Pharmacological Potential of Sea Cucumbers
Abstract
:1. Introduction
2. Biologically Active Compounds with Anticoagulant and Antithrombotic Activity
3. Biologically Active Compounds with Anticancer Activity
3.1. Triterpene Glycosides
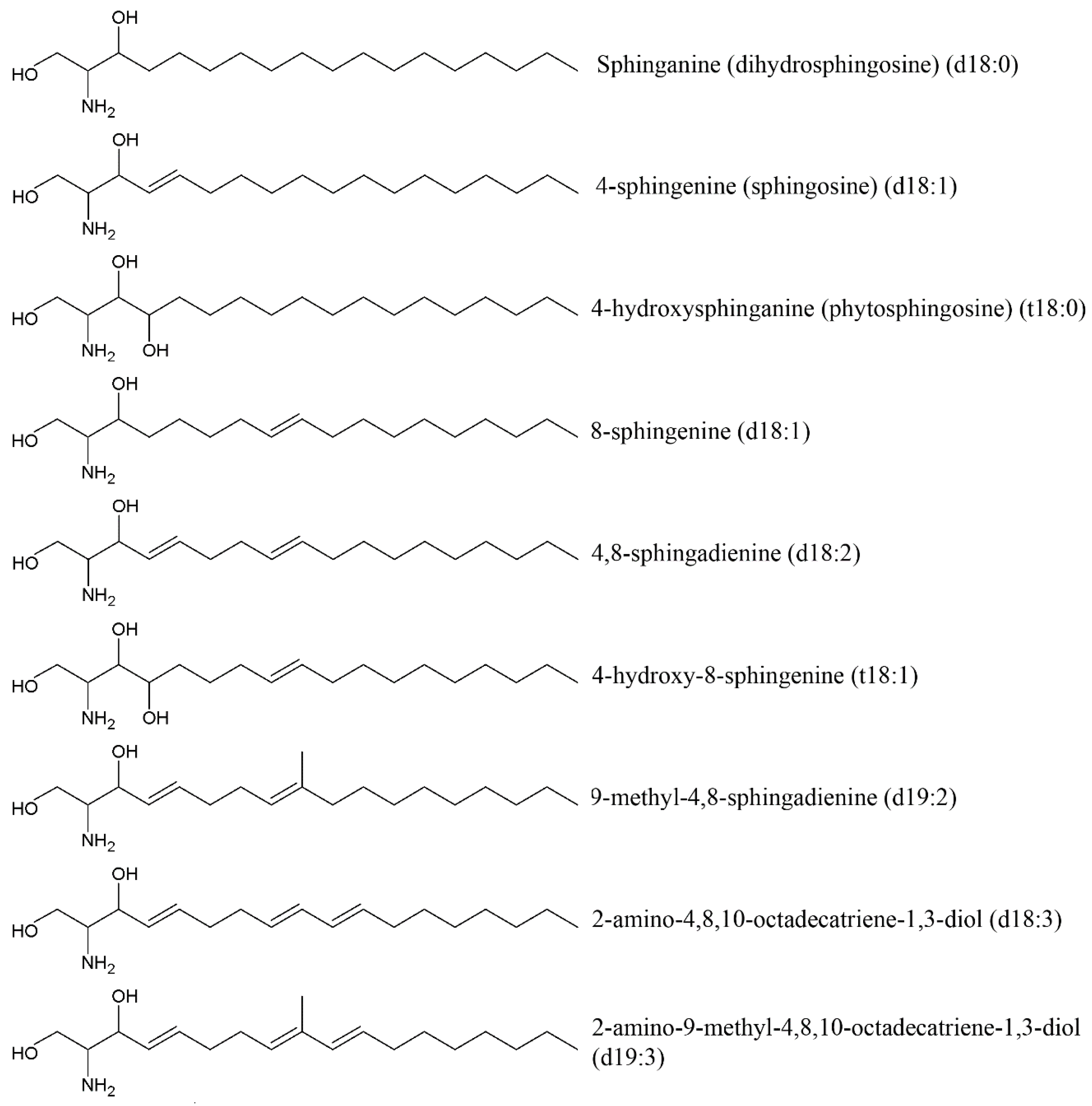
3.2. Cerebrosides
3.3. Fucosylated Chondroitin Sulfates
4. Antiprotozoal, Antibacterial, Antiviral Agents
4.1. Antimalarial Agents
4.2. Anti-Leishmania Agents
4.3. Antibacterial and Antifungal Agents
4.4. Antiviral Compounds
5. Antioxidants
6. Angiotensin-Converting Enzyme Inhibitors
7. Immunity Stimulating Agents
8. Antihyperlipidemic Agents
9. Anti-Diabetic and Glucose Lowering Activity
10. Final Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | Azino-bis-3-ehtylbenzothiazoline-6-sulfonic acid |
| APTT | Activated partial thromboplastin time |
| AT III | Antithrombin III |
| EPA-enriched PL | Eicosapentaenoic acid-enriched phospholipids |
| Fuc | α-l-Fucose |
| FuCS | Fucosylated chondroitin sulfate |
| GalNAc | N-Acetyl-β-d-galactosamine |
| GlcUA | β-d-Glucuronic acid |
| HC II | Heparin cofactor II |
| HFSD | High-fat high-sucrose |
| MIC | Minimum inhibitory concentration |
| NAFLD | Non-alcoholic fatty liver disease |
| PPAR | Peroxisome proliferator-activated receptor |
| PT | Prothrombin time |
| SCD | Stearoyl CoA desaturase |
| SREBP | Sterol-regulatory element binding protein |
| TT | Thrombin time |
References
- Adrianov, A.V. Current problems in marine biodiversity studies. Russ. J. Mar. Biol. 2004, 30, 1–16. [Google Scholar] [CrossRef]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Altmann, K.H. Drugs from the oceans: Marine natural products as leads for drug discovery. Chimia 2017, 71, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Khotimchenko, Y.S. The nutritional value of holothurians. Russ. J. Mar. Biol. 2015, 41, 409–423. [Google Scholar] [CrossRef]
- Gao, N.; Lu, F.; Xiao, C.; Yang, L.; Chen, K.; Zhou, K.; Wen, D.; Li, Z.; Wu, Z.; Jiang, J.; et al. β-Eliminative depolymerization of the fucosylated chondroitin sulfate and anticoagulant activities of resulting fragments. Carbohydr. Polym. 2015, 127, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Marques, J.; Vilanova, E.; Paulo, A.S.; Mourão, P.E.S.; Fernàndez-Busquets, X. Marine organism sulfated polysaccharides exhibiting significant antimalarial activity and inhibition of red blood cell invasion by Plasmodium. Sci. Rep. 2016, 6, 24368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, F.-J.; Xue, Y.; Liu, X.-F.; Xue, C.-H.; Wang, J.-F.; Du, L.; Takahashi, K.; Wang, Y.-M. The protective effect of eicosapentaenoic acid-enriched phospholipids from sea cucumber Cucumaria frondosa on oxidative stress in PC12 cells and SAMP8 mice. Neurochem. Int. 2014, 64, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Sila, A.; Bougatef, A. Antioxidant peptides from marine by-products: Isolation, identification and application in food systems. A review. J. Funct. Foods 2016, 21, 10–26. [Google Scholar] [CrossRef]
- Cuong, N.C.; Vien, L.T.; Hoang, L.; Hanh, T.T.H.; Thao, D.T.; Thanh, N.V.; Nam, N.H.; Thung, D.C.; Kiem, P.V.; Minh, C.V. Cytotoxic triterpene diglycosides from the sea cucumber Stichopus horrens. Bioorg. Med. Chem. Lett. 2017, 27, 2939–2942. [Google Scholar] [CrossRef] [PubMed]
- Yue, Z.; Wang, A.; Zhu, Z.; Tao, L.; Li, Y.; Zhou, L.; Chen, W.; Lu, Y. Holothurian glycosaminoglycan inhibits metastasis via inhibition of P-selectin in B16F10 melanoma cells. Mol. Cell. Biochem. 2015, 410, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Gao, Z.; Zhang, L.; Guo, F.; Chen, Y.; Li, Y.; Huang, C. Saponin-enriched sea cucumber extracts exhibit an antiobesity effect through inhibition of pancreatic lipase activity and upregulation of LXR-β signaling. Pharm. Biol. 2016, 54, 1312–1325. [Google Scholar] [CrossRef] [PubMed]
- 12 Manan, W.Z.W.A.; Mahalingam, S.R.; Arshad, K.; Bukhari, S.I.; Ming, L.C. Safety and efficacy of sea cucumber containing products. Arch. Pharm. Pract. 2016, 7, 48–52. [Google Scholar] [CrossRef]
- Mourão, P.A.; Bastos, I.G. Highly acidic glycans from sea cucumbers. Isolation and fractionation of fucose-rich sulfated polysaccharides from the body wall of Ludwigothurea grisea. Eur. J. Biochem. 1987, 166, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Vieira, R.P.; Mourão, P.A. Occurrence of a unique fucose-branched chondroitin sulfate in the body wall of a sea cucumber. J. Biol. Chem. 1988, 263, 18176–18183. [Google Scholar] [PubMed]
- Mulloy, B.; Ribeiro, A.C.; Vieira, R.P.; Mourao, P.A. Structural analysis of sulfated fucans by high-field NMR. Braz. J. Med. Biol. Res. 1994, 27, 515–521. [Google Scholar] [PubMed]
- Pomin, V.H. Holothurian fucosylated chondroitin sulfate. Mar. Drugs 2014, 12, 232–254. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Wu, M.; Xu, L.; Lian, W.; Xiang, J.; Lu, F.; Gao, N.; Xiao, C.; Wang, S.; Zhao, J. Comparison of physicochemical characteristics and anticoagulant activities of polysaccharides from three sea cucumbers. Mar. Drugs 2013, 11, 399–417. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Xu, L.; Zhao, L.; Xiao, C.; Gao, N.; Luo, L.; Yang, L.; Li, Z.; Chen, L.; Zhao, J. Structural Analysis and Anticoagulant Activities of the Novel Sulfated Fucan Possessing a Regular Well-Defined Repeating Unit from Sea Cucumber. Mar. Drugs 2015, 13, 2063–2084. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.C.; Vieira, R.P.; Mourao, P.A.; Mulloy, B. A sulfated alpha-l-fucan from sea cucumber. Carbohydr. Res. 1994, 255, 225–240. [Google Scholar] [CrossRef]
- Chen, S.; Hu, Y.; Ye, X.; Li, G.; Yu, G.; Xue, C.; Chai, W. Sequence determination and anticoagulant and antithrombotic activities of a novel sulfated fucan isolated from the sea cucumber Isostichopus badionotus. BBA Gen. Subj. 2012, 1820, 989–1000. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Mulloy, B.; Imai, K.; Tominaga, A.; Kaneko, T.; Asari, A.; Suzuki, K.; Masuda, H.; Kyogashima, M.; Ishii, T. Isolation and partial characterization of fucan sulfates from the body wall of sea cucumber Stichopus japonicus and their ability to inhibit osteoclastogenesis. Carbohydr. Res. 2004, 339, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-H.; Li, S.; Zhi, Z.-J.; Yan, L.-F.; Ye, X.-Q.; Ding, T.; Yan, L.; Linhardt, R.J.; Chen, S.-G. Depolymerization of Fucosylated Chondroitin Sulfate with a Modified Fenton-System and Anticoagulant Activity of the Resulting Fragments. Mar. Drugs 2016, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Mourão, P.A.S.; Pereira, M.S.G.; Pavão, M.S.G.; Mulloy, B.; Tollefsen, D.M.; Mowinckel, M.C.; Abildgaard, U. Structure and anticoagulant activity of a fucosylated chondroitin sulfate from Echinoderm. Sulfated fucose branches on the polysaccharide account for its high anticoagulant action. J. Biol. Chem. 1996, 271, 23973–23984. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Xue, C.; Yin, L.; Tang, Q.; Yu, G.; Chai, W. Comparison of structures and anticoagulant activities of fucosylated chondroitin sulfates from different sea cucumbers. Carbohydr. Polym. 2011, 83, 688–696. [Google Scholar] [CrossRef]
- Yang, J.; Wang, Y.; Jiang, T.; Lv, Z. Novel branch patterns and anticoagulant activity of glycosaminoglycan from sea cucumber Apostichopus japonicus. Int. J. Biol. Macromol. 2015, 72, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Mou, J.; Ding, D.; Wang, X. In vivo and in vitro antithrombus activities of depolymerized holothurian polysaccharides. Int. J. Biol. Macromol. 2017, 94, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Vieira, R.P.; Mulloy, B.; Mourão, P.A.S. Structure of a fucose-branched chondroitin sulfate from sea cucumber. Evidence for the presence of 3-O-sulfo-beta-d-glucuronosyl residues. J. Biol. Chem. 1991, 266, 13530–13536. [Google Scholar] [PubMed]
- Yoshida, K.; Minami, Y.; Nemoto, H.; Numata, K.; Yamanaka, E. Structure of DHG, depolymerized glycosaminoglycan from sea cucumber, Stichopus japonicus. Tetrahedron Lett. 1992, 33, 4959–4962. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Nifantiev, N.E.; Usov, A.I. Two fucosylated chondroitin sulfates from the sea cucumber Eupentacta fraudatrix. Carbohydr. Polym. 2017, 164, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. The structure of a fucosylated chondroitin sulfate from the sea cucumber Cucumaria frondosa. Carbohydr. Polym. 2017, 165, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, Y.; Hao, J.; Zhao, X.; Lang, Y.; Fan, F.; Cai, C.; Li, G.; Zhang, L.; Yu, G. In vivo anti-cancer mechanism of low-molecular-weight fucosylated chondroitin sulfate (LFCS) from sea cucumber Cucumaria frondosa. Molecules 2016, 21, 625. [Google Scholar] [CrossRef] [PubMed]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Borodina, E.Y.; Stonik, V.A.; Nifantiev, N.E.; Usov, A.I. A highly regular fucosylated chondroitin sulfate from the sea cucumber Massinium magnum: Structure and effects on coagulation. Carbohydr. Polym. 2017, 167, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Mou, J.; Wang, C.; Li, W.; Yang, J. Purification, structural characterization and anticoagulant properties of fucosylated chondroitin sulfate isolated from Holothuria mexicana. Int. J. Biol. Macromol. 2017, 98, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Mourao, P.A.S.; Pereira, M.S. Searching for Alternatives to Heparin: Sulfated Fucans from Marine Invertebrates. Trends Cardiovasc. Med. 1999, 9, 225–232. [Google Scholar] [CrossRef]
- Glauser, B.F.; Pereira, M.S.; Monteiro, R.Q.; Mourão, P.A.S. Serpin-independent anticoagulant activity of a fucosylated chondroitin sulfate. Thromb. Haemost. 2008, 100, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H.; Mourão, P.A.S. Structure, biology, evolution and medical importance of sulfated fucans and galactans. Glycobiology 2008, 18, 1016–1027. [Google Scholar] [CrossRef] [PubMed]
- Buyue, Y.; Sheehan, J.P. Fucosylated chondroitin sulfate inhibits plasma thrombin generation via targeting of the factor IXa heparin-binding exosite. Blood 2009, 114, 3092–3100. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, R.J.; Mourao, P.A. Fucosylated chondroitin sulfate as a new oral antithrombotic agent. Thromb. Haemost. 2006, 96, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wen, D.; Gao, N.; Xiao, C.; Yang, L.; Xu, L.; Lian, W.; Peng, W.; Jiang, J.; Zhao, J. Anticoagulant and antithrombotic evaluation of native fucosylated chondroitin sulfates and their derivatives as selective inhibitors of intrinsic factor Xase. Eur. J. Med. Chem. 2015, 92, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Panagos, C.G.; Thomson, D.S.; Moss, C.; Hughes, A.D.; Kelly, M.S.; Liu, Y.; Chai, W.; Venkatasamy, R.; Spina, D.; Page, C.P.; et al. Fucosylated chondroitin sulfates from the body wall of the sea cucumber Holothuria forskali: Conformation, selectin binding, and biological activity. J. Biol. Chem. 2014, 289, 28284–28298. [Google Scholar] [CrossRef] [PubMed]
- Melo, F.R.; Pereira, M.S.; Foguel, D.; Mourão, P.A.S. Antithrombin-mediated anticoagulant activity of sulfated polysaccharides. J. Biol. Chem. 2004, 279, 20824–20835. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Xu, S.; Zhao, J.; Kang, H.; Ding, H. Free-radical depolymerization of glycosaminoglycan from sea cucumber Thelenota ananas by hydrogen peroxide and copper ions. Carbohydr. Polym. 2010, 80, 1116–1124. [Google Scholar] [CrossRef]
- Wu, M.; Xu, S.; Zhao, J.; Kang, H.; Ding, H. Physicochemical characteristics and anticoagulant activities of low molecular weight fractions by free-radical depolymerization of a fucosylated chondroitin sulphate from sea cucumber Thelenata ananas. Food Chem. 2010, 122, 716–723. [Google Scholar] [CrossRef]
- Wu, M.; Huang, R.; Wen, D.; Gao, N.; He, J.; Li, Z.; Zhao, J. Structure and effect of sulfated fucose branches on anticoagulant activity of the fucosylated chondroitin sulfate from sea cucumber Thelenota ananas. Carbohydr. Polym. 2012, 87, 862–868. [Google Scholar] [CrossRef]
- Haroun-Bouhedja, F.; Ellouali, M.; Sinquin, C.; Boisson-Vidal, C. Relationship between sulfate groups and biological activities of fucans. Thromb. Res. 2000, 100, 453–459. [Google Scholar] [CrossRef]
- Fonseca, R.J.C.; Santos, G.R.C.; Mourao, P.A.S. Effects of polysaccharides enriched in 2,4-disulfated fucose units on coagulation, thrombosis and bleeding. Thromb. Haemost. 2009, 102, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Mourão, P.A.S.; Boisson-Vidal, C.; Tapon-Bretaudiere, J.; Drouet, B.; Bros, A.; Fischer, A. Inactivation of thrombin by a fucosylated chondroitin sulfate from echinoderm. Thromb. Res. 2001, 102, 167–176. [Google Scholar] [CrossRef]
- Mourão, P.A. Use of sulfated fucans as anticoagulant and antithrombotic agents: Future perspectives. Curr. Pharm. Des. 2004, 10, 967–981. [Google Scholar] [CrossRef] [PubMed]
- Nagase, H.; Enjyoji, K.; Minamiguchi, K.; Kitazato, K.T.; Kitazato, K.; Saito, H.; Kato, H. Depolymerized holothurian glycosaminoglycan with novel anticoagulant actions: Antithrombin III- and heparin cofactor II-independent inhibition of factor X activation by factor IXa-factor VIIIa complex and heparin cofactor II-dependent inhibition of thrombin. Blood 1995, 85, 1527–1534. [Google Scholar] [PubMed]
- Kitazato, K.; Kitazato, K.T.; Sasaki, E.; Minamiguchi, K.; Nagase, H. Prolonged bleeding time induced by anticoagulant glycosaminoglycans in dogs is associated with the inhibition of thrombin-induced platelet aggregation. Thromb. Res. 2003, 112, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, J.; Walke, E. Depolymerized holothurian glycosaminoglycan and heparin inhibit the intrinsic tenase complex by a common antithrombin-independent mechanism. Blood 2006, 107, 3876–3882. [Google Scholar] [CrossRef] [PubMed]
- Hirsh, J.; Warkentin, T.E.; Shaughnessy, S.G.; Anand, S.S.; Halperin, J.L.; Raschke, R.; Granger, C.; Ohman, E.M.; Dalen, J.E. Heparin and low-molecular-weight heparin: Mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest 2001, 119, 64S–94S. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hao, J.; Shan, X.; Zhang, X.; Zhao, X.; Li, Q.; Wang, X.; Cai, C.; Li, G.; Yu, G. Antithrombotic activities of fucosylated chondroitin sulfates and their depolymerized fragments from two sea cucumbers. Carbohydr. Polym. 2016, 152, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Lai, S.; Huang, R.; Wu, M.; Gao, N.; Xu, L.; Qin, H.; Peng, W.; Zhao, J. Structure and anticoagulant activity of fucosylated glycosaminoglycan degraded by deaminative cleavage. Carbohydr. Polym. 2013, 98, 1514–1523. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Wu, M.; Liu, S.; Lian, W.; Li, Z.; Zhao, J. Preparation and characterization of O-acylated fucosylated chondroitin sulfate from sea cucumber. Mar. Drugs 2012, 10, 1647–1661. [Google Scholar] [CrossRef] [PubMed]
- Nigrelli, R.F. The effects of holothurin on fish and mice with sarcoma 180. Zoologica 1952, 37, 89–90. [Google Scholar]
- Sullivan, T.D.; Nigrelli, R.F. The antitumorous action of biologics of marine origin I. Survival of Swiss mice inoculated with Krebs-2 ascites tumor and treated with holothurin, a steroid saponin from the sea cucumber, Actinopyga agassizi. Proc. Am. Assoc. Cancer Res. 1956, 2, 151–155. [Google Scholar]
- Friess, S.L.; Standaert, F.G.; Whitcomb, E.R.; Nigrelli, R.F.; Chanley, J.D.; Sobotka, H. Some pharmacologic properties of holothurin A, a glycosidic mixture from the sea cucumber. Ann. N. Y. Acad. Sci. 1960, 90, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Mondol, M.A.M.; Shin, H.J.; Rahman, M.A.; Islam, M.T. Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties. Mar. Drugs 2017, 15, 317. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Menchinskaya, E.S.; Pisliagin, E.A.; Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I. Anticancer Activity of Sea Cucumber Triterpene Glycosides. Mar. Drugs 2015, 13, 1202–1223. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-S.; Yi, Y.-H.; Li, L.; Zhang, S.-L.; Han, H.; Weng, Y.-Y.; Pan, M.-X. Arguside A: A new cytotoxic triterpene glycoside from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2007, 4, 2845–2851. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-S.; Yi, Y.-H.; Li, L.; Sun, P.; Yuan, W.-H.; Sun, G.-Q.; Yan, H.; Xue, M. Argusides B and C, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2008, 5, 1288–1297. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-S.; Yi, Y.-H.; Li, L.; Sun, P.; Han, H.; Sun, G.-Q.; Wang, X.-H.; Wang, Z.-L. Argusides D and E, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2008, 5, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Liu, B.-S.; Yi, Y.-H.; Li, L.; Giu, M.; Tang, H.-F.; Zhang, D.-Z.; Zhang, S.-L. A new cytotoxic lanostane-type triterpene glycoside from the sea cucumber Holothuria impatiens. Chem. Biodivers. 2007, 4, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yi, Y. Studies on antitumor activities of triterpene glycoside colochiroside A 1515 from sea cucumber Colochirus anceps. Zhongguo Zhong Yao Za Zhi 2011, 36, 504–507. [Google Scholar] [PubMed]
- Jin, J.-O.; Shastina, V.V.; Shin, S.-W.; Xu, Q.; Park, J.-I.; Rasskazov, V.A.; Avilov, S.A.; Fedorov, S.N.; Stonik, V.A.; Kwak, J.-Y. Differential effects of triterpene glycosides, frondoside A and cucumarioside A2–2 isolated from sea cucumbers on caspase activation and apoptosis of human leukemia cells. FEBS Lett. 2009, 583, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Han, H.; Chen, X.; Yi, Y.; Sun, H. Cytotoxic and apoptosis-inducing activity of triterpene glycosides from Holothuria scabra and Cucumaria frondosa against HepG2 cells. Mar. Drugs 2014, 12, 4274–4290. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Xue, Y.; Wang, J.-F.; Li, H.; Long, T.-T.; Li, Z.; Wang, Y.-M.; Dong, P.; Xue, C.-H. In vitro and in vivo anti-tumor activities of echinoside A and ds-echinoside A from Pearsonothuria graeffei. J. Sci. Food Agric. 2012, 92, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, Z.-D.; Xue, Y.; Wang, J.-F.; Li, H.; Tang, Q.-J.; Wang, Y.-M.; Dong, P.; Xue, C.-H. Ds-echinoside A, a new triterpene glycoside derived from sea cucumber, exhibits antimetastatic activity via the inhibition of NF-κB-dependent MMP-9 and VEGF expressions. J. Zhejiang Univ. Sci. B 2011, 12, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Xue, Y.; Liu, Z.-D.; Li, H.; Wang, J.-F.; Li, Z.-J.; Wang, Y.-M.; Dong, P.; Xue, C.-H. Differential effects of sulfated triterpene glycosides, Holothurin A1, and 24-dehydroechinoside A, on antimetastasic activity via regulation of the MMP-9 signal pathway. J. Food Sci. 2010, 75, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Ye, X.; Huang, H.; Peng, R.; Su, Z.; Lian, X.-Y.; Zhang, Z. Bioactive sulfated saponins from sea cucumber Holothuria moebii. Planta Med. 2015, 81, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Roginsky, A.B.; Ding, X.-Z.; Woodward, C.; Collin, P.; Newman, R.A.; Bell, R.H., Jr.; Adrian, T.E. Review of the apoptosis pathways in pancreatic cancer and the anti-apoptotic effects of the novel sea cucumber compound, frondoside A. Ann. N. Y. Acad. Sci. 2008, 1138, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Arafat, K.; Gelaude, A.; Al Sultan, M.A.; Bracke, M.; Collin, P.; Takahashi, T.; Adrian, T.E.; De Wever, O. Frondoside A suppressive effects on lung cancer survival, tumor growth, angiogenesis, invasion, and metastasis. PLoS ONE 2013, 8, e53087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, B.C.Q.; Yoshimura, K.; Kumazawa, S.; Tawata, S.; Maruta, H. Frondoside A from sea cucumber and nymphaeols from Okinawa propolis: Natural anti-cancer agents that selectively inhibit PAK1 in vitro. Drug Discov. Ther. 2017, 11, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-Y.; Yi, Y.-H.; Tang, H.-F. Bioactive triterpene glycosides from the sea cucumber Holothuria fuscocinerea. J. Nat. Prod. 2006, 69, 1492–1495. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.-Q.; Li, L.; Yi, Y.-H.; Yuan, W.-H.; Liu, B.-S.; Weng, Y.-Y.; Zhang, S.-L.; Sun, P.; Wang, Z.-L. Two new cytotoxic nonsulfated pentasaccharide holostane (=20-Hydroxylanostan-18-oic Acid γ-Lactone) glycosides from the sea cucumber Holothuria grisea. Helv. Chim. Acta 2008, 91, 1453–1460. [Google Scholar] [CrossRef]
- Wu, J.; Yi, Y.-H.; Tang, H.-F.; Zou, Z.-R.; Wu, H.-M. Structure and cytotoxicity of a new lanostane-type triterpene glycoside from the sea cucumber Holothuria hilla. Chem. Biodivers. 2006, 3, 1249–1254. [Google Scholar] [CrossRef] [PubMed]
- Dang, N.H.; Van Thanh, N.; Van Kiem, P.; Huong, L.M.; Van Minh, C.; Kim, Y.H. Two new triterpene glycosides from the Vietnamese sea cucumber Holothuria scabra. Arch. Pharm. Res. 2007, 30, 1387–1391. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.-R.; Yi, Y.-H.; Wu, H.-M.; Wu, J.-H.; Liaw, C.-C.; Lee, K.-H. Intercedensides A–C, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2003, 66, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- 80 Zhang, Z.; Yu, S.; Chen, L.; Lian, X. Moebioside A as well as preparation and application thereof. Patent CN 104151392A, 19 November 2014. [Google Scholar]
- Careaga, V.P.; Bueno, C.; Muniain, C.; Alche, L.; Maier, M.S. Antiproliferative, cytotoxic and hemolytic activities of a triterpene glycoside from Psolus patagonicus and its desulfated analog. Chemotherapy 2009, 55, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Xu, Q.-Z.; Tang, H.-F.; Yi, Y.-H.; Gong, W. Cytotoxic holostane-type triterpene glycosides from the sea cucumber Pentacta quadrangularis. Planta Med. 2010, 76, 1900–1904. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Zhang, X.; Tian, F.; Yi, Y.; Xu, Q.; Li, L.; Tong, L.; Lin, L.; Ding, J. Philinopside A, a novel marine-derived compound possessing dual anti-angiogenetic and anti-tumor effects. Int. J. Cancer 2005, 114, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-L.; Li, L.; Yi, Y.-H.; Sun, P. Philinopsides E and F, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Nat. Prod. Res. 2006, 20, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Zhang, X.W.; Tong, Y.G.; Yi, Y.; Zhang, S.L.; Li, L.; Sun, P.; Lin, L.P.; Ding, J. PE, a new sulfated saponin from sea cucumber, exhibits anti-angiogenic and anti-tumor activities in vitro and in vivo. Cancer Biol. Ther. 2005, 48, 874–882. [Google Scholar] [CrossRef]
- Yun, S.-H.; Park, E.-S.; Shin, S.-W.; Na, Y.-W.; Han, J.-Y.; Jeong, J.-S.; Shastina, V.V.; Stonik, V.A.; Park, J.-I.; Kwak, J.-Y. Stichoposide C induces apoptosis through the generation of ceramide in leukemia and colorectal cancer cells and shows in vivo antitumor activity. Clin. Cancer Res. 2012, 18, 5934–5948. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-Y.; Yi, Y.-H.; Tang, H.-F. Cytotoxic Sulfated Triterpene Glycosides from the Sea Cucumber Pseudocolochirus violaceus. Chem. Biodivers. 2006, 3, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.R.; Kundu, N.; Collin, P.D.; Goloubeva, O.; Fulton, A.M. Frondoside A inhibits breast cancer metastasis and antagonizes prostaglandin E receptors EP4 and EP2. Breast Cancer Res. Treat. 2012, 132, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Al Shemaili, J.; Parekh, K.A.; Newman, R.N.; Hellman, B.; Woodward, C.; Adem, A.; Collin, P.; Adrian, T.E. Pharmacokinetics in mouse and comparative effects of frondosides in pancreatic Cancer. Mar. Drugs 2016, 14, 115. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Li, L.; Yi, Y.-H.; Wang, X.-H.; Pan, M.-X. Triterpene glycosides from sea cucumber Holothuria scabra with cytotoxic activity. Chin. Herb. Med. 2012, 4, 183–188. [Google Scholar]
- Assawasuparerk, K.; Vanichviriyakit, R.; Chotwiwatthanakun, C.; Nobsathian, S.; Rawangchue, T.; Wittayachumnankul, D. Scabraside D Extracted from Holothuria scabra Induces Apoptosis and Inhibits Growth of Human Cholangiocarcinoma Xenografts in Mice. Asian Pac. J. Cancer Prev. 2016, 17, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Ye, X.; Chen, L.; Xie, X.; Zhou, Q.; Lian, X.-Y.; Zhang, Z. Cytotoxic and anti-colorectal tumor effects of sulfated saponins from sea cucumber Holothuria moebii. Phytomedicine 2015, 22, 22–1112. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Aida, K.; Duan, J.; Hirata, T. Analysis of glucosylceramides from various sources by liquid chromatography-ion trap mass. J. Oleo Sci. 2010, 59, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Duan, J.; Xue, C.; Feng, T.; Dong, P.; Sugawara, T.; Hirata, T. Analysis and comparison of glucocerebroside species from three edible sea cucumbers using liquid chromatography-ion trap-time-of-flight mass spectrometry. J. Agric. Food Chem. 2011, 59, 12246–12253. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Xu, J.; Xue, Y.; Gao, Z.; Li, Z.J.; Leng, K.L.; Wang, J.F.; Xue, C.H.; Wang, Y.M. Sea cucumber cerebrosides and long-chain bases from Acaudina molpadioides protect against high fat diet-induced metabolic disorders in mice. Food Funct. 2015, 6, 3428–3436. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Zaima, N.; Yamamoto, A.; Sakai, S.; Noguch, R.; Hirata, T. Isolation of sphingoid bases of sea cucumber cerebrosides and their cytotoxicity against human colon cancer cells. Biosci. Biotechnol. Biochem. 2006, 70, 2906–2912. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Li, S.; Cong, P.; Wang, Y.; Sugawara, T.; Xue, C.; Xu, J. High throughput analysis of cerebrosides from the sea cucumber Pearsonothria graeffei by liquid chromatography-quadrupole-time-of-flight mass spectrometry. J. Oleo Sci. 2015, 64, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, Y.; Inagaki, M.; Yamada, K.; Zhang, X.W.; Zhang, B.; Miyamoto, T.; Higuchi, R. Isolation and structure of a galactocerebroside from the sea cucumber Bohadschia argus. Chem. Pharm. Bull. 2009, 57, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Guo, S.; Du, L.; Wang, Y.-M.; Sugawara, T.; Hirata, T.; Xue, C.-H. Isolation of cytotoxic glucocerebrosides and long-chain bases from sea cucumber Cucumaria frondosa using high speed counter-current chromatography. J. Oleo Sci. 2013, 62, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Li, Z.-J.; Xu, J.; Wang, J.-F.; Xue, Y.; Xue, C.-H.; Takahashi, K.; Wang, Y.-M. The anti-tumor activities of cerebrosides derived from sea cucumber Acaudina molpadioides and starfish Asterias amurensis in vitro and in vivo. J. Oleo Sci. 2012, 61, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Hossain, Z.; Sugawara, T.; Hirata, T. Sphingoid bases from sea cucumber induce apoptosis in human hepatoma HepG2 cells through p-AKT and DR5. Oncol. Rep. 2013, 29, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Tao, L.; Wang, Y.; Zhang, F.; Li, M.; Huang, S.; Wang, A.; Chen, W.; Yue, Z.; Chen, L.; et al. Downregulation of integrins in cancer cells and anti-platelet properties are involved in holothurian glycosaminoglycan-mediated disruption of the interaction of cancer cells and platelets in hematogenous metastasis. J. Vasc. Res. 2015, 52, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.F.; Albrecht, L.; Kozlowski, E.O.; Lopes, S.C.P.; Blanco, Y.C.; Carlos, B.C.; Castiñeiras, C.C.; Vicente, C.P.; Wermeck, C.C.; Wunderlich, G.; et al. Fucosylated chondroitin sulfate inhibits plasmodium falciparum cytoadhesion and merozoite invasion. Antimicrob. Agents Chemother. 2014, 58, 1862–1871. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Kumar, R.; Gupta, S.; Dube, A.; Lakshmi, V. Antileishmanial activity in vitro and in vivo of constituents of sea cucumber Actinopyga lecanora. Parasitol. Res. 2008, 103, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Chaturvedi, A.K.; Shukla, P.K.; Lakshmi, V. Antifungal activity in triterpene glycosides from the sea cucumber Actinopyga lecanora. Bioorg. Med. Chem. Lett. 2007, 17, 4387–4391. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.-H.; Yi, Y.; Tang, H.-F.; Liu, B.-S.; Wang, Z.-L.; Sun, G.-Q.; Zhang, W.; Li, L.; Sun, P. Antifungal triterpene glycosides from the sea cucumber Bohadschia marmorata. Planta Med. 2009, 75, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi, V.; Srivastava, S.; Mishra, S.K.; Shukla, P.K. Antifungal activity of bivittoside-D from Bohadschia vitiensis (Semper). Nat. Prod. Res. 2012, 26, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Yano, A.; Abe, A.; Aizawa, F.; Yamada, H.; Minami, K.; Matsui, M.; Kishi, M. The Effect of eating sea cucumber jelly on candida load in the oral cavity of elderly individuals in a nursing home. Mar. Drugs 2013, 11, 4993–5007. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Wu, M.-Y.; Zheng, C.-B.; Zhu, L.; Zhao, J.-H.; Zheng, Y.-T. The depolymerized fucosylated chondroitin sulfate from sea cucumber potently inhibits HIV replication via interfering with virus entry. Carbohydr. Res. 2013, 380, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Khademvatan, S.; Eskandari, A.; Saki, J.; Foroutan-Rad, M. Cytotoxic activity of Holothuria leucospilota Extract against Leishmania infantum in vitro. Adv. Pharmacol. Sci. 2016, 8195381. [Google Scholar] [CrossRef]
- Farjami, B.; Nematollahi, M.A.; Moradi, Y.; Shakouri, A.; Ameri, A. Antibacterial activity of the sea cucumber Holothuria leucospilota. Int. J. Mol. Clin. Microbiol. 2013, 1, 225–230. [Google Scholar]
- Adibpour, N.; Nasr, F.; Nematpour, F.; Shakouri, A.; Ameri, A. Antibacterial and antifungal activity of Holothuria leucospilota isolated from Persian gulf and Oman sea. Jundishapur J. Microbiol. 2014, 7, e8708. [Google Scholar] [CrossRef] [PubMed]
- Jawahar, A.T.; Nagarajan, J.; Shanmugam, S.A. Antimicrobial sub-stances of potential biomedical importance from Holothurian species. Ind. J. Mar. Sci. 2002, 31, 161–164. [Google Scholar]
- Mohammadizadeh, F.; Ehsanpor, M.; Afkhami, M.; Mokhlesi, A.; Khazaali, A.; Montazeri, S. Evaluation of antibacterial, antifungal and cytotoxic effects of Holothuria scabra from the North Coast of the Persian Gulf. J. Mycol. Med. 2013, 23, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Mohammadizadeh, F.; Ehsanpor, M.; Afkhami, M.; Mokhlesi, A.; Khazaali, A.; Montazeri, S. Antibacterial, antifungal and cytotoxic effects of a sea cucumber Holothuria leucospilota, from the north coast of the Persian Gulf. J. Mar. Biol. Assoc. UK 2013, 93, 1401–1405. [Google Scholar] [CrossRef]
- Ghanbari, R.; Ebrahimpour, A.; Abdul-Hamid, A.; Ismail, A.; Saari, N. Actinopyga lecanora hydrolysates as natural antibacterial agents. Int. J. Mol. Sci. 2012, 13, 16796–16811. [Google Scholar] [CrossRef] [PubMed]
- Farshadpour, F.; Gharibi, S.; Taherzadeh, M.; Amirinejad, R.; Taherkhani, R.; Habibian, A.; Zandi, K. Antiviral activity of Holothuria sp. a sea cucumber against herpes simplex virus type 1 (HSV-1). Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 333–337. [Google Scholar] [PubMed]
- Esmat, A.Y.; Said, M.M.; Soliman, A.A.; El-Masry, K.S.H.; Badiea, E.A. Bioactive compounds, antioxidant potential, and hepatoprotective activity of sea cucumber (Holothuria atra) against thioacetamide intoxication in rats. Nutrition 2013, 29, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Dakrory, A.I.; Fahmy, S.R.; Soliman, A.M.; Mohamed, A.S.; Amer, S.A.M. Protective and curative effects of the sea cucumber Holothuria atra extract against DMBA-induced hepatorenal diseases in rats. Biomed. Res. Int. 2015, 563652. [Google Scholar] [CrossRef]
- Fahmy, S.R.; Mohamed, A.S. Holoturia arenicola extract modulates bile duct ligation-induced oxidative stress in rat kidney. Int. J. Clin. Exp. Pathol. 2015, 8, 1649–1657. [Google Scholar] [PubMed]
- Roggatz, C.C.; Gonzáles-Wangüemert, M.; Pereira, H.; Rodrigues, M.J.; da Silva, M.M.; Barreira, L.; Varela, J.; Custódio, L. First report of the nutritional profile and antioxidant potential of Holothuria arguinensis, a new resource for aquaculture in Europe. Nat. Prod. Res. 2016, 30, 2034–2040. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, J.; Zhao, Y.; Hu, S.; Shi, D.; Xue, C. Fucoidan from sea cucumber Cucumaria frondosa exhibits anti-hyperglycemic effects in insulin resistant mice via activating the PI3K/PKB pathway and GLUT4. J. Biosci. Bioeng. 2016, 121, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, Y.; Furukawa, A.; Shigai, I.; Muroi, Y.; Ishii, T.; Hongo, Y.; Takahashi, S.; Sugawara, T.; Koshino, H.; Ohnishi, M. Cytoprotective effects of lysophospholipids from sea cucumber Holothuria atra. PLoS ONE 2015, 10, e0135701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soltani, M.; Parivar, K.; Baharara, J.; Kerachian, M.A.; Asili, J. Putative mechanism for apoptosis-inducing properties of crude saponin isolated from sea cucumber (Holothuria leucospilota) as an antioxidant compound. Iran. J. Basic Med. Sci. 2015, 18, 180–187. [Google Scholar] [PubMed]
- Wu, F.-J.; Xue, Y.; Tang, Q.-J.; Xu, J.; Du, L.; Xue, C.-H.; Takahashi, K.; Wang, Y.-M. The protective effect of cerebrosides from sea cucumber and starfish on the oxidative damage in PC12 cells. J. Oleo Sci. 2013, 62, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Che, H.; Du, L.; Cong, P.; Tao, S.; Ding, N.; Wu, F.; Xue, C.; Xu, J.; Wang, Y. Cerebrosides from sea cucumber protect against oxidative stress in SAMP8 mice and PC12 cells. J. Med. Food. 2017, 20, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Chai, T.-T.; Law, Y.-C.; Wong, F.-C.; Kim, S.-K. Enzyme-assisted discovery of antioxidant peptides from edible marine invertebrates: A review. Mar. Drugs 2017, 15, 42. [Google Scholar] [CrossRef] [PubMed]
- Zou, T.-B.; He, T.-P.; Li, H.-B.; Tang, H.-W.; Xia, E.-Q. The structure-activity relationship of the antioxidant peptides from natural proteins. Molecules 2016, 21, 72. [Google Scholar] [CrossRef] [PubMed]
- Abedin, M.Z.; Karim, A.A.; Latiff, A.A.; Gan, C.-Y.; Ghazali, F.C.; Barzideh, Z.; Ferdosh, S.; Akanda, M.J.H.; Zaman, W.; Karim, M.R.; et al. Biochemical and radical-scavenging properties of sea cucumber (Stichopus vastus) collagen hydrolysates. Nat. Prod. Res. 2014, 28, 1302–1305. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Vega, J.A.; Olivera-Castillo, L.; Gómez-Ruiz, J.A.; Hernández-Ledesma, B. Release of multifunctional peptides by gastrointestinal digestion of sea cucumber (Isostichopus badionotus). J. Funct. Foods 2013, 5, 869–877. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, C.; Jiang, A. Antioxidant peptides isolated from sea cucumber Stichopus Japonicus. Eur. Food Res. Technol. 2012, 234, 441–447. [Google Scholar] [CrossRef]
- Ghanbari, R.; Zarei, M.; Ebrahimpour, A.; Abdul-Hamid, A.; Ismail, A.; Saari, N. Angiotensin-I converting enzyme (ace) inhibitory and anti-oxidant activities of sea cucumber (Actinopyga lecanora) hydrolysates. Int. J. Mol. Sci. 2015, 16, 28870–28885. [Google Scholar] [CrossRef] [PubMed]
- Auwal, S.M.; Zarei, M.; Abdul-Hamid, A.; Saari, N. Optimization of bromelain-aided production of angiotensin I-converting enzyme inhibitory hydrolysates from stone fish using response surface methodology. Mar. Drugs 2017, 15, 104. [Google Scholar] [CrossRef] [PubMed]
- Forghani, B.; Ebrahimpour, A.; Bakar, J.; Abdul Hamid, A.; Hassan, Z.; Saari, N. Enzyme hydrolysates from Stichopus horrens as a new source for angiotensin-converting enzyme inhibitory peptides. Evid.-Based Complement. Altern. Med. 2012, 236384. [Google Scholar] [CrossRef]
- Vishkaei, M.S.; Ebrahimpour, A.; Abdul-Hamid, A.; Ismail, A.; Saari, N. Angiotensin-I converting enzyme (ACE) inhibitory and anti-hypertensive effect of protein hydrolysate from Actinopyga lecanora (Sea Cucumber) in Rats. Mar Drugs 2016, 14, 176. [Google Scholar] [CrossRef] [PubMed]
- Agafonova, I.G.; Aminin, D.L.; Avilov, S.A.; Stonik, V.A. Influence of cucumariosides upon intracellular [Ca2+] and lysosomal activity of macrophages. J. Agric. Food Chem. 2003, 51, 6982–6986. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Silchenko, A.S.; Avilov, S.A.; Stepanov, V.G.; Kalinin, V.I. Immunomodulatory action of monosulfated triterpene glycosides from the sea cucumber Cucumaria okhotensis: Stimulation of activity of mouse peritoneal macrophages. Nat. Prod. Commun. 2010, 5, 1877–1880. [Google Scholar] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Woodward, C. Immunomodulatory properties of frondoside A, a major triterpene glycoside from the North Atlantic commercially harvested sea cucumber Cucumaria frondosa. J. Med. Food 2008, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, F.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Menchinskaya, E.S.; Aminin, D.L.; Kalinin, V.I. Structure of cucumarioside I2 from the sea cucumber Eupentacta fraudatrix (Djakonov et Baranova) and cytotoxic and immunostimulatory activities of this saponin and relative compounds. Nat. Prod. Res. 2013, 27, 1776–1783. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Jin, S.-J.; Cui, L.-H.; Ji, X.-J.; Yang, F.-G. Immunomodulatory effect of Stichopus japonicas acid mucopolysaccharide on experimental hepatocellular carcinoma in rats. Molecules 2013, 18, 7179–7193. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Li, X.; Cao, B.; Zuo, T.; Wu, J.; Wang, J.; Xue, C.; Tang, Q. Dietary Apostichopus japonicus enhances the respiratory and intestinal mucosal immunity in immunosuppressive mice. Biosci. Biotechnol. Biochem. 2015, 79, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, Z.; Zhang, M.; Meng, X.; Xia, X.; Yuan, W.; Xue, F.; Liu, C. Antioxidant and antihyperlipidemic activities of polysaccharides from sea cucumber Apostichopus japonicas. Carbohydr. Polym. 2012, 90, 1664–1670. [Google Scholar] [CrossRef] [PubMed]
- Olivera-Castillo, L.; Davalos, A.; Grant, G.; Valadez-Gonzalez, N.; Montero, J.; Barrera-Perez, H.A.M.; Chim-Chi, Y.; Olvera-Novoa, M.A.; Ceja-Moreno, V.; Acereto-Escoffie, P.; et al. Diets containing sea cucumber (Isostichopus badionotus) meals are hypocholesterolemic in young rats. PLoS ONE 2015, 10, e0125451. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Q.; Wang, Y.-M.; Wang, J.-F.; Xue, Y.; Li, Z.-J.; Nagao, K.; Yanagita, T.; Xue, C.-H. Dietary saponins of sea cucumber alleviate orotic acid-induced fatty liver in rats via PPARa and SREBP-1c signaling. Lipids Health Dis. 2010, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Li, Z.; Xue, Y.; Xu, J.; Xue, C.; Wang, J.; Wang, Y. Dietary saponins of sea cucumber ameliorate obesity, hepatic steatosis, and glucose intolerance in high-fat diet-fed mice. J. Med. Food 2012, 15, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Q.; Xu, J.; Xue, Y.; Li, Z.-J.; Wang, J.-F.; Wang, J.-H.; Xue, C.-H.; Wang, Y.-M. Effects of bioactive components of sea cucumber on the serum, liver lipid profile and lipid absorption. Biosci. Biotechnol. Biochem. 2012, 76, 2214–2218. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, Y.-M.; Feng, T.-Y.; Zhang, B.; Sugawara, T.; Xue, C.-H. Isolation and anti-fatty liver activity of a novel cerebroside from the sea cucumber Acaudina molpadioides. Biosci. Biotechnol. Biochem. 2011, 75, 1466–1471. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, J.; Zhang, X.; Li, Z.; Wang, Y.; Xue, C. Inhibitory effect of fucosylated chondroitin sulfate from the sea cucumber Acaudina molpadioides on adipogenesis is dependent on Wnt/b-catenin pathway. J. Biosci. Bioeng. 2015, 119, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Zhang, Y.; Ye, X.; Hu, Y.; Ding, T.; Chen, S. Sulfation pattern of fucose branches affects the anti-hyperlipidemic activities of fucosylated chondroitin sulfate. Carbohydr. Polym. 2016, 147, 1–7. [Google Scholar] [CrossRef] [PubMed]
- El Barky, A.R.; Hussein, S.A.; Alm-Eldeen, A.A.; Hafez, Y.A.; Mohamed, T.M. Anti-diabetic activity of Holothuria thomasi saponin. Biomed. Pharmacother. 2016, 84, 1472–1487. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Wen, M.; Han, X.; Yanagita, T.; Xue, Y.; Wang, J.; Xue, C.; Wang, Y. Effect and potential mechanism of action of sea cucumber saponins on postprandial blood glucose in mice. Biosci. Biotechnol. Biochem. 2016, 80, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Chang, Y.; He, M.; Wang, J.; Wang, Y.; Xue, C. Fucosylated chondroitin sulfate from sea cucumber improves insulin sensitivity via activation of PI3K/PKB pathway. J. Food Sci. 2014, 79, H1424–H1429. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Chang, Y.; Wang, J.; Xue, C.; Li, Z.; Wang, Y. Fucosylated chondroitin sulfate from sea cucumber in combination with rosiglytazone improved glucose metabolism in the liver of the insulin-resistent mice. Biosci. Biotechnol. Biochem. 2013, 77, 2263–2268. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Um, B.H.; Kim, S.M. two unsaturated fatty acids with potent α-glucosidase inhibitory activity purified from the body wall of sea cucumber (Stichopus japonicus). J. Food Sci. 2011, 76, H208–H214. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Xu, L.; Shi, D.; Wang, J.; Wang, Y.; Lou, Q.; Xue, C. Eicosapentaenoic acid-enriched phosphatidylcholine isolated from Cucumaria frondosa exhibits anti-hyperglycemic effects via activating phosphoinositide 3-kinase/protein kinase B signal pathway. J. Biosci. Bioeng. 2014, 117, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.W.; Samyn, Y.; Conand, C. Commercially important sea cucumbers of the world. In FAO Species Catalogue for Fishery Purposes; FAO: Rome, Italy, 2012; Volume 6, p. 150. [Google Scholar]
- Bordbar, S.; Anwar, F.; Saari, N. High-value components and bioactives from sea cucumbers for functional Foods—A review. Mar. Drugs 2011, 9, 1761–1805. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Hu, C.; Fan, S. Chemical composition and nutritional quality of sea cucumbers. J. Sci. Food Agric. 2010, 90, 2469–2474. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F. Nutraceuticals and functional foods: Whole versus processed foods. Trends Food Sci. Technol. 2009, 20, 376–387. [Google Scholar] [CrossRef]
- Pavão, M.S.G. Glycosaminoglycans analogs from marine invertebrates: Structure, biological effects, and potential as new therapeutics. Front. Cell. Infect. Microbiol. 2014, 4, 123. [Google Scholar] [CrossRef] [PubMed]
- Blagodatski, A.; Cherepanov, V.; Koval, A.; Kharlamenko, V.I.; Khotimchenko, Y.S.; Katanaev, V.L. High-throughput targeted screening in triple-negative breast cancer cells identifies Wnt-inhibiting activities in Pacific brittle stars. Sci. Rep. 2017, 7, 11964. [Google Scholar] [CrossRef] [PubMed]
- Olivon, F.; Allard, P.-M.; Koval, A.; Righi, D.; Genta-Jouve, G.; Neyts, J.; Apel, C.; Pannecouque, C.; Nothias, L.-F.; Cachet, X.; et al. Bioactive natural products prioritization using massive multi-informational molecular networks. ACS Chem. Biol. 2017, 12, 2644–2651. [Google Scholar] [CrossRef] [PubMed]
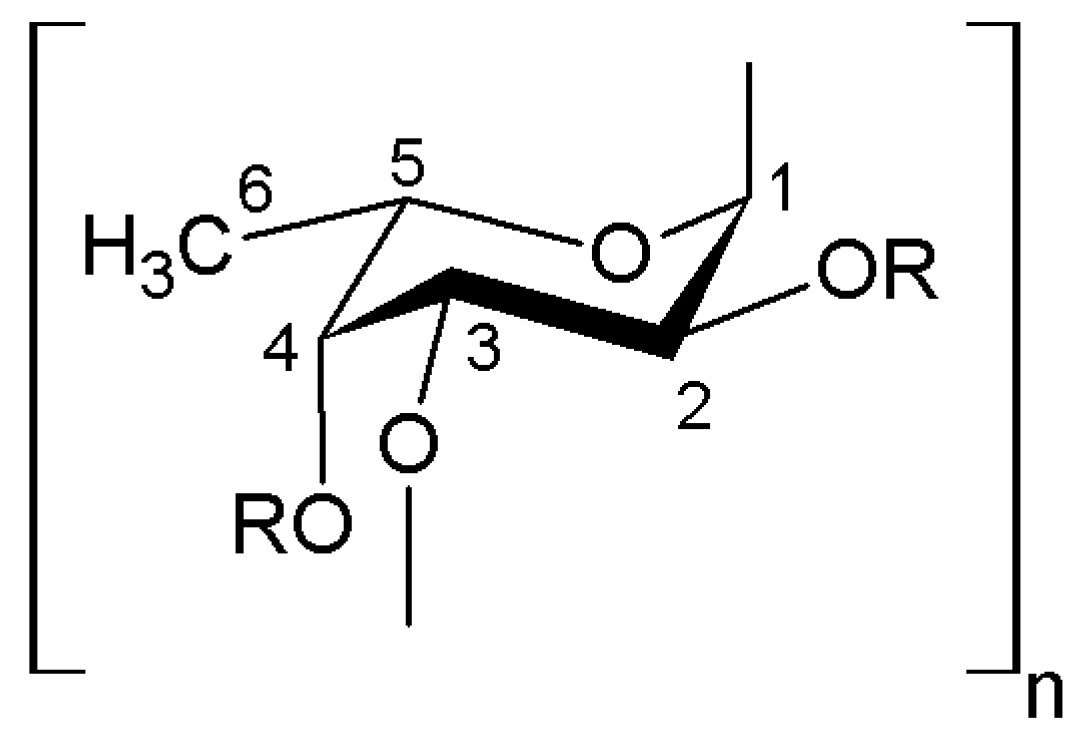
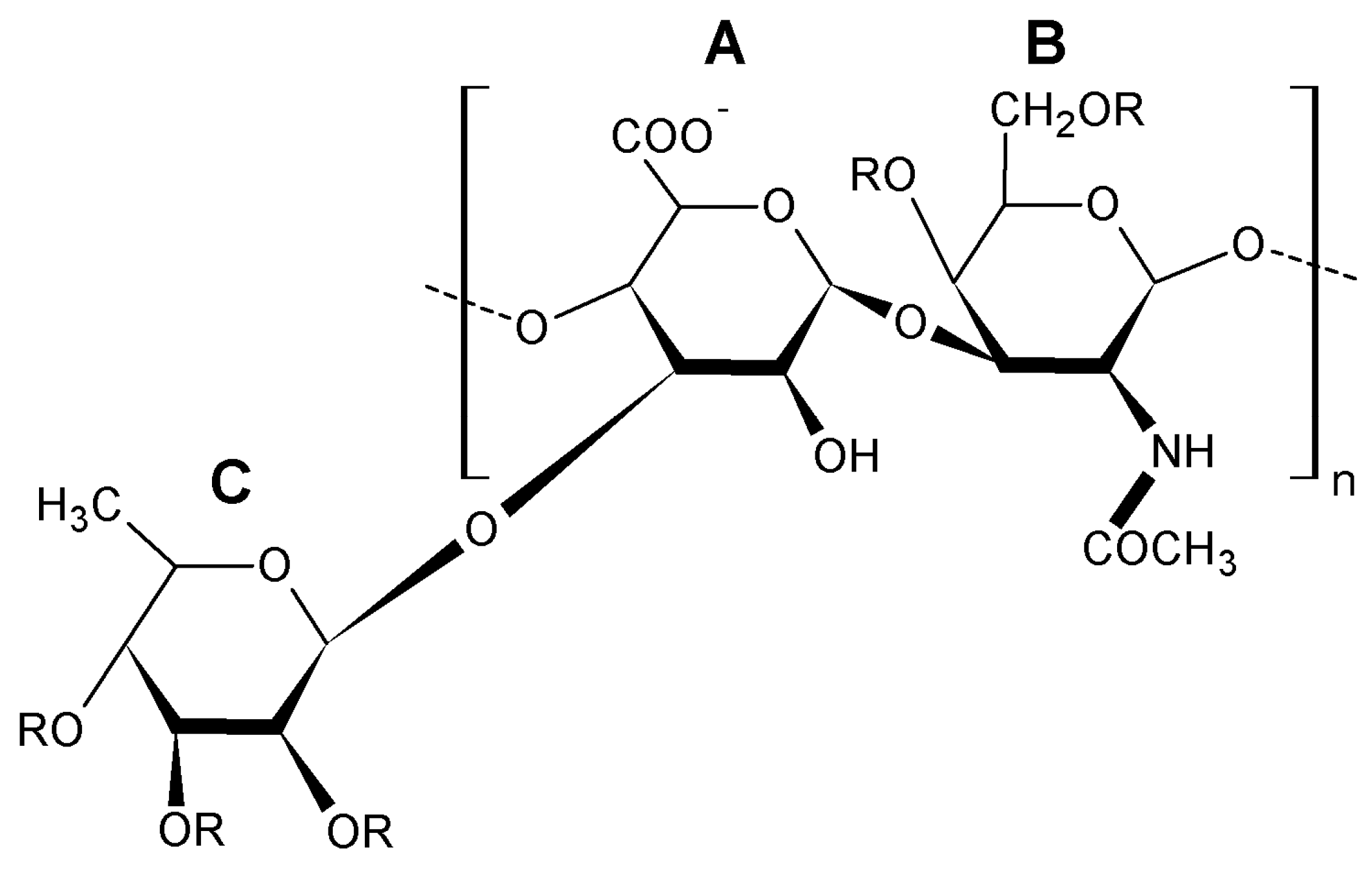
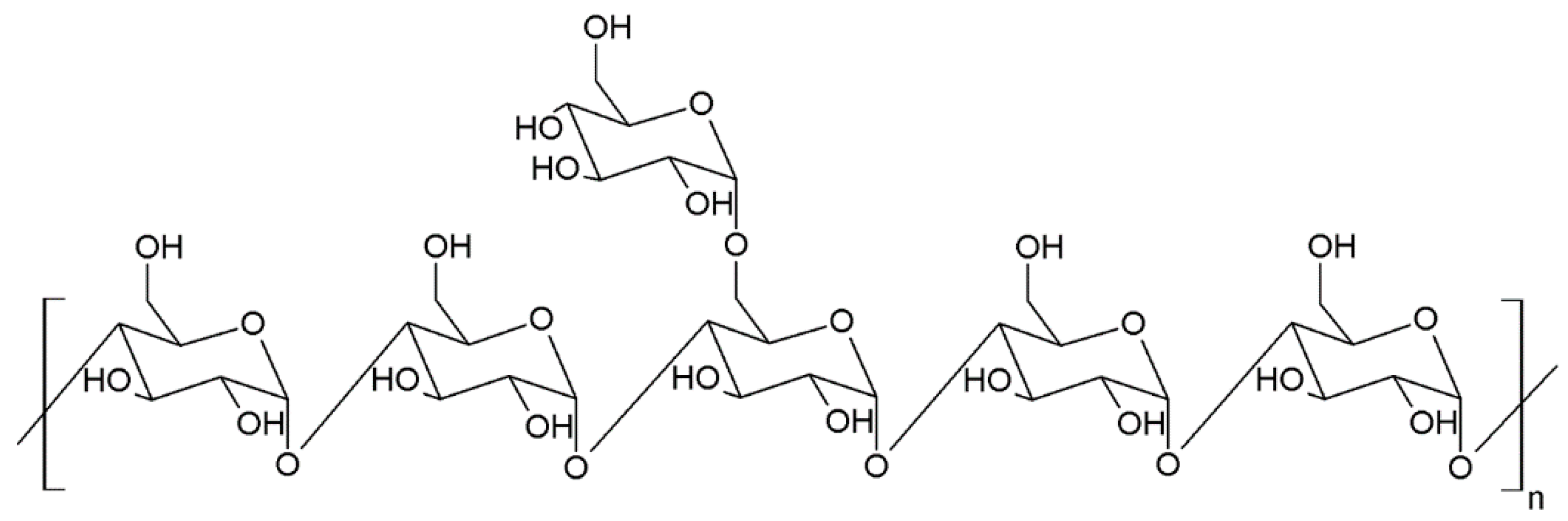
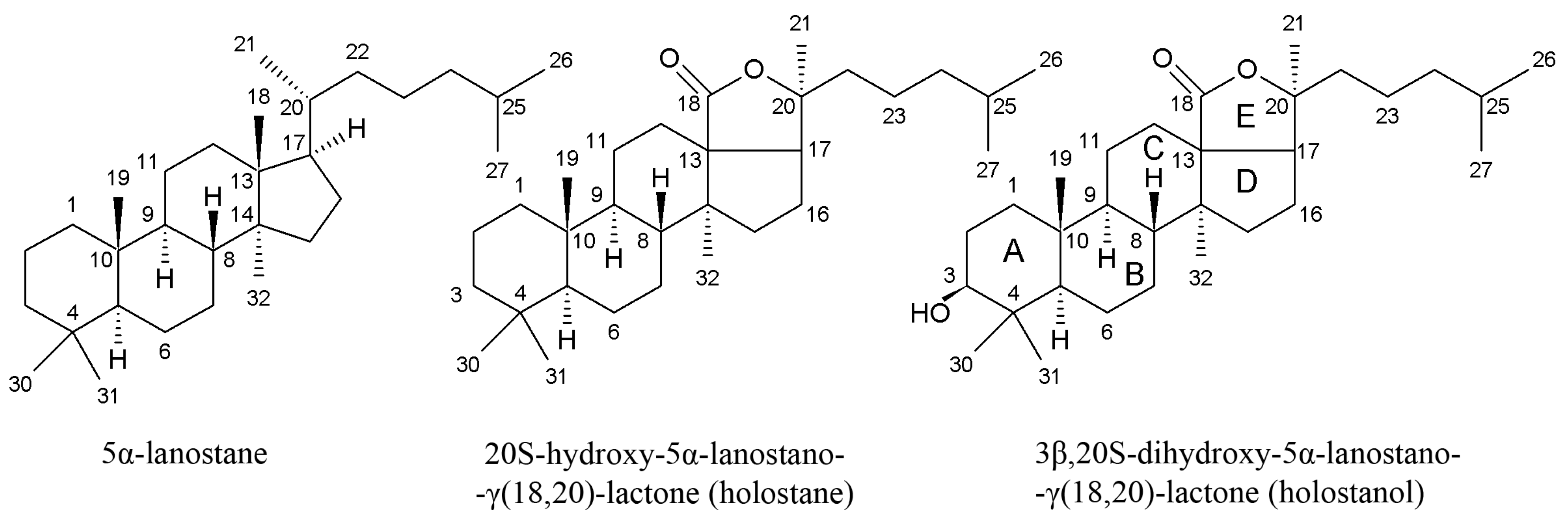
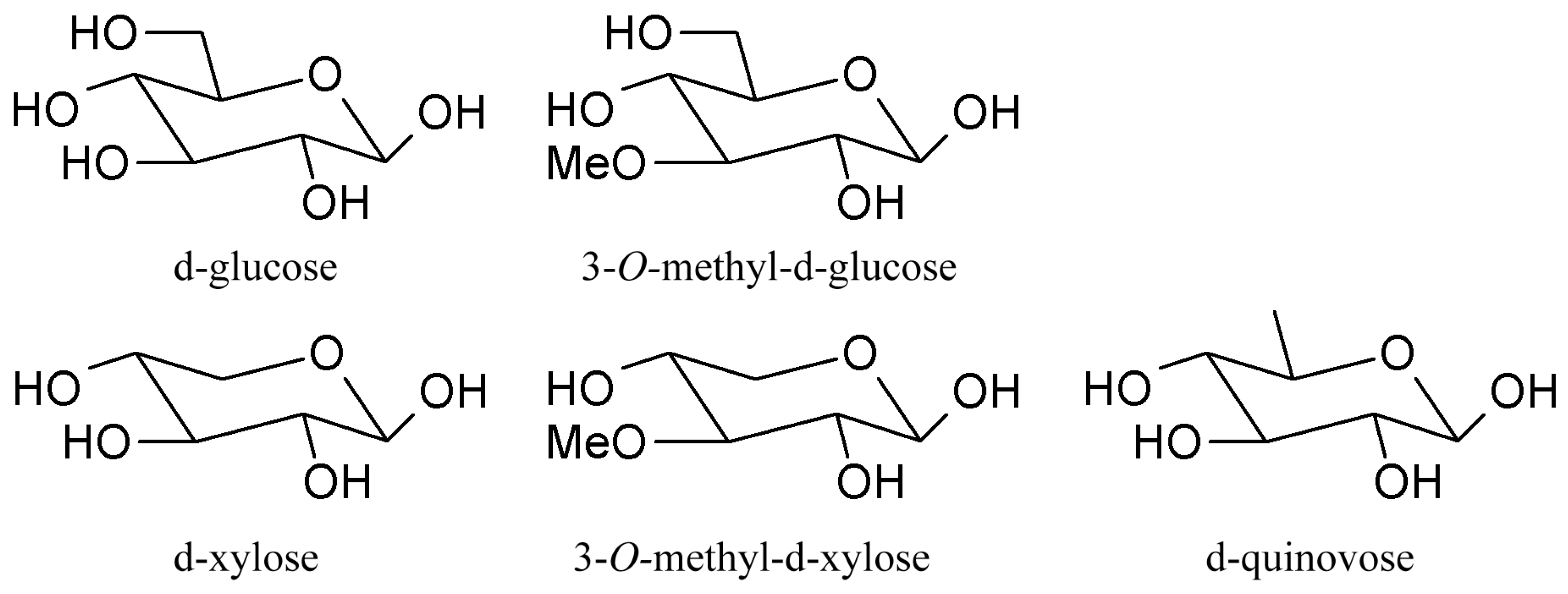
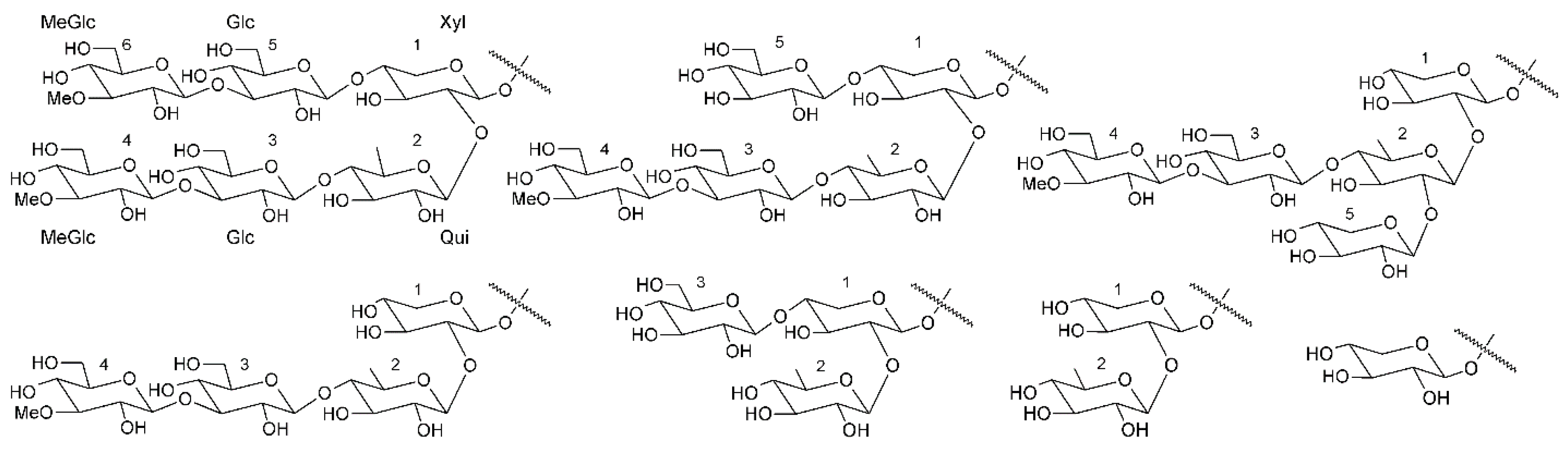
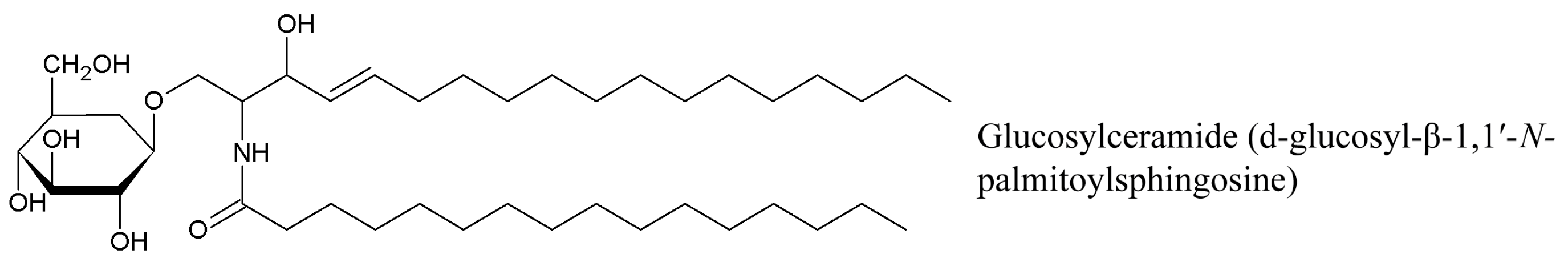
| Species * | Bioactive Compounds |
|---|---|
| Order Dendrochirotida Family Cucumariidae | |
| Cercodemas anceps (Colochirus anceps) | Triterpene glycoside: colochiroside A |
| Cucumaria frondosa | Polysaccharides: fucosylated chondroitin sulfate; Triterpene glycosides: frondosides A, A1, A6, 24-dehydrofrondoside A6; Cerebrosides; Phospholipids |
| Cucumaria frondosa japonica (Cucumaria japonica) | Triterpene glycosides: cucumariosides A2-2, A4-2 |
| Colochirus quadrangularis (Pentacta quadrangularis) | Triterpene glycosides: pentactasides I, II, III, philinopsides A, B, E, |
| Pseudocolochirus violaceus | Triterpene glycosides: violaceusides A, B |
| Mensamaria intercedens | Triterpene glycoside: intercedensides A, B, C |
| Family Phyllophoridae | |
| Massinium magnum | Polysaccharides: fucosylated chondroitin sulfate |
| Family Psolidae | |
| Psolus patagonicus | Triterpene glycoside: patagonicoside A |
| Family Sclerodactylidae | |
| Eupentacta fraudatrix | Polysaccharides: fucosylated chondroitin sulfate; Triterpene glycosides: cucumariosides A5, B2, I2 |
| Order Holothuriida Family Holothuriidae | |
| Actinopyga agassizi | Holothurin A |
| Actinopyga echinites | Ethanol extract |
| Actinopyga lecanora | Peptide hydrolysates, Triterpenes Holothurin A, B, methanol extract |
| Actinopyga miliaris | Ethanol extract |
| Bohadschia argus | Triterpene glycosides: argusides A, B, C, D, E; Galactocerebroside |
| Bohadschia marmorata | Triterpene glycosides: marmoratoside A, impatienside A, 17α-hydroxy impatienside A, bivittoside D |
| Holothuria arenicola | Phenolic compounds |
| Holothuria arguinensis | Polyunsaturated fatty acids: arachidonic, eicosapentaenoic, docosahexaenoic acids; Amino acids |
| Holothuria atra | Phospholipids: lysoplatelet activating factor, lysophosphatidylcholine, ethanol extract |
| Holothuria edulis | Polysaccharides: sulfated fucan, fucosylated chondroitin sulfate |
| Holothuria forskali | Polysaccharides: fucosylated chondroitin sulfate |
| Holothuria fuscocinerea | Triterpene glycosides: fuscocinerosides A, B, C |
| Holothuria grisea (Ludwigothuria grisea) | Triterpene glycosides: 17-dehydroxyholothurinoside A, griseaside A |
| Holothuria hilla | Triterpene glycoside: hillaside C |
| Holothuria impatiens | Triterpene glycosides: impatienside A, 17α-hydroxyimpatienside A |
| Holothuria leucospilota | Polysaccharides: glycosaminoglycan; Saponin complex |
| Holothuria mexicana | Polysaccharides: fucosylated chondroitin sulfate |
| Holothuria moebii | Triterpene glycosides: moebioside A, holothurin A, holothurin B, 24-dehydroechinoside B |
| Holothuria nobilis | Polysaccharides: sulfated fucan, fucosylated chondroitin sulfate |
| Holothuria scabra | Triterpene glycosides: scabrasides B, D, echinoside A, 24-dehydroechinoside A, HS-1, holothurins A, A1, A3, A4, B, B4, fuscocineroside C |
| Holothuria thomasi | Saponin complex |
| Pearsonothuria graeffei | Triterpene glycosides: echinoside A, Ds-echinoside A, 24-dehydroechinoside A, holothurin A; Polysaccharides: fucosylated chondroitin sulfate; Cerebrosides |
| Order Molpadiida Family Caudinidae | |
| Acaudina molpadioides | Polysaccharide: fucosylated chondroitin sulfate; Cerebrosides |
| Order Synallactida Family Stichopodidae | |
| Apostichopus japonicus (Stichopus japonicus) | Polysaccharides: sulfated fucan, fucosylated chondroitin sulfate; Triterpene glycosides: holotoxins A, B, D, E, F, G, H; Cerebrosides; Fatty acids: 7(Z)-octadecenoic acid, 7(Z),10(Z)-octadecadienoic acid; Active proteins |
| Isostichopus badionotus | Polysaccharides: fucosylated chondroitin sulfate, sulfated fucan; Low molecular peptides |
| Stichopus horrens (Stichopus variegates) | Triterpene diglycosides: stichorrenosides A, B, C, D, stichoposides A, B, 3β-O-[β-d-xylopyranosyl-(1→2)-β-d-xylopyranosyl]-23S-acetoxyholost-7-ene, 3β-O-[β-d-xylopyranosyl-(1→2)-β-d-xylopyranosyl]-23S-hydroxyholost-7-en; Enzyme hydrolysates; Cerebrosides |
| Stichopus vastus | Collagen hydrolysates |
| Thelenota ananas | Polysaccharides: fucosylated chondroitin sulfate |
| Thelenota anax | Triterpene glycosides: stichoposide C, cucumarioside A2-2, cucumarioside A4-2 |
| Sample | Source | Chemical Composition (Molar Ratios) | Mn (kDa) | MW (kDa) | MW/Mn | Reference | |||
|---|---|---|---|---|---|---|---|---|---|
| GlcA | GalNAc | Fuc | Glc | ||||||
| Sulfated fucan | Holothuria edulis | <0.01 | <0.01 | 1.00 | <0.01 | 517.90 | 615.50 | 1.19 | [17] |
| Sulfated fucan | Apostichopus japonicas | <0.01 | <0.01 | 1.00 | <0.01 | 225.90 | 419.90 | 1.86 | |
| Sulfated fucan | Holothuria nobilis | <0.01 | <0.01 | 1.00 | <0.01 | 289.40 | 475.80 | 1.64 | |
| Fucosylated chondroitin sulfate | Holothuria edulis | 1 | 1.28 | 0.82 | <0.01 | 41.34 | 51.09 | 1.24 | |
| Fucosylated chondroitin sulfate | Apostichopus japonicas | 1 | 1.05 | 1.03 | <0.01 | 46.76 | 56.82 | 1.22 | |
| Fucosylated chondroitin sulfate | Holothuria nobilis | 1 | 0.96 | 0.82 | <0.01 | 42.46 | 55.32 | 1.30 | |
| Neutral glucan | Holothuria edulis | <0.01 | <0.01 | <0.01 | 1.00 | 197.90 | 253.30 | 1.28 | |
| Heparin (commercial drug) | Porcine intestine | ND | ND | ND | <0.01 | 20.23 | 26.26 | 1.30 | |
| Neutral glucan | Holothuria edulis | <0.01 | <0.01 | <0.01 | 1.00 | 197.90 | 253.30 | 1.28 | |
| Fucosylated chondroitin sulfate | Isostichopus badionotus | 1.43 | 1 | 1.71 | ND | 94.78 | 109 | 1.15 | [22] |
| Depolymerized fucosylated chondroitin sulfate | 1.35 | 1 | 1.70 | ND | 4.60 | 7.4 | 1.61 | ||
| Depolymerized fucosylated chondroitin sulfate | 1.32 | 1 | 1.72 | ND | 2.89 | 5.2 | 1.80 | ||
| Depolymerized fucosylated chondroitin sulfate | 1.30 | 1 | 1.71 | ND | 2.38 | 4.3 | 1.80 | ||
| Low molecular weight heparin (commercial drug) | Porcine intestine | 5.0 | 6.4 | 1.28 | |||||
| Sample | Source | MW (kDa) | APTT (IU/mg) | TT (IU/mg) | TP (IU/mg) | Reference |
|---|---|---|---|---|---|---|
| Sulfated fucan | Holothuria edulis | 615.50 | 13 | <1 | <1 | [17] |
| Sulfated fucan | Apostichopus japonicas | 419.90 | 19 | <1 | <1 | |
| Sulfated fucan | Holothuria nobilis | 475.80 | 9 | <1 | <1 | |
| Fucosylated chondroitin sulfate | Holothuria edulis | 51.09 | 89 | 6 | <1 | |
| Fucosylated chondroitin sulfate | Apostichopus japonicas | 56.82 | 116 | 7 | <1 | |
| Fucosylated chondroitin sulfate | Holothuria nobilis | 55.32 | 59 | 4 | <1 | |
| Neutral glucan | Holothuria edulis | 253.30 | <1 | <1 | <1 | |
| Heparin | Porcine intestine (commercial drug) | 26.26 | 212 | 212 | 212 | |
| Sulfated fucan | Isostichopus badionotus | 450 | 9 | 6 | [20] | |
| Fucosylated chondroitin sulfate | Isostichopus badionotus | Not specified | 183 | 157 | ||
| Heparin | Porcine intestine (commercial drug) | 150 | 150 | |||
| Fucosylated chondroitin sulfate | Isostichopus badionotus | 109 | 187 | 157 | [22] | |
| Depolymerized fucosylated chondroitin sulfate * | Isostichopus badionotus | 7.4 | 103.8 | 34.3 | ||
| Depolymerized fucosylated chondroitin sulfate * | Isostichopus badionotus | 5.2 | 60.5 | <1 | ||
| Depolymerized fucosylated chondroitin sulfate * | Isostichopus badionotus | 4.3 | 34.8 | <1 | ||
| Low molecular weight heparin | Porcine intestine (commercial drug) | 6.4 | 69 | 64 | ||
| Heparin | Porcine intestine (commercial drug) | 18.6 | 212 | 212 |
| Thrombogenesis in the Artificial Blood Vessel | ||||||
|---|---|---|---|---|---|---|
| Sample | Source | MW (kDa) | Length of Thrombus (cm) | Weight of Thrombus (mg) | Reference | |
| Normal blood | 2.16 ± 0.52 | 110.47 ± 9.49 | [20] | |||
| Sulfated fucan (0.5 mg/mL) | Isostichopus badionotus | 450 | 2.02 ± 0.24 | 40.58 ± 8.05 a | ||
| Sulfated fucan (1.0 mg/mL) | 2.05 ± 0.26 | 35.08 ± 5.10 a | ||||
| Fucosylated chondroitin sulfate (0.3 mg/mL) | Not specified | 1.80 ± 0.33 | 35.40 ± 7.15 a | |||
| Fucosylated chondroitin sulfate (0.5 mg/mL) | 1.73 ± 0.42 | 32.73 ± 5.35 a | ||||
| Heparin (Jiangsu Pharmacia, China) (0.3 mg/mL) | ND | ND | ||||
| Antithrombotic Potency Values | ||||||
| Factor Xa/Antithrombin * | Thrombin/Antithrombin * | Thrombin/HCII ** | ||||
| IU/1 mg of sample | [40] | |||||
| Native fucosylated chondroitin sulfate | Holothuria forskali | 120–140 | 0.40 | 0.56 | 120 | |
| Depolymerized fucosylated chondroitin sulfate | 6 | 0.46 | <limit of detection | 7.8 | ||
| Depolymerized fucosylated chondroitin sulfate | 2.5 | 0.181 | <limit of detection | 0.65 | ||
| Heparin | 203 | 196 | 206 | |||
| IC50 (μg/mL) | [18] | |||||
| Sulfated fucan | Holothuria edulis | 616 | >1000 | 78.7 ± 6.2 | 0.7 ± 0.02 | |
| Sulfated fucan | Holothuria grisea | 564 | >1500 | 66.5 ± 11 | 0.5 ± 0.03 | |
| Heparin | 0.03 ± 0.003 | 0.015 ± 0.001 | 0.2 ± 0.01 | |||
| IC50 (μg/mL) | [20] | |||||
| Sulfated fucan | Isostichopus badionotus | 450 | 0.53 | 0.32 | 2.55 | |
| Fucosylatedchondroitin sulfate | Not specified | 2.58 | 0.56 | 0.05 | ||
| Heparin | 0.005 | 0.01 | 0.98 | |||
| Compounds | Type of Tumor Cell Lines | Activity Result, IC50 | Reference |
|---|---|---|---|
| Arguside A | Human leukemia HL-60, human colon cancer HCT-116, human hepatocellular carcinoma BEL-7402, human stomach adenocarcinoma MKN45 | 0.14–4.42 μM | [61] |
| Arguside B | Human lung cancer A549, HCT-116, human hepatocellular carcinoma human breast cancer MCF-7 | 0.46–2.60 μg/mL | [62] |
| Arguside C | A549, HCT-116, HepG2, MCF-7 | 0.38–2.56 μg/mL | [62] |
| Arguside D | A549, HCT-116, HepG2, MCF-7 | 3.45–7.77 μg/mL | [63] |
| Arguside E | A549, HCT-116, HepG2, MCF-7 | 3.36–7.53 μg/mL | [63] |
| Bivittoside D | Colon carcinoma HT-29, HCT-116, human prostate cancer DU-145, MCF-7, human epidermoid carcinoma KB, A549, HepG2 | 0.37–2.46 μg/mL | [64] |
| Colochiroside A | Murine leukemia P-388, HL-60, A549, lung adenocarcinoma SPC-A4, stomach adenocarcinoma MKN28, gastric carcinoma SGC-7901 | 3.61 ± 0.55 μg/mL | [65] |
| Cucumarioside A2-2 | HL-60, human leukemia THP-1, human leukemia NB-4, human leukemia K562 | Effective concentration: ≥5 μM | [66] |
| Cucumarioside A4-2 | HL-60, THP-1, NB-4, human leukemia K562 | Effective concentration: ≥5 μM | [66] |
| Echinoside A | Human cervical cancer HeLa, HepG2, K562 | 1.25–1.61 μg/mL | [67] |
| HepG2 | 2.70 μmol/L | [68] | |
| Ds-echinoside A | HepG2, | 2.65 μmol/L | [69] |
| 24-Dehydroechinoside A | HepG2 | 3.14 μM | [70] |
| HeLa, HepG2, K562 | 1.95–6.15 μg/mL | [67] | |
| 24-Dehydroechinoside B | Rat glioma C6, human glioma U-87-MG, human glioma U251, human glioma SHG-44 | 1.99–6.10 μM | [71] |
| Frondoside A | Human pancreatic cancer AsPC-1 | Effective concentration: 4 μg/mL | [72] |
| Human lung cancer LNM35, A549, human lung carcinoma NCI-H460-Luc2, human melanoma MDA-MB-435, human mammary adenocarcinoma MCF-7, HepG2 | 1.7–2.5 μM | [73] | |
| A549 | 0.6 μM | [74] | |
| HeLa, HepG2, K562 | 3.30–4.14 μg/mL | [67] | |
| Frondoside A1 | HeLa, HepG2, K562 | 1.91–2.21 μg/mL | [67] |
| Fuscocineroside A | HL-60, BEL-7402 | 0.58–0.88 μg/mL | [75] |
| Fuscocineroside B | HL-60, BEL-7402 | 0.58–0.88 μg/mL | [75] |
| Fuscocineroside C | HL-60, BEL-7402 | 0.58–0.88 μg/mL | [75] |
| Griseaside A | A549, HL-60, BEL-7402, human acute lymphoblastic leukemia Molt-4 | 0.427–2.60 μM | [76] |
| Hillaside C | A549, MCF-7, human lung carcinoma IA9, human clear cell carcinoma CAKI-1, human prostate adenocarcinoma PC-3, KB, nasopharyngeal cancer KB-VIN, HCT-8 | 0.15–3.20 μg/mL | [77] |
| Holothurin A | HL-60, BEL-7402 | 0.58–0.88 μg/mL | [75] |
| HeLa, HepG2, K562 | 3.46–8.94 μg/mL | [67] | |
| C6, U87-MG, U251, SHG-44 | 0.99–4.03 μM | [71] | |
| Holothurin A1 | HepG2, | 3.40 μM | [70] |
| Holothurin A1 | HeLa, HepG2, K562 | 2.84–6.50 μg/mL | [67] |
| Holothurin A3 | KB, HepG2 | 0.32–0.87 μg/mL | [78] |
| Holothurin A4 | KB, HepG2 | 0.57–1,12 μg/mL | [78] |
| Holothurin B | HeLa, HepG2, K562 | 1.79–3.64 μg/mL | [67] |
| Holothurins B | C6, U87-MG, U251, SHG-44 | 1.39–8.64 μM | [71] |
| Holothurin B4 | HeLa, HepG2, K562 IC50 = 2.71–3.55 | 2.71–3.55 μg/mL | [67] |
| Dehydroxyholothurinoside A | A549, HL-60, BEL-7402, Molt-4 | 0.245–0.97 μM | [76] |
| Impatienside A | HT-29, HCT-116, DU-145, MCF-7, KB, A549, HepG2 | 0.353–2.72 μg/mL | [64] |
| Intercedensides A, B, C | A549, MCF-7, IA9, CAKI-1, U-87-MG, PC-3, KB, KB-VIN, human skin melanoma SK-MEL-2, HCT-8 | 0.60–4.00 μg/mL | [79] |
| Moebioside A | C6, U87-MG, U251, SHG-44 | 1.22–4.39 μM | [71,80] |
| Patagonicoside A | Human hepatocarcinoma Hep3B, breast cancer MDA-MB-231, A549 | 0~80 μM | [81] |
| Pervicoside C | HCT-116 and A549 | 18.7–28.6 μg/mL | [64] |
| Pentactasides I, II, III | P-388, A549, MCF-7, MKN28, HCT-116, U87-MG | 0.60–3.95 μM | [82] |
| Philinopside A | P-388, A549, MCF-7, MKN28, HCT-116, U87MG | 0.60–3.95 μM | [82] |
| BEL-7402, MCF-7, ovarian epitheloid carcinoma HO-8910, mouse Sarcoma-180 | 1.5–2.4 μM | [83] | |
| Philinopside B | P-388, A549, MCF-7, MKN28, HCT-116, U87MG | 0.60–3.95 μM | [82] |
| Philinopside E | MKN28, P-388, BEL-7402, HL-60, SPC-A4, A549, SGC7901, human ovarian carcinoma HO8901, human fetal lung fibroblasts W138, human epithelial carcinoma A431 | 0.75–3.50 μg/mL | [84] |
| BEL-7402, SPC-A4, ovarian epitheloid carcinoma HO-8910 | 2.4–4.1 μM | [85] | |
| Scabraside B | HeLa, HepG2, K562 | 4.44–11.85 μg/mL | [67] |
| Stichoposide C | HL-60, K562, NB-4, THP-1, colorectal cancer CT-26, HT-29, colon adenocercinoma SNU-C4 | Effective concentrations: 0.3–1.5 μmol/L | [86] |
| Scabraside D | HeLa, HepG2, K562 | 3.33–10.06 μg/mL | [67] |
| Stichorrenosides A, B, C, D | KB, HepG2, MCF-7, human prostate adenocarcinoma LNCaP cells, SK-Mel-2 | 1.31–3.53 μM | [9] |
| Violaceusides I, II, III | MKN45, HCT-116 | 0.068–0.352 μM | [87] |
| Compounds | Source | Type of Tumor Cell Lines | Pharmacological Effect, Anticancer Mechanism | Reference |
|---|---|---|---|---|
| Sphingoid bases | Stichopus horrens | Human colon cancer cells Caco-2, DLD-1 and WiDr | Reduction of cell viability, induction of apoptosis, increasing caspase-3 activity | [96] |
| Glucocerebrosides, sphingoid bases | Cucumaria frondosa | Caco-2 | Inhibition of cell proliferation | [99] |
| Glucocerebrosides | Acaudina molpadioides | Murine sarcoma cells S180 | Induction of apoptosis | [100] |
| Sphingoid bases | Species not specified | Human hepatoma cells Hep-G2 | Reduction of cell viability, induction of apoptosis, upregulation of death receptor-5, apoptosis inducer protein Bax, growth arrest and DNA-damage-inducible protein DNA-damage-inducible gene 45 and peroxisome proliferator-activated receptor-γ, downregulation of protein kinase p-AKT, increasing of caspase-3 and caspase-8 activities | [101] |
| Compounds | Source | Targets | Reference |
|---|---|---|---|
| Fucosylated chondroitin sulfate | Holothuria grisea | Plasmodium falciparum | [6,103] |
| Fucosylated chondroitin sulfate | Isostichopus badionotus | Plasmodium falciparum | [6] |
| Holothurin B | Actinopyga lecanora | Leishmania donovani | [104] |
| Holothurin B | Actinopyga lecanora | Candida albicans, Candida neoformans, Sporothrix schenckii, Trychophyton mentagrophytes, Aspergillus fumigatus | [105] |
| Marmoratoside A, bivittoside D, impatienside A, 17α-hydroxy impatienside | Bohadschia marmorata | Candida albicans, Candida neoformans, Sporothrix schenckii, Trichophyton mentagrophytes, Aspergillus fumigatus, Candida parapsilosis | [106,107] |
| Holotoxins B, A, A1 | Apostichopus japonicus | Candida species | [108] |
| Fucosylated chondroitin sulfate | Thelenota ananas | Human immunodeficiency virus (HIV) | [109] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khotimchenko, Y. Pharmacological Potential of Sea Cucumbers. Int. J. Mol. Sci. 2018, 19, 1342. https://doi.org/10.3390/ijms19051342
Khotimchenko Y. Pharmacological Potential of Sea Cucumbers. International Journal of Molecular Sciences. 2018; 19(5):1342. https://doi.org/10.3390/ijms19051342
Chicago/Turabian StyleKhotimchenko, Yuri. 2018. "Pharmacological Potential of Sea Cucumbers" International Journal of Molecular Sciences 19, no. 5: 1342. https://doi.org/10.3390/ijms19051342