Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives
Abstract
1. Introduction
2. Methods
3. Results
4. Cartilage
4.1. Hyaline Cartilage
4.2. Fibrocartilage (Menisci)
4.3. Elastic Cartilage (Auricular Cartilage)
5. Bone
6. Skeletal Muscle
7. Tendon
8. Adipose Tissues
9. Heart
9.1. Myocardium
9.2. Pericardium
9.3. Heart Valves
10. Vessels
10.1. Arteries
10.2. Veins
11. Lung
12. Gingiva
13. Dental Pulp
14. Schneiderian Membrane
15. Intestine
16. Liver
17. Pancreas
18. Kidney
19. Bladder
20. Male Reproductive System
21. Female Reproductive System
22. Products of Childbirth: Umbilical Cord, Placenta and Amniotic Membrane
22.1. Umbilical Cord (Wharton’s Jelly)
22.2. Placenta
22.3. Amniotic Membrane
23. Cornea
24. Vocal Folds
25. Peripheral Nerves
26. Complex Composite Structures
27. Conclusions and Future Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- Gao, L.P.; Du, M.J.; Lv, J.J.; Schmull, S.; Huang, R.T.; Li, J. Use of human aortic extracellular matrix as a scaffold for construction of a patient-specific tissue engineered vascular patch. Biomed. Mater. 2017, 12, 065006. [Google Scholar] [CrossRef] [PubMed]
- Kartus, J.; Movin, T.; Karlsson, J. Donor-site morbidity and anterior knee problems after anterior cruciate ligament reconstruction using autografts. Arthroscopy 2001, 17, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Konofaos, P.; Ver Halen, J.P. Nerve repair by means of tubulization: Past, present, future. J. Reconstr. Microsurg. 2013, 29, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Tanaka, M. Designing Smart Biomaterials for Tissue Engineering. Int. J. Mol. Sci. 2017, 19, 17. [Google Scholar] [CrossRef]
- Bryksin, A.V.; Brown, A.C.; Baksh, M.M.; Finn, M.G.; Barker, T.H. Learning from nature—Novel synthetic biology approaches for biomaterial design. Acta Biomater. 2014, 10, 1761–1769. [Google Scholar] [CrossRef] [PubMed]
- Dan, P.; Velot, É.; Francius, G.; Menu, P.; Decot, V. Human-derived extracellular matrix from Wharton’s jelly: An untapped substrate to build up a standardized and homogeneous coating for vascular engineering. Acta Biomater. 2017, 48, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Schenke-Layland, K.; Nerem, R.M. In vitro human tissue models—moving towards personalized regenerative medicine. Adv. Drug. Deliv. Rev. 2011, 63, 195–196. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.W.; Shin, S.C.; Jang, J.Y.; Park, H.Y.; Lee, J.C.; Wang, S.G.; Lee, B.J. Decellularization of Human Nasal Septal Cartilage for the Novel Filler Material of Vocal Fold Augmentation. J. Voice 2017, 31, 127.e1–127.e6. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.H.; Enayati, M.; Grasl, C.; Walter, I.; Budinsky, L.; Zebic, G.; Kaun, C.; Wagner, A.; Kratochwill, K.; Redl, H.; et al. Acellular vascular matrix grafts from human placenta chorion: Impact of ECM preservation on graft characteristics, protein composition and in vivo performance. Biomaterials 2018, 177, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Macchi, V.; Porzionato, A.; Stecco, C.; Tiengo, C.; Parenti, A.; Cestrone, A.; De Caro, R. Body parts removed during surgery: A useful training source. Anat. Sci. Educ. 2011, 4, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Porzionato, A.; Macchi, V.; Stecco, C.; Mazzi, A.; Rambaldo, A.; Sarasin, G.; Parenti, A.; Scipioni, A.; De Caro, R. Quality management of Body Donation Program at the University of Padova. Anat. Sci. Educ. 2012, 5, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Porzionato, A.; Macchi, V.; Stecco, C.; De Caro, R. The body donation program of the University of Padua: Organizing an anatomical biobank for medical education. In New Insights on Biobanks; Caenazzo, L., Ed.; CLEUP: Padova, Italy, 2013; pp. 155–171. ISBN 9788867871216. [Google Scholar]
- Porzionato, A.; Sfriso, M.M.; Macchi, V.; Rambaldo, A.; Lago, G.; Lancerotto, L.; Vindigni, V.; De Caro, R. Decellularized omentum as novel biologic scaffold for reconstructive surgery and regenerative medicine. Eur. J. Histochem. 2013, 24, e4. [Google Scholar] [CrossRef] [PubMed]
- Porzionato, A.; Sfriso, M.M.; Pontini, A.; Macchi, V.; Petrelli, L.; Pavan, P.G.; Natali, A.N.; Bassetto, F.; Vindigni, V.; De Caro, R. Decellularized Human Skeletal Muscle as Biologic Scaffold for Reconstructive Surgery. Int. J. Mol. Sci. 2015, 16, 14808–14831. [Google Scholar] [CrossRef] [PubMed]
- Porzionato, A.; Sfriso, M.M.; Pontini, A.; Macchi, V.; Buompensiere, M.I.; Petrelli, L.; Bassetto, F.; Vindigni, V.; De Caro, R. Development of small-diameter vascular grafts through decellularization of human blood vessels. J. Biomat. Tissue Eng. 2017, 7, 101–110. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Groen, W.M.; Diloksumpan, P.; van Weeren, P.R.; Levato, R.; Malda, J. From intricate to integrated: Biofabrication of articulating joints. J. Orthop. Res. 2017, 35, 2089–2097. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, B.; Mashayekhan, S. Fabrication of porous scaffolds with decellularized cartilage matrix for tissue engineering application. Biologicals 2017, 48, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Azhim, A.; Ono, T.; Fukui, Y.; Morimoto, Y.; Furukawa, K.; Ushida, T. Preparation of decellularized meniscal scaffolds using sonication treatment for tissue engineering. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2013, 2013, 6953–6956. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, J.; Zhang, Z.; Lou, K.; Zhang, Q.; Wang, S.; Ni, J.; Liu, W.; Fan, S.; Lin, X. Current advances in the development of natural meniscus scaffolds: Innovative approaches to decellularization and recellularization. Cell Tissue Res. 2017, 370, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Korpershoek, J.V.; de Windt, T.S.; Hagmeijer, M.H.; Vonk, L.A.; Saris, D.B. Cell-Based Meniscus Repair and Regeneration: At the Brink of Clinical Translation?: A Systematic Review of Preclinical Studies. Orthop. J. Sports. Med. 2017, 5, 2325967117690131. [Google Scholar] [CrossRef] [PubMed]
- Haykal, S.; Salna, M.; Waddell, T.K.; Hofer, S.O. Advances in tracheal reconstruction. Plast. Reconstr. Surg. Glob. Open 2014, 2, e178. [Google Scholar] [CrossRef] [PubMed]
- Graham, M.E.; Gratzer, P.F.; Bezuhly, M.; Hong, P. Development and characterization of decellularized human nasoseptal cartilage matrix for use in tissue engineering. Laryngoscope 2016, 126, 2226–2231. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Peng, J.; Guo, Q.; Huang, J.; Zhang, L.; Yao, J.; Yang, F.; Wang, S.; Xu, W.; Wang, A.; et al. A cartilage ECM-derived 3-D porous acellular matrix scaffold for in vivo cartilage tissue engineering with PKH26-labeled chondrogenic bone marrow-derived mesenchymal stem cells. Biomaterials 2008, 29, 2378–2387. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Lehmann, J.; van Osch, G.J.; Hildner, F.; Teuschl, A.; Monforte, X.; Miosga, D.; Heimel, P.; Priglinger, E.; Redl, H.; et al. Systematic Comparison of Protocols for the Preparation of Human Articular Cartilage for Use as Scaffold Material in Cartilage Tissue Engineering. Tissue Eng. Part C Methods 2016, 22, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Sandmann, G.H.; Eichhorn, S.; Vogt, S.; Adamczyk, C.; Aryee, S.; Hoberg, M.; Milz, S.; Imhoff, A.B.; Tischer, T. Generation and characterization of a human acellular meniscus scaffold for tissue engineering. J. Biomed. Mater. Res. A 2009, 91, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Baiguera, S.; Gonfiotti, A.; Jaus, M.; Comin, C.E.; Paglierani, M.; Del Gaudio, C.; Bianco, A.; Ribatti, D.; Macchiarini, P. Development of bioengineered human larynx. Biomaterials 2011, 32, 4433–4442. [Google Scholar] [CrossRef] [PubMed]
- Macchiarini, P.; Jungebluth, P.; Go, T.; Asnaghi, M.A.; Rees, L.E.; Cogan, T.A.; Dodson, A.; Martorell, J.; Bellini, S.; Parnigotto, P.P.; et al. Clinical transplantation of a tissue-engineered airway. Lancet 2008, 372, 2023–2030. [Google Scholar] [CrossRef]
- Butler, C.R.; Hynds, R.E.; Crowley, C.; Gowers, K.H.; Partington, L.; Hamilton, N.J.; Carvalho, C.; Platé, M.; Samuel, E.R.; Burns, A.J.; et al. Vacuum-assisted decellularization: An accelerated protocol to generate tissue-engineered human tracheal scaffolds. Biomaterials 2017, 124, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.J.; De Coppi, P.; Speggiorin, S.; Roebuck, D.; Butler, C.R.; Samuel, E.; Crowley, C.; McLaren, C.; Fierens, A.; Vondrys, D.; et al. Stem-cell-based, tissue engineered tracheal replacement in a child: A 2-year follow-up study. Lancet 2012, 380, 994–1000. [Google Scholar] [CrossRef]
- Hamilton, N.J.; Kanani, M.; Roebuck, D.J.; Hewitt, R.J.; Cetto, R.; Culme-Seymour, E.J.; Toll, E.; Bates, A.J.; Comerford, A.P.; McLaren, C.A.; et al. Tissue-Engineered Tracheal Replacement in a Child: A 4-Year Follow-Up Study. Am. J. Transplant. 2015, 15, 2750–2777. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.; Koerber, L.; Elsaesser, A.F.; Goldberg-Bockhorn, E.; Seitz, A.M.; Dürselen, L.; Ignatius, A.; Walther, P.; Breiter, R.; Rotter, N. Decellularized cartilage matrix as a novel biomatrix for cartilage tissue-engineering applications. Tissue Eng. Part A 2012, 18, 2195–2209. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, A.J.; Converse, G.L.; Hopkins, R.A.; Detamore, M.S. The bioactivity of cartilage extracellular matrix in articular cartilage regeneration. Adv. Healthc. Mater. 2015, 4, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.W.; Solorio, L.D.; Alsberg, E. Decellularized tissue and cell-derived extracellular matrices as scaffolds for orthopaedic tissue engineering. Biotechnol. Adv. 2014, 32, 462–484. [Google Scholar] [CrossRef] [PubMed]
- Kiyotake, E.A.; Beck, E.C.; Detamore, M.S. Cartilage extracellular matrix as a biomaterial for cartilage regeneration. Ann. N. Y. Acad. Sci. 2016, 1383, 139–159. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Dalzoppo, D.; Lora, S.; Sartore, L.; Folin, M.; Parnigotto, P.P.; Grandi, C. Tailored PVA/ECM scaffolds for cartilage regeneration. Biomed. Res. Int. 2014, 2014, 762189. [Google Scholar] [CrossRef] [PubMed]
- Gonfiotti, A.; Jaus, M.O.; Barale, D.; Baiguera, S.; Comin, C.; Lavorini, F.; Fontana, G.; Sibila, O.; Rombolà, G.; Jungebluth, P.; et al. The first tissue-engineered airway transplantation: 5-year follow-up results. Lancet 2014, 383, 238–244. [Google Scholar] [CrossRef]
- Baiguera, S.; Jungebluth, P.; Burns, A.; Mavilia, C.; Haag, J.; Coppi, D.E.; Macchiarini, P. Tissue engineered human tracheas for in vivo implantation. Biomaterials 2010, 31, 8931–8938. [Google Scholar] [CrossRef] [PubMed]
- Tint, D.; Stabler, C.T.; Hanifi, A.; Yousefi, F.; Linkov, G.; Hy, K.; Soliman, A.M.S.; Pleshko, N. Spectroscopic Analysis of Human Tracheal Tissue during Decellularization. Otolaryngol. Head Neck Surg. 2018, 194599818806271. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Park, D.Y.; Cho, Y.H.; Chang, J.W.; Choi, J.W.; Park, J.K.; Min, B.H.; Shin, Y.S.; Kim, C.H. Cultured chondrocyte and porcine cartilage-derived substance (PCS) construct as a possible dorsal augmentation material in rhinoplasty: A preliminary animal study. J. Plast. Reconstr. Aesthet. Surg. 2015, 68, 659–666. [Google Scholar] [CrossRef] [PubMed]
- San-Marina, S.; Sharma, A.; Voss, S.G.; Janus, J.R.; Hamilton, G.S., 3rd. Assessment of Scaffolding Properties for Chondrogenic Differentiation of Adipose-Derived Mesenchymal Stem Cells in Nasal Reconstruction. JAMA Facial Plast. Surg. 2017, 19, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Oseni, A.; Crowley, C.; Lowdell, M.; Birchall, M.; Butler, P.E.; Seifalian, A.M. Advancing nasal reconstructive surgery: The application of tissue engineering technology. J. Tissue Eng. Regen. Med. 2012, 6, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Kohl, B.; Kokozidou, M.; Arens, S.; Schulze-Tanzil, G. Establishment of a Cytocompatible Cell-Free Intervertebral Disc Matrix for Chondrogenesis with Human Bone Marrow-Derived Mesenchymal Stromal Cells. Cells Tissues Organs 2016, 201, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Godkin, O.; Schulze-Tanzil, G. The Challenge in Using Mesenchymal Stromal Cells for Recellularization of Decellularized Cartilage. Stem Cell Rev. 2017, 13, 50–67. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.O.; Han, J.W.; Kim, J.M.; Cho, H.J.; Park, C.; Lee, N.; Kim, D.W.; Yoon, Y.S. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ. Res. 2011, 108, 1340–1347. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A. Endogenous and exogenous stem cells: A role in lung repair and use in airway tissue engineering and transplantation. J. Biomed. Sci. 2010, 17, 92. [Google Scholar] [CrossRef] [PubMed]
- Batioglu-Karaaltin, A.; Karaaltin, M.V.; Ovali, E.; Yigit, O.; Kongur, M.; Inan, O.; Bozkurt, E.; Cansiz, H. In vivo tissue-engineered allogenic trachea transplantation in rabbits: A preliminary report. Stem Cell Rev. 2015, 11, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.H.; Su, C.H.; Lin, S.E.; Tseng, H. Preliminary experiences in trachea scaffold tissue engineering with segmental organ decellularization. Laryngoscope 2016, 126, 2520–2527. [Google Scholar] [CrossRef] [PubMed]
- Go, T.; Jungebluth, P.; Baiguero, S.; Asnaghi, A.; Martorelli, J.; Ostertag, H.; Mantero, S.; Birchall, M.; Bader, A.; Macchiarini, P. Both epithelial cells and mesenchymal stem cell-derived chondrocytes contribute to the survival of tissue-engineered airway transplants in pigs. J. Thorac. Cardiovasc. Surg. 2010, 139, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.W.; Murphy, S.V.; Feng, X.; Wright, S.C., Jr. Tracheal reconstruction in a canine model. Otolaryngol. Head Neck Surg. 2014, 150, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Minehara, H.; Urabe, K.; Naruse, K.; Mehlhorn, A.T.; Uchida, K.; Sudkamp, N.P.; Itoman, M. A new technique for seeding chondrocytes onto solvent-preserved human meniscus using the chemokinetic effect of recombinant human bone morphogenetic protein-2. Cell Tissue Bank. 2011, 12, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, R.C.; Charoenpanich, A.; Vaughn, C.E.; Griffith, E.H.; Fisher, M.B.; Cole, J.H.; Spang, J.T.; Loboa, E.G. Enhanced cellular infiltration of human adipose-derived stem cells in allograft menisci using a needle-punch method. J. Orthop. Surg. Res. 2016, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Idrees, E.; Szojka, A.R.A.; Andrews, S.H.J.; Kunze, M.; Mulet-Sierra, A.; Jomha, N.M.; Adesida, A.B. Chondrogenic differentiation of synovial fluid mesenchymal stem cells on human meniscus-derived decellularized matrix requires exogenous growth factors. Acta Biomater. 2018, 80, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Utomo, L.; Pleumeekers, M.M.; Nimeskern, L.; Nürnberger, S.; Stok, K.S.; Hildner, F.; van Osch, G.J. Preparation and characterization of a decellularized cartilage scaffold for ear cartilage reconstruction. Biomed. Mater. 2015, 10, 015010. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Griffin, M.; Naik, A.; Szarko, M.; Butler, P.E.M. Optimising the decellularization of human elastic cartilage with trypsin for future use in ear reconstruction. Sci. Rep. 2018, 8, 3097. [Google Scholar] [CrossRef] [PubMed]
- Duisit, J.; Amiel, H.; Wuthrich, T.; Taddeo, A.; Dedriche, A.; Destoop, V.; Pardoen, T.; Bouzin, C.; Joris, V.; Magee, D.; et al. Perfusion-decellularization of human ear grafts enables ECM-based scaffolds for auricular vascularized composite tissue engineering. Acta biomater. 2018, 73, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Elkins, R.C.; Dawson, P.E.; Goldstein, S.; Walsh, S.P.; Black, K.S. Decellularized human valve allografts. Ann. Thorac. Surg. 2001, 71, S428–S432. [Google Scholar] [CrossRef]
- Da Costa, F.D.; Dohmen, P.M.; Duarte, D.; von Glenn, C.; Lopes, S.V.; Filho, H.H.; da Costa, M.B.; Konertz, W. Immunological and echocardiographic evaluation of decellularized versus cryopreserved allografts during the Ross operation. Eur. J. Cardio-Thorac. Surg. 2005, 27, 572–578. [Google Scholar] [CrossRef]
- Zehr, K.J.; Yagubyan, M.; Connolly, H.M.; Nelson, S.M.; Schaff, H.V. Aortic root replacement with a novel decellularized cryopreserved aortic homograft: Postoperative immunoreactivity and early results. J. Thorac. Cardiovasc. Surg. 2005, 130, 1010–1015. [Google Scholar] [CrossRef] [PubMed]
- Cebotari, S.; Lichtenberg, A.; Tudorache, I.; Hilfiker, A.; Mertsching, H.; Leyh, R.; Breymann, T.; Kallenbach, K.; Maniuc, L.; Batrinac, A.; et al. Clinical application of tissue engineered human heart valves using autologous progenitor cells. Circulation 2006, 114, I132–I137. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.; Dohmen, P.; Vieira, E.; Lopes, S.V.; Colatusso, C.; Pereira, E.W.; Matsuda, C.N.; Cauduro, S. Ross Operation with decellularized pulmonary allografts: Medium-term results. Rev. Bras. Cir. Cardiovasc. 2007, 22, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.W.; Ruzmetov, M.; Rodefeld, M.D.; Turrentine, M.W. Right ventricular outflow tract reconstruction in Ross patients: Does the homograft fare better? Ann. Thorac. Surg. 2008, 86, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, F.D.; Costa, A.C.; Prestes, R.; Domanski, A.C.; Balbi, E.M.; Ferreira, A.D.; Lopes, S.V. The early and midterm function of decellularized aortic valve allografts. Ann. Thorac. Surg. 2010, 90, 1854–1860. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.W.; Ruzmetov, M.; Eltayeb, O.; Rodefeld, M.D.; Turrentine, M.W. Performance of SynerGraft decellularized pulmonary homograft in patients undergoing a Ross procedure. Ann. Thorac. Surg. 2011, 91, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Cebotari, S.; Tudorache, I.; Ciubotaru, A.; Boethig, D.; Sarikouch, S.; Goerler, A.; Lichtenberg, A.; Cheptanaru, E.; Barnaciuc, S.; Cazacu, A.; et al. Use of fresh decellularized allografts for pulmonary valve replacement may reduce the reoperation rate in children and young adults: Early report. Circulation 2011, 124, S115–S123. [Google Scholar] [CrossRef] [PubMed]
- Kneib, C.; von Glehn, C.Q.; Costa, F.D.; Costa, M.T.; Susin, M.F. Evaluation of humoral immune response to donor HLA after implantation of cellularized versus decellularized human heart valve allografts. Tissue Antigens 2012, 80, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Sarikouch, S.; Horke, A.; Tudorache, I.; Beerbaum, P.; Westhoff-Bleck, M.; Boethig, D.; Repin, O.; Maniuc, L.; Ciubotaru, A.; Haverich, A.; et al. Decellularized fresh homografts for pulmonary valve replacement: A decade of clinical experience. Eur. J. Cardiothorac. Surg. 2016, 50, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Bibevski, S.; Ruzmetov, M.; Fortuna, R.S.; Turrentine, M.W.; Brown, J.W.; Ohye, R.G. Performance of SynerGraft Decellularized Pulmonary Allografts Compared with Standard Cryopreserved Allografts: Results From Multiinstitutional Data. Ann. Thorac. Surg. 2017, 103, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, H.; Ueno, T.; Taira, M.; Toda, K.; Kuratani, T.; Sawa, Y. Application of a Fresh Decellularized Pulmonary Allograft for Pulmonary Valve Replacement in Japan. Circ. J. 2018, 82, 1526–1533. [Google Scholar] [CrossRef] [PubMed]
- Lofland, G.K.; O’Brien, J.E., Jr.; Gandy, K.L.; Dennis, P.A.; Marshall, J.A.; Mastbergen, R.K.; Hopkins, R.A. Initial pediatric cardiac experience with decellularized allograft patches. Ann. Thorac. Surg. 2012, 93, 968–971. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, R.A.; Lofland, G.K.; Marshall, J.; Connelly, D.; Acharya, G.; Dennis, P.; Stroup, R.; McFall, C.; O’Brien, J.E., Jr. Pulmonary arterioplasty with decellularized allogeneic patches. Ann. Thorac. Surg. 2014, 97, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Olausson, M.; Kuna, V.K.; Travnikova, G.; Backdahl, H.; Patil, P.B.; Saalman, R.; Borg, H.; Jeppsson, A.; Sumitran-Holgersson, S. In Vivo Application of Tissue-Engineered Veins Using Autologous Peripheral Whole Blood: A Proof of Concept Study. EBioMedicine 2014, 1, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Madden, R.; Lipkowitz, G.; Benedetto, B.; Kurbanov, A.; Miller, M.; Bow, L. Decellularized cadaver vein allografts used for hemodialysis access do not cause allosensitization or preclude kidney transplantation. Am. J. Kidney Dis. 2002, 40, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Atala, A.; Guzman, L.; Retik, A.B. A novel inert collagen matrix for hypospadias repair. J. Urol. 1999, 162, 1148–1151. [Google Scholar] [CrossRef]
- El-Kassaby, A.W.; Retik, A.B.; Yoo, J.J.; Atala, A. Urethral stricture repair with an off-the-shelf collagen matrix. J. Urol. 2003, 169, 170–173. [Google Scholar] [CrossRef]
- Atala, A.; Bauer, S.B.; Soker, S.; Yoo, J.J.; Retik, A.B. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 2006, 367, 1241–1246. [Google Scholar] [CrossRef]
- El-Kassaby, A.; AbouShwareb, T.; Atala, A. Randomized comparative study between buccal mucosal and acellular bladder matrix grafts in complex anterior urethral strictures. J. Urol. 2008, 179, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- Letendre, S.; LaPorta, G.; O’Donnell, E.; Dempsey, J.; Leonard, K. Pilot Trial of Biovance Collagen-Based Wound Covering for Diabetic Ulcers. Adv. Skin. Wound Care 2009, 22, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Alio Del Barrio, J.L.; El Zarif, M.; Azaar, A.; Makdissy, N.; Khalil, C.; Harb, W.; El Achkar, I.; Jawad, Z.A.; de Miguel, M.P.; Alio, J.L. Corneal Stroma Enhancement with Decellularized Stromal Laminas With or Without Stem Cell Recellularization for Advanced Keratoconus. Am. J. Ophthalmol. 2018, 186, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Ebraheim, N.A.; Elgafy, H.; Xu, R. Bone-graft harvesting from iliac and fibular donor sites: Techniques and complications. J. Am. Acad. Orthop. Surg. 2001, 9, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Varettas, K.; Taylor, P. Bioburden assessment of banked bone used for allografts. Cell Tissue Bank. 2011, 12, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Richardson, S.M.; Eagle, M.J.; Rooney, P.; Board, T.; Hoyland, J.A. The use of a novel bone allograft wash process to generate a biocompatible, mechanically stable and osteoinductive biological scaffold for use in bone tissue engineering. J. Tissue Eng. Regen. Med. 2015, 9, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Board, T.N.; Rooney, P.; Eagle, M.J.; Richardson, S.M.; Hoyland, J.A. Human decellularized bone scaffolds from aged donors show improved osteoinductive capacity compared to young donor bone. PLoS ONE 2017, 12, e0177416. [Google Scholar] [CrossRef] [PubMed]
- Sladkova, M.; Cheng, J.J.; Palmer, M.; Chen, S.; Lin, C.; Xia, W.; Yu, Y.E.; Zhou, B.; Engqvist, H.; de Peppo, G.M. Comparison of Decellularized Cow and Human Bone for Engineering Bone Grafts with human iPS cells. Tissue Eng. Part A 2018. [Google Scholar] [CrossRef] [PubMed]
- Urciuolo, A.; De Coppi, P. Decellularized Tissue for Muscle Regeneration. Int. J. Mol. Sci. 2018, 19, 2392. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.; Terlouw, A.; Roberts, K.; Wolchok, J.C. The characterization of decellularized human skeletal muscle as a blueprint for mimetic scaffolds. J. Mater. Sci. Mater. Med. 2016, 27, 125. [Google Scholar] [CrossRef] [PubMed]
- Davari, H.R.; Rahim, M.B.; Tanideh, N.; Sani, M.; Tavakoli, H.R.; Rasekhi, A.R.; Monabati, A.; Koohi-Hosseinabadi, O.; Gholami, S. Partial replacement of left hemidiaphragm in dogs by either cryopreserved or decellularized heterograft patch. Interact. Cardiovasc. Thorac. Surg. 2016, 23, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Pridgen, B.; Woon, C.; Kim, M.; Thorfinn, J.; Lindsey, D.; Pham, H.; Chang, J. Flexor tendon tissue engineering: Acellularization of human flexor tendons with preservation of biomechanical properties and biocompatibility. Tissue Eng. Part C 2011, 17, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Woon, C.Y.L.; Pridgen, B.; Kraus, A.; Bari, S.; Pham, H.; Chang, J. Optimization of human tendon tissue engineering: Peracetic acid oxidation for enhanced reseeding of acellularized intrasynovial tendon. Plast. Reconstr. Surg. 2011, 127, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, S.S.; Woon, C.Y.; Kraus, A.; Megerle, K.; Choi, M.S.; Pridgen, B.C.; Pham, H.; Chang, J. Human flexor tendon tissue engineering: Decellularization of human flexor tendons reduces immunogenicity in vivo. Tissue Eng. Part A 2012, 18, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, T.; Fox, P.M.; Woon, C.Y.; Farnebo, S.J.; Bronstein, J.A.; Behn, A.; Pham, H.; Chang, J. Human flexor tendon tissue engineering: In vivo effects of stem cell reseeding. Plast. Reconstr. Surg. 2013, 132, 567e–576e. [Google Scholar] [CrossRef] [PubMed]
- Martinello, T.; Bronzini, I.; Volpin, A.; Vindigni, V.; Maccatrozzo, L.; Caporale, G.; Bassetto, F.; Patruno, M. Successful recellularization of human tendon scaffolds using adipose-derived mesenchymal stem cells and collagen gel. J. Tissue Eng. Regen. Med. 2014, 8, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Long, C.; Galvez, M.G.; Legrand, A.; Joubert, L.-M.; Wang, Z.; Chattopadhyay, A.; Chang, J.; Fox, P.M. Intratendinous Injection of Hydrogel for Reseeding Decellularized Human Flexor Tendons. Plast. Reconstr. Surg. 2017, 139, 1305e–1314e. [Google Scholar] [CrossRef] [PubMed]
- Le, W.; Cheah, A.E.-J.; Yao, J. Ex-vivo Tendon Repair Augmented with Bone Marrow Derived Mesenchymal Stem Cells Stimulated with Myostatin for Tenogenesis. J. Hand Surg. Asian-Pac. Vol. 2018, 23, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Whitlock, P.W.; Seyler, T.M.; Parks, G.D.; Ornelles, D.A.; Smith, T.L.; Van Dyke, M.E.; Poehling, G.G. A novel process for optimizing musculoskeletal allograft tissue to improve safety, ultrastructural properties, and cell infiltration. J. Bone Joint Surg. Am. 2012, 94, 1458–1467. [Google Scholar] [CrossRef] [PubMed]
- Chiou, G.J.; Crowe, C.; McGoldrick, R.; Hui, K.; Pham, H.; Chang, J. Optimization of an injectable tendon hydrogel: The effects of platelet-rich plasma and adipose-derived stem cells on tendon healing in vivo. Tissue Eng. Part A 2015, 21, 1579–1586. [Google Scholar] [CrossRef] [PubMed]
- Bronstein, J.A.; Woon, C.Y.L.; Farnebo, S.; Behn, A.W.; Schmitt, T.; Pham, H.; Castillo, A.B.; Chang, J. Physicochemical decellularization of composite flexor tendon-bone interface grafts. Plast. Reconstr. Surg. 2013, 132, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Fox, P.M.; Farnebo, S.; Lindsey, D.; Chang, J.; Schmitt, T.; Chang, J. Decellularized human tendon-bone grafts for composite flexor tendon reconstruction: A cadaveric model of initial mechanical properties. J. Hand Surg. Am. 2013, 38, 2323–2328. [Google Scholar] [CrossRef] [PubMed]
- Farnebo, S.; Woon, C.Y.; Schmitt, T.; Joubert, L.M.; Kim, M.; Pham, H.; Chang, J. Design and characterization of an injectable tendon hydrogel: A novel scaffold for guided tissue regeneration in the musculoskeletal system. Tissue Eng. Part A 2014, 20, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; John, T.M.; Gosiewska, A.; Buensuceso, C.S.; Colter, D.C.; Seyda, A.; Shissias, S.R. Decellularized Omentum Matrix and Uses Thereof. U.S. Patent 20140271784A1, 18 September 2014. [Google Scholar]
- Soffer-Tsur, N.; Shevach, M.; Shapira, A.; Peer, D.; Dvir, T. Optimizing the biofabrication process of omentum-based scaffolds for engineering autologous tissues. Biofabrication 2014, 6, 035023. [Google Scholar] [CrossRef] [PubMed]
- Omidi, E.; Fuetterer, L.; Reza Mousavi, S.; Armstrong, R.C.; Flynn, L.E.; Samani, A. Characterization and assessment of hyperelastic and elastic properties of decellularized human adipose tissues. J. Biomech. 2014, 47, 3657–3663. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.A.; Muir, L.A.; Washabaugh, A.R.; Neeley, C.K.; Chen, S.Y.; Flesher, C.G.; Vorwald, J.; Finks, J.F.; Ghaferi, A.A.; Mulholland, M.W.; et al. Diabetes-Specific Regulation of Adipocyte Metabolism by the Adipose Tissue Extracellular Matrix. J. Clin. Endocrinol. Metab. 2017, 102, 1032–1043. [Google Scholar] [CrossRef] [PubMed]
- Flynn, L.E. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials 2010, 31, 4715–4724. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, B.S.; Kim, J.Y.; Kim, J.D.; Choi, Y.C.; Yang, H.J.; Park, K.; Lee, H.Y.; Cho, Y.W. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J. Biomed. Mater. Res. A 2011, 97, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Young, D.A.; Ibrahim, D.O.; Hu, D.; Christman, K.L. Injectable hydrogel scaffold from decellularized human lipoaspirate. Acta Biomater. 2011, 7, 1040–1049. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.E.; Flynn, L.E. Design and characterization of tissue-specific extracellular matrix-derived microcarriers. Tissue Eng. Part C Methods 2012, 18, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.E.; Yu, C.; Bianco, J.; Watkins, J.F.; Flynn, L.E. The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials 2012, 33, 4490–4499. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.; Nahas, Z.; Kimmerling, K.A.; Rosson, G.D.; Elisseeff, J.H. An injectable adipose matrix for soft-tissue reconstruction. Plast. Reconstr. Surg. 2012, 129, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Q.; Fan, J.; Gao, J.H.; Zhang, C.; Bai, S.L. Comparison of in vivo adipogenic capabilities of two different extracellular matrix microparticle scaffolds. Plast. Reconstr. Surg. 2013, 131, 174e–187e. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Johnson, J.A.; Zhang, Q.; Beahm, E.K. Combining decellularized human adipose tissue extracellular matrix and adipose-derived stem cells for adipose tissue engineering. Acta Biomater. 2013, 9, 8921–8931. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Bianco, J.; Brown, C.; Fuetterer, L.; Watkins, J.F.; Samani, A.; Flynn, L.E. Porous decellularized adipose tissue foams for soft tissue regeneration. Biomaterials 2013, 34, 3290–3302. [Google Scholar] [CrossRef] [PubMed]
- Cheung, H.K.; Han, T.T.; Marecak, D.M.; Watkins, J.F.; Amsden, B.G.; Flynn, L.E. Composite hydrogel scaffolds incorporating decellularized adipose tissue for soft tissue engineering with adipose-derived stem cells. Biomaterials 2014, 35, 1914–1923. [Google Scholar] [CrossRef] [PubMed]
- Han, T.T.; Toutounji, S.; Amsden, B.G.; Flynn, L.E. Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds. Biomaterials 2015, 72, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.F.; Yan, J.; Han, T.T.; Marecak, D.M.; Amsden, B.G.; Flynn, L.E. Effect of decellularized adipose tissue particle size and cell density on adipose-derived stem cell proliferation and adipogenic differentiation in composite methacrylated chondroitin sulphate hydrogels. Biomed. Mater. 2015, 10, 045010. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Kornmuller, A.; Brown, C.; Hoare, T.; Flynn, L.E. Decellularized adipose tissue microcarriers as a dynamic culture platform for human adipose-derived stem/stromal cell expansion. Biomaterials 2017, 120, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Shridhar, A.; Gillies, E.; Amsden, B.G.; Flynn, L.E. Composite Bioscaffolds Incorporating Decellularized ECM as a Cell-Instructive Component Within Hydrogels as In Vitro Models and Cell Delivery Systems. Methods Mol. Biol. 2017. [Google Scholar] [CrossRef]
- Kayabolen, A.; Keskin, D.; Aykan, A.; Karslioglu, Y.; Zor, F.; Tezcaner, A. Native extracellular matrix/fibroin hydrogels for adipose tissue engineering with enhanced vascularization. Biomed. Mater. 2017, 12, 035007. [Google Scholar] [CrossRef] [PubMed]
- Morissette Martin, P.; Grant, A.; Hamilton, D.W.; Flynn, L.E. Matrix composition in 3-D collagenous bioscaffolds modulates the survival and angiogenic phenotype of human chronic wound dermal fibroblasts. Acta Biomater. 2018. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Lin, M.; Wang, X.; Guan, J.; Dong, Z.; Lu, F.; Xing, M.; Feng, C.; Li, X. Optimized adipose tissue engineering strategy based on a neo-mechanical processing method. Wound Repair Regen. 2018, 26, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Thomas-Porch, C.; Li, J.; Zanata, F.; Martin, E.C.; Pashos, N.; Genemaras, K.; Poche, J.N.; Totaro, N.P.; Bratton, M.R.; Gaupp, D.; et al. Comparative proteomic analyses of human adipose extracellular matrices decellularized using alternative procedures. J. Biomed. Mater. Res. Part A 2018, 106, 2481–2493. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Poche, J.N.; Liu, Y.; Scherr, T.; McCann, J.; Forghani, A.; Smoak, M.; Muir, M.; Berntsen, L.; Chen, C.; et al. Hybrid Synthetic-Biological Hydrogel System for Adipose Tissue Regeneration. Macromol. Biosci. 2018, 18, e1800122. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Liu, Y.; Hui, L. Preparation and characterization of acellular adipose tissue matrix using a combination of physical and chemical treatments. Mol. Med. Rep. 2018, 17, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Fan, J.; Bai, S. Biocompatibility of injectable hydrogel from decellularized human adipose tissue in vitro and in vivo. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018. [Google Scholar] [CrossRef] [PubMed]
- Giatsidis, G.; Succar, J.; Haddad, A.; Lago, G.; Schaffer, C.; Wang, X.; Schilling, B.; Chnari, E.; Matsumine, H.; Orgill, D.P. Preclinical Optimization of a Shelf-Ready, Injectable, Human-Derived, Decellularized Allograft Adipose Matrix. Tissue Eng. Part A 2018. [Google Scholar] [CrossRef] [PubMed]
- Sano, H.; Orbay, H.; Terashi, H.; Hyakusoku, H.; Ogawa, R. Acellular adipose matrix as a natural scaffold for tissue engineering. J. Plast. Reconstr. Aesthet. Surg. 2014, 67, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Dunne, L.W.; Huang, Z.; Meng, W.; Fan, X.; Zhang, N.; Zhang, Q.; An, Z. Human decellularized adipose tissue scaffold as a model for breast cancer cell growth and drug treatments. Biomaterials 2014, 35, 4940–4949. [Google Scholar] [CrossRef] [PubMed]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Adams, R.J.; Berry, J.D.; Brown, T.M.; Carnethon, M.R.; Dai, S.; de Simone, G.; Ford, E.S.; et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics-2011 update: A report from the American Heart Association. Circulation 2011, 123, e18–e209. [Google Scholar] [CrossRef] [PubMed]
- Tonsho, M.; Michel, S.; Ahmed, Z.; Alessandrini, A.; Madsen, J.C. Heart transplantation: Challenges facing the field. Cold Spring Harb. Perspect. Med. 2014, 4, A015636. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Lemcke, H.; David, R. Stem cell therapy in heart diseases—Cell types, mechanisms and improvement strategies. Cell. Physiol. Biochem. 2018, 48, 2607–2655. [Google Scholar] [CrossRef] [PubMed]
- Perez-Estenaga, I.; Prosper, F.; Pelacho, B. Allogeneic mesenchymal stem cells and biomaterials: The perfect match for cardiac repair? Int. J. Mol. Sci. 2018, 19, 3236. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, R.; Hong, Y.; Takanari, K.; Fujimoto, K.L.; Tobita, K.; Wagner, W.R. The effect of polymer degradation time on functional outcomes of temporary elastic patch support in ischemic cardiomyopathy. Biomaterials 2013, 34, 7353–7363. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.D.; Ko, M.C.; Wu, S.T.; Li, S.F.; Hu, J.F.; Lai, Y.J.; Harn, H.I.; Laio, I.C.; Yeh, M.L.; Yeh, H.I.; et al. A nanopatterned cell-seeded cardiac patch prevents electro-uncoupling and improves the therapeutic efficacy of cardiac repair. Biomater. Sci. 2014, 2, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Menasché, P.; Vanneaux, V.; Hagège, A.; Bel, A.; Cholley, B.; Cacciapuoti, I.; Parouchev, A.; Benhamouda, N.; Tachdjian, G.; Tosca, L.; et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: First clinical case report. Eur. Heart J. 2015, 36, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.; Maring, J.A.; Oberwallner, B.; Kappler, B.; Klein, O.; Falk, V.; Stamm, C. Processing of human cardiac tissue toward extracellular matrix self-assembling hydrogel for in vitro and in vivo applications. J. Vis. Exp. 2017, 130, e56419. [Google Scholar] [CrossRef] [PubMed]
- Moroni, F.; Mirabella, T. Decellularized matrices for cardiovascular tissue engineering. Am. J. Stem Cells 2014, 3, 1–20. [Google Scholar] [PubMed]
- Sánchez, P.L.; Fernández-Santos, M.E.; Costanza, S.; Climent, A.M.; Moscoso, I.; Gonzalez-Nicolas, M.A.; Sanz-Ruiz, R.; Rodríguez, H.; Kren, S.M.; Garrido, G.; et al. Acellular human heart matrix: A critical step toward whole heart grafts. Biomaterials 2015, 61, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Oberwallner, B.; Brodarac, A.; Choi, Y.H.; Saric, T.; Anić, P.; Morawietz, L.; Stamm, C. Preparation of cardiac extracellular matrix scaffolds by decellularization of human myocardium. J. Biomed. Mater. Res. A 2014, 102, 3263–3272. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, P.L.; Fernández-Santos, M.E.; Espinosa, M.A.; González-Nicolas, M.A.; Acebes, J.R.; Costanza, S.; Moscoso, I.; Rodríguez, H.; García, J.; Romero, J.; et al. Data from acellular human heart matrix. Data Brief 2016, 8, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Garreta, E.; de Oñate, L.; Fernández-Santos, M.E.; Oria, R.; Tarantino, C.; Climent, A.M.; Marco, A.; Samitier, M.; Martínez, E.; Valls-Margarit, M.; et al. Myocardial commitment from human pluripotent stem cells: Rapid production of human heart grafts. Biomaterials 2016, 98, 64–78. [Google Scholar] [CrossRef] [PubMed]
- Guyette, J.P.; Charest, J.M.; Mills, R.W.; Jank, B.J.; Moser, P.T.; Gilpin, S.E.; Gershlak, J.R.; Okamoto, T.; Gonzalez, G.; Milan, D.J.; et al. Bioengineering human myocardium on native extracellular matrix. Circ. Res. 2016, 118, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Guhathakurta, S.; Mathapati, S.; Bishi, D.K.; Rallapalli, S.; Cherian, K.M. Nanofiber-reinforced myocardial tissue-construct as ventricular assist device. Asian Cardiovasc. Thorac. Ann. 2014, 22, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Kappler, B.; Anic, P.; Becker, M.; Bader, A.; Klose, K.; Klein, O.; Oberwallner, B.; Choi, Y.H.; Falk, V.; Stamm, C. The cytoprotective capacity of processed human cardiac extracellular matrix. J. Mater. Sci. Mater. Med. 2016, 27, 120. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.; Maring, J.A.; Schneider, M.; Herrera Martin, A.X.; Seifert, M.; Klein, O.; Braun, T.; Falk, V.; Stamm, C. Towards a Novel Patch Material for Cardiac Applications: Tissue-Specific Extracellular Matrix Introduces Essential Key Features to Decellularized Amniotic Membrane. Int. J. Mol. Sci. 2018, 19, 1032. [Google Scholar] [CrossRef] [PubMed]
- Godier-Furnémont, A.F.; Martens, T.P.; Koeckert, M.S.; Wan, L.; Parks, J.; Arai, K.; Zhang, G.; Hudson, B.; Homma, S.; Vunjak-Novakovic, G. Composite scaffold provides a cell delivery platform for cardiovascular repair. Proc. Natl. Acad. Sci. USA 2011, 108, 7974–7979. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.D.; Hill, R.C.; Dzieciatkowska, M.; Nigam, V.; Behfar, A.; Christman, K.L.; Hansen, K.C. Quantification of decellularized human myocardial matrix: A comparison of six patients. Proteom. Clin. Appl. 2016, 10, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.M.; Johnson, T.D.; He, J.; Rong, Z.; Wong, M.; Nigam, V.; Behfar, A.; Xu, Y.; Christman, K.L. Humanized mouse model for assessing the human immune response to xenogeneic and allogeneic decellularized biomaterials. Biomaterials 2017, 129, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, F.; Nurzynska, D.; Romano, V.; Miraglia, R.; Belviso, I.; Sacco, A.M.; Barbato, V.; Di Gennaro, M.; Granato, G.; Maiello, C.; et al. Optimization of human myocardium decellularization method for the construction of implantable patches. Tissue Eng. Part C Methods 2017, 23, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Mirsadraee, S.; Wilcox, H.E.; Watterson, K.G.; Kearney, J.N.; Hunt, J.; Fisher, J.; Ingham, E. Biocompatibility of acellular human pericardium. J. Surg. Res. 2007, 143, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Mirsadraee, S.; Wilcox, H.E.; Korossis, S.A.; Kearney, J.N.; Watterson, K.G.; Fisher, J.; Ingham, E. Development and characterization of an acellular human pericardial matrix for tissue engineering. Tissue Eng. 2006, 12, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Vinci, M.C.; Tessitore, G.; Castiglioni, L.; Prandi, F.; Soncini, M.; Santoro, R.; Consolo, F.; Colazzo, F.; Micheli, B.; Sironi, L.; et al. Mechanical compliance and immunological compatibility of fixative-free decellularized/cryopreserved human pericardium. PLoS ONE 2013, 8, e64769. [Google Scholar] [CrossRef] [PubMed]
- Rajabi-Zeleti, S.; Jalili-Firoozinezhad, S.; Azarnia, M.; Khayyatan, F.; Vahdat, S.; Nikeghbalian, S.; Khademhosseini, A.; Baharvand, H.; Aghdami, N. The behavior of cardiac progenitor cells on macroporous pericardium-derived scaffolds. Biomaterials 2014, 35, 970–982. [Google Scholar] [CrossRef] [PubMed]
- Van Steenberghe, M.; Schubert, T.; Guiot, Y.; Bouzin, C.; Bollen, X.; Gianello, P. Enhanced vascular biocompatibility of decellularized xeno-/allogeneic matrices in a rodent model. Cell Tissue Bank. 2017, 18, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Van Steenberghe, M.; Schubert, T.; Xhema, D.; Bouzin, C.; Guiot, Y.; Duisit, J.; Abdelhamid, K.; Gianello, P. Enhanced vascular regeneration with chemically/physically treated bovine/human pericardium in rodents. J. Surg. Res. 2018, 222, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Van Steenberghe, M.; Schubert, T.; Bouzin, C.; Caravaggio, C.; Guiot, Y.; Xhema, D.; Gianello, P. Enhanced vascular biocompatibility and remodeling of decellularized and secured xenogeneic/allogeneic matrices in a porcine model. Eur. Surg. Res. 2018, 59, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Seif-Naraghi, S.B.; Horn, D.; Schup-Magoffin, P.A.; Madani, M.M.; Christman, K.L. Patient-to-patient variability in autologous pericardial matrix scaffolds for cardiac repair. J. Cardiovasc. Transl. Res. 2011, 4, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Prat-Vidal, C.; Gálvez-Montón, C.; Puig-Sanvicens, V.; Sanchez, B.; Díaz-Güemes, I.; Bogónez-Franco, P.; Perea-Gil, I.; Casas-Solà, A.; Roura, S.; Llucià-Valldeperas, A.; et al. Online monitoring of myocardial bioprosthesis for cardiac repair. Int. J. Cardiol. 2014, 174, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Monton, C.; Bragos, R.; Soler-Botija, C.; Diaz-Guemes, I.; Prat-Vidal, C.; Crisostomo, V.; Sanchez-Margallo, F.M.; Llucia-Valldeperas, A.; Bogonez-Franco, P.; Perea-Gil, I.; et al. Noninvasive Assessment of an Engineered Bioactive Graft in Myocardial Infarction: Impact on Cardiac Function and Scar Healing. Stem Cells Transl. Med. 2017, 6, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Perea-Gil, I.; Galvez-Monton, C.; Prat-Vidal, C.; Jorba, I.; Segu-Verges, C.; Roura, S.; Soler-Botija, C.; Iborra-Egea, O.; Revuelta-Lopez, E.; Fernandez, M.A.; et al. Head-to-head comparison of two engineered cardiac grafts for myocardial repair: From scaffold characterization to pre-clinical testing. Sci. Rep. 2018, 8, 6708. [Google Scholar] [CrossRef] [PubMed]
- Cheung, D.Y.; Duan, B.; Butcher, J.T. Current progress in tissue engineering of heart valves: Multiscale problems; multiscale solutions. Expert Opin. Biol. Ther. 2015, 15, 1155–1172. [Google Scholar] [CrossRef] [PubMed]
- Naso, F.; Gandaglia, A. Different approaches to heart valve decellularization: A comprehensive overview of the past 30 years. Xenotransplantation 2018, 25. [Google Scholar] [CrossRef] [PubMed]
- Dainese, L.; Guarino, A.; Burba, I.; Esposito, G.; Pompilio, G.; Polvani, G.; Rossini, A. Heart valve engineering: Decellularized aortic homograft seeded with human cardiac stromal cells. J. Heart Valve Dis. 2012, 21, 125–134. [Google Scholar] [PubMed]
- Koenig, F.; Lee, J.S.; Akra, B.; Hollweck, T.; Wintermantel, E.; Hagl, C.; Thierfelder, N. Is Transcatheter Aortic Valve Implantation of Living Tissue-Engineered Valves Feasible? An In Vitro Evaluation Utilizing a Decellularized and Reseeded Biohybrid Valve. Artif. Organs 2016, 40, 727–737. [Google Scholar] [CrossRef] [PubMed]
- VeDepo, M.C.; Buse, E.E.; Quinn, R.W.; Williams, T.D.; Detamore, M.S.; Hopkins, R.A.; Converse, G.L. Species-specific effects of aortic valve decellularization. Acta Biomater. 2017, 50, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Iop, L.; Paolin, A.; Aguiari, P.; Trojan, D.; Cogliati, E.; Gerosa, G. Decellularized Cryopreserved Allografts as Off-the-Shelf Allogeneic Alternative for Heart Valve Replacement: In Vitro Assessment Before Clinical Translation. J. Cardiovasc. Transl. Res. 2017, 10, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Rieder, E.; Seebacher, G.; Kasimir, M.T.; Eichmair, E.; Winter, B.; Dekan, B.; Wolner, E.; Simon, P.; Weigel, G. Tissue engineering of heart valves: Decellularized porcine and human valve scaffolds differ importantly in residual potential to attract monocytic cells. Circulation 2005, 111, 2792–2797. [Google Scholar] [CrossRef] [PubMed]
- Iop, L.; Renier, V.; Naso, F.; Piccoli, M.; Bonetti, A.; Gandaglia, A.; Pozzobon, M.; Paolin, A.; Ortolani, F.; Marchini, M.; et al. The influence of heart valve leaflet matrix characteristics on the interaction between human mesenchymal stem cells and decellularized scaffolds. Biomaterials 2009, 30, 4104–4116. [Google Scholar] [CrossRef] [PubMed]
- Cebotari, S.; Mertsching, H.; Kallenbach, K.; Kostin, S.; Repin, O.; Batrinac, A.; Kleczka, C.; Ciubotaru, A.; Haverich, A. Construction of autologous human heart valves based on an acellular allograft matrix. Circulation 2002, 106, I63–I68. [Google Scholar] [PubMed]
- Gerson, C.J.; Elkins, R.C.; Goldstein, S.; Heacox, A.E. Structural integrity of collagen and elastin in SynerGraft(R) decellularized-cryopreserved human heart valves. Cryobiology 2012, 64, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Jiao, T.; Clifton, R.J.; Converse, G.L.; Hopkins, R.A. Measurements of the effects of decellularization on viscoelastic properties of tissues in ovine, baboon, and human heart valves. Tissue Eng. Part A 2012, 18, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Weymann, A.; Schmack, B.; Okada, T.; Soós, P.; Istók, R.; Radovits, T.; Straub, B.; Barnucz, E.; Loganathan, S.; Pätzold, I.; et al. Reendothelialization of human heart valve neoscaffolds using umbilical cord-derived endothelial cells. Circ. J. 2013, 77, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Vafaee, T.; Thomas, D.; Desai, A.; Jennings, L.M.; Berry, H.; Rooney, P.; Kearney, J.; Fisher, J.; Ingham, E. Decellularization of human donor aortic and pulmonary valved conduits using low concentration sodium dodecyl sulfate. J. Tissue Eng. Regen. Med. 2018, 12, e841–e853. [Google Scholar] [CrossRef] [PubMed]
- Weymann, A.; Radovits, T.; Schmack, B.; Li, S.; Korkmaz, S.; Soós, P.; Istók, R.; Veres, G.; Chaimow, N.; Karck, M.; Szabó, G. In vitro generation of atrioventricular heart valve neoscaffolds. Artif. Organs 2014, 38, E118–E128. [Google Scholar] [CrossRef] [PubMed]
- Iablonskii, P.; Cebotari, S.; Tudorache, I.; Granados, M.; Morticelli, L.; Goecke, T.; Klein, N.; Korossis, S.; Hilfiker, A.; Haverich, A. Tissue-engineered mitral valve: Morphology and biomechanics. Interact. Cardiovasc. Thorac. Surg. 2015, 20, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Li, S.; Lei, D.; Huang, P.; Yuan, Z.; You, Z.; Ye, X.; Zhao, Q. Tissue-engineered mitral valve chordae tendineae: Biomechanical and biological characterization of decellularized porcine chordae. J. Mech. Behav. Biomed. Mater. 2016, 56, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Gunning, G.M.; Murphy, B.P. The effects of decellularization and cross-linking techniques on the fatigue life and calcification of mitral valve chordae tendineae. J. Mech. Behav. Biomed. Mater. 2016, 57, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Granados, M.; Morticelli, L.; Andriopoulou, S.; Kalozoumis, P.; Pflaum, M.; Iablonskii, P.; Glasmacher, B.; Harder, M.; Hegermann, J.; Wrede, C.; et al. Development and characterization of a porcine mitral valve scaffold for tissue engineering. J. Cardiovasc. Transl. Res. 2017, 10, 374–390. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Chen, Y.; Wang, Z.; Wang, W.; Schmull, S.; Dong, J.; Xue, S.; Imboden, H.; Li, J. Human heart valve-derived scaffold improves cardiac repair in a murine model of myocardial infarction. Sci. Rep. 2017, 7, 39988. [Google Scholar] [CrossRef] [PubMed]
- Teebken, O.E.; Haverich, A. Tissue Engineering of Small Diameter Vascular Grafts. Eur. J. Vasc. Endovasc. Surg. 2002, 23, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Gui, L.; Muto, A.; Chan, S.A.; Breuer, C.K.; Niklason, L.E. Development of decellularized human umbilical arteries as small-diameter vascular grafts. Tissue Eng. Part A 2009, 15, 2665. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Wu, G.; Wu, M.; Zhang, X.; Velot, E.; Decot, V.; Cui, W.; Huang, Y.; Stoltz, J.F.; Du, J.; et al. Construction of biocompatible porous tissue scaffold from the decellularized umbilical artery. Biomed. Mater. Eng. 2015, 25, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Huang, R.; Zhang, X.; Xin, Y.; Li, J.; Huang, Y.; Cui, W.; Stoltz, J.F.; Zhou, Y.; Kong, Q. Stem cells cardiac patch from decellularized umbilical artery improved heart function after myocardium infarction. Biomed. Mater. Eng. 2017, 28, S87–S94. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rodríguez, V.E.; Martínez-González, B.; Quiroga-Garza, A.; Reyes-Hernández, C.G.; de la Fuente-Villarreal, D.; de la Garza-Castro, O.; Guzmán-López, S.; Elizondo-Omaña, R.E. Human Umbilical Vessels: Choosing the Optimal Decellularization Method. ASAIO J. 2018, 64, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Tuan-Mu, H.Y.; Yu, C.H.; Hu, J.J. On the decellularization of fresh or frozen human umbilical arteries: Implications for small-diameter tissue engineered vascular grafts. Ann. Biomed. Eng. 2014, 42, 1305–1318. [Google Scholar] [CrossRef] [PubMed]
- Mallis, P.; Gontika, I.; Poulogiannopoulos, T.; Zoidakis, J.; Vlahou, A.; Michalopoulos, E.; Chatzistamatiou, T.; Papassavas, A.; Stavropoulos-Giokas, C. Evaluation of decellularization in umbilical cord artery. Transplant. Proc. 2014, 46, 3232. [Google Scholar] [CrossRef] [PubMed]
- Mallis, P.; Michalopoulos, E.; Dinou, A.; Vlachou, M.S.; Panagouli, E.; Papapanagiotou, A.; Kassi, E.; Giokas, C.S. Development of HLA-matched vascular grafts utilizing decellularized human umbilical artery. Hum. Immunol. 2018, 79, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.; Juran, C.; McClendon, M.; Eyadiel, C.; McFetridge, P.S. Development of a mechanically tuneable 3D scaffold for vascular reconstruction. J. Biomed. Mater. Res. Part A 2012, 100, 3480–3489. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Li, J.; Dong, P. Utilization of pulsatile flow to decellularize the human umbilical arteries to make small-caliber blood vessel scaffolds. Acta Cardiol. Sin. 2013, 29, 451. [Google Scholar] [PubMed]
- Wilshaw, S.P.; Rooney, P.; Berry, H.; Kearney, J.N.; Homer-Vanniasinkam, S.; Fisher, J.; Ingham, E. Development and characterization of acellular allogeneic arterial matrices. Tissue Eng. Part A 2012, 18, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Kajbafzadeh, A.M.; Khorramirouz, R.; Kameli, S.M.; Hashemi, J.; Bagheri, A. Decellularization of Human Internal Mammary Artery: Biomechanical Properties and Histopathological Evaluation. Biores. Open Access 2017, 6, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.G.; Hu, Y.; Xu, Q.; Jahangiri, M. Stem cells accumulate on a decellularized arterial xenograft in vivo. Ann. Thorac. Surg. 2014, 97, 2104–2110. [Google Scholar] [CrossRef] [PubMed]
- Quint, C.; Arief, M.; Muto, A.; Dardik, A.; Niklason, L.E. Allogeneic human tissue-engineered blood vessel. J. Vasc. Surg. 2012, 55, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Daniel, J.; Abe, K.; McFetridge, P.S. Development of the human umbilical vein scaffold for cardiovascular tissue engineering applications. ASAIO J. 2005, 5, 252–261. [Google Scholar] [CrossRef]
- Goktas, S.; Matuska, A.M.; Pierre, N.; Gibson, T.M.; Dmytryk, J.J.; McFetridge, P.S. Decellularization method influences early remodeling of an allogenic tissue scaffold. J. Biomed. Mater. Res. A 2014, 102, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Uzarski, J.S.; Van De Walle, A.B.; McFetridge, P.S. Preimplantation processing of ex vivo-derived vascular biomaterials: Effects on peripheral cell adhesion. J. Biomed. Mater. Res. A 2013, 101, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Van de Walle, A.B.; Uzarski, J.S.; McFetridge, P.S. The consequence of biologic graft processing on blood interface biocompatibility and mechanics. Cardiovasc. Eng. Technol. 2015, 6, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Mangold, S.; Schrammel, S.; Huber, G.; Niemeyer, M.; Schmid, C.; Stangassinger, M.; Hoenicka, M. Evaluation of decellularized human umbilical vein (HUV) for vascular tissue engineering—Comparison with endothelium-denuded HUV. J. Tissue Eng. Regen. Med. 2015, 9, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Abousleiman, R.I.; Reyes, Y.; McFetridge, P.; Sikavitsas, V. The human umbilical vein: A novel scaffold for musculoskeletal soft tissue regeneration. Artif. Organs 2008, 32, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Abousleiman, R.I.; Reyes, Y.; McFetridge, P.; Sikavitsas, V. Tendon tissue engineering using cell-seeded umbilical veins cultured in a mechanical stimulator. Tissue Eng. Part A 2009, 15, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Montoya, C.V.; McFetridge, P.S. Preparation of ex vivo–based biomaterials using convective flow decellularization. Tissue Eng. Part C Methods 2009, 15, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Issa, R.I.; Engebretson, B.; Rustom, L.; McFetridge, P.S.; Sikavitsas, V.I. The effect of cell seeding density on the cellular and mechanical properties of a mechanostimulated tissue-engineered tendon. Tissue Eng. Part A 2011, 17, 1479–1487. [Google Scholar] [CrossRef] [PubMed]
- Hoenicka, M.; Schrammel, S.; Bursa, J.; Huber, G.; Bronger, H.; Schmid, C.; Birnbaum, D.E. Development of endothelium-denuded human umbilical veins as living scaffolds for tissue-engineered small-calibre vascular grafts. J. Tissue Eng. Regen. Med. 2013, 7, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Hoenicka, M.; Kaspar, M.; Schmid, C.; Liebold, A.; Schrammel, S. Contact-free monitoring of vessel graft stiffness—Proof of concept as a tool for vascular tissue engineering. J. Tissue Eng. Regen. Med. 2017, 11, 2828–2835. [Google Scholar] [CrossRef] [PubMed]
- Hsia, K.; Yang, M.J.; Chen, W.M.; Yao, C.L.; Lin, C.H.; Loong, C.C.; Huang, Y.L.; Lin, Y.T.; Lander, A.D.; Lee, H.; et al. Sphingosine-1-phosphate improves endothelialization with reduction of thrombosis in recellularized human umbilical vein graft by inhibiting syndecan-1 shedding in vitro. Acta Biomater. 2017, 51, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Schaner, P.J.; Martin, N.D.; Tulenko, T.N.; Shapiro, I.M.; Talora, N.A.; Leichter, R.F.; Carabasi, R.A.; Dimuzio, P.J. Decellularized vein as a potential scaffold for vascular tissue engineering. J. Vasc. Surg. 2004, 40, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Kumar Kuna, V.; Xu, B.; Sumitran-Holgersson, S. Decellularization and Recellularization Methodology for Human Saphenous Veins. J. Vis. Exp. 2018, 27, 57803. [Google Scholar] [CrossRef] [PubMed]
- Rambol, M.H.; Hisdal, J.; Sundhagen, J.O.; Brinchmann, J.E.; Rosales, A. Recellularization of Decellularized Venous Grafts Using Peripheral Blood: A Critical Evaluation. EBioMedicine 2018, 32, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Hassan, N.H.; Sulong, A.F.; Ng, M.H.; Htwe, O.; Idrus, R.B.; Roohi, S.; Naicker, A.S.; Abdullah, S. Neural-differentiated mesenchymal stem cells incorporated into muscle stuffed vein scaffold forms a stable living nerve conduit. J. Orthop. Res. 2012, 30, 1674–1681. [Google Scholar] [CrossRef] [PubMed]
- McIlhenny, S.; Zhang, P.; Tulenko, T.; Comeau, J.; Fernandez, S.; Policha, A.; Ferroni, M.; Faul, E.; Bagameri, G.; Shapiro, I.; et al. eNOS transfection of adipose-derived stem cells yields bioactive nitric oxide production and improved results in vascular tissue engineering. J. Tissue. Eng. Regen. Med. 2015, 9, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J.D.; Anfang, R.; Anandappa, A.; Costa, J.; Javidfar, J.; Wobma, H.M.; Singh, G.; Freytes, D.O.; Bacchetta, M.D.; Sonett, J.R.; et al. Decellularization of human and porcine lung tissues for pulmonary tissue engineering. Ann. Thorac. Surg. 2013, 96, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- Ghaedi, M.; Calle, E.A.; Mendez, J.J.; Gard, A.L.; Balestrini, J.; Booth, A.; Bove, P.F.; Gui, L.; White, E.S.; Niklason, L.E. Human iPS cell-derived alveolar epithelium repopulates lung extracellular matrix. J. Clin. Investig. 2013, 123, 4950–4962. [Google Scholar] [CrossRef] [PubMed]
- Bruzauskaite, I.; Raudoniute, J.; Denkovskij, J.; Bagdonas, E.; Meidute-Abaraviciene, S.; Simonyte, V.; Bironaite, D.; Siaurys, A.; Bernotiene, E.; Aldonyte, R. Native matrix-based human lung alveolar tissue model in vitro: Studies of the reparatory actions of mesenchymal stem cells. Cytotechnology 2017, 69, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Rosmark, O.; Ahrman, E.; Muller, C.; Elowsson Rendin, L.; Eriksson, L.; Malmstrom, A.; Hallgren, O.; Larsson-Callerfelt, A.K.; Westergren-Thorsson, G.; Malmstrom, J. Quantifying extracellular matrix turnover in human lung scaffold cultures. Sci. Rep. 2018, 8, 5409. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.E.; Niles, J.; Riddle, M.; Vargs, G.; Schilagard, T.; Ma, L.; Edward, K.; La Francesca, S.; Sakamoto, J.; Vega, S.; et al. Production and assessment of decellularized pig and human lung scaffolds. Tissue Eng. Part A 2013, 19, 2045–2062. [Google Scholar] [CrossRef] [PubMed]
- Wagner, D.E.; Bonenfant, N.R.; Sokocevic, D.; DeSarno, M.J.; Borg, Z.D.; Parsons, C.S.; Brooks, E.M.; Platz, J.J.; Khalpey, Z.I.; Hoganson, D.M.; et al. Three-dimensional scaffolds of acellular human and porcine lungs for high throughput studies of lung disease and regeneration. Biomaterials 2014, 35, 2664–2679. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, S.E.; Guyette, J.P.; Gonzalez, G.; Ren, X.; Asara, J.M.; Mathisen, D.J.; Vacanti, J.P.; Ott, H.C. Perfusion decellularization of human and porcine lungs: Bringing the matrix to clinical scale. J. Heart Lung Transplant. 2014, 33, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Zvarova, B.; Uhl, F.E.; Uriarte, J.J.; Borg, Z.D.; Coffey, A.L.; Bonenfant, N.R.; Weiss, D.J.; Wagner, D.E. Residual Detergent Detection Method for Nondestructive Cytocompatibility Evaluation of Decellularized Whole Lung Scaffolds. Tissue Eng. Part C Methods 2016, 22, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Balestrini, J.L.; Gard, A.L.; Gerhold, K.A.; Wilcox, E.C.; Liu, A.; Schwan, J.; Le, A.V.; Baevova, P.; Dimitrievska, S.; Zhao, L.; et al. Comparative biology of decellularized lung matrix: Implications of species mismatch in regenerative medicine. Biomaterials 2016, 102, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.E.; La Francesca, S.; Vega, S.P.; Niles, J.A.; Argueta, L.B.; Riddle, M.; Sakamoto, J.; Vargas, G.; Pal, R.; Woodson, L.; et al. Giving new life to old lungs: Methods to produce and assess whole human paediatric bioengineered lungs. J. Tissue. Eng. Regen. Med. 2017, 11, 2136–2152. [Google Scholar] [CrossRef] [PubMed]
- Lwebuga-Mukasa, J.S.; Ingbar, D.H.; Madri, J.A. Repopulation of a human alveolar matrix by adult rat type II pneumocytes in vitro. A novel system for type II pneumocyte culture. Exp. Cell Res. 1986, 162, 423–435. [Google Scholar] [CrossRef]
- Wagner, D.E.; Bonenfant, N.R.; Parsons, C.S.; Sokocevic, D.; Brooks, E.M.; Borg, Z.D.; Lathrop, M.J.; Wallis, J.D.; Daly, A.B.; Lam, Y.W.; et al. Comparative decellularization and recellularization of normal versus emphysematous human lungs. Biomaterials 2014, 35, 3281–3297. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.X.; Islam, M.N.; O’ Neill, J.; Hu, Z.; Yang, Y.G.; Chen, Y.W.; Mumau, M.; Green, M.D.; Vunjak-Novakovic, G.; Bhattacharya, J.; et al. Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nat. Biotechnol. 2014, 32, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.H.; Calle, E.A.; Zhao, L.; Lee, E.J.; Gui, L.; Raredon, M.B.; Gavrilov, K.; Yi, T.; Zhuang, Z.W.; Breuer, C.; et al. Tissue-engineered lungs for in vivo implantation. Science 2010, 329, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Moser, P.T.; Gilpin, S.E.; Okamoto, T.; Wu, T.; Tapias, L.F.; Mercier, F.E.; Xiong, L.; Ghawi, R.; Scadden, D.T.; et al. Engineering pulmonary vasculature in decellularized rat and human lungs. Nat. Biotechnol. 2015, 33, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, S.E.; Charest, J.M.; Ren, X.; Tapias, L.F.; Wu, T.; Evangelista-Leite, D.; Mathisen, D.J.; Ott, H.C. Regenerative potential of human airway stem cells in lung epithelial engineering. Biomaterials 2016, 108, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, S.E.; Wagner, D.E. Acellular human lung scaffolds to model lung disease and tissue regeneration. Eur. Respir. Rev. 2018, 27, 180021. [Google Scholar] [CrossRef] [PubMed]
- Mendez, J.J.; Ghaedi, M.; Steinbacher, D.; Niklason, L.E. Epithelial cell differentiation of human mesenchymal stromal cells in decellularized lung scaffolds. Tissue Eng. Part A 2014, 20, 1735–1746. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.J.; Hadley, R.; Cornett, A.M.; Dreffs, A.A.; Matthes, S.A.; Tsui, J.L.; Weiss, K.; Horowitz, J.C.; Fiore, V.F.; Barker, T.H.; et al. Acellular normal and fibrotic human lung matrices as a culture system for in vitro investigation. Am. J. Respir. Crit. Care Med. 2012, 186, 866–876. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.W.; Rossi, D.; Peterson, M.; Smith, K.; Sikstrom, K.; White, E.S.; Connett, J.E.; Henke, C.A.; Larsson, O.; Bitterman, P.B. Fibrotic extracellular matrix activates a profibrotic positive feedback loop. J. Clin. Investig. 2014, 124, 1622–1635. [Google Scholar] [CrossRef] [PubMed]
- Tjin, G.; White, E.S.; Faiz, A.; Sicard, D.; Tschumperlin, D.J.; Mahar, A.; Kable, E.P.W.; Burgess, J.K. Lysyl oxidases regulate fibrillar collagen remodelling in idiopathic pulmonary fibrosis. Dis. Model. Mech. 2017, 10, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Sava, P.; Ramanathan, A.; Dobronyi, A.; Peng, X.; Sun, H.; Ledesma-Mendoza, A.; Herzog, E.L.; Gonzalez, A.J. Human pericytes adopt myofibroblast properties in the microenvironment of the IPF lung. JCI Insight 2017, 2, e96352. [Google Scholar] [CrossRef] [PubMed]
- Van der Velden, J.L.; Wagner, D.E.; Lahue, K.G.; Abdalla, S.T.; Lam, Y.W.; Weiss, D.J.; Janssen-Heininger, Y.M.W. TGF-β1-induced deposition of provisional extracellular matrix by tracheal basal cells promotes epithelial-to-mesenchymal transition in a JNK1-dependent manner. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L984–L997. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhu, Y.; Pan, H.; Chen, X.; Balestrini, J.L.; Lam, T.T.; Kanyo, J.E.; Eichmann, A.; Gulati, M.; Fares, W.H.; et al. Netrin-1 regulates fibrocyte accumulation in the decellularized fibrotic sclerodermatous lung microenvironment and in bleomycin-induced pulmonary fibrosis. Arthritis Rheumatol. 2016, 68, 1251–1261. [Google Scholar] [CrossRef] [PubMed]
- Mahdavishahri, N.; Moghatam Matin, M.; Fereidoni, M.; Yarjanli, Z.; Banihashem Rad, S.A.; Khajeh Ahmadi, S. In vitro Assay of Human Gingival Scaffold in Differentiation of Rat’s Bone Marrow Mesenchymal Stem Cells to Keratinocystes. Iran J. Basic Med. Sci. 2012, 15, 1185–1190. [Google Scholar] [PubMed]
- Naderi, S.; Khayat Zadeh, J.; Mahdavi Shahri, N.; Nejad Shahrokh Abady, K.; Cheravi, M.; Baharara, J.; Banihashem Rad, S.A.; Bahrami, A.R. Three-dimensional scaffold from decellularized human gingiva for cell cultures: Glycoconjugates and cell behavior. Cell J. 2013, 15, 166–175. [Google Scholar] [PubMed]
- Sangkert, S.; Meesane, J.; Kamonmattayakul, S.; Chai, W.L. Modified silk fibroin scaffolds with collagen/decellularized pulp for bone tissue engineering in cleft palate: Morphological structures and biofunctionalities. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 1138–1149. [Google Scholar] [CrossRef] [PubMed]
- Sangkert, S.; Kamonmattayakul, S.; Chai, W.L.; Meesane, J. Modified porous scaffolds of silk fibroin with mimicked microenvironment based on decellularized pulp/fibronectin for designed performance biomaterials in maxillofacial bone defect. J. Biomed. Mater. Res. A 2017, 105, 1624–1636. [Google Scholar] [CrossRef] [PubMed]
- Song, J.S.; Takimoto, K.; Jeon, M.; Vadakekalam, J.; Ruparel, N.B.; Diogenes, A. Decellularized Human Dental Pulp as a Scaffold for Regenerative Endodontics. J. Dent. Res. 2017, 96, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Matoug-Elwerfelli, M.; Duggal, M.S.; Nazzal, H.; Esteves, F.; Raïf, E. A biocompatible decellularized pulp scaffold for regenerative endodontics. Int. Endod. J. 2018, 51, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Wilshaw, S.P.; Kearney, J.N.; Fisher, J.; Ingham, E. Production of an acellular amniotic membrane matrix for use in tissue engineering. Tissue Eng. 2006, 12, 2117–2129. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Alfaro, F.; Torradeflot, M.M.; Marti, C. Prevalence and management of Schneiderian membrane perforations during sinus-lift procedures. Clin. Oral Implants Res. 2008, 19, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Meleo, D.; Mangione, F.; Corbi, S.; Pacifici, L. Management of the Schneiderian membrane perforation during the maxillary sinus elevation procedure: A case report. Ann. Stomatol. 2012, 3, 24–30. [Google Scholar]
- Rahpeyma, A.; Khajehahmadi, S.; Khalife, H.H. Decellularized human Schneiderian membrane: Electron microscopic study as a bioscaffold and preliminary cell seeding. Pathol. Res. Pract. 2014, 210, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Grandi, F.; Stocco, E.; Barbon, S.; Rambaldo, A.; Contran, M.; Fascetti Leon, F.; Gamba, P.; Parnigotto, P.P.; Macchi, V.; De Caro, R.; et al. Composite Scaffolds Based on Intestinal Extracellular Matrices and Oxidized Polyvinyl Alcohol: A Preliminary Study for a New Regenerative Approach in Short Bowel Syndrome. Biomed. Res. Int. 2018, 2018, 7824757. [Google Scholar] [CrossRef] [PubMed]
- Patil, P.B.; Chougule, P.B.; Kumar, V.K.; Almström, S.; Bäckdahl, H.; Banerjee, D.; Herlenius, G.; Olausson, M.; Sumitran-Holgersson, S. Recellularization of acellular human small intestine using bone marrow stem cells. Stem Cells Transl. Med. 2013, 2, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Kajbafzadeh, A.M.; Khorramirouz, R.; Masoumi, A.; Keihani, S.; Nabavizadeh, B. Decellularized human fetal intestine as a bioscaffold for regeneration of the rabbit bladder submucosa. J. Pediatr. Surg. 2018, 53, 1781–1788. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; et al. Disability–adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- Murray, C.J.; Lopez, A.D. Alternative projections of mortality and disability by cause 1990–2020: Global Burden of Disease Study. Lancet 1997, 349, 1498–1504. [Google Scholar] [CrossRef]
- Mazza, G.; Rombouts, K.; Rennie Hall, A.; Urbani, L.; Vinh Luong, T.; Al-Akkad, W.; Longato, L.; Brown, D.; Maghsoudlou, P.; Dhillon, A.P.; et al. Decellularized human liver as a natural 3D-scaffold for liver bioengineering and transplantation. Sci. Rep. 2015, 5, 13079. [Google Scholar] [CrossRef] [PubMed]
- Verstegen, M.M.A.; Willemse, J.; van den Hoek, S.; Kremers, G.J.; Luider, T.M.; van Huizen, N.A.; Willemssen, F.E.J.A.; Metselaar, H.J.; IJzermans, J.N.M.; van der Laan, L.J.W.; et al. Decellularization of Whole Human Liver Grafts Using Controlled Perfusion for Transplantable Organ Bioscaffolds. Stem Cells Dev. 2017, 26, 1304–1315. [Google Scholar] [CrossRef] [PubMed]
- Mattei, G.; Magliaro, C.; Pirone, A.; Ahluwalia, A. Decellularized human liver is too heterogeneous for designing a generic extracellular matrix mimic hepatic scaffold. Artif. Organs 2017, 41, E347–E355. [Google Scholar] [CrossRef] [PubMed]
- Mazza, G.; Al-Akkad, W.; Telese, A.; Longato, L.; Urbani, L.; Robinson, B.; Hall, A.; Kong, K.; Frenguelli, L.; Marrone, G.; et al. Rapid production of human liver scaffolds for functional tissue engineering by high shear stress oscillation-decellularization. Sci. Rep. 2017, 7, 5534. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, M.; Yeh, H.; Yarmush, M.L.; Uygun, B.E. Decellularized human liver extracellular matrix (hDLM)-mediated hepatic differentiation of human induced pluripotent stem cells (hIPSCs). J. Tissue Eng. Regen. Med. 2018, 12, 1962–1973. [Google Scholar] [CrossRef] [PubMed]
- Nemets, E.A.; Kirsanova, L.A.; Basok, J.B.; Lymareva, M.V.; Schagidulin, M.J.; Sevastianov, V.I. Decellularized human liver tissue fragments to create cell- and tissue-engineered liver constructs. Curr. Trends Biomed. Eng. Biosci. 2017, 8, 555747. [Google Scholar]
- Baiocchini, A.; Montaldo, C.; Conigliaro, A.; Grimaldi, A.; Correani, V.; Mura, F.; Ciccosanti, F.; Rotiroti, N.; Brenna, A.; Montalbano, M.; et al. Extracellular matrix molecular remodeling in human liver fibrosis evolution. PLoS ONE 2016, 11, e0151736. [Google Scholar] [CrossRef] [PubMed]
- Peloso, A.; Urbani, L.; Cravedi, P.; Katari, R.; Maghsoudlou, P.; Fallas, M.E.; Sordi, V.; Citro, A.; Purroy, C.; Niu, G.; et al. The human pancreas as a source of protolerogenic extracellular matrix scaffold for a new-generation bioartificial endocrine pancreas. Ann. Surg. 2016, 264, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Sackett, S.D.; Tremmel, D.M.; Ma, F.; Feeney, A.K.; Maguire, R.M.; Brown, M.E.; Zhou, Y.; Li, X.; O’Brien, C.; Li, L.; et al. Extracellular matrix scaffold and hydrogel derived from decellularized and delipidized human pancreas. Sci. Rep. 2018, 8, 10452. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.; Yang, C.W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Stewart, D.E.; Garcia, V.C.; Rosendale, J.D.; Klassen, D.K.; Carrico, B.J. Diagnosing the decades-long rise in the deceased donor kidney discard rate in the United States. Transplantation 2017, 101, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Orlando, G.; Booth, C.; Wang, Z.; Totonelli, G.; Ross, C.L.; Moran, E.; Salvatori, M.; Maghsoudlou, P.; Turmaine, M.; Delario, G.; et al. Discarded human kidneys as a source of ECM scaffold for kidney regeneration technologies. Biomaterials 2013, 34, 5915–5925. [Google Scholar] [CrossRef] [PubMed]
- Bombelli, S.; Meregalli, C.; Scalia, C.; Bovo, G.; Torsello, B.; De Marco, S.; Cadamuro, M.; Viganò, P.; Strada, G.; Cattoretti, G.; et al. Nephrosphere-derived cells are induced to multilineage differentiation when cultured on human decellularized kidney scaffolds. Am. J. Pathol. 2018, 188, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Song, J.J.; Guyette, J.P.; Gilpin, S.E.; Gonzalez, G.; Vacanti, J.P.; Ott, H.C. Regeneration and experimental orthotopic transplantation of a bioengineered kidney. Nat. Med. 2013, 19, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Peloso, A.; Petrosyan, A.; Da Sacco, S.; Booth, C.; Zambon, J.P.; O’Brien, T.; Aardema, C.; Robertson, J.; De Filippo, R.E.; Soker, S.; et al. Renal extracellular matrix scaffolds from discarded kidneys maintain glomerular morphometry and vascular resilience and retains critical growth factors. Transplantation 2015, 99, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Nagao, R.J.; Xu, J.; Luo, P.; Xue, J.; Wang, Y.; Kotha, S.; Zeng, W.; Fu, X.; Himmelfarb, J.; Zheng, Y. Decellularized Human Kidney Cortex Hydrogels Enhance Kidney Microvascular Endothelial Cell Maturation and Quiescence. Tissue Eng. Part A 2016, 22, 1140–1150. [Google Scholar] [CrossRef] [PubMed]
- Hiraki, H.L.; Nagao, R.J.; Himmelfarb, J.; Zheng, Y. Fabricating a Kidney Cortex Extracellular Matrix-Derived Hydrogel. J. Vis. Exp. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Yoo, J.J.; Atala, A. Experimental and clinical experience using tissue regeneration for urethral reconstruction. World J. Urol. 2000, 18, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Pokrywczynska, M.; Gubanska, I.; Drewa, G.; Drewa, T. Application of bladder acellular matrix in urinary bladder regeneration: The state of the art and future directions. Biomed. Res. Int. 2015, 2015, 613439. [Google Scholar] [CrossRef] [PubMed]
- Dahms, S.E.; Piechota, H.J.; Dahiya, R.; Lue, T.F.; Tanagho, E.A. Composition and biomechanical properties of the bladder acellular matrix graft: Comparative analysis in rat, pig and human. Br. J. Urol. 1998, 82, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Sievert, K.D.; Fandel, T.; Wefer, J.; Gleason, C.A.; Nunes, L.; Dahiya, R.; Tanagho, E.A. Collagen I:III ratio in canine heterologous bladder acellular matrix grafts. World J. Urol. 2006, 24, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Del Vento, F.; Vermeulen, M.; de Michele, F.; Giudice, M.G.; Poels, J.; des Rieux, A.; Wyns, C. Tissue engineering to improve immature testicular tissue and cell transplantation outcomes: One step closer to fertility restoration for prepubertal boys exposed to gonadotoxic treatments. Int. J. Mol. Sci. 2018, 19, 286. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, M.; Del Vento, F.; de Michele, F.; Poels, J.; Wyns, C. Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality. Int. J. Mol. Sci. 2018, 19, 227. [Google Scholar] [CrossRef] [PubMed]
- Baert, Y.; Stukenborg, J.B.; Landreh, M.; De Kock, J.; Jörnvall, H.; Söder, O.; Goossens, E. Derivation and characterization of a cytocompatible scaffold from human testis. Hum. Reprod. 2015, 30, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Baert, Y.; De Kock, J.; Alves-Lopes, J.P.; Söder, O.; Stukenborg, J.B.; Goossens, E. Primary human testicular cells self-organize into organoids with testicular properties. Stem Cell Rep. 2017, 8, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.G.; Filho, A.M.; Damião, R.; da Silva, E.A. Human acellular matrix graft of tunica albuginea for penile reconstruction. J. Sex. Med. 2011, 8, 3196–3203. [Google Scholar] [CrossRef] [PubMed]
- Egydio, F.M.; Filho Freitas, L.G.; Sayeg, K.; Laks, M.; Oliveira, A.S.; Almeida, F.G. Acellular human glans extracellular matrix as a scaffold for tissue engineering: In vitro cell support and biocompatibility. Int. Braz. J. Urol. 2015, 41, 990–1001. [Google Scholar] [CrossRef] [PubMed]
- Kajbafzadeh, A.M.; Abbasioun, R.; Sabetkish, N.; Sabetkish, S.; Habibi, A.A.; Tavakkolitabassi, K. In vivo human corpus cavernosum regeneration: Fabrication of tissue-engineered corpus cavernosum in rat using the body as a natural bioreactor. Int. Urol. Nephrol. 2017, 49, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Kajbafzadeh, A.M.; Abbasioun, R.; Sabetkish, S.; Sabetkish, N.; Rahmani, P.; Tavakkolitabassi, K.; Arshadi, H. Future Prospects for Human Tissue Engineered Urethra Transplantation: Decellularization and Recellularization-Based Urethra Regeneration. Ann. Biomed. Eng. 2017, 45, 1795–1806. [Google Scholar] [CrossRef] [PubMed]
- Cazzaniga, W.; Nebuloni, M.; Longhi, E.; Locatelli, I.; Allevi, R.; Lucianò, R.; Senatore, G.; Ventimiglia, E.; Cucchiara, V.; Genovese, L.; et al. Human Prostate Tissue-derived Extracellular Matrix as a Model of Prostate Microenvironment. Eur. Urol. Focus 2016, 2, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Hassanpour, A.; Talaei-Khozani, T.; Kargar-Abarghouei, E.; Razban, V.; Vojdani, Z. Decellularized human ovarian scaffold based on a sodium lauryl ester sulfate (SLES)-treated protocol, as a natural three-dimensional scaffold for construction of bioengineered ovaries. Stem Cell Res. Ther. 2018, 9, 252. [Google Scholar] [CrossRef] [PubMed]
- Young, R.C.; Goloman, G. Allo- and xeno-reassembly of human and rat myometrium from cells and scaffolds. Tissue Eng. Part A 2013, 19, 2112–2119. [Google Scholar] [CrossRef] [PubMed]
- Olalekan, S.A.; Burdette, J.E.; Getsios, S.; Woodruff, T.K.; Kim, J.J. Development of a novel human recellularized endometrium that responds to a 28-day hormone treatment. Biol. Reprod. 2017, 96, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Chen, G.; Liu, J.; Ti, D.; Zhao, Y.; Xu, S.; Fu, X.; Han, W. Culturing on Wharton’s jelly extract delays mesenchymal stem cell senescence through p53 and p16INK4a/ pRb pathways. PLoS ONE 2013, 8, e58314. [Google Scholar] [CrossRef] [PubMed]
- Jadalannagari, S.; Converse, G.; McFall, C.; Buse, E.; Filla, M.; Villar, M.T.; Artigues, A.; Mellot, A.J.; Wang, J.; Detamore, M.S.; et al. Decellularized Wharton’s Jelly from human umbilical cord as a novel 3D scaffolding material for tissue engineering applications. PLoS ONE 2017, 12, e0172098. [Google Scholar] [CrossRef] [PubMed]
- Beiki, B.; Zeynali, B.; Seyedjafari, E. Fabrication of a three dimensional spongy scaffold using human Wharton’s jelly derived extra cellular matrix for wound healing. Mater. Sci. Eng. C. Mater. Biol. Appl. 2017, 78, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Converse, G.L.; Li, D.; Buse, E.E.; Hopkins, R.A.; Aljitawi, O.S. Wharton’s Jelly Matrix Decellularization for Tissue Engineering Applications. Methods Mol. Biol. 2017. [Google Scholar] [CrossRef]
- Wildman, D.E. Review: Toward an integrated evolutionary understanding of the mammalian placenta. Placenta 2011, 32, S142–S145. [Google Scholar] [CrossRef] [PubMed]
- Flynn, L.; Semple, J.L.; Woodhouse, K.A. Decellularized placental matrices for adipose tissue engineering. J. Biomed. Mater. Res A 2006, 79, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Flynn, L.; Prestwich, G.D.; Semple, J.L.; Woodhouse, K.A. Adipose tissue engineering with naturally derived scaffolds and adipose-derived stem cells. Biomaterials 2007, 28, 3834–3842. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, J.D.; Yoon, H.S.; Cho, Y.W. Full-thickness skin wound healing using human placenta-derived extracellular matrix containing bioactive molecules. Tissue Eng. Part A 2013, 19, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Kakabadze, A.; Kakabadze, Z. Prospect of using decellularized human placenta and cow placentome for creation of new organs: Targeting the liver (part I: Anatomic study). Transplant. Proc. 2015, 47, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Kakabadze, Z.; Mardaleishvili, K.; Loladze, G.; Javakhishvili, I.; Chakhunasvili, K.; Karalashvili, L.; Sukhitashvili, N.; Chutkerashvili, G.; Kakabadze, A.; Chakhunasvili, D. Clinical application of decellularized and lyophilized human amnion/chorion membrane grafts for closing post-laryngectomy pharyngocutaneous fistulas. J. Surg. Oncol. 2016, 113, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Francis, M.P.; Breathwaite, E.; Bulysheva, A.A.; Varghese, F.; Rodriguez, R.U.; Dutta, S.; Semenov, I.; Ogle, R.; Huber, A.; Tichy, A.M.; et al. Human placenta hydrogel reduces scarring in a rat model of cardiac ischemia and enhances cardiomyocyte and stem cell cultures. Acta Biomater. 2017, 52, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhou, Z.; Lin, H.; Wu, J.; Ginn, B.; Choi, J.S.; Jiang, X.; Elisseeff, J.H.; Yiu, S.; Mao, H.Q. Synthetic Nanofiber-Reinforced Amniotic Membrane via Interfacial Bonding. ACS Appl. Mater. Interfaces 2018, 10, 14559–14569. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, M.; Pereira, M.; Rana, H.; Stout, B.; Lewis, C.; Abramson, S.; Labazzo, K.; Ray, C.; Liu, Q.; Hofgartner, W.; et al. The Mechanism of Cell Interaction and Response on Decellularized Human Amniotic Membrane: Implications in Wound Healing. Wounds 2007, 19, 207–217. [Google Scholar] [PubMed]
- Lim, L.S.; Poh, R.W.Y.; Riau, A.K.; Beuerman, R.W.; Tan, D.; Mehta, J.S. Biological and ultrastructural properties of acelagraft, a freeze-dried γ-irradiated human amniotic membrane. Arch. Ophthalmol. 2010, 128, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Saghizadeh, M.; Winkler, M.A.; Kramerov, A.A.; Hemmati, D.M.; Ghiam, C.A.; Dimitrijevich, S.D.; Sareen, D.; Ornelas, L.; Ghiasi, H.; Brunken, W.J.; et al. A simple alkaline method for decellularizing human amniotic membrane for cell culture. PLoS ONE 2013, 8, e79632. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Sameni, M.; Radenkovic, D.; Mozafari, M.; Mossahebi-Mohammadi, M.; Seifalian, A. Decellularized human amniotic membrane: How viable is it as a delivery system for human adipose tissue-derived stromal cells? Cell Prolif. 2016, 49, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Sanluis-Verdes, A.; Sanluis-Verdes, N.; Manso-Revilla, M.J.; Castro-Castro, A.M.; Pombo-Otero, J.; Fraga-Mariño, M.; Sanchez-Ibañez, J.; Domenech, N.; Rendal-Vaszquez, M.E. Tissue engineering for neurodegenerative diseases using human amniotic membrane and umbilical cord. Cell Tissue Bank. 2017, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Motamed, M.; Sadr, Z.; Valojerdi, M.R.; Moini, A.; Oryan, S.; Totonchi, M.; Ebrahimi, B.; Maroufizadeh, S.; Taghiabadi, E. Tissue Engineered Human Amniotic Membrane Application in Mouse Ovarian Follicular Culture. Ann. Biomed. Eng. 2017, 45, 1664–1675. [Google Scholar] [CrossRef] [PubMed]
- Hasmad, H.; Yusof, M.R.; Mohd Razi, Z.R.; Hj Idrus, R.B.; Chowdhury, S.R. Human Amniotic Membrane with Aligned Electrospun Fiber as Scaffold for Aligned Tissue Regeneration. Tissue Eng. Part C Methods 2018, 24, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Gao, M.; Shen, Q.; Hou, L.; Zhu, Y.; Wang, J. Biocompatible surgical meshes based on decellularized human amniotic membrane. Mater. Sci. Eng. C 2015, 54, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Swim, M.M.; Albertario, A.; Iacobazzi, D.; Caputo, M.; Ghorbel, M.T. Amnion-based scaffold with enhanced strength and biocompatibility for in vivo vascular repair. Tissue Eng. Part A 2018. [Google Scholar] [CrossRef] [PubMed]
- Amensag, S.; McFetridge, P.S. Rolling the Human Amnion to Engineer Laminated Vascular Tissues. Tissue Eng. Part C Methods 2012, 18, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Amensag, S.; McFetridge, P.S. Tuning scaffold mechanics by laminating native extracellular matrix membranes and effects on early cellular remodeling. J. Biomed. Mater. Res. A 2014, 102, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Sharifiaghdas, F.; Moghadasali, R.; Baharvand, H.; Hosseini-Moghaddam, S.M.; Mahmoudnejad, N. Special characteristics of culturing mature human bladder smooth muscle cells on human amniotic membrane as a suitable matrix. Urol. J. 2009, 6, 283–288. [Google Scholar] [PubMed]
- Chen, C.; Zheng, S.; Zhang, X.; Dai, P.; Gao, Y.; Nan, L.; Zhang, Y. Transplantation of Amniotic Scaffold-Seeded Mesenchymal Stem Cells and/or Endothelial Progenitor Cells From Bone Marrow to Efficiently Repair 3-cm Circumferential Urethral Defect in Model Dogs. Tissue Eng. Part A 2018, 24, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Poghosyan, T.; Gaujoux, S.; Vanneaux, V.; Bruneval, P.; Domet, T.; Lecourt, S.; Jarraya, M.; Sfeir, R.; Larghero, J.; Cattan, P. In Vitro Development and Characterization of a Tissue-Engineered Conduit Resembling Esophageal Wall Using Human and Pig Skeletal Myoblast, Oral Epithelial Cells, and Biologic Scaffolds. Tissue Eng Part A 2013, 19, 2242–2252. [Google Scholar] [CrossRef] [PubMed]
- Dorazehi, F.; Nabiuni, M.; Jalali, H. Potential use of amniotic membrane—Derived scaffold for cerebrospinal fluid applications. Int. J. Mol. Cell. Med. 2018, 7, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Sun, J.; Li, X.; Mao, L.; Zhou, Y.; Cui, L.; Bai, W. Antifibrotic Effects of Decellularized and Lyophilized Human Amniotic Membrane Transplant on the Formation of Intrauterine Adhesion. Exp. Clin. Transplant. 2018. [Google Scholar] [CrossRef]
- Song, M.; Wang, W.; Ye, Q.; Bu, S.; Shen, Z.; Zhu, Y. The repairing of full-thickness skin deficiency and its biological mechanism using decellularized human amniotic membrane as the wound dressing. Mater. Sci. Eng. C 2017, 77, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Wilshaw, S.-P.; Kearney, J.; Fisher, J.; Ingham, E. Biocompatibility and Potential of Acellular Human Amniotic Membrane to Support the Attachment and Proliferation of Allogeneic Cells. Tissue Eng. Part A 2008, 14, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Samadikuchaksaraei, A.; Seifalian, A.M.; Urbanska, A.M.; Ghanbarian, H.; Hardy, J.G.; Omrani, M.D.; Mozafari, M.; Reis, R.L.; Kundu, S.C. Silk fibroin/amniotic membrane 3D bi-layered artificial skin. Biomed. Mater. 2018, 13, 035003. [Google Scholar] [CrossRef] [PubMed]
- Mallik, A.S.; Fichter, M.A.; Rieder, S.; Bilic, G.; Stergioula, S.; Henke, J.; Schneider, K.T.; Kurmanavicius, J.; Biemer, E.; Zimmermann, R.; et al. Fetoscopic closure of punctured fetal membranes with acellular human amnion plugs in a rabbit model. Obstet. Gynecol. 2007, 110, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Salah, R.A.; Mohamed, I.K.; El-Badri, N. Development of decellularized amniotic membrane as a bioscaffold for bone marrow-derived mesenchymal stem cells: Ultrastructural study. J. Mol. Histol. 2018, 49, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Mozafari, M.; Salehi, M.; Seifalian, A.; Bandehpour, M.; Ghanbarian, H.; Urbanska, A.M.; Sameni, M.; Samadikuchaksaraei, A.; Seifalian, A.M. Development of a cost-effective and simple protocol for decellularization and preservation of human amniotic membrane as a soft tissue replacement and delivery system for bone marrow stromal cells. Adv. Healthc. Mater. 2015, 4, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Bandehpour, M.; Mozafari, M.; Hashemi, A.; Ghanbarian, H.; Sameni, M.; Salimi, M.; Gholami, M.; Samadikuchaksaraei, A. Decellularized human amniotic membrane: More is needed for an efficient dressing for protection of burns against antibiotic-resistant bacteria isolated from burn patients. Burns 2015, 41, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Seifalian, A.M.; Urbanska, A.M.; Omrani, M.D.; Hardy, J.G.; Madjd, Z.; Hashemi, S.M.; Ghanbarian, H.; Brouki Milan, P.; Mozafari, M.; et al. 3D Protein-Based Bilayer Artificial Skin for the Guided Scarless Healing of Third-Degree Burn Wounds in Vivo. Biomacromolecules 2018, 19, 2409–2422. [Google Scholar] [CrossRef] [PubMed]
- Sarumathi, G.; Siti Solehah, Z.; Siti Fatmah, A.; Mohamad Nabil, I.A.; Rajasegaran, M.; Hanis Nazihah, H.; Mohd Reusmaazran Bin, Y.; Salina, H.; Haslinda, A.A.; Lokanathan, Y. Decellularization and Genipin Crosslinking of Amniotic Membrane Suitable for Tissue Engineering Applications. J. Biomater Sci. Polym. Ed. 2018, 0, 1–27. [Google Scholar] [CrossRef]
- Roy, R.; Haase, T.; Ma, N.; Bader, A.; Becker, M.; Seifert, M.; Choi, Y.H.; Falk, V.; Stamm, C. Decellularized amniotic membrane attenuates postinfarct left ventricular remodeling. J. Surg. Res. 2016, 200, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yu, K.; Bai, J.; Tian, D.; Liu, G. Experimental study of tendon sheath repair via decellularized amnion to prevent tendon adhesion. PLoS ONE 2018, 13, e0205811. [Google Scholar] [CrossRef] [PubMed]
- Van den Bogerd, B.; Ní Dhubhghaill, S.; Zakaria, N. Characterizing human decellularized crystalline lens capsules as a scaffold for corneal endothelial tissue engineering. J. Tissue Eng. Regen. Med. 2018, 12, e2020–e2028. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Williams, J.K.; Greven, M.; Walter, K.A.; Laber, P.W.; Khang, G.; Soker, S. Bioengineering endothelialized neo-corneas using donor-derived corneal endothelial cells and decellularized corneal stroma. Biomaterials 2010, 31, 6738–6745. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, C.E.; Rnjak-Kovacina, J.; Kaplan, D.L. Corneal tissue engineering: Recent advances and future perspectives. Tissue Eng. Part B Rev. 2015, 21, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.L.; Sidney, L.E.; Dunphy, S.E.; Dua, H.S.; Hopkinson, A. Corneal decellularization: A method of recycling unsuitable donor tissue for clinical translation? Curr. Eye Res. 2016, 41, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Lynch, A.P.; Ahearne, M. Strategies for developing decellularized corneal scaffolds. Exp. Eye Res. 2013, 108, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Niu, G.; Choi, J.S.; Giegengack, M.; Atala, A.; Soker, S. Bioengineered multilayered human corneas from discarded human corneal tissue. Biomed. Mater. 2015, 10, 035012. [Google Scholar] [CrossRef] [PubMed]
- Shafiq, M.A.; Gemeinhart, R.A.; Yue, B.Y.; Djalilian, A.R. Decellularized human cornea for reconstructing the corneal epithelium and anterior stroma. Tissue Eng. Part C Methods 2012, 18, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Genicio, N.; Gallo Paramo, J.; Shortt, A.J. Quantum dot labeling and tracking of cultured limbal epithelial cell transplants in vitro. Investig. Ophthalmol. Vis. Sci. 2015, 56, 3051–3059. [Google Scholar] [CrossRef] [PubMed]
- Alio del Barrio, J.L.; Chiesa, M.; Garagorri, N.; Garcia-Urquia, N.; Fernandez-Delgado, J.; Bataille, L.; Rodriguez, A.; Arnalich-Montiel, F.; Zarnowski, T.; Álvarez de Toledo, J.P.; et al. Acellular human corneal matrix sheets seeded with human adipose-derived mesenchymal stem cells integrate functionally in an experimental animal model. Exp. Eye Res. 2015, 132, 91–100. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Forest, F.; Bernard, A.; Gauthier, A.S.; Montard, R.; Peoc’h, M.; Jumelle, C.; Courrier, E.; Perrache, C.; Gain, P.; et al. Cutting and decellularization of multiple corneal stromal lamellae for the bioengineering of endothelial grafts. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6639–6651. [Google Scholar] [CrossRef] [PubMed]
- Yam, G.H.; Yusoff, N.Z.; Goh, T.W.; Setiawan, M.; Lee, X.W.; Liu, Y.C.; Mehta, J.S. Decellularization of human stromal refractive lenticules for corneal tissue engineering. Sci. Rep. 2016, 6, 26339. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Qiu, P.; Wu, F.; Zhang, W.; Teng, W.; Qin, Z.; Li, C.; Zhou, J.; Fang, Z.; Tang, Q.; et al. Construction of a Corneal Stromal Equivalent with SMILE-Derived Lenticules and Fibrin Glue. Sci. Rep. 2016, 6, 33848. [Google Scholar] [CrossRef] [PubMed]
- Bhogal, M.; Lwin, C.N.; Seah, X.-Y.; Peh, G.; Mehta, J.S. Allogeneic Descemet’s membrane transplantation enhances corneal endothelial monolayer formation and restores functional integrity following Descemet’s stripping. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4249–4260. [Google Scholar] [CrossRef] [PubMed]
- Long, J.L. Tissue engineering for treatment of vocal fold scar. Curr. Opin. Otolaryngol. Head Neck Surg. 2010, 18, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Welham, N.V.; Chang, Z.; Smith, L.M.; Frey, B.L. Proteomic analysis of a decellularized human vocal fold mucosa scaffold using 2D electrophoresis and high-resolution mass spectrometry. Biomaterials 2013, 34, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Tse, J.R.; Long, J.L. Microstructure characterization of a decellularized vocal fold scaffold for laryngeal tissue engineering. Laryngoscope 2014, 124, E326–E331. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chang, Z.; Oliveira, G.; Xiong, M.; Smith, L.M.; Frey, B.L.; Welham, N.V. Protein turnover during in vitro tissue engineering. Biomaterials 2016, 81, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Lora, L.; Grandi, F.; Sartore, L.; Tiengo, C.; Petrelli, L.; Dalzoppo, D.; Parnigotto, P.P.; Macchi, V.; et al. Partially oxidized polyvinyl alcohol conduitfor peripheral nerve regeneration. Sci. Rep. 2018, 8, 604. [Google Scholar] [CrossRef] [PubMed]
- Szynkaruk, M.; Kemp, S.W.; Wood, M.D.; Gordon, T.; Borschel, G.H. Experimental and clinical evidence for use of decellularized nerve allografts in peripheral nerve gap reconstruction. Tissue Eng. Part B Rev. 2013, 19, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.J.; Mackinnon, S.E.; Levi, A.D.; Wade, J.A.; Hunter, D.A.; Nakao, Y.; Midha, R. Cold preserved nerve allografts: Changes in basement membrane, viability, immunogenicity, and regeneration. Muscle Nerve 1998, 21, 1507–1522. [Google Scholar] [CrossRef]
- Gulati, A.K. Evaluation of acellular and cellular nerve grafts in repair of rat peripheral nerve. J. Neurosurg. 1988, 68, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Ide, C.; Tohyama, K.; Yokota, R.; Nitatori, T.; Onodera, S. Schwann cell basal lamina and nerve regeneration. Brain Res. 1983, 288, 61–75. [Google Scholar] [CrossRef]
- Haase, S.C.; Rovak, J.M.; Dennis, R.G.; Kuzon, W.M., Jr.; Cederna, P.S. Recovery of muscle contractile function following nerve gap repair with chemically acellularized peripheral nerve grafts. J. Reconstr. Microsurg. 2003, 19, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Hudson, T.W.; Zawko, S.; Deister, C.; Lundy, S.; Hu, C.Y.; Lee, K.; Schmidt, C.E. Optimized Acellular Nerve Graft Is Immunologically Tolerated and Supports Regeneration. Tissue Eng. 2004, 10, 1641–1651. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, S.E.; Hudson, A.R.; Falk, R.E.; Kline, D.; Hunter, D. Peripheral nerve allograft: An immunological assessment of pretreatment methods. Neurosurgery 1984, 14, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, S.E.; Hudson, A.R.; Falk, R.E.; Kline, D.; Hunter, D. Peripheral nerve allograft: An assessment of regeneration across pretreated nerve allografts. Neurosurgery 1984, 15, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.C.; Duhamel, R.C.; Meezan, E.; Brendel, K. Preparation of cell-free extracellular matrix from human peripheral nerve. Muscle Nerve 1982, 5, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Sondell, M.; Lundborg, G.; Kanje, M. Regeneration of the rat sciatic nerve into allografts made acellular through chemical extraction. Brain Res. 1998, 795, 44–54. [Google Scholar] [CrossRef]
- Whitlock, E.L.; Tuffaha, S.H.; Luciano, J.P.; Yan, Y.; Hunter, D.A.; Magill, C.K.; Moore, A.M.; Tong, A.Y.; Mackinnon, S.E.; Borschel, G.H. Processed allografts and type I collagen conduits for repair of peripheral nerve gaps. Muscle Nerve 2009, 39, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Rivlin, M.; Sheikh, E.; Isaac, R.; Beredjiklian, P.K. The role of nerve allografts and conduits for nerve injuries. Hand Clin. 2010, 26, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Wolford, L.M.; Rodrigues, D.B. Autogenous grafts/allografts/conduits for bridging peripheral trigeminal nerve gaps. Atlas Oral Maxillofac. Surg. Clin. N. Am. 2011, 19, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Shanti, R.M.; Ziccardi, V.B. Use of decellularized nerve allograft for inferior alveolar nerve reconstruction: A case report. J. Oral Maxillofac. Surg. 2011, 69, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Gerli, M.F.M.; Guyette, J.P.; Evangelista-Leite, D.; Ghoshhajra, B.B.; Ott, H.C. Perfusion decellularization of a human limb: A novel platform for composite tissue engineering and reconstructive surgery. PLoS ONE 2018, 13, e0191497. [Google Scholar] [CrossRef] [PubMed]
- Giatsidis, G.; Sinha, I.; Pomahac, B. Reflections on a decade of face transplantation. Ann. Surg. 2017, 265, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Duisit, J.; Maistriaux, L.; Taddeo, A.; Orlando, G.; Joris, V.; Coche, E.; Behets, C.; Lerut, J.; Dessy, C.; Cossu, G.; et al. Bioengineering a Human Face Graft: The Matrix of Identity. Ann. Surg. 2017, 266, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Pati, F.; Cho, D.W. Bioprinting of 3D Tissue Models Using Decellularized Extracellular Matrix Bioink. Methods Mol. Biol. 2017, 1612, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Aljohani, W.; Ullah, M.W.; Zhang, X.; Yang, G. Bioprinting and its applications in tissue engineering and regenerative medicine. Int. J. Biol. Macromol. 2018, 107, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Bejleri, D.; Streeter, B.W.; Nachlas, A.L.Y.; Brown, M.E.; Gaetani, R.; Christman, K.L.; Davis, M.E. A Bioprinted Cardiac Patch Composed of Cardiac-Specific Extracellular Matrix and Progenitor Cells for Heart Repair. Adv. Health Mater. 2018, 31, e1800672. [Google Scholar] [CrossRef] [PubMed]
- Mora-Boza, A.; Lopez-Donaire, M.L. Preparation of Polymeric and Composite Scaffolds by 3D Bioprinting. Adv. Exp. Med. Biol. 2018, 1058, 221–245. [Google Scholar] [CrossRef] [PubMed]
- Pati, F.; Jang, J.; Ha, D.H.; Won Kim, S.; Rhie, J.W.; Shim, J.H.; Kim, D.H.; Cho, D.W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 2014, 5, 3935. [Google Scholar] [CrossRef] [PubMed]
- Hiller, T.; Berg, J.; Elomaa, L.; Röhrs, V.; Ullah, I.; Schaar, K.; Dietrich, A.C.; Al-Zeer, M.A.; Kurtz, A.; Hocke, A.C.; et al. Generation of a 3D Liver Model Comprising Human Extracellular Matrix in an Alginate/Gelatin-Based Bioink by Extrusion Bioprinting for Infection and Transduction Studies. Int. J. Mol. Sci. 2018, 19, 3129. [Google Scholar] [CrossRef] [PubMed]
- Kuna, V.K.; Rosales, A.; Hisdal, J.; Osnes, E.K.; Sundhagen, J.O.; Bäckdahl, H.; Sumitran-Holgersson, S.; Jørgensen, J.J. Successful tissue engineering of competent allogeneic venous valves. J. Vasc. Surg. Venous Lymphat. Disord. 2015, 3, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Chauhan, V.M.; Ghaemmaghami, A.M.; Aylott, J.W. New generation of bioreactors that advance extracellular matrix modelling and tissue engineering. Biotechnol. Lett. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, D.A.; Gomide, V.; Monteiro, F.J. The role of perfusion bioreactors in bone tissue engineering. Biomatter 2012, 2, 167–175. [Google Scholar] [CrossRef] [PubMed]
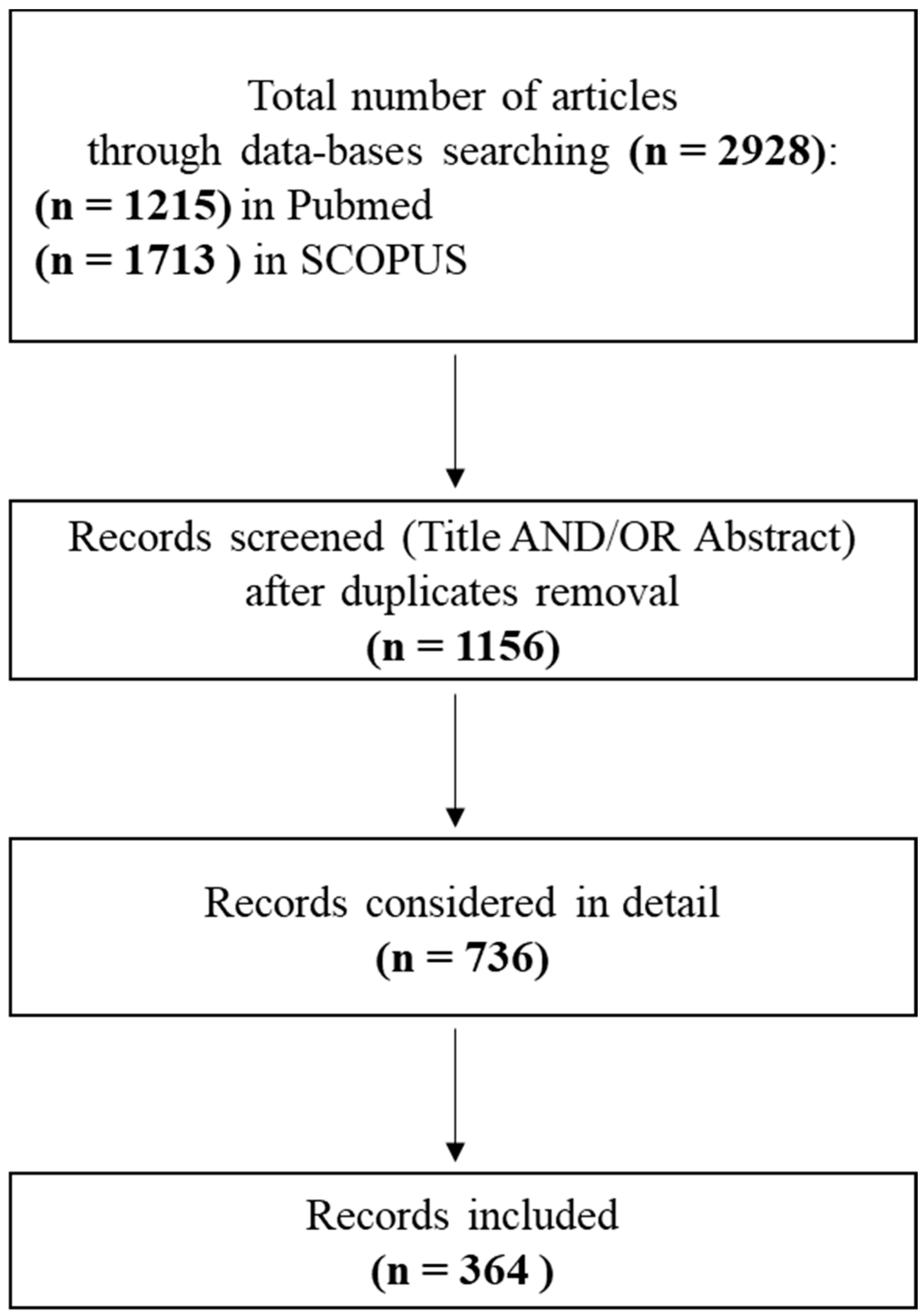
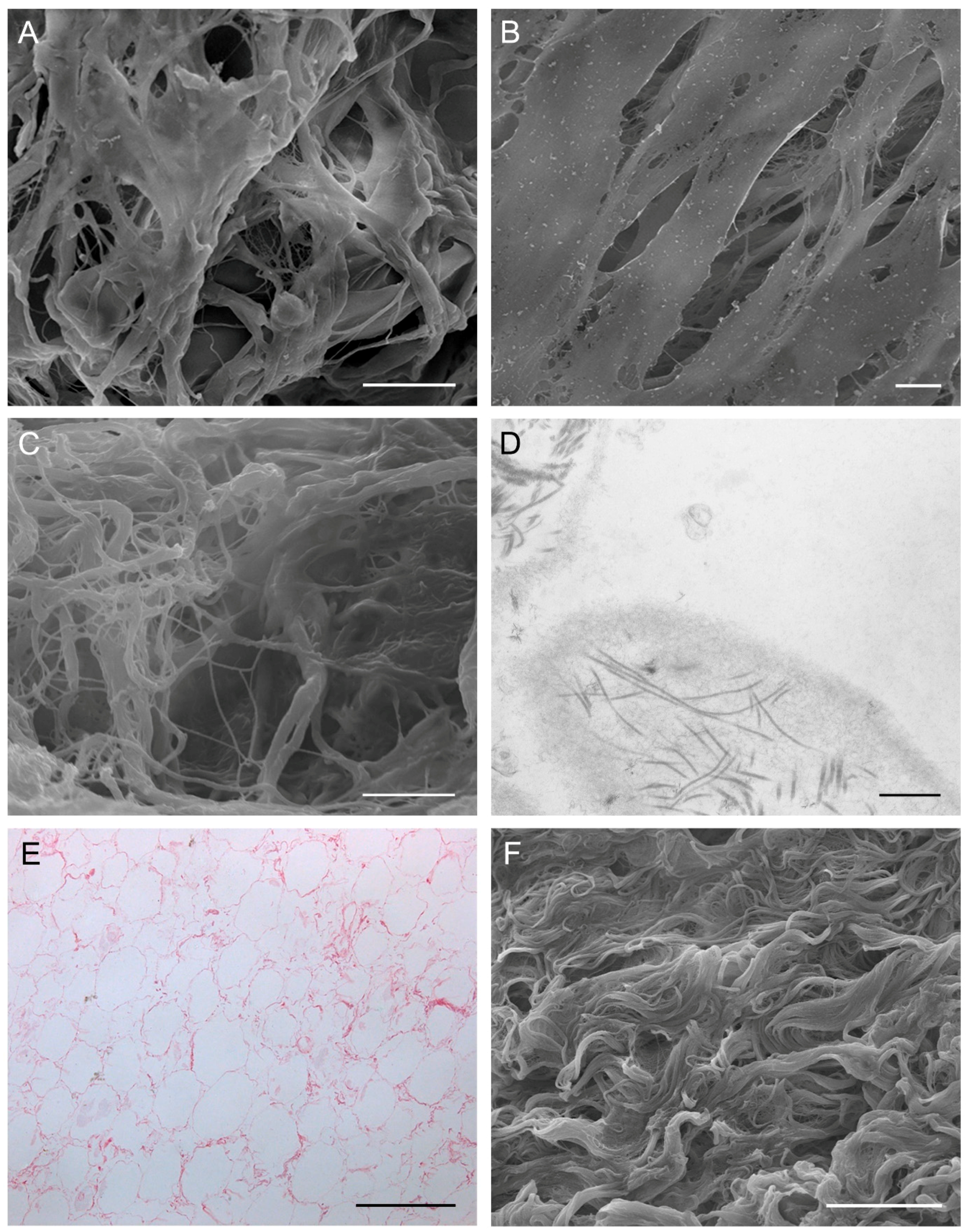
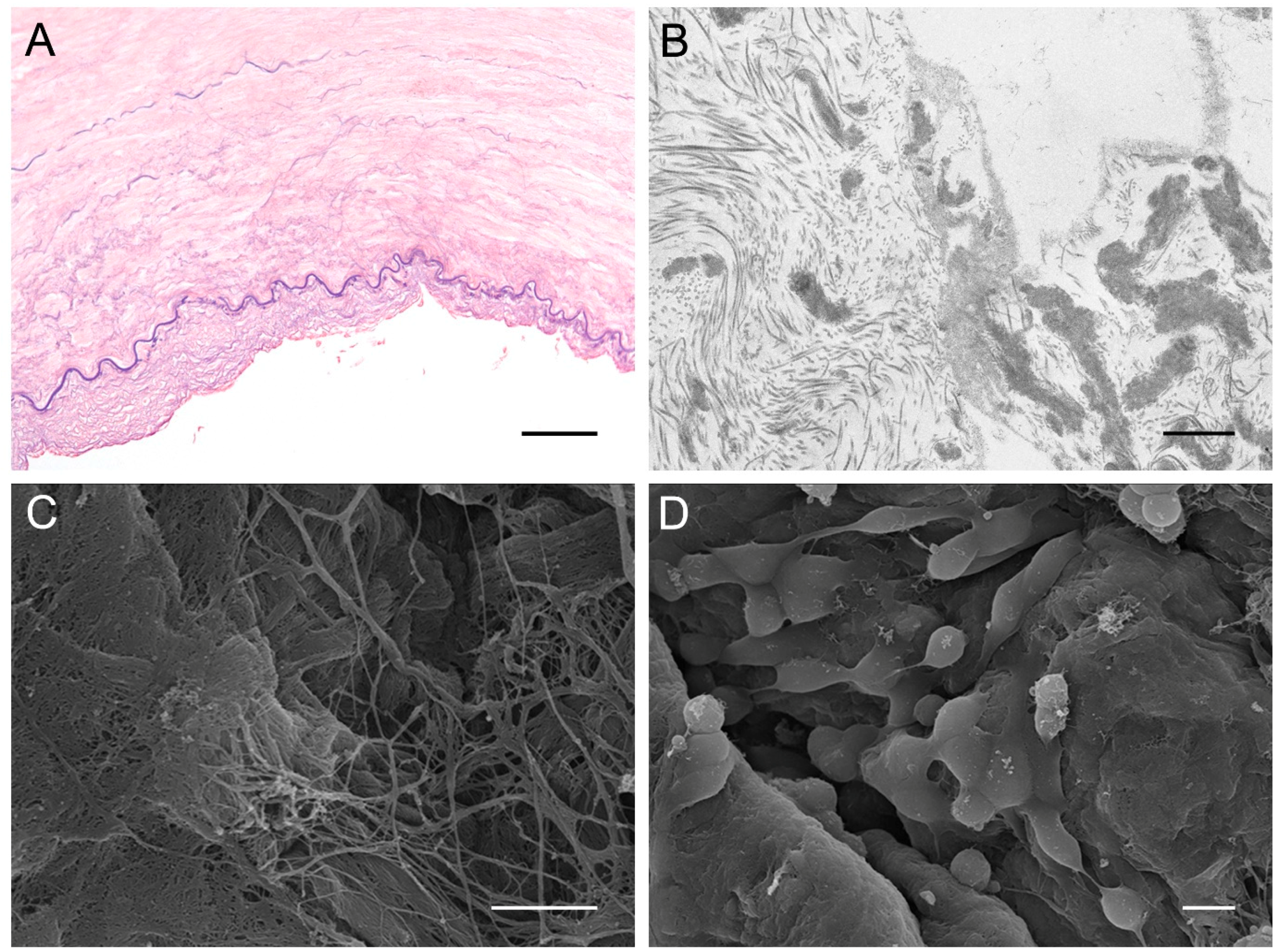
| Implanted Tissue | Pathology | Type of Scaffolds | Recipient(s) | Follow-up | Findings/Complications | Reference |
|---|---|---|---|---|---|---|
| Trachea | - End-stage bronchomalacia | Decellularized trachea by detergent enzymatic method + autologous epithelial cells and mesenchymal stem-cell-derived chondrocytes | n = 1 (age 30 years) | 4, 14 days; 2, 4 months |
| Macchiarini et al., 2008 [28] |
| 5 years Approximately every 3 months: multidetector CT scan and bronchoscopy; every 6 months mucosal biopsy samples |
| Gonfiotti et al., 2014 [37] | ||||
| Long-segment congenital tracheal stenosis and pulmonary sling | Decellularized trachea by detergent enzymatic method + bone marrow mesenchymal stem cells + patches of autologous epithelium | n = 1 (age 10 years) | 2 years |
| Elliott et al., 2012 [30] | |
| 4 years |
| Hamilton et al., 2015 [31] | ||||
| Heart valves | - Aortic or pulmonary dysfunctions | Decellularized pulmonary valves for Ross aortic valve or dysfunctional pulmonary allograft replacements | n = 36 (age 0.27–51.2 years) | 1 and 3 months |
| Elkins et al., 2001 [57] |
| Decellularized pulmonary allografts for right ventricular outflow tract reconstruction during the Ross procedure | n = 11, decellularized graft (mean age 23.0 ± 9.04 years) n = 9, cryopreserved graft (mean age 24.3 ± 8.06 years) | 5, 10 days; 1, 3, 6, 12 months |
| da Costa et al., 2005 [58] | |
| Decellularized aortic valve conduit for aortic root replacement | n = 22 (mean age 53 ± 14 years) | 1, 3 months; 1 year |
| Zehr et al., 2005 [59] | |
| Congenital pulmonary valve failure | Pulmonary heart valves engineered with autologous endothelial progenitor cells | 2 pediatric patients (age 11 and 13 years) | 3, 6, 12, 18, 24, 30, 36, and 42 months |
| Cebotari et al., 2006 [60] | |
| Decellularized pulmonary allografts in patients undergoing a Ross procedure | n = 68 (mean age 30.3 ± 11.2 years) | 4 years |
| Costa et al., 2007 [61] | |
| Isolated aortic valve disease or multilevel left ventricular outflow tract obstruction | Decellularized pulmonary conduit implanted during Ross operation with right ventricular outflow tract reconstruction | n = 183 (mean age 23.3 ± 15.2 years): n = 156, cryopreserved homograft n = 22, decellularized homograft n = 5, bovine jugular vein | 5.7 ± 3.3 years (mean follow-up) |
| Brown et al., 2008 [62] | |
| Decellularized aortic valve conduit for aortic root replacement | n = 41 (mean age 34 years) | 19 months |
| da Costa et al., 2010 [63] | |
| Decellularized pulmonary allografts in patients undergoing a Ross procedure | n = 63 (mean age 28.6 ± 16.0 years): n = 29, decellularized allograft n = 34, standard cryopreserved allograft | 4.9 ± 2.7 years (mean follow-up) |
| Brown et al., 2011 [64] | |
| Decellularized pulmonary homografts | n = 38, decellularized homograft (mean age 16.4 ± 11.4 years) n = 38, cryopreserved homograft (mean age 16.6 ± 11.3 years) n = 38, bovine jugular vein (mean age 17.9 ± 12.5 years) | 5 years |
| Cebotari et al., 2011 [65] | |
| Cellularized and decellularized aortic/pulmonary allografts for aortic valve replacement | n = 6, cellularized allograft (median age 59 years) n = 6, decellularized allograft (median age 38.6 years) | 5, 10 days; 1, 3 months | Decellularized grafts elicited lower levels of anti-HLA class I and II antibody formation after implantation than cellularized allografts | Kneib et al., 2012 [66] | |
| Congenital pulmonary valve malformations | Decellularized pulmonary homografts for pulmonary valve replacement | n = 93, decellularized homograft (mean age 15.8 ± 10.21 years) n = 93, cryopreserved homograft (mean age 15.9 ± 10.4 years) n = 93, bovine jugular vein conduit (mean age 15.6 ± 9.9 years) | 10 years | Reduced reoperation rates for decellularized pulmonary homografts in comparison with cryopreserved and bovine homograft | Sarikouch et al., 2016 [67] | |
| Decellularized pulmonary allografts for pulmonary valve replacement | n = 163, decellularized allograft (mean age 207.6 ± 197.8 months) n = 124, standard allograft (mean age 151.5 ± 171.5 months) | 60.1 ± 37.1 months (mean follow-up) |
| Bibevski et al., 2017 [68] | |
| Decellularized pulmonary valves | n = 5 adult patients n = 1 pediatric patients | 3, 6 months; 1, 2, 3 years |
| Ozawa et al., 2018 [69] | |
| Vessels-pulmonary artery | Right ventricular outflow tract reconstruction in neonates and infants | MatrACELL (LifeNet Health, Inc.) supplied in three forms:
| n = 44 (mean age 290 ± 343 days) n = 26, thin patches n = 1, 2 thin patches n = 10, thick patches n = 1, thick and thin patch n = 9, hemipulmonary artery | 230.3 days |
| Lofland et al., 2011 [70] |
| Congenital cardiac reconstructions | MatrACELL | n = 108 (mean age 367 ± 655 days) | 687 patient-days |
| Hopkins et al., 2014 [71] | |
| Vessels-iliac or mammary artery | Hepatic portal vein thrombosis | Human iliac or mammary veins from deceased organ donors Decellularization by 1% Triton X-100 and 1% tri-n-butyl phosphate and 4 mg/L DNAse. Recellularization by autologous blood-perfusion. | n = 2 (4 years and 2 years) | 1 year and 9 months 1 year and 7 months |
| Olausson et al., 2014 [72] |
| Cadaver vein | Patients with renal failure without adequate vasculature for creation of a native arteriovenous fistula | Placement of an synergraft-processed cadaver vein allograft for hemodialysis access. | n = 20 | 1 year |
| Madden et al., 2002 [73] |
| Bladder | Hypospadias | Decellularized bladder submucosal, collagen matrix for urethral reconstruction | n = 4 (4–20 years) | 22 months |
| Atala et al., 1999 [74] |
| Urethral strictures | Decellularized bladder submucosal collagen matrix for urethral reconstruction | n = 28 (mean age 40.4 years) | 36, 48 months |
| El-Kassaby et al., 2003 [75] | |
| Myelomeningocele resulting in a poorly compliant bladder | Decellularized bladder matrix seeded with autologous urothelial and muscle cells | n = 7 (4–19 years) | 22, 61 months |
| Atala et al., 2006 [76] | |
| Urethral strictures | Decellularized bladder submucosal, collagen matrix for urethral reconstruction | n = 30 (mean age 36.2 years): n = 15, buccal mucosa-derived graft n = 15, bladder-derived graft | 18, 6 months |
| El-Kassaby et al., 2008 [77] | |
| Amniotic membrane | Chronic nonhealing recalcitrant diabetic partial- or full-thickness foot ulcers. | Biovance (Celgene Cellular Therapeutics) | n = 13 patients (+1) * (mean age 63.4 years) * 1 patient received Biovance on bilateral feet on separate occasions so that was considered as 2 separate subjects. | 12 weeks | - Nine completed the study without deviation/adverse reactions n = 5, 100% wound closure; n = 3, significant decrease in wound size (i.e., 50 to 80%); n = 1, <50% wound clousure | Letendre et al., 2009 [78] |
| Cornea | Keratoconus | Decellularized corneal stromal lamina seeded with autologous adipose-derived adult stem cells | n = 9 (mean age: 34 years): n = 5, acellular graft n = 4, recellularized graft | 1 day; 1 week; 1, 3, 6 months | General improvement of:
| Alió del Barrio et al., 2018 [79] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In vivo Implant | References |
|---|---|---|---|---|---|
| Tibialis anterior and abdominal rectus muscle | 4% SDS + DNase I | Uniaxial tensile tests | - | Rabbits: surgical defect of the abdominal wall | Porzionato et al., 2015 [14] |
| 0.05% trypsin and 0.02% EDTA + 2% Triton X-100 and 0.8% NH4OH (*) | |||||
| Rectus femoris and supraspinatus muscles | 1% SDS with 1 % EDTA in Tris–HCl + DNase/RNase buffer (1 kU/mL) | Uniaxial tensile tests | - | Outbred mice: dorsal subcutaneous pouch | Wilson et al., 2016 [86] |
| Hemidiaphgram | Freezing at −80 °C | - | - | Dog: native diaphgram replacement | Davari et al., 2016 [87] |
| dH2O + 4% sodium deoxycholate and 4000 KU of DNase-1 in 1 mol/L NaCl (*) | |||||
| Upper limb flexor tendons | 0.1% EDTA + 1% Triton X-100 and 0.1% EDTA | Uniaxial tensile tests | Seeding of human dermal fibroblasts | - | Pridgen et al., 2011 [88] |
| 0.1% EDTA + 1% Tri-n-butyl-phosphate and 0.1% EDTA | |||||
| 0.1% EDTA + 1% SDS and 0.1% EDTA | |||||
| 0.1% EDTA + 0.1% SDS and 0.1% EDTA (*) | |||||
| Upper limb flexor tendons | 0.1% EDTA + 0.1% SDS and 0.1% EDTA (2%, 5%, and 10% peracetic acid solutions) | Uniaxial tensile tests | Seeding of human dermal fibroblasts | - | Woon et al., 2011 [89] |
| Upper limb flexor tendons | 0.1% EDTA + 0.1% SDS | Uniaxial tensile tests | - | Rats: dorsal subcutaneous tissue anchored to spinal ligaments (immunogenicity test) | Raghavan et al., 2012 [90] |
| Upper limb flexor tendons | 0.1% EDTA + 0.1% SDS and 0.1% EDTA | Uniaxial tensile tests on unseeded and reseeded tendons after 2 or 4 weeks from surgical implantation | Seeding of adipose-derived mesenchymal stem cells | Athymic rats: unseeded and reseeded sutured tendons in dorsal subcutaneous tissue | Schmitt et al., 2013 [91] |
| Upper limb flexor tendons | 0.1% EDTA + 0.1% SDS + DNase and 1M NaCl | - | Injection of adipose-derived mesenchymal stem cells | - | Martinello et al., 2014 [92] |
| Upper limb flexor tendons | 0.1% EDTA + 0.1% SDS and 0.1% EDTA | - | Seeding or injection with FBS or hydrogel of adipose-derived mesenchymal stem cells | - | Long et al., 2017 [93] |
| Upper limb flexor tendons from cadavers | Trypsin and 0.05% EDTA | - | Seeding on coated sutures of bone marrow-derived mesenchymal stem cells | - | Le et al., 2018 [94] |
| Flexor digitorum profundus tendon with attached distal phalanx | 5% peracetic acid + 0.1% EDTA + 0.1% SDS | Uniaxial tensile tests | - | - | Bronstein et al., 2013 [97] Fox et al., 2013 [98] |
| ultrasonication (3 min) | |||||
| ultrasonication (10 min) | |||||
| ultrasonication (10 min) + 5% peracetic acid + 0.1% EDTA + 0.1% SDS (*) | |||||
| Achilles tendon | hypotonic aqueous solutions + trypsin digestion + peracetic acid + Triton X-100 | Tensile testing | Seeding of NIH 3T3 | Rabbits: anterior cruciate ligament reconstruction | Whitlock et al., 2012 [95] |
| Flexor digitorum profundus, superficialis, and pollicis longus tendons from fresh-frozen, human cadaveric hands and upper extremities | 0.1% EDTA + 0.1% EDTA and 0.1% SDS + lyophilized + milled + pepsin/hydrochloric acid + gelation | - | - | Rats: full-thickness defect within the mid-substance of each Achilles tendon | Chiou et al., 2015 [96] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|---|
| Subcutaneous adipose tissue from abdomen, breast, omentum, pericardial depot, thymic remnants | Freezing–thawing cycles + 0.25% trypsin and 0.1% EDTA + 99.9 2-propanol + 8 g/L NaCl, 200 mg/L KCl, 1 g/L Na2HPO4, 200 mg/L KH2PO4 + 55 mM Na2HPO4, 17 mM KH2PO4, 4.9 mM MgSO4, DNase, RNase, lipase | Indentation and compression tests | Static/dynamic seeding on ECM fragments/microcarriers/bead foams or encapsulation into hydrogel scaffolds of:
| Rats and athymic mice: subcutaneous implantation | Flynn, 2010 [104] Turner and Flynn, 2012 [107] Turner et al., 2012 [108] Porzionato et al., 2013 [13] Yu et al., 2013 [112] Omidi et al., 2014 [102] Cheung et al., 2014 [113] Han et al., 2015 [114] Brown et al., 2015 [115] Baker et al., 2017 [103] Yu et al., 2017 [116] Shridhar et al., 2017 [117] Morissette Martin et al., 2018 [119] |
| 1% SDS + 2.5 mM sodium deoxycholate, lipase, and colipase | - | Seeding on hydrogel scaffolds of human adipose-derived stem cells | Athymic mice: injection of acellular adipose matrix in the subcutaneous dorsal region | Young et al., 2011 [106] | |
| 2.5 mM sodium deoxycholate + lipase and colipase | |||||
| Mechanical agitation + 0.1, 1, 3, 5% peracetic acid + 1% Triton X-100 in 2 mM EDTA + 600 U/mL DNase | - | Seeding on hydrogel ECM of human adipose-derived stem cells | Rats: subcutaneous injection | Wu et al., 2012 [109] | |
| Adipose tissue surgically sampled from abdomen, breast and forearm | Ultrasonic homogenization + 0.25% pancreatin | - | Culture of human adipose-derived stem cells with ECM microparticles | Nude mice: subcutaneous implantation of cell/scaffold complexes | Wang et al., 2013a [110] |
| Freezing–thawing cycles + 0.5-1 M NaCl + 0.25% trypsin/EDTA + 1% Triton X-100 | - | Seeding on adipose tissue microparticles of human adipose-derived stem cells | Nude rats: subcutaneous injection | Wang et al., 2013b [111] | |
| Decellularization according to Flynn, 2010 [104] | - | - | - | Sano et al., 2014 [126] | |
| Freezing–thawing cycles | |||||
| DNase + 4% SDS + 1% sodium azide | |||||
| 1% Triton X-100 + DNase | |||||
| Freezing–thawing cycles + 0.25% trypsin/EDTA + 1% Triton X-100 + isopropanol | - | Seeding of breast cancer cells | - | Dunne et al., 2014 [127] | |
| 1% SDS + 2.5 mM sodium deoxycholate + 100 µg/ml lipase and 50 ng/ml colipase | Compression tests | Encapsulation into the composite hydrogels of rat adipose-derived stem cells | Rats: subcutaneous tissue | Kayabolen et al., 2017 [118] | |
| Freezing–thawing cycles + 0.5% trypsin + 99.9% isopropanol + 500 U/mL Benzonase® | - | - | Mice: subcutaneous dorsal tissue | He et al., 2018 [120] | |
| Decellularization according to Flynn, 2010 [104] | - | - | GFP+ transgenic mice: subcutaneous tissue | Thomas-Porch et al., 2018 [121] | |
| 1 M NaCl + 1 mM EDTA + Lysis buffer (1% tergitol type NP-40, 0.1% SDS, 5 mM EDTA, 0.4M NaCl, 50 mM Tris-HCl pH 8, 1 mM PMSF) | |||||
| Homogenization + 2 M urea buffer | |||||
| Subcutaneous adipose tissue from cadavers | Organic solvent + surfactant/ethanol based solution | - | - | Immunocompetent and athimic mice: subcutaneous tissue | Giatsidis et al., 2018 [125] |
| Adipose tissue from cosmetic, abdominoplasty and liposuction surgery | Homogenization + freezing in 2 M urea buffer + 0.5% pepsin in 0.5 M acetic acid | Compression tests | Encapsulation into ECM-PEG hydrogels of human adipose-derived stem cells | Mice: subcutaneous implant of non-seeded ECM-PEG hydrogels | Li et al., 2018 [122] |
| Freezing–thawing cycles + 99.9% isopropanol 0.05% trypsin/0.05% EDTA, 20 ng/mL DNase and 20 ng/ml RNase | Tensile tests | Seeding of human adipose-derived stem cells | - | Song et al., 2018 [123] | |
| Freezing–thawing cycles + homogenization + 1% Triton X-100 + 100 U/mL DNase and 100 µg/ml RNase | - | Encapsulation into ECM hydrogels of human adipose-derived stem cells | Rats: subcutaneous injection of unseeded hydrogel scaffolds | Zhao et al., 2018 [124] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implants | References |
|---|---|---|---|---|---|
| Myocardium | 10 µM Tris buffer and 0.1% EDTA + 0.5% SDS + 50 U/mL DNase and 1 U/mL RNase | Uniaxial tensile test | Seeding on composite scaffolds (acellular myocardium/fibrin hydrogel) of mesenchymal progenitor cells | Nude rat model of acute and chronic cardiac infarction: acellular and recellularized composite scaffolds as patches on the infarcted myocardium | Godier-Furnémont et al., 2011 [145] |
| Myocardium (300 µm thick-sections) | 0.5% SDS | - | Seeding of:
| - | Oberwallner et al., 2014 [138] |
| 5% Triton X-100 | |||||
| 4% sodium deoxycholate | |||||
| 10 mM Tris and 0.1% EDTA + 0.5% SDS + FBS (*) | |||||
| Myocardium | 1% deoxycholic acid + DNase and RNase | - | Injection of cord blood mononuclear cells on nanofibers-coated myocardial ECM | Sheep models of myocardial infarction: ischemic myocardial apex | Guhathakurta et al., 2014 [142] |
| Heart (whole organ) | Perfusion with: 1% SDS | Pressure–volume measurements | Seeding on slices of decellularized human leftventricle of:
| - | Sánchez et al., 2015 [137] Sánchez et al., 2016 [139] |
| Heart (whole organ) | Decellularization according to Sanchez et al., 2015 [97] | - | Seeding on myocardial left ventricle slices of cardiac-like cells derived from induced pluripotent stem cells | - | Garreta et al., 2016 [140] |
| Heart (whole organ) | Perfusion with: 1% SDS + dH2O + 1% Triton X-100 + 25 U/mL DNase | Biaxial tensile test | Perfusion of whole acellular heart / seeding of decellularized cardiac slices with cardiac myocytes derived from human induced pluripotent stem cells | Rats: decellularized myocardium fragments into subcutaneous tissue | Guyette et al., 2016 [141] |
| Myocardium | 1% SDS + DNase and RNase | - | - | - | Johnson et al., 2016 [146] |
| Myocardium (300 μm thick-slices) | 10 mM Tris and 0.1% EDTA + 0.5% SDS + FBS | - | Murine HL-1 cardiomyocyte cultures | - | Kappler et al., 2016 [143] Becker et al., 2017 [144] |
| Myocardium (350 μm thick-sections) | 10 mM Tris and 0.1% EDTA + 0.5% SDS + FBS | - | Seeding of human cardiac progenitor cells | - | Di Meglio et al., 2017 [148] |
| 10 µM Tris and 0.1% EDTA + 0.5% SDS + 50 U/mL DNase and 1 U/mL RNase | |||||
| 10 mM Tris-HCl and protease inhibitors + 0.1% SDS + 50 U/mL DNase and 1 U/mL RNase | |||||
| 1% SDS + dH2O + 1% Triton X-100 | |||||
| 1% SDS and L1% Triton X-100 (*) | |||||
| Myocardium | Decellularization according to Johnson et al., 2016 [103] | - | - | Humanized mice: injection of acellular ECM hydrogels in the subcutaneous dorsal tissue | Wang et al., 2017 [147] |
| Myocardium | Decellularization according to Becker et al., 2017 [96] | Uniaxial pulling tests | Seeding on myocardial ECM hydrogel/amniotic membrane composite scaffolds of:
| - | Becker et al., 2018 [144] |
| Pericardium | 10 mM Tris-HCl and protease inhibitors + 0.1% SDS + 50 U/mL DNase and 1 U/mL RNase | Uniaxial tensile test | - | - | Mirsadraee et al., 2006 [150] |
| Decellularization according to Mirsadraee et al., 2006 [106] | - | Seeding of human dermal fibroblasts from human skin of cadaveric donors | Mice: acellular pericardia into the subcutaneous tissue | Mirsadraee et al., 2007 [149] | |
| Decellularization according to Mirsadraee et al., 2006 [106] | Uniaxial tensile test | - | Immune-competent mice: subcutis | Vinci et al., 2013 [151] | |
| Hypotonic/hypertonic solutions + 1% SDS | - | - | Rats: injectable matrix gels into the left ventricular wall | Seif-Naraghi et al., 2011 [156] | |
| 0.1% SDS and protease inhibitors + DNase/RNase | Uniaxial compression and tensile tests | Seeding on gel scaffolds of human cardiac progenitor cells | Rats: cell-loaded scaffolds into the subcutaneous tissue | Rajabi-Zeleti et al., 2014 [152] | |
| 0.1% SDS + Triton X-100 + 0.1 mg/mL DNase | Tensile tests | Seeding on ECM/hydrogel RAD16-I scaffolds of porcine mediastinal adipose-derived progenitor cells | Porcine models of myocardial infarction: repopulated scaffolds on the ischemic myocardium | Prat-Vidal et al., 2014 [157] Gálvez-Montón et al., 2017 [158] Perea-Gil et al., 2018 [159] | |
| Acetone + Ethanol + 1 N NaOH + 7% NaCl + H2O2 | Uniaxial mechanical resistance tests | - | Rats: decellularized human pericardium patches on the abdominal aorta | van Steenberghe et al., 2017 [153] | |
| Decellularization according to van Steenberghe et al., 2017 [109] | Uniaxial mechanical resistance tests | - | In vivo implant according to van Steenberghe et al., 2017 [153] | van Steenberghe et al., 2018 [154] | |
| Decellularization according to van Steenberghe et al., 2017 [109] | - | - | Vietnamese pigs: decellularized ECM fragments/patches into subcutaneous dorsal tissue or on carotid/aorta | van Steenberghe et al., 2018 [155] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implants | References |
|---|---|---|---|---|---|
| Aortic and pulmonary valves/conduits | SynerGraft treatment: dH2O + DNase and RNase | Uniaxial tensile test | - | Patients with heart valve disfunctions | Elkins et al., 2001 [57] Zehr et al., 2005 [59] Brown et al., 2008 [62] Brown et al., 2011 [64] Bibevski et al., 2017 [68] |
| Aortic and pulmonary valves | 0.5% Trypsin and 0.2% EDTA | - | Dynamic seeding of human endothelial cells from saphenous vein | - | Cebotari et al., 2002 [168] Cebotari et al., 2006 [65] |
| Pulmonary valve conduits | 0.05% Triton 100-X, 0.05% sodium deoxycholate and 0.05% octylphenyl-polyethylene glycol + 150 IU/DNase and 100 µg/ml RNase | - | - | - | Rieder et al., 2005 [166] |
| 1% deoxycholic acid + 70/80% ethanol | - | - | Patients with heart valve disfunctions | da Costa et al., 2005 [58] Costa et al., 2007 [61] | |
| 0.1% SDS | . | . | Patients with heart valve disfunctions | Costa et al., 2007 [61] da Costa et al., 2010 [63] Kneib et al., 2012 [66] | |
| Pulmonary valves | Hypo/hypertonic solutions 0.1–1% Triton X-100 + 10 mM Sodium cholate + Benzonase® | - | Seeding of human bone marrow mesenchymal stem cells | - | Iop et al., 2009 [167] |
| 0.5% sodium deoxycholate and 0.5% SDS + 0.9% NaCl | - | - | Patients with heart valve disfunctions | Cebotari et al., 2011 [65] Sarikouch et al., 2016 [67] Ozawa et al., 2018 [69] | |
| Aortic valves | 0.5% Trypsin + 20 mg/mL RNase | - | Seeding of cardiac mesenchymal stromal cells from human auricle fragments | - | Dainese et al., 2012 [162] |
| Aortic and pulmonary valves | SynerGraft treatment: dH2O + DNase and RNase | - | - | - | Gerson et al., 2012 [169] |
| Hypo/hypertonic solutions + 0.05% Triton 100-X + Benzonase® | Torsional wave experiments | - | - | Jiao et al., 2012 [170] | |
| 1% SDS and 0.05% NaN3 | Tensile strength measurement | Dynamic perfusion of human umbilical vein endothelial cells on the luminal surface | - | Weymann et al., 2013 [171] | |
| Biohybrid aortic valves | 0.5% sodium deoxycholate and 0.5% SDS | - | Dynamic seeding of fibroblasts and endothelial cells from human saphenous vein | - | Koening et al., 2016 [163] |
| Aortic valves Aortic valves | Hypertonic salt solution + 0.05% Triton X-100 + Benzonase® | Biaxial mechanical testing | - | - | VeDepo et al., 2017 [164] |
| Hypo/hypertonic solutions 0.1–1% Triton X-100 + 10 mM Sodium cholate + Benzonase® | Seeding of human bone marrow mesenchymal stem cells | Iop et al., 2017 [165] | |||
| Aortic and pulmonary valves | 10 mM Tris, 0.1% EDTA, 10 KIU/L aprotinin + 0.1% SDS + Nuclease treatment | Uniaxial tensile test | - | Mouse: subcutis | Vafaee et al., 2018 [172] |
| Mitral valves | 0.5–1% SDS | - | Seeding of post-infarct murine bone marrow c-kit+ cells | Murine models of myocardial infarction: repopulated scaffolds on the infarcted epicardium/myocardium | Wan et al., 2017 [178] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | References |
|---|---|---|---|---|
| Gingiva | Liquid nitrogen + 1% SDS + 1% Triton X-100 | - | Seeding of rat bone marrow mesenchymal stem cells | Mahdavishahri et al., 2012 [234] |
| Liquid nitrogen + 0.1% SDS | - | Seeding of rabbit blastema cells | Naderi et al., 2013 [235] | |
| Liquid nitrogen + 0.5% SDS | ||||
| Liquid nitrogen + 1% SDS (*) | ||||
| Dental pulp | Collagenase and dispase | Stress-strain and Young’s modulus | Seeding of human osteoblast-like cells (MG-63) | Sangkert et al., 2016 [236] |
| Collagenase and dispase | - | Seeding of human osteoblast-like cells (MG-63) | Sangkert et al., 2017 [237] | |
| 2% Triton X-100 and 0.1% NH4OH | - | Seeding of human stem cells of the apical papilla cell-line | Song et al., 2017 [238] | |
| 0.01 M Tris-HCl and 1 mm EDTA followed by 3 cycles of: 1% SDS + 1% Triton X-100 (*) | ||||
| 1% SDS + 1% Triton X-100 | ||||
| 10 mM Tris and protease inhibitors + [0.1% EDTA and aprotinin (10 KIU mL-1)] and 0.03% SDS and wash in Tris-buffered saline and Tris-hydrochloric acid (50 mM) and DNase (50 U/mL) + RNase (1 U/mL) | - | Seeding of human dental pulp stem cells | Matoug-Elwerfelli M. et al., 2018 [239] | |
| Schneiderian membrane | Liquid nitrogen + 1% SDS | - | Seeding of human adipose tissue mesenchymal stem cells | Rahpeyma et al., 2014 [243] |
| Tissues | Decellularization Methods | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|
| Liver (whole organ or left lobe) | Perfusion with: dH2O + 0.025% Trypsin/EDTA + 3% Triton X-100 + 0.01%, 0.1%, 1% SDS | Seeding on cubic scaffolds of:
| Immunocompetent mice: acellular scaffolds into subcutaneous tissue or omentum | Mazza et al., 2015 [249] |
| Liver (biopsies from HCV-infected patients) | 0.5 M NaCl and 10 mM Tris Base + 1% SDS + Mechanical agitation | - | - | Baiocchini et al., 2016 [255] |
| Liver (whole organ) | Perfusion with: 4% Triton X-100 and 1% NH4OH + 0.9% NaCl + DNase solution | Seeding on ECM sections of human umbilical vein endothelial cells | - | Verstegen et al., 2017 [250] |
| Liver (fragments obtained by a mechanical homogenizer) | 1% Triton X-100 and 0.1% SDS + 2% Triton X-100 and 0.1% SDS + 3% Triton X-100 and 0.1% SDS + Mechanical agitation | - | - | Nemets et al., 2017 [254] |
| Liver (cylindrical tissue samples) | 1–0.1% Triton X-100 | - | - | Mattei et al., 2017 [251] |
| 0.1% SDS | ||||
| Liver (tissue biopsies sectioned into 50 µm-slices) | 0.01% SDS + 0.1% SDS + 0.2% SDS + 0.5% SDS + 1% Triton X-100 | Seeding on ECM gel of human induced pluripotent stem cells | - | Jaramillo et al., 2018 [253] |
| Liver (tissue biopsies sectioned into 50 µm-slices) | dH2O + 1% Triton X-100 and 0.1% SDS + 2% Triton X-100 and 0.1% SDS + 3% Triton X-100 and 0.1% SDS + High g-force agitation | Seeding on cubic scaffolds of:
| - | Mazza et al., 2017 [252] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|---|
| Pancreas (whole organ) | Perfusion with: 1% Triton X-100 and 0.1% NH4OH | Uniaxial tensile test | Seeding on scaffolds of:
| - | Peloso et al., 2016 [256] |
| Pancreas (1 cm3 specimens or homogenized tissue) | 2.5 mM sodium deoxycholate | - | Seeding on ECM hydrogel-coated plates of:
| Humanized mice: acellular ECM pre-gel into the dorsum | Sackett et al., 2018 [257] |
| Tissues | Decellularization Methods | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|
| Kidney (whole organ) | Perfusion with: 0.5% SDS | - | - | Orlando et al., 2013 [260] |
| Perfusion with: 1% SDS + 1% Triton X-100 | - | - | Song et al., 2013 [262] | |
| Perfusion with: 0.5% SDS + DNase | - | - | Peloso et al., 2015 [263] | |
| Kidney (cortex samples) | 1% SDS | Seeding on ECM gels of:
| - | Nagao et al., 2016 [264] Hiraki et al., 2018 [265] |
| Kidney (2 mm-thick tissue slices) | 0.02% Trypsin + 2% Tween-20 + 4% Sodium deoxycholate + 1% SDS | Seeding of renal stem/progenitor-like cells (nephrosphere) | - | Bombelli et al., 2018 [261] |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|---|
| Testis | 1% Triton X-100 | - | - | - | Baert et al., 2015 [272] Baert et al., 2017 [273] |
| 1% SDS (*) | Seeding of human testicular cells from orchidectomy | ||||
| 1% Triton X-100 + 1% SDS | - | ||||
| Glans | 1% Triton X-100 and 0.1% ammonium hydroxide | Uniaxial tensile test | Perfusion of rat mesenchymal stem cells | - | Egydio et al., 2015 [275] |
| Corpus cavernosum | 1% Triton X-100 + 5% SDS | - | Rats: sections of decellularized scaffold transplanted into the omentum and located into the scrotum | Kajbafzadeh et al., 2017 [276] | |
| Urethra and Corpus spongiosum | 1% Triton X-100 + 3% SDS | Uniaxial tensile test | Seeding or perfusion of preputial mesenchymal stem cells | In vivo implant according to Kajbafzadeh et al., 2017 [276] | Kajbafzadeh et al., 2017 [277] |
| Penile tunica albuginea | PEG 1000 | Uniaxial strength test | - | - | da Silva et al., 2011 [274] |
| Triton X-100 | |||||
| Prostate | 5 mM EDTA and 10% dimethyl sulfoxide + 1% TrytonX-100 + 0.5 M NaCl + 10 mM sodium cholate + 50 mM Tris-HCl + 100 U/mL Benzonase® | - | Seeding of primary prostate cancer cells | Cazzaniga et al., 2016 [278] | |
| Ovary | 1% sodium lauryl ester sulfate + DNase I | - | Seeding of rat primary ovarian cells | Immature female rats: acellular and recellularized scaffolds onto the renal fat pad after ovariectomy | Hassanpour et al., 2018 [279] |
| Myometrium | 70% Ethanol + 0.25% Trypsin/EDTA | - | Seeding of human myometrial cells | - | Young and Goloman, 2013 [280] |
| Endometrium | 0.25% Triton X-100 and 0.25% SDS + ribonuclease and DNase I | - | Seeding of human endometrial cells | - | Olalekan et al., 2017 [281] |
| Tissue | Decellularization Methods | Biomechanical tests | In Vitro Recellularization | In Vivo implant | References |
|---|---|---|---|---|---|
| Wharton’s jelly | dH2O + 4% sodium deoxycholate + 2000 KU DNase-I + homogenation in 10% acetic acid solution (2.5 M) | - | Seeding of human primary chondrocytes | - | Stocco et al., 2014 [36] |
| Hypertonic salt solution + hypotonic solution (0.005% Triton X-100) + anionic detergent (sodium lauryl) and sodium succinate + Benzonase® + 40% ethyl alcohol | - | Seeding of:
| Full-thickness parietal bone defect—craniotomy on transgenic mice. | Jadalannagari et al., 2017 [283] | |
| TBS 10 mM and 0.1% w/v EDTA + 0.03% SDS in TBS and EDTA | Compressive and tensile properties | Seeding of human fibroblasts cell line (HSF-PI 18) | Regeneration of full-thickness wound in mice | Beiki et al., 2017 [284] | |
| 0.05% Triton X-100 + hypertonic salt solution + 250 U/µL Benzonase® + N-lauroylsarcosine + ethanol solution + saline mannitol solution | - | Seeding of:
| - | Converse et al., 2017 [287] |
| Tissue | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|---|
| Placenta (Intact and highly vascularized portion) | Cyclic perfusion or soaking with these solutions: (A) hypotonic Tris solution: 10 mM Tris base, 5 mM EDTA, 1% PMSF + (B) detergent extraction: 50 mM Tris base, 1.5 M KCl, 5 mM EDTA, 1% lauroyl sarcosine, 1% PMSF (C) detergent extraction: 50 mM Tris base, 1% lauroyl sarcosine Two enzymatic digestion phases in 15,000 U DNAse Type II, 12.5 mg RNAse Type III A also occurred. | - | Seeding of primary human adipose precursor cells | - | Flynn et al., 2006 [287] |
| Placenta (Intact and highly vascularized portion) | Perfusive and diffusive protocol according to Flynn et al., 2006 [209] | - | Seeding of primary human adipose precursor cells | - | Flynn et al., 2007 [288] |
| Placenta (entire) | Homogenation in dH2O + 0.5% SDS + dH2O + 0.2% DNase (2000 U) and 200 μg/ml RNase | Tensile testing | - | Cutaneous wounds in rats | Choi et al., 2013 [289] |
| Placenta (entire) | Perfusion with: SDS 0.01% + 0.1% + 1% + dH2O + 1% Triton X-100 | - | - | - | Kakabadze and Kakabadze, 2015 [290] |
| Placental vessels | FT + Perfusion with: 1.2% NaCl (hypertonic solution) + 0.4% NaCl (hypotonic solution) + 1% Triton X-100 + 0.02% w/w EDTA + DNAse I (200 IU/mL) | Tensile testing | Seeding of human umbilical vein endothelial cells | - | Schneider et al., 2016 [9] |
| Placenta (entire) | 2% lauryl sarcosine + homogenation in 0.1 HCl with pepsin | - | Seeding of:
| Rat acute myocardial infarction model | Francis et al., 2017 [292] |
| Placenta (entire) | Perfusion with: 0.01% + 0.1% + 1% SDS + dH2O + 1% Triton X-100 | Evaluation of mechanical strength of vessels | - | Heterotopic transplantation of hepatized placenta in sheep | Kakabadze et al., 2016 [291] |
| Placental vessels | Perfusion with: | - | Seeding of macrophages | Implantation of grafts into the infrarenal aorta of rats | Schneider et al., 2018 [9] |
| 1% Triton X-100 and 0.02% w/w EDTA + heparin cross-linking | |||||
| 0.5% SDS and 0.02% w/w EDTA + heparin cross-linking |
| Tissues | Decellularization Methods | Biomechanical Tests | In Vitro Recellularization | In Vivo Implant | References |
|---|---|---|---|---|---|
| Corneal stroma (120–200 µm thickness slices) | 2% Triton X-100 and 0.1% NH4OH | Uniaxial tensile tests | Seeding of human corneal endothelial cells | - | Choi et al., 2010 [322] |
| Cornea | 0.1–1% Triton X-100 | - | Seeding of:
| - | Shafiq et al., 2012 [327] |
| 0.1–1% SDS | |||||
| liquid nitrogen + hypoxic environment | |||||
| PEG | |||||
| 10.0mM Tris + 1% Triton X-100, 1.5 M KCl, and 10.0 mM Tris + 1% SDS and 10.0 mM Tris + 1% Triton X-100 | |||||
| 1.5 M NaCl + 5 U/mL DNAse and 5 U/mL RNAse (*) | |||||
| Corneal stroma (90 µm thick sheets) | 1% SDS and protease inhibitors + 6.5 U/mL DNAse | - | Seeding of human adipose derived adult stem cells | Rabbits: recellularized corneal sheets into the cornea | Alio del Barrio et al., 2015 [329] Alió del Barrio et al., 2018 [79] |
| Cornea | 2% Triton X-100 and 0.1% NH4OH +/− 5 U/mL DNase | - | Seeding of:
| - | Zhang et al., 2015 [326] |
| 1.5 M NaCl +/− 5 U/mL Dnase | |||||
| Corneo-scleral rims | 100mM EDTA + mechanical abrasion of the epithelium | - | Transplantation of human limbal epithelial cell sheets onto the limbus of decellularised corneal scleral rims | - | Genicio et al., 2015 [328] |
| Cornea | 1.5M NaCl +/− 5 U/mL DNase and 5 U/mL Rnase | - | - | - | Wilson et al., 2016 [324] |
| 0.5% SDS +/− 5 U/mL DNase and 5 U/mL RNase | |||||
| 1% w/v Triton-X100 +/− 5 U/mL DNase and 5 U/mL RNase | |||||
| 2.4 U/mL Dispase II +/− 5 U/mL DNase and 5 U/mL Rnase | |||||
| Mechanical agitation +/− 5 U/mL DNase and 5 U/mL RNase | |||||
| Corneal stromal lenticules | 1.5 M NaCl + Mechanical agitation | - | Seeding of primary human stromal fibroblasts | Rabbits: decellularized 70 μm lenticules into a corneal stromal pocket | Yam et al., 2016 [331] |
| 0.1% SDS (*) | |||||
| 0.1% Triton X-100 | |||||
| 0.1% SDS and 0.1% Triton X-100 | |||||
| 1.5 M NaCl + 2; 5; 10 U/mL nuclease | |||||
| 1.5 M NaCl + 2; 5; 10 U/mL nuclease + 0.1% SDS | |||||
| Corneal lamellae | Mechanical agitation (sonification) | - | Seeding of human corneal endothelial cell line | - | He et al., 2016 [330] |
| freezing/thawing (liquid nitrogen/37 °C) | |||||
| freezing in liquid nitrogen + hypoxia in nitrogen | |||||
| 1.5 M NaCl + 0.02% EDTA/0.05% trypsin | |||||
| 0.1% SDS | |||||
| 1% SDS + Mechanical agitation | |||||
| 1% SDS + DNase + Mechanical agitation (*) | |||||
| Corneal stromal lenticules | 1.5 M NaCl + 5 U/mL DNAse and 5 U/mL RNAse | - | - | Rabbits: fibrin glue-adhered lenticules into the corneal stroma | Yin et al., 2016 [332] |
| Cornea | freezing/thawing | - | - | Rabbits: Descemet’s membrane transplantation into the wounded corneal endothelium | Bhogal et al., 2017 [333] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porzionato, A.; Stocco, E.; Barbon, S.; Grandi, F.; Macchi, V.; De Caro, R. Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. Int. J. Mol. Sci. 2018, 19, 4117. https://doi.org/10.3390/ijms19124117
Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R. Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. International Journal of Molecular Sciences. 2018; 19(12):4117. https://doi.org/10.3390/ijms19124117
Chicago/Turabian StylePorzionato, Andrea, Elena Stocco, Silvia Barbon, Francesca Grandi, Veronica Macchi, and Raffaele De Caro. 2018. "Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives" International Journal of Molecular Sciences 19, no. 12: 4117. https://doi.org/10.3390/ijms19124117
APA StylePorzionato, A., Stocco, E., Barbon, S., Grandi, F., Macchi, V., & De Caro, R. (2018). Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. International Journal of Molecular Sciences, 19(12), 4117. https://doi.org/10.3390/ijms19124117










