Protein–Protein Interactions with Connexin 43: Regulation and Function
Abstract
1. Introduction
2. Direct Interactions with Cx43 and Their Functional Consequence
2.1. Interactions that Promote Synthesis, Trafficking to the Gap Junction Plaque, and Channel Opening
2.2. Interactions that Promote Channel Closure, Gap Junction Disassembly, Internalization and Degradation
3. The Intercalated Disc as a Hub of Cx43 Mediated Protein–Protein Interactions
3.1. Nedd4 Regulates the Cx43 Content of Cardiac Gap Junctions
3.2. Cx43 Regulates the Forward Trafficking of the Cardiac Sodium Channel NaV1.5
3.3. Cx43, the Area Composita and the Connexome
4. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | amino acids |
| AGS8 | Activator of G protein signaling 8 |
| AKT | protein kinase B |
| AMSH | associated molecule with the SH3 domain of STAM |
| AP2 | adaptor protein 2 |
| Atg | Autophagy-related protein |
| β-tub | β-tubulin |
| CaM | Calmodulin |
| CaMKII | Ca2+/calmodulin-dependent protein kinase II |
| CASK | Ca2+/calmodulin-activated serine kinase |
| CCN3 | CYR61/CTGF/NOV |
| CDK1 | Cyclin-dependent kinase 1 |
| CIP75 | connexin interacting protein 75 kDa |
| CIP85 | Cx43-interacting protein of 85-kDa |
| CK1 | Casein kinase 1 |
| co-Loc | co-localization |
| CT | carboxyl terminal |
| Cx43 | connexin 43 |
| Cx43CT | Cx43 carboxyl terminal |
| Dlg | Discs-large |
| DMPK | dystrophia myotonica protein kinase |
| Drebrin | Developmentally Regulated Brain Protein 1 |
| EB1 | End binding 1 |
| Eps15 | Epidermal growth factor receptor substrate 15 |
| ER | endoplasmic reticulum |
| ERp29 | Endoplasmic reticulum protein 29 |
| FERM domain | Domain found in 4.1 protein (F), Ezrin, Radixin and Moesin |
| FW | Far-Western |
| Hrs | hepatocyte growth factor-regulated tyrosine kinase substrate |
| HSP70 | heat shock protein 70 |
| HSP90 | heat shock protein 90 |
| ID | intercalated disc |
| IP | immunoprecipitation |
| IV | in vitro assay |
| Lin-7 | linage-7 |
| MAPK | Mitogen-activated protein kinase |
| MMP7 | matrix metalloproteinase-7 |
| Nedd4 | Neural precursor cell expressed developmentally down-regulated protein 4 |
| NMR | nuclear magnetic resonance |
| NOV | nephroblastoma overexpressed |
| p120ctn | p120-catenin |
| PD | pull-down |
| PIP2 | Phosphatidylinositol-bisphosphate |
| PKA | protein kinase A |
| PKC | protein kinase C |
| PKG | protein kinase G |
| PLA | proximity ligation assay |
| PP | protein phosphatase |
| RPTPµ | receptor-like protein tyrosine phosphatase µ |
| SH3 | Src homology 3 domain |
| Smurf2 | Smad ubiquitination regulatory factor-2 |
| SPR | surface plasmon resonance |
| STAMBP | Signal transducing adapter molecule 1 binding protein |
| TC-PTP | T-cell protein tyrosine phosphatase |
| TEM | transmission electron microscopy |
| TOM20 | mitochondrial outer membrane receptor 20 |
| TRIM21 | Tripartite motif-containing protein 21 |
| Tsg101 | Tumor susceptibility gene 101 protein |
| Tyk2 | Tyrosine kinase 2 |
| UBA | Ubiquitin-associating domain |
| UEV | Ubiquitin E2 variant domain |
| USP8 | Ubiquitin specific protease 8 |
| VEGF | Vascular endothelial growth factor |
| ZO-1 | Zonula occludens-1 |
| ZO-2 | Zonula occludens-2 |
References
- Laird, D.W. The gap junction proteome and its relationship to disease. Trends Cell Biol. 2010, 20, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Lampe, P.D.; Lau, A.F. The effects of connexin phosphorylation on gap junctional communication. Int. J. Biochem. Cell Biol. 2004, 36, 1171–1186. [Google Scholar] [CrossRef]
- Herve, J.; Bourmeyster, N.; Sarrouilhe, D.; Duffy, H. Gap junctional complexes: From partners to functions. Progress. Biophys. Mol. Biol. 2007, 94, 29–65. [Google Scholar] [CrossRef] [PubMed]
- Thevenin, A.F.; Kowal, T.J.; Fong, J.T.; Kells, R.M.; Fisher, C.G.; Falk, M.M. Proteins and mechanisms regulating gap-junction assembly, internalization, and degradation. Physiology (Bethesda) 2013, 28, 93–116. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.S.; Axelsen, L.N.; Sorgen, P.L.; Verma, V.; Delmar, M.; Holstein-Rathlou, N.H. Gap junctions. Compr. Physiol. 2012, 2, 1981–2035. [Google Scholar] [PubMed]
- Moreno, A.P.; Chanson, M.; Elenes, S.; Anumonwo, J.; Scerri, I.; Gu, H.; Taffet, S.M.; Delmar, M. Role of the carboxyl terminal of connexin43 in transjunctional fast voltage gating. Circ. Res. 2002, 90, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Morley, G.E.; Taffet, S.M.; Delmar, M. Intramolecular interactions mediate ph regulation of connexin43 channels. Biophys. J. 1996, 70, 1294–1302. [Google Scholar] [CrossRef]
- Anumonwo, J.M.; Taffet, S.M.; Gu, H.; Chanson, M.; Moreno, A.P.; Delmar, M. The carboxyl terminal domain regulates the unitary conductance and voltage dependence of connexin40 gap junction channels. Circ. Res. 2001, 88, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Revilla, A.; Castro, C.; Barrio, L.C. Molecular dissection of transjunctional voltage dependence in the connexin-32 and connexin-43 junctions. Biophys. J. 1999, 77, 1374–1383. [Google Scholar] [CrossRef]
- Sorgen, P.L.; Duffy, H.S.; Sahoo, P.; Coombs, W.; Delmar, M.; Spray, D.C. Structural changes in the carboxyl terminus of the gap junction protein connexin43 indicates signaling between binding domains for c-src and zonula occludens-1. J. Biol. Chem. 2004, 279, 54695–54701. [Google Scholar] [CrossRef] [PubMed]
- Bouvier, D.; Kieken, F.; Kellezi, A.; Sorgen, P.L. Structural changes in the carboxyl terminus of the gap junction protein connexin 40 caused by the interaction with c-src and zonula occludens-1. Cell Commun. Adhes. 2008, 15, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Stauch, K.; Kieken, F.; Sorgen, P. Characterization of the structure and intermolecular interactions between the connexin 32 carboxyl-terminal domain and the protein partners synapse-associated protein 97 and calmodulin. J. Biol. Chem. 2012, 287, 27771–27788. [Google Scholar] [CrossRef] [PubMed]
- Nelson, T.K.; Sorgen, P.L.; Burt, J.M. Carboxy terminus and pore-forming domain properties specific to cx37 are necessary for cx37-mediated suppression of insulinoma cell proliferation. Am. J. Physiol. Cell Physiol. 2013, 305, C1246–C1256. [Google Scholar] [CrossRef] [PubMed]
- Gilleron, J.; Carette, D.; Chevallier, D.; Segretain, D.; Pointis, G. Molecular connexin partner remodeling orchestrates connexin traffic: From physiology to pathophysiology. Crit. Rev. Biochem. Mol. Biol. 2012, 47, 407–423. [Google Scholar] [CrossRef] [PubMed]
- Leithe, E.; Mesnil, M.; Aasen, T. The connexin 43 c-terminus: A tail of many tales. Biochim. Biophys. Acta 2018, 1860, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Solan, J.L.; Lampe, P.D. Spatio-temporal regulation of connexin43 phosphorylation and gap junction dynamics. Biochim. Biophys. Acta 2018, 1860, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Falk, M.M.; Bell, C.L.; Kells Andrews, R.M.; Murray, S.A. Molecular mechanisms regulating formation, trafficking and processing of annular gap junctions. BMC Cell Biol. 2016, 17 (Suppl. 1), 22. [Google Scholar] [CrossRef] [PubMed]
- Axelsen, L.N.; Calloe, K.; Holstein-Rathlou, N.H.; Nielsen, M.S. Managing the complexity of communication: Regulation of gap junctions by post-translational modification. Front. Pharmacol. 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Romero, P.R.; Noutsou, M.; Maurice, M.M.; Rudiger, S.G.; William, A.M., Jr.; Mizianty, M.J.; Kurgan, L.; Uversky, V.N.; Dunker, A.K. Stochastic machines as a colocalization mechanism for scaffold protein function. FEBS Lett. 2013, 587, 1587–1591. [Google Scholar] [CrossRef] [PubMed]
- Grosely, R.; Kopanic, J.L.; Nabors, S.; Kieken, F.; Spagnol, G.; Al-Mugotir, M.; Zach, S.; Sorgen, P.L. Effects of phosphorylation on the structure and backbone dynamics of the intrinsically disordered connexin43 c-terminal domain. J. Biol. Chem. 2013, 288, 24857–24870. [Google Scholar] [CrossRef] [PubMed]
- Yamane, Y.; Shiga, H.; Asou, H.; Haga, H.; Kawabata, K.; Abe, K.; Ito, E. Dynamics of astrocyte adhesion as analyzed by a combination of atomic force microscopy and immuno-cytochemistry: The involvement of actin filaments and connexin 43 in the early stage of adhesion. Arch. Histol. Cytol. 1999, 62, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Squecco, R.; Sassoli, C.; Nuti, F.; Martinesi, M.; Chellini, F.; Nosi, D.; Zecchi-Orlandini, S.; Francini, F.; Formigli, L.; Meacci, E. Sphingosine 1-phosphate induces myoblast differentiation through cx43 protein expression: A role for a gap junction-dependent and -independent function. Mol. Biol. Cell 2006, 17, 4896–4910. [Google Scholar] [CrossRef] [PubMed]
- Wall, M.E.; Otey, C.; Qi, J.; Banes, A.J. Connexin 43 is localized with actin in tenocytes. Cell Motil. Cytoskelet. 2007, 64, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Jiao, Q.; Honda, T.; Kurotani, R.; Toyota, E.; Okumura, S.; Takeya, T.; Minamisawa, S.; Lanier, S.M.; Ishikawa, Y. Activator of g protein signaling 8 (ags8) is required for hypoxia-induced apoptosis of cardiomyocytes: Role of g betagamma and connexin 43 (cx43). J. Biol. Chem. 2009, 284, 31431–31440. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Kong, X.; Zhuang, W.; Teng, B.; Yu, X.; Hua, S.; Wang, S.; Liang, F.; Ma, D.; Zhang, S.; et al. Dynamic changes in protein interaction between akap95 and cx43 during cell cycle progression of a549 cells. Sci. Rep. 2016, 6, 21224. [Google Scholar] [CrossRef] [PubMed]
- Sato, P.Y.; Coombs, W.; Lin, X.; Nekrasova, O.; Green, K.J.; Isom, L.L.; Taffet, S.M.; Delmar, M. Interactions between ankyrin-g, plakophilin-2, and connexin43 at the cardiac intercalated disc. Circ. Res. 2011, 109, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Denuc, A.; Nunez, E.; Calvo, E.; Loureiro, M.; Miro-Casas, E.; Guaras, A.; Vazquez, J.; Garcia-Dorado, D. New protein-protein interactions of mitochondrial connexin 43 in mouse heart. J. Cell. Mol. Med. 2016, 20, 794–803. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, E.; Yuste, A.; Patel, B.; Stout, R.F., Jr.; Spray, D.C.; Cuervo, A.M. Connexins modulate autophagosome biogenesis. Nat. Cell Biol. 2014, 16, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhao, X.; Yao, Y.; Qi, X.; Yuan, Y.; Hu, Y. Connexin 43 interacts with bax to regulate apoptosis of pancreatic cancer through a gap junction-independent pathway. Int. J. Oncol. 2012, 41, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Bivi, N.; Lezcano, V.; Romanello, M.; Bellido, T.; Plotkin, L.I. Connexin43 interacts with betaarrestin: A pre-requisite for osteoblast survival induced by parathyroid hormone. J. Cell. Biochem. 2011, 112, 2920–2930. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Ueki, T.; Furukawa, H.; Sato, K. The identification of novel protein, brain-derived integrating factor-1 (bdif1), which interacts with astrocytic gap junctional protein. Neurosci. Res. 2011, 70, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Marquez-Rosado, L.; Singh, D.; Rincon-Arano, H.; Solan, J.L.; Lampe, P.D. Cask (lin2) interacts with cx43 in wounded skin and their coexpression affects cell migration. J. Cell Sci. 2012, 125, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Schubert, A.L.; Schubert, W.; Spray, D.C.; Lisanti, M.P. Connexin family members target to lipid raft domains and interact with caveolin-1. Biochemistry 2002, 41, 5754–5764. [Google Scholar] [CrossRef] [PubMed]
- Langlois, S.; Cowan, K.N.; Shao, Q.; Cowan, B.J.; Laird, D.W. Caveolin-1 and -2 interact with connexin43 and regulate gap junctional intercellular communication in keratinocytes. Mol. Biol. Cell 2008, 19, 912–928. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Y.; Lin, J.; Liang, Q.; Sheng, X.; Wu, J.; Huang, R.; Liu, S.; Li, Y. Connexin43 interacts with caveolin-3 in the heart. Mol. Biol. Rep. 2010, 37, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.D.; Horackova, M.; Pressler, M.L. Changes in the expression and distribution of connexin 43 in isolated cultured adult guinea pig cardiomyocytes. Exp. Cell Res. 1996, 228, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, K.; Chiba, H.; Fujita, H.; Kojima, T.; Saito, T.; Endo, T.; Sawada, N. Possible involvement of gap junctions in the barrier function of tight junctions of brain and lung endothelial cells. J. Cell. Physiol. 2006, 208, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Lan, Z.; Kurata, W.E.; Martyn, K.D.; Jin, C.; Lau, A.F. Novel rab gap-like protein, cip85, interacts with connexin43 and induces its degradation. Biochemistry 2005, 44, 2385–2396. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Del Castillo, F.J.; Cohen-Salmon, M.; Charollais, A.; Caille, D.; Lampe, P.D.; Chavrier, P.; Meda, P.; Petit, C. Consortin, a trans-golgi network cargo receptor for the plasma membrane targeting and recycling of connexins. Hum. Mol. Genet. 2010, 19, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, S.R.; Kroncke, B.M.; Straub, A.C.; Best, A.K.; Dunn, C.A.; Mitchell, L.A.; Peskova, Y.; Nakamoto, R.K.; Koval, M.; Lo, C.W.; et al. Mapk phosphorylation of connexin 43 promotes binding of cyclin e and smooth muscle cell proliferation. Circ. Res. 2012, 111, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Gehmlich, K.; Lambiase, P.D.; Asimaki, A.; Ciaccio, E.J.; Ehler, E.; Syrris, P.; Saffitz, J.E.; McKenna, W.J. A novel desmocollin-2 mutation reveals insights into the molecular link between desmosomes and gap junctions. Heart Rhythm 2011, 8, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, A.I.; Sun, P.; Hernandez-Lopez, H.; Aasen, T.; Hodgins, M.B.; Edward, M.; Roberts, S.; Massimi, P.; Thomas, M.; Banks, L.; et al. A functional interaction between the maguk protein hdlg and the gap junction protein connexin 43 in cervical tumour cells. Biochem. J. 2012, 446, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Gilleron, J.; Carette, D.; Fiorini, C.; Dompierre, J.; Macia, E.; Denizot, J.P.; Segretain, D.; Pointis, G. The large gtpase dynamin2: A new player in connexin 43 gap junction endocytosis, recycling and degradation. Int. J. Biochem. Cell Biol. 2011, 43, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Shaw, R.M.; Fay, A.J.; Puthenveedu, M.A.; von Zastrow, M.; Jan, Y.N.; Jan, L.Y. Microtubule plus-end-tracking proteins target gap junctions directly from the cell interior to adherens junctions. Cell 2007, 128, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Girao, H.; Catarino, S.; Pereira, P. Eps15 interacts with ubiquitinated cx43 and mediates its internalization. Exp. Cell Res. 2009, 315, 3587–3597. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Smith, T.D.; Sarma, J.D.; Ritzenthaler, J.D.; Maza, J.; Kaplan, B.E.; Cunningham, L.A.; Suaud, L.; Hubbard, M.J.; Rubenstein, R.C.; et al. Erp29 restricts connexin43 oligomerization in the endoplasmic reticulum. Mol. Biol. Cell 2009, 20, 2593–2604. [Google Scholar] [CrossRef] [PubMed]
- Leithe, E.; Kjenseth, A.; Sirnes, S.; Stenmark, H.; Brech, A.; Rivedal, E. Ubiquitylation of the gap junction protein connexin-43 signals its trafficking from early endosomes to lysosomes in a process mediated by hrs and tsg101. J. Cell Sci. 2009, 122, 3883–3893. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, T.; Dai, P.; Harada, Y.; Hino, H.; Tsukahara, F.; Maru, Y.; Otsuji, E.; Takamatsu, T. Connexin43 functions as a novel interacting partner of heat shock cognate protein 70. Sci. Rep. 2013, 3, 2719. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Sinovas, A.; Boengler, K.; Cabestrero, A.; Gres, P.; Morente, M.; Ruiz-Meana, M.; Konietzka, I.; Miro, E.; Totzeck, A.; Heusch, G.; et al. Translocation of connexin 43 to the inner mitochondrial membrane of cardiomyocytes through the heat shock protein 90-dependent tom pathway and its importance for cardioprotection. Circ. Res. 2006, 99, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, E.; Girao, H.; Yuste, A.; Patel, B.; Marques, C.; Spray, D.C.; Pereira, P.; Cuervo, A.M. Autophagy modulates dynamics of connexins at the plasma membrane in a ubiquitin-dependent manner. Mol. Biol. Cell 2012, 23, 2156–2169. [Google Scholar] [CrossRef] [PubMed]
- Martins-Marques, T.; Catarino, S.; Zuzarte, M.; Marques, C.; Matafome, P.; Pereira, P.; Girao, H. Ischaemia-induced autophagy leads to degradation of gap junction protein connexin43 in cardiomyocytes. Biochem. J. 2015, 467, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Lampe, P.D. Identification of connexin-43 interacting proteins. Cell Commun. Adhes. 2003, 10, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Piehl, M.; Lehmann, C.; Gumpert, A.; Denizot, J.P.; Segretain, D.; Falk, M.M. Internalization of large double-membrane intercellular vesicles by a clathrin-dependent endocytic process. Mol. Biol. Cell 2007, 18, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, G.; Furlan, S.; Marin, O.; Salvatori, S. Myotonic dystrophy protein kinase of the cardiac muscle: Evaluation using an immunochemical approach. Microsc. Res. Tech. 2002, 58, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, J.D.; Thyagarajan, V.; Chen, C.; Isom, L.L. Tyrosine-phosphorylated and nonphosphorylated sodium channel beta1 subunits are differentially localized in cardiac myocytes. J. Biol. Chem. 2004, 279, 40748–40754. [Google Scholar] [CrossRef] [PubMed]
- Akar, F.G.; Spragg, D.D.; Tunin, R.S.; Kass, D.A.; Tomaselli, G.F. Mechanisms underlying conduction slowing and arrhythmogenesis in nonischemic dilated cardiomyopathy. Circ. Res. 2004, 95, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.T.; Bechberger, J.F.; Ozog, M.A.; Perbal, B.; Naus, C.C. Ccn3 (nov) interacts with connexin43 in c6 glioma cells: Possible mechanism of connexin-mediated growth suppression. J. Biol. Chem. 2004, 279, 36943–36950. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Li, W.E.; Huang, G.Y.; Meyer, R.; Chen, T.; Luo, Y.; Thomas, M.P.; Radice, G.L.; Lo, C.W. Modulation of mouse neural crest cell motility by n-cadherin and connexin 43 gap junctions. J. Cell Biol. 2001, 154, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Fortes, F.S.; Pecora, I.L.; Persechini, P.M.; Hurtado, S.; Costa, V.; Coutinho-Silva, R.; Braga, M.B.; Silva-Filho, F.C.; Bisaggio, R.C.; De Farias, F.P.; et al. Modulation of intercellular communication in macrophages: Possible interactions between gap junctions and p2 receptors. J. Cell Sci. 2004, 117, 4717–4726. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, D.A.; Suadicani, S.O.; Iacobas, S.; Chrisman, C.; Cohen, M.A.; Spray, D.C.; Scemes, E. Gap junction and purinergic p2 receptor proteins as a functional unit: Insights from transcriptomics. J. Membr. Biol. 2007, 217, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Kwak, B.R.; Saez, J.C.; Wilders, R.; Chanson, M.; Fishman, G.I.; Hertzberg, E.L.; Spray, D.C.; Jongsma, H.J. Effects of cgmp-dependent phosphorylation on rat and human connexin43 gap junction channels. Pflugers Arch. 1995, 430, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Li, M.W.; Mruk, D.D.; Lee, W.M.; Cheng, C.Y. Connexin 43 and plakophilin-2 as a protein complex that regulates blood-testis barrier dynamics. Proc. Natl. Acad. Sci. USA 2009, 106, 10213–10218. [Google Scholar] [CrossRef] [PubMed]
- Ai, X.; Pogwizd, S.M. Connexin 43 downregulation and dephosphorylation in nonischemic heart failure is associated with enhanced colocalized protein phosphatase type 2a. Circ. Res. 2005, 96, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Lezcano, V.; Bellido, T.; Plotkin, L.I.; Boland, R.; Morelli, S. Osteoblastic protein tyrosine phosphatases inhibition and connexin 43 phosphorylation by alendronate. Exp. Cell Res. 2014, 324, 30–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fykerud, T.A.; Kjenseth, A.; Schink, K.O.; Sirnes, S.; Bruun, J.; Omori, Y.; Brech, A.; Rivedal, E.; Leithe, E. Smad ubiquitination regulatory factor-2 controls gap junction intercellular communication by modulating endocytosis and degradation of connexin43. J. Cell Sci. 2012, 125, 3966–3976. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Rodrigues, T.M.; Catarino, S.; Marques, C.; Ferreira, J.V.; Martins-Marques, T.; Pereira, P.; Girao, H. Amsh-mediated deubiquitination of cx43 regulates internalization and degradation of gap junctions. FASEB J. 2014, 28, 4629–4641. [Google Scholar] [CrossRef] [PubMed]
- Chen, V.C.; Kristensen, A.R.; Foster, L.J.; Naus, C.C. Association of connexin43 with e3 ubiquitin ligase trim21 reveals a mechanism for gap junction phosphodegron control. J. Proteome Res. 2012, 11, 6134–6146. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Hu, Q.; Peng, H.; Peng, C.; Zhou, L.; Lu, J.; Huang, C. The ubiquitin-specific protease usp8 deubiquitinates and stabilizes cx43. J. Biol. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Basheer, W.A.; Harris, B.S.; Mentrup, H.L.; Abreha, M.; Thames, E.L.; Lea, J.B.; Swing, D.A.; Copeland, N.G.; Jenkins, N.A.; Price, R.L.; et al. Cardiomyocyte-specific overexpression of the ubiquitin ligase wwp1 contributes to reduction in connexin 43 and arrhythmogenesis. J. Mol. Cell Cardiol. 2015, 88, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Solan, J.L.; Taffet, S.M.; Javier, R.; Lampe, P.D. Connexin 43 interacts with zona occludens-1 and -2 proteins in a cell cycle stage-specific manner. J. Biol. Chem. 2005, 280, 30416–30421. [Google Scholar] [CrossRef] [PubMed]
- Musil, L.S.; Goodenough, D.A. Multisubunit assembly of an integral plasma membrane channel protein, gap junction connexin43, occurs after exit from the er. Cell 1993, 74, 1065–1077. [Google Scholar] [CrossRef]
- Kieken, F.; Spagnol, G.; Su, V.; Lau, A.F.; Sorgen, P.L. Nmr structure note: Uba domain of cip75. J. Biomol. NMR 2010, 46, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Kopanic, J.L.; Schlingmann, B.; Koval, M.; Lau, A.F.; Sorgen, P.L.; Su, V.F. Degradation of gap junction connexins is regulated by the interaction with cx43-interacting protein of 75 kda (cip75). Biochem. J. 2015, 466, 571–585. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Su, V.; Kurata, W.E.; Jin, C.; Lau, A.F. A novel connexin43-interacting protein, cip75, which belongs to the ubl-uba protein family, regulates the turnover of connexin43. J. Biol. Chem. 2008, 283, 5748–5759. [Google Scholar] [CrossRef] [PubMed]
- Su, V.; Hoang, C.; Geerts, D.; Lau, A.F. Cip75 (connexin43-interacting protein of 75 kda) mediates the endoplasmic reticulum dislocation of connexin43. Biochem. J. 2014, 458, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Su, V.; Nakagawa, R.; Koval, M.; Lau, A.F. Ubiquitin-independent proteasomal degradation of endoplasmic reticulum-localized connexin43 mediated by cip75. J. Biol. Chem. 2010, 285, 40979–40990. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.; Jordan, K.; Simek, J.; Shao, Q.; Jedeszko, C.; Walton, P.; Laird, D.W. Mechanisms of cx43 and cx26 transport to the plasma membrane and gap junction regeneration. J. Cell Sci. 2005, 118, 4451–4462. [Google Scholar] [CrossRef] [PubMed]
- Akhmanova, A.; Steinmetz, M.O. Microtubule +tips at a glance. J. Cell Sci. 2010, 123, 3415–3419. [Google Scholar] [CrossRef] [PubMed]
- Welte, M.A. Bidirectional transport along microtubules. Curr. Biol. 2004, 14, R525–R537. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.S.; Shaw, R.M. Trafficking highways to the intercalated disc: New insights unlocking the specificity of connexin 43 localization. Cell Commun. Adhes. 2014, 21, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Smyth, J.W.; Shaw, R.M. Autoregulation of connexin43 gap junction formation by internally translated isoforms. Cell Rep. 2013, 5, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Basheer, W.A.; Xiao, S.; Epifantseva, I.; Fu, Y.; Kleber, A.G.; Hong, T.; Shaw, R.M. Gja1-20k arranges actin to guide cx43 delivery to cardiac intercalated discs. Circ. Res. 2017, 121, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Dukic, A.R.; Gerbaud, P.; Guibourdenche, J.; Thiede, B.; Tasken, K.; Pidoux, G. Ezrin-anchored pka phosphorylates serine 369 and 373 on connexin 43 to enhance gap junction assembly, communication, and cell fusion. Biochem. J. 2017, 475, 455–476. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, M.M.; Lampe, P.D.; Lin, H.H.; Kollander, R.; Li, X.R.; Kiang, D.T. Cyclic amp modifies the cellular distribution of connexin43 and induces a persistent increase in the junctional permeability of mouse mammary tumor cells. J. Cell Sci. 1995, 108, 3079–3090. [Google Scholar] [PubMed]
- Spray, D.C.; Moreno, A.P.; Kessler, J.A.; Dermietzel, R. Characterization of gap junctions between cultured leptomeningeal cells. Brain Res. 1991, 568, 1–14. [Google Scholar] [CrossRef]
- Pidoux, G.; Gerbaud, P.; Dompierre, J.; Lygren, B.; Solstad, T.; Evain-Brion, D.; Tasken, K. A pka-ezrin-cx43 signaling complex controls gap junction communication and thereby trophoblast cell fusion. J. Cell Sci. 2014, 127, 4172–4185. [Google Scholar] [CrossRef] [PubMed]
- Thevenin, A.F.; Margraf, R.A.; Fisher, C.G.; Kells-Andrews, R.M.; Falk, M.M. Phosphorylation regulates connexin43/zo-1 binding and release, an important step in gap junction turnover. Mol. Biol. Cell 2017, 28, 3595–3608. [Google Scholar] [CrossRef] [PubMed]
- Dunn, C.A.; Lampe, P.D. Injury-triggered akt phosphorylation of cx43: A zo-1-driven molecular switch that regulates gap junction size. J. Cell Sci. 2014, 127, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Dunn, C.A.; Su, V.; Lau, A.F.; Lampe, P.D. Activation of akt, not connexin 43 protein ubiquitination, regulates gap junction stability. J. Biol. Chem. 2012, 287, 2600–2607. [Google Scholar] [CrossRef] [PubMed]
- Hunter, A.W.; Barker, R.J.; Zhu, C.; Gourdie, R.G. Zonula occludens-1 alters connexin43 gap junction size and organization by influencing channel accretion. Mol. Biol. Cell 2005, 16, 5686–5698. [Google Scholar] [CrossRef] [PubMed]
- Rhett, J.M.; Jourdan, J.; Gourdie, R.G. Connexin 43 connexon to gap junction transition is regulated by zonula occludens-1. Mol. Biol. Cell 2011, 22, 1516–1528. [Google Scholar] [CrossRef] [PubMed]
- Batra, N.; Riquelme, M.A.; Burra, S.; Jiang, J.X. 14-3-3theta facilitates plasma membrane delivery and function of mechanosensitive connexin 43 hemichannels. J. Cell Sci. 2014, 127, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Park, D.J.; Freitas, T.A.; Wallick, C.J.; Guyette, C.V.; Warn-Cramer, B.J. Molecular dynamics and in vitro analysis of connexin43: A new 14-3-3 mode-1 interacting protein. Protein Sci. 2006, 15, 2344–2355. [Google Scholar] [CrossRef] [PubMed]
- Park, D.J.; Wallick, C.J.; Martyn, K.D.; Lau, A.F.; Jin, C.; Warn-Cramer, B.J. Akt phosphorylates connexin43 on ser373, a “mode-1” binding site for 14-3-3. Cell Commun. Adhes. 2007, 14, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Solan, J.L.; Lampe, P.D. Connexin43 phosphorylation: Structural changes and biological effects. Biochem. J. 2009, 419, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.D.; Lampe, P.D. Casein kinase 1 regulates connexin-43 gap junction assembly. J. Biol. Chem. 2002, 277, 44962–44968. [Google Scholar] [CrossRef] [PubMed]
- Remo, B.F.; Qu, J.; Volpicelli, F.M.; Giovannone, S.; Shin, D.; Lader, J.; Liu, F.Y.; Zhang, J.; Lent, D.S.; Morley, G.E.; et al. Phosphatase-resistant gap junctions inhibit pathological remodeling and prevent arrhythmias. Circ. Res. 2011, 108, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Giepmans, B.N. Role of connexin43-interacting proteins at gap junctions. Adv. Cardiol. 2006, 42, 41–56. [Google Scholar] [PubMed]
- Giepmans, B.N. Gap junctions and connexin-interacting proteins. Cardiovasc. Res. 2004, 62, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Kanemitsu, M.Y.; Loo, L.W.; Simon, S.; Lau, A.F.; Eckhart, W. Tyrosine phosphorylation of connexin 43 by v-src is mediated by sh2 and sh3 domain interactions. J. Biol. Chem. 1997, 272, 22824–22831. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Warn-Cramer, B.J.; Kurata, W.E.; Lau, A.F. V-src phosphorylation of connexin 43 on tyr247 and tyr265 disrupts gap junctional communication. J. Cell Biol. 2001, 154, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Solan, J.L.; Lampe, P.D. Connexin 43 in la-25 cells with active v-src is phosphorylated on y247, y265, s262, s279/282, and s368 via multiple signaling pathways. Cell Commun. Adhes. 2008, 15, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Swenson, K.I.; Piwnica-Worms, H.; McNamee, H.; Paul, D.L. Tyrosine phosphorylation of the gap junction protein connexin43 is required for the pp60v-src-induced inhibition of communication. Cell Regul. 1990, 1, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Kasperek, E.M.; Nicholson, B.J. Dissection of the molecular basis of pp60(v-src) induced gating of connexin 43 gap junction channels. J. Cell Biol. 1999, 144, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, C.; Ren, C.; Spagnol, G.; Cavin, G.; Cone, A.; Grintsevich, E.E.; Sosinsky, G.E.; Sorgen, P.L. Connexin43 forms supramolecular complexes through non-overlapping binding sites for drebrin, tubulin, and zo-1. PLoS ONE 2016, 11, e0157073. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Li.; Li, H.; Spagnol, G.; Patel, K.; Sorgen, P.L. Src phosphorylation of Cx43 residue Y313 contributes to inhibiting the interaction with Drebrin and gap junction disassembly. J. Mol. Cell. Cardiol. (under review).
- Francis, R.; Xu, X.; Park, H.; Wei, C.J.; Chang, S.; Chatterjee, B.; Lo, C. Connexin43 modulates cell polarity and directional cell migration by regulating microtubule dynamics. PLoS ONE 2011, 6, e26379. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Spagnol, G.; Naslavsky, N.; Caplan, S.; Sorgen, P.L. Tc-ptp directly interacts with connexin43 to regulate gap junction intercellular communication. J. Cell Sci. 2014, 127, 3269–3279. [Google Scholar] [CrossRef] [PubMed]
- Ai, Z.; Fischer, A.; Spray, D.C.; Brown, A.M.; Fishman, G.I. Wnt-1 regulation of connexin43 in cardiac myocytes. J. Clin. Investig. 2000, 105, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, T.; Ohkusa, T.; Okamoto, Y.; Yoshida, M.; Lee, J.K.; Mizukami, Y.; Yano, M. Rapid electrical stimulation causes alterations in cardiac intercellular junction proteins of cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 2014, 306, H1324–H1333. [Google Scholar] [CrossRef] [PubMed]
- Swope, D.; Cheng, L.; Gao, E.; Li, J.; Radice, G.L. Loss of cadherin-binding proteins beta-catenin and plakoglobin in the heart leads to gap junction remodeling and arrhythmogenesis. Mol. Cell. Biol. 2012, 32, 1056–1067. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.X.; Gillio-Meina, C.; Chen, S.; Gong, X.Q.; Li, T.Y.; Bai, D.; Kidder, G.M. The canonical wnt2 pathway and fsh interact to regulate gap junction assembly in mouse granulosa cells. Biol. Reprod. 2013, 89, 39. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, F.; Hartfield, E.M.; Crompton, L.A.; Badger, J.L.; Glover, C.P.; Kelly, C.M.; Rosser, A.E.; Uney, J.B.; Caldwell, M.A. Cross-regulation of connexin43 and beta-catenin influences differentiation of human neural progenitor cells. Cell Death Dis. 2014, 5, e1017. [Google Scholar] [CrossRef] [PubMed]
- Spagnol, G.; Trease, A.J.; Zheng, Li.; Phillips, A.; Sorgen, P.L. Regulation of Connexin43 by the direct interaction with β-catenin. Int. J. Mol. Sci. (under review).
- Giepmans, B.N.; Hengeveld, T.; Postma, F.R.; Moolenaar, W.H. Interaction of c-src with gap junction protein connexin-43. Role in the regulation of cell-cell communication. J. Biol. Chem. 2001, 276, 8544–8549. [Google Scholar] [CrossRef] [PubMed]
- Gilleron, J.; Fiorini, C.; Carette, D.; Avondet, C.; Falk, M.M.; Segretain, D.; Pointis, G. Molecular reorganization of cx43, zo-1 and src complexes during the endocytosis of gap junction plaques in response to a non-genomic carcinogen. J. Cell Sci. 2008, 121, 4069–4078. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.S.; Xu, J.; Nicholson, B.J. Coregulation of multiple signaling mechanisms in pp60v-src-induced closure of cx43 gap junction channels. J. Membr. Biol. 2012, 245, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Pahujaa, M.; Anikin, M.; Goldberg, G.S. Phosphorylation of connexin43 induced by src: Regulation of gap junctional communication between transformed cells. Exp. Cell Res. 2007, 313, 4083–4090. [Google Scholar] [CrossRef] [PubMed]
- Toyofuku, T.; Akamatsu, Y.; Zhang, H.; Kuzuya, T.; Tada, M.; Hori, M. C-src regulates the interaction between connexin-43 and zo-1 in cardiac myocytes. J. Biol. Chem. 2001, 276, 1780–1788. [Google Scholar] [CrossRef] [PubMed]
- Homma, N.; Alvarado, J.L.; Coombs, W.; Stergiopoulos, K.; Taffet, S.M.; Lau, A.F.; Delmar, M. A particle-receptor model for the insulin-induced closure of connexin43 channels. Circ. Res. 1998, 83, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, G.T.; Lin, R.; Warn-Cramer, B.J.; Lau, A.F.; Burt, J.M. Mechanism of v-src- and mitogen-activated protein kinase-induced reduction of gap junction communication. Am. J. Physiol. Cell Physiol. 2003, 284, C511–C520. [Google Scholar] [CrossRef] [PubMed]
- Saidi Brikci-Nigassa, A.; Clement, M.J.; Ha-Duong, T.; Adjadj, E.; Ziani, L.; Pastre, D.; Curmi, P.A.; Savarin, P. Phosphorylation controls the interaction of the connexin43 c-terminal domain with tubulin and microtubules. Biochemistry 2012, 51, 4331–4342. [Google Scholar] [CrossRef] [PubMed]
- Butkevich, E.; Hulsmann, S.; Wenzel, D.; Shirao, T.; Duden, R.; Majoul, I. Drebrin is a novel connexin-43 binding partner that links gap junctions to the submembrane cytoskeleton. Curr. Biol. 2004, 14, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Kieken, F.; Mutsaers, N.; Dolmatova, E.; Virgil, K.; Wit, A.L.; Kellezi, A.; Hirst-Jensen, B.J.; Duffy, H.S.; Sorgen, P.L. Structural and molecular mechanisms of gap junction remodeling in epicardial border zone myocytes following myocardial infarction. Circ. Res. 2009, 104, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Spagnol, G.; Zheng, L.; Stauch, K.L.; Sorgen, P.L. Regulation of connexin43 function and expression by tyrosine kinase 2. J. Biol. Chem. 2016, 291, 15867–15880. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Saharinen, P.; Pesu, M.; Holt, V.E., 3rd; Silvennoinen, O.; O’Shea, J.J. The janus kinases (jaks). Genome Biol. 2004, 5, 253. [Google Scholar] [CrossRef] [PubMed]
- Girault, J.A.; Labesse, G.; Mornon, J.P.; Callebaut, I. The n-termini of fak and jaks contain divergent band 4.1 domains. Trends Biochem. Sci. 1999, 24, 54–57. [Google Scholar] [CrossRef]
- Haan, C.; Kreis, S.; Margue, C.; Behrmann, I. Jaks and cytokine receptors—An intimate relationship. Biochem. Pharmacol. 2006, 72, 1538–1546. [Google Scholar] [CrossRef] [PubMed]
- Wilks, A.F.; Harpur, A.G.; Kurban, R.R.; Ralph, S.J.; Zurcher, G.; Ziemiecki, A. Two novel protein-tyrosine kinases, each with a second phosphotransferase-related catalytic domain, define a new class of protein kinase. Mol. Cell Biol. 1991, 11, 2057–2065. [Google Scholar] [CrossRef] [PubMed]
- Huibregtse, J.M.; Scheffner, M.; Beaudenon, S.; Howley, P.M. A family of proteins structurally and functionally related to the e6-ap ubiquitin-protein ligase. Proc. Natl. Acad. Sci. USA 1995, 92, 2563–2567. [Google Scholar] [CrossRef] [PubMed]
- Leykauf, K.; Salek, M.; Bomke, J.; Frech, M.; Lehmann, W.D.; Durst, M.; Alonso, A. Ubiquitin protein ligase nedd4 binds to connexin43 by a phosphorylation-modulated process. J. Cell Sci. 2006, 119, 3634–3642. [Google Scholar] [CrossRef] [PubMed]
- Spagnol, G.; Kieken, F.; Kopanic, J.L.; Li, H.; Zach, S.; Stauch, K.L.; Grosely, R.; Sorgen, P.L. Structural studies of the nedd4 ww domains and their selectivity for the connexin43 (cx43) carboxyl terminus. J. Biol. Chem. 2016, 291, 7637–7650. [Google Scholar] [CrossRef] [PubMed]
- Auth, T.; Schluter, S.; Urschel, S.; Kussmann, P.; Sonntag, S.; Hoher, T.; Kreuzberg, M.M.; Dobrowolski, R.; Willecke, K. The tsg101 protein binds to connexins and is involved in connexin degradation. Exp. Cell Res. 2009, 315, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Fong, J.T.; Kells, R.M.; Falk, M.M. Two tyrosine-based sorting signals in the cx43 c-terminus cooperate to mediate gap junction endocytosis. Mol. Biol. Cell 2013, 24, 2834–2848. [Google Scholar] [CrossRef] [PubMed]
- Solan, J.L.; Marquez-Rosado, L.; Sorgen, P.L.; Thornton, P.J.; Gafken, P.R.; Lampe, P.D. Phosphorylation at s365 is a gatekeeper event that changes the structure of cx43 and prevents down-regulation by pkc. J. Cell Biol. 2007, 179, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Ek-Vitorin, J.F.; King, T.J.; Heyman, N.S.; Lampe, P.D.; Burt, J.M. Selectivity of connexin 43 channels is regulated through protein kinase c-dependent phosphorylation. Circ. Res. 2006, 98, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Nimlamool, W.; Andrews, R.M.; Falk, M.M. Connexin43 phosphorylation by pkc and mapk signals vegf-mediated gap junction internalization. Mol. Biol. Cell 2015, 26, 2755–2768. [Google Scholar] [CrossRef] [PubMed]
- Cone, A.C.; Cavin, G.; Ambrosi, C.; Hakozaki, H.; Wu-Zhang, A.X.; Kunkel, M.T.; Newton, A.C.; Sosinsky, G.E. Protein kinase cdelta-mediated phosphorylation of connexin43 gap junction channels causes movement within gap junctions followed by vesicle internalization and protein degradation. J. Biol. Chem. 2014, 289, 8781–8798. [Google Scholar] [CrossRef] [PubMed]
- Fong, J.T.; Nimlamool, W.; Falk, M.M. Egf induces efficient cx43 gap junction endocytosis in mouse embryonic stem cell colonies via phosphorylation of ser262, ser279/282, and ser368. FEBS Lett. 2014, 588, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.E.; Mitra, S.; Katoch, P.; Kelsey, L.S.; Johnson, K.R.; Mehta, P.P. Phosphorylation on ser-279 and ser-282 of connexin43 regulates endocytosis and gap junction assembly in pancreatic cancer cells. Mol. Biol. Cell 2013, 24, 715–733. [Google Scholar] [CrossRef] [PubMed]
- Lampe, P.D. Analyzing phorbol ester effects on gap junctional communication: A dramatic inhibition of assembly. J. Cell Biol. 1994, 127, 1895–1905. [Google Scholar] [CrossRef] [PubMed]
- Lampe, P.D.; TenBroek, E.M.; Burt, J.M.; Kurata, W.E.; Johnson, R.G.; Lau, A.F. Phosphorylation of connexin43 on serine368 by protein kinase c regulates gap junctional communication. J. Cell Biol. 2000, 149, 1503–1512. [Google Scholar] [CrossRef] [PubMed]
- Kirchhausen, T.; Owen, D.; Harrison, S.C. Molecular structure, function, and dynamics of clathrin-mediated membrane traffic. Cold Spring Harb. Perspect. Biol. 2014, 6, a016725. [Google Scholar] [CrossRef] [PubMed]
- Bonifacino, J.S.; Traub, L.M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 2003, 72, 395–447. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.A.; Zosso, N.; Scerri, I.; Demaurex, N.; Chanson, M.; Staub, O. A tyrosine-based sorting signal is involved in connexin43 stability and gap junction turnover. J. Cell Sci. 2003, 116, 2213–2222. [Google Scholar] [CrossRef] [PubMed]
- Kittler, J.T.; Chen, G.; Kukhtina, V.; Vahedi-Faridi, A.; Gu, Z.; Tretter, V.; Smith, K.R.; McAinsh, K.; Arancibia-Carcamo, I.L.; Saenger, W.; et al. Regulation of synaptic inhibition by phospho-dependent binding of the ap2 complex to a yecl motif in the gabaa receptor gamma2 subunit. Proc. Natl. Acad. Sci. USA 2008, 105, 3616–3621. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Y.; Laing, J.G.; Kanter, E.M.; Berthoud, V.M.; Bao, M.; Rohrs, H.W.; Townsend, R.R.; Yamada, K.A. Identification of camkii phosphorylation sites in connexin43 by high-resolution mass spectrometry. J. Proteome Res. 2011, 10, 1098–1109. [Google Scholar] [CrossRef] [PubMed]
- Procida, K.; Jorgensen, L.; Schmitt, N.; Delmar, M.; Taffet, S.M.; Holstein-Rathlou, N.H.; Nielsen, M.S.; Braunstein, T.H. Phosphorylation of connexin43 on serine 306 regulates electrical coupling. Heart Rhythm 2009, 6, 1632–1638. [Google Scholar] [CrossRef] [PubMed]
- Shifman, J.M.; Choi, M.H.; Mihalas, S.; Mayo, S.L.; Kennedy, M.B. Ca2+/calmodulin-dependent protein kinase ii (camkii) is activated by calmodulin with two bound calciums. Proc. Natl. Acad. Sci. USA 2006, 103, 13968–13973. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.P.; Schulman, H. The multifunctional calcium/calmodulin-dependent protein kinase: From form to function. Annu. Rev. Physiol. 1995, 57, 417–445. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, W.; Lurtz, M.M.; Ye, Y.; Huang, Y.; Lee, H.W.; Chen, Y.; Louis, C.F.; Yang, J.J. Identification of the calmodulin binding domain of connexin 43. J. Biol. Chem. 2007, 282, 35005–35017. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Salarian, M.; Chen, Y.; Veenstra, R.; Louis, C.F.; Yang, J.J. Gap junction regulation by calmodulin. FEBS Lett. 2014, 588, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Spagnol, G.; Chenavas, S.; Trease, A.; Li, H.; Kieken, F.; Brownell, S.; Sorgen, P.L. Characterizing the interaction between calmodulin and the Cx43 cytoplasmic domains (manuscript in preparation).
- Dbouk, H.A.; Mroue, R.M.; El-Sabban, M.E.; Talhouk, R.S. Connexins: A myriad of functions extending beyond assembly of gap junction channels. Cell Commun. Signal. 2009, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Boassa, D.; Solan, J.L.; Papas, A.; Thornton, P.; Lampe, P.D.; Sosinsky, G.E. Trafficking and recycling of the connexin43 gap junction protein during mitosis. Traffic 2010, 11, 1471–1486. [Google Scholar] [CrossRef] [PubMed]
- Kanemitsu, M.Y.; Jiang, W.; Eckhart, W. Cdc2-mediated phosphorylation of the gap junction protein, connexin43, during mitosis. Cell Growth Differ. 1998, 9, 13–21. [Google Scholar] [PubMed]
- Lampe, P.D.; Kurata, W.E.; Warn-Cramer, B.J.; Lau, A.F. Formation of a distinct connexin43 phosphoisoform in mitotic cells is dependent upon p34cdc2 kinase. J. Cell Sci. 1998, 111, 833–841. [Google Scholar] [PubMed]
- Stein, L.S.; Boonstra, J.; Burghardt, R.C. Reduced cell-cell communication between mitotic and nonmitotic coupled cells. Exp. Cell Res. 1992, 198, 1–7. [Google Scholar] [CrossRef]
- Lindsey, M.L.; Escobar, G.P.; Mukherjee, R.; Goshorn, D.K.; Sheats, N.J.; Bruce, J.A.; Mains, I.M.; Hendrick, J.K.; Hewett, K.W.; Gourdie, R.G.; et al. Matrix metalloproteinase-7 affects connexin-43 levels, electrical conduction, and survival after myocardial infarction. Circulation 2006, 113, 2919–2928. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Mohammad, G.; dos Santos, J.M.; Zhong, Q. Abrogation of mmp-9 gene protects against the development of retinopathy in diabetic mice by preventing mitochondrial damage. Diabetes 2011, 60, 3023–3033. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, G.; Kowluru, R.A. Novel role of mitochondrial matrix metalloproteinase-2 in the development of diabetic retinopathy. Invest. Ophthalmol. Vis. Sci. 2011, 52, 3832–3841. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Huang, W.; Luo, G.; Alain, L.A. Hypoxia induces connexin 43 dysregulation by modulating matrix metalloproteinases via mapk signaling. Mol. Cell Biochem. 2013, 384, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Vermij, S.H.; Abriel, H.; van Veen, T.A. Refining the molecular organization of the cardiac intercalated disc. Cardiovasc. Res. 2017, 113, 259–275. [Google Scholar] [CrossRef] [PubMed]
- Martins-Marques, T.; Anjo, S.I.; Pereira, P.; Manadas, B.; Girao, H. Interacting network of the gap junction (gj) protein connexin43 (cx43) is modulated by ischemia and reperfusion in the heart. Mol. Cell Proteom. 2015, 14, 3040–3055. [Google Scholar] [CrossRef] [PubMed]
- Martins-Marques, T.; Catarino, S.; Marques, C.; Matafome, P.; Ribeiro-Rodrigues, T.; Baptista, R.; Pereira, P.; Girao, H. Heart ischemia results in connexin43 ubiquitination localized at the intercalated discs. Biochimie 2015, 112, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Mollerup, S.; Hofgaard, J.P.; Braunstein, T.H.; Kjenseth, A.; Leithe, E.; Rivedal, E.; Holstein-Rathlou, N.H.; Nielsen, M.S. Norepinephrine inhibits intercellular coupling in rat cardiomyocytes by ubiquitination of connexin43 gap junctions. Cell Commun. Adhes. 2011, 18, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Hofgaard, J.P.; Banach, K.; Mollerup, S.; Jorgensen, H.K.; Olesen, S.P.; Holstein-Rathlou, N.H.; Nielsen, M.S. Phosphatidylinositol-bisphosphate regulates intercellular coupling in cardiac myocytes. Pflugers Arch. 2008, 457, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Totland, M.Z.; Bergsland, C.H.; Fykerud, T.A.; Knudsen, L.M.; Rasmussen, N.L.; Eide, P.W.; Yohannes, Z.; Sorensen, V.; Brech, A.; Lothe, R.A.; et al. The e3 ubiquitin ligase nedd4 induces endocytosis and lysosomal sorting of connexin 43 to promote loss of gap junctions. J. Cell Sci. 2017, 130, 2867–2882. [Google Scholar] [CrossRef] [PubMed]
- Stroemlund, L.W.; Jensen, C.F.; Qvortrup, K.; Delmar, M.; Nielsen, M.S. Gap junctions-guards of excitability. Biochem. Soc. Trans. 2015, 43, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Gutstein, D.E.; Morley, G.E.; Tamaddon, H.; Vaidya, D.; Schneider, M.D.; Chen, J.; Chien, K.R.; Stuhlmann, H.; Fishman, G.I. Conduction slowing and sudden arrhythmic death in mice with cardiac-restricted inactivation of connexin43. Circ. Res. 2001, 88, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Van Rijen, H.V.; Eckardt, D.; Degen, J.; Theis, M.; Ott, T.; Willecke, K.; Jongsma, H.J.; Opthof, T.; de Bakker, J.M. Slow conduction and enhanced anisotropy increase the propensity for ventricular tachyarrhythmias in adult mice with induced deletion of connexin43. Circulation 2004, 109, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Desplantez, T.; McCain, M.L.; Beauchamp, P.; Rigoli, G.; Rothen-Rutishauser, B.; Parker, K.K.; Kleber, A.G. Connexin43 ablation in foetal atrial myocytes decreases electrical coupling, partner connexins, and sodium current. Cardiovasc. Res. 2012, 94, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.A.; Noorman, M.; Musa, H.; Stein, M.; de Jong, S.; van der Nagel, R.; Hund, T.J.; Mohler, P.J.; Vos, M.A.; van Veen, T.A.; et al. Reduced heterogeneous expression of cx43 results in decreased NaV1.5 expression and reduced sodium current that accounts for arrhythmia vulnerability in conditional cx43 knockout mice. Heart Rhythm 2012, 9, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Lubkemeier, I.; Requardt, R.P.; Lin, X.; Sasse, P.; Andrie, R.; Schrickel, J.W.; Chkourko, H.; Bukauskas, F.F.; Kim, J.S.; Frank, M.; et al. Deletion of the last five c-terminal amino acid residues of connexin43 leads to lethal ventricular arrhythmias in mice without affecting coupling via gap junction channels. Basic Res. Cardiol. 2013, 108, 348. [Google Scholar] [CrossRef] [PubMed]
- Agullo-Pascual, E.; Lin, X.; Leo-Macias, A.; Zhang, M.; Liang, F.X.; Li, Z.; Pfenniger, A.; Lubkemeier, I.; Keegan, S.; Fenyo, D.; et al. Super-resolution imaging reveals that loss of the c-terminus of connexin43 limits microtubule plus-end capture and NaV1.5 localization at the intercalated disc. Cardiovasc. Res. 2014, 104, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Rhett, J.M.; Gourdie, R.G. The perinexus: A new feature of cx43 gap junction organization. Heart Rhythm 2012, 9, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Franke, W.W.; Borrmann, C.M.; Grund, C.; Pieperhoff, S. The area composita of adhering junctions connecting heart muscle cells of vertebrates. I. Molecular definition in intercalated disks of cardiomyocytes by immunoelectron microscopy of desmosomal proteins. Eur. J. Cell Biol. 2006, 85, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Agullo-Pascual, E.; Reid, D.A.; Keegan, S.; Sidhu, M.; Fenyo, D.; Rothenberg, E.; Delmar, M. Super-resolution fluorescence microscopy of the cardiac connexome reveals plakophilin-2 inside the connexin43 plaque. Cardiovasc. Res. 2013, 100, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Agullo-Pascual, E.; Delmar, M. The noncanonical functions of cx43 in the heart. J. Membr. Biol. 2012, 245, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Oxford, E.M.; Musa, H.; Maass, K.; Coombs, W.; Taffet, S.M.; Delmar, M. Connexin43 remodeling caused by inhibition of plakophilin-2 expression in cardiac cells. Circ. Res. 2007, 101, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Agullo-Pascual, E.; Cerrone, M.; Delmar, M. Arrhythmogenic cardiomyopathy and brugada syndrome: Diseases of the connexome. FEBS Lett. 2014, 588, 1322–1330. [Google Scholar] [CrossRef] [PubMed]
- Leo-Macias, A.; Agullo-Pascual, E.; Delmar, M. The cardiac connexome: Non-canonical functions of connexin43 and their role in cardiac arrhythmias. Semin. Cell Dev. Biol. 2016, 50, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Beyer, E.C.; Paul, D.L.; Goodenough, D.A. Connexin43: A protein from rat heart homologous to a gap junction protein from liver. J. Cell Biol. 1987, 105, 2621–2629. [Google Scholar] [CrossRef] [PubMed]
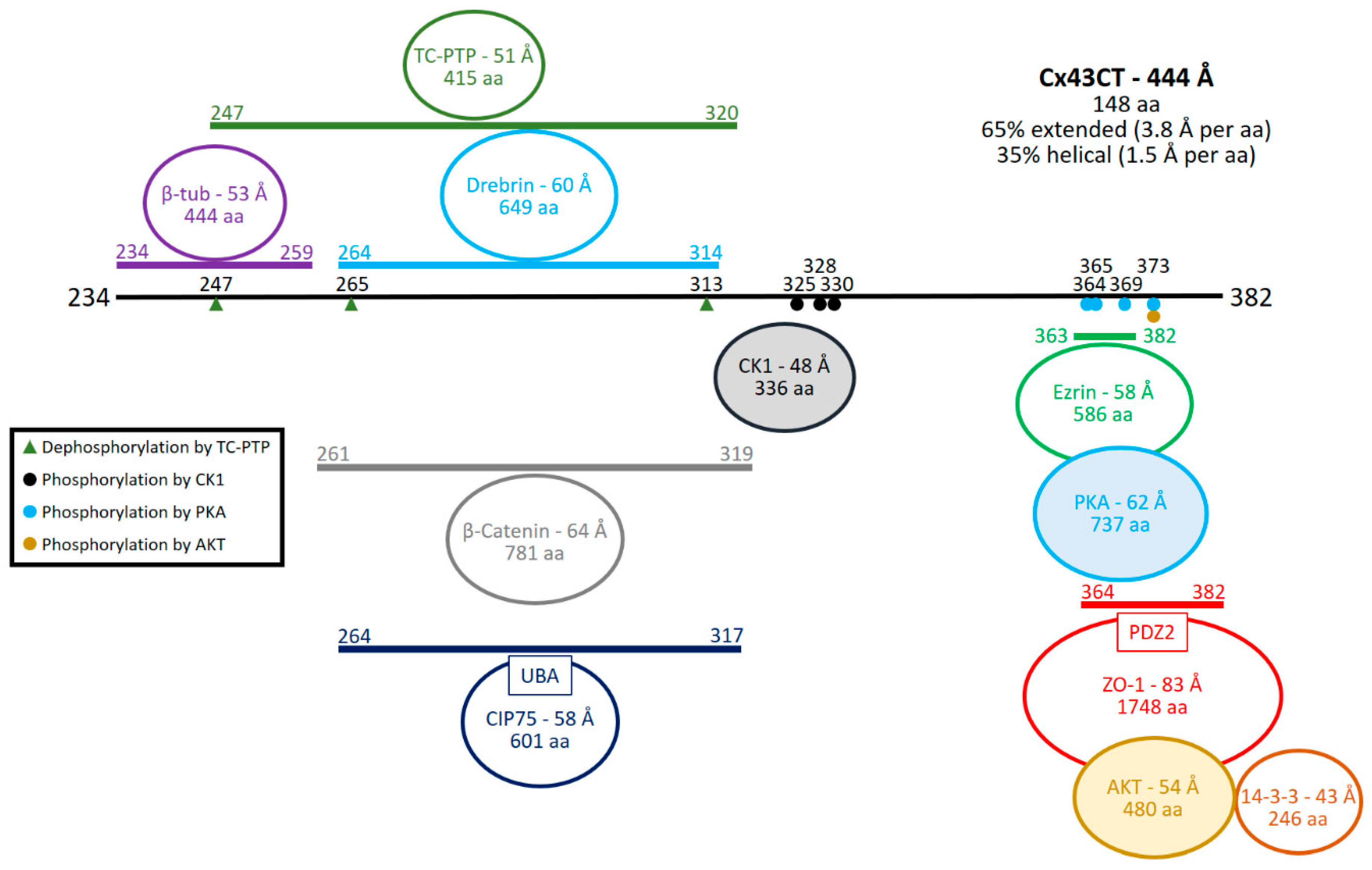
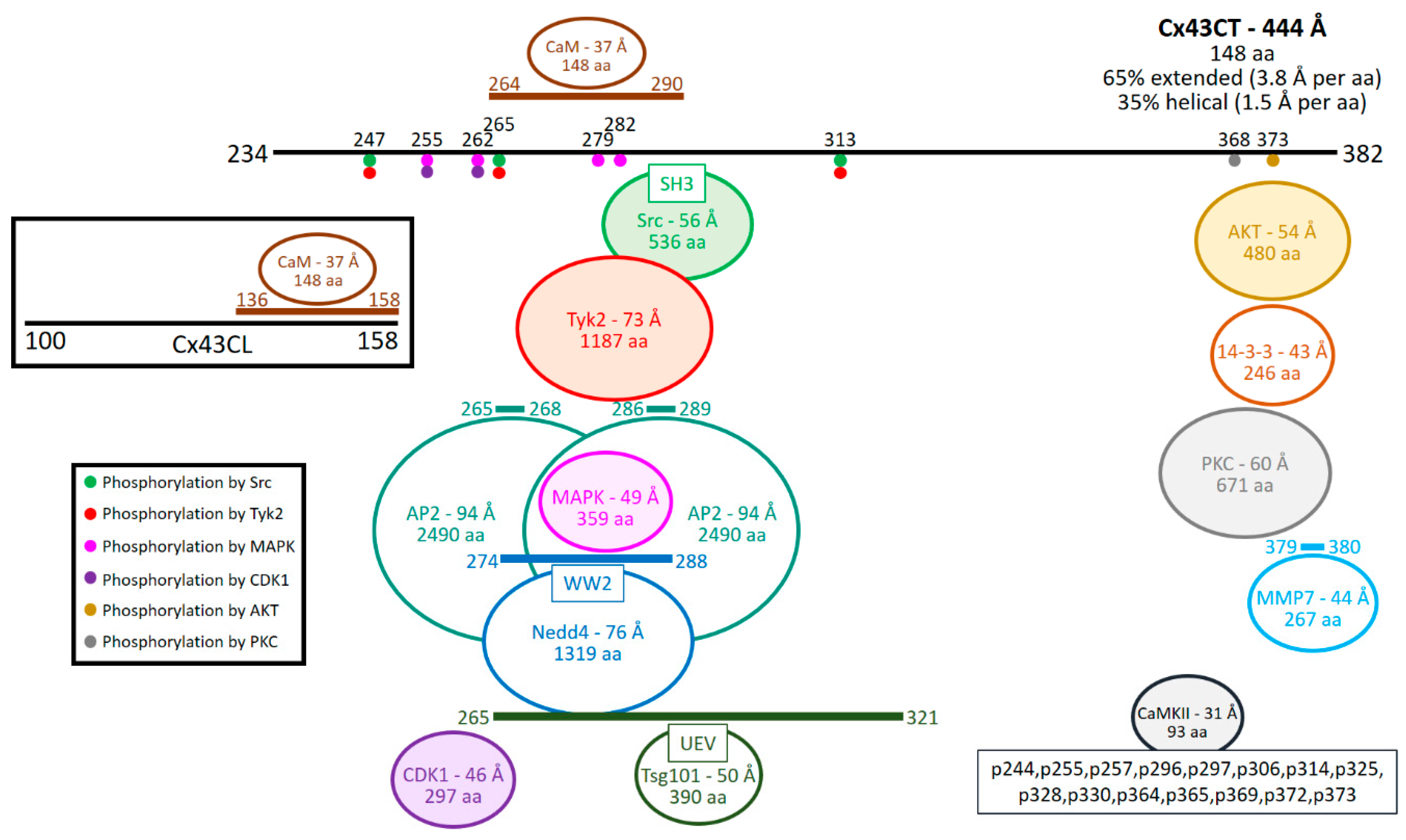
| Interacting Protein | Type of Detection | References |
|---|---|---|
| Actin | co-Loc | [21,22,23] |
| AGS8 | IP, co-Loc | [24] |
| A-kinase anchoring protein 95 | IP, co-Loc | [25] |
| Ankyrin G | IP | [26] |
| Apoptosis-inducing factor | IP, co-Loc, PLA | [27] |
| Atg16L/Atg14/Atg9/Vps34 | IP, co-Loc | [28] |
| Bax | IP, co-Loc | [29] |
| β-arrestin | IP, co-Loc | [30] |
| β-subunit of the electron-transfer protein | IP, co-Loc, PLA | [27] |
| Brain-derived integrating factor-1 | IP, co-Loc | [31] |
| CASK (LIN2) | IP, co-Loc | [32] |
| Caveolin-1,2,3 | IP, co-Loc | [33,34,35] |
| Clathrin | IP, co-Loc | [36] |
| Claudin 5 | IP, co-Loc | [37] |
| CIP85 | IP, co-Loc | [38] |
| Consortin | IP, co-Loc | [39] |
| Cyclin E | IP, PLA, TEM | [40] |
| Desmocollin-2a | PD | [41] |
| Dlg | co-Loc | [42] |
| Dynamin | IP, co-Loc | [43] |
| EB1 | IP | [44] |
| Eps15 | IP, co-Loc | [45] |
| ERp29 | IP, co-Loc | [46] |
| Hrs | co-Loc | [47] |
| HSP70 | IP, PD | [48] |
| HSP90 | IP, co-Loc | [49] |
| Light chain 3 | IP, co-Loc | [50,51] |
| Lin-7 | PD | [52] |
| Myosin-VI | co-Loc | [53] |
| DMPK | IP, co-Loc | [54] |
| NaV1.5 | co-Loc | [55] |
| N-cadherin | co-Loc | [56] |
| NOV/CCN3 | IP, PD | [57] |
| Occludin | IP, co-Loc | [37] |
| p120ctn | co-Loc | [58] |
| P2X7 | IP, co-Loc | [59,60] |
| P62 | IP | [50] |
| PKG | IV | [61] |
| Plakophilin-2 | co-Loc | [62] |
| PP1/PP2A | IP, co-Loc | [63] |
| RPTPµ | IP | [64] |
| Smurf2 | IP, co-Loc | [65] |
| STAMBP (AMSH) | IP, co-Loc | [66] |
| TOM20 | IP, co-Loc | [49] |
| TRIM21 | IP, co-Loc | [67] |
| USP8 | IP | [68] |
| Vinculin | IP, co-Loc | [60] |
| Wwp1 | IP | [69] |
| ZO-2 | IP, co-Loc, PD, FW | [52,70] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorgen, P.L.; Trease, A.J.; Spagnol, G.; Delmar, M.; Nielsen, M.S. Protein–Protein Interactions with Connexin 43: Regulation and Function. Int. J. Mol. Sci. 2018, 19, 1428. https://doi.org/10.3390/ijms19051428
Sorgen PL, Trease AJ, Spagnol G, Delmar M, Nielsen MS. Protein–Protein Interactions with Connexin 43: Regulation and Function. International Journal of Molecular Sciences. 2018; 19(5):1428. https://doi.org/10.3390/ijms19051428
Chicago/Turabian StyleSorgen, Paul L., Andrew J. Trease, Gaelle Spagnol, Mario Delmar, and Morten S. Nielsen. 2018. "Protein–Protein Interactions with Connexin 43: Regulation and Function" International Journal of Molecular Sciences 19, no. 5: 1428. https://doi.org/10.3390/ijms19051428
APA StyleSorgen, P. L., Trease, A. J., Spagnol, G., Delmar, M., & Nielsen, M. S. (2018). Protein–Protein Interactions with Connexin 43: Regulation and Function. International Journal of Molecular Sciences, 19(5), 1428. https://doi.org/10.3390/ijms19051428




