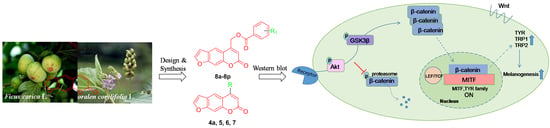
Novel Furocoumarin Derivatives Stimulate Melanogenesis in B16 Melanoma Cells by Up-Regulation of MITF and TYR Family via Akt/GSK3β/β-Catenin Signaling Pathways
Abstract
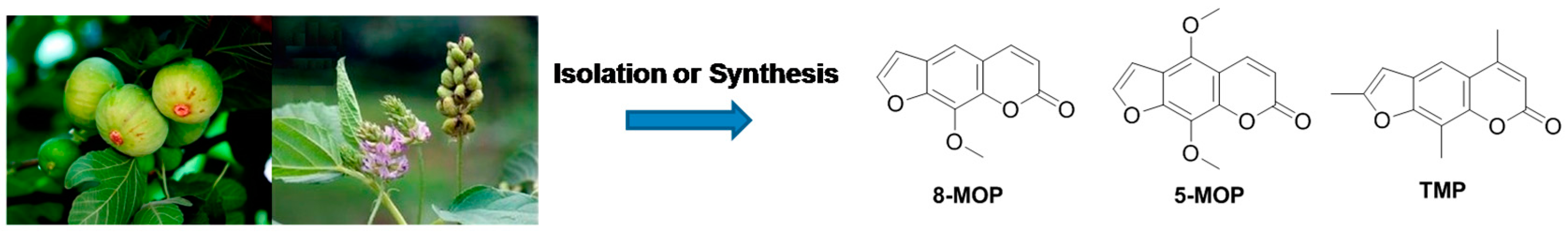
:1. Introduction
2. Results and Discussion
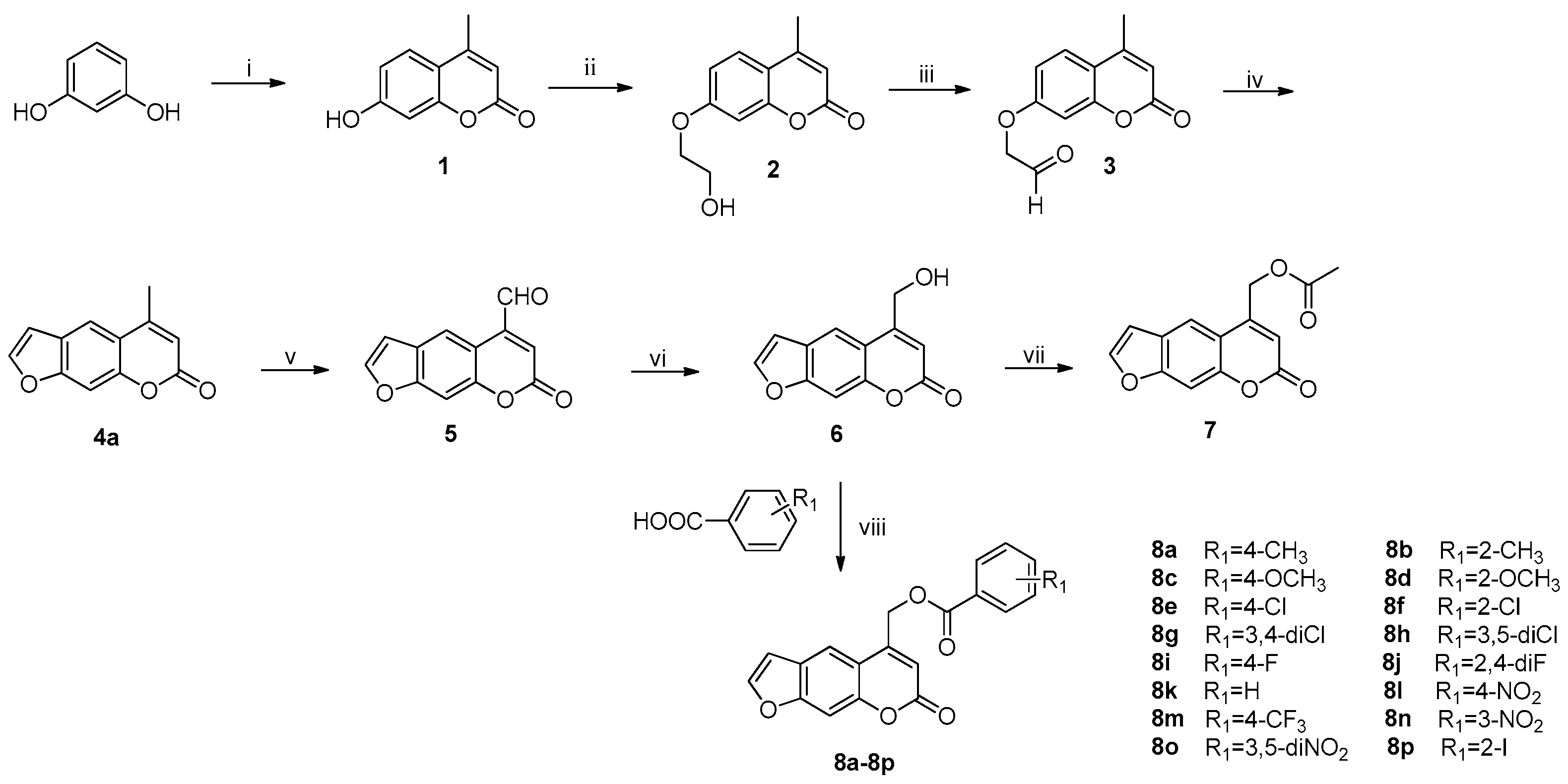
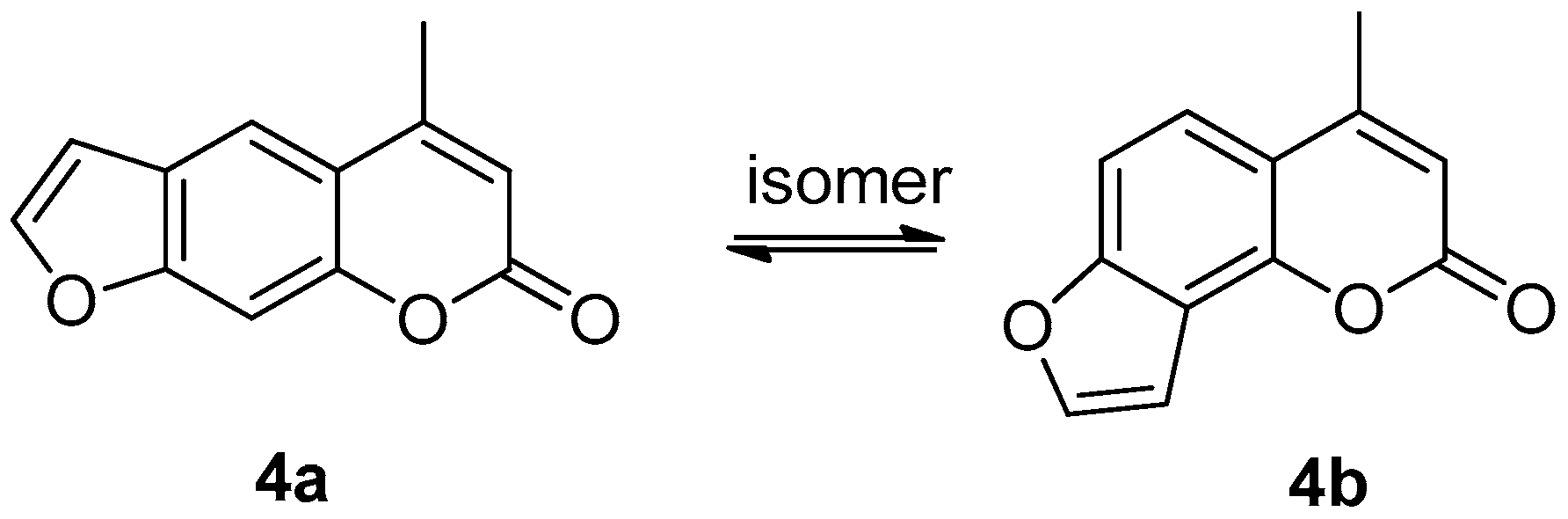
2.1. Synthesis
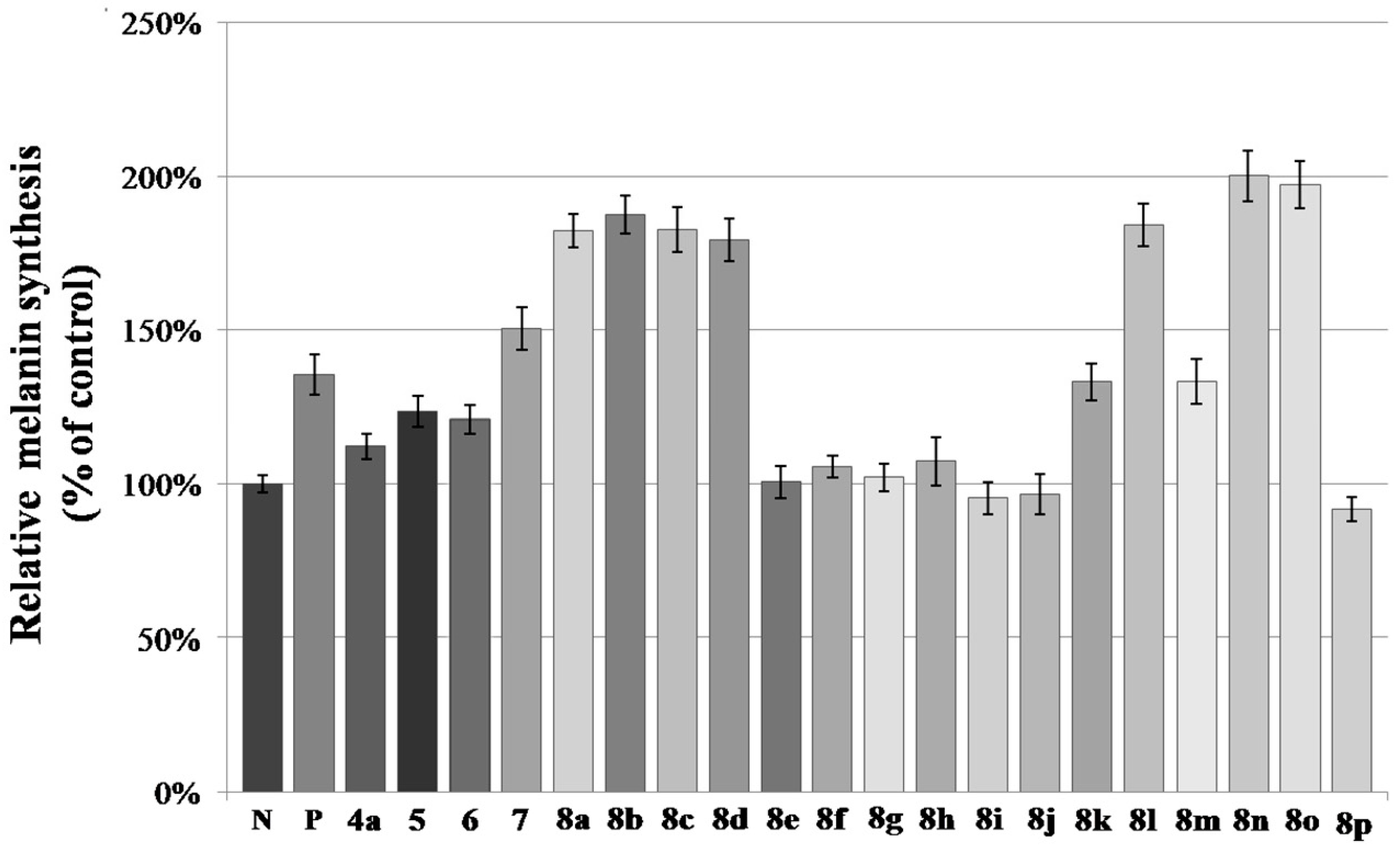
2.2. Melanin Synthesis Evaluation of 8a–8p
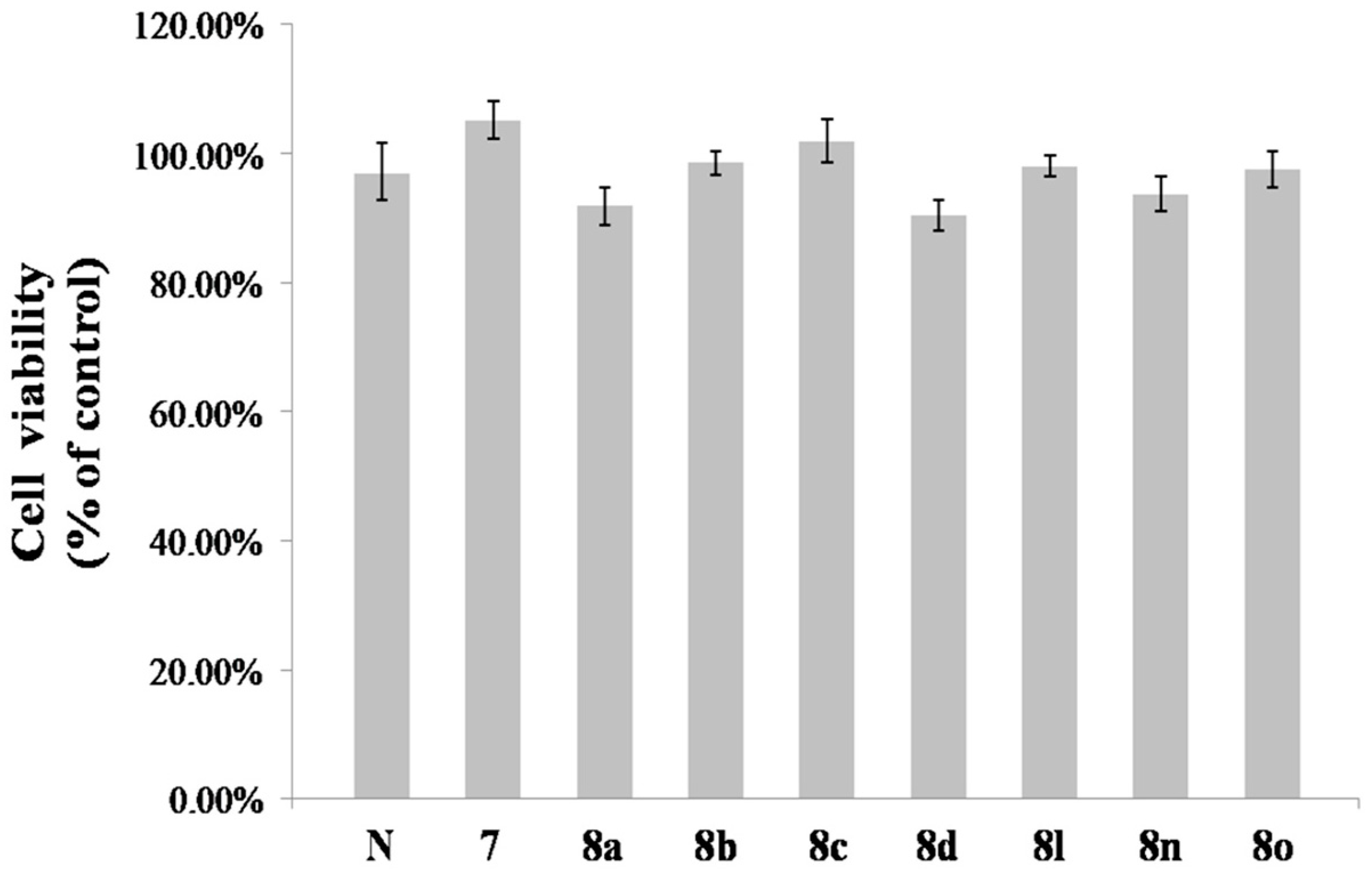
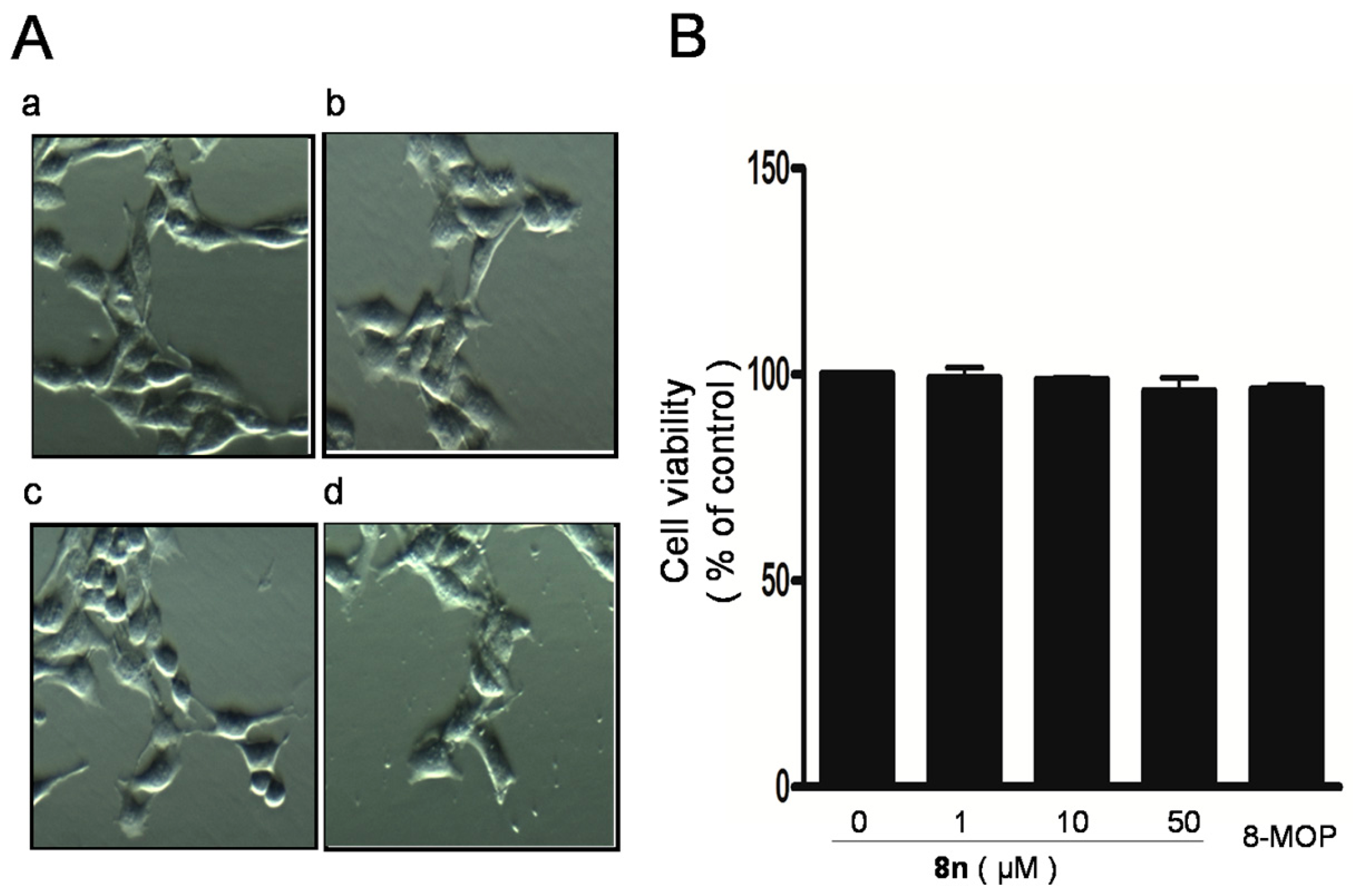
2.3. Effect of 8n on B16 Melanoma Cells Viability
2.4. Effect of 8n on Melanogenesis and Tyrosinase in B16 Melanoma Cells
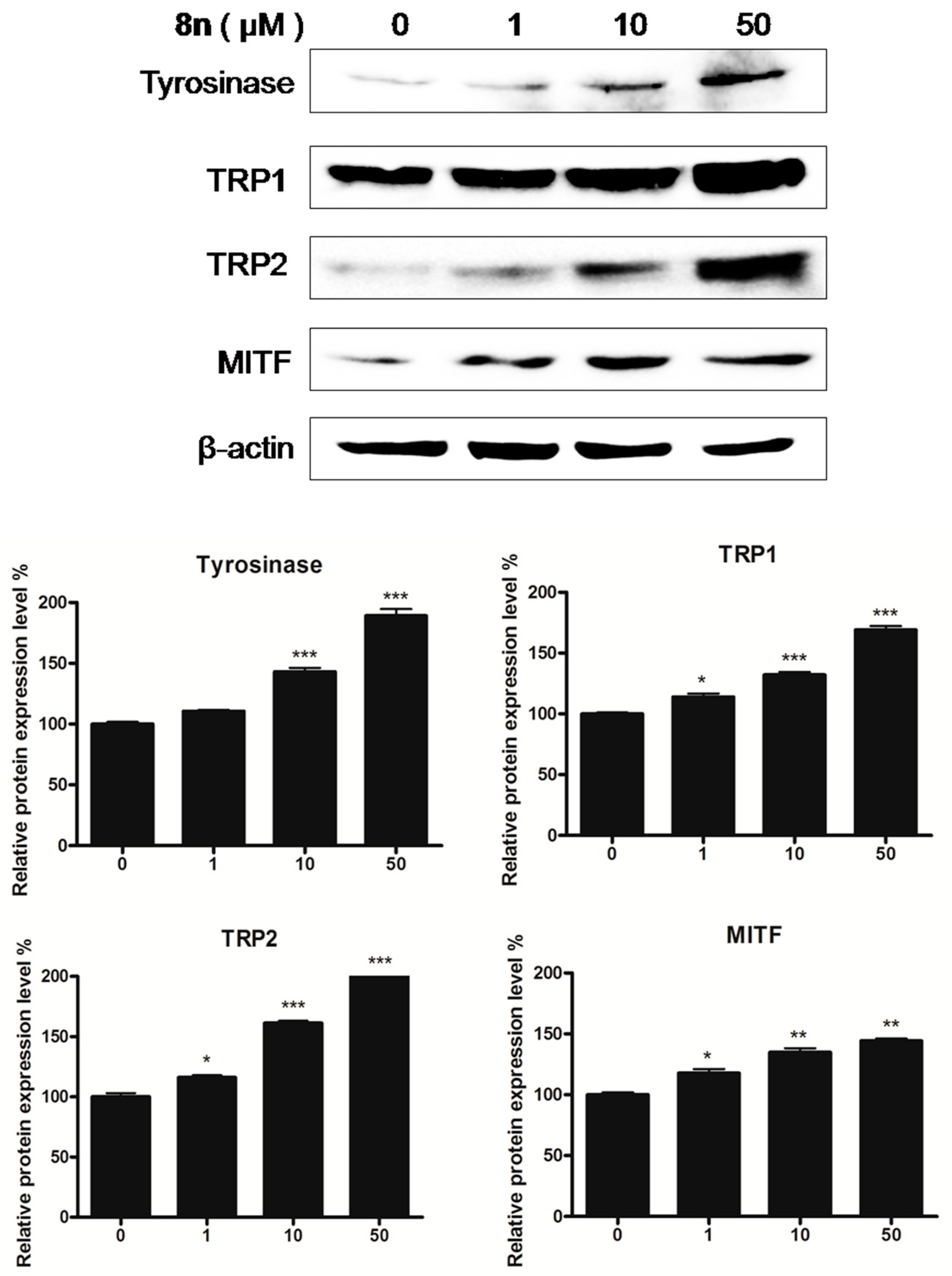
2.5. Effects of 8n on TYR Family and MITF Expression Levels in B16 Melanoma Cells
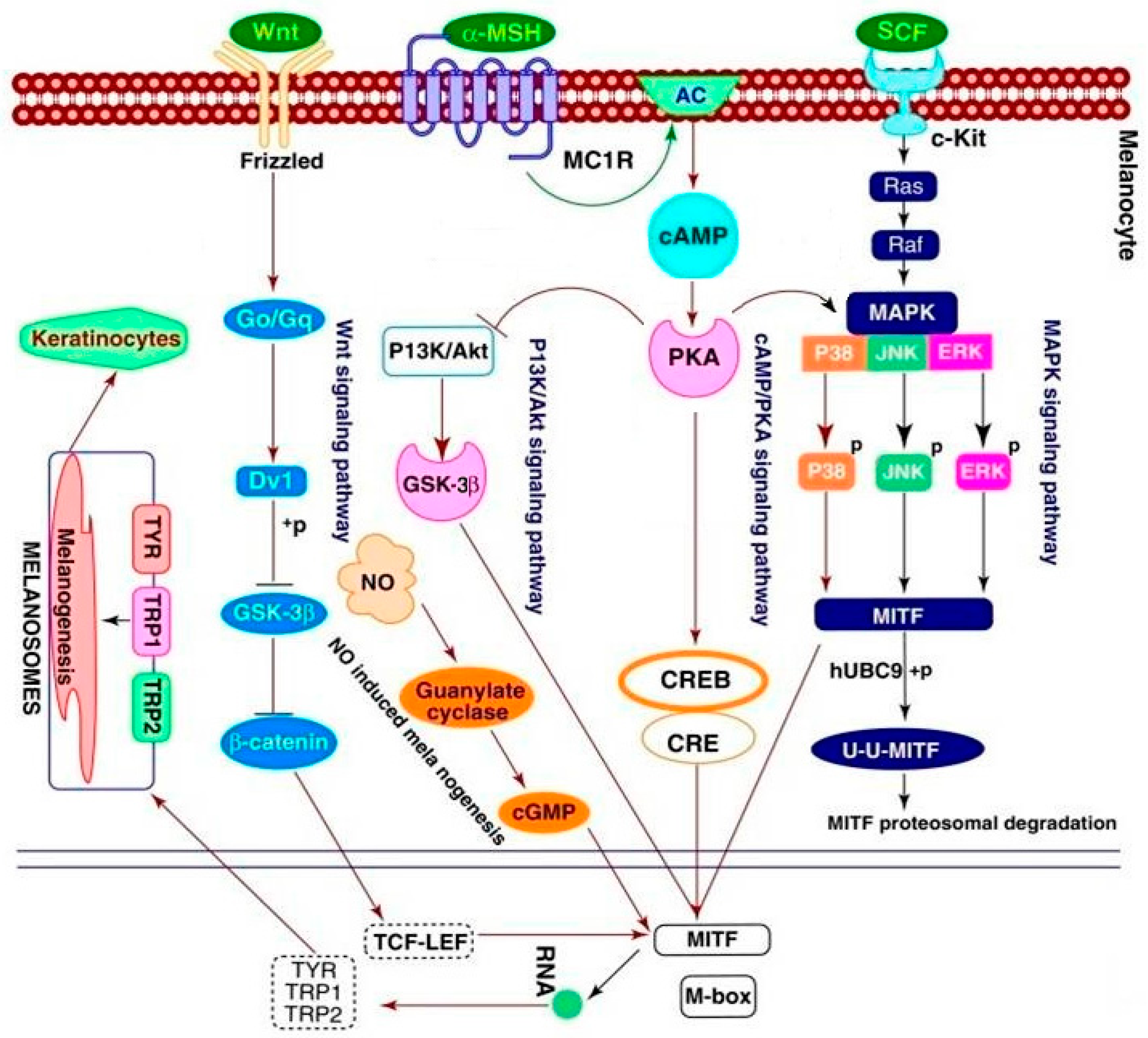
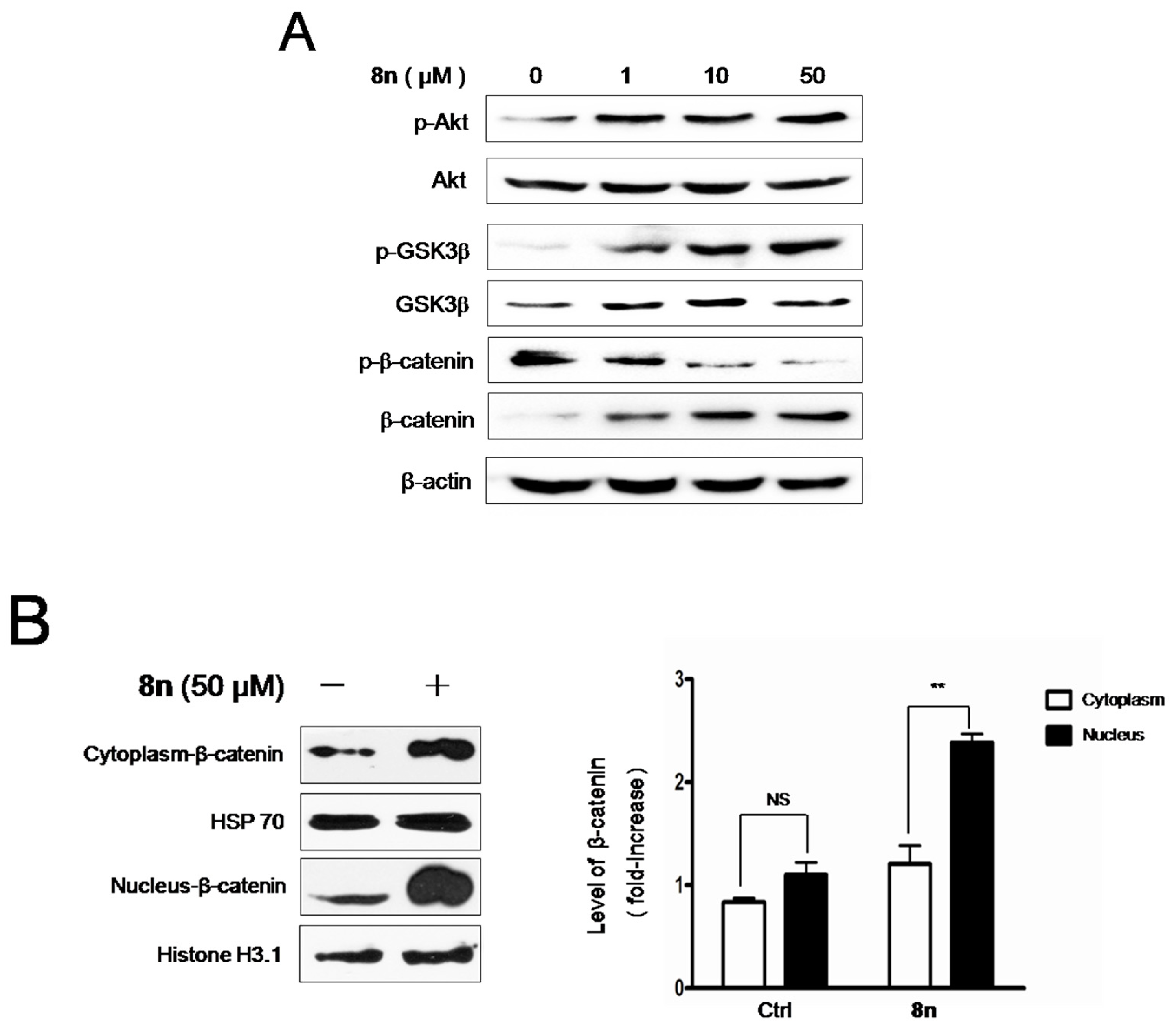
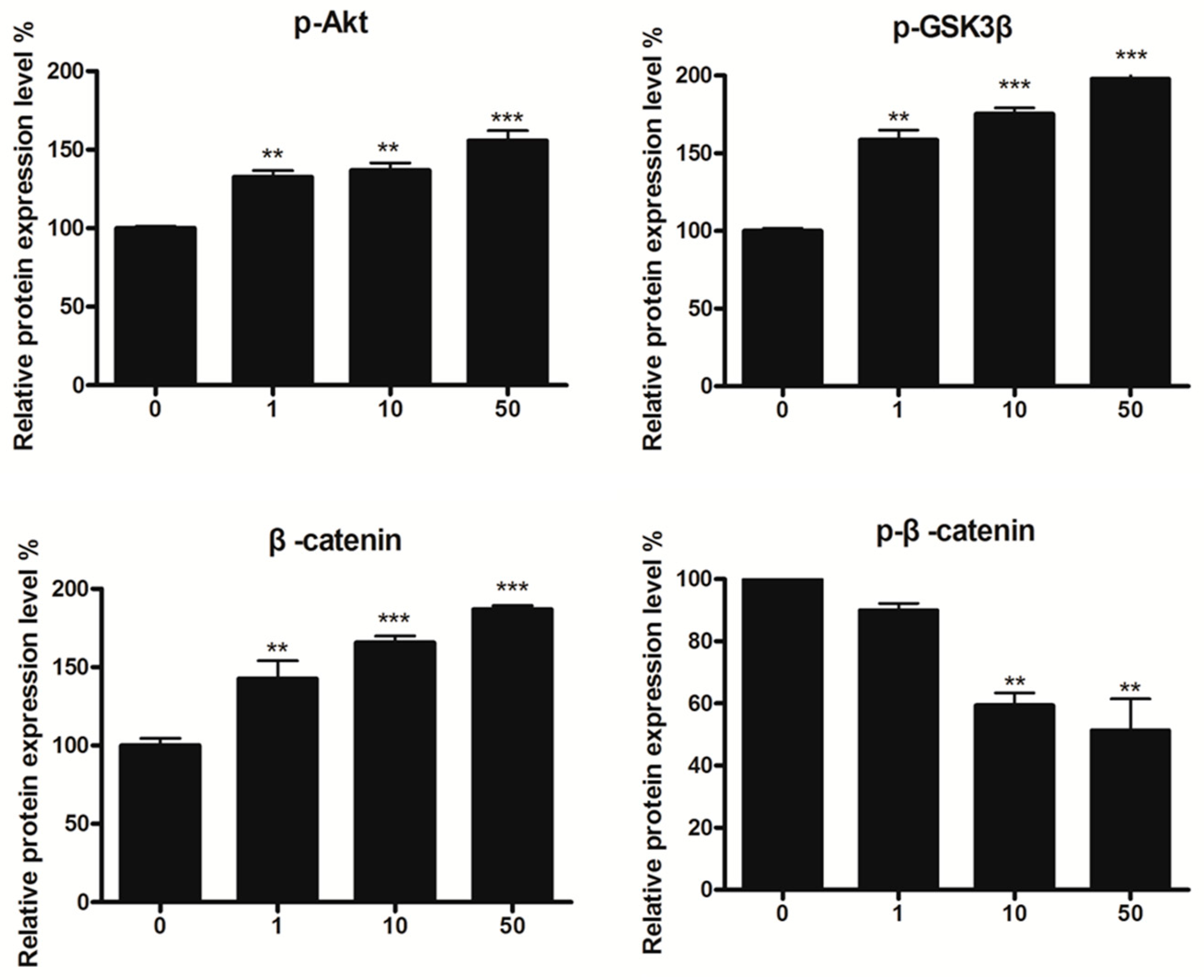
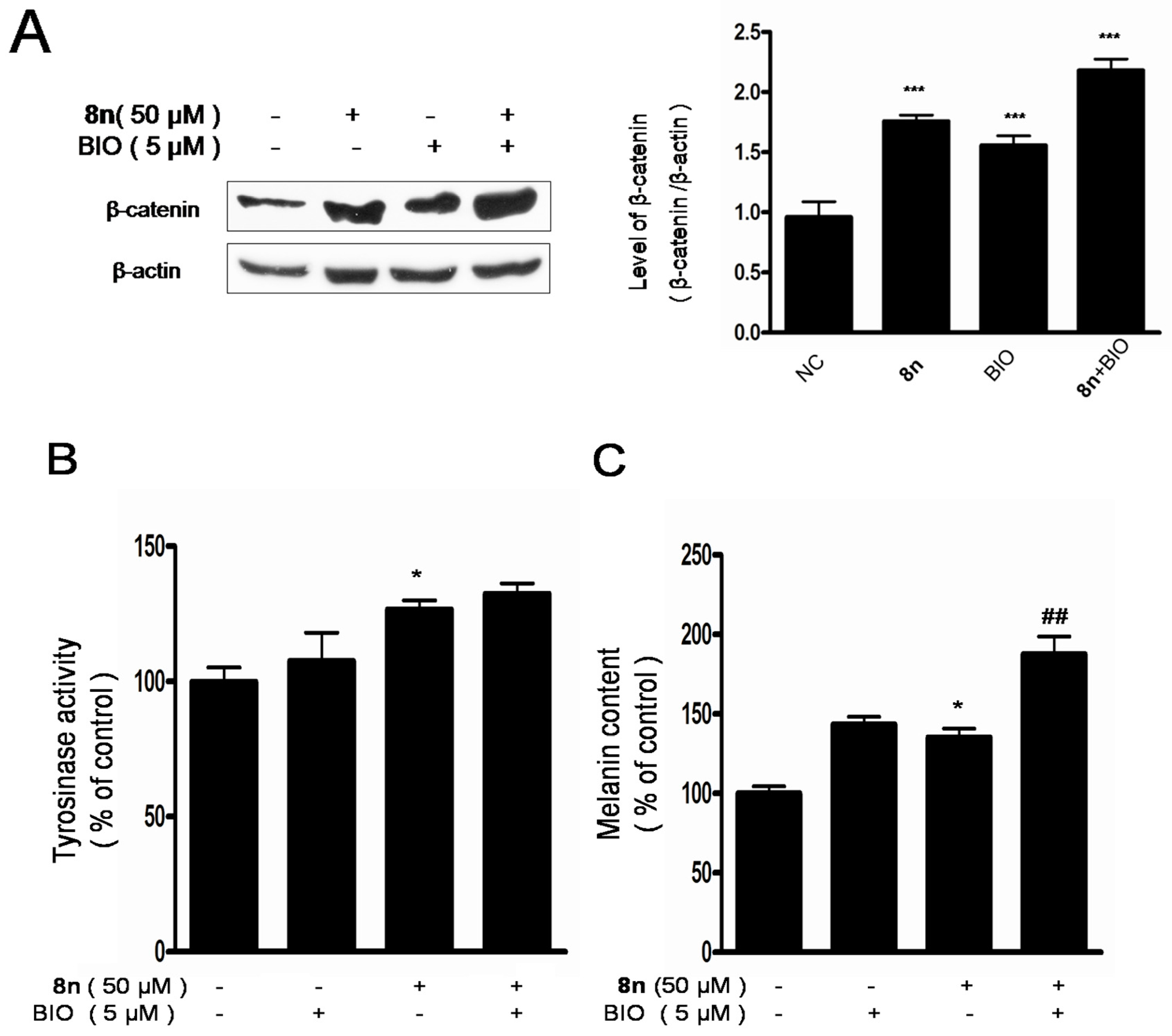
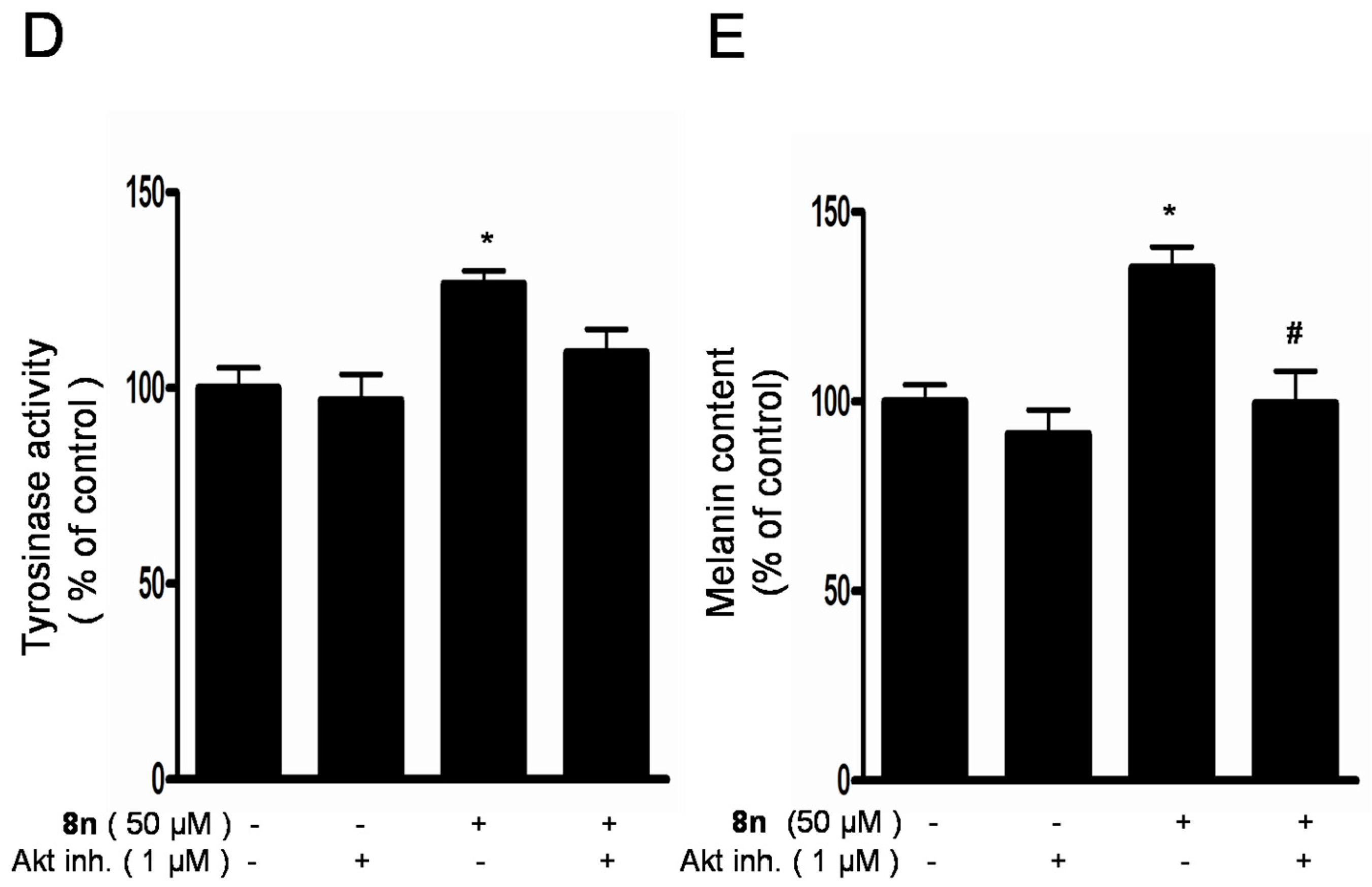
2.6. Effect of 8n on Akt and Wnt/β-Catenin Signaling Pathways Involved Inmelanogenesis
3. Materials and Methods
3.1. Chemistry
3.1.1. Preparation of 4-Methylumbelliferone (1)
3.1.2. Preparation of 7-(2-Hydroxyethoxy)-4-methyl-2H-chromen-2-one (2)
3.1.3. Preparation of 7-(2-Oxoethoxy)-4-methyl-2H-chromen-2-one (3)
3.1.4. Preparation of Compounds 4
3.1.5. Preparation of 7-Oxo-7H-furo[3,2-g]chromene-5-carbaldehyde (5)
3.1.6. Preparation of 5-(Hydroxymethyl)-7H-furo[3,2-g]chromen-7-one (6)
3.1.7. Preparation of (7-Oxo-7H-furo[3,2-g]chromen-5-yl)methyl acetate (7)
3.1.8. General Procedure Of preparation of Esters 8a–8p
3.2. Biological Activity
3.2.1. Cell Cultures
3.2.2. Cell Morphology and Cell Viability Measurement
3.2.3. Melanin Measurement
3.2.4. Tyrosinase Activity Assay
3.2.5. Western Blot Analysis
3.2.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ezzedine, K.; Sheth, V.; Rodrigues, M.; Eleftheriadou, V.; Harris, J.E.; Hamzavi, I.H.; Pandya, A.G. Vitiligo is not a cosmetic disease. J. Am. Acad. Dermatol. 2015, 73, 883–885. [Google Scholar] [CrossRef] [PubMed]
- Halder, R.M.; Chappell, J.L. Vitiligo Update. Semin. Cutan. Med. Surg. 2009, 28, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Namazi, M.R. Neurogenic dysregulation, oxidative stress, autoimmunity, and melanocytorrhagy in vitiligo: Can they be interconnected? Pigment Cell Res. 2007, 20, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Ralf Paus, L.; Schallreuter, K.U.; Bahadoran, P.; Picardo, M.; Slominski, A.; Elassiuty, Y.E.; Kemp, E.H.; Giachino, C.; Liu, J.B.; Luiten, R.M.; et al. Vitiligo pathogenesis: Autoimmune disease, genetic defect, excessive reactive oxygen species, calcium imbalance, or what else? Exp. Dermatol. 2008, 17, 139–140. [Google Scholar] [CrossRef]
- Schallreuter, K.U.; Kothari, S.; Chavan, B.; Spencer, J.D. Regulation of melanogenesis—Controversies and new concepts. Exp. Dermatol. 2008, 17, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Sandoval-Cruz, M.; García-Carrasco, M.; Sánchez-Porras, R.; Mendoza-Pinto, C.; Jiménez-Hernández, M.; Munguía-Realpozo, P.; Ruiz-Argüelles, A. Immunopathogenesis of vitiligo. Autoimmun. Rev. 2011, 10, 762–765. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Molina, M.M.; Muñoz-Muñoz, J.L.; Garcia-Molina, F.; García-Ruiz, P.A.; Garcia-Canovas, F. Action of Tyrosinase on Ortho-Substituted Phenols: Possible Influence on Browning and Melanogenesis. J. Agric. Food Chem. 2012, 60, 6447–6453. [Google Scholar] [CrossRef] [PubMed]
- Ismaya, W.T.; Rozeboom, H.J.; Weijn, A.; Mes, J.J.; Fusetti, F.; Wichers, H.J.; Dijkstra, B.W. Crystal structure of Agaricus bisporus mushroom tyrosinase: Identity of the tetramer subunits and interaction with tropolone. Biochemistry 2011, 50, 5477–5486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vachtenheim, J.; Borovanský, J. “Transcription physiology” of pigment formation in melanocytes: Central role of MITF. Exp. Dermatol. 2010, 19, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Pillaiyar, T.; Manickam, M.; Jung, S.H. Downregulation of melanogenesis: Drug discovery and therapeutic options. Drug Discov. Today 2017, 22, 282–298. [Google Scholar] [CrossRef] [PubMed]
- Del Marmol, V.; Beermann, F. Tyrosinase and related proteins in mammalian pigmentation. FEBS Lett. 1996, 381, 165–168. [Google Scholar] [CrossRef]
- Liu, J.J.; Fisher, D.E. Lighting a path to pigmentation: Mechanisms of MITF induction by UV. Pigment Cell Melanoma Res. 2010, 23, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Sun, Z.; Ye, Y.; Han, X.; Song, X.; Liu, S. Psoralen inhibits bone metastasis of breast cancer in mice. Fitoterapia 2013, 91, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Harzallah, A.; Bhouri, A.M.; Amri, Z.; Soltana, H.; Hammami, M. Phytochemical content and antioxidant activity of different fruit parts juices of three figs (Ficus carica L.) varieties grown in Tunisia. Ind. Crops Prod. 2016, 83, 255–267. [Google Scholar] [CrossRef]
- Wang, Y.F.; Liu, Y.N.; Xiong, W.; Yan, D.M.; Zhu, Y.; Gao, X.M.; Xu, Y.T.; Qi, A.D. A UPLC–MS/MS method for in vivo and in vitro pharmacokinetic studies of psoralenoside, isopsoralenoside, psoralen and isopsoralen from Psoralea corylifolia extract. J. Ethnopharmacol. 2014, 151, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Pei, T.; Zheng, C.; Huang, C.; Chen, X.; Guo, Z.; Fu, Y.; Liu, J.; Wang, Y. Systematic understanding the mechanisms of vitiligo pathogenesis and its treatment by Qubaibabuqi formula. J. Ethnopharmacol. 2016, 190, 272–287. [Google Scholar] [CrossRef] [PubMed]
- Jois, H.S.; Manjunath, B.L.; Venkatarao, S. Chemical examination of the seeds of Psoralea corylifolia. J. Indian Chem. Soc. 1933, 10, 41–43. [Google Scholar]
- Späth, E. Constitution and synthesis of foeniculin. Ber. Deut. Chem. Ges. 1937, 70, 83–87. [Google Scholar] [CrossRef]
- Fowucs, W.L.; Griffith, D.G.; Oginsky, E.L. Photosensitization of bacteria by furocoumarins and related compounds. Nature 1958, 181, 571–572. [Google Scholar]
- El Mofty, A.M. Vitiligo and Psoralens; Pergamon Press: Oxford, UK, 1968. [Google Scholar]
- Fitzpatrick, T.B.; Parrish, J.A.; Pathak, M.A. Phototherapy of Vitiligo (Idiopatic Leukodermia) in Sunlight and Man; Tokyo University Press: Tokyo, Japan, 1974. [Google Scholar]
- Parrish, J.A.; Fitzpatrick, T.B.; Shea, C.; Pathak, M.A. Photochemotherapy of vitiligo: Use of orally administered psoralens and a high-intensity long-wave ultraviolet light system. Arch. Dermatol. 1976, 112, 1531–1534. [Google Scholar] [CrossRef] [PubMed]
- Felsten, L.M.; Alikhan, A.; Petronic-Rosic, V. Vitiligo: A comprehensive overview: Part II: Treatment options and approach to treatment. J. Am. Acad. Dermatol. 2011, 65, 493–514. [Google Scholar] [CrossRef] [PubMed]
- Tippisetty, S.; Goudi, D.; Mohammed, A.W.; Jahan, P. Repair efficiency and PUVA therapeutic response variation in patients with vitiligo. Toxicology 2013, 27, 438–440. [Google Scholar] [CrossRef] [PubMed]
- Whitton, M.E.; Ashcroft, D.M.; González, U. Therapeutic interventions for vitiligo. J. Am. Acad. Dermatol. 2008, 59, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Aisa, H.A. Upregulation of Melanogenesis and Tyrosinase Activity: Potential Agents for Vitiligo. Molecules 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Li, G.; Tuerxuntayi, A.; Aisa, H.A. Synthesis and Bioactivity of New Chalcone Derivatives as Potential Tyrosinase Activator Based on the Click Chemistry. Chin. J. Chem. 2015, 33, 486–494. [Google Scholar] [CrossRef]
- Niu, C.; Pang, G.X.; Li, G.; Dou, J.; Nie, L.F.; Himit, H.; Kabas, M.; Aisa, H.A. Synthesis and biological evaluation of furocoumarin derivatives on melanin synthesis in murine B16 cells for the treatment of vitiligo. Biorg. Med. Chem. 2016, 24, 5960–5968. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Tuerxuntayi, A.; Li, G.; Kabas, M.; Dong, C.Z.; Aisa, H.A. Design, synthesis and bioactivity of chalcones and its analogues. Chin. Chem. Lett. 2017, 28, 1533–1538. [Google Scholar] [CrossRef]
- Niu, C.; Yin, L.; Nie, L.F.; Dou, J.; Zhao, J.Y.; Li, G.; Aisa, H.A. Synthesis and bioactivity of novel isoxazole chalcone derivatives on tyrosinase and melanin synthesis in murine B16 cells for the treatment of vitiligo. Biorg. Med. Chem. 2016, 24, 5440–5448. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Li, G.; Madina, K.; Aisa, H.A. Synthesis and activity on tyrosinase of novel chalcone derivatives. Chem. J. Chin. Univ. 2014, 35, 1204–1211. [Google Scholar] [CrossRef]
- Zhang, B.L.; Fan, C.Q.; Dong, L.; Wang, F.D.; Yue, J.M. Structural modification of a specific antimicrobial lead against Helicobacter pylori discovered from traditional Chinese medicine and a structure–activity relationship study. Eur. J. Med. Chem. 2010, 45, 5258–5264. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zou, B.; Chen, Z.; Pan, Y.; Wang, H.; Liang, H.; Yi, X. Synthesis and antioxidant activities of novel 4-Schiff base-7-benzyloxy-coumarin derivatives. Bioorg. Med. Chem. Lett. 2011, 21, 6811–6815. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Peng, Q.; Shao, J.L.; Liu, X.F.; Huang, Z.S.; Pu, X.Z.; Ma, L.; Li, Y.M.; Chan, A.S.C.; Gu, L.Q. Synthesis and biological evaluation of functionalized coumarins as acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2005, 40, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Schönleber, R.O.; Bendig, J.; Hagen, V.; Giese, B. Rapid photolytic release of cytidine 5′-diphosphate from a coumarin derivative: A new tool for the investigation of ribonucleotide reductases. Biorg. Med. Chem. 2002, 10, 97–101. [Google Scholar] [CrossRef]
- Fournier, L.; Aujard, I.; Le Saux, T.; Maurin, S.; Beaupierre, S.; Baudin, J.B.; Jullien, L. Coumarinylmethyl Caging Groups with Redshifted Absorption. Chem. Eur. J. 2013, 19, 17494–17507. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Kim, J.S.; Woo, J.T.; Lee, I.S.; Cha, B.Y. Hyperpigmentation mechanism of methyl 3,5-di-caffeoylquinate through activation of p38 and MITF induction of tyrosinase. Acta Biochim. Biophys. Sin. 2015, 47, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Bellei, B.; Flori, E.; Izzo, E.; Maresca, V.; Picardo, M. GSK3β inhibition promotes melanogenesis in mouse B16 melanoma cells and normal human melanocytes. Cell. Signal. 2008, 20, 1750–1761. [Google Scholar] [CrossRef] [PubMed]
- Bellei, B.; Pitisci, A.; Catricalà, C.; Larue, L.; Picardo, M. Wnt/β-catenin signaling is stimulated by α-melanocyte-stimulating hormone in melanoma and melanocyte cells: Implication in cell differentiation. Pigment Cell Melanoma Res. 2011, 24, 309–325. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Takemoto, C.; Kobayashi, I.; Watanabe, A.; Nobukuni, Y.; Fisher, D.E.; Tachibana, M. Ser298 of MITF, a mutation site in Waardenburg syndrome type 2, is a phosphorylation site with functional significance. Hum. Mol. Genet. 2000, 9, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Debowska, R.; Pasikowska, M.; Bazela, K.; Szczepanowska, J.; Ciescinska, C.; Vincent, C.; Napierala, M.; Szewczyk, A.; Lewandowska, M.; Eris, I. Plant flavonoid activating potassium channels—Naringenin for vitiligo skin care. J. Investig. Dermatol. 2016, 136. [Google Scholar] [CrossRef]
- Lin, J.J.; Yu, J.; Zhao, J.Y.; Zhang, K.; Zheng, J.C.; Wang, J.L.; Huang, C.H.; Zhang, J.R.; Yan, X.J.; Gerwick, W.H.; et al. Fucoxanthin, a Marine Carotenoid, Attenuates beta-Amyloid Oligomer-Induced Neurotoxicity Possibly via Regulating the PI3K/Akt and the ERK Pathways in SH-SY5Y Cells. Oxid. Med. Cell. Longev. 2017, 2017, 6792543. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.W.; Wang, Z.; Liu, C.L.; Shen, H.T.; Chen, Z.Q.; Yin, J.; Zuo, G.; Duan, X.C.; Li, H.Y.; Chen, G. Pramipexole-Induced Hypothermia Reduces Early Brain Injury via PI3K/AKT/GSK3β pathway in Subarachnoid Hemorrhage rats. Sci. Rep. 2016, 6, 23817. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.J.; Song, D.D.; Wang, M.; Chen, K.Y.; Zhang, T.Z. Alpha7 nicotinic acetylcholine receptor agonist attenuates the cerebral injury in a rat model of cardiopulmonary bypass by activating the Akt/GSK3beta pathway. Mol. Med. Rep. 2017, 16, 7979–7986. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhao, L.H. Calycosin ameliorates diabetes-induced cognitive impairments in rats by reducing oxidative stress via the PI3K/Akt/GSK-3β signaling pathway. Biochem. Biophys. Res. Commun. 2016, 473, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Zong, Z.K.; Pang, H.; Yu, R.T.; Jiao, Y.Q. PCDH8 inhibits glioma cell proliferation by negatively regulating the AKT/GSK3beta/beta-catenin signaling pathway. Oncol. Lett. 2017, 14, 3357–3362. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Park, S.Y.; Park, O.J.; Kim, Y.M. Curcumin suppresses migration and proliferation of Hep3B hepatocarcinoma cells through inhibition of the Wnt signaling pathway. Mol. Med. Rep. 2013, 8, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Yam, J.W.; Wong, C.M.; Ng, I.O. Molecular and functional genetics of hepatocellular carcinoma. Front. Biosci. 2010, 2, 117–134. [Google Scholar]
- Fukuda, K.; Hibiya, Y.; Mutoh, M.; Koshiji, M.; Akao, S.; Fujiwara, H. Inhibition by berberine of cyclooxygenase-2 transcriptional activity in human colon cancer cells. J. Ethnopharmacol. 1999, 66, 227–233. [Google Scholar] [CrossRef]
- Kong, W.J.; Wei, J.; Abidi, P.; Lin, M.H.; Inaba, S.; Li, C.; Wang, Y.L.; Wang, Z.Z.; Si, S.Y.; Pan, H.N.; et al. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat. Med. 2004, 10, 1344. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yin, J.H.; Gao, H.F.; Xu, L.X.; Wang, Y.; Xu, L.; Li, M. Berberine improves insulin sensitivity by inhibiting fat store and adjusting adipokines profile in human preadipocytes and metabolic syndrome patients. Evid.-Based Complement. Alternat. Med. 2012, 2012, 363845. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jung, K.; Kim, Y.S.; Park, D. Diosgenin inhibits melanogenesis through the activation of phosphatidylinositol-3-kinase pathway (PI3K) signaling. Life Sci. 2007, 81, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.H.; Tsai, Y.T.; Yen, M.H.; Lin, C.C.; Liang, C.J.; Yang, T.H.; Lee, C.W.; Yen, F.L. Norartocarpetin from a folk medicine Artocarpus communis plays a melanogenesis inhibitor without cytotoxicity in B16F10 cell and skin irritation in mice. BMC Complement. Altern. Med. 2013, 13, 348. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Kim, D.H.; Oh, I.Y.; Kim, S.Y.; Lim, Y.Y.; Kim, H.M.; Kim, Y.H.; Choi, Y.M.; Kim, S.E.; Kim, B.J.; et al. Inhibitory Effects of Saururi chinensis Extracts on Melanin Biosynthesis in B16F10 Melanoma Cells. Biol. Pharm. Bull. 2013, 36, 772–779. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, C.; Yin, L.; Aisa, H.A. Novel Furocoumarin Derivatives Stimulate Melanogenesis in B16 Melanoma Cells by Up-Regulation of MITF and TYR Family via Akt/GSK3β/β-Catenin Signaling Pathways. Int. J. Mol. Sci. 2018, 19, 746. https://doi.org/10.3390/ijms19030746
Niu C, Yin L, Aisa HA. Novel Furocoumarin Derivatives Stimulate Melanogenesis in B16 Melanoma Cells by Up-Regulation of MITF and TYR Family via Akt/GSK3β/β-Catenin Signaling Pathways. International Journal of Molecular Sciences. 2018; 19(3):746. https://doi.org/10.3390/ijms19030746
Chicago/Turabian StyleNiu, Chao, Li Yin, and Haji Akber Aisa. 2018. "Novel Furocoumarin Derivatives Stimulate Melanogenesis in B16 Melanoma Cells by Up-Regulation of MITF and TYR Family via Akt/GSK3β/β-Catenin Signaling Pathways" International Journal of Molecular Sciences 19, no. 3: 746. https://doi.org/10.3390/ijms19030746
APA StyleNiu, C., Yin, L., & Aisa, H. A. (2018). Novel Furocoumarin Derivatives Stimulate Melanogenesis in B16 Melanoma Cells by Up-Regulation of MITF and TYR Family via Akt/GSK3β/β-Catenin Signaling Pathways. International Journal of Molecular Sciences, 19(3), 746. https://doi.org/10.3390/ijms19030746