Amelioration of Behavioral Impairments and Neuropathology by Antiepileptic Drug Topiramate in a Transgenic Alzheimer’s Disease Model Mice, APP/PS1
Abstract
1. Introduction
2. Results
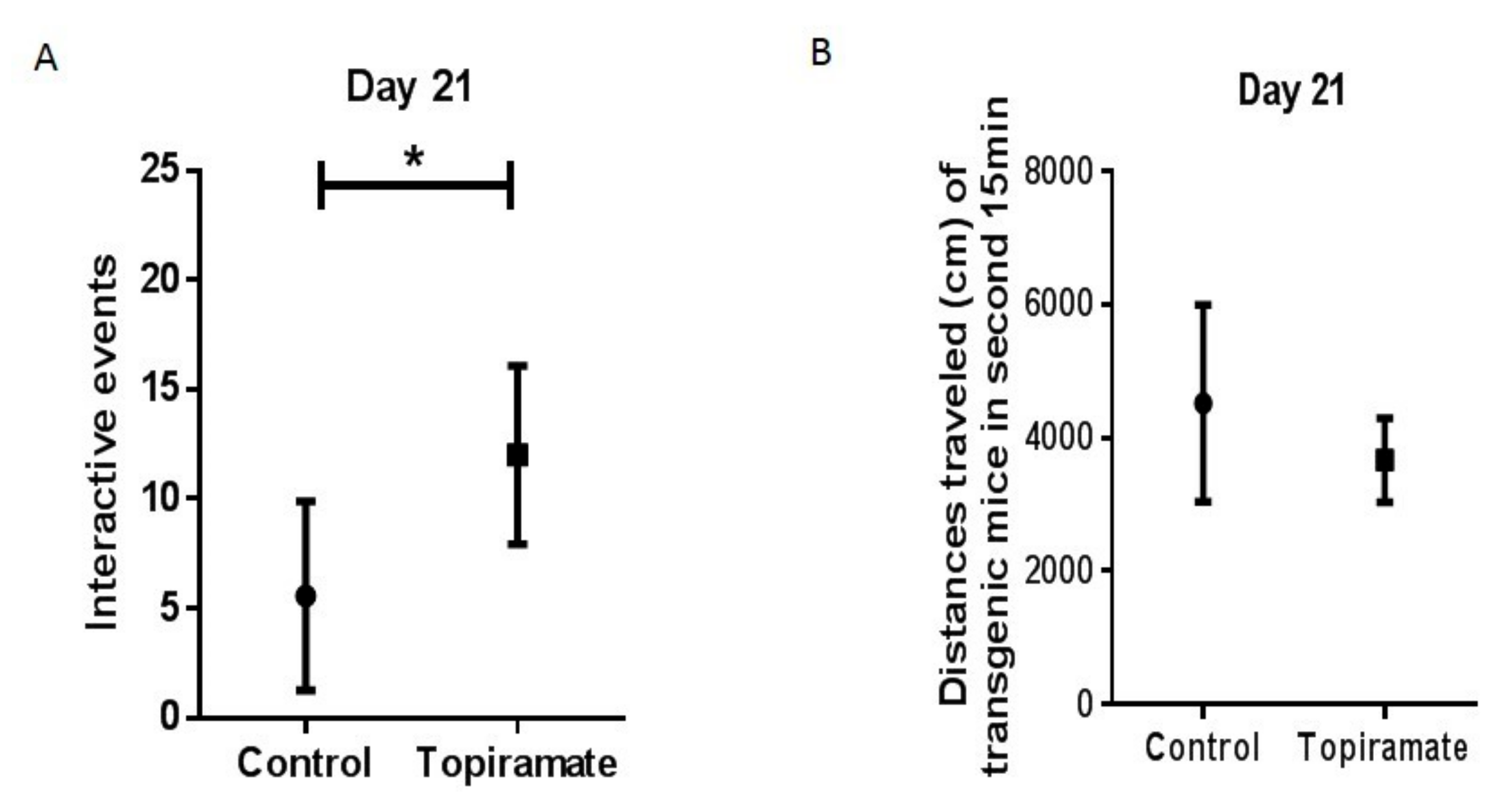
2.1. Effect of Topiramate Treatment on Social Interaction
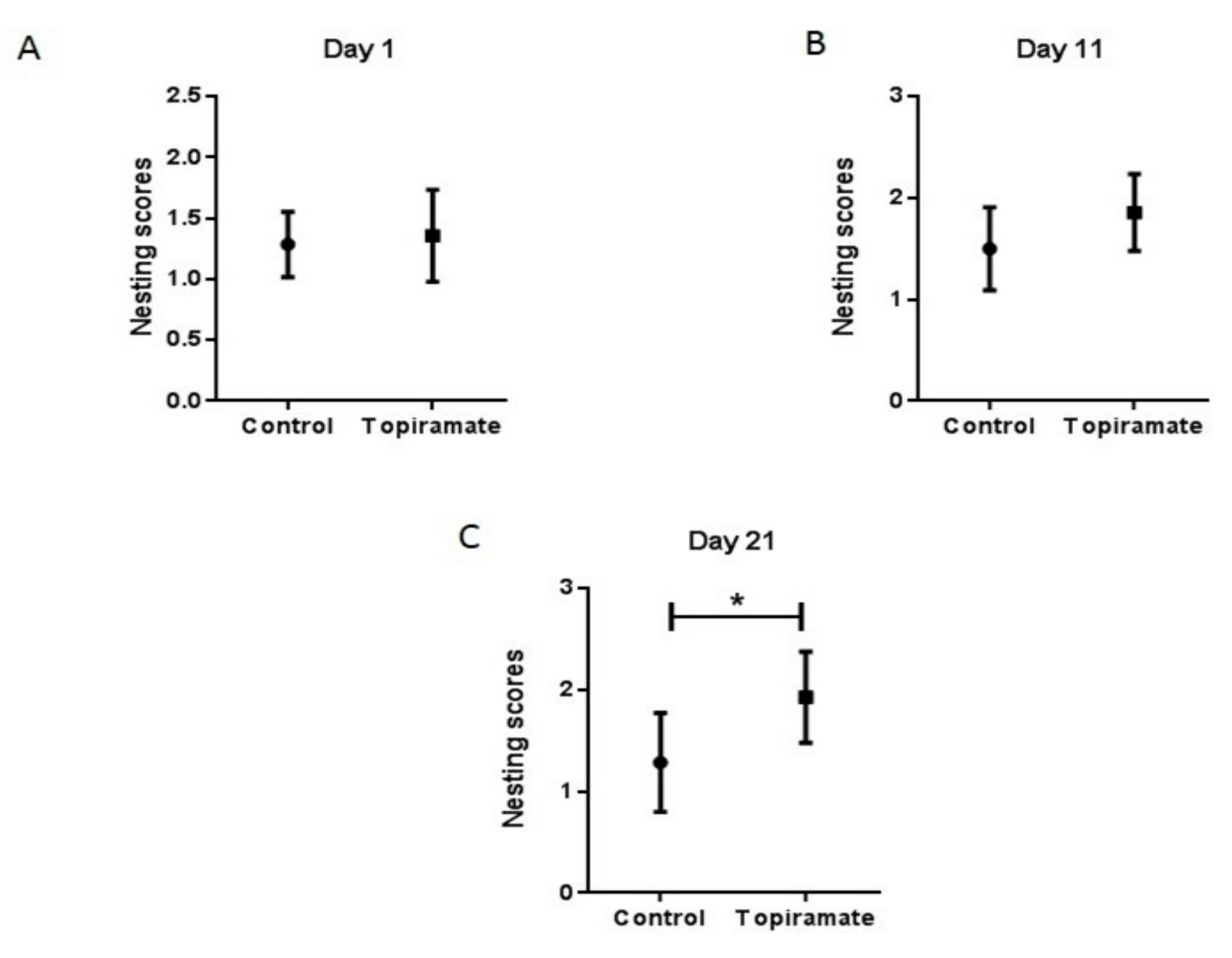
2.2. Effect of Topiramate Treatment on Behavioral Impairment (Nest Construction Assay)
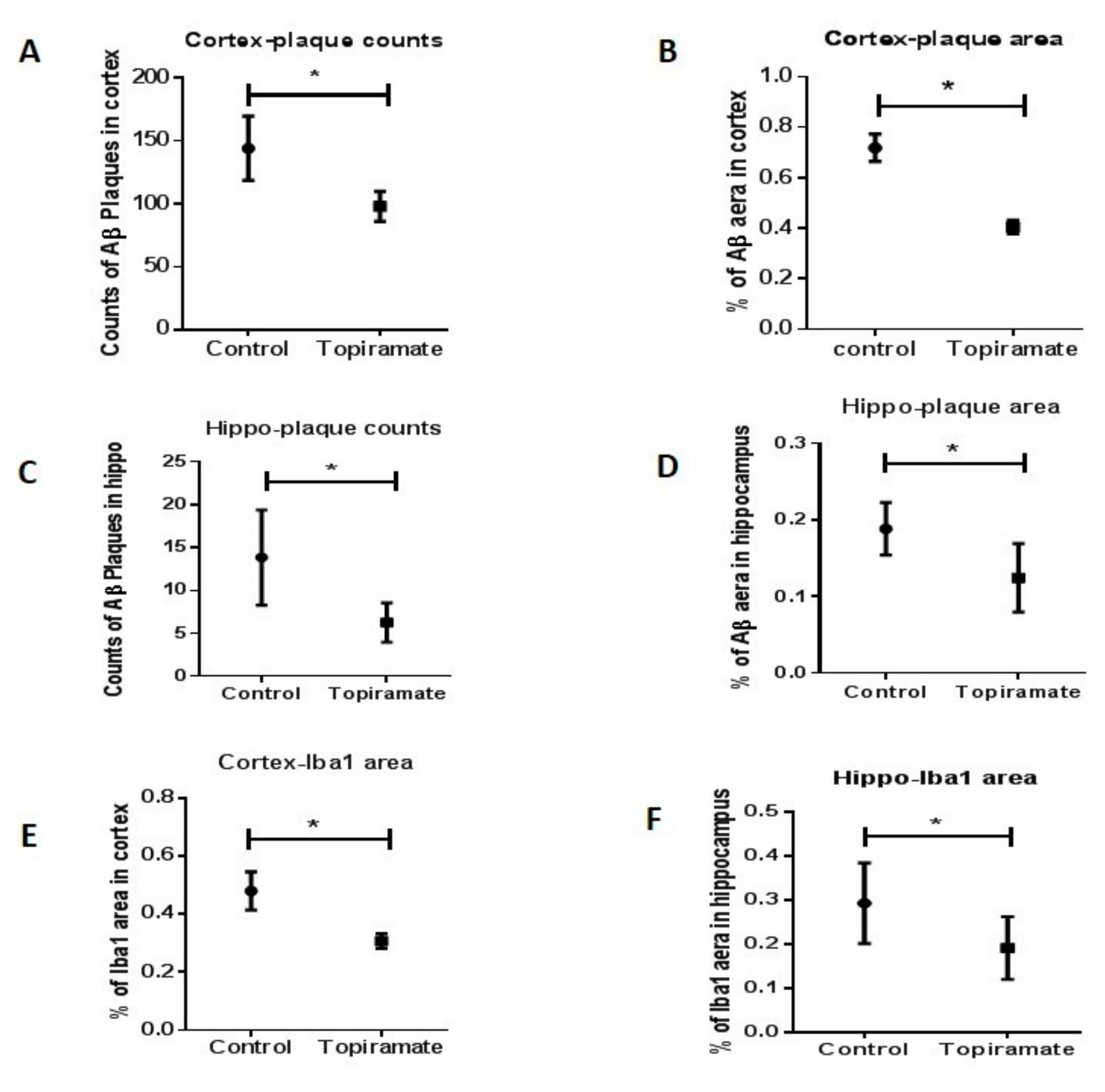
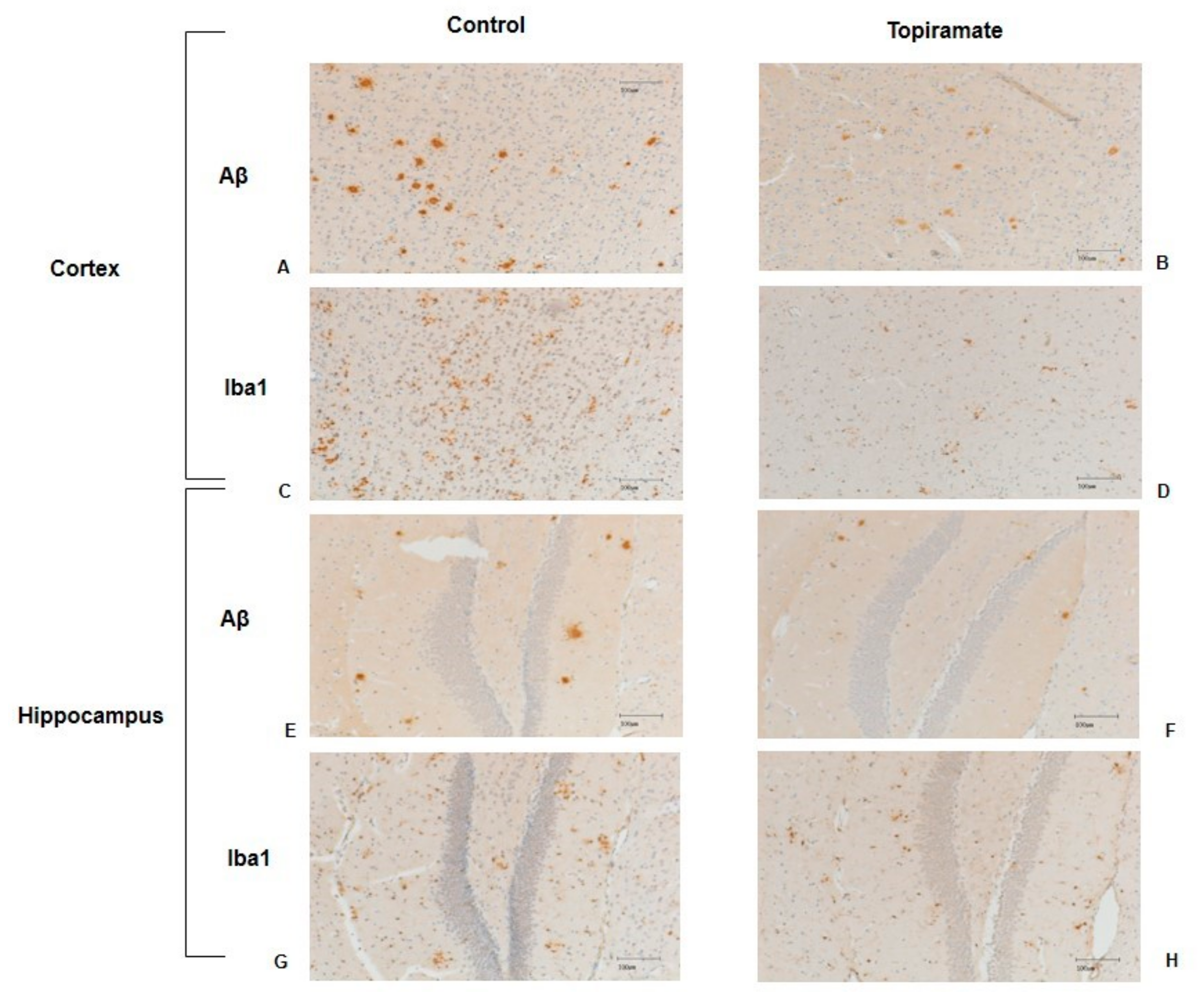
2.3. Effects of Topiramate on Neuroinflammation and Amyloidosis
3. Discussion
4. Materials and Methods
4.1. Animals
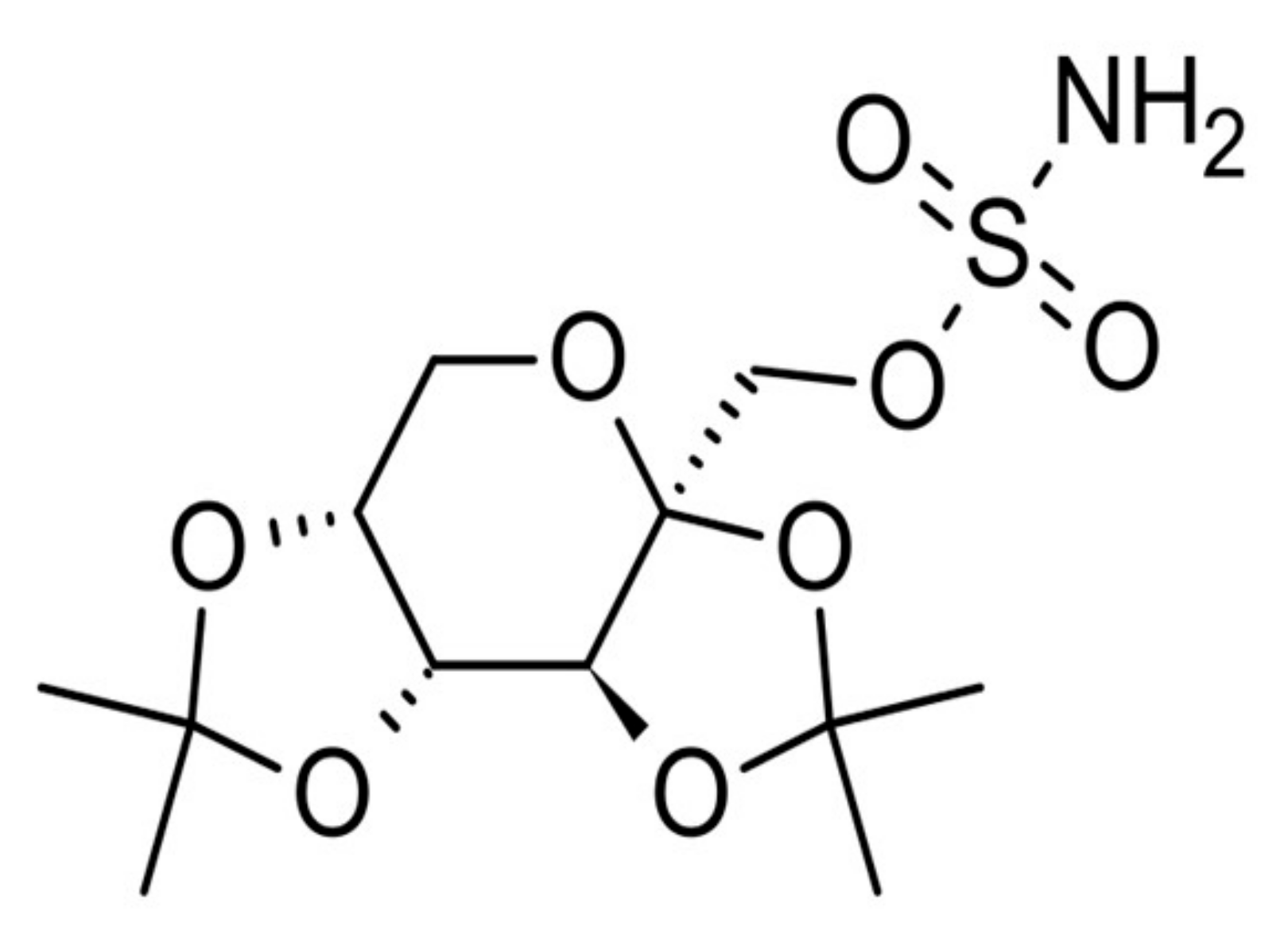
4.2. Materials
4.3. Treatment with Topiramate
4.4. Social Interaction Assay
4.5. Design and Evaluation of Nest Construction Assay
4.6. Immunohistochemistry and Image Evaluation/Analysis
4.7. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Schreiner, B.; Hedskog, L.; Wiehager, B.; Ankarcrona, M. Amyloid-β peptides are generated in mitochondria-associated endoplasmic reticulum membranes. J. Alzheimer’s Dis. 2015, 43, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Li, W.; Luo, J.; Lee, N.T.K.; Li, M.; Tsim, K.W.K.; Pang, Y.; Youdim, M.B.H.; Han, Y. Promising anti-Alzheimer’s dimer bis(7)-tacrine reduces beta-amyloid generation by directly inhibiting BACE-1 activity. Biochem. Biophys. Res. Commun. 2008, 366, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Imfeld, P.; Bodmer, M.; Schuerch, M.; Jick, S.S.; Meier, C.R. Seizures in patients with Alzheimer’s disease or vascular dementia: A population-based nested case-control analysis. Epilepsia 2013, 54, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Mendez, M.F.; Catanzaro, P.; Doss, R.C.; Arguello, R.; Frey, W.H. Seizures in Alzheimer’s Disease: Clinicopathologic Study. J. Geriatr. Psychiatry Neurol. 1994, 7, 230–233. [Google Scholar] [CrossRef]
- Qing, H.; He, G.; Ly, P.T.T.; Fox, C.J.; Staufenbiel, M.; Cai, F.; Zhang, Z.; Wei, S.; Sun, X.; Chen, C.-H.; et al. Valproic acid inhibits Abeta production, neuritic plaque formation, and behavioral deficits in Alzheimer’s disease mouse models. J. Exp. Med. 2008, 205, 2781–2789. [Google Scholar] [CrossRef] [PubMed]
- Boison, D. The adenosine kinase hypothesis of epileptogenesis. Prog. Neurobiol. 2008, 84, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Spiro, A.S.; Jenner, A.M.; Garner, B.; Karl, T. Long-Term Cannabidiol Treatment Prevents the Development of Social Recognition Memory Deficits in Alzheimer’s Disease Transgenic Mice. J. Alzheimer’s Dis. Jad. 2014, 42, 1383–1396. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Y.; Daniels, R.; Schluesener, H.J. Oridonin ameliorates neuropathological changes and behavioural deficits in a mouse model of cerebral amyloidosis. J. Cell. Mol. Med. 2013, 17, 1566–1576. [Google Scholar] [CrossRef]
- Colonna, M.; Wang, Y. TREM2 variants: New keys to decipher Alzheimer disease pathogenesis. Nat. Rev. Neurosci. 2016, 17, 201–207. [Google Scholar] [CrossRef]
- Starkstein, S.E.; Jorge, R.; Mizrahi, R.; Robinson, R.G. The Construct of Minor and Major Depression in Alzheimer’s Disease. Am. J. Psychiatry 2005, 162, 2086–2093. [Google Scholar] [CrossRef]
- Holcomb, L.; Gordon, M.N.; McGowan, E.; Yu, X.; Benkovic, S.; Jantzen, P.; Wright, K.; Saad, I.; Mueller, R.; Morgan, D.; et al. Accelerated Alzheimer-type phenotype in transgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgenes. Nat. Med. 1998, 4, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Bromley-Brits, K.; Deng, Y.; Song, W. Morris water maze test for learning and memory deficits in Alzheimer’s disease model mice. J. Vis. Exp. Jove. 2011, 53, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Filali, M.; Lalonde, R.; Rivest, S. Cognitive and non-cognitive behaviors in an APPswe/PS1 bigenic model of Alzheimer’s disease. Genes Brain Behav. 2009, 8, 143–148. [Google Scholar] [CrossRef] [PubMed]
- García-Mesa, Y.; López-Ramos, J.C.; Giménez-Llort, L.; Revilla, S.; Guerra, R.; Gruart, A.; Laferla, F.M.; Cristòfol, R.; Delgado-García, J.M.; Sanfeliu, C. Physical exercise protects against Alzheimer’s disease in 3xTg-AD mice. J. Alzheimer’s Dis. 2011, 24, 421–454. [Google Scholar] [CrossRef] [PubMed]
- Torres-Lista, V.; Giménez-Llort, L. Impairment of nesting behaviour in 3xTg-AD mice. Behav. Brain Res. 2013, 247, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Kranzler, H.R.; Armeli, S.; Feinn, R.; Tennen, H.; Gelernter, J.; Covault, J. GRIK1 genotype moderates topiramate’s effects on daily drinking level, expectations of alcohol’s positive effects and desire to drink. Int. J. Neuropsychopharmacol. 2014, 17, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.-L.; Li, M.-K. Effect of topiramate on apoptosis-related protein expression of hippocampus in model rats with Alzheimers disease. Eur. Rev. Med Pharmacol. Sci. 2014, 18, 761–768. [Google Scholar] [PubMed]
- Katz, I.R.; Jeste, D.V.; Mintzer, J.E.; Clyde, C.; Napolitano, J.; Brecher, M. Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: A randomized, double-blind trial. J. Clin. Psychiatry 1999, 60, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Okuyaz, C.; Kursel, O.; Komur, M.; Tamer, L. Evaluation of appetite-stimulating hormones in prepubertal children with epilepsy during topiramate treatment. Pediatric Neurol. 2012, 47, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.Q.; Wang, B.R.; Tian, Y.Y.; Xu, J.; Gao, L.; Zhao, S.L.; Jiang, T.; Xie, H.G.; Zhang, Y.D. Antiepileptics topiramate and levetiracetam alleviate behavioral deficits and reduce neuropathology in APPswe/PS1dE9 transgenic mice. Cns Neurosci. Ther. 2013, 19, 871–881. [Google Scholar] [CrossRef]
- Pinheiro, D. Anticonvulsant mood stabilizers in the treatment of behavioral and psychological symptoms of dementia (BPSD). L’Encéphale 2008, 34, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.A.; Ait-Daoud, N.; Bowden, C.L.; DiClemente, C.C.; Roache, J.D.; Lawson, K.; Javors, M.A.; Ma, J.Z. Oral topiramate for treatment of alcohol dependence: A randomised controlled trial. Lancet 2003, 361, 1677–1685. [Google Scholar] [CrossRef]
- Johnson, B.A.; Roache, J.D.; Ait-Daoud, N.; Wallace, C.; Wells, L.T.; Wang, Y. Effects of isradipine on methamphetamine-induced changes in attentional and perceptual-motor skills of cognition. Psychopharmacology 2005, 178, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Umbricht, A.; DeFulio, A.; Winstanley, E.L.; Tompkins, D.A.; Peirce, J.; Mintzer, M.Z.; Strain, E.C.; Bigelow, G.E. Topiramate for cocaine dependence during methadone maintenance treatment: A randomized controlled trial. Drug Alcohol Depend. 2014, 140, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Villarejo, M.V.; López-Pedrajas, R.; Sánchez-Vallejo, V.; Genovés, J.M.; Muriach, M.; Barcia, J.M.; Romero, F.J.; Miranda, M. Chronic cocaine effects in retinal metabolism and electrophysiology: Treatment with topiramate. Curr. Eye Res. 2014, 39, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Appolinario, J.C.; Fontenelle, L.F.; Papelbaum, M.; Bueno, J.R.; Coutinho, W. Topiramate use in obese patients with binge eating disorder: An open study. Can. J. Psychiatry. 2002, 47, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Phelan, S. Long-term weight loss maintenance. Am. J. Clin. Nutr. 2005, 82, 222S–225S. [Google Scholar] [CrossRef] [PubMed]
- Artemenko, A.R.; Kurenkov, A.L.; Filatova, E.G.; Nikitin, S.S.; Kaube, H.; Katsarava, Z. Effects of topiramate on migraine frequency and cortical excitability in patients with frequent migraine. Cephalalgia Int. J. Headache 2008, 28, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Salisbury-Afshar, E. Topiramate for the prophylaxis of episodic migraine in adults. Am. Fam. Phys. 2014, 90, 24. [Google Scholar]
- Burton, L.A.; Harden, C. Effect of topiramate on attention. Epilepsy Res. 1997, 27, 29–32. [Google Scholar] [CrossRef]
- Medagama, A.; Senaratne, T.; Bandara, J.M.R.P.; Abeysekera, R.A.; Imbulpitiya, I.V.B. Topiramate-induced acute onset myopia: A case report. BMC Res. Notes 2014, 7, 665. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.Y.; Kim, C.H.; Kim, J.M.; Cheong, J.H.; Park, S.G. A muliticenter retrospective study of the actual using patterns and clinical effects of topiramate in patients with neurosurgical disease. J. Epilepsy Res. 2014, 4, 62–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mouallem, M.; Potashnik, Y. Beer potomania. Harefuah 2004, 143, 12–13, 87. [Google Scholar] [PubMed]
- Rigoulot, M.A. Neuroprotective Properties of Topiramate in the Lithium-Pilocarpine Model of Epilepsy. J. Pharmacol. Exp. Ther. 2004, 308, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; O’Banion, M.K. Inflammatory processes in Alzheimer’s disease. J. Neuroimmunol. 2007, 184, 69–91. [Google Scholar] [CrossRef] [PubMed]
- Wesson, D.W.; Wilson, D.A. Age and gene overexpression interact to abolish nesting behavior in Tg2576 amyloid precursor protein (APP) mice. Behav. Brain Res. 2011, 216, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zug, C.; Qu, H.; Schluesener, H.; Zhang, Z. Hesperidin ameliorates behavioral impairments and neuropathology of transgenic APP/PS1 mice. Behav. Brain Res. 2015, 281, 32–42. [Google Scholar] [CrossRef]
- Hsiao, Y.H.; Chen, P.S.; Chen, S.H.; Gean, P.W. The involvement of Cdk5 activator p35 in social isolation-triggered onset of early alzheimers disease-related cognitive deficit in the transgenic mice. Neuropsychopharmacology 2011, 36, 1848–1858. [Google Scholar] [CrossRef]
- Pietropaolo, S.; Delage, P.; Lebreton, F.; Crusio, W.E.; Cho, Y.H. Early development of social deficits in APP and APP-PS1 mice. Neurobiol. Aging 2012, 33, e17–e27. [Google Scholar] [CrossRef]
- Eyal, S.; Yagen, B.; Sobol, E.; Altschuler, Y.; Shmuel, M.; Bialer, M. The activity of antiepileptic drugs as histone deacetylase inhibitors. Epilepsia 2004, 45, 737–744. [Google Scholar] [CrossRef]
- Kudin, A.P.; Debska-Vielhaber, G.; Vielhaber, S.; Elger, C.E.; Kunz, W.S. The mechanism of neuroprotection by topiramate in an animal model of epilepsy. Epilepsia 2004, 45, 1478–1487. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Owona, B.A.; Zug, C.; Schluesener, H.J.; Zhang, Z.-Y. Amelioration of Behavioral Impairments and Neuropathology by Antiepileptic Drug Topiramate in a Transgenic Alzheimer’s Disease Model Mice, APP/PS1. Int. J. Mol. Sci. 2019, 20, 3003. https://doi.org/10.3390/ijms20123003
Owona BA, Zug C, Schluesener HJ, Zhang Z-Y. Amelioration of Behavioral Impairments and Neuropathology by Antiepileptic Drug Topiramate in a Transgenic Alzheimer’s Disease Model Mice, APP/PS1. International Journal of Molecular Sciences. 2019; 20(12):3003. https://doi.org/10.3390/ijms20123003
Chicago/Turabian StyleOwona, Brice Ayissi, Caroline Zug, Hermann J. Schluesener, and Zhi-Yuan Zhang. 2019. "Amelioration of Behavioral Impairments and Neuropathology by Antiepileptic Drug Topiramate in a Transgenic Alzheimer’s Disease Model Mice, APP/PS1" International Journal of Molecular Sciences 20, no. 12: 3003. https://doi.org/10.3390/ijms20123003
APA StyleOwona, B. A., Zug, C., Schluesener, H. J., & Zhang, Z.-Y. (2019). Amelioration of Behavioral Impairments and Neuropathology by Antiepileptic Drug Topiramate in a Transgenic Alzheimer’s Disease Model Mice, APP/PS1. International Journal of Molecular Sciences, 20(12), 3003. https://doi.org/10.3390/ijms20123003



