PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas
Abstract
1. Introduction
2. Results
2.1. Immunohistochemistry
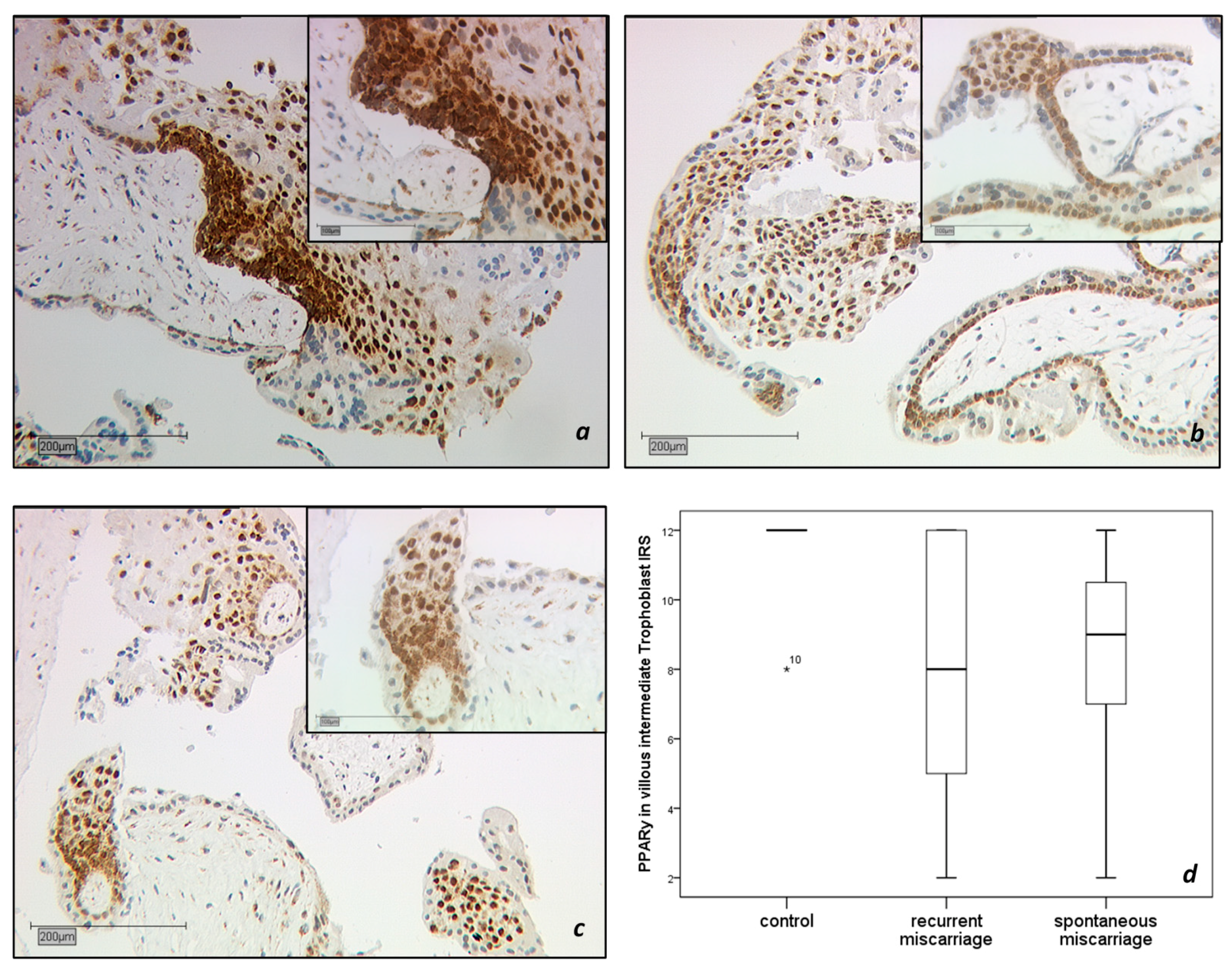
2.1.1. PPARγ-Expression in the Trophoblast
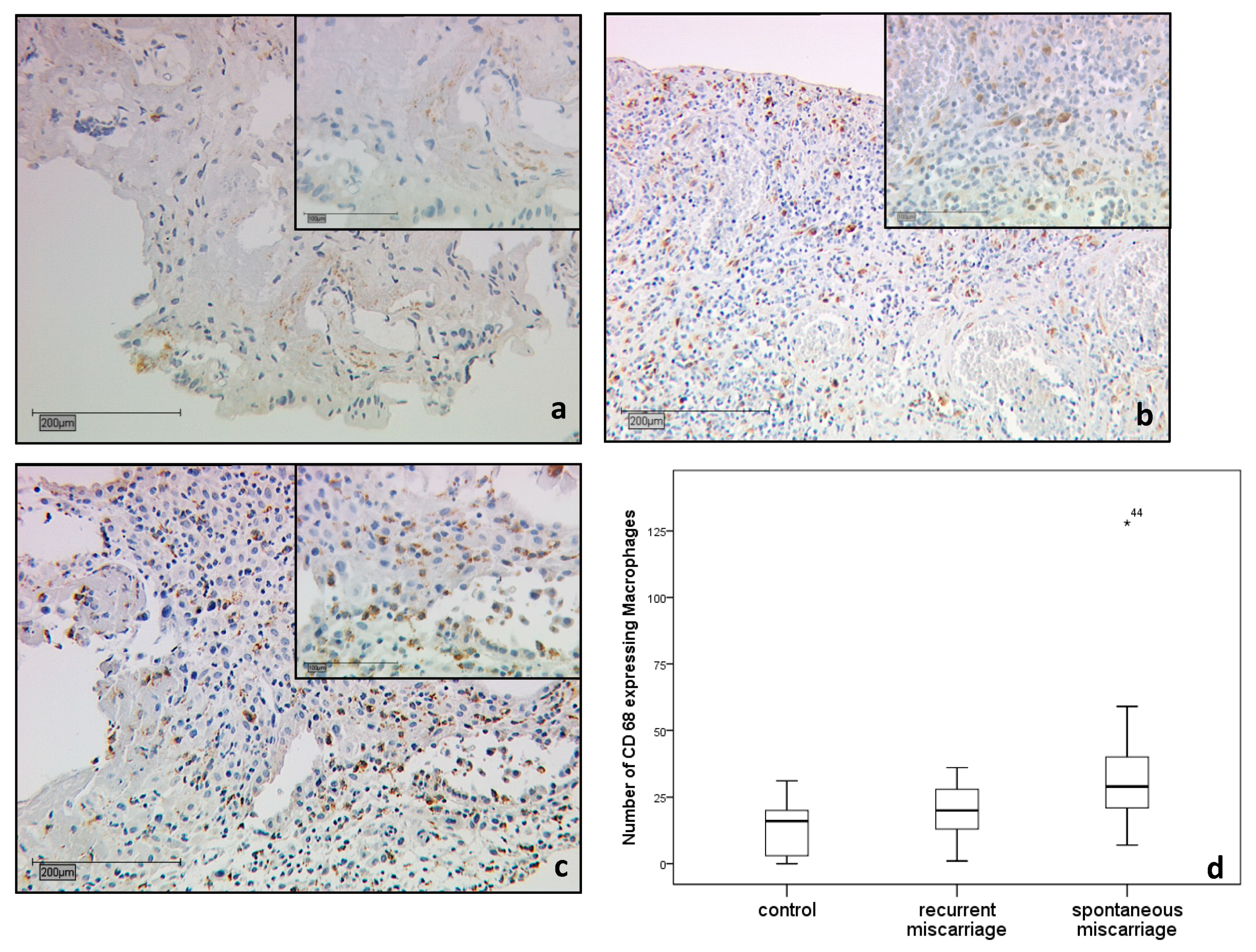
2.1.2. CD68 Positive Decidual Macrophages in the Decidua
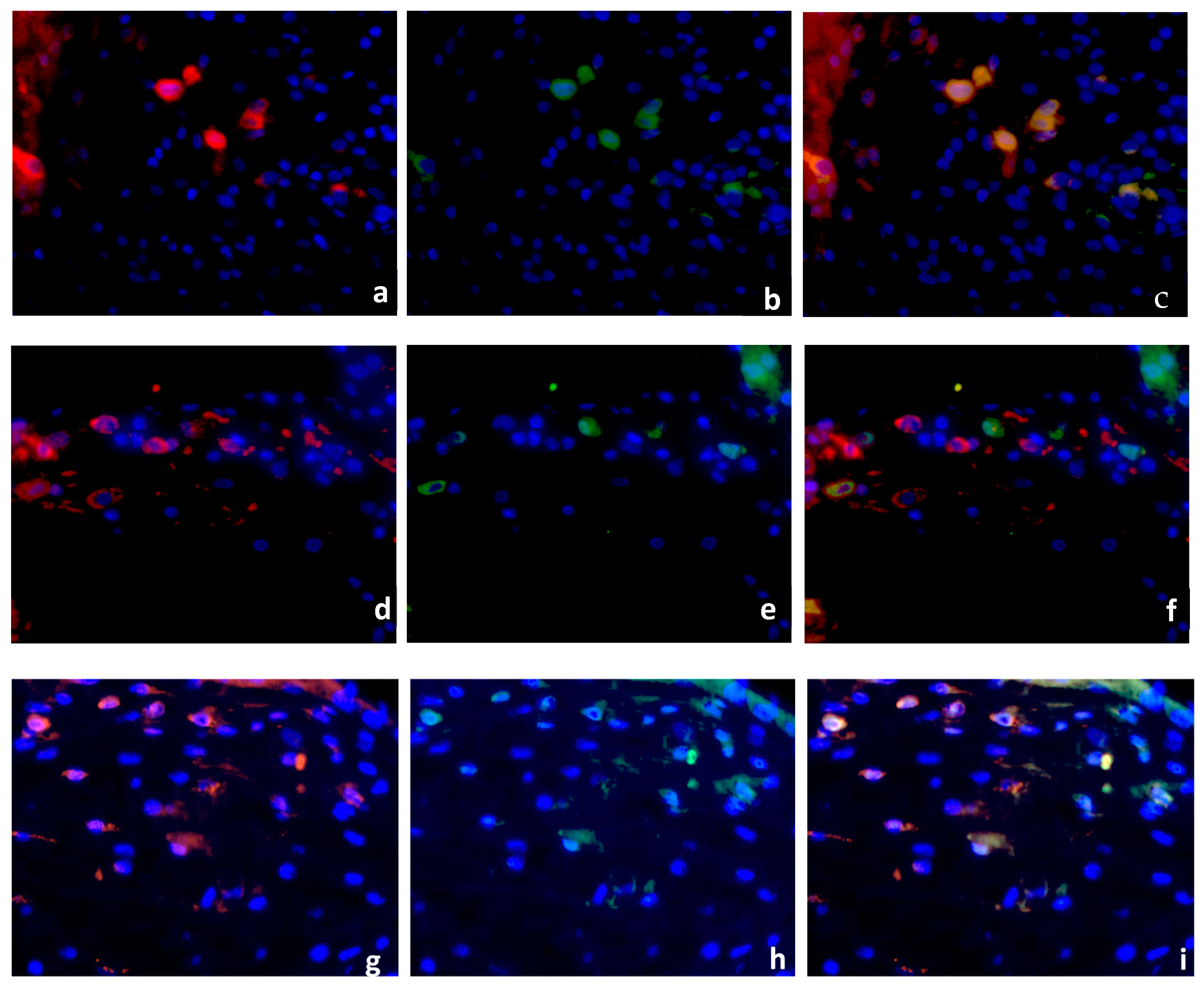
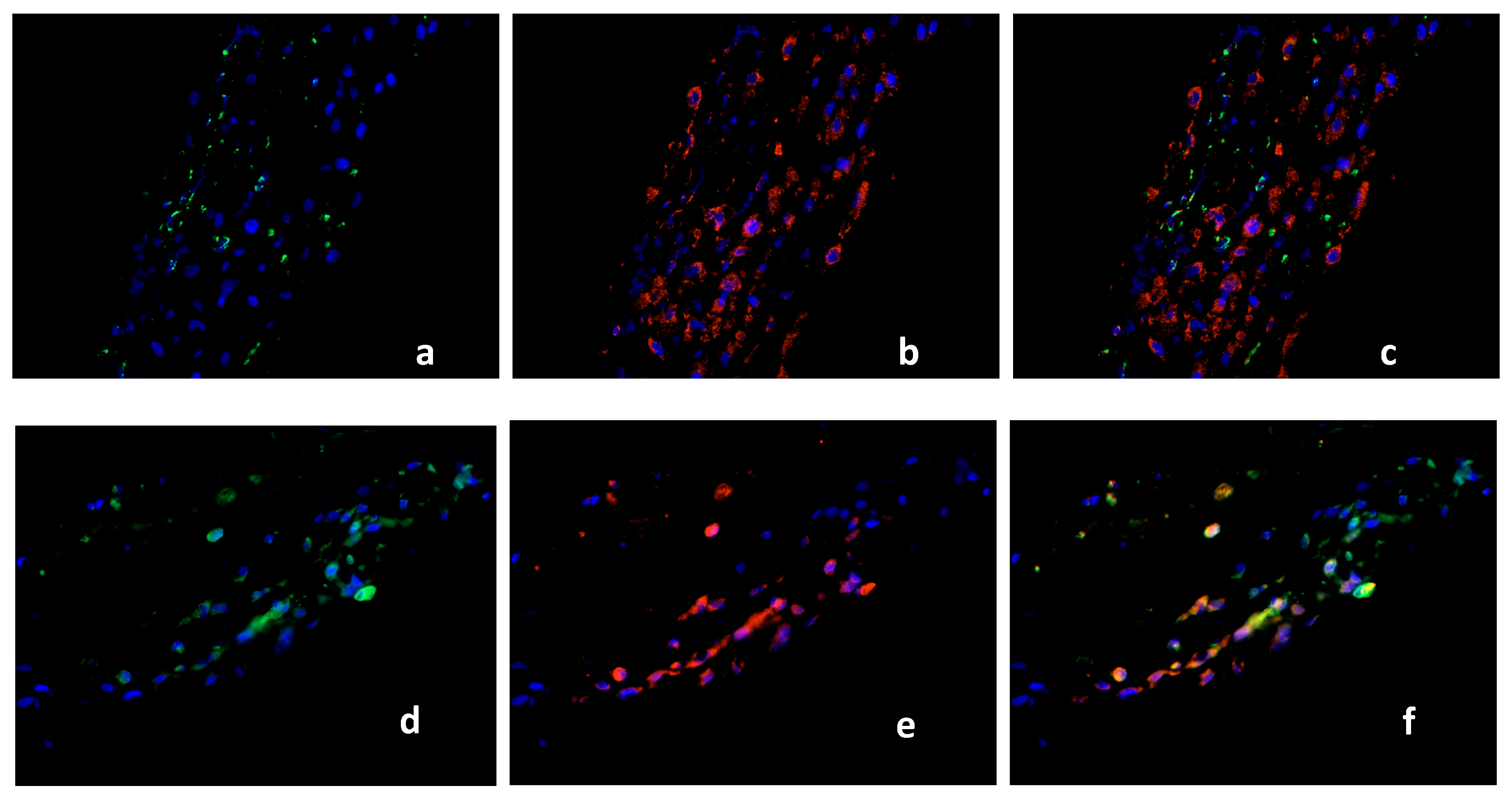
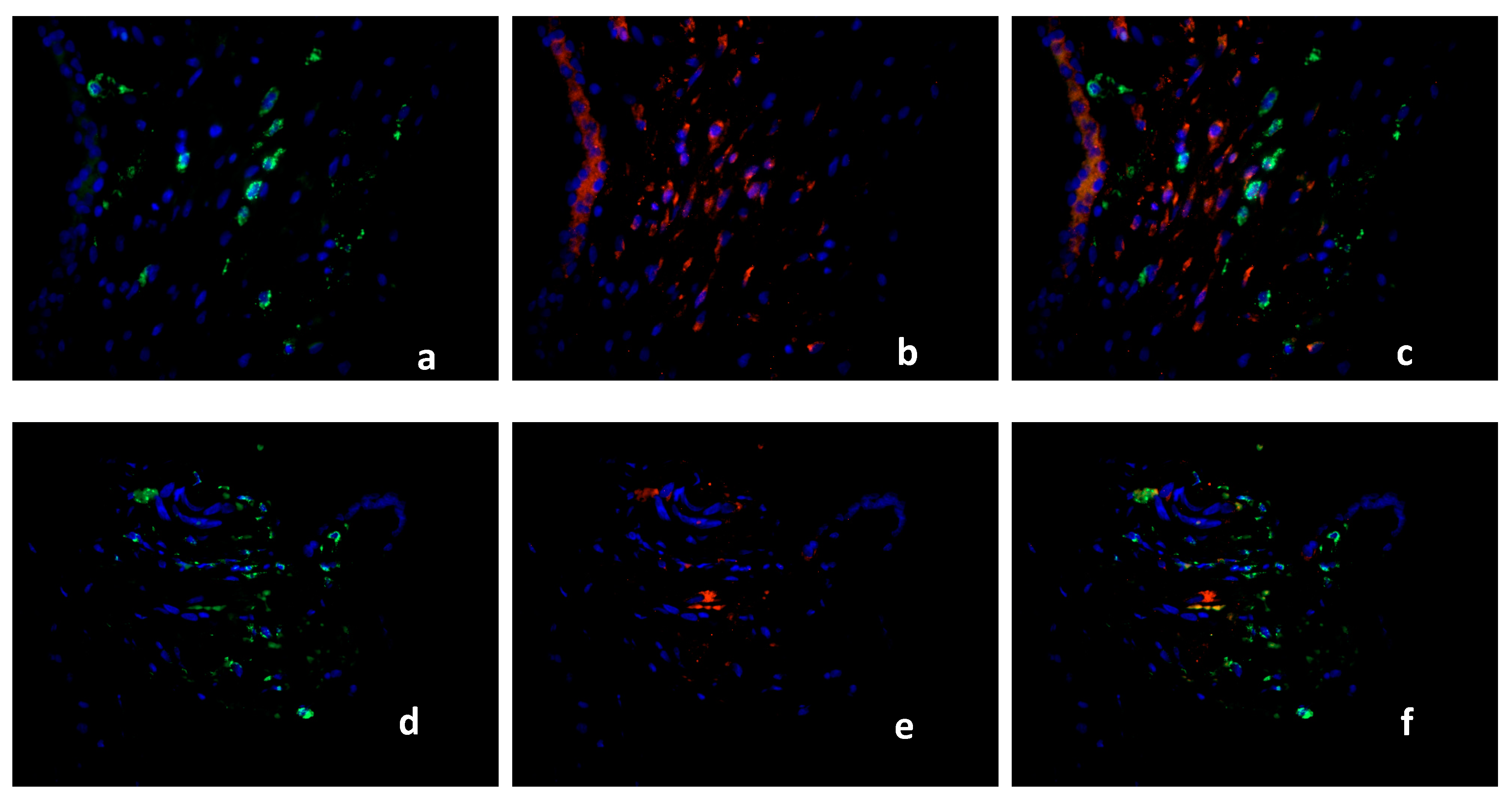
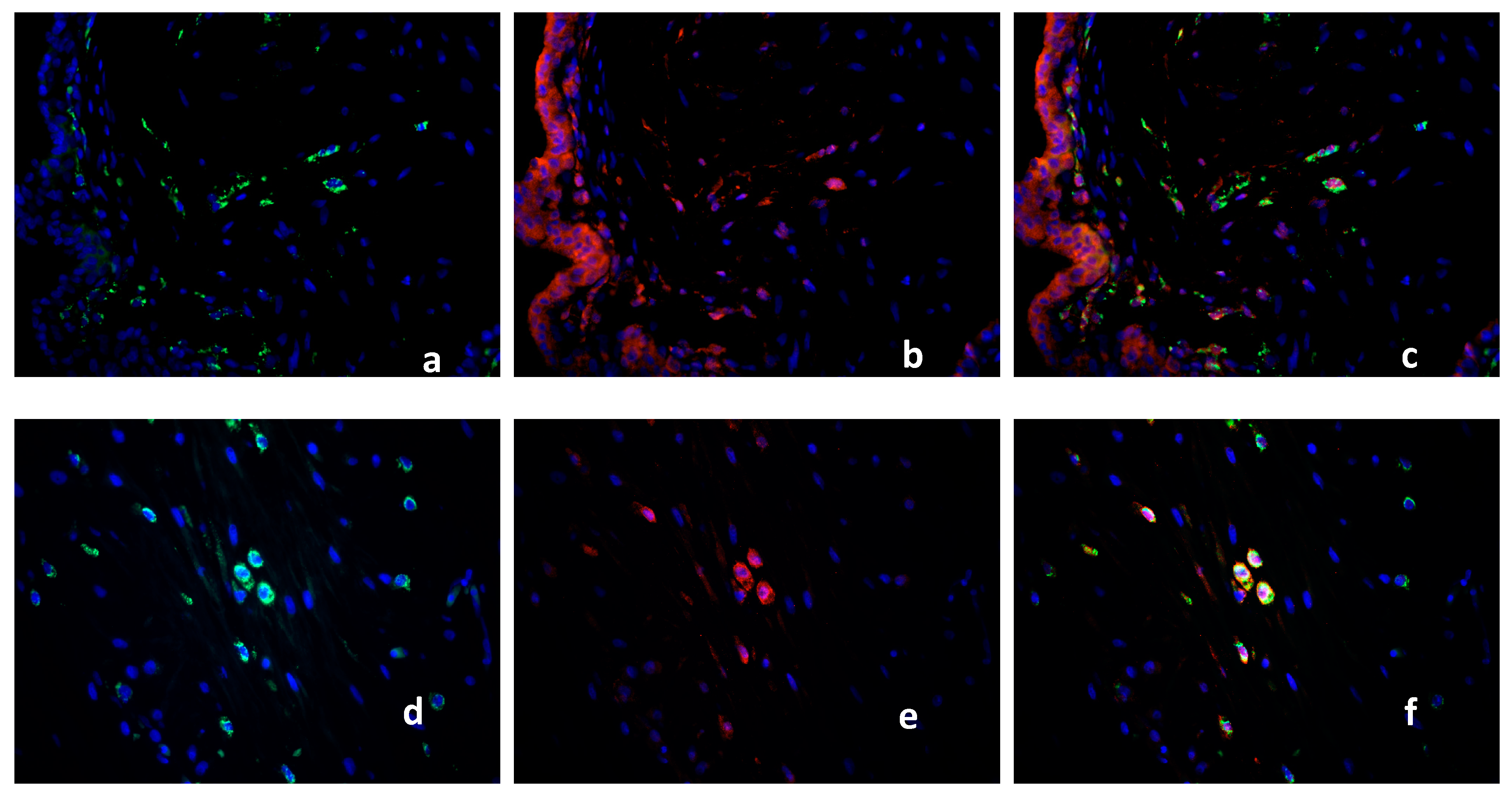
2.2. Double Immunofluorescence
Identification of PPARγ-Expressing Cells in the Decidua Basalis
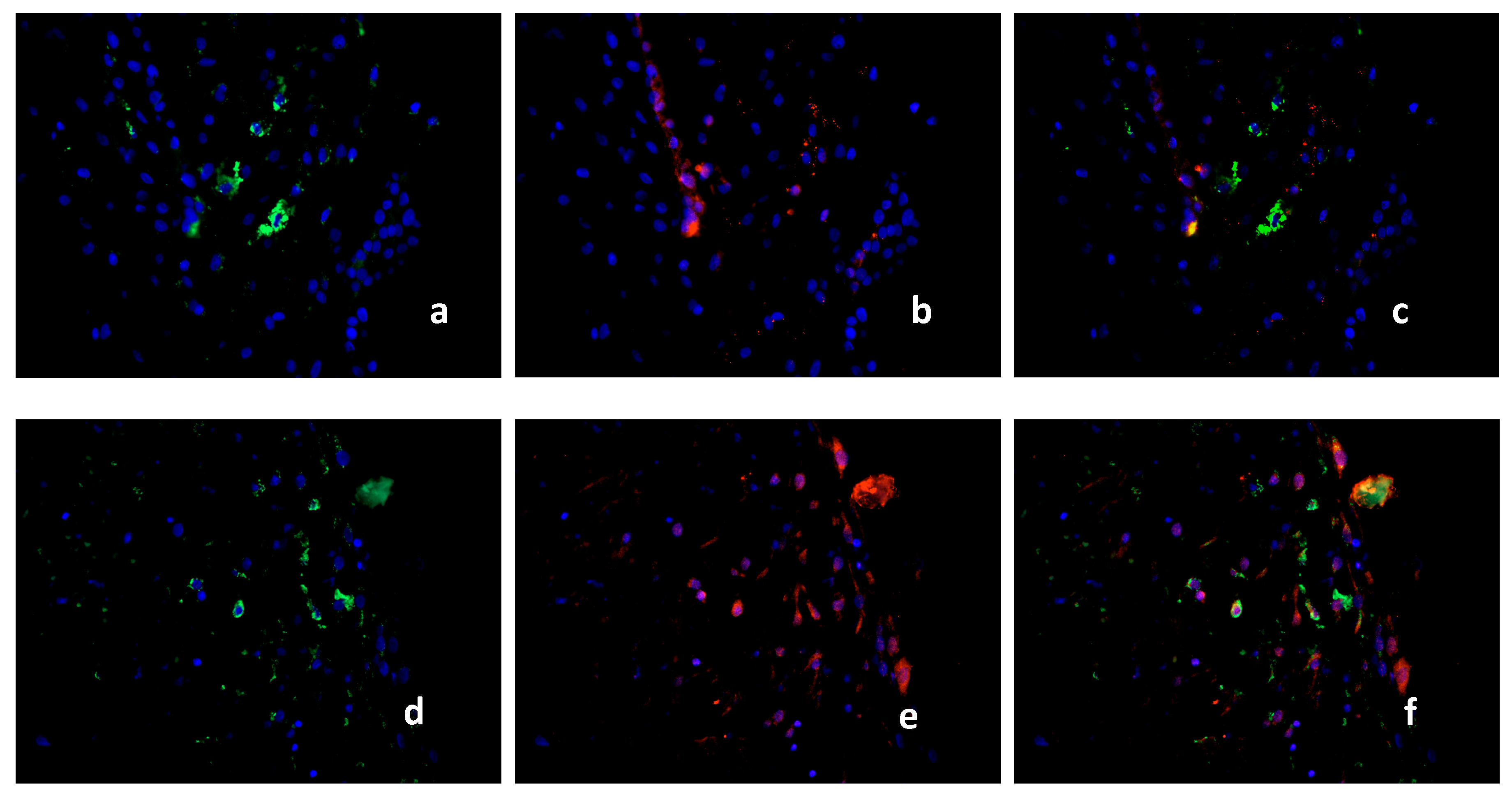
2.3. Characterization of the Macrophage Population in Recurrent Miscarriage Cases
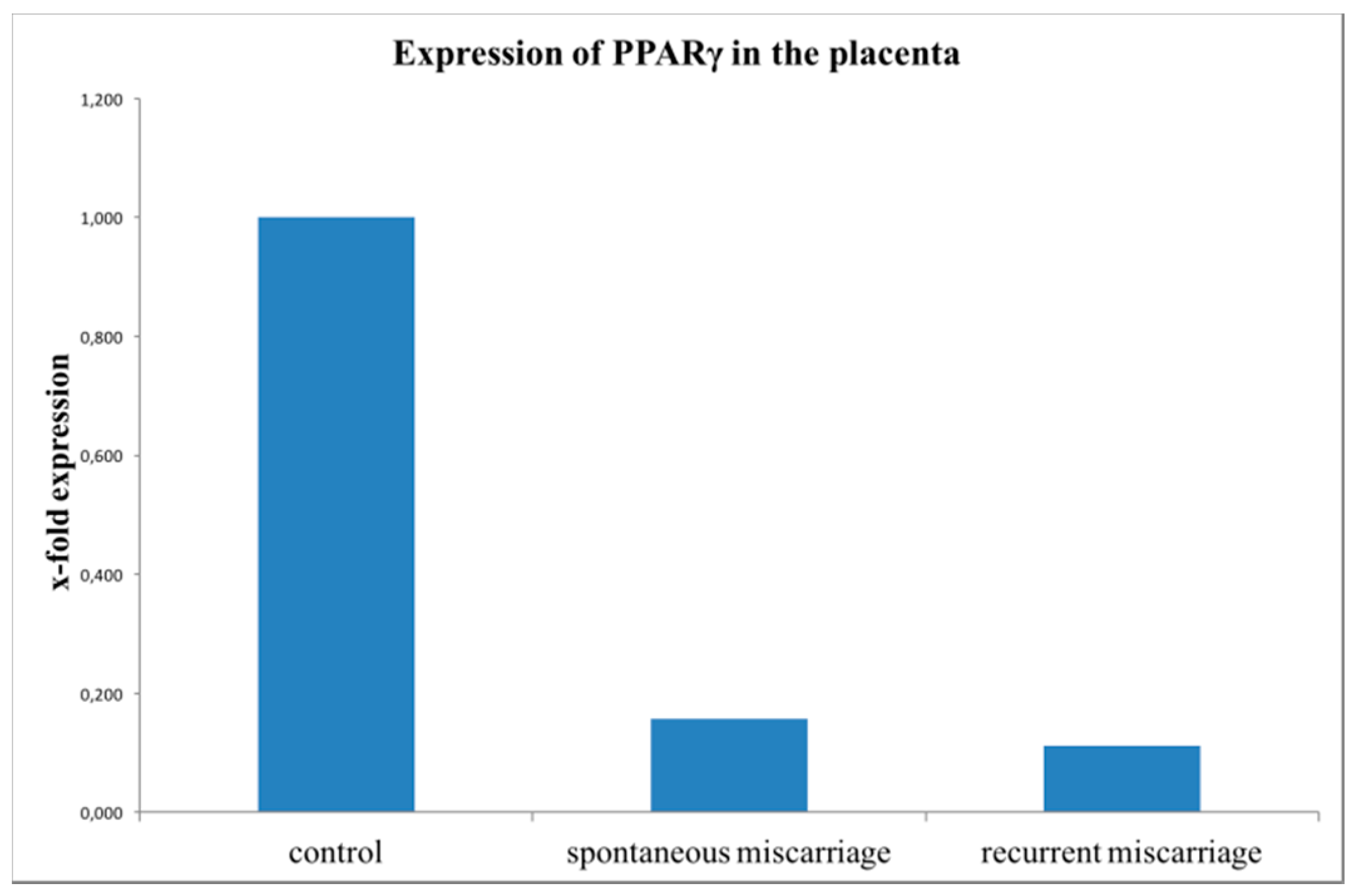
2.4. Evaluation of PPARγ Expression with Real-Time RT-PCR (TaqMan)
3. Discussion
4. Materials and Methods
4.1. Patient Data
4.2. Immunohistochemistry
4.3. Immunofluorescence
4.3.1. Evaluation of PPARγ-Expressing Cells as Macrophages
4.3.2. Evaluation of M1/M2 Marker on Decidual Macrophages
4.4. Evaluation of PPARγ with Real-Time RT-PCR (Taq Man)
4.4.1. RNA Extraction from Placental Tissue
4.4.2. Reverse Transcription
4.4.3. Real-Time Reverse Transcription PCR
4.5. Statistics
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| PPAR | Peroxisome-Proliferator-activated Receptor |
| SM | Spontaneous miscarriage |
| RM | Recurrent miscarriage |
References
- Makrigiannakis, A.; Petsas, G.; Toth, B.; Relakis, K.; Jeschke, U. Recent advances in understanding immunology of reproductive failure. J. Reprod. Immunol. 2011, 90, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Knabl, J.; Vattai, A.; Huttenbrenner, R.; Hutter, S.; Karsten, M.; Jeschke, U. Rxralpha is upregulated in first trimester endometrial glands of spontaneous abortions unlike LXR and PPARγ. Eur. J. Histochem. 2016, 60, 2665. [Google Scholar] [CrossRef] [PubMed]
- Toth, B.; Jeschke, U.; Rogenhofer, N.; Scholz, C.; Wurfel, W.; Thaler, C.J.; Makrigiannakis, A. Recurrent miscarriage: Current concepts in diagnosis and treatment. J. Reprod. Immunol. 2010, 85, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Arck, P.C.; Rucke, M.; Rose, M.; Szekeres-Bartho, J.; Douglas, A.J.; Pritsch, M.; Blois, S.M.; Pincus, M.K.; Barenstrauch, N.; Dudenhausen, J.W.; et al. Early risk factors for miscarriage: A prospective cohort study in pregnant women. Reprod. Biomed. Online 2008, 17, 101–113. [Google Scholar] [CrossRef]
- Pestka, A.; Fitzgerald, J.S.; Toth, B.; Markert, U.R.; Jeschke, U. Nuclear hormone receptors and female reproduction. Curr. Mol. Med. 2013, 13, 1066–1078. [Google Scholar] [CrossRef] [PubMed]
- Guibourdenche, J.; Alsat, E.; Soncin, F.; Rochette-Egly, C.; Evain-Brion, D. Retinoid receptors expression in human term placenta: Involvement of rxr alpha in retinoid induced-hcg secretion. J. Clin. Endoc. Metab. 1998, 83, 1384–1387. [Google Scholar]
- Toth, B.; Hornung, D.; Scholz, C.; Djalali, S.; Friese, K.; Jeschke, U. Peroxisome proliferator-activated receptors: New players in the field of reproduction. Am. J. Reprod. Immunol. 2007, 58, 289–310. [Google Scholar] [CrossRef] [PubMed]
- Arck, P.; Toth, B.; Pestka, A.; Jeschke, U. Nuclear receptors of the peroxisome proliferator-activated receptor (PPAR) family in gestational diabetes: From animal models to clinical trials. Biol. Reprod. 2010, 83, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Pestka, A.; Toth, B.; Kuhn, C.; Hofmann, S.; Wiest, I.; Wypior, G.; Friese, K.; Jeschke, U. Retinoid x receptor alpha and retinoids are key regulators in apoptosis of trophoblasts of patients with recurrent miscarriages. J. Mol. Endocrinol. 2011, 47, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Knabl, J.; Pestka, A.; Huttenbrenner, R.; Plosch, T.; Ensenauer, R.; Welbergen, L.; Hutter, S.; Gunthner-Biller, M.; Jeschke, U. The liver x receptor in correlation with other nuclear receptors in spontaneous and recurrent abortions. PPAR Res. 2013, 2013, 575604. [Google Scholar] [CrossRef] [PubMed]
- Vattai, A.; Ziegelmuller, B.; Kost, B.; Kuhn, C.; Hofmann, S.; Bayer, B.; Anslinger, K.; Jeschke, U.; Ditsch, N. The expression of thyroid hormone receptors (THR) is regulated by the progesterone receptor system in first trimester placental tissue and in bewo cells in vitro. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 195, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Moradin, A.; Schlessinger, D.; Nagaraja, R. RXRα and LXR activate two promoters in placenta- and tumor-specific expression of plac1. Placenta 2011, 32, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Hutter, S.; Knabl, J.; Andergassen, U.; Jeschke, U. The role of ppars in placental immunology: A systematic review of the literature. PPAR Res. 2013, 2013, 970276. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Romero, R.; Miller, D.; Kadam, L.; Mial, T.N.; Plazyo, O.; Garcia-Flores, V.; Hassan, S.S.; Xu, Z.; Tarca, A.L.; et al. An m1-like macrophage polarization in decidual tissue during spontaneous preterm labor that is attenuated by rosiglitazone treatment. J. Immunol. 2016, 196, 2476–2491. [Google Scholar] [CrossRef] [PubMed]
- Hutter, S.; Heublein, S.; Knabl, J.; Andergassen, U.; Vrekoussis, T.; Makrigiannakis, A.; Friese, K.; Mayr, D.; Jeschke, U. Macrophages: Are they involved in endometriosis, abortion and preeclampsia and how? J. Nippon Med. Sch. 2013, 80, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Slukvin, I.I.; Breburda, E.E.; Golos, T.G. Dynamic changes in primate endometrial leukocyte populations: Differential distribution of macrophages and natural killer cells at the rhesus monkey implantation site and in early pregnancy. Placenta 2004, 25, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Heublein, S.; Vrekoussis, T.; Kuhn, C.; Friese, K.; Makrigiannakis, A.; Mayr, D.; Lenhard, M.; Jeschke, U. Inducers of g-protein coupled estrogen receptor (GPER) in endometriosis: Potential implications for macrophages and follicle maturation. J. Reprod. Immunol. 2013, 97, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Stremmel, C.; Schuchert, R.; Wagner, F.; Thaler, R.; Weinberger, T.; Pick, R.; Mass, E.; Ishikawa-Ankerhold, H.C.; Margraf, A.; Hutter, S.; et al. Yolk sac macrophage progenitors traffic to the embryo during defined stages of development. Nat. Commun. 2018, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Petsas, G.; Jeschke, U.; Richter, D.U.; Minas, V.; Hammer, A.; Kalantaridou, S.; Toth, B.; Tsatsanis, C.; Friese, K.; Makrigiannakis, A. Aberrant expression of corticotropin-releasing hormone in pre-eclampsia induces expression of fasl in maternal macrophages and extravillous trophoblast apoptosis. Mol. Hum. Reprod. 2012, 18, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Guenther, S.; Vrekoussis, T.; Heublein, S.; Bayer, B.; Anz, D.; Knabl, J.; Navrozoglou, I.; Dian, D.; Friese, K.; Makrigiannakis, A.; et al. Decidual macrophages are significantly increased in spontaneous miscarriages and over-express fasl: A potential role for macrophages in trophoblast apoptosis. Int. J. Mol. Sci. 2012, 13, 9069–9080. [Google Scholar] [CrossRef] [PubMed]
- Hannan, N.J.; Jones, R.L.; Critchley, H.O.; Kovacs, G.J.; Rogers, P.A.; Affandi, B.; Salamonsen, L.A. Coexpression of fractalkine and its receptor in normal human endometrium and in endometrium from users of progestin-only contraception supports a role for fractalkine in leukocyte recruitment and endometrial remodeling. J. Clin. Endocrinol. Metab. 2004, 89, 6119–6129. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Feng, Y.; Xia, Z.; Li, X.; Rong, J. Omega-alkynyl arachidonic acid promotes anti-inflammatory macrophage m2 polarization against acute myocardial infarction via regulating the cross-talk between PKM2, HIF-1α and iNOS. Biochim. Biophys. Acta 2017, 1862, 1595–1605. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, L.; Yu, C.; Yang, X.F.; Wang, H. Monocyte and macrophage differentiation: Circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark. Res. 2014, 2, 1. [Google Scholar] [CrossRef] [PubMed]
- Tontonoz, P.; Spiegelman, B.M. Fat and beyond: The diverse biology of pparγ. Annu. Rev. Biochem. 2008, 77, 289–312. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, C.; Mjosberg, J.; Matussek, A.; Geffers, R.; Matthiesen, L.; Berg, G.; Sharma, S.; Buer, J.; Ernerudh, J. Gene expression profiling of human decidual macrophages: Evidence for immunosuppressive phenotype. PLoS ONE 2008, 3, e2078. [Google Scholar] [CrossRef] [PubMed]
- Svensson, J.; Jenmalm, M.C.; Matussek, A.; Geffers, R.; Berg, G.; Ernerudh, J. Macrophages at the fetal-maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J. Immunol. 2011, 187, 3671–3682. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.M.; Moore, L.B.; Smith-Oliver, T.A.; Wilkison, W.O.; Willson, T.M.; Kliewer, S.A. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor γ (PPARγ). J. Biolo. Chem. 1995, 270, 12953–12956. [Google Scholar] [CrossRef]
- Bouhlel, M.A.; Derudas, B.; Rigamonti, E.; Dievart, R.; Brozek, J.; Haulon, S.; Zawadzki, C.; Jude, B.; Torpier, G.; Marx, N.; et al. PPARγ activation primes human monocytes into alternative M2 macrophages with anti-inflammatory properties. Cell Metab. 2007, 6, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Lavinsky, R.M.; Jepsen, K.; Heinzel, T.; Torchia, J.; Mullen, T.M.; Schiff, R.; Del-Rio, A.L.; Ricote, M.; Ngo, S.; Gemsch, J.; et al. Diverse signaling pathways modulate nuclear receptor recruitment of n-cor and smrt complexes. Proc. Natl. Acad. Sci. USA. 1998, 95, 2920–2925. [Google Scholar] [CrossRef] [PubMed]
- Ricote, M.; Huang, J.; Fajas, L.; Li, A.; Welch, J.; Najib, J.; Witztum, J.L.; Auwerx, J.; Palinski, W.; Glass, C.K. Expression of the peroxisome proliferator-activated receptor γ (PPARγ) in human atherosclerosis and regulation in macrophages by colony stimulating factors and oxidized low density lipoprotein. Proc. Natl. Acad. Sci. USA 1998, 95, 7614–7619. [Google Scholar] [CrossRef] [PubMed]
- Ricote, M.; Li, A.C.; Willson, T.M.; Kelly, C.J.; Glass, C.K. The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 1998, 391, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Wongweragiat, S.; Searle, R.F.; Bulmer, J.N. Expression of fas/fas ligand by decidual leukocytes in hydatidiform mole. Biol. Reprod. 2001, 64, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Hammer, A.; Blaschitz, A.; Daxbock, C.; Walcher, W.; Dohr, G. Fas and fas-ligand are expressed in the uteroplacental unit of first-trimester pregnancy. Am. J. Reprod. Immunol. 1999, 41, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Hammer, A.; Dohr, G. Expression of fas-ligand in first trimester and term human placental villi. J. Reprod. Immunol. 2000, 46, 83–90. [Google Scholar] [CrossRef]
- Aplin, J.D. Developmental cell biology of human villous trophoblast: Current research problems. Int. J. Dev. Biol. 2010, 54, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Tarrade, A.; Rochette-Egly, C.; Guibourdenche, J.; Evain-Brion, D. The expression of nuclear retinoid receptors in human implantation. Placenta 2000, 21, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Toth, B.; Bastug, M.; Mylonas, I.; Scholz, C.; Makovitzky, J.; Kunze, S.; Thaler, C.; Friese, K.; Jeschke, U. Peroxisome proliferator-activated receptor-γ in normal human pregnancy and miscarriage. Acta Histochem. 2009, 111, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Toth, B.; Bastug, M.; Scholz, C.; Arck, P.; Schulze, S.; Kunze, S.; Friese, K.; Jeschke, U. Leptin and peroxisome proliferator-activated receptors: Impact on normal and disturbed first trimester human pregnancy. Histol. Histopathol. 2008, 23, 1465–1475. [Google Scholar] [PubMed]
- Fournier, T.; Guibourdenche, J.; Handschuh, K.; Tsatsaris, V.; Rauwel, B.; Davrinche, C.; Evain-Brion, D. Pparγ and human trophoblast differentiation. J. Reprod. Immunol. 2011, 90, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Knabl, J.; Huttenbrenner, R.; Hutter, S.; Gunthner-Biller, M.; Vrekoussis, T.; Karl, K.; Friese, K.; Kainer, F.; Jeschke, U. Peroxisome proliferator-activated receptor-γ (PPARγ) is down regulated in trophoblast cells of gestational diabetes mellitus (GDM) and in trophoblast tumour cells bewo in vitro after stimulation with pparγ agonists. J. Perinat. Med. 2014, 42, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Ziegelmuller, B.; Vattai, A.; Kost, B.; Kuhn, C.; Hofmann, S.; Bayer, B.; Toth, B.; Jeschke, U.; Ditsch, N. Expression of thyroid hormone receptors in villous trophoblasts and decidual tissue at protein and mrna levels is downregulated in spontaneous and recurrent miscarriages. J. Histochem. Cytochem. 2015, 63, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Barak, Y.; Nelson, M.C.; Ong, E.S.; Jones, Y.Z.; Ruiz-Lozano, P.; Chien, K.R.; Koder, A.; Evans, R.M. PPARγ is required for placental, cardiac, and adipose tissue development. Mol. Cell 1999, 4, 585–595. [Google Scholar] [CrossRef]
| Gestational Age | Normal Pregnancy | Spontaneous Miscarriage | Recurrent Miscarriage |
|---|---|---|---|
| 4th week | 0 | 0 | 1 |
| 7th week | 2 | 1 | 3 |
| 8th week | 4 | 5 | 5 |
| 9th week | 2 | 2 | 4 |
| 10th week | 3 | 3 | 0 |
| 11th week | 0 | 3 | 2 |
| 12th week | 3 | 1 | 1 |
| 13th week | 1 | 0 | 0 |
| n = 15 | n = 15 | n = 16 |
| Characteristics * | Normal Pregnancy n = 15 | Spontaneous Miscarriage n= 15 | Recurrent Miscarriage n= 16 | p Value (Kruskal Wallis Test) |
|---|---|---|---|---|
| Maternal age (years) | 31.18 ± 8.06 (18.7−43.3) | 37.8 ± 4.51 (29.2–43.2) | 35.76 ± 4.8 (29.5–46.9) | 0.049 |
| Gestational age (weeks) | 9.53 ± 1.95 (7–13) | 8.4 ± 1.89 (7–12) | 9.3 ± 1.49 (4–12) | 0.276 |
| Gravidity | 4 ± 1.8 (1–7) | 1.6 ± 0.9 (1–4) | 3.1 ± 1.1 (2–5) | 0.001 |
| Parity | 2 ± 1.1 (0–4) | 0.9 ± 0.8 (0–2) | 0.3 ± 0.6 (0–2) | 0.003 |
| Antibody | Isotype | Clone | Dilution | Source |
|---|---|---|---|---|
| PPARγ a,b | rabbit IgG | polyclonal | 1:500 in PBS a 1:500 in Dako b | Abcam Serotec, Cambridge, UK DAKO (S322); Carpenteira, CA, USA |
| CD 68 a,b | mouse IgG1 | monoclonal | 1:8000 in PBS a 1:8000 in Dako b | Sigma Aldrich (CL1346), St. Louis, MO, USA DAKO (S322); Carpenteira, CA, USA |
| iNOS b | Rabbit IgG | polyclonal | 1:3000 in Dako | Thermo Scientific, (NPA3-030A) DAKO (S322); Carpenteira, CA, USA |
| TLR2 b | Rabbit IgG | polyclonal | 1:750 in Dako | Sigma Aldrich, St. Louis, MO, USA Dako (S322); Carpenteira, CA, USA |
| CCL1 b | Rabbit IgG | polyclonal | 1: 50 in Dako | Sigma Aldrich, St. Louis, , MO, USA Dako (S322); Carpenteira, CA, USA |
| CX3CR1 b | Rabbit IgG | polyclonal | 1: 400 in Dako | Abcam Serotec, Cambridge, UK Dako (S322); Carpenteira, CA, USA |
| Cy-2 or -3 b | goat IgG anti-mouse | polyclonal | 1:500 b | Dianova, Hamburg, Germany |
| Cy-2 or -3 b | goat IgG anti-rabbit | polyclonal | 1:100 b | Dianova, Hamburg, Germany |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolben, T.M.; Rogatsch, E.; Vattai, A.; Hester, A.; Kuhn, C.; Schmoeckel, E.; Mahner, S.; Jeschke, U.; Kolben, T. PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas. Int. J. Mol. Sci. 2018, 19, 1872. https://doi.org/10.3390/ijms19071872
Kolben TM, Rogatsch E, Vattai A, Hester A, Kuhn C, Schmoeckel E, Mahner S, Jeschke U, Kolben T. PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas. International Journal of Molecular Sciences. 2018; 19(7):1872. https://doi.org/10.3390/ijms19071872
Chicago/Turabian StyleKolben, Theresa Maria, Elisabeth Rogatsch, Aurelia Vattai, Anna Hester, Christina Kuhn, Elisa Schmoeckel, Sven Mahner, Udo Jeschke, and Thomas Kolben. 2018. "PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas" International Journal of Molecular Sciences 19, no. 7: 1872. https://doi.org/10.3390/ijms19071872
APA StyleKolben, T. M., Rogatsch, E., Vattai, A., Hester, A., Kuhn, C., Schmoeckel, E., Mahner, S., Jeschke, U., & Kolben, T. (2018). PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas. International Journal of Molecular Sciences, 19(7), 1872. https://doi.org/10.3390/ijms19071872






