Modified Methacrylate Hydrogels Improve Tissue Repair after Spinal Cord Injury
Abstract
:1. Introduction
2. Results
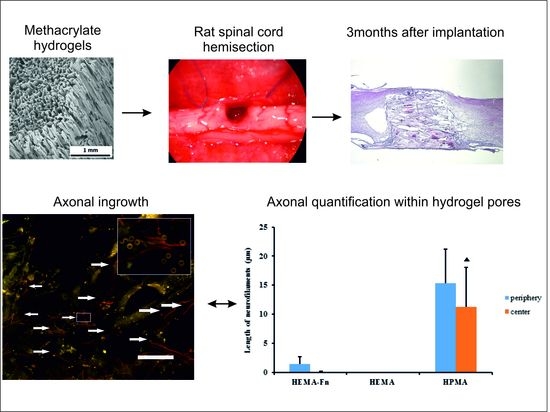
2.1. Hydrogel Scaffold
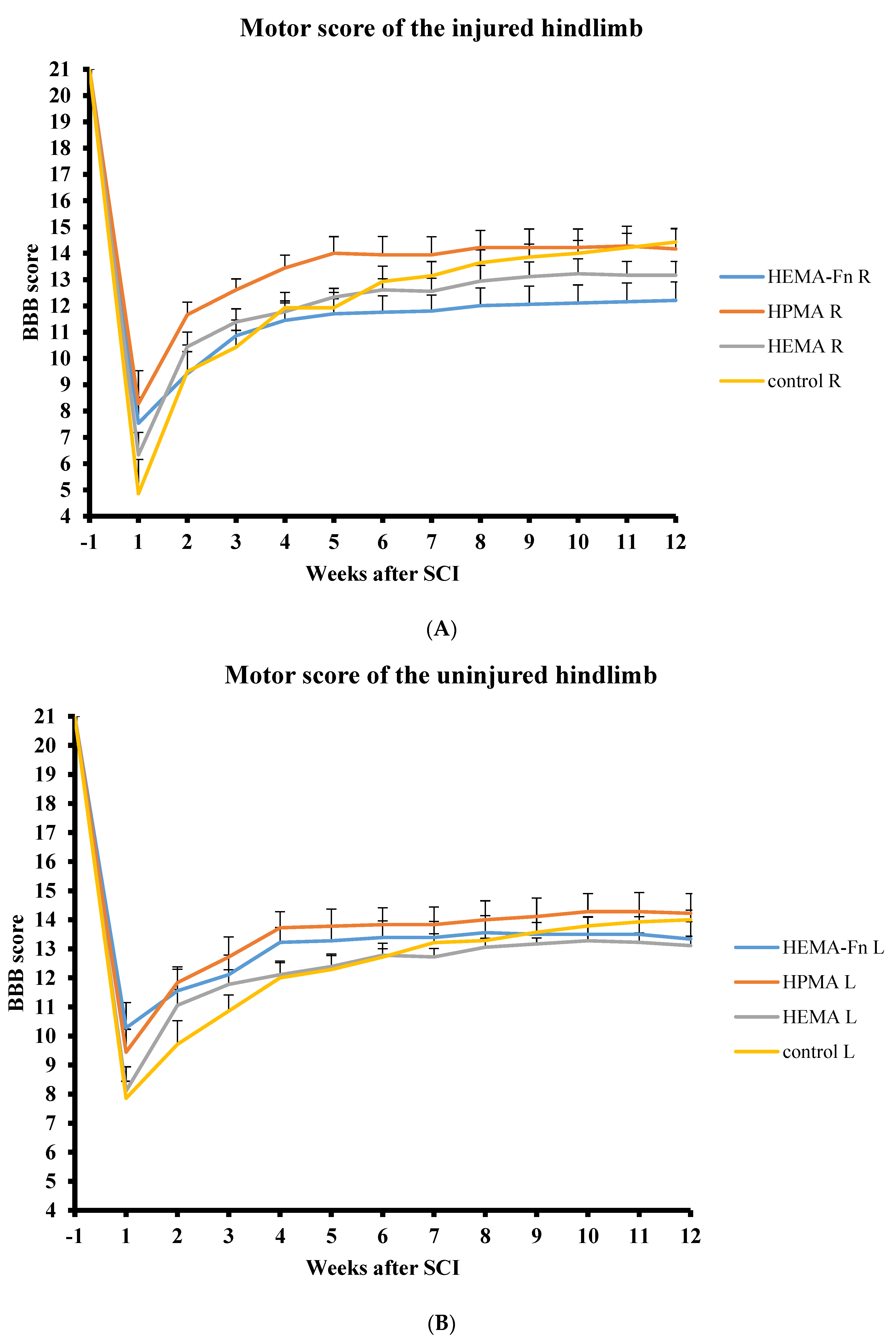
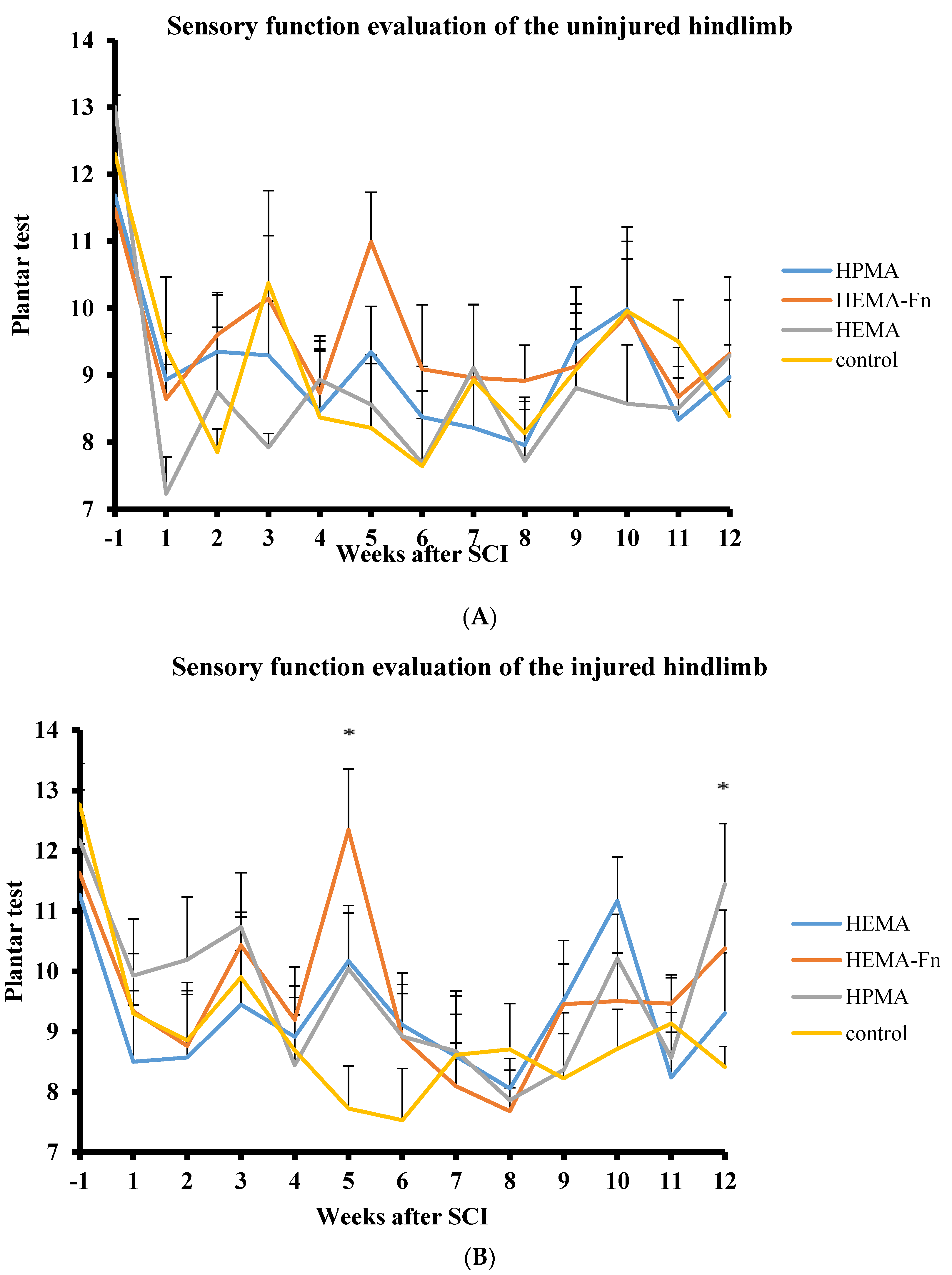
2.2. Functional Tests
2.3. BBB
2.4. Plantar Test
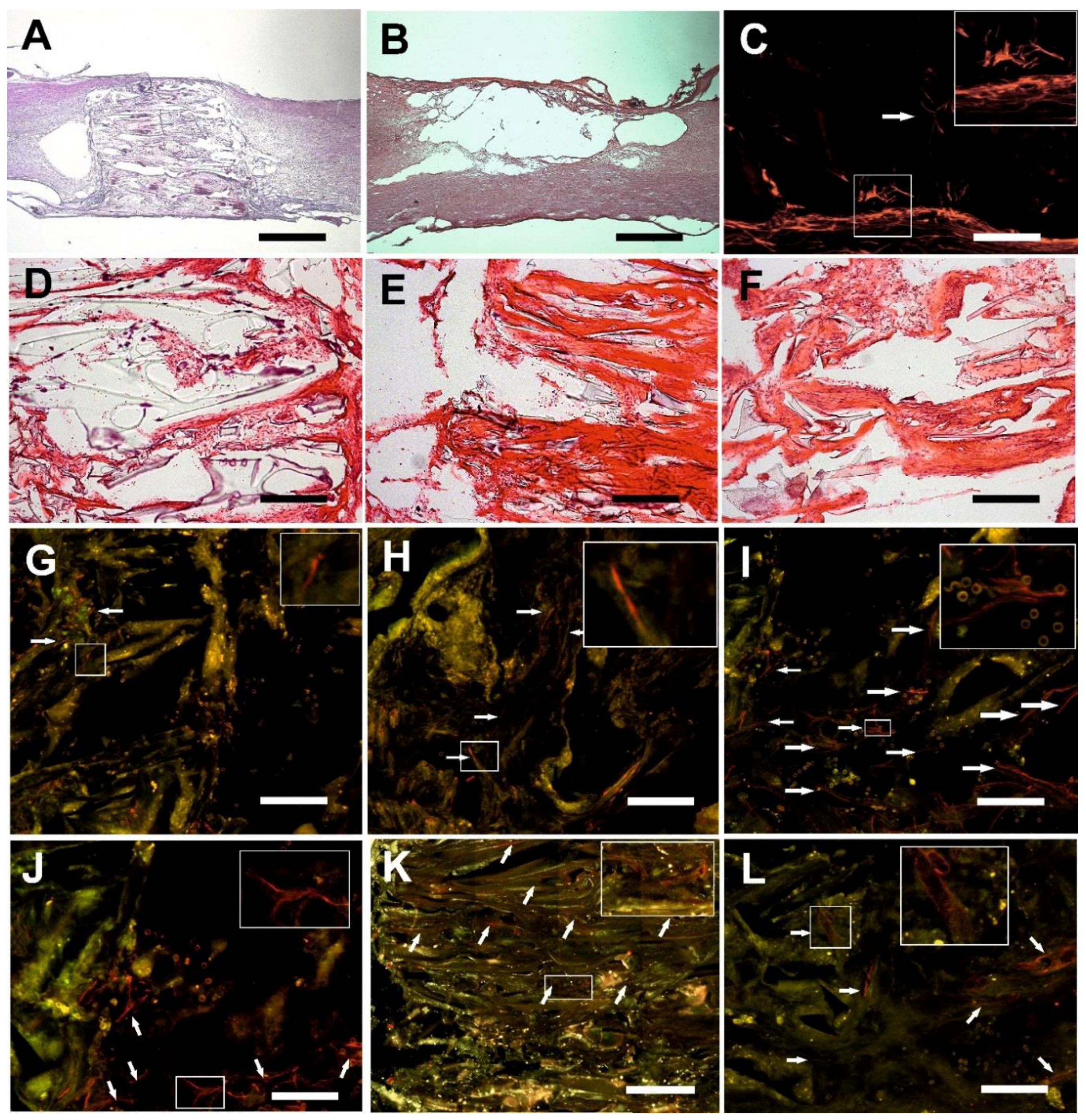
2.5. Microscopic Evaluation of the Hydrogel Bridge
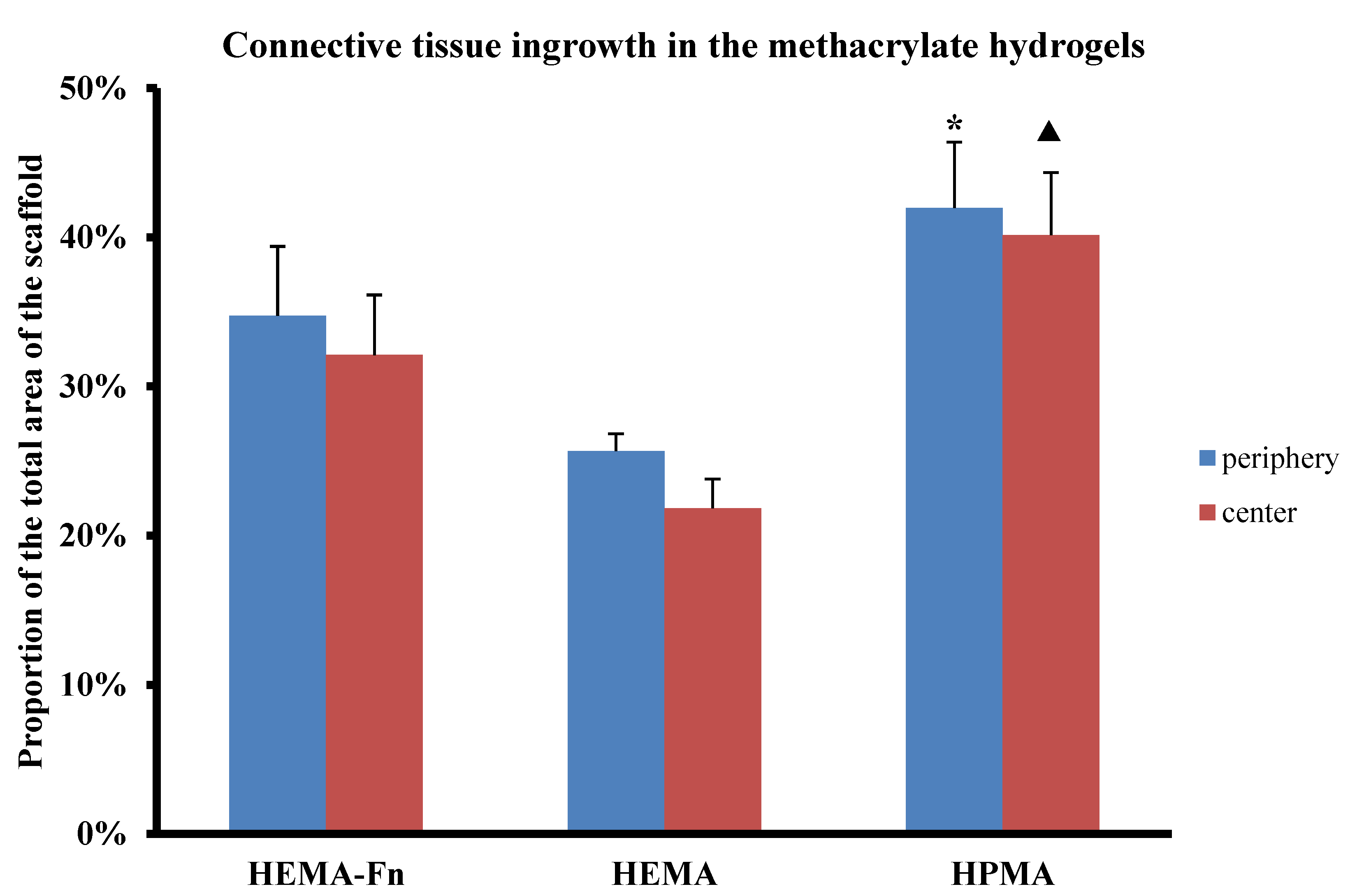
2.6. Connective Tissue
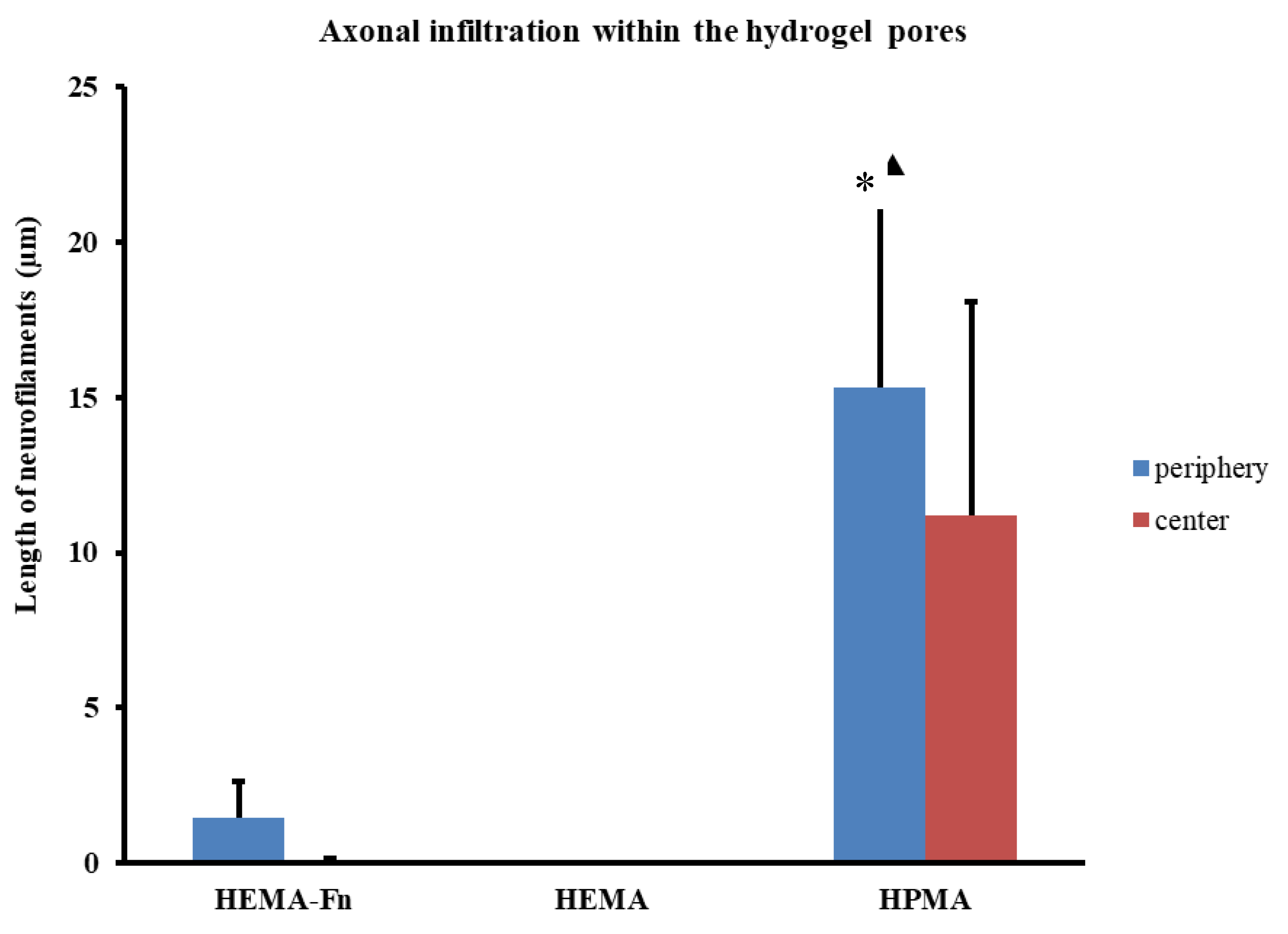
2.7. Axonal Regeneration
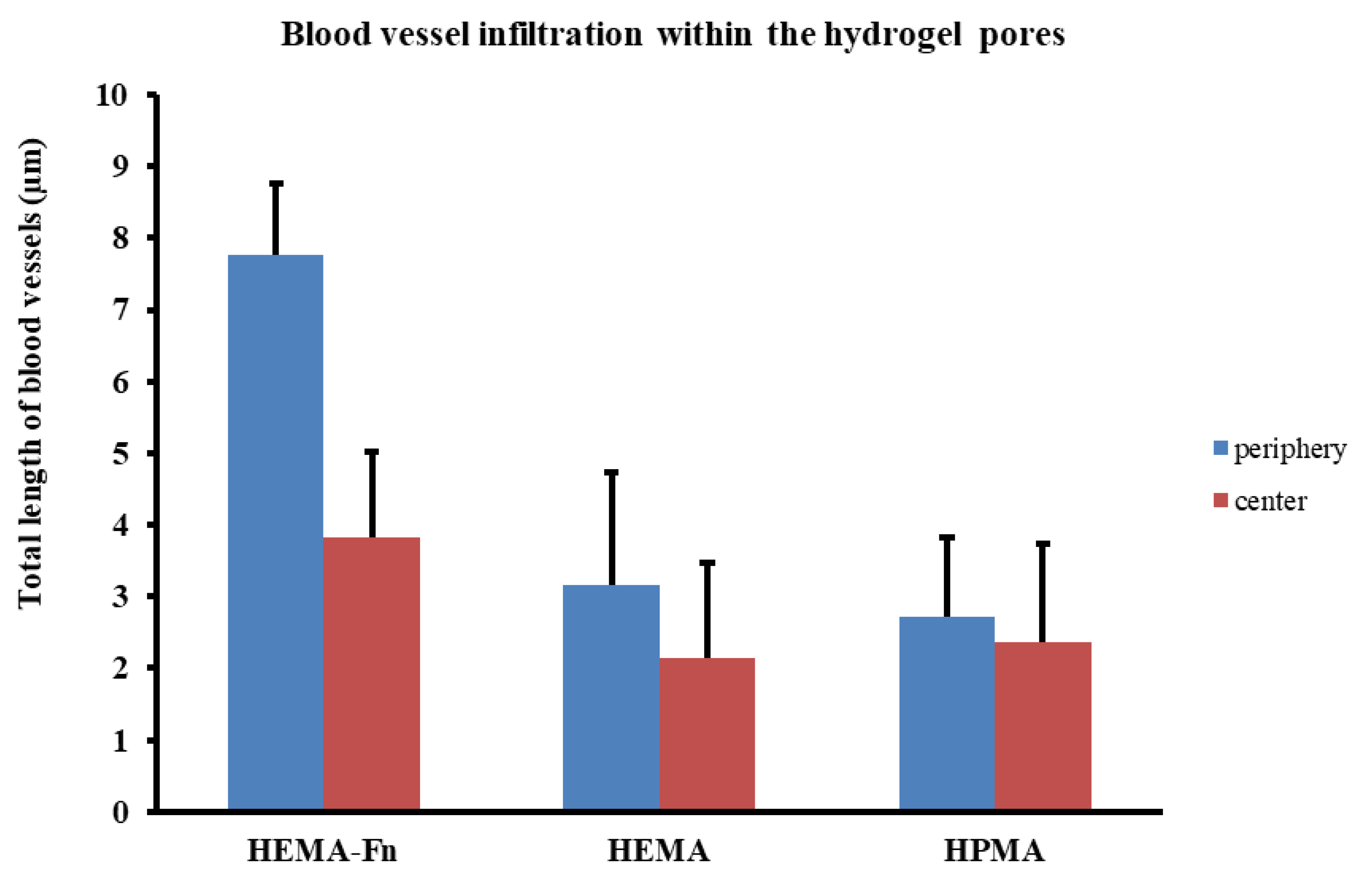
2.8. Growth of Blood Vessels in the Hydrogels
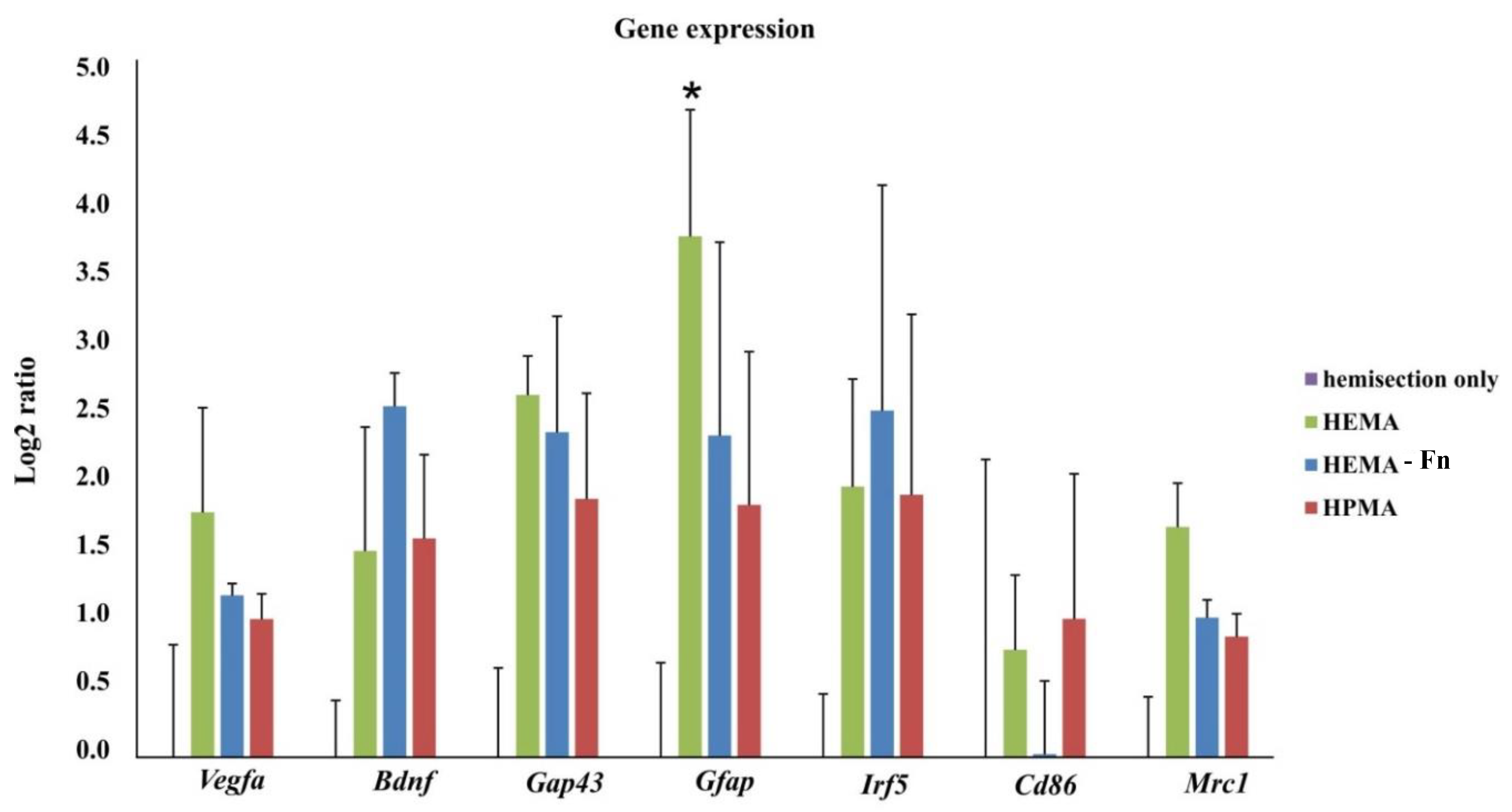
2.9. Gene Expression
3. Discussion
4. Materials and Methods
4.1. Hydrogel Scaffolds
Preparation of Superporouspoly (2-Hydroxyethyl Methacrylate) (HEMA), HEMA-Fn, and Poly [(N-(2-Hydroxypropyl) Methacrylamide] (HPMA) Hydrogels
4.2. Animal Handling and Surgery
4.3. Spinal Cord Injury and Hydrogel Implantation
4.4. Behavioral Testing
BBB Test
4.5. Plantar
4.6. Tissue Processing and Histology
4.7. Tissue Quantification
4.8. qPCR
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Atala, R.; Langer, R.; Thomson, J.; Nerem, R. Principles of Regenerative Medicine; Academic Press: Burlington, MA, USA, 2008. [Google Scholar]
- Hu, B.H.; Su, J.; Messersmith, P.B. Hydrogels cross-linked by native chemical ligation. Biomacromolecules 2009, 10, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in regenerative medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2002, 54, 3–12. [Google Scholar] [CrossRef]
- Shoichet, M. Polymer scaffolds for Biomaterials Applications. Macromolecules 2010, 43, 581–591. [Google Scholar] [CrossRef]
- Ahuja, C.S.; Fehlings, M. Concise Review: Bridging the Gap: Novel Neuroregenerative and Neuroprotective Strategies in Spinal Cord Injury. Stem Cells Transl. Med. 2016, 5, 914–924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Estrada, V.; Brazda, N.; Schmitz, C.; Heller, S.; Blazyca, H.; Martini, R.; Muller, H.W. Long-lasting significant functional improvement in chronic severe spinal cord injury following scar resection and polyethylene glycol implantation. Neurobiol. Dis. 2014, 67, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.R.; Euteneuer, S.; Chavez, E.; Mullen, L.M.; Hui, E.E.; Bhatia, S.N.; Ryan, A.F. Laminin and fibronectin modulate inner ear spiral ganglion neurite outgrowth in an in vitro alternate choice assay. Dev. Neurobiol. 2007, 67, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.K.; Knight, A.M.; de Ruiter, G.C.; Spinner, R.J.; Yaszemski, M.J.; Currier, B.L.; Windebank, A.J. Axon regeneration through scaffold into distal spinal cord after transection. J. Neurotrauma 2009, 26, 1759–1771. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.; Shanks, H.; Witt, A.; Rutkowski, G.; Mallapragada, S. Oriented Schwann cell growth on micropatterned biodegradable polymer substrates. Biomaterials 2001, 22, 1263–1269. [Google Scholar] [CrossRef]
- Zhu, Y.; Soderblom, C.; Trojanowsky, M.; Lee, D.H.; Lee, J.K. Fibronectin Matrix Assembly after Spinal Cord Injury. J. Neurotrauma 2015, 32, 1158–1167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Novikova, L.N.; Pettersson, J.; Brohlin, M.; Wiberg, M.; Novikov, L.N. Biodegradable poly-beta-hydroxybutyrate scaffold seeded with Schwann cells to promote spinal cord repair. Biomaterials 2008, 29, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Venstrom, K.A.; Reichardt, L.F. Extracellular matrix. 2: Role of extracellular matrix molecules and their receptors in the nervous system. Off. Publ. Fed. Am. Soc. Exp. Biol. 1993, 7, 996–1003. [Google Scholar] [CrossRef]
- Tysseling-Mattiace, V.M.; Sahni, V.; Niece, K.L.; Birch, D.; Czeisler, C.; Fehlings, M.G.; Stupp, S.I.; Kessler, J.A. Self-assembling nanofibers inhibit glial scar formation and promote axon elongation after spinal cord injury. J. Neurosci. 2008, 28, 3814–3823. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Lesny, P.; Pradny, M.; Michalek, J.; Jendelova, P.; Stulik, J.; Sykova, E. Biocompatible hydrogels in spinal cord injury repair. Physiol. Res. 2008, 57 (Suppl. 3), S121–S132. [Google Scholar] [PubMed]
- Pradny, M.; Michalek, J.; Lesny, P.; Hejcl, A.; Vacik, J.; Slouf, M.; Sykova, E. Macroporous hydrogels based on 2-hydroxyethyl methacrylate. Part 5: Hydrolytically degradable materials. J. Mater. Sci. Mater. Med. 2006, 17, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Kubinova, S.; Horak, D.; Hejcl, A.; Plichta, Z.; Kotek, J.; Sykova, E. Highly superporous cholesterol-modified poly(2-hydroxyethyl methacrylate) scaffolds for spinal cord injury repair. J. Biomed. Mater. Res. A 2011, 99, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Zaviskova, K.; Tukmachev, D.; Dubisova, J.; Vackova, I.; Hejcl, A.; Bystronova, J.; Pravda, M.; Scigalkova, I.; Sulakova, R.; Velebny, V.; et al. Injectable hydroxyphenyl derivative of hyaluronic acid hydrogel modified with rgd as scaffold for spinal cord injury repair. J. Biomed. Mater. Res. A 2018, 106, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Sykova, E.; Jendelova, P.; Urdzikova, L.; Lesny, P.; Hejcl, A. Bone marrow stem cells and polymer hydrogels—Two strategies for spinal cord injury repair. Cell Mol. Neurobiol. 2006, 26, 1113–1129. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Urdzikova, L.; Sedy, J.; Lesny, P.; Pradny, M.; Michalek, J.; Burian, M.; Hajek, M.; Zamecnik, J.; Jendelova, P.; et al. Acute and delayed implantation of positively charged 2-hydroxyethyl methacrylate scaffolds in spinal cord injury in the rat. J. Neurosurg. Spine 2008, 8, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Kubinova, S.; Horak, D.; Hejcl, A.; Plichta, Z.; Kotek, J.; Proks, V.; Forostyak, S.; Sykova, E. SIKVAV-modified highly superporous PHEMA scaffolds with oriented pores for spinal cord injury repair. J. Tissue Eng. Regen. Med. 2015, 9, 1298–1309. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Ruzicka, J.; Kapcalova, M.; Turnovcova, K.; Krumbholcova, E.; Pradny, M.; Michalek, J.; Cihlar, J.; Jendelova, P.; Sykova, E. Adjusting the chemical and physical properties of hydrogels leads to improved stem cell survival and tissue ingrowth in spinal cord injury reconstruction: A comparative study of four methacrylate hydrogels. Stem Cells Dev. 2013, 22, 2794–2805. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Lesny, P.; Pradny, M.; Sedy, J.; Zamecnik, J.; Jendelova, P.; Michalek, J.; Sykova, E. Macroporous hydrogels based on 2-hydroxyethyl methacrylate. Part 6: 3D hydrogels with positive and negative surface charges and polyelectrolyte complexes in spinal cord injury repair. J. Mater. Sci. Mater. Med. 2009, 20, 1571–1577. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Sedy, J.; Kapcalova, M.; Toro, D.A.; Amemori, T.; Lesny, P.; Likavcanova-Masinova, K.; Krumbholcova, E.; Pradny, M.; Michalek, J.; et al. HPMA-RGD hydrogels seeded with mesenchymal stem cells improve functional outcome in chronic spinal cord injury. Stem Cells Dev. 2010, 19, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Filippov, S.; Hruby, M.; Konak, C.; Mackova, H.; Spirkova, M.; Stepanek, P. Novel pH-responsive nanoparticles. Langmuir 2008, 24, 9295–9301. [Google Scholar] [CrossRef] [PubMed]
- Bruzauskaite, I.; Bironaite, D.; Bagdonas, E.; Bernotiene, E. Scaffolds and cells for tissue regeneration: Different scaffold pore sizes-different cell effects. Cytotechnology 2016, 68, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Flynn, L.; Dalton, P.D.; Shoichet, M.S. Fiber templating of poly(2-hydroxyethyl methacrylate) for neural tissue engineering. Biomaterials 2003, 24, 4265–4272. [Google Scholar] [CrossRef]
- Woerly, S.; Pinet, E.; de Robertis, L.; Van Diep, D.; Bousmina, M. Spinal cord repair with PHPMA hydrogel containing RGD peptides (NeuroGel). Biomaterials 2001, 22, 1095–1111. [Google Scholar] [CrossRef]
- Hejcl, A.; Jendelova, P.; Sykova, E. Experimental reconstruction of the injured spinal cord. Adv. Technol. Stand. Neurosurg. 2011, 37, 65–95. [Google Scholar]
- Loh, N.K.; Woerly, S.; Bunt, S.M.; Wilton, S.D.; Harvey, A.R. The regrowth of axons within tissue defects in the CNS is promoted by implanted hydrogel matrices that contain BDNF and CNTF producing fibroblasts. Exp. Neurol. 2001, 170, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Plant, G.W.; Woerly, S.; Harvey, A.R. Hydrogels containing peptide or aminosugar sequences implanted into the rat brain: Influence on cellular migration and axonal growth. Exp. Neurol. 1997, 143, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.E.; Baumann, M.D.; Tator, C.H.; Shoichet, M.S. Localized and sustained delivery of fibroblast growth factor-2 from a nanoparticle-hydrogel composite for treatment of spinal cord injury. Cells Tissues Organs 2013, 197, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Danen, E.H.; Yamada, K.M. Fibronectin, integrins, and growth control. J. Cell Physiol. 2001, 189, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- To, W.S.; Midwood, K.S. Plasma and cellular fibronectin: Distinct and independent functions during tissue repair. Fibrog. Tissue Repair 2011, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- King, V.R.; Alovskaya, A.; Wei, D.Y.; Brown, R.A.; Priestley, J.V. The use of injectable forms of fibrin and fibronectin to support axonal ingrowth after spinal cord injury. Biomaterials 2010, 31, 4447–4456. [Google Scholar] [CrossRef] [PubMed]
- Sakai, T.; Johnson, K.J.; Murozono, M.; Sakai, K.; Magnuson, M.A.; Wieloch, T.; Cronberg, T.; Isshiki, A.; Erickson, H.P.; Fassler, R. Plasma fibronectin supports neuronal survival and reduces brain injury following transient focal cerebral ischemia but is not essential for skin-wound healing and hemostasis. Nat. Med. 2001, 7, 324–330. [Google Scholar] [CrossRef] [PubMed]
- King, V.R.; Hewazy, D.; Alovskaya, A.; Phillips, J.B.; Brown, R.A.; Priestley, J.V. The neuroprotective effects of fibronectin mats and fibronectin peptides following spinal cord injury in the rat. Neuroscience 2010, 168, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Hejcl, A.; Ruzicka, J.; Proks, V.; Mackova, H.; Kubinova, S.; Tukmachev, D.; Cihlar, J.; Horak, D.; Jendelova, P. Dynamics of tissue ingrowth in SIKVAV-modified highly superporous PHEMA scaffolds with oriented pores after bridging a spinal cord transection. J. Mater. Sci. Mater. Med. 2018, 29, 89. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Nakagawa, M.; Asano, T.; Fukushima, C.; Tachi, K. Free vascularized lateral femoral cutaneous nerve graft with anterolateral thigh flap for reconstruction of facial nerve defects. J. Reconstr. Microsurg. 2006, 22, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Glaser, J.; Gonzalez, R.; Sadr, E.; Keirstead, H.S. Neutralization of the chemokine CXCL10 reduces apoptosis and increases axon sprouting after spinal cord injury. J. Neurosci. Res. 2006, 84, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Macaya, D.; Spector, M. Injectable hydrogel materials for spinal cord regeneration: A review. Biomed. Mater. 2012, 7, 012001. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.M.; Beattie, M.S.; Bresnahan, J.C. A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 1995, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hejčl, A.; Růžička, J.; Kekulová, K.; Svobodová, B.; Proks, V.; Macková, H.; Jiránková, K.; Kárová, K.; Machová Urdziková, L.; Kubinová, Š.; et al. Modified Methacrylate Hydrogels Improve Tissue Repair after Spinal Cord Injury. Int. J. Mol. Sci. 2018, 19, 2481. https://doi.org/10.3390/ijms19092481
Hejčl A, Růžička J, Kekulová K, Svobodová B, Proks V, Macková H, Jiránková K, Kárová K, Machová Urdziková L, Kubinová Š, et al. Modified Methacrylate Hydrogels Improve Tissue Repair after Spinal Cord Injury. International Journal of Molecular Sciences. 2018; 19(9):2481. https://doi.org/10.3390/ijms19092481
Chicago/Turabian StyleHejčl, Aleš, Jiří Růžička, Kristýna Kekulová, Barbora Svobodová, Vladimír Proks, Hana Macková, Kateřina Jiránková, Kristýna Kárová, Lucia Machová Urdziková, Šárka Kubinová, and et al. 2018. "Modified Methacrylate Hydrogels Improve Tissue Repair after Spinal Cord Injury" International Journal of Molecular Sciences 19, no. 9: 2481. https://doi.org/10.3390/ijms19092481
APA StyleHejčl, A., Růžička, J., Kekulová, K., Svobodová, B., Proks, V., Macková, H., Jiránková, K., Kárová, K., Machová Urdziková, L., Kubinová, Š., Cihlář, J., Horák, D., & Jendelová, P. (2018). Modified Methacrylate Hydrogels Improve Tissue Repair after Spinal Cord Injury. International Journal of Molecular Sciences, 19(9), 2481. https://doi.org/10.3390/ijms19092481