Oral Administration of Porphyromonas gingivalis, a Major Pathogen of Chronic Periodontitis, Promotes Resistance to Paclitaxel in Mouse Xenografts of Oral Squamous Cell Carcinoma
Abstract
1. Introduction
2. Results
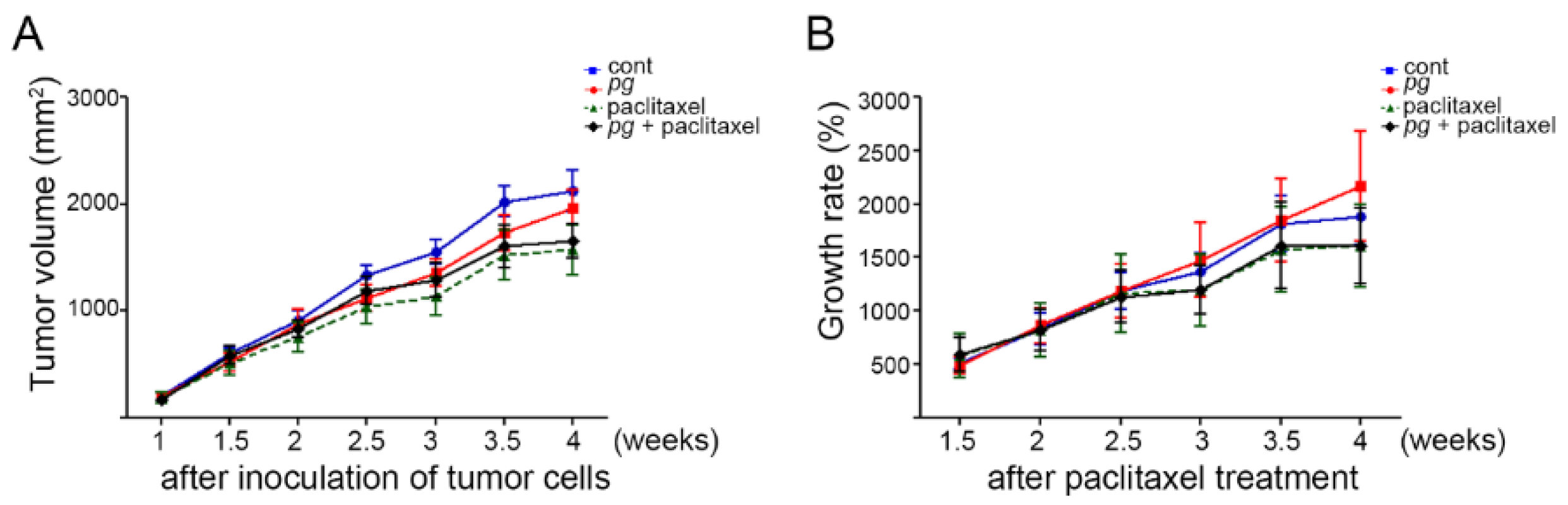
2.1. Oral Administration of P. gingivalis Reduces the Effect of Paclitaxel on OSCC Xenografts
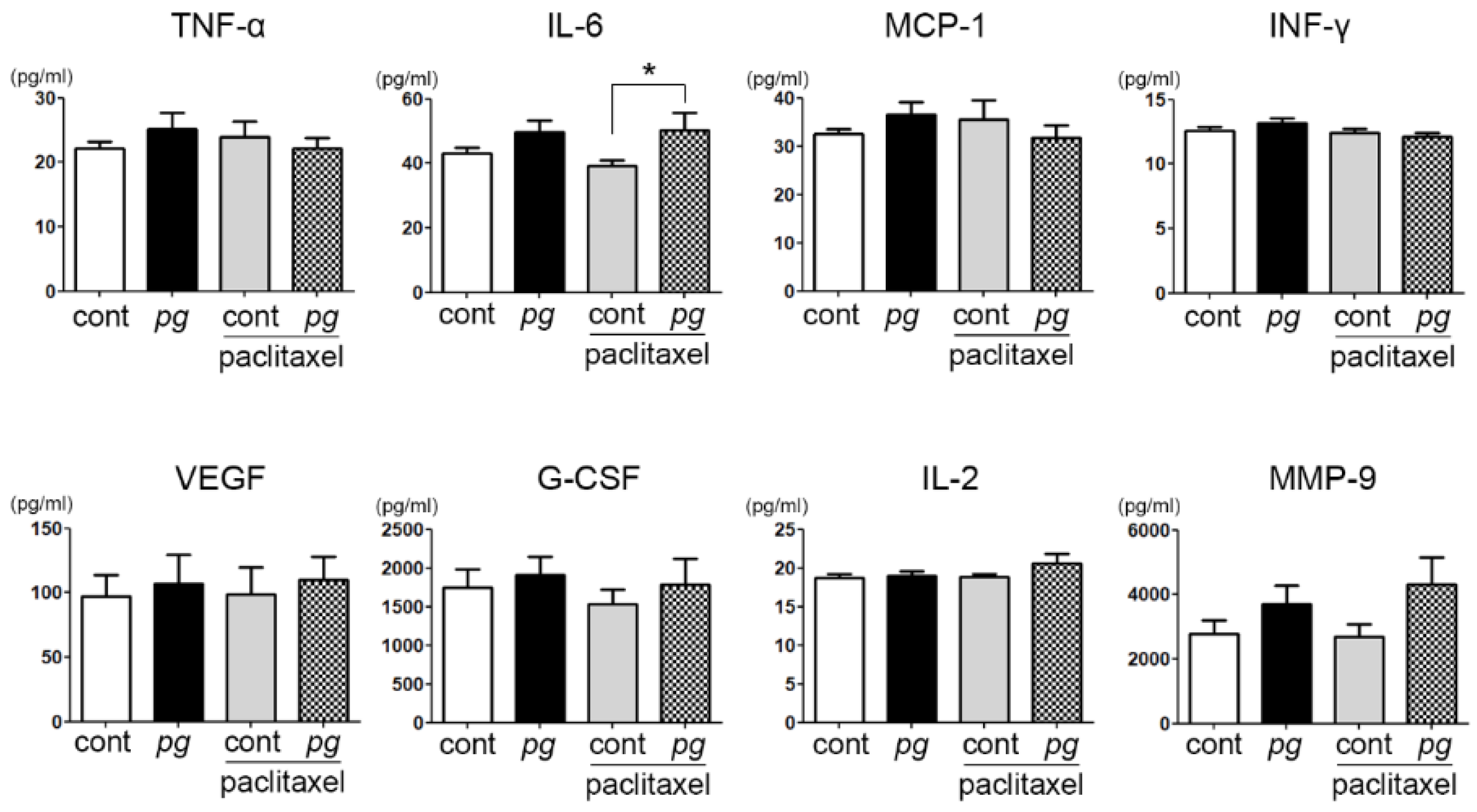
2.2. Increase in Serum IL-6 Level with P. gingivalis Administration in Mice
2.3. Suppression of Inflammation Mitigates the Resistance of OSCC Xenografts to Paclitaxel in P. gingivalis-Treated Mice
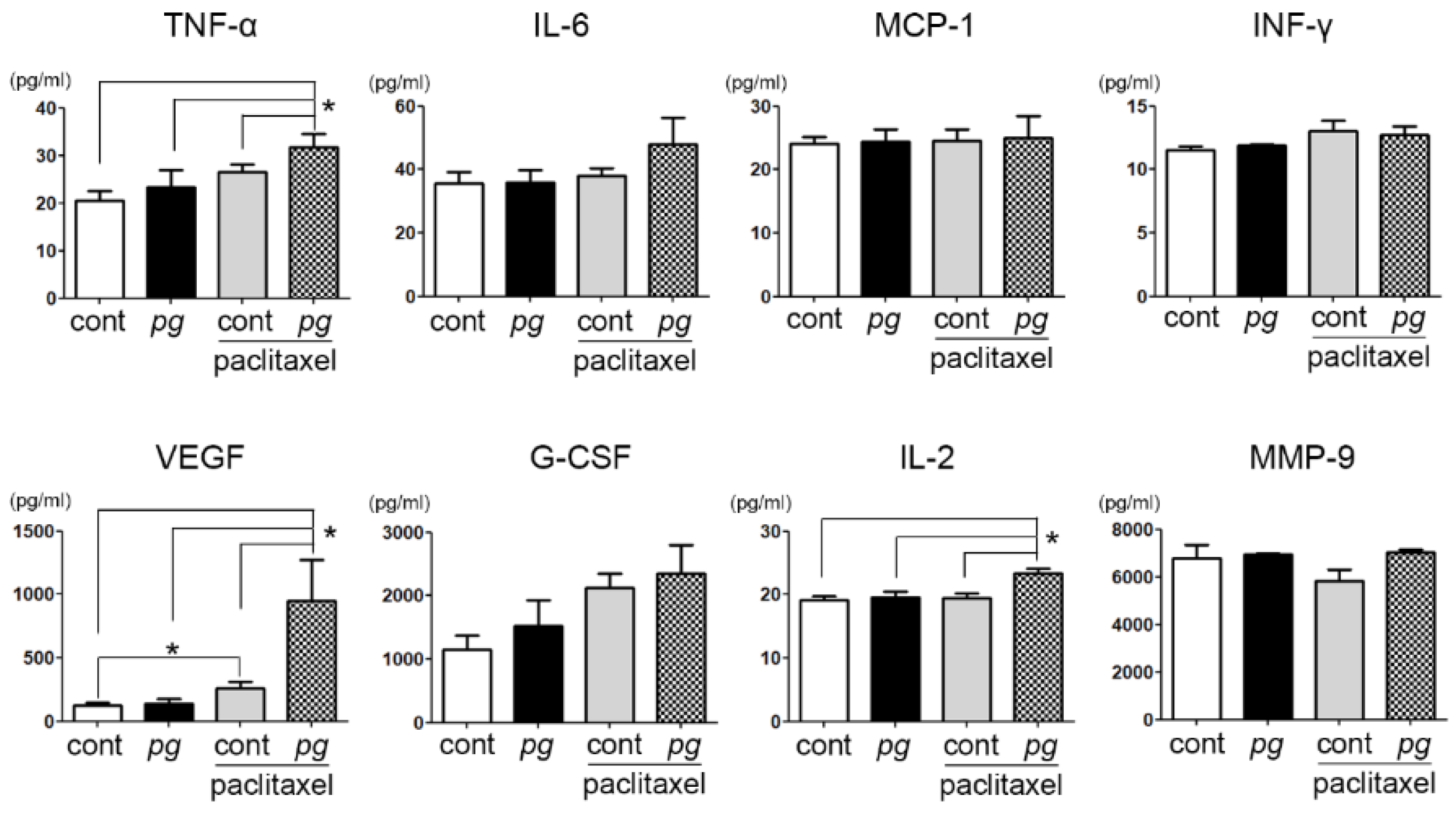
2.4. Prophylactic Use of Ibuprofen Significantly Suppresses Serum Levels of MCP-1 While Increasing VEGF and MMP-9 Expression
3. Discussion
4. Materials and Methods
4.1. Cell and Bacterial Cultures
4.2. Animals and Experimental Design
4.3. Histopathological Analysis
4.4. Multiplex Beads Assay
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| OSCC | Oral Squamous Cell Carcinoma |
| P. gingivalis | Porphyromonas gingivalis |
| IL-6 | Interleukin-6 |
| MMP | Matrix metalloproteinase |
References
- Rimessi, A.; Patergnani, S.; Ioannidi, E.; Pinton, P. Chemoresistance and Cancer-Related Inflammation: Two Hallmarks of Cancer Connected by an Atypical Link, PKCzeta. Front. Oncol. 2013, 3, 232. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, J.; Lin, P.C.; Zhou, B.P. Inflammation fuels tumor progress and metastasis. Curr. Pharm. Des. 2015, 21, 3032–3040. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Pappan, L.; Galliher-Beckley, A.; Shi, J. IL-1β promotes stemness and invasiveness of colon cancer cells through Zeb1 activation. Mol. Cancer 2012, 11, 87. [Google Scholar] [CrossRef]
- Bharti, R.; Dey, G.; Mandal, M. Cancer development, chemoresistance, epithelial to mesenchymal transition and stem cells: A snapshot of IL-6 mediated involvement. Cancer Lett. 2016, 375, 51–61. [Google Scholar] [CrossRef]
- Abdulkareem, A.A.; Shelton, R.M.; Landini, G.; Cooper, P.R.; Milward, M.R. Periodontal pathogens promote epithelial–mesenchymal transition in oral squamous carcinoma cells in vitro. Cell Adh. Migr. 2018, 12, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Bessède, E.; Staedel, C.; Acuña Amador, L.A.; Nguyen, P.H.; Chambonnier, L.; Hatakeyama, M.; Belleannée, G.; Mégraud, F.; Varon, C. Helicobacter pylori generates cells with cancer stem cell properties via epithelial–mesenchymal transition-like changes. Oncogene 2014, 33, 4123–4131. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Beaugerie, L.; Svrcek, M.; Seksik, P.; Bouvier, A.M.; Simon, T.; Allez, M.; Brixi, H.; Gornet, J.M.; Altwegg, R.; Beau, P.; et al. Risk of colorectal high-grade dysplasia and cancer in a prospective observational cohort of patients with inflammatory bowel disease. Gastroenterology 2013, 145, 166–175.e8. [Google Scholar] [CrossRef]
- Said, R.; Tsimberidou, A.M. Pharmacokinetic evaluation of vincristine for the treatment of lymphoid malignancies. Expert. Opin. Drug Metab. Toxicol. 2014, 10, 483–494. [Google Scholar] [CrossRef]
- Gao, G.; Chu, H.; Zhao, L.; Gui, T.; Xu, Q.; Shi, J. A meta-analysis of paclitaxel-based chemotherapies administered once every week compared with once every 3 weeks first-line treatment of advanced non-small-cell lung cancer. Lung Cancer 2012, 76, 380–386. [Google Scholar] [CrossRef]
- Pulte, D.; Brenner, H. Changes in survival in head and neck cancers in the late 20th and early 21st century: A period analysis. Oncologist 2010, 15, 994–1001. [Google Scholar] [CrossRef]
- Cooper, J.S.; Zhang, Q.; Pajak, T.F.; Forastiere, A.A.; Jacobs, J.; Saxman, S.B.; Kish, J.A.; Kim, H.E.; Cmelak, A.J.; Rotman, M.; et al. Long-term follow-up of the RTOG 9501/intergroup phase III trial: Postoperative concurrent radiation therapy and chemotherapy in high-risk squamous cell carcinoma of the head and neck. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 1198–1205. [Google Scholar] [CrossRef]
- Kiyota, N.; Tahara, M.; Fujii, M. Adjuvant treatment for post-operative head and neck squamous cell carcinoma. JPN J. Clin. Oncol. 2015, 45, 2–6. [Google Scholar] [CrossRef]
- Ferlazzo, N.; Curro, M.; Zinellu, A.; Caccamo, D.; Isola, G.; Ventura, V.; Carru, C.; Matarese, G.; Ientile, R. Influence of MTHFR Genetic Background on p16 and MGMT Methylation in Oral Squamous Cell Cancer. Int. J. Mol. Sci. 2017, 18, 724. [Google Scholar] [CrossRef]
- Tezal, M.; Sullivan, M.A.; Hyland, A.; Marshall, J.R.; Stoler, D.; Reid, M.E.; Loree, T.R.; Rigual, N.R.; Merzianu, M.; Hauck, L.; et al. Chronic periodontitis and the incidence of head and neck squamous cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 2406–2412. [Google Scholar] [CrossRef]
- Hujoel, P.P.; Drangsholt, M.; Spiekerman, C.; Weiss, N.S. An exploration of the periodontitis-cancer association. Ann Epidemiol. 2003, 13, 312–316. [Google Scholar] [CrossRef]
- Meyer, M.S.; Joshipura, K.; Giovannucci, E.; Michaud, D.S. A review of the relationship between tooth loss, periodontal disease, and cancer. Cancer Causes Control. 2008, 19, 895–907. [Google Scholar] [CrossRef]
- Osorio, R.; Alfonso-Rodriguez, C.A.; Osorio, E.; Medina-Castillo, A.L.; Alaminos, M.; Toledano-Osorio, M.; Toledano, M. Novel potential scaffold for periodontal tissue engineering. Clin. Oral. Investig. 2017, 21, 2695–2707. [Google Scholar] [CrossRef]
- Briguglio, F.; Briguglio, E.; Briguglio, R.; Cafiero, C.; Isola, G. Treatment of infrabony periodontal defects using a resorbable biopolymer of hyaluronic acid: A randomized clinical trial. Quintessence Int. 2013, 44, 231–240. [Google Scholar]
- Inaba, H.; Sugita, H.; Kuboniwa, M.; Iwai, S.; Hamada, M.; Noda, T.; Morisaki, I.; Lamont, R.J.; Amano, A. Porphyromonas gingivalis promotes invasion of oral squamous cell carcinoma through induction of proMMP9 and its activation. Cell Microbiol. 2014, 16, 131–145. [Google Scholar] [CrossRef]
- Sahingur, S.E.; Yeudall, W.A. Chemokine function in periodontal disease and oral cavity cancer. Front. Immunol. 2015, 6, 214. [Google Scholar] [CrossRef]
- Woo, B.H.; Kim, D.J.; Choi, J.I.; Kim, S.J.; Park, B.S.; Song, J.M.; Lee, J.H.; Park, H.R. Oral cancer cells sustainedly infected with Porphyromonas gingivalis exhibit resistance to Taxol and have higher metastatic potential. Oncotarget 2017, 8, 46981–46992. [Google Scholar] [CrossRef]
- Olsen, I.; Yilmaz, O. Possible role of Porphyromonas gingivalis in orodigestive cancers. J. Oral. Microbiol. 2019, 11, 1563410. [Google Scholar] [CrossRef]
- Wilensky, A.; Polak, D.; Awawdi, S.; Halabi, A.; Shapira, L.; Houri-Haddad, Y. Strain-dependent activation of the mouse immune response is correlated with Porphyromonas gingivalis-induced experimental periodontitis. J. Clin. Periodontol. 2009, 36, 915–921. [Google Scholar] [CrossRef]
- Mohammed, H.; Varoni, E.; Cochis, A.; Cordaro, M.; Gallenzi, P.; Patini, R.; Staderini, E.; Lajolo, C.; Rimondini, L.; Rocchetti, V. Oral Dysbiosis in Pancreatic Cancer and Liver Cirrhosis: A Review of the Literature. Biomedicines 2018, 6, 115. [Google Scholar] [CrossRef]
- Tsai, W.C.; Hsu, C.C.; Chang, H.N.; Lin, Y.C.; Lin, M.S.; Pang, J.H. Ibuprofen upregulates expressions of matrix metalloproteinase-1, -8, -9, and -13 without affecting expressions of types I and III collagen in tendon cells. J. Orthop. Res. 2010, 28, 487–491. [Google Scholar] [CrossRef]
- Matarese, G.; Isola, G.; Anastasi, G.; Cutroneo, G.; Cordasco, G.; Favaloro, A.; Vita, G.; Vermiglio, G.; Milardi, D.; Zizzari, V. Transforming Growth Factor β1 and Vascular Endothelial Growth Factor levels in the pathogenesis of periodontal disease. Eur. J. Inflamm. 2013, 11, 479–488. [Google Scholar] [CrossRef]
- Ha, N.H.; Park, D.G.; Woo, B.H.; Choi, J.I.; Park, B.S.; Kim, Y.D.; Lee, J.H.; Park, H.R. Porphyromonas gingivalis increases the invasiveness of oral cancer cells by upregulating IL-8 and MMPs. Cytokine 2016, 86, 64–72. [Google Scholar] [CrossRef]
- Patini, R.; Staderini, E.; Lajolo, C.; Lopetuso, L.; Mohammed, H.; Rimondini, L.; Rocchetti, V.; Franceschi, F.; Cordaro, M.; Gallenzi, P. Relationship between oral microbiota and periodontal disease: A systematic review. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5775–5788. [Google Scholar]
- Patini, R.; Gallenzi, P.; Spagnuolo, G.; Cordaro, M.; Cantiani, M.; Amalfitano, A.; Arcovito, A.; Callà, C.; Mingrone, G.; Nocca, G. Correlation Between Metabolic Syndrome, Periodontitis and Reactive Oxygen Species Production. A Pilot Study. Open Dent. J. 2017, 11, 621. [Google Scholar] [CrossRef]
- Kuboniwa, M.; Hasegawa, Y.; Mao, S.; Shizukuishi, S.; Amano, A.; Lamont, R.J.; Yilmaz, O.P. P. gingivalis accelerates gingival epithelial cell progression through the cell cycle. Microbes Infect. 2008, 10, 122–128. [Google Scholar] [CrossRef]
- Inaba, H.; Kuboniwa, M.; Bainbridge, B.; Yilmaz, O.; Katz, J.; Shiverick, K.T.; Amano, A.; Lamont, R.J. Porphyromonas gingivalis invades human trophoblasts and inhibits proliferation by inducing G1 arrest and apoptosis. Cell Microbiol. 2009, 11, 1517–1532. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Li, L.; Guo, X.; Jin, X.; Sun, W.; Zhang, X.; Xu, R.C. Interleukin-6 signaling regulates anchorage-independent growth, proliferation, adhesion and invasion in human ovarian cancer cells. Cytokine 2012, 59, 228–236. [Google Scholar] [CrossRef]
- You, W.; Tang, Q.; Zhang, C.; Wu, J.; Gu, C.; Wu, Z.; Li, X. IL-26 promotes the proliferation and survival of human gastric cancer cells by regulating the balance of STAT1 and STAT3 activation. PLoS ONE 2013, 8, e63588. [Google Scholar] [CrossRef]
- Cho, T.J.; Wee, S.W.; Woo, V.H.; Choi, J.I.; Kim, S.J.; Shin, H.I.; Lee, J.H.; Park, H.R. Porphyromonas gingivalis-induced autophagy suppresses cell proliferation through G1 arrest in oral cancer cells. Arch. Oral Biol. 2014, 59, 370–378. [Google Scholar] [CrossRef]
- Puig, I.; Tenbaum, S.P.; Chicote, I.; Arques, O.; Martinez-Quintanilla, J.; Cuesta-Borras, E.; Ramirez, L.; Gonzalo, P.; Soto, A.; Aguilar, S.; et al. TET2 controls chemoresistant slow-cycling cancer cell survival and tumor recurrence. J. Clin. Invest. 2018, 128, 3887–3905. [Google Scholar] [CrossRef]

| Control | Mean ± SD (pg/mL) | p Value | |
| Ibuprofen (−) | Ibuprofen (+) | ||
| TNF-α | 22.100 ± 3.665 | 20.625 ± 5.528 | 0.529 |
| MCP-1 *** | 32.450 ± 3.685 | 24.125 ± 2.800 | <0.001 |
| VEGF | 97.150 ± 52.341 | 122.563 ± 72.128 | 0.315 |
| IL-2 | 18.700 ± 1.494 | 19.125 ± 1.808 | 0.573 |
| IL-6 | 43.000 ± 6.429 | 35.625 ± 9.768 | 0.101 |
| IFN-γ * | 12.600 ± 0.843 | 11.500 ± 0.926 | 0.034 |
| G-CSF | 1750.950 ± 743.944 | 1156.500 ± 637.470 | 0.092 |
| MMP-9 *** | 2767.100 ± 1407.780 | 6794.313 ± 1534.532 | <0.001 |
| Pg | Mean ± SD (pg/mL) | p Value | |
| Ibuprofen (−) | Ibuprofen (+) | ||
| TNF-α | 25.200 ± 8.011 | 23.250 ± 10.306 | 0.657 |
| MCP-1 ** | 36.600 ± 7.863 | 24.375 ± 5.553 | 0.006 |
| VEGF *** | 106.800 ± 71.311 | 138.188 ± 105.538 | <0.001 |
| IL-2 | 18.900 ± 2.283 | 19.563 ± 2.665 | 0.633 |
| IL-6 * | 49.850 ± 10.970 | 35.750 ± 11.668 | 0.021 |
| IFN-γ ** | 13.200 ± 1.033 | 11.875 ± 0.354 | 0.006 |
| G-CSF | 1909.000 ± 750.013 | 1523.063 ± 1140.469 | 0.400 |
| MMP-9 *** | 3697.850 ± 1861.348 | 6952.750 ± 124.409 | <0.001 |
| Paclitaxel | Mean ± SD (pg/mL) | p Value | |
| Ibuprofen (−) | Ibuprofen (+) | ||
| TNF-α | 24.000 ± 7.557 | 26.500 ± 4.375 | 0.420 |
| MCP-1 * | 35.550 ± 12.348 | 24.563 ± 5.039 | 0.016 |
| VEGF ** | 99.100 ± 64.781 | 258.000 ± 161.402 | 0.009 |
| IL-2 | 18.800 ± 1.229 | 19.375 ± 2.134 | 0.460 |
| IL-6 | 39.300 ± 5.599 | 38.062 ± 6.774 | 0.408 |
| IFN-γ | 12.450 ± 0.832 | 13.000 ± 2.507 | 0.522 |
| G-CSF | 1538.350 ± 591.133 | 2126.875 ± 626.152 | 0.083 |
| MMP-9 ** | 2694.600 ± 1204.875 | 5833.687 ± 1307.932 | 0.001 |
| Pg + Paclitaxel | Mean ± SD (pg/mL) | p Value | |
| Ibuprofen (−) | Ibuprofen (+) | ||
| TNF-α ** | 22.100 ± 5.607 | 31.750 ± 8.013 | 0.008 |
| MCP-1 | 31.750 ± 8.244 | 25.063 ± 9.563 | 0.274 |
| VEGF ** | 109.900 ± 57.058 | 951.500 ± 899.341 | 0.001 |
| IL-2 * | 20.600 ± 3.864 | 23.375 ± 2.066 | 0.043 |
| IL-6 | 50.300 ± 17.385 | 47.938 ± 23.501 | 0.696 |
| IFN-γ | 12.100 ± 1.101 | 12.750 ± 1.832 | 0.364 |
| G-CSF | 1782.600 ± 1050.864 | 2348.000 ± 1268.862 | 0.316 |
| MMP-9 * | 4310.300 ± 2671.717 | 7027.625 ± 360.735 | 0.011 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.M.; Woo, B.H.; Lee, J.H.; Yoon, S.; Cho, Y.; Kim, Y.-D.; Park, H.R. Oral Administration of Porphyromonas gingivalis, a Major Pathogen of Chronic Periodontitis, Promotes Resistance to Paclitaxel in Mouse Xenografts of Oral Squamous Cell Carcinoma. Int. J. Mol. Sci. 2019, 20, 2494. https://doi.org/10.3390/ijms20102494
Song JM, Woo BH, Lee JH, Yoon S, Cho Y, Kim Y-D, Park HR. Oral Administration of Porphyromonas gingivalis, a Major Pathogen of Chronic Periodontitis, Promotes Resistance to Paclitaxel in Mouse Xenografts of Oral Squamous Cell Carcinoma. International Journal of Molecular Sciences. 2019; 20(10):2494. https://doi.org/10.3390/ijms20102494
Chicago/Turabian StyleSong, Jae Min, Bok Hee Woo, Ji Hye Lee, Sanggyeong Yoon, Youngseuk Cho, Yong-Deok Kim, and Hae Ryoun Park. 2019. "Oral Administration of Porphyromonas gingivalis, a Major Pathogen of Chronic Periodontitis, Promotes Resistance to Paclitaxel in Mouse Xenografts of Oral Squamous Cell Carcinoma" International Journal of Molecular Sciences 20, no. 10: 2494. https://doi.org/10.3390/ijms20102494
APA StyleSong, J. M., Woo, B. H., Lee, J. H., Yoon, S., Cho, Y., Kim, Y.-D., & Park, H. R. (2019). Oral Administration of Porphyromonas gingivalis, a Major Pathogen of Chronic Periodontitis, Promotes Resistance to Paclitaxel in Mouse Xenografts of Oral Squamous Cell Carcinoma. International Journal of Molecular Sciences, 20(10), 2494. https://doi.org/10.3390/ijms20102494






