Cytoskeleton, Transglutaminase and Gametophytic Self-Incompatibility in the Malinae (Rosaceae)
Abstract
1. Introduction: The Molecular Basis of S-RNase-Based Gametophytic Self-Incompatibility in the Malinae
2. Pollen Tube Growth and How It Is Impacted by Self-Incompatibility
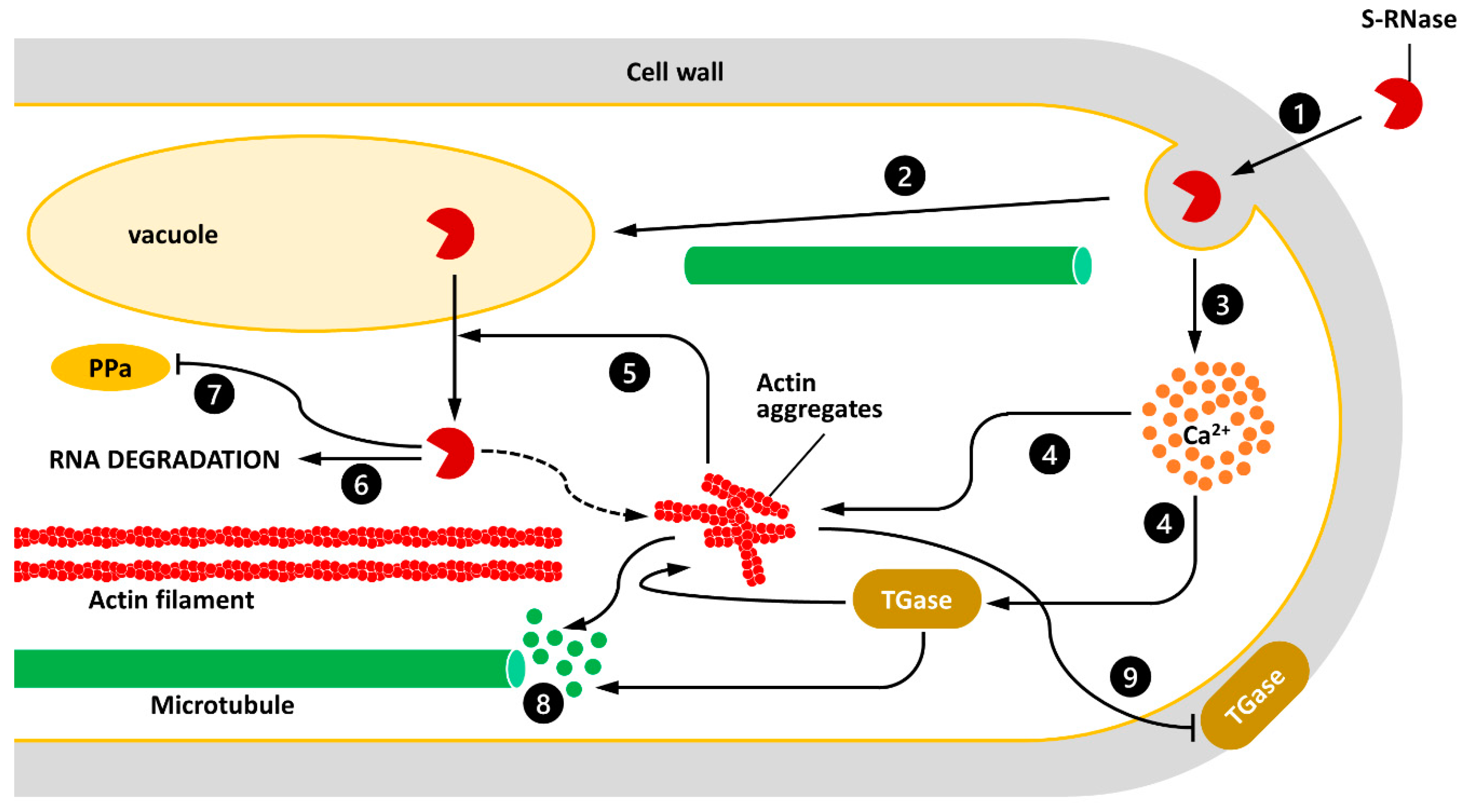
3. The TGase-Cytoskeleton Interplay as a Crucial Player of the SI Process
4. Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Barrett, S.C.; Harder, L.D. The ecology of mating and its evolutionary consequences in seed plants. Ann. Rev. Ecol. Evol. Syst. 2017, 48, 135–157. [Google Scholar] [CrossRef]
- Barrett, S.C. Mating strategies in flowering plants: The outcrossing-selfing paradigm and beyond. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003, 358, 991–1004. [Google Scholar] [PubMed]
- Ashman, T.L.; Knight, T.M.; Steets, J.A.; Amarasekare, P.; Burd, M.; Campbell, D.R.; Dudash, M.R.; Johnston, M.O.; Mazer, S.J.; Mitchell, R.J. Pollen limitation of plant reproduction: Ecological and evolutionary causes and consequences. Ecology 2004, 85, 2408–2421. [Google Scholar]
- Qu, H.; Guan, Y.; Wang, Y.; Zhang, S. PLC-mediated signaling pathway in pollen tubes regulates the gametophytic self-incompatibility of Pyrus species. Front. Plant Sci. 2017, 8, 1164. [Google Scholar] [CrossRef]
- Serrano, I.; Romero-Puertas, M.C.; Sandalio, L.M.; Olmedilla, A. The role of reactive oxygen species and nitric oxide in programmed cell death associated with self-incompatibility. J. Exp. Bot. 2015, 66, 2869–2876. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Kubo, K.I.; Takayama, S. Non-self-and self-recognition models in plant self-incompatibility. Nat. Plants 2016, 2, 16130. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.J.; de Graaf, B.H.J.; Hadjiosif, N.; Perry, R.M.; Poulter, N.S.; Osman, K.; Vatovec, S.; Harper, A.; Franklin, F.C.; Franklin-Tong, V.E. Identification of the pollen self-incompatibility determinant in Papaver rhoeas. Nature 2009, 459, 992–995. [Google Scholar] [CrossRef] [PubMed]
- Kear, P.J.; McClure, B. How did Flowering Plants Learn to Avoid Blind Date Mistakes? Springer: New York, NY, USA, 2012; pp. 108–123. [Google Scholar]
- De Franceschi, P.; Dondini, L.; Sanzol, J. Molecular bases and evolutionary dynamics of self-incompatibility in the Pyrinae (Rosaceae). J. Exp. Bot. 2012, 63, 4015–4032. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, P.; Pierantoni, L.; Dondini, L.; Grandi, M.; Sanzol, J.; Sansavini, S. Cloning and mapping multiple S-locus F-box genes in European pear (Pyrus communis L.). Tree Gen. Genomes 2011, 7, 231–240. [Google Scholar] [CrossRef]
- Li, W.; Meng, D.; Gu, Z.; Yang, Q.; Yuan, H.; Li, Y.; Chen, Q.; Yu, J.; Liu, C.; Li, T. Apple S-RNase triggers inhibition of tRNA aminoacylation by interacting with a soluble inorganic pyrophosphatase in growing self-pollen tubes in vitro. New Phytol. 2018, 218, 579–593. [Google Scholar]
- Liu, Z.Q.; Xu, G.H.; Zhang, S.L. Pyrus pyrifolia stylar S-RNase induces alterations in the actin cytoskeleton in self-pollen and tubes in vitro. Protoplasma 2007, 232, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Meng, D.; Gu, Z.; Li, W.; Chen, Q.; Li, Y.; Yuan, H.; Yu, J.; Liu, C.; Li, T. Apple S-RNase interacts with an actin-binding protein, MdMVG, to reduce pollen tube growth by inhibiting its actin-severing activity at the early stage of self-pollination induction. Plant J. 2018, 95, 41–56. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Gao, Y.; Zhou, H.; Chen, J.; Wu, J.; Zhang, S. Apoplastic calmodulin promotes self-incompatibility pollen tube growth by enhancing calcium influx and reactive oxygen species concentration in Pyrus pyrifolia. Plant Cell Rep. 2014, 33, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Eaves, D.J.; Flores-Ortiz, C.; Haque, T.; Lin, Z.; Teng, N.; Franklin-Tong, V.E. Self-incompatibility in Papaver: Advances in integrating the signalling network. Biochem. Soc. Trans. 2014, 42, 370–376. [Google Scholar] [PubMed]
- Thomas, S.G.; Huang, S.; Li, S.; Staiger, C.J.; Franklin-Tong, V.E. Actin depolymerization is sufficient to induce programmed cell death in self-incompatible pollen. J. Cell Biol. 2006, 174, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Poulter, N.S.; Vatovec, S.; Franklin-Tong, V.E. Microtubules are a target for self-incompatibility signaling in Papaver pollen. Plant Physiol. 2008, 146, 1358–1367. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Jiang, Y.; Chang, M.; Liu, X.; Zhang, R.; Huang, S. Organization and regulation of the actin cytoskeleton in the pollen tube. Front Plant Sci. 2015, 5, 786. [Google Scholar] [PubMed]
- Fu, Y. The cytoskeleton in the pollen tube. Curr. Opin. Plant Biol. 2015, 28, 111–119. [Google Scholar] [CrossRef]
- Cai, G.; Parrotta, L.; Cresti, M. Organelle trafficking, the cytoskeleton, and pollen tube growth. J. Int. Plant Biol. 2015, 57, 63–78. [Google Scholar] [CrossRef]
- Cole, R.A.; Fowler, J.E. Polarized growth: Maintaining focus on the tip. Curr. Opin. Plant Biol. 2006, 9, 579–588. [Google Scholar] [CrossRef]
- Gu, F.; Nielsen, E. Targeting and Regulation of Cell Wall Synthesis During Tip Growth in Plants. J. Int. Plant Biol. 2013, 55, 835–846. [Google Scholar]
- Hepler, P.K.; Rounds, C.M.; Winship, L.J. Control of cell wall extensibility during pollen tube growth. Mol. Plant 2013, 6, 998–1017. [Google Scholar] [PubMed]
- Chebli, Y.; Kaneda, M.; Zerzour, R.; Geitmann, A. The cell wall of the Arabidopsis pollen tube—Spatial distribution, recycling, and network formation of polysaccharides. Plant Physiol. 2012, 160, 1940–1955. [Google Scholar] [CrossRef] [PubMed]
- Kroeger, J.H.; Zerzour, R.; Geitmann, A. Regulator or driving force? The role of turgor pressure in oscillatory plant cell growth. PLoS ONE 2011, 6, e18549. [Google Scholar] [CrossRef] [PubMed]
- Kaya, H.; Nakajima, R.; Iwano, M.; Kanaoka, M.M.; Kimura, S.; Takeda, S.; Kawarazaki, T.; Senzaki, E.; Hamamura, Y.; Higashiyama, T.; et al. Ca2+-activated reactive oxygen species production by Arabidopsis RbohH and RbohJ is essential for proper pollen tube tip growth. Plant Cell 2014, 26, 1069–1080. [Google Scholar] [CrossRef]
- Sewelam, N.; Kazan, K.; Schenk, P.M. Global plant stress signaling: Reactive oxygen species at the cross-road. Front Plant Sci. 2016, 7, 187. [Google Scholar] [CrossRef]
- Wang, C.L.; Wu, J.; Xu, G.H.; Gao, Y.B.; Chen, G.; Wu, J.Y.; Wu, H.Q.; Zhang, S.L. S-RNase disrupts tip-localized reactive oxygen species and induces nuclear DNA degradation in incompatible pollen tubes of Pyrus pyrifolia. J. Cell Sci. 2010, 123, 4301–4309. [Google Scholar] [CrossRef]
- Aloisi, I.; Cai, G.; Tumiatti, V.; Minarini, A.; Del Duca, S.D. Natural polyamines and synthetic analogues modify the growth and the morphology of Pyrus communis pollen tubes affecting ROS levels and causing cell death. Plant Sci. 2015, 239, 92–105. [Google Scholar]
- Speranza, A.; Crinelli, R.; Scoccianti, V.; Geitmann, A. Reactive oxygen species are involved in pollen tube initiation in kiwifruit. Plant Biol. 2012, 14, 64–76. [Google Scholar] [CrossRef]
- Daher, F.B.; Geitmann, A. Actin is involved in pollen tube tropism through redefining the spatial targeting of secretory vesicles. Traffic 2011, 12, 1537–1551. [Google Scholar] [CrossRef]
- Mori, I.C.; Schroeder, J.I. Reactive oxygen species activation of plant Ca2+ channels. A signaling mechanism in polar growth, hormone transduction, stress signaling, and hypothetically mechanotransduction. Plant Physiol. 2004, 135, 702–708. [Google Scholar] [PubMed]
- Griffin, M.; Casadio, R.; Bergamini, C.M. Transglutaminases: nature’s biological glues. Biochem. J. 2002, 368, 377–396. [Google Scholar] [PubMed]
- Del Duca, S.; Serafini-Fracassini, D. Transglutaminases of higher, lower plants and fungi. Prog. Exp. Tumor Res. 2005, 38, 223–247. [Google Scholar] [PubMed]
- Aloisi, I.; Cai, G.; Serafini-Fracassini, D.; Del, D.S. Transglutaminase as polyamine mediator in plant growth and differentiation. Amino Acids 2016, 48, 2467–2478. [Google Scholar] [CrossRef] [PubMed]
- Del Duca, S.; Creus, J.A.; D’Orazi, D.; Dondini, L.; Bregoli, A.M.; Serafini-Fracassini, D. Tuber vegetative stages and cell cycle in Helianthus tuberosus: Protein pattern and their modification by spermidine. J. Plant Physiol. 2000, 156, 17–25. [Google Scholar] [CrossRef]
- Serafini-Fracassini, D.; Della Mea, M.; Tasco, G.; Casadio, R.; Del Duca, S. Plant and animal transglutaminases: Do similar functions imply similar structures? Amino Acids 2009, 36, 643–657. [Google Scholar] [CrossRef]
- Serafini-Fracassini, D.; Del Duca, S. Transglutaminases: Widespread cross-linking enzymes in plants. Ann. Bot. 2008, 102, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Del Duca, S.; Faleri, C.; Iorio, R.A.; Cresti, M.; Serafini-Fracassini, D.; Cai, G. Distribution of transglutaminase in pear pollen tubes in relation to cytoskeleton and membrane dynamics. Plant Physiol. 2013, 161, 1706–1721. [Google Scholar] [CrossRef]
- Di Sandro, A.; Del Duca, S.; Verderio, E.; Hargreaves, A.J.; Scarpellini, A.; Cai, G.; Cresti, M.; Faleri, C.; Iorio, R.A.; Shigehisa, H.; et al. An extracellular transglutaminase is required for apple pollen tube growth. Biochem. J. 2010, 429, 261–271. [Google Scholar]
- Klausner, R.D.; Donaldson, J.G.; Lippincott-Schwartz, J. Brefeldin A: Insights into the control of membrane traffic and organelle structure. J. Cell Biol. 1992, 116, 1071–1080. [Google Scholar]
- Del Duca, S.; Cai, G.; Di Sandro, A.; Serafini-Fracassini, D. Compatible and self-incompatible pollination in Pyrus communis displays different polyamine levels and transglutaminase activity. Amino Acids 2010, 38, 659–667. [Google Scholar] [CrossRef]
- Gentile, A.; Antognoni, F.; Iorio, R.A.; Distefano, G.; Las, C.G.; La, M.S.; Serafini-Fracassini, D.; Del Duca, S. Polyamines and transglutaminase activity are involved in compatible and self-incompatible pollination of Citrus grandis. Amino Acids 2012, 42, 1025–1035. [Google Scholar] [CrossRef]
- Della Mea, M.; Serafini-Fracassini, D.; Del Duca, S. Programmed cell death: Similarities and differences in animals and plants. A flower paradigm. Amino Acids 2007, 33, 395–404. [Google Scholar]
- Fésus, L.; Szondy, Z. Transglutaminase 2 in the balance of cell death and survival. FEBS Lett. 2005, 579, 3297–3302. [Google Scholar] [CrossRef]
- Del Duca, S.; Serafini-Fracassini, D.; Bonner, P.L.; Cresti, M.; Cai, G. Effects of post-translational modifications catalyzed by pollen transglutaminase on the functional properties of microtubules and actin filaments. Biochem. J. 2009, 418, 651–664. [Google Scholar] [CrossRef] [PubMed]
- Di Sandro, A.; Serafini-Fracassini, D.; Del Duca, S.; Della Mea, M.; Faleri, C.; Cai, G.; De Franceschi, P.; Dondini, L.; Sansavini, S. Pollen transglutaminase in pear self incompatibility and relationship with S-RNases and S-allele variability. Acta Hortic. 2008, 800, 423–430. [Google Scholar] [CrossRef]
- Del Duca, S.; Bregoli, A.M.; Bergamini, C.; Serafini-Fracassini, D. Transglutaminase-catalyzed modification of cytoskeletal proteins by polyamines during the germination of Malus domestica pollen. Sex. Plant. Reprod. 1997, 10, 89–95. [Google Scholar] [CrossRef]
- Cai, G.; Faleri, C.; Cresti, M.; Di Sandro, A.; Serafini-Fracassini, D.; Del Duca, S. Pollen tube cytoskeleton, transglutaminases and self-incompatibility. Acta Hortic. 2012, 967, 149–156. [Google Scholar] [CrossRef]
- Poulter, N.S.; Bosch, M.; Franklin-Tong, V. Proteins implicated in mediating self-incompatibility-induced alterations to the actin cytoskeleton of Papaver pollen. Ann. Bot. 2011, 108, 659–675. [Google Scholar] [CrossRef]
- Meng, D.; Gu, Z.; Yuan, H.; Wang, A.; Li, W.; Yang, Q.; Zhu, Y.; Li, T. The microtubule cytoskeleton and pollen tube Golgi-vesicle system are required for in vitro S-RNase internalization and gametic self incompatibility in apple. Plant Cell Physiol. 2014, 55, 977–989. [Google Scholar] [CrossRef]
- Bosch, M.; Franklin-Tong, V.E. Self-incompatibility in Papaver: Signalling to trigger PCD in incompatible pollen. J. Exp. Bot. 2008, 59, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Idilli, A.I.; Morandini, P.; Onelli, E.; Rodighiero, S.; Caccianiga, M.; Moscatelli, A. Microtubule depolymerization affects endocytosis and exocytosis in the tip and influences endosome movement in tobacco pollen tubes. Mol. Plant 2013, 6, 1109–1130. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Miller, G.; Morales, J.; Shulaev, V.; Torres, M.A.; Mittler, R. Respiratory burst oxidases: the engines of ROS signaling. Curr. Opin. Plant Biol. 2011, 2011 14, 691–699. [Google Scholar] [CrossRef]
- Qu, H.Y.; Zhang, Z.; Wu, F.; Wang, Y. The role of Ca(2+) and Ca(2+) channels in the gametophytic self-incompatibility of Pyrus pyrifolia. Cell Calcium 2016, 60, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Potocky, M.; Pejchar, P.; Gutkowska, M.; Jimenez-Quesada, M.J.; Potocka, A.; Alche, J.D.; Kost, B.; Zarsky, V. NADPH oxidase activity in pollen tubes is affected by calcium ions, signaling phospholipids and Rac/Rop GTPases. J. Plant Physiol. 2012, 169, 1654–1663. [Google Scholar] [CrossRef]
- Roldán, J.A.; Rojas, H.J.; Goldraij, A. Disorganization of F-actin cytoskeleton precedes vacuolar disruption in pollen tubes during the in vivo self-incompatibility response in Nicotiana alata. Ann. Bot. 2012, 110, 787–795. [Google Scholar] [CrossRef]
- Roldán, J.; Rojas, H.J.; Goldraij, A. In vitro inhibition of incompatible pollen tubes in Nicotiana alata involves the uncoupling of the F-actin cytoskeleton and the endomembrane trafficking system. Protoplasma 2015, 252, 63–75. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Del Duca, S.; Aloisi, I.; Parrotta, L.; Cai, G. Cytoskeleton, Transglutaminase and Gametophytic Self-Incompatibility in the Malinae (Rosaceae). Int. J. Mol. Sci. 2019, 20, 209. https://doi.org/10.3390/ijms20010209
Del Duca S, Aloisi I, Parrotta L, Cai G. Cytoskeleton, Transglutaminase and Gametophytic Self-Incompatibility in the Malinae (Rosaceae). International Journal of Molecular Sciences. 2019; 20(1):209. https://doi.org/10.3390/ijms20010209
Chicago/Turabian StyleDel Duca, Stefano, Iris Aloisi, Luigi Parrotta, and Giampiero Cai. 2019. "Cytoskeleton, Transglutaminase and Gametophytic Self-Incompatibility in the Malinae (Rosaceae)" International Journal of Molecular Sciences 20, no. 1: 209. https://doi.org/10.3390/ijms20010209
APA StyleDel Duca, S., Aloisi, I., Parrotta, L., & Cai, G. (2019). Cytoskeleton, Transglutaminase and Gametophytic Self-Incompatibility in the Malinae (Rosaceae). International Journal of Molecular Sciences, 20(1), 209. https://doi.org/10.3390/ijms20010209





