Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism
Abstract
1. Introduction
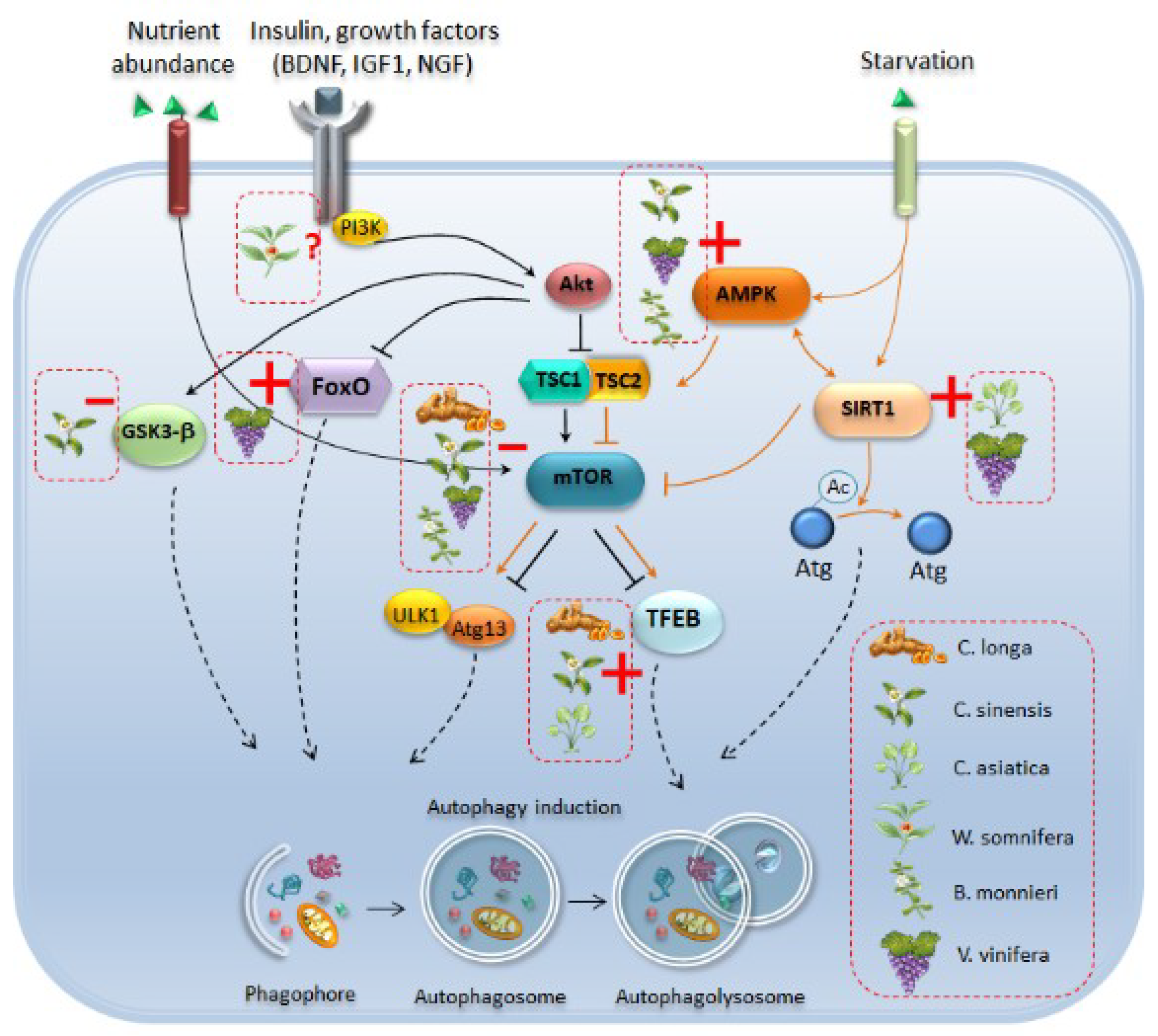
2. Eukaryotic Cell Clearing Pathways: A Focus on Autophagy
3. Autophagy Failure in Parkinson’s Disease Patients and Experimental Models
4. Phytochemicals: Autophagy-Based Effects and Related Potential for Alpha-Synuclein Clearance in Experimental Parkinsonism
4.1. Introduction to Phytochemicals and Rough Classification
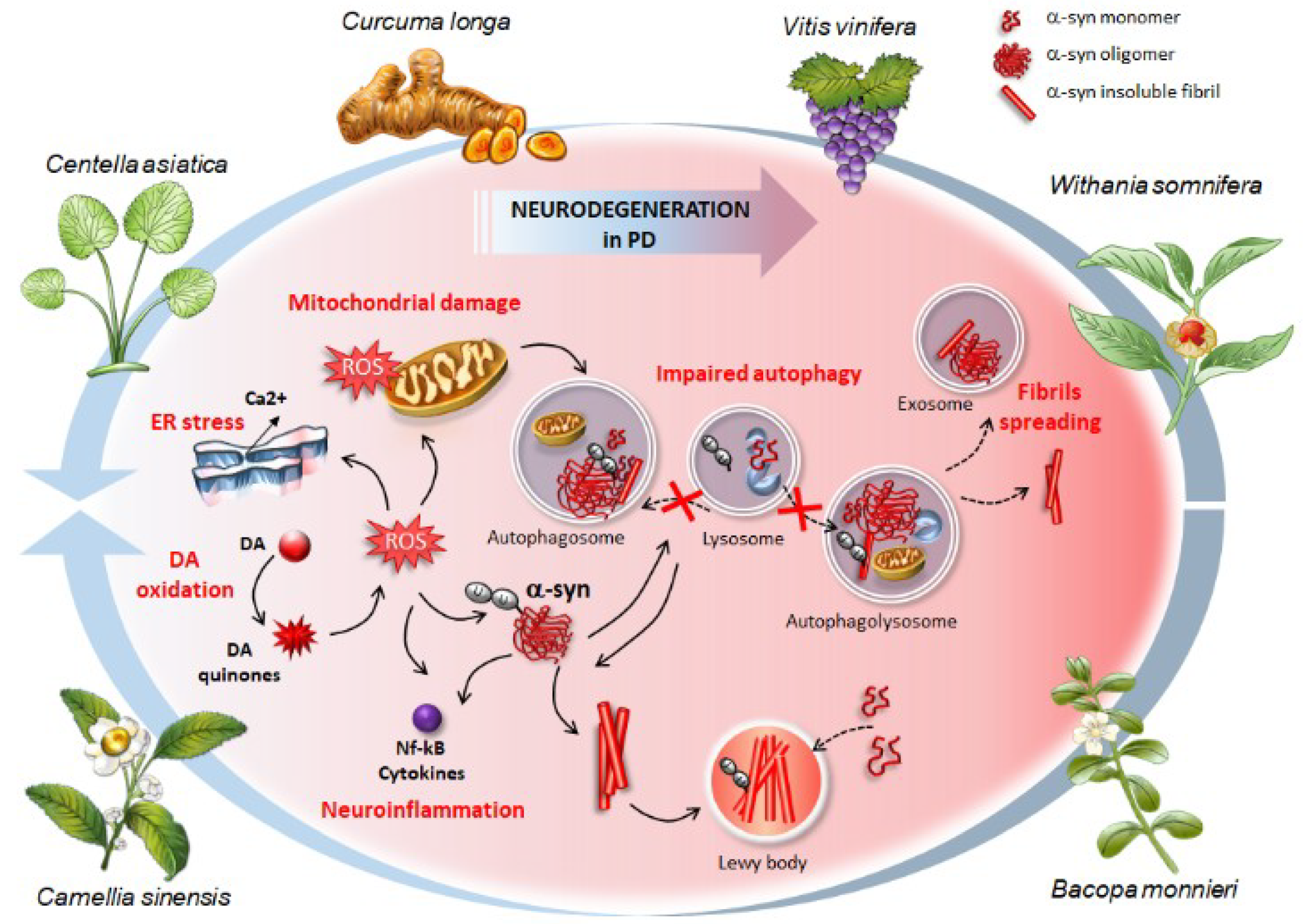
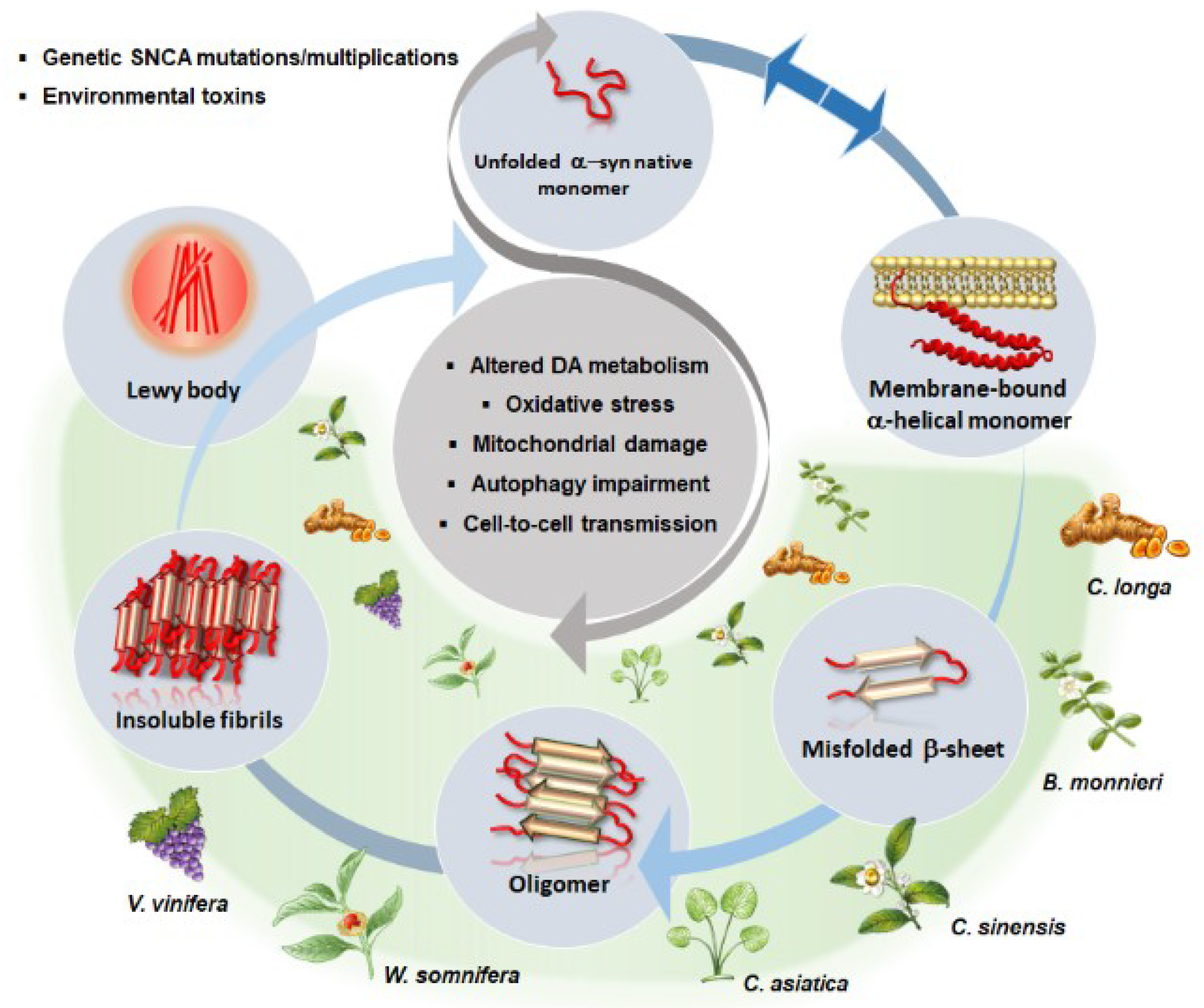
4.2. Autophagy and Alpha-Synuclein Clearance as Common Effects Induced by Phytochemicals
4.2.1. Curcumin from Curcuma longa
4.2.2. Bacosides and Bacopasides from Bacopa monnieri
4.2.3. Green Tea Catechins from Camellia sinensis
4.2.4. Gallic Acids, Asiatic Acids, and Madecassosides from Centella asiatica
4.2.5. Withanolides and Withaferin from Withania somnifera, ashwagandha
4.2.6. Resveratrol from Vitis vinifera
5. Conclusions and Future Directions
Funding
Conflicts of Interest
Abbreviations
| 3-MA | 3-Methyladenine |
| 6-OHDA | 6-Hydroxydopamine |
| AD | Alzheimer’s Disease |
| ALS | Amyotrophic Lateral Sclerosis |
| AMPK | 5′ AMP-activated Protein Kinase |
| Atg | Autophagy-Related-Gene |
| BAD | Bcl-2-Associated Death Promoter |
| BDNF | Brain-Derived Neurotrophic Factor |
| CAT | Catalase |
| CNS | Central Nervous System |
| COX-2 | Cyclooxygenase-2 |
| Cyt | Cytochrome |
| DA | Dopamine |
| DAT | Dopamine Transporter |
| DLB | Dementia with Lewy Bodies |
| EGCG | Epigallocatechin Gallate |
| ER | Endoplasmic Reticulum |
| ERK | Extracellular Signal–Regulated Kinase |
| FOXO3 | Forkhead Box O3 |
| GDNF | Glial Cell Line-Derived Neurotrophic Factor |
| GFAP | Glial Fibrillary Acidic Protein |
| GFP | Green Fluorescent Protein |
| Gpx | Glutathione Peroxidase |
| GR | Glutathione Reductase |
| GSH | Glutathione |
| GSk3-β | Glycogen Synthase Kinase 3 Beta |
| GST | Glutathione S-Transferase |
| HDAC6 | Histone Deacetylase 6 |
| HIF-1 | Hypoxia-Inducible Factor 1 |
| HMGB1 | High Mobility Group Box 1 |
| IFNγ | Interferon Gamma |
| IIS | Insulin/Insulin-Like Growth Factor Signaling |
| IL-1β | Interleukine 1 Beta |
| IL-1β/a | Interleukine-1 beta/alpha |
| iNOS | inducible Nitric Oxide Synthase |
| JNK | c-Jun N-Terminal Kinase |
| LAMP-2A | Lysosomal-Associated Membrane Protein Type 2a |
| LPS | Lipopolysaccharide |
| LRRK2 | Leucine-Rich Repeat Kinase 2 |
| LSD | Lysosomal Storage Diseases |
| MALAT1 | Metastasis-Associated Lung Adenocarcinoma Transcript 1 |
| MAPK | Mitogen-Activated Protein Kinase |
| MDA | Malondialdehyde |
| Meth | Methamphetamine |
| MMP | Mitochondrial Membrane Potential |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| MSA | Multisystem Atrophy |
| mTOR | Mammalian Target of Rapamycin |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate Hydrogen |
| Nf-Kb | Nuclear Factor K Beta |
| NGF | Neurotrophic Growth Factor |
| NO | Nitric Oxide |
| Nrf2 | Nuclear Factor Erythroid 2-Related Factor 2 |
| PAF | Pure Autonomic Failure |
| PARP | Poly (ADP-ribose) Polymerase |
| PD | Parkinson’s Disease |
| PGC-1α | Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha |
| PINK1 | PTEN-induced kinase 1 |
| PKC α | Protein Kinase C alpha |
| Rab GTPase | Gtp Bound Ras Proteins in Brain |
| ROS | Reactive Oxygen Species |
| SDH | Succinate Dehydrogenase |
| SIRT1 | NAD-dependent deacetylase Sirtuin-1 |
| SNARE | Soluble Nsf Attachment Protein Receptor |
| SNpc | Substantia Nigra Pars Compacta |
| SOD | Superoxide Dismutase |
| SQSTM1 | Sequestosome-1 |
| SVZ | Subventricular Zone |
| TBARS | Thiobarbituric Acid Reactive Substance |
| TFEB | Transcription Factor EB |
| TGF-b1 | Transforming Growth Factor Beta 1 |
| TH | Tyrosine Hydroxylase |
| TNFα | Tumor Necrosis Factor Alpha |
| Trk A/B | Tyrosine Receptor Kinase A/B |
| UCH-LI | Ubiquitin carboxy-terminal hydrolase L1 |
| VEGF | Vascular-Endothelial Growth Factor |
| VTA | Ventral Tegmental Area |
| YFP | Yellow Fluorescent Protein |
| α-syn | alpha-synuclein |
References
- Aronson, J.K. Defining ‘nutraceuticals’: Neither nutritious nor pharmaceutical. Br. J. Clin. Pharmacol. 2017, 1, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Andrew, R.; Izzo, A.A. Principles of pharmacological research of nutraceuticals. Br. J. Pharmacol. 2017, 11, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Campos-Vega, R.; Oomah, B.D. Chemistry and classification of phytochemicals. In Handbook of Plant Food Phytochemicals; Tiwari, B., Brunton, N.P., Brennan, C.S., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “Curecumin”: From kitchen to clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, A.A.; Farooqui, T.; Madan, A.; Ong, J.H.; Ong, W.Y. Ayurvedic Medicine for the Treatment of Dementia: Mechanistic Aspects. Evid. Based Complement. Alternat. Med. 2018, 2018, 2481076. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, C.; Fornai, M.; Antonioli, L.; Blandizzi, C.; Calderone, V. Phytochemicals as Novel Therapeutic Strategies for NLRP3 Inflammasome-Related Neurological, Metabolic, and Inflammatory Diseases. Int. J. Mol. Sci. 2019, 20, 2876. [Google Scholar] [CrossRef] [PubMed]
- Rajasekar, J.; Perumal, M.K.; Vallikannan, B. A critical review on anti-angiogenic property of phytochemicals. J. Nutr. Biochem. 2019, 71, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Banach, M. Botanicals and phytochemicals active on cognitive decline: The clinical evidence. Pharmacol. Res. 2018, 130, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Brondino, N.; Re, S.; Boldrini, A.; Cuccomarino, A.; Lanati, N.; Barale, F.; Politi, P. Curcumin as a therapeutic agent in dementia: A mini systematic review of human studies. Sci. World J. 2014, 2014, 174282. [Google Scholar] [CrossRef]
- Kongkeaw, C.; Dilokthornsakul, P.; Thanarangsarit, P.; Limpeanchob, N.; Norman Scholfield, C. Meta-analysis of randomized controlled trials on cognitive effects of Bacopa monnieri extract. J. Ethnopharmacol. 2014, 151, 528–535. [Google Scholar] [CrossRef]
- Pase, M.P.; Kean, J.; Sarris, J.; Neale, C.; Scholey, A.B.; Stough, C. The cognitive-enhancing effects of Bacopa monnieri: A systematic review of randomized, controlled human clinical trials. J. Altern. Complement. Med. 2012, 18, 647–652. [Google Scholar] [CrossRef]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. Alzheimer’s Disease Cooperative Study. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, D.; Bhattacharyya, S.; Bose, S. Efficacy and Safety of Ashwagandha (Withania somnifera (L.) Dunal) Root Extract in Improving Memory and Cognitive Functions. J. Diet Suppl. 2017, 14, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Cascella, M.; Bimonte, S.; Muzio, M.R.; Schiavone, V.; Cuomo, A. The efficacy of Epigallocatechin-3-gallate (green tea) in the treatment of Alzheimer’s disease: An overview of pre-clinical studies and translational perspectives in clinical practice. Infect. Agent Cancer 2017, 12, 36. [Google Scholar] [CrossRef] [PubMed]
- Chico, L.; Ienco, E.C.; Bisordi, C.; Lo Gerfo, A.; Petrozzi, L.; Petrucci, A.; Mancuso, M.; Siciliano, G. Amyotrophic Lateral Sclerosis and Oxidative Stress: A Double-Blind Therapeutic Trial After Curcumin Supplementation. CNS Neurol. Disord. Drug Targets 2018, 17, 767–779. [Google Scholar] [CrossRef]
- Singh, M.; Arseneault, M.; Sanderson, T.; Murthy, V.; Ramassamy, C. Challenges for research on polyphenols from foods in Alzheimer’s disease: Bioavailability, metabolism, and cellular and molecular mechanisms. J. Agric. Food Chem. 2008, 56, 4855–4873. [Google Scholar] [CrossRef] [PubMed]
- Andrade, S.; Ramalho, M.J.; Pereira, M.D.C.; Loureiro, J.A. Resveratrol Brain Delivery for Neurological Disorders Prevention and Treatment. Front. Pharmacol. 2018, 9, 1261. [Google Scholar] [CrossRef]
- Ji, H.F.; Shen, L. Can improving bioavailability improve the bioactivity of curcumin? Trends Pharmacol. Sci. 2014, 35, 265–266. [Google Scholar] [CrossRef]
- Garcia-Alloza, M.; Borrelli, L.A.; Rozkalne, A.; Hyman, B.T.; Bacskai, B.J. Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J. Neurochem. 2007, 102, 1095–1104. [Google Scholar] [CrossRef]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar] [CrossRef]
- Rajan, K.E.; Preethi, J.; Singh, H.K. Molecular and Functional Characterization of Bacopa monniera: A Retrospective Review. Evid. Based Complement. Alternat. Med. 2015, 2015, 945217. [Google Scholar] [CrossRef] [PubMed]
- De, K.; Chandra, S.; Misra, M. Evaluation of the biological effect of brahmi (Bacopa monnieri Linn) extract on the biodistribution of technetium-99m radiopharmaceuticals. Life Sci. J. 2008, 5, 45–49. [Google Scholar]
- Suganuma, M.; Okabe, S.; Oniyama, M.; Tada, Y.; Ito, H.; Fujiki, H. Wide distribution of (-)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998, 19, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Anukunwithaya, T.; Tantisira, M.H.; Tantisira, B.; Khemawoot, P. Pharmacokinetics of a Standardized Extract of Centella asiatica ECa 233 in Rats. Planta Med. 2017, 83, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Vareed, S.K.; Bauer, A.K.; Nair, K.M.; Liu, Y.; Jayaprakasam, B.; Nair, M.G. Blood-brain barrier permeability of bioactive withanamides present in Withania somnifera fruit extract. Phytother. Res. 2014, 28, 1260–1264. [Google Scholar] [CrossRef]
- Rege, S.D.; Geetha, T.; Griffin, G.D.; Broderick, T.L.; Babu, J.R. Neuroprotective effects of resveratrol in Alzheimer disease pathology. Front. Aging Neurosci. 2014, 6, 218. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Yan, F.; Liang, X.; Wu, M.; Shen, Y.; Chen, M.; Xu, Y.; Zou, G.; Jiang, P.; Tang, C.; et al. Localized delivery of curcumin into brain with polysorbate 80-modified cerasomes by ultrasound-targeted microbubble destruction for improved Parkinson’s disease therapy. Theranostics 2018, 8, 2264–2277. [Google Scholar] [CrossRef]
- Jinfeng, L.; Yunliang, W.; Xinshan, L.; Yutong, W.; Shanshan, W.; Peng, X.; Xiaopeng, Y.; Zhixiu, X.; Qingshan, L.; Honglei, Y.; et al. Therapeutic Effects of CUR-Activated Human Umbilical Cord Mesenchymal Stem Cells on 1-Methyl-4-phenylpyridine-Induced Parkinson’s Disease Cell Model. Biomed. Res. Int. 2016, 2016, 9140541. [Google Scholar] [CrossRef]
- Wu, Y.; Liang, S.; Xu, B.; Zhang, R.; Xu, L. Protective effect of curcumin on dopamine neurons in Parkinson’s disease and its mechanism. J. Zhejiang Univ. Med. Sci. 2018, 47, 480–486. [Google Scholar]
- Yu, S.; Zheng, W.; Xin, N.; Chi, Z.H.; Wang, N.Q.; Nie, Y.X.; Feng, W.Y.; Wang, Z.Y. Curcumin prevents dopaminergic neuronal death through inhibition of the c-Jun N-terminal kinase pathway. Rejuvenation Res. 2010, 13, 55–64. [Google Scholar] [CrossRef]
- Pan, J.; Li, H.; Ma, J.F.; Tan, Y.Y.; Xiao, Q.; Ding, J.Q.; Chen, S.D. Curcumin inhibition of JNKs prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease through suppressing mitochondria dysfunction. Transl. Neurodegener. 2012, 1, 16. [Google Scholar] [CrossRef] [PubMed]
- He, X.J.; Uchida, K.; Megumi, C.; Tsuge, N.; Nakayama, H. Dietary curcumin supplementation attenuates 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxicity in C57BL mice. J. Toxicol. Pathol. 2015, 28, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.J.; Lian, Y.G.; Zhao, H.Y.; Xu, Q.L. Curcumin protects from oxidative stress and inhibits α-synuclein aggregation in MPTP induced parkinsonian mice. Int. J. Clin. Exp. Med. 2016, 9, 2654–2665. [Google Scholar]
- Wang, Y.L.; Ju, B.; Zhang, Y.Z.; Yin, H.L.; Liu, Y.J.; Wang, S.S.; Zeng, Z.L.; Yang, X.P.; Wang, H.T.; Li, J.F. Protective Effect of Curcumin Against Oxidative Stress-Induced Injury in Rats with Parkinson’s Disease Through the Wnt/β-Catenin Signaling Pathway. Cell. Physiol. Biochem. 2017, 43, 2226–2241. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Nie, Q.; Li, Z.; Du, G. Curcumin improves neurofunctions of 6-OHDA-induced parkinsonian rats. Pathol. Res. Pract. 2016, 212, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Du, X.X.; Jiang, H.; Xie, J.X. Curcumin attenuates 6-hydroxydopamine-induced cytotoxicity by anti-oxidation and nuclear factor-kappa B modulation in MES23.5 cells. Biochem. Pharmacol. 2009, 78, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Jaisin, Y.; Thampithak, A.; Meesarapee, B.; Ratanachamnong, P.; Suksamrarn, A.; Phivthong-Ngam, L.; Phumala-Morales, N.; Chongthammakun, S.; Govitrapong, P.; Sanvarinda, Y. Curcumin I protects the dopaminergic cell lines SH-SY5Y from 6-hydroxydopamine-induced neurotoxicity through attenuation of p53-mediated apoptosis. Neurosci. Lett. 2011, 489, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Meesarapee, B.; Thampithak, A.; Jaisin, Y.; Sanvarinda, P.; Suksamrarn, A.; Tuchinda, P.; Morales, N.P.; Sanvarinda, Y. Curcumin I mediates neuroprotective effect through attenuation of quinoprotein formation, p-p38 MAPK expression, and caspase-3 activation in 6-hydroxydopamine treated SH-SY5Y cells. Phytother. Res. 2014, 28, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.K.; Park, H.Y.; Go, J.; Kim, Y.H.; Hwang, J.H.; Choi, D.H.; Noh, J.R.; Rhee, M.; Han, P.L.; Lee, C.H.; et al. Effects of histone acetyltransferase inhibitors on L-DOPA-induced dyskinesia in a murine model of Parkinson’s disease. J. Neural Transm. 2018, 125, 1319–1331. [Google Scholar] [CrossRef]
- Ramkumar, M.; Rajasankar, S.; Gobi, V.V.; Dhanalakshmi, C.; Manivasagam, T.; Justin Thenmozhi, A.; Essa, M.M.; Kalandar, A.; Chidambaram, R. Neuroprotective effect of Demethoxycurcumin, a natural derivative of Curcumin on rotenone induced neurotoxicity in SH-SY 5Y Neuroblastoma cells. BMC Complement. Altern. Med. 2017, 17, 217. [Google Scholar] [CrossRef]
- Cui, Q.; Li, X.; Zhu, H. Curcumin ameliorates dopaminergic neuronal oxidative damage via activation of the Akt/Nrf2 pathway. Mol. Med. Rep. 2016, 13, 1381–1388. [Google Scholar] [CrossRef] [PubMed]
- Khatri, D.K.; Juvekar, A.R. Neuroprotective effect of curcumin as evinced by abrogation of rotenone-induced motor deficits, oxidative and mitochondrial dysfunctions in mouse model of Parkinson’s disease. Pharmacol. Biochem. Behav. 2016, 150–151, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Abbaoui, A.; Chatoui, H.; El Hiba, O.; Gamrani, H. Neuroprotective effect of curcumin-I in copper-induced dopaminergic neurotoxicity in rats: A possible link with Parkinson’s disease. Neurosci. Lett. 2017, 66, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Abbaoui, A.; Gamrani, H. Neuronal, astroglial and locomotor injuries in subchronic copper intoxicated rats are repaired by curcumin: A possible link with Parkinson’s disease. Acta Histochem. 2018, 120, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Nehru, B. Curcumin affords neuroprotection and inhibits α-synuclein aggregation in lipopolysaccharide-induced Parkinson’s disease model. Inflammopharmacology 2018, 26, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.S.; Boddapati, S.; Emadi, S.; Sierks, M.R. Curcumin reduces alpha-synuclein induced cytotoxicity in Parkinson’s disease cell model. BMC Neurosci. 2010, 11, 57. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.F.; Zhang, Y.J.; Zhou, H.Y.; Wang, H.M.; Tian, L.P.; Liu, J.; Ding, J.Q.; Chen, S.D. Curcumin ameliorates the neurodegenerative pathology in A53T α-synuclein cell model of Parkinson’s disease through the downregulation of mTOR/p70S6K signaling and the recovery of macroautophagy. J. Neuroimmune Pharmacol. 2013, 8, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yu, Y.; Li, X.; Ross, C.A.; Smith, W.W. Curcumin protects against A53Talpha-synuclein-induced toxicity in a PC12 inducible cell model for Parkinsonism. Pharmacol. Res. 2011, 63, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Siddique, Y.H.; Naz, F.; Jyoti, S. Effect of curcumin on lifespan, activity pattern, oxidative stress, and apoptosis in the brains of transgenic Drosophila model of Parkinson’s disease. BioMed Res. Int. 2014, 2014, 606928. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Vuu, M.D.; Huynh, M.A.; Yamaguchi, M.; Tran, L.T.; Dang, T.P.T. Curcumin Effectively Rescued Parkinson’s Disease-Like Phenotypes in a Novel Drosophila melanogaster Model with dUCH Knockdown. Oxid. Med. Cell. Longev. 2018, 2018, 2038267. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.; Terpstra, K.J.; Bureau, Y.; Hou, J.; Raheb, H.; Cernvosky, Z.; Badmeav, V.; Copen, J.; Husni, M.; Woodbury-Farina, M. Liposomal-formulated curcumin [Lipocurc™] targeting HDAC (histone deacetylase) prevents apoptosis and improves motor deficits in park 7 (DJ-1)-knockout rat model of Parkinson’s disease: Implications for epigenetics-based nanotechnology-driven drug platform. J. Complement. Integr. Med. 2013, 10, 75–88. [Google Scholar] [CrossRef] [PubMed]
- van der Merwe, C.; van Dyk, H.C.; Engelbrecht, L.; van der Westhuizen, F.H.; Kinnear, C.; Loos, B.; Bardien, S. Curcumin Rescues a PINK1 Knock Down SH-SY5Y Cellular Model of Parkinson’s Disease from Mitochondrial Dysfunction and Cell Death. Mol. Neurobiol. 2017, 54, 2752–2762. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Li, T.; Liu, Z.; Arbez, N.; Yan, J.; Moran, T.H.; Ross, C.A.; Smith, W.W. LRRK2 kinase activity mediates toxic interactions between genetic mutation and oxidative stress in a Drosophila model: Suppression by curcumin. Neurobiol. Dis. 2012, 47, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Murthy, V.; Ramassamy, C. Standardized extracts of Bacopa monniera protect against MPP+- and paraquat-induced toxicity by modulating mitochondrial activities, proteasomal functions, and redox pathways. Toxicol. Sci. 2012, 125, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Pandey, S.; Verma, R.; Ansari, J.A.; Mahdi, A.A. Comparative evaluation of extract of Bacopa monnieri and Mucuna pruriens as neuroprotectant in MPTP model of Parkinson’s disease. Indian J. Exp. Biol. 2016, 54, 758–766. [Google Scholar]
- Singh, B.; Pandey, S.; Yadav, S.K.; Verma, R.; Singh, S.P.; Mahdi, A.A. Role of ethanolic extract of Bacopa monnieri against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) induced mice model via inhibition of apoptotic pathways of dopaminergic neurons. Brain Res. Bull. 2017, 135, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Nellore, J.; Pauline, C.; Amarnath, K. Bacopa monnieri Phytochemicals Mediated Synthesis of Platinum Nanoparticles and Its Neurorescue Effect on 1-Methyl 4-Phenyl 1,2,3,6 Tetrahydropyridine-Induced Experimental Parkinsonism in Zebrafish. J. Neurodegener. Dis. 2013, 2013, 972391. [Google Scholar] [CrossRef]
- Hosamani, R.; Muralidhara. Prophylactic treatment with Bacopa monnieri leaf powder mitigates paraquat-induced oxidative perturbations and lethality in Drosophila melanogaster. Indian J. Biochem. Biophys. 2010, 47, 75–82. [Google Scholar]
- Singh, M.; Murthy, V.; Ramassamy, C. Neuroprotective mechanisms of the standardized extract of Bacopa monniera in a paraquat/diquat-mediated acute toxicity. Neurochem. Int. 2013, 62, 530–539. [Google Scholar] [CrossRef]
- Hosamani, R.; Krishna, G.; Muralidhara. Standardized Bacopa monnieri extract ameliorates acute paraquat-induced oxidative stress, and neurotoxicity in prepubertal mice brain. Nutr. Neurosci. 2016, 19, 434–446. [Google Scholar] [CrossRef]
- Krishna, G.; Hosamani, R.; Muralidhara. Bacopa monnieri Supplements Offset Paraquat-Induced Behavioral Phenotype and Brain Oxidative Pathways in Mice. Cent. Nerv. Syst. Agents Med. Chem. 2019, 19, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Srivastav, S.; Fatima, M.; Mondal, A.C. Bacopa monnieri alleviates paraquat induced toxicity in Drosophila by inhibiting jnk mediated apoptosis through improved mitochondrial function and redox stabilization. Neurochem. Int. 2018, 121, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Shobana, C.; Kumar, R.R.; Sumathi, T. Alcoholic extract of Bacopa monniera Linn. protects against 6-hydroxydopamine-induced changes in behavioral and biochemical aspects: A pilot study. Cell. Mol. Neurobiol. 2012, 32, 1099–1112. [Google Scholar] [PubMed]
- Jadiya, P.; Khan, A.; Sammi, S.R.; Kaur, S.; Mir, S.S.; Nazir, A. Anti-Parkinsonian effects of Bacopa monnieri: Insights from transgenic and pharmacological Caenorhabditis elegans models of Parkinson’s disease. Biochem. Biophys. Res. Commun. 2011, 413, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Shinomol, G.K.; Mythri, R.B.; Srinivas Bharath, M.M.; Muralidhara. Bacopa monnieri extract offsets rotenone-induced cytotoxicity in dopaminergic cells and oxidative impairments in mice brain. Cell. Mol. Neurobiol. 2012, 32, 455–465. [Google Scholar] [CrossRef]
- Hosamani, R.; Muralidhara. Neuroprotective efficacy of Bacopa monnieri against rotenone induced oxidative stress and neurotoxicity in Drosophila melanogaster. Neurotoxicology 2009, 30, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Jansen, R.L.; Brogan, B.; Whitworth, A.J.; Okello, E.J. Effects of five Ayurvedic herbs on locomotor behaviour in a Drosophila melanogaster Parkinson’s disease model. Phytother. Res. 2014, 28, 1789–1795. [Google Scholar] [CrossRef]
- Ye, Q.; Ye, L.; Xu, X.; Huang, B.; Zhang, X.; Zhu, Y.; Chen, X. Epigallocatechin-3-gallate suppresses 1-methyl-4-phenyl-pyridine-induced oxidative stress in PC12 cells via the SIRT1/PGC-1α signaling pathway. BMC Complement. Altern. Med. 2012, 12, 82. [Google Scholar] [CrossRef]
- Levites, Y.; Weinreb, O.; Maor, G.; Youdim, M.B.H.; Mandel, S. Green tea polyphenol (-)-epigallocatechin-3gallate prevents N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced dopaminergic neurodegeneration. J. Neurochem. 2001, 78, 1073–1082. [Google Scholar] [CrossRef]
- Mandel, S.; Maor, G.; Youdim, M.B. Iron and alpha-synuclein in the substantia nigra of MPTP-treated mice: Effect of neuroprotective drugs R-apomorphine and green tea polyphenol (-)-epigallocatechin-3-gallate. J. Mol. Neurosci. 2004, 24, 401–416. [Google Scholar] [CrossRef]
- Xu, Q.; Langley, M.; Kanthasamy, A.G.; Reddy, M.B. Epigallocatechin Gallate Has a Neurorescue Effect in a Mouse Model of Parkinson Disease. J. Nutr. 2017, 147, 1926–1931. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Zhu, M.; Liang, Z. (-)-Epigallocatechin-3-gallate modulates peripheral immunity in the MPTP-induced mouse model of Parkinson’s disease. Mol. Med. Rep. 2018, 17, 4883–4888. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Perez, D.A.; Jimenez-Del-Rio, M.; Velez-Pardo, C. Epigallocatechin-3-Gallate Protects and Prevents Paraquat-Induced Oxidative Stress and Neurodegeneration in Knockdown dj-1-β Drosophila melanogaster. Neurotox. Res. 2018, 34, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Bonilla-Ramirez, L.; Jimenez-Del-Rio, M.; Velez-Pardo, C. Low doses of paraquat and polyphenols prolong life span and locomotor activity in knock-down parkin Drosophila melanogaster exposed to oxidative stress stimuli: Implication in autosomal recessive juvenile parkinsonism. Gene 2013, 512, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Levites, Y.; Youdim, M.B.; Maor, G.; Mandel, S. Attenuation of 6-hydroxydopamine (6-OHDA)-induced nuclear factor-kappaB (NF-kappaB) activation and cell death by tea extracts in neuronal cultures. Biochem. Pharmacol. 2002, 63, 21–29. [Google Scholar] [CrossRef]
- Bitu Pinto, N.; da Silva Alexandre, B.; Neves, K.R.; Silva, A.H.; Leal, L.K.; Viana, G.S. Neuroprotective Properties of the Standardized Extract from Camellia sinensis (Green Tea) and Its Main Bioactive Components, Epicatechin and Epigallocatechin Gallate, in the 6-OHDA Model of Parkinson’s Disease. Evid. Based Complement. Alternat. Med. 2015, 2015, 161092. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Chen, L.; Hu, X.; Cao, S.; Yang, J. Effects and mechanism of epigallocatechin-3-gallate on apoptosis and mTOR/AKT/GSK-3β pathway in substantia nigra neurons in Parkinson rats. Neuroreport 2019, 30, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Kamalden, T.A.; Ji, D.; Osborne, N.N. Rotenone-induced death of RGC-5 cells is caspase independent, involves the JNK and p38 pathways and is attenuated by specific green tea flavonoids. Neurochem. Res. 2012, 37, 1091–1101. [Google Scholar] [CrossRef]
- Ng, C.H.; Guan, M.S.; Koh, C.; Ouyang, X.; Yu, F.; Tan, E.K.; O’Neill, S.P.; Zhang, X.; Chung, J.; Lim, K.L. AMP kinase activation mitigates dopaminergic dysfunction and mitochondrial abnormalities in Drosophila models of Parkinson’s disease. J. Neurosci. 2012, 32, 14311–14317. [Google Scholar] [CrossRef]
- Siddique, Y.H.; Jyoti, S.; Naz, F. Effect of epicatechin gallate dietary supplementation on transgenic Drosophila model of Parkinson’s disease. J. Diet Suppl. 2014, 11, 121–130. [Google Scholar] [CrossRef]
- Nataraj, J.; Manivasagam, T.; Justin Thenmozhi, A.; Essa, M.M. Neurotrophic Effect of Asiatic acid, a Triterpene of Centella asiatica Against Chronic 1-Methyl 4-Phenyl 1, 2, 3, 6-Tetrahydropyridine Hydrochloride/Probenecid Mouse Model of Parkinson’s disease: The Role of MAPK, PI3K-Akt-GSK3β and mTOR Signalling Pathways. Neurochem. Res. 2017, 42, 1354–1365. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, M.; Goel, I.; Roy, T.; Shukla, S.D.; Khurana, S. Complete Comparison Display (CCD) evaluation of ethanol extracts of Centella asiatica and Withania somnifera shows that they can non-synergistically ameliorate biochemical and behaviouraldamages in MPTP induced Parkinson’s model of mice. PLoS ONE 2017, 12, e0177254. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.L.; Qu, R.; Zhang, J.; Li, L.F.; Ma, S.P. Neuroprotective effects of madecassoside in early stage of Parkinson’s disease induced by MPTP in rats. Fitoterapia 2013, 90, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Haleagrahara, N.; Ponnusamy, K. Neuroprotective effect of Centella asiatica extract (CAE) on experimentally induced parkinsonism in aged Sprague-Dawley rats. J. Toxicol. Sci. 2010, 35, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Nataraj, J.; Manivasagam, T.; Justin Thenmozhi, A.; Essa, M.M. Neuroprotective effect of asiatic acid on rotenone-induced mitochondrial dysfunction and oxidative stress-mediated apoptosis in differentiated SH-SYS5Y cells. Nutr. Neurosci. 2017, 20, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Teerapattarakan, N.; Benya-Aphikul, H.; Tansawat, R.; Wanakhachornkrai, O.; Tantisira, M.H.; Rodsiri, R. Neuroprotective effect of a standardized extract of Centella asiatica ECa233 in rotenone-induced parkinsonism rats. Phytomedicine 2018, 44, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Siddique, Y.H.; Naz, F.; Jyoti, S.; Fatima, A.; Khanam, S.; Rahul Ali, F.; Mujtaba, S.F.; Faisal, M. Effect of Centella asiatica Leaf Extract on the Dietary Supplementation in Transgenic Drosophila Model of Parkinson’s Disease. Parkinson’s Dis. 2014, 2014, 262058. [Google Scholar] [CrossRef]
- Sankar, S.R.; Manivasagam, T.; Krishnamurti, A.; Ramanathan, M. The neuroprotective effect of Withania somnifera root extract in MPTP-intoxicated mice: An analysis of behavioral and biochemical variables. Cell. Mol. Biol. Lett. 2007, 12, 473–481. [Google Scholar] [CrossRef]
- Rajasankar, S.; Manivasagam, T.; Sankar, V.; Prakash, S.; Muthusamy, R.; Krishnamurti, A.; Surendran, S. Withania somnifera root extract improves catecholamines and physiological abnormalities seen in a Parkinson’s disease model mouse. J. Ethnopharmacol. 2009, 125, 369–373. [Google Scholar] [CrossRef]
- Rajasankar, S.; Manivasagam, T.; Surendran, S. Ashwagandha leaf extract: A potential agent in treating oxidative damage and physiological abnormalities seen in a mouse model of Parkinson’s disease. Neurosci. Lett. 2009, 454, 11–15. [Google Scholar] [CrossRef]
- Prakash, J.; Chouhan, S.; Yadav, S.K.; Westfall, S.; Rai, S.N.; Singh, S.P. Withania somnifera alleviates parkinsonian phenotypes by inhibiting apoptotic pathways in dopaminergic neurons. Neurochem. Res. 2014, 39, 2527–2536. [Google Scholar] [CrossRef] [PubMed]
- Prakash, J.; Yadav, S.K.; Chouhan, S.; Singh, S.P. Neuroprotective role of Withania somnifera root extract in maneb-paraquat induced mouse model of parkinsonism. Neurochem. Res. 2013, 38, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Saleem, S.; Ahmad, A.S.; Ansari, M.A.; Yousuf, S.; Hoda, M.N.; Islam, F. Neuroprotective effects of Withania somnifera on 6-hydroxydopamine induced Parkinsonism in rats. Hum. Exp. Toxicol. 2005, 24, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Manjunath, M.J.; Muralidhara. Standardized extract of Withania somnifera (Ashwagandha) markedly offsets rotenone-induced locomotor deficits, oxidative impairments and neurotoxicity in Drosophila melanogaster. J. Food Sci. Technol. 2015, 52, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- De Rose, F.; Marotta, R.; Poddighe, S.; Talani, G.; Catelani, T.; Setzu, M.D.; Solla, P.; Marrosu, F.; Sanna, E.; Kasture, S.; et al. Functional and Morphological Correlates in the Drosophila LRRK2 loss-of-function Model of Parkinson’s Disease: Drug Effects of Withania somnifera (Dunal) Administration. PLoS ONE 2016, 11, e0146140. [Google Scholar] [CrossRef] [PubMed]
- Xia, D.; Sui, R.; Zhang, Z. Administration of resveratrol improved Parkinson’s disease-like phenotype by suppressing apoptosis of neurons via modulating the MALAT1/miR-129/SNCA signaling pathway. J. Cell. Biochem. 2019, 120, 4942–4951. [Google Scholar] [CrossRef]
- Blanchet, J.; Longpré, F.; Bureau, G.; Morissette, M.; DiPaolo, T.; Bronchti, G.; Martinoli, M.G. Resveratrol, a red wine polyphenol, protects dopaminergic neurons in MPTP-treated mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 1243–1250. [Google Scholar] [CrossRef]
- Liu, Q.; Zhu, D.; Jiang, P.; Tang, X.; Lang, Q.; Yu, Q.; Zhang, S.; Che, Y.; Feng, X. Resveratrol synergizes with low doses of L-DOPA to improve MPTP-induced Parkinson disease in mice. Behav. Brain Res. 2019, 367, 10–18. [Google Scholar] [CrossRef]
- Guo, Y.J.; Dong, S.Y.; Cui, X.X.; Feng, Y.; Liu, T.; Yin, M.; Kuo, S.H.; Tan, E.K.; Zhao, W.J.; Wu, Y.C. Resveratrol alleviates MPTP-induced motor impairments and pathological changes by autophagic degradation of α-synuclein via SIRT1-deacetylated LC3. Mol. Nutr. Food Res. 2016, 60, 2161–2175. [Google Scholar] [CrossRef]
- Abolaji, A.O.; Adedara, A.O.; Adie, M.A.; Vicente-Crespo, M.; Farombi, E.O. Resveratrol prolongs lifespan and improves 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced oxidative damage and behavioural deficits in Drosophila melanogaster. Biochem. Biophys. Res. Commun. 2018, 503, 1042–1048. [Google Scholar] [CrossRef]
- Albani, D.; Polito, L.; Batelli, S.; De Mauro, S.; Fracasso, C.; Martelli, G.; Colombo, L.; Manzoni, C.; Salmona, M.; Caccia, S.; et al. The SIRT1 activator resveratrol protects SK-N-BE cells from oxidative stress and against toxicity caused by α-synuclein or amyloid-beta (1-42) peptide. J. Neurochem. 2009, 110, 1445–1456. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Wu, Q.; Lu, Y.F.; Gong, Q.H.; Shi, J.S. Neuroprotective effect of resveratrol on 6-OHDA-induced Parkinson’s disease in rats. Eur. J. Pharmacol. 2008, 600, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.L.; Lin, K.J.; Wang, P.W.; Chuang, J.H.; Lin, H.Y.; Chen, S.D.; Chuang, Y.C.; Huang, S.T.; Tiao, M.M.; Chen, J.B.; et al. Resveratrol provides neuroprotective effects through modulation of mitochondrial dynamics and ERK1/2 regulated autophagy. Free Radic. Res. 2018, 52, 1371–1386. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.K.; Chen, S.D.; Chuang, Y.C.; Lin, H.Y.; Huang, C.R.; Chuang, J.H.; Wang, P.W.; Huang, S.T.; Tiao, M.M.; Chen, J.B.; et al. Resveratrol partially prevents rotenone-induced neurotoxicity in dopaminergic SH-SY5Y cells through induction of heme oxygenase-1 dependent autophagy. Int. J. Mol. Sci. 2014, 15, 1625–1646. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Dong, X.; Liu, Z.; Zhu, S.; Liu, H.; Fan, W.; Hu, Y.; Hu, T.; Yu, Y.; Li, Y.; et al. Resveratrol Suppresses Rotenone-induced Neurotoxicity Through Activation of SIRT1/Akt1 Signaling Pathway. Anat. Rec. 2018, 301, 1115–1125. [Google Scholar] [CrossRef]
- Wu, Y.; Li, X.; Zhu, J.X.; Xie, W.; Le, W.; Fan, Z.; Jankovic, J.; Pan, T. Resveratrol-activated AMPK/SIRT1/autophagy in cellular models of Parkinson’s disease. Neurosignals 2011, 19, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Palle, S.; Neerati, P. Improved neuroprotective effect of resveratrol nanoparticles as evinced by abrogation of rotenone-induced behavioral deficits and oxidative and mitochondrial dysfunctions in rat model of Parkinson’s disease. Naunyn. Schmiedebergs Arch. Pharmacol. 2018, 391, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Gaballah, H.H.; Zakaria, S.S.; Elbatsh, M.M.; Tahoon, N.M. Modulatory effects of resveratrol on endoplasmic reticulum stress-associated apoptosis and oxido-inflammatory markers in a rat model of rotenone-induced Parkinson’s disease. Chem. Biol. Interact. 2016, 251, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.F.; Yu, X.L.; Ji, M.; Liu, S.Y.; Wu, X.L.; Wang, Y.J.; Liu, R.T. Resveratrol alleviates motor and cognitive deficits and neuropathology in the A53T α-synuclein mouse model of Parkinson’s disease. Food Funct. 2018, 9, 6414–6426. [Google Scholar] [CrossRef]
- Wu, Z.; Wu, A.; Dong, J.; Sigears, A.; Lu, B. Grape skin extract improves muscle function and extends lifespan of a Drosophila model of Parkinson’s disease through activation of mitophagy. Exp. Gerontol. 2018, 113, 10–17. [Google Scholar] [CrossRef]
- Abrahams, S.; Haylett, W.L.; Johnson, G.; Carr, J.A.; Bardien, S. Antioxidant effects of curcumin in models of neurodegeneration, aging, oxidative and nitrosative stress: A review. Neuroscience 2019, 406, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.S.; Zhang, Z.R.; Zhang, M.M.; Sun, M.X.; Wang, W.W.; Xie, C.L. Neuroprotective properties of curcumin in toxin-base animal models of Parkinson’s disease: A systematic experiment literatures review. BMC Complement. Altern. Med. 2017, 17, 412. [Google Scholar] [CrossRef] [PubMed]
- Srivastav, S.; Fatima, M.; Mondal, A.C. Important medicinal herbs in Parkinson’s disease pharmacotherapy. Biomed. Pharmacother. 2017, 92, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, B.K.; Rathinasamy, B.; Lohanathan, B.P.; Thiyagarajan, V.; Weng, C.F. Neuroprotective Role of Phytochemicals. Molecules 2018, 23, 2485. [Google Scholar] [CrossRef] [PubMed]
- Valero, T. Mitochondrial biogenesis: Pharmacological approaches. Curr. Pharm. Des. 2014, 20, 5507–5509. [Google Scholar] [CrossRef] [PubMed]
- Mathur, D.; Goyal, K.; Koul, V.; Anand, A. The Molecular Links of Re-Emerging Therapy: A Review of Evidence of Brahmi (Bacopa monniera). Front. Pharmacol. 2016, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Yadav, R.S. Efficacy of Natural Compounds in Neurodegenerative Disorders. Adv. Neurobiol. 2016, 12, 107–123. [Google Scholar] [CrossRef]
- Aguiar, S.; Borowski, T. Neuropharmacological review of the nootropic herb Bacopa monnieri. Rejuvenation Res. 2013, 16, 313–326. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Bahramsoltani, R.; Abbasabadi, Z.; Braidy, N.; Nabavi, S.M. Role of green tea catechins in prevention of age-related cognitive decline: Pharmacological targets and clinical perspective. J. Cell. Physiol. 2019, 234, 2447–2459. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Ohishi, T.; Tanabe, H.; Miyoshi, N.; Nakamura, Y. Beneficial Effects of Green Tea Catechins on Neurodegenerative Diseases. Molecules 2018, 23, 1297. [Google Scholar] [CrossRef]
- Gray, N.E.; Harris, C.J.; Quinn, J.F.; Soumyanath, A. Centella asiatica modulates antioxidant and mitochondrial pathways and improves cognitive function in mice. J. Ethnopharmacol. 2016, 180, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E. Centella asiatica (L.) Urban: From Traditional Medicine to Modern Medicine with Neuroprotective Potential. Evid. Based Complement. Alternat. Med. 2012, 2012, 946259. [Google Scholar] [CrossRef] [PubMed]
- Ur Rasheed, M.S.; Tripathi, M.K.; Mishra, A.K.; Shukla, S.; Singh, M.P. Resveratrol Protects from Toxin-Induced Parkinsonism: Plethora of Proofs Hitherto Petty Translational Value. Mol. Neurobiol. 2016, 53, 2751–2760. [Google Scholar] [CrossRef] [PubMed]
- Lynch-Day, M.A.; Mao, K.; Wang, K.; Zhao, M.; Klionsky, D.J. The role of autophagy in Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a009357. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, S.; Ferese, R.; Biagioni, F.; Busceti, C.L.; Campopiano, R.; Griguoli, A.M.P.; Limanaqi, F.; Novelli, G.; Storto, M.; Fornai, F. The Monoamine Brainstem Reticular Formation as a Paradigm for Re-Defining Various Phenotypes of Parkinson’s Disease Owing Genetic and Anatomical Specificity. Front. Cell. Neurosci. 2017, 11, 102. [Google Scholar] [CrossRef]
- Ferrucci, M.; Pasquali, L.; Ruggieri, S.; Paparelli, A.; Fornai, F. Alpha-synuclein and autophagy as common steps in neurodegeneration. Parkinsonism Relat. Disord. 2008, 14 (Suppl. 2), S180–S184. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, L.; Ruggieri, S.; Murri, L.; Paparelli, A.; Fornai, F. Does autophagy worsen or improve the survival of dopaminergic neurons? Parkinsonism Relat. Disord. 2009, 15 (Suppl. 4), S24–S27. [Google Scholar] [CrossRef]
- Limanaqi, F.; Biagioni, F.; Gambardella, S.; Ryskalin, L.; Fornai, F. Interdependency Between Autophagy and Synaptic Vesicle Trafficking: Implications for Dopamine Release. Front. Mol. Neurosci. 2018, 11, 299. [Google Scholar] [CrossRef] [PubMed]
- Obergasteiger, J.; Frapporti, G.; Pramstaller, P.P.; Hicks, A.A.; Volta, M. A new hypothesis for Parkinson’s disease pathogenesis: GTPase-p38 MAPK signaling and autophagy as convergence points of etiology and genomics. Mol. Neurodegener. 2018, 13, 40. [Google Scholar] [CrossRef]
- Ryskalin, L.; Busceti, C.L.; Limanaqi, F.; Biagioni, F.; Gambardella, S.; Fornai, F. A Focus on the Beneficial Effects of Alpha Synuclein and a Re-Appraisal of Synucleinopathies. Curr. Protein Pept. Sci. 2018, 19, 598–611. [Google Scholar] [CrossRef]
- Rubinsztein, D.C.; Bento, C.F.; Deretic, V. Therapeutic targeting of autophagy in neurodegenerative and infectious diseases. J. Exp. Med. 2015, 212, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Giordano, S.; Darley-Usmar, V.; Zhang, J. Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biol. 2013, 2, 82–90. [Google Scholar] [CrossRef]
- Giorgi, F.S.; Bandettini di Poggio, A.; Battaglia, G.; Pellegrini, A.; Murri, L.; Ruggieri, S.; Paparelli, A.; Fornai, F. A short overview on the role of alpha-synuclein and proteasome in experimental models of Parkinson’s disease. J. Neural. Transm. Suppl. 2006, 70, 105–109. [Google Scholar]
- Limanaqi, F.; Biagioni, F.; Gaglione, A.; Busceti, C.L.; Fornai, F. A Sentinel in the Crosstalk Between the Nervous and Immune System: The (Immuno)-Proteasome. Front. Immunol. 2019, 10, 628. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Uchihara, T.; Fukuda, T.; Noda, S.; Kondo, H.; Saiki, S.; Komatsu, M.; Uchiyama, Y.; Tanaka, K.; Hattori, N. Loss of autophagy in dopaminergic neurons causes Lewy pathology and motor dysfunction in aged mice. Sci. Rep. 2018, 8, 2813. [Google Scholar] [CrossRef] [PubMed]
- Limanaqi, F.; Biagioni, F.; Busceti, C.L.; Ryskalin, L.; Soldani, P.; Frati, A.; Fornai, F. Cell Clearing Systems Bridging Neuro-Immunity and Synaptic Plasticity. Int. J. Mol. Sci. 2019, 20, 2197. [Google Scholar] [CrossRef]
- Ryskalin, L.; Limanaqi, F.; Biagioni, F.; Frati, A.; Esposito, V.; Calierno, M.T.; Lenzi, P.; Fornai, F. The emerging role of m-TOR up-regulation in brain Astrocytoma. Histol. Histopathol. 2017, 32, 413–431. [Google Scholar] [CrossRef] [PubMed]
- Ryskalin, L.; Limanaqi, F.; Frati, A.; Busceti, C.L.; Fornai, F. mTOR-Related Brain Dysfunctions in Neuropsychiatric Disorders. Int. J. Mol. Sci. 2018, 19, 2226. [Google Scholar] [CrossRef]
- Ferrucci, M.; Biagioni, F.; Ryskalin, L.; Limanaqi, F.; Gambardella, S.; Frati, A.; Fornai, F. Ambiguous Effects of Autophagy Activation Following Hypoperfusion/Ischemia. Int. J. Mol. Sci. 2018, 19, 2756. [Google Scholar] [CrossRef]
- Limanaqi, F.; Biagioni, F.; Busceti, C.L.; Ryskalin, L.; Fornai, F. The effects of proteasome on baseline and methamphetamine-dependent dopamine transmission. Neurosci. Biobehav. Rev. 2019, 102, 308–317. [Google Scholar] [CrossRef]
- Lenzi, P.; Lazzeri, G.; Biagioni, F.; Busceti, C.L.; Gambardella, S.; Salvetti, A.; Fornai, F. The Autophagoproteasome a Novel Cell Clearing Organelle in Baseline and Stimulated Conditions. Front. Neuroanat. 2016, 10, 78. [Google Scholar] [CrossRef] [PubMed]
- Lazzeri, G.; Biagioni, F.; Fulceri, F.; Busceti, C.L.; Scavuzzo, M.C.; Ippolito, C.; Salvetti, A.; Lenzi, P.; Fornai, F. mTOR Modulates Methamphetamine-Induced Toxicity through Cell Clearing Systems. Oxid. Med. Cell. Longev. 2018, 2018, 6124745. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K. Organellophagy: Eliminating cellular building blocks via selective autophagy. J. Cell. Biol. 2014, 205, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Tooze, S.A.; Schiavo, G. Liaisons dangereuses: Autophagy, neuronal survival and neurodegeneration. Curr. Opin. Neurobiol. 2008, 18, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N. Autophagy in protein and organelle turnover. In Cold Spring Harbor Symposia on Quantitative Biology; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2011. [Google Scholar]
- Xie, Z.; Klionsky, D.J. Autophagosome formation: Core machinery and adaptations. Nat. Cell. Biol. 2007, 9, 1102–1109. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Chen, Y.; Tooze, S.A. Autophagy pathway: Cellular and molecular mechanisms. Autophagy 2017, 14, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Kamada, Y.; Yoshino, K.; Kondo, C.; Kawamata, T.; Oshiro, N.; Yonezawa, K.; Ohsumi, Y. Tor directly controls the Atg1 kinase complex to regulate autophagy. Mol. Cell. Biol. 2010, 30, 1049–1058. [Google Scholar] [CrossRef]
- Pasquali, L.; Busceti, C.L.; Fulceri, F.; Paparelli, A.; Fornai, F. Intracellular pathways underlying the effects of lithium. Behav. Pharmacol. 2010, 21, 473–492. [Google Scholar] [CrossRef]
- Zhou, J.; Tan, S.H.; Nicolas, V.; Bauvy, C.; Yang, N.D.; Zhang, J.; Xue, Y.; Codogno, P.; Shen, H.M. Activation of lysosomal function in the course of autophagy via mTORC1 suppression and autophagosome-lysosome fusion. Cell Res. 2013, 23, 508–523. [Google Scholar] [CrossRef]
- Pietrocola, F.; Mariño, G.; Lissa, D.; Vacchelli, E.; Malik, S.A.; Niso-Santano, M.; Zamzami, N.; Galluzzi, L.; Maiuri, M.C.; Kroemer, G. Pro-autophagic polyphenols reduce the acetylation of cytoplasmic proteins. Cell Cycle 2012, 11, 3851–3860. [Google Scholar] [CrossRef]
- Janda, E.; Lascala, A.; Carresi, C.; Parafati, M.; Aprigliano, S.; Russo, V.; Savoia, C.; Ziviani, E.; Musolino, V.; Morani, F.; et al. Parkinsonian toxin-induced oxidative stress inhibits basal autophagy in astrocytes via NQO2/quinone oxidoreductase 2: Implications for neuroprotection. Autophagy 2015, 11, 1063–1080. [Google Scholar] [CrossRef] [PubMed]
- Ao, X.; Zou, L.; Wu, Y. Regulation of autophagy by the Rab GTPase network. Cell Death Differ. 2014, 21, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Filomeni, G.; De Zio, D.; Cecconi, F. Oxidative stress and autophagy: The clash between damage and metabolic needs. Cell Death Differ. 2015, 22, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Dokladny, K.; Myers, O.B.; Moseley, P.L. Heat shock response and autophagy—Cooperation and control. Autophagy 2015, 11, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Tsapras, P.; Nezis, I.P. Caspase involvement in autophagy. Cell Death Differ. 2017, 24, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Song, J.X.; Lu, J.H.; Liu, L.F.; Chen, L.L.; Durairajan, S.S.; Yue, Z.; Zhang, H.Q.; Li, M. HMGB1 is involved in autophagy inhibition caused by SNCA/alpha-synuclein overexpression: A process modulated by the natural autophagy inducer corynoxine B. Autophagy 2014, 10, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, A.M.; Stefanis, L.; Fredenburg, R.; Lansbury, P.T.; Sulzer, D. Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science 2004, 305, 1292–1295. [Google Scholar] [CrossRef]
- Martinez-Vicente, M.; Talloczy, Z.; Kaushik, S.; Massey, A.C.; Mazzulli, J.; Mosharov, E.V.; Hodara, R.; Fredenburg, R.; Wu, D.C.; Follenzi, A.; et al. Dopamine-modified alpha-synuclein blocks chaperone-mediated autophagy. J. Clin. Investig. 2008, 118, 777–788. [Google Scholar] [CrossRef]
- Anglade, P.; Vyas, S.; Javoy-Agid, F.; Herrero, M.T.; Michel, P.P.; Marquez, J.; Mouatt-Prigent, A.; Ruberg, M.; Hirsch, E.C.; Agid, Y. Apoptosis and autophagy in nigral neurons of patients with Parkinson’s disease. Histol. Histopathol. 1997, 12, 25–31. [Google Scholar]
- Mayer, R.J.; Lowe, J.; Lennox, G.; Doherty, F.; Landon, M. Intermediate filaments and ubiquitin: A new thread in the understanding of chronic neurodegenerative diseases. Prog. Clin. Biol. Res. 1989, 317, 809–818. [Google Scholar]
- Mayer, R.J.; Lowe, J.; Lennox, G.; Landon, M.; MacLennan, K.; Doherty, F.J. Intermediate filament-ubiquitin diseases: Implications for cell sanitization. Biochem. Soc. Symp. 1989, 55, 193–201. [Google Scholar] [PubMed]
- Zhu, J.H.; Guo, F.; Shelburne, J.; Watkins, S.; Chu, C.T. Localization of phosphorylated ERK/MAP kinases to mitochondria and autophagosomes in Lewy body diseases. Brain Pathol. 2003, 13, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Dodiya, H.; Aebischer, P.; Olanow, C.W.; Kordower, J.H. Alterations in lysosomal and proteasomal markers in Parkinson’s disease: Relationship to alpha-synuclein inclusions. Neurobiol. Dis. 2009, 35, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Dehay, B.; Bove, J.; Rodriguez-Muela, N.; Perier, C.; Recasens, A.; Boya, P.; Vila, M. Pathogenic lysosomal depletion in Parkinson’s disease. J. Neurosci. 2010, 30, 12535–12544. [Google Scholar] [CrossRef] [PubMed]
- Klucken, J.; Poehler, A.M.; Ebrahimi-Fakhari, D.; Schneider, J.; Nuber, S.; Rockenstein, E.; Schlötzer-Schrehardt, U.; Hyman, B.T.; McLean, P.J.; Masliah, E.; et al. Alpha-synuclein aggregation involves a bafilomycin A 1-sensitive autophagy pathway. Autophagy 2012, 8, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.M.; Chan, B.K.; Park, J.S.; Hill, K.J.; Aitken, J.B.; Cottle, L.; Farghaian, H.; Cole, A.R.; Lay, P.A.; Sue, C.M.; et al. Parkinson’s disease-linked human PARK9/ATP13A2 maintains zinc homeostasis and promotes alpha-Synuclein externalization via exosomes. Hum. Mol. Genet. 2014, 23, 2816–2833. [Google Scholar] [CrossRef] [PubMed]
- Crews, L.; Spencer, B.; Desplats, P.; Patrick, C.; Paulino, A.; Rockenstein, E.; Hansen, L.; Adame, A.; Galasko, D.; Masliah, E. Selective molecular alterations in the autophagy pathway in patients with Lewy body disease and in models of alpha-synucleinopathy. PLoS ONE 2010, 5, e9313. [Google Scholar] [CrossRef] [PubMed]
- Stefanis, L.; Larsen, K.E.; Rideout, H.J.; Sulzer, D.; Greene, L.A. Expression of A53T mutant but not wild-type alpha-synuclein in PC12 cells induces alterations of the ubiquitin-dependent degradation system, loss of dopamine release, and autophagic cell death. J. Neurosci. 2001, 21, 9549–9560. [Google Scholar] [CrossRef]
- Webb, J.L.; Ravikumar, B.; Atkins, J.; Skepper, J.N.; Rubinsztein, D.C. Alpha-Synuclein is degraded by both autophagy and the proteasome. J. Biol. Chem. 2003, 278, 25009–25013. [Google Scholar] [CrossRef]
- Vogiatzi, T.; Xilouri, M.; Vekrellis, K.; Stefanis, L. Wild type alpha-synuclein is degraded by chaperone-mediated autophagy and macroautophagy in neuronal cells. J. Biol. Chem. 2008, 283, 23542–23556. [Google Scholar] [CrossRef]
- Yu, W.H.; Dorado, B.; Figueroa, H.Y.; Wang, L.; Planel, E.; Cookson, M.R.; Clark, L.N.; Duff, K.E. Metabolic activity determines efficacy of macroautophagic clearance of pathological oligomeric alpha-synuclein. Am. J. Pathol. 2009, 175, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Castino, R.; Lazzeri, G.; Lenzi, P.; Bellio, N.; Follo, C.; Ferrucci, M.; Fornai, F.; Isidoro, C. Suppression of autophagy precipitates neuronal cell death following low doses of methamphetamine. J. Neurochem. 2008, 106, 1426–1439. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Chandramani-Shivalingappa, P.; Jin, H.; Ghosh, A.; Anantharam, V.; Ali, S.; Kanthasamy, A.G.; Kanthasamy, A. Methamphetamine-induced neurotoxicity linked to ubiquitin-proteasome system dysfunction and autophagy-related changes that can be modulated by protein kinase C delta in dopaminergic neuronal cells. Neuroscience 2012, 210, 308–332. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Wan, J.; Meng, J.; Banerjee, S.; Ramakrishnan, S.; Roy, S. Methamphetamine induces autophagy as a pro-survival response against apoptotic endothelial cell death through the Kappa opioid receptor. Cell Death Dis. 2014, 5, e1099. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.H.; Wu, W.R.; Lee, L.M.; Huang, P.R.; Chen, J.C. mTOR signaling in the nucleus accumbens mediates behavioral sensitization to methamphetamine. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 86, 331–339. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Yuan, W.; Li, Z.; Hou, Y.; Liu, F.; Feng, J. 6-Hydroxydopamine induces autophagic flux dysfunction by impairing transcription factor EB activation and lysosomal function in dopaminergic neurons and SH-SY5Y cells. Toxicol. Lett. 2018, 283, 58–68. [Google Scholar] [CrossRef]
- Pal, R.; Bajaj, L.; Sharma, J.; Palmieri, M.; Di Ronza, A.; Lotfi, P.; Chaudhury, A.; Neilson, J.; Sardiello, M.; Rodney, G.G. NADPH oxidase promotes Parkinsonian phenotypes by impairing autophagic flux in an mTORC1-independent fashion in a cellular model of Parkinson’s disease. Sci. Rep. 2016, 6, 22866. [Google Scholar] [CrossRef]
- Pan, T.; Rawal, P.; Wu, Y.; Xie, W.; Jankovic, J.; Le, W. Rapamycin protects against rotenone-induced apoptosis through autophagy induction. Neuroscience 2009, 164, 541–551. [Google Scholar] [CrossRef]
- Jiang, J.; Jiang, J.; Zuo, Y.; Gu, Z. Rapamycin protects the mitochondria against oxidative stress and apoptosis in a rat model of Parkinson’s disease. Int. J. Mol. Med. 2013, 31, 825–832. [Google Scholar] [CrossRef]
- Hu, Z.Y.; Chen, B.; Zhang, J.P.; Ma, Y.Y. Up-regulation of autophagy-related gene 5 (ATG5) protects dopaminergic neurons in a zebrafish model of Parkinson’s disease. J. Biol. Chem. 2017, 292, 18062–18074. [Google Scholar] [CrossRef]
- Hara, T.; Nakamura, K.; Matsui, M.; Yamamoto, A.; Nakahara, Y.; Suzuki-Migishima, R.; Yokoyama, M.; Mishima, K.; Saito, I.; Okano, H.; et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 2006, 441, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, M.; Waguri, S.; Chiba, T.; Murata, S.; Iwata, J.; Tanida, I.; Ueno, T.; Koike, M.; Uchiyama, Y.; Kominami, E.; et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 2006, 441, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, M.; Wang, Q.J.; Holstein, G.R.; Friedrich, V.L., Jr.; Iwata, J.; Kominami, E.; Chait, B.T.; Tanaka, K.; Yue, Z. Essential role for autophagy protein Atg7 in the maintenance of axonal homeostasis and the prevention of axonal degeneration. Proc. Natl. Acad. Sci. USA 2007, 104, 14489–14494. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Cho, E.D.; Lee, K.W.; Kim, J.H.; Cho, S.G.; Lee, S.J. Autophagic failure promotes the exocytosis and intercellular transfer of α-synuclein. Exp. Mol. Med. 2013, 45, e22. [Google Scholar] [CrossRef] [PubMed]
- Vekrellis, K.; Xilouri, M.; Emmanouilidou, E.; Rideout, H.J.; Stefanis, L. Pathological roles of α-synuclein in neurological disorders. Lancet Neurol. 2011, 10, 1015–1025. [Google Scholar] [CrossRef]
- Xilouri, M.; Brekk, O.R.; Stefanis, L. Autophagy and Alpha-Synuclein: Relevance to Parkinson’s Disease and Related Synucleopathies. Mov. Disord. 2016, 31, 178–192. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Mizushima, N. Autophagy and human diseases. Cell Res. 2013, 24, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Quan, L.; Ishikawa, T.; Michiue, T.; Li, D.R.; Zhao, D.; Oritani, S.; Zhu, B.L.; Maeda, H. Ubiquitin-immunoreactive structures in the midbrain of methamphetamine abusers. Leg. Med. 2005, 7, 144–150. [Google Scholar] [CrossRef]
- Limanaqi, F.; Gambardella, S.; Biagioni, F.; Busceti, C.L.; Fornai, F. Epigenetic Effects Induced by Methamphetamine and Methamphetamine-Dependent Oxidative Stress. Oxid. Med. Cell. Longev. 2018, 2018, 4982453. [Google Scholar] [CrossRef]
- Figueira, I.; Menezes, R.; Macedo, D.; Costa, I.; Dos Santos, C.N. Polyphenols Beyond Barriers: A Glimpse into the Brain. Curr. Neuropharmacol. 2017, 15, 562–594. [Google Scholar] [CrossRef]
- Lokanathan, Y.; Omar, N.; Ahmad Puzi, N.N.; Saim, A.; Hj Idrus, R. Recent Updates in Neuroprotective and Neuroregenerative Potential of Centella asiatica. Malays J. Med. Sci. 2016, 23, 4–14. [Google Scholar] [PubMed]
- Bohlmann, J.; Keeling, C.I. Terpenoid biomaterials. Plant J. 2008, 54, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, R.; Konar, A.; Kaul, S.C. Nootropic potential of Ashwagandha leaves: Beyond traditional root extracts. Neurochem. Int. 2016, 95, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Javed, H.; Nagoor Meeran, M.F.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant Extracts and Phytochemicals Targeting α-Synuclein Aggregation in Parkinson’s Disease Models. Front. Pharmacol. 2019, 9, 1555. [Google Scholar] [CrossRef] [PubMed]
- Lan, D.; Wang, W.; Zhuang, J.; Zhao, Z. Proteasome inhibitor-induced autophagy in PC12 cells overexpressing A53T mutant α-synuclein. Mol. Med. Rep. 2015, 11, 1655–1660. [Google Scholar] [CrossRef] [PubMed]
- Heebkaew, N.; Rujanapun, N.; Kunhorm, P.; Jaroonwitchawan, T.; Chaicharoenaudomrung, N.; Promjantuek, W.; Noisa, P. Curcumin Induces Neural Differentiation of Human Pluripotent Embryonal Carcinoma Cells through the Activation of Autophagy. Biomed. Res. Int. 2019, 2019, 4378710. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Zhao, Y.; Han, S.; Zhang, N.; Chen, H.; Wang, X. Autophagy-ERK1/2-Involved Disinhibition of Hippocampal Neurons Contributes to the Pre-Synaptic Toxicity Induced by Aβ42 Exposure. J. Alzheimers Dis. 2017, 59, 851–869. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Pan, X.Y.; Xu, Y.; Xiao, Y.; An, Y.; Tie, L.; Pan, Y.; Li, X.J. Curcumin induces autophagy to protect vascular endothelial cell survival from oxidative stress damage. Autophagy 2012, 8, 812–825. [Google Scholar] [CrossRef]
- Guo, S.; Long, M.; Li, X.; Zhu, S.; Zhang, M.; Yang, Z. Curcumin activates autophagy and attenuates oxidative damage in EA. hy926 cells via the Akt/mTOR pathway. Mol. Med. Rep. 2016, 13, 2187–2193. [Google Scholar] [CrossRef]
- Taebnia, N.; Morshedi, D.; Yaghmaei, S.; Aliakbari, F.; Rahimi, F.; Arpanaei, A. Curcumin-loaded amine-functionalized mesoporous silica nanoparticles inhibit α-synuclein fibrillation and reduce its cytotoxicity-associated effects. Langmuir 2016, 32, 13394–13402. [Google Scholar] [CrossRef]
- Bollimpelli, V.S.; Kumar, P.; Kumari, S.; Kondapi, A.K. Neuroprotective effect of curcumin-loaded lactoferrin nano particles against rotenone induced neurotoxicity. Neurochem. Int. 2016, 95, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Jaroonwitchawan, T.; Chaicharoenaudomrung, N.; Namkaew, J.; Noisa, P. Curcumin attenuates paraquat-induced cell death in human neuroblastoma cells through modulating oxidative stress and autophagy. Neurosci. Lett. 2017, 636, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Ma, K.; Wu, H.Y.; Wu, Y.P.; Li, B.X. Isoflavones Induce BEX2-Dependent Autophagy to Prevent ATR-Induced Neurotoxicity in SH-SY5Y Cells. Cell. Physiol. Biochem. 2017, 43, 1866–1879. [Google Scholar] [CrossRef] [PubMed]
- Song, J.X.; Sun, Y.R.; Peluso, I.; Zeng, Y.; Yu, X.; Lu, J.H.; Xu, Z.; Wang, M.Z.; Liu, L.F.; Huang, Y.Y.; et al. A novel curcumin analog binds to and activates TFEB in vitro and in vivo independent of MTOR inhibition. Autophagy 2016, 12, 1372–1389. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, J.; Xu, J.; Lu, Y.; Jiang, J.; Wang, L.; Shen, H.M.; Xia, D. Curcumin targets the TFEB-lysosome pathway for induction of autophagy. Oncotarget 2016, 7, 75659–75671. [Google Scholar] [CrossRef] [PubMed]
- Decressac, M.; Mattsson, B.; Weikop, P.; Lundblad, M.; Jakobsson, J.; Björklund, A. TFEB-mediated autophagy rescues midbrain dopamine neurons from α-synuclein toxicity. Proc. Natl. Acad. Sci. USA 2013, 110, E1817–E1826. [Google Scholar] [CrossRef] [PubMed]
- Kundu, P.; Das, M.; Tripathy, K.; Sahoo, S.K. Delivery of dual drug loaded lipid based nanoparticles across the blood-brain barrier imparts enhanced neuroprotection in a rotenone induced mouse model of Parkinson’s disease. ACS Chem. Neurosci. 2016, 7, 1658–1670. [Google Scholar] [CrossRef]
- Das, D.N.; Naik, P.P.; Nayak, A.; Panda, P.K.; Mukhopadhyay, S.; Sinha, N.; Bhutia, S.K. Bacopa monnieri-Induced Protective Autophagy Inhibits Benzo[a]pyrene-Mediated Apoptosis. Phytother. Res. 2016, 30, 1794–1801. [Google Scholar] [CrossRef]
- Smith, E.; Palethorpe, H.M.; Tomita, Y.; Pei, J.V.; Townsend, A.R.; Price, T.J.; Young, J.P.; Yool, A.J.; Hardingham, J.E. The Purified Extract from the Medicinal Plant Bacopa monnieri, Bacopaside II, Inhibits Growth of Colon Cancer Cells In Vitro by Inducing Cell Cycle Arrest and Apoptosis. Cells 2018, 7, 81. [Google Scholar] [CrossRef]
- Kim, H.S.; Quon, M.J.; Kim, J.A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014, 2, 187–195. [Google Scholar] [CrossRef]
- Prasanth, M.I.; Sivamaruthi, B.S.; Chaiyasut, C.; Tencomnao, T. A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy. Nutrients 2019, 11, 474. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.C.; Bartlett, J.J.; Perez, L.J.; Brunt, K.R.; Legare, J.F.; Hassan, A.; Kienesberger, P.C.; Pulinilkunnil, T. Glucolipotoxicity diminishes cardiomyocyte TFEB and inhibits lysosomal autophagy during obesity and diabetes. Biochim. Biophys. Acta 2016, 1861, 1893–1910. [Google Scholar] [CrossRef] [PubMed]
- Holczer, M.; Besze, B.; Zámbó, V.; Csala, M.; Bánhegyi, G.; Kapuy, O. Epigallocatechin-3-Gallate (EGCG) Promotes Autophagy-Dependent Survival via Influencing the Balance of mTOR-AMPK Pathways upon Endoplasmic Reticulum Stress. Oxid. Med. Cell. Longev. 2018, 2018, 6721530. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, S.; Xu, J.; Li, W.; Duan, G.; Wang, H.; Yang, H.; Yang, Z.; Zhou, R. A new molecular mechanism underlying the EGCG-mediated autophagic modulation of AFP in HepG2 cells. Cell Death Dis. 2017, 8, e3160. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Moon, J.H.; Kim, S.W.; Jeong, J.K.; Nazim, U.M.; Lee, Y.J.; Seol, J.W.; Park, S.Y. EGCG-mediated autophagy flux has a neuroprotection effect via a class III histone deacetylase in primary neuron cells. Oncotarget 2015, 6, 9701–9717. [Google Scholar] [CrossRef]
- Li, W.; Zhu, S.; Li, J.; Assa, A.; Jundoria, A.; Xu, J.; Fan, S.; Eissa, N.T.; Tracey, K.J.; Sama, A.E.; et al. EGCG stimulates autophagy and reduces cytoplasmic HMGB1 levels in endotoxin-stimulated macrophages. Biochem. Pharmacol. 2011, 81, 1152–1163. [Google Scholar] [CrossRef]
- Kim, H.S.; Montana, V.; Jang, H.J.; Parpura, V.; Kim, J.A. Epigallocatechin gallate (EGCG) stimulates autophagy in vascular endothelial cells: A potential role for reducing lipid accumulation. J. Biol. Chem. 2013, 288, 22693–22705. [Google Scholar] [CrossRef]
- Zhou, J.; Farah, B.L.; Sinha, R.A.; Wu, Y.; Singh, B.K.; Bay, B.H.; Yang, C.S.; Yen, P.M. Epigallocatechin-3-Gallate (EGCG), a green tea polyphenol, stimulates hepatic autophagy and lipid clearance. PLoS ONE 2014, 9, e87161. [Google Scholar] [CrossRef]
- Chen, C.M.; Wu, C.T.; Yang, T.H.; Chang, Y.A.; Sheu, M.L.; Liu, S.H. Green Tea Catechin Prevents Hypoxia/Reperfusion-Evoked Oxidative Stress-Regulated Autophagy-Activated Apoptosis and Cell Death in Microglial Cells. J. Agric. Food Chem. 2016, 64, 4078–4085. [Google Scholar] [CrossRef]
- Wei, R.; Mao, L.; Xu, P.; Zheng, X.; Hackman, R.M.; Mackenzie, G.G.; Wang, Y. Suppressing glucose metabolism with epigallocatechin-3-gallate (EGCG) reduces breast cancer cell growth in preclinical models. Food Funct. 2018, 9, 5682–5696. [Google Scholar] [CrossRef]
- Grube, S.; Ewald, C.; Kögler, C.; Lawson McLean, A.; Kalff, R.; Walter, J. Achievable Central Nervous System Concentrations of the Green Tea Catechin EGCG Induce Stress in Glioblastoma Cells in Vitro. Nutr. Cancer 2018, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Sakagami, H. Induction of apoptosis by epigallocatechin gallate and autophagy inhibitors in a mouse macrophage-like cell line. Anticancer Res. 2008, 28, 1713–1718. [Google Scholar] [PubMed]
- Renaud, J.; Nabavi, S.F.; Daglia, M.; Nabavi, S.M.; Martinoli, M.G. Epigallocatechin-3-Gallate, a Promising Molecule for Parkinson’s Disease? Rejuvenation Res. 2015, 18, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.H.; Kim, S.H.; Kwon, H.; Park, Y.; Kim, K.S.; Song, C.W.; Kim, J.; Kim, M.H.; Yu, H.J.; Henkel, J.S.; et al. Epigallocatechin gallate protects nerve growth factor differentiated PC12 cells from oxidative-radical-stress-induced apoptosis through its effect on phosphoinositide 3-kinase/Akt and glycogen synthase kinase-3. Brain Res. Mol. Brain. Res. 2003, 118, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Šneideris, T.; Baranauskiene, L.; Cannon, J.G.; Rutkiene, R.; Meškys, R.; Smirnovas, V. Looking for a generic inhibitor of amyloid-like fibril formation among flavone derivatives. Peer J. 2015, 3, e1271. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, O.; Mandel, S.; Youdim, M.B.H.; Amit, T. Targeting dysregulation of brain iron homeostasis in Parkinson’s disease by iron chelators. Free Radic. Biol. Med. 2013, 62, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Bosch-Marce, M.; Shimoda, L.A.; Tan, Y.S.; Baek, J.H.; Wesley, J.B.; Gonzalez, F.J.; Semenza, G.L. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J. Biol. Chem. 2008, 283, 10892–91003. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishna, N.; Meeker, H.C.; Brown, W.T. Novel Epigenetic Regulation of Alpha-Synuclein Expression in Down Syndrome. Mol. Neurobiol. 2016, 53, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Ehrnhoefer, D.E.; Bieschke, J.; Boeddrich, A.; Herbst, M.; Masino, L.; Lurz, R.; Engemann, S.; Pastore, A.; Wanker, E.E. EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat. Struct. Mol. Biol. 2008, 15, 558–566. [Google Scholar] [CrossRef]
- Bae, S.Y.; Kim, S.; Hwang, H.; Kim, H.K.; Yoon, H.C.; Kim, J.H.; Lee, S.; Kim, T.D. Amyloid formation and disaggregation of α-synuclein and its tandem repeat (α-TR). Biochem. Biophys. Res. Commun. 2010, 400, 531–536. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, S.; Shi, D.; Bai, Q.; Liu, H.; Yao, X. Influence of EGCG on α-synuclein (αS) aggregation and identification of their possible binding mode: A computational study using molecular dynamics simulation. Chem. Biol. Drug Des. 2018, 91, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.; Gong, Q.; Xiong, X.; Sun, L.; Zhao, W.; Zhu, W.; Lu, Y. Protective effect of madecassoside on H2O2-induced oxidative stress and autophagy activation in human melanocytes. Oncotarget 2017, 8, 51066–51075. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.; Cao, Q.X.; Zhai, F.R.; Yang, S.Q.; Zhang, H.X. Asiatic acid exerts anticancer potential in human ovarian cancer cells via suppression of PI3K/Akt/mTOR signalling. Pharm. Biol. 2016, 54, 2377–2382. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.F.; Xiong, Y.Y.; Liu, J.K.; Qian, J.J.; Zhu, L.; Gao, J. Asiatic acid, a pentacyclic triterpene in Centella asiatica, attenuates glutamate-induced cognitive deficits in mice and apoptosis in SH-SY5Y cells. Acta Pharmacol. Sin. 2012, 33, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Berrocal, R.; Vasudevaraju, P.; Indi, S.S.; Sambasiva Rao, K.R.; Rao, K.S. In vitro evidence that an aqueous extract of Centella asiatica modulates α-synuclein aggregation dynamics. J. Alzheimers Dis. 2014, 39, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Carver, J.A.; Calabrese, A.N.; Pukala, T.L. Gallic acid interacts with α-synuclein to prevent the structural collapse necessary for its aggregation. Biochim. Biophys. Acta 2014, 1844, 1481–1485. [Google Scholar] [CrossRef] [PubMed]
- Ardah, M.T.; Paleologou, K.E.; Lv, G.; Abul Khair, S.B.; Kazim, A.S.; Minhas, S.T.; Al-Tel, T.H.; Al-Hayani, A.A.; Haque, M.E.; Eliezer, D.; et al. Structure activity relationship of phenolic acid inhibitors of α-synuclein fibril formation and toxicity. Front. Aging Neurosci. 2014, 6, 197. [Google Scholar] [CrossRef] [PubMed]
- Akhoon, B.A.; Pandey, S.; Tiwari, S.; Pandey, R. Withanolide A offers neuroprotection, ameliorates stress resistance and prolongs the life expectancy of Caenorhabditis elegans. Exp. Gerontol. 2016, 78, 47–56. [Google Scholar] [CrossRef]
- Kuboyama, T.; Tohda, C.; Komatsu, K. Neuritic regeneration and synaptic reconstruction induced by withanolide A. Br. J. Pharmacol. 2005, 144, 961–971. [Google Scholar] [CrossRef]
- Okamoto, S.; Tsujioka, T.; Suemori, S.; Kida, J.; Kondo, T.; Tohyama, Y.; Tohyama, K. Withaferin A suppresses the growth of myelodysplasia and leukemia cell lines by inhibiting cell cycle progression. Cancer Sci. 2016, 107, 1302–1314. [Google Scholar] [CrossRef]
- Dutta, K.; Patel, P.; Julien, J.P. Protective effects of Withania somnifera extract in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Exp. Neurol. 2018, 309, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhu, F.; Jiang, J.; Sun, C.; Zhong, Q.; Shen, M.; Wang, X.; Tian, R.; Shi, C.; Xu, M.; et al. Simultaneous inhibition of the ubiquitin-proteasome system and autophagy enhances apoptosis induced by ER stress aggravators in human pancreatic cancer cells. Autophagy 2016, 12, 1521–1537. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; De, S.; Mukherjee, S.; Das, S.; Ghosh, A.N.; Sengupta, S.B. Withaferin A induced impaired autophagy and unfolded protein response in human breast cancer cell-lines MCF-7 and MDA-MB-231. Toxicol. In Vitro 2017, 44, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Muniraj, N.; Siddharth, S.; Nagalingam, A.; Walker, A.; Woo, J.; Gyorffy, B.; Gabrielson, E.; Saxena, N.K.; Sharma, D. Withaferin A inhibits lysosomal activity to block autophagic flux and induces apoptosis via energetic impairment in breast cancer cells. Carcinogenesis 2019, in press. [Google Scholar] [CrossRef]
- Siddharth, S.; Muniraj, N.; Saxena, N.K.; Sharma, D. Concomitant Inhibition of Cytoprotective Autophagy Augments the Efficacy of Withaferin A in Hepatocellular Carcinoma. Cancers 2019, 11, 453. [Google Scholar] [CrossRef]
- Rah, B.; ur Rasool, R.; Nayak, D.; Yousuf, S.K.; Mukherjee, D.; Kumar, L.D.; Goswami, A. PAWR-mediated suppression of BCL2 promotes switching of 3-azido withaferin A (3-AWA)-induced autophagy to apoptosis in prostate cancer cells. Autophagy 2015, 11, 314–331. [Google Scholar] [CrossRef] [PubMed]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar] [CrossRef]
- Park, D.; Jeong, H.; Lee, M.N.; Koh, A.; Kwon, O.; Yang, Y.R.; Noh, J.; Suh, P.G.; Park, H.; Ryu, S.H. Resveratrol induces autophagy by directly inhibiting mTOR through ATP competition. Sci. Rep. 2016, 6, 21772. [Google Scholar] [CrossRef]
- Suvorova, I.I.; Knyazeva, A.R.; Petukhov, A.V.; Aksenov, N.D.; Pospelov, VA. Resveratrol enhances pluripotency of mouse embryonic stem cells by activating AMPK/Ulk1 pathway. Cell Death Discov. 2019, 5, 61. [Google Scholar] [CrossRef]
- Joe, I.S.; Jeong, S.G.; Cho, G.W. Resveratrol-induced SIRT1 activation promotes neuronal differentiation of human bone marrow mesenchymal stem cells. Neurosci. Lett. 2015, 584, 97–102. [Google Scholar] [CrossRef]
- Yang, Q.B.; He, Y.L.; Zhong, X.W.; Xie, W.G.; Zhou, J.G. Resveratrol ameliorates gouty inflammation via upregulation of sirtuin 1 to promote autophagy in gout patients. Inflammopharmacology 2019, 27, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, J.; Cao, N.; Li, Z.; Han, J.; Li, L. Resveratrol, an activator of SIRT1, induces protective autophagy in non-small-cell lung cancer via inhibiting Akt/mTOR and activating p38-MAPK. Onco Targets Ther. 2018, 11, 7777–7786. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Han, X. Resveratrol alleviates early brain injury following subarachnoid hemorrhage: Possible involvement of the AMPK/SIRT1/autophagy signaling pathway. Biol. Chem. 2018, 399, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Li, Z.; Wang, Y.; Hou, Y.; Li, L.; Zhao, J. Resveratrol alleviates cerebral ischemia/reperfusion injury in rats by inhibiting NLRP3 inflammasome activation through Sirt1-dependent autophagy induction. Int. Immunopharmacol. 2017, 50, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Bai, L.; Lu, W.; Gao, Y.; Bi, Y.; Lv, G. Regulation of autophagy by AMP-activated protein kinase/sirtuin 1 pathway reduces spinal cord neurons damage. Iran. J. Basic Med. Sci. 2017, 20, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Chen, S.; Gao, K.; Zhou, Z.; Wang, C.; Shen, Z.; Guo, Y.; Li, Z.; Wan, Z.; Liu, C.; et al. Resveratrol protects against spinal cord injury by activating autophagy and inhibiting apoptosis mediated by the SIRT1/AMPK signaling pathway. Neuroscience 2017, 348, 241–251. [Google Scholar] [CrossRef]
- Deng, H.; Mi, M.T. Resveratrol Attenuates Aβ25-35 Caused Neurotoxicity by Inducing Autophagy Through the TyrRS-PARP1-SIRT1 Signaling Pathway. Neurochem. Res. 2016, 41, 2367–2379. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cao, X.; Zhu, W.; Liu, Z.; Liu, H.; Zhou, Y.; Cao, Y.; Liu, C.; Xie, Y. Resveratrol Enhances Autophagic Flux and Promotes Ox-LDL Degradation in HUVECs via Upregulation of SIRT1. Oxid. Med. Cell. Longev. 2016, 2016, 7589813. [Google Scholar] [CrossRef]
- Al Azzaz, J.; Rieu, A.; Aires, V.; Delmas, D.; Chluba, J.; Winckler, P.; Bringer, M.A.; Lamarche, J.; Vervandier-Fasseur, D.; Dalle, F.; et al. Resveratrol-Induced Xenophagy Promotes Intracellular Bacteria Clearance in Intestinal Epithelial Cells and Macrophages. Front. Immunol. 2019, 9, 3149. [Google Scholar] [CrossRef]
- Du, L.; Chen, E.; Wu, T.; Ruan, Y.; Wu, S. Resveratrol attenuates hydrogen peroxide-induced aging through upregulation of autophagy in human umbilical vein endothelial cells. Drug Des. Devel. Ther. 2019, 13, 747–755. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, T.; Li, W.; Gao, N.; Zhang, T. Resveratrol attenuates oxidative damage through activating mitophagy in an in vitro model of Alzheimer’s disease. Toxicol. Lett. 2018, 282, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Sivanesam, K.; Andersen, N.H. Modulating the Amyloidogenesis of α-Synuclein. Curr. Neuropharmacol. 2016, 14, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Kang, S.J.; Poncz, M.; Song, K.J.; Park, K.S. Resveratrol protects SH-SY5Y neuroblastoma cells from apoptosis induced by dopamine. Exp. Mol. Med. 2007, 39, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.C.; Wei, Y.; An, Z.; Zou, Z.; Xiao, G.; Bhagat, G.; White, M.; Reichelt, J.; Levine, B. Akt-mediated regulation of autophagy and tumorigenesis through Beclin 1 phosphorylation. Science 2012, 338, 956–959. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; She, H.; Zhang, T.; Xu, H.; Cheng, L.; Yepes, M.; Zhao, Y.; Mao, Z. p38 MAPK inhibits autophagy and promotes microglial inflammatory responses by phosphorylating ULK1. J. Cell Biol. 2018, 217, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Yang, S.; Li, S.; Hang, C. Melatonin Upregulates Nuclear Factor Erythroid-2 Related Factor 2 (Nrf2) and Mediates Mitophagy to Protect Against Early Brain Injury After Subarachnoid Hemorrhage. Med. Sci. Monit. 2018, 24, 6422–6430. [Google Scholar] [CrossRef]
- Manzoni, C.; Mamais, A.; Dihanich, S.; Abeti, R.; Soutar, M.P.M.; Plun-Favreau, H.; Giunti, P.; Tooze, S.A.; Bandopadhyay, R.; Lewis, P.A. Inhibition of LRRK2 kinase activity stimulates macroautophagy. Biochim. Biophys. Acta 2013, 1833, 2900–2910. [Google Scholar] [CrossRef]
- Wang, D.; Ji, X.; Liu, J.; Li, Z.; Zhang, X. Dopamine Receptor Subtypes Differentially Regulate Autophagy. Int. J. Mol. Sci. 2018, 19, 1540. [Google Scholar] [CrossRef]
- Chen, A.; Xiong, L.J.; Tong, Y.; Mao, M. Neuroprotective effect of brain-derived neurotrophic factor mediated by autophagy through the PI3K/Akt/mTOR pathway. Mol. Med. Rep. 2013, 8, 1011–1016. [Google Scholar] [CrossRef]
- Pandey, U.B.; Nie, Z.; Batlevi, Y.; McCray, B.A.; Ritson, G.P.; Nedelsky, N.B.; Schwartz, S.L.; DiProspero, N.A.; Knight, M.A.; Schuldiner, O.; et al. HDAC6 rescues neurodegeneration and provides an essential link between autophagy and the UPS. Nature 2007, 447, 859–863. [Google Scholar] [CrossRef]
- Natale, G.; Pasquali, L.; Paparelli, A.; Fornai, F. Parallel manifestations of neuropathologies in the enteric and central nervous systems. Neurogastroenterol. Motil. 2011, 23, 1056–1065. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, Y.; Chen, Z.; Leng, S.X. Connection between Systemic Inflammation and Neuroinflammation Underlies Neuroprotective Mechanism of Several Phytochemicals in Neurodegenerative Diseases. Oxid. Med. Cell. Longev. 2018, 2018, 1972714. [Google Scholar] [CrossRef] [PubMed]
- Akintunde, J.K.; Farouk, A.A.; Mogbojuri, O. Metabolic treatment of syndrome linked with Parkinson’s disease and hypothalamus pituitary gonadal hormones by turmeric curcumin in Bisphenol-A induced neuro-testicular dysfunction of wistar rat. Biochem. Biophys. Rep. 2018, 17, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Forni, C.; Facchiano, F.; Bartoli, M.; Pieretti, S.; Facchiano, A.; D’Arcangelo, D.; Norelli, S.; Valle, G.; Nisini, R.; Beninati, S.; et al. Beneficial Role of Phytochemicals on Oxidative Stress and Age-Related Diseases. Biomed. Res. Int. 2019, 2019, 8748253. [Google Scholar] [CrossRef] [PubMed]
| PD Model | Cell-Based Models | In Vivo Models |
|---|---|---|
| MPTP [28,29,30,31,32,33,34] | Nanoparticle-loaded curcumin in SH-SY5Y [28] ↓cytotoxicity and necrotic-like morphologic alterations ↑DA and tyrosine hydroxylase (TH) levels ↓α-syn aggregation Serum from Curcumin-activated human mesenchymal stem cells in PC12 cells [29] ↓apoptosis ↑neuronal differentiation, DAT and TH expression ↓pro-inflammatory cytokine release ↓nitric oxide (NO), and inducible NO synthase (iNOS) levels Curcumin in SH-SY5Y [30,31] ↓cytotoxicity [30,31] ↓α-syn protein and mRNA levels [30] ↑LAMP2 and LC3II and TFEB-dependent autophagy [30] ↓c-Jun, c-Jun N-Terminal Kinase (JNK), and caspase-3 [31] | Nanoparticle-loaded [28] or free curcumin [31,32,33,34] in mice ↓Parkinsonian motor symptoms [28,31,32,34] ↓loss of TH-positive neurons, depletion of DA levels and dopamine transporter (DAT)-positive fibers in the striatum [28,31,32,33,34] ↓α-syn positive Lewy Bodies [34] ↓lipid peroxidation [34] ↑antioxidant markers superoxide dismutase (SOD) and glutathione (GSH) [34] ↓JNK and caspase-related apoptotic pathways [31,32] ↑regeneration of neuroblasts in the subventricular zone (SVZ) [33] ↑growth-derived neurotrophic factor (GDNF) and transforming growth factor beta 1 (TGF-β1) levels in the SVZ [33] |
| 6-OHDA [35,36,37,38,39,40] | Curcumin in deutocerebrum primary cells [35] ↑survival, antioxidant defense, and adhesive ability ↑Wnt/β-catenin signaling pathway Curcumin in MES23.5 and SH-SY5Y cells [37,38] ↓neurotoxicity ↓ROS accumulation ↓p53-mediated apoptosis ↓Nuclear Factor K Beta (NF-kβ) nuclear translocation ↑antioxidant enzyme levels and mitochondrial membrane potential (MMP) Curcumin in SH-SY5Y [39] ↓toxicity ↓loss of TH ↓toxic quinone formation ↓p38-Mitogen-Activated Protein Kinase (MAPK) and caspase-3 | Curcumin in rats [35,36] ↓neurotoxicity ↓behavioral alterations ↑TH and DAT expression ↓Glial Fibrillary Acidic Protein (GFAP), Heat shock protein 70 (HSP70), and Malondialdehyde (MDA) content ↑antioxidant markers SOD and GSH ↑Wnt3/b-catenin pathway, neurotrophic growth factors (NGF) and tyrosine receptor kinase A (TrkA) expression Curcumin in mice [40] ↓L-DOPA-induced dyskinesia ↓Extracellular Signal-Regulated Kinase (ERK)-related AP-1 family transcription factors c-Fos, Fra, FosB, and c-Jun |
| Rotenone [41,42,43] | Demethoxycurcumin (DMC), a derivative of curcumin, in SH-SY5Y cells [41] ↓toxicity ↓ intracellular ROS ↓proapoptotic proteins Bax, BAD, caspase-3, -6, -8, -9 in mitochondria, and cytochrome (Cyt)-c in the cytosol ↑MMP ↑antiapoptotic markers Bcl-2, Bcl-xL, and Cyt-c in mitochondria | Curcumin in rats [42] ↓motor dysfunction ↑TH activity ↓GSH, Heme Oxygenase-1, and Nicotinamide Adenine Dinucleotide Phosphate Hydrogen (NADPH):quinone oxidoreductase 1 levels ↓ROS and MDA ↑Akt-Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2) pathway Curcumin in mice [43] ↓parkinsonian behavior ↓lipid peroxidation and nitrite levels ↑antioxidant enzymes SOD, catalase (CAT), and GSH ↑Succinate Dehydrogenase (SDH) activity and mitochondrial enzyme complex activity |
| Copper [44,45] | Curcumin in mice [44,45] ↑locomotor activity ↑TH expression within SNc, ventral tegmental area (VTA), and dorsal striatum ↓loss of GFAP levels | |
| LPS [46] | Curcumin in rats [46] ↓iron deposition ↓α-syn aggregation ↓pro-apoptotic markers Bax, Caspase 3, and Caspase 9 ↓inflammatory response markers GFAP, NF-kβ, tumor necrosis factor alpha (TNF-α), interleukine (IL)-1β and 1α, and iNOS ↑NADPH oxidase complex and GSH | |
| α-Syn overexpression/mutation [47,48,49,50] | Curcumin in SH-SY5Y cells [47] ↓cytotoxicity induced by either extracellularly administered or intracellularly overexpressed α-syn ↓cytotoxicity of aggregated α-syn ↓ROS ↓caspase-3 activation and apoptosis Curcumin in SH-SY5Y cells [48] ↓A53T α-syn-induced cytotoxicity↓ cytoskeletal pathology ↓α-syn overload ↓mTOR ↑autophagy, LC3II levels and co-localization of LC3-α-syn puncta Curcumin in PC12 cells [49] ↓A53T α-syn-induced cytotoxicity ↓ROS, Cyt-c release, caspase-9 and -3 activation, and mitochondrial depolarization | Curcumin in Drosophila models expressing human α-syn [50] ↑lifespan and activity pattern ↓oxidative stress, apoptosis, lipid peroxidation protein carbonyl overload |
| dUCH Knockout [51] | Curcumin in Drosophila ubiquitin carboxy-terminal hydrolase (UCH)-KO [51] ↓locomotor defects ↓loss of TH-positive neurons and DA levels ↓ROS | |
| DJ-1 Knockout [52] | Liposomal-formulated curcumin in DJ-1-KO rats [52] ↑motor activity ↓apoptosis ↑stimulates DA neurogenesis through targeting histone deacetylase (HDAC) inhibition | |
| PINK1 siRNA [53] | Curcumin in PINK1 siRNA SH-SY5Y cells [53] ↓apoptosis ↑MMP and maximal respiration Curcumin in PINK1 siRNA SH-SY5Y cells exposed to paraquat [53] ↔apoptosis and mitochondrial dysfunctions | |
| LRRK2 mutation [54] | Curcumin in LRRK2-transfected HEK293T cells and primary neurons treated with H2O2 [54] ↓combined cytotoxicity ↓LRRK2 kinase activity | Curcumin in LRRK2-transgenic Drosophila exposed to H2O2 [54] ↑survival and locomotor activity ↓loss of DA neurons ↓oxidized protein levels and LRRK2 kinase activity |
| PD Model | Cell-Based Models | In Vivo Models |
|---|---|---|
| MPTP [55,56,57,58] | B. monnieri in SH-SY5Y cells [55] ↓toxicity and morphologic alterations ↑mitochondrial functions, MMP, NADH dehydrogenase, mitochondrial complex I activity ↑proteasome activity and GSH levels ↑pAkt/total Akt ratio, and activation of Nrf2 | B. monnieri in mice [56,57] ↓Parkinsonian motor abnormalities ↓TH-positive cell loss ↑DA and its metabolite levels ↑neurogenic genes in the SNc ↓lipid peroxidation and nitrite levels ↑antioxidant enzymes CAT, glutathione reductase and peroxidase (GR and GPx), ↓apoptotic enzymes caspase-3 and Bax ↑antiapoptotic enzyme Bcl-2 Nanoparticle-loaded B. monnieri in zebrafish [58] ↓Parkinsonian motor symptoms ↑DA and its metabolites levels ↑GSH, GPx, CAT, SOD, and mitochondrial complex-I ↓lipid peroxidation, MDA levels |
| Paraquat [55,59,60,61,62,63] | B. monnieri in SH-SY5Y cells [55] ↓toxicity ↓ROS and superoxide anione levels ↑GSH and antioxidant enzymes levels ↑pAkt/total Akt ratio and Nrf2 activation B. monnieri in PC12 cells [60] ↓toxicity ↑TH levels ↓ROS, superoxide anion, MMP ↑antioxidant systems glutamylcysteine synthetase (GCS) and thioredoxin1 (Trx1) levels ↓activation of Akt and HSP90 | B. monnieri in Drosophila [59,63] ↓oxidative stress, mitochondrial dysfunctions, and lethality [59,63] ↑survival and locomotor activity [63] ↓MDA, ROS and H2O2 levels [59,63] ↓apoptosis-associated genes and proteins JNK, caspase-3 [63] ↑SDH, mitochondrial complex I-III and II-III enzymes, CAT, and ATP [59,63] B. monnieri in mice [61,62] ↓behavioral alterations in mice ↓oxidative stress, mitochondrial dysfunctions, and neurotransmitter alterations ↓ROS, MDA and H2O2 levels ↑SDH and mitochondrial complex enzymes activities ↑cholinergic enzymes activity and striatal DA levels |
| 6-OHDA [64,65] | B. monnieri in rats ↓behavioral alterations ↓lipid peroxidation ↑GSH content, and the amount and activities of the antioxidant GPx, GST, SOD, and CAT enzymes [64]. B. monnieri in C. elegans ↓loss of GFP-tagged DA neurons [65] | |
| Rotenone [66,67] | B. monnieri in N27 DA-cells [66] ↓toxicity ↓ROS and H2O2 levels ↑GSH levels | B. monnieri in flies and mice [66,67] ↓toxicity and motor alterations ↑cholinergic enzymes activity and striatal DA levels ↓lipid peroxidation, MDA and H2O2 levels, protein carbonyl content ↑GSH, SOD and CAT content |
| PINK1-KO [68] | B. monnieri in PINK1-KO flies [68] ↑climbing ability | |
| α-Syn Overexpression [65] | B. monnieri in C. elegans transgenic models overexpressing human α-syn [65] ↓α-syn aggregation ↑lipid content |
| PD Model | Cell-Based Models | In Vivo Models |
|---|---|---|
| MPTP [69,70,71,72,73] | EGCG in PC12 cells [69] ↓cytotoxicity ↓ROS production ↑antioxidant enzymes SOD1 and GPx ↑SIRT1/ Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-alpha (PGC-1α) pathway | EGCG in mice [70,71,72,73] ↓motor abnormalities [72,73] ↓loss of TH-positive neurons [70,71,73] ↑striatal DA levels, TH amount and activity [70,71,72] ↓α-syn accumulation [70,71] ↑Bcl-2 ↓Bax [70,71] ↑Protein Kinase C alpha (PKC-α) overexpression [70,71] ↓oxidative stress and protein carbonyl content [72] ↑ iron-export protein ferroportin [72] ↓the ratio of CD3+CD4+ to CD3+CD8+ T lymphocytes in the peripheral blood [73] ↓TNF-α and IL-6 in the serum [73] |
| DJ-1-KO/Parkin-KO + Paraquat [74,75] | Catechins (EGCG and propyl gallate, PG) in paraquat-exposed DJ-1-KO or Parkin-KO Drosophila [74,75] ↑life-span and locomotor activity ↓degeneration of TH-positive neurons ↓lipid peroxidation | |
| 6-OHDA [76,77,78] | EGCG in PC12 and SH-SY5Y cells [76] ↓toxicity ↓Nf-kβ nuclear translocation and binding activity | C. sinensis extracts and catechins in rats [77] ↓behavioral alterations ↑TH- and cyclooxygenase (COX)-2 immunopositivity ↑DA and its metabolites levels in the striatum ↓lipid peroxidation, nitrite levels, and iNOS immunopositivity EGCG in rats [78] ↓motor alterations and apoptosis in the SN ↓α-syn, mTOR, AKT, and GSK3β levels |
| Rotenone [79] | EGCG in RGC-5 [79] ↓toxicity ↓lipid peroxidation ↓MAPK, c-Jun, JNK, and p38 | |
| Parkin-KO and/or LRRK2 mutation [80] | EGCG in Parkin-null or LRRK2-mutated Drosophila [80] ↑climbing scores ↓loss of DA neurons ↑mitochondrial integrity ↑activation of AMPK | |
| α-Syn overexpression [81] | EGCG in Drosophila expressing human α-syn in the brain [81] ↑climbing ability ↓apoptosis and lipid peroxidation |
| PD Model | Cell-Based Models | In Vivo Models |
|---|---|---|
| MPTP [82,83,84,85] | C. asiatica in mice [82,83] ↓motor abnormalities [82,83] ↑DA levels, DAT and vesicular monoamine transporter type 2 (VMAT2) in the SN and striatum [82] ↑Brain-Derived and Vascular-Endothelial Growth Factors (BDNF, VEGF), GDNF, and TrKB [82] ↓MAPK-P38 related activation of JNK and ERK [82] ↑SOD, CAT, GPx, and GSH [83] ↓lipid peroxidation [83] C. asiatica in rats [84,85] ↓motor abnormalities [84] ↑DA and its metabolite levels [84] ↓lipid peroxidation, MDA, and protein carbonyl content [84,85] ↑GSH, Bcl-2/Bax ratio, BDNF [84] ↑SOD, CAT, GPx, and GSH [85] | |
| Rotenone [86,87] | C. asiatica in SH-SY5Y cells [86] ↓cytotoxicity, ROS, apoptosis, and DNA damage ↑MPP, Bcl-2 ↓Bax, Cyt-c, caspases-3, -6, -8, and -9 | C. asiatica in rats [87] ↓motor deficits ↓loss of TH-immunopositivity in the SN and striatum ↓ lipid peroxidation, MDA levels ↑mitochondrial complex I activity, SOD, and CAT |
| α-syn overexpression [88] | C. asiatica in Drosophila expressing human α-syn in the brain [88] ↑climbing ability and activity pattern ↓lipid peroxidation, MDA, and protein carbonyl content ↑GSH | |
| PINK1-KO [68] | C. asiatica in PINK1-KO Drosophila [68] ↑climbing ability |
| PD Model | In Vivo Models |
|---|---|
| MPTP [83,89,90,91] | Ashwagandha in mice [83,89,90,91] ↓Parkinsonian motor abnormalities [83,89,90,91] ↑DA and its metabolite levels [89,90,91] ↑GSH, GPx, SOD, and CAT [83,89,90,91] ↓lipid peroxidation and thiobarbituric acid reactive substance (TBARS) [83,89,90,91] |
| Maneb-Paraquat [92,93] | Ashwagandha in mice [92,93] ↓behavioral alterations and TH loss ↓ROS, lipid peroxidation, iNOS, Bax, GFAP ↑Bcl-2, CAT |
| 6-OHDA [94] | Ashwagandha in rats [94] ↓behavioral alterations ↑TH expression, DA and its metabolite levels, DA D2 receptor binding ↓lipid peroxidation ↑GSH, GPx, GR, GST, SOD, and CAT |
| Rotenone [95] | Ashwagandha in Drosophila [95] ↓toxicity and motor alterations ↑striatal DA levels ↓ROS, lipid peroxidation, and H2O4 ↑GSH, GST, SOD, and CAT ↑SDH, mitochondrial complex-I-III and complex-II-III |
| LRRK2 mutation [96] | Ashwagandha in adult Drosophila [96] ↑lifespan, locomotor activity, muscle electrophysiological response to stimuli ↓mitochondria degeneration |
| PINK1-KO [68] | Ashwagandha in PINK1-KO Drosophila [68] ↑climbing ability |
| PD Model | Cell-Based Models | In Vivo Models |
|---|---|---|
| MPTP [97,98,99,100,101] | Resveratrol in SH-SY5Y cells [97] ↓cytotoxicity and apoptosis ↓α-syn mRNA levels ↓metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) and miR-129 expression | Resveratrol in mice [97,98,99,100] ↓Parkinsonian motor symptoms [98,99,100] ↓loss of TH-positive neurons and striatal DA depletion [97,98,99,100] ↓α-syn levels [97,99,100] ↓apoptosis, Bax and Caspase 3 [97,99] ↓MALAT1 and miR-129 expression [97] ↓proinflammatory cytokine IL-1β and GFAP [99] ↑pAkt/Akt ratio [99] ↓p62 levels [100] ↑SIRT1 and autophagy [100] Resveratrol in Drosophila [101] ↓behavioral deficits and brain histopathology ↑survival rate and life-span ↓H2O2 and nitric oxide (NO) ↑GST and CAT |
| 6-OHDA [102,103] | Resveratrol in SK-N-BE cells [102] ↓cytotoxicity ↓ROS ↑SIRT1-dependent autophagy | Resveratrol in rats [103] ↓behavioral alterations induced by apomorphine-and 6-OHDA ↓ultrastructural alterations: chromatin condensation and clumping, mitochondrial tumefaction, and vacuolization ↓COX-2 and TNF-α |
| Rotenone [104,105,106,107,108,109] | Resveratrol in SH-SY5Y and PC12 cells [104,105,106,107] ↓cytotoxicity and mitochondrial damage [104,105,106,107] ↓ROS and apoptosis [104,105,106] ↓histone-associated DNA fragmentation [107] ↓α-syn aggregation [107] ↓cleaved Poly ADP-ribose Polymerase (PARP) [107] ↑p-ERK1/2/ERK1/2 ratio [104,105] ↑autophagy [104,105,106,107] ↑Heme Oxygenase-1-dependent autophagy [104,105] ↑SIRT1 pathway and autophagy [106,107] | Nanoparticle-loaded [108] and free resveratrol [109] in rats ↓Parkinsonian motor dysfunction [108,109] and nigral histopathology [108] ↓striatal DA depletion [109] ↓lipid peroxidation, MDA [108] ↓ER stress markers CHOP and GRP78 [109] ↓caspase 3 activity, IL-1β level, protein carbonyl content [109] ↑SDH, citrate synthase, aconitase, and mitochondrial complex I activity [108] ↑antioxidant GSH, CAT, GPx [108,109] ↑Nrf2 DNA-binding activity [109] |
| α-Syn mutation [102,110] | Resveratrol in SK-N-BE cells [102] ↓A30P α-syn-induced cytotoxicity ↑SIRT1-dependent autophagy | Resveratrol in mice [110] ↓A53T α-syn-induced neurotoxicity ↓motor and cognitive deficits ↓total α-syn and oligomers, α-syn aggregation ↓neuroinflammation and oxidative stress |
| PINK1 mutation [111] | Resveratrol in PINK1 mutated Drosophila [111] ↑lifespan, locomotor activity, and muscle ATP production ↓DA neuron loss and abnormal wing posture ↓mitochondrial aggregates ↑autophagy and mitophagy |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Limanaqi, F.; Biagioni, F.; Busceti, C.L.; Ryskalin, L.; Polzella, M.; Frati, A.; Fornai, F. Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism. Int. J. Mol. Sci. 2019, 20, 3274. https://doi.org/10.3390/ijms20133274
Limanaqi F, Biagioni F, Busceti CL, Ryskalin L, Polzella M, Frati A, Fornai F. Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism. International Journal of Molecular Sciences. 2019; 20(13):3274. https://doi.org/10.3390/ijms20133274
Chicago/Turabian StyleLimanaqi, Fiona, Francesca Biagioni, Carla Letizia Busceti, Larisa Ryskalin, Maico Polzella, Alessandro Frati, and Francesco Fornai. 2019. "Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism" International Journal of Molecular Sciences 20, no. 13: 3274. https://doi.org/10.3390/ijms20133274
APA StyleLimanaqi, F., Biagioni, F., Busceti, C. L., Ryskalin, L., Polzella, M., Frati, A., & Fornai, F. (2019). Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism. International Journal of Molecular Sciences, 20(13), 3274. https://doi.org/10.3390/ijms20133274






