Individual versus Combinatorial Effects of Silicon, Phosphate, and Iron Deficiency on the Growth of Lowland and Upland Rice Varieties
Abstract
1. Introduction
2. Results
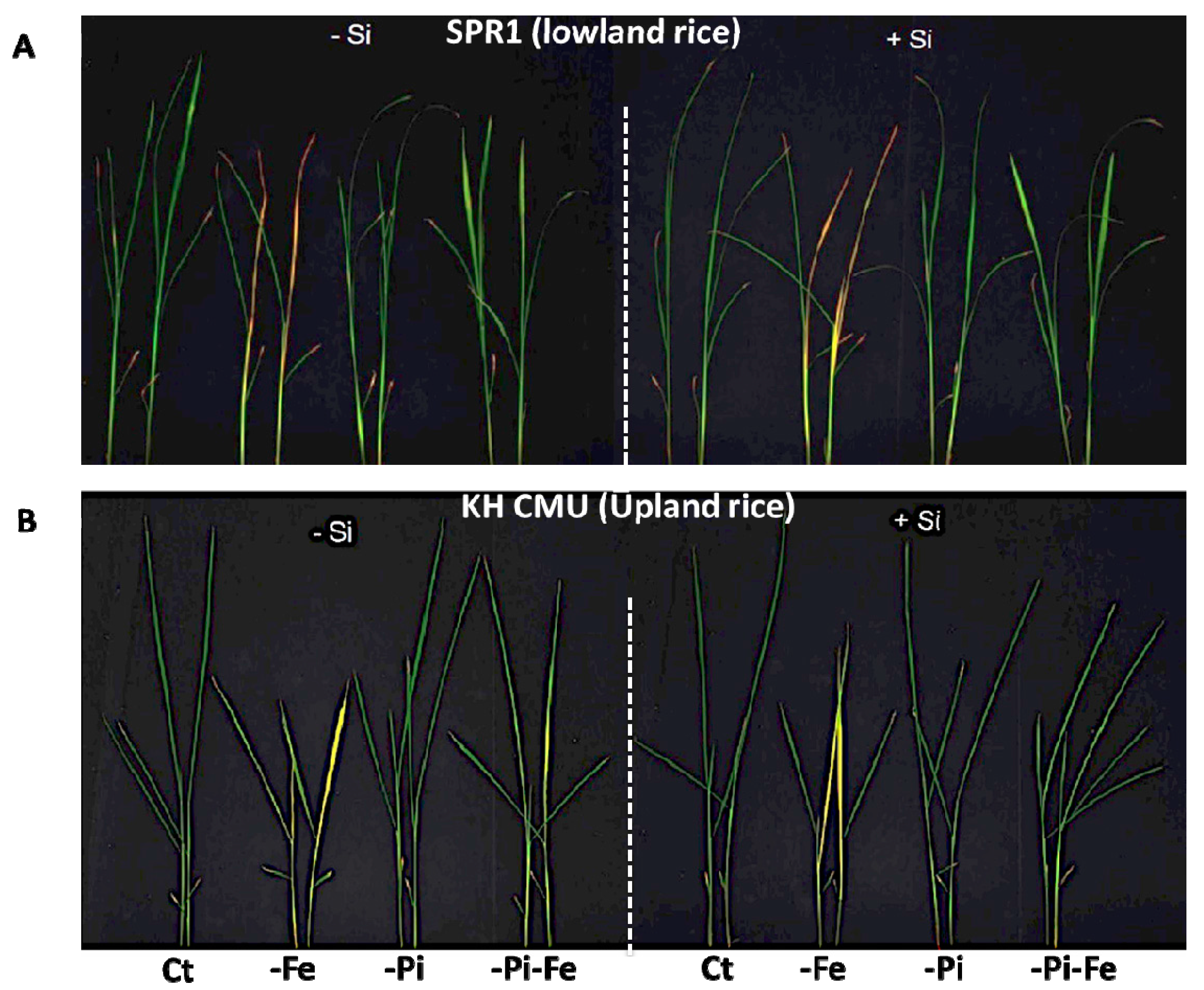
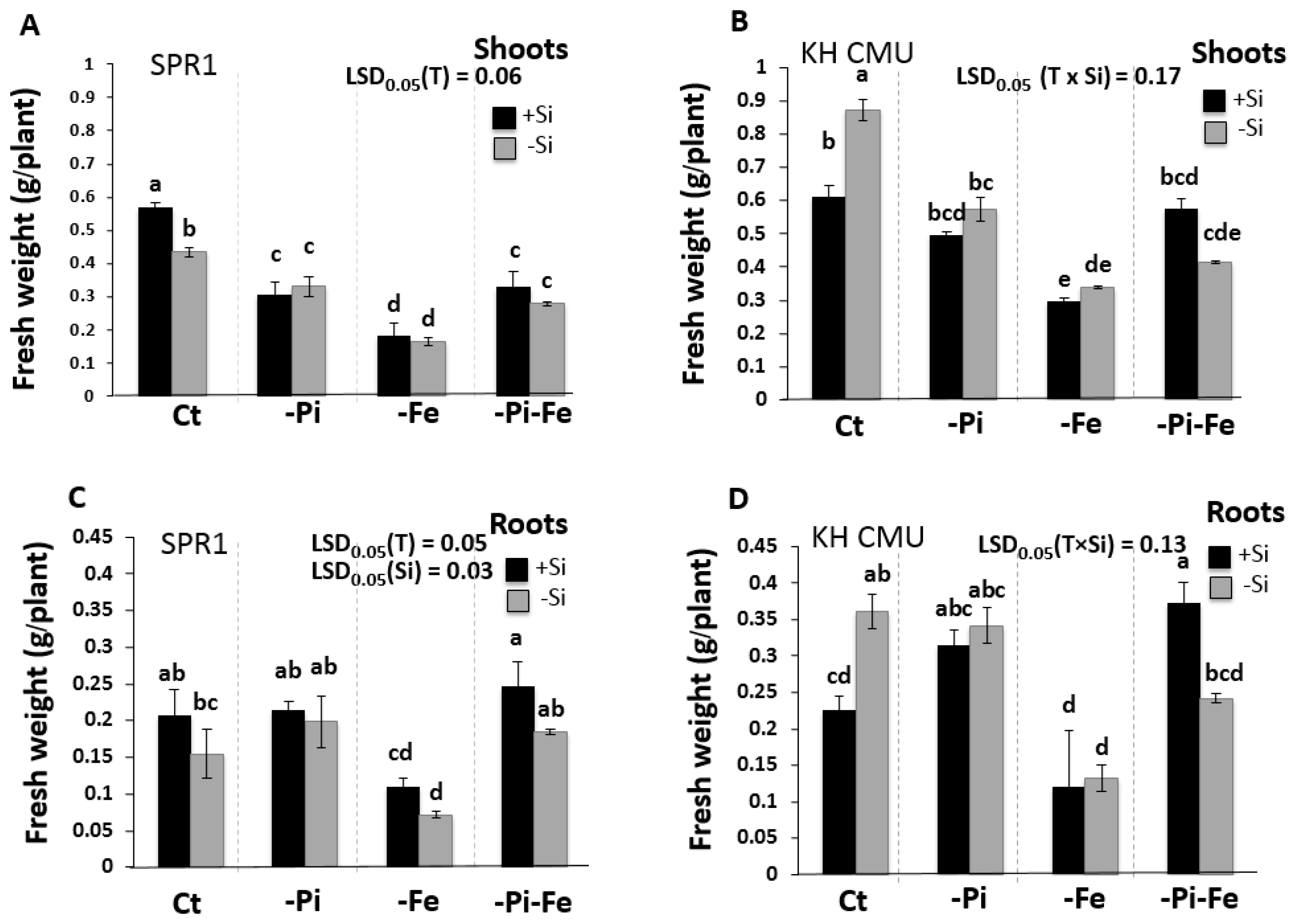
2.1. The Effect of Silicon Deficiency on Rice Biomass Varies with the Availability of Pi and Fe
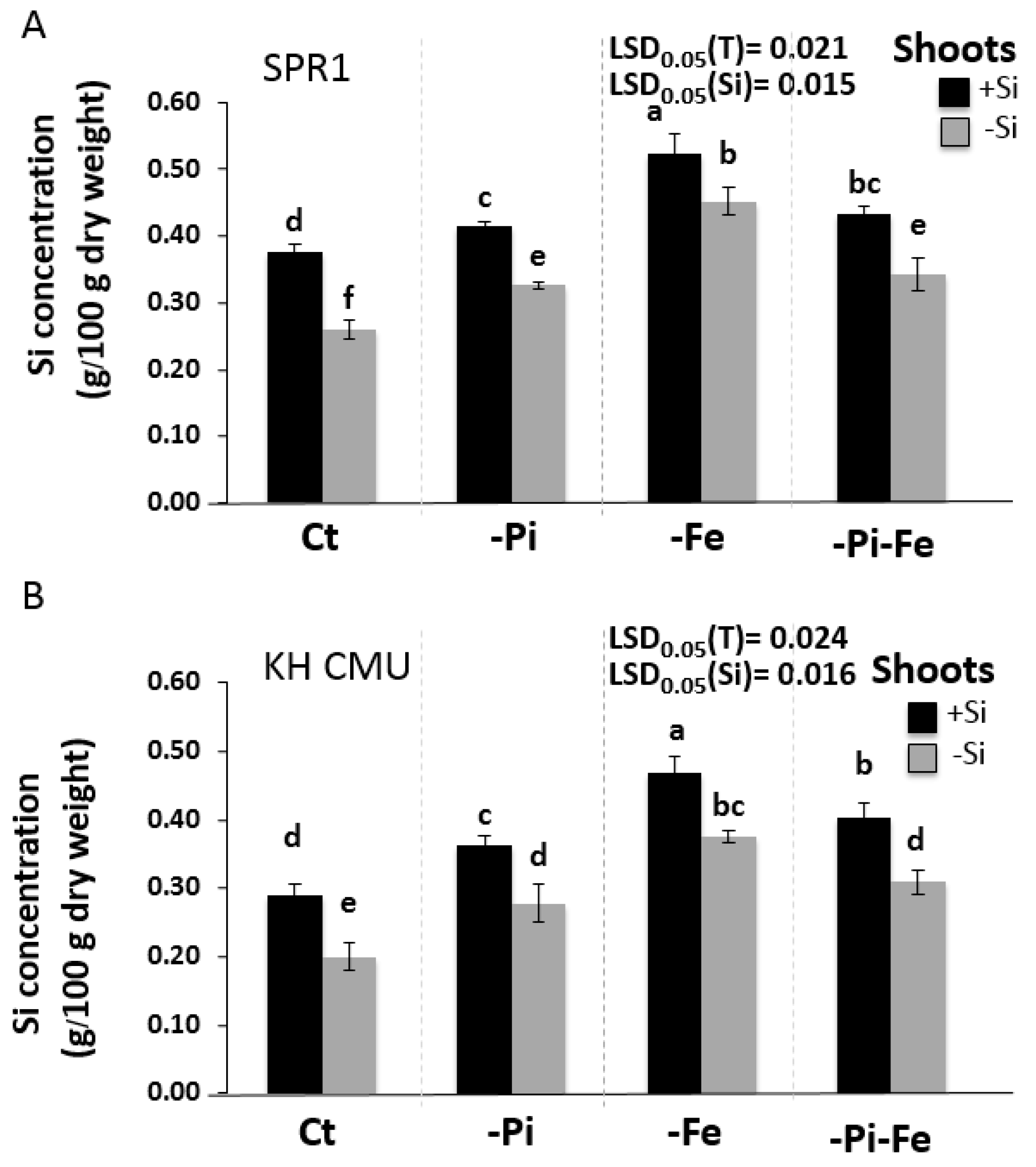
2.2. The Availability of Pi and Fe Affects Si Accumulation in Rice
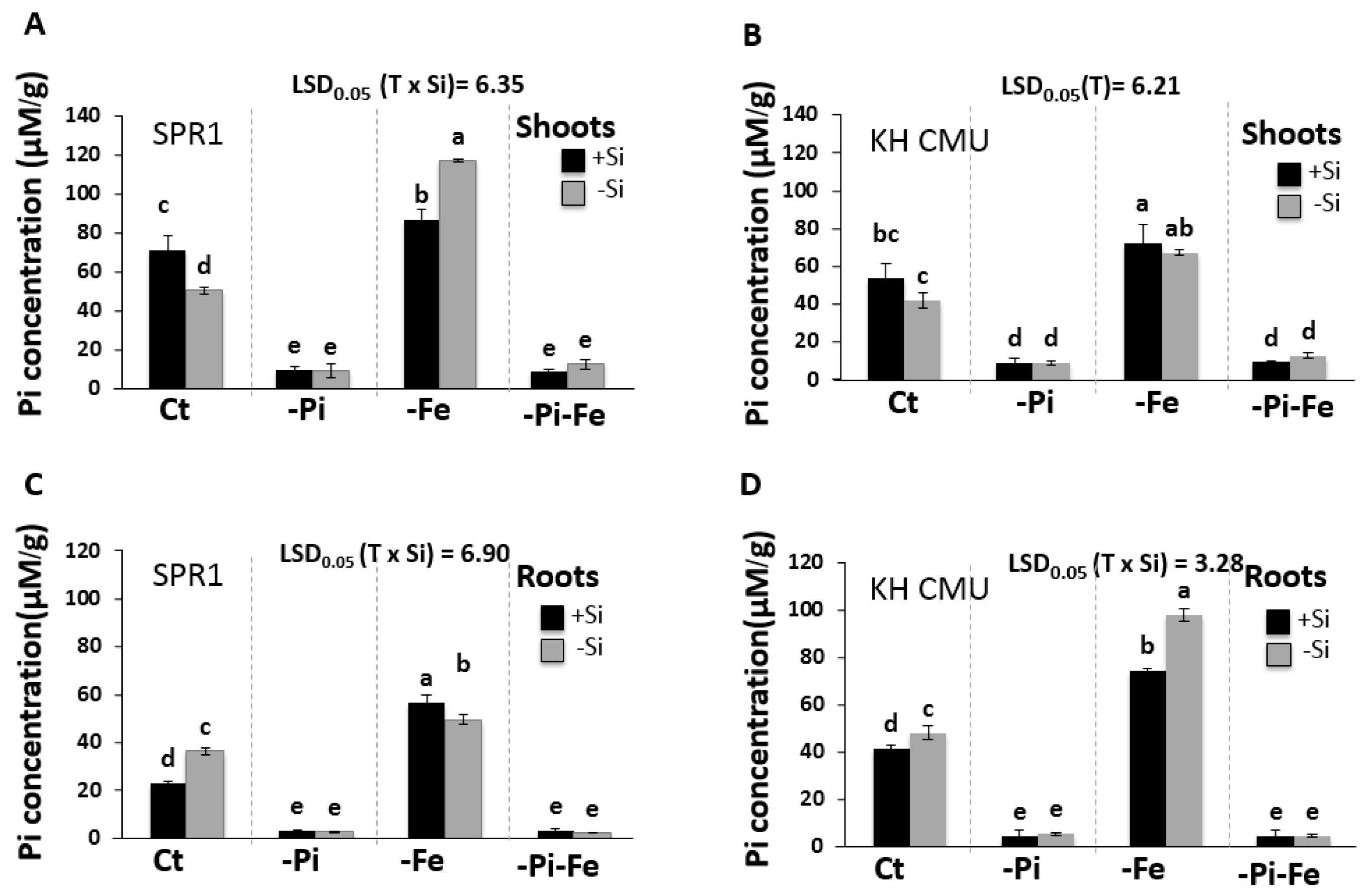
2.3. The Supply of Si and Fe Modulates Pi Accumulation in Rice
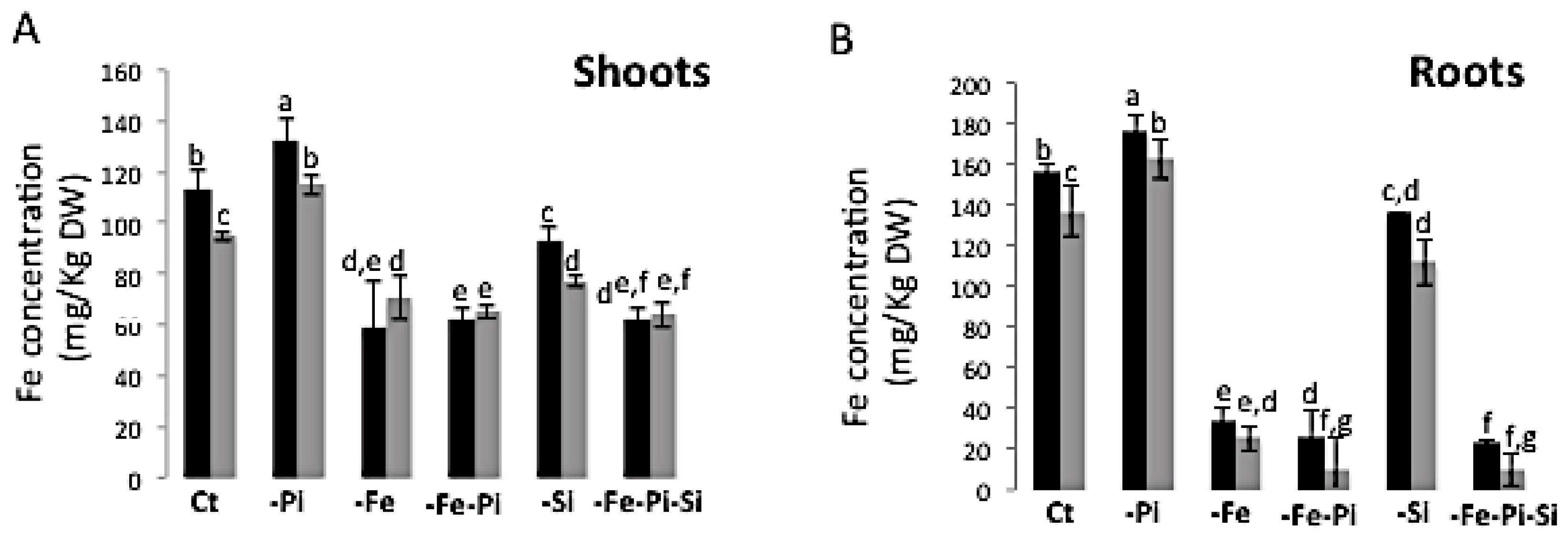
2.4. The Availability of Pi and Si Affects Fe Accumulation in Rice
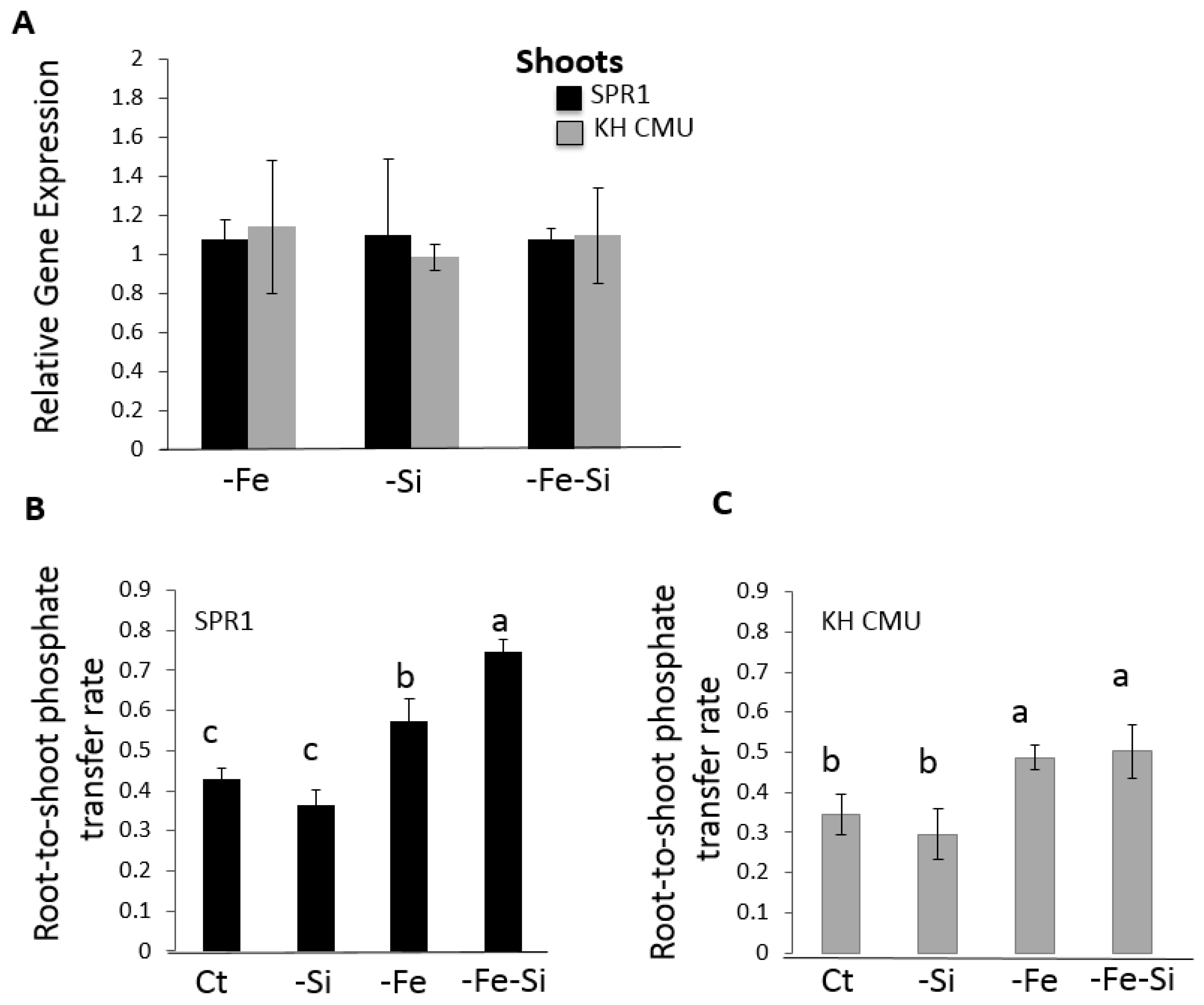
2.5. Si and Fe Homeostases Interact and Differentially Modulate Pi Root-to-Shoot Transfer in the Two Rice Varieties
3. Discussion
4. Materials and Methods
4.1. Rice Growth Condition
4.2. Sample Collection
4.3. Nutrient Concentration Analysis
4.4. Real-Time Quantitative Reverse-Transcription PCR
4.5. Phosphate Root-to-Shoot Translocation Measurements
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Reynolds, O.L.; Keeping, M.G.; Meyer, J.H. Silicon-augmented resistance of plants to herbivorous insects: A review. Ann. Appl. Biol. 2009, 155, 171–186. [Google Scholar] [CrossRef]
- Meharg, C.; Meharg, A.A. Silicon, the silver bullet for mitigating biotic and abiotic stress, and improving grain quality, in rice? Environ. Exp. Bot. 2015, 120, 8–17. [Google Scholar] [CrossRef]
- Keeping, M.G.; Meyer, J.H. Silicon-mediated resistance of sugarcane to Eldana saccharina Walker (Lepidoptera: Pyralidae): Effects of silicon source and cultivar. J. Appl. Entomol. 2006, 130, 410–420. [Google Scholar] [CrossRef]
- Massey, F.P.; Hartley, S.E. Physical defences wear you down: Progressive and irreversible impacts of silica on insect herbivores. J. Anim. Ecol. 2009, 78, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Fawe, A.B.; Abou-Zaid, M.; Menzies, J.G.; Bélanger, R.R. Silicon-mediated accumulation of flavonoid phytoalexins in cucumber. Phytopathology 1998, 88, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.B.; Moraes, J.C.; Santos, C.D.; Goussain, M.M. Resistance induction in wheat plants by silicon and aphids. Sci. Agric. 2005, 62, 547–551. [Google Scholar] [CrossRef]
- Fauteux, F.; Chain, F.; Belzile, F.; Menzies, J.G.; Bélanger, R.R. The protective role of silicon in the Arabidopsis-powdery mildew pathosystem. Proc. Natl. Acad. Sci. USA 2006, 103, 17554–17559. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Song, Y.Y.; Long, J.; Wang, R.L.; Baerson, S.R.; Pan, Z.Q. Priming of jasmonate-mediated antiherbivore defense responses in rice by silicon. Proc. Natl. Acad. Sci. USA 2013, 110, 3631–3639. [Google Scholar] [CrossRef] [PubMed]
- Kvedaras, O.L.; An, M.; Choi, Y.S.; Gurr, G.M. Silicon enhances natural enemy attraction and biological control through induced plant defenses. Bull. Entomol. Res. 2010, 100, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Khan, A.L.; Waqas, M.; Lee, I.J. Silicon Regulates Antioxidant Activities of Crop Plants under Abiotic-Induced Oxidative Stress: A Review. Front. Plant Sci. 2017, 8, 510. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sardans, J.; Lai, D.Y.F.; Wang, C.; Zeng, C.; Tong, C.; Liang, Y.; Penuelas, J. Effects of steel slag application on greenhouse gas emissions and crop yield over multiple growing seasons in a subtropical paddy field in China. Field Crops Res. 2015, 171, 146–156. [Google Scholar] [CrossRef]
- Shi, Y.; Zhang, Y.; Han, W.; Feng, R.; Hu, Y.; Guo, J.; Gong, H. Silicon enhances water stress tolerance by improving root hydraulic conductance in Solanum lycopersicum L. Front. Plant Sci. 2016, 7, 196. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Meunier, J.D.; Davidian, J.C.; Pokrovsky, O.S.; Bovet, N.; Keller, C. Silicon alleviates Cd stress of wheat seedlings (Triticum turgidum L. cv. Claudio) grown in hydroponics. Environ. Sci. Pollut. Res. 2016, 23, 1414–1427. [Google Scholar] [CrossRef] [PubMed]
- Pontigo, S.; Godoy, K.; Jiménez, H.; Gutiérrez-Moraga, A.; de la Luz Mora, M.; Cartes, P. Silicon-mediated alleviation of aluminum toxicity by modulation of Al/Si uptake and antioxidant performance in ryegrass plants. Front. Plant Sci. 2017, 8, 642. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, J.; Samardzic, J.; Maksimović, V.; Timotijevic, G.; Stevic, N.; Laursen, K.H.; Hansen, T.H.; Husted, S.; Schjoerring, J.K.; Liang, Y.; et al. Silicon alleviates iron deficiency in cucumber by promoting mobilization of iron in the root apoplast. New Phytol. 2013, 198, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, R.K.; Ma, J.F.; Belanger, R. Role of Silicon in Plants. Front. Plant Sci. 2017, 8, 1858. [Google Scholar] [CrossRef] [PubMed]
- Epstein, E. Silicon. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 641–664. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Yamaji, N. Silicon uptake and accumulation in higher plants. Trends Plant Sci. 2006, 11, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Takahashi, E. The effect of silicic acid on rice in a P-deficient soil. Plant Soil. 1990, 126, 121–125. [Google Scholar] [CrossRef]
- Kewai, H.; Yan, L.; Guan, L.Z. Effect of supply silicon on adsorption and desorption action of phosphorus in paddy soil. Plant Nutr. Fert. Sci. 2002, 8, 214–218. [Google Scholar]
- Zheng, L.; Huang, F.; Narsai, R.; Wu, J.; Giraud, E.; He, F.; Cheng, L.; Wang, F.; Wu, P.; Whelan, J.; et al. Physiological and transcriptome analysis of iron and phosphorus Interaction in rice seedlings. Plant Physiol. 2009, 151, 262–274. [Google Scholar] [CrossRef] [PubMed]
- Saenchai, C.; Bouain, N.; Kisko, M.; Prom-u-thai, C.; Doumas, P.; Rouached, H. The involvement of OsPHO1;1 in the regulation of iron transport through integration of phosphate and zinc deficiency signaling. Front. Plant Sci. 2016, 7, 396. [Google Scholar] [CrossRef] [PubMed]
- Rouached, H.; Rhee, S.Y. System-level understanding of plant mineral nutrition in the big data era. Curr. Opin. Syst. Biol. 2017, 4, 71–77. [Google Scholar] [CrossRef]
- Mongon, J.; Chaiwong, N.; Bouain, N.; Prom-u-Thai, C.; Secco, D.; Rouached, H. Phosphorus and Iron Deficiencies Influences Rice Shoot Growth in an Oxygen Dependent Manner: Insight from Upland and Lowland Rice. Int. J. Mol. Sci. 2017, 18, 607. [Google Scholar] [CrossRef] [PubMed]
- Briat, J.; Rouached, H.; Tissot, N.; Gaymard, F.; Dubos, C. Integration of P, S, Fe and Zn nutrition signals in Arabidopsis thaliana: Potential involvement of PHOSPHATE STARVATION RESPONSE1 (PHR1). Front. Plant Sci. 2015, 6, 290. [Google Scholar] [CrossRef] [PubMed]
- Secco, D.; Baumann, A.; Poirier, Y. Characterization of the rice PHO1 gene family reveals a key role for OsPHO1;2 in phosphate homeostasis and the evolution of a distinct clade in dicotyledons. Plant physiol. 2010, 152, 1693–1704. [Google Scholar] [CrossRef] [PubMed]
- Jabnoune, M.; Secco, D.; Lecampion, C.; Robaglia, C.; Shu, Q.; Poirier, Y. A rice cis-natural antisense RNA acts as a translational enhancer for its cognate mRNA and contributes to phosphate homeostasis and plant fitness. Plant Cell. 2013, 25, 4166–4182. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S. Fundamentals of Rice Crop Science; International Rice Research Institute: Los Banos, Philippines, 1981. [Google Scholar]
- Ponnamperuma, F.N. Growth-limiting factors of aerobic soils. In Major Research in Upland Rice; IRRI: Los Banos, Laguna, Philippines, 1975; pp. 40–43. [Google Scholar]
- Drew, M.D.; Mullins, A.P.; Rice, D.A. Synthesis, characterization and structural properties of some copper (II) trans-cinnamates and related compounds. Polyhedron 1994, 13, 1631–1637. [Google Scholar] [CrossRef]
- Mongon, J.; Jantasorn, A.; Oupkaew, P.; Prom-u-Thai, C.; Rouached, H. The Time of Flooding Occurrence is Critical for Yield Production in Rice and Vary in a Genotype-Dependent Manner. Online J Biol Sci. 2017, 17, 58–65. [Google Scholar] [CrossRef][Green Version]
- Bityutskii, N.; Pavlovic, J.; Yakkonen, K.; Maksimović, V.; Nikolic, M. Contrasting effect of silicon on iron, zinc and manganese status and accumulation of metal-mobilizing compounds in micronutrient-deficient cucumber. Plant Physiol. Biochem 2014, 74, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, J.; Samardzic, J.; Kostic, L.; Laursen, K.H.; Natic, M.; Timotijevic, G.; Schjoerring, J.K.; Nikolic, M. Silicon enhances leaf remobilization of iron in cucumber under limited iron conditions. Ann. Bot. 2016, 118, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Secco, D.; Bouain, N.; Rouached, A.; Prom, U.T.C.; Hanin, M.; Pandey, A.K.; Rouached, H. Phosphate, phytate and phytases in plants: From fundamental knowledge gained in Arabidopsis to potential biotechnological applications in wheat. Crit. Rev. Biotechnol. 2017, 37, 898–910. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Kisko, M.; Dubos, C.; Lacombe, B.; Berthomieu, P.; Krouk, G.; Rouached, H. TransDetect identifies a new regulatory module controlling phosphate accumulation in Arabidopsis. Plant Physiol. 2017, 175, 916–926. [Google Scholar] [PubMed]
- Bouain, N.; Doumas, P.; Rouached, H. Recent Advances in Understanding the Molecular Mechanisms Regulating the Root System Response to Phosphate Deficiency in Arabidopsis. Curr. Genom. 2016, 17, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Kisko, M.; Bouain, N.; Safi, A.; Medici, A.; Akkers, R.C.; Secco, D.; Fouret, G.; Krouk, G.; Aarts, M.G.; Busch, W.; et al. LPCAT1 controls phosphate homeostasis in a zinc-dependent manner. eLife 2018, 7, e32077. [Google Scholar] [CrossRef] [PubMed]
- Rouached, H. Multilevel coordination of phosphate and sulfate homeostasis in plants. Plant Signal Behav. 2011, 6, 952–955. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Foorno, D.A.; Cock, J.H.; Gomez, K.A. Laboratory Manual for Physiological Studies of Rice, 3rd ed.; International Rice Research Institute: Los Baños, Philippines, 1976; pp. 61–63. [Google Scholar]
- Ames, B.N. Assay of inorganic phosphate, total phosphate and phosphatases. Methods Enzymol. 1966, 8, 115–118. [Google Scholar]
- Dai, W.M.; Zhang, K.Q.; Duan, B.W.; Sun, C.X.; Zheng, K.L.; Cai, R.; Zhuang, J.Y. Rapid determination of silicon content in rice. Rice Sci. 2005, 12, 145–147. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods. 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Rouached, H.; Wirtz, M.; Alary, R.; Hell, R.; Arpat, A.B.; Davidian, J.C.; Fourcroy, P.; Berthomieu, P. Differential regulation of the expression of two high affinity sulfate transporters, SULTR1.1 and SULTR1.2, in Arabidopsis. Plant Physiol. 2008, 147, 897–911. [Google Scholar] [CrossRef] [PubMed]
- Krouk, G.; Lacombe, B.; Bielach, A.; Perrine-Walker, F.; Malinska, K.; Mounier, E.; Hoyerova, K.; Tillard, P.; Leon, S.; Ljung, K.; Zazimalova, E. Nitrate-regulated auxin transport by NRT1.1 defines a mechanism for nutrient sensing in plants. Developmental cell. 2010, 18, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Bouain, N.; Kisko, M.; Rouached, A.; Dauzat, M.; Lacombe, B.; Belgaroui, N.; Ghnaya, T.; Davidian, J.C.; Berthomieu, P.; Abdelly, C.; et al. Phosphate/zinc interaction analysis in two lettuce varieties reveals contrasting effects on biomass, photosynthesis, and dynamics of Pi transport. Bio. Med. Res. Int. 2014. [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaiwong, N.; Prom-u-thai, C.; Bouain, N.; Lacombe, B.; Rouached, H. Individual versus Combinatorial Effects of Silicon, Phosphate, and Iron Deficiency on the Growth of Lowland and Upland Rice Varieties. Int. J. Mol. Sci. 2018, 19, 899. https://doi.org/10.3390/ijms19030899
Chaiwong N, Prom-u-thai C, Bouain N, Lacombe B, Rouached H. Individual versus Combinatorial Effects of Silicon, Phosphate, and Iron Deficiency on the Growth of Lowland and Upland Rice Varieties. International Journal of Molecular Sciences. 2018; 19(3):899. https://doi.org/10.3390/ijms19030899
Chicago/Turabian StyleChaiwong, Nanthana, Chanakan Prom-u-thai, Nadia Bouain, Benoit Lacombe, and Hatem Rouached. 2018. "Individual versus Combinatorial Effects of Silicon, Phosphate, and Iron Deficiency on the Growth of Lowland and Upland Rice Varieties" International Journal of Molecular Sciences 19, no. 3: 899. https://doi.org/10.3390/ijms19030899
APA StyleChaiwong, N., Prom-u-thai, C., Bouain, N., Lacombe, B., & Rouached, H. (2018). Individual versus Combinatorial Effects of Silicon, Phosphate, and Iron Deficiency on the Growth of Lowland and Upland Rice Varieties. International Journal of Molecular Sciences, 19(3), 899. https://doi.org/10.3390/ijms19030899







