Diet-Independent Correlations between Bacteria and Dysfunction of Gut, Adipose Tissue, and Liver: A Comprehensive Microbiota Analysis in Feces and Mucosa of the Ileum and Colon in Obese Mice with NAFLD
Abstract
1. Introduction
2. Results
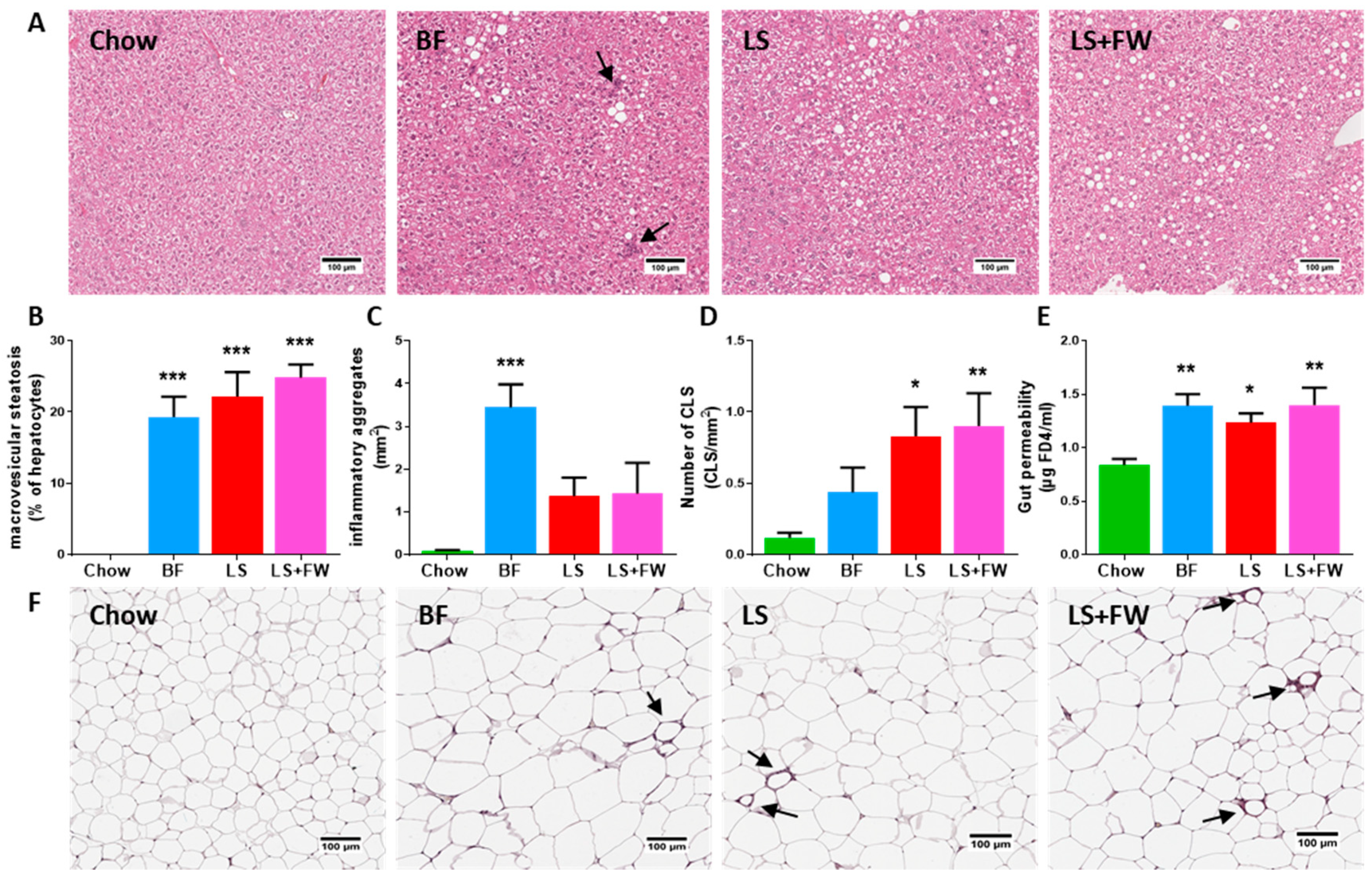
2.1. All ED Diets Resulted in NAFLD in the Context of Obesity, White Adipose Tissue Dysfunction, and Gut Dysfunction
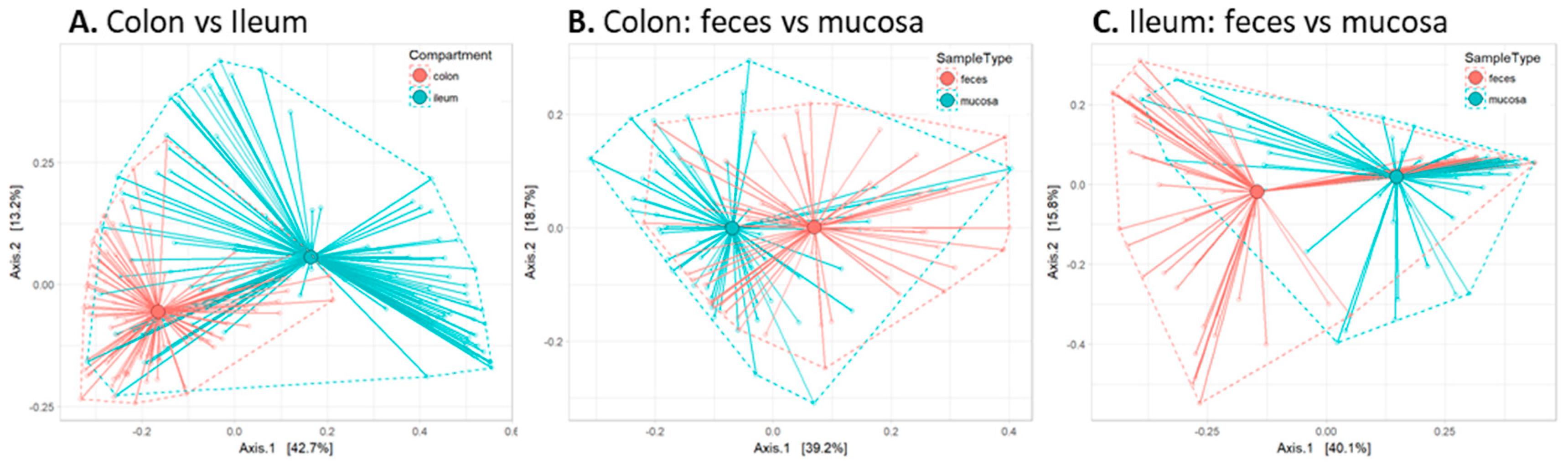
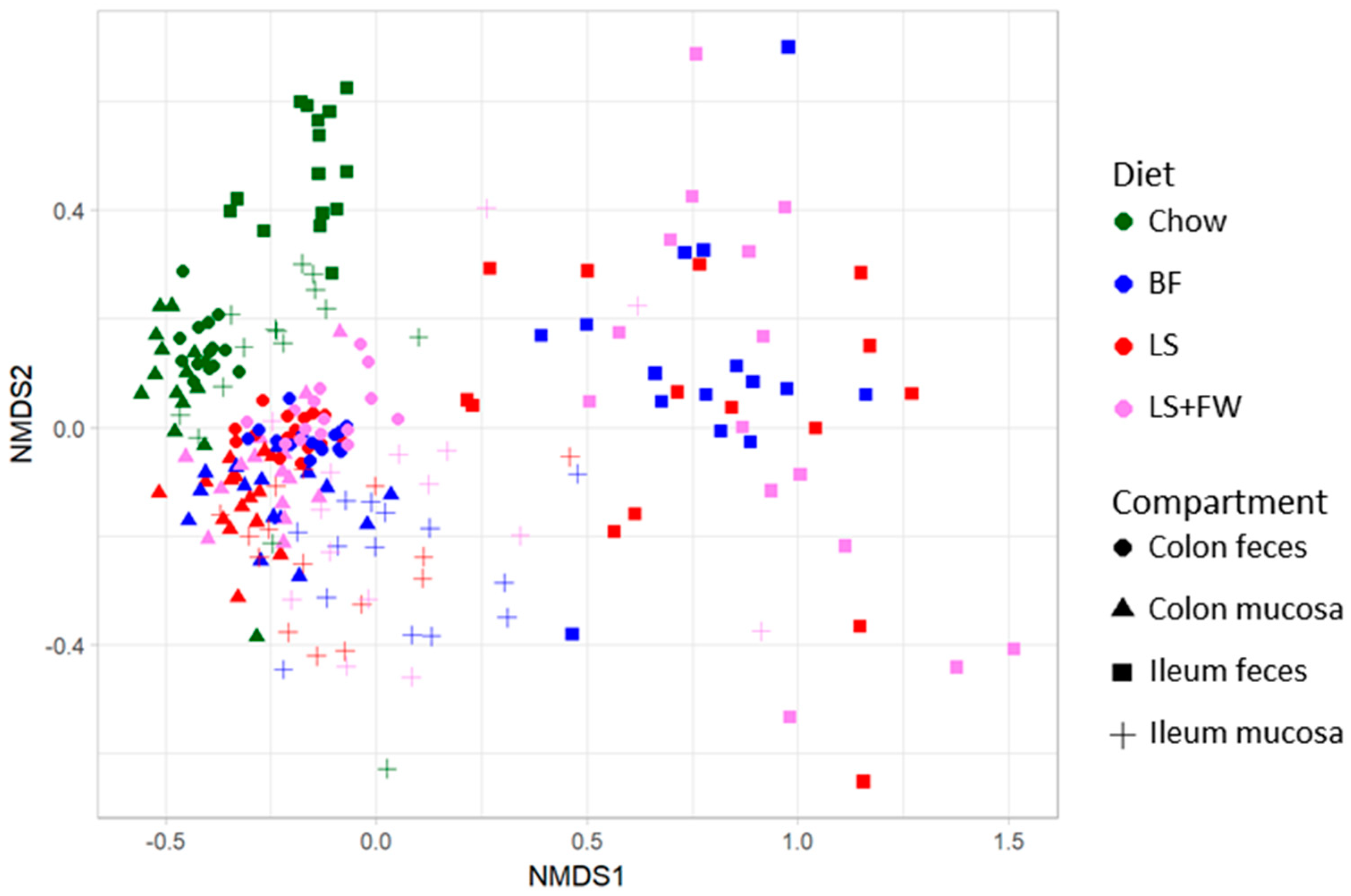
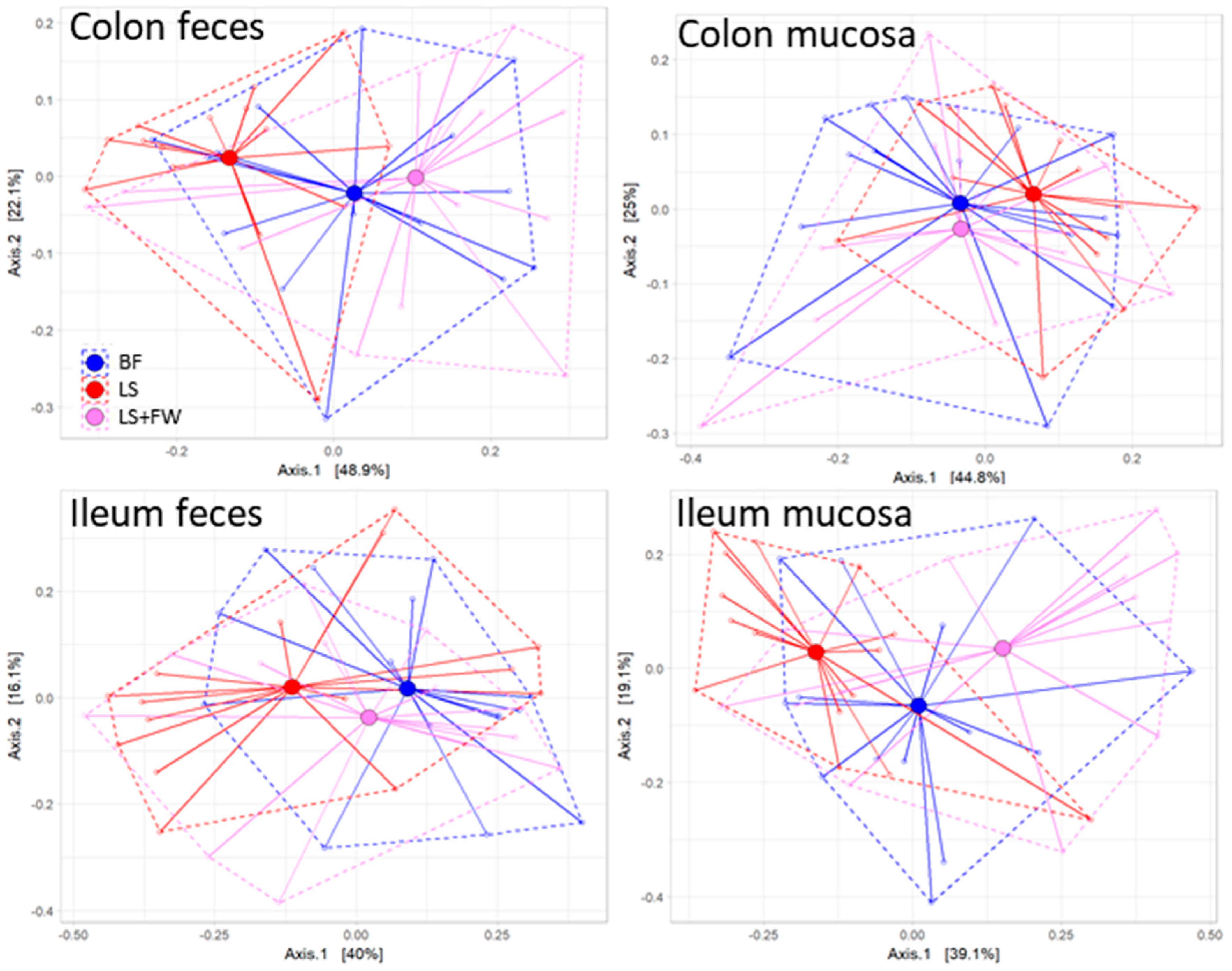
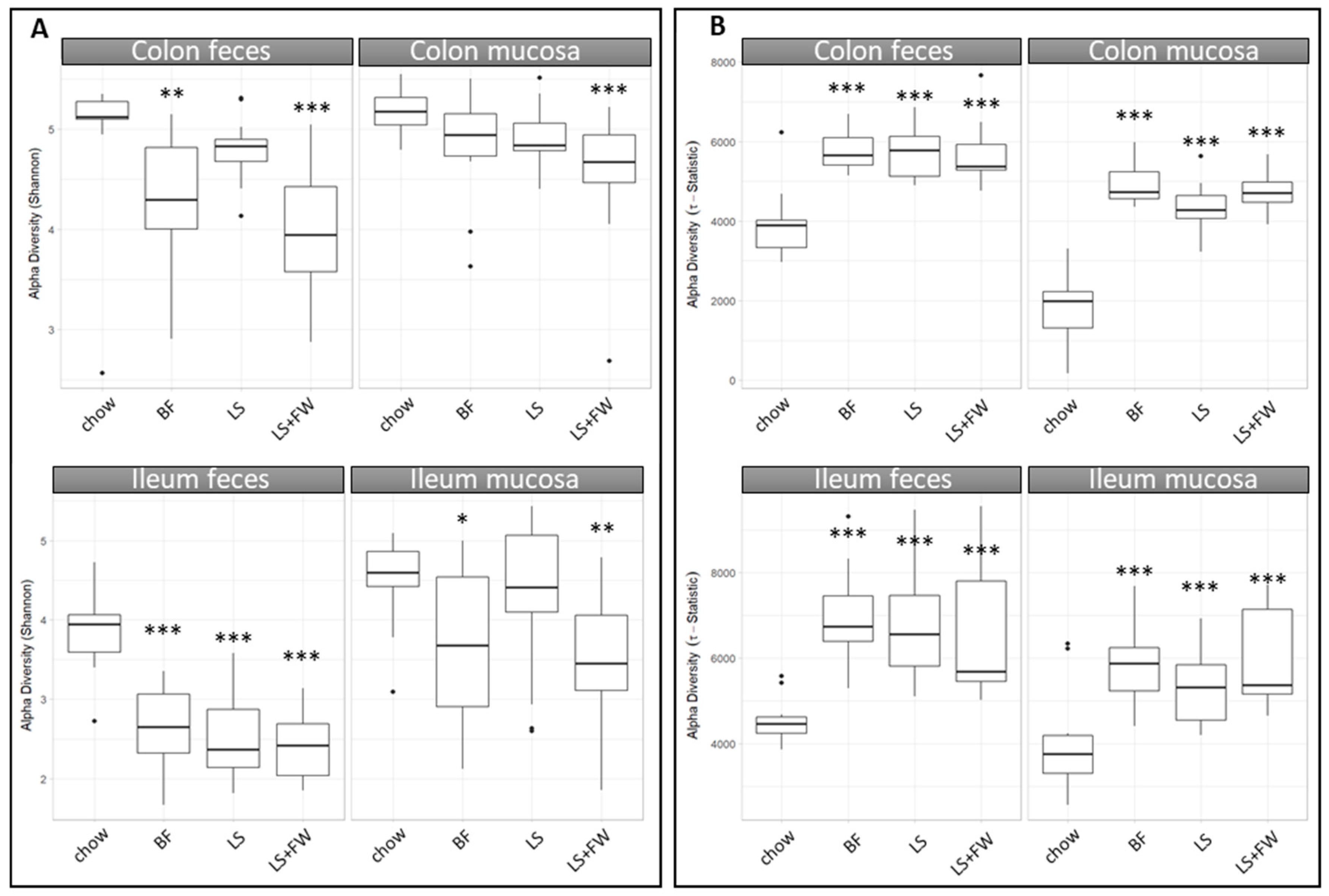
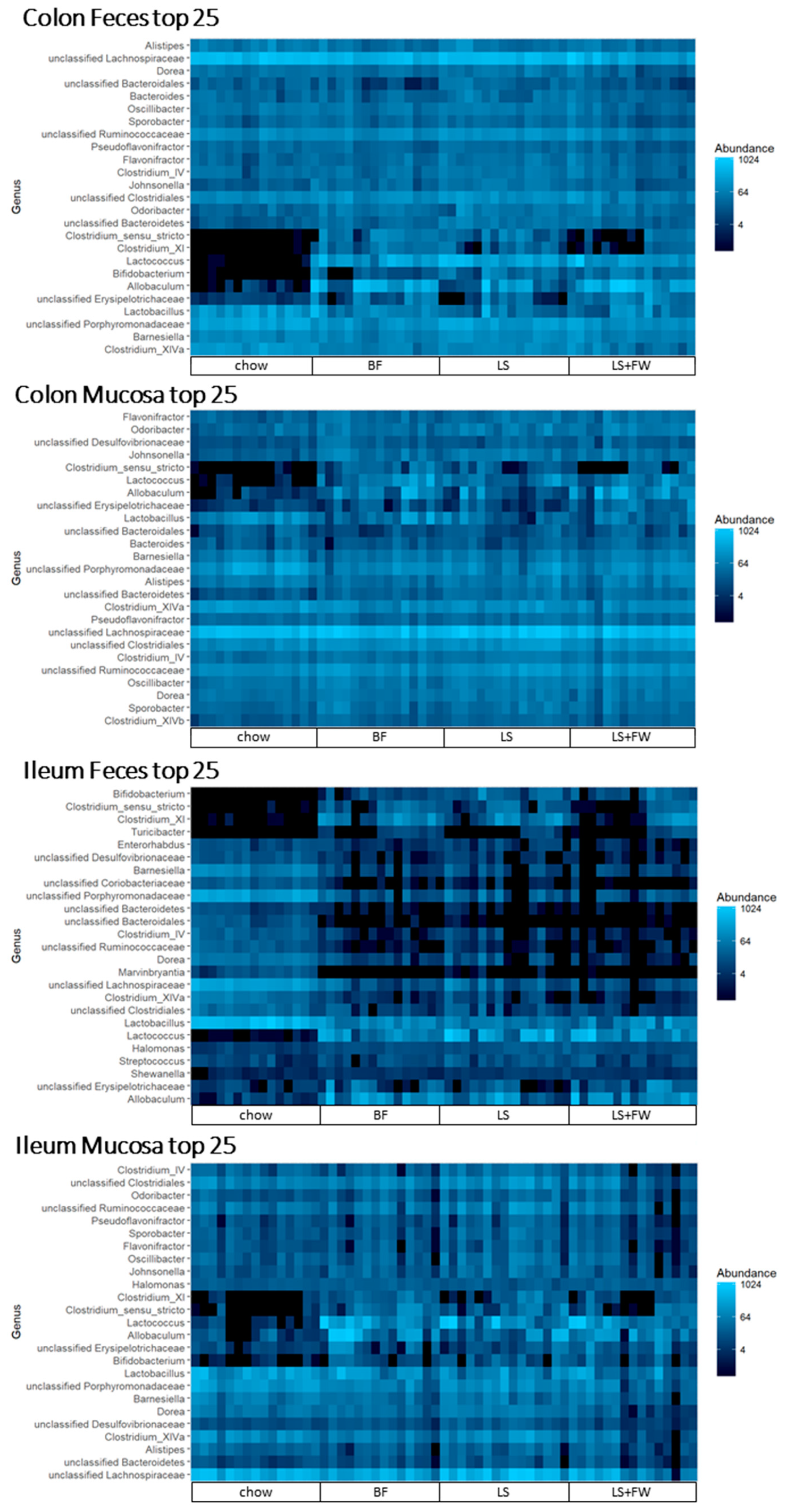
2.2. Global Analysis of Fecal and Mucosal Microbiota Composition in Colon and Ileum
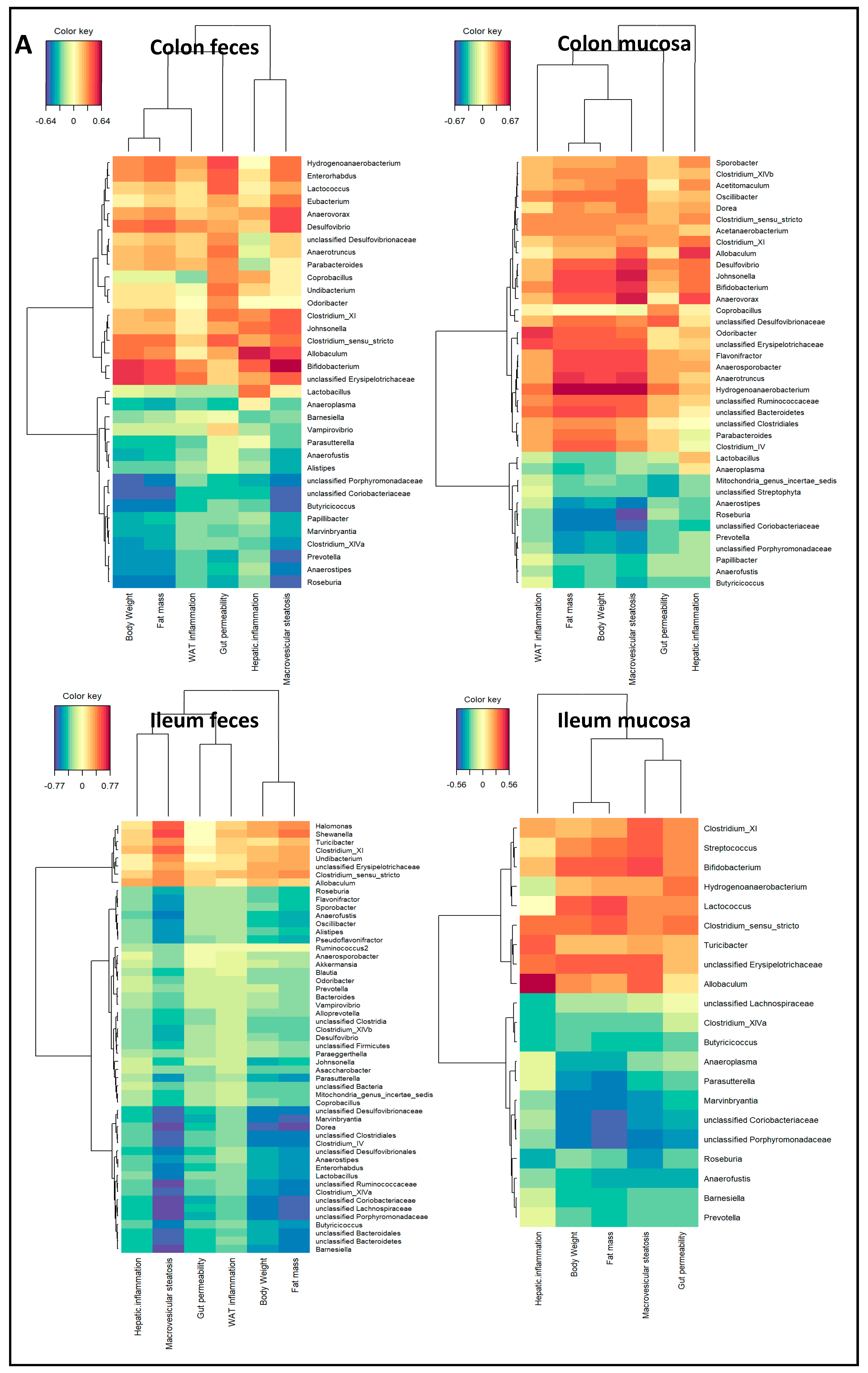
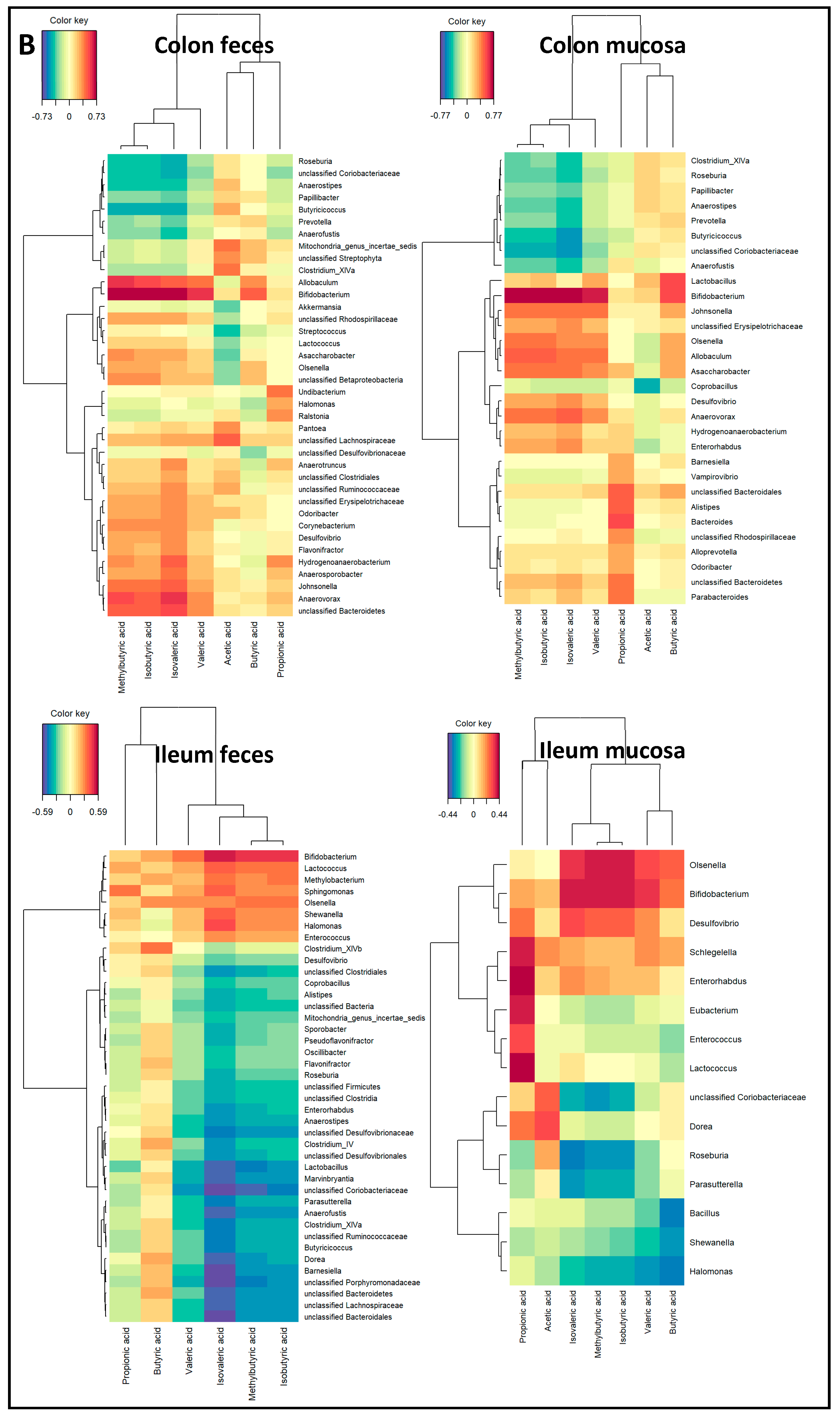
2.3. Microbiota Composition Associations with NAFLD Disease Endpoints
3. Discussion
4. Materials and Methods
4.1. Animals, Diets, and Study Design
4.2. Blood Chemistry
4.3. Histological Analysis of Adipose Tissue and Liver
4.4. FD4 Gut Permeability Assay
4.5. Statistical Analysis
4.6. Gut Microbiota Analysis
4.6.1. Collection and DNA Isolation
4.6.2. Metagenomic Sequencing and Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NAFLD | Non-alcoholic fatty liver disease |
| NASH | Non-alcoholic steatohepatitis |
| Ldlr−/− | Low-density lipoprotein receptor knockout |
| ED | Energy-dense |
| BF | Butter fat fructose |
| LS | Lard fat sucrose |
| LS+FW | Lard fat sucrose and fructose water |
| HFD | High-fat diet |
| SCFA | Short-chain fatty acid |
| ALT | Alanine-aminotransferase |
| TG | Triglycerides |
| WAT | White adipose tissue |
| CLS | Crown-like structures |
| FD4 | Fluorescein isothiocyanate-labeled dextran 4 |
| LC-MS | Liquid chromatography–mass spectrometry |
| NMDS | Non-metric multidimensional scaling |
| CV-score | Cross-validation score |
| rCCA | Regularized canonical correlation analysis |
| IR | Insulin resistance |
| UPLC | Ultraperformance liquid chromatography |
| HR-MS | High-resolution mass spectrometry |
References
- Estes, C.; Anstee, Q.M.; Arias-Loste, M.T.; Bantel, H.; Bellentani, S.; Caballeria, J.; Colombo, M.; Craxi, A.; Crespo, J.; Day, C.P.; et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J. Hepatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.; Suzuki, A.; Guy, C.; Unalp-Arida, A.; Colvin, R.; Johnson, R.D.A. Increased fructose consumption is associated with fibrosis severity in patients with nonalcoholic fatty liver disease. Hepatology 2011, 51, 1961–1971. [Google Scholar] [CrossRef] [PubMed]
- Jang, C.; Hui, S.; Lu, W.; Cowan, A.J.; Morscher, R.J.; Lee, G.; Liu, W.; Tesz, G.J.; Birnbaum, M.J.; Rabinowitz, J.D. The Small Intestine Converts Dietary Fructose into Glucose and Organic Acids. Cell Metab. 2018, 27, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Mulder, P.; Morrison, M.C.; Wielinga, P.Y.; Van Duyvenvoorde, W.; Kooistra, T.; Kleemann, R. Surgical removal of inflamed epididymal white adipose tissue attenuates the development of non-alcoholic steatohepatitis in obesity. Int. J. Obes. 2016, 40, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Boursier, J.; Mueller, O.; Barret, M.; Machado, M.; Fizanne, L.; Araujo-Perez, F.; Guy, C.D.; Seed, P.C.; Rawls, J.F.; David, L.A.; et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology 2016, 63, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Altamirano-Barrera, A.; Uribe, M.; Chávez-Tapia, N.C.; Nuño-Lámbarri, N. The role of the gut microbiota in the pathology and prevention of liver disease. J. Nutr. Biochem. 2018, 60, 1–8. [Google Scholar] [CrossRef]
- Boursier, J.; Diehl, A.M. Nonalcoholic Fatty Liver Disease and the Gut Microbiome. Clin. Liver Dis. 2016, 20, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Nascimbeni, F.; Targher, G.; Bernardi, M.; Bonino, F.; Bugianesi, E.; Casini, A.; Gastaldelli, A.; Marchesini, G.; Marra, F.; et al. AISF position paper on nonalcoholic fatty liver disease (NAFLD): Updates and future directions. Dig. Liver Dis. 2017, 49, 471–483. [Google Scholar] [CrossRef]
- Korpela, K. Diet, Microbiota, and Metabolic Health: Trade-Off Between Saccharolytic and Proteolytic Fermentation. Annu. Rev. Food Sci. Technol. 2018, 9. [Google Scholar] [CrossRef]
- Aron-Wisnewsky, J.; Doré, J.; Clement, K. The importance of the gut microbiota after bariatric surgery. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 590–598. [Google Scholar] [CrossRef]
- Rangel, I.; Sundin, J.; Fuentes, S.; Repsilber, D.; de Vos, W.M.; Brummer, R.J. The relationship between faecal-associated and mucosal-associated microbiota in irritable bowel syndrome patients and healthy subjects. Aliment. Pharmacol. Ther. 2015, 42, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Menke, A.L.; Driessen, A.; Koek, G.H.; Lindeman, J.H.; Stoop, R.; Havekes, L.M.; Kleemann, R.; van den Hoek, A.M. Establishment of a general NAFLD scoring system for rodent models and comparison to human liver pathology. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.C.; Kleemann, R.; van Koppen, A.; Hanemaaijer, R.; Verschuren, L. Key inflammatory processes in human NASH are reflected in Ldlr−/−.Leiden mice: A translational gene profiling study. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- van Koppen, A.; Verschuren, L.; van den Hoek, A.M.; Verheij, J.; Morrison, M.C.; Li, K.; Nagabukuro, H.; Costessi, A.; Caspers, M.P.; van den Broek, T.J.; et al. Uncovering a Predictive Molecular Signature for the Onset of NASH-Related Fibrosis in a Translational NASH Mouse Model. CMGH 2018, 5, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Graf, D.; Di Cagno, R.; Fåk, F.; Flint, H.J.; Nyman, M.; Saarela, M.; Watzl, B. Contribution of diet to the composition of the human gut microbiota. Microb. Ecol. Health Dis. 2015, 26, 26164. [Google Scholar] [CrossRef] [PubMed]
- den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed]
- Pickens, M.K.; Ogata, H.; Soon, R.K.; Grenert, J.P.; Maher, J.J. Dietary fructose exacerbates hepatocellular injury when incorporated into a methionine-choline-deficient diet. Liver Int. 2010, 30, 1229–1239. [Google Scholar] [CrossRef]
- Ishimoto, T.; Lanaspa, M.A.; Rivard, C.J.; Roncal-Jimenez, C.A.; Orlicky, D.J.; Cicerchi, C.; McMahan, R.H.; Abdelmalek, M.F.; Rosen, H.R.; Jackman, M.R.; et al. High-fat and high-sucrose (western) diet induces steatohepatitis that is dependent on fructokinase. Hepatology 2013, 58, 1632–1643. [Google Scholar] [CrossRef]
- Morrison, M.C.; Mulder, P.; Stavro, P.M.; Suárez, M.; Arola-Arnal, A.; Van Duyvenvoorde, W.; Kooistra, T.; Wielinga, P.Y.; Kleemann, R. Replacement of dietary saturated fat by PUFA-rich pumpkin seed oil attenuates non-alcoholic fatty liver disease and atherosclerosis development, with additional health effects of virgin over refined oil. PLoS ONE 2015. [Google Scholar] [CrossRef]
- Kiziltas, S. Toll-like receptors in pathophysiology of liver diseases. World J. Hepatol. 2016, 8, 1354–1369. [Google Scholar] [CrossRef]
- Liang, W.; Lindeman, J.H.; Menke, A.L.; Koonen, D.P.; Morrison, M.; Havekes, L.M.; Van Den Hoek, A.M.; Kleemann, R. Metabolically induced liver inflammation leads to NASH and differs from LPS-or IL-1β-induced chronic inflammation. Lab. Investig. 2014, 94, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.C.; Verschuren, L.; Salic, K.; Verheij, J.; Menke, A.; Wielinga, P.Y.; Iruarrizaga-Lejarreta, M.; Gole, L.; Yu, W.M.; Turner, S.; et al. Obeticholic Acid Modulates Serum Metabolites and Gene Signatures Characteristic of Human NASH and Attenuates Inflammation and Fibrosis Progression in Ldlr−/−.Leiden Mice. Hepatol. Commun. 2018. [Google Scholar] [CrossRef] [PubMed]
- Teufel, A.; Itzel, T.; Erhart, W.; Brosch, M.; Wang, X.Y.; Kim, Y.O.; von Schönfels, W.; Herrmann, A.; Brückner, S.; Stickel, F.; Dufour, J.F. Comparison of Gene Expression Patterns Between Mouse Models of Nonalcoholic Fatty Liver Disease and Liver Tissues From Patients. Gastroenterology 2016. [Google Scholar] [CrossRef] [PubMed]
- Tidjani Alou, M.; Lagier, J.C.; Raoult, D. Diet influence on the gut microbiota and dysbiosis related to nutritional disorders. Hum. Microbiome J. 2016, 1, 3–11. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Possemiers, S.; Druart, C.; Van de Wiele, T.; De Backer, F.; Cani, P.D.; Larondelle, Y.; Delzenne, N.M. Prebiotic effects of wheat Arabinoxylan related to the increase in bifidobacteria, roseburia and bacteroides/prevotella in diet-induced obese mice. PLoS ONE 2011. [Google Scholar] [CrossRef] [PubMed]
- Brinkworth, G.D.; Noakes, M.; Clifton, P.M.; Bird, A.R. Comparative effects of very low-carbohydrate, high-fat and high-carbohydrate, low-fat weight-loss diets on bowel habit and faecal short-chain fatty acids and bacterial populations. Br. J. Nutr. 2009. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.R.; Gratz, S.W.; Duncan, S.H.; Holtrop, G.; Ince, J.; Scobbie, L.; Duncan, G.; Johnstone, A.M.; Lobley, G.E.; Wallace, R.J.; et al. High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am. J. Clin. Nutr. 2011. [Google Scholar] [CrossRef]
- Aguirre, M.; Eck, A.; Koenen, M.E.; Savelkoul, P.H.M.; Budding, A.E.; Venema, K. Diet drives quick changes in the metabolic activity and composition of human gut microbiota in a validated in vitro gut model. Res. Microbiol. 2016. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Mulders, R.J.; de Git, K.C.G.; Schéle, E.; Dickson, S.L.; Sanz, Y.; Adan, R.A.H. Microbiota in obesity: Interactions with enteroendocrine, immune and central nervous systems. Obes. Rev. 2018. [Google Scholar] [CrossRef]
- De Wit, N.; Derrien, M.; Bosch-Vermeulen, H.; Oosterink, E.; Keshtkar, S.; Duval, C.; de Vogel-van den Bosch, J.; Kleerebezem, M.; Müller, M.; van der Meer, R. Saturated fat stimulates obesity and hepatic steatosis and affects gut microbiota composition by an enhanced overflow of dietary fat to the distal intestine. AJP Gastrointest. Liver Physiol. 2012, 303, G589–G599. [Google Scholar] [CrossRef]
- Vaiserman, A.M.; Koliada, A.K.; Marotta, F. Gut microbiota: A player in aging and a target for anti-aging intervention. Ageing Res. Rev. 2017, 35, 36–45. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, P.W.; Jeffery, I.B. Gut microbiota and aging. Science 2015, 350, 1214–1215. [Google Scholar] [CrossRef] [PubMed]
- Cornick, S.; Tawiah, A.; Chadee, K. Roles and regulation of the mucus barrier in the gut. Tissue Barriers 2015. [Google Scholar] [CrossRef]
- Li, H.; Limenitakis, J.P.; Fuhrer, T.; Geuking, M.B.; Lawson, M.A.; Wyss, M.; Brugiroux, S.; Keller, I.; Macpherson, J.A.; Rupp, S.; et al. The outer mucus layer hosts a distinct intestinal microbial niche. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.S.; Poles, J.; Leung, J.M.; Wolff, M.J.; Davenport, M.; Lee, S.C.; Lim, Y.A.; Chua, K.H.; Loke, P.N.; Cho, I. Inferred metagenomic comparison of mucosal and fecal microbiota from individuals undergoing routine screening colonoscopy reveals similar differences observed during active inflammation. Gut Microbes 2015. [Google Scholar] [CrossRef]
- Yin, Y.N.; Yu, Q.F.; Fu, N.; Liu, X.W.; Lu, F.G. Effects of four Bifidobacteria on obesity in high-fat diet induced rats. World J. Gastroenterol. 2010. [Google Scholar] [CrossRef]
- Solga, S.F.; Buckley, G.; Clark, J.M.; Horska, A.; Diehl, A.M. The effect of a probiotic on hepatic steatosis. J. Clin. Gastroenterol. 2008, 42, 1117–1119. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gibson, G.R.; Walton, G.E. Impact of high fat diets, prebiotics and probiotics on gut microbiota and immune function, with relevance to elderly populations. Nutr. Aging 2016, 3, 171–192. [Google Scholar] [CrossRef]
- Jiang, W.; Wu, N.; Wang, X.; Chi, Y.; Zhang, Y.; Qiu, X.; Hu, Y.; Li, J.; Liu, Y. Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Sci. Rep. 2015. [Google Scholar] [CrossRef]
- Woting, A.; Blaut, M. Small Intestinal Permeability and Gut-Transit Time. Nutrients 2018, 10, 685. [Google Scholar] [CrossRef]
- Jung, M.J.; Lee, J.; Shin, N.R.; Kim, M.S.; Hyun, D.W.; Yun, J.H.; Kim, P.S.; Whon, T.W.; Bae, J.W. Chronic Repression of mTOR Complex 2 Induces Changes in the Gut Microbiota of Diet-induced Obese Mice. Sci. Rep. 2016. [Google Scholar] [CrossRef]
- Mathur, R.; Amichai, M.; Chua, K.S.; Mirocha, J.; Barlow, G.M.; Pimentel, M. Methane and hydrogen positivity on breath test is associated with greater body mass index and body fat. J. Clin. Endocrinol. Metab. 2013. [Google Scholar] [CrossRef] [PubMed]
- Boursier, J.; Diehl, A.M. Implication of gut microbiota in nonalcoholic fatty liver disease. PLoS Pathog. 2015, 11, e1004559. [Google Scholar] [CrossRef]
- Newgard, C.B. Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab. 2012, 15, 606–614. [Google Scholar] [CrossRef]
- Beck, H.C. Branched-chain fatty acid biosynthesis in a branched-chain amino acid aminotransferase mutant of Staphylococcus carnosus. FEMS Microbiol. Lett. 2005. [Google Scholar] [CrossRef] [PubMed]
- Windey, K.; de Preter, V.; Verbeke, K. Relevance of protein fermentation to gut health. Mol. Nutr. Food Res. 2012, 56, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Jakobsdottir, G.; Bjerregaard, J.H.; Skovbjerg, H.; Nyman, M. Fasting serum concentration of short-chain fatty acids in subjects with microscopic colitis and celiac disease: No difference compared with controls, but between genders. Scand. J. Gastroenterol. 2013. [Google Scholar] [CrossRef]
- Kleiner, D.E.; Brunt, E.M. Nonalcoholic fatty liver disease: Pathologic patterns and biopsy evaluation in clinical research. Semin. Liver Dis. 2012, 32, 3–13. [Google Scholar] [CrossRef]
- Schoemaker, M.H.; Kleemann, R.; Morrison, M.C.; Verheij, J.; Salic, K.; van Tol, E.A.; Kooistra, T.; Wielinga, P.Y. A casein hydrolysate based formulation attenuates obesity and associated nonalcoholic fatty liver disease and atherosclerosis in Ldlr−/−.Leiden mice. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Nejdfors, P.; Ekelund, M.; Weström, B.R.; Willén, R.; Jeppsson, B. Intestinal permeability in humans is increased after radiation therapy. Dis. Colon Rectum 2000, 43, 1582–1587. [Google Scholar] [CrossRef]
- Patel, R.M.; Myers, L.S.; Kurundkar, A.R.; Maheshwari, A.; Nusrat, A.; Lin, P.W. Probiotic bacteria induce maturation of intestinal claudin 3 expression and barrier function. Am. J. Pathol. 2012, 180, 626–635. [Google Scholar] [CrossRef]
- Dong, C.X.; Zhao, W.; Solomon, C.; Rowland, K.J.; Ackerley, C.; Robine, S.; Holzenberger, M.; Gonska, T.; Brubaker, P.L. The intestinal epithelial insulin-like growth factor-1 receptor links glucagon-like peptide-2 action to gut barrier function. Endocrinology 2014, 155, 370–379. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15. [Google Scholar] [CrossRef]
- Oksanen, J. Vegan: Community Ecology Package, Version 1.17-2; R Package 2010. Available online: http://cran.r-project.org (accessed on 7 May 2018).
- Friedman, J.H.; Rafsky, L.C. Multivariate Generalizations of the Wald-Wolfowitz and Smirnov Two-Sample Tests. Ann. Stat. 1979, 7, 697–717. [Google Scholar] [CrossRef]
- Schilling, M.F. Multivariate two-sample tests based on nearest neighbors. J. Am. Stat. Assoc. 1986, 81, 799–806. [Google Scholar] [CrossRef]
- Methods, B.; Tests, P. Bootstrap Methods and Permutation Tests. Introd. Pract. Stat. 2005, 5, 1–70. [Google Scholar]
- Fukuyama, J.; Holmes, S. phyloseqGraphTest: Graph-Based Permutation Tests for Microbiome Data. R Package Version 0.0.2. 2018. Available online: https://cran.r-project.org/package=phyloseqGraphTest (accessed on 7 May 2018).
- Rencher, A.C. Canonical Correlation. Methods Multivar. Anal. 2003, 361–379. [Google Scholar] [CrossRef]
- González, I.; Lê Cao, K.A.; Déjean, S. MixOmics: Omics Data Integration Project. 2011. Available online: http://www.mixomics.org (accessed on 7 May 2018).
| Chow | BF | LS | LS+FW | |
|---|---|---|---|---|
| Body weight (g) | 29.1 ± 2.2 | 34.5 ± 3.8 ** | 37.8 ± 6.0 *** | 40.1 ± 5.1 *** |
| Total caloric intake (kcal/day) | 11.0 ± 0.7 | 11.7 ± 1.1 | 11.5 ± 0.5 | 12.6 ± 0.8 |
| Total fat mass (g) | 3.7 ± 1.0 | 9.1 ± 1.9 *** | 11.8 ± 4.0 *** | 13.4 ± 4.8 *** |
| Plasma cholesterol (mM) | 8.9 ± 1.8 | 50.9 ± 10.9 *** | 20.3 ± 6.2 *** | 24.3 ± 10.8 *** |
| Plasma triglycerides (mM) | 1.6 ± 0.4 | 10.3 ± 2.4 *** | 4.9 ± 1.9 *** | 4.5 ± 2.6 *** |
| Plasma insulin (ng/mL) | 1.3 ± 0.6 | 4.2 ± 1.2 | 4.6 ± 2.1 * | 8.0 ± 6.8 * |
| Blood glucose (mM) | 7.2 ± 1.0 | 6.3 ± 0.9 | 7.6 ± 2.1 | 7.7 ± 1.5 ** |
| ALT (U/L) | 32.6 ± 7.3 | 139.7 ± 76.4 *** | 78.9 ± 50.0 | 137.1 ± 99.3 *** |
| Chow | BF | LS | LS+FW | |
|---|---|---|---|---|
| Acetate (nM) | 5966.6 ± 874.1 | 4828.2 ± 891.8 ** | 6468.3 ± 1361.3 | 4977.9 ± 810.9 ** |
| Butyrate (nM) | 123.7 ± 67.1 | 55.6 ± 14.4 ** | 142.3 ± 90.7 | 45.8 ± 7.3 ** |
| Propionate (nM) | 257.8 ± 103.3 | 310.8 ± 127.1 | 464.6 ± 214.9 ** | 388.3 ± 171.6 |
| Isobutyrate (nM) | 33.5 ± 7.3 | 45.7 ± 11.6 | 88.9 ± 42.7 *** | 57.6 ± 12.2 * |
| Methylbutyrate (nM) | 36.6 ± 8.5 | 46.9 ± 8.7 | 81.9 ± 34.6 *** | 54.8 ± 8.9 * |
| Isovalerate (nM) | 26.9 ± 4.6 | 33.3 ± 7.7 | 66.7 ± 16.7 *** | 60.9 ± 19.8 *** |
| Valerate (nM) | 10.8 ± 5.7 | 19.9 ± 11.8 | 36.4 ± 14.4 *** | 23.7 ± 10.2 ** |
| Histological and Functional Readouts | Metabolites | ||
|---|---|---|---|
| Colon | Feces | 0.56 | 0.43 |
| Mucosa | 0.71 | 0.62 | |
| Ileum | Feces | 0.60 | 0.62 |
| Mucosa | 0.52 | 0.41 | |
| Chow | BF | LS | LS+FW | |
|---|---|---|---|---|
| Fat | 9% kcal (Cereal grains and soy) | 41% kcal (Butter fat) | 45% kcal (Lard) | Same as LS |
| Protein | 24% kcal (Soy and cereal grains) | 14% kcal (Casein) | 20% kcal (Casein) | Same as LS |
| Carbohydrate | 67% kcal (Starch) | 44% kcal (Fructose) | 35% kcal (Sucrose) | Same as LS |
| Cholesterol | - | - | - | - |
| kcal/gram | 3.2 kcal/g | 4.5 kcal/g | 4.7 kcal/g | Same as LS |
| Fiber | 4.9% w/w | 5.1% w/w | 5.8% w/w | Same as LS |
| Drink | Water | Water | Water | 10% w/v fructose in water |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gart, E.; Souto Lima, E.; Schuren, F.; De Ruiter, C.G.F.; Attema, J.; Verschuren, L.; Keijer, J.; Salic, K.; Morrison, M.C.; Kleemann, R. Diet-Independent Correlations between Bacteria and Dysfunction of Gut, Adipose Tissue, and Liver: A Comprehensive Microbiota Analysis in Feces and Mucosa of the Ileum and Colon in Obese Mice with NAFLD. Int. J. Mol. Sci. 2019, 20, 1. https://doi.org/10.3390/ijms20010001
Gart E, Souto Lima E, Schuren F, De Ruiter CGF, Attema J, Verschuren L, Keijer J, Salic K, Morrison MC, Kleemann R. Diet-Independent Correlations between Bacteria and Dysfunction of Gut, Adipose Tissue, and Liver: A Comprehensive Microbiota Analysis in Feces and Mucosa of the Ileum and Colon in Obese Mice with NAFLD. International Journal of Molecular Sciences. 2019; 20(1):1. https://doi.org/10.3390/ijms20010001
Chicago/Turabian StyleGart, Eveline, Everton Souto Lima, Frank Schuren, Christa G. F. De Ruiter, Joline Attema, Lars Verschuren, Jaap Keijer, Kanita Salic, Martine C. Morrison, and Robert Kleemann. 2019. "Diet-Independent Correlations between Bacteria and Dysfunction of Gut, Adipose Tissue, and Liver: A Comprehensive Microbiota Analysis in Feces and Mucosa of the Ileum and Colon in Obese Mice with NAFLD" International Journal of Molecular Sciences 20, no. 1: 1. https://doi.org/10.3390/ijms20010001
APA StyleGart, E., Souto Lima, E., Schuren, F., De Ruiter, C. G. F., Attema, J., Verschuren, L., Keijer, J., Salic, K., Morrison, M. C., & Kleemann, R. (2019). Diet-Independent Correlations between Bacteria and Dysfunction of Gut, Adipose Tissue, and Liver: A Comprehensive Microbiota Analysis in Feces and Mucosa of the Ileum and Colon in Obese Mice with NAFLD. International Journal of Molecular Sciences, 20(1), 1. https://doi.org/10.3390/ijms20010001





