Hydroquinone Exhibits In Vitro and In Vivo Anti-Cancer Activity in Cancer Cells and Mice
Abstract
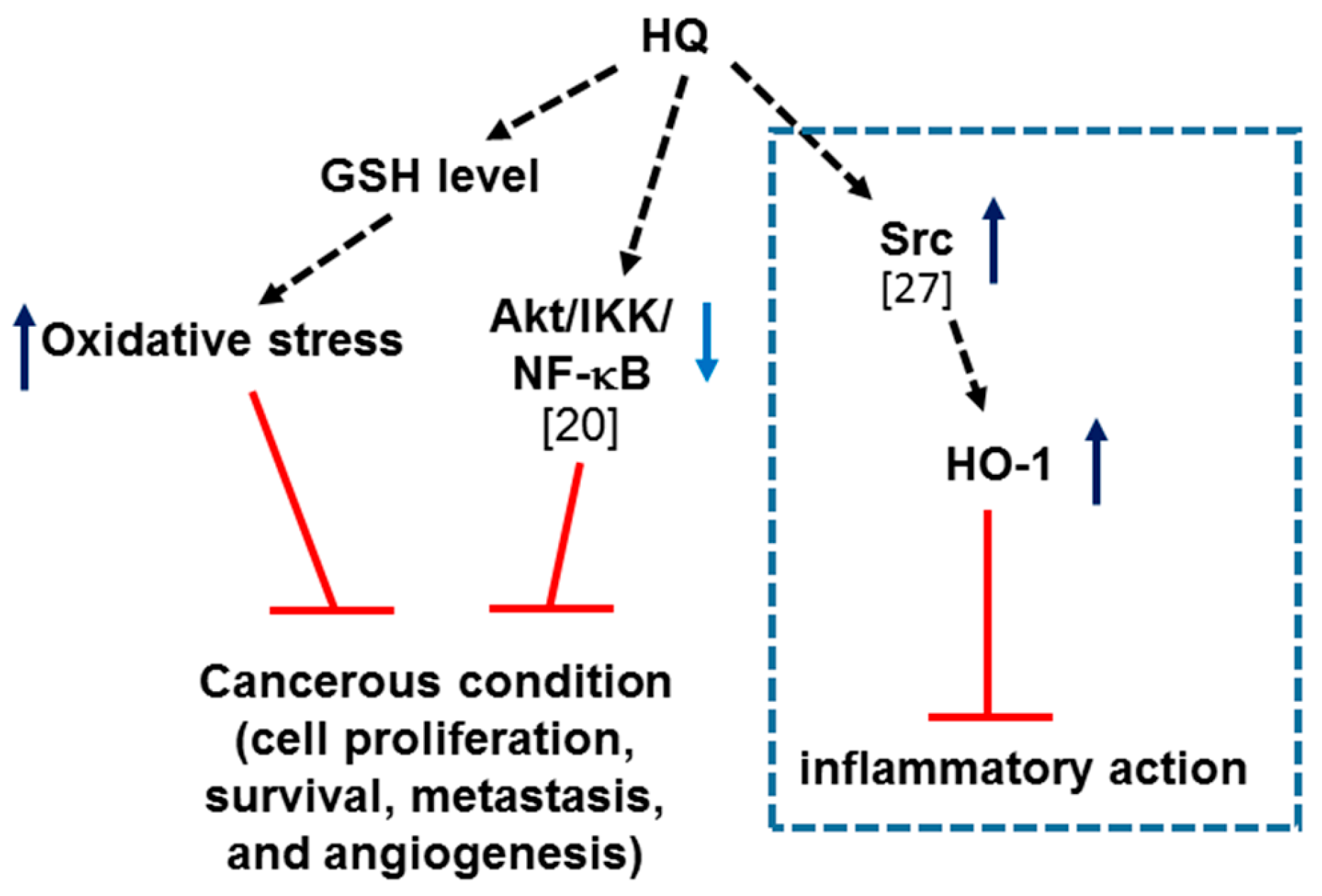
1. Introduction
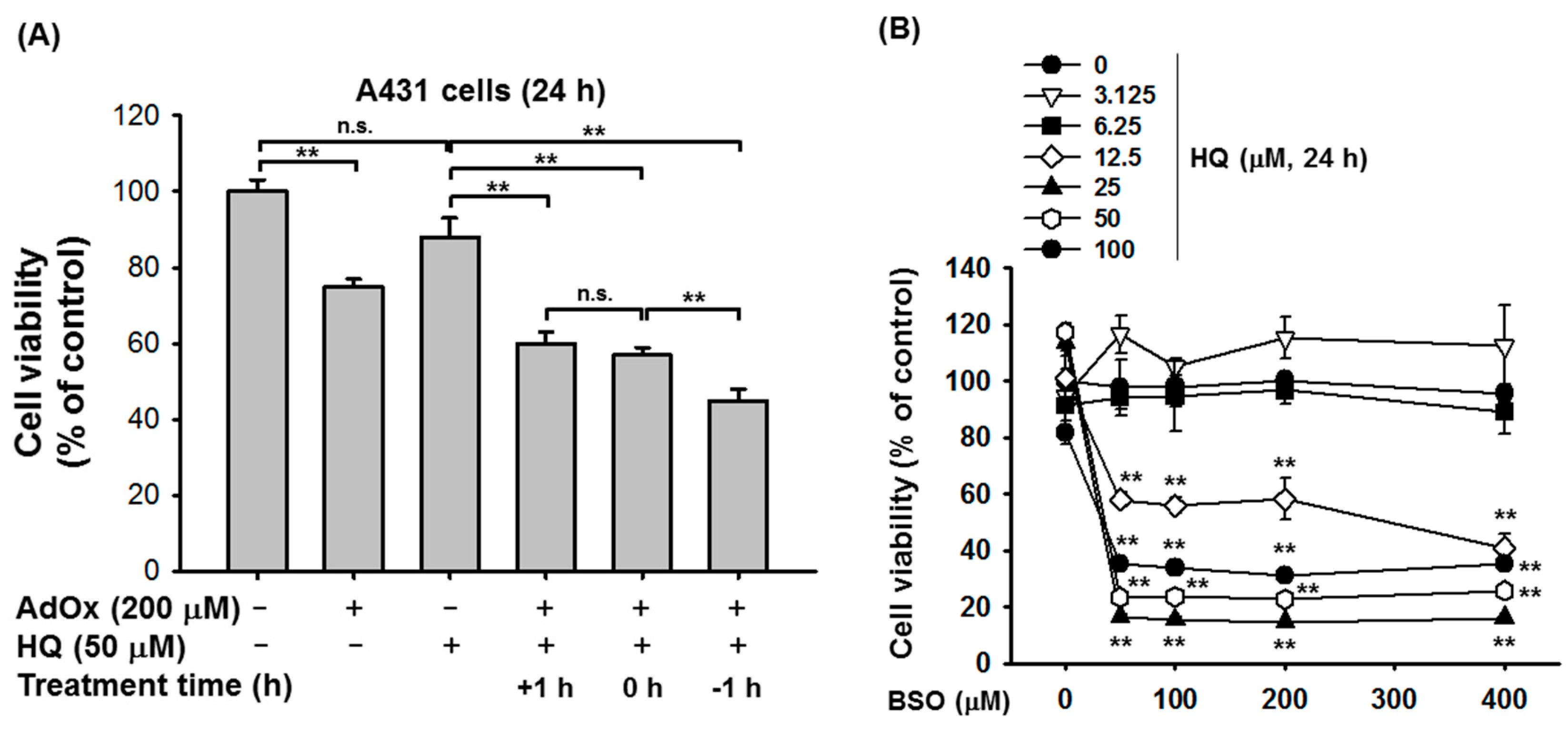
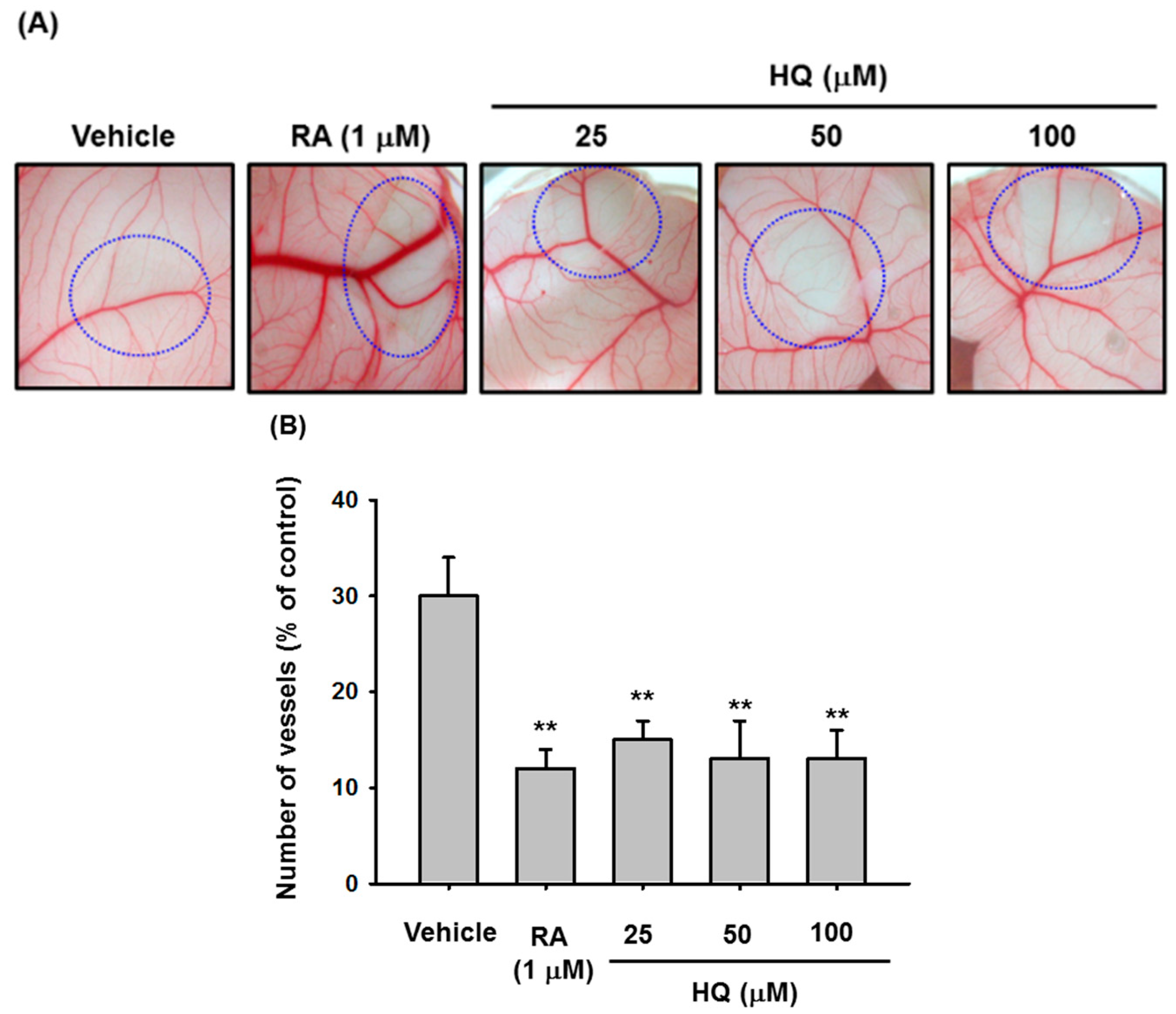
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Animal
3.3. Cell Culture
3.4. Cell Viability Assay
3.5. CAM Assay in Fertilized Chicken Eggs
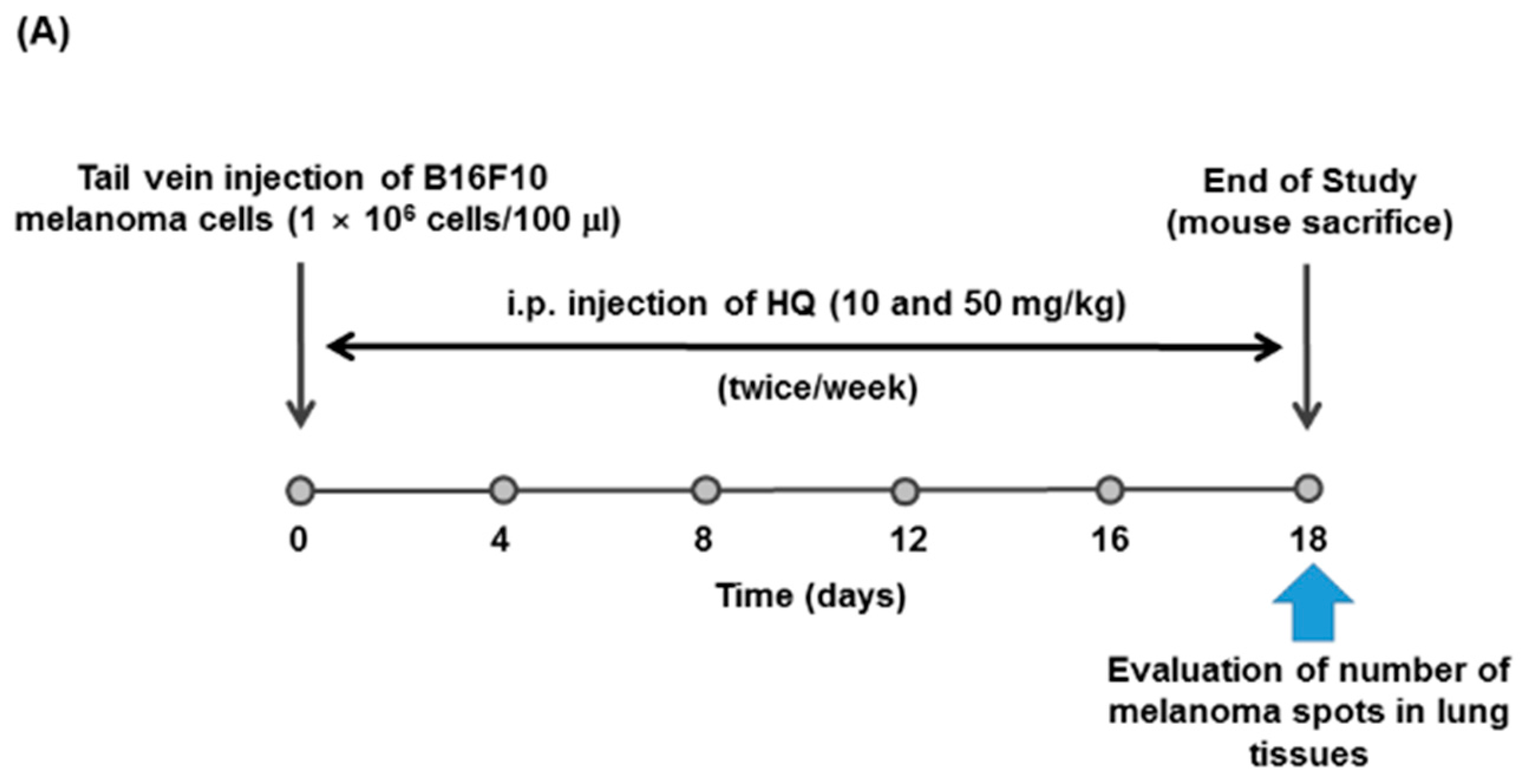
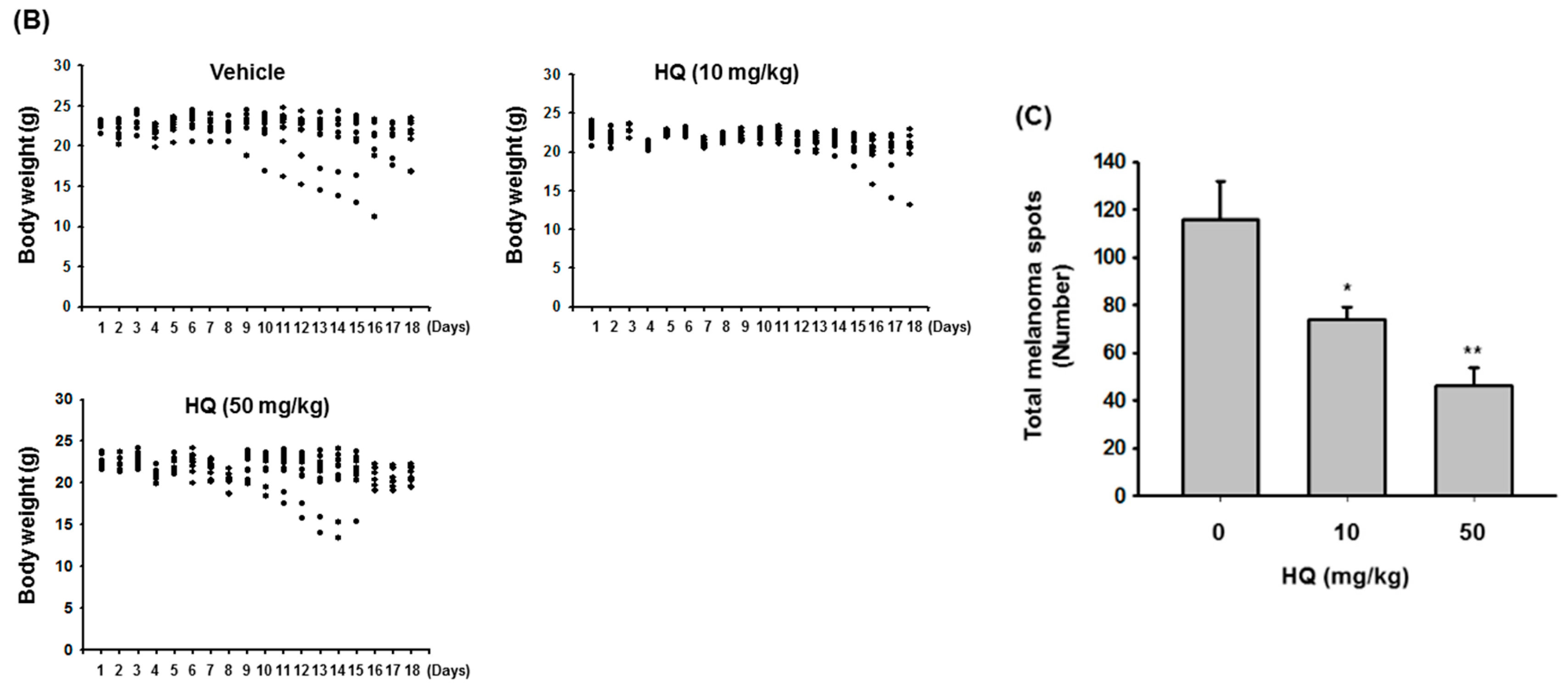
3.6. B16F10 Melanoma Lung Metastasis Model
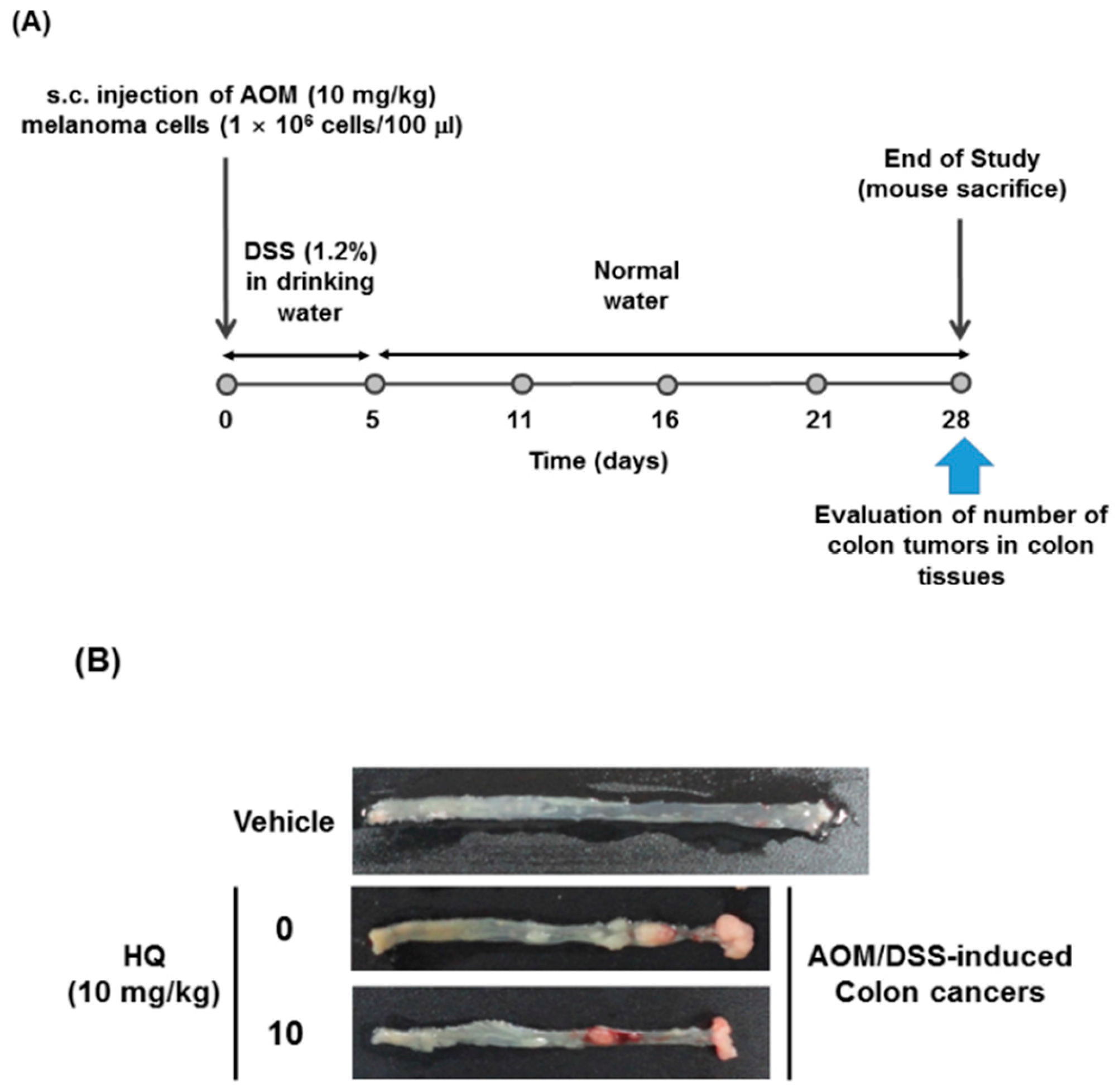
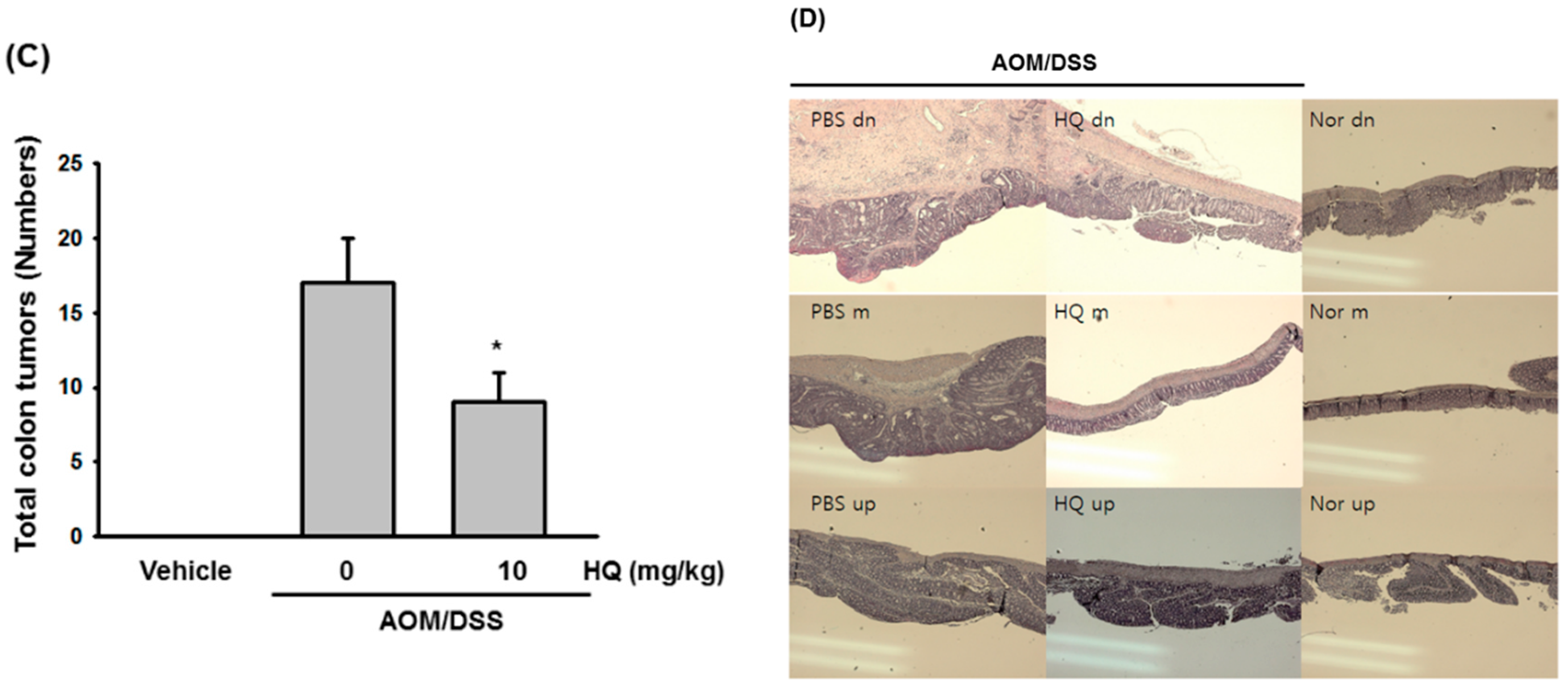
3.7. AOM/DSS-Induced Colon Cancer Model
3.8. Hematoxylin and Eosin Staining
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Singh, D.; Arora, R.; Kaur, P.; Singh, B.; Mannan, R.; Arora, S. Overexpression of hypoxia-inducible factor and metabolic pathways: Possible targets of cancer. Cell Biosci. 2017, 7, 62. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.S.; Yi, Y.S.; Son, Y.J.; Jeong, D.; Sung, N.Y.; Aravinthan, A.; Kim, J.H.; Cho, J.Y. Comparison of anticancer activities of Korean Red Ginseng-derived fractions. J. Ginseng. Res. 2017, 41, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Yi, Y.S.; Kim, D.; Kim, M.H.; Park, J.G.; Kim, E.; Lee, S.Y.; Yoon, K.; Kim, J.H.; Park, J.; et al. Nuclear factor kappa-B- and activator protein-1-mediated immunostimulatory activity of compound K in monocytes and macrophages. J. Ginseng. Res. 2017, 41, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Son, Y.J.; Aravinthan, A.; Kim, J.H.; Cho, J.Y. Korean Red Ginseng water extract arrests growth of xenografted lymphoma cells. J. Ginseng. Res. 2016, 40, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Kang, W.S.; Park, K.T.; Park, D.J.; Aravinthan, A.; Kim, J.H.; Cho, J.Y. Anticancer effect of joboksansam, Korean wild ginseng germinated from bird feces. J. Ginseng. Res. 2016, 40, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Son, Y.J.; Lee, T.H.; Baek, N.J.; Yoon, D.H.; Kim, T.W.; Aravinthan, A.; Hong, S.; Kim, J.H.; Sung, G.H.; et al. Anticancer Efficacy of Cordyceps militaris Ethanol Extract in a Xenografted Leukemia Model. Evid. Based Complement. Alternat. Med. 2017, 2017, 8474703. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, Y.; Kim, M.Y.; Cho, J.Y. 4-(Tert-butyl)-2,6-bis(1-phenylethyl)phenol induces pro-apoptotic activity. Korean J. Physiol. Pharmacol. 2016, 20, 253–259. [Google Scholar] [CrossRef] [PubMed][Green Version]
- DeCaprio, A.P. The toxicology of hydroquinone—Relevance to occupational and environmental exposure. Crit. Rev. Toxicol. 1999, 29, 283–330. [Google Scholar] [CrossRef] [PubMed]
- McDonald, T.A.; Holland, N.T.; Skibola, C.; Duramad, P.; Smith, M.T. Hypothesis: Phenol and hydroquinone derived mainly from diet and gastrointestinal flora activity are causal factors in leukemia. Leukemia 2001, 15, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Hadzi-Taskovic Sukalovic, V.; Kukavica, B.; Vuletic, M. Hydroquinone peroxidase activity of maize root mitochondria. Protoplasma 2007, 231, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.M.; Zhou, Q.; Lei, T.C.; Ding, S.F.; Xu, S.Z. Effects of hydroquinone and its glucoside derivatives on melanogenesis and antioxidation: Biosafety as skin whitening agents. J. Dermatol. Sci. 2009, 55, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, C.W.; Ahn, J.H.; Choi, S.Y.; Shin, M.C.; Cho, J.H.; Lee, T.K.; Kim, I.H.; Lee, J.C.; Kim, Y.H.; et al. Neuroprotection and reduced gliosis by pre- and post-treatments of hydroquinone in a gerbil model of transient cerebral ischemia. Chem. Biol. Interact. 2017, 118, e87. [Google Scholar] [CrossRef] [PubMed]
- Ha Park, J.; Yoo, K.Y.; Hye Kim, I.; Cho, J.H.; Lee, J.C.; Hyeon Ahn, J.; Jin Tae, H.; Chun Yan, B.; Won Kim, D.; Kyu Park, O.; et al. Hydroquinone Strongly Alleviates Focal Ischemic Brain Injury via Blockage of Blood-Brain Barrier Disruption in Rats. Toxicol. Sci. 2016, 154, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Geiselhart, L.; Mittler, J.N.; Mudzinski, S.P.; Lawrence, D.A.; Freed, B.M. Inhibition of human T lymphoblast proliferation by hydroquinone. Toxicol. Appl. Pharmacol. 1996, 139, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.M.; Cho, Y.C.; Cho, W.J.; Kim, T.S.; Kang, B.Y. Hydroquinone, a major component in cigarette smoke, reduces IFN-gamma production in antigen-primed lymphocytes. Arch. Pharm. Res. 2008, 31, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Chung, S.W.; Kang, B.Y.; Kim, K.M.; Kim, T.S. Hydroquinone, a reactive metabolite of benzene, enhances interleukin-4 production in CD4+ T cells and increases immunoglobulin E levels in antigen-primed mice. Immunology 2002, 106, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.Y. Suppressive effect of hydroquinone, a benzene metabolite, on in vitro inflammatory responses mediated by macrophages, monocytes, and lymphocytes. Mediat. Inflamm. 2008, 2008, 298010. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.R.; Cho, J.Y.; Lee, J.Y.; Choi, J.S.; Chung, H.Y. Hydroquinone modulates reactivity of peroxynitrite and nitric oxide production. J. Pharm. Pharmacol. 2005, 57, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Pyatt, D.W.; Yang, Y.; Stillman, W.S.; Cano, L.L.; Irons, R.D. Hydroquinone inhibits PMA-induced activation of NFkappaB in primary human CD19+ B lymphocytes. Cell Biol. Toxicol. 2000, 16, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Lee, Y.G.; Lee, J.; Yang, K.J.; Kim, A.R.; Kim, J.Y.; Won, M.H.; Park, J.; Yoo, B.C.; Kim, S.; et al. Akt Cys-310-targeted inhibition by hydroxylated benzene derivatives is tightly linked to their immunosuppressive effects. J. Biol. Chem. 2010, 285, 9932–9948. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Kinneer, K.; Ye, J.; Chen, B.J. Inhibition of nuclear factor kappaB by phenolic antioxidants: Interplay between antioxidant signaling and inflammatory cytokine expression. Mol. Pharmacol. 2003, 64, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Kim, J.Y.; Lee, Y.G.; Shin, W.C.; Chun, T.; Rhee, M.H.; Cho, J.Y. Hydroquinone, a reactive metabolite of benzene, reduces macrophage-mediated immune responses. Mol. Cells 2007, 23, 198–206. [Google Scholar] [PubMed]
- Sumalatha, K.; Gowda, M.; Meenakshisundaram, S. ROS-mediated induction of apoptosis by benzoquinone embelin in human colon adenocarcinoma cells HT-29. J. Complement. Integr. Med. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Zappavigna, S.; Scuotto, M.; Cossu, A.M.; Ingrosso, D.; De Rosa, M.; Schiraldi, C.; Filosa, R.; Caraglia, M. The 1,4 benzoquinone-featured 5-lipoxygenase inhibitor RF-Id induces apoptotic death through downregulation of IAPs in human glioblastoma cells. J. Exp. Clin. Cancer Res. 2016, 35, 167. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, Y.G.; Yoo, S.; Oh, J.; Jeong, D.; Song, W.K.; Yoo, B.C.; Rhee, M.H.; Park, J.; Cha, S.H.; et al. Involvement of Src and the actin cytoskeleton in the antitumorigenic action of adenosine dialdehyde. Biochem. Pharmacol. 2013, 85, 1042–1056. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, S.C.; Yi, Y.S.; Yang, W.S.; Yang, Y.; Kim, H.G.; Lee, J.Y.; Kim, K.H.; Yoo, B.C.; Hong, S.; et al. Adenosine dialdehyde suppresses MMP-9-mediated invasion of cancer cells by blocking the Ras/Raf-1/ERK/AP-1 signaling pathway. Biochem. Pharmacol. 2013, 86, 1285–1300. [Google Scholar] [CrossRef] [PubMed]
- Byeon, S.E.; Yu, T.; Yang, Y.; Lee, Y.G.; Kim, J.H.; Oh, J.; Jeong, H.Y.; Hong, S.; Yoo, B.C.; Cho, W.J.; et al. Hydroquinone regulates hemeoxygenase-1 expression via modulation of Src kinase activity through thiolation of cysteine residues. Free Radic. Biol. Med. 2013, 57, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Defty, C.L.; Marsden, J.R. Melphalan in regional chemotherapy for locally recurrent metastatic melanoma. Curr. Top. Med. Chem. 2012, 12, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Bauer, P.I.; Mendeleyeva, J.; Kirsten, E.; Comstock, J.A.; Hakam, A.; Buki, K.G.; Kun, E. Anti-cancer action of 4-iodo-3-nitrobenzamide in combination with buthionine sulfoximine: Inactivation of poly(ADP-ribose) polymerase and tumor glycolysis and the appearance of a poly(ADP-ribose) polymerase protease. Biochem. Pharmacol. 2002, 63, 455–462. [Google Scholar] [CrossRef]
- Tsutsui, K.; Komuro, C.; Ono, K.; Nishidai, T.; Shibamoto, Y.; Takahashi, M.; Abe, M. Chemosensitization by buthionine sulfoximine in vivo. Int. J. Radiat. Oncol. Biol. Phys. 1986, 12, 1183–1186. [Google Scholar] [CrossRef]
- McGregor, D. Hydroquinone: An evaluation of the human risks from its carcinogenic and mutagenic properties. Crit. Rev. Toxicol. 2007, 37, 887–914. [Google Scholar] [CrossRef] [PubMed]
- Marhall, A.; Kazi, J.U.; Ronnstrand, L. The Src family kinase LCK cooperates with oncogenic FLT3/ITD in cellular transformation. Sci. Rep. 2017, 7, 13734. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, S.; Verzoni, E.; Prinzi, N.; Mennitto, A.; Femia, D.; Grassi, P.; Concas, L.; Vernieri, C.; Lo Russo, G.; Procopio, G. Everolimus treatment for neuroendocrine tumors: Latest results and clinical potential. Ther. Adv. Med. Oncol. 2017, 9, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Augereau, P.; Patsouris, A.; Bourbouloux, E.; Gourmelon, C.; Abadie Lacourtoisie, S.; Berton Rigaud, D.; Soulie, P.; Frenel, J.S.; Campone, M. Hormonoresistance in advanced breast cancer: A new revolution in endocrine therapy. Ther. Adv. Med. Oncol. 2017, 9, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.G.; Lee, J.; Byeon, S.E.; Yoo, D.S.; Kim, M.H.; Lee, S.Y.; Cho, J.Y. Functional role of Akt in macrophage-mediated innate immunity. Front. Biosci. 2011, 16, 517–530. [Google Scholar] [CrossRef]
- Hossen, M.J.; Hong, Y.D.; Baek, K.S.; Yoo, S.; Hong, Y.H.; Kim, J.H.; Lee, J.O.; Kim, D.; Park, J.; Cho, J.Y. In vitro antioxidative and anti-inflammatory effects of the compound K-rich fraction BIOGF1K, prepared from Panax ginseng. J. Ginseng. Res. 2017, 41, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.F.; Mirando, A.C.; Wilkinson, B.; Francklyn, C.S.; Lounsbury, K.M. Secreted Threonyl-tRNA synthetase stimulates endothelial cell migration and angiogenesis. Sci. Rep. 2013, 3, 1317. [Google Scholar] [CrossRef] [PubMed]
- Gerlier, D.; Thomasset, N. Use of MTT colorimetric assay to measure cell activation. J. Immunol. Methods 1986, 94, 57–63. [Google Scholar] [CrossRef]
- Ambasta, R.K.; Jha, S.K.; Kumar, D.; Sharma, R.; Jha, N.K.; Kumar, P. Comparative study of anti-angiogenic activities of luteolin, lectin and lupeol biomolecules. J. Transl. Med. 2015, 13, 307. [Google Scholar] [CrossRef] [PubMed]
- Overwijk, W.W.; Restifo, N.P. B16 as a mouse model for human melanoma. Curr. Protoc. Immunol. 2001. [Google Scholar] [CrossRef]
- De Robertis, M.; Massi, E.; Poeta, M.L.; Carotti, S.; Morini, S.; Cecchetelli, L.; Signori, E.; Fazio, V.M. The AOM/DSS murine model for the study of colon carcinogenesis: From pathways to diagnosis and therapy studies. J. Carcinog. 2011, 10, 9. [Google Scholar] [CrossRef] [PubMed]
- Thaker, A.I.; Shaker, A.; Rao, M.S.; Ciorba, M.A. Modeling colitis-associated cancer with azoxymethane (AOM) and dextran sulfate sodium (DSS). J. Vis. Exp. 2012. [Google Scholar] [CrossRef] [PubMed]
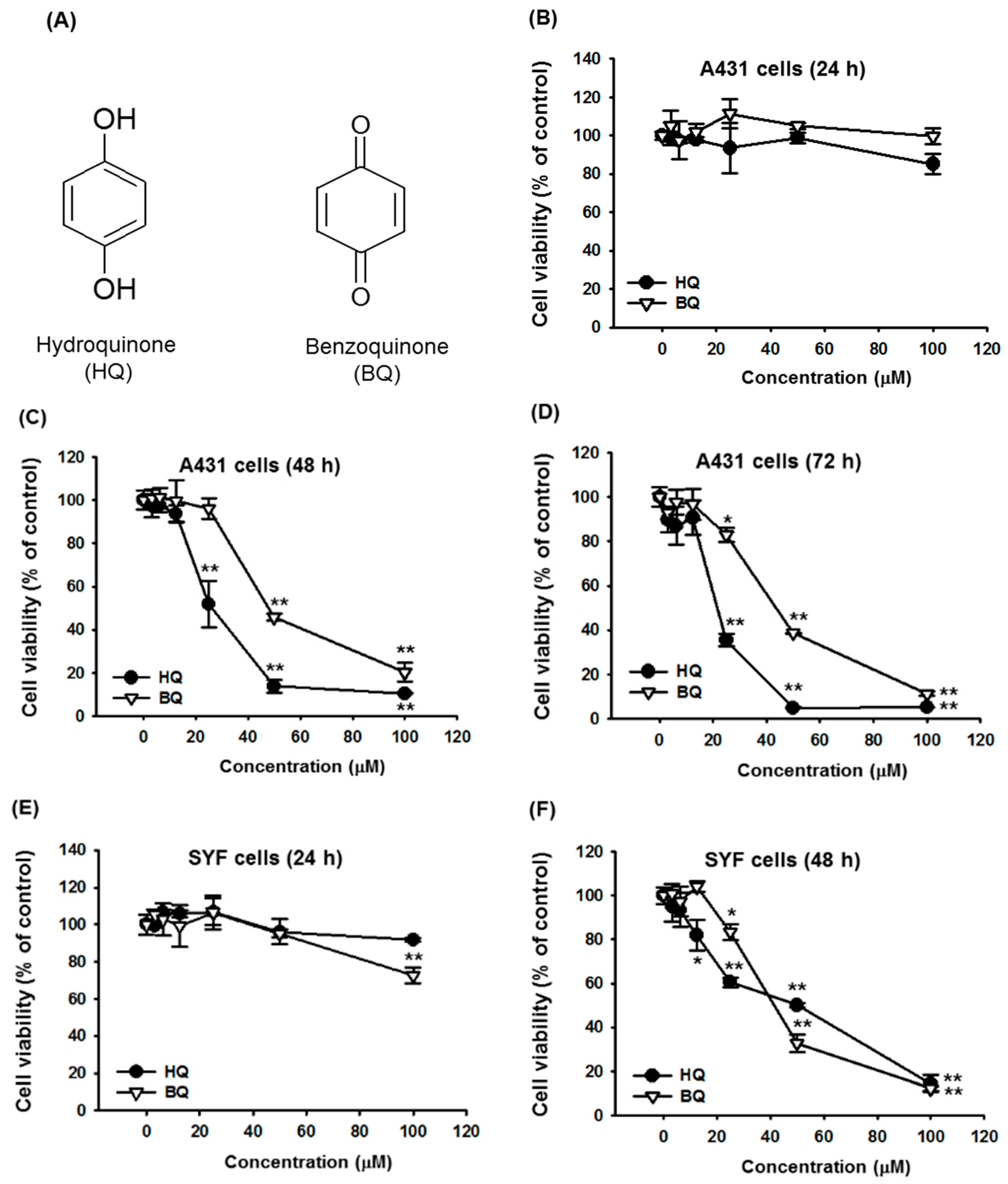
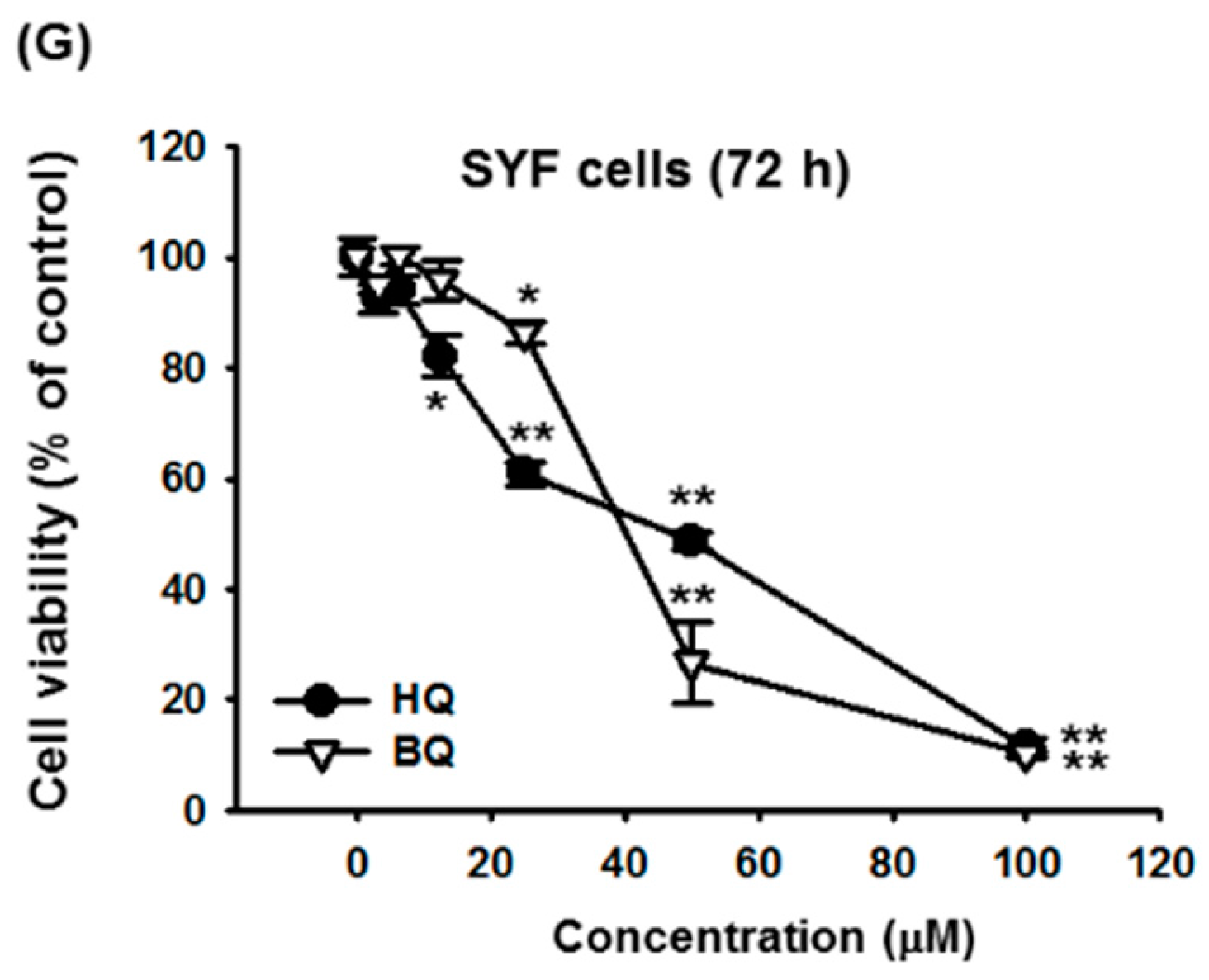

| Compound | IC50 Value (μM) at 72 h | |
|---|---|---|
| A431 Cells | SYF Cells | |
| HQ | 23.3 | 37.5 |
| BQ | 54.1 | 52.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byeon, S.E.; Yi, Y.-S.; Lee, J.; Yang, W.S.; Kim, J.H.; Kim, J.; Hong, S.; Kim, J.-H.; Cho, J.Y. Hydroquinone Exhibits In Vitro and In Vivo Anti-Cancer Activity in Cancer Cells and Mice. Int. J. Mol. Sci. 2018, 19, 903. https://doi.org/10.3390/ijms19030903
Byeon SE, Yi Y-S, Lee J, Yang WS, Kim JH, Kim J, Hong S, Kim J-H, Cho JY. Hydroquinone Exhibits In Vitro and In Vivo Anti-Cancer Activity in Cancer Cells and Mice. International Journal of Molecular Sciences. 2018; 19(3):903. https://doi.org/10.3390/ijms19030903
Chicago/Turabian StyleByeon, Se Eun, Young-Su Yi, Jongsung Lee, Woo Seok Yang, Ji Hye Kim, Jooyoung Kim, Suntaek Hong, Jong-Hoon Kim, and Jae Youl Cho. 2018. "Hydroquinone Exhibits In Vitro and In Vivo Anti-Cancer Activity in Cancer Cells and Mice" International Journal of Molecular Sciences 19, no. 3: 903. https://doi.org/10.3390/ijms19030903
APA StyleByeon, S. E., Yi, Y.-S., Lee, J., Yang, W. S., Kim, J. H., Kim, J., Hong, S., Kim, J.-H., & Cho, J. Y. (2018). Hydroquinone Exhibits In Vitro and In Vivo Anti-Cancer Activity in Cancer Cells and Mice. International Journal of Molecular Sciences, 19(3), 903. https://doi.org/10.3390/ijms19030903








