RBM4a-SRSF3-MAP4K4 Splicing Cascade Constitutes a Molecular Mechanism for Regulating Brown Adipogenesis
Abstract
1. Introduction
2. Results
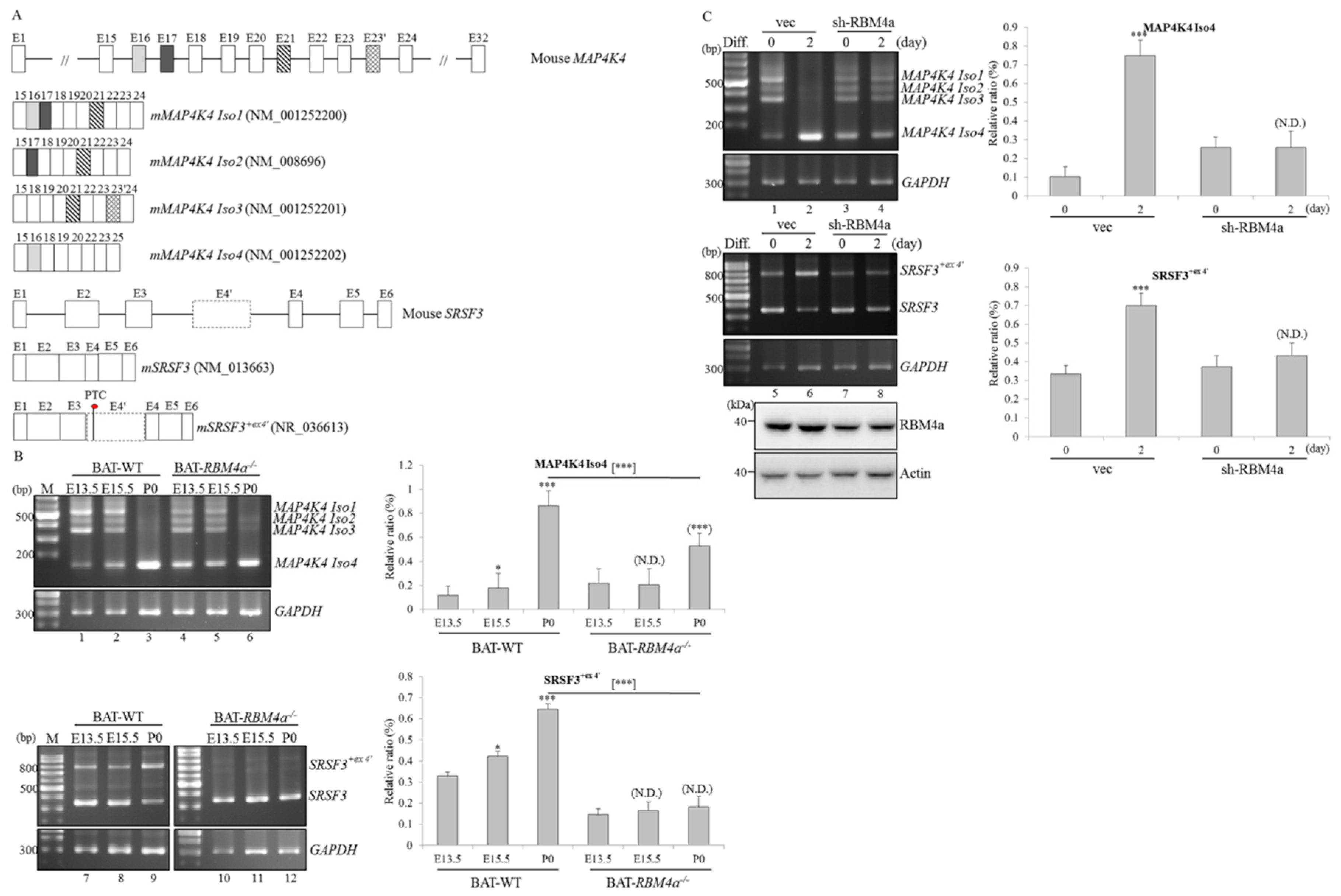
2.1. Transcriptome Analyses Reveal Differential Expressions and Splicing Profiles of MAP4K4 and SRSF3 in Differentiating and RBM4a−/− BAs
2.2. Splicing and Expression Profiles of MAP4K4 and SRSF3 Transcripts are Reprogrammed Throughout BAT Development
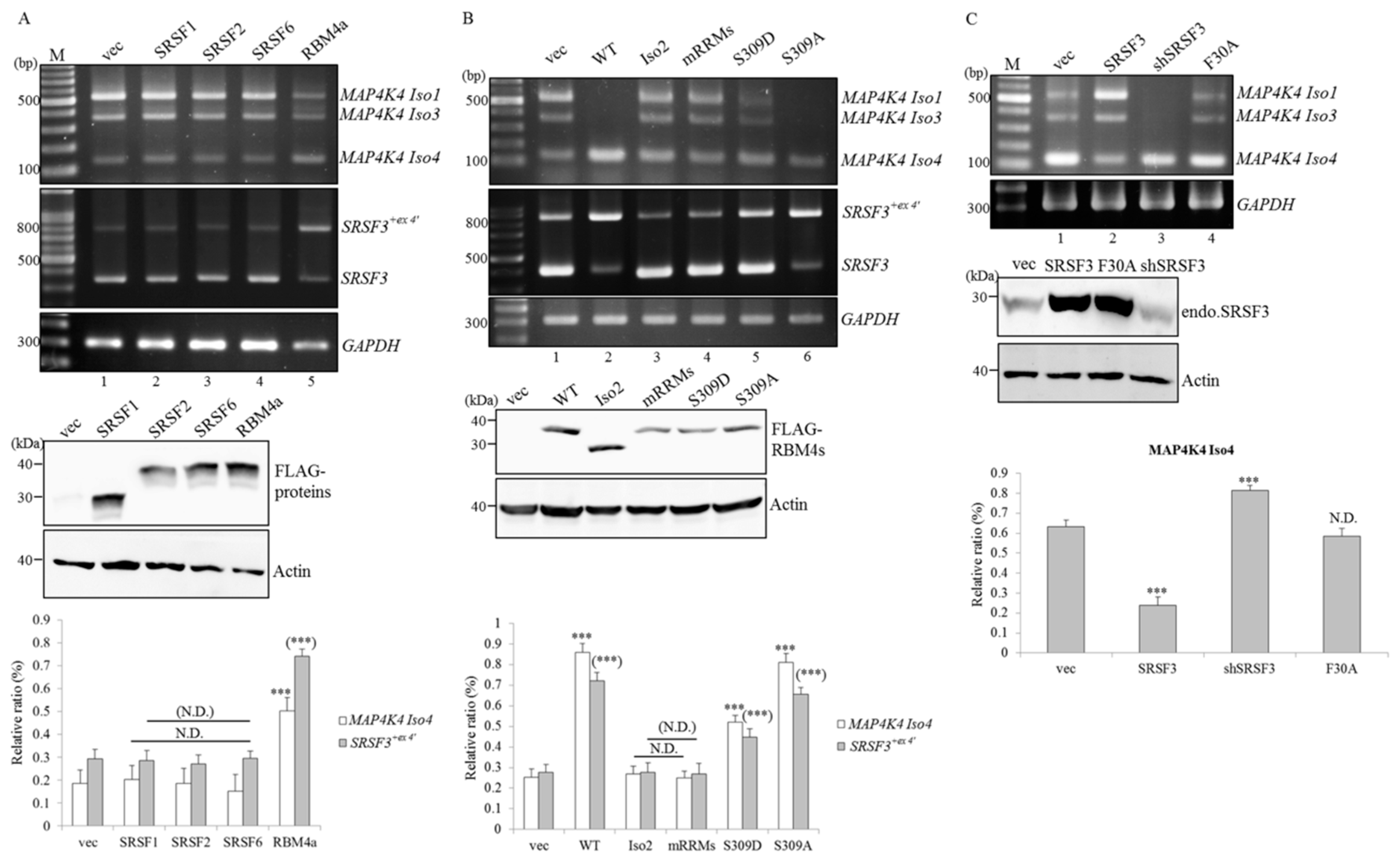
2.3. RBM4a Exerts a Specific Influence on Modulating Splicing Profiles of MAP4K4 and SRSF3 Transcripts
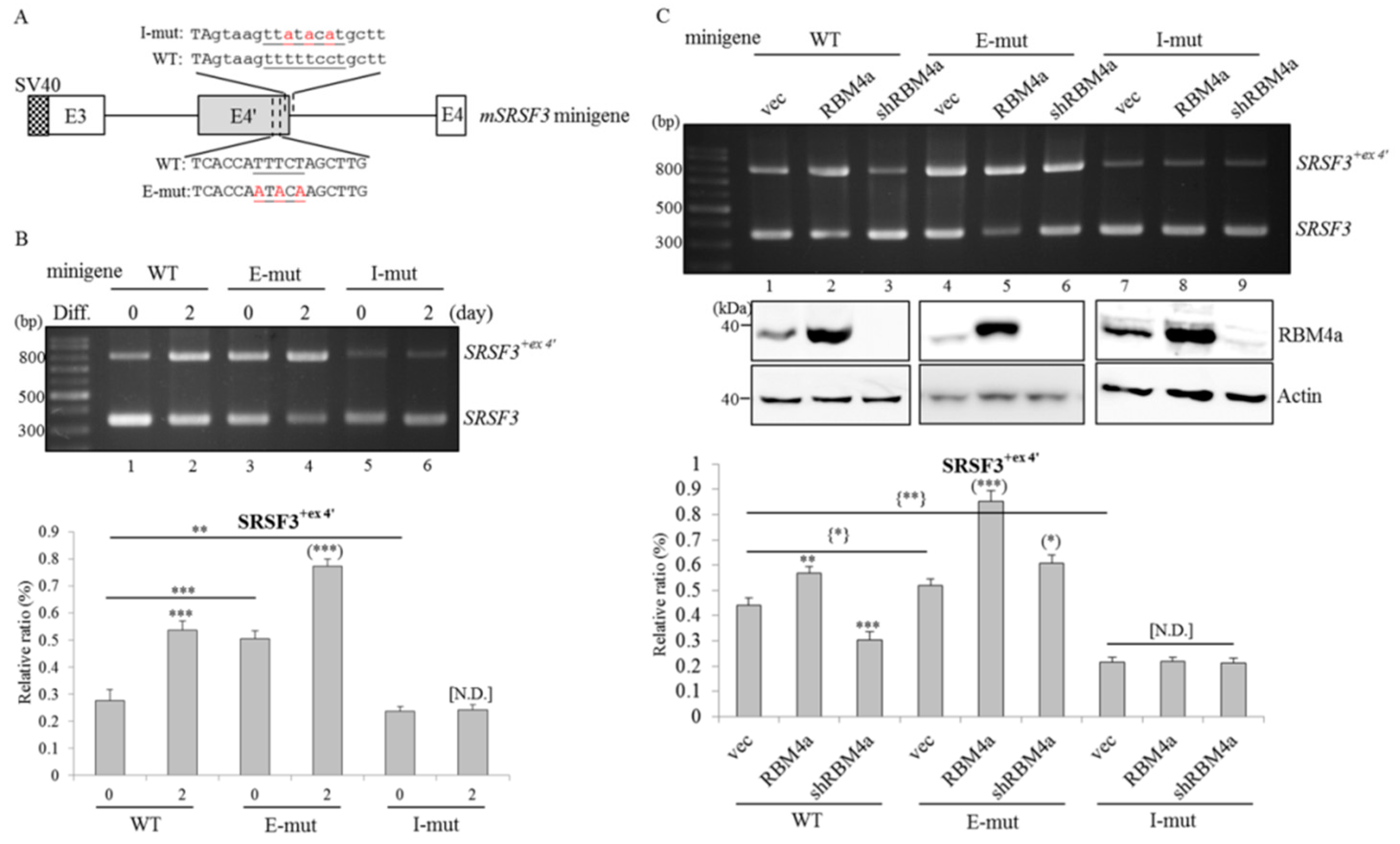
2.4. RBM4a Modulates the Alternative Splicing of SRSF3 Transcripts in a Sequence-Specific Manner
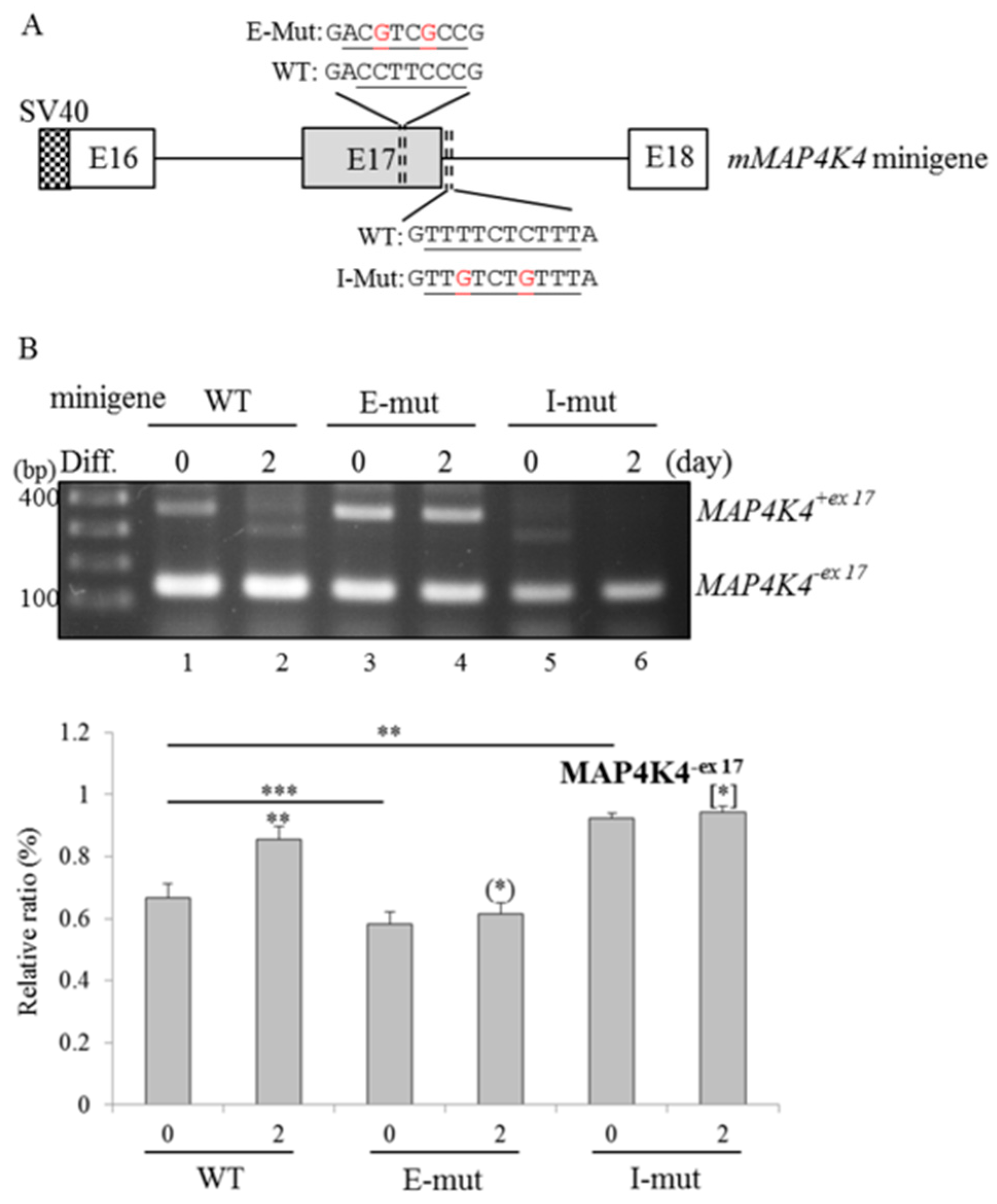
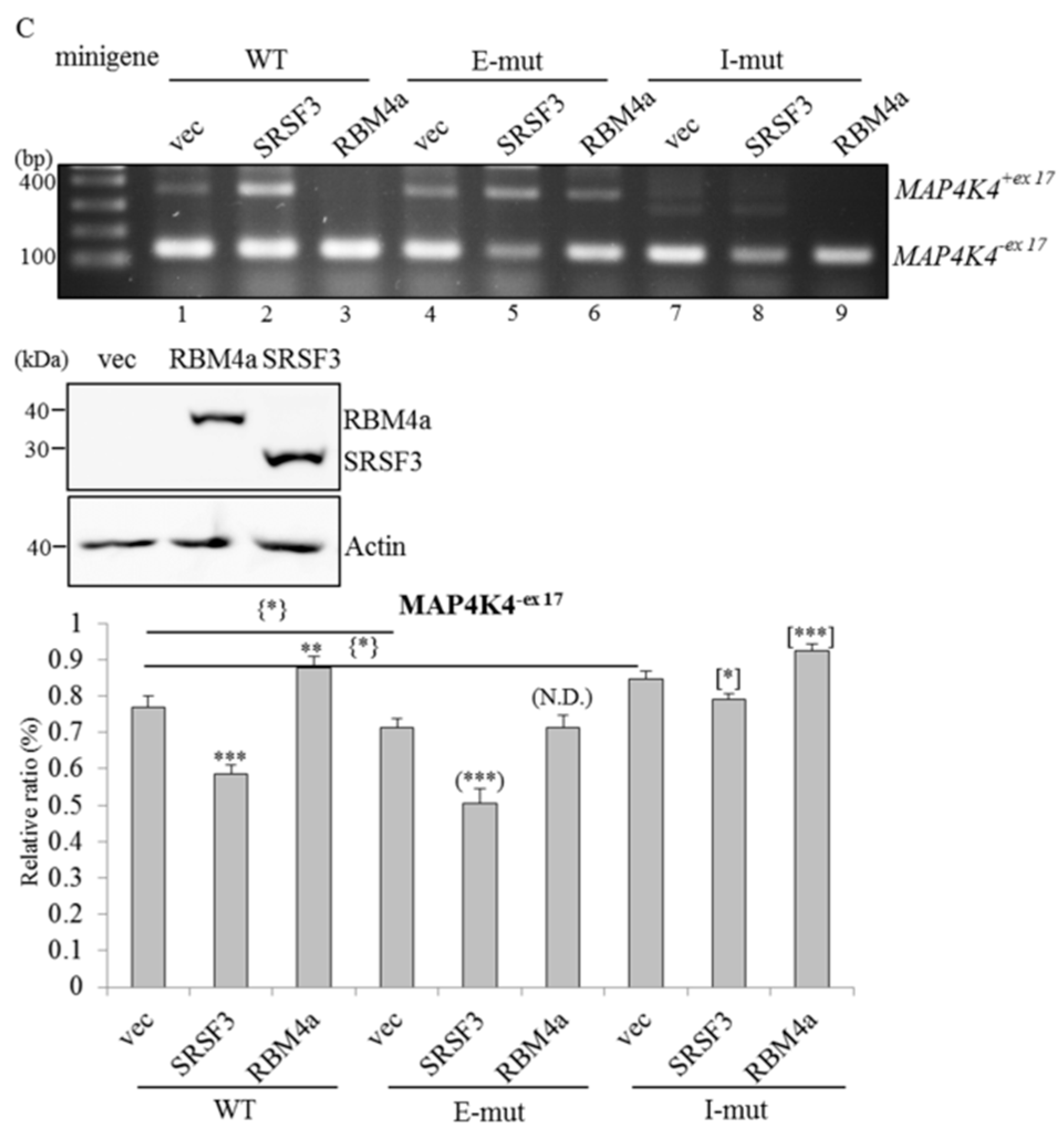
2.5. RBM4a and SRSF3 Exert Antagonistic Effects on Utilization of MAP4K4 Exon 17
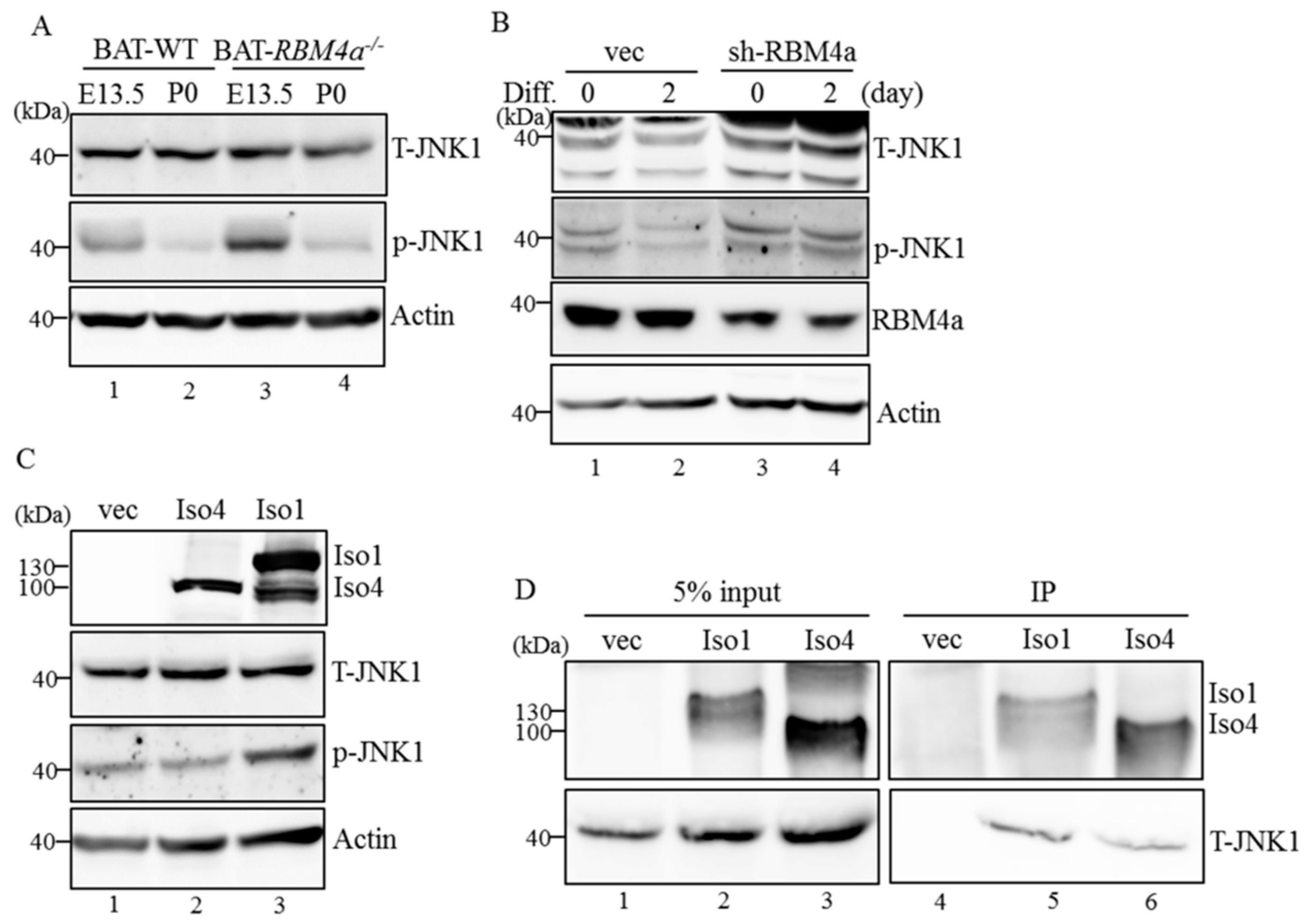
2.6. MAP4K4 Isoforms Differentially Modulate Activity of the Brown Adipogenesis-Related Signaling Pathway
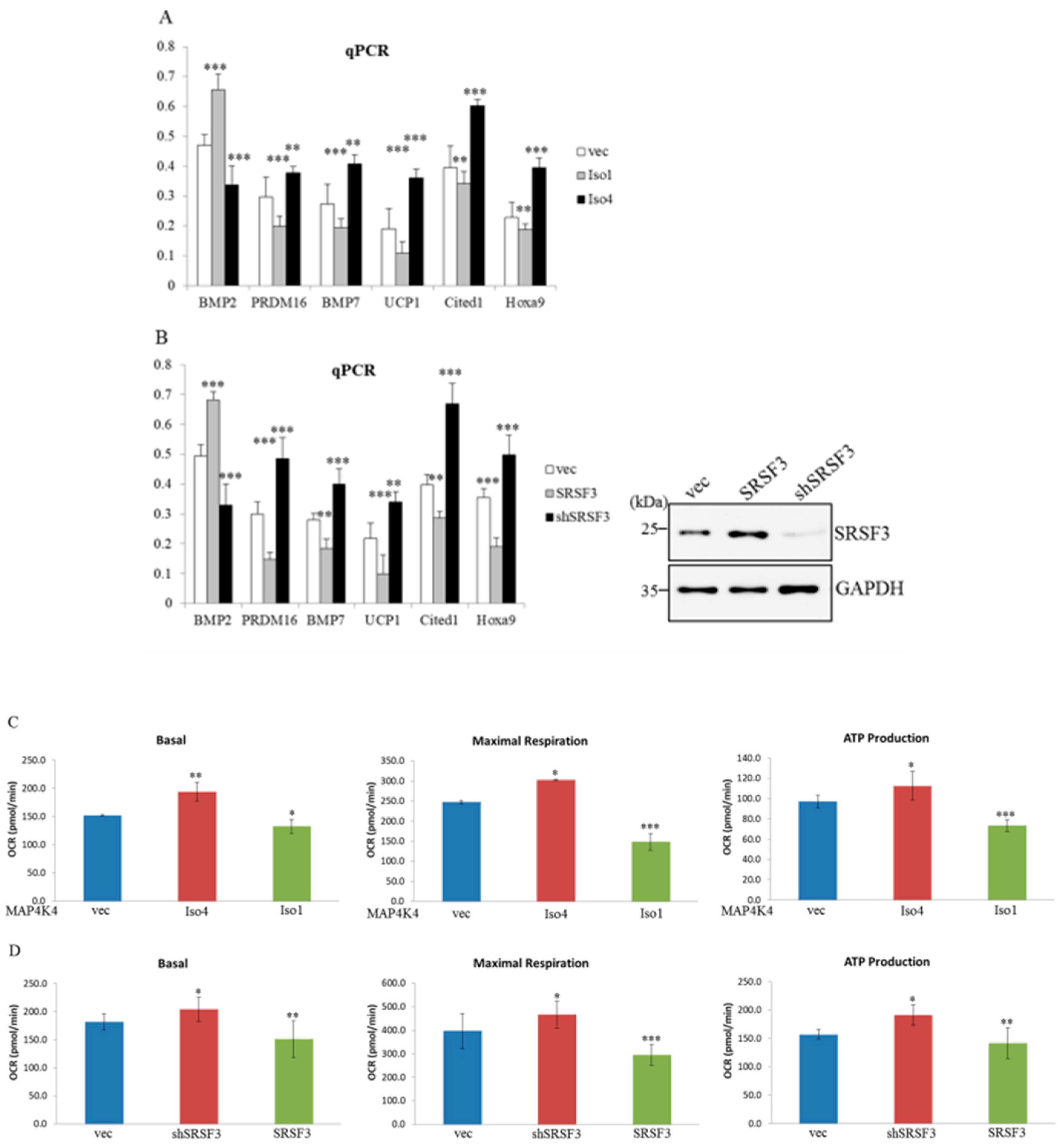
2.7. MAP4K4 Isoforms and SRSF3 Exert Discriminative Effects on Brown Adipogenesis
3. Discussion
4. Materials and Methods
4.1. Ethics Statement for Animal Research
4.2. Transcriptome Analyses
4.3. Cell Culture and In Vitro Differentiation
4.4. Plasmid Construction
4.5. Transient Transfection, Reverse-Transcription (RT)-PCR, and Quantitative (q)PCR Analyses
4.6. Co-Immunoprecipitation (Co-IP) and Immunoblot Assays
4.7. Mitochondrial Respiration Assay
4.8. Statistical Analyses
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kim, S.P.; Frey, J.L.; Li, Z.; Kushwaha, P.; Zoch, M.L.; Tomlinson, R.E.; Da, H.; Aja, S.; Noh, H.L.J.; Kim, K.; et al. Sclerostin influences body composition by regulating catabolic and anabolic metabolism in adipocytes. Proc. Natl. Acad. Sci. USA 2017, 114, E11238–E11247. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.; Bronec, C.; Napoli, E.; Graham, J.; Stanhope, K.L.; Marsilio, L.; Giron, M.C.; Havel, P.J.; Giulivi, C. Adipose depot-specific effects of ileal interposition surgery in UCD-T2D rats: Unexpected implications for obesity and diabetes. Biochem. J. 2018, 475, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Banfi, S.; Gusarova, V.; Gromada, J.; Cohen, J.C.; Hobbs, H.H. Increased thermogenesis by a noncanonical pathway in ANGPTL3/8-deficient mice. Proc. Natl. Acad. Sci. USA 2018, 115, E1249–E1258. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Yue, F.; Jia, Z.; Gao, Y.; Jin, W.; Hu, K.; Zhang, Y.; Zhu, D.; Yang, G.; Kuang, S. A novel brown adipocyte-enriched long non-coding RNA that is required for brown adipocyte differentiation and sufficient to drive thermogenic gene program in white adipocytes. Biochim. Biophys. Acta 2018, 1863, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Reza, M.M.; Subramaniyam, N.; Sim, C.M.; Ge, X.; Sathiakumar, D.; McFarlane, C.; Sharma, M.; Kambadur, R. Irisin is a pro-myogenic factor that induces skeletal muscle hypertrophy and rescues denervation-induced atrophy. Nat. Commun. 2017, 8, 1104. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Federspiel, C.K.; Borup, A.; Christensen, P.M.; Madsen, A.N.; Heine, M.; Nielsen, C.H.; Kjaer, A.; Holst, B.; Heeren, J.; et al. The apolipoprotein M/S1P axis controls triglyceride metabolism and brown fat activity. Cell Rep. 2018, 22, 175–188. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Wang, G.; Diao, Y.; Long, Y.; Fu, X.; Weng, M.; Zhou, L.; Sun, K.; Cheung, T.H.; Ip, N.Y.; et al. A molecular switch regulating cell fate choice between muscle progenitor cells and brown adipocytes. Dev. Cell 2017, 41, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, T.W.; Graveley, B.R. Expansion of the eukaryotic proteome by alternative splicing. Nature 2010, 463, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, Y.; Luo, J.; Zhao, W.; Zhou, X. Deep learning of the splicing (epi)genetic code reveals a novel candidate mechanism linking histone modifications to ESC fate decision. Nucleic Acids Res. 2017, 45, 12100–12112. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Luo, C.; Shen, L.; Liu, Y.; Wang, Q.; Zhang, C.; Guo, R.; Zhang, Y.; Xie, Z.; Wei, N. SRSF1 prevents DNA damage and promotes tumorigenesis through regulation of DBF4B pre-mrna splicing. Cell Rep. 2017, 21, 3406–3413. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Chen, M.; Dai, R.; Zhang, Y.; Zhao, H.; Song, Z.; Zhang, L.; Li, Z.; Feng, Y.; Gao, H.; et al. SRSF1 promotes vascular smooth muscle cell proliferation through a Δ133p53/EGR1/KLF5 pathway. Nat. Commun. 2017, 8, 16016. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Yuan, J.; Zhang, Z.; Chang, X. Cytoplasmic poly(A)-binding protein 1 (PABPC1) interacts with the RNA-binding protein hnRNPLL and thereby regulates immunoglobulin secretion in plasma cells. J. Biol. Chem. 2017, 292, 12285–12295. [Google Scholar] [CrossRef] [PubMed]
- Parikshak, N.N.; Swarup, V.; Belgard, T.G.; Irimia, M.; Ramaswami, G.; Gandal, M.J.; Hartl, C.; Leppa, V.; Ubieta, L.T.; Huang, J.; et al. Genome-wide changes in lncRNA, splicing, and regional gene expression patterns in autism. Nature 2016, 540, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Tarn, W.Y.; Hsieh, W.K. Emerging role for RNA binding motif protein 4 in the development of brown adipocytes. Biochim. Biophys. Acta 2014, 1843, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C. Multi-posttranscriptional regulations lessen the repressive effect of SRPK1 on brown adipogenesis. Biochim. Biophys. Acta 2018, 1863, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Yan, Y.T.; Hsieh, W.K.; Peng, P.J.; Su, C.H.; Tarn, W.Y. RBM4 promotes pancreas cell differentiation and insulin expression. Mol. Cell. Biol. 2013, 33, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Jia, J.; Jia, R. PTBP1 and PTBP2 impaired autoregulation of SRSF3 in cancer cells. Sci. Rep. 2015, 5, 14548. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Tarn, W.Y. RBM4 down-regulates PTB and antagonizes its activity in muscle cell-specific alternative splicing. J. Cell Biol. 2011, 193, 509–520. [Google Scholar] [CrossRef] [PubMed]
- García-Casarrubios, E.; de Moura, C.; Arroba, A.I.; Pescador, N.; Calderon-Dominguez, M.; Garcia, L.; Herrero, L.; Serra, D.; Cadenas, S.; Reis, F.; et al. Rapamycin negatively impacts insulin signaling, glucose uptake and uncoupling protein-1 in brown adipocytes. Biochim. Biophys. Acta 2016, 1861, 1929–1941. [Google Scholar] [CrossRef] [PubMed]
- Guntur, K.V.; Guilherme, A.; Xue, L.; Chawla, A.; Czech, M.P. Map4k4 negatively regulates peroxisome proliferator-activated receptor (PPAR) gamma protein translation by suppressing the mammalian target of rapamycin (mTOR) signaling pathway in cultured adipocytes. J. Biol. Chem. 2010, 285, 6595–6603. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Wang, Y.; Hui, L.; Li, X.; Jiang, X. Silencing SOX2 Expression by RNA Interference Inhibits Proliferation, Invasion and Metastasis, and Induces Apoptosis through MAP4K4/JNK Signaling Pathway in Human Laryngeal Cancer TU212 Cells. J. Histochem. Cytochem. 2015, 63, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Vuong, J.K.; Lin, C.H.; Zhang, M.; Chen, L.; Black, D.L.; Zheng, S. PTBP1 and PTBP2 Serve Both Specific and Redundant Functions in Neuronal Pre-mRNA Splicing. Cell Rep. 2016, 17, 2766–2775. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.H.; Wang, X.; Manning, G.; LaMere, B.J.; Le, P.; Zhu, S.; Khatry, D.; Flanagan, P.M.; Buckley, S.D.; Whyte, D.B.; et al. The STE20 kinase HGK is broadly expressed in human tumor cells and can modulate cellular transformation, invasion, and adhesion. Mol. Cell. Biol. 2003, 23, 2068–2082. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.C.; Han, J.; Xu, S.; Cobb, M.; Skolnik, E.Y. NIK is a new Ste20-related kinase that binds NCK and MEKK1 and activates the SAPK/JNK cascade via a conserved regulatory domain. EMBO J. 1997, 16, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Becker, E.; Huynh-Do, U.; Holland, S.; Pawson, T.; Daniel, T.O.; Skolnik, E.Y. Nck-interacting Ste20 kinase couples Eph receptors to c-Jun N-terminal kinase and integrin activation. Mol. Cell. Biol. 2000, 20, 1537–1545. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Guilherme, A.; Chakladar, A.; Powelka, A.M.; Konda, S.; Virbasius, J.V.; Nicoloro, S.M.; Straubhaar, J.; Czech, M.P. An RNA interference-based screen identifies MAP4K4/NIK as a negative regulator of PPARgamma, adipogenesis, and insulin-responsive hexose transport. Proc. Natl. Acad. Sci. USA 2006, 103, 2087–2092. [Google Scholar] [CrossRef] [PubMed]
- Tesz, G.J.; Guilherme, A.; Guntur, K.V.; Hubbard, A.C.; Tang, X.; Chawla, A.; Czech, M.P. Tumor necrosis factor alpha (TNFalpha) stimulates Map4k4 expression through TNFalpha receptor 1 signaling to c-Jun and activating transcription factor 2. J. Biol. Chem. 2007, 282, 19302–19312. [Google Scholar] [CrossRef] [PubMed]
- Isakson, P.; Hammarstedt, A.; Gustafson, B.; Smith, U. Impaired preadipocyte differentiation in human abdominal obesity: Role of Wnt, tumor necrosis factor-alpha, and inflammation. Diabetes 2009, 58, 1550–1557. [Google Scholar] [CrossRef] [PubMed]
- Danai, L.V.; Guilherme, A.; Guntur, K.V.; Straubhaar, J.; Nicoloro, S.M.; Czech, M.P. Map4k4 suppresses Srebp-1 and adipocyte lipogenesis independent of JNK signaling. J. Lipid Res. 2013, 54, 2697–2707. [Google Scholar] [CrossRef] [PubMed]
- Roth Flach, R.J.; Danai, L.V.; DiStefano, M.T.; Kelly, M.; Menendez, L.G.; Jurczyk, A.; Sharma, R.B.; Jung, D.Y.; Kim, J.H.; Kim, J.K.; et al. Protein Kinase Mitogen-activated Protein Kinase Kinase Kinase Kinase 4 (MAP4K4) Promotes Obesity-induced Hyperinsulinemia. J. Biol. Chem. 2016, 291, 16221–16230. [Google Scholar] [CrossRef] [PubMed]
- Harhouri, K.; Navarro, C.; Depetris, D.; Mattei, M.G.; Nissan, X.; Cau, P.; De Sandre-Giovannoli, A.; Lévy, N. MG132-induced progerin clearance is mediated by autophagy activation and splicing regulation. EMBO Mol. Med. 2017, 9, 1294–1313. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Bossing, T. Single neuron transcriptomics identify SRSF/SR protein B52 as a regulator of axon growth and Choline acetyltransferase splicing. Sci. Rep. 2016, 6, 34952. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, D.; Caponnetto, A.; Cirnigliaro, M.; Brex, D.; Barbagallo, C.; D’Angeli, F.; Morrone, A.; Caltabiano, R.; Barbagallo, G.M.; Ragusa, M.; et al. CircSMARCA5 Inhibits Migration of Glioblastoma Multiforme Cells by Regulating a Molecular Axis Involving Splicing Factors SRSF1/SRSF3/PTB. Int. J. Mol. Sci. 2018, 19, 480. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Jumaa, H.; Webster, N.J. Splicing factor SRSF3 is crucial for hepatocyte differentiation and metabolic function. Nat. Commun. 2013, 4, 1336. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, S.; Song, T.; Guan, X.; Zhang, R.; Chen, X. Trichostatin a Protects Dendritic Cells against Oxygen-Glucose Deprivation via the SRSF3/PKM2/Glycolytic Pathway. Front. Pharmacol. 2018, 9, 612. [Google Scholar] [CrossRef] [PubMed]
| (A) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample | Symbol | Accession No. | Locus | Length | Coverage | FPKM | p-Value | Percentage Isoform Index (NM_001177995) |
| WT BAT (E13.5) | PRDM16 | NM_001177995 | chr4:154316124-154636873 | 8433 | 2.3435 | 1.95142 | 0.0231 | 35.18% |
| PRDM16 | NM_027504 | chr4:154316124-154636873 | 8605 | 3.4659 | 3.59551 | 0.0347 | ||
| RBM4a−/− BAT (E13.5) | PRDM16 | NM_001177995 | chr4:154316124-154636873 | 8433 | 0 | 1.69 × 10−5 | 0.0219 | <1% |
| PRDM16 | NM_027504 | chr4:154316124-154636873 | 8605 | 4.35909 | 4.12531 | 0.0147 | ||
| WT BAT (P0) | PRDM16 | NM_001177995 | chr4:154316124-154636873 | 8433 | 4.112475 | 2.04078 | 0.0341 | >99% |
| PRDM16 | NM_027504 | chr4:154316124-154636873 | 8605 | 0 | 2.71 × 10−5 | 0.0157 | ||
| RBM4a−/− BAT (P0) | PRDM16 | NM_001177995 | chr4:154316124-154636873 | 8433 | 2.102418 | 1.65639 | 0.0267 | 77.24% |
| PRDM16 | NM_027504 | chr4:154316124-154636873 | 8605 | 1.112425 | 0.487891 | 0.0271 | ||
| (B) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Gene | Locus | Sample-1 | Sample-2 | FPKM-1 | FPKM-2 | Log 2 (Fold Change) | p-Value | q-Value |
| PRDM16 (E13.5) | chr4:154316124-154636873 | BAT-WT | BAT- RBM4a−/− | 8.21412 | 6.21324 | 0.403 | 0.0397 | 0.0455 |
| PRDM16 (P0) | chr4:154316124-154636873 | BAT-WT | BAT- RBM4a−/− | 12.0365 | 7.54704 | 0.674 | 0.0241 | 0.0361 |
| p = 0.0207 | p > 0.05 | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, H.-Y.; Liang, Y.-C.; Tan, T.-H.; Chuang, H.-C.; Lin, Y.-J.; Lin, J.-C. RBM4a-SRSF3-MAP4K4 Splicing Cascade Constitutes a Molecular Mechanism for Regulating Brown Adipogenesis. Int. J. Mol. Sci. 2018, 19, 2646. https://doi.org/10.3390/ijms19092646
Peng H-Y, Liang Y-C, Tan T-H, Chuang H-C, Lin Y-J, Lin J-C. RBM4a-SRSF3-MAP4K4 Splicing Cascade Constitutes a Molecular Mechanism for Regulating Brown Adipogenesis. International Journal of Molecular Sciences. 2018; 19(9):2646. https://doi.org/10.3390/ijms19092646
Chicago/Turabian StylePeng, Hui-Yu, Yu-Chih Liang, Tse-Hua Tan, Huai-Chia Chuang, Ying-Ju Lin, and Jung-Chun Lin. 2018. "RBM4a-SRSF3-MAP4K4 Splicing Cascade Constitutes a Molecular Mechanism for Regulating Brown Adipogenesis" International Journal of Molecular Sciences 19, no. 9: 2646. https://doi.org/10.3390/ijms19092646
APA StylePeng, H.-Y., Liang, Y.-C., Tan, T.-H., Chuang, H.-C., Lin, Y.-J., & Lin, J.-C. (2018). RBM4a-SRSF3-MAP4K4 Splicing Cascade Constitutes a Molecular Mechanism for Regulating Brown Adipogenesis. International Journal of Molecular Sciences, 19(9), 2646. https://doi.org/10.3390/ijms19092646





