Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles
Abstract
1. Introduction
2. Defense Mechanisms against Different Sizes of Metal and Metal Oxide Nanoparticles.
3. Defense Mechanisms against the Surface Charge of Metal and Metal Oxide Nanoparticles
4. Defense Mechanisms against Metal Ions Release from Metal and Metal Oxide Nanoparticles
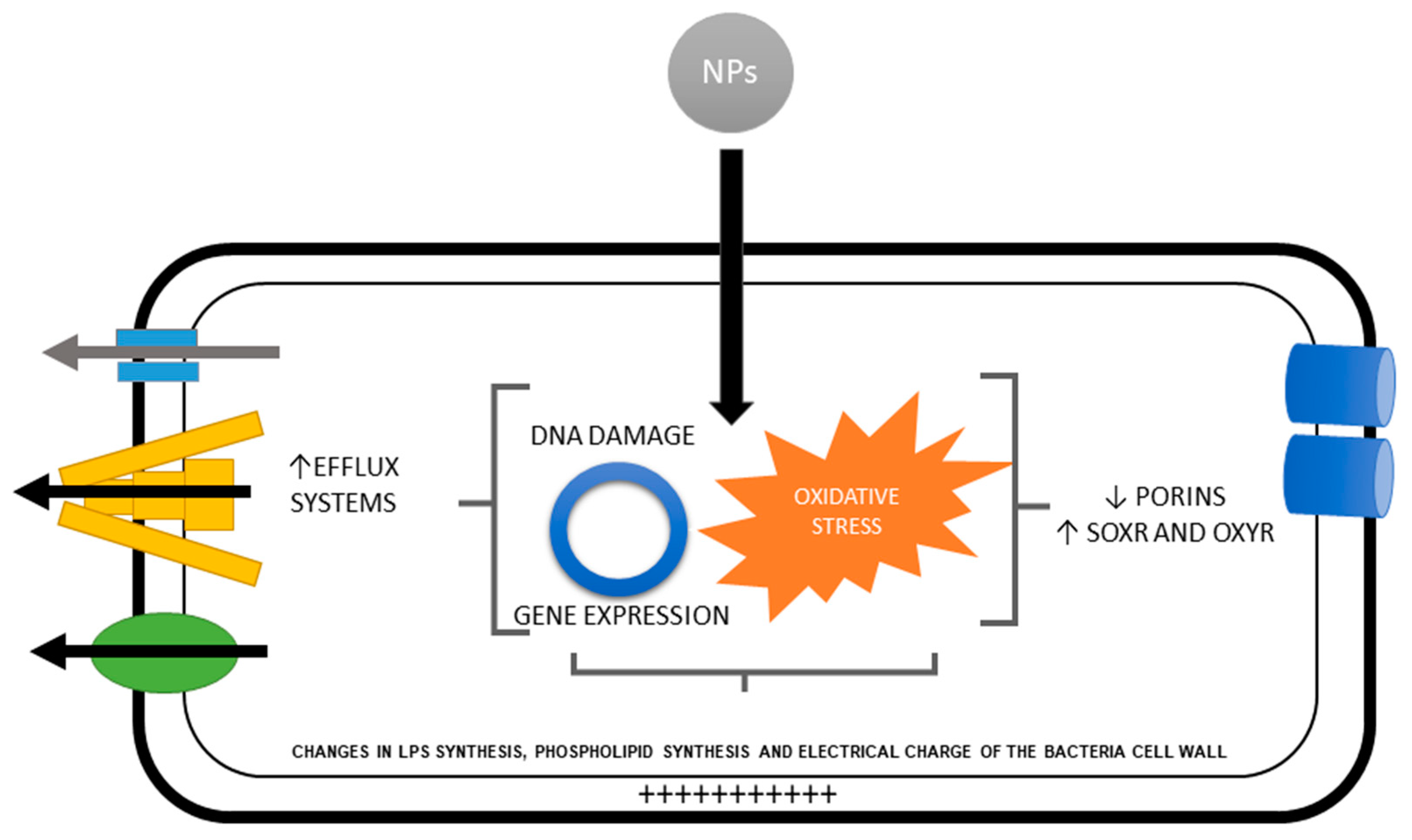
5. Defense Mechanisms against the Production of ROS and Oxidative Stress by Metal and Metal Oxide Nanoparticles.
6. Defense Mechanisms of Biofilms against Metal and Metal Oxide Nanoparticles.
7. Future Directions
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ZnO NPs | Zinc oxide nanoparticles |
| CuO NPs | Copper oxide nanoparticles |
| Fe2O3 NPs | Iron oxide nanoparticles |
| ECS | Extracellular substances |
| Ag NPs | Silver nanoparticles |
| MgO NPs | Magnesium oxide nanoparticles |
| Al2O3 NPs | Aluminum oxide nanoparticles |
| TiO2 NPs | Titanium dioxide nanoparticles |
| CeO2 NPs | Cerium oxide nanoparticles |
| CAMP | Cationic antimicrobial peptides |
| ESR | Envelope stress response |
| LPS | Lipopolysaccharide |
| PEA | Phosphoethanolamine |
| Cl- | Chloride |
| NOM | Natural organic matter |
| RND | Resistance-nodulation-cell division transporters |
| CDF | Cation diffusion facilitator transporters |
| QDs | Quantum dots |
| MFP | Membrane fusion protein family |
| OMF | Outer membrane factors |
| SiO2 NPs | Silicon oxide nanoparticles |
| Fe3O4 NPs | Ferric oxide nanoparticles |
| NiO NPs | Nickel oxide nanoparticles |
| TRx | Thioredoxin reductase |
| GPx | Glutathione peroxidase |
| CS-SeNPs | Chitosan-modified selenium nanoparticles |
| Cpx | Pilus expression response |
| Rcs | Regulation of capsular synthesis phosphorelay |
| Bae | Bacterial adaptive response |
| PSP | Phage shock protein response |
| ECF | Extracytoplasmic function |
| ROS | Reactive oxygen species |
| Au NPs | Gold nanoparticles |
| ECP | Extracellular polymers |
| PVP-AgNPs | Polyvinylpyrrolidone-coated silver nanoparticles |
| ARGs | Antibiotic resistance genes |
References
- Allahverdiyev, A.M.; Abamor, E.S.; Bagirova, M.; Rafailovich, M. Antimicrobial effects of TiO2 and Ag2O nanoparticles against drug-resistant bacteria and leishmania parasites. Future Microbiol. 2011, 6, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Klemm, E.J.; Wong, V.K.; Dougan, G. Emergence of dominant multidrug-resistant bacterial clades: Lessons from history and whole-genome sequencing. Proc. Natl. Acad. Sci. USA 2018, 115, 12872–12877. [Google Scholar] [CrossRef] [PubMed]
- Sugden, R.; Kelly, R.; Davies, S. Combatting antimicrobial resistance globally. Nat Microbiol 2016, 1, 16187. [Google Scholar] [CrossRef] [PubMed]
- Besinis, A.; De Peralta, T.; Handy, R.D. The antibacterial effects of silver, titanium dioxide and silica dioxide nanoparticles compared to the dental disinfectant chlorhexidine on Streptococcus mutans using a suite of bioassays. Nanotoxicology 2014, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Mathieu, J.M.; Chattopadhyay, S.; Miller, J.T.; Wu, T.; Shibata, T.; Guo, W.; Alvarez, P.J. Defense mechanisms of Pseudomonas aeruginosa PAO1 against quantum dots and their released heavy metals. ACS Nano 2012, 6, 6091–6098. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Carlsson, F.; Edman, M.; Hummelgård, M.; Jonsson, B.G.; Bylund, D.; Olin, H. Escherichia coli Bacteria Develop Adaptive Resistance to Antibacterial ZnO Nanoparticles. Adv. Biosyst. 2018, 2, 1800019. [Google Scholar] [CrossRef]
- Graves Jr, J.L.; Tajkarimi, M.; Cunningham, Q.; Campbell, A.; Nonga, H.; Harrison, S.H.; Barrick, J.E. Rapid evolution of silver nanoparticle resistance in Escherichia coli. Front. Genet. 2015, 6, 42. [Google Scholar] [CrossRef]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.; Memic, A. Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and-negative bacterial strains. Int. J. Nanomed. 2012, 7, 3527. [Google Scholar] [CrossRef]
- Agnihotri, S.; Mukherji, S.; Mukherji, S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. Rsc Adv. 2014, 4, 3974–3983. [Google Scholar] [CrossRef]
- Raza, M.; Kanwal, Z.; Rauf, A.; Sabri, A.; Riaz, S.; Naseem, S. Size-and shape-dependent antibacterial studies of silver nanoparticles synthesized by wet chemical routes. Nanomaterials 2016, 6, 74. [Google Scholar] [CrossRef] [PubMed]
- Helmlinger, J.; Sengstock, C.; Groß-Heitfeld, C.; Mayer, C.; Schildhauer, T.; Köller, M.; Epple, M. Silver nanoparticles with different size and shape: Equal cytotoxicity, but different antibacterial effects. Rsc Adv. 2016, 6, 18490–18501. [Google Scholar] [CrossRef]
- Khalandi, B.; Asadi, N.; Milani, M.; Davaran, S.; Abadi, A.J.N.; Abasi, E.; Akbarzadeh, A. A review on potential role of silver nanoparticles and possible mechanisms of their actions on bacteria. Drug Res 2017, 11, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Butler, K.S.; Peeler, D.J.; Casey, B.J.; Dair, B.J.; Elespuru, R.K. Silver nanoparticles: Correlating nanoparticle size and cellular uptake with genotoxicity. Mutagenesis 2015, 30, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Lawson, R.; Ray, P.C.; Yu, H. Toxic effects of gold nanoparticles on Salmonella typhimurium bacteria. Toxicol. Ind. Health 2011, 27, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Panáček, A.; Kvítek, L.; Smékalová, M.; Večeřová, R.; Kolář, M.; Röderová, M.; Dyčka, F.; Šebela, M.; Prucek, R.; Tomanec, O. Bacterial resistance to silver nanoparticles and how to overcome it. Nat. Nanotechnol. 2018, 13, 65. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Anaya, N.; Astudillo-Castro, C.; Oyanedel-Craver, V. Kinetic, metabolic and macromolecular response of bacteria to chronic nanoparticle exposure in continuous culture. Environ. Sci. Nano 2018, 5, 1386–1396. [Google Scholar] [CrossRef]
- Siemer, S.; Westmeier, D.; Barz, M.; Eckrich, J.; Wünsch, D.; Seckert, C.; Thyssen, C.; Schilling, O.; Hasenberg, M.; Pang, C. Biomolecule-corona formation confers resistance of bacteria to nanoparticle-induced killing: Implications for the design of improved nanoantibiotics. Biomaterials 2019, 192, 551–559. [Google Scholar] [CrossRef]
- Li, X.-Z.; Nikaido, H.; Williams, K.E. Silver-resistant mutants of Escherichia coli display active efflux of Ag+ and are deficient in porins. J. Bacteriol. 1997, 179, 6127–6132. [Google Scholar] [CrossRef]
- Hachicho, N.; Hoffmann, P.; Ahlert, K.; Heipieper, H.J. Effect of silver nanoparticles and silver ions on growth and adaptive response mechanisms of Pseudomonas putida mt-2. Fems Microbiol. Lett. 2014, 355, 71–77. [Google Scholar] [CrossRef]
- Feris, K.; Otto, C.; Tinker, J.; Wingett, D.; Punnoose, A.; Thurber, A.; Kongara, M.; Sabetian, M.; Quinn, B.; Hanna, C. Electrostatic interactions affect nanoparticle-mediated toxicity to gram-negative bacterium Pseudomonas aeruginosa PAO1. Langmuir 2009, 26, 4429–4436. [Google Scholar] [CrossRef] [PubMed]
- Stoimenov, P.K.; Klinger, R.L.; Marchin, G.L.; Klabunde, K.J. Metal oxide nanoparticles as bactericidal agents. Langmuir 2002, 18, 6679–6686. [Google Scholar] [CrossRef]
- Haggstrom, J.A.; Klabunde, K.J.; Marchin, G.L. Biocidal properties of metal oxide nanoparticles and their halogen adducts. Nanoscale 2010, 2, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Nabavizadeh, M.; Abbaszadegan, A.; Gholami, A.; Kadkhoda, Z.; Mirhadi, H.; Ghasemi, Y.; Safari, A.; Hemmateenejad, B.; Dorostkar, S.; Sharghi, H. Antibiofilm Efficacy of Positively Charged Imidazolium-Based Silver Nanoparticles in Enterococcus faecalis Using Quantitative Real-Time PCR. Jundishapur J. Microbiol. 2017, 10. [Google Scholar] [CrossRef]
- Abbaszadegan, A.; Ghahramani, Y.; Gholami, A.; Hemmateenejad, B.; Dorostkar, S.; Nabavizadeh, M.; Sharghi, H. The effect of charge at the surface of silver nanoparticles on antimicrobial activity against gram-positive and gram-negative bacteria: A preliminary study. J. Nanomater. 2015, 16, 53. [Google Scholar] [CrossRef]
- Kumariya, R.; Sood, S.K.; Rajput, Y.S.; Saini, N.; Garsa, A.K. Increased membrane surface positive charge and altered membrane fluidity leads to cationic antimicrobial peptide resistance in Enterococcus faecalis. Biochim. Et Biophys. Acta (Bba)-Biomembr. 2015, 1848, 1367–1375. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.; Hutchings, M.I.; Mascher, T. Cell envelope stress response in Gram-positive bacteria. Fems Microbiol. Rev. 2008, 32, 107–146. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, Y.-L.; Ambrose, K.D.; Zughaier, S.; Zhou, X.; Miller, Y.K.; Shafer, W.M.; Stephens, D.S. Cationic antimicrobial peptide resistance in Neisseria meningitidis. J. Bacteriol. 2005, 187, 5387–5396. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Gao, Y.; Lin, Z.; Yao, Z.; Zhang, W. The joint effects on Photobacterium phosphoreum of metal oxide nanoparticles and their most likely coexisting chemicals in the environment. Aquat. Toxicol. 2014, 154, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Bakshi, S.; He, Z.L.; Harris, W.G. Natural nanoparticles: Implications for environment and human health. Crit. Rev. Environ. Sci. Technol. 2015, 45, 861–904. [Google Scholar] [CrossRef]
- Meyer, D.E.; Curran, M.A.; Gonzalez, M.A. An examination of existing data for the industrial manufacture and use of nanocomponents and their role in the life cycle impact of nanoproducts. ACS Publications: 2009. Environ. Sci. Technol. 2009, 43, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Torabifard, M.; Arjmandi, R.; Rashidi, A.; Nouri, J.; Mohammadfam, I. Inherent health and environmental risk assessment of nanostructured metal oxide production processes. Environ. Monit. Assess. 2018, 190, 73. [Google Scholar] [CrossRef] [PubMed]
- Louie, S.M.; Pettibone, J.M. Research highlights: Probing adsorbed organic coatings on nanoparticle surfaces. Environ. Sci. Nano 2015, 2, 417–420. [Google Scholar] [CrossRef]
- Sharma, V.K.; Sayes, C.M.; Guo, B.; Pillai, S.; Parsons, J.G.; Wang, C.; Yan, B.; Ma, X. Interactions between silver nanoparticles and other metal nanoparticles under environmentally relevant conditions: A review. Sci. Total Environ. 2019, 653, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Louie, S.M.; Ma, R.; Lowry, G.V. Transformations of nanomaterials in the environment. In Frontiers of Nanoscience; Elsevier: Amsterdam, The Netherlands, 2014; Volume 7, pp. 55–87. [Google Scholar]
- Levard, C.m.; Mitra, S.; Yang, T.; Jew, A.D.; Badireddy, A.R.; Lowry, G.V.; Brown, G.E., Jr. Effect of chloride on the dissolution rate of silver nanoparticles and toxicity to E. coli. Environ. Sci. Technol. 2013, 47, 5738–5745. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.L.; Lowry, G.V.; Casman, E.A. Modeling nanosilver transformations in freshwater sediments. Environ. Sci. Technol. 2013, 47, 12920–12928. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Greden, K.; Alvarez, P.J.; Gregory, K.B.; Lowry, G.V. Adsorbed polymer and NOM limits adhesion and toxicity of nano scale zerovalent iron to E. coli. Environ. Sci. Technol. 2010, 44, 3462–3467. [Google Scholar] [CrossRef]
- Guo, J.; Gao, S.-H.; Lu, J.; Bond, P.L.; Verstraete, W.; Yuan, Z. Copper oxide nanoparticles induce lysogenic bacteriophage and metal-resistance genes in Pseudomonas aeruginosa PAO1. ACS Appl. Mater. Interfaces 2017, 9, 22298–22307. [Google Scholar] [CrossRef]
- Brown, N.L.; Misra, T.K.; Winnie, J.N.; Schmidt, A.; Seiff, M.; Silver, S. The nucleotide sequence of the mercuric resistance operons of plasmid R100 and transposon Tn501: Further evidence for mer genes which enhance the activity of the mercuric ion detoxification system. Mol. Gen. Genet. MGG 1986, 202, 143–151. [Google Scholar] [CrossRef]
- Saier, M.; Tam, R.; Reizer, A.; Reizer, J. Two novel families of bacterial membrane proteins concerned with nodulation, cell division and transport. Mol. Microbiol. 1994, 11, 841–847. [Google Scholar] [CrossRef]
- Johnson, J.M.; Church, G.M. Alignment and structure prediction of divergent protein families: Periplasmic and outer membrane proteins of bacterial efflux pumps1. J. Mol. Biol. 1999, 287, 695–715. [Google Scholar] [CrossRef] [PubMed]
- Kolaj-Robin, O.; Russell, D.; Hayes, K.A.; Pembroke, J.T.; Soulimane, T. Cation diffusion facilitator family: Structure and function. Febs Lett. 2015, 589, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Arguello, J.M.; Padilla-Benavides, T.; Collins, J.M. Transport Mechanism and Cellular Functions of Bacterial Cu (I)-ATPases. Met. Cells 2016, 155. [Google Scholar]
- Imran, M.; Das, K.R.; Naik, M.M. Co-selection of multi-antibiotic resistance in bacterial pathogens in metal and microplastic contaminated environments: An emerging health threat. Chemosphere 2018. [Google Scholar] [CrossRef] [PubMed]
- Randall, C.P.; Gupta, A.; Jackson, N.; Busse, D.; O’neill, A.J. Silver resistance in Gram-negative bacteria: A dissection of endogenous and exogenous mechanisms. J. Antimicrob. Chemother. 2015, 70, 1037–1046. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Kang, F.; Gao, Y.; Mao, X.; Hu, X. Sequestration of nanoparticles by an EPS matrix reduces the particle-specific bactericidal activity. Sci. Rep. 2016, 6, 21379. [Google Scholar] [CrossRef]
- Ellis, D.H.; Maurer-Gardner, E.I.; Sulentic, C.E.; Hussain, S.M. Silver nanoparticle antibacterial efficacy and resistance development in key bacterial species. Biomed. Phys. Eng. Express 2018, 5, 015013. [Google Scholar] [CrossRef]
- Contreras, F.; Vargas, E.; Jimenez, K.; Muñoz-Villagrán, C.; Figueroa, M.; Vásquez, C.; Arenas, F. Reduction of gold (III) and tellurium (IV) by Enterobacter cloacae MF01 results in nanostructure formation both in aerobic and anaerobic conditions. Front. Microbiol. 2018, 9, 3118. [Google Scholar] [CrossRef]
- Sari, A.P.; Nurhidayati, F.; Harsojo, E.S. Silver Nanoparticles Biotransforming Bacteria Isolated from Silver-Craft Waste. Trans. Sci. Technol. 2017, 4, 92–97. [Google Scholar]
- Ramos-Zúñiga, J.; Gallardo, S.; Martínez-Bussenius, C.; Norambuena, R.; Navarro, C.A.; Paradela, A.; Jerez, C.A. Response of the biomining Acidithiobacillus ferrooxidans to high cadmium concentrations. J. Proteom. 2019, 198, 132–144. [Google Scholar]
- Palomo-Siguero, M.; Gutiérrez, A.M.a.; Pérez-Conde, C.; Madrid, Y. Effect of selenite and selenium nanoparticles on lactic bacteria: A multi-analytical study. Microchem. J. 2016, 126, 488–495. [Google Scholar] [CrossRef]
- Chandrangsu, P.; Rensing, C.; Helmann, J.D. Metal homeostasis and resistance in bacteria. Nat. Rev. Microbiol. 2017, 15, 338. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, F.; Zhao, J.; Xu, Y.; Mao, D.; Zhu, X.; Luo, Y.; Alvarez, P. Bacterial exposure to ZnO nanoparticles facilitates horizontal transfer of antibiotic resistance genes. NanoImpact 2018, 10, 61–67. [Google Scholar] [CrossRef]
- Qiu, Z.; Shen, Z.; Qian, D.; Jin, M.; Yang, D.; Wang, J.; Zhang, B.; Yang, Z.; Chen, Z.; Wang, X. Effects of nano-TiO2 on antibiotic resistance transfer mediated by RP4 plasmid. Nanotoxicology 2015, 9, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Flores-Kim, J.; Darwin, A.J. Regulation of bacterial virulence gene expression by cell envelope stress responses. Virulence 2014, 5, 835–851. [Google Scholar] [CrossRef] [PubMed]
- Cronholm, P.; Karlsson, H.L.; Hedberg, J.; Lowe, T.A.; Winnberg, L.; Elihn, K.; Wallinder, I.O.; Möller, L. Intracellular uptake and toxicity of Ag and CuO nanoparticles: A comparison between nanoparticles and their corresponding metal ions. Small 2013, 9, 970–982. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhu, L.; Lin, D. Toxicity of ZnO nanoparticles to Escherichia coli: Mechanism and the influence of medium components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef]
- Cui, Y.; Zhao, Y.; Tian, Y.; Zhang, W.; Lü, X.; Jiang, X. The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials 2012, 33, 2327–2333. [Google Scholar] [CrossRef]
- Tang, S.; Zheng, J. Antibacterial Activity of Silver Nanoparticles: Structural Effects. Adv. Healthc. Mater. 2018, 1701503. [Google Scholar] [CrossRef]
- Yu, S.; Liu, J.; Yin, Y.; Shen, M. Interactions between engineered nanoparticles and dissolved organic matter: A review on mechanisms and environmental effects. J. Environ. Sci. 2018, 63, 198–217. [Google Scholar] [CrossRef]
- Carré, G.; Hamon, E.; Ennahar, S.; Estner, M.; Lett, M.-C.; Horvatovich, P.; Gies, J.-P.; Keller, V.; Keller, N.; Andre, P. TiO2 photocatalysis damages lipids and proteins in Escherichia coli. Appl. Environ. Microbiol. 2014, 80, 2573–2581. [Google Scholar] [CrossRef] [PubMed]
- Xiu, Z.-m.; Zhang, Q.-b.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yang, P.; Yuan, Z.; Guo, J. Aerobic condition enhances bacteriostatic effects of silver nanoparticles in aquatic environment: An antimicrobial study on Pseudomonas aeruginosa. Sci. Rep. 2017, 7, 7398. [Google Scholar] [CrossRef] [PubMed]
- Garuglieri, E.; Meroni, E.; Cattò, C.; Villa, F.; Cappitelli, F.; Erba, D. Effects of sub-lethal concentrations of silver nanoparticles on a simulated intestinal prokaryotic–eukaryotic interface. Front. Microbiol. 2018, 8, 2698. [Google Scholar] [CrossRef]
- Čáp, M.; Váchová, L.; Palková, Z. Reactive oxygen species in the signaling and adaptation of multicellular microbial communities. Oxidative Med. Cell. Longev. 2012, 2012. [Google Scholar] [CrossRef]
- Van der Heijden, J.; Vogt, S.L.; Reynolds, L.A.; Peña-Díaz, J.; Tupin, A.; Aussel, L.; Finlay, B.B. Exploring the redox balance inside gram-negative bacteria with redox-sensitive GFP. Free Radic. Biol. Med. 2016, 91, 34–44. [Google Scholar] [CrossRef]
- Loi, V.V.; Rossius, M.; Antelmann, H. Redox regulation by reversible protein S-thiolation in bacteria. Front. Microbiol. 2015, 6, 187. [Google Scholar] [CrossRef]
- Rochat, T.; Nicolas, P.; Delumeau, O.; Rabatinová, A.; Korelusova, J.; Leduc, A.; Bessieres, P.; Dervyn, E.; Krásný, L.; Noirot, P. Genome-wide identification of genes directly regulated by the pleiotropic transcription factor Spx in Bacillus subtilis. Nucleic Acids Res. 2012, 40, 9571–9583. [Google Scholar] [CrossRef]
- Tkachenko, A. Stress Responses of Bacterial Cells as Mechanism of Development of Antibiotic Tolerance. Appl. Biochem. Microbiol. 2018, 54, 108–127. [Google Scholar] [CrossRef]
- Gou, N.; Onnis-Hayden, A.; Gu, A.Z. Mechanistic toxicity assessment of nanomaterials by whole-cell-array stress genes expression analysis. Environ. Sci. Technol. 2010, 44, 5964–5970. [Google Scholar] [CrossRef]
- Sheng, Z.; Van Nostrand, J.D.; Zhou, J.; Liu, Y. The effects of silver nanoparticles on intact wastewater biofilms. Front. Microbiol. 2015, 6, 680. [Google Scholar] [CrossRef] [PubMed]
- Peulen, T.-O.; Wilkinson, K.J. Diffusion of nanoparticles in a biofilm. Environ. Sci. Technol. 2011, 45, 3367–3373. [Google Scholar] [CrossRef] [PubMed]
- Nevius, B.A.; Chen, Y.P.; Ferry, J.L.; Decho, A.W. Surface-functionalization effects on uptake of fluorescent polystyrene nanoparticles by model biofilms. Ecotoxicology 2012, 21, 2205–2213. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-C.; Wang, Y.-J.; Dang, F.; Zhou, D.-M. Mechanistic understanding of reduced AgNP phytotoxicity induced by extracellular polymeric substances. J. Hazard. Mater. 2016, 308, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Yu, C.-P.; Fernández, G.E.; Hu, Z. Interactions of nanosilver with Escherichia coli cells in planktonic and biofilm cultures. Water Res. 2010, 44, 6095–6103. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Mezgebe, B.; Hassan, A.A.; Sahle-Demessie, E.; Sorial, G.A.; Bennett-Stamper, C. Experimental and modeling studies of sorption of ceria nanoparticle on microbial biofilms. Bioresour. Technol. 2014, 161, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Sahle-Demessie, E.; Tadesse, H. Kinetics and equilibrium adsorption of nano-TiO2 particles on synthetic biofilm. Surf. Sci. 2011, 605, 1177–1184. [Google Scholar] [CrossRef]
- You, G.; Wang, P.; Hou, J.; Wang, C.; Xu, Y.; Miao, L.; Lv, B.; Yang, Y.; Liu, Z.; Zhang, F. Insights into the short-term effects of CeO2 nanoparticles on sludge dewatering and related mechanism. Water Res. 2017, 118, 93–103. [Google Scholar] [CrossRef]
- Yang, Y.; Alvarez, P.J. Sublethal concentrations of silver nanoparticles stimulate biofilm development. Environ. Sci. Technol. Lett. 2015, 2, 221–226. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, C.; Hou, J.; Wang, P.; You, G.; Miao, L. Effects of cerium oxide nanoparticles on bacterial growth and behaviors: Induction of biofilm formation and stress response. Environ. Sci. Pollut. Res. 2019, 1–12. [Google Scholar] [CrossRef]
- Finley, P.J.; Norton, R.; Austin, C.; Mitchell, A.; Zank, S.; Durham, P. Unprecedented silver resistance in clinically isolated Enterobacteriaceae: Major implications for burn and wound management. Antimicrob. Agents Chemother. 2015, 59, 4734–4741. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Lu, X.; Sun, P.; Hu, Z.; Wang, R.; Lou, C.; Han, J. Understanding the performance of microbial community induced by ZnO nanoparticles in enhanced biological phosphorus removal system and its recoverability. Bioresour. Technol. 2017, 225, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Yu, Y.; Chen, Z.; Jin, M.; Yang, D.; Zhao, Z.; Wang, J.; Shen, Z.; Wang, X.; Qian, D. Nanoalumina promotes the horizontal transfer of multiresistance genes mediated by plasmids across genera. Proc. Natl. Acad. Sci. USA 2012, 201107254. [Google Scholar] [CrossRef] [PubMed]
- Qayyum, S.; Khan, A.U. Nanoparticles vs. biofilms: A battle against another paradigm of antibiotic resistance. MedChemComm 2016, 7, 1479–1498. [Google Scholar] [CrossRef]
- Roe, D.; Karandikar, B.; Bonn-Savage, N.; Gibbins, B.; Roullet, J.-B. Antimicrobial surface functionalization of plastic catheters by silver nanoparticles. J. Antimicrob. Chemother. 2008, 61, 869–876. [Google Scholar] [CrossRef]
- Gomez-Carretero, S.; Nybom, R.; Richter-Dahlfors, A. Electroenhanced antimicrobial coating based on conjugated polymers with covalently coupled silver nanoparticles prevents Staphylococcus aureus biofilm formation. Adv. Healthc. Mater. 2017, 6, 1700435. [Google Scholar] [CrossRef]
- Applerot, G.; Lellouche, J.; Perkas, N.; Nitzan, Y.; Gedanken, A.; Banin, E. ZnO nanoparticle-coated surfaces inhibit bacterial biofilm formation and increase antibiotic susceptibility. Rsc Adv. 2012, 2, 2314–2321. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niño-Martínez, N.; Salas Orozco, M.F.; Martínez-Castañón, G.-A.; Torres Méndez, F.; Ruiz, F. Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles. Int. J. Mol. Sci. 2019, 20, 2808. https://doi.org/10.3390/ijms20112808
Niño-Martínez N, Salas Orozco MF, Martínez-Castañón G-A, Torres Méndez F, Ruiz F. Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles. International Journal of Molecular Sciences. 2019; 20(11):2808. https://doi.org/10.3390/ijms20112808
Chicago/Turabian StyleNiño-Martínez, Nereyda, Marco Felipe Salas Orozco, Gabriel-Alejandro Martínez-Castañón, Fernando Torres Méndez, and Facundo Ruiz. 2019. "Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles" International Journal of Molecular Sciences 20, no. 11: 2808. https://doi.org/10.3390/ijms20112808
APA StyleNiño-Martínez, N., Salas Orozco, M. F., Martínez-Castañón, G.-A., Torres Méndez, F., & Ruiz, F. (2019). Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles. International Journal of Molecular Sciences, 20(11), 2808. https://doi.org/10.3390/ijms20112808




