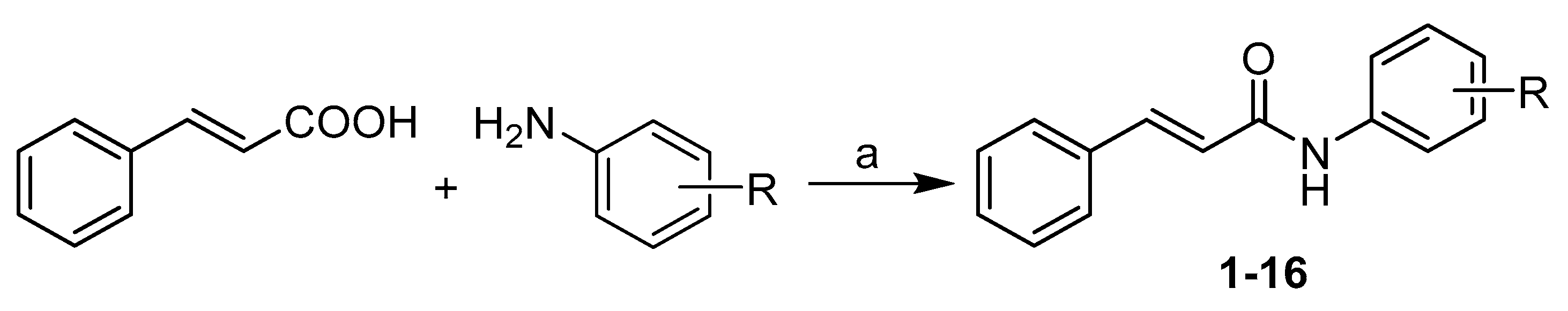
Synthesis and Spectrum of Biological Activities of Novel N-arylcinnamamides
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemistry and Physicochemical Properties
2.2. In Vitro Antibacterial Susceptibility Testing
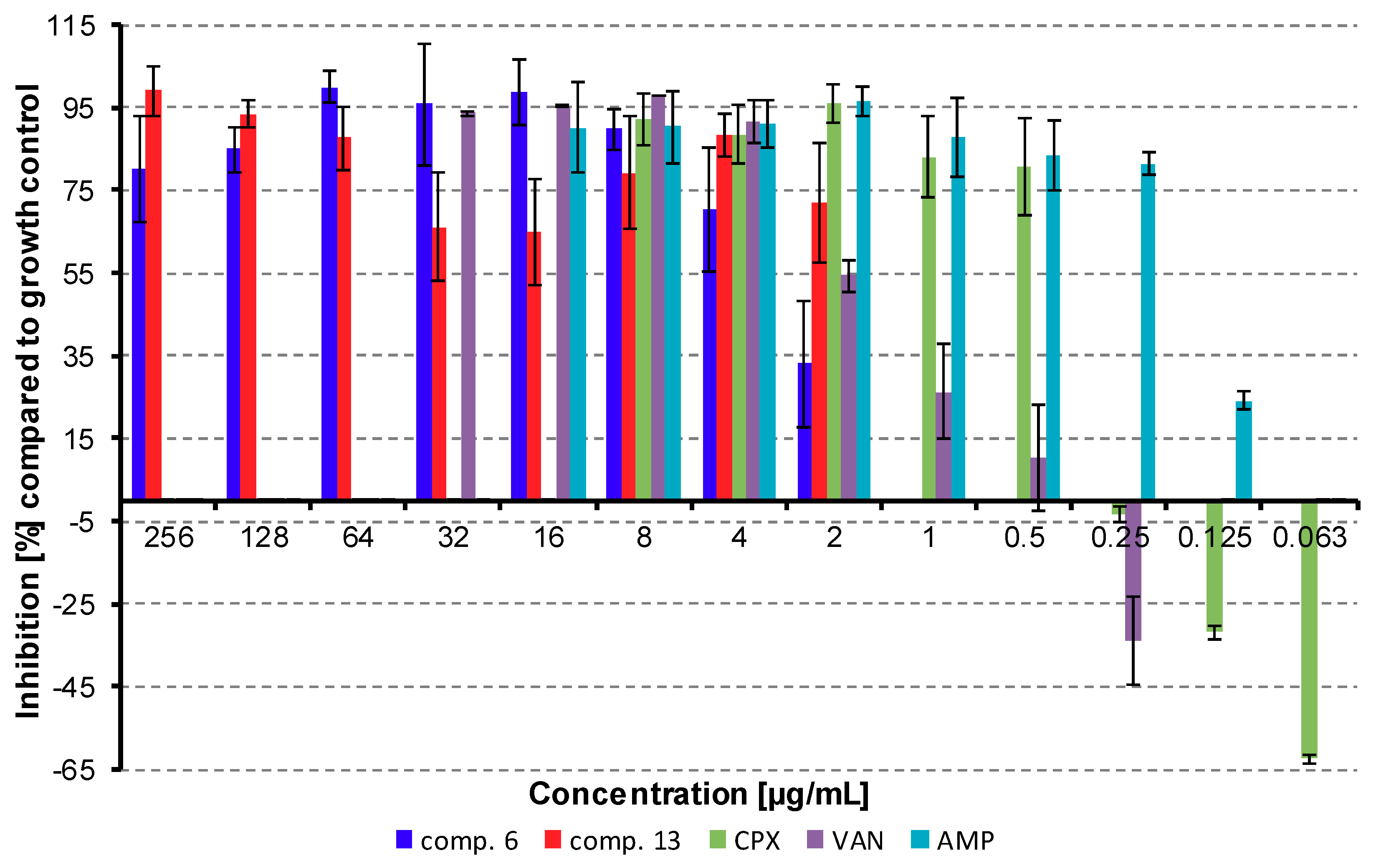
2.2.1. Synergy Effect with Clinically Used Drugs against MRSA
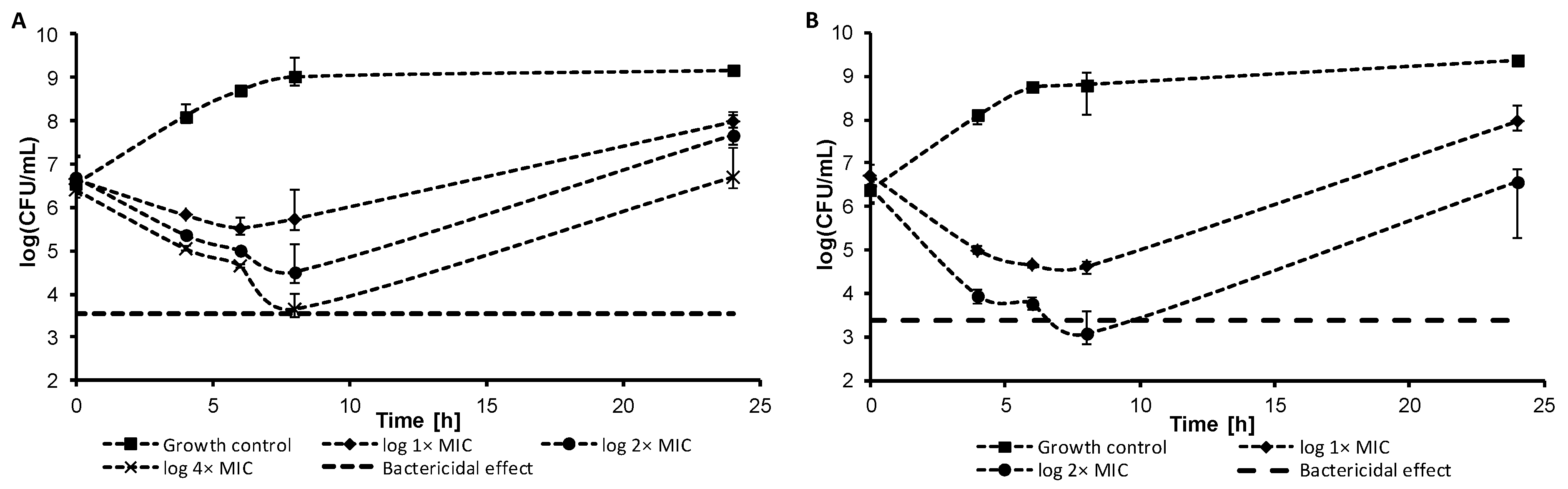
2.2.2. Dynamics of Antibacterial Activity
2.2.3. Inhibition of Bofilm Formation
2.3. In Vitro Antitubercular Activity
2.4. In Vitro Activity against Plant Pathogenic Fungi
Inhibition of B. sorokiniana Germination
2.5. In Vitro Antiproliferative Assay
2.6. Inhibition of Photosynthetic Electron Transport (PET) in Spinach Chloroplasts
2.7. In Vivo Toxicity against Plant Cells
3. Materials and Methods
3.1. Chemistry—General Information
Synthesis
3.2. Lipophilicity Determination by HPLC (Capacity Factor k/Calculated log k)
3.3. Biological Testing
3.3.1. In Vitro Antibacterial Evaluation
3.3.2. Synergy Effect with Clinically Used Drugs
3.3.3. Dynamics of Antibacterial Effect
3.3.4. Biofilm Inhibition Assay
3.3.5. In Vitro Antimycobacterial Evaluation
3.3.6. MTT Assay
3.3.7. In Vitro Antifungal Activity
3.3.8. Inhibition of Bipolaris sorokiniana Germination
3.3.9. In Vitro Antiproliferative Assay
3.3.10. Study of Inhibition of Photosynthetic Electron Transport (PET) in Spinach Chloroplasts
3.3.11. In Vivo Toxicity against Plant Cells
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lichtenthaler, H.K.; Schweiger, J. Cell wall bound ferulic acid, the major substance of the blue-green fluorescence emission of plants. J. Plant Physiol. 1998, 152, 272–282. [Google Scholar] [CrossRef]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Adisakwattana, S.; Chantarasinlapin, P.; Thammarat, H.; Yibchok-Anun, S. A series of cinnamic acid derivatives and their inhibitory activity on intestinal alpha-glucosidase. J. Enzyme Inhib. Med. Chem. 2009, 24, 1194–1200. [Google Scholar] [CrossRef] [PubMed]
- Berrin, O.; Murat, K.; Ilkay, O. Cytotoxicity, antiviral and antimicrobial activities of alkaloids, flavonoids, and phenolic acids. Pharm. Biol. 2011, 49, 396–402. [Google Scholar]
- Sharma, P. Cinnamic acid derivatives: A new chapter of various pharmacological activities. J. Chem. Pharm. Res. 2011, 3, 403–423. [Google Scholar]
- Sova, M. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef] [PubMed]
- Korosec, B.; Sova, M.; Turk, S.; Krasevec, N.; Novak, M.; Lah, L.; Stojan, J.; Podobnik, B.; Berne, S.; Zupanec, N.; et al. Antifungal activity of cinnamic acid derivatives involves inhibition of benzoate 4-hydroxylase (CYP53). J. Appl. Microbiol. 2014, 116, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Guzman, J.D. Natural cinnamic acids, synthetic derivatives and hybrids with antimicrobial activity. Molecules 2014, 19, 19292–19349. [Google Scholar] [CrossRef] [PubMed]
- Peperidou, A.; Kapoukranidou, D.; Kontogiorgis, C.; Hadjipavlou-Litina, D. Multitarget molecular hybrids of cinnamic acids. Molecules 2014, 19, 20197–20226. [Google Scholar] [CrossRef] [PubMed]
- Pontiki, E.; Hadjipavlou-Litina, D.; Litinas, K.; Geromichalos, G. Novel cinnamic acid derivatives as antioxidant and anticancer agents: Design, synthesis and modeling studies. Molecules 2014, 19, 9655–9674. [Google Scholar] [CrossRef] [PubMed]
- Hadjipavlou-Litina, D.; Pontiki, E. Aryl-acetic and cinnamic acids as lipoxygenase inhibitors with antioxidant, anti-inflammatory, and anticancer activity. Methods Mol. Biol. 2015, 1208, 361–377. [Google Scholar] [PubMed]
- Su, P.; Shi, Y.; Wang, J.; Shen, X.; Zhang, J. Anticancer agents derived from natural cinnamic acids. Anticancer Agents Med. Chem. 2015, 15, 980–987. [Google Scholar] [CrossRef] [PubMed]
- De Vita, D.; Simonetti, G.; Pandolfi, F.; Costi, R.; Di Santo, R.; D’Auria, F.D.; Scipione, L. Exploring the anti-biofilm activity of cinnamic acid derivatives in Candida albicans. Bioorg. Med. Chem. Lett. 2016, 26, 5931–5935. [Google Scholar] [CrossRef] [PubMed]
- Peperidou, A.; Pontiki, E.; Hadjipavlou-Litina, D.; Voulgari, E.; Avgoustakis, K. Multifunctional cinnamic acid derivatives. Molecules 2017, 22, 1247. [Google Scholar] [CrossRef] [PubMed]
- Lima, T.C.; Ferreira, A.R.; Silva, D.F.; Lima, E.O.; de Sousa, D.P. Antifungal activity of cinnamic acid and benzoic acid esters against Candida albicans strains. Nat. Prod. Res. 2018, 32, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Dolab, J.G.; Lima, B.; Spaczynska, E.; Kos, J.; Cano, N.H.; Feresin, G.; Tapia, A.; Garibotto, F.; Petenatti, E.; Olivella, M.; et al. Antimicrobial activity of Annona emarginata (Schltdl.) H. Rainer and most active isolated compound against clinically important bacteria. Molecules 2018, 23, 1187. [Google Scholar] [CrossRef] [PubMed]
- FRAC Code List 2018. Available online: http://www.frac.info/docs/default-source/publications/frac-code-list/frac_code_list_2018-final.pdf?sfvrsn=6144b9a_2 (accessed on 18 June 2018).
- WHO. Global Antimicrobial Resistance Surveillance System (GLASS) Report; HO Press: Geneva, Switzerland, 2017. [Google Scholar]
- Gonec, T.; Bobal, P.; Sujan, J.; Pesko, M.; Guo, J.; Kralova, K.; Pavlacka, L.; Vesely, L.; Kreckova, E.; Kos, J.; et al. Investigating the spectrum of biological activity of substituted quinoline-2-carboxamides and their isosteres. Molecules 2012, 17, 613–644. [Google Scholar] [CrossRef] [PubMed]
- Kos, J.; Nevin, E.; Soral, M.; Kushkevych, I.; Gonec, T.; Bobal, P.; Kollar, P.; Coffey, A.; O’Mahony, J.; Liptaj, T.; et al. Synthesis and antimycobacterial properties of ring-substituted 6-hydroxynaphthalene-2-carboxanilides. Bioorg. Med. Chem. 2015, 23, 2035–2043. [Google Scholar] [CrossRef] [PubMed]
- Tischer, W.; Strotmann, H. Relationship between inhibitor binding by chloroplasts and inhibition of photosynthetic electron-transport. Biochim. Biophys. Acta 1977, 460, 113–125. [Google Scholar] [CrossRef]
- Trebst, A.; Draber, W. Structure activity correlations of recent herbicides in photosynthetic reactions. In Advances in Pesticide Science; Greissbuehler, H., Ed.; Pergamon Press: Oxford, UK, 1979; pp. 223–234. [Google Scholar]
- Bowyer, J.R.; Camilleri, P.; Vermaas, W.F.J. Herbicides, Topics in Photosynthesis; Baker, N.R., Percival, M.P., Eds.; Elsevier: Amsterdam, The Netherlands, 1991; Volume 10, pp. 27–85. [Google Scholar]
- Izawa, S. Acceptors and donors for chloroplast electron transport. In Methods in Enzymology; Colowick, P., Kaplan, N.O., Eds.; Academic Press: New York, NY, USA; London, UK, 1980; Volume 69, Part C; pp. 413–434. [Google Scholar]
- Good, N.E. Inhibitors of the Hill reaction. Plant Physiol. 1961, 36, 788–803. [Google Scholar] [CrossRef] [PubMed]
- Otevrel, J.; Mandelova, Z.; Pesko, M.; Guo, J.; Kralova, K.; Sersen, F.; Vejsova, M.; Kalinowski, D.; Kovacevic, Z.; Coffey, A.; et al. Investigating the spectrum of biological activity of ring-substituted salicylanilides and carbamoylphenylcarbamates. Molecules 2010, 15, 8122–8142. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Kos, J.; Zadrazilova, I.; Pesko, M.; Keltosova, S.; Tengler, J.; Bobal, P.; Kollar, P.; Cizek, A.; Kralova, K.; et al. Antimycobacterial and herbicidal activity of ring-substituted 1-hydroxynaphthalene-2-carboxanilides. Bioorg. Med. Chem. 2013, 21, 6531–6541. [Google Scholar] [CrossRef] [PubMed]
- Kralova, K.; Perina, M.; Waisser, K.; Jampilek, J. Structure-activity relationships of N-benzylsalicylamides for inhibition of photosynthetic electron transport. Med. Chem. 2015, 11, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Kralova, K.; Pesko, M.; Jampilek, J. Antimycobacterial N-Alkoxyphenylhydroxynaphthalene-carboxamides Affecting Photosystem II. Bioorg. Med. Chem. Lett. 2017, 27, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Kos, J.; Pesko, M.; Dohanosova, J.; Oravec, M.; Liptaj, T.; Kralova, K.; Jampilek, J. Halogenated 1-Hydroxynaphthalene-2-carboxanilides Affecting Photosynthetic Electron Transport in Photosystem II. Molecules 2017, 22, 1709. [Google Scholar] [CrossRef] [PubMed]
- Shaner, D.L. Herbicide safety relative to common targets in plants and mammals. Pest. Manag. Sci. 2004, 60, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Delaney, J.; Clarke, E.; Hughes, D.; Rice, M. Modern agrochemical research: A missed opportunity for drug discovery? Drug Discov. Today 2006, 11, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O. Herbicide and pharmaceutical relationships. Weed Sci. 2010, 58, 334–339. [Google Scholar] [CrossRef]
- Myung, K.; Klittich, C.J. Can agricultural fungicides accelerate the discovery of human antifungal drugs? Drug Discov. Today 2015, 20, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Jampilek, J. Potential of agricultural fungicides for antifungal drug discovery. Expert Opin. Drug Dis. 2016, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pliska, V. Methods and Principles in Medicinal Chemistry. In Lipophilicity in Drug Action and Toxicology, 1st ed.; Pliska, V., Testa, B., van der Waterbeemd, H., Eds.; Wiley-VCH: Weinheim, Germany, 1996; Volume 4. [Google Scholar]
- Zadrazilova, I.; Pospisilova, S.; Pauk, K.; Imramovsky, A.; Vinsova, J.; Cizek, A.; Jampilek, J. In vitro bactericidal activity of 4- and 5-chloro-2-hydroxy-N-[1-oxo-1-(phenylamino)alkan-2-yl]benzamides against MRSA. Biomed Res. Int. 2015, 2015, 349534. [Google Scholar] [CrossRef] [PubMed]
- Zadrazilova, I.; Pospisilova, S.; Masarikova, M.; Imramovsky, A.; Monreal-Ferriz, J.; Vinsova, J.; Cizek, A.; Jampilek, J. Salicylanilide carbamates: Promising antibacterial agents with high in vitro activity against methicillin-resistant Staphylococcus aureus. Eur. J. Pharm. Sci. 2015, 77, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Oravcova, V.; Zurek, L.; Townsend, A.; Clark, A.B.; Ellis, J.C.; Cizek, A. American crows as carriers of vancomycin-resistant enterococci with vanA gene. Environ. Microbiol. 2014, 16, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Pospisilova, S.; Michnova, H.; Kauerova, T.; Pauk, K.; Kollar, P.; Vinsova, J.; Imramovsky, A.; Cizek, A.; Jampilek, J. In vitro activity of salicylamide derivatives against vancomycin-resistant enterococci. Bioorg. Med. Chem. Lett. 2018, 28, 2184–2188. [Google Scholar] [CrossRef] [PubMed]
- Schwalbe, R.; Steele-Moore, L.; Goodwin, A.C. Antimicrobial Susceptibility Testing Protocols; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Bonapace, C.R.; Bosso, J.A.; Friedrich, L.V.; White, R.L. Comparison of methods of interpretation of checkerboard synergy testing. Diagn. Microbiol. Infect. Dis. 2002, 44, 363–366. [Google Scholar] [CrossRef]
- Helander, I.M.; Alakomi, H.L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; Gorris, L.G.M.; von Wright, A. Characterization of the action of selected essential oil components on gram negative bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Ultee, A.; Bennik, M.H.J.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Gill, A.O.; Holley, R.A. Inhibition of membrane bound ATPases of Escherichia coli and Listeria monocytogenes by plant oil aromatics. Int. J. Food Microbiol. 2006, 3, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Langeveld, W.T.; Veldhuizen, E.J.; Burt, S.A. Synergy between essential oil components and antibiotics: A review. Crit. Rev. Microbiol. 2014, 40, 76–94. [Google Scholar] [CrossRef] [PubMed]
- Hemaiswarya, S.; Doble, M. Synergistic interaction of phenylpropanoids with antibiotics against bacteria. J. Med. Microbiol. 2010, 59, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.G.; Lee, J.H.; Kim, S.I.; Baek, K.H.; Lee, J. Cinnamon bark oil and its components inhibit biofilm formation and toxin production. Int. J. Food Microbiol. 2015, 195, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Defoirdt, T.; Miyamoto, C.; Bossier, P.; Van Calenbergh, S.; Nelis, H.; Coenye, T. Cinnamaldehyde and cinnamaldehyde derivatives reduce virulence in Vibrio spp. by decreasing the DNA-binding activity of the quorum sensing response regulator LuxR. BMC Microbiol. 2008, 8, 149. [Google Scholar] [CrossRef] [PubMed]
- Zodrow, K.R.; Schiffman, J.D.; Elimelech, M. Biodegradable polymer (PLGA) coatings featuring cinnamaldehyde and carvacrol mitigate biofilm formation. Langmuir 2012, 28, 13993–13999. [Google Scholar] [CrossRef] [PubMed]
- Jia, P.; Xue, Y.J.; Duan, X.J.; Shao, S.H. Effect of cinnamaldehyde on biofilm formation and sarA expression by methicillin-resistant Staphylococcus aureus. Lett. Appl. Microbiol. 2011, 53, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Coenye, T. Quorum sensing inhibitors as anti-biofilm agents. Curr. Pharm. Des. 2015, 21, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Afre, S.; Gilbert, E.S. Subinhibitory concentrations of cinnamaldehyde interfere with quorum sensing. Lett. Appl. Microbiol. 2006, 43, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Nuryastuti, T.; van der Mei, H.C.; Busscher, H.J.; Iravati, S.; Aman, A.T.; Krom, B.P. Effect of cinnamon oil on icaA expression and biofilm formation by Staphylococcus epidermidis. Appl. Environ. Microbiol. 2009, 75, 6850–6855. [Google Scholar] [CrossRef] [PubMed]
- Budzynska, A.; Wieckowska-Szakiel, M.; Sadowska, B.; Kalemba, D.; Rozalska, B. Antibiofilm activity of selected plant essential oils and their major components. Pol. J. Microbiol. 2011, 60, 35–41. [Google Scholar] [PubMed]
- Kaplan, J.B. Antibiotic-induced biofilm formation. Int. J. Artif. Organs 2011, 34, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Mirani, Z.A.; Jamil, N. Effect of sub-lethal doses of vancomycin and oxacillin on biofilm formation by vancomycin intermediate resistant Staphylococcus aureus. J. Basic Microbiol. 2011, 51, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Lu, L.; Wang, B.; Pu, S.; Zhang, X.; Zhu, G.; Shi, W.; Zhang, L.; Wang, H.; Wang, S.; et al. Genetic basis of virulence attenuation revealed by comparative genomic analysis of Mycobacterium tuberculosis strain H37Ra versus H37Rv. PLoS ONE 2008, 3, e2375. [Google Scholar] [CrossRef] [PubMed]
- Bueno, J. Antitubercular in vitro drug discovery: Tools for begin the search. In Understanding Tuberculosis-New Approaches to Fighting against Drug Resistance; Cardona, P.J., Ed.; InTech: Rijeka, Croatia, 2012; pp. 147–168. [Google Scholar]
- Kos, J.; Zadrazilova, I.; Nevin, E.; Soral, M.; Gonec, T.; Kollar, P.; Oravec, M.; Coffey, A.; O’Mahony, J.; Liptaj, T.; et al. Ring-substituted 8-Hydroxyquinoline-2-carboxanilides as potential antimycobacterial agents. Bioorg. Med. Chem. 2015, 23, 4188–4196. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Zadrazilova, I.; Nevin, E.; Kauerova, T.; Pesko, M.; Kos, J.; Oravec, M.; Kollar, P.; Coffey, A.; O’Mahony, J.; et al. Synthesis and biological evaluation of N-alkoxyphenyl-3-hydroxynaphthalene-2-carbox-anilides. Molecules 2015, 20, 9767–9787. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Pospisilova, S.; Kauerova, T.; Kos, J.; Dohanosova, J.; Oravec, M.; Kollar, P.; Coffey, A.; Liptaj, T.; Cizek, A.; et al. N-Alkoxyphenylhydroxynaphthalenecarboxamides and their antimycobacterial activity. Molecules 2016, 21, 1068. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Nahid, P.; Cole, S.T. Advances in the development of new tuberculosis drugs and treatment regimens. Nat. Rev. Drug Discov. 2013, 12, 388–404. [Google Scholar] [CrossRef] [PubMed]
- Upadhayaya, R.S.; Vandavasi, J.K.; Kardile, R.A.; Lahore, S.V.; Dixit, S.S.; Deokar, H.S.; Shinde, P.D.; Sarmah, M.P.; Chattopadhyaya, J. Novel quinoline and naphthalene derivatives as potent antimycobacterial agents. Eur. J. Med. Chem. 2010, 45, 1854–1867. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, L.; Zhou, Z.; Geng, Y.; Chen, Y.; Sun, T. Design, synthesis, configuration research, and in vitro antituberculosis activities of two chiral naphthylamine substituted analogs of bedaquiline. J. Heterocycl. Chem. 2017, 54, 1024–1030. [Google Scholar] [CrossRef]
- Tong, A.S.T.; Choi, P.J.; Blaser, A.; Sutherland, H.S.; Tsang, S.K.Y.; Guillemont, J.; Motte, M.; Cooper, C.B.; Andries, K.; van den Broeck, W.; et al. 6-Cyano analogues of bedaquiline as less lipophilic and potentially safer diarylquinolines for tuberculosis. ACS Med. Chem. Lett. 2017, 8, 1019–10242. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Huang, S.T.; Sun, F.M.; Chiang, Y.L.; Chiang, C.J.; Tsai, C.M.; Weng, C.J. Transformation of cinnamic acid from trans- to cis-form raises a notable bactericidal and synergistic activity against multiple-drug resistant Mycobacterium tuberculosis. Eur. J. Pharm. Sci. 2011, 43, 188–194. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Koumba, Y.G.; Constant, P.; Bedos-Belval, F.; Duran, H.; Saffon, N.; Daffe, M.; Baltas, M. Design, synthesis, and biological evaluation of new cinnamic derivatives as antituberculosis agents. J. Med. Chem. 2011, 54, 1449–1461. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Veau, D.; Bedos-Belval, F.; Chassaing, S.; Baltas, M. Cinnamic derivatives in tuberculosis. In Understanding Tuberculosis-New Approaches to Fighting against Drug Resistance; Cardona, P.J., Ed.; InTech: Rijeka, Croatia, 2012; pp. 337–362. [Google Scholar]
- Adeniji, S.E.; Uba, S.; Uzairu, A. Quantitative structure–activity relationship and molecular docking of 4-alkoxy-cinnamic analogues as anti-mycobacterium tuberculosis. J. King Saud Uni. Sci. 2018. [Google Scholar] [CrossRef]
- Degola, F.; Morcia, C.; Bisceglie, F.; Mussi, F.; Tumino, G.; Ghizzoni, R.; Pelosi, G.; Terzi, V.; Buschini, A.; Restivo, F.M.; et al. In vitro evaluation of the activity of thiosemicarbazone derivatives against mycotoxigenic fungi affecting cereals. Int. J. Food Microbiol. 2015, 200, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Chen, D.; Li, B.; Zhang, B.; Miao, F.; Zhou, L. Bioactivity and structure-activity relationship of cinnamic acid esters and their derivatives as potential antifungal agents for plant protection. PLoS ONE 2017, 12, e0176189. [Google Scholar] [CrossRef] [PubMed]
- Krishnendu, A.; Dutta, A.K.; Pradhan, P. Bipolaris sorokiniana’(Sacc.) Shoem.: The most destructive wheat fungal pathogen in the warmer areas. Aust. J. Crop Sci. 2011, 5, 1064–1071. [Google Scholar]
- Saari, E.E. Leaf blight disease and associated soil borne fungal pathogens of wheat in South and Southeast Asia. In Helminthosporium Blights of Wheat: Spot Blotch and Tan Spot; Duveiller, E., Dubin, H.J., Reeves, J., McNab, A., Eds.; CIMMYT: Texcoco de Mora, Mexico, 1998; pp. 37–51. [Google Scholar]
- Sundheim, L.; Brodal, G.; Hofgaard, I.S.; Rafoss, T. Temporal variation of mycotoxin producing fungi in norwegian cereals. Microorganisms 2013, 1, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Placinta, C.M.; D’Mello, J.P.F.; MacDonald, A.M.C. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed Sci. Technol. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- Tortora, G.J.; Funke, B.R.; Case, C.L. Microbiology: An introduction, 10th ed.; Benjamin Cummings: San Francisco, CA, USA, 2010. [Google Scholar]
- ROCHE. Cell proliferation reagent WST-1. Roche Diagnostics GmbH, Mannheim, Germany. 2011. Available online: https://www.sigmaaldrich.com/content/dam/sigma-aldrich/docs/Roche/Bulletin/1/cellprorobul.pdf (accessed on 26 June 2018).
- Kauerova, T.; Kos, J.; Gonec, T.; Jampilek, J.; Kollar, P. Antiproliferative and pro-apoptotic effect of novel nitro-substituted hydroxynaphthanilides on human cancer cell lines. Int. J. Mol. Sci. 2016, 17, 1219. [Google Scholar] [CrossRef] [PubMed]
- Suffness, M.; Douros, J. Current status of the NCI plant and animal product program. J. Nat. Prod. 1982, 45, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Jampilek, J.; Kralova, K.; Pesko, M.; Kos, J. Ring-substituted 8-hydroxyquinoline-2-carboxanilides as photosystem II inhibitors. Bioorg. Med. Chem. Lett. 2016, 26, 3862–3865. [Google Scholar] [CrossRef] [PubMed]
- Fajkusova, D.; Pesko, M.; Keltosova, S.; Guo, J.; Oktabec, Z.; Vejsova, M.; Kollar, P.; Coffey, A.; Csollei, J.; Kralova, K.; et al. Anti-infective and herbicidal activity of N-substituted 2-aminobenzothiazoles. Bioorg. Med. Chem. 2012, 20, 7059–7068. [Google Scholar] [CrossRef] [PubMed]
- Gonec, T.; Kos, J.; Zadrazilova, I.; Pesko, M.; Govender, R.; Keltosova, S.; Kollar, B.; Imramovsky, A.; O’Mahony, J.; Coffey, A.; et al. Antibacterial and herbicidal activity of ring-substituted 2-hydroxynaphthalene-1-carboxanilides. Molecules 2013, 18, 9397–9419. [Google Scholar] [CrossRef] [PubMed]
- Kralova, K.; Sersen, F.; Cizmarik, J. Inhibitory effect of piperidinoethylesters of alkoxyphenylcarbamic acids on photosynthesis. Gen. Physiol. Biophys. 1992, 11, 261–267. [Google Scholar]
- Kralova, K.; Bujdakova, H.; Kuchta, T.; Loos, D. Correlation between biological activity and the structure of 6-amino-2-R-thiobenzothiazoles. Anti-yeast activity and inhibition of photochemical activity of chloroplasts. Pharmazie 1994, 49, 460–461. [Google Scholar] [PubMed]
- Kralova, K.; Kallova, J.; Loos, D.; Devinsky, F. Correlation between biological activity and the structure of N,N’-bis(alkyldimethyl)-1,6-hexanediammonium dibromides. Antibacterial activity and inhibition of photochemical activity of chloroplasts. Pharmazie 1994, 49, 857–858. [Google Scholar] [PubMed]
- Kralova, K.; Bujdakova, H.; Cizmarik, J. Antifungal and antialgal activity of piperidinopropyl esters of alkoxy substituted phenylcarbamic acids. Pharmazie 1995, 50, 440–441. [Google Scholar] [PubMed]
- Szabo, E. Isolation and characterization of EBR specific induced chitinases from tobacco (Nicotiana tabacum). Acta Biol. Szeged. 2008, 52, 251–252. [Google Scholar]
- Zhang, M.; Lu, X.; Zhang, H.J.; Li, N.; Xiao, Y.; Zhu, H.L.; Ye, Y.H. Synthesis, structure, and biological assay of cinnamic amides as potential EGFR kinase inhibitors. Med. Chem. Res. 2013, 22, 986–994. [Google Scholar] [CrossRef]
- Lee, C.C.; Lo, Y.; Ho, L.J.; Lai, J.H.; Lien, S.B.; Lin, L.C.; Chen, C.L.; Chen, T.C.; Liu, F.C.; Huang, H.S. A new application of parallel synthesis strategy for discovery of amide-linked small molecules as potent chondroprotective agents in TNF-α-stimulated chondrocytes. PLoS ONE 2016, 11, e0149317. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; The 8th Informational Supplement Document; CLSI: Wayne, PA, USA, 2012; M100-S22. [Google Scholar]
- Abate, G.; Mshana, R.N.; Miorner, H. Evaluation of a colorimetric assay based on 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) for rapid detection of rifampicin resistance in Mycobacterium tuberculosis. Int. J. Tuberc. Lung Dis. 1998, 2, 1011–1016. [Google Scholar] [PubMed]
- De Lucca, A.J.; Walsh, T.J.; Daigle, D.J. N-acetylcysteine inhibits germination of conidia and growth of Aspergillus spp. and Fusarium spp. Antimicrob. Agents Chemother. 1996, 40, 1274–1276. [Google Scholar] [PubMed]
- Masarovicova, E.; Kralova, K. Approaches to Measuring Plant Photosynthesis Activity. In Handbook of Photosynthesis, 2nd ed.; Pessarakli, M., Ed.; Taylor & Francis Group: Boca Raton, FL, USA, 2005; pp. 617–656. [Google Scholar]
- Kralova, K.; Sersen, F.; Sidoova, E. Photosynthesis inhibition produced by 2-alkylthio-6-R-benzothiazoles. Chem. Pap. 1992, 46, 348–350. [Google Scholar]
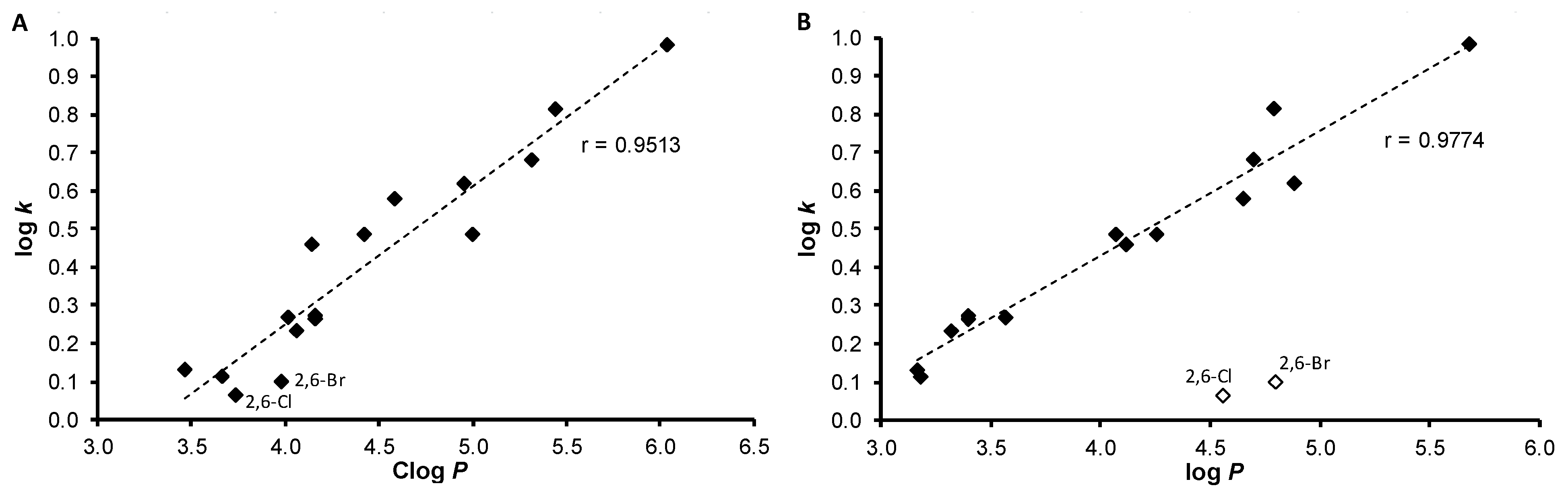
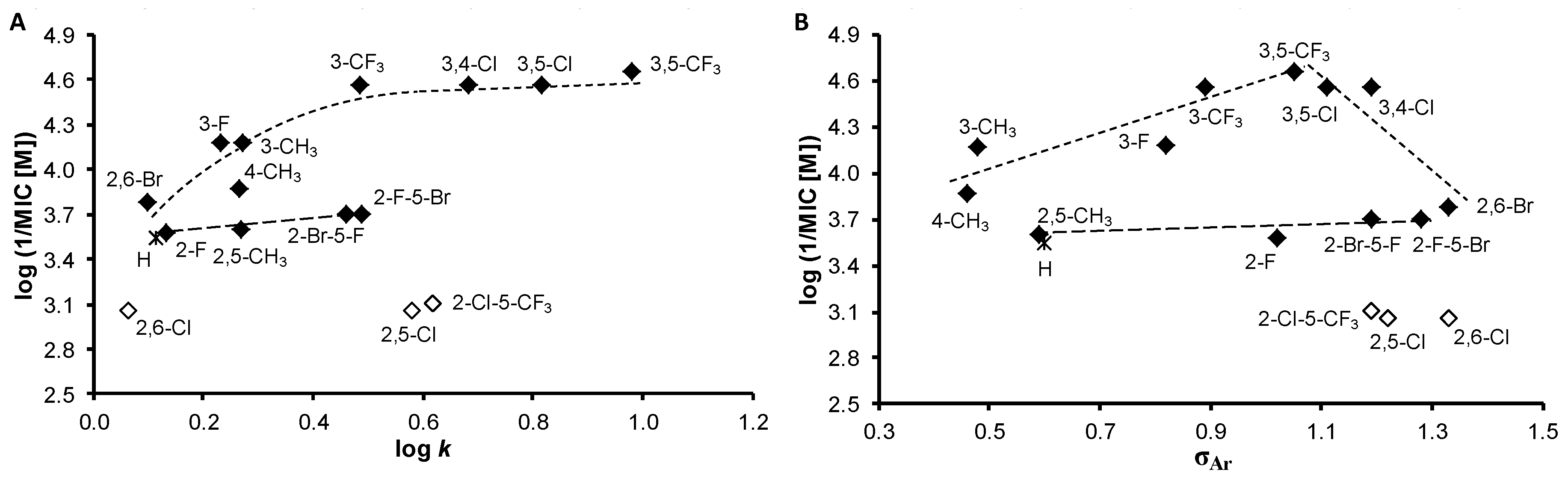
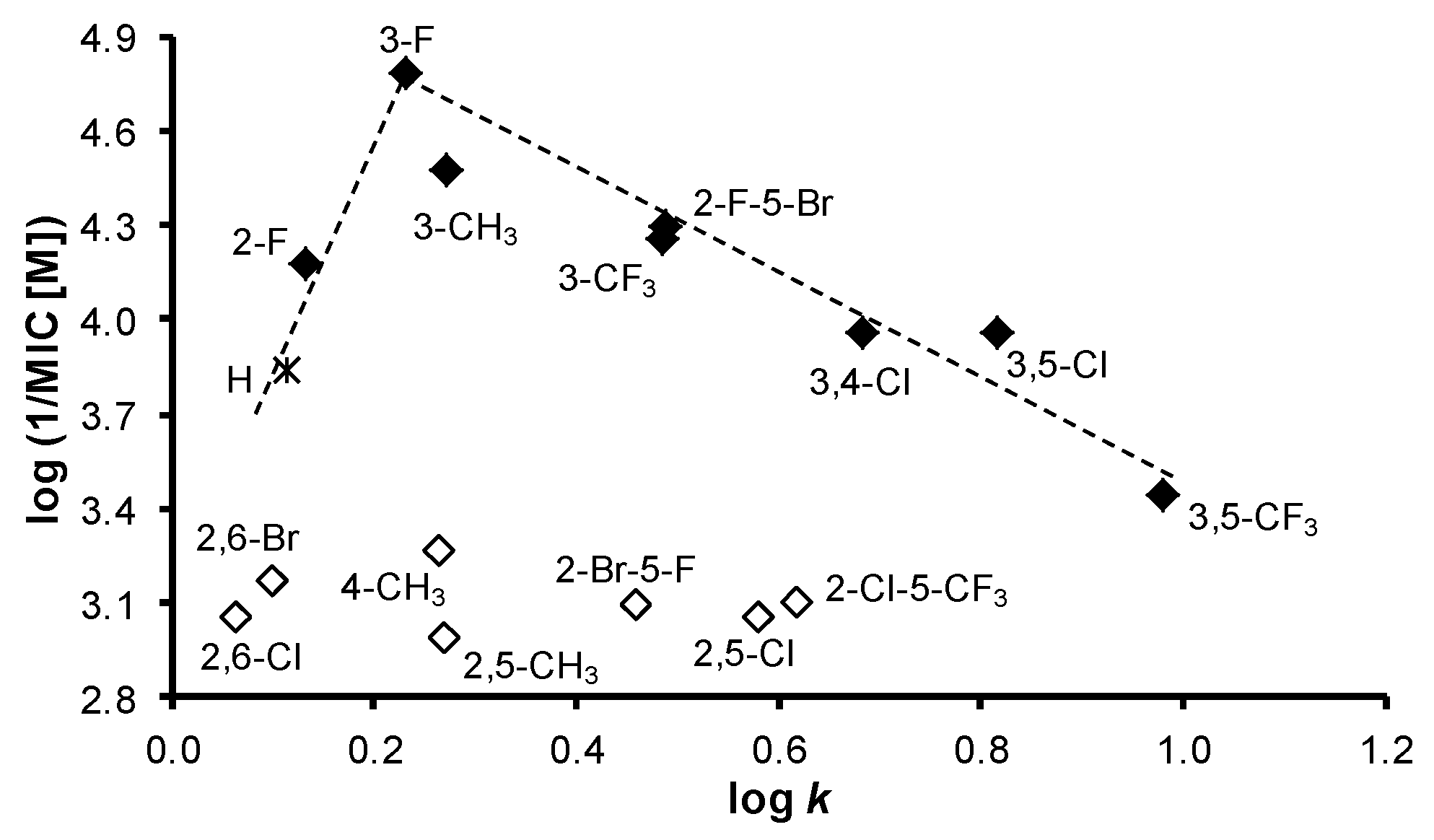


| Comp. | R | log k | Clog P a | log P b | σAr b |
|---|---|---|---|---|---|
| 1 | H | 0.1146 | 3.6640 | 3.18 | 0.60 |
| 2 | 3-CH3 | 0.2729 | 4.1630 | 3.40 | 0.48 |
| 3 | 4-CH3 | 0.2640 | 4.1630 | 3.40 | 0.46 |
| 4 | 2-F | 0.1330 | 3.4646 | 3.17 | 1.02 |
| 5 | 3-F | 0.2327 | 4.0646 | 3.32 | 0.82 |
| 6 | 3-CF3 | 0.4859 | 4.9978 | 4.26 | 0.89 |
| 7 | 2,5-CH3 | 0.2691 | 4.0120 | 3.57 | 0.59 |
| 8 | 2,5-Cl | 0.5799 | 4.5878 | 4.65 | 1.22 |
| 9 | 2,6-Cl | 0.0632 | 3.7378 | 4.56 | 1.33 |
| 10 | 3,4-Cl | 0.6821 | 5.3178 | 4.70 | 1.19 |
| 11 | 3,5-Cl | 0.8155 | 5.4378 | 4.79 | 1.11 |
| 12 | 2,6-Br | 0.0992 | 3.9778 | 4.80 | 1.33 |
| 13 | 3,5-CF3 | 0.9814 | 6.0386 | 5.68 | 1.05 |
| 14 | 2-F-5-Br | 0.4875 | 4.4178 | 4.07 | 1.28 |
| 15 | 2-Br-5-F | 0.4588 | 4.1378 | 4.12 | 1.19 |
| 16 | 2-Cl-5-CF3 | 0.6178 | 4.9509 | 4.88 | 1.19 |
| Comp. | R | MIC (μM (µg/mL)) | Tox IC50 (μM) | PET IC50 (μM) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SA | MRSA 63718 | MRSA SA 630 | MRSA SA 3202 | Mtb | FA | BS | ||||
| 1 | H | >1146 (>256) | >1146 (>256) | >1146 (>256) | >1146 (>256) | 286 (64) | 1146 (256) | 143 (32) | – | 250 |
| 2 | 3-CH3 | >1078 (>256) | >1078 (>256) | >1078 (>256) | >1078 (>256) | 67.43 (16) | 270 (64) | 33.71 (8) | >30 | 343 |
| 3 | 4-CH3 | >1078 (>256) | >1078 (>256) | >1078 (>256) | >1078 (>256) | 134 (32) | 1078 (256) | 539 (128) | – | 320 |
| 4 | 2-F | >1061 (>256) | >1061 (>256) | >1061 (>256) | >1061 (>256) | 265 (64) | 1061 (256) | 66.32 (16) | – | 223 |
| 5 | 3-F | >1061 (>256) | >1061 (>256) | >1061 (>256) | >1061 (>256) | 66.31 (16) | 531 (128) | 16.58 (4) | >30 | 165 |
| 6 | 3-CF3 | 27.47 (8) | 27.47 (8) | 27.47 (8) | 27.47 (8) | 27.47 (8) | 54.93 (16) | 54.93 (16) | 22.72 ± 1.73 | 189 |
| 7 | 2,5-CH3 | >1018 (>256) | >1018 (>256) | >1018 (>256) | >1018 (>256) | 254 (64) | 1019 (256) | 1019 (256) | – | 338 |
| 8 | 2,5-Cl | >876 (>256) | >876 (>256) | >876 (>256) | >876 (>256) | 876 (256) | 876 (256) | 876 (256) | – | 67.1 |
| 9 | 2,6-Cl | >876 (>256) | >876 (>256) | >876 (>256) | >876 (>256) | 876 (256) | 876 (256) | 876 (256) | – | 1380 |
| 10 | 3,4-Cl | 438 (128) | 876 (256) | 438 (128) | 876 (256) | 27.38 (8) | 219 (64) | 110 (32) | 29.81 ± 0.31 | 54.9 |
| 11 | 3,5-Cl | 438 (128) | 876 (256) | 109 (64) | 438 (128) | 27.38 (8) | 219 (64) | 110 (32) | 29.44 ± 1.73 | 5.1 |
| 12 | 2,6-Br | >671 (256) | >671 (256) | >671 (256) | >671 (256) | 167 (64) | 671 (256) | 671 (256) | – | 732 |
| 13 | 3,5-CF3 | 22.27 (8) | 22.27 (8) | 22.27 (8) | 22.27 (8) | 22.27 (8)) | 713 (256) | 356 (128) | 22.59 ± 1.88 | 111 |
| 14 | 2-F-5-Br | >799 (>256) | >799 (>256) | >799 (>256) | >799 (>256) | 199 (64) | 799 (256) | 49.98 (16) | – | 188 |
| 15 | 2-Br-5-F | >799 (>256) | >799 (>256) | >799 (>256) | >799 (>256) | 199 (64) | 799 (256) | 799 (256) | – | 205 |
| 16 | 2-Cl-5-CF3 | >785 (>256) | >785 (>256) | >785 (>256) | >785 (>256) | 785 (256) | 785 (256) | 785 (256) | – | 63.2 |
| AMP | – | 5.72 (2) | 45.81 (16) | 45.81 (16) | 45.81 (16) | – | – | – | – | – |
| INH | – | – | – | – | – | 36.55 (5) | – | – | – | – |
| BNM | – | – | – | – | – | – | 1.94 (0.5) | 17.22 (5) | – | – |
| CMP | – | – | – | – | – | – | – | – | 0.16 ± 0.07 | – |
| DCMU | – | – | – | – | – | – | – | – | – | 2.1 |
| Isolate | Combination of Compds. | Separate MIC (μg/mL) | FIC Index | Concentration (μg/mL) Causing Synergistic Effect | Concentration (μg/mL) Causing Additive Effect |
|---|---|---|---|---|---|
| MRSA 63718 | 6/TET | 8/128 | 1.004–2.250 | – | 2/64; 8/32 |
| 6/CPX | 16/16 | 0.75–1.125 | – | 8/4; 4/8 | |
| 6/VAN | 32/2 | 1.000–1.250 | – | – | |
| MRSA SA 3202 | 6/TET | 16/64 | 1.002–1.25 | – | – |
| 6/CPX | 8/8 | 1.000–1.250 | – | – | |
| 6/VAN | 8/1 | 0.750–1.256 | – | 4/0.25 | |
| 13/TET | 32/64 | 0.500–1.125 | 8/16 | 16/16; 4/32; 2/64 | |
| 13/CPX | 32/8 | 0.375–1.250 | 8/1 | 2/4 | |
| 13/VAN | 32/1 | 0.750–1.25 | – | 16/0.25 | |
| MRSA SA 630 | 6/CPX | 8/256 | 0.625–1.125 | – | 4/64; 1/128 |
| 6/VAN | 8/1 | 0.750–1.250 | – | 2/0.5 | |
| 13/CPX | 8/256 | 0.375–1.004 | 2/32; 1/64 | 4/8 | |
| 13/VAN | 4/1 | 0.562–1.250 | – | 0.25/0.5 |
| Comp. | Concentration (μg/mL) | Inhibition (%) Compared to Negative Control | Comp. | Concentration (μg/mL) | Inhibition (%) Compared to Negative Control |
|---|---|---|---|---|---|
| 1 | 256 | 59.6 | 9 | 256 | 82.5 |
| 128 | 74.2 | 128 | 76.3 | ||
| 2 | 256 | 64.7 | 10 | 256 | 84.3 |
| 128 | 59.1 | 128 | 51.3 | ||
| 3 | 256 | 88.7 | 11 | 256 | 84.8 |
| 128 | 89.1 | 128 | 82.6 | ||
| 4 | 256 | 60.8 | 12 | 256 | 91.3 |
| 128 | 58.7 | 128 | 82.1 | ||
| 5 | 256 | 93.6 | 13 | 256 | 76.3 |
| 128 | 95.4 | 128 | 75.5 | ||
| 6 | 256 | 18.4 | 14 | 256 | 81.8 |
| 128 | 26.7 | 128 | 77.6 | ||
| 7 | 256 | 73.5 | 15 | 256 | 100 |
| 128 | 35.1 | 128 | 60.9 | ||
| 8 | 256 | 92.7 | 16 | 256 | 86.2 |
| 128 | 88.7 | 128 | 61.2 | ||
| BNM | 10 | 100 | BNM | 10 | 100 |
| 5 | 100 | 5 | 100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pospisilova, S.; Kos, J.; Michnova, H.; Kapustikova, I.; Strharsky, T.; Oravec, M.; Moricz, A.M.; Bakonyi, J.; Kauerova, T.; Kollar, P.; et al. Synthesis and Spectrum of Biological Activities of Novel N-arylcinnamamides. Int. J. Mol. Sci. 2018, 19, 2318. https://doi.org/10.3390/ijms19082318
Pospisilova S, Kos J, Michnova H, Kapustikova I, Strharsky T, Oravec M, Moricz AM, Bakonyi J, Kauerova T, Kollar P, et al. Synthesis and Spectrum of Biological Activities of Novel N-arylcinnamamides. International Journal of Molecular Sciences. 2018; 19(8):2318. https://doi.org/10.3390/ijms19082318
Chicago/Turabian StylePospisilova, Sarka, Jiri Kos, Hana Michnova, Iva Kapustikova, Tomas Strharsky, Michal Oravec, Agnes M. Moricz, Jozsef Bakonyi, Tereza Kauerova, Peter Kollar, and et al. 2018. "Synthesis and Spectrum of Biological Activities of Novel N-arylcinnamamides" International Journal of Molecular Sciences 19, no. 8: 2318. https://doi.org/10.3390/ijms19082318
APA StylePospisilova, S., Kos, J., Michnova, H., Kapustikova, I., Strharsky, T., Oravec, M., Moricz, A. M., Bakonyi, J., Kauerova, T., Kollar, P., Cizek, A., & Jampilek, J. (2018). Synthesis and Spectrum of Biological Activities of Novel N-arylcinnamamides. International Journal of Molecular Sciences, 19(8), 2318. https://doi.org/10.3390/ijms19082318






