Evolution from Natural β-Carboline Alkaloids to Obtain 1,2,4,9-tetrahydro-3-thia-9-aza-fluorene Derivatives as Potent Fungicidal Agents against Rhizoctonia solani
Abstract
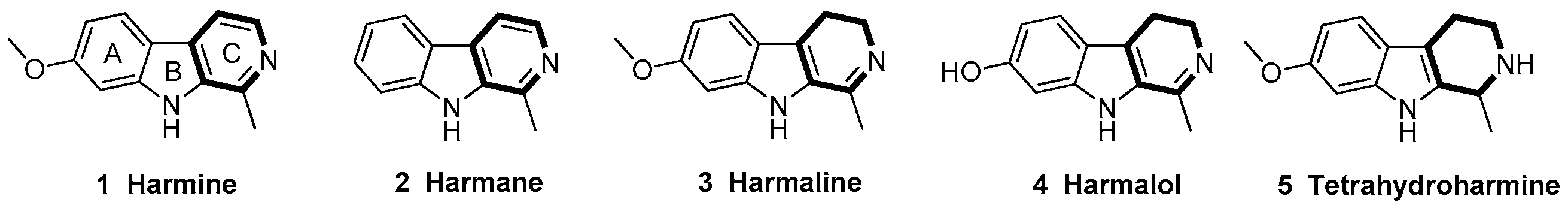
1. Introduction
2. Results and Discussion
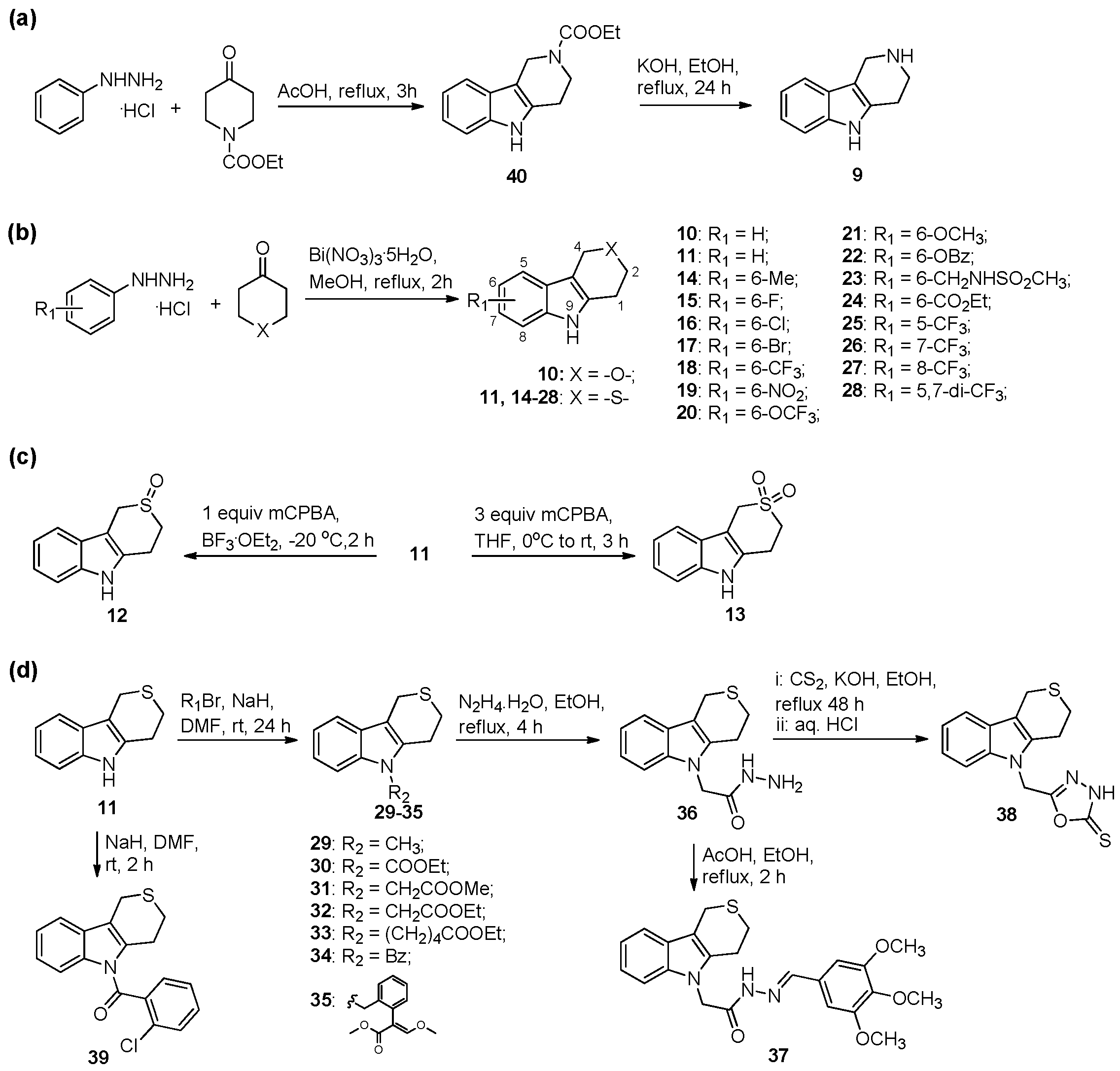
2.1. Chemistry
2.2. Biological Activity
2.2.1. In Vitro Fungicidal Activity of Compounds 6–39 against R. solani
2.2.2. In Vivo Protective and Curative Effects against Rice Sheath Blight
2.3. Preliminary Mode of Action of Compound 18 against R. solani
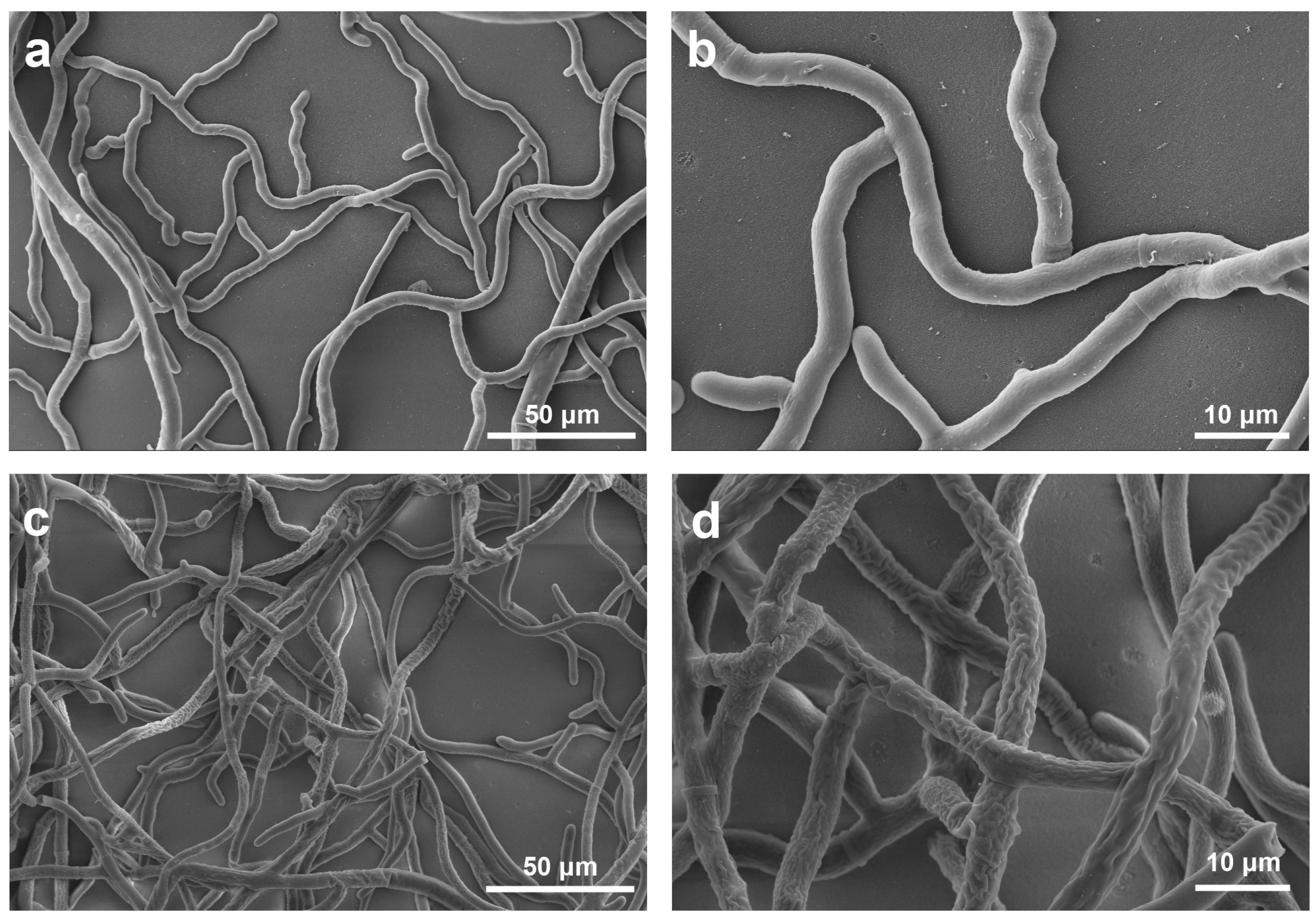
2.3.1. Effect of 18 on the hyphae morphology of R. solani
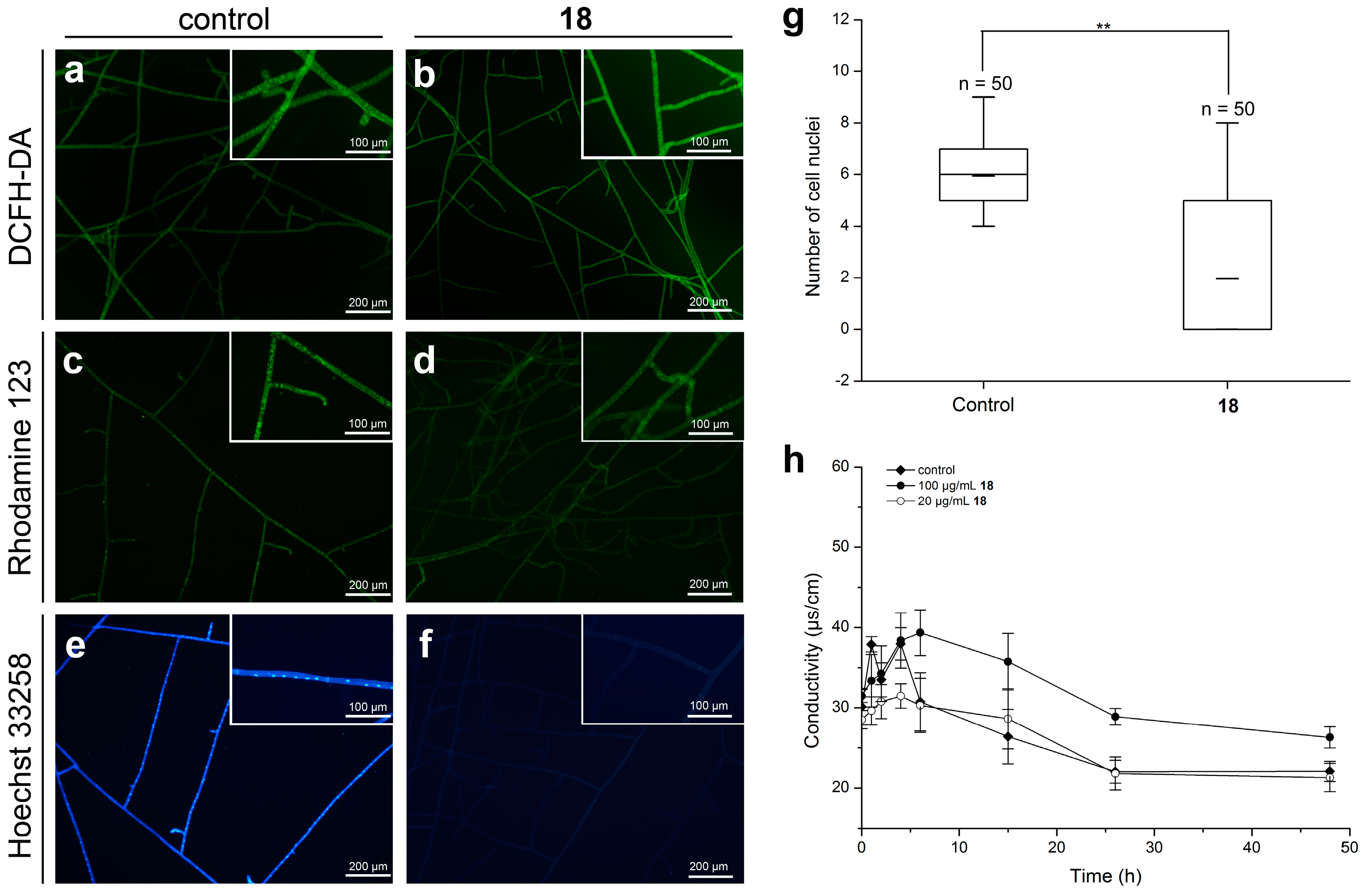
2.3.2. Effect of 18 on the Endogenous ROS Production and Cell Membrane Permeability
2.3.3. Effect of 18 on the mitochondrial membrane potential (MMP)
2.3.4. Effect of 18 on the Nuclear Morphology of R. solani
3. Materials and Methods
3.1. Chemicals
3.2. Biological Assay
3.2.1. In Vitro Fungicidal Activity against R. solani
3.2.2. In Vivo Protective Activity against R. solani using Detached Leaf Assay
3.2.3. In Vivo Protective and Curative Activities against R. solani using Greenhouse Experiment
3.3. Scanning Electron Microscopy (SEM)
3.4. Transmission Electron Microscopy (TEM)
3.5. Determination of ROS Generation
3.6. Determination of MMP
3.7. Karyological Analysis
3.8. Detection of Cell Membrane Permeability
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Miyake, I. Studien ueber die Pilze der Reispflanze in Japan. J. Coll. Agric. Tokyo 1910, 11, 237–251. [Google Scholar]
- Groth, D.E. Azoxystrobin rate and timing effects on rice sheath blight incidence and severity and rice grain and milling yields. Plant Dis. 2005, 89, 1171–1174. [Google Scholar] [CrossRef]
- Lee, F.N.; Rush, M.C. Rice sheath blight: A major rice disease. Plant Dis. 1983, 67, 829–832. [Google Scholar] [CrossRef]
- Marchetti, M.A. Potential impact of sheath blight on yield and milling quality of short-statured rice lines in the Southern United States. Plant Dis. 1983, 67, 162–165. [Google Scholar] [CrossRef]
- Boukaew., S.; Klinmanee, C.; Prasertsan, P. Potential for the integration of biological and chemical control of sheath blight disease caused by Rhizoctonia solani on rice. World J. Microbiol. Biotechnol. 2013, 29, 1885–1893. [Google Scholar] [CrossRef] [PubMed]
- Mew, T.W. Applying rice seed-associated antagonistic bacteria to manage rice sheath blight in developing countries. Plant Dis. 2004, 88, 557–564. [Google Scholar] [CrossRef]
- Zhang, C.Q.; Liu, Y.H.; Ma, X.Y.; Feng, Z.; Ma, Z.H. Characterization of sensitivity of Rhizoctonia solani, causing rice sheath blight, to mepronil and boscalid. Crop Prot. 2009, 28, 381–386. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Ann. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef]
- Rosell, G.; Quero, C.; Coll, J.; Guerrero, A. Biorational insecticides in pest management. J. Pestic. Sci. 2008, 33, 103–121. [Google Scholar] [CrossRef]
- Li, S.P.; Cheng, X.M.; Wang, C.H. A review on traditional uses, phytochemistry, pharmacology, pharmacokinetics and toxicology of the genus Peganum. J. Ethnopharmacol. 2017, 203, 127–162. [Google Scholar] [CrossRef]
- Song, H.; Liu, Y.; Liu, Y.; Wang, L.; Wang, Q. Synthesis and antiviral and fungicidal activity evaluation of β-carboline, dihydro-β-carboline, tetrahydro-β-carboline alkaloids, and their derivatives. J. Agric. Food Chem. 2014, 62, 1010–1018. [Google Scholar] [CrossRef]
- Li, Z.B.; Chen, S.H.; Zhu, S.W.; Luo, J.J.; Zhang, Y.M.; Weng, Q.F. Synthesis and fungicidal activity of β-carboline alkaloids and their derivatives. Molecules 2015, 20, 13941–13957. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Zeng, Y.; Jiang, Z.Y.; Shu, B.S.; Sethuraman, V.; Zhong, G.H. Design, synthesis, fungicidal property and QSAR studies of novel β-carbolines containing urea, benzoylthiourea and benzoylurea for the control of rice sheath blight. Pest Manag. Sci. 2018, 74, 1736–1746. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Jiang, Z.Y.; Zhu, Q.; Zhong, G.H. Discovery of β-carboline oxadiazole derivatives as fungicidal agents against rice sheath blight. J. Agric. Food Chem. 2018, 66, 9598–9607. [Google Scholar] [CrossRef] [PubMed]
- De Vreese, R.; Depetter, Y.; Verhaeghe, T.; Desmet, T.; Benoy, V.; Haeck, W.; Van Den Bosch, L.; D’hooghe, M. Synthesis and SAR assessment of novel Tubathian analogs in the pursuit of potent and selective HDAC6 inhibitors. Org. Biomol. Chem. 2016, 14, 2537–2549. [Google Scholar] [CrossRef] [PubMed]
- Khorana, N.; Purohit, A.; Herrick-Davis, K.; Teitler, M.; Glennon, R.A. gamma-Carbolines: Binding at 5-HT5A serotonin receptors. Bioorg. Med. Chem. 2003, 11, 717–722. [Google Scholar] [CrossRef]
- Liu, Y.; Song, H.; Huang, Y.; Li, J.; Zhao, S.; Song, Y.; Yang, P.; Xiao, Z.; Liu, Y.; Li, Y.; et al. Design, synthesis, and antiviral, fungicidal, and insecticidal activities of tetrahydro-β-carboline- 3-carbohydrazide derivatives. J. Agric. Food Chem. 2014, 62, 9987–9999. [Google Scholar] [CrossRef]
- Shen, Q.; Zhou, W.; Li, H.; Hu, L.; Mo, H. ROS involves the fungicidal actions of thymol against spores of Aspergillus flavus via the induction of nitric oxide. PLoS ONE 2016. [Google Scholar] [CrossRef]
- Ghosh, P.; Roy, A.; Hess, D.; Ghosh, A.; Das, S. Deciphering the mode of action of a mutant Allium sativum Leaf Agglutinin (mASAL), a potent antifungal protein on Rhizoctonia solani. BMC Microbiol. 2015, 15, 1–16. [Google Scholar] [CrossRef]
- Li, Y.; Chang, W.; Zhang, M.; Li, X.; Jiao, Y.; Lou, H. Diorcinol D exerts fungicidal action against Candida albicans through cytoplasm membrane destruction and ROS accumulation. PLoS ONE 2015. [Google Scholar] [CrossRef]
- Childress, E.S.; Kharel, Y.; Brown, A.M.; Bevan, D.R.; Lynch, K.R.; Santos, W.L. Transforming sphingosine kinase 1 inhibitors into dual and sphingosine kinase 2 selective inhibitors: Design, synthesis, and in vivo activity. J. Med. Chem. 2017, 60, 3933–3957. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Liang, X.; Jin, S.; Lv, J.; Yu, C.; Qi, W.; Li, B.; Yuan, H.; Qi, S.; Shi, Y. Primary study on mode of action for macrocyclic fungicide candidates (7B3, D1) against Rhizoctonia solani Kühn. J. Agric. Food Chem. 2010, 58, 2726–2729. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Z.; Shu, B.; Cui, G.; Zhong, G. Cytotoxic and apoptotic activity of the novel harmine derivative ZC-14 in Sf9 cells. Int. J. Mol. Sci. 2018, 19, 811. [Google Scholar] [CrossRef] [PubMed]



| Compound | Percentage Inhibition (%) | EC50 (μg/mL) | 95% CI b | |
|---|---|---|---|---|
| 100 μg/mL | 10 μg/mL | |||
| harmine (1) | 30.25 ± 1.11 | 18.28 ± 0.76 | 318.56 | 255.10–441.98 |
| harmaline (3) | 29.51 ± 1.81 | 14.45 ± 3.76 | - c | - c |
| tetrahydroharmine (5) | 28.31 ± 6.84 | 9.03 ± 1.04 | - c | - c |
| β-carboline (6) | 34.04 ± 1.21 | 16.67 ± 1.34 | 241.52 | 109.44–303.76 |
| carbazole (7) | 56.67 ± 1.69 | 26.67 ± 2.21 | - c | - c |
| tetrahydro-β-carboline (8) | 5.37 ± 5.45 | 0 | - c | - c |
| tetrahydro-γ-carboline (9) | 63.95 ± 1.33 | 10.11 ± 0.08 | 79.22 | 71.08–89.41 |
| 10 | 69.66 ± 1.21 | 22.47 ± 1.01 | 66.89 | 38.42–94.17 |
| 11 | 91.02 ± 2.31 | 32.45 ± 3.87 | 38.58 | 25.45–51.97 |
| 12 | 3.67 ± 1.69 | 0.00 | >1000 | - c |
| 13 | 6.35 ± 1.54 | 0.00 | >1000 | - c |
| validamycin A | 36.68 ± 1.09 | 18.91 ± 0.49 | 183.00 | 162.62–210.66 |
| Compound | Percentage Inhibition (%) | EC50 (μg/mL) | 95% CI c | |
|---|---|---|---|---|
| 100 μg/mL | 10 μg/mL | |||
| 14 | 68.27 ± 1.37 | 48.05 ± 2.10 | 37.02 | 15.40–52.18 |
| 15 | 90.80 ± 0.80 | 57.24 ± 2.76 | 10.55 | 0.10–19.73 |
| 16 | 89.88 ± 0.80 | 66.89 ± 2.38 | 6.84 | 1.91–13.04 |
| 17 | 93.56 ± 0.80 | 81.61 ± 2.11 | 2.94 | 0.23–6.34 |
| 18b | 88.38 ± 0.87 | 82.83 ± 0.87 | 2.35 | 0.42–5.72 |
| 19 | 89.89 ± 1.33 | 72.15 ± 3.08 | 3.79 | 0.19–7.60 |
| 20b | 75.53 ± 0.73 | 55.27 ± 2.64 | 10.86 | 2.41–24.47 |
| 21 | 68.54 ± 1.36 | 37.09 ± 1.33 | 40.15 | 16.41–71.36 |
| 22 | 8.44 ± 3.65 | 0.00 | - d | - d |
| 23b | 0.00 | 0.00 | - d | - d |
| 24 | 79.77 ± 1.69 | 43.82 ± 1.33 | 26.69 | 14.50–37.00 |
| 25b | 91.92 ± 1.75 | 52.53 ± 1.75 | 14.85 | 1.15–25.21 |
| 26b | 83.84 ± 3.15 | 40.91 ± 1.52 | 22.81 | 9.56–33.56 |
| 27b | 84.85 ± 0.00 | 59.09 ± 1.51 | 7.46 | 4.50–11.21 |
| 28b | 64.65 ± 0.87 | 28.28 ± 2.31 | 54.55 | 39.01–74.92 |
| 29 | 75.39 ± 1.21 | 36.18 ± 1.34 | 50.93 | 29.23–83.72 |
| 30b | 50.65 ± 2.25 | 42.85 ± 1.33 | 93.08 | 34.27–172.92 |
| 31b | 67.41 ± 1.69 | 38.20 ± 2.33 | 40.38 | 11.35–56.72 |
| 32b | 50.66 ± 2.97 | 20.21 ± 0.08 | 93.42 | 56.84–372.27 |
| 33b | 87.64 ± 1.63 | 49.43 ± 3.72 | 25.65 | 6.42–46.82 |
| 34 | 56.18 ± 1.69 | 38.20 ± 1.21 | 67.85 | 36.64–140.78 |
| 35b | 56.18 ± 1.33 | 29.21 ± 0.87 | 83.84 | 64.84–122.16 |
| 36b | 4.12 ± 0.71 | 0.00 | - d | - d |
| 37b | 6.33 ± 1.21 | 0.00 | - d | - d |
| 38b | 37.37 ± 0.74 | 4.12 ± 0.91 | 120.49 | 110.03–135.48 |
| 39b | 38.82 ± 1.47 | 21.94 ± 3.18 | 149.44 | 117.28–226.41 |
| Treatment | Concentration (μg/mL) | Lesion Length a (cm ± SE) | Control Efficacy (%) |
|---|---|---|---|
| 11 | 200 | 0.61 ± 0.40 ** | 92.47 |
| 100 | 1.12 ± 0.45 ** | 86.17 | |
| 14 | 200 | 0.77 ± 0.38 ** | 90.49 |
| 100 | 1.69 ± 0.55 ** | 79.14 | |
| 15 | 200 | 0.29 ± 0.19 ** | 96.42 |
| 100 | 1.31 ± 0.56 ** | 83.83 | |
| 16 | 200 | 0.17 ± 0.17 ** | 97.90 |
| 100 | 0.90 ± 0.45 ** | 88.89 | |
| 17 | 200 | 0 ** | 100 |
| 100 | 0.21 ± 0.24 ** | 97.41 | |
| 18 | 200 | 0 ** | 100 |
| 100 | 0.01 ± 0.05 ** | 99.87 | |
| 19 | 200 | 0 ** | 100 |
| 100 | 0 ** | 100 | |
| 21 | 200 | 0.71 ± 0.31 ** | 91.23 |
| 100 | 1.69 ± 0.46 ** | 79.14 | |
| validamycin A | 200 | 0 ** | 100 |
| 100 | 0 ** | 100 | |
| control | 0 | 8.10 ± 1.43 | - b |
| Treatment | Concentration (μg/mL) | Protective Effect | Curative Effect | ||
|---|---|---|---|---|---|
| Lesion Length a (cm ± SE) | Control Efficacy (%) | Lesion Length (cm ± SE) | Control Efficacy (%) | ||
| 17 | 200 | 0.92 ± 0.59 ** | 80.30 | 0.99± 0.33 ** | 78.80 |
| 100 | 1.55 ± 0.73 ** | 66.81 | 1.76 ± 0.88 ** | 62.31 | |
| 18 | 200 | 0.88 ± 0.47 ** | 81.16 | 0.86 ± 0.33 ** | 81.58 |
| 100 | 1.23 ± 0.61 ** | 73.66 | 1.14 ± 0.39 ** | 75.59 | |
| 19 | 200 | 0.96 ± 0.50 ** | 79.44 | 0.98 ± 0.40 ** | 79.01 |
| 100 | 1.63 ± 0.45 ** | 65.10 | 1.48 ± 0.50 ** | 68.31 | |
| validamycin A | 200 | 0.99 ± 0.43 ** | 78.80 | 1.13 ± 0.51 ** | 75.80 |
| 100 | 1.60 ± 0.60 ** | 65.74 | 1.52 ± 0.64** | 67.45 | |
| control | 0 | 4.67 ± 0.97 | 4.67 ± 0.97 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xi, J.; Zhang, Z.; Zhu, Q.; Zhong, G. Evolution from Natural β-Carboline Alkaloids to Obtain 1,2,4,9-tetrahydro-3-thia-9-aza-fluorene Derivatives as Potent Fungicidal Agents against Rhizoctonia solani. Int. J. Mol. Sci. 2018, 19, 4044. https://doi.org/10.3390/ijms19124044
Xi J, Zhang Z, Zhu Q, Zhong G. Evolution from Natural β-Carboline Alkaloids to Obtain 1,2,4,9-tetrahydro-3-thia-9-aza-fluorene Derivatives as Potent Fungicidal Agents against Rhizoctonia solani. International Journal of Molecular Sciences. 2018; 19(12):4044. https://doi.org/10.3390/ijms19124044
Chicago/Turabian StyleXi, Junmin, Zhijun Zhang, Qi Zhu, and Guohua Zhong. 2018. "Evolution from Natural β-Carboline Alkaloids to Obtain 1,2,4,9-tetrahydro-3-thia-9-aza-fluorene Derivatives as Potent Fungicidal Agents against Rhizoctonia solani" International Journal of Molecular Sciences 19, no. 12: 4044. https://doi.org/10.3390/ijms19124044
APA StyleXi, J., Zhang, Z., Zhu, Q., & Zhong, G. (2018). Evolution from Natural β-Carboline Alkaloids to Obtain 1,2,4,9-tetrahydro-3-thia-9-aza-fluorene Derivatives as Potent Fungicidal Agents against Rhizoctonia solani. International Journal of Molecular Sciences, 19(12), 4044. https://doi.org/10.3390/ijms19124044





