A Child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 Mutation Associated with a Severe Early Infantile Epileptic Encephalopathy
Abstract
1. Introduction
2. Discussion
3. Materials and Methods
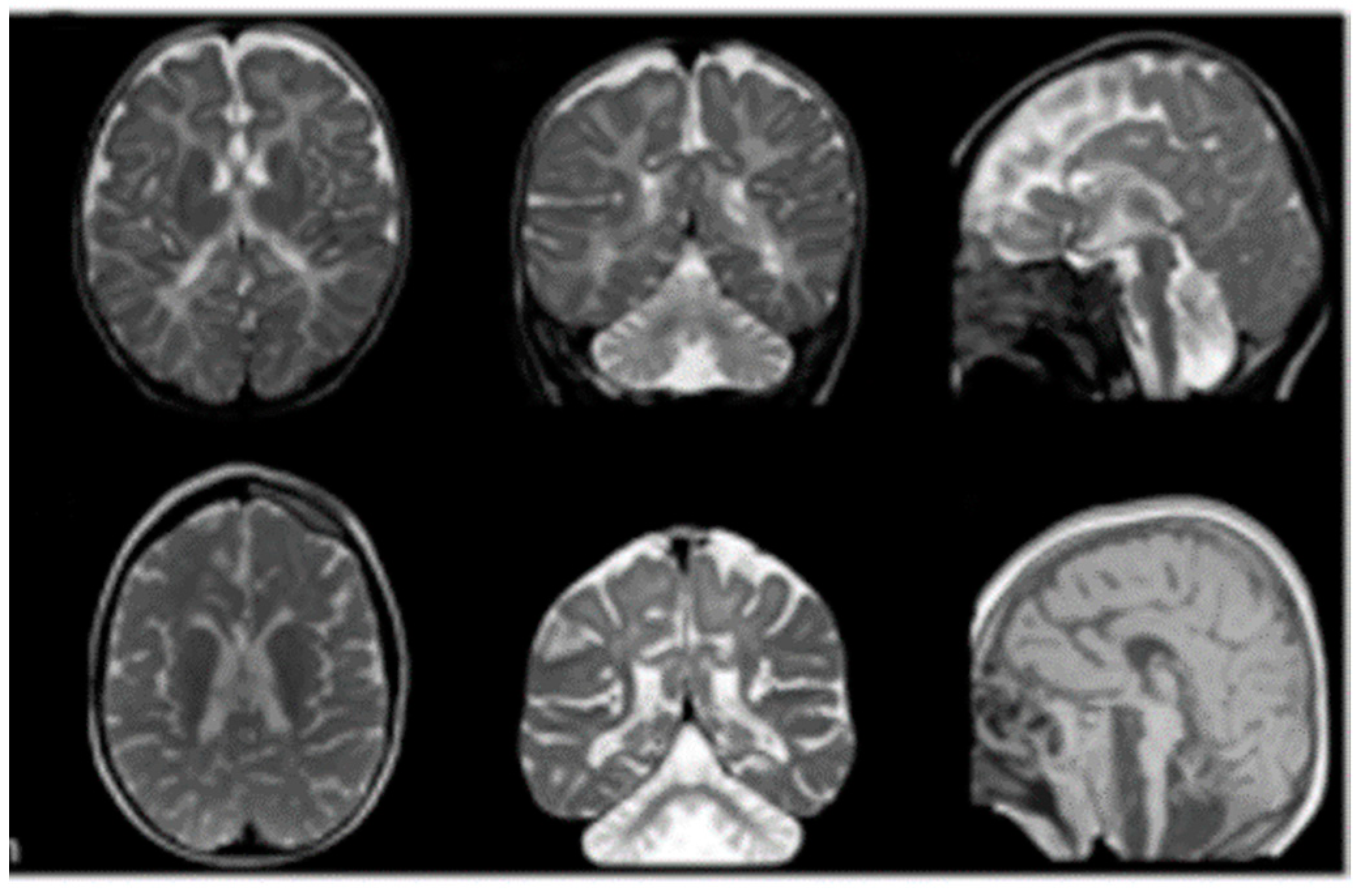
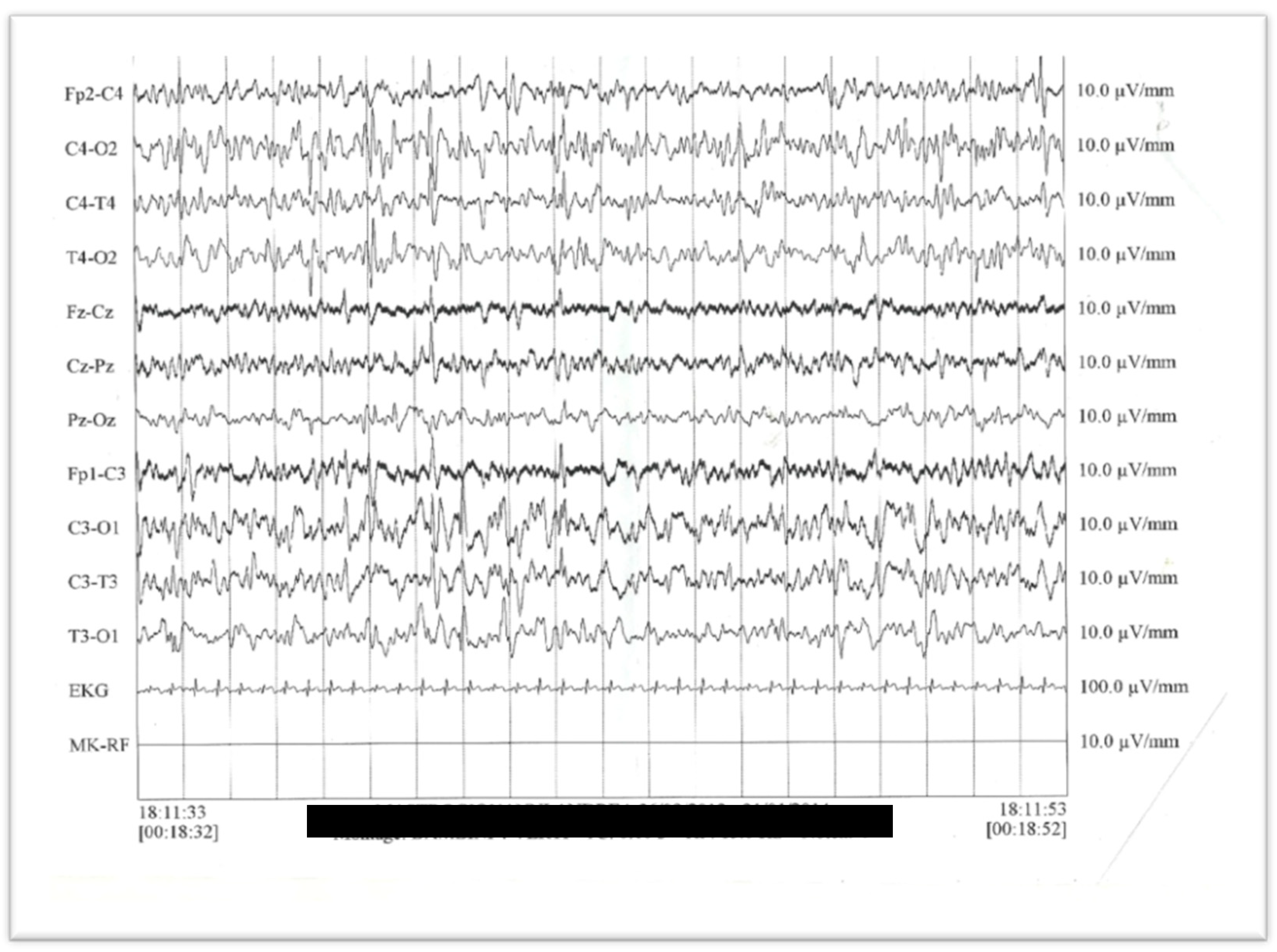
3.1. Case Report
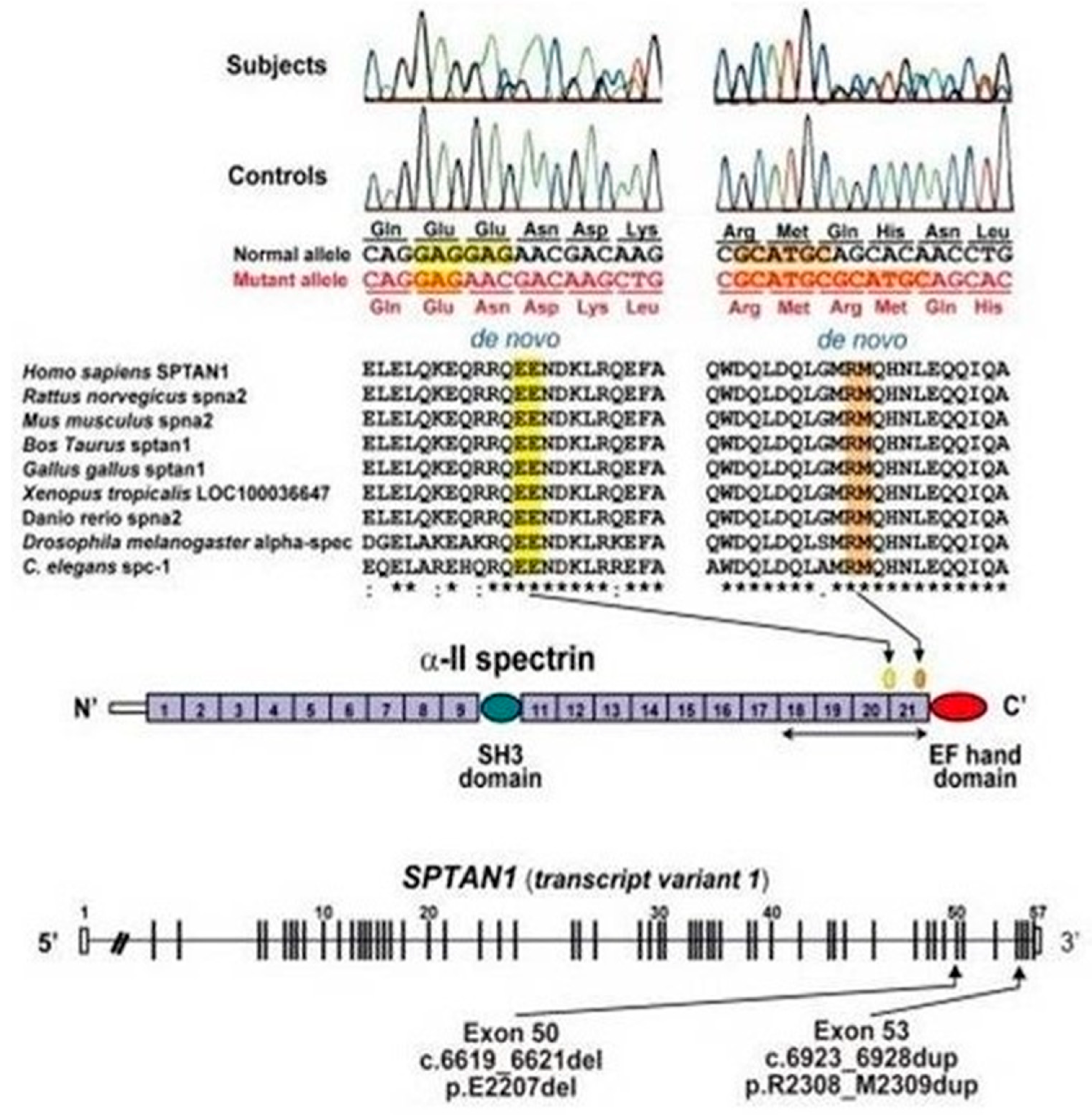
3.2. Genetic Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Berg, A.T.; Berkovic, S.F.; Brodie, M.J.; Buchhalter, J.; Cross, J.H.; van Emde Boas, W.; Engel, J.; French, J.; Glauser, T.A.; Mathern, G.W.; et al. Revised terminology and concepts for organization of seizures and epilepsies: Report of the ILAE Commission on Classification and Terminology, 2005–2009. Epilepsia 2010, 51, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Radaelli, G.; de Souza Santos, F.; Borelli, W.V.; Pisani, L.; Nunes, M.L.; Scorza, F.A.; da Costa, J.C. Causes of mortality in early infantile epileptic encephalopathy: A systematic review. Epilepsy Behav. 2018, 85, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Pavone, P.; Corsello, G.; Ruggieri, M.; Marino, S.; Marino, S.; Falsaperla, R. Benign and severe early-life seizures: A round in the first year of life. Ital. J. Pediatr. 2018, 44, 54. [Google Scholar] [CrossRef] [PubMed]
- Nieh, S.E.; Sherr, E.H. Epileptic encephalopathies: New genes and new pathways. Neurotherapeutics 2014, 11, 796–806. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Zhang, C.; Zhao, Q.; Li, D. Spectrin: Structure, function and disease. Sci. China Life Sci. 2013, 56, 1076–1085. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ji, T.; Nelson, A.D.; Glanowska, K.; Murphy, G.G.; Jenkins, P.M.; Parent, J.M. Critical roles of αII spectrin in brain development and epileptic encephalopathy. J. Clin. Investig. 2018, 128, 760–773. [Google Scholar] [CrossRef] [PubMed]
- Begg, G.E.; Harper, S.L.; Morris, M.B.; Speicher, D.W. Initiation of spectrin dimerization involves complementary electrostatic interactions between paired triple-helical bundles. J. Biol. Chem. 2000, 275, 3279–3287. [Google Scholar] [CrossRef] [PubMed]
- Syrbe, S.; Harms, F.L.; Parrini, E.; Montomoli, M.; Mütze, U.; Helbig, K.L.; Polster, T.; Albrecht, B.; Bernbeck, U.; Van Binsbergen, E.; et al. Delineating SPTAN1 associated phenotypes: From isolated epilepsy to encephalopathy with progressive brain atrophy. Brain 2017, 140, 2322–2336. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.H. Epilepsy syndromes during the first year of life and the usefulness of an epilepsy gene panel. Korean J. Pediatr. 2018, 61, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Bennett, V.; Baines, A.J. Spectrin and ankyrin-based pathways: Metazoan inventions for integrating cells into tissues. Physiol. Rev. 2001, 81, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Leterrier, C. The axon initial segment: An updated viewpoint. J. Neurosci. 2018, 38, 2135–2145. [Google Scholar] [CrossRef] [PubMed]
- Tohyama, J.; Nakashima, M.; Nabatame, S.; Gaik-Siew, C.; Miyata, R.; Rener-Primec, Z.; Kato, M.; Matsumoto, N.; Saitsu, H. SPTAN1 encephalopathy: Distinct phenotypes and genotypes. J. Hum. Genet. 2015, 60, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Saitsu, H.; Tohyama, J.; Kumada, T.; Egawa, K.; Hamada, K.; Okada, I.; Mizuguchi, T.; Osaka, H.; Miyata, R.; Furukawa, T.; et al. Dominant-negative mutations in α-II spectrin cause West syndrome with severe cerebral hypomyelination, spastic quadriplegia, and developmental delay. Am. J. Hum. Genet. 2010, 86, 881–891. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rapaccini, V.; Esposito, S.; Strinati, F.; Allegretti, M.; Manfroi, E.; Miconi, F.; Pitzianti, M.; Prontera, P.; Principi, N.; Pasini, A. A Child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 Mutation Associated with a Severe Early Infantile Epileptic Encephalopathy. Int. J. Mol. Sci. 2018, 19, 1976. https://doi.org/10.3390/ijms19071976
Rapaccini V, Esposito S, Strinati F, Allegretti M, Manfroi E, Miconi F, Pitzianti M, Prontera P, Principi N, Pasini A. A Child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 Mutation Associated with a Severe Early Infantile Epileptic Encephalopathy. International Journal of Molecular Sciences. 2018; 19(7):1976. https://doi.org/10.3390/ijms19071976
Chicago/Turabian StyleRapaccini, Valentina, Susanna Esposito, Francesco Strinati, Mariella Allegretti, Elisabetta Manfroi, Francesco Miconi, Mariabernarda Pitzianti, Paolo Prontera, Nicola Principi, and Augusto Pasini. 2018. "A Child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 Mutation Associated with a Severe Early Infantile Epileptic Encephalopathy" International Journal of Molecular Sciences 19, no. 7: 1976. https://doi.org/10.3390/ijms19071976
APA StyleRapaccini, V., Esposito, S., Strinati, F., Allegretti, M., Manfroi, E., Miconi, F., Pitzianti, M., Prontera, P., Principi, N., & Pasini, A. (2018). A Child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 Mutation Associated with a Severe Early Infantile Epileptic Encephalopathy. International Journal of Molecular Sciences, 19(7), 1976. https://doi.org/10.3390/ijms19071976





