Inhibitory Effect of Opuntia humifusa Fruit Water Extract on Solar Ultraviolet-Induced MMP-1 Expression
Abstract
1. Introduction
2. Results
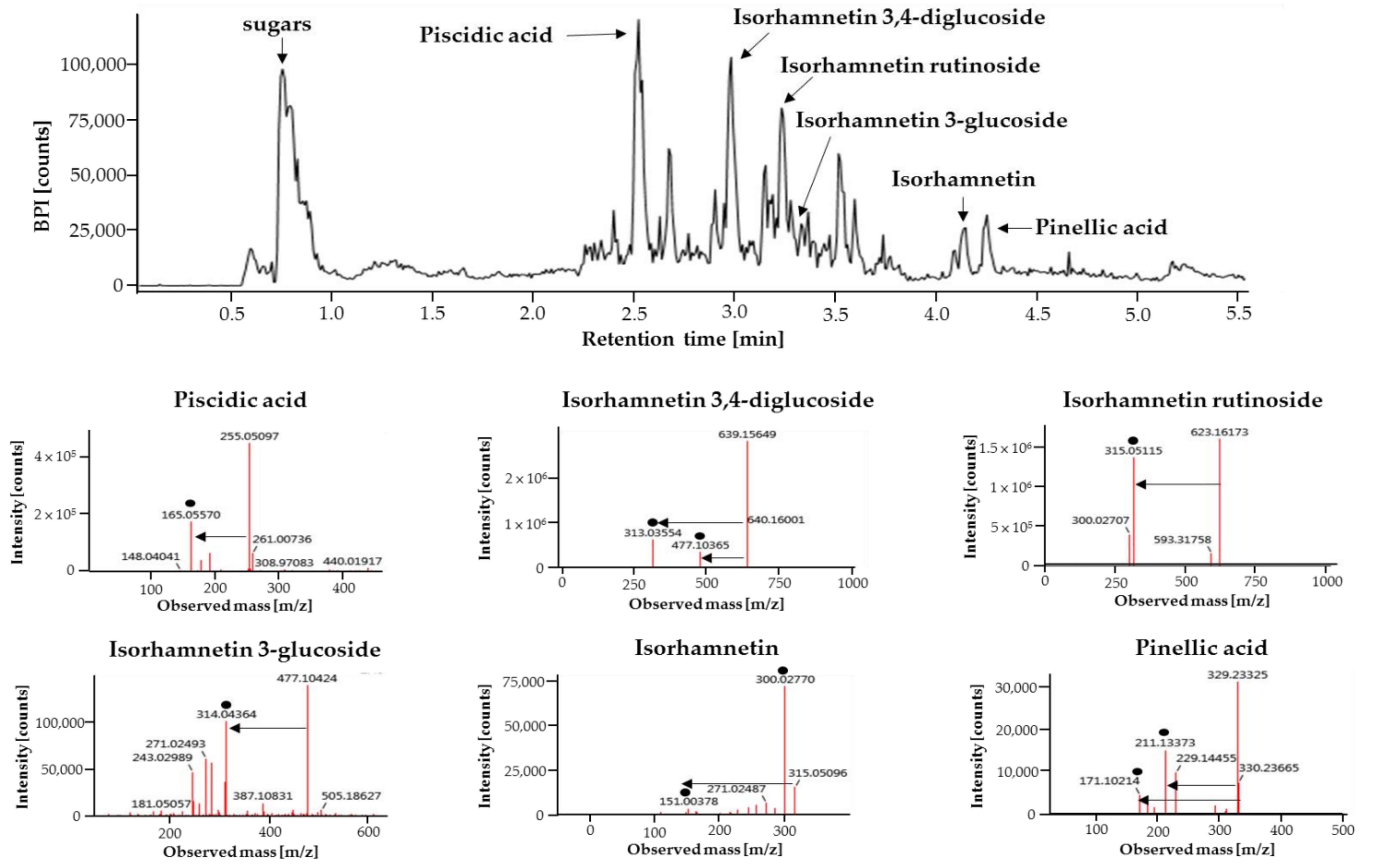
2.1. Analysis of Major Phytochemicals in OHE
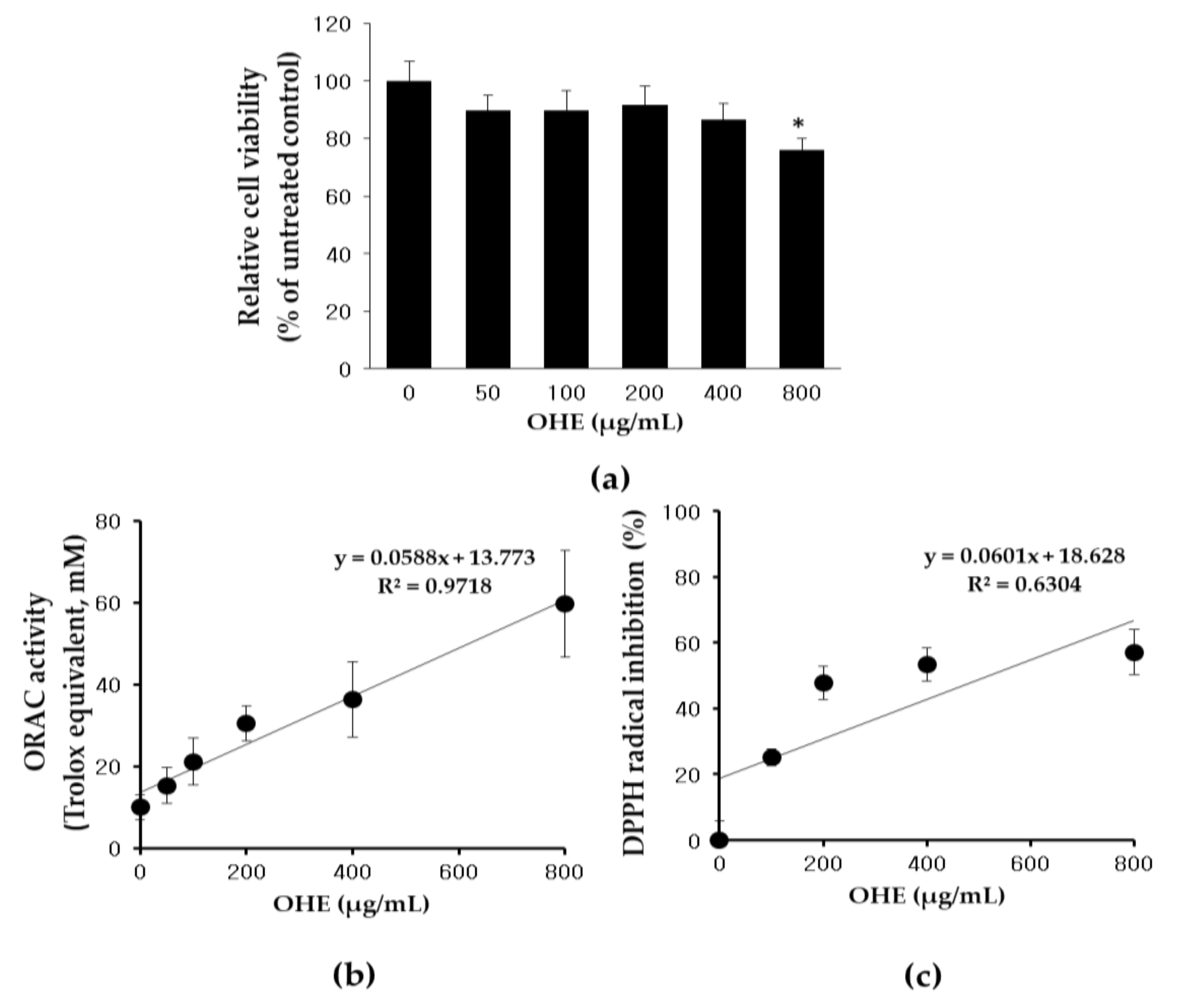
2.2. Effects of OHE on Cell Viability and Antioxidative Activity
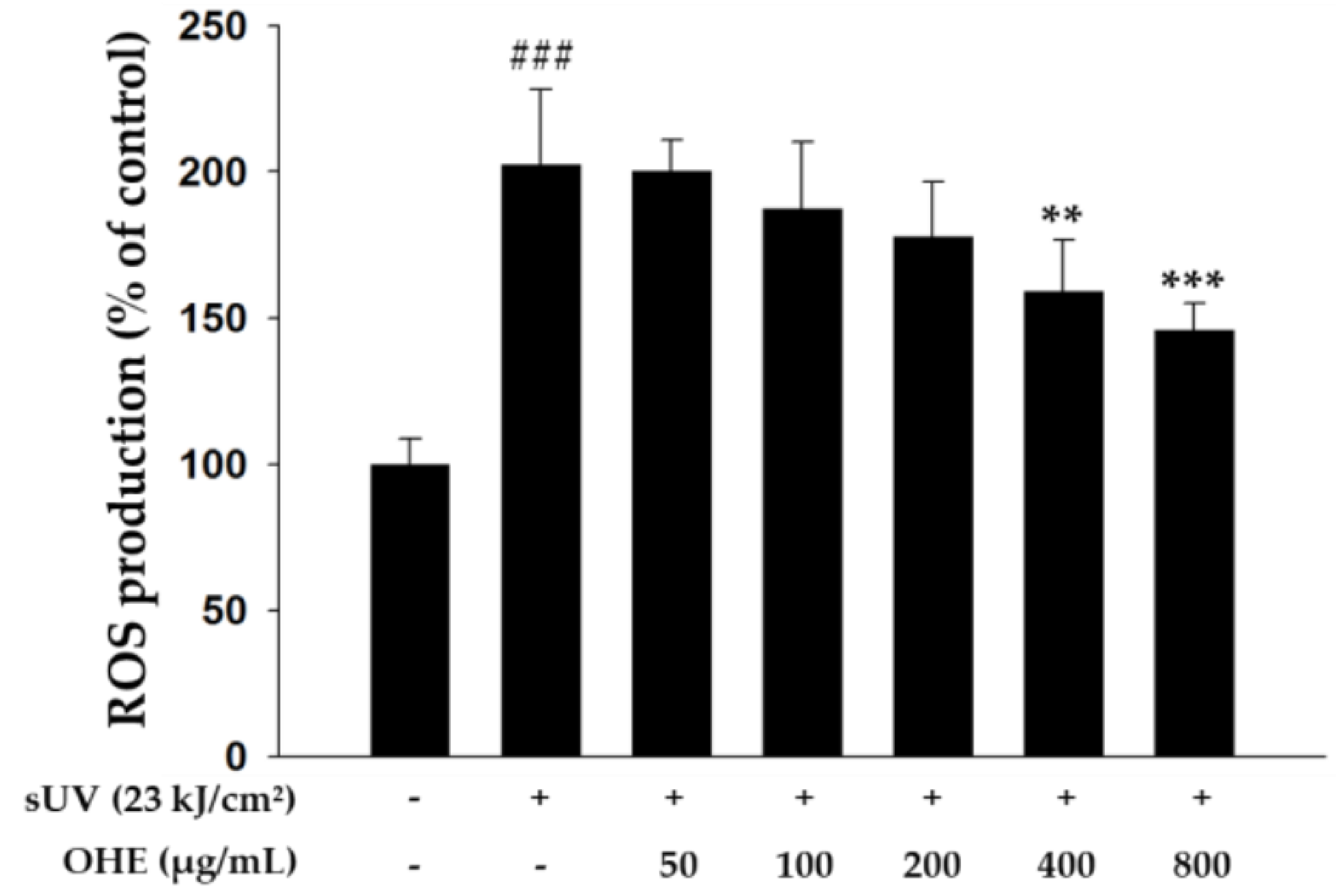
2.3. Effects of OHE on Intracellular ROS Production in Primary HDFs
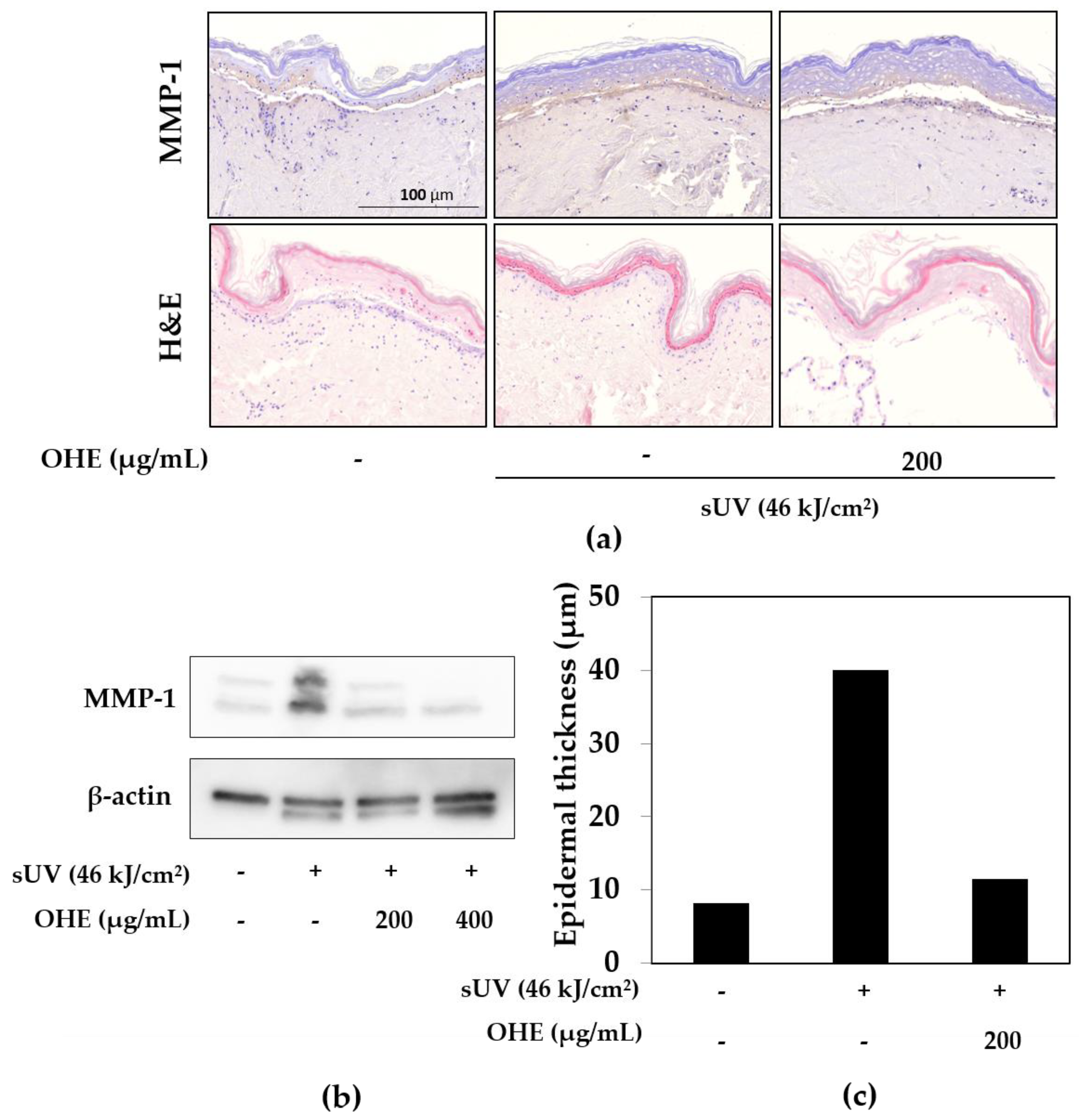
2.4. Effects of OHE on sUV-Induced Photoaging in Human Skin
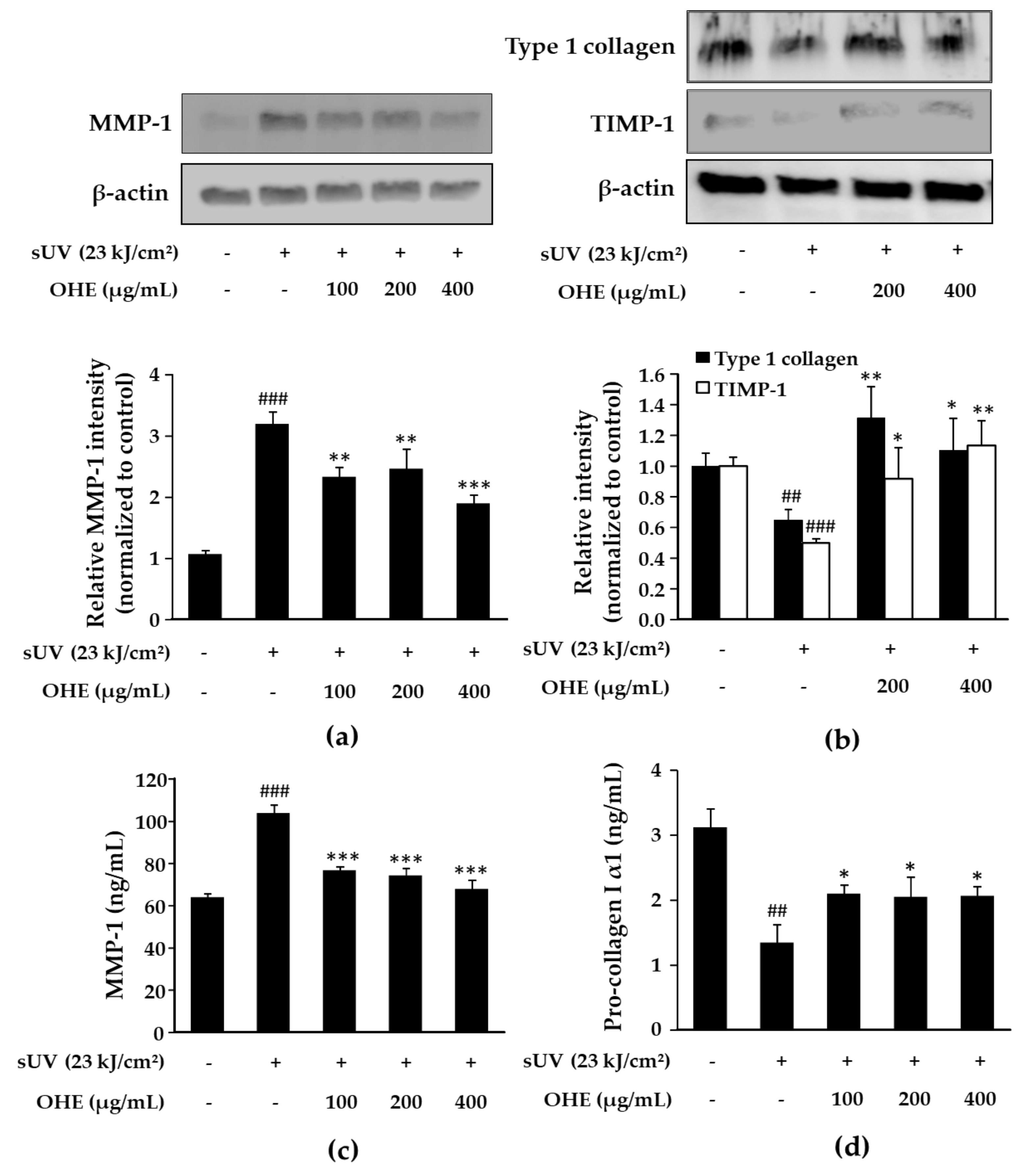
2.5. Effects of OHE on MMP-1, Type 1 Collagen, and Tissue Inhibitor of Matrix Metalloproteinase 1 (TIMP-1) Expression in sUV-Exposed Primary HDFs
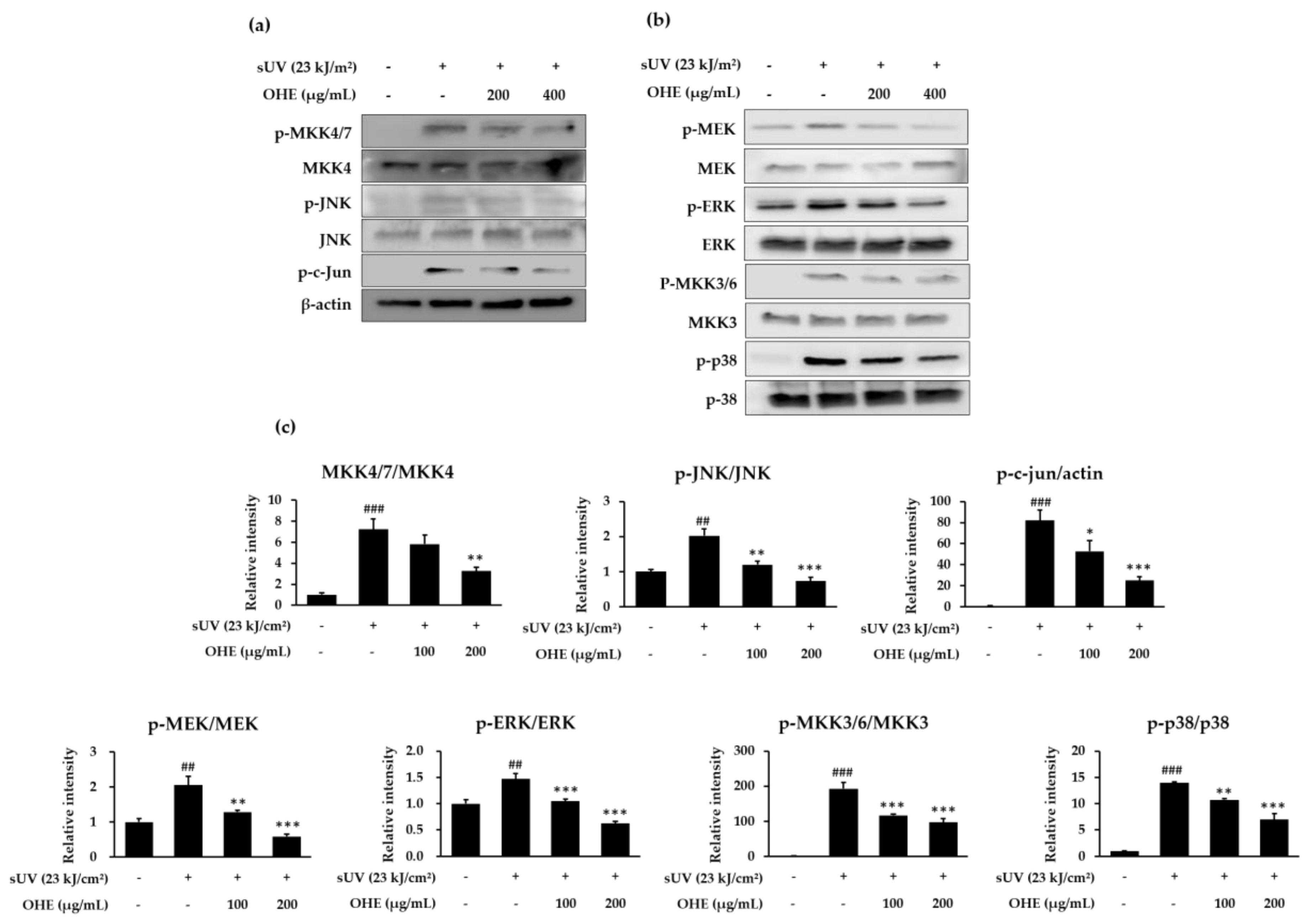
2.6. OHE Suppresses sUV-Induced MKK4/JNK/c-Jun, MEK/ERK, and MKK3/6/p38 Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Sample Preparation and Extraction Procedure
4.3. Analysis of Phytochemicals in OHE
4.4. Cell Culture
4.5. sUV Light Exposure
4.6. Cell Viability Assay
4.7. Measurement of MMP-1 and Type 1 Procollagen Production
4.8. Western Blot
4.9. DPPH Radical Scavenging Assay
4.10. Measurement of Intracellular ROS Production
4.11. ORAC Assay
4.12. Human Skin Sample Preparation
4.13. Immunohistochemical Staining
4.14. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AAPH | 2,20-azobis-(2-amidinopropane) dihydrochloride |
| AP-1 | Activator protein 1 |
| DMEM | Dulbecco’s modified Eagle medium |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ECM | Extracellular matrix |
| ERK | Extracellular signal-regulated kinases |
| ESI | Electrospray ionization |
| FA | Formic acid |
| FBS | Fetal bovine serum |
| HDFs | Human dermal fibroblasts |
| H&E | Hematoxylin and eosin |
| JNK | c-Jun N-terminal kinases |
| LC | Liquid chromatography |
| MAPK | Mitogen-activated protein kinase |
| MAPKK | MAPK kinase |
| MMP-1 | Matrix metalloproteinase-1 |
| MS | Mass spectrometry |
| MS/MS | Tandem mass spectrometry |
| MTS | 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H tetrazolium |
| OHE | Opuntia humifusa fruit water extract |
| ORAC | Oxygen radical absorbance capacity |
| ROS | Reactive oxygen species |
| SD | Standard deviation |
| sUV | Solar ultraviolet |
| TIMP-1 | Tissue inhibitor of matrix metalloproteinase 1 |
| UPLC-Q-TOF | Ultra-performance liquid chromatography–quadrupole-time of flight |
| UV | Ultraviolet |
References
- Poljšak, B.; Dahmane, R. Free radicals and extrinsic skin aging. Dermatol. Res. Pract. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Kang, S.; Varani, J.; Bata-Csorgo, Z.; Wan, Y.; Datta, S.; Voorhees, J.J. Mechanisms of photoaging and chronological skin aging. Arch. Dermatol. 2002, 138, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Kohl, E.; Steinbauer, J.; Landthaler, M.; Szeimies, R.M. Skin ageing. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 873–884. [Google Scholar] [CrossRef] [PubMed]
- Kammeyer, A.; Luiten, R. Oxidation events and skin aging. Ageing Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.-Y.; Choi, J.-S.; Choi, Y.-J.; Shin, S.-Y.; Kang, S.-W.; Han, S.J.; Kang, Y.-H. (−)Epigallocatechin gallate hampers collagen destruction and collagenase activation in ultraviolet-B-irradiated human dermal fibroblasts: Involvement of mitogen-activated protein kinase. Food Chem. Toxicol. 2008, 46, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Karin, M. The regulation of AP-1 activity by mitogen-activated protein kinases. J. Biol. Chem. 1995, 270, 16483–16486. [Google Scholar] [CrossRef] [PubMed]
- Rittié, L.; Fisher, G.J. UV-light-induced signal cascades and skin aging. Ageing Res. Rev. 2002, 1, 705–720. [Google Scholar] [CrossRef]
- Brennan, M.; Bhatti, H.; Nerusu, K.C.; Bhagavathula, N.; Kang, S.; Fisher, G.J.; Varani, J.; Voorhees, J.J. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem. Photobiol. 2003, 78, 43–48. [Google Scholar] [CrossRef]
- Zorgui, L.; Ayed-Boussema, I.; Ayed, Y.; Bacha, H.; Hassen, W. The antigenotoxic activities of cactus (Opuntia ficus-indica) cladodes against the mycotoxin zearalenone in Balb/c mice: Prevention of micronuclei, chromosome aberrations and DNA fragmentation. Food Chem. Toxicol. 2009, 47, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.-J.; You, S.-H. Bioactive characteristics of extracts of Opuntia humifusa fruit as functional cosmetic ingredients. Asian J. Beauty Cosmetol. 2016, 14, 463–472. [Google Scholar] [CrossRef]
- Han, S.H.; Park, K.; Kim, E.Y.; Ahn, S.H.; Lee, H.-S.; Suh, H.J. Cactus (Opuntia humifusa) water extract ameliorates loperamide-induced constipation in rats. BMC Complement. Altern. Med. 2017, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- Cha, M.-N.; Jun, H.-I.; Lee, W.-J.; Kim, M.-J.; Kim, M.-K.; Kim, Y.-S. Chemical composition and antioxidant activity of Korean cactus (Opuntia humifusa) fruit. Food Sci. Biotechnol. 2013, 22, 523–529. [Google Scholar] [CrossRef]
- Cho, J.Y.; Park, S.C.; Kim, T.W.; Kim, K.S.; Song, J.C.; Lee, H.M.; Sung, H.J.; Rhee, M.H.; Kim, S.K.; Park, H.J. Radical scavenging and anti-inflammatory activity of extracts from Opuntia humifusa Raf. J. Pharm. Pharmacol. 2006, 58, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Feugang, J.M.; Konarski, P.; Zou, D.; Stintzing, F.C.; Zou, C. Nutritional and medicinal use of Cactus pear (Opuntia spp.) cladodes and fruits. Front. Biosci. 2006, 11, 2574–2589. [Google Scholar] [CrossRef] [PubMed]
- Hahm, S.-W.; Park, J.; Oh, S.-Y.; Lee, C.-W.; Park, K.-Y.; Kim, H.; Son, Y.-S. Anticancer properties of extracts from Opuntia humifusa against human cervical carcinoma cells. J. Med. Food 2015, 18, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Choi, H.-S.; Hong, Y.H.; Jung, E.Y.; Suh, H.J. Cactus cladodes (Opuntia humifusa) extract minimizes the effects of UV irradiation on keratinocytes and hairless mice. Pharm. Biol. 2017, 55, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Cawston, T. Metalloproteinase inhibitors and the prevention of connective tissue breakdown. Pharmacol. Ther. 1996, 70, 163–182. [Google Scholar] [CrossRef]
- Cho, H.S.; Lee, M.H.; Lee, J.W.; No, K.O.; Park, S.K.; Lee, H.S.; Kang, S.; Cho, W.G.; Park, H.J.; Oh, K.W. Anti-wrinkling effects of the mixture of vitamin C, vitamin E, pycnogenol and evening primrose oil, and molecular mechanisms on hairless mouse skin caused by chronic ultraviolet B irradiation. Photodermatol. Photoimmunol. Photomed. 2007, 23, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.-G.; Kim, J.-E.; Jung, S.K.; Li, Y.; Bode, A.M.; Park, J.-S.; Yeom, M.H.; Dong, Z.; Lee, K.W. MLK3 is a direct target of biochanin A, which plays a role in solar UV-induced COX-2 expression in human keratinocytes. Biochem. Pharmacol. 2013, 86, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Millis, A.J.; Hoyle, M.; McCue, H.M.; Martini, H. Differential expression of metalloproteinase and tissue inhibitor of metalloproteinase genes in aged human fibroblasts. Exp. Cell Res. 1992, 201, 373–379. [Google Scholar] [CrossRef]
- Kim, J.M.; Noh, E.M.; Kwon, K.B.; Hwang, B.M.; Hwang, J.K.; You, Y.O.; Kim, M.S.; Lee, W.; Lee, J.H.; Kim, H.J. Dihydroavenanthramide D prevents UV-irradiated generation of reactive oxygen species and expression of matrix metalloproteinase-1 and-3 in human dermal fibroblasts. Exp. Dermatol. 2013, 22, 759–761. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Wang, Z.; Datta, S.C.; Varani, J.; Kang, S.; Voorhees, J.J. Pathophysiology of premature skin aging induced by ultraviolet light. N. Engl. J. Med. 1997, 337, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, G.; Nobel, P.S. Water relations and low-temperature acclimation for cactus species varying in freezing tolerance. Plant Physiol. 1994, 104, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Kim, T. Korean Resources Plants; Seoul National University: Seoul, Korea, 1996; p. 169. [Google Scholar]
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C. Food colorants: Challenges, opportunities and current desires of agro-industries to ensure consumer expectations and regulatory practices. Trends Food Sci. Technol. 2016, 52, 1–15. [Google Scholar] [CrossRef]
- Kuti, J.O. Antioxidant compounds from four Opuntia cactus pear fruit varieties. Food Chem. 2004, 85, 527–533. [Google Scholar] [CrossRef]
- Serra, A.T.; Poejo, J.; Matias, A.A.; Bronze, M.R.; Duarte, C.M. Evaluation of Opuntia spp. derived products as antiproliferative agents in human colon cancer cell line (HT29). Food Res. Int. 2013, 54, 892–901. [Google Scholar] [CrossRef]
- Takahira, M.; Kusano, A.; Shibano, M.; Kusano, G.; Miyase, T. Piscidic acid and fukiic acid esters from Cimicifuga simplex. Phytochemistry 1998, 49, 2115–2119. [Google Scholar] [CrossRef]
- Igarashi, K.; Ohmuma, M. Effects of isorhamnetin, rhamnetin, and quercetin on the concentrations of cholesterol and lipoperoxide in the serum and liver and on the blood and liver antioxidative enzyme activities of rats. Biosci. Biotechnol. Biochem. 1995, 59, 595–601. [Google Scholar] [CrossRef] [PubMed]
- González-Peña, D.; Colina-Coca, C.; Char, C.D.; Cano, M.P.; de Ancos, B.A.; Sánchez-Moreno, C.N. Hyaluronidase inhibiting activity and radical scavenging potential of flavonols in processed onion. J. Agric. Food Chem. 2013, 61, 4862–4872. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.Y.; Zheng, H.C.; Zhang, H.W.; Zhang, J.Y.; Ma, C.M. Comparison of Antioxidant Constituents of Agriophyllum squarrosum Seed with Conventional Crop Seeds. J. Food Sci. 2018, 83, 1823–1831. [Google Scholar] [CrossRef] [PubMed]
- Devi, V.G.; Rooban, B.; Sasikala, V.; Sahasranamam, V.; Abraham, A. Isorhamnetin-3-glucoside alleviates oxidative stress and opacification in selenite cataract in vitro. Toxicol. In Vitro 2010, 24, 1662–1669. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-E.; Roh, E.; Lee, M.H.; Yu, D.H.; Kim, D.J.; Lim, T.-G.; Jung, S.K.; Peng, C.; Cho, Y.-Y.; Dickinson, S. Fyn is a redox sensor involved in solar ultraviolet light-induced signal transduction in skin carcinogenesis. Oncogene 2016, 35, 4091–4101. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.K.; Koide, M.; Rao, T.P.; Okubo, T.; Ogasawara, Y.; Juneja, L.R. ORAC and DPPH assay comparison to assess antioxidant capacity of tea infusions: Relationship between total polyphenol and individual catechin content. Int. J. Food Sci. Nutr. 2010, 61, 109–124. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, A.-R.; Lim, T.-G.; Song, Y.-R.; Jang, M.; Rhee, Y.K.; Hong, H.-D.; Kim, M.-H.; Kim, H.-J.; Cho, C.-W. Inhibitory Effect of Opuntia humifusa Fruit Water Extract on Solar Ultraviolet-Induced MMP-1 Expression. Int. J. Mol. Sci. 2018, 19, 2503. https://doi.org/10.3390/ijms19092503
Han A-R, Lim T-G, Song Y-R, Jang M, Rhee YK, Hong H-D, Kim M-H, Kim H-J, Cho C-W. Inhibitory Effect of Opuntia humifusa Fruit Water Extract on Solar Ultraviolet-Induced MMP-1 Expression. International Journal of Molecular Sciences. 2018; 19(9):2503. https://doi.org/10.3390/ijms19092503
Chicago/Turabian StyleHan, Ah-Ram, Tae-Gyu Lim, Young-Ran Song, Mi Jang, Young Kyoung Rhee, Hee-Do Hong, Mi-Hyun Kim, Hyun-Jin Kim, and Chang-Won Cho. 2018. "Inhibitory Effect of Opuntia humifusa Fruit Water Extract on Solar Ultraviolet-Induced MMP-1 Expression" International Journal of Molecular Sciences 19, no. 9: 2503. https://doi.org/10.3390/ijms19092503
APA StyleHan, A.-R., Lim, T.-G., Song, Y.-R., Jang, M., Rhee, Y. K., Hong, H.-D., Kim, M.-H., Kim, H.-J., & Cho, C.-W. (2018). Inhibitory Effect of Opuntia humifusa Fruit Water Extract on Solar Ultraviolet-Induced MMP-1 Expression. International Journal of Molecular Sciences, 19(9), 2503. https://doi.org/10.3390/ijms19092503





