int. Soc. Anal. Cytol. 2013, 83, 714. [Google Scholar] [CrossRef]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 2013, 15, 641. [Google Scholar] [PubMed]
- Salgado, A.J.; Reis, R.L.; Sousa, N.J.; Gimble, J.M. Adipose tissue derived stem cells secretome: Soluble factors and their roles in regenerative medicine. Curr. Stem Cell Res. 2010, 5, 103. [Google Scholar] [CrossRef]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004, 109, 1292. [Google Scholar] [CrossRef] [PubMed]
- Natesan, S.; Zhang, G.; Baer, D.G.; Walters, T.J.; Christy, R.J.; Suggs, L.J. A bilayer construct controls adipose-derived stem cell differentiation into endothelial cells and pericytes without growth factor stimulation. Tissue Eng. Part A 2011, 17, 941. [Google Scholar] [CrossRef] [PubMed]
- Gadelkarim, M.; Abushouk, A.I.; Ghanem, E.; Hamaad, A.M.; Saad, A.M.; Abdel-Daim, M.M. Adipose-derived stem cells: Effectiveness and advances in delivery in diabetic wound healing. Biomed. Pharm. Biomed. Pharm. 2018, 107, 625. [Google Scholar] [CrossRef]
- Rigotti, G.; Marchi, A.; Galie, M.; Baroni, G.; Benati, D.; Krampera, M.; Pasini, A.; Sbarbati, A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: A healing process mediated by adipose-derived adult stem cells. Plast. Reconstr. Surg. 2007, 119, 1409. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Zhang, L.; Hu, J.; Sun, Y. Mesenchymal stem cells: A potential targeted-delivery vehicle for anti-cancer drug, loaded nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 174. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, R.; Tsuji, W.; Gorantla, V.S.; Marra, K.G.; Rubin, J.P.; Plock, J.A. The role of adipose-derived stem cells in breast cancer progression and metastasis. Stem Cells Int. 2015, 2015, 120949. [Google Scholar] [CrossRef] [PubMed]
- Freese, K.E.; Kokai, L.; Edwards, R.P.; Philips, B.J.; Sheikh, M.A.; Kelley, J.; Comerci, J.; Marra, K.G.; Rubin, J.P.; Linkov, F. Adipose-derived stems cells and their role in human cancer development, growth, progression, and metastasis: A systematic review. Cancer Res. 2015, 75, 1161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bellows, C.F.; Kolonin, M.G. Adipose tissue-derived progenitor cells and cancer. World J. Stem Cells 2010, 2, 103. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xu, H.; Qian, C. C-Kit-Positive Adipose Tissue-Derived Mesenchymal Stem Cells Promote the Growth and Angiogenesis of Breast Cancer. BioMed Res. Int. 2017, 2017, 7407168. [Google Scholar] [CrossRef] [PubMed]
- Salha, S.; Gehmert, S.; Brebant, V.; Anker, A.; Loibl, M.; Prantl, L.; Gehmert, S. PDGF regulated migration of mesenchymal stem cells towards malignancy acts via the PI3K signaling pathway. Clin. Hemorheol. Microcirc. 2018, 70, 543. [Google Scholar] [CrossRef]
- Preisner, F.; Leimer, U.; Sandmann, S.; Zoernig, I.; Germann, G.; Koellensperger, E. Impact of Human Adipose Tissue-Derived Stem Cells on Malignant Melanoma Cells in An In Vitro Co-culture Model. Stem Cell Rev. 2018, 14, 125. [Google Scholar] [CrossRef]
- Campbell, K. Contribution of epithelial-mesenchymal transitions to organogenesis and cancer metastasis. Curr. Opin. Cell Biol. 2018, 55, 30. [Google Scholar] [CrossRef]
- Ritter, A.; Friemel, A.; Fornoff, F.; Adjan, M.; Solbach, C.; Yuan, J.; Louwen, F. Characterization of adipose-derived stem cells from subcutaneous and visceral adipose tissues and their function in breast cancer cells. Oncotarget 2015, 6, 34475. [Google Scholar] [CrossRef]
- Kucerova, L.; Skolekova, S.; Matuskova, M.; Bohac, M.; Kozovska, Z. Altered features and increased chemosensitivity of human breast cancer cells mediated by adipose tissue-derived mesenchymal stromal cells. BMC Cancer 2013, 13, 535. [Google Scholar] [CrossRef] [PubMed]
- Strong, A.L.; Ohlstein, J.F.; Biagas, B.A.; Rhodes, L.V.; Pei, D.T.; Tucker, H.A.; Llamas, C.; Bowles, A.C.; Dutreil, M.F.; Zhang, S.; et al. Leptin produced by obese adipose stromal/stem cells enhances proliferation and metastasis of estrogen receptor positive breast cancers. Breast Cancer Res. BCR 2015, 17, 112. [Google Scholar] [CrossRef] [PubMed]
- DiMeo, T.A.; Anderson, K.; Phadke, P.; Fan, C.; Perou, C.M.; Naber, S.; Kuperwasser, C. A novel lung metastasis signature links Wnt signaling with cancer cell self-renewal and epithelial-mesenchymal transition in basal-like breast cancer. Cancer Res. 2009, 69, 5364. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Wang, S.; Zhao, R.C. Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Mol. Cell. Biochem. 2013, 383, 13. [Google Scholar] [CrossRef] [PubMed]
- Visweswaran, M.; Keane, K.N.; Arfuso, F.; Dilley, R.J.; Newsholme, P.; Dharmarajan, A. The Influence of Breast Tumour-Derived Factors and Wnt Antagonism on the Transformation of Adipose-Derived Mesenchymal Stem Cells into Tumour-Associated Fibroblasts. Cancer Microenviron. Off. J. Int. Cancer Microenviron. Soc. 2018, 11, 71. [Google Scholar] [CrossRef]
- Yu, Y.; Xiao, C.H.; Tan, L.D.; Wang, Q.S.; Li, X.Q.; Feng, Y.M. Cancer-associated fibroblasts induce epithelial-mesenchymal transition of breast cancer cells through paracrine TGF-beta signalling. Br. J. Cancer 2014, 110, 724. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.H.; Warncke, C.; Choi, S.J.; Choi, S.; Chiou, A.E.; Ling, L.; Liu, H.Y.; Daniel, S.; Antonyak, M.A.; Cerione, R.A.; et al. Breast cancer-derived extracellular vesicles stimulate myofibroblast differentiation and pro-angiogenic behavior of adipose stem cells. Matrix Biol. J. Int. Soc. for Matrix Biol. 2017, 60, 190–205. [Google Scholar] [CrossRef]
- Razmkhah, M.; Mansourabadi, Z.; Mohtasebi, M.S.; Talei, A.R.; Ghaderi, A. Cancer and normal adipose-derived mesenchymal stem cells (ASCs): Do they have differential effects on tumor and immune cells? Cell Biol. Int. 2018, 42, 334. [Google Scholar] [CrossRef]
- Xishan, Z.; Bin, Z.; Haiyue, Z.; Xiaowei, D.; Jingwen, B.; Guojun, Z. Jagged-2 enhances immunomodulatory activity in adipose derived mesenchymal stem cells. Sci. Rep. 2015, 5, 14284. [Google Scholar] [CrossRef]
- Zimmerlin, L.; Park, T.S.; Zambidis, E.T.; Donnenberg, V.S.; Donnenberg, A.D. Mesenchymal stem cell secretome and regenerative therapy after cancer. Biochimie 2013, 95, 2235. [Google Scholar] [CrossRef]
- Banas, A.; Teratani, T.; Yamamoto, Y.; Tokuhara, M.; Takeshita, F.; Osaki, M.; Kawamata, M.; Kato, T.; Okochi, H.; Ochiya, T. IFATS collection: In vivo therapeutic potential of human adipose tissue mesenchymal stem cells after transplantation into mice with liver injury. Stem Cells (Dayt.Ohio) 2008, 26, 2705. [Google Scholar] [CrossRef] [PubMed]
- Anjanappa, M.; Burnett, R.; Zieger, M.A.; Merfeld-Clauss, S.; Wooden, W.; March, K.; Tholpady, S.; Nakshatri, H. Distinct Effects of Adipose-Derived Stem Cells and Adipocytes on Normal and Cancer Cell Hierarchy. Mol. Cancer Res. MCR 2016, 14, 660. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yang, Y.; Liu, Y.; Hao, Y.; Zhang, Y.; Hu, Y.; Jiang, L.; Gong, Y.; Wu, K.; Liu, Y. Upregulation of PAG1/Cbp contributes to adipose-derived mesenchymal stem cells promoted tumor progression and chemoresistance in breast cancer. Biochem. Biophys. Res. Commun. 2017, 494, 719. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Daquinag, A.; Traktuev, D.O.; Amaya-Manzanares, F.; Simmons, P.J.; March, K.L.; Pasqualini, R.; Arap, W.; Kolonin, M.G. White adipose tissue cells are recruited by experimental tumors and promote cancer progression in mouse models. Cancer Res. 2009, 69, 5259. [Google Scholar] [CrossRef] [PubMed]
- Stamatopoulos, A.; Stamatopoulos, T.; Gamie, Z.; Kenanidis, E.; Ribeiro, R.D.C.; Rankin, K.S.; Gerrand, C.; Dalgarno, K.; Tsiridis, E. Mesenchymal stromal cells for bone sarcoma treatment: Roadmap to clinical practice. J. Bone Oncol. 2019, 16, 100231. [Google Scholar] [CrossRef]
- Chulpanova, D.S.; Kitaeva, K.V.; Tazetdinova, L.G.; James, V.; Rizvanov, A.A.; Solovyeva, V.V. Application of Mesenchymal Stem Cells for Therapeutic Agent Delivery in Anti-tumor Treatment. Front. Pharmacol. 2018, 9, 259. [Google Scholar] [CrossRef]
- Chen, W.C.; Park, T.S.; Murray, I.R.; Zimmerlin, L.; Lazzari, L.; Huard, J.; Peault, B. Cellular kinetics of perivascular MSC precursors. Stem Cells Int. 2013, 2013, 983059. [Google Scholar] [CrossRef]
- Cammarota, F.; Laukkanen, M.O. Mesenchymal Stem/Stromal Cells in Stromal Evolution and Cancer Progression. Stem Cells Int. 2016, 2016, 4824573. [Google Scholar] [CrossRef]
- Papaccio, F.; Paino, F.; Regad, T.; Papaccio, G.; Desiderio, V.; Tirino, V. Concise Review: Cancer Cells, Cancer Stem Cells, and Mesenchymal Stem Cells: Influence in Cancer Development. Stem Cells Transl. Med. 2017, 6, 2115. [Google Scholar] [CrossRef]
- Klopp, A.H.; Zhang, Y.; Solley, T.; Amaya-Manzanares, F.; Marini, F.; Andreeff, M.; Debeb, B.; Woodward, W.; Schmandt, R.; Broaddus, R.; et al. Omental adipose tissue-derived stromal cells promote vascularization and growth of endometrial tumors. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 771. [Google Scholar] [CrossRef]
- Poltavets, V.; Kochetkova, M.; Pitson, S.M.; Samuel, M.S. The Role of the Extracellular Matrix and Its Molecular and Cellular Regulators in Cancer Cell Plasticity. Front. Oncol. 2018, 8, 431. [Google Scholar] [CrossRef] [PubMed]
- Rowan, B.G.; Gimble, J.M.; Sheng, M.; Anbalagan, M.; Jones, R.K.; Frazier, T.P.; Asher, M.; Lacayo, E.A.; Friedlander, P.L.; Kutner, R.; et al. Human adipose tissue-derived stromal/stem cells promote migration and early metastasis of triple negative breast cancer xenografts. PLoS ONE 2014, 9, e89595. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.H.; Shon, S.H.; Shan, M.; Stroock, A.D.; Fischbach, C. Adipose-derived stem cells increase angiogenesis through matrix metalloproteinase-dependent collagen remodeling. Integr. Biol. Quant. Biosci. Nano Macro 2016, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Tang, H.; Guo, Y.; Guo, J.; Huang, B.; Fang, F.; Cai, J.; Wang, Z. Adipose-derived mesenchymal stem cells promote cell proliferation and invasion of epithelial ovarian cancer. Exp. Cell Res. 2015, 337, 16. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Zhang, J.; Lu, X.; Wang, W.; Chen, H.; Robinson, M.K.; Cheng, J.; Tang, G.; Medeiros, L.J. Coexistent genetic alterations involving ALK, RET, ROS1 or MET in 15 cases of lung adenocarcinoma. Mod. Pathol. 2018, 31, 307. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chu, Y.; Yue, B.; Ma, X.; Zhang, G.; Xiang, H.; Liu, Y.; Wang, T.; Wu, X.; Chen, B. Adipose-derived mesenchymal stem cells promote osteosarcoma proliferation and metastasis by activating the STAT3 pathway. Oncotarget 2017, 8, 23803. [Google Scholar] [CrossRef] [PubMed]
- Strong, A.L.; Semon, J.A.; Strong, T.A.; Santoke, T.T.; Zhang, S.; McFerrin, H.E.; Gimble, J.M.; Bunnell, B.A. Obesity-associated dysregulation of calpastatin and MMP-15 in adipose-derived stromal cells results in their enhanced invasion. Stem Cells (Dayt. Ohio) 2012, 30, 2774. [Google Scholar] [CrossRef]
- Azuma, T.; Koths, K.; Flanagan, L.; Kwiatkowski, D. Gelsolin in complex with phosphatidylinositol 4,5-bisphosphate inhibits caspase-3 and -9 to retard apoptotic progression. J. Biol. Chem. 2000, 275, 3761. [Google Scholar] [CrossRef]
- Katsuda, T.; Kosaka, N.; Takeshita, F.; Ochiya, T. The therapeutic potential of mesenchymal stem cell-derived extracellular vesicles. Proteomics 2013, 13, 1637. [Google Scholar] [CrossRef]
- Thery, C.; Ostrowski, M.; Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009, 9, 581. [Google Scholar] [CrossRef]
- Baglio, S.R.; Rooijers, K.; Koppers-Lalic, D.; Verweij, F.J.; Perez Lanzon, M.; Zini, N.; Naaijkens, B.; Perut, F.; Niessen, H.W.; Baldini, N.; et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res. Ther. 2015, 6, 127. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.; Kim, S.M.; Woo, E.Y.; Han, K.C.; Park, E.J.; Ko, S.; Choi, E.W.; Jang, M. Stemness-Attenuating miR-503-3p as a Paracrine Factor to Regulate Growth of Cancer Stem Cells. Stem Cells Int. 2018, 2018, 4851949. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.F.; Yip, H.K.; Zhen, Y.Y.; Lee, C.C.; Lee, C.C.; Huang, C.C.; Ng, S.H.; Lin, J.W. Adipose-Derived Mesenchymal Stem Cell Exosomes Suppress Hepatocellular Carcinoma Growth in a Rat Model: Apparent Diffusion Coefficient, Natural Killer T-Cell Responses, and Histopathological Features. Stem Cells Int. 2015, 2015, 853506. [Google Scholar] [CrossRef] [PubMed]
- Reza, A.; Choi, Y.J.; Yasuda, H.; Kim, J.H. Human adipose mesenchymal stem cell-derived exosomal-miRNAs are critical factors for inducing anti-proliferation signalling to A2780 and SKOV-3 ovarian cancer cells. Sci. Rep. 2016, 6, 38498. [Google Scholar] [CrossRef] [PubMed]
- Takahara, T.; Kojima, T.; Miyabayashi, C.; Inoue, K.; Sasaki, H.; Muragaki, Y.; Ooshima, A. Collagen production in fat-sorting cells after carbon tetrachloride intoxication in the rat. Immunoelectron microscopic observation of type I, type III collagens, and prolyl hydroxylase. Lab. Investig. 1988, 59, 509. [Google Scholar] [PubMed]
- Iser, I.C.; Ceschini, S.M.; Onzi, G.R.; Bertoni, A.P.; Lenz, G.; Wink, M.R. Conditioned Medium from Adipose-Derived Stem Cells (ADSCs) Promotes Epithelial-to-Mesenchymal-Like Transition (EMT-Like) in Glioma Cells In vitro. Mol. Neurobiol. 2016, 53, 7184. [Google Scholar] [CrossRef] [PubMed]
- Onzi, G.R.; Ledur, P.F.; Hainzenreder, L.D.; Bertoni, A.P.; Silva, A.O.; Lenz, G.; Wink, M.R. Analysis of the safety of mesenchymal stromal cells secretome for glioblastoma treatment. Cytotherapy 2016, 18, 828. [Google Scholar] [CrossRef]
- Liu, Z.; Jiang, Z.; Huang, J.; Huang, S.; Li, Y.; Yu, S.; Yu, S.; Liu, X. miR-7 inhibits glioblastoma growth by simultaneously interfering with the PI3K/ATK and Raf/MEK/ERK pathways. Int. J. Oncol. 2014, 44, 1571. [Google Scholar] [CrossRef]
- Yang, C.; Lei, D.; Ouyang, W.; Ren, J.; Li, H.; Hu, J.; Huang, S. Conditioned media from human adipose tissue-derived mesenchymal stem cells and umbilical cord-derived mesenchymal stem cells efficiently induced the apoptosis and differentiation in human glioma cell lines in vitro. Biomed. Res. Int. 2014, 2014, 109389. [Google Scholar] [CrossRef]
- Del Fattore, A.; Luciano, R.; Saracino, R.; Battafarano, G.; Rizzo, C.; Pascucci, L.; Alessandri, G.; Pessina, A.; Perrotta, A.; Fierabracci, A.; et al. Differential effects of extracellular vesicles secreted by mesenchymal stem cells from different sources on glioblastoma cells. Expert Opin. Biol. Ther. 2015, 15, 495. [Google Scholar] [CrossRef]
- Park, Y.M.; Yoo, S.H.; Kim, S.H. Adipose-derived stem cells induced EMT-like changes in H358 lung cancer cells. Anticancer Res. 2013, 33, 4421. [Google Scholar] [PubMed]
- Cho, J.A.; Park, H.; Lim, E.H.; Lee, K.W. Exosomes from breast cancer cells can convert adipose tissue-derived mesenchymal stem cells into myofibroblast-like cells. Int. J. Oncol. 2012, 40, 130. [Google Scholar] [PubMed]
- Cho, J.A.; Park, H.; Lim, E.H.; Kim, K.H.; Choi, J.S.; Lee, J.H.; Shin, J.W.; Lee, K.W. Exosomes from ovarian cancer cells induce adipose tissue-derived mesenchymal stem cells to acquire the physical and functional characteristics of tumor-supporting myofibroblasts. Gynecol. Oncol. 2011, 123, 379. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Contreras, M.; Vera-Donoso, C.D.; Hernandez-Andreu, J.M.; Garcia-Verdugo, J.M.; Oltra, E. Therapeutic potential of human adipose-derived stem cells (ADSCs) from cancer patients: A pilot study. PLoS ONE 2014, 9, e113288. [Google Scholar] [CrossRef] [PubMed]
- Abd Elmageed, Z.Y.; Yang, Y.; Thomas, R.; Ranjan, M.; Mondal, D.; Moroz, K.; Fang, Z.; Rezk, B.M.; Moparty, K.; Sikka, S.C.; et al. Neoplastic reprogramming of patient-derived adipose stem cells by prostate cancer cell-associated exosomes. Stem Cells (Dayt. Ohio) 2014, 32, 983. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, X.; Xu, M.; Wang, J.; Zhao, R.C. Reduced adipogenesis after lung tumor exosomes priming in human mesenchymal stem cells via TGFbeta signaling pathway. Mol. Cell. Biochem. 2017, 435, 59. [Google Scholar] [CrossRef] [PubMed]
- Pessina, A.; Cocce, V.; Pascucci, L.; Bonomi, A.; Cavicchini, L.; Sisto, F.; Ferrari, M.; Ciusani, E.; Crovace, A.; Falchetti, M.L.; et al. Mesenchymal stromal cells primed with Paclitaxel attract and kill leukaemia cells, inhibit angiogenesis and improve survival of leukaemia-bearing mice. Br. J. Haematol. 2013, 160, 766. [Google Scholar] [CrossRef] [PubMed]
- Pascucci, L.; Cocce, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Vigano, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: A new approach for drug delivery. J. Control. Release Off. J. Control. Release Soc. 2014, 192, 262. [Google Scholar] [CrossRef]
- Bonomi, A.; Cocce, V.; Cavicchini, L.; Sisto, F.; Dossena, M.; Balzarini, P.; Portolani, N.; Ciusani, E.; Parati, E.; Alessandri, G.; et al. Adipose tissue-derived stromal cells primed in vitro with paclitaxel acquire anti-tumor activity. Int. J. Immunopathol. Pharm. 2013, 26, 33. [Google Scholar] [CrossRef]
- Bonomi, A.; Steimberg, N.; Benetti, A.; Berenzi, A.; Alessandri, G.; Pascucci, L.; Boniotti, J.; Cocce, V.; Sordi, V.; Pessina, A.; et al. Paclitaxel-releasing mesenchymal stromal cells inhibit the growth of multiple myeloma cells in a dynamic 3D culture system. Hematol. Oncol. 2017, 35, 693. [Google Scholar] [CrossRef]
- Cocce, V.; Balducci, L.; Falchetti, M.L.; Pascucci, L.; Ciusani, E.; Brini, A.T.; Sisto, F.; Piovani, G.; Alessandri, G.; Parati, E.; et al. Fluorescent Immortalized Human Adipose Derived Stromal Cells (hASCs-TS/GFP+) for Studying Cell Drug Delivery Mediated by Microvesicles. Anti-Cancer Agents Med. Chem. 2017, 17, 1578. [Google Scholar] [CrossRef] [PubMed]
- Petrella, F.; Cocce, V.; Masia, C.; Milani, M.; Sale, E.O.; Alessandri, G.; Parati, E.; Sisto, F.; Pentimalli, F.; Brini, A.T.; et al. Paclitaxel-releasing mesenchymal stromal cells inhibit in vitro proliferation of human mesothelioma cells. Biomed. Pharm. Biomed. Pharm. 2017, 87, 755. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. How Taxol/paclitaxel kills cancer cells. Mol. Biol. Cell 2014, 25, 2677. [Google Scholar] [CrossRef] [PubMed]
- Scioli, M.G.; Artuso, S.; D’Angelo, C.; Porru, M.; D’Amico, F.; Bielli, A.; Gentile, P.; Cervelli, V.; Leonetti, C.; Orlandi, A. Adipose-derived stem cell-mediated paclitaxel delivery inhibits breast cancer growth. PLoS ONE 2018, 13, e0203426. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, Y.; Tang, Y.; Wang, S.; Wang, C.; Li, Y.; Su, X.; Tian, J.; Tian, Y.; Pan, J.; et al. Synergistic Chemo-Photothermal Therapy of Breast Cancer by Mesenchymal Stem Cell-Encapsulated Yolk-Shell GNR@HPMO-PTX Nanospheres. ACS Appl. Mater. Interfaces 2016, 8, 17927. [Google Scholar] [CrossRef]
- Huang, W.C.; Lu, I.L.; Chiang, W.H.; Lin, Y.W.; Tsai, Y.C.; Chen, H.H.; Chang, C.W.; Chiang, C.S.; Chiu, H.C. Tumortropic adipose-derived stem cells carrying smart nanotherapeutics for targeted delivery and dual-modality therapy of orthotopic glioblastoma. J. Control. Release Off. J. Control. Release Soc. 2017, 254, 119. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Sun, S.; Dangelmajer, S.; Zhang, Q.; Wang, J.; Hu, F.; Dong, F.; Kahlert, U.D.; Zhu, M.; Lei, T. Exploiting tumor-intrinsic signals to induce mesenchymal stem cell-mediated suicide gene therapy to fight malignant glioma. Stem Cell Res. Ther. 2019, 10, 88. [Google Scholar] [CrossRef]
- Choi, S.A.; Yun, J.W.; Joo, K.M.; Lee, J.Y.; Kwak, P.A.; Lee, Y.E.; You, J.R.; Kwon, E.; Kim, W.H.; Wang, K.C.; et al. Preclinical Biosafety Evaluation of Genetically Modified Human Adipose Tissue-Derived Mesenchymal Stem Cells for Clinical Applications to Brainstem Glioma. Stem Cells Dev. 2016, 25, 897. [Google Scholar] [CrossRef]
- Li, M.; Zeng, L.; Liu, S.; Dangelmajer, S.; Kahlert, U.D.; Huang, H.; Han, Y.; Chi, X.; Zhu, M.; Lei, T. Transforming Growth Factor-beta Promotes Homing and Therapeutic Efficacy of Human Mesenchymal Stem Cells to Glioblastoma. J. Neuropathol. Exp. Neurol. 2019, 78, 315. [Google Scholar] [CrossRef]
- Tyciakova, S.; Matuskova, M.; Bohovic, R.; Polakova, K.; Toro, L.; Skolekova, S.; Kucerova, L. Genetically engineered mesenchymal stromal cells producing TNFalpha have tumour suppressing effect on human melanoma xenograft. J. Gene Med. 2015, 17, 54. [Google Scholar] [CrossRef]
- Mirzaei, H.; Salehi, H.; Oskuee, R.K.; Mohammadpour, A.; Mirzaei, H.R.; Sharifi, M.R.; Salarinia, R.; Darani, H.Y.; Mokhtari, M.; Masoudifar, A.; et al. The therapeutic potential of human adipose-derived mesenchymal stem cells producing CXCL10 in a mouse melanoma lung metastasis model. Cancer Lett. 2018, 419, 30. [Google Scholar] [CrossRef] [PubMed]
- Kucerova, L.; Altanerova, V.; Matuskova, M.; Tyciakova, S.; Altaner, C. Adipose tissue-derived human mesenchymal stem cells mediated prodrug cancer gene therapy. Cancer Res. 2007, 67, 6304. [Google Scholar] [CrossRef] [PubMed]
- Altaner, C.; Altanerova, V.; Cihova, M.; Ondicova, K.; Rychly, B.; Baciak, L.; Mravec, B. Complete regression of glioblastoma by mesenchymal stem cells mediated prodrug gene therapy simulating clinical therapeutic scenario. Int. J. Cancer 2014, 134, 1458. [Google Scholar] [CrossRef] [PubMed]
- Kucerova, L.; Skolekova, S.; Demkova, L.; Bohovic, R.; Matuskova, M. Long-term efficiency of mesenchymal stromal cell-mediated CD-MSC/5FC therapy in human melanoma xenograft model. Gene Ther. 2014, 21, 874. [Google Scholar] [CrossRef] [PubMed]
- Matuskova, M.; Hlubinova, K.; Pastorakova, A.; Hunakova, L.; Altanerova, V.; Altaner, C.; Kucerova, L. HSV-tk expressing mesenchymal stem cells exert bystander effect on human glioblastoma cells. Cancer Lett. 2010, 290, 58. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.H.; Peng, B.Y.; Chang, C.C.; Dubey, N.K.; Lo, W.C.; Cheng, H.C.; Wang, J.R.; Wei, H.J.; Deng, W.P. Tumor-Targeted Immunotherapy by Using Primary Adipose-Derived Stem Cells and an Antigen-Specific Protein Vaccine. Cancers 2018, 10, 446. [Google Scholar] [CrossRef] [PubMed]
- Mangraviti, A.; Tzeng, S.Y.; Gullotti, D.; Kozielski, K.L.; Kim, J.E.; Seng, M.; Abbadi, S.; Schiapparelli, P.; Sarabia-Estrada, R.; Vescovi, A.; et al. Non-virally engineered human adipose mesenchymal stem cells produce BMP4, target brain tumors, and extend survival. Biomaterials 2016, 100, 53. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Fitch, S.; Wang, C.; Wilson, C.; Li, J.; Grant, G.A.; Yang, F. Nanoparticle engineered TRAIL-overexpressing adipose-derived stem cells target and eradicate glioblastoma via intracranial delivery. Proc. Natl. Acad. Sci. USA 2016, 113, 13857. [Google Scholar] [CrossRef] [PubMed]
- Baranova, A.; Maltseva, D.; Tonevitsky, A. Adipose may actively delay progression of NAFLD by releasing tumor-suppressing, anti-fibrotic miR-122 into circulation. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2019, 20, 108. [Google Scholar] [CrossRef]
- Lee, H.K.; Finniss, S.; Cazacu, S.; Bucris, E.; Ziv-Av, A.; Xiang, C.; Bobbitt, K.; Rempel, S.A.; Hasselbach, L.; Mikkelsen, T.; et al. Mesenchymal stem cells deliver synthetic microRNA mimics to glioma cells and glioma stem cells and inhibit their cell migration and self-renewal. Oncotarget 2013, 4, 346. [Google Scholar] [CrossRef]
- Lou, G.; Song, X.; Yang, F.; Wu, S.; Wang, J.; Chen, Z.; Liu, Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J. Hematol. Oncol. 2015, 8, 122. [Google Scholar] [CrossRef] [PubMed]
- Karlitepe, A.; Kabadayi, H.; Vatansever, S.; Gurdal, M.; Gunduz, C.; Ercan, G. Anti-cancer efficiency of natural killer cells differentiated from human adipose tissue-derived mesenchymal stem cells and transfected with miRNA150. Exp. Oncol. 2017, 39, 212. [Google Scholar] [CrossRef]
- Lee, K.S.; Choi, J.S.; Cho, Y.W. Reprogramming of cancer stem cells into non-tumorigenic cells using stem cell exosomes for cancer therapy. Biochem. Biophys. Res. Commun. 2019, 512, 511. [Google Scholar] [CrossRef] [PubMed]
- Bateman, M.E.; Strong, A.L.; Gimble, J.M.; Bunnell, B.A. Concise Review: Using Fat to Fight Disease: A Systematic Review of Nonhomologous Adipose-Derived Stromal/Stem Cell Therapies. Stem Cells (Dayt. Ohio) 2018, 36, 1311. [Google Scholar] [CrossRef] [PubMed]
- Nicolay, N.H.; Lopez Perez, R.; Debus, J.; Huber, P.E. Mesenchymal stem cells—A new hope for radiotherapy-induced tissue damage? Cancer Lett. 2015, 366, 133. [Google Scholar] [CrossRef] [PubMed]
- Gronhoj, C.; Jensen, D.H.; Vester-Glowinski, P.; Jensen, S.B.; Bardow, A.; Oliveri, R.S.; Fog, L.M.; Specht, L.; Thomsen, C.; Darkner, S.; et al. Safety and Efficacy of Mesenchymal Stem Cells for Radiation-Induced Xerostomia: A Randomized, Placebo-Controlled Phase 1/2 Trial (MESRIX). Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 581. [Google Scholar] [CrossRef]
- Dufrane, D.; Docquier, P.L.; Delloye, C.; Poirel, H.A.; Andre, W.; Aouassar, N. Scaffold-free Three-dimensional Graft From Autologous Adipose-derived Stem Cells for Large Bone Defect Reconstruction: Clinical Proof of Concept. Medicine 2015, 94, e2220. [Google Scholar] [CrossRef]
- Veriter, S.; Andre, W.; Aouassar, N.; Poirel, H.A.; Lafosse, A.; Docquier, P.L.; Dufrane, D. Human Adipose-Derived Mesenchymal Stem Cells in Cell Therapy: Safety and Feasibility in Different “Hospital Exemption” Clinical Applications. PLoS ONE 2015, 10, e0139566. [Google Scholar] [CrossRef]
- Pennati, A.; Riggio, E.; Marano, G.; Biganzoli, E. Autologous fat grafting after sarcoma surgery: Evaluation of oncological safety. J. Plast. Reconstr. Aesthet. Surg. JPRAS 2018, 71, 1723. [Google Scholar] [CrossRef]
- Furia, S.; Cadenelli, P.; Andriani, F.; Scanagatta, P.; Duranti, L.; Spano, A.; Galeone, C.; Porcu, L.; Pastorino, U. Autologous fat tissue grafting improves pulmonary healing after laser metastasectomy. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2017, 43, 2315. [Google Scholar] [CrossRef]
- Coleman, S.R. Facial recontouring with lipostructure. Clin. Plast Surg. 1997, 24, 347. [Google Scholar] [PubMed]
- Gutowski, K.A. Current applications and safety of autologous fat grafts: A report of the ASPS fat graft task force. Plast. Reconstr. Surg. 2009, 124, 272. [Google Scholar] [CrossRef] [PubMed]
- Eaves, F.F., 3rd; Haeck, P.C.; Rohrich, R.J. ASAPS/ASPS Position statement on stem cells and fat grafting. Plast. Reconstr. Surg. 2012, 129, 285. [Google Scholar] [CrossRef] [PubMed]
- Illouz, Y.G.; Sterodimas, A. Autologous fat transplantation to the breast: A personal technique with 25 years of experience. Aesthet. Plast. Surg. 2009, 33, 706. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.W.; McCulley, S.J.; Macmillan, R.D. Autologous fat transfer—A review of the literature with a focus on breast cancer surgery. J. Plast. Reconstr. Aesthet. Surg. JPRAS 2008, 61, 1438. [Google Scholar] [CrossRef] [PubMed]
- Rigotti, G.; Marchi, A.; Stringhini, P.; Baroni, G.; Galie, M.; Molino, A.M.; Mercanti, A.; Micciolo, R.; Sbarbati, A. Determining the oncological risk of autologous lipoaspirate grafting for post-mastectomy breast reconstruction. Aesthet. Plast. Surg. 2010, 34, 475. [Google Scholar] [CrossRef]
- Petit, J.Y.; Botteri, E.; Lohsiriwat, V.; Rietjens, M.; De Lorenzi, F.; Garusi, C.; Rossetto, F.; Martella, S.; Manconi, A.; Bertolini, F.; et al. Locoregional recurrence risk after lipofilling in breast cancer patients. Ann. Oncol. Off. J. Eur. Soc. Med Oncol. 2012, 23, 582. [Google Scholar] [CrossRef]
- Perez-Cano, R.; Vranckx, J.J.; Lasso, J.M.; Calabrese, C.; Merck, B.; Milstein, A.M.; Sassoon, E.; Delay, E.; Weiler-Mithoff, E.M. Prospective trial of adipose-derived regenerative cell (ADRC)-enriched fat grafting for partial mastectomy defects: The RESTORE-2 trial. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2012, 38, 382. [Google Scholar] [CrossRef]
- Petit, J.Y.; Rietjens, M.; Botteri, E.; Rotmensz, N.; Bertolini, F.; Curigliano, G.; Rey, P.; Garusi, C.; De Lorenzi, F.; Martella, S.; et al. Evaluation of fat grafting safety in patients with intraepithelial neoplasia: A matched-cohort study. Ann. Oncol. Off. J. Eur. Soc. Med Oncol. 2013, 24, 1479. [Google Scholar] [CrossRef]
- Gale, K.L.; Rakha, E.A.; Ball, G.; Tan, V.K.; McCulley, S.J.; Macmillan, R.D. A case-controlled study of the oncologic safety of fat grafting. Plast. Reconstr. Surg. 2015, 135, 1263. [Google Scholar] [CrossRef]
- Kronowitz, S.J.; Mandujano, C.C.; Liu, J.; Kuerer, H.M.; Smith, B.; Garvey, P.; Jagsi, R.; Hsu, L.; Hanson, S.; Valero, V. Lipofilling of the Breast Does Not Increase the Risk of Recurrence of Breast Cancer: A Matched Controlled Study. Plast. Reconstr. Surg. 2016, 137, 385. [Google Scholar] [CrossRef] [PubMed]
- Myckatyn, T.M.; Wagner, I.J.; Mehrara, B.J.; Crosby, M.A.; Park, J.E.; Qaqish, B.F.; Moore, D.T.; Busch, E.L.; Silva, A.K.; Kaur, S.; et al. Cancer Risk after Fat Transfer: A Multicenter Case-Cohort Study. Plast. Reconstr. Surg. 2017, 139, 11. [Google Scholar] [CrossRef] [PubMed]
- Cohen, O.; Lam, G.; Karp, N.; Choi, M. Determining the Oncologic Safety of Autologous Fat Grafting as a Reconstructive Modality: An Institutional Review of Breast Cancer Recurrence Rates and Surgical Outcomes. Plast. Reconstr. Surg. 2017, 140, 382e. [Google Scholar] [CrossRef] [PubMed]
- Fertsch, S.; Hagouan, M.; Munder, B.; Schulz, T.; Abu-Ghazaleh, A.; Schaberick, J.; Stambera, P.; Aldeeri, M.; Andree, C.; Thamm, O.C. Increased risk of recurrence associated with certain risk factors in breast cancer patients after DIEP-flap reconstruction and lipofilling-a matched cohort study with 200 patients. Gland Surg. 2017, 6, 315. [Google Scholar] [CrossRef] [PubMed]
- Gennari, R.; Griguolo, G.; Dieci, M.V.; Guarneri, V.; Tavaniello, B.; Sibilio, A.; Conte, P. Fat grafting for breast cancer patients: From basic science to clinical studies. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2016, 42, 1088. [Google Scholar] [CrossRef] [PubMed]
- Agha, R.A.; Fowler, A.J.; Herlin, C.; Goodacre, T.E.; Orgill, D.P. Use of autologous fat grafting for breast reconstruction: A systematic review with meta-analysis of oncological outcomes. J. Plast. Reconstr. Aesthet. Surg. JPRAS 2015, 68, 143. [Google Scholar] [CrossRef] [PubMed]
- Wazir, U.; El Hage Chehade, H.; Headon, H.; Oteifa, M.; Kasem, A.; Mokbel, K. Oncological Safety of Lipofilling in Patients with Breast Cancer: A Meta-analysis and Update on Clinical Practice. Anticancer Res. 2016, 36, 4521. [Google Scholar] [CrossRef]
- Groen, J.W.; Negenborn, V.L.; Twisk, D.; Rizopoulos, D.; Ket, J.C.F.; Smit, J.M.; Mullender, M.G. Autologous fat grafting in onco-plastic breast reconstruction: A systematic review on oncological and radiological safety, complications, volume retention and patient/surgeon satisfaction. J. Plast. Reconstr. Aesthet. Surg. JPRAS 2016, 69, 742. [Google Scholar] [CrossRef]
- Charvet, H.J.; Orbay, H.; Wong, M.S.; Sahar, D.E. The Oncologic Safety of Breast Fat Grafting and Contradictions Between Basic Science and Clinical Studies: A Systematic Review of the Recent Literature. Ann. Plast. Surg. 2015, 75, 471. [Google Scholar] [CrossRef]
- Waked, K.; Colle, J.; Doornaert, M.; Cocquyt, V.; Blondeel, P. Systematic review: The oncological safety of adipose fat transfer after breast cancer surgery. Breast (Edinb. Scotl.) 2017, 31, 128. [Google Scholar] [CrossRef]
| Category | Type | Mechanism of Action | Target | References |
|---|---|---|---|---|
| Drug loading | PTX | anti-mitotic activity | human leukaemia MOLT-4 cells human osteosarcoma SK-ES-I cells human prostatic carcinoma DU145 cells human neuroblastoma GI-LI-N and SH-SY5Y human breast cancer CG5 cells | [89] [89] [89] [89] [94] |
| Drug-loaded NPs | PTX | anti-mitotic activity + high frequency magnetic field | murine brain astrocytoma ALTS1C1 | [96] |
| Gene delivery by viral vectors | TRAIL overexpression | suicide inductor | primary patient-derived glioblastoma human glioma U-87MG primary patient-derived glioblastoma | [97] [98] [99] |
| TNF-α overexpression | apoptotic activity | human breast cancer SKBR3 cells human melanoma A375 cells | [110] | |
| IP-10 overexpression | antitumor activity | murine metastatic melanoma | [101] | |
| cytosine deaminase | cytotoxicity to 5-fluorouracil | human colon cancer HT-29 cells rattus brain glioma C6 cells human melanoma A375 cells | [102] [103] [104] | |
| thymidine kinase | cytotoxicity to ganciclovir | human glioblastoma cells 8-MG-BA, 42-MG-BA and U-118 MG | [105] | |
| modified E6/E7 antigen | immunomodulatory activity | murine lung carcinoma LLC1 cells murine colon carcinoma CT26 cells | [106] | |
| Gene delivery by non-viral vectors | BMP4 plasmid-loaded NPs | antitumor activity | primary patient-derived glioma | [107] |
| TRAIL plasmid-loaded NPs | suicide inductor | primary patient-derived glioblastoma | [108] | |
| Micro-RNA transfection | miR-124 e miR-145 | reducing cell migration and self-renewal | primary patient-derived glioma and glioma stem cells | [110] |
| miR-122 | apoptotic activity | human hepatocellular carcinoma HepG2 cells | [111] | |
| miR-150 | immunomodulatory activity | human pancreatic cancer cells PANC1 | [112] |
| Clinical study | Year | Design of the Study | Control | N° Patients | Median Follow-Up (Months) | Results with fat Grafting | Reference |
|---|---|---|---|---|---|---|---|
| Osteosarcoma and soft tissue sarcomas | 2015 | Retrospective case series | no | 17 | 32.5 | LR and DM not reported | [118] |
| 2018 | Retrospective case series | no | 60 | 28.6 | not increased recurrence risk (LR and DM) | [119] | |
| Radiotherapy induced xerosthomia | 2018 | Randomized placebo controlled phase I/II | yes | 30 | 4 | LR and DM not reported | [116] |
| Pulmonary metastasis | 2017 | Prospective cohort study | yes | 40 | 61 | LR and DM not reported | [120] |
| Breast cancer | 2010 | Retrospective cohort | no | 137 | 91 | not increased LR, DM not reported | [126] |
| 2012 | Retrospective matched cohort | yes | 321 | 26 | not increased recurrence risk, higher risk of LR for in-situ cancer subgroup | [127] | |
| 2012 | Prospective single-arm trial | no | 67 | 12 | no LR, DM not reported | [128] | |
| 2013 | Retrospective matched cohort | yes | 59 | 42 | increased LR for in-situ cancer patients, DM not reported | [129] | |
| 2015 | Retrospective matched cohort | yes | 211 | 32 | not increased recurrence risk (LR and DM) | [130] | |
| 2016 | Retrospective matched cohort | yes | 719 | 60 | not increased recurrence risk, higher risk of LR for HT subgroup | [131] | |
| 2017 | Retrospective case-control | yes | 225 | / | cases are cancer recurrence (2006-2011), not increased recurrence risk | [132] | |
| 2017 | Retrospective cohort | yes | 248 | 45.6 | not increased recurrence risk (LR and DM) | [133] | |
| 2017 | Retrospective matched cohort | yes | 100 | 31 | increased LR for positive nodal status and high-grade neoplasia, DM not reported, similar disease-free survival and overall recurrence | [134] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scioli, M.G.; Storti, G.; D’Amico, F.; Gentile, P.; Kim, B.-S.; Cervelli, V.; Orlandi, A. Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities. Int. J. Mol. Sci. 2019, 20, 3296. https://doi.org/10.3390/ijms20133296
Scioli MG, Storti G, D’Amico F, Gentile P, Kim B-S, Cervelli V, Orlandi A. Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities. International Journal of Molecular Sciences. 2019; 20(13):3296. https://doi.org/10.3390/ijms20133296
Chicago/Turabian StyleScioli, Maria Giovanna, Gabriele Storti, Federico D’Amico, Pietro Gentile, Bong-Sung Kim, Valerio Cervelli, and Augusto Orlandi. 2019. "Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities" International Journal of Molecular Sciences 20, no. 13: 3296. https://doi.org/10.3390/ijms20133296
APA StyleScioli, M. G., Storti, G., D’Amico, F., Gentile, P., Kim, B.-S., Cervelli, V., & Orlandi, A. (2019). Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities. International Journal of Molecular Sciences, 20(13), 3296. https://doi.org/10.3390/ijms20133296