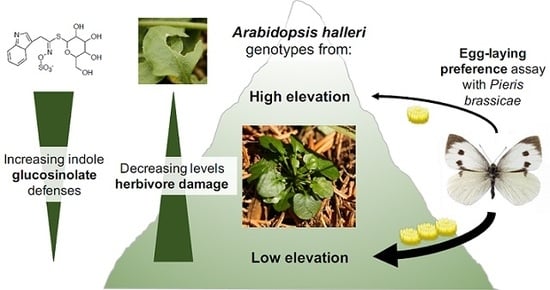
Divergence in Glucosinolate Profiles between High- and Low-Elevation Populations of Arabidopsis halleri Correspond to Variation in Field Herbivory and Herbivore Behavioral Preferences
Abstract
1. Introduction
2. Results
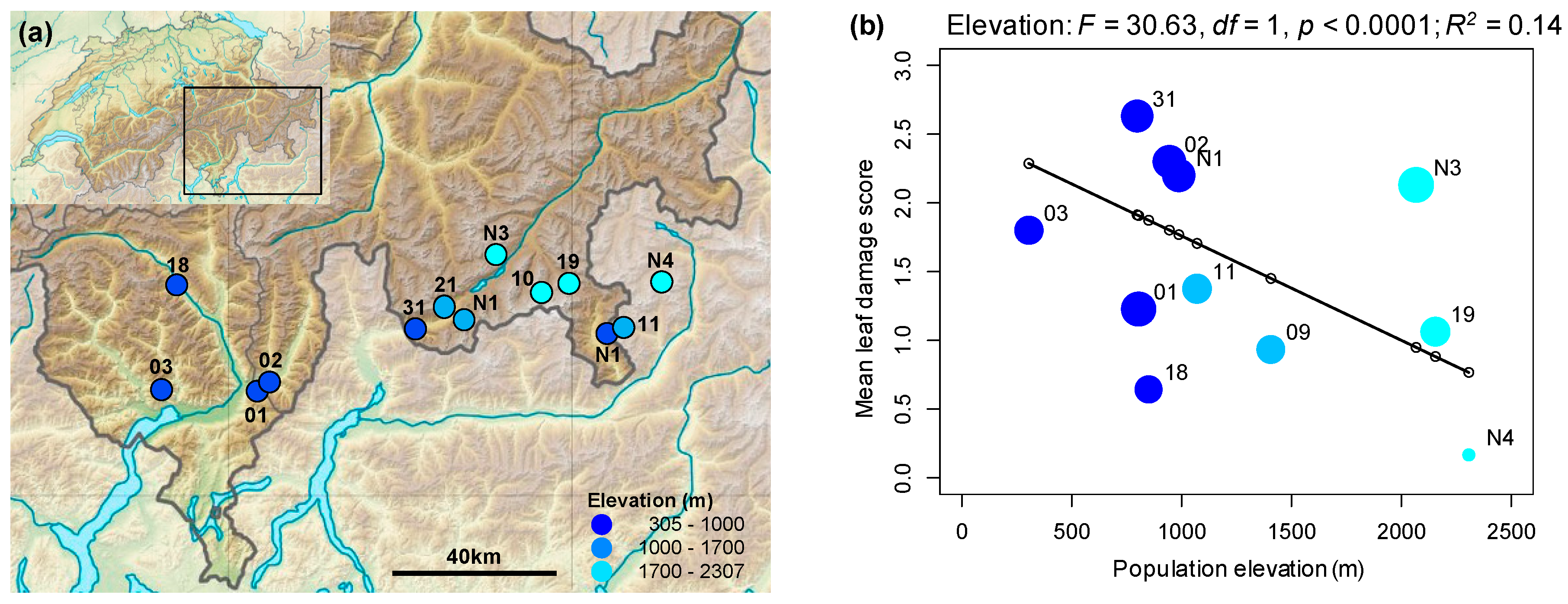
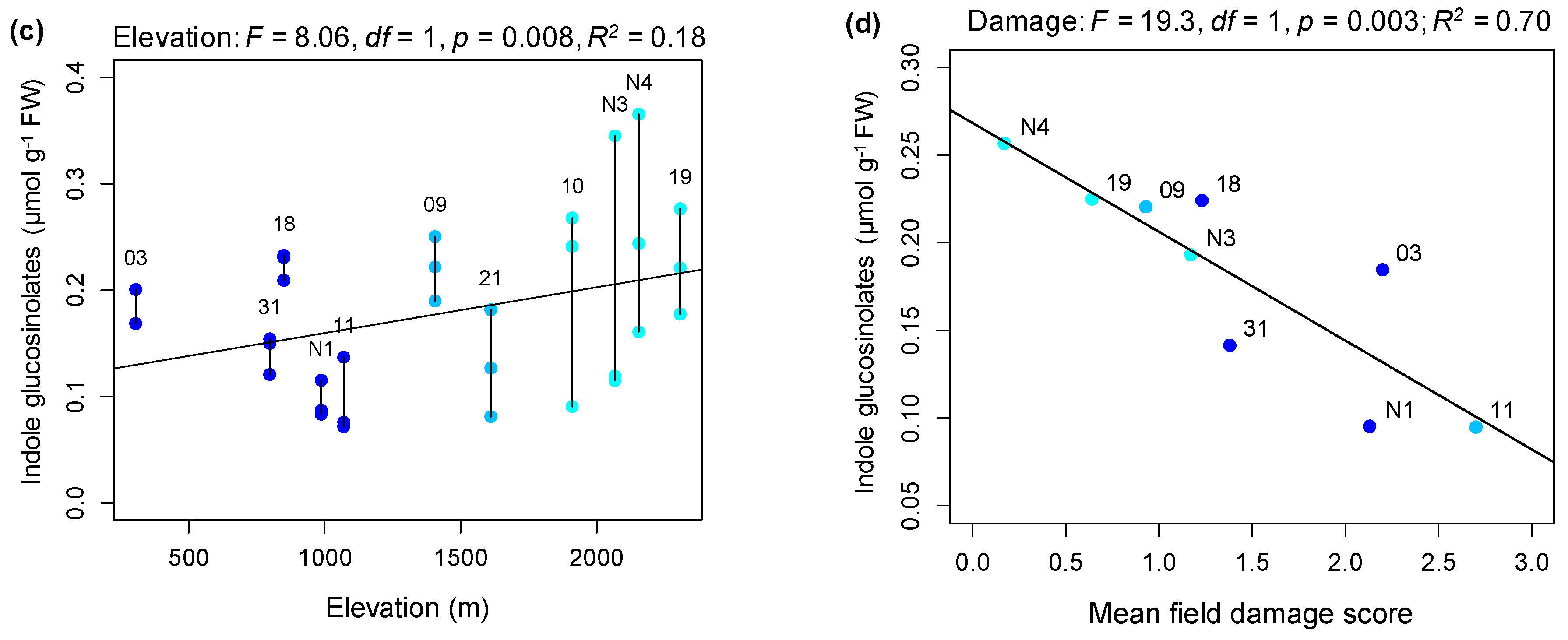
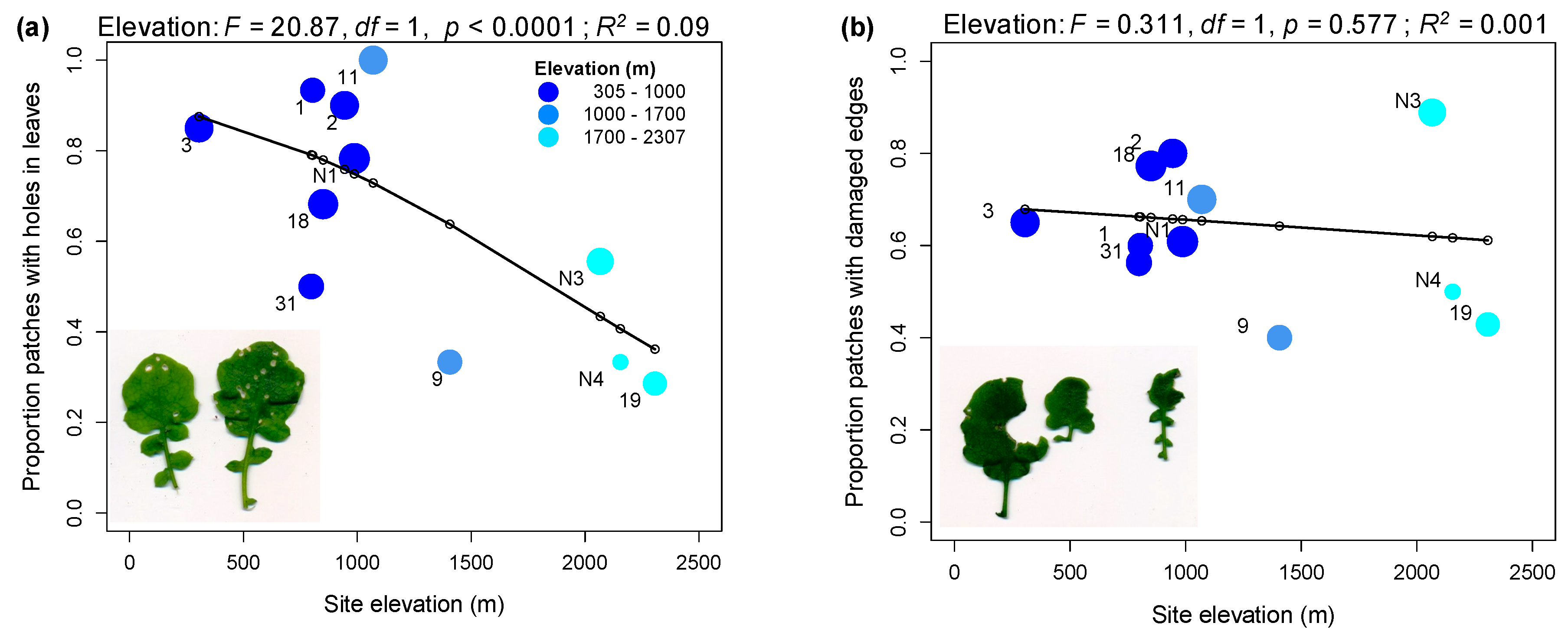
2.1. Rates of Herbivore Damage in the Field Decline with Elevation and Are Associated with Increasing Investment in Indole Glucosinolate Production
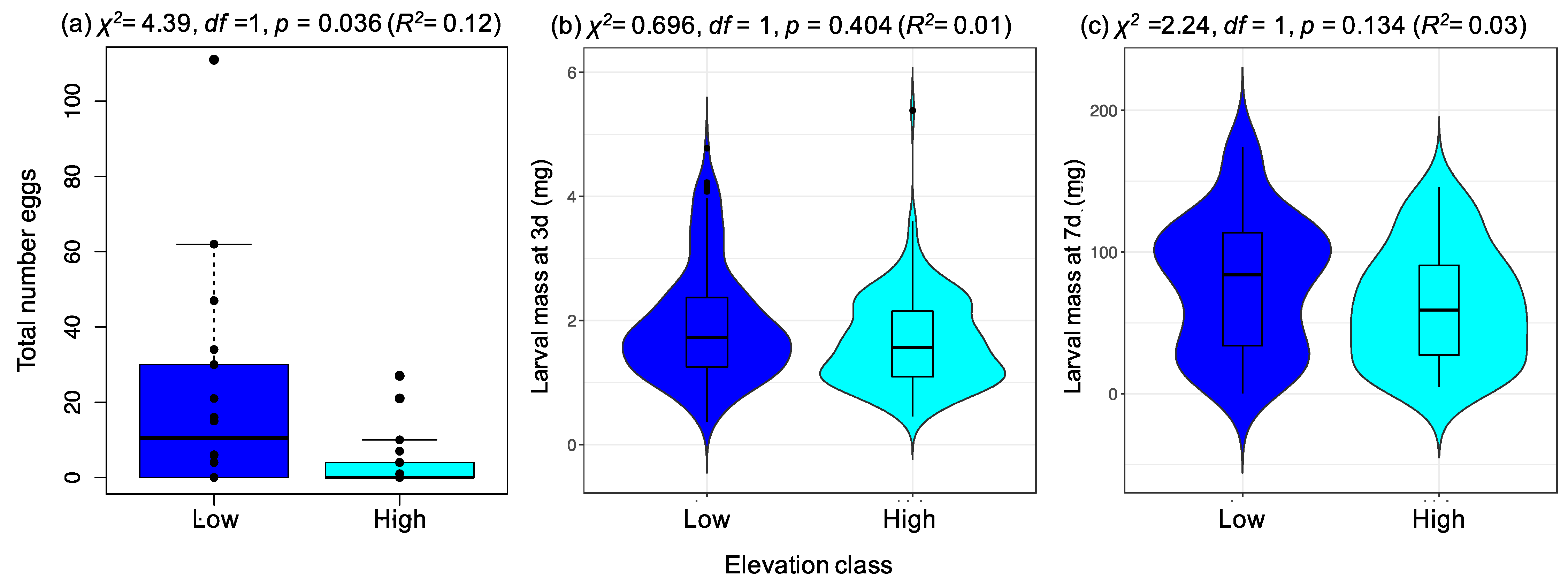
2.2. A Specialist Herbivore (Pieris brassicae) Prefers Plants from Low-Elevation Populations, despite No Associated Enhancement of Offspring Performance
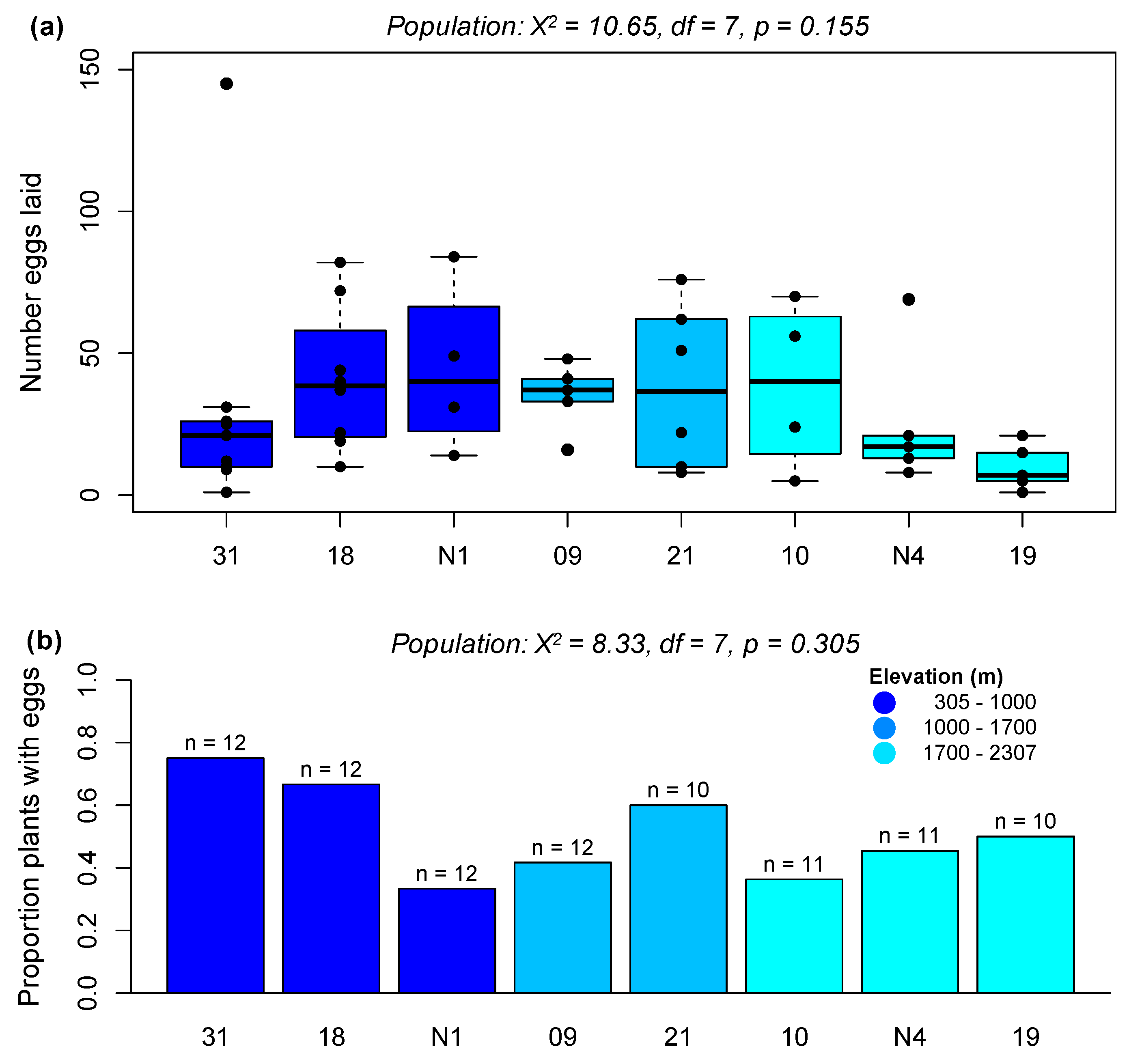
2.2.1. Preference Experiment 1: Assessing Population-Level Effects on Preference
2.2.2. Preference Experiment 2: Assessing Elevation Effects on P. brassicae Preference
3. Discussion
3.1. Rates of Herbivore Damage in the Field Decline with Elevation and Are Associated with Increasing Investment in Indole Glucosinolate Production
3.2. A Specialist Herbivore Prefers Plants from Low-Elevation Populations despite No Associated Enhancement of Offspring Performance
4. Materials and Methods
4.1. Establishing Parental Plant Stock Population in the Greenhouse
4.2. Assessing Variation in Herbivore Damage across Field Populations
4.3. Assessing the Relationship between Constitutive Glucosinolates in the Greenhouse and Elevation
4.4. Screening Populations of A. halleri for Variation in Butterfly Oviposition Preference
4.4.1. Preference Experiment 1: Assessing Population-Level Effects on Preference
4.4.2. Preference Experiment 2: Assessing Elevation Effects on P. brassicae Preference
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Urban, M.C.; Bocedi, G.; Hendry, A.P.; Mihoub, J.B.; Pe’er, G.; Singer, A.; Bridle, J.R.; Crozier, L.G.; De Meester, L.; Godsoe, W.; et al. Improving the forecast for biodiversity under climate change. Science 2016, 353, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Moreira, X.; Petry, W.K.; Mooney, K.A.; Rasmann, S.; Abdala-Roberts, L. Elevational gradients in plant defences and insect herbivory: Recent advances in the field and prospects for future research. Ecography 2018, 41, 1485–1496. [Google Scholar] [CrossRef]
- Rasmann, S.; Pellissier, L.; Defossez, E.; Jactel, H.; Kunstler, G.; Bailey, J.K. Climate-driven change in plant-insect interactions along elevation gradients. Funct. Ecol. 2014, 28, 46–54. [Google Scholar] [CrossRef]
- Descombes, P.; Marchon, J.; Pradervand, J.-N.; Bilat, J.; Guisan, A.; Rasmann, S.; Pellissier, L.; Whitney, K. Community-level plant palatability increases with elevation as insect herbivore abundance declines. J. Ecol. 2017, 105, 142–151. [Google Scholar] [CrossRef]
- Pellissier, L.; Fiedler, K.; Ndribe, C.; Dubuis, A.; Pradervand, J.N.; Guisan, A.; Rasmann, S. Shifts in species richness, herbivore specialization, and plant resistance along elevation gradients. Ecol. Evol. 2012, 2, 1818–1825. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.; Widmer, A.; Mescher, M.C.; De Moraes, C.M. Variation in growth and defense traits among populations of an alpine plant may facilitate responses to climate change. J. Ecol. 2018, submitted. [Google Scholar]
- Mauricio, R. Cost of resistance to natural enemies in field populations of the annual plant Arabidopsis thaliana. Am. Nat. 1998, 151, 20–28. [Google Scholar] [CrossRef]
- Züst, T.; Agrawal, A.A. Trade-offs between plant growth and defense against insect herbivory: A mechanistic synthesis. Annu. Rev. Plant Biol. 2017, 68, 513–534. [Google Scholar] [CrossRef]
- Züst, T.; Rasmann, S.; Agrawal, A.A. Growth-defense tradeoffs for two major anti-herbivore traits of the common milkweed Asclepias syriaca. Oikos 2015, 124, 1404–1415. [Google Scholar] [CrossRef]
- Anstett, D.N.; Nunes, K.A.; Baskett, C.; Kotanen, P.M. Sources of controversy surrounding latitudinal patterns in herbivory and defense. Trends Ecol. Evol. 2016, 31, 789–802. [Google Scholar] [CrossRef]
- Abdala-Roberts, L.; Moreira, X.; Rasmann, S.; Parra-Tabla, V.; Mooney, K.A.; Lau, J. Test of biotic and abiotic correlates of latitudinal variation in defences in the perennial herb Ruellia nudiflora. J. Ecol. 2016, 104, 580–590. [Google Scholar] [CrossRef]
- Flanders, K.L.; Hawkes, J.G.; Radcliffe, E.B.; Lauer, F.I. Insect resistance in potatoes: Sources, evolutionary relationships, morphological and chemical defenses, and ecogeographical associations. Euphytica 1992, 61, 83–111. [Google Scholar] [CrossRef]
- Hahn, P.G.; Maron, J.L. A framework for predicting intraspecific variation in plant defense. Trends Ecol. Evol. 2016, 31, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Donohue, K.; Jacquemyn, H. Local adaptation and plasticity of Erysimum capitatum to altitude: Its implications for responses to climate change. J. Ecol. 2013, 101, 796–805. [Google Scholar] [CrossRef]
- Körner, C.; Neumayer, M.; Menendez-Riedl, S.P.; Smeets-Scheel, A. Functional morphology of mountain plants. Flora 1989, 182, 353–383. [Google Scholar] [CrossRef]
- Defossez, E.; Pellissier, L.; Rasmann, S. The unfolding of plant growth form-defence syndromes along elevation gradients. Ecol. Lett. 2018, 21, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Kergunteuil, A.; Descombes, P.; Glauser, G.; Pellissier, L.; Rasmann, S. Plant physical and chemical defence variation along elevation gradients: A functional trait-based approach. Oecologia 2018, 187, 561–571. [Google Scholar] [CrossRef]
- Halbritter, A.H.; Fior, S.; Keller, I.; Billeter, R.; Edwards, P.; Holderegger, R.; Karrenberg, S.; Pluess, A.R.; Widmer, A.; Alexander, J.M. Trait differentiation and adaptation of plants along elevation gradients. J. Evol. Biol. 2018, 31, 784–800. [Google Scholar] [CrossRef]
- Pellissier, L.; Roger, A.; Bilat, J.; Rasmann, S. High elevation Plantago lanceolata plants are less resistant to herbivory than their low elevation conspecifics: Is it just temperature? Ecography 2014, 37, 950–959. [Google Scholar] [CrossRef]
- Rasmann, S.; Buri, A.; Gallot-Lavallée, M.; Joaquim, J.; Purcell, J.; Pellissier, L.; Heard, M. Differential allocation and deployment of direct and indirect defences by Vicia sepium along elevation gradients. J. Ecol. 2014, 102, 930–938. [Google Scholar] [CrossRef]
- Dostalek, T.; Rokaya, M.B.; Marsik, P.; Rezek, J.; Skuhrovec, J.; Pavela, R.; Munzbergova, Z. Trade-off among different anti-herbivore defence strategies along an altitudinal gradient. AoB PLANTS 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Rokaya, M.B.; Dostálek, T.; Münzbergová, Z. Plant-herbivore interactions along elevational gradient: Comparison of field and common garden data. Acta Oecol. 2016, 77, 168–175. [Google Scholar] [CrossRef]
- Salgado, A.L.; Suchan, T.; Pellissier, L.; Rasmann, S.; Ducrest, A.L.; Alvarez, N. Differential phenotypic and genetic expression of defence compounds in a plant-herbivore interaction along elevation. R. Soc. Open Sci. 2016, 3, 160226. [Google Scholar] [CrossRef] [PubMed]
- Moreira, X.; Mooney, K.A.; Rasmann, S.; Petry, W.K.; Carrillo-Gavilán, A.; Zas, R.; Sampedro, L.; Novotny, V. Trade-offs between constitutive and induced defences drive geographical and climatic clines in pine chemical defences. Ecol. Lett. 2014, 17, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Pellissier, L.; Moreira, X.; Danner, H.; Serrano, M.; Salamin, N.; van Dam, N.M.; Rasmann, S.; Bartomeus, I. The simultaneous inducibility of phytochemicals related to plant direct and indirect defences against herbivores is stronger at low elevation. J. Ecol. 2016, 104, 1116–1125. [Google Scholar] [CrossRef]
- Abdala-Roberts, L.; Rasmann, S.; Berny-Mier, Y.T.J.C.; Covelo, F.; Glauser, G.; Moreira, X. Biotic and abiotic factors associated with altitudinal variation in plant traits and herbivory in a dominant oak species. Am. J. Bot. 2016, 103, 2070–2078. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Tonsor, S.J.; Traw, M.B. A geographic cline in leaf salicylic acid with increasing elevation in Arabidopsis thaliana. Plant Signal. Behav. 2015, 10, e992741. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Kitzberger, T.; Chaneton, E.J. Environmental and genetic control of insect adundance and herbivory along a forest elevational gradient. Oecologia 2011, 167, 117–129. [Google Scholar] [CrossRef]
- Horgan, F.G.; Quiring, D.T.; Lagnaoui, A.; Pelletier, Y. Effects of altitude of origin on trichome-mediated anti-herbivore resistance in wild Andean potatoes. Flora 2009, 204, 49–62. [Google Scholar] [CrossRef]
- Scheidel, U.; Bruelheide, H. Altitudinal differences in herbivory on montane Compositae species. Oecologia 2001, 129, 75–86. [Google Scholar] [CrossRef]
- Gripenberg, S.; Mayhew, P.J.; Parnell, M.; Roslin, T. A meta-analysis of preference-performance relationships in phytophagous insects. Ecol. Lett. 2010, 13, 383–393. [Google Scholar] [CrossRef]
- Kelly, C.A.; Bowers, D.A. Preference and performance of generalist and specialist herbivores on chemically defended host plants. Ecol. Entomol. 2016, 41, 308–316. [Google Scholar] [CrossRef]
- Sato, Y.; Kudoh, H. Tests of associational defence provided by hairy plants for glabrous plants of Arabidopsis halleri subsp. gemmifera against insect herbivores. Ecol. Entomol. 2015, 40, 269–279. [Google Scholar]
- Sletvold, N.; Huttunen, P.; Handley, R.; Kärkkäinen, K.; Ågren, J. Cost of trichome production and resistance to a specialist insect herbivore in Arabidopsis lyrata. Evol. Ecol. 2010, 24, 1307–1319. [Google Scholar] [CrossRef]
- Fei, M.; Harvey, J.A.; Yin, Y.; Gols, R. Oviposition preference for young plants by the large cabbage butterfly (Pieris brassicae) does not strongly correlate with caterpillar performance. J. Chem. Ecol. 2017, 43, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Reudler Talsma, J.H.; Biere, A.; Harvey, J.A.; van Nouhuys, S. Oviposition cues for a specialist butterfly—Plant chemistry and size. J. Chem.Ecol. 2008, 34, 1202–1212. [Google Scholar] [CrossRef]
- Van Leur, H.; Vet, L.E.M.; van der Putten, W.H.; van Dam, N.M. Barbarea vulgaris glucosinolate phenotypes differentially affect performance and preference of two different species of Lepidopteran herbivores. J. Chem. Ecol. 2008, 34, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Fortuna, T.M.; Woelke, J.B.; Hordijk, C.A.; Jansen, J.J.; van Dam, N.M.; Vet, L.E.M.; Harvey, J.A. A tritrophic approach to the preference-performance hypothesis involving an exotic and a native plant. Biol. Invasions 2013, 15, 2387–2401. [Google Scholar] [CrossRef]
- Newton, E.L.; Bullock, J.M.; Hodgson, D.J. Glucosinolate polymorphism in wild cabbage (Brassica oleracea) influences the structure of herbivore communities. Oecologia 2009, 160, 63–76. [Google Scholar] [CrossRef]
- Poelman, E.H.; van Dam, N.M.; van Loon, J.J.A.; Vet, L.E.M.; Dicke, M. Chemical diversity in Brassica oleraceae affects biodiversity of insect herbivores. Ecology 2009, 90, 1863–1877. [Google Scholar] [CrossRef]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Windsor, A.J.; Reichelt, M.; Figuth, A.; Svatos, A.; Kroymann, J.; Kliebenstein, D.J.; Gershenzon, J.; Mitchell-Olds, T. Geographic and evolutionary diversification of glucosinolates among near relatives of Arabidopsis thaliana (Brassicaceae). Phytochemistry 2005, 66, 1321–1333. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.E.; Huang, X.C.; Hansen, C.I.C.; Cipollini, D.; Orgaard, M.; Matthes, A.; Geu-Flores, F.; Koch, M.A.; Agerbirk, N. Glucosinolate diversity within a phylogenetic framework of the tribe Cardamineae (Brassicaceae) unraveled with HPLC-MS/MS and NMR-based analytical distinction of 70 desulfoglucosinolates. Phytochemistry 2016, 132, 33–56. [Google Scholar] [CrossRef] [PubMed]
- Agerbirk, N.; Olsen, C.E. Glucosinolate structures in evolution. Phytochemistry 2012, 77, 16–45. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.; de Vos, M.; Sun, J.Y.; Sonderby, I.E.; Halkier, B.A.; Wittstock, U.; Jander, G. Differential effects of indole and aliphatic glucosinolates on lepidopteran herbivores. J. Chem. Ecol. 2010, 36, 905–913. [Google Scholar] [CrossRef]
- Giamoustaris, A.; Mithen, R. The effect of modifying the glucosinolate content of leaves of oilseed rape (Brassica napus ssp. oleifera) on its interaction with specialist and generalist pests. Ann. Appl. Bot. 1995, 126, 347–363. [Google Scholar]
- Lankau, R.A. Specialist and generalist herbivores exert opposing selection on a chemical defense. New Phytol. 2007, 175, 176–184. [Google Scholar] [CrossRef]
- Jeschke, V.; Gershenzon, J.; Vassão, D.G. Insect Detoxification of Glucosinolates and Their Hydrolysis Products. Adv. Botan. Res. 2016, 80, 199–245. [Google Scholar]
- Van Loon, J.J.A.; Blaakmeer, A.; Griepink, F.C.; van Beek, T.A.; Schoonhoven, L.M.; de Groot, A. Leaf surface compound from Brassica oleracea (Cruciferae) induces oviposition by Pieris brassicae (Lepidoptera: Pieridae). Chemoecology 1992, 3, 39–44. [Google Scholar] [CrossRef]
- Sun, J.Y.; Sonderby, I.E.; Halkier, B.A.; Jander, G.; de Vos, M. Non-volatile intact indole glucosinolates are host recognition cues for ovipositing Plutella xylostella. J. Chem. Ecol. 2009, 35, 1427–1436. [Google Scholar] [CrossRef]
- Bidart-Bouzat, M.G.; Kliebenstein, D.J. Differential levels of insect herbivory in the field associated with genotypic variation in glucosinolates in Arabidopsis thaliana. J. Chem. Ecol. 2008, 34, 1026–1037. [Google Scholar] [CrossRef] [PubMed]
- Smallegange, R.C.; van Loon, J.J.; Blatt, S.E.; Harvey, J.A.; Agerbirk, N.; Dicke, M. Flower vs. leaf feeding by Pieris brassicae: Glucosinolate-rich flower tissues are preferred and sustain higher growth rate. J. Chem. Ecol. 2007, 33, 1831–1844. [Google Scholar] [CrossRef] [PubMed]
- Gloss, A.D.; Brachi, B.; Feldmann, M.J.; Groen, S.C.; Bartoli, C.; Gouzy, J.; LaPlante, E.R.; Meyer, C.G.; Pyon, H.S.; Rogan, S.C.; et al. Genetic variants affecting plant size and chemcial defenses jointly shape herbivory in Arabidopsis. BioArXiv 2017. [Google Scholar] [CrossRef]
- Robin, A.H.K.; Hossain, M.R.; Park, J.I.; Kim, H.R.; Nou, I.S. Glucosinolate Profiles in Cabbage Genotypes Influence the Preferential Feeding of Diamondback Moth (Plutella xylostella). Front. Plant Sci. 2017, 8, 1244. [Google Scholar] [CrossRef]
- De Vos, M.; Kriksunov, K.L.; Jander, G. Indole-3-acetonitrile production from indole glucosinolates deters oviposition by Pieris rapae. Plant Physiol. 2008, 146, 916–926. [Google Scholar] [CrossRef] [PubMed]
- Gols, R.; Wagenaar, R.; Bukovinszky, T.; van Dam, N.M.; Dicke, M.; Bullock, J.M.; Harvey, J.A. Genetic variation in defense chemistry in wild cabbages affects herbivores and endoparasitoids. Ecology 2008, 89, 1616–1626. [Google Scholar] [CrossRef] [PubMed]
- Stolpe, C.; Krämer, U.; Müller, C. Heavy metal (hyper)accumulation in leaves of Arabidopsis halleri is accompanied by a reduced performance of herbivores and shifts in leaf glucosinolate and element concentrations. Environ. Exp. Bot. 2017, 133, 78–86. [Google Scholar] [CrossRef]
- Fischer, M.C.; Rellstab, C.; Tedder, A.; Zoller, S.; Gugerli, F.; Shimizu, K.K.; Holderegger, R.; Widmer, A. Population genomic footprints of selection and associations with climate in natural populations of Arabidopsis halleri from the Alps. Mol. Ecol. 2013, 22, 5594–5607. [Google Scholar] [CrossRef]
- Rellstab, C.; Fischer, M.C.; Zoller, S.; Graf, R.; Tedder, A.; Shimizu, K.K.; Widmer, A.; Holderegger, R.; Gugerli, F. Local adaptation (mostly) remains local: Reassessing environmental associations of climate-related candidate SNPs in Arabidopsis halleri. Heredity 2017, 118, 193–201. [Google Scholar] [CrossRef]
- Galmán, A.; Abdala-Roberts, L.; Zhang, S.; Berny-Mier y Teran, J.C.; Rasmann, S.; Moreira, X.; Randall Hughes, A. A global analysis of elevational gradients in leaf herbivory and its underlying drivers: Effects of plant growth form, leaf habit and climatic correlates. J. Ecol. 2018, 106, 413–421. [Google Scholar] [CrossRef]
- Smith, J.D.; Woldemariam, M.G.; Mescher, M.C.; Jander, G.; De Moraes, C.M. Glucosinolates from Host Plants Influence Growth of the Parasitic Plant Cuscuta gronovii and Its Susceptibility to Aphid Feeding. Plant Physiol. 2016, 172, 181–197. [Google Scholar] [CrossRef]
- Schlaeppi, K.; Abou-Mansour, E.; Buchala, A.; Mauch, F. Disease resistance of Arabidopsis to Phytophthora brassicae is established by the sequential action of indole glucosinolates and camalexin. Plant J. 2010, 62, 840–851. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.A.; van Dam, N.M.; Raaijmakers, C.E.; Bullock, J.M.; Gols, R. Tri-trophic effects of population variation in defence chemistry of wild cabbage (Brassica oleracea). Oecologia 2011, 166, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Abdalsamee, M.K.; Muller, C. Effects of indole glucosinolates on performance and sequestration by the sawfly Athalia rosae and consequences of feeding on the plant defense system. J. Chem. Ecol. 2012, 38, 1366–1375. [Google Scholar] [CrossRef] [PubMed]
- Agerbirk, N.; De Vos, M.; Kim, J.H.; Jander, G. Indole glucosinolate breakdown and its biological effects. Phytochem. Rev. 2008, 8, 101–120. [Google Scholar] [CrossRef]
- Metz, J.; Ribbers, K.; Tielbörger, K.; Müller, C. Long- and medium-term effects of aridity on the chemical defence of a widespread Brassicaceae in the Mediterranean. Environ. Exp. Bot. 2014, 105, 39–45. [Google Scholar] [CrossRef]
- Cacho, N.I.; Kliebenstein, D.J.; Strauss, S.Y. Macroevolutionary patterns of glucosinolate defense and tests of defense-escalation and resource availability hypotheses. New Phytol. 2015, 208, 915–927. [Google Scholar] [CrossRef]
- Körner, C. The use of ‘altitude’ in ecological research. Trends Ecol. Evol. 2007, 22, 569–574. [Google Scholar] [CrossRef]
- Anstett, D.N.; Ahern, J.R.; Glinos, J.; Nawar, N.; Salminen, J.P.; Johnson, M.T. Can genetically based clines in plant defence explain greater herbivory at higher latitudes? Ecol. Lett. 2015, 18, 1376–1386. [Google Scholar] [CrossRef]
- Løe, G.; Toräng, P.; Gaudeul, M.; Ågren, J. Trichome production and spatiotemporal variation in herbivory in the perennial herb Arabidopsis lyrata. Oikos 2007, 116, 134–142. [Google Scholar] [CrossRef]
- Schulz, E.; Tohge, T.; Zuther, E.; Fernie, A.R.; Hincha, D.K. Flavonoids are determinants of freezing tolerance and cold acclimation in Arabidopsis thaliana. Sci. Rep. 2016, 6, 34027. [Google Scholar] [CrossRef] [PubMed]
- Beradi, A.; Taylor, D. The Role of the Plant Flavonoid Pathway in Adaptation to Elevation. Ph.D. Thesis, University of Virginia, Charlottesville, VA, USA, 2014. [Google Scholar]
- Simmonds, M.S.J. Importance of flavonoids in insect-plant interactions: Feeding and oviposition. Phytochemistry 2001, 56, 245–252. [Google Scholar] [CrossRef]
- Harvey, J.A.; Gols, R.; Wagenaar, R.; Bezemer, T.M. Development of an insect herbivore and its pupal parasitoid reflect differences in direct plant defense. J. Chem. Ecol. 2007, 33, 1556–1569. [Google Scholar] [CrossRef] [PubMed]
- Kazemi-Dinan, A.; Thomaschky, S.; Stein, R.J.; Kramer, U.; Muller, C. Zinc and cadmium hyperaccumulation act as deterrents towards specialist herbivores and impede the performance of a generalist herbivore. New Phytol. 2014, 202, 628–639. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Grosser, K.; van Dam, N.M. A Straightforward Method for Glucosinolate Extraction and Analysis with High-pressure Liquid Chromatography (HPLC). J. Vis. Exp. 2017, 121, e55425. [Google Scholar] [CrossRef] [PubMed]
- Jackman, S. pscl: Classes and Methods for R Developed in the Political Science Computational Laboratory; R Package Version 1.5.2; United States Studies Centre, University of Sydney: Sydney, New South Wales, Australia, 2017. [Google Scholar]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Lefcheck, J.S. piecewiseSEM: Piecewise structural equation modeling in R for ecology, evolution, and systematics. Methods Ecol. Evol. 2015, 7, 573–579. [Google Scholar] [CrossRef]
| % Leaves Damaged | Score |
|---|---|
| <1% | 0 |
| 1–5% | 1 |
| 5–10% | 2 |
| 10–25% | 3 |
| 25–50% | 4 |
| >50% | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buckley, J.; Pashalidou, F.G.; Fischer, M.C.; Widmer, A.; Mescher, M.C.; De Moraes, C.M. Divergence in Glucosinolate Profiles between High- and Low-Elevation Populations of Arabidopsis halleri Correspond to Variation in Field Herbivory and Herbivore Behavioral Preferences. Int. J. Mol. Sci. 2019, 20, 174. https://doi.org/10.3390/ijms20010174
Buckley J, Pashalidou FG, Fischer MC, Widmer A, Mescher MC, De Moraes CM. Divergence in Glucosinolate Profiles between High- and Low-Elevation Populations of Arabidopsis halleri Correspond to Variation in Field Herbivory and Herbivore Behavioral Preferences. International Journal of Molecular Sciences. 2019; 20(1):174. https://doi.org/10.3390/ijms20010174
Chicago/Turabian StyleBuckley, James, Foteini G. Pashalidou, Martin C. Fischer, Alex Widmer, Mark C. Mescher, and Consuelo M. De Moraes. 2019. "Divergence in Glucosinolate Profiles between High- and Low-Elevation Populations of Arabidopsis halleri Correspond to Variation in Field Herbivory and Herbivore Behavioral Preferences" International Journal of Molecular Sciences 20, no. 1: 174. https://doi.org/10.3390/ijms20010174
APA StyleBuckley, J., Pashalidou, F. G., Fischer, M. C., Widmer, A., Mescher, M. C., & De Moraes, C. M. (2019). Divergence in Glucosinolate Profiles between High- and Low-Elevation Populations of Arabidopsis halleri Correspond to Variation in Field Herbivory and Herbivore Behavioral Preferences. International Journal of Molecular Sciences, 20(1), 174. https://doi.org/10.3390/ijms20010174