Effect of Melatonin on Rat Heart Mitochondria in Acute Heart Failure in Aged Rats
Abstract
:1. Introduction
2. Results
2.1. Histological Analysis of Cryosections of the Left Ventricle of Rat Heart
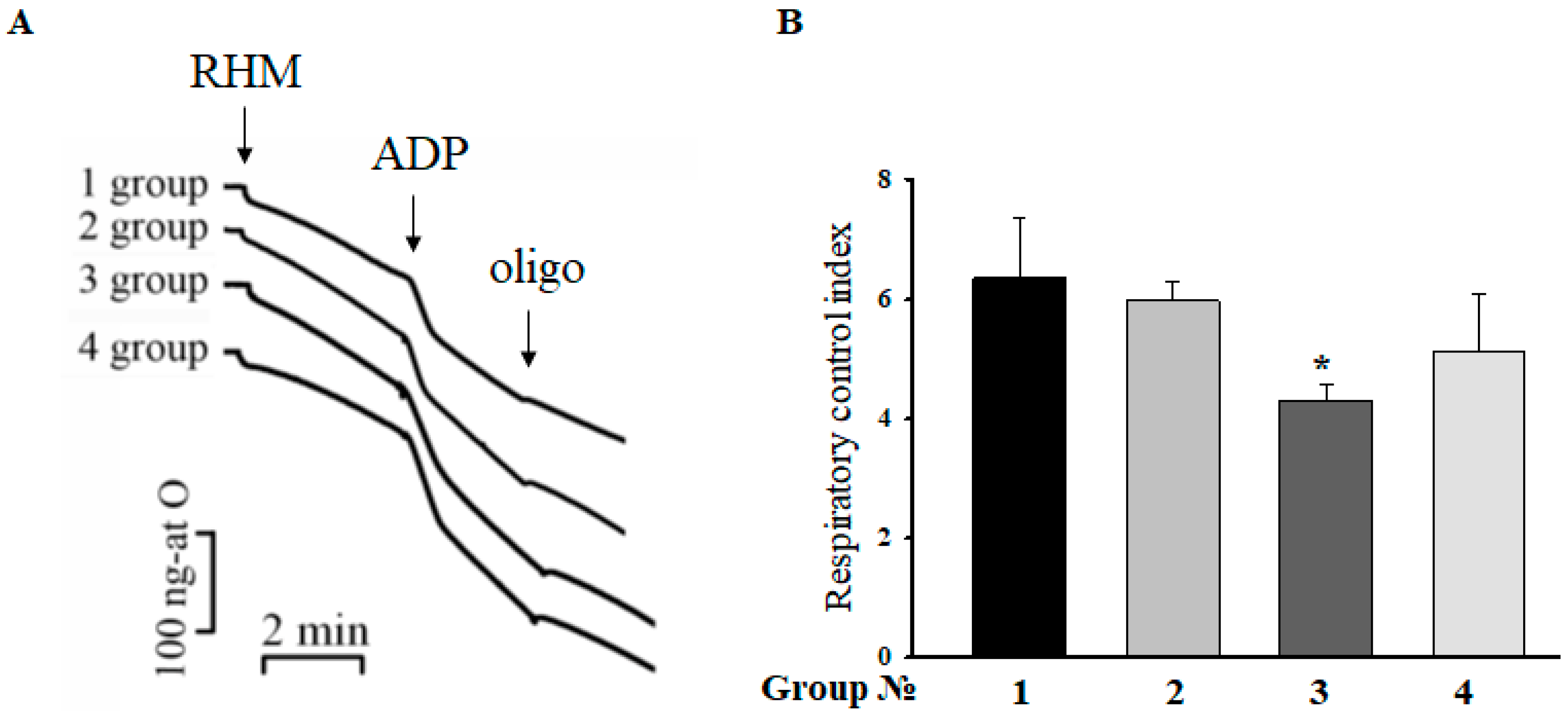
2.2. Respiratory Control Index (RCI) of RHM
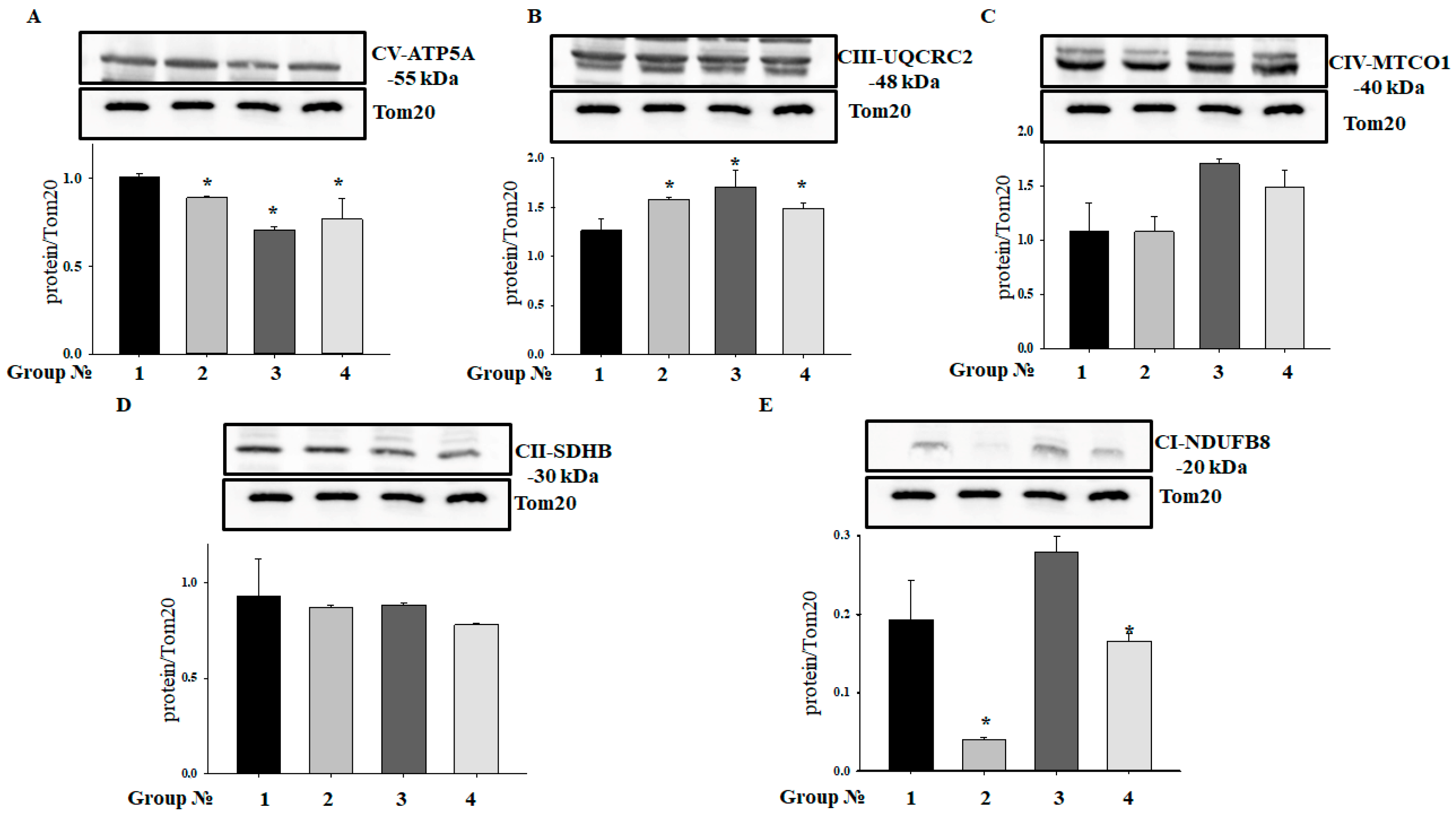
2.3. Effect of MEL and ISO on the Level of Enzymes in the Electron Transport Chain in RHM
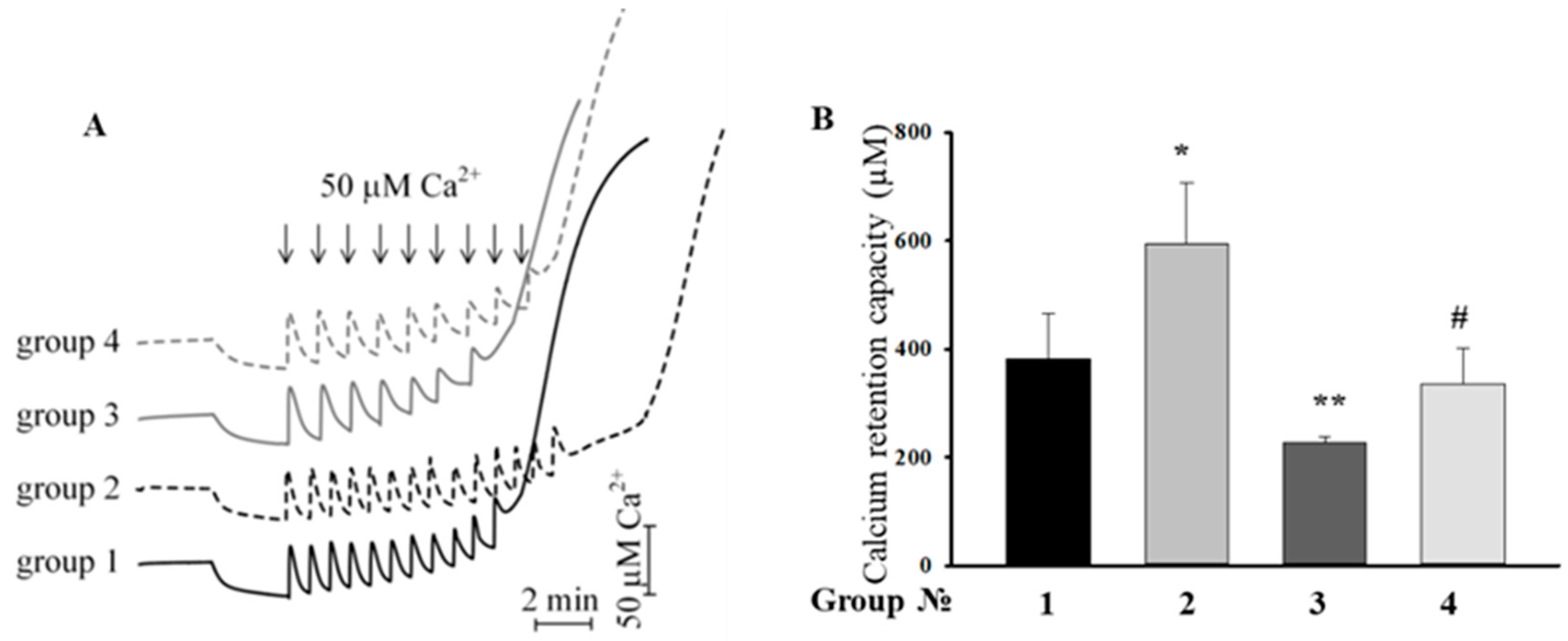
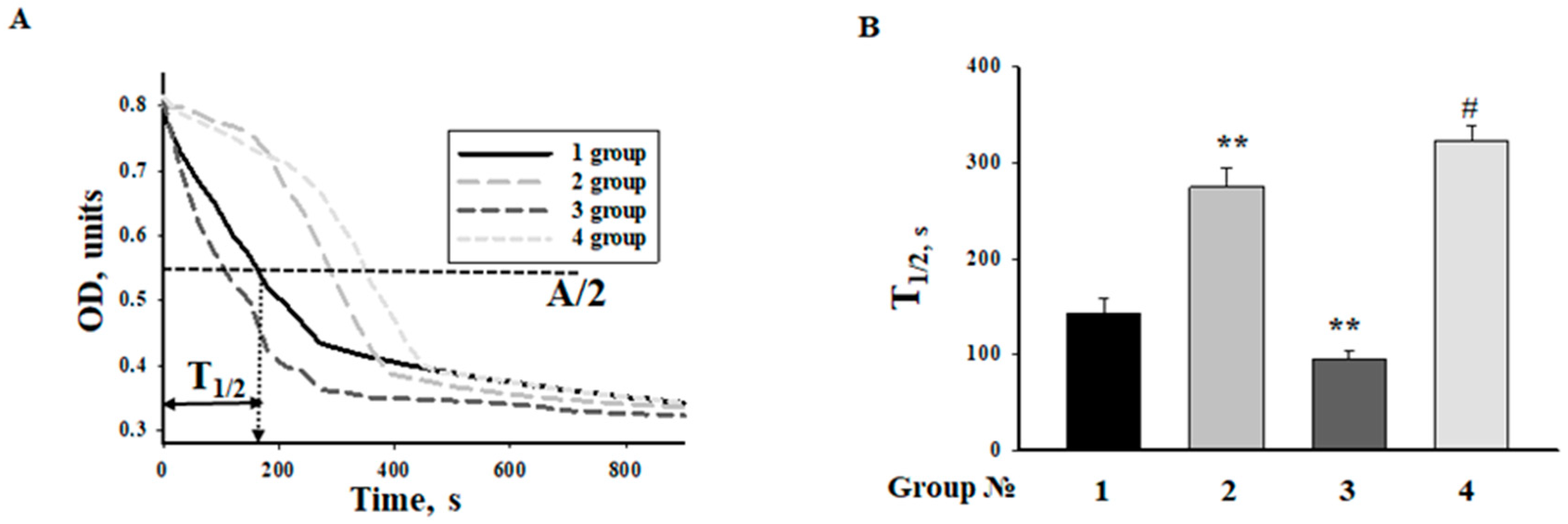
2.4. Effect of the MEL, ISO, and the Combined Effect of MEL and ISO on Changes in the Calcium Retention Capacity
2.5. Effect of MEL and ISO on the Swelling of RHM
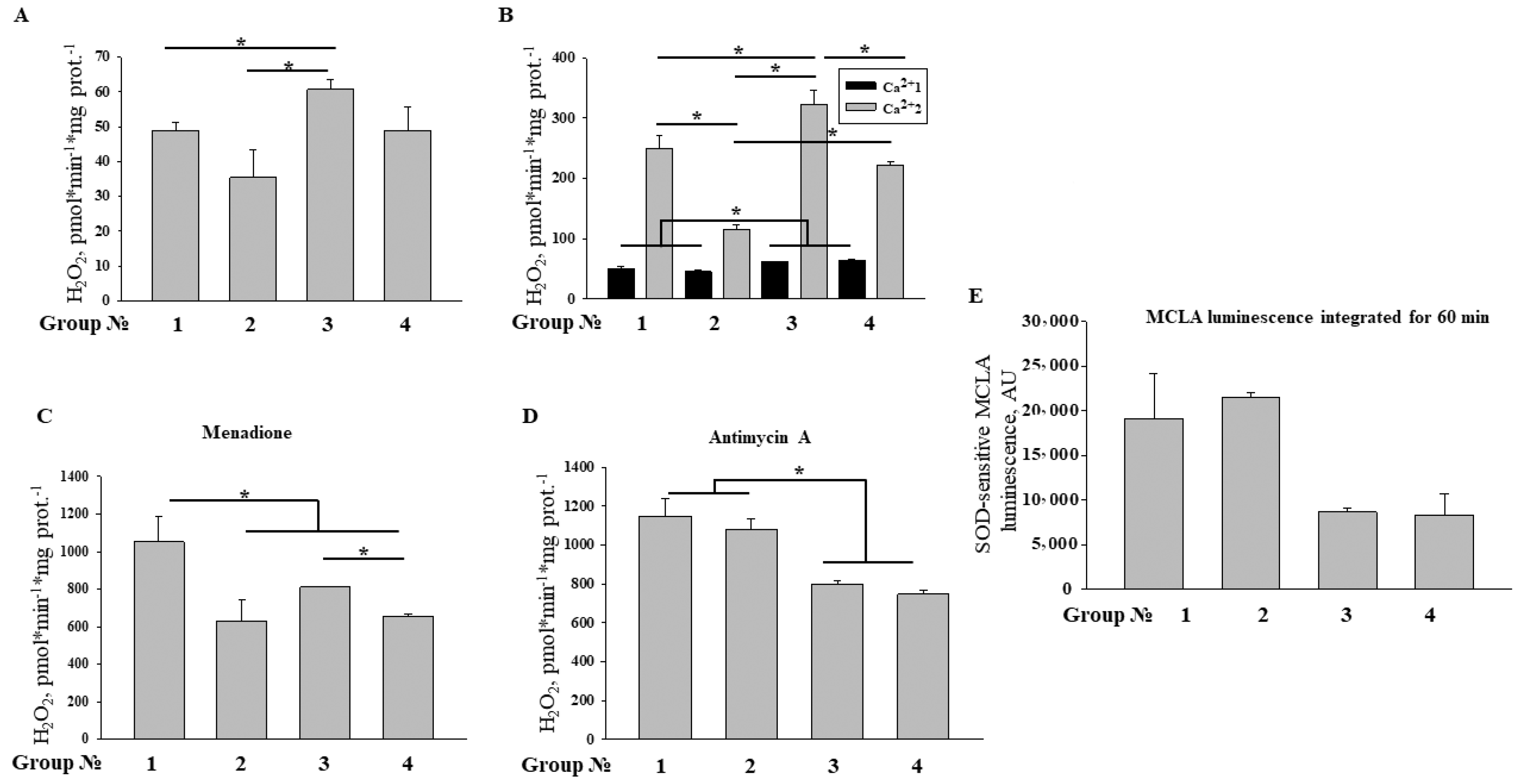
2.6. Effect of MEL and ISO on ROS Production upon mPTP Opening in RHM
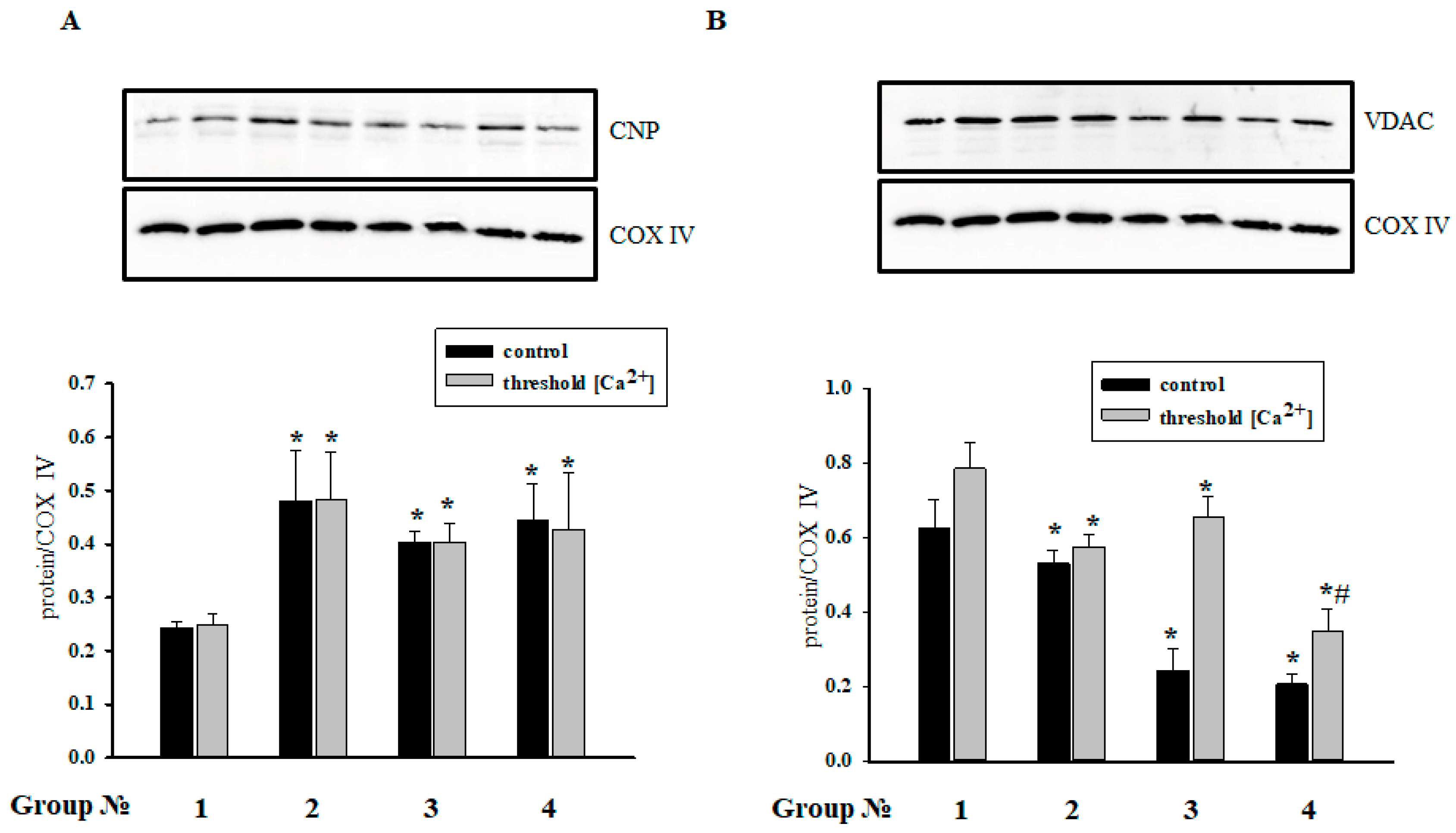
2.7. The Effect of MEL and ISO on the Levels of CNPase and VDAC in RHM
3. Discussion
4. Materials and Methods
4.1. Animal and Treatment
4.2. Histological Analysis
4.3. Isolation of Rat Heart Mitochondria
4.4. Evaluation of Mitochondrial Functions
4.5. ROS Measurement in Heart Mitochondria
4.6. Sample Preparation, Electrophoresis, and Immunoblotting of Mitochondrial Proteins
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RHM | rat heart mitochondria |
| VDAC | voltage dependent anion cannel |
| CNPase | 2′,3′-cyclic nucleotide-3′-phosphodiesterase |
| ROS | reactive oxygen species |
| mPTP | mitochondrial permeability transition pore |
| ISO | isoprenaline hydrochloride |
| MEL | melatonin |
| RCI | respiratory control index |
| ETC | electronic transport chain |
References
- Katz, A.M.; Konstam, M.A. Heart Failure: Pathophysiology, Molecular Biology, and Clinical Management, 2nd ed.; DeStefano, F.R., Ed.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2009. [Google Scholar]
- Dickstein, K.; Cohen-Solal, A.; Filippatos, G.; McMurray, J.J.; Ponikowski, P.; Poole-Wilson, P.A.; Stromberg, A.; van Veldhuisen, D.J.; Atar, D.; Hoes, A.W.; et al. Esc guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: The task force for the diagnosis and treatment of acute and chronic heart failure 2008 of the european society of cardiology. Developed in collaboration with the heart failure association of the esc (hfa) and endorsed by the european society of intensive care medicine (esicm). Eur. Heart J. 2008, 29, 2388–2442. [Google Scholar] [PubMed]
- Griffiths, E.J. Mitochondria and heart disease. Adv. Exp. Med. Biol. 2012, 942, 249–267. [Google Scholar] [PubMed]
- Rostovtseva, T.K.; Bezrukov, S.M. Vdac inhibition by tubulin and its physiological implications. Biochim. Biophys. Acta 2012, 1818, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Azarashvili, T.; Krestinina, O.; Galvita, A.; Grachev, D.; Baburina, Y.; Stricker, R.; Evtodienko, Y.; Reiser, G. Ca2+-dependent permeability transition regulation in rat brain mitochondria by 2′,3′-cyclic nucleotides and 2′,3′-cyclic nucleotide 3′-phosphodiesterase. Am. J. Physiol. Cell Physiol. 2009, 296, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- Krestinina, O.; Azarashvili, T.; Baburina, Y.; Galvita, A.; Grachev, D.; Stricker, R.; Reiser, G. In aging, the vulnerability of rat brain mitochondria is enhanced due to reduced level of 2′,3′-cyclic nucleotide-3′-phosphodiesterase (cnp) and subsequently increased permeability transition in brain mitochondria in old animals. Neurochem. Int. 2015, 80, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Macias, M.; Escames, G.; Leon, J.; Acuna-Castroviejo, D. Melatonin but not vitamins c and e maintains glutathione homeostasis in t-butyl hydroperoxide-induced mitochondrial oxidative stress. FASEB J. 2000, 14, 1677–1679. [Google Scholar] [CrossRef] [PubMed]
- Menendez-Pelaez, A.; Reiter, R.J. Distribution of melatonin in mammalian tissues: The relative importance of nuclear versus cytosolic localization. J. Pineal Res. 1993, 15, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Suofu, Y.; Li, W.; Jean-Alphonse, F.G.; Jia, J.; Khattar, N.K.; Li, J.; Baranov, S.V.; Leronni, D.; Mihalik, A.C.; He, Y.; et al. Dual role of mitochondria in producing melatonin and driving gpcr signaling to block cytochrome c release. Proc. Natl. Acad. Sci. USA 2017, 114, E7997–E8006. [Google Scholar] [CrossRef] [PubMed]
- Brzezinski, A. Melatonin in humans. N. Engl. J. Med. 1997, 336, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Karasek, M.; Reiter, R.J. Melatonin and aging. Neuro Endocrinol. Lett. 2002, 23 (Suppl. 1), 14–16. [Google Scholar] [PubMed]
- Zhao, Z.Y.; Xie, Y.; Fu, Y.R.; Bogdan, A.; Touitou, Y. Aging and the circadian rhythm of melatonin: A cross-sectional study of Chinese subjects 30–110 yr of age. Chronobiol. Int. 2002, 19, 1171–1182. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J. Melatonin. Clin. Endocrinol. Oxf. 1988, 29, 205–229. [Google Scholar] [CrossRef] [PubMed]
- Scholtens, R.M.; van Munster, B.C.; van Kempen, M.F.; de Rooij, S.E. Physiological melatonin levels in healthy older people: A systematic review. J. Psychosom. Res. 2016, 86, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Baburina, Y.; Odinokova, I.; Azarashvili, T.; Akatov, V.; Lemasters, J.J.; Krestinina, O. 2′,3′-Cyclic nucleotide 3′-phosphodiesterase as a messenger of protection of the mitochondrial function during melatonin treatment in aging. Biochim. Biophys. Acta 2017, 1859, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Simko, F.; Paulis, L. Melatonin as a potential antihypertensive treatment. J. Pineal Res. 2007, 42, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Upaganlawar, A.; Zalawadia, R.; Balaraman, R. Cardioprotective effect of melatonin against isoproterenol induced myocardial infarction in rats: A biochemical, electrocardiographic and histoarchitectural evaluation. Eur. J. Pharmacol. 2010, 644, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Sahna, E.; Acet, A.; Ozer, M.K.; Olmez, E. Myocardial ischemia-reperfusion in rats: Reduction of infarct size by either supplemental physiological or pharmacological doses of melatonin. J. Pineal Res. 2002, 33, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Girouard, H.; Denault, C.; Chulak, C.; de Champlain, J. Treatment by N-acetylcysteine and melatonin increases cardiac baroreflex and improves antioxidant reserve. Am. J. Hypertens. 2004, 17, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Jiang, S.; Dong, Y.; Fan, C.; Zhao, L.; Yang, X.; Li, J.; Di, S.; Yue, L.; Liang, G.; et al. Melatonin prevents cell death and mitochondrial dysfunction via a sirt1-dependent mechanism during ischemic-stroke in mice. J. Pineal Res. 2015, 58, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Ghosh, A.K.; Bandyopadhyay, A.; Basu, A.; Datta, S.; Pattari, S.K.; Reiter, R.J.; Bandyopadhyay, D. Melatonin protects against isoproterenol-induced alterations in cardiac mitochondrial energy-metabolizing enzymes, apoptotic proteins, and assists in complete recovery from myocardial injury in rats. J. Pineal Res. 2012, 53, 166–179. [Google Scholar] [CrossRef] [PubMed]
- Hibaoui, Y.; Roulet, E.; Ruegg, U.T. Melatonin prevents oxidative stress-mediated mitochondrial permeability transition and death in skeletal muscle cells. J. Pineal Res. 2009, 47, 238–252. [Google Scholar] [CrossRef] [PubMed]
- Petrosillo, G.; Fattoretti, P.; Matera, M.; Ruggiero, F.M.; Bertoni-Freddari, C.; Paradies, G. Melatonin prevents age-related mitochondrial dysfunction in rat brain via cardiolipin protection. Rejuvenation Res. 2008, 11, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; O’Neill, R.C.; Park, M.W.; Gravel, M.; Braun, P.E. Mitochondrial localization of cnp2 is regulated by phosphorylation of the N-terminal targeting signal by pkc: Implications of a mitochondrial function for cnp2 in glial and non-glial cells. Mol. Cell Neurosci. 2006, 31, 446–462. [Google Scholar] [CrossRef] [PubMed]
- Baburina, Y.; Azarashvili, T.; Grachev, D.; Krestinina, O.; Galvita, A.; Stricker, R.; Reiser, G. Mitochondrial 2′, 3′-cyclic nucleotide 3′-phosphodiesterase (cnp) interacts with mptp modulators and functional complexes (І–V) coupled with release of apoptotic factors. Neurochem. Int. 2015, 90, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Odinokova, I.V.; Baburina, Y.L.; Kruglov, A.G.; Santalova, I.M.; Azarashvili, T.S.; Krestinina, O.V. Operation of the permeability transition pore in rat heart mitochondria in aging. Biol. Membrany 2018, 35, 42–51. [Google Scholar] [CrossRef]
- Acuna-Castroviejo, D.; Martin, M.; Macias, M.; Escames, G.; Leon, J.; Khaldy, H.; Reiter, R.J. Melatonin, mitochondria, and cellular bioenergetics. J. Pineal Res. 2001, 30, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.G.; Khand, A.; Clark, A. The heart failure epidemic: Exactly how big is it? Eur. Heart J. 2001, 22, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.; Lee, K.S. Study of heart mitochondria and glycolytic metabolism in experimentally induced cardiac failure. Circ. Res. 1962, 10, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Crompton, M. Mitochondria and aging: A role for the permeability transition? Aging Cell 2004, 3, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Di Lisa, F.; Bernardi, P. Mitochondrial function and myocardial aging. A critical analysis of the role of permeability transition. Cardiovasc. Res. 2005, 66, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Szalai, G.; Krishnamurthy, R.; Hajnoczky, G. Apoptosis driven by IP3-linked mitochondrial calcium signals. EMBO J. 1999, 18, 6349–6361. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, P. The mitochondrial permeability transition pore: A mystery solved? Front. Physiol. 2013, 4, 95. [Google Scholar] [CrossRef] [PubMed]
- Krestinina, O.V.; Kruglov, A.G.; Grachev, D.E.; Baburina, Y.L.; Evtodienko, Y.V.; Moshkov, D.A.; Santalova, I.M.; Azarashvili, T.S. Age-related changes of mitochondrial functions under the conditions of ca2+-induced opening of permeability transition pore. Biol. Membr. 2010, 27, 177–183. [Google Scholar]
- Osterholt, M.; Nguyen, T.D.; Schwarzer, M.; Doenst, T. Alterations in mitochondrial function in cardiac hypertrophy and heart failure. Heart Fail. Rev. 2013, 18, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Zago, E.B.; Castilho, R.F.; Vercesi, A.E. The redox state of endogenous pyridine nucleotides can determine both the degree of mitochondrial oxidative stress and the solute selectivity of the permeability transition pore. FEBS Lett. 2000, 478, 29–33. [Google Scholar] [CrossRef]
- Akila, P.; Asaikumar, L.; Vennila, L. Chlorogenic acid ameliorates isoproterenol-induced myocardial injury in rats by stabilizing mitochondrial and lysosomal enzymes. Biomed. Pharmacother. 2017, 85, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Teplova, V.V.; Belosludtsev, K.N.; Kruglov, A.G. Mechanism of triclosan toxicity: Mitochondrial dysfunction including complex І inhibition, superoxide release and uncoupling of oxidative phosphorylation. Toxicol. Lett. 2017, 275, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Petrosillo, G.; Colantuono, G.; Moro, N.; Ruggiero, F.M.; Tiravanti, E.; di Venosa, N.; Fiore, T.; Paradies, G. Melatonin protects against heart ischemia-reperfusion injury by inhibiting mitochondrial permeability transition pore opening. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Krestinina, O.V.; Baburina, Y.L.; Azarashvili, T.S. Effect of melatonin on stress-induced opening of non-selective pore in mitochondria from brain of young and old rats. Biochem. Moscow Suppl. Ser. A 2015, 9, 116. [Google Scholar] [CrossRef]
- Krestinina, O.V.; Odinokova, I.V.; Baburina, Y.L.; Azarashvili, T.S. Age-related effect of melatonin on permeability transition pore opening in rat brain mitochondria. Biochem. Moscow Suppl. Ser. A 2013, 7, 286. [Google Scholar] [CrossRef]
- Cheshchevik, V.T.; Dremza, I.K.; Lapshina, E.A.; Zabrodskaya, S.V.; Kujawa, J.; Zavodnik, I.B. Corrections by melatonin of liver mitochondrial disorders under diabetes and acute intoxication in rats. Cell Biochem. Funct. 2011, 29, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Petrosillo, G.; di Venosa, N.; Pistolese, M.; Casanova, G.; Tiravanti, E.; Colantuono, G.; Federici, A.; Paradies, G.; Ruggiero, F.M. Protective effect of melatonin against mitochondrial dysfunction associated with cardiac ischemia-reperfusion: Role of cardiolipin. FASEB J. 2006, 20, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Kruglov, A.G.; Subbotina, K.B.; Saris, N.E. Redox-cycling compounds can cause the permeabilization of mitochondrial membranes by mechanisms other than ros production. Free Radic. Biol. Med. 2008, 44, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Azarashvili, T.; Krestinina, O.; Galvita, A.; Grachev, D.; Baburina, Y.; Stricker, R.; Reiser, G. Identification of phosphorylated form of 2′, 3′-cyclic nucleotide 3′-phosphodiesterase (cnpase) as 46 kda phosphoprotein in brain non-synaptic mitochondria overloaded by calcium. J. Bioenerg. Biomembr. 2014, 46, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Beckman, J.S. Oxidative damage and tyrosine nitration from peroxynitrite. Chem. Res. Toxicol. 1996, 9, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Petrosillo, G.; Moro, N.; Paradies, V.; Ruggiero, F.M.; Paradies, G. Increased susceptibility to Ca2+-induced permeability transition and to cytochrome c release in rat heart mitochondria with aging: Effect of melatonin. J. Pineal Res. 2010, 48, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.H.; Jacobson, K.A.; Rose, J.; Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. 2008, 2008. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G.; Petrosillo, G.; Pistolese, M.; Di Venosa, N.; Federici, A.; Ruggiero, F.M. Decrease in mitochondrial complex І activity in ischemic/reperfused rat heart: Involvement of reactive oxygen species and cardiolipin. Circ. Res. 2004, 94, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Azarashvili, T.; Grachev, D.; Krestinina, O.; Evtodienko, Y.; Yurkov, I.; Papadopoulos, V.; Reiser, G. The peripheral-type benzodiazepine receptor is involved in control of ca2+-induced permeability transition pore opening in rat brain mitochondria. Cell Calcium 2007, 42, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, A.B.; Saris, N.E.; Kruglov, A.G. External mitochondrial nadh-dependent reductase of redox cyclers: Vdac1 or cyb5r3? Free Radic. Biol. Med. 2014, 74, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Kambayashi, Y.; Ogino, K. Reestimation of cypridina luciferin analogs (mcla) as a chemiluminescence probe to detect active oxygen species-Cautionary note for use of mcla. J. Toxicol. Sci. 2003, 28, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Teplova, V.V.; Kruglov, A.G.; Kovalyov, L.I.; Nikiforova, A.B.; Fedotcheva, N.I.; Lemasters, J.J. Glutamate contributes to alcohol hepatotoxicity by enhancing oxidative stress in mitochondria. J. Bioenerg. Biomembr. 2017, 49, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Stricker, R.; Lottspeich, F.; Reiser, G. The myelin protein cnp (2′,3′-cyclic nucleotide 3′-phosphodiesterase): Immunoaffinity purification of cnp from pig and rat brain using a monoclonal antibody and phosphorylation of cnp by cyclic nucleotide-dependent protein kinases. Biol. Chem. Hoppe Seyler 1994, 375, 205–209. [Google Scholar] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odinokova, I.; Baburina, Y.; Kruglov, A.; Fadeeva, I.; Zvyagina, A.; Sotnikova, L.; Akatov, V.; Krestinina, O. Effect of Melatonin on Rat Heart Mitochondria in Acute Heart Failure in Aged Rats. Int. J. Mol. Sci. 2018, 19, 1555. https://doi.org/10.3390/ijms19061555
Odinokova I, Baburina Y, Kruglov A, Fadeeva I, Zvyagina A, Sotnikova L, Akatov V, Krestinina O. Effect of Melatonin on Rat Heart Mitochondria in Acute Heart Failure in Aged Rats. International Journal of Molecular Sciences. 2018; 19(6):1555. https://doi.org/10.3390/ijms19061555
Chicago/Turabian StyleOdinokova, Irina, Yulia Baburina, Alexey Kruglov, Irina Fadeeva, Alena Zvyagina, Linda Sotnikova, Vladimir Akatov, and Olga Krestinina. 2018. "Effect of Melatonin on Rat Heart Mitochondria in Acute Heart Failure in Aged Rats" International Journal of Molecular Sciences 19, no. 6: 1555. https://doi.org/10.3390/ijms19061555
APA StyleOdinokova, I., Baburina, Y., Kruglov, A., Fadeeva, I., Zvyagina, A., Sotnikova, L., Akatov, V., & Krestinina, O. (2018). Effect of Melatonin on Rat Heart Mitochondria in Acute Heart Failure in Aged Rats. International Journal of Molecular Sciences, 19(6), 1555. https://doi.org/10.3390/ijms19061555