Vitamin D and Uterine Fibroids—Review of the Literature and Novel Concepts
Abstract
1. Introduction
2. Materials and Methods
3. Discussion
3.1. Uterine Fibroid Biology—Overview
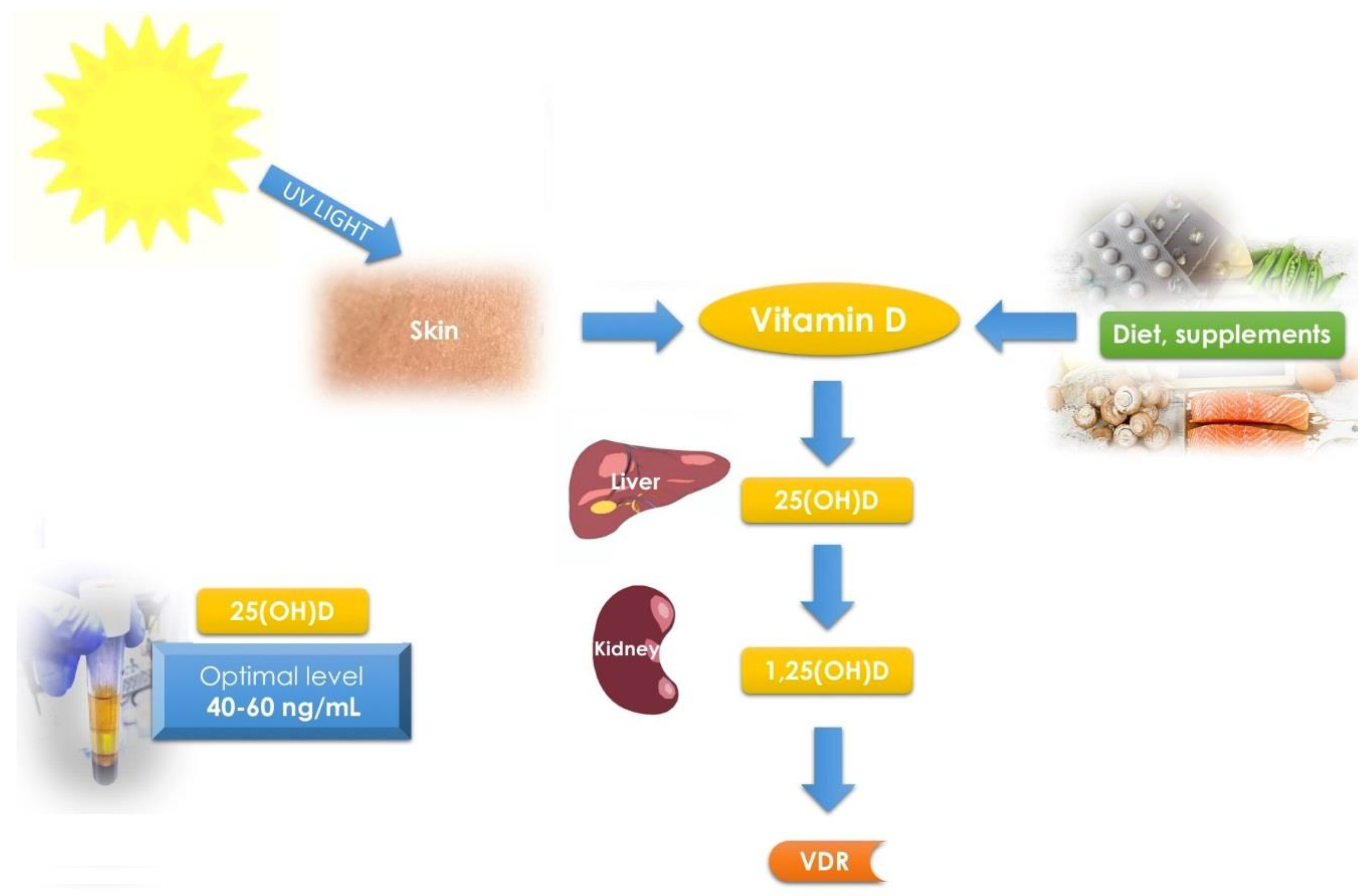
3.2. Vitamin D and Its Receptor—Overview
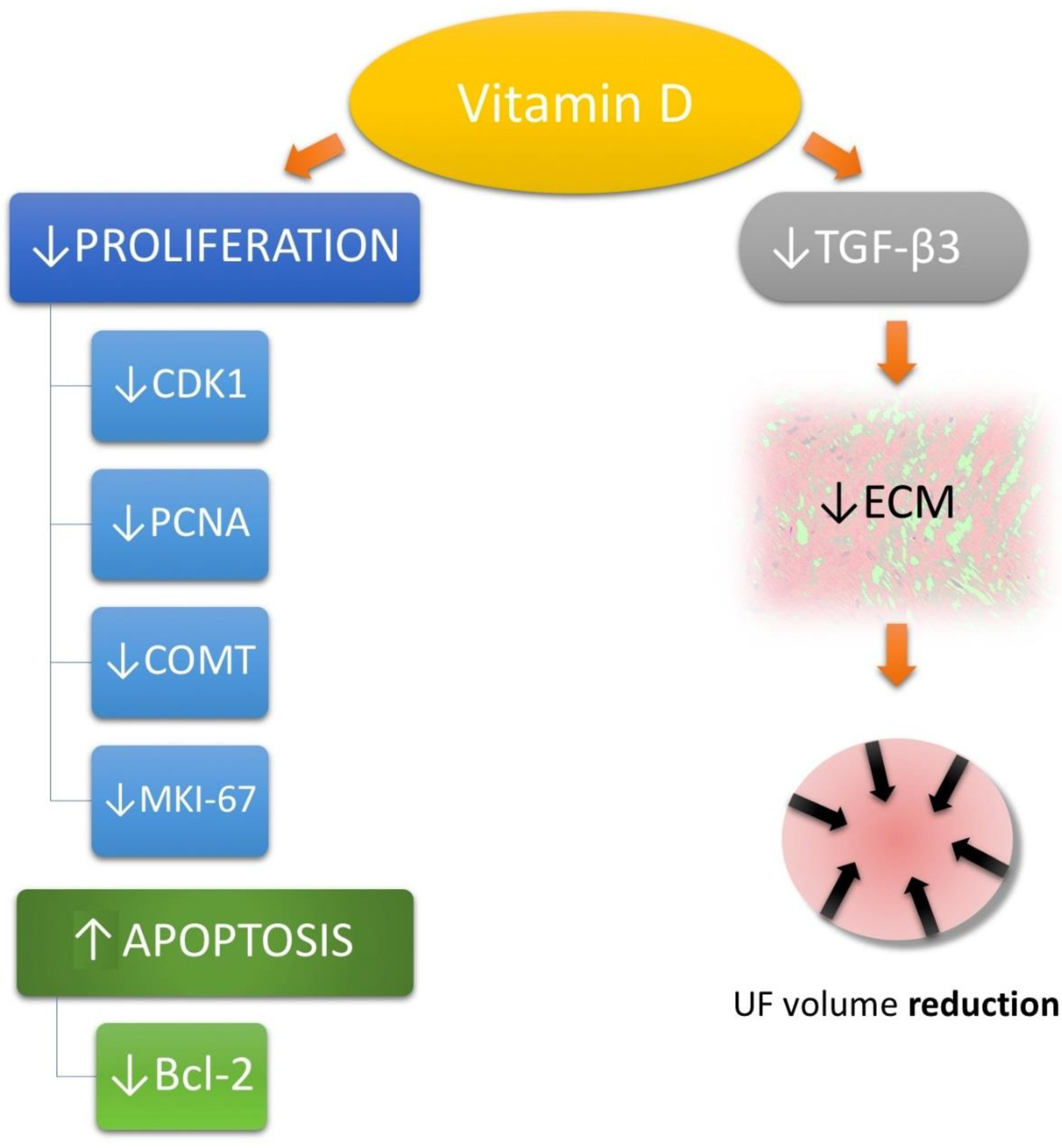
3.3. Vitamin D in Uterine Fibroid Biology
3.4. Vitamin D Receptor and Uterine Fibroids—Gene Polymorphisms
3.5. Vitamin D—Potential Uterine Fibroid Prophylaxis or Treatment Method
3.5.1. Vitamin D—Optimal Levels and Supplementation against Uterine Fibroids
3.5.2. The Use of Paricalcitol in Uterine Fibroid Management
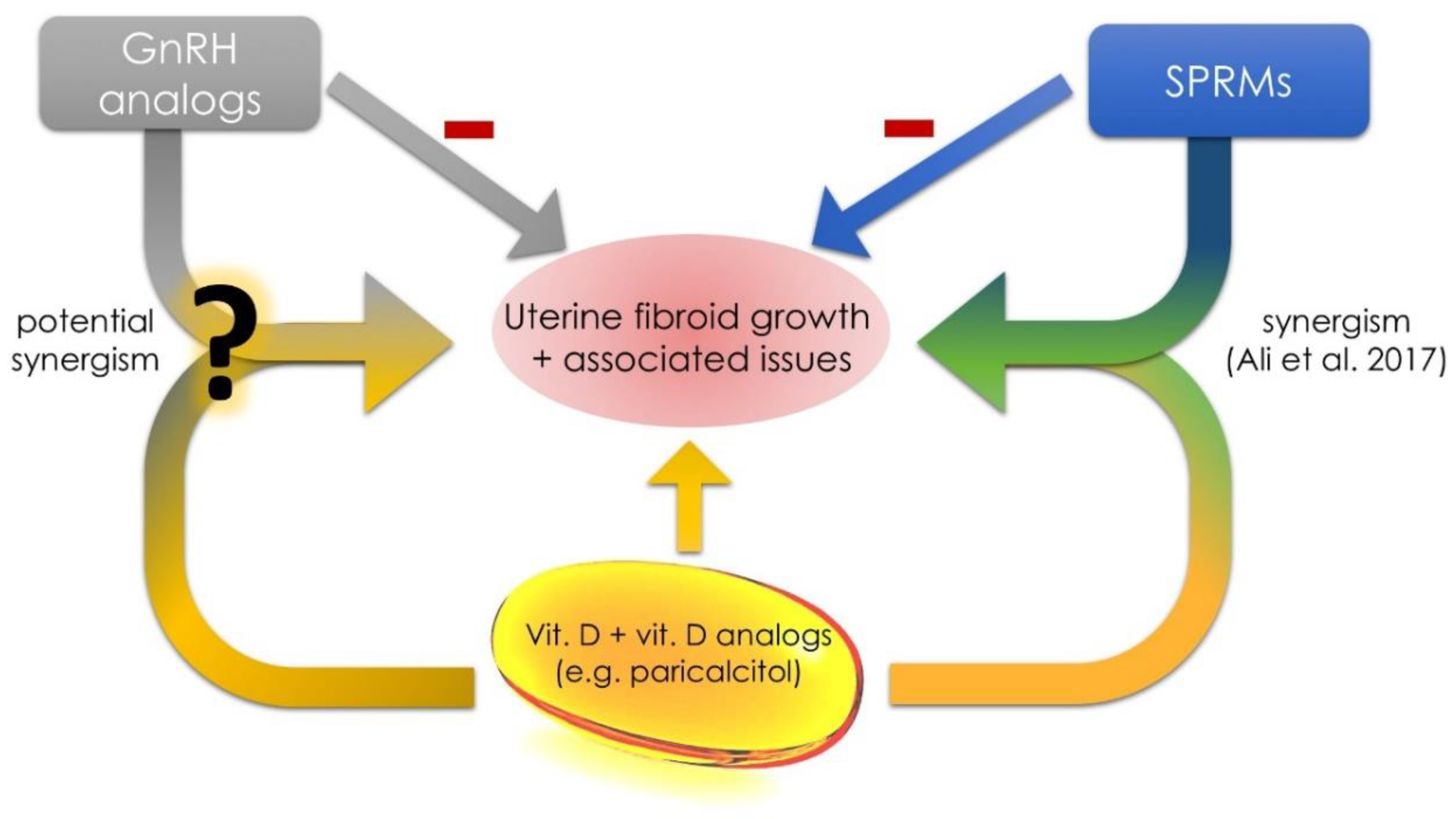
3.6. Future Concepts in the Area of Uterine Fibroids and Vitamin D
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 1,25(OH)D | 1,25-dihydroxyvitamin D |
| 25(OH)D | 25-hydroxyvitamin D |
| BMI | body mass index |
| CDK1 | cyclin-dependent kinase 1 |
| COC | combined oral contraception |
| COMT | catechol-O-methyltransferase |
| ECM | extracellular matrix |
| EMA | European Medicines Agency |
| FEN-1 | flap structure-specific endonuclease 1 |
| GnRH | Gonadotropin releasing hormone |
| MMP | matrix metalloproteinase |
| mTOR | mechanistic target of rapamycin |
| NHANES | National Health and Nutrition Examination Survey |
| PCNA | proliferating cell nuclear antigen |
| QoL | quality of life |
| SELF | Study of environment lifestyle and fibroids |
| SNP | single nucleotide polymorphism |
| SPRM | selective progesterone receptor modulator |
| TGF-β | transforming growth factor beta |
| TIMP | tissue inhibitor of metalloproteinase |
| UF | uterine fibroid |
| UPA | ulipristal acetate |
| VDBP | vitamin D binding protein |
| VDR | vitamin D receptor |
References
- Stewart, E.A. Uterine fibroids. Lancet 2001, 357, 293–298. [Google Scholar] [CrossRef]
- Stewart, E.A.; Laughlin-Tommaso, S.K.; Catherino, W.H.; Lalitkumar, S.; Gupta, D.; Vollenhoven, B. Uterine fibroids. Nat. Rev. Dis. Primers 2016, 2, 16043. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.A.; Cookson, C.L.; Gandolfo, R.A.; Schulze-Rath, R. Epidemiology of uterine fibroids: A systematic review. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 1501–1512. [Google Scholar] [CrossRef] [PubMed]
- Al-Hendy, A.; Myers, E.R.; Stewart, E. Uterine fibroids: Burden and unmet medical need. Semin. Reprod. Med. 2017, 35, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Dunson, D.B.; Hill, M.C.; Cousins, D.; Schectman, J.M. High cumulative incidence of uterine leiomyoma in black and white women: Ultrasound evidence. Am. J. Obstet. Gynecol. 2003, 188, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Wlodarczyk, M.; Slabuszewska-Jozwiak, A.; Nowicka, G.; Jakiel, G. Influence of vitamin D and transforming growth factor beta3 serum concentrations, obesity, and family history on the risk for uterine fibroids. Fertil. Steril. 2016, 106, 1787–1792. [Google Scholar] [CrossRef] [PubMed]
- Sabry, M.; Halder, S.K.; Allah, A.S.; Roshdy, E.; Rajaratnam, V.; Al-Hendy, A. Serum vitamin D3 level inversely correlates with uterine fibroid volume in different ethnic groups: A cross-sectional observational study. Int. J. Women’s Health 2013, 5, 93–100. [Google Scholar] [CrossRef]
- Paffoni, A.; Somigliana, E.; Vigano, P.; Benaglia, L.; Cardellicchio, L.; Pagliardini, L.; Papaleo, E.; Candiani, M.; Fedele, L. Vitamin D status in women with uterine leiomyomas. J. Clin. Endocrinol. Metab. 2013, 98, E1374–1378. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Hill, M.C.; Schectman, J.M.; Hollis, B.W. Vitamin D and the risk of uterine fibroids. Epidemiology 2013, 24, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A. Study of environment lifestyle and fibroids (self): Advancing the field of fibroid epidemiology. J. Women’s Health 2015, 24, 862–864. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Harmon, Q.E.; Upson, K.; Moore, K.R.; Barker-Cummings, C.; Baker, S.; Cooper, T.; Wegienka, G. A prospective, ultrasound-based study to evaluate risk factors for uterine fibroid incidence and growth: Methods and results of recruitment. J. Women’s Health 2015, 24, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.K.; Osteen, K.G.; Al-Hendy, A. 1,25-dihydroxyvitamin D3 reduces extracellular matrix-associated protein expression in human uterine fibroid cells. Biol. Reprod. 2013, 89, 150. [Google Scholar] [CrossRef] [PubMed]
- Brakta, S.; Diamond, J.S.; Al-Hendy, A.; Diamond, M.P.; Halder, S.K. Role of vitamin D in uterine fibroid biology. Fertil. Steril. 2015, 104, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.H. Etiology, symptomatology, and diagnosis of uterine myomas. Fertil. Steril. 2007, 87, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Kjerulff, K.H.; Langenberg, P.; Seidman, J.D.; Stolley, P.D.; Guzinski, G.M. Uterine leiomyomas. Racial differences in severity, symptoms and age at diagnosis. J. Reprod. Med. 1996, 41, 483–490. [Google Scholar] [PubMed]
- Buttram, V.C., Jr.; Reiter, R.C. Uterine leiomyomata: Etiology, symptomatology, and management. Fertil. Steril. 1981, 36, 433–445. [Google Scholar] [PubMed]
- Khan, A.T.; Shehmar, M.; Gupta, J.K. Uterine fibroids: Current perspectives. Int. J. Women’s Health 2014, 6, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Soave, I.; Marci, R. Uterine leiomyomata: The snowball effect. Curr. Med. Res. Opin. 2017, 33, 1909–1911. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.M.; Margolis, M.K.; Castelli-Haley, J.; Fuldeore, M.J.; Owens, C.D.; Coyne, K.S. Impact of uterine fibroid symptoms on health-related quality of life of us women: Evidence from a cross-sectional survey. Curr. Med. Res. Opin. 2017, 33, 1971–1978. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.M.; Yang, H.; Du, E.X.; Kelkar, S.S.; Winkel, C. The direct and indirect costs of uterine fibroid tumors: A systematic review of the literature between 2000 and 2013. Am. J. Obstet. Gynecol. 2015, 213, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, E.R.; Clark, A.D.; Banks, N.K.; Henne, M.B.; Stegmann, B.J.; Segars, J.H. The estimated annual cost of uterine leiomyomata in the United States. Am. J. Obstet. Gynecol. 2012, 206, 211.e1–211.e9. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.Y.; Wang, K.C. Minimally invasive approaches to the surgical management of fibroids. Semin. Reprod. Med. 2017, 35, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Donnez, J.; Dolmans, M.M. Uterine fibroid management: From the present to the future. Hum. Reprod. Update 2016, 22, 665–686. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, C.M.; Steiner, C.A. Hysterectomy rates in the United States 1990–1997. Obstet. Gynecol. 2002, 99, 229–234. [Google Scholar] [PubMed]
- Taylor, D.K.; Holthouser, K.; Segars, J.H.; Leppert, P.C. Recent scientific advances in leiomyoma (uterine fibroids) research facilitates better understanding and management. F1000Research 2015, 4, 183. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.I.; Gargiulo, A.R. The role of hysteroscopic and robot-assisted laparoscopic myomectomy in the setting of infertility. Clin. Obstet. Gynecol. 2016, 59, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Borah, B.J.; Yao, X.; Laughlin-Tommaso, S.K.; Heien, H.C.; Stewart, E.A. Comparative effectiveness of uterine leiomyoma procedures using a large insurance claims database. Obstet. Gynecol. 2017, 130, 1047–1056. [Google Scholar] [CrossRef] [PubMed]
- Faustino, F.; Martinho, M.; Reis, J.; Aguas, F. Update on medical treatment of uterine fibroids. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 216, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Sefton, E.C. The role of progesterone signaling in the pathogenesis of uterine leiomyoma. Mol. Cell. Endocrinol. 2012, 358, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E. Uterine fibroids. N. Engl. J. Med. 2013, 369, 1344–1355. [Google Scholar] [CrossRef] [PubMed]
- Donnez, J.; Donnez, O.; Courtoy, G.E.; Dolmans, M.M. The place of selective progesterone receptor modulators in Myoma therapy. Minerva Ginecol. 2016, 68, 313–320. [Google Scholar] [PubMed]
- Kalampokas, T.; Kamath, M.; Boutas, I.; Kalampokas, E. Ulipristal acetate for uterine fibroids: A systematic review and meta-analysis. Gynecol. Endocrinol. 2016, 32, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Agency, E.M. Women Taking Esmya for Uterine Fibroids to Have Regular Liver Tests While EMA Review Is Ongoing; European Medicines Agency: London, UK, 2018. [Google Scholar]
- Ciebiera, M.; Lukaszuk, K.; Meczekalski, B.; Ciebiera, M.; Wojtyla, C.; Slabuszewska-Jozwiak, A.; Jakiel, G. Alternative oral agents in prophylaxis and therapy of uterine fibroids-an up-to-date review. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Fritton, K.; Borahay, M.A. New and emerging therapies for uterine fibroids. Semin. Reprod. Med. 2017, 35, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D: A millenium perspective. J. Cell. Biochem. 2003, 88, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Blauer, M.; Rovio, P.H.; Ylikomi, T.; Heinonen, P.K. Vitamin D inhibits myometrial and leiomyoma cell proliferation in vitro. Fertil. Steril. 2009, 91, 1919–1925. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Holick, M.F.; Grant, W.B.; Konstantynowicz, J.; Mascarenhas, M.R.; Haq, A.; Povoroznyuk, V.; Balatska, N.; Barbosa, A.P.; Karonova, T.; et al. Vitamin D supplementation guidelines. J. Steroid Biochem. Mol. Biol. 2018, 175, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Protic, O.; Toti, P.; Islam, M.S.; Occhini, R.; Giannubilo, S.R.; Catherino, W.H.; Cinti, S.; Petraglia, F.; Ciavattini, A.; Castellucci, M.; et al. Possible involvement of inflammatory/reparative processes in the development of uterine fibroids. Cell Tissue Res. 2016, 364, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.; Al-Hendy, A. Hypovitaminosis D and high serum transforming growth factor beta-3: Important biomarkers for uterine fibroids risk. Fertil. Steril. 2016, 106, 1648–1649. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.L.; Segars, J.H. Is vitamin D the answer for prevention of uterine fibroids? Fertil. Steril. 2015, 104, 559–560. [Google Scholar] [CrossRef] [PubMed]
- Elkafas, H.; Qiwei, Y.; Al-Hendy, A. Origin of uterine fibroids: Conversion of myometrial stem cells to tumor-initiating cells. Semin. Reprod. Med. 2017, 35, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Holdsworth-Carson, S.J.; Zaitseva, M.; Vollenhoven, B.J.; Rogers, P.A. Clonality of smooth muscle and fibroblast cell populations isolated from human fibroid and myometrial tissues. Mol. Hum. Reprod. 2014, 20, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Ciavattini, A.; Petraglia, F.; Castellucci, M.; Ciarmela, P. Extracellular matrix in uterine leiomyoma pathogenesis: A potential target for future therapeutics. Hum. Reprod. Update 2018, 24, 59–85. [Google Scholar] [CrossRef] [PubMed]
- Sozen, I.; Arici, A. Interactions of cytokines, growth factors, and the extracellular matrix in the cellular biology of uterine leiomyomata. Fertil. Steril. 2002, 78, 1–12. [Google Scholar] [CrossRef]
- Rafique, S.; Segars, J.H.; Leppert, P.C. Mechanical signaling and extracellular matrix in uterine fibroids. Semin. Reprod. Med. 2017, 35, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Borahay, M.A.; Asoglu, M.R.; Mas, A.; Adam, S.; Kilic, G.S.; Al-Hendy, A. Estrogen receptors and signaling in fibroids: Role in pathobiology and therapeutic implications. Reprod. Sci. 2017, 24, 1235–1244. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Ishi, K.; Serna, V.A.; Kakazu, R.; Bulun, S.E.; Kurita, T. Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology 2010, 151, 2433–2442. [Google Scholar] [CrossRef] [PubMed]
- Ciarmela, P.; Islam, M.S.; Reis, F.M.; Gray, P.C.; Bloise, E.; Petraglia, F.; Vale, W.; Castellucci, M. Growth factors and myometrium: Biological effects in uterine fibroid and possible clinical implications. Hum. Reprod. Update 2011, 17, 772–790. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Protic, O.; Stortoni, P.; Grechi, G.; Lamanna, P.; Petraglia, F.; Castellucci, M.; Ciarmela, P. Complex networks of multiple factors in the pathogenesis of uterine leiomyoma. Fertil. Steril. 2013, 100, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Garrett, T.A.; Laughlin, S.K.; Davis, B.; Semelka, R.C.; Peddada, S.D. Short-term change in growth of uterine leiomyoma: Tumor growth spurts. Fertil. Steril. 2011, 95, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Chill, H.H.; Safrai, M.; Reuveni Salzman, A.; Shushan, A. The rising phoenix-progesterone as the main target of the medical therapy for leiomyoma. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Moravek, M.B.; Bulun, S.E. Endocrinology of uterine fibroids: Steroid hormones, stem cells, and genetic contribution. Curr. Opin. Obstet. Gynecol. 2015, 27, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Donnez, J.; Tatarchuk, T.F.; Bouchard, P.; Puscasiu, L.; Zakharenko, N.F.; Ivanova, T.; Ugocsai, G.; Mara, M.; Jilla, M.P.; Bestel, E.; et al. Ulipristal acetate versus placebo for fibroid treatment before surgery. N. Engl. J. Med. 2012, 366, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Chwalisz, K.; Taylor, H. Current and emerging medical treatments for uterine fibroids. Semin. Reprod. Med. 2017, 35, 510–522. [Google Scholar] [CrossRef] [PubMed]
- Borahay, M.A.; Al-Hendy, A.; Kilic, G.S.; Boehning, D. Signaling pathways in leiomyoma: Understanding pathobiology and implications for therapy. Mol. Med. 2015, 21, 242–256. [Google Scholar] [CrossRef] [PubMed]
- Makinen, N.; Mehine, M.; Tolvanen, J.; Kaasinen, E.; Li, Y.; Lehtonen, H.J.; Gentile, M.; Yan, J.; Enge, M.; Taipale, M.; et al. Med12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 2011, 334, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, H.R.; Sarvilinna, N.S.; Sjoberg, J.; Kampjarvi, K.; Pitkanen, E.; Vahteristo, P.; Makinen, N.; Aaltonen, L.A. Med12 mutation frequency in unselected sporadic uterine leiomyomas. Fertil. Steril. 2014, 102, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.K.; Laknaur, A.; Miller, J.; Layman, L.C.; Diamond, M.; Al-Hendy, A. Novel med12 gene somatic mutations in women from the southern united states with symptomatic uterine fibroids. Mol. Genet. Genom. 2015, 290, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.; Balk, E.M.; Brendel, M.; Ip, S.; Lau, J.; Lee, J.; Lichtenstein, A.; Patel, K.; Raman, G.; Tatsioni, A.; et al. Vitamin D and calcium: A systematic review of health outcomes. Evid. Rep. Technol. Assess. (Full Rep.) 2009, 183, 1–420. [Google Scholar]
- Norman, A.W. From vitamin D to hormone d: Fundamentals of the vitamin D endocrine system essential for good health. Am. J. Clin. Nutr. 2008, 88, 491S–499S. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Christakos, S. Biology and mechanisms of action of the vitamin D hormone. Endocrinol. Metab. Clin. N. Am. 2017, 46, 815–843. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin. Proc. 2006, 81, 353–373. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R. Who, what, where and when-influences on cutaneous vitamin D synthesis. Prog. Biophys. Mol. Biol. 2006, 92, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Verboven, C.; Rabijns, A.; De Maeyer, M.; Van Baelen, H.; Bouillon, R.; De Ranter, C. A structural basis for the unique binding features of the human vitamin d-binding protein. Nat. Struct. Biol. 2002, 9, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Hong, C.C.; Bandera, E.V.; Zhu, Q.; Liu, S.; Cheng, T.D.; Zirpoli, G.; Haddad, S.A.; Lunetta, K.L.; Ruiz-Narvaez, E.A.; et al. Demographic, lifestyle, and genetic determinants of circulating concentrations of 25-hydroxyvitamin D and vitamin d-binding protein in African American and European American women. Am. J. Clin. Nutr. 2017, 105, 1362–1371. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Lee, J.W.; Park, P.J.; Shin, Y.S.; Lee, W.Y.; Lee, K.A.; Ye, S.; Hyun, H.; Kang, K.N.; Yeo, D.; et al. The multiplex bead array approach to identifying serum biomarkers associated with breast cancer. Breast Cancer Res. 2009, 11, R22. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.P.; Chen, Y.W.; Liu, W.H.; Chou, H.C.; Chang, Y.P.; Lin, S.T.; Li, J.M.; Jian, S.F.; Lee, Y.R.; Chan, H.L. Proteomic identification of plasma biomarkers in uterine leiomyoma. Mol. Biosyst. 2012, 8, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Kato, S. The function of vitamin D receptor in vitamin D action. J. Biochem. 2000, 127, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Ylikomi, T.; Laaksi, I.; Lou, Y.R.; Martikainen, P.; Miettinen, S.; Pennanen, P.; Purmonen, S.; Syvala, H.; Vienonen, A.; Tuohimaa, P. Antiproliferative action of vitamin D. Vitam. Horm. 2002, 64, 357–406. [Google Scholar] [PubMed]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Spiro, A.; Buttriss, J.L. Vitamin d: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [PubMed]
- Nesby-O’Dell, S.; Scanlon, K.S.; Cogswell, M.E.; Gillespie, C.; Hollis, B.W.; Looker, A.C.; Allen, C.; Doughertly, C.; Gunter, E.W.; Bowman, B.A. Hypovitaminosis D prevalence and determinants among african american and white women of reproductive age: Third national health and nutrition examination survey, 1988–1994. Am. J. Clin. Nutr. 2002, 76, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Zadshir, A.; Tareen, N.; Pan, D.; Norris, K.; Martins, D. The prevalence of hypovitaminosis D among us adults: Data from the Nhanes III. Ethn. Dis. 2005, 15, S5-97–S5-101. [Google Scholar] [PubMed]
- Rosen, C.J. Clinical practice. Vitamin D insufficiency. N. Engl. J. Med. 2011, 364, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Guidelines for preventing and treating vitamin D deficiency and insufficiency revisited. J. Clin. Endocrinol. Metab. 2012, 97, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Trummer, C.; Pandis, M.; Schwetz, V.; Aberer, F.; Grubler, M.; Verheyen, N.; Tomaschitz, A.; Marz, W. Vitamin D: Current guidelines and future outlook. Anticancer Res. 2018, 38, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B. A review of the evidence supporting the vitamin D-cancer prevention hypothesis in 2017. Anticancer Res. 2018, 38, 1121–1136. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, M.; von Versen-Hoynck, F. Vitamin D—Roles in women’s reproductive health? Reprod. Biol. Endocrinol. 2011, 9, 146. [Google Scholar] [CrossRef] [PubMed]
- Lerchbaum, E.; Rabe, T. Vitamin D and female fertility. Curr. Opin. Obstet. Gynecol. 2014, 26, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Dabrowski, F.A.; Grzechocinska, B.; Wielgos, M. The role of vitamin D in reproductive health—A trojan horse or the golden fleece? Nutrients 2015, 7, 4139–4153. [Google Scholar] [CrossRef] [PubMed]
- Skowronska, P.; Pastuszek, E.; Kuczynski, W.; Jaszczol, M.; Kuc, P.; Jakiel, G.; Woclawek-Potocka, I.; Lukaszuk, K. The role of vitamin D in reproductive dysfunction in women—A systematic review. Ann. Agric. Environ. Med. 2016, 23, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Whynott, R.M.; Vaught, K.C.C.; Segars, J.H. The effect of uterine fibroids on infertility: A systematic review. Semin. Reprod. Med. 2017, 35, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Jukic, A.M.; Steiner, A.Z.; Baird, D.D. Lower plasma 25-hydroxyvitamin D is associated with irregular menstrual cycles in a cross-sectional study. Reprod. Biol. Endocrinol. 2015, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Jukic, A.M.; Steiner, A.Z.; Baird, D.D. Association between serum 25-hydroxyvitamin D and ovarian reserve in premenopausal women. Menopause 2015, 22, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Oskovi Kaplan, Z.A.; Tasci, Y.; Topcu, H.O.; Erkaya, S. 25-hydroxy vitamin D levels in premenopausal turkish women with uterine leiomyoma. Gynecol. Endocrinol. 2018, 34, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Ford, E.S.; Tsai, J.; Li, C.; Croft, J.B. Factors associated with vitamin D deficiency and inadequacy among women of childbearing age in the united states. ISRN Obstet. Gynecol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Catherino, W.H.; Eltoukhi, H.M.; Al-Hendy, A. Racial and ethnic differences in the pathogenesis and clinical manifestations of uterine leiomyoma. Semin. Reprod. Med. 2013, 31, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Mitro, S.D.; Zota, A.R. Vitamin D and uterine leiomyoma among a sample of us women: Findings from nhanes, 2001–2006. Reprod. Toxicol. 2015, 57, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Sharan, C.; Halder, S.K.; Thota, C.; Jaleel, T.; Nair, S.; Al-Hendy, A. Vitamin D inhibits proliferation of human uterine leiomyoma cells via catechol-o-methyltransferase. Fertil. Steril. 2011, 95, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C. Pcna: A silent housekeeper or a potential therapeutic target? Trends Pharmacol. Sci. 2014, 35, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Enserink, J.M.; Kolodner, R.D. An overview of cdk1-controlled targets and processes. Cell Div. 2010, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Hardwick, J.M.; Soane, L. Multiple functions of bcl-2 family proteins. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, E.; de Aquino Castro, R.; Gomes, M.T.; da Silva, I.D.; Baracat, E.C.; de Lima, G.R.; Sartori, M.G.; Girao, M.J. The catechol-O-methyltransferase (comt) gene polymorphism and prevalence of uterine fibroids. Maturitas 2008, 60, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.K.; Goodwin, J.S.; Al-Hendy, A. 1,25-dihydroxyvitamin D3 reduces TGF-beta3-induced fibrosis-related gene expression in human uterine leiomyoma cells. J. Clin. Endocrinol. Metab. 2011, 96, E754–762. [Google Scholar] [CrossRef] [PubMed]
- Joseph, D.S.; Malik, M.; Nurudeen, S.; Catherino, W.H. Myometrial cells undergo fibrotic transformation under the influence of transforming growth factor beta-3. Fertil. Steril. 2010, 93, 1500–1508. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Wlodarczyk, M.; Wrzosek, M.; Slabuszewska-Jozwiak, A.; Nowicka, G.; Jakiel, G. Ulipristal acetate decreases transforming growth factor beta3 serum and tumor tissue concentrations in patients with uterine fibroids. Fertil. Steril. 2018, 109, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Wlodarczyk, M.; Wrzosek, M.; Meczekalski, B.; Nowicka, G.; Lukaszuk, K.; Ciebiera, M.; Slabuszewska-Jozwiak, A.; Jakiel, G. Role of transforming growth factor beta in uterine fibroid biology. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Norian, J.; McCarthy-Keith, D.; Britten, J.; Catherino, W.H. Why leiomyomas are called fibroids: The central role of extracellular matrix in symptomatic women. Semin. Reprod. Med. 2010, 28, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Norian, J.M.; Malik, M.; Parker, C.Y.; Joseph, D.; Leppert, P.C.; Segars, J.H.; Catherino, W.H. Transforming growth factor beta3 regulates the versican variants in the extracellular matrix-rich uterine leiomyomas. Reprod. Sci. 2009, 16, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.K.; Sharan, C.; Al-Hendy, A. 1,25-dihydroxyvitamin D3 treatment shrinks uterine leiomyoma tumors in the Eker rat model. Biol. Reprod. 2012, 86, 116. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.K.; Sharan, C.; Al-Hendy, O.; Al-Hendy, A. Paricalcitol, a vitamin D receptor activator, inhibits tumor formation in a murine model of uterine fibroids. Reprod. Sci. 2014, 21, 1108–1119. [Google Scholar] [CrossRef] [PubMed]
- Al-Hendy, A.; Diamond, M.P.; El-Sohemy, A.; Halder, S.K. 1,25-dihydroxyvitamin D3 regulates expression of sex steroid receptors in human uterine fibroid cells. J. Clin. Endocrinol. Metab. 2015, 100, E572–582. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Greco, S.; Janjusevic, M.; Ciavattini, A.; Giannubilo, S.R.; D’Adderio, A.; Biagini, A.; Fiorini, R.; Castellucci, M.; Ciarmela, P. Growth factors and pathogenesis. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 34, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Protic, O.; Islam, M.S.; Greco, S.; Giannubilo, S.R.; Lamanna, P.; Petraglia, F.; Ciavattini, A.; Castellucci, M.; Hinz, B.; Ciarmela, P. Activin A in inflammation, tissue repair, and fibrosis: Possible role as inflammatory and fibrotic mediator of uterine fibroid development and growth. Semin. Reprod. Med. 2017, 35, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Al-Hendy, A.; Laknaur, A.; Diamond, M.P.; Ismail, N.; Boyer, T.G.; Halder, S.K. Silencing med12 gene reduces proliferation of human leiomyoma cells mediated via WNT/beta-catenin signaling pathway. Endocrinology 2017, 158, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Leppert, P.C.; Baginski, T.; Prupas, C.; Catherino, W.H.; Pletcher, S.; Segars, J.H. Comparative ultrastructure of collagen fibrils in uterine leiomyomas and normal myometrium. Fertil. Steril. 2004, 82, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Oblak, M.; Mlinsek, G.; Kandus, A.; Buturovic-Ponikvar, J.; Arnol, M. Effects of paricalcitol on biomarkers of inflammation and fibrosis in kidney transplant recipients: Results of a randomized controlled trial. Clin. Nephrol. 2017, 88, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Al-Hendy, A.; Diamond, M.P.; Boyer, T.G.; Halder, S.K. Vitamin D3 inhibits WNT/beta-catenin and MTOR signaling pathways in human uterine fibroid cells. J. Clin. Endocrinol. Metab. 2016, 101, 1542–1551. [Google Scholar] [CrossRef] [PubMed]
- Gene, N. Fen1 Flap Structure-Specific Endonuclease 1 [Homo Sapiens (Human)]. 8.04.2018. Available online: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=2237 (accessed on 10 June 2018).
- Singh, P.; Yang, M.; Dai, H.; Yu, D.; Huang, Q.; Tan, W.; Kernstine, K.H.; Lin, D.; Shen, B. Overexpression and hypomethylation of flap endonuclease 1 gene in breast and other cancers. Mol. Cancer Res. 2008, 6, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Deng, L.; Chen, Y.; Li, G.C.; Shao, C.; Tischfield, J.A. Modulation of DNA end joining by nuclear proteins. J. Biol. Chem. 2005, 280, 31442–31449. [Google Scholar] [CrossRef] [PubMed]
- Dou, Q.; Tarnuzzer, R.W.; Williams, R.S.; Schultz, G.S.; Chegini, N. Differential expression of matrix metalloproteinases and their tissue inhibitors in leiomyomata: A mechanism for gonadotrophin releasing hormone agonist-induced tumour regression. Mol. Hum. Reprod. 1997, 3, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Brew, K.; Dinakarpandian, D.; Nagase, H. Tissue inhibitors of metalloproteinases: Evolution, structure and function. Biochim. Biophys. Acta 2000, 1477, 267–283. [Google Scholar] [CrossRef]
- Halder, S.K.; Osteen, K.G.; Al-Hendy, A. Vitamin D3 inhibits expression and activities of matrix metalloproteinase-2 and -9 in human uterine fibroid cells. Hum. Reprod. 2013, 28, 2407–2416. [Google Scholar] [CrossRef] [PubMed]
- Artaza, J.N.; Norris, K.C. Vitamin D reduces the expression of collagen and key profibrotic factors by inducing an antifibrotic phenotype in mesenchymal multipotent cells. J. Endocrinol. 2009, 200, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A.; Ruiz-Narvaez, E.A.; Haddad, S.A.; Rosenberg, L.; Palmer, J.R. Polymorphisms in vitamin D-related genes and risk of uterine leiomyomata. Fertil. Steril. 2014, 102, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi, S. Exploring the link between VDR rs2228570 and uterine leiomyoma in Iranian women. Egypt. J. Med. Hum. Genet. 2016, 17, 115–118. [Google Scholar] [CrossRef]
- Gulec Yilmaz, S.; Gul, T.; Attar, R.; Yildirim, G.; Isbir, T. Association between fok1 polymorphism of vitamin D receptor gene with uterine leiomyoma in Turkish populations. J. Turk. Ger. Gynecol. Assoc. 2018. [Google Scholar] [CrossRef]
- Parazzini, F.; Di Martino, M.; Candiani, M.; Vigano, P. Dietary components and uterine leiomyomas: A review of published data. Nutr. Cancer 2015, 67, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Akhtar, M.M.; Segars, J.H.; Castellucci, M.; Ciarmela, P. Molecular targets of dietary phytochemicals for possible prevention and therapy of uterine fibroids: Focus on fibrosis. Crit. Rev. Food Sci. Nutr. 2017, 57, 3583–3600. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Karczmarewicz, E.; Bayer, M.; Carter, G.; Chlebna-Sokol, D.; Czech-Kowalska, J.; Debski, R.; Decsi, T.; Dobrzanska, A.; Franek, E.; et al. Practical guidelines for the supplementation of vitamin D and the treatment of deficits in central Europe—Recommended vitamin D intakes in the general population and groups at risk of vitamin D deficiency. Endokrynol. Pol. 2013, 64, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Tebben, P.J.; Singh, R.J.; Kumar, R. Vitamin d-mediated hypercalcemia: Mechanisms, diagnosis, and treatment. Endocr. Rev. 2016, 37, 521–547. [Google Scholar] [CrossRef] [PubMed]
- Ciavattini, A.; Delli Carpini, G.; Serri, M.; Vignini, A.; Sabbatinelli, J.; Tozzi, A.; Aggiusti, A.; Clemente, N. Hypovitaminosis D and “small burden” uterine fibroids: Opportunity for a vitamin D supplementation. Medicine 2016, 95, e5698. [Google Scholar] [CrossRef] [PubMed]
- Vojinovic, J. Vitamin D receptor agonists’ anti-inflammatory properties. Ann. N. Y. Acad. Sci. 2014, 1317, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Anami, Y.; Itoh, T. Development of vitamin D analogs modulating the pocket structure of vitamin D receptor. Curr. Top. Med. Chem. 2014, 14, 2378–2387. [Google Scholar] [CrossRef] [PubMed]
- Trillini, M.; Cortinovis, M.; Ruggenenti, P.; Reyes Loaeza, J.; Courville, K.; Ferrer-Siles, C.; Prandini, S.; Gaspari, F.; Cannata, A.; Villa, A.; et al. Paricalcitol for secondary hyperparathyroidism in renal transplantation. J. Am. Soc. Nephrol. 2015, 26, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kong, J.; Deb, D.K.; Chang, A.; Li, Y.C. Vitamin D receptor attenuates renal fibrosis by suppressing the renin-angiotensin system. J. Am. Soc. Nephrol. 2010, 21, 966–973. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Nino, M.D.; Bozic, M.; Cordoba-Lanus, E.; Valcheva, P.; Gracia, O.; Ibarz, M.; Fernandez, E.; Navarro-Gonzalez, J.F.; Ortiz, A.; Valdivielso, J.M. Beyond proteinuria: VDR activation reduces renal inflammation in experimental diabetic nephropathy. Am. J. Physiol. Ren. Physiol. 2012, 302, F647–657. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Li, Y.; Liu, Y. Paricalcitol attenuates renal interstitial fibrosis in obstructive nephropathy. J. Am. Soc. Nephrol. 2006, 17, 3382–3393. [Google Scholar] [CrossRef] [PubMed]
- Stavenuiter, A.W.; Farhat, K.; Vila Cuenca, M.; Schilte, M.N.; Keuning, E.D.; Paauw, N.J.; ter Wee, P.M.; Beelen, R.H.; Vervloet, M.G. Protective effects of paricalcitol on peritoneal remodeling during peritoneal dialysis. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Poveda, J.; Sanz, A.B.; Fernandez-Fernandez, B.; Carrasco, S.; Ruiz-Ortega, M.; Cannata-Ortiz, P.; Ortiz, A.; Sanchez-Nino, M.D. Mxra5 is a tgf-beta1-regulated human protein with anti-inflammatory and anti-fibrotic properties. J. Cell. Mol. Med. 2017, 21, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Bartels, C.B.; Cayton, K.C.; Chuong, F.S.; Holthouser, K.; Arian, S.E.; Abraham, T.; Segars, J.H. An evidence-based approach to the medical management of fibroids: A systematic review. Clin. Obstet. Gynecol. 2016, 59, 30–52. [Google Scholar] [CrossRef] [PubMed]
- Buggio, L.; Roncella, E.; Somigliana, E.; Vercellini, P. Vitamin D and benign gynaecological diseases: A critical analysis of the current evidence. Gynecol. Endocrinol. 2016, 32, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Kashani, B.N.; Centini, G.; Morelli, S.S.; Weiss, G.; Petraglia, F. Role of medical management for uterine leiomyomas. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 34, 85–103. [Google Scholar] [CrossRef] [PubMed]
- Al-Hendy, A.; Badr, M. Can vitamin D reduce the risk of uterine fibroids? Women’s Health 2014, 10, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.L.A.; Shaheen, S.; Sabri, M.; Al-Hendy, A. Vitamin D synergizes the antiproliferative, apoptotic, antifibrotic and anti-inflammatory effects of ulipristal acetate against human uterine fibroids. Fertil. Steril. 2017, 108, e66. [Google Scholar] [CrossRef]
- Ali, M.; Chaudhry, Z.T.; Al-Hendy, A. Successes and failures of uterine leiomyoma drug discovery. Expert Opin. Drug Discov. 2018, 13, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C.; Haq, A. The concept of the personal vitamin D response index. J. Steroid Biochem. Mol. Biol. 2018, 175, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Harmon, Q.E.; Umbach, D.M.; Baird, D.D. Use of estrogen-containing contraception is associated with increased concentrations of 25-hydroxy vitamin D. J. Clin. Endocrinol. Metab. 2016, 101, 3370–3377. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.W.M.; Słabuszewska-Jóźwiak, A.; Ciebiera, M.; Nowicka, G.; Jakiel, G. Vitamin D serum levels in women using contraception containing drospirenone—A preliminary study. Arch. Med. Sci. 2017. [Google Scholar] [CrossRef]
- Morch, L.S.; Skovlund, C.W.; Hannaford, P.C.; Iversen, L.; Fielding, S.; Lidegaard, O. Contemporary hormonal contraception and the risk of breast cancer. N. Engl. J. Med. 2017, 377, 2228–2239. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Green, J.; Berrington de Gonzalez, A.; Appleby, P.; Peto, J.; Plummer, M.; Franceschi, S.; Beral, V. Cervical cancer and use of hormonal contraceptives: A systematic review. Lancet 2003, 5, 1159–1167. [Google Scholar] [CrossRef]
- Iversen, L.; Sivasubramaniam, S.; Lee, A.J.; Fielding, S.; Hannaford, P.C. Lifetime cancer risk and combined oral contraceptives: The royal college of general practitioners’ oral contraception study. Am. J. Obstet. Gynecol. 2017, 216, 580.e1–580.e9. [Google Scholar] [CrossRef] [PubMed]
- McLaren, J.S.; Morris, E.; Rymer, J. Gonadotrophin receptor hormone analogues in combination with add-back therapy: An update. Menopause Int. 2012, 18, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Somekawa, Y.; Chigughi, M.; Harada, M.; Ishibashi, T. Use of vitamin K2 (menatetrenone) and 1,25-dihydroxyvitamin D3 in the prevention of bone loss induced by leuprolide. J. Clin. Endocrinol. Metab. 1999, 84, 2700–2704. [Google Scholar] [CrossRef] [PubMed]
- Lerchbaum, E. Vitamin D and menopause—A narrative review. Maturitas 2014, 79, 3–7. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciebiera, M.; Włodarczyk, M.; Ciebiera, M.; Zaręba, K.; Łukaszuk, K.; Jakiel, G. Vitamin D and Uterine Fibroids—Review of the Literature and Novel Concepts. Int. J. Mol. Sci. 2018, 19, 2051. https://doi.org/10.3390/ijms19072051
Ciebiera M, Włodarczyk M, Ciebiera M, Zaręba K, Łukaszuk K, Jakiel G. Vitamin D and Uterine Fibroids—Review of the Literature and Novel Concepts. International Journal of Molecular Sciences. 2018; 19(7):2051. https://doi.org/10.3390/ijms19072051
Chicago/Turabian StyleCiebiera, Michał, Marta Włodarczyk, Magdalena Ciebiera, Kornelia Zaręba, Krzysztof Łukaszuk, and Grzegorz Jakiel. 2018. "Vitamin D and Uterine Fibroids—Review of the Literature and Novel Concepts" International Journal of Molecular Sciences 19, no. 7: 2051. https://doi.org/10.3390/ijms19072051
APA StyleCiebiera, M., Włodarczyk, M., Ciebiera, M., Zaręba, K., Łukaszuk, K., & Jakiel, G. (2018). Vitamin D and Uterine Fibroids—Review of the Literature and Novel Concepts. International Journal of Molecular Sciences, 19(7), 2051. https://doi.org/10.3390/ijms19072051





