Genome Editing in Agriculture: Technical and Practical Considerations
Abstract
1. Introduction
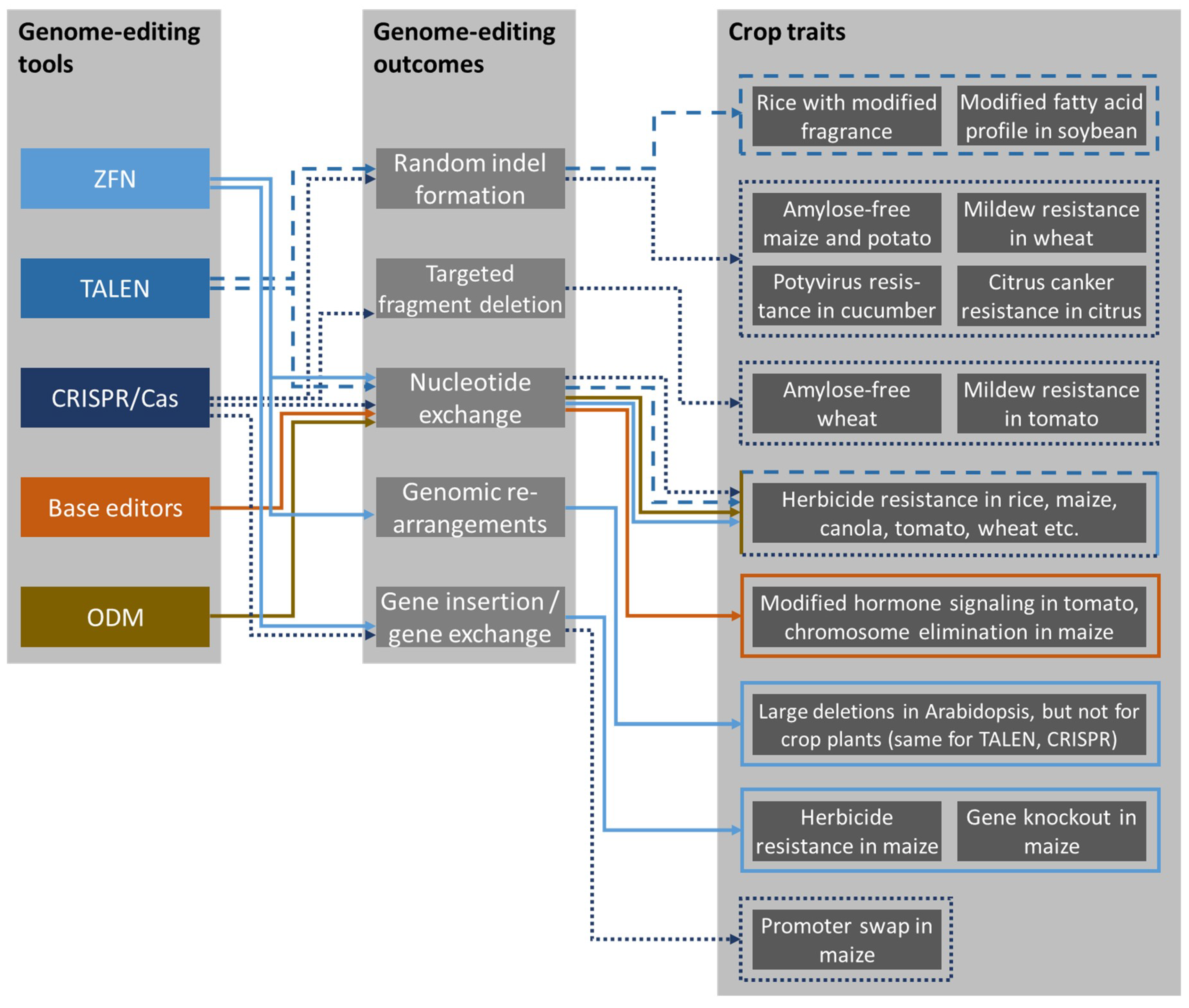
2. Genome-Editing Tools
2.1. Programmable Sequence-Specific Nucleases
2.2. Base Editors
2.3. Oligonucleotide-Directed Mutagenesis
3. Outcomes of Genome Editing
3.1. Random Indel Formation
3.2. Targeted Fragment Deletion
3.3. Targeted Nucleotide Exchange
3.4. Genomic Rearrangements
3.5. Gene Insertion and Gene Exchange
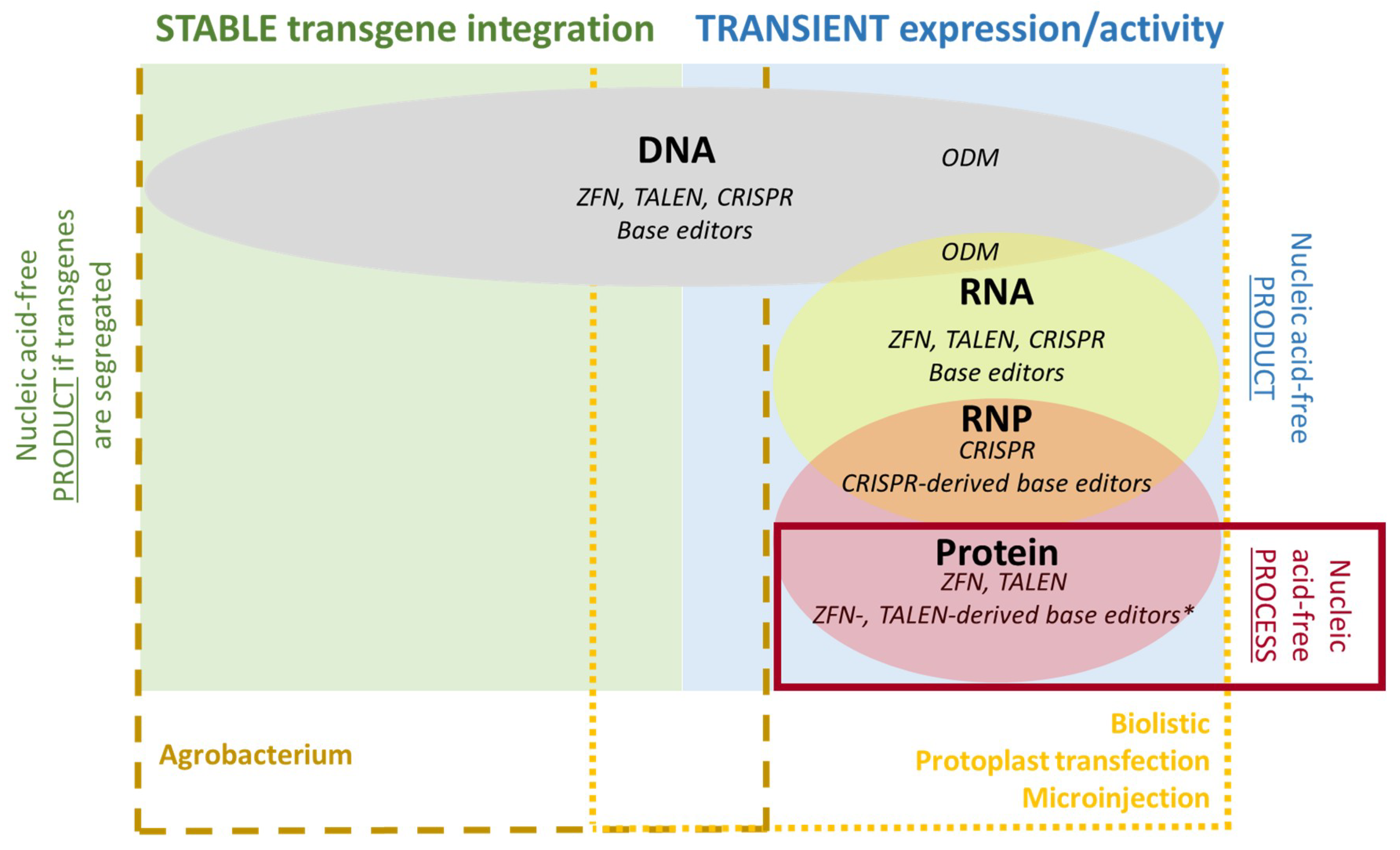
4. Delivery Methods
4.1. DNA
4.1.1. Agrobacterium-Mediated Transformation
4.1.2. Particle Bombardment
4.1.3. Protoplast Transfection
4.1.4. Electroporation
4.1.5. Other Delivery Methods
4.2. RNA
4.2.1. Particle Bombardment
4.2.2. Protoplast Transfection
4.3. Proteins and RNPs
4.3.1. Particle Bombardment
4.3.2. Protoplast Transfection
5. Selection
5.1. Selectable Marker Genes
5.2. Visual/Screenable Phenotype
5.3. High-Throughput Screening of Edited Lines
6. Regeneration
6.1. Suitable Tissues
6.2. Timelines
7. Regulatory Aspects of Plant Genome Editing
8. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hiom, K. Coping with DNA double strand breaks. DNA Repair 2010, 9, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Puchta, H. The repair of double-strand breaks in plants: Mechanisms and consequences for genome evolution. J. Exp. Bot. 2005, 56, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Gorbunova, L. How plants make ends meet: DNA double-strand break repair. Trends Plant Sci. 1999, 4, 263–269. [Google Scholar] [CrossRef]
- Sander, J.D.; Dahlborg, E.J.; Goodwin, M.J.; Cade, L.; Zhang, F.; Cifuentes, D.; Curtin, S.J.; Blackburn, J.S.; Thibodeau-Beganny, S.; Qi, Y.; et al. Selection-free zinc-finger-nuclease engineering by context-dependent assembly (CoDA). Nat. Method. 2011, 8, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Guilinger, J.P.; Pattanayak, V.; Reyon, D.; Tsai, S.Q.; Sander, J.D.; Joung, J.K.; Liu, D.R. Broad specificity profiling of TALENs results in engineered nucleases with improved DNA-cleavage specificity. Nat. Method. 2014, 11, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Doyon, Y.; Vo, T.D.; Mendel, M.C.; Greenberg, S.G.; Wang, J.; Xia, D.F.; Miller, J.C.; Urnov, F.D.; Gregory, P.D.; Holmes, M.C. Enhancing zinc-finger-nuclease activity with improved obligate heterodimeric architectures. Nat. Method. 2011, 8, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.C.; Holmes, M.C.; Wang, J.; Guschin, D.Y.; Lee, Y.L.; Rupniewski, I.; Beausejour, C.M.; Waite, A.J.; Wang, N.S.; Kim, K.A.; et al. An improved zinc-finger nuclease architecture for highly specific genome editing. Nat. Biotechnol. 2007, 25, 778–785. [Google Scholar] [CrossRef]
- Szczepek, M.; Brondani, V.; Buchel, J.; Serrano, L.; Segal, D.J.; Cathomen, T. Structure-based redesign of the dimerization interface reduces the toxicity of zinc-finger nucleases. Nat. Biotechnol. 2007, 25, 786–793. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, H.J.; Kim, H.; Cho, S.W.; Kim, J.S. Targeted genome editing in human cells with zinc finger nucleases constructed via modular assembly. Genome Res. 2009, 19, 1279–1288. [Google Scholar] [CrossRef]
- Cermak, T.; Doyle, E.L.; Christian, M.; Wang, L.; Zhang, Y.; Schmidt, C.; Baller, J.A.; Somia, N.V.; Bogdanove, A.J.; Voytas, D.F. Efficient design and assembly of custom TALEN and other TAL effector-based constructs for DNA targeting. Nucleic Acids Res. 2011, 39, e82. [Google Scholar] [CrossRef]
- Bibikova, M.; Carroll, D.; Segal, D.J.; Trautman, J.K.; Smith, J.; Kim, Y.-G.; Chandrasegaran, S. Stimulation of Homologous Recombination through Targeted Cleavage by Chimeric Nucleases. Mol. Cell Biol. 2001, 21, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Bortesi, L.; Baysal, C.; Twyman, R.M.; Fischer, R.; Capell, T.; Schillberg, S.; Christou, P. Characteristics of Genome Editing Mutations in Cereal Crops. Trends Plant Sci. 2017, 22, 38–52. [Google Scholar] [CrossRef] [PubMed]
- D’Halluin, K.; Vanderstraeten, C.; Stals, E.; Cornelissen, M.; Ruiter, R. Homologous recombination: A basis for targeted genome optimization in crop species such as maize. Plant Biotechnol. J. 2008, 6, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kweon, J.; Kim, J.S. TALENs and ZFNs are associated with different mutation signatures. Nat. Method. 2013, 10, 185. [Google Scholar] [CrossRef] [PubMed]
- Jaganathan, D.; Ramasamy, K.; Sellamuthu, G.; Jayabalan, S.; Venkataraman, G. CRISPR for Crop Improvement: An Update Review. Front Plant Sci. 2018, 9, 985. [Google Scholar] [CrossRef] [PubMed]
- Shukla, V.K.; Doyon, Y.; Miller, J.C.; Dekelver, R.C.; Moehle, E.A.; Worden, S.E.; Mitchell, J.C.; Arnold, N.L.; Gopalan, S.; Meng, X.; et al. Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 2009, 459, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Smith, J.; Yang, M.; Jones, S.; Djukanovic, V.; Nicholson, M.G.; West, A.; Bidney, D.; Falco, S.C.; Jantz, D.; et al. Heritable targeted mutagenesis in maize using a designed endonuclease. Plant J. 2010, 61, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Char, S.N.; Unger-Wallace, E.; Frame, B.; Briggs, S.A.; Main, M.; Spalding, M.H.; Vollbrecht, E.; Wang, K.; Yang, B. Heritable site-specific mutagenesis using TALENs in maize. Plant Biotechnol. J. 2015, 13, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Mussolino, C.; Morbitzer, R.; Lutge, F.; Dannemann, N.; Lahaye, T.; Cathomen, T. A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res. 2011, 39, 9283–9293. [Google Scholar] [CrossRef]
- Zischewski, J.; Fischer, R.; Bortesi, L. Detection of on-target and off-target mutations generated by CRISPR/Cas9 and other sequence-specific nucleases. Biotechnol. Adv. 2017, 35, 95–104. [Google Scholar] [CrossRef]
- Kannan, B.; Jung, J.H.; Moxley, G.W.; Lee, S.M.; Altpeter, F. TALEN-mediated targeted mutagenesis of more than 100 COMT copies/alleles in highly polyploid sugarcane improves saccharification efficiency without compromising biomass yield. Plant Biotechnol. J. 2018, 16, 856–866. [Google Scholar] [CrossRef] [PubMed]
- Bortesi, L.; Fischer, R. The CRISPR/Cas9 system for plant genome editing and beyond. Biotechnol. Adv. 2015, 33, 41–52. [Google Scholar] [CrossRef]
- Chen, K.; Gao, C. Developing CRISPR Technology in Major Crop Plants. In Advances in New Technology for Targeted Modification of Plant Genomes; Zhang, F., Puchta, H., Thomson, J.G., Eds.; Springer: New York, NY, USA, 2015; pp. 145–159. [Google Scholar]
- Gasiunas, G.; Barrangou, R.; Horvath, P.; Siksnys, V. Cas9–crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc. Natl. Acad. Sci. USA 2012, 109, E2579–E2586. [Google Scholar] [CrossRef] [PubMed]
- Nishimasu, H.; Ran, F.A.; Hsu, P.D.; Konermann, S.; Shehata, S.I.; Dohmae, N.; Ishitani, R.; Zhang, F.; Nureki, O. Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell 2014, 156, 935–949. [Google Scholar] [CrossRef] [PubMed]
- Xie, K.; Zhang, J.; Yang, Y. Genome-wide prediction of highly specific guide RNA spacers for CRISPR-Cas9-mediated genome editing in model plants and major crops. Mol. Plant. 2014, 7, 923–926. [Google Scholar] [CrossRef]
- Friedland, A.E.; Baral, R.; Singhal, P.; Loveluck, K.; Shen, S.; Sanchez, M.; Marco, E.; Gotta, G.M.; Maeder, M.L.; Kennedy, E.M.; et al. Characterization of Staphylococcus aureus Cas9: A smaller Cas9 for all-in-one adeno-associated virus delivery and paired nickase applications. Genome Biol. 2015, 16, 257. [Google Scholar] [CrossRef]
- Hu, X.; Wang, C.; Fu, Y.; Liu, Q.; Jiao, X.; Wang, K. Expanding the Range of CRISPR/Cas9 Genome Editing in Rice. Mol. Plant. 2016, 9, 943–945. [Google Scholar] [CrossRef]
- Mali, P.; Yang, L.; Esvelt, K.M.; Aach, J.; Guell, M.; DiCarlo, J.E.; Norville, J.E.; Church, G.M. RNA-guided human genome engineering via Cas9. Science 2013, 339, 823–826. [Google Scholar] [CrossRef]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; et al. Multiplex Genome Engineering Using CRISPR/Cas Systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef]
- Vazquez-Vilar, M.; Bernabé-Orts, J.M.; Fernandez-Del-Carmen, A.; Ziarsolo, P.; Blanca, J.; Granell, A.; Orzaez, D. A modular toolbox for gRNA-Cas9 genome engineering in plants based on the GoldenBraid standard. Plant Method. 2016, 12, 10. [Google Scholar] [CrossRef]
- Xing, H.-L.; Dong, L.; Wang, Z.-P.; Zhang, H.-Y.; Han, C.-Y.; Liu, B.; Wang, X.-C.; Chen, Q.-J. A CRISPR/Cas9 toolkit for multiplex genome editing in plants. BMC Plant Biol. 2014, 14, 327. [Google Scholar] [CrossRef] [PubMed]
- Xie, K.; Minkenberg, B.; Yang, Y. Boosting CRISPR/Cas9 multiplex editing capability with the endogenous tRNA-processing system. Proc. Nat. Acad. Sci. USA 2015, 112, 3570–3575. [Google Scholar] [CrossRef] [PubMed]
- Bortesi, L.; Zhu, C.; Zischewski, J.; Perez, L.; Bassie, L.; Nadi, R.; Forni, G.; Lade, S.B.; Soto, E.; Jin, X.; et al. Patterns of CRISPR/Cas9 activity in plants, animals and microbes. Plant Biotechnol. J. 2016, 14, 2203–2216. [Google Scholar] [CrossRef] [PubMed]
- Brooks, C.; Nekrasov, V.; Lippman, Z.B.; Van Eck, J. Efficient gene editing in tomato in the first generation using the clustered regularly interspaced short palindromic repeats/CRISPR-associated9 system. Plant Physiol. 2014, 166, 1292–1297. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.S.; Mahfouz, M.M.; Mansoor, S. CRISPR-Cpf1: A New Tool for Plant Genome Editing. Trends Plant Sci. 2017, 22, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Zetsche, B.; Gootenberg, J.S.; Abudayyeh, O.O.; Slaymaker, I.M.; Makarova, K.S.; Essletzbichler, P.; Volz, S.E.; Joung, J.; van der Oost, J.; Regev, A.; et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 2015, 163, 759–771. [Google Scholar] [CrossRef]
- Tang, X.; Lowder, L.G.; Zhang, T.; Malzahn, A.A.; Zheng, X.; Voytas, D.F.; Zhong, Z.; Chen, Y.; Ren, Q.; Li, Q.; et al. A CRISPR-Cpf1 system for efficient genome editing and transcriptional repression in plants. Nat. Plants 2017, 3, 17018. [Google Scholar] [CrossRef] [PubMed]
- Malzahn, A.A.; Tang, X.; Lee, K.; Ren, Q.; Sretenovic, S.; Zhang, Y.; Chen, H.; Kang, M.; Bao, Y.; Zheng, X.; et al. Application of CRISPR-Cas12a temperature sensitivity for improved genome editing in rice, maize, and Arabidopsis. BMC Biol. 2019, 17, 9. [Google Scholar] [CrossRef]
- Feng, Z.; Mao, Y.; Xu, N.; Zhang, B.; Wei, P.; Yang, D.L.; Wang, Z.; Zhang, Z.; Zheng, R.; Yang, L.; et al. Multigeneration analysis reveals the inheritance, specificity, and patterns of CRISPR/Cas-induced gene modifications in Arabidopsis. Proc. Natl. Acad. Sci. USA 2014, 111, 4632–4637. [Google Scholar] [CrossRef]
- Hahn, F.; Nekrasov, V. CRISPR/Cas precision: Do we need to worry about off-targeting in plants? Plant Cell Rep. 2018. [Google Scholar] [CrossRef]
- Minkenberg, B.; Zhang, J.; Xie, K.; Yang, Y. CRISPR-PLANT v2: An online resource for highly specific guide RNA spacers based on improved off-target analysis. Plant Biotechnol. J. 2019, 17, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Komor, A.C.; Kim, Y.B.; Packer, M.S.; Zuris, J.A.; Liu, D.R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 2016, 533, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Zhu, J.K. Precise Editing of a Target Base in the Rice Genome Using a Modified CRISPR/Cas9 System. Mol. Plant. 2017, 10, 523–525. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, J.M.; Cervantes, O.; Clement, M.K.; Wu, Y.; Zeng, J.; Bauer, D.E.; Pinello, L.; Joung, J.K. An APOBEC3A-Cas9 base editor with minimized bystander and off-target activities. Nat. Biotechnol. 2018, 36, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Zhang, F.; Karcher, D.; Bock, R. Engineering of high-precision base editors for site-specific single nucleotide replacement. Nat. Commun. 2019, 10, 439. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Sretenovic, S.; Ren, Q.; Yang, L.; Bao, Y.; Qi, C.; Yuan, M.; He, Y.; Liu, S.; Liu, X.; et al. Improving Plant Genome Editing with High-Fidelity xCas9 and Non-canonical PAM-Targeting Cas9-NG. Mol. Plant. 2019. [Google Scholar] [CrossRef]
- Hua, K.; Tao, X.; Han, P.; Wang, R.; Zhu, J.-K. Genome Engineering in Rice Using Cas9 Variants that Recognize NG PAM Sequences. Mol. Plant. 2019. [Google Scholar] [CrossRef]
- Gaudelli, N.M.; Komor, A.C.; Rees, H.A.; Packer, M.S.; Badran, A.H.; Bryson, D.I.; Liu, D.R. Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 2017, 551, 464–471. [Google Scholar] [CrossRef]
- Li, C.; Zong, Y.; Wang, Y.; Jin, S.; Zhang, D.; Song, Q.; Zhang, R.; Gao, C. Expanded base editing in rice and wheat using a Cas9-adenosine deaminase fusion. Genome. Biol. 2018, 19, 59. [Google Scholar] [CrossRef]
- Zuo, E.; Sun, Y.; Wei, W.; Yuan, T.; Ying, W.; Sun, H.; Yuan, L.; Steinmetz, L.M.; Li, Y.; Yang, H. Cytosine base editor generates substantial off-target single-nucleotide variants in mouse embryos. Science 2019. [Google Scholar] [CrossRef]
- Kang, B.C.; Yun, J.Y.; Kim, S.T.; Shin, Y.; Ryu, J.; Choi, M.; Woo, J.W.; Kim, J.S. Precision genome engineering through adenine base editing in plants. Nat. Plants 2018, 4, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Shimatani, Z.; Kashojiya, S.; Takayama, M.; Terada, R.; Arazoe, T.; Ishii, H.; Teramura, H.; Yamamoto, T.; Komatsu, H.; Miura, K.; et al. Targeted base editing in rice and tomato using a CRISPR-Cas9 cytidine deaminase fusion. Nat. Biotechnol. 2017, 35, 441–443. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Kuang, Y.; Ren, B.; Wang, J.; Zhang, D.; Lin, H.; Yang, B.; Zhou, X.; Zhou, H. Highly Efficient, A.T to G.C Base Editing by Cas9n-Guided tRNA Adenosine Deaminase in Rice. Mol. Plant 2018, 11, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Zong, Y.; Wang, Y.; Li, C.; Zhang, R.; Chen, K.; Ran, Y.; Qiu, J.L.; Wang, D.; Gao, C. Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion. Nat. Biotechnol. 2017, 35, 438–440. [Google Scholar] [CrossRef] [PubMed]
- Rees, H.A.; Wilson, C.; Doman, J.L.; Liu, D.R. Analysis and minimization of cellular RNA editing by DNA adenine base editors. Sci. Adv. 2019, 5, eaax5717. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lu, Z.; Yang, G.; Huang, S.; Li, G.; Feng, S.; Liu, Y.; Li, J.; Yu, W.; Zhang, Y.; et al. Efficient generation of mouse models of human diseases via ABE- and BE-mediated base editing. Nat. Commun. 2018, 9, 2338. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Yu, L.; Zhang, X.; Xin, C.; Huang, S.; Bai, L.; Chen, W.; Gao, R.; Li, J.; Pan, S.; et al. Highly efficient and precise base editing by engineered dCas9-guide tRNA adenosine deaminase in rats. Cell Discov. 2018, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Sauer, N.J.; Mozoruk, J.; Miller, R.B.; Warburg, Z.J.; Walker, K.A.; Beetham, P.R.; Schopke, C.R.; Gocal, G.F. Oligonucleotide-directed mutagenesis for precision gene editing. Plant Biotechnol. J. 2016, 14, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Ruiter, R.; van den Brande, I.; Stals, E.; Delaure, S.; Cornelissen, M.; D’Halluin, K. Spontaneous mutation frequency in plants obscures the effect of chimeraplasty. Plant Mol. Biol. 2003, 53, 675–689. [Google Scholar] [CrossRef]
- Sauer, N.J.; Narvaez-Vasquez, J.; Mozoruk, J.; Miller, R.B.; Warburg, Z.J.; Woodward, M.J.; Mihiret, Y.A.; Lincoln, T.A.; Segami, R.E.; Sanders, S.L.; et al. Oligonucleotide-Mediated Genome Editing Provides Precision and Function to Engineered Nucleases and Antibiotics in Plants. Plant Physiol. 2016, 170, 1917–1928. [Google Scholar] [CrossRef] [PubMed]
- Kertesz, S.; Kerenyi, Z.; Merai, Z.; Bartos, I.; Palfy, T.; Barta, E.; Silhavy, D. Both introns and long 3’-UTRs operate as cis-acting elements to trigger nonsense-mediated decay in plants. Nucleic. Acids Res. 2006, 34, 6147–6157. [Google Scholar] [CrossRef] [PubMed]
- Shan, Q.; Zhang, Y.; Chen, K.; Zhang, K.; Gao, C. Creation of fragrant rice by targeted knockout of the OsBADH2 gene using TALEN technology. Plant Biotechnol. J. 2015. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M. US gives food manufacturers three years to ban trans fats. Brit. Med. J. 2015, 350, h3315. [Google Scholar] [CrossRef] [PubMed]
- Brownell, K.D.; Pomeranz, J.L. The Trans-Fat Ban—Food Regulation and Long-Term Health. N. Engl. J. Med. 2014, 370, 1773–1775. [Google Scholar] [CrossRef] [PubMed]
- Demorest, Z.L.; Coffman, A.; Baltes, N.J.; Stoddard, T.J.; Clasen, B.M.; Luo, S.; Retterath, A.; Yabandith, A.; Gamo, M.E.; Bissen, J.; et al. Direct stacking of sequence-specific nuclease-induced mutations to produce high oleic and low linolenic soybean oil. BMC Plant Biol. 2016, 16, 225. [Google Scholar] [CrossRef] [PubMed]
- Haun, W.; Coffman, A.; Clasen, B.M.; Demorest, Z.L.; Lowy, A.; Ray, E.; Retterath, A.; Stoddard, T.; Juillerat, A.; Cedrone, F.; et al. Improved soybean oil quality by targeted mutagenesis of the fatty acid desaturase 2 gene family. Plant Biotechnol. J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Shure, M.; Wessler, S.; Fedoroff, N. Molecular identification and isolation of the Waxy locus in maize. Cell 1983, 35, 225–233. [Google Scholar] [CrossRef]
- Andersson, M.; Turesson, H.; Olsson, N.; Fält, A.-S.; Ohlsson, P.; Gonzalez Matías, N.; Samuelsson, M.; Hofvander, P. Genome editing in potato via CRISPR-Cas9 ribonucleoprotein delivery. Physiol. Plant 2018, 164, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cheng, X.; Shan, Q.; Zhang, Y.; Liu, J.; Gao, C.; Qiu, J.-L. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotech. 2014, 32, 947–951. [Google Scholar] [CrossRef]
- Chandrasekaran, J.; Brumin, M.; Wolf, D.; Leibman, D.; Klap, C.; Pearlsman, M.; Sherman, A.; Arazi, T.; Gal-On, A. Development of broad virus resistance in non-transgenic cucumber using CRISPR/Cas9 technology. Mol. Plant Pathol. 2016, 17, 1140–1153. [Google Scholar] [CrossRef]
- Peng, A.; Chen, S.; Lei, T.; Xu, L.; He, Y.; Wu, L.; Yao, L.; Zou, X. Engineering canker-resistant plants through CRISPR/Cas9-targeted editing of the susceptibility gene CsLOB1 promoter in citrus. Plant Biotechnol. J. 2017, 15, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Cigan, A.M.; Gadlage, M.J.; Gao, H.; Meeley, R.B.; Young, J.K. Waxy corn. WO2017132239A1, 3 August 2017. [Google Scholar]
- Nekrasov, V.; Wang, C.; Win, J.; Lanz, C.; Weigel, D.; Kamoun, S. Rapid generation of a transgene-free powdery mildew resistant tomato by genome deletion. Sci. Rep. 2017, 7, 482. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, B.; Chen, C.Y.; Yang, B. TALEN-Mediated Homologous Recombination Produces Site-Directed DNA Base Change and Herbicide-Resistant Rice. J. Genet. Genom. 2016, 43, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Ran, Y.; Patron, N.; Kay, P.; Wong, D.; Buchanan, M.; Cao, Y.Y.; Sawbridge, T.; Davies, J.P.; Mason, J.; Webb, S.R.; et al. Zinc finger nuclease-mediated precision genome editing of an endogenous gene in hexaploid bread wheat (Triticum aestivum) using a DNA repair template. Plant Biotechnol. J. 2018, 16, 2088–2101. [Google Scholar] [CrossRef] [PubMed]
- Svitashev, S.; Schwartz, C.; Lenderts, B.; Young, J.K.; Mark Cigan, A. Genome editing in maize directed by CRISPR-Cas9 ribonucleoprotein complexes. Nat. Commun. 2016, 7, 13274. [Google Scholar] [CrossRef] [PubMed]
- Svitashev, S.; Young, J.K.; Schwartz, C.; Gao, H.; Falco, S.C.; Cigan, A.M. Targeted Mutagenesis, Precise Gene Editing, and Site-Specific Gene Insertion in Maize Using Cas9 and Guide RNA. Plant Physiol. 2015, 169, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Puchta, H. Applying CRISPR/Cas for genome engineering in plants: The best is yet to come. Curr. Opin. Plant Biol. 2017, 36, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Li, X.; Zhang, Y.; Starker, C.G.; Baltes, N.J.; Zhang, F.; Sander, J.D.; Reyon, D.; Joung, J.K.; Voytas, D.F. Targeted deletion and inversion of tandemly arrayed genes in Arabidopsis thaliana using zinc finger nucleases. G3 (Bethesda, Md.) Genes Genom. Genet. 2013, 3, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Liu, B.; Weeks, D.P.; Spalding, M.H.; Yang, B. Large chromosomal deletions and heritable small genetic changes induced by CRISPR/Cas9 in rice. Nucleic Acid Res. 2014, 42, 10903–10914. [Google Scholar] [CrossRef] [PubMed]
- Lynagh, P.G.; Inagaki, S.; Amundson, K.R.; Marimithu, M.P.A.; Pike, B.R.; Henry, I.M.; Tan, E.H.; Comai, L. Translocation and duplication from CRISPR-Cas9 editing in Arabidopsis thaliana. BioRXiv 2018. [Google Scholar] [CrossRef]
- Shan, Q.; Wang, Y.; Chen, K.; Liang, Z.; Li, J.; Zhang, Y.; Zhang, K.; Liu, J.; Voytas, D.F.; Zheng, X.; et al. Rapid and efficient gene modification in rice and Brachypodium using TALENs. Mol. Plant. 2013, 6, 1365–1368. [Google Scholar] [CrossRef]
- Liang, G.; Zhang, H.; Lou, D.; Yu, D. Selection of highly efficient sgRNAs for CRISPR/Cas9-based plant genome editing. Sci. Rep. 2016, 6, 21451. [Google Scholar] [CrossRef] [PubMed]
- Ainley, W.M.; Sastry-Dent, L.; Welter, M.E.; Murray, M.G.; Zeitler, B.; Amora, R.; Corbin, D.R.; Miles, R.R.; Arnold, N.L.; Strange, T.L.; et al. Trait stacking via targeted genome editing. Plant Biotech. J. 2013, 11, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Wright, T.R.; Shan, G.; Walsh, T.A.; Lira, J.M.; Cui, C.; Song, P.; Zhuang, M.; Arnold, N.L.; Lin, G.; Yau, K.; et al. Robust crop resistance to broadleaf and grass herbicides provided by aryloxyalkanoate dioxygenase transgenes. Proc. Natl. Acad. Sci. USA 2010, 107, 20240–20245. [Google Scholar] [CrossRef] [PubMed]
- Bonawitz, N.D.; Ainley, W.M.; Itaya, A.; Chennareddy, S.R.; Cicak, T.; Effinger, K.; Jiang, K.; Mall, T.K.; Marri, P.R.; Samuel, J.P.; et al. Zinc finger nuclease-mediated targeting of multiple transgenes to an endogenous soybean genomic locus via non-homologous end joining. Plant Biotechnol. J. 2019, 17, 750–761. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.; Schiermeyer, A.; Dolls, A.; Koch, N.; Herwartz, D.; Kirchhoff, J.; Fischer, R.; Russell, S.M.; Cao, Z.; Corbin, D.R.; et al. Targeted gene exchange in plant cells mediated by a zinc finger nuclease double cut. Plant Biotechnol. J. 2016, 14, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Gao, H.; Wang, H.; Lafitte, H.R.; Archibald, R.L.; Yang, M.; Hakimi, S.M.; Mo, H.; Habben, J.E. ARGOS8 variants generated by CRISPR-Cas9 improve maize grain yield under field drought stress conditions. Plant Biotechnol. J. 2017, 15, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Hoekema, A.; Hirsch, P.R.; Hooykaas, P.J.; Schilperoort, R.A. A binary plant vector strategy based on separation of vir-and T-region of the Agrobacterium tumefaciens Ti-plasmid. Nature 1983, 303, 179. [Google Scholar] [CrossRef]
- Lee, L.-Y.; Gelvin, S.B. T-DNA binary vectors and systems. Plant Physiol. 2008, 146, 325–332. [Google Scholar] [CrossRef]
- Chilton, M.-D. Agrobacterium Ti Plasmids as a Tool for Genetic Engineering in Plants. In Genetic Engineering of Osmoregulation: Impact on Plant Productivity for Food, Chemicals, and Energy; Rains, D.W., Valentine, R.C., Hollaender, A., Eds.; Springer: Boston, MA, USA, 1980; pp. 23–31. [Google Scholar]
- Chen, Q.; Lai, H.; Hurtado, J.; Stahnke, J.; Leuzinger, K.; Dent, M. Agroinfiltration as an Effective and Scalable Strategy of Gene Delivery for Production of Pharmaceutical Proteins. Adv. Tech. Biol. Med. 2013, 1, 103. [Google Scholar] [CrossRef]
- Valvekens, D.; Montagu, M.V.; Lijsebettens, M.V. Agrobacterium tumefaciens-mediated transformation of Arabidopsis thaliana root explants by using kanamycin selection. Proc. Natl. Acad. Sci. USA 1988, 85, 5536–5540. [Google Scholar] [CrossRef] [PubMed]
- An, G. High Efficiency Transformation of Cultured Tobacco Cells. Plant Physiol. 1985, 79, 568–570. [Google Scholar] [CrossRef] [PubMed]
- Deblaere, R.; Bytebier, B.; De Greve, H.; Deboeck, F.; Schell, J.; Van Montagu, M.; Leemans, J. Efficient octopine Ti plasmid-derived vectors for Agrobacterium-mediated gene transfer to plants. Nucleic Acid. Res. 1985, 13, 4777–4788. [Google Scholar] [CrossRef] [PubMed]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.; Giritch, A.; Bartels, D.; Bortesi, L.; Gleba, Y. A novel and fully scalable Agrobacterium spray-based process for manufacturing cellulases and other cost-sensitive proteins in plants. Plant Biotechnol. J. 2015, 13, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Sood, P.; Bhattacharya, A.; Sood, A. Problems and possibilities of monocot transformation. Biol. Plant. 2011, 55, 1–15. [Google Scholar] [CrossRef]
- Ratanasut, K.; Rod-In, W.; Sujipuli, K. In planta Agrobacterium-Mediated Transformation of Rice. Rice Sci. 2017, 24, 181–186. [Google Scholar] [CrossRef]
- Barker, R.F.; Idler, K.B.; Thompson, D.V.; Kemp, J.D. Nucleotide sequence of the T-DNA region from the Agrobacterium tumefaciens octopine Ti plasmid pTi15955. Plant Mol. Biol. 1983, 2, 335–350. [Google Scholar] [CrossRef]
- Hamilton, C.M.; Frary, A.; Lewis, C.; Tanksley, S.D. Stable transfer of intact high molecular weight DNA into plant chromosomes. Proc. Natl. Acad. Sci. USA 1996, 93, 9975–9979. [Google Scholar] [CrossRef]
- Preston, A. Choosing a cloning vector. Methods Mol. Biol. 2003, 235, 19–26. [Google Scholar] [CrossRef]
- Che, P.; Anand, A.; Wu, E.; Sander Jeffry, D.; Simon Marissa, K.; Zhu, W.; Sigmund Amy, L.; Zastrow-Hayes, G.; Miller, M.; Liu, D.; et al. Developing a flexible, high-efficiency Agrobacterium-mediated sorghum transformation system with broad application. Plant Biotechnol. J. 2018, 16, 1388–1395. [Google Scholar] [CrossRef] [PubMed]
- Verma, K.; Saini, R.; Rani, A. Recent advances in the regeneration and genetic transformation of soybean. J. Innov. Biol. 2014, 1, 15–26. [Google Scholar]
- Lor, V.S.; Starker, C.G.; Voytas, D.F.; Weiss, D.; Olszewski, N.E. Targeted Mutagenesis of the Tomato PROCERA Gene Using Transcription Activator-Like Effector Nucleases. Plant Physiol. 2014, 166, 1288–1291. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, W.; Katin-Grazzini, L.; Ding, J.; Gu, X.; Li, Y.; Gu, T.; Wang, R.; Lin, X.; Deng, Z.; et al. A method for the production and expedient screening of CRISPR/Cas9-mediated non-transgenic mutant plants. Hortic. Res. 2018, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Jansing, J.; Sack, M.; Augustine, S.M.; Fischer, R.; Bortesi, L. CRISPR/Cas9-mediated knockout of six glycosyltransferase genes in Nicotiana benthamiana for the production of recombinant proteins lacking beta-1,2-xylose and core alpha-1,3-fucose. Plant Biotechnol. J. 2018, 17, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Chaparro-Garcia, A.; Kamoun, S.; Nekrasov, V. Boosting plant immunity with CRISPR/Cas. Genome Biol. 2015, 16, 254. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.; Turesson, H.; Nicolia, A.; Falt, A.S.; Samuelsson, M.; Hofvander, P. Efficient targeted multiallelic mutagenesis in tetraploid potato (Solanum tuberosum) by transient CRISPR-Cas9 expression in protoplasts. Plant Cell Rep. 2017, 36, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Baltes, N.J.; Gil-Humanes, J.; Cermak, T.; Atkins, P.A.; Voytas, D.F. DNA Replicons for Plant Genome Engineering. Plant Cell. 2014, 26, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Voytas, D.F. Plant genome engineering with sequence-specific nucleases. Ann. Rev. Plant. Biol. 2013, 64, 327–350. [Google Scholar] [CrossRef]
- Ali, Z.; Abul-Faraj, A.; Piatek, M.; Mahfouz, M.M. Activity and specificity of TRV-mediated gene editing in plants. Plant Signal. Behav. 2015, 10, e1044191. [Google Scholar] [CrossRef]
- Chujo, T.; Yoshikawa, M.; Ariga, H.; Endo, M.; Toki, S.; Ishibashi, K. A removable virus vector suitable for plant genome editing. Plant J. 2017, 91, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Cody, W.; Scholthof, H.B.; Mirkov, T.E. Multiplexed gene editing and protein over-expression using a Tobacco mosaic virus viral vector. Plant Physiol. 2017, 175, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.S.; Mansoor, S. Viral Vectors for Plant Genome Engineering. Front. Plant Sci. 2017, 8, 539. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.; Abul-Faraj, A.; Li, L.; Ghosh, N.; Piatek, M.; Mahjoub, A.; Aouida, M.; Piatek, A.; Baltes, N.J.; Voytas, D.F. Efficient Virus-Mediated Genome Editing in Plants Using the CRISPR/Cas9 System. Mol. Plant. 2015, 8, 1288–1291. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Han, T.; Liu, G.; Chen, T.; Wang, Y.; Yu, A.Y.; Liu, Y. A geminivirus-based guide RNA delivery system for CRISPR/Cas9 mediated plant genome editing. Sci. Rep. 2015, 5, 14926. [Google Scholar] [CrossRef] [PubMed]
- Gil-Humanes, J.; Wang, Y.; Liang, Z.; Shan, Q.; Ozuna, C.V.; Sanchez-Leon, S.; Baltes, N.J.; Starker, C.; Barro, F.; Gao, C.; et al. High efficiency gene targeting in hexaploid wheat using DNA replicons and CRISPR/Cas9. Plant J. 2016, 89, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Sanford, J.C. Biolistic plant transformation. Physiol. Plant. 1990, 79, 206–209. [Google Scholar] [CrossRef]
- Vidal, J.R.; Kikkert, J.R.; Wallace, P.G.; Reisch, B.I. High-efficiency biolistic co-transformation and regeneration of ‘Chardonnay’ (Vitis vinifera L.) containing npt-II and antimicrobial peptide genes. Plant. Cell Rep. 2003, 22, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Ismagul, A.; Yang, N.; Maltseva, E.; Iskakova, G.; Mazonka, I.; Skiba, Y.; Bi, H.; Eliby, S.; Jatayev, S.; Shavrukov, Y.; et al. A biolistic method for high-throughput production of transgenic wheat plants with single gene insertions. BMC Plant Biol. 2018, 18, 135. [Google Scholar] [CrossRef] [PubMed]
- Baltes, N.J.; Gil-Humanes, J.; Voytas, D.F. Chapter One—Genome Engineering and Agriculture: Opportunities and Challenges. In Progress in Molecular Biology and Translational Science; Weeks, D.P., Yang, B., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 149, pp. 1–26. [Google Scholar]
- Darbani, B.; Farajnia, S.; Toorchi, M.; Zakerbostanabad, S.; Noeparvar, S.; Stewart, C. DNA-delivery methods to produce transgenic plants. Biotechnology 2008, 7, 385–402. [Google Scholar] [CrossRef]
- Bernard, G.; Gagneul, D.; Alves Dos Santos, H.; Etienne, A.; Hilbert, J.-L.; Rambaud, C. Efficient Genome Editing Using CRISPR/Cas9 Technology in Chicory. Int. J. Mol. Sci. 2019, 20, 1155. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Chen, K.; Li, T.; Zhang, Y.; Wang, Y.; Zhao, Q.; Liu, J.; Zhang, H.; Liu, C.; Ran, Y.; et al. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat. Commun. 2017, 8, 14261. [Google Scholar] [CrossRef] [PubMed]
- Potrykus, I.; Bilang, R.; Futterer, J.; Sautter, C.; Schrott, M.; Spangenberg, G. Genetic engineering of crop plants. In Agricultural Biotechnology; CRC Press: Boca Raton, FL, USA, 1998; pp. 119–159. [Google Scholar]
- Nicolia, A.; Proux-Wéra, E.; Åhman, I.; Onkokesung, N.; Andersson, M.; Andreasson, E.; Zhu, L.-H. Targeted gene mutation in tetraploid potato through transient TALEN expression in protoplasts. J. Biotechnol. 2015, 204, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Demirer, G.S.; Landry, M.P. Delivering genes to plants. Chem. Eng. Prog. 2017, 113, 40–45. [Google Scholar]
- Hauptmann, R.M.; Ozias-Akins, P.; Vasil, V.; Tabaeizadeh, Z.; Rogers, S.G.; Horsch, R.B.; Vasil, I.K.; Fraley, R.T. Transient expression of electroporated DNA in monocotyledonous and dicotyledonous species. Plant. Cell Rep. 1987, 6, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Ou-Lee, T.-M.; Turgeon, R.; Wu, R. Expression of a foreign gene linked to either a plant-virus or a Drosophila promoter, after electroporation of protoplasts of rice, wheat, and sorghum. Proc. Natl. Acad. Sci. USA 1986, 83, 6815–6819. [Google Scholar] [CrossRef] [PubMed]
- Que, Q.; Elumalai, S.; Li, X.; Zhong, H.; Nalapalli, S.; Schweiner, M.; Fei, X.; Nuccio, M.; Kelliher, T.; Gu, W.; et al. Maize transformation technology development for commercial event generation. Front Plant. Sci. 2014, 5. [Google Scholar] [CrossRef]
- Yadava, P.; Abhishek, A.; Singh, R.; Singh, I.; Kaul, T.; Pattanayak, A.; Agrawal, P.K. Advances in Maize Transformation Technologies and Development of Transgenic Maize. Front. Plant. Sci. 2017, 7. [Google Scholar] [CrossRef]
- Furuhata, Y.; Sakai, A.; Murakami, T.; Morikawa, M.; Nakamura, C.; Yoshizumi, T.; Fujikura, U.; Nishida, K.; Kato, Y. A method using electroporation for the protein delivery of Cre recombinase into cultured Arabidopsis cells with an intact cell wall. Sci. Rep. 2019, 9, 2163. [Google Scholar] [CrossRef]
- Bhowmik, P.; Ellison, E.; Polley, B.; Bollina, V.; Kulkarni, M.; Ghanbarnia, K.; Song, H.; Gao, C.; Voytas, D.F.; Kagale, S. Targeted mutagenesis in wheat microspores using CRISPR/Cas9. Sci. Rep. 2018, 8, 6502. [Google Scholar] [CrossRef]
- Kaeppler, H.; Somers, D.; Rines, H.; Cockburn, A. Silicon carbide fiber-mediated stable transformation of plant cells. Theor. Appl. Genet. 1992, 84, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Petolino, J.F.; Arnold, N.L. Whiskers-mediated maize transformation. In Transgenic Maize; Springer: Berlin, Germany, 2009; pp. 59–67. [Google Scholar]
- Neuhaus, G.; Spangenberg, G. Plant transformation by microinjection techniques. Physiol. Plant. 1990, 79, 213–217. [Google Scholar] [CrossRef]
- Barampuram, S.; Zhang, Z.J. Recent advances in plant transformation. In Plant Chromosome Engineering; Springer: Berlin, Germany, 2011; pp. 1–35. [Google Scholar]
- Zhang, Y.; Liang, Z.; Zong, Y.; Wang, Y.; Liu, J.; Chen, K.; Qiu, J.-L.; Gao, C. Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat. Commun. 2016, 7, 12617. [Google Scholar] [CrossRef] [PubMed]
- Beetham, P.R.; Kipp, P.B.; Sawycky, X.L.; Arntzen, C.J.; May, G.D. A tool for functional plant genomics: Chimeric RNA/DNA oligonucleotides cause in vivo gene-specific mutations. Proc. Nat. Acad. Sci. USA 1999, 96, 8774–8778. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Peterson, D.J.; Tagliani, L.; Clair, G.S.; Baszczynski, C.L.; Bowen, B. Targeted manipulation of maize genes in vivo using chimeric RNA/DNA oligonucleotides. Proc. Nat. Acad. Sci. USA 1999, 96, 8768–8773. [Google Scholar] [CrossRef]
- Okuzaki, A.; Toriyama, K. Chimeric RNA/DNA oligonucleotide-directed gene targeting in rice. Plant Cell. Rep. 2004, 22, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Beetham, P.; Vincent, K.; Sharp, P. Oligonucleotide-directed gene repair in wheat using a transient plasmid gene repair assay system. Plant Cell. Rep. 2006, 25, 457–465. [Google Scholar] [CrossRef]
- Stoddard, T.J.; Clasen, B.M.; Baltes, N.J.; Demorest, Z.L.; Voytas, D.F.; Zhang, F.; Luo, S. Targeted Mutagenesis in Plant Cells through Transformation of Sequence-Specific Nuclease mRNA. PLoS ONE 2016, 11, e0154634. [Google Scholar] [CrossRef]
- Gocal, G. Non-transgenic trait development in crop plants using oligo-directed mutagenesis: Cibus’ rapid trait development system. In NABC Report 26; Boyce Thompson Institute: Ithaca, CA, USA, 2014. [Google Scholar]
- Woo, J.W.; Kim, J.; Kwon, S.I.; Corvalan, C.; Cho, S.W.; Kim, H.; Kim, S.-G.; Kim, S.-T.; Choe, S.; Kim, J.-S. DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins. Nat. Biotech. 2015, 33, 1162. [Google Scholar] [CrossRef]
- Malnoy, M.; Viola, R.; Jung, M.-H.; Koo, O.-J.; Kim, S.; Kim, J.-S.; Velasco, R.; Nagamangala Kanchiswamy, C. DNA-Free Genetically Edited Grapevine and Apple Protoplast Using CRISPR/Cas9 Ribonucleoproteins. Front Plant. Sci. 2016, 7. [Google Scholar] [CrossRef]
- Subburaj, S.; Chung, S.J.; Lee, C.; Ryu, S.-M.; Kim, D.H.; Kim, J.-S.; Bae, S.; Lee, G.-J. Site-directed mutagenesis in Petunia × hybrida protoplast system using direct delivery of purified recombinant Cas9 ribonucleoproteins. Plant Cell Rep. 2016, 35, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Miki, B.; McHugh, S. Selectable marker genes in transgenic plants: Applications, alternatives and biosafety. J. Biotechnol. 2004, 107, 193–232. [Google Scholar] [CrossRef] [PubMed]
- Breyer, D.; Kopertekh, L.; Reheul, D. Alternatives to Antibiotic Resistance Marker Genes for In Vitro Selection of Genetically Modified Plants–Scientific Developments, Current Use, Operational Access and Biosafety Considerations. Critl. Rev. Plant Sci. 2014, 33, 286–330. [Google Scholar] [CrossRef]
- Rosellini, D. Selectable Markers and Reporter Genes: A Well Furnished Toolbox for Plant Science and Genetic Engineering. Crit Rev. Plant. Sci. 2012, 31, 401–453. [Google Scholar] [CrossRef]
- Goodwin, J.L.; Pastori, G.M.; Davey, M.R.; Jones, H.D. Selectable markers: Antibiotic and herbicide resistance. In Transgenic Plants: Methods and Protocols; Springer: Berlin, Germany, 2005; pp. 191–201. [Google Scholar]
- Park, J.; Lee, Y.K.; Kang, B.K.; Chung, W.I. Co-transformation using a negative selectable marker gene for the production of selectable marker gene-free transgenic plants. Theor. Appl. Genet. 2004, 109, 1562–1567. [Google Scholar] [CrossRef] [PubMed]
- Erikson, O.; Hertzberg, M.; Näsholm, T. A conditional marker gene allowing both positive and negative selection in plants. Nat. Biotechnol. 2004, 22, 455. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.A.; Tinland, B.; Gilbertson, L.A.; Staub, J.M.; Bannon, G.A.; Goodman, R.E.; McCoy, R.L.; Silvanovich, A. Human safety and genetically modified plants: A review of antibiotic resistance markers and future transformation selection technologies. J. Appl. Microbiol. 2005, 99, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Hiei, Y.; Ishida, Y.; Komari, T. Progress of cereal transformation technology mediated by Agrobacterium tumefaciens. Front. Plant. Sci. 2014, 5. [Google Scholar] [CrossRef]
- Barrell Philippa, J.; Meiyalaghan, S.; Jacobs Jeanne, M.E.; Conner Anthony, J. Applications of biotechnology and genomics in potato improvement. Plant Biotech. J. 2013, 11, 907–920. [Google Scholar] [CrossRef]
- Twyman, R.M.; Stöger, E.; Kohli, A.; Capell, T.; Christou, P. Selectable and Screenable Markers for Rice Transformation. In Testing for Genetic Manipulation in Plants; Jackson, J.F., Linskens, H.F., Eds.; Springer: Berlin, Germany, 2002; pp. 1–17. [Google Scholar]
- Hiei, Y.; Komari, T. Agrobacterium-mediated transformation of rice using immature embryos or calli induced from mature seed. Nat. Protoc. 2008, 3, 824. [Google Scholar] [CrossRef]
- Harwood, W. Barley as a Cereal Model for Biotechnology Applications. In Biotechnology of Major Cereals; Jones, H.D., Ed.; CABI: Wallingford, UK, 2016. [Google Scholar]
- Marthe, C.; Kumlehn, J.; Hensel, G. Barley (Hordeum vulgare L.) transformation using immature embryos. In Agrobacterium Protocols; Springer: Berlin, Germany, 2015; pp. 71–83. [Google Scholar]
- Ishida, Y.; Hiei, Y.; Komari, T. High Efficiency Wheat Transformation Mediated by Agrobacterium tumefaciens. In Advances in Wheat Genetics: From Genome to Field; Springer: Tokyo, Japan, 2015; pp. 167–173. [Google Scholar]
- Ortiz, J.P.; Reggiardo, M.I.; Ravizzini, R.A.; Altabe, S.G.; Cervigni, G.D.; Spitteler, M.A.; Morata, M.M.; Elias, F.E.; Vallejos, R.H. Hygromycin resistance as an efficient selectable marker for wheat stable transformation. Plant Cell. Rep. 1996, 15, 877–881. [Google Scholar] [CrossRef]
- Hu, L.; Li, H.; Qin, R.; Xu, R.; Li, J.; Li, L.; Wei, P.; Yang, J. Plant phosphomannose isomerase as a selectable marker for rice transformation. Sci. Rep. 2016, 6, 25921. [Google Scholar] [CrossRef]
- Wei, Z.; Wang, X.; Xing, S. Current progress of biosafe selectable markers in plant transformation. J. Plant Breed. Crop. Sci. 2012, 4, 1–8. [Google Scholar]
- Ziemienowicz, A. Plant selectable markers and reporter genes. Acta Physiol. Plant. 2001, 23, 363–374. [Google Scholar] [CrossRef]
- Kortstee, A.J.; Khan, S.A.; Helderman, C.; Trindade, L.M.; Wu, Y.; Visser, R.G.F.; Brendolise, C.; Allan, A.; Schouten, H.J.; Jacobsen, E. Anthocyanin production as a potential visual selection marker during plant transformation. Transgenic Res. 2011, 20, 1253–1264. [Google Scholar] [CrossRef] [PubMed]
- Mikami, M.; Toki, S.; Endo, M. Comparison of CRISPR/Cas9 expression constructs for efficient targeted mutagenesis in rice. Plant Mol. Biol. 2015, 88, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Ueta, R.; Abe, C.; Watanabe, T.; Sugano, S.S.; Ishihara, R.; Ezura, H.; Osakabe, Y.; Osakabe, K. Rapid breeding of parthenocarpic tomato plants using CRISPR/Cas9. Sci. Rep. 2017, 7, 507. [Google Scholar] [CrossRef] [PubMed]
- Shan, Q.; Wang, Y.; Li, J.; Zhang, Y.; Chen, K.; Liang, Z.; Zhang, K.; Liu, J.; Xi, J.J.; Qiu, J.-L.; et al. Targeted genome modification of crop plants using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 686–688. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.K.; Kumar, J.; Alok, A.; Tuli, R. RNA-guided genome editing for target gene mutations in wheat. G3 (Bethesda, Md.) 2013, 3, 2233–2238. [Google Scholar] [CrossRef]
- Fan, D.; Liu, T.; Li, C.; Jiao, B.; Li, S.; Hou, Y.; Luo, K. Efficient CRISPR/Cas9-mediated Targeted Mutagenesis in Populus in the First Generation. Sci. Rep. 2015, 5, 12217. [Google Scholar] [CrossRef] [PubMed]
- Odipio, J.; Alicai, T.; Ingelbrecht, I.; Nusinow, D.A.; Bart, R.; Taylor, N.J. Efficient CRISPR/Cas9 Genome Editing of Phytoene desaturase in Cassava. Front. Plant. Sci. 2017, 8, 1780. [Google Scholar] [CrossRef]
- Nishitani, C.; Hirai, N.; Komori, S.; Wada, M.; Okada, K.; Osakabe, K.; Yamamoto, T.; Osakabe, Y. Efficient Genome Editing in Apple Using a CRISPR/Cas9 system. Sci. Rep. 2016, 6, 31481. [Google Scholar] [CrossRef] [PubMed]
- Qin, G.; Gu, H.; Ma, L.; Peng, Y.; Deng, X.W.; Chen, Z.; Qu, L.-J. Disruption of phytoene desaturase gene results in albino and dwarf phenotypes in Arabidopsis by impairing chlorophyll, carotenoid, and gibberellin biosynthesis. Cell Res. 2007, 17, 471. [Google Scholar] [CrossRef] [PubMed]
- Delporte, F.; Jacquemin, J.-M.; Masson, P.; Watillon, B. Insights into the regenerative property of plant cells and their receptivity to transgenesis. Plant Signal. Behav. 2012, 7, 1608–1620. [Google Scholar] [CrossRef] [PubMed]
- Ikeuchi, M.; Ogawa, Y.; Iwase, A.; Sugimoto, K. Plant regeneration: Cellular origins and molecular mechanisms. Development 2016, 143, 1442–1451. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, D.; Kareem, A.; Durgaprasad, K.; Sreeraj, E.; Sugimoto, K.; Prasad, K. Shoot regeneration: A journey from acquisition of competence to completion. Curr. Opin. Plant Biol. 2018, 41, 23–31. [Google Scholar] [CrossRef]
- Birch, R.G. Plant Transformation: Problems and Strategies for Practical Application. Ann. Rev. Plant Physiol. Plant Mo.l Biol. 1997, 48, 297–326. [Google Scholar] [CrossRef]
- Kelliher, T.; Starr, D.; Su, X.; Tang, G.; Chen, Z.; Carter, J.; Wittich, P.E.; Dong, S.; Green, J.; Burch, E.; et al. One-step genome editing of elite crop germplasm during haploid induction. Nat. Biotechnol. 2019, 37, 287–292. [Google Scholar] [CrossRef]
- Wang, B.; Zhu, L.; Zhao, B.; Zhao, Y.; Xie, Y.; Zheng, Z.; Li, Y.; Sun, J.; Wang, H. Development of a Haploid-Inducer Mediated Genome Editing System for Accelerating Maize Breeding. Mol. Plant. 2019, 12, 597–602. [Google Scholar] [CrossRef]
- Rhodes, C.A.; Lowe, K.S.; Ruby, K.L. Plant Regeneration from Protoplasts Isolated from Embryogenic Maize Cell Cultures. Nat. Biotechnol. 1988, 6, 56. [Google Scholar] [CrossRef]
- Lin, C.S.; Hsu, C.T.; Yang, L.H.; Lee, L.Y.; Fu, J.Y.; Cheng, Q.W.; Wu, F.H.; Hsiao Han, C.W.; Zhang, Y.; Zhang, R.; et al. Application of protoplast technology to CRISPR/Cas9 mutagenesis: From single-cell mutation detection to mutant plant regeneration. Plant Biotechnol. J. 2017, 16, 1295–1310. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Mohanty, A. Rice transformation for crop improvement and functional genomics. Plant Sci. 2000, 158, 1–18. [Google Scholar] [CrossRef]
- Mikami, M.; Toki, S.; Endo, M. Parameters affecting frequency of CRISPR/Cas9 mediated targeted mutagenesis in rice. Plant Cell Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.D. Wheat transformation: Current technology and applications to grain development and composition. J. Cereal Sci. 2005, 41, 137–147. [Google Scholar] [CrossRef]
- Li, J.; Ye, X.; An, B.; Du, L.; Xu, H. Genetic transformation of wheat: Current status and future prospects. Plant Biotechnol. Rep. 2012, 6, 183–193. [Google Scholar] [CrossRef]
- Parrott, D.L.; Anderson, A.J.; Carman, J.G. Agrobacterium induces plant cell death in wheat (Triticum aestivum L.). Physiol. Mol. Plant. Pathol. 2002, 60, 59–69. [Google Scholar] [CrossRef]
- Chauhan, H.; Khurana, P. Wheat Genetic Transformation Using Mature Embryos as Explants. In Wheat Biotechnology: Methods and Protocols; Bhalla, P.L., Singh, M.B., Eds.; Springer: New York, NY, USA, 2017; pp. 153–167. [Google Scholar]
- Aadel, H.; Abdelwahd, R.; Udupa, S.; Diria, G.; El Mouhtadi, A.; Ahansal, K.; Gaboun, F.; Douira, A.; Iraqi, D. Agrobacterium-mediated transformation of mature embryo tissues of bread wheat (Triticum aestivum L.) genotypes. Cereal Res. Commun. 2018, 46, 10–20. [Google Scholar] [CrossRef]
- Harwood, W.A. Advances and remaining challenges in the transformation of barley and wheat. J. Exp. Bot. 2012, 63, 1791–1798. [Google Scholar] [CrossRef]
- Holme, I.B.; Brinch-Pedersen, H.; Lange, M.; Holm, P.B. Transformation of different barley (Hordeum vulgare L.) cultivars by Agrobacterium tumefaciens infection of in vitro cultured ovules. Plant Cell Rep. 2008, 27, 1833–1840. [Google Scholar] [CrossRef]
- Kapusi, E.; Corcuera-Gomez, M.; Melnik, S.; Stoger, E. Heritable Genomic Fragment Deletions and Small Indels in the Putative ENGase Gene Induced by CRISPR/Cas9 in Barley. Front. Plant Sci. 2017, 8, 540. [Google Scholar] [CrossRef]
- Altpeter, F.; Springer, N.M.; Bartley, L.E.; Blechl, A.E.; Brutnell, T.P.; Citovsky, V.; Conrad, L.J.; Gelvin, S.B.; Jackson, D.P.; Kausch, A.P.; et al. Advancing Crop Transformation in the Era of Genome Editing. Plant Cell 2016, 28, 1510–1520. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Godwin, I.D. Highly efficient sorghum transformation. Plant Cell Rep. 2012, 31, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Han, E.; Lee, M. An efficient transformation method for a potato (Solanum tuberosum L. var. Atlantic). J. Plant Biotechnol. 2015, 42, 77–82. [Google Scholar] [CrossRef]
- Bradshaw, J.E. Chapter 8—Potato-Breeding Strategy. In Potato Biology and Biotechnology; Vreugdenhil, D., Bradshaw, J., Gebhardt, C., Govers, F., Mackerron, D.K.L., Taylor, M.A., Ross, H.A., Eds.; Elsevier Science, B.V.: Amsterdam, The Netherlands, 2007; pp. 157–177. [Google Scholar]
- Hartman, G.L.; West, E.D.; Herman, T.K. Crops that feed the World 2. Soybean—Worldwide production, use, and constraints caused by pathogens and pests. Food Secur. 2011, 3, 5–17. [Google Scholar] [CrossRef]
- Lee, H.; Park, S.-Y.; Zhang, Z.J. An overview of genetic transformation of soybean. In A Comprehensive Survey of International Soybean Research-Genetics, Physiology, Agronomy and Nitrogen Relationships; InTech: London, UK, 2013. [Google Scholar]
- Homrich, M.S.; Wiebke-Strohm, B.; Weber, R.L.M.; Bodanese-Zanettini, M.H. Soybean genetic transformation: A valuable tool for the functional study of genes and the production of agronomically improved plants. Genet Mol. Biol. 2012, 35, 998–1010. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Yi, B.-Y.; Kim, K.-H.; Kim, J.-B.; Suh, S.-C.; Woo, H.-J.; Shin, K.-S.; Kweon, S.-J. Development of efficient transformation protocol for soybean (Glycine max L.) and characterization of transgene expression after Agrobacterium-mediated gene transfer. J. Korean Soc. Appl. Biol. Chem. 2011, 54, 37–45. [Google Scholar] [CrossRef]
- Limei, Z.; Depu, L.; Huan, S.; Ying, Y.; Mei, H. A Sterile Material of Soybean gained by Introducing Exogenous DNA. Soybean Sci. 1995, 01, 83–87. [Google Scholar]
- Liu, J.; Su, Q.; An, L.; Yang, A. Transfer of a minimal linear marker-free and vector-free smGFP cassette into soybean via ovary-drip transformation. Biotechnol. Lett. 2009, 31, 295–303. [Google Scholar] [CrossRef]
- Shou, H.; Palmer, R.G.; Wang, K. Irreproducibility of the soybean pollen-tube pathway transformation procedure. Plant Mol. Biol. Rep. 2002, 20, 325–334. [Google Scholar] [CrossRef]
- Du, H.; Zeng, X.; Zhao, M.; Cui, X.; Wang, Q.; Yang, H.; Cheng, H.; Yu, D. Efficient targeted mutagenesis in soybean by TALENs and CRISPR/Cas9. J. Biotechnol. 2016, 217, 90–97. [Google Scholar] [CrossRef]
- Mariashibu, T.S.; Anbazhagan, V.R.; Jiang, S.-Y.; Ganapathi, A.; Ramachandran, S. In vitro regeneration and genetic transformation of soybean: Current status and future prospects. In A Comprehensive Survey of International Soybean Research-Genetics, Physiology, Agronomy and Nitrogen Relationships; Board, J.E., Ed.; InTechOpen: London, UK, 2013. [Google Scholar]
- Cai, Y.; Chen, L.; Liu, X.; Guo, C.; Sun, S.; Wu, C.; Jiang, B.; Han, T.; Hou, W. CRISPR/Cas9-mediated targeted mutagenesis of GmFT2a delays flowering time in soybean. Plant Biotechnol. J. 2017. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Z.B.; Xing, A.; Moon, B.P.; Koellhoffer, J.P.; Huang, L.; Ward, R.T.; Clifton, E.; Falco, S.C.; Cigan, A.M. Cas9-guide RNA Directed Genome Editing in Soybean. Plant Physiol. 2015, 169, 960–970. [Google Scholar] [CrossRef] [PubMed]
- Curtin, S.J.; Xiong, Y.; Michno, J.M.; Campbell, B.W.; Stec, A.O.; Cermak, T.; Starker, C.; Voytas, D.F.; Eamens, A.L.; Stupar, R.M. CRISPR/Cas9 and TALENs generate heritable mutations for genes involved in small RNA processing of Glycine max and Medicago truncatula. Plant Biotechnol. J. 2018, 16, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Bull, S.E.; Owiti, J.A.; Niklaus, M.; Beeching, J.R.; Gruissem, W.; Vanderschuren, H. Agrobacterium-mediated transformation of friable embryogenic calli and regeneration of transgenic cassava. Nat. Protoc. 2009, 4, 1845. [Google Scholar] [CrossRef] [PubMed]
- Zainuddin, I.M.; Schlegel, K.; Gruissem, W.; Vanderschuren, H. Robust transformation procedure for the production of transgenic farmer-preferred cassava landraces. Plant Method. 2012, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Fondong, V.N.; Rey, C. Recent Biotechnological Advances in the Improvement of Cassava. In Cassava; Waisundara, V., Ed.; InTechOpen: London, UK, 2018. [Google Scholar]
- Gomez, M.A.; Lin, Z.D.; Moll, T.; Chauhan, R.D.; Hayden, L.; Renninger, K.; Beyene, G.; Taylor, N.J.; Carrington, J.C.; Staskawicz, B.J.; et al. Simultaneous CRISPR/Cas9-mediated editing of cassava eIF4E isoforms nCBP-1 and nCBP-2 reduces cassava brown streak disease symptom severity and incidence. Plant Biotechnol. J. 2019, 17, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Weeks, J.T.; Anderson, O.D.; Blechl, A.E. Rapid Production of Multiple Independent Lines of Fertile Transgenic Wheat (Triticum aestivum). Plant Physiol. 1993, 102, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, D.; Khurana, P. Wheat biotechnology: A minireview. Electron. J. Biotechnol. 2001, 4, 7–8. [Google Scholar]
- Reuzeau, C.; Pen, J.; Frankard, V.; de Wolf, J.; Peerbolte, R.; Broekaert, W.; van Camp, W. TraitMill: A discovery engine for identifying yield-enhancement genes in cereals. Mol. Plant Breed 2010, 3, 753–759. [Google Scholar] [CrossRef]
- Nishimura, A.; Aichi, I.; Matsuoka, M. A protocol for Agrobacterium-mediated transformation in rice. Nat. Protoc. 2007, 1, 2796. [Google Scholar] [CrossRef]
- Chennareddy, S.; Cicak, T.; Mall, T.; Effinger, K.; Sardesai, N.; Pareddy, D.; Sarria, R. Improved direct transformation via particle bombardment of split-immature embryo explants in soybean (Glycine max). Plant Cell Tissue Organ Cult 2018, 135, 22–35. [Google Scholar] [CrossRef]
- Watson, A.; Ghosh, S.; Williams, M.J.; Cuddy, W.S.; Simmonds, J.; Rey, M.-D.; Asyraf Md Hatta, M.; Hinchliffe, A.; Steed, A.; Reynolds, D.; et al. Speed breeding is a powerful tool to accelerate crop research and breeding. Nat. Plant. 2018, 4, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Eckerstorfer, M.F.; Engelhard, M.; Heissenberger, A.; Simon, S.; Teichmann, H. Plants Developed by New Genetic Modification Techniques-Comparison of Existing Regulatory Frameworks in the EU and Non-EU Countries. Front. Bioeng. Biotechnol. 2019, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Strauss, S.H.; Sax, J.K. Ending event-based regulation of GMO crops. Nat. Biotechnol. 2016, 34, 474–477. [Google Scholar] [CrossRef] [PubMed]
- McHughen, A. A critical assessment of regulatory triggers for products of biotechnology: Product vs. process. GM Crops Food 2016, 7, 125–158. [Google Scholar] [CrossRef] [PubMed]
- Court of Justice of the European Union. Press Release. Organisms obtained by mutagenesis are GMOs and are, in principle, subject to obligations laid down by the GMO Directive, Luxembourg. 25 July 2018. [Google Scholar]
- Grohmann, L.; Keilwagen, J.; Duensing, N.; Dagand, E.; Hartung, F.; Wilhelm, R.; Bendiek, J.; Sprink, T. Detection and Identification of Genome Editing in Plants: Challenges and Opportunities. Front. Plant Sci. 2019, 10, 236. [Google Scholar] [CrossRef] [PubMed]
- Lowder, L.G.; Malzahn, A.; Qi, Y. Plant Gene Regulation Using Multiplex CRISPR-dCas9 Artificial Transcription Factors. Method. Mol. Biol. 2018, 1676, 197–214. [Google Scholar] [CrossRef]
- Rodriguez-Leal, D.; Lemmon, Z.H.; Man, J.; Bartlett, M.E.; Lippman, Z.B. Engineering Quantitative Trait Variation for Crop Improvement by Genome Editing. Cell 2017, 171, 470–480. [Google Scholar] [CrossRef]
- Gallego-Bartolome, J.; Liu, W.; Kuo, P.H.; Feng, S.; Ghoshal, B.; Gardiner, J.; Zhao, J.M.; Park, S.Y.; Chory, J.; Jacobsen, S.E. Co-targeting RNA Polymerases IV and V Promotes Efficient De Novo DNA Methylation in Arabidopsis. Cell 2019, 176, 1068–1082 e1019. [Google Scholar] [CrossRef]
- Papikian, A.; Liu, W.; Gallego-Bartolome, J.; Jacobsen, S.E. Site-specific manipulation of Arabidopsis loci using CRISPR-Cas9 SunTag systems. Nat. Commun. 2019, 10, 729. [Google Scholar] [CrossRef]
- Puchta, H. Using CRISPR/Cas in three dimensions: Towards synthetic plant genomes, transcriptomes and epigenomes. Plant. J. 2016, 87, 5–15. [Google Scholar] [CrossRef] [PubMed]
| Delivery Method | Cargo | Plant Species | Tissue | Selection | Mutation Efficiency | Calculation | Reference |
|---|---|---|---|---|---|---|---|
| Agrobacterium-mediated transformation | DNA (transient) CRISPR/Cas9 | Tobacco Nicotiana tabacum | Leaf disks | No | 2.57% | Mutated plants/total regenerated shoots | [107] |
| No | 17.2% | Non-transgenic plants/total mutant plants | |||||
| Particle bombardment | DNA CRISPR/Cas9 | Wheat Triticum aestivum | Immature embryos | No | 3.3%, 2/26 plants homozygous and transgene-free | Mutated plants/bombarded embryos | [140] |
| IVT mRNA CRISPR/Cas9 | No | 1.1%, 6/17 plants homozygous and transgene-free | Mutated plants/bombarded embryos | ||||
| Particle bombardment | RNP CRISPR/Cas9 | Maize Zea mays | Immature embryos | No | 2.4–9.7%, 9.6–12.9% of mutated plants biallelic | Mutated plants/analyzed plants | [77] |
| Particle bombardment | RNP CRISPR/Cas9 | Wheat Triticum aestivum | Immature embryos | No | 4.4% | Mutated plants/bombarded embryos | [126] |
| Protoplast transfection (PEG) | DNA TALEN | Potato Solanum tuberosum | Protoplasts | No | 11–13% | Mutated callus/total protoplast-derived callus | [128] |
| Protoplast transfection (PEG) | DNA TALEN | Tobacco Nicotiana benthamiana | Protoplasts | n.a. | 70.5% | Deep sequencing of protoplasts | [145] |
| mRNA TALEN | n.a. | 5.8–16.9% | Without/with UTR | ||||
| Protoplast transfection (PEG) | RNP CRISPR/Cas9 | Lettuce Lactuca sativa | Protoplasts | No | 46%, 6% mono-, 40% biallelic | Mutated callus/analyzed callus | [147] |
| Protoplast transfection (PEG) | RNP CRISPR/Cas9 | Grapevine Vitis vinifera cv. Chardonnay | Protoplasts | n.a. | 0.1% | Deep sequencing of protoplasts | [148] |
| Apple Malus domestica cv. Golden delicious | Protoplasts | n.a. | 0.5–6.7% | Deep sequencing of protoplasts | |||
| Protoplast transfection (PEG) | RNP CRISPR/Cas9 | Petunia Petunia x hybrida | Protoplasts | n.a. | 5.3–17.8% | Deep sequencing of protoplasts | [149] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jansing, J.; Schiermeyer, A.; Schillberg, S.; Fischer, R.; Bortesi, L. Genome Editing in Agriculture: Technical and Practical Considerations. Int. J. Mol. Sci. 2019, 20, 2888. https://doi.org/10.3390/ijms20122888
Jansing J, Schiermeyer A, Schillberg S, Fischer R, Bortesi L. Genome Editing in Agriculture: Technical and Practical Considerations. International Journal of Molecular Sciences. 2019; 20(12):2888. https://doi.org/10.3390/ijms20122888
Chicago/Turabian StyleJansing, Julia, Andreas Schiermeyer, Stefan Schillberg, Rainer Fischer, and Luisa Bortesi. 2019. "Genome Editing in Agriculture: Technical and Practical Considerations" International Journal of Molecular Sciences 20, no. 12: 2888. https://doi.org/10.3390/ijms20122888
APA StyleJansing, J., Schiermeyer, A., Schillberg, S., Fischer, R., & Bortesi, L. (2019). Genome Editing in Agriculture: Technical and Practical Considerations. International Journal of Molecular Sciences, 20(12), 2888. https://doi.org/10.3390/ijms20122888






