Antioxidant Properties and the Formation of Iron Coordination Complexes of 8-Hydroxyquinoline
Abstract
1. Introduction
2. Results
2.1. Mass Spectrometry
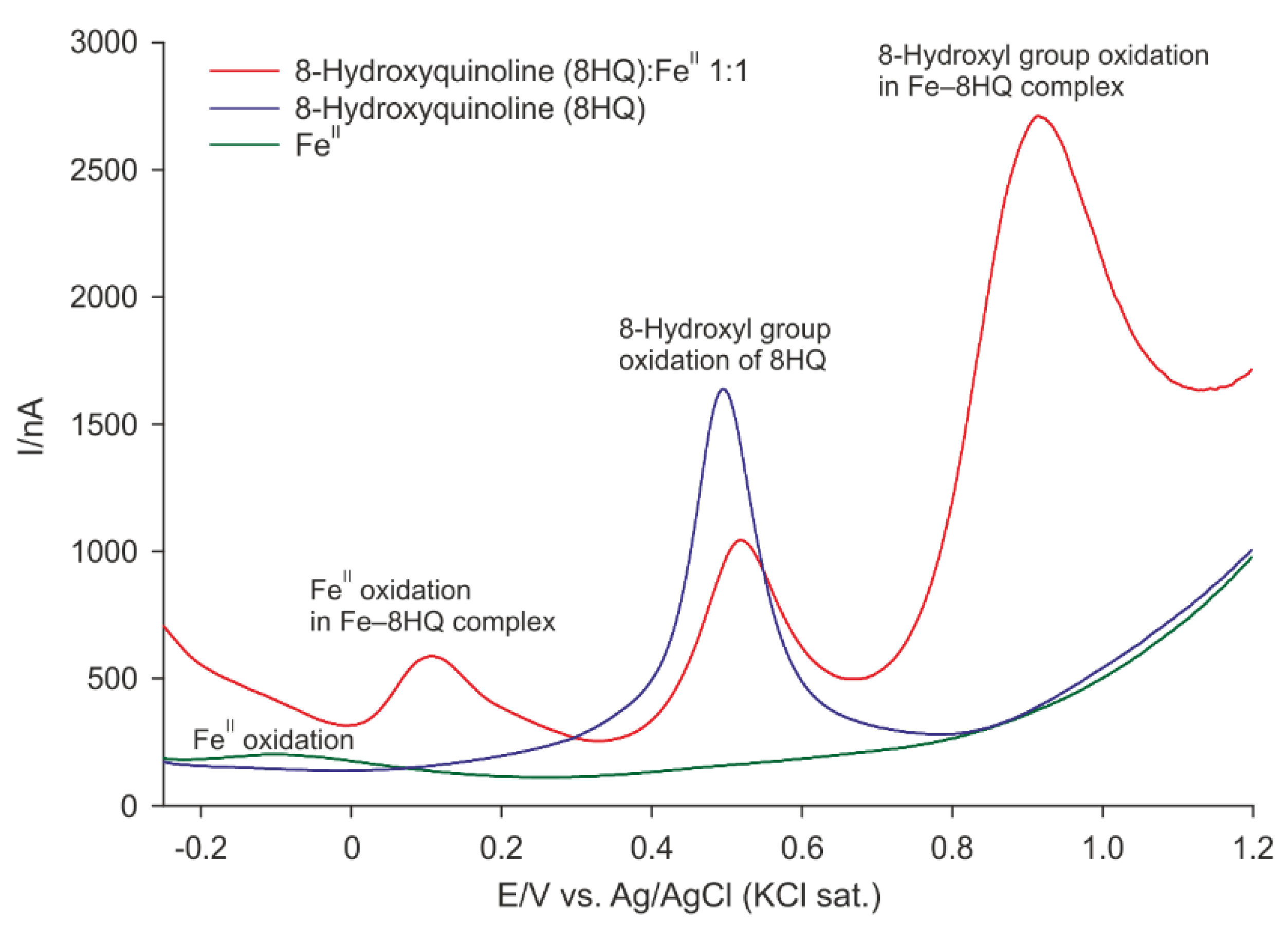
2.2. Differential Pulse Voltammetry
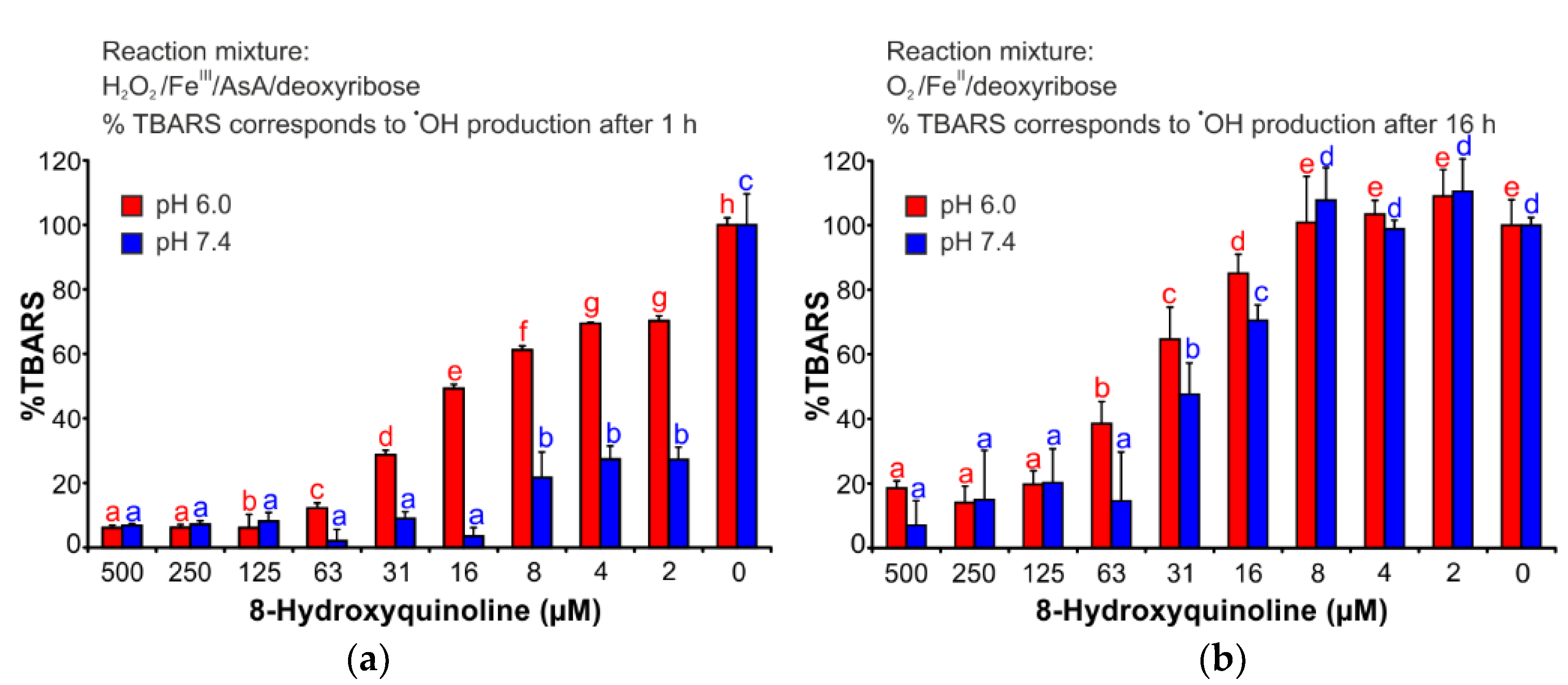
2.3. Deoxyribose Degradation Assay
2.4. Iron (II) Autoxidation Assay
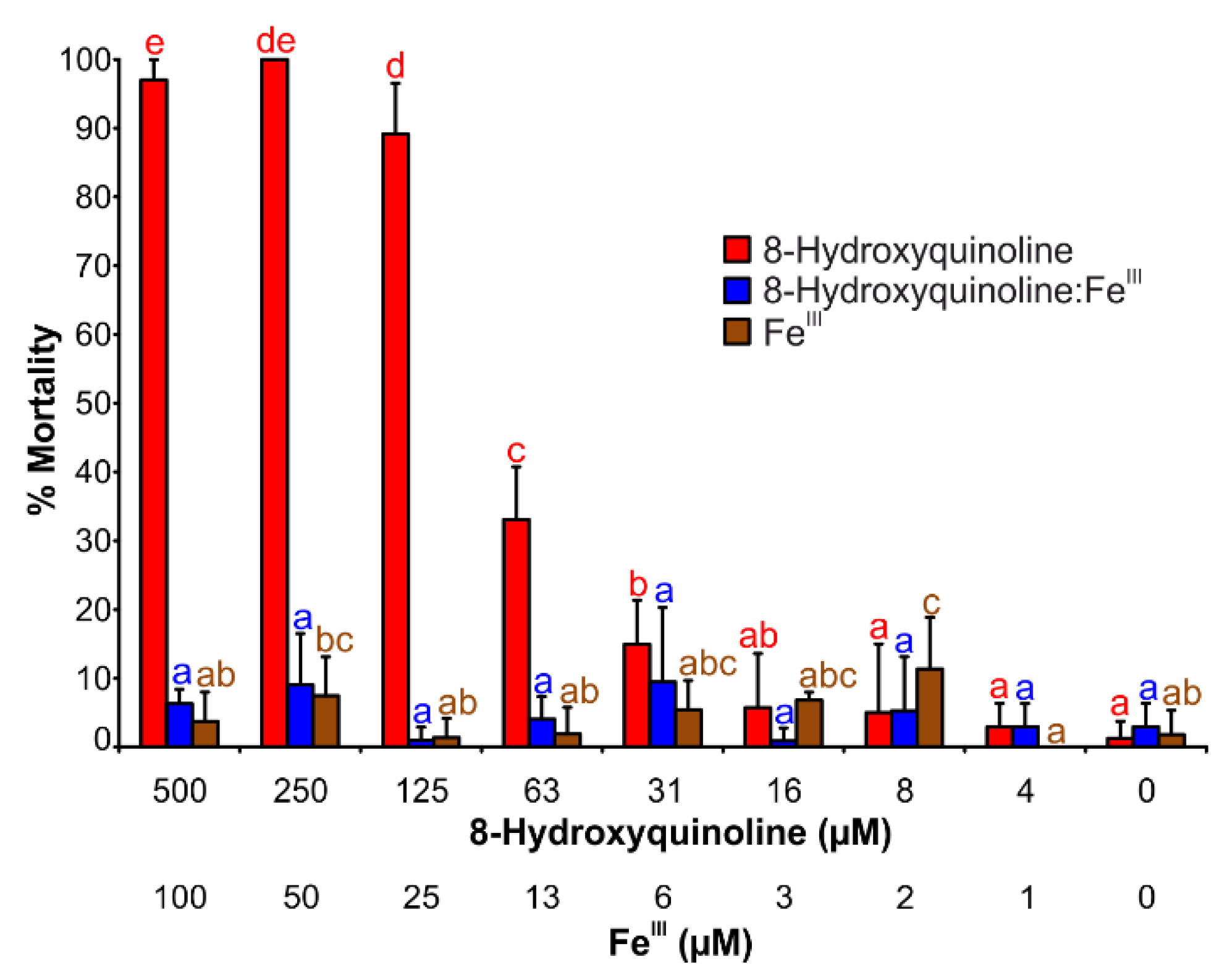
2.5. Brine Shrimp Mortality Assay
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Mass Spectrometry
4.3. Differential Pulse Voltammetry
4.4. Deoxyribose Degradation Assay
4.5. Iron (II) Autoxidation Assay
4.6. Brine Shrimp Mortality Assay
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AsA | Ascorbic acid |
| BBB | Blood−brain barrier |
| DPV | Differential pulse voltammetry |
| 8HQ | 8-Hydroxyquinoline |
| L | Ligand |
| Nano-ESI−MS | Nano-electrospray−mass spectrometry |
| ROS | Reactive oxygen species |
| S.D. | Standard deviation |
| TBARS | Thiobarbituric acid reactive species |
References
- Tharayil, N.; Bhowmik, P.; Alpert, P.; Walker, E.; Amarasiriwardena, D.; Xing, B. Dual purpose secondary compounds: Phytotoxin of Centaurea diffusa also facilitates nutrient uptake. New Phytol. 2009, 181, 424–434. [Google Scholar] [CrossRef]
- Lee, C.H.; Jeon, J.H.; Lee, S.G.; Lee, H.S. Insecticidal properties of Euphorbiaceae: Sebastiania corniculata-derived 8-hydroxyquinoline and its derivatives against three plant hopper species (Hemiptera: Delphacidae). J. Korean Soc. Appl. Biol. Chem. 2010, 53, 464–469. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K.; Pesko, M.; Kos, J. Ring-substituted 8-hydroxyquinoline-2-carboxanilides as photosystem II inhibitors. Bioorg. Med. Chem. Lett. 2016, 26, 3862–3865. [Google Scholar] [CrossRef]
- Novakova, J.; Dzunkova, M.; Musilova, S.; Vlkova, E.; Kokoska, L.; Moya, A.; D’Auria, G. Selective growth-inhibitory effect of 8-hydroxyquinoline towards Clostridium difficile and Bifidobacterium longum subsp longum in co-culture analysed by flow cytometry. J. Med. Microbiol. 2014, 63, 1663–1669. [Google Scholar] [CrossRef]
- Skrivanova, E.; Van Immerseel, F.; Hovorkova, P.; Kokoska, L. In vitro selective growth-Inhibitory effect of 8-hydroxyquinoline on Clostridium perfringens versus Bifidobacteria in a medium containing chicken ileal digesta. PLoS ONE 2016, 11, e0167638. [Google Scholar] [CrossRef]
- Pippi, B.; Reginatto, P.; Machado, G.D.M.; Bergamo, V.Z.; Dalla Lana, D.F.; Teixeira, M.L.; Franco, L.L.; Alves, R.J.; Andrade, S.F.; Fuentefria, A.M. Evaluation of 8-hydroxyquinoline derivatives as hits for antifungal drug design. Med. Mycol. 2017, 55, 763–773. [Google Scholar] [CrossRef]
- Oliveri, V.; Vecchio, G. 8-Hydroxyquinolines in medicinal chemistry: A structural perspective. Eur. J. Med. Chem. 2016, 120, 252–274. [Google Scholar] [CrossRef]
- Prachayasittikul, V.; Prachayasittikul, S.; Ruchirawat, S. 8-Hydroxyquinolines: A review of their metal chelating properties and medicinal applications. Drug Des. Devel. Ther. 2013, 7, 1157–1178. [Google Scholar] [CrossRef]
- Kell, D.B. Iron behaving badly: Inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases. BMC Med. Genom. 2009, 2, 2. [Google Scholar] [CrossRef]
- Chobot, V.; Drage, S.; Hadacek, F. Redox properties of 8-quinolinol and implications for its mode of action. Nat. Prod. Commun. 2011, 6, 597–602. [Google Scholar]
- Kell, D.B. Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson’s, Huntington’s, Alzheimer’s, prions, bactericides, chemical toxicology and others as examples. Arch. Toxicol. 2010, 84, 825–889. [Google Scholar] [CrossRef]
- Murakami, K.; Haneda, M.; Yoshino, M. Prooxidant action of xanthurenic acid and quinoline compounds: Role of transition metals in the generation of reactive oxygen species and enhanced formation of 8-hydroxy-2 ‘-deoxyguanosine in DNA. Biometals 2006, 19, 429–435. [Google Scholar] [CrossRef]
- Leanderson, P.; Tagesson, C. Iron bound to the lipophilic iron chelator, 8-hydroxyquinoline, causes DNA strand breakage in cultured lung cells. Carcinogenesis 1996, 17, 545–550. [Google Scholar] [CrossRef]
- Fang, S.L.; Yu, X.N.; Ding, H.X.; Han, J.A.; Feng, J. Effects of intracellular iron overload on cell death and identification of potent cell death inhibitors. Biochem. Biophys. Res. Commun. 2018, 503, 297–303. [Google Scholar] [CrossRef]
- Kayyali, R.; Pannala, A.S.; Khodr, H.; Hider, R.C. Comparative radical scavenging ability of bidentate iron(III) chelators. Biochem. Pharmacol. 1998, 55, 1327–1332. [Google Scholar] [CrossRef]
- Lange, B. Kolorimetrische Analyse mit Besonderer Besichtigung der Lichtelektrischen Kolorimetrie, 1st ed.; Verlag Chemie, G.M.B.H: Berlin, Germany, 1941. [Google Scholar]
- Chobot, V. Simultaneous detection of pro- and antioxidative effects in the variants of the deoxyribose degradation assay. J. Agric. Food Chem. 2010, 58, 2088–2094. [Google Scholar] [CrossRef]
- Chobot, V.; Hadacek, F.; Kubicova, L. Effects of selected dietary secondary metabolites on reactive oxygen species production caused by iron(II) autoxidation. Molecules 2014, 19, 20023–20033. [Google Scholar] [CrossRef]
- Kubicova, L.; Sustr, M.; Kral’ova, K.; Chobot, V.; Vytlacilova, J.; Jahodar, L.; Vuorela, P.; Machacek, M.; Kaustova, J. Synthesis and biological evaluation of quinazoline-4-thiones. Molecules 2003, 8, 756–769. [Google Scholar] [CrossRef]
- Nunes, B.S.; Carvalho, F.D.; Guilhermino, L.M.; Van Stappen, G. Use of the genus Artemia in ecotoxicity testing. Environ. Pollut. 2006, 144, 453–462. [Google Scholar] [CrossRef]
- Stevic, M.C.; Ignjatovic, L.M.; Ciric-Marjanovic, G.; Stanisic, S.M.; Stankovic, D.M.; Zima, J. Voltammetric behaviour and determination of 8-hydroxyquinoline using a glassy carbon paste electrode and the theoretical study of its electrochemical oxidation mechanism. Int. J. Electrochem. Sci. 2011, 6, 2509–2525. [Google Scholar]
- Chobot, V.; Huber, C.; Trettenhahn, G.; Hadacek, F. (+/−)-Catechin: Chemical weapon, antioxidant, or stress regulator? J. Chem. Ecol. 2009, 35, 980–996. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Bareggi, S.R.; Cornelli, U. Clioquinol: Review of its mechanisms of action and clinical uses in neurodegenerative disorders. CNS Neurosci. Ther. 2012, 18, 41–46. [Google Scholar] [CrossRef]
- Matthews, R.S. Artemia salina as a test organism for measuring superoxide-mediated toxicity. Free Radical Biol. Med. 1995, 18, 919–922. [Google Scholar] [CrossRef]
- Chobot, V.; Vytlacilova, J.; Kubicova, L.; Opletal, L.; Jahodaar, L.; Laakso, I.; Vuorela, P. Phototoxic activity of a thiophene polyacetylene from Leuzea carthamoides. Fitoterapia 2006, 77, 194–198. [Google Scholar] [CrossRef]
- Tewari, R.K.; Bachmann, G.; Hadacek, F. Iron in complex with the alleged phytosiderophore 8-hydroxyquinoline induces functional iron deficiency and non-autolytic programmed cell death in rapeseed plants. Environ. Exp. Bot. 2015, 109, 151–160. [Google Scholar] [CrossRef]
- Montagne, A.; Zhao, Z.; Zlokovic, B.V. Alzheimer’s disease: A matter of blood−brain barrier dysfunction? J. Exp. Med. 2017, 214, 3151–3169. [Google Scholar] [CrossRef]
- Kubicova, L.; Hadacek, F.; Weckwerth, W.; Chobot, V. Effects of endogenous neurotoxin quinolinic acid on reactive oxygen species production by Fenton reaction catalyzed by iron or copper. J. Organomet. Chem. 2015, 782, 111–115. [Google Scholar] [CrossRef]
- Kubicova, L.; Hadacek, F.; Chobot, V. Quinolinic Acid: Neurotoxin or oxidative stress modulator? Int. J. Mol. Sci. 2013, 14, 21328–21338. [Google Scholar] [CrossRef]
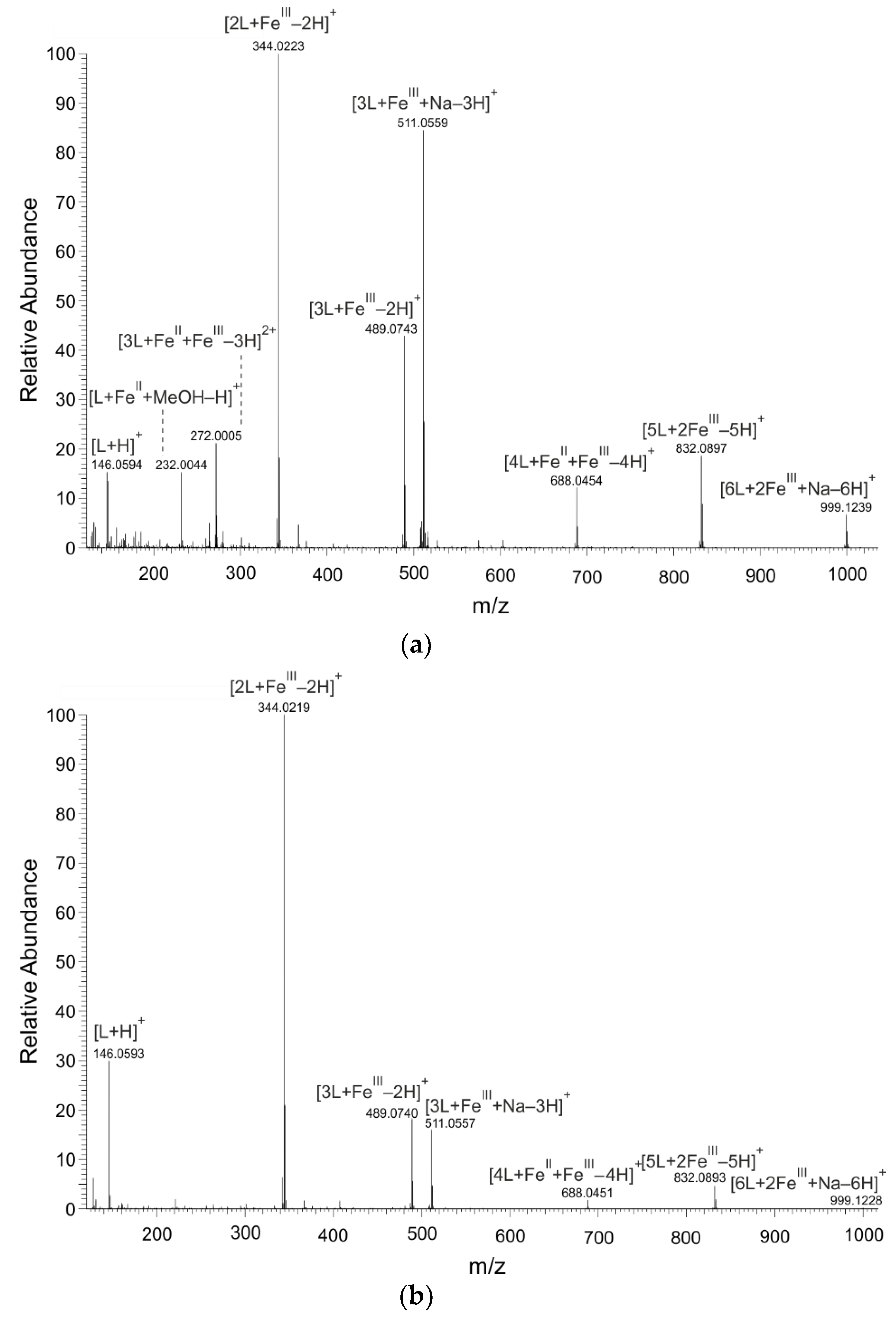
| L:FeII 2:1 | L:FeIII 2:1 | |||||
|---|---|---|---|---|---|---|
| Composition | Formula | m/z Calculated | m/z Found | Δ (ppm) | m/z Found | Δ (ppm) |
| [L + H]+ | [C9H8NO]+ | 146.0600 | 146.0594 | −0.59 | 146.0593 | −0.71 |
| [L + Na]+ | [C9H7NNaO]+ | 168.0420 | 168.0413 | −0.71 | 168.0412 | −0.82 |
| [L + FeII + MeOH–H]+ | [C10H10 FeNO2]+ | 232.0055 | 232.0044 | −4.76 | − | − |
| [3L + FeII + FeIII–3H]2+ | [C27H18 Fe2N3O3]2+ | 272.0018 | 272.0005 | −4.86 | − | − |
| [2L + FeIII–2H]+ | [C18H12 FeN2O2]+ | 344.0243 | 344.0223 | −5.87 | 344.0219 | −6.98 |
| [3L + FeIII–2H]+ | [C27H19 FeN3O3]+ | 489.0770 | 489.0743 | −5.54 | 489.0740 | −6.29 |
| [3L + FeIII + Na–3H]+ | [C27H18 FeN3NaO3]+ | 511.0590 | 511.0559 | −5.93 | 511.0557 | −6.44 |
| [4L + FeII + FeIII–4H]+ | [C36H24 Fe2N4O4]+ | 688.0491 | 688.0454 | −5.30 | 688.0451 | −5.84 |
| [5L + 2FeIII–5H]+ | [C45H30 Fe2N5O5]+ | 832.0940 | 832.0897 | −5.16 | 832.0893 | −5.74 |
| [6L + 2FeIII + Na–6H]+ | [C54H36 Fe2N6NaO6]+ | 999.1287 | 999.1239 | −4.86 | 999.1228 | −5.99 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chobot, V.; Hadacek, F.; Bachmann, G.; Weckwerth, W.; Kubicova, L. Antioxidant Properties and the Formation of Iron Coordination Complexes of 8-Hydroxyquinoline. Int. J. Mol. Sci. 2018, 19, 3917. https://doi.org/10.3390/ijms19123917
Chobot V, Hadacek F, Bachmann G, Weckwerth W, Kubicova L. Antioxidant Properties and the Formation of Iron Coordination Complexes of 8-Hydroxyquinoline. International Journal of Molecular Sciences. 2018; 19(12):3917. https://doi.org/10.3390/ijms19123917
Chicago/Turabian StyleChobot, Vladimir, Franz Hadacek, Gert Bachmann, Wolfram Weckwerth, and Lenka Kubicova. 2018. "Antioxidant Properties and the Formation of Iron Coordination Complexes of 8-Hydroxyquinoline" International Journal of Molecular Sciences 19, no. 12: 3917. https://doi.org/10.3390/ijms19123917
APA StyleChobot, V., Hadacek, F., Bachmann, G., Weckwerth, W., & Kubicova, L. (2018). Antioxidant Properties and the Formation of Iron Coordination Complexes of 8-Hydroxyquinoline. International Journal of Molecular Sciences, 19(12), 3917. https://doi.org/10.3390/ijms19123917