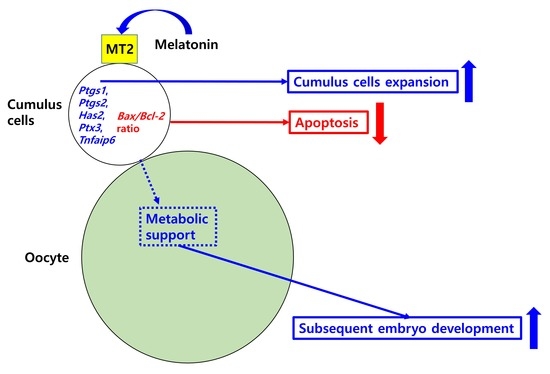
Stimulatory Effects of Melatonin on Porcine In Vitro Maturation Are Mediated by MT2 Receptor
Abstract
:1. Introduction
2. Results
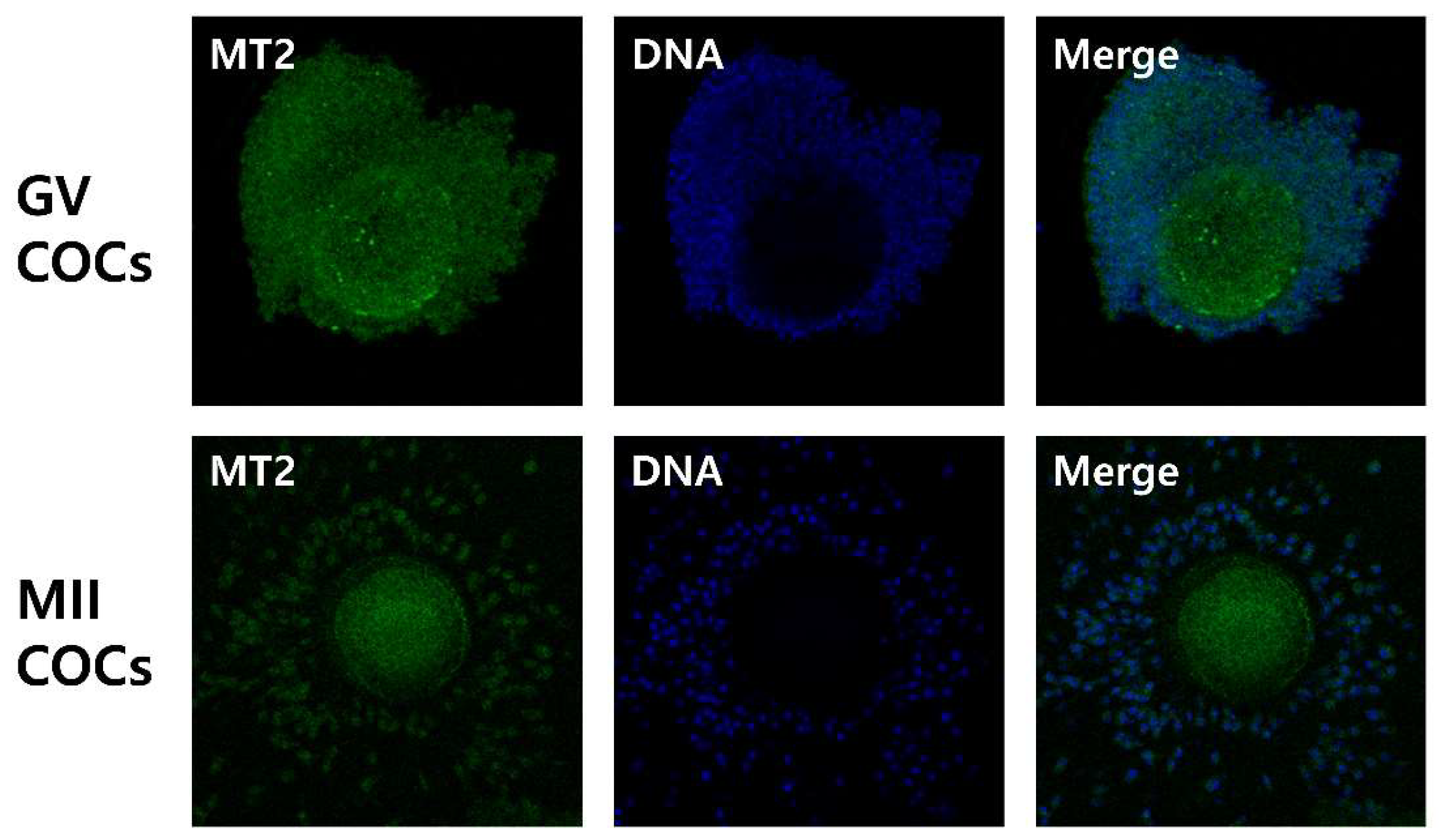
2.1. Detection of Melatonin Membrane Receptor 2 (MT2) in Porcine Cumulus-Oocyte Complexes
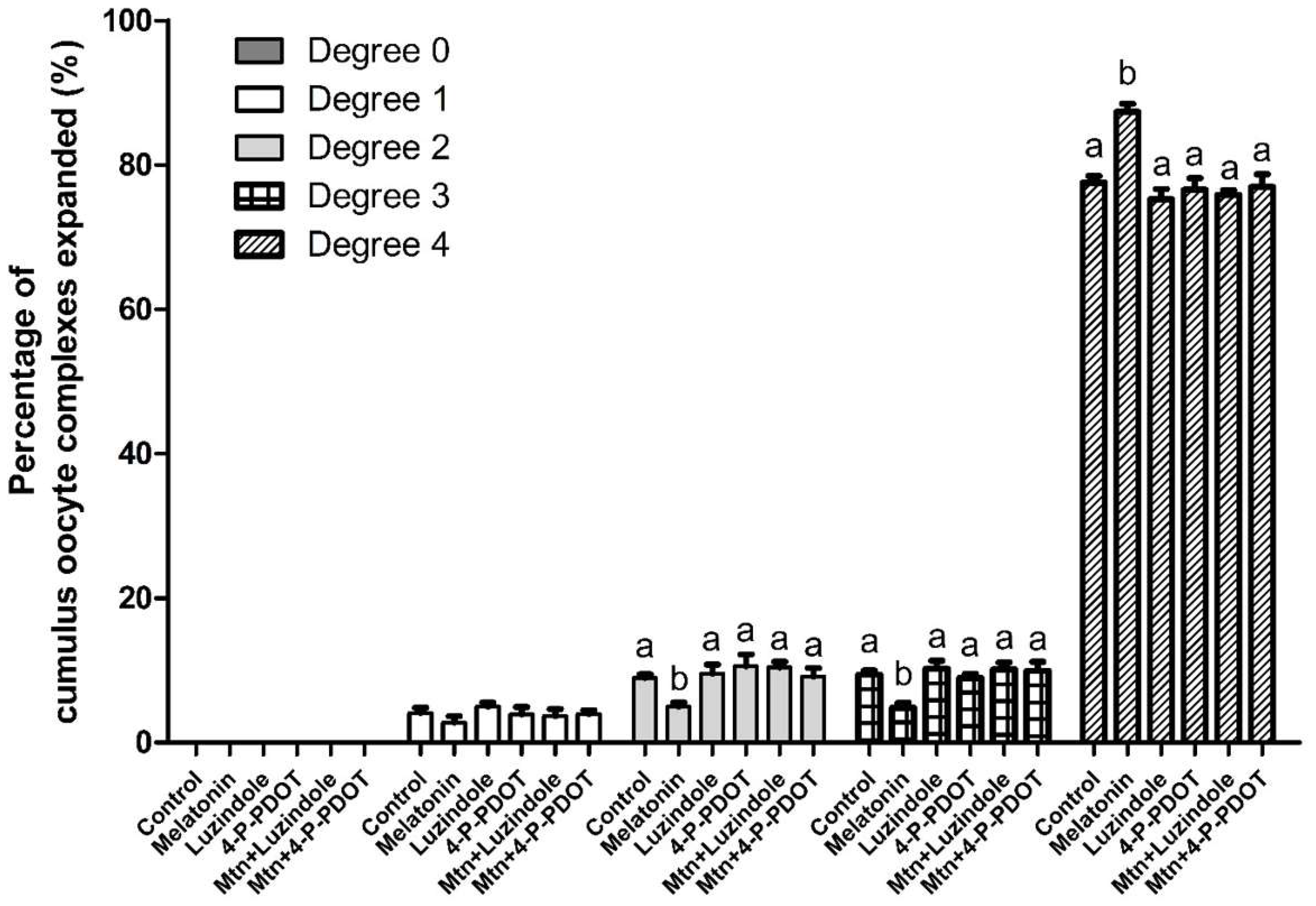
2.2. Effects of Melatonin with or without Selective MT2 Antagonists on Cumulus Expansion
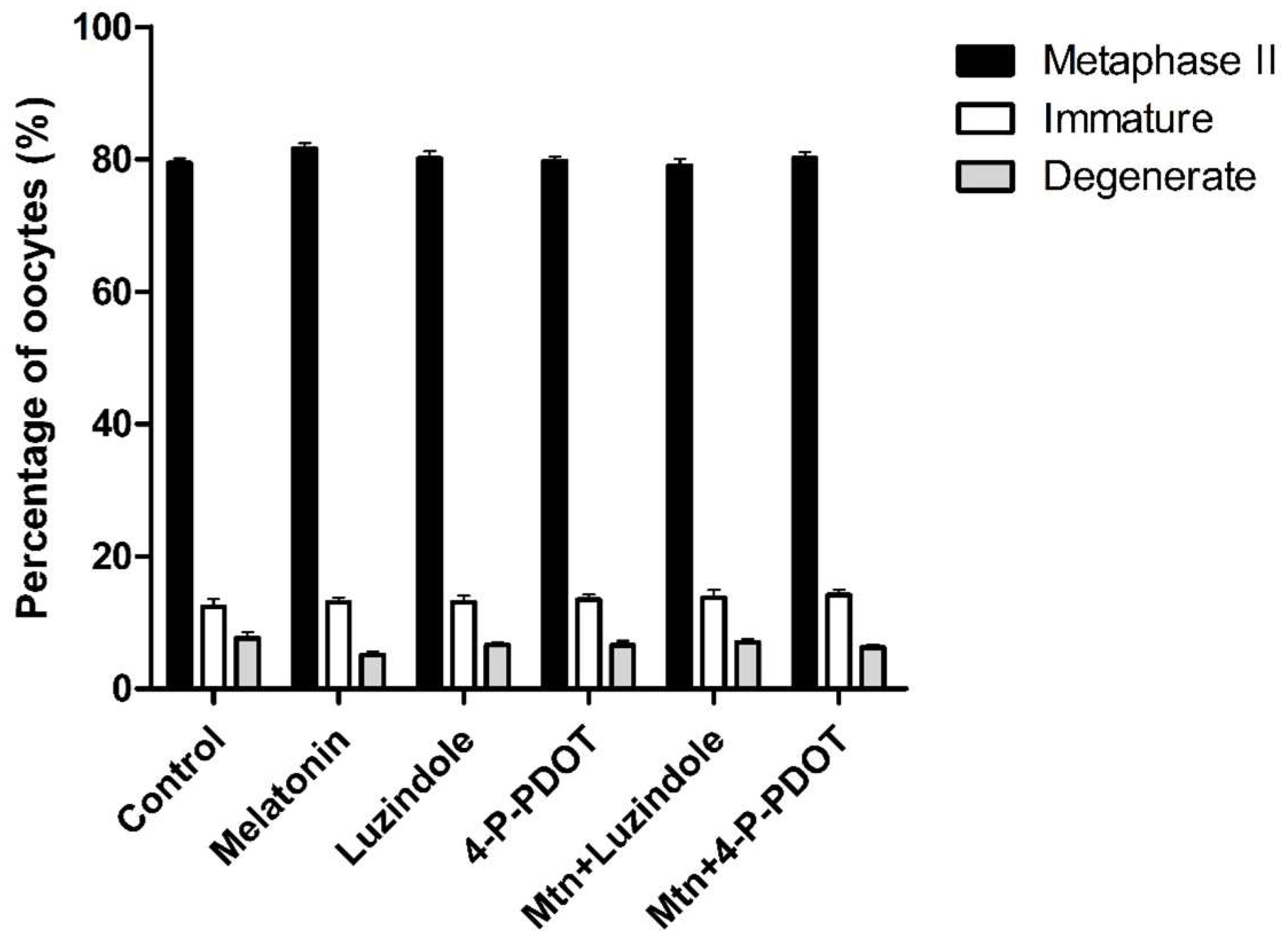
2.3. Effects of Melatonin with or without Selective MT2 Antagonists on Oocyte Nuclear Maturation
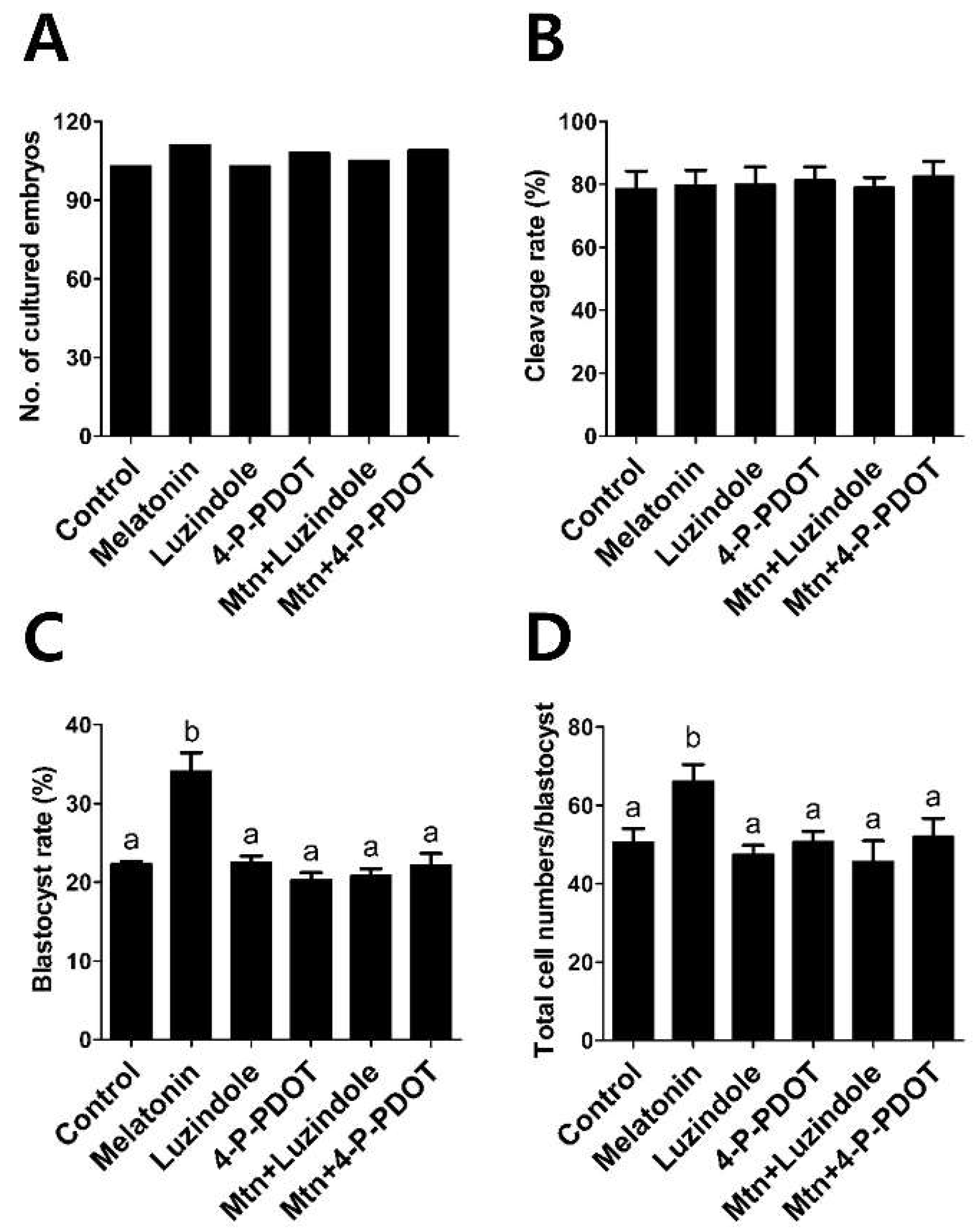
2.4. Effects of Melatonin with or without Selective MT2 Antagonists Treatment during IVM on Subsequent Development of PA Embryos
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Oocyte Recovery and In Vitro Maturation
4.3. Detection of Melatonin Receptor 2 (MT2) in COCs by Immunofluorescence
4.4. Cumulus Expansion Assessment
4.5. Assessment of Nuclear Maturation
4.6. Parthenogenetic Activation (PA) of Oocytes
4.7. Embryo Evaluation and Total Cell Counts
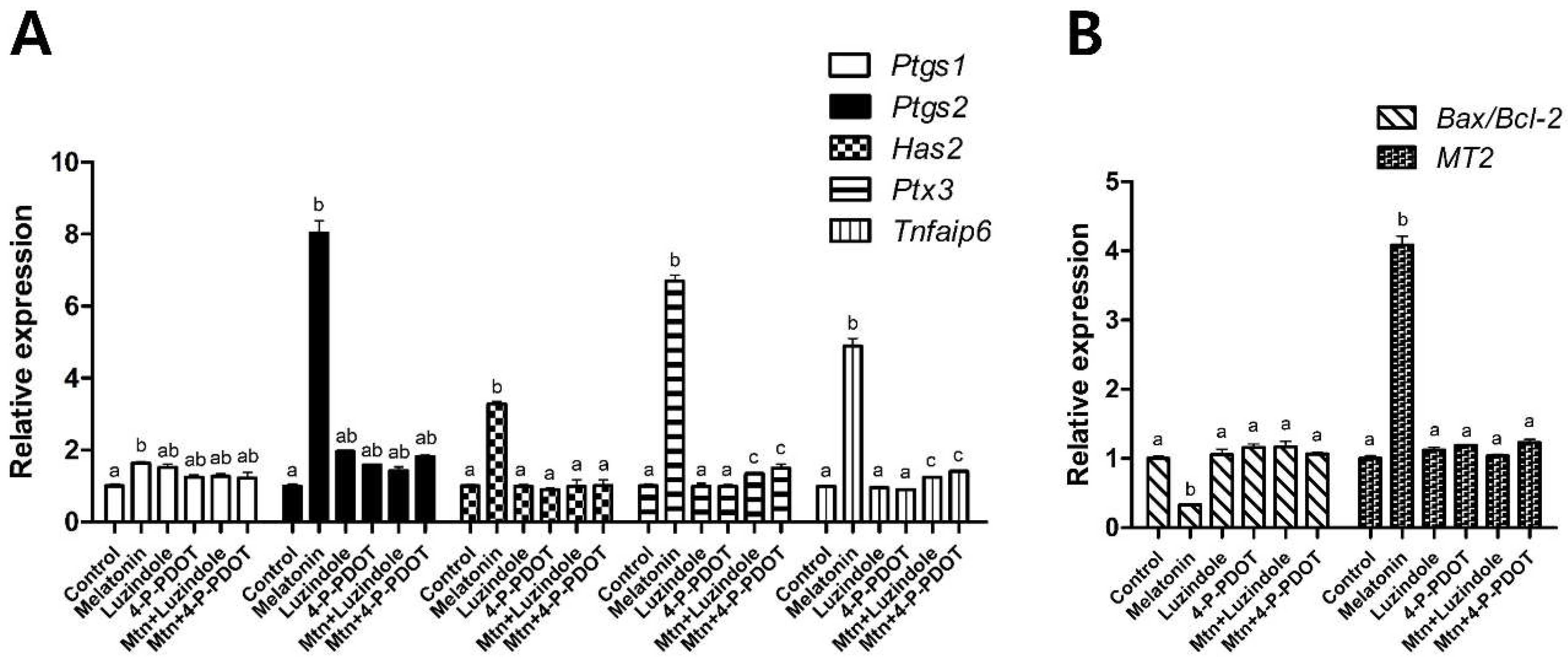
4.8. Gene Expression Analysis by Real-Time PCR
4.9. Experimental Design
4.10. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| Ptgs1 | Prostaglandin-endoperoxide synthase 1 |
| Ptgs2 | Prostaglandin-endoperoxide synthase 2 |
| Has2 | Hyaluronan synthase 2 |
| Ptx3 | Pentraxin 3 |
| Tnfaip6 | Tumor necrosis factor α induced protein 6 |
| Bcl-2 | B cell leukemia/lymphoma 2 |
| Bax | Bcl-2 associated X |
References
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Zhang, M.; Weintraub, S.T.; Cabrera, J.; Sainz, R.M.; Mayo, J.C. Identification of highly elevated levels of melatonin in bone marrow: Its origin and significance. Biochim. Biophys. Acta 1999, 1472, 206–214. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Calvo, J.R.; Abreu, P.; Lardone, P.J.; Garcia-Maurino, S.; Reiter, R.J.; Guerrero, J.M. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: Possible role as intracrine, autocrine, and/or paracrine substance. FASEB J. 2004, 18, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Iuvone, P.M.; Tosini, G.; Pozdeyev, N.; Haque, R.; Klein, D.C.; Chaurasia, S.S. Circadian clocks, clock networks, arylalkylamine N-acetyltransferase, and melatonin in the retina. Prog. Retin. Eye Res. 2005, 24, 433–456. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Zhuang, J.; Zhu, H.Y.; Shen, Y.X.; Tan, Z.L.; Zhou, J.N. Cultured rat cortical astrocytes synthesize melatonin: Absence of a diurnal rhythm. J. Pineal Res. 2007, 43, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Naranjo, M.C.; Guerrero, J.M.; Rubio, A.; Lardone, P.J.; Carrillo-Vico, A.; Carrascosa-Salmoral, M.P.; Jimenez-Jorge, S.; Arellano, M.V.; Leal-Noval, S.R.; Leal, M.; et al. Melatonin biosynthesis in the thymus of humans and rats. Cell. Mol. Life Sci. 2007, 64, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tamura, H.; Tan, D.X.; Xu, X.Y. Melatonin and the circadian system: Contributions to successful female reproduction. Fertil. Steril. 2014, 102, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.M.; Tian, X.Z.; Zhou, G.B.; Wang, L.; Gao, C.; Zhu, S.E.; Zeng, S.M.; Tian, J.H.; Liu, G.S. Melatonin exists in porcine follicular fluid and improves in vitro maturation and parthenogenetic development of porcine oocytes. J. Pineal Res. 2009, 47, 318–323. [Google Scholar] [CrossRef] [PubMed]
- El-Raey, M.; Geshi, M.; Somfai, T.; Kaneda, M.; Hirako, M.; Abdel-Ghaffar, A.E.; Sosa, G.A.; El-Roos, M.E.; Nagai, T. Evidence of melatonin synthesis in the cumulus oocyte complexes and its role in enhancing oocyte maturation in vitro in cattle. Mol. Reprod. Dev. 2011, 78, 250–262. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, K.; Itoh, M.T.; Takahashi, N.; Tarumi, W.; Ishizuka, B. The rat oocyte synthesises melatonin. Reprod. Fertil. Dev. 2013, 25, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Nakamura, Y.; Korkmaz, A.; Manchester, L.C.; Tan, D.X.; Sugino, N.; Reiter, R.J. Melatonin and the ovary: Physiological and pathophysiological implications. Fertil. Steril. 2009, 92, 328–343. [Google Scholar] [CrossRef] [PubMed]
- Pacchiarotti, A.; Carlomagno, G.; Antonini, G.; Pacchiarotti, A. Effect of myo-inositol and melatonin versus myo-inositol, in a randomized controlled trial, for improving in vitro fertilization of patients with polycystic ovarian syndrome. Gynecol. Endocrinol. 2016, 32, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Melatonin: Lowering the High Price of Free Radicals. Physiology 2000, 15, 246–250. [Google Scholar] [CrossRef]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Grossini, E.; Molinari, C.; Uberti, F.; Mary, D.A.; Vacca, G.; Caimmi, P.P. Intracoronary melatonin increases coronary blood flow and cardiac function through beta-adrenoreceptors, MT1/MT2 receptors, and nitric oxide in anesthetized pigs. J. Pineal Res. 2011, 51, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Rivara, S.; Lorenzi, S.; Mor, M.; Plazzi, P.V.; Spadoni, G.; Bedini, A.; Tarzia, G. Analysis of structure-activity relationships for MT2 selective antagonists by melatonin MT1 and MT2 receptor models. J. Med. Chem. 2005, 48, 4049–4060. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Cunha, M.C.; Mesquita, L.G.; Bressan, F.; Collado, M.D.; Balieiro, J.C.; Schwarz, K.R.; de Castro, F.C.; Watanabe, O.Y.; Watanabe, Y.F.; de Alencar Coelho, L.; et al. Effects of melatonin during IVM in defined medium on oocyte meiosis, oxidative stress, and subsequent embryo development. Theriogenology 2016, 86, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, Z.; He, C.; Zhu, K.; Xu, Z.; Ma, T.; Tao, J.; Liu, G. Melatonin protects porcine oocyte in vitro maturation from heat stress. J. Pineal Res. 2015, 59, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, R.B.; Ritter, L.J.; Armstrong, D.T. Oocyte-somatic cell interactions during follicle development in mammals. Anim. Reprod. Sci. 2004, 82–83, 431–446. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.Q.; Sugiura, K.; Eppig, J.J. Mouse oocyte control of granulosa cell development and function: Paracrine regulation of cumulus cell metabolism. Semin. Reprod. Med. 2009, 27, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Manabe, N.; Goto, Y.; Matsuda-Minehata, F.; Inoue, N.; Maeda, A.; Sakamaki, K.; Miyano, T. Regulation mechanism of selective atresia in porcine follicles: Regulation of granulosa cell apoptosis during atresia. J. Reprod. Dev. 2004, 50, 493–514. [Google Scholar] [CrossRef] [PubMed]
- Eppig, J.J. Intercommunication between mammalian oocytes and companion somatic cells. Bioessays 1991, 13, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jin, J.X.; Taweechaipaisankul, A.; Kim, G.A.; Ahn, C.; Lee, B.C. Melatonin influences the sonic hedgehog signaling pathway in porcine cumulus oocyte complexes. J. Pineal Res. 2017, 63. [Google Scholar] [CrossRef] [PubMed]
- Russell, M.C.; Cowan, R.G.; Harman, R.M.; Walker, A.L.; Quirk, S.M. The hedgehog signaling pathway in the mouse ovary. Biol. Reprod. 2007, 77, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Marchal, R.; Caillaud, M.; Martoriati, A.; Gerard, N.; Mermillod, P.; Goudet, G. Effect of growth hormone (GH) on in vitro nuclear and cytoplasmic oocyte maturation, cumulus expansion, hyaluronan synthases, and connexins 32 and 43 expression, and GH receptor messenger RNA expression in equine and porcine species. Biol. Reprod. 2003, 69, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- He, Y.M.; Deng, H.H.; Shi, M.H.; Bodinga, B.M.; Chen, H.L.; Han, Z.S.; Jiang, Z.L.; Li, Q.W. Melatonin modulates the functions of porcine granulosa cells via its membrane receptor MT2 in vitro. Anim. Reprod. Sci. 2016, 172, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Browning, C.; Beresford, I.; Fraser, N.; Giles, H. Pharmacological characterization of human recombinant melatonin MT(1) and MT(2) receptors. Br. J. Pharmacol. 2000, 129, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Tunstall, R.R.; Shukla, P.; Grazul-Bilska, A.; Sun, C.; O’Rourke, S.T. MT2 receptors mediate the inhibitory effects of melatonin on nitric oxide-induced relaxation of porcine isolated coronary arteries. J. Pharmacol. Exp. Ther. 2011, 336, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Vanderhyden, B.C.; Caron, P.J.; Buccione, R.; Eppig, J.J. Developmental pattern of the secretion of cumulus expansion-enabling factor by mouse oocytes and the role of oocytes in promoting granulosa cell differentiation. Dev. Biol. 1990, 140, 307–317. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.A.; Manchester, L.C.; Tan, D.X. Peripheral reproductive organ health and melatonin: Ready for prime time. Int. J. Mol. Sci. 2013, 14, 7231–7272. [Google Scholar] [CrossRef] [PubMed]
- Abeydeera, L.R.; Wang, W.H.; Prather, R.S.; Day, B.N. Maturation in vitro of pig oocytes in protein-free culture media: Fertilization and subsequent embryo development in vitro. Biol. Reprod. 1998, 58, 1316–1320. [Google Scholar] [CrossRef] [PubMed]
- Algriany, O.; Bevers, M.; Schoevers, E.; Colenbrander, B.; Dieleman, S. Follicle size-dependent effects of sow follicular fluid on in vitro cumulus expansion, nuclear maturation and blastocyst formation of sow cumulus oocytes complexes. Theriogenology 2004, 62, 1483–1497. [Google Scholar] [CrossRef] [PubMed]
- Sutton-McDowall, M.L.; Gilchrist, R.B.; Thompson, J.G. Cumulus expansion and glucose utilisation by bovine cumulus-oocyte complexes during in vitro maturation: The influence of glucosamine and follicle-stimulating hormone. Reproduction 2004, 128, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Russell, P.T.; Larsen, W.J. Functional significance of cumulus expansion in the mouse: Roles for the preovulatory synthesis of hyaluronic acid within the cumulus mass. Mol. Reprod. Dev. 1993, 34, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Kumei, Y.; Morita, S.; Shimokawa, H.; Ohya, K.; Shinomiya, K. Antagonism between apoptotic (Bax/Bcl-2) and anti-apoptotic (IAP) signals in human osteoblastic cells under vector-averaged gravity condition. Ann. N. Y. Acad. Sci. 2003, 1010, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Park, E.J.; Moon, J.H.; Kim, S.J.; Song, K.; Lee, B.C. Sequential treatment with resveratrol-trolox improves development of porcine embryos derived from parthenogenetic activation and somatic cell nuclear transfer. Theriogenology 2015, 84, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, E.S.; Racowsky, C. In Vitro Fertilization: A Comprehensive Guide; Springer: New York, NY, USA, 2012; p. 240. ISBN 978-971-4419-9847-7. [Google Scholar]
- Nomura, M.; Iwase, A.; Furui, K.; Kitagawa, T.; Matsui, Y.; Yoshikawa, M.; Kikkawa, F. Preferable correlation to blastocyst development and pregnancy rates with a new embryo grading system specific for day 3 embryos. J. Assist. Reprod. Genet. 2007, 24, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Knijn, H.M.; Gjorret, J.O.; Vos, P.L.; Hendriksen, P.J.; van der Weijden, B.C.; Maddox-Hyttel, P.; Dieleman, S.J. Consequences of in vivo development and subsequent culture on apoptosis, cell number, and blastocyst formation in bovine embryos. Biol. Reprod. 2003, 69, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Wang, F.; He, C.; Zhang, L.; Tan, D.; Reiter, R.J.; Xu, J.; Ji, P.; Liu, G. Beneficial effects of melatonin on bovine oocytes maturation: A mechanistic approach. J. Pineal Res. 2014, 57, 239–247. [Google Scholar] [CrossRef] [PubMed]
| Gene | Primer Sequences (5′-3′) | Product Size (bp) | GenBank Accession Number |
|---|---|---|---|
| GAPDH | F: GTCGGTTGTGGATCTGACCT R: TTGACGAAGTGGTCGTTGAG | 207 | NM_001206359 |
| Ptgs1 | F: CAACACGGCACACGACTACA R: CTGCTTCTTCCCTTTGGTCC | 121 | XM_001926129 |
| Ptgs2 | F: ACAGGGCCATGGGGTGGACT R: CCACGGCAAAGCGGAGGTGT | 194 | NM_214321 |
| Has2 | F: AGTTTATGGGCAGCCAATGTAGTT R: GCACTTGGACCGAGCTGTGT | 101 | AB050389 |
| Ptx3 | F: GGCCAGGGATGAATTTTAC R: GCTATCCTCTCCAACAAGTGA | 185 | NM_001244783 |
| Tnfaip6 | F: AGAAGCGAAAGATGGGATGCT R: CATTTGGGAAGCCTGGAGATT | 106 | NM_001159607 |
| Bax | F: TGCCTCAGGATGCATCTACC R: AAGTAGAAAAGCGCGACCAC | 199 | XM_003127290 |
| Bcl-2 | F: AGGGCATTCAGTGACCTGAC R: CGATCCGACTCACCAATACC | 193 | NM_214285 |
| MT2 | F: CGGTCGTGTGCTTCTGTTACC R: AGCAGACGGCGAAGATCA | 151 | XM_013979266 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Jin, J.-X.; Taweechaipaisankul, A.; Kim, G.-A.; Lee, B.-C. Stimulatory Effects of Melatonin on Porcine In Vitro Maturation Are Mediated by MT2 Receptor. Int. J. Mol. Sci. 2018, 19, 1581. https://doi.org/10.3390/ijms19061581
Lee S, Jin J-X, Taweechaipaisankul A, Kim G-A, Lee B-C. Stimulatory Effects of Melatonin on Porcine In Vitro Maturation Are Mediated by MT2 Receptor. International Journal of Molecular Sciences. 2018; 19(6):1581. https://doi.org/10.3390/ijms19061581
Chicago/Turabian StyleLee, Sanghoon, Jun-Xue Jin, Anukul Taweechaipaisankul, Geon-A Kim, and Byeong-Chun Lee. 2018. "Stimulatory Effects of Melatonin on Porcine In Vitro Maturation Are Mediated by MT2 Receptor" International Journal of Molecular Sciences 19, no. 6: 1581. https://doi.org/10.3390/ijms19061581
APA StyleLee, S., Jin, J.-X., Taweechaipaisankul, A., Kim, G.-A., & Lee, B.-C. (2018). Stimulatory Effects of Melatonin on Porcine In Vitro Maturation Are Mediated by MT2 Receptor. International Journal of Molecular Sciences, 19(6), 1581. https://doi.org/10.3390/ijms19061581