Biological Activities and Safety of Citrus spp. Essential Oils
Abstract
1. Introduction
2. Biological Properties
2.1. Sweet Orange (Citrus sinensis L.) Essential Oil
2.2. Bitter Orange (Citrus aurantium L.) Essential Oil
2.3. Neroli (Citrus aurantium L.) Essential Oil
2.4. Orange Petitgrain (Citrus aurantium L.) Essential Oil
2.5. Mandarin (Citrus reticulata Blanco) Essential Oil
2.6. Lemon (Citrus limon Osbeck) Essential Oil
2.7. Key Lime (Citrus aurantifolia) Essential Oil
2.8. Grapefruit (Citrus × paradisi Macfady) Essential Oil
2.9. Bergamot (Citrus bergamia Risso & Poit) Essential Oil
2.10. Yuzu or Yuja (Citrus junos Sieb. ex Tanaka) Essential Oil
2.11. Kumquat (Citrus japonica Thunb) Essential Oil
3. Safety of Citrus Oils
4. Bioactivity and Safety of Individual Key Components
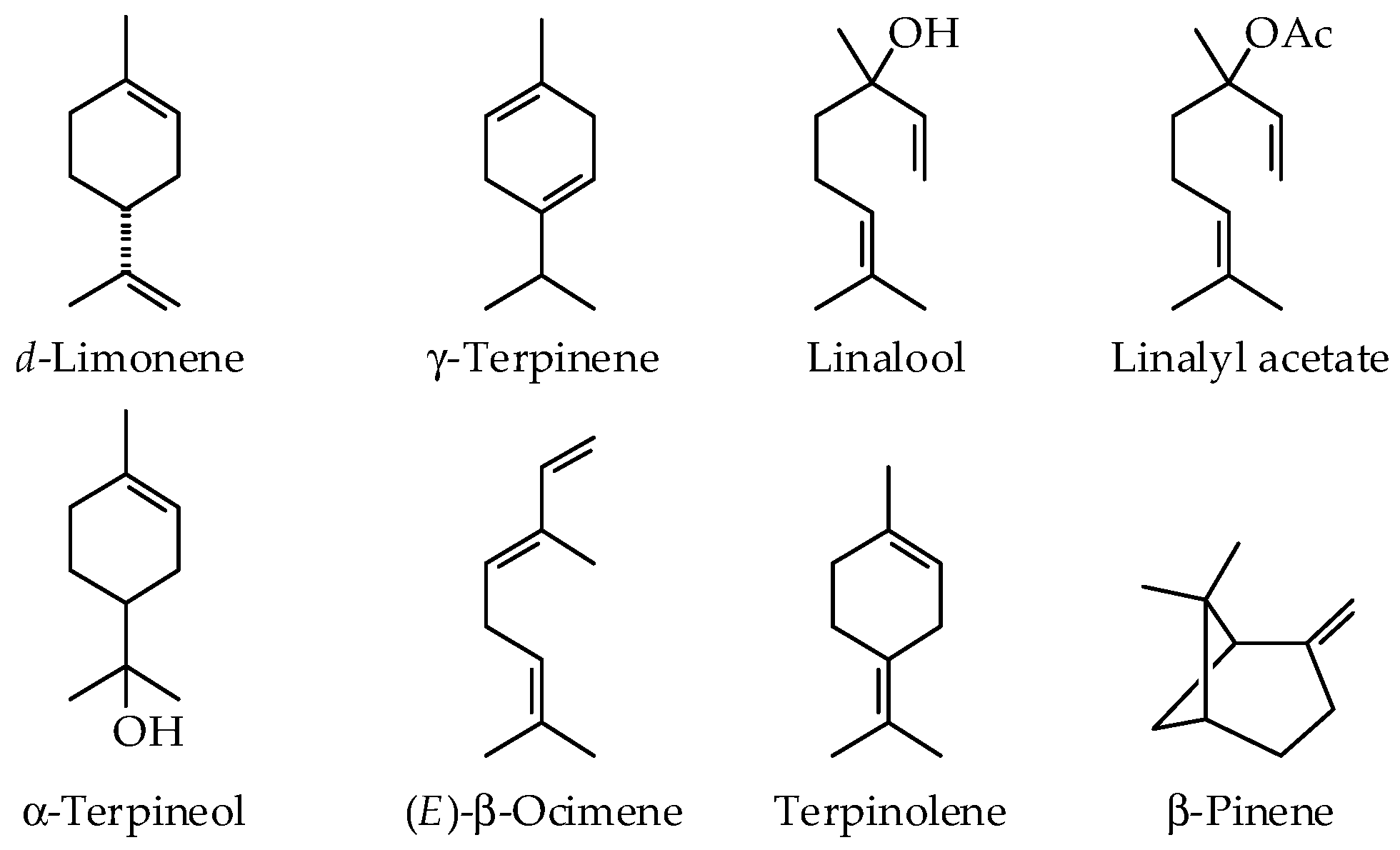
4.1. d-Limonene
4.2. γ-Terpinene
4.3. Linalool
4.4. Linalyl Acetate
4.5. α-Terpineol
4.6. Geranyl Acetate
4.7. Terpinolene
4.8. β-Pinene
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 5-HT | serotonin |
| ACC | anterior cingulate cortex |
| AMPK | AMP-activated protein kinase |
| BLM | bleomycin |
| COX-2 | cyclooxygenase-2 |
| CTGF | connective tissue growth factor |
| DA | dopamine |
| DBP | Dibenzo-[α]-pyrene |
| DENA | N-nitrosodiethylamine |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| EO | essential oil |
| EPM | elevated plus-maze |
| ERK | extracellular signal-regulated protein kinase |
| FCF | furanocoumarin-free |
| GABA | gamma-aminobutyric acid |
| GRAS | generally recognized as safe |
| GSH | glutathione |
| HELFs | human embryonic lung fibroblasts |
| HL-60 | human leukemia cells |
| HPA | hypothalamic-pituitary-adrenal |
| IC50 | median inhibitory concentration |
| IR | insulin resistance |
| LD50 | median lethal dose |
| LDL | low-density lipoprotein |
| LNCaP | human prostate acedocarcinoma cells |
| MI | myocardial infarction |
| NDMA | N-nitrosodimethylamine |
| NNK | 4-(methylnitrosoamine)-1-(3-pyridyl)-1-butanone |
| NVP | nausea and vomiting of pregnancy |
| phospho-Akt | phosphorylated protein kinase B |
| phospho-GSK-3β | phosphorylated glycogen synthase kinase 3 beta |
| PMS | premenstrual syndrome |
| PPAR-γ | peroxisome proliferator-activated receptor gamma |
| ppm | parts per million |
| ROS | reactive oxygen species |
| RyR | ryanodine receptor |
| SH-SY5Y | human neuroblasoma cells |
| WAT | white adipose tissue |
References
- Moore, G.A. Oranges and lemons: Clues to the taxonomy of Citrus from molecular markers. Trends Genet. 2001, 17, 536–540. [Google Scholar] [CrossRef]
- Mabberley, D.J. Citrus (Rutaceae): A review of recent advances in etymology, systematics and medical applications. Blumea 2004, 49, 481–498. [Google Scholar] [CrossRef]
- Anwar, F.; Naseer, R.; Bhanger, M.I.; Ashraf, S.; Talpur, F.N.; Aladedunye, F.A. Physico-chemical characteristics of citrus seeds and seed oils from Pakistan. J. Am. Oil Chem. Soc. 2008, 85, 321–330. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Cho, M.H.; Lee, Y.R. Converting citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.A.; Siles, J.A.; Chica, A.F.; Martín, A. Biomethanization of orange peel waste. Bioresour. Technol. 2010, 101, 8993–8999. [Google Scholar] [CrossRef] [PubMed]
- Rezzadori, K.; Benedetti, S.; Amante, E.R. Proposals for the residues recovery: Orange waste as raw material for new products. Food Bioprod. Process. 2012, 90, 606–614. [Google Scholar] [CrossRef]
- Ferhat, M.A.; Meklati, B.Y.; Smadja, J.; Chemat, F. An improved microwave Clevenger apparatus for distillation of essential oils from orange peel. J. Chromatogr. A 2006, 1112, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Tisserand, R.; Young, R. Essential Oil Safety, 2nd ed.; Elsevier: New York, NY, USA, 2014. [Google Scholar]
- Mitropoulou, G.; Fitsiou, E.; Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Bardouki, H.; Vamvakias, M.; Panas, P.; Chlichlia, K.; Pappa, A.; Kourkoutas, Y. Citrus medica essential oil exhibits significant antimicrobial and antiproliferative activity. LWT Food Sci. Technol. 2017, 84, 344–352. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Perez-Álvarez, J. Antifungal activity of lemon (Citrus limon L.), mandarin (Citrus reticulata L.), grapefruit (Citrus paradisi L.) and orange (Citrus sinensis L.) essential oils. Food Control 2008, 19, 1130–1138. [Google Scholar] [CrossRef]
- Ali, N.; Chhetri, B.; Dosoky, N.; Shari, K.; Al-Fahad, A.; Wessjohann, L.; Setzer, W. Antimicrobial, antioxidant, and cytotoxic activities of Ocimum forskolei and Teucrium yemense (Lamiaceae) essential oils. Medicines 2017, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Yan, J.; Sun, Z. D-limonene exhibits anti-inflammatory and antioxidant properties in an ulcerative colitis rat model via regulation of iNOS, COX-2, PGE2 and ERK signaling pathways. Mol. Med. Rep. 2017, 15, 2339–2346. [Google Scholar] [CrossRef] [PubMed]
- Roberto, D.; Micucci, P.; Sebastian, T.; Graciela, F.; Anesini, C. Antioxidant activity of limonene on normal murine lymphocytes: Relation to H2O2 modulation and cell proliferation. Basic Clin. Pharmacol. Toxicol. 2010, 106, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I.; Bhatia, S.; Grigorov, P.; Balkansky, S.; Parmar, V.S.; Prasad, A.K.; Saso, L. Coumarins as antioxidants. Curr. Med. Chem. 2011, 18, 3929–3951. [Google Scholar] [CrossRef] [PubMed]
- Caccioni, D.R.; Guizzardi, M.; Biondi, D.M.; Renda, A.; Ruberto, G. Relationship between volatile components of citrus fruit essential oils and antimicrobial action on Penicillium digitatum and Penicillium italicum. Int. J. Food Microbiol. 1998, 43, 73–79. [Google Scholar] [CrossRef]
- De Pasquale, F.; Siragusa, M.; Abbate, L.; Tusa, N.; De Pasquale, C.; Alonzo, G. Characterization of five sour orange clones through molecular markers and leaf essential oils analysis. Sci. Hortic. 2006, 109, 54–59. [Google Scholar] [CrossRef]
- Dosoky, N.S.; Moriarity, D.M.; Setzer, W.N. Phytochemical and biological investigations of Conradina canescens. Nat. Prod. Commun. 2016, 11, 25–28. [Google Scholar] [PubMed]
- Dosoky, N.S.; Stewart, C.D.; Setzer, W.N. Identification of essential oil components from Conradina canescens. Am. J. Essent. Oils Nat. Prod. 2014, 2, 24–28. [Google Scholar]
- da Silva, J.K.; da Trindade, R.; Moreira, E.C.; Maia, J.G.S.; Dosoky, N.S.; Miller, R.S.; Cseke, L.J.; Setzer, W.N. Chemical diversity, biological activity, and genetic aspects of three Ocotea species from the Amazon. Int. J. Mol. Sci. 2017, 18, 1081. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.H.; Bouajila, J.; Lebrihi, A.; Mathieu, F.; Romdhane, M.; Zagrouba, F. Chemical composition and in vitro antimicrobial and antioxidant activities of Citrus aurantium L. flowers essential oil (Neroli oil). Pak. J. Biol. Sci. 2012, 15, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.F.; Li, Y.H.; Li, B.L.; Lu, B.Y.; Xia, N.H. Aromatic Plants and Their Essential Constitutes; South China Inst. Bot., Chinese Academy of Sciences, Peace Book Co.: Hong Kong, China, 1993. [Google Scholar]
- Kubeczka, K.-H. Essential Oils Analysis by Capillary Gas Chromatography and Carbon-13 NMR Spectroscopy; Wiley: Chichester, UK, 2002. [Google Scholar]
- Pino, J.A.; Rosado, A. Comparative investigation of the distilled lime oils (Citrus aurantifolia Swingle and Citrus latifolia Tanaka) from Cuba. J. Essent. Oil Res. 2001, 13, 179–180. [Google Scholar] [CrossRef]
- Dugo, P.; Mondello, L.; Proteggente, A.R.; Cavazza, A.; Dugo, G. Oxygen heterocyclic compounds of bergamot essential oils. Riv. Ital. EPPOS 1999, 27, 31–41. [Google Scholar]
- Verzera, A.; Trozzi, A.; Stagno d’Alcontres, I.; Mondello, L.; Dugo, G.; Sebastiani, E. The composition of the volatile fraction of calabrian bergamot essential oil. Riv. Ital. EPPOS 1998, 25, 17–38. [Google Scholar]
- Dugo, P.; Mondello, L.; Sebastiani, E.; Ottanà, R.; Errante, G.; Dugo, G. Identification of minor oxygen heterocyclic compounds of citrus essential oils by liquid chromatography-atmospheric pressure chemical ionisation mass spectrometry. J. Liq. Chromatogr. Relat. Technol. 1999, 22, 2991–3005. [Google Scholar] [CrossRef]
- Sawamura, M.; Hasegawa, K.; Kashiwagi, T.; Nguyen Thi, L.-P.; Wada, M.; Kumagai, C. Determination of bergapten in Japanese citrus essential oils. Jpn. J. Aromather. 2009, 9, 30–37. [Google Scholar]
- Hata, T.; Sakaguchi, I.; Mori, M.; Ikeda, N.; Kato, Y.; Minamino, M.; Watabe, K. Induction of apoptosis by Citrus paradisi essential oil in human leukemic (HL-60) cells. In Vivo 2003, 17, 553–559. [Google Scholar] [PubMed]
- Chidambara Murthy, K.N.; Jayaprakasha, G.K.; Patil, B.S. D-limonene rich volatile oil from blood oranges inhibits angiogenesis, metastasis and cell death in human colon cancer cells. Life Sci. 2012, 91, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Ikei, H.; Song, C.; Miyazaki, Y. Effects of olfactory stimulation with rose and orange oil on prefrontal cortex activity. Complement. Ther. Med. 2014, 22, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Goes, T.C.; Antunes, F.D.; Alves, P.B.; Teixeira-Silva, F. Effect of sweet orange aroma on experimental anxiety in humans. J. Altern. Complement. Med. 2012, 18, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Faturi, C.B.; Leite, J.R.; Alves, P.B.; Canton, A.C.; Teixeira-Silva, F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Lehrner, J.; Eckersberger, C.; Walla, P.; Potsch, G.; Deecke, L. Ambient odor of orange in a dental office reduces anxiety and improves mood in female patients. Physiol. Behav. 2000, 71, 83–86. [Google Scholar] [CrossRef]
- Yip, Y.B.; Tam, A.C.Y. An experimental study on the effectiveness of massage with aromatic ginger and orange essential oil for moderate-to-severe knee pain among the elderly in Hong Kong. Complement. Ther. Med. 2008, 16, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Bodake, H.B.; Panicker, K.N.; Kailaje, V.; Rao, K.V. Chemopreventive effect of orange oil on the development of hepatic preneoplastic lesions induced by N-nitrosodiethylamine in rats: An ultrastructural study. Indian J. Exp. Biol. 2002, 40, 245–251. [Google Scholar] [PubMed]
- Homburger, F.; Treger, A.; Boger, E. Inhibition of murine subcutaneous and intravenous benzo(rst)pentaphene carcinogenesis by sweet orange oils and d-limonene. Oncology 1971, 25, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Asjad, H.; Akhtar, M.; Bashir, S.; Gulzar, B.; Khalid, R.; Asad, M. Phenol, flavonoid contents and antioxidant activity of six common citrus plants in Pakistan. J. Pharm. Cosmet. Sci. 2013, 1, 1–5. [Google Scholar]
- Rimini, S.; Petracci, M.; Smith, D.P. The use of thyme and orange essential oils blend to improve quality traits of marinated chicken meat. Poult. Sci. 2014, 93, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Matiz, G.; Osorio, M.R.; Camacho, F.; Atencia, M.; Herazo, J. Effectiveness of antimicrobial formulations for acne based on orange (Citrus sinensis) and sweet basil (Ocimum basilicum L.) essential oils. Biomedica 2012, 32, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Franco-Vega, A.; Reyes-Jurado, F.; Cardoso-Ugarte, G.A.; Sosa-Morales, M.E.; Palou, E.; Lopez-Malo, A. Sweet Orange (Citrus Sinensis) Oils; Elsevier Inc.: New York, NY, USA, 2015; ISBN 9780124166448. [Google Scholar]
- Settani, L.; Palazzolo, E.; Guarrasi, V.; Aleo, A.; Mammina, C.; Moschetti, G.; Germaná, M. Inhibition of foodborne pathogen bacteria by essential oils extracted from citrus fruits cultivated in Sicily. Food Control 2012, 26, 326–330. [Google Scholar] [CrossRef]
- Lin, C.M.; Sheu, S.R.; Hsu, S.C.; Tsai, Y.H. Determination of bactericidal efficacy of essential oil extracted from orange peel on the food contact surfaces. Food Control 2010, 21, 1710–1715. [Google Scholar] [CrossRef]
- Bourgou, S.; Zohra, F.; Ourghemmi, I.; Saidani, M. Changes of peel essential oil composition of four Tunisian citrus during fruit maturation. Sci. World J. 2012, 2012, 528593. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Shukla, R.; Prakash, B.; Kumar, A.; Singh, S.; Mishra, P.K.; Dubey, N.K. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, dl-limonene. Food Chem. Toxicol. 2010, 48, 1734–1740. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Tripathi, A. Effects of Citrus sinensis (L.) Osbeck epicarp essential oil on growth and morphogenesis of Aspergillus niger (L.) Van Tieghem. Microbiol. Res. 2008, 163, 337–344. [Google Scholar] [CrossRef] [PubMed]
- El-Akhal, F.; Lalamia, A.E.O.; Guemmouh, R. Larvicidal activity of essential oils of Citrus sinensis and Citrus aurantium (Rutaceae) cultivated in Morocco against the malaria vector Anopheles labranchiae (Diptera: Culicidae). Asian Pac. J. Trop. Dis. 2015, 5, 458–462. [Google Scholar] [CrossRef]
- Galvão, J.G.; Silva, V.F.; Ferreira, S.G.; França, F.R.M.; Santos, D.A.; Freitas, L.S.; Alves, P.B.; Araújo, A.A.S.; Cavalcanti, S.C.H.; Nunes, R.S. β-Cyclodextrin inclusion complexes containing Citrus sinensis (L.) Osbeck essential oil: An alternative to control Aedes aegypti larvae. Thermochim. Acta 2015, 608, 14–19. [Google Scholar] [CrossRef]
- Rossi, Y.E.; Palacios, S.M. Fumigant toxicity of Citrus sinensis essential oil on Musca domestica L. adults in the absence and presence of a P450 inhibitor. Acta Trop. 2013, 127, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Ezeonu, F.C.; Chidume, G.I.; Udedi, S.C. Insecticidal properties of volatile extracts of orange peels. Bioresour. Technol. 2001, 76, 273–274. [Google Scholar] [CrossRef]
- Raina, A.; Bland, J.; Doolittle, M.; Lax, A.; Folkins, M.; Raina, A.; Bland, J.; Doolittle, M.; Lax, A.; Boopathy, R.A.J.; et al. Effect of orange oil extract on the Formosan subterranean termite (Isoptera: Rhinotermitidae). J. Econ. Entomol. 2007, 100, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Gaínza, Y.A.; Domingues, L.F.; Perez, O.P.; Rabelo, M.D.; López, E.R.; de Souza Chagas, A.C. Anthelmintic activity in vitro of Citrus sinensis and Melaleuca quinquenervia essential oil from Cuba on Haemonchus contortus. Ind. Crop. Prod. 2015, 76, 647–652. [Google Scholar] [CrossRef]
- Acar, U.; Kesbiç, O.S.; Yilmaz, S.; Gültepe, N.; Türker, A. Evaluation of the effects of essential oil extracted from sweet orange peel (Citrus sinensis) on growth rate of tilapia (Oreochromis mossambicus) and possible disease resistance against Streptococcus iniae. Aquaculture 2015, 437, 282–286. [Google Scholar] [CrossRef]
- Anwar, S.; Ahmed, N.; Speciale, A.; Cimino, F.; Saija, A. Bitter Orange (Citrus Aurantium L.) Oils; Elsevier Inc.: New York, NY, USA, 2015; ISBN 9780124166448. [Google Scholar]
- Carvalho-Freitas, M.I.R.; Costa, M. Anxiolytic and sedative effects of extracts and essential oil from Citrus aurantium L. Biol. Pharm. Bull. 2002, 25, 1629–1633. [Google Scholar] [CrossRef] [PubMed]
- De Moraes Pultrini, A.; Almeida Galindo, L.; Costa, M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci. 2006, 78, 1720–1725. [Google Scholar] [CrossRef] [PubMed]
- Pimenta, F.C.F.; Alves, M.F.; Pimenta, M.B.F.; Melo, S.A.L.; de Almeida, A.A.F.; Leite, J.R.; Pordeus, L.C.D.M.; Diniz, M.D.F.F.M.; de Almeida, R.N. Anxiolytic effect of Citrus aurantium L. on patients with chronic myeloid leukemia. Phyther. Res. 2016, 30, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.A.R.A.; Cury, T.C.; Cassettari, B.O.; Takahira, R.K.; Florio, J.C.; Costa, M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. BMC Complement. Altern. Med. 2013, 13, 42. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.-T.; Xu, H.-L.; Feng, J.; Zhan, X.; Zhou, L.-P.; Cui, C.-C. Involvement of monoaminergic systems in the antidepressant like effect of nobiletin. Physiol. Behav. 2011, 102, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Namazi, M.; Ali Akbari, S.A.; Mojab, F.; Talebi, A.; Majd, H.A.; Jannesari, S. Effects of Citrus aurantium (bitter orange) on the severity of first-stage labor pain. Iran. J. Pharm. Res. 2014, 13, 1011–1018. [Google Scholar] [PubMed]
- Moraes, T.M.; Kushima, H.; Moleiro, F.C.; Santos, R.C.; Machado Rocha, L.R.; Marques, M.O.; Vilegas, W.; Hiruma-Lima, C.A. Effects of limonene and essential oil from Citrus aurantium on gastric mucosa: Role of prostaglandins and gastric mucus secretion. Chem. Biol. Interact. 2009, 180, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.X.; Zhao, S.P.; Zhang, J.Y.; Zhang, H.; Xie, Z.H.; Cai, G.M.; Jiang, W.H. Effect of orange peel essential oil on oxidative stress in AOM animals. Int. J. Biol. Macromol. 2012, 50, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Ullah, N.; Khan, M.A.; Khan, T.; Ahmad, W. Nephroprotective potentials of Citrus aurantium: A prospective pharmacological study on experimental models. Pak. J. Pharm. Sci. 2014, 27, 505–510. [Google Scholar] [PubMed]
- Friedman, M.; Henika, P.R.; Levin, C.E.; Mandrell, R.E. Antibacterial activities of plant essential oils and their components against Escherichia coli O157:H7 and Salmonella enterica in apple juice. J. Agric. Food Chem. 2004, 52, 6042–6048. [Google Scholar] [CrossRef] [PubMed]
- Iturriaga, L.; Olabarrieta, I.; de Marañón, I.M. Antimicrobial assays of natural extracts and their inhibitory effect against Listeria innocua and fish spoilage bacteria, after incorporation into biopolymer edible films. Int. J. Food Microbiol. 2012, 158, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.A.C.; Zambrana, J.R.M.; di Iorio, F.B.R.; Pereira, C.A.; Jorge, A.O.C. The antimicrobial effects of Citrus limonum and Citrus aurantium essential oils on multi-species biofilms. Braz. Oral Res. 2014, 28, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, W.; Mourad, B.; Ibrahim, S.; Sonbol, F. Oil of bitter orange: New topical antifungal agent. Int. J. Dermatol. 1996, 35, 448–449. [Google Scholar] [CrossRef] [PubMed]
- Zarrad, K.; Hamouda, A.B.; Chaieb, I.; Laarif, A.; Jemâa, J.M. Ben Chemical composition, fumigant and anti-acetylcholinesterase activity of the Tunisian Citrus aurantium L. essential oils. Ind. Crop. Prod. 2015, 76, 121–127. [Google Scholar] [CrossRef]
- Battaglia, S. The Complete Guide to Aromatherapy Brisbane; The International Centre of Holistic Aromatherapy: Brisbane, Australia, 2003.
- Akhlaghi, M.; Shabanian, G.; Rafieian-Kopaei, M.; Parvin, N.; Saadat, M.; Akhlaghi, M. Citrus aurantium blossom and preoperative anxiety. Rev. Bras. Anestesiol. 2011, 61, 702–712. [Google Scholar] [CrossRef]
- Farshbaf-Khalili, A.; Kamalifard, M.; Namadian, M. Comparison of the effect of lavender and bitter orange on anxiety in postmenopausal women: A triple-blind, randomized, controlled clinical trial. Complement. Ther. Clin. Pract. 2018, 31, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Azanchi, T.; Shafaroodi, H.; Asgarpanah, J. Anticonvulsant activity of Citrus aurantium blossom essential oil (neroli): Involvment of the GABAergic system. Nat. Prod. Commun. 2014, 9, 1615–1618. [Google Scholar] [PubMed]
- Rahnama, S.; Rabiei, Z.; Alibabaei, Z.; Mokhtari, S.; Rafieian-Kopaei, M.; Deris, F. Antiamnesic activity of Citrus aurantium flowers extract against scopolamine-induced memory impairments in rats. Neurol. Sci. 2014, 36, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Khodabakhsh, P.; Shafaroodi, H.; Asgarpanah, J. Analgesic and anti-inflammatory activities of Citrus aurantium L. blossoms essential oil (neroli): Involvement of the nitric oxide/cyclic-guanosine monophosphate pathway. J. Nat. Med. 2015, 69, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Kang, P.; Lee, H.S.; Seol, G.H. Effects of inhalation of essential oil of Citrus aurantium L. var. amara on menopausal symptoms, stress, and estrogen in postmenopausal women: A randomized controlled trial. Evid.-Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Heydari, N.; Abootalebi, M.; Jamalimoghadam, N.; Kasraeian, M.; Emamghoreishi, M.; Akbarzade, M. Investigation of the effect of aromatherapy with Citrus aurantium blossom essential oil on premenstrual syndrome in university students: A clinical trial study. Complement. Ther. Clin. Pract. 2018, 32, 1–5. [Google Scholar] [CrossRef]
- Kang, P.; Ryu, K.H.; Lee, J.M.; Kim, H.K.; Seol, G.H. Endothelium- and smooth muscle-dependent vasodilator effects of Citrus aurantium L. var. amara: Focus on Ca2+ modulation. Biomed. Pharmacother. 2016, 82, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.H.; Kim, C.; Seong, K.; Hur, M.H.; Lim, H.M.; Lee, M.S. Essential oil inhalation on blood pressure and salivary cortisol levels in prehypertensive and hypertensive subjects. Evid.-Based Complement. Altern. Med. 2012, 2012, 984203. [Google Scholar] [CrossRef] [PubMed]
- Ao, Y.; Satoh, K.; Shibano, K.; Kawahito, Y.; Shioda, S. Singlet oxygen scavenging activity and cytotoxicity of essential oils from Rutaceae. J. Clin. Biochem. Nutr. 2008, 43, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Ben Hsouna, A.; Hamdi, N.; Ben Halima, N.; Abdelkafi, S. Characterization of essential oil from Citrus aurantium L. flowers: Antimicrobial and antioxidant activities. J. Oleo Sci. 2013, 62, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Sarrou, E.; Chatzopoulou, P.; Dimassi-Theriou, K.; Therios, I. Volatile constituents and antioxidant activity of peel, flowers and leaf oils of Citrus aurantium L. growing in Greece. Molecules 2013, 18, 10639–10647. [Google Scholar] [CrossRef] [PubMed]
- Ellouze, I.; Abderrabba, M.; Sabaou, N.; Mathieu, F.; Lebrihi, A.; Bouajila, J. Season’s variation impact on Citrus aurantium leaves essential oil: Chemical composition and biological activities. J. Food Sci. 2012, 77, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.M.; Zhao, Y.; He, C.C.; Li, J.X. Preventive effects of Citrus reticulata essential oil on bleomycin-induced pulmonary fibrosis in rats and the mechanism. J. Chin. Integr. Med. 2012, 10, 200–209. [Google Scholar] [CrossRef]
- Yi, F.; Jin, R.; Sun, J.; Ma, B.; Bao, X. Evaluation of mechanical-pressed essential oil from Nanfeng mandarin (Citrus reticulata Blanco cv. Kinokuni) as a food preservative based on antimicrobial and antioxidant activities. LWT Food Sci. Technol. 2018, 95, 346–353. [Google Scholar] [CrossRef]
- Tao, N.; Jia, L.; Zhou, H. Anti-fungal activity of Citrus reticulata Blanco essential oil against Penicillium italicum and Penicillium digitatum. Food Chem. 2014, 153, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Matan, N.; Matan, N. Antifungal activities of anise oil, lime oil, and tangerine oil against molds on rubberwood (Hevea brasiliensis). Int. Biodeterior. Biodegrad. 2008, 62, 75–78. [Google Scholar] [CrossRef]
- Wu, T.; Cheng, D.; He, M.; Pan, S.; Yao, X.; Xu, X. Antifungal action and inhibitory mechanism of polymethoxylated flavones from Citrus reticulata Blanco peel against Aspergillus niger. Food Control 2014, 35, 354–359. [Google Scholar] [CrossRef]
- Chutia, M.; Deka Bhuyan, P.; Pathak, M.G.; Sarma, T.C.; Boruah, P. Antifungal activity and chemical composition of Citrus reticulata Blanco essential oil against phytopathogens from North East India. LWT Food Sci. Technol. 2009, 42, 777–780. [Google Scholar] [CrossRef]
- Komiya, M.; Takeuchi, T.; Harada, E. Lemon oil vapor causes an anti-stress effect via modulating the 5-HT and DA activities in mice. Behav. Brain Res. 2006, 172, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Ogeturk, M.; Kose, E.; Sarsilmaz, M.; Akpinar, B.; Kus, I.; Meydan, S. Effects of lemon essential oil aroma on the learning behaviors of rats. Neurosciences 2010, 15, 292–293. [Google Scholar] [PubMed]
- Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of ten essential oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 cancer cells. Molecules 2010, 15, 3200–3210. [Google Scholar] [CrossRef] [PubMed]
- Wattenberg, L.; Coccia, J.B. Inhibition of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone carcinogenesis in mice by D-limonene and citrus fruit oils. Carcinogenesis 1991, 12, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Niijima, A.; Nagai, K. Effect of Olfactory stimulation with flavor of grapefruit oil and lemon oil on the activity of sympathetic branch in the white adipose tissue of the epididymis. Exp. Biol. Med. 2003, 228, 1190–1192. [Google Scholar] [CrossRef]
- Campêlo, L.M.; Moura Gonçalves, F.C.; Feitosa, C.M.; de Freitas, R.M. Antioxidant activity of Citrus limon essential oil in mouse hippocampus. Pharm. Biol. 2011, 49, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.-S.; Song, H.S.; Ukeda, H.; Sawamura, M. Radical-scavenging activities of citrus essential oils and their components: Detection using 1,1-diphenyl-2-picrylhydrazyl. J. Agric. Food Chem. 2000, 48, 4156–4161. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, R.M.; Campêlo, L.M.L.; de Almeida, A.A.C.; de Freitas, R.L.M.; Cerqueira, G.S.; de Sousa, G.F.; Saldanha, G.B.; Feitosa, C.M. Antioxidant and antinociceptive effects of Citrus limon essential oil in mice. J. Biomed. Biotechnol. 2011. [Google Scholar] [CrossRef]
- Ceccarelli, I.; Lariviere, W.R.; Fiorenzani, P.; Sacerdote, P.; Aloisi, A.M. Effects of long-term exposure of lemon essential oil odor on behavioral, hormonal and neuronal parameters in male and female rats. Brain Res. 2004, 1001, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, I.; Masi, F.; Fiorenzani, P.; Aloisi, A.M. Sex differences in the citrus lemon essential oil-induced increase of hippocampal acetylcholine release in rats exposed to a persistent painful stimulation. Neurosci. Lett. 2002, 330, 25–28. [Google Scholar] [CrossRef]
- Ikeda, H.; Takasu, S.; Murase, K. Contribution of anterior cingulate cortex and descending pain inhibitory system to analgesic effect of lemon odor in mice. Mol. Pain 2014, 10, 14. [Google Scholar] [CrossRef] [PubMed]
- Yavari Kia, P.; Safajou, F.; Shahnazi, M.; Nazemiyeh, H. The effect of lemon inhalation aromatherapy on nausea and vomiting of pregnancy: A double-blinded, randomized, controlled clinical trial. Iran. Red Crescent Med. J. 2014, 16. [Google Scholar] [CrossRef] [PubMed]
- Akpinar, B. The effects of olfactory stimuli on scholastic performance. Ir. J. Educ. 2005, 36, 86–90. [Google Scholar]
- Valgimigli, L.; Gabbanini, S.; Berlini, E.; Lucchi, E.; Beltramini, C.; Bertarelli, Y.L. Lemon (Citrus limon, Burm.f.) essential oil enhances the trans-epidermal release of lipid-(A, E) and water-(B6, C) soluble vitamins from topical emulsions in reconstructed human epidermis. Int. J. Cosmet. Sci. 2012, 34, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Perez-Álvarez, J. Antibacterial activity of lemon (Citrus limon L.), mandarin (Citrus reticulata L.), grapefruit (Citrus paradisi L.) and orange (Citrus sinensis L.) essential oils. J. Food Saf. 2008, 28, 567–576. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Mohamady, M.A.; Fernández-López, J.; Abd ElRazik, K.A.; Omer, E.A.; Pérez-Alvarez, J.A.; Sendra, E. In vitro antioxidant and antibacterial activities of essentials oils obtained from Egyptian aromatic plants. Food Control 2011, 22, 1715–1722. [Google Scholar] [CrossRef]
- Oshaghi, M.A.; Ghalandari, R.; Vatandoost, H.; Shayeghi, M.; Abolhassani, M.; Hashemzadeh, M. Repellent effect of extracts and essential oils of Citrus limon (Rutaceae) and Melissa officinalis (Labiatae) against main malaria vector, Anopheles stephensi (Diptera: Culicidae). Iran. J. Public Health 2003, 32, 47–52. [Google Scholar]
- Aboelhadid, S.M.; Mahrous, L.N.; Hashem, S.A.; Abdel-Kafy, E.-S.M.; Miller, R.J. In vitro and in vivo effect of Citrus limon essential oil against sarcoptic mange in rabbits. Parasitol. Res. 2016, 115, 3013–3020. [Google Scholar] [CrossRef] [PubMed]
- Asnaashari, S.; Delazar, A.; Habibi, B.; VasÒ, R.; Nahar, L.; Hamedeyazdan, S.; Sarker, S.D. Essential oil from Citrus aurantifolia prevents ketotifen-induced weight-gain in mice. Phyther. Res. 2010, 24, 1893–1897. [Google Scholar] [CrossRef] [PubMed]
- Shafreen, R.B.; Lubinska, M.; Różańska, A.; Dymerski, T.; Namieśnik, J.; Katrich, E.; Gorinstein, S. Human serum interactions with phenolic and aroma substances of Kaffir (Citrus hystrix) and Key lime (Citrus aurantifolia) juices. J. Lumin. 2018. [Google Scholar] [CrossRef]
- Spadaro, F.; Costa, R.; Circosta, C.; Occhiuto, F. Volatile composition and biological activity of key lime Citrus aurantifolia essential oil. Nat. Prod. Commun. 2012, 7, 1523–1526. [Google Scholar] [PubMed]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Colica, C.; Menichini, F. Comparative study on the antioxidant capacity and cholinesterase inhibitory activity of Citrus aurantifolia Swingle, C. aurantium L., and C. bergamia Risso and Poit. peel essential oils. J. Food Sci. 2012, 77, H40–H46. [Google Scholar] [CrossRef] [PubMed]
- Amorim, J.L.; Simas, D.L.R.; Pinheiro, M.M.G.; Moreno, D.S.A.; Alviano, C.S.; Da Silva, A.J.R.; Fernandes, P.D. Anti-inflammatory properties and chemical characterization of the essential oils of four Citrus species. PLoS ONE 2016, 11, e0153643. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Valenzuela, M.R.; Tapia-Rodriguez, M.R.; Vazquez-Armenta, F.J.; Silva-Espinoza, B.A.; Ayala-Zavala, J.F. Lime (Citrus aurantifolia) Oils; Elsevier Inc.: New York, NY, USA, 2015; ISBN 9780124166448. [Google Scholar]
- Ruberto, G. Analysis of Volatile Components of Citrus Fruit Essential Oils. In Analysis of Taste and Aroma; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Fagodia, S.K.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Phytotoxicity and cytotoxicity of Citrus aurantiifolia essential oil and its major constituents: Limonene and citral. Ind. Crop. Prod. 2017, 108, 708–715. [Google Scholar] [CrossRef]
- Fouad, H.A.; da Camara, C.A.G. Chemical composition and bioactivity of peel oils from Citrus aurantiifolia and Citrus reticulata and enantiomers of their major constituent against Sitophilus zeamais (Coleoptera: Curculionidae). J. Stored Prod. Res. 2017, 73, 30–36. [Google Scholar] [CrossRef]
- Nagai, K.; Niijima, A.; Horii, Y.; Shen, J.; Tanida, M. Olfactory stimulatory with grapefruit and lavender oils change autonomic nerve activity and physiological function. Auton. Neurosci. Basic Clin. 2014, 185, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Stiles, K.G. The Essential Oils Complete Reference Guide: Over 250 Recipes for Natural Wholesome Aromatherapy; Page Street Publishing: Salem, MA, USA, 2017; ISBN 1624143067. [Google Scholar]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer Science & Business Media: New York, NY, USA, 2012; Volume 4, ISBN 978-94-007-4052-5. [Google Scholar]
- Okunowo, W.O.; Oyedeji, O.; Afolabi, L.O.; Matanmi, E. Essential oil of grape fruit (Citrus paradisi) peels and its antimicrobial activities. Am. J. Plant Sci. 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Churata-Oroya, D.E.; Ramos-Perfecto, D.; Moromi-Nakata, H.; Martínez-Cadillo, E.; Castro-Luna, A.; Garcia-de-la-Guarda, R. Antifungal effect of Citrus paradisi “grapefruit”on strains of Candida albicans isolated from patients with denture stomatitis. Rev. Estomatol. Hered. 2016, 26, 78–84. [Google Scholar] [CrossRef]
- Tirillini, B. Grapefruit: The last decade acquisitions. Fitoterapia 2000, 71. [Google Scholar] [CrossRef]
- Ruiz, M.J.; Juaìrez, M.L.; Alzogaray, R.A.; Arrighi, F.; Arroyo, L.; Gastaminza, G.; Willink, E.; del Valle Bardoìn, A.; Vera, T. Toxic effect of citrus peel constituents on Anastrepha fraterculus Wiedemann and Ceratitis capitata Wiedemann immature stages. J. Agric. Food Chem. 2014, 62, 10084–10091. [Google Scholar] [CrossRef] [PubMed]
- Ivoke, N.; Ogbonna, P.C.; Ekeh, F.N.; Ezenwaji, N.E.; Atama, C.I.; Ejere, V.C.; Onoja, U.S.; Eyo, J.E. Effects of grapefruit (Citrus paradisi MACF) (Rutaceae) peel oil against developmental stages of Aedes aegypti (Diptera: Culicidae). Southeast Asian J. Trop. Med. Public Health 2013, 44, 970–978. [Google Scholar] [PubMed]
- Sanei-Dehkord, A.; Sedaghat, M.M.; Vatandoost, H.; Abai, M.R. Chemical compositions of the peel essential oil of Citrus aurantium and its natural larvicidal activity against the malaria vector Anopheles stephensi (Diptera: Culicidae) in comparison with Citrus paradisi. J. Arthropod Borne Dis. 2016, 10, 577–585. [Google Scholar]
- Pérez, A.; Alcala, Y.; Salem, A.Z.M.; Alberti, A.B. Anticoccidial efficacy of naringenin and a grapefruit peel extract in growing lambs naturally-infected with Eimeria spp. Vet. Parasitol. 2016, 232, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Russo, R.; Cassiano, M.G.V.; Ciociaro, A.; Adornetto, A.; Varano, G.P.; Chiappini, C.; Berliocchi, L.; Tassorelli, C.; Bagetta, G.; Corasaniti, M.T. Role of d-limonene in autophagy induced by bergamot essential oil in SH-SY5Y neuroblastoma cells. PLoS ONE 2014, 9, e0113682. [Google Scholar] [CrossRef] [PubMed]
- Moysan, A.; Morlière, P.; Averbeck, D.; Dubertret, L. Evaluation of phototoxic and photogenotoxic risk associated with the use of photosensitizers in suntan preparations: Application to tanning preparations containing bergamot oil. Skin Pharmacol. Physiol. 1993, 6, 282–291. [Google Scholar] [CrossRef]
- Rombolà, L.; Amantea, D.; Russo, R.; Adornetto, A.; Berliocchi, L.; Tridico, L.; Corasaniti, M.; Sakurada, S.; Sakurada, T.; Bagetta, G.; et al. Rational basis for the use of bergamot essential oil in complementary medicine to treat chronic pain. Mini-Rev. Med. Chem. 2016, 16, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Lauro, F.; Ilari, S.; Giancotti, L.A.; Morabito, C.; Malafoglia, V.; Gliozzi, M.; Palma, E.; Salvemini, D.; Muscoli, C. The protective role of bergamot polyphenolic fraction on several animal models of pain. PharmaNutrition 2016, 4, S35–S40. [Google Scholar] [CrossRef]
- Sakurada, T.; Mizoguchi, H.; Kuwahata, H.; Katsuyama, S.; Komatsu, T.; Morrone, L.A.; Corasaniti, M.T.; Bagetta, G.; Sakurada, S. Intraplantar injection of bergamot essential oil induces peripheral antinociception mediated by opioid mechanism. Pharmacol. Biochem. Behav. 2011, 97, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Katsuyama, S.K.; Towa, A.O.; Amio, S.K.; Ato, K.S.; Agi, T.Y.; Ishikawa, Y.K.; Omatsu, T.K.; Agetta, G.B.; Akurada, T.S.; Akamura, H.N. Effect of plantar subcutaneous administration of bergamot essential oil and linalool on formalin-induced nociceptive behavior in mice. Biomed. Res. 2015, 36, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Kuwahata, H.; Komatsu, T.; Katsuyama, S.; Corasaniti, M.T.; Bagetta, G.; Sakurada, S.; Sakurada, T.; Takahama, K. Peripherally injected linalool and bergamot essential oil attenuate mechanical allodynia via inhibiting spinal ERK phosphorylation. Pharmacol. Biochem. Behav. 2013, 103, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, M.; Luini, A.; Bombelli, R.; Corasaniti, M.T.; Bagetta, G.; Marino, F. The essential oil of bergamot stimulates reactive oxygen species production in human polymorphonuclear leukocytes. Phyther. Res. 2014, 28, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Navarra, M.; Ferlazzo, N.; Cirmi, S.; Trapasso, E.; Bramanti, P.; Lombardo, G.E.; Minciullo, P.L.; Calapai, G.; Gangemi, S. Effects of bergamot essential oil and its extractive fractions on SH-SY5Y human neuroblastoma cell growth. J. Pharm. Pharmacol. 2015, 67, 1042–1053. [Google Scholar] [CrossRef] [PubMed]
- Berliocchi, L.; Ciociaro, A.; Russo, R.; Cassiano, M.G.V.; Blandini, F.; Rotiroti, D.; Morrone, L.A.; Corasaniti, M.T. Toxic profile of bergamot essential oil on survival and proliferation of SH-SY5Y neuroblastoma cells. Food Chem. Toxicol. 2011, 49, 2780–2792. [Google Scholar] [CrossRef] [PubMed]
- Celia, C.; Trapasso, E.; Locatelli, M.; Navarra, M.; Ventura, C.A.; Wolfram, J.; Carafa, M.; Morittu, V.M.; Britti, D.; Di Marzio, L.; et al. Anticancer activity of liposomal bergamot essential oil (BEO) on human neuroblastoma cells. Colloids Surf. B Biointerfaces 2013, 112, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Sawamura, M. Citrus Essential Oils: Flavor and Fragrance; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Bagetta, G.; Morrone, L.A.; Rombolà, L.; Amantea, D.; Russo, R.; Berliocchi, L.; Sakurada, S.; Sakurada, T.; Rotiroti, D.; Corasaniti, M.T. Neuropharmacology of the essential oil of bergamot. Fitoterapia 2010, 81, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Amantea, D.; Fratto, V.; Maida, S.; Rotiroti, D.; Ragusa, S.; Corasaniti, M.T. Prevention of glutamate accumulation and upregulation of phospho-Akt may account for neuroprotection afforded by bergamot essential oil against brain injury induced by focal cerebral ischemia in rat. Int. Rev. Neurobiol. 2009, 85, 389–405. [Google Scholar] [PubMed]
- Saiyudthong, S.; Mekseepralard, C. Effect of Inhaling bergamot oil on depression-related behaviors in chronic stressed rats. J. Med. Assoc. Thail. 2015, 98, S152–S159. [Google Scholar]
- Saiyudthong, S.; Marsden, C.A. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phyther. Res. 2011, 25, 858–862. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Gibson, J.; Eggett, D.L.; Parker, T.L. Bergamot (Citrus bergamia) essential oil inhalation improves positive feelings in the waiting room of a mental health treatment center: A pilot study. Phyther. Res. 2017, 31, 812–816. [Google Scholar] [CrossRef] [PubMed]
- Avila-Sosa, R.; Navarro-Cruz, A.R.; Sosa-Morales, M.E.; López-Malo, A.; Palou, E. Bergamot (Citrus Bergamia) Oils; Elsevier Inc.: New York, NY, USA, 2015; ISBN 9780124166448. [Google Scholar]
- Kirbaslar, F.G.; Tavman, A.; Dülger, B.; Türker, G. Antimicrobial activity of Turkish citrus peel oils. Pak. J. Bot. 2009, 41, 3207–3212. [Google Scholar]
- Fisher, K.; Phillips, C.A. The effect of lemon, orange and bergamot essential oils and their components on the survival of Campylobacter jejuni, Escherichia coli O157, Listeria monocytogenes, Bacillus cereus and Staphylococcus aureus in vitro and in food systems. J. Appl. Microbiol. 2006, 101, 1232–1240. [Google Scholar] [CrossRef] [PubMed]
- Stević, T.; Berić, T.; Šavikin, K.; Soković, M.; Gođevac, D.; Dimkić, I.; Stanković, S. Antifungal activity of selected essential oils against fungi isolated from medicinal plant. Ind. Crop. Prod. 2014, 55, 116–122. [Google Scholar] [CrossRef]
- Sanguinetti, M.; Posteraro, B.; Romano, L.; Battaglia, F.; Lopizzo, T.; De Carolis, E.; Fadda, G. In vitro activity of Citrus bergamia (bergamot) oil against clinical isolates of dermatophytes. J. Antimicrob. Chemother. 2007, 59, 305–308. [Google Scholar] [CrossRef] [PubMed]
- El-Ashmawy, W.R.; Elsaeed, M.; Gebely, M. Randomized clinical trial on evaluation of the effect of bergamot oil on treatment of ring worm infection in calves and cats. Int. J. Infect. Dis. 2016, 45, 312–313. [Google Scholar] [CrossRef]
- Furneri, P.M.; Mondello, L.; Mandalari, G.; Paolino, D.; Dugo, P.; Garozzo, A.; Bisignano, G. In vitro antimycoplasmal activity of Citrus bergamia essential oil and its major components. Eur. J. Med. Chem. 2012, 52, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Sawamura, M.; Wu, Y.; Fujiwara, C.; Urushibata, M. Inhibitory effect of yuzu essential oil on the formation of N-nitrosodimethylamine in vegetables. J. Agric. Food Chem. 2005, 53, 4281–4287. [Google Scholar] [CrossRef] [PubMed]
- Hirota, R.; Roger, N.N.; Nakamura, H.; Song, H.S.; Sawamura, M.; Suganuma, N. Anti-inflammatory effects of limonene from yuzu (Citrus junos Tanaka) essential oil on eosinophils. J. Food Sci. 2010, 75, 20492298. [Google Scholar] [CrossRef] [PubMed]
- Ueki, S.; Niinomi, K.; Takashima, Y.; Kimura, R.; Komai, K.; Murakami, K.; Fujiwara, C. Effectiveness of aromatherapy in decreasing maternal anxiety for a sick child undergoing infusion in a paediatric clinic. Complement. Ther. Med. 2014, 22, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kimura, T.; Hayashi, T. Aromatic effects of a Japanese citrus fruit-yuzu (Citrus junos Sieb. ex Tanaka)-on psychoemotional states and autonomic nervous system activity during the menstrual cycle: A single-blind randomized controlled crossover study. Biopsychosoc. Med. 2016, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kimura, T.; Hayashi, T. Does Japanese citrus fruit yuzu (Citrus junos Sieb. ex Tanaka) fragrance have lavender-like therapeutic effects that alleviate premenstrual emotional symptoms? A single-blind randomized crossover Study. J. Altern. Complement. Med. 2017, 23, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, K.; Takahashi, E.; Nishibori, K. Suppressing effect of yuzu peel on the odor of Niboshi soup stock. Bull. Jpn. Soc. Sci. Fish. 1993, 59, 673–675. [Google Scholar] [CrossRef]
- Kim, S.H.; Shin, E.J.; Hur, H.J.; Park, J.H.; Sung, M.J.; Kwon, D.Y.; Hwang, J.T. Citrus junos Tanaka peel extract attenuates experimental colitis and inhibits tumour growth in a mouse xenograft model. J. Funct. Foods 2014, 8, 301–308. [Google Scholar] [CrossRef]
- Hwang, J.T.; Shin, E.J. Ethanol extract of Citrus junos Tanaka exerts hypocholesterolemic effect in mice fed a high cholesterol diet. Atherosclerosis 2013, 241, e195. [Google Scholar] [CrossRef]
- Kim, S.H.; Hur, H.J.; Yang, H.J.; Kim, H.J.; Kim, M.J.; Park, J.H.; Sung, M.J.; Kim, M.S.; Kwon, D.Y.; Hwang, J.T. Citrus junos Tanaka peel extract exerts antidiabetic effects via AMPK and PPAR-γboth in vitro and in vivo in mice fed a high-fat diet. Evid.-Based Complement. Altern. Med. 2013, 2013, 921012. [Google Scholar]
- Zang, L.; Shimada, Y.; Kawajiri, J.; Tanaka, T.; Nishimura, N. Effects of yuzu (Citrus junos Siebold ex Tanaka) peel on the diet-induced obesity in a zebrafish model. J. Funct. Foods 2014, 10, 499–510. [Google Scholar] [CrossRef]
- Yu, H.Y.; Park, S.W.; Chung, I.M.; Jung, Y.S. Anti-platelet effects of yuzu extract and its component. Food Chem. Toxicol. 2011, 49, 3018–3024. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Ahn, J.H.; Park, S.W.; Jung, Y.-S. Preventive effect of yuzu and hesperidin on left ventricular remodeling and dysfunction in rat permanent left anterior descending coronary artery occlusion model. PLoS ONE 2015, 10, e110596. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasha, G.; Murthy, K.C.; Demarais, R.; Patil, B. Inhibition of prostate cancer (LNCaP) cell proliferation by volatile components from Nagami kumquats. Planta Med. 2012, 78, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Nouri, A.; Shafaghatlonbar, A. Chemical constituents and antioxidant activity of essential oil and organic extract from the peel and kernel parts of Citrus japonica Thunb. (kumquat) from Iran. Nat. Prod. Res. 2016, 30, 1093–1097. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Zeng, W.C.; Xu, P.Y.; Lan, Y.J.; Zhu, R.X.; Zhong, K.; Huang, Y.N.; Gao, H. Chemical composition and antimicrobial activity of the essential oil of kumquat (Fortunella crassifolia Swingle) peel. Int. J. Mol. Sci. 2012, 13, 3382–3393. [Google Scholar] [CrossRef] [PubMed]
- Md Othman, S.; Hassan, M.; Nahar, L.; Basar, N.; Jamil, S.; Sarker, S. Essential Oils from the Malaysian Citrus (Rutaceae) medicinal plants. Medicines 2016, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Morrone, L.A.; Rombolà, L.; Pelle, C.; Corasaniti, M.T.; Zappettini, S.; Paudice, P.; Bonanno, G.; Bagetta, G. The essential oil of bergamot enhances the levels of amino acid neurotransmitters in the hippocampus of rat: Implication of monoterpene hydrocarbons. Pharmacol. Res. 2007, 55, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Volpato, G.T.; Francia-Farje, L.A.D.; Damasceno, D.C.; Renata, V.O.; Clélia, A.H.-L.; Wilma, G.K. Effect of essential oil from Citrus aurantium in maternal reproductive outcome and fetal anomaly frequency in rats. An. Acad. Bras. Ciênc. 2015, 87, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Opdyke, D.L.J. Monographs on fragrance raw materials. Food Cosmet. Toxicol. 1974, 12, 807–1016. [Google Scholar] [CrossRef]
- Naganuma, M.; Hirose, S.; Nakayama, Y.; Nakajima, K.; Someya, T. A study of the phototoxicity of lemon oil. Arch. Dermatol. Res. 1985, 278, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Rudzki, E.; Grzywa, Z.; Bruo, W.S. Sensitivity to 35 essential oils. Contact Dermat. 1976, 2, 196–200. [Google Scholar] [CrossRef]
- Santucci, B.; Cristaudo, A.; Cannistraci, C.; Picardo, M. Contact dermatitis to fragrances. Contact Dermat. 1987, 16, 93–95. [Google Scholar] [CrossRef]
- Menenghini, C.L.; Rantuccio, F.; Lomuto, M. Additives, vehicles and active drugs of topical medicaments as causes of delayed-type allergic dermatitis. Dermatologica 1971, 143, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Young, A.R.; Walker, S.L.; Kinley, J.S.; Plastow, S.R.; Averbeck, D.; Morlière, P.; Dubertret, L. Phototumorigenesis studies of 5-methoxypsoralen in bergamot oil: Evaluation and modification of risk of human use in an albino mouse skin model. J. Photochem. Photobiol. B 1990, 7, 231–250. [Google Scholar] [CrossRef]
- Opdyke, D.L.S. Fragrance raw materials Monographs. Food Cosmet. Toxicol. 1973, 11, 873–874. [Google Scholar] [CrossRef]
- Bouhlal, K.; Meynadier, J.; Peyron, J.L.; Meynadier, J.; Peyron, L.; Senaux, M.S. The cutaneous effects of the common concretes and absolutes used in the perfume industry. In The Antimicrobial/Biological Activity of Essential Oils; Lawrence, B.M., Ed.; Allured: Carol Stream, IL, USA, 2005; pp. 10–23. [Google Scholar]
- Christensson, J.B.; Forsstrom, P.; Wennberg, A.M.; Karlberg, A.T. Air oxidation increases skin irritation from fragrance terpenes. Contact Dermat. 2009, 60, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Liu, Z.Q. Unusual antioxidant behavior of alpha- and gamma-terpinene in protecting methyl linoleate, DNA, and erythrocyte. J. Agric. Food Chem. 2009, 57, 3943–3948. [Google Scholar] [CrossRef] [PubMed]
- Tisserand, R.; Balacs, T. Essential Oil Safety—A Guide for Health Care Professionals; Harcourt: Glasgow, UK, 1999. [Google Scholar]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological activities of lavender essential oil. Phyther. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.M.; Priestley, C.M.; Burgess, I.F. An investigation and comparison of the bioactivity of selected essential oils on human lice and house dust mites. Fitoterapia 2007, 78, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.K.R.; Maia, J.G.S.; Dosoky, N.S.; Setzer, W.N. Antioxidant, antimicrobial, and cytotoxic properties of Aniba parviflora essential oils from the Amazon. Nat. Prod. Commun. 2016, 11, 1025–1028. [Google Scholar]
- Matura, M.; Skold, M.; Borje, A.; Andersen, K.E.; Bruze, M.; Frosch, P.; Goossens, A.; Johansen, J.D.; Svedman, C.; White, I.R.; et al. Selected oxidized fragrance terpenes are common contact allergens. Contact Dermat. 2005, 52, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Bickers, D.; Calow, P.; Greim, H.; Hanifin, J.M.; Rogers, A.E.; Saurat, J.H.; Sipes, I.G.; Smith, R.L.; Tagami, H. A toxicologic and dermatologic assessment of linalool and related esters when used as fragrance ingredients. Food Chem. Toxicol. 2003, 41, 919–942. [Google Scholar] [CrossRef]
- Jenner, P.M.; Hagan, E.C.; Taylor, J.M.; Cook, E.L.; Fitzhugh, O.G. Food flavorings and compounds of related structure. I. acute oral toxicity. Food Cosmet. Toxicol. 1964, 2, 327–343. [Google Scholar] [CrossRef]
- Letizia, C.S.; Cocchiara, J.; Lalko, J.; Api, A.M. Fragrance material review on linalool. Food Chem. Toxicol. 2003, 41, 943–964. [Google Scholar] [CrossRef]
- Powers, K.A.; Beasley, V.R. Toxicolgical aspects of linalool: A review. Vet. Hum. Toxicol. 1985, 27, 484–486. [Google Scholar] [PubMed]
- Fujii, T.; Furukawa, S.; Suzuki, S. Studies on compounded perfumes for toilet goods. On the non-irritative compounded perfumes for soaps. Yukagaku 1972, 21, 904–908. [Google Scholar]
- Bicas, J.L.; Neri-Numa, I.A.; Ruiz, A.L.; De Carvalho, J.E.; Pastore, G.M. Evaluation of the antioxidant and antiproliferative potential of bioflavors. Food Chem. Toxicol. 2011, 49, 1610–1615. [Google Scholar] [CrossRef] [PubMed]
- Placzek, M.; Frömel, W.; Eberlein, B.; Gilbertz, K.P.; Przybilla, B. Evaluation of phototoxic properties of fragrances. Acta Derm. Venereol. 2007, 87, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.J.; Cruz, M.T.; Tavares, A.C.; Cavaleiro, C.; Lopes, M.C.; Canhoto, J.; Salgueiro, L. Composition and biological activity of the essential oil from Thapsia minor, a new source of geranyl acetate. Ind. Crop. Prod. 2012, 35, 166–171. [Google Scholar] [CrossRef]
- Kakarla, S.; Ganjewala, D. Antimicrobial activity of essential oils of four lemongrass (Cymbopogon flexuosus Steud) varieties. Med. Aromat. Plant Sci. Biotechnol. 2009, 3, 107–109. [Google Scholar]
- Mortelmans, K.; Haworth, S.; Lawlor, T.; Speck, W.; Tainer, B.; Zeiger, E. Salmonella mutagenicity tests: II. Results from the testing of 270 chemicals. Environ. Mutagen. 1986, 8, 1–119. [Google Scholar] [CrossRef] [PubMed]
- Shelby, M.D.; Erexson, G.L.; Hook, G.J.; Tice, R.R. Evaluation of a three-exposure mouse bone marrow micronucleus protocol: Results with 49 chemicals. Environ. Mol. Mutagen. 1993, 21, 160–179. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Chen, F.; Wu, C.; Wang, X.; Chung, H.Y.; Jin, Z. Evaluation of antioxidant activity of Australian tea tree (Melaleuca alternifolia) oil and its components. J. Agric. Food Chem. 2004, 52, 2849–2854. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.L.; Yeung, C.M.; Chiu, L.C.; Cen, Y.Z.; Ooi, V.E. Chemical composition and antiproliferative activity of essential oil from the leaves of a medicinal herb, Schefflera heptaphylla. Phytother. Res. 2009, 23, 140–142. [Google Scholar] [CrossRef] [PubMed]
- Pirila, V.; Siltanen, E.; Pirila, L. On the chemical nature of the eczematogenic agent in oil of turpentine. IV. the primary irritant effect of terpenes. Dermatologica 1964, 128, 16–21. [Google Scholar] [CrossRef]
- Schlede, E.; Aberer, W.; Fuchs, T.; Gerner, I.; Lessmann, H.; Maurer, T.; Rossbacher, R.; Stropp, G.; Wagner, E.; Kayser, D. Chemical substances and contact allergy—244 substances ranked according to allergenic potency. Toxicology 2003, 193, 219–259. [Google Scholar] [CrossRef]
| Citrus EO | Sweet Orange [8,21] | Bitter Orange [8] | Neroli (Egyptian) [8] | Petitgrain [8] | Mandarin [8] | Lemon (D) [8] | Lemon (Ex) [8] | Lime (D) [8,22,23] | Lime (Ex) [8,22] | Bergamot (FCF) [8,24] | Bergamot (Ex) [8,24,25] | Grapefruit [8,26] | Yuzu [8,27] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Plant Part | Fruit Peel | Fruit Peel | Flower | Leaf | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | Fruit Peel | |
| Essential oil Composition | d-Limonene | 83.9–95.9% | 89.7–94.7% | 6.0–10.2% | 0.4–8.0% | 65.3–74.2% | 64.0–70.5% | 56.6–76.0% | 40.4–49.4% | 48.2% | 28.0–45.0% | 27.4–52.0% | 84.8–95.4% | 63.1% |
| Linalool | 0–5.6% | 0.1–2.0% | 43.7–54.3% | 12.3–24.2% | 4.0–20.0% | 1.7–20.6% | 2–8% | |||||||
| Linalyl acetate | 3.5–8.6% | 51.0–71.0% | 18.0–28.0% | 17.1–40.4% | ||||||||||
| β-Pinene | 3.5–5.3% | 0.3–2.7% | 1.4–2.1% | 8.2–14.0% | 6.0–17.0% | 2.0–2.9% | 21.1% | 4.0–11.0% | 4.4–11.0% | 1.1% | ||||
| γ-Terpinene | 16.4–22.7% | 8.4–10.7% | 3.0–13.3% | 9.5–10.7% | 8.1% | 3.0–12.0% | 5.0–11.4% | 12.5% | ||||||
| α-Pinene | 0.6–1.0% | 2.0–2.7% | 1.1–2.1% | 1.3–4.4% | 1.2–2.1% | 2.5% | 1.0–1.8% | 0.7–2.2% | 0.2–1.6% | 2.7% | ||||
| β-Myrcene | 1.3–3.3% | 1.6–2.4% | 1.4–2.1% | 0–2.0% | 1.5–1.8% | 1.4–1.6% | tr–2.2% | 1.3–2.1% | 1.3% | 0.6–1.8% | 1.4–3.6% | 3.2% | ||
| α-Terpineol | 3.9–5.8% | 2.1–5.2% | 0.1–8.0% | 5.4–12.7% | ||||||||||
| (E)-β-Ocimene | 4.6–5.8% | 0.2–2.2% | ||||||||||||
| Sabinene | 0.2–1.0% | 0.4–1.6% | 0.8–1.7% | 0.5–2.4% | 3.1% | 0.4–1.0% | ||||||||
| Neral | 0–1.3% | 0.5–1.5% | 0.4–2.0% | 1.4% | ||||||||||
| Geranial | 0–1.8% | 0.7–2.2% | 0.5–4.3% | 2.4% | ||||||||||
| Bicyclogermacrene | 2.0% | |||||||||||||
| (E)-β-Farnesene | 1.3% | |||||||||||||
| Geranyl acetate | 3.4–4.1% | 1.9–3.4% | ||||||||||||
| Terpinolene | 0.7–1.0% | 8.1–8.7% | ||||||||||||
| (E)-Nerolidol | 1.3–4.0% | |||||||||||||
| Geraniol | 2.8–3.6% | 1.4–2.3% | ||||||||||||
| Nerol | 1.1–1.3% | 0.4–1.1% | ||||||||||||
| p-Cymene | 0.1–1.4% | tr–2.3% | 1.6–2.5% | |||||||||||
| (E,E)-Farnesol | 1.6–3.2% | |||||||||||||
| (E,Z)-Farnesol | ||||||||||||||
| Neryl acetate | 1.7–2.1% | 0–2.6% | 0.1–1.5% | 0.1–1.2% | ||||||||||
| Terpinen-4-ol | tr–1.9% | 0.7–1.9% | ||||||||||||
| (Z)-β-Ocimene | 0.7–1.0% | |||||||||||||
| α-Thujene | 0.7–1.0% | |||||||||||||
| 1,4-Cineole | 2.0–3.0% | |||||||||||||
| Terpinen-1-ol | 1.0–2.3% | |||||||||||||
| (Z)-β-Terpineol | 0.5–2.2% | |||||||||||||
| α-Terpinene | tr–2.1% | |||||||||||||
| β-Bisabolene | 1.6–1.8% | 1.8% | ||||||||||||
| α-Fenchol | 0.6–1.4% | |||||||||||||
| Borneol | 0.5–1.4% | |||||||||||||
| Camphene | 0.5–1.3% | |||||||||||||
| γ-Terpineol | 0.8–1.6% | |||||||||||||
| (E)-α-Bergamotene | 1.1% | |||||||||||||
| β-Caryophyllene | 1.0% | |||||||||||||
| (2E,6E)-α-Farnesene | 1.0% | |||||||||||||
| β-Phellandrene | 5.4% | |||||||||||||
| Nootkatone | 0.1–0.8% | |||||||||||||
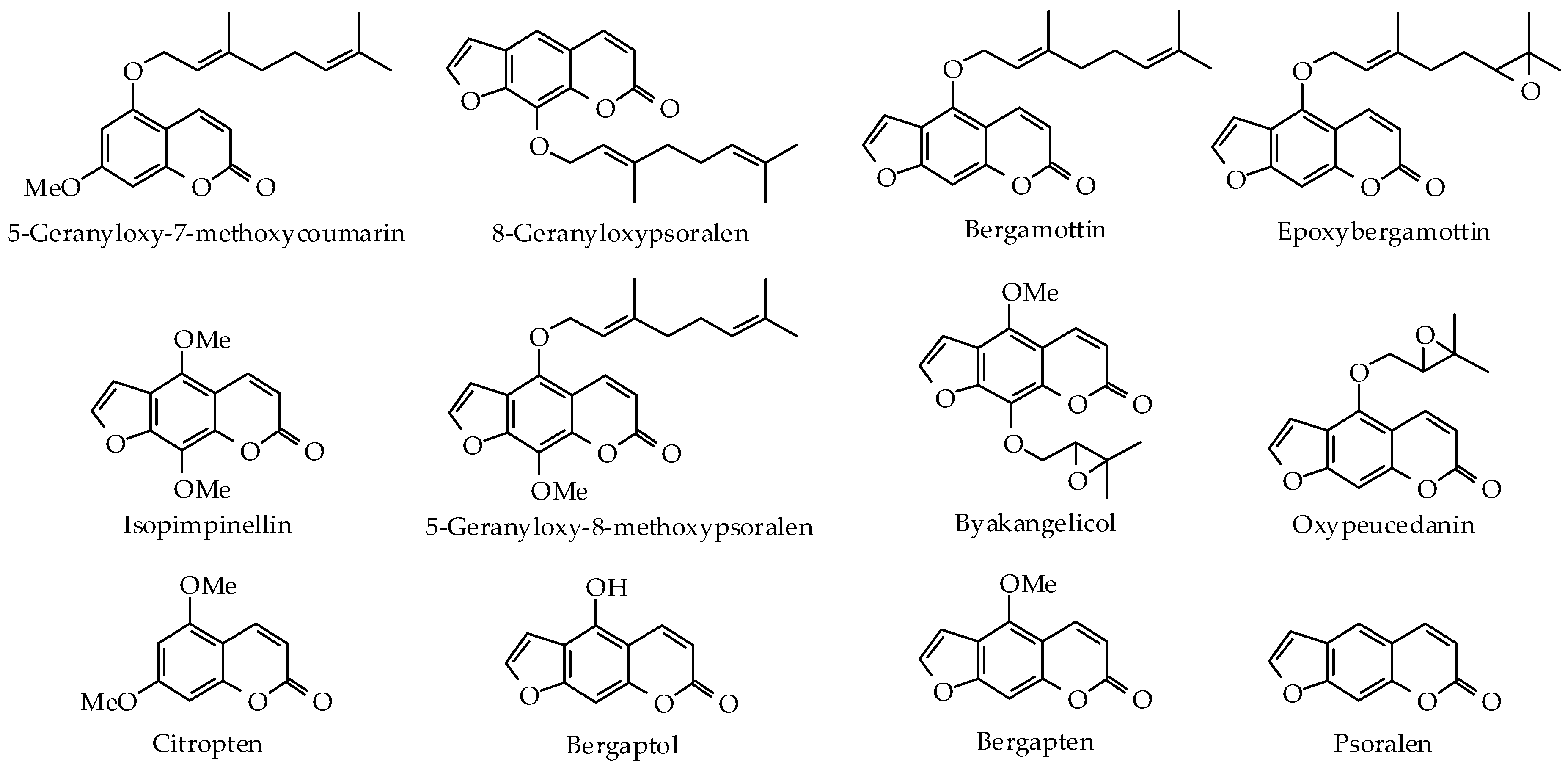
| Non-Volatile Components | Bitter Orange [8,26] | Lemon [8] | Lime [8,26] | Grapefruit [8,26] | Bergamot [8,24,25] | Bergamot (FCF) [8,24] | Mandarin [8] |
|---|---|---|---|---|---|---|---|
| Bergamottin | - | 0.16–0.54% | 1.7–3.0% | <0.11% | 0.68–2.75% | 0–1.625% | 0–0.001% |
| Bergapten | 0.035–0.073% | 0.0001–0.035% | 0.17–0.33% | 0.012–0.19% | 0.11–0.33% | 0–0.0091% | 0–0.0003% |
| Oxypeucedanin | - | 0.09–0.82% | 0.02–0.3% | - | - | - | - |
| 5-Geranloxy-7-methoxycoumarin | - | 0.18–0.28% | 1.7–3.2% | - | 0.08–0.68% | 0–0.19% | - |
| Citropten | - | 0.05–0.17% | 0.4–2.2% | - | 0.01–0.35% | 0–0.0052% | - |
| Byakangelicol | - | 0.006–0.16% | - | - | - | - | - |
| 8-Geranyloxypsoralen | - | 0.01–0.045% | 0.10–0.14% | - | - | - | - |
| Isopimpinellin | - | 0–0.011% | 0.1–1.3% | - | - | - | - |
| 5-Geranoxy-8-methoxypsoralen | - | - | 0.2–0.9% | - | - | - | - |
| Epoxybergamottin | 0.082% | - | - | 0.1126% | - | - | - |
| Psoralen | 0.007% | - | - | - | 0–0.0026% | - | - |
| Bergaptol | - | - | - | - | 0–0.19% | - | - |
| Essential Oil | Biological Activity | Ref. |
|---|---|---|
| Sweet orange | Anticarcinogenic | [28,29] |
| Relaxant | [30] | |
| Anxiolytic | [31,32,33] | |
| Pain relief | [34] | |
| Hepatocarcinogenesis suppressant | [35] | |
| Anti-tumor | [36] | |
| Antioxidant | [37] | |
| Food preservative | [38] | |
| Acne treatment (with sweet basil oil) | [39] | |
| Antibacterial | [40,41,42,43] | |
| Antifungal | [10,44,45] | |
| Anti-aflatoxigenic (at 500 ppm) | [44] | |
| Larvicidal | [46,47] | |
| Insecticidal | [48,49,50] | |
| Anthelmintic | [51] | |
| Growth promoter (in Tilapia) | [52] | |
| Bitter orange | Mild sedative, hypnotic, soothing, calming, and motor relaxant | [53] |
| Sleep inducer | [54] | |
| Anxiolytic and antidepressant | [53,55,56,57,58] | |
| Pain relief | [34,59] | |
| Antiseizure and anticonvulsant agent | [54] | |
| Anti-spasmodic and sexual desire enhancer | [59] | |
| Gastroprotective and ulcer healing | [60] | |
| Digestive disorders treatment | [53] | |
| Hepatocarcinogenesis suppressant | [35] | |
| Antioxidant | [53,61] | |
| Nephroprotective | [62] | |
| Antibacterial | [53,63,64,65] | |
| Pimple and acne treatment | [53] | |
| Antifungal | [15,53,66] | |
| Fumigant and anti-cholinesterase | [67] | |
| Larvicidal | [46] | |
| Neroli | Sedative, soothing, calming, and motor relaxant | [55,68] |
| Anxiolytic and antidepressant | [53,57,69,70] | |
| Antiseizure and anticonvulsant | [71,72] | |
| Central and peripheral antinociceptive effects | [73] | |
| Anti-inflammatory | [73] | |
| Menopausal symptoms relief | [74] | |
| Premenstrual syndrome (PMS) relief | [75] | |
| Sexual desire enhancer | [59] | |
| Endothelium- and smooth muscle-dependent vasodilator | [76] | |
| Hypotensive | [77] | |
| Antioxidant | [20,78] | |
| Anti-amnesic | [72] | |
| Antibacterial | [53,79] | |
| Antifungal | [20,53,75,79] | |
| Orange petitgrain | Antioxidant | [78,80] |
| Antibacterial | [81] | |
| Antifungal | [81] | |
| Mandarin | Anti-proliferative | [82] |
| Chemoprotective | [82] | |
| Antioxidant | [83] | |
| Antibacterial | [83,84] | |
| Antifungal | [84,85,86,87] | |
| Lemon | Stress relief | [88,89] |
| Cytotoxic | [28,90] | |
| Chemoprotective | [91] | |
| Anti-obesity | [92] | |
| Antioxidant | [93] | |
| Neuroprotective | [94,95] | |
| Anti-anxiety | [96] | |
| Creativity and mood enhancer | [97] | |
| Analgesic | [98] | |
| Relief of nausea and vomiting of pregnancy | [99] | |
| Anti-spasmodic | [89] | |
| Attention level, concentration, cognitive performance, mood, and memory enhancer | [89,100] | |
| Skin penetration enhancer | [101] | |
| Antibacterial | [102,103] | |
| Antifungal | [10] | |
| Insect repellent | [104] | |
| Miticidal | [105] | |
| Lime | Anti-obesity | [106] |
| Spasmolytic agent | [107,108] | |
| Selective acetylcholinesterase and buytrylcholinesterase inhibitor | [109] | |
| Antioxidant | [109] | |
| Anti-inflammatory | [110] | |
| Flavoring agent | [111,112] | |
| Antibacterial | [111,113] | |
| Antifungal | [111,113] | |
| Insecticidal | [114] | |
| Phytotoxic | [113] | |
| Grapefruit | Anti-obesity | [92,115,116,117] |
| Cravings and hunger reducer (mixed with patchouli oil) | [116] | |
| Body cleansing promoter | [116] | |
| Cytotoxic | [28,90] | |
| Antibacterial | [118,119] | |
| Antifungal | [118,119,120] | |
| Larvicidal | [121,122,123,124] | |
| Bergamot | Melanogenic component in suntan preparations | [125,126] |
| Pain relief | [127,128,129] | |
| Peripheral antinociceptive | [129,130] | |
| Antiallodynic | [127,131] | |
| Wound healing | [132] | |
| Cytotoxic | [125,133,134,135] | |
| Anti-tumor | [136] | |
| Neuroprotective | [137,138] | |
| Sedative, calming, and soothing | [139] | |
| Anxiolytic | [139,140] | |
| Mood enhancer | [141] | |
| Antioxidant | [109] | |
| Antibacterial | [142,143,144] | |
| Antifungal | [142,143,145] | |
| Anti-dermatophyte | [146,147] | |
| Antimycoplasmal | [148] | |
| Yuzu | Anti-carcinogenic | [149] |
| Anti-inflammatory | [150] | |
| Anti-anxiety | [151] | |
| Mood disturbance, tension-anxiety, anger-hostility, and fatigue reducer | [152,153] | |
| Mind and body health promoter | [152] | |
| Odor suppressant | [154] | |
| Anti-cancer | [155] | |
| Hypocholesterolemic | [156] | |
| Anti-diabetic | [157] | |
| Anti-obesity | [158] | |
| Platelet aggregation inhibitor | [159] | |
| Heart failure treatment | [160] | |
| Kumquat | Antiproliferative | [161] |
| Antioxidant | [161,162] | |
| Antibacterial | [163] | |
| Antifungal | [163] |
| Acute Toxicity | Phototoxicity Risk [167] | Irritation of Undiluted Oil [8] | Acute Dermal LD50 in Rabbits (g/kg) [167] | Acute Oral LD50 in Rats (g/kg) [167] | Maximum Dermal Use Level [8] |
|---|---|---|---|---|---|
| Sweet orange EO | Low risk | Moderately irritating to rabbits but not irritating to mice | >5 | >5 | - |
| Bitter orange EO | low risk | Moderately irritating to rabbits | >10 | >5 | 1.25% |
| Neroli EO | Not phototoxic | Not irritating | >5 | 4.55 | - |
| Petitgrain EO | Not phototoxic | Slightly irritating to rabbits, but not irritating to mice or pigs | <2 | >5 | - |
| Lemon EO (distilled) | Not phototoxic | Moderately irritating to rabbits and slightly irritating to mice | >5 | >5 | 20% |
| Lemon EO (expressed) | Low risk | Not irritating | >5 | >5 | 2% |
| Lime EO (distilled) | Not phototoxic | Slightly irritating to rabbits | >5 | >5 | - |
| Lime EO (expressed) | moderate risk | No data available | >5 | >5 | 0.7% |
| Grapefruit EO | Low risk | Slightly irritating to rabbits, but not irritating to mice or pigs | >5 | >5 | 4% |
| Bergamot EO (FCF) | Not phototoxic | Mildly irritating to rabbits | >20 | >10 | 0.4% |
| Bergamot EO (expressed) | Moderate risk | Moderately irritating to rabbits | - | - | - |
| Yuzu EO | Not phototoxic | Not irritating | - | - | - |
| Mandarin | Not phototoxic | Moderately irritating (produces slight edema and erythema) to rabbits, mice, and pigs | >5 | >5 | 30% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dosoky, N.S.; Setzer, W.N. Biological Activities and Safety of Citrus spp. Essential Oils. Int. J. Mol. Sci. 2018, 19, 1966. https://doi.org/10.3390/ijms19071966
Dosoky NS, Setzer WN. Biological Activities and Safety of Citrus spp. Essential Oils. International Journal of Molecular Sciences. 2018; 19(7):1966. https://doi.org/10.3390/ijms19071966
Chicago/Turabian StyleDosoky, Noura S., and William N. Setzer. 2018. "Biological Activities and Safety of Citrus spp. Essential Oils" International Journal of Molecular Sciences 19, no. 7: 1966. https://doi.org/10.3390/ijms19071966
APA StyleDosoky, N. S., & Setzer, W. N. (2018). Biological Activities and Safety of Citrus spp. Essential Oils. International Journal of Molecular Sciences, 19(7), 1966. https://doi.org/10.3390/ijms19071966







