Intracerebral Delivery of Brain-Derived Neurotrophic Factor Using HyStem®-C Hydrogel Implants Improves Functional Recovery and Reduces Neuroinflammation in a Rat Model of Ischemic Stroke
Abstract
1. Introduction
2. Results
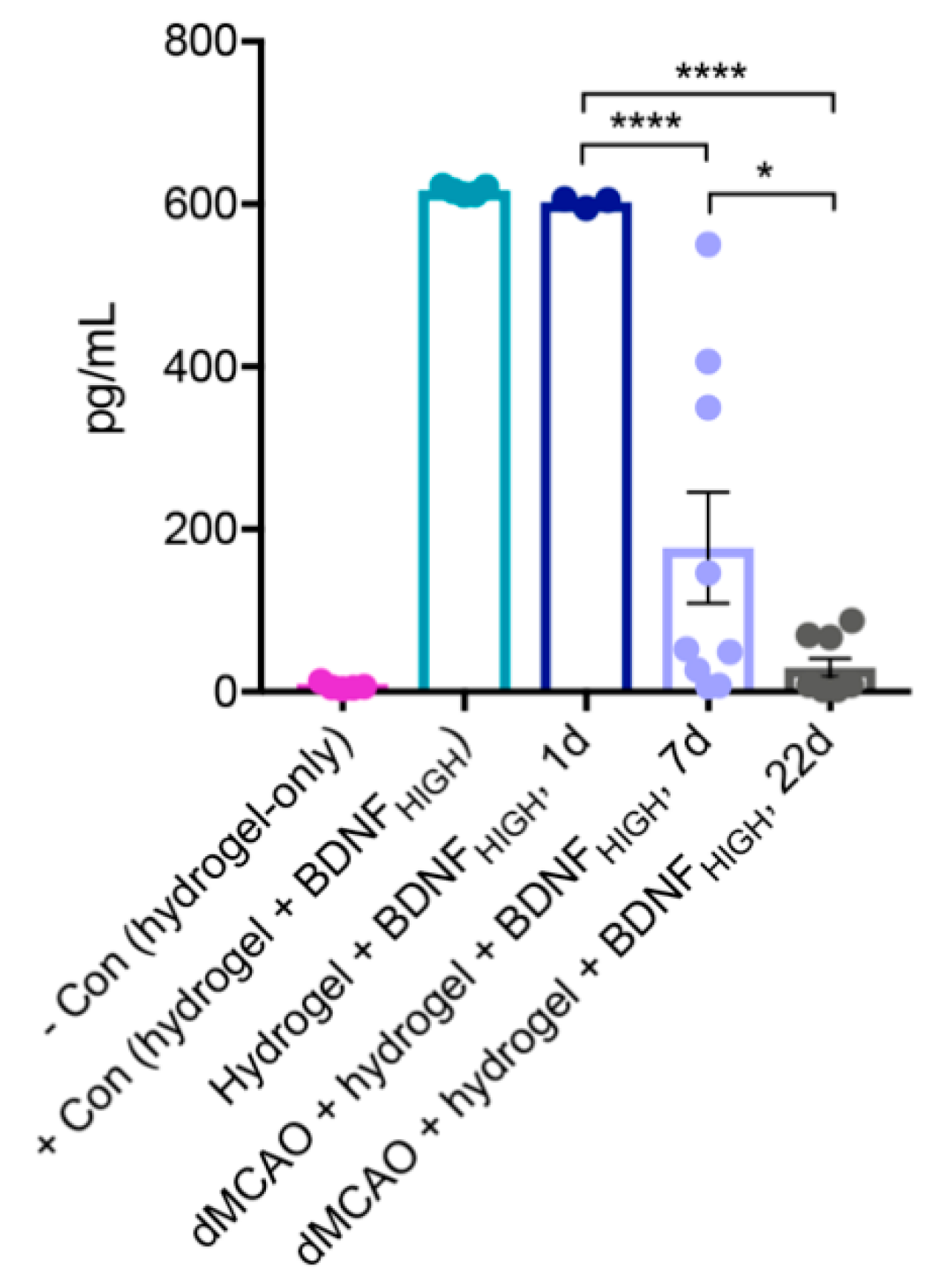
2.1. BDNF Released from Fully Gelated Hydrogel Over Time
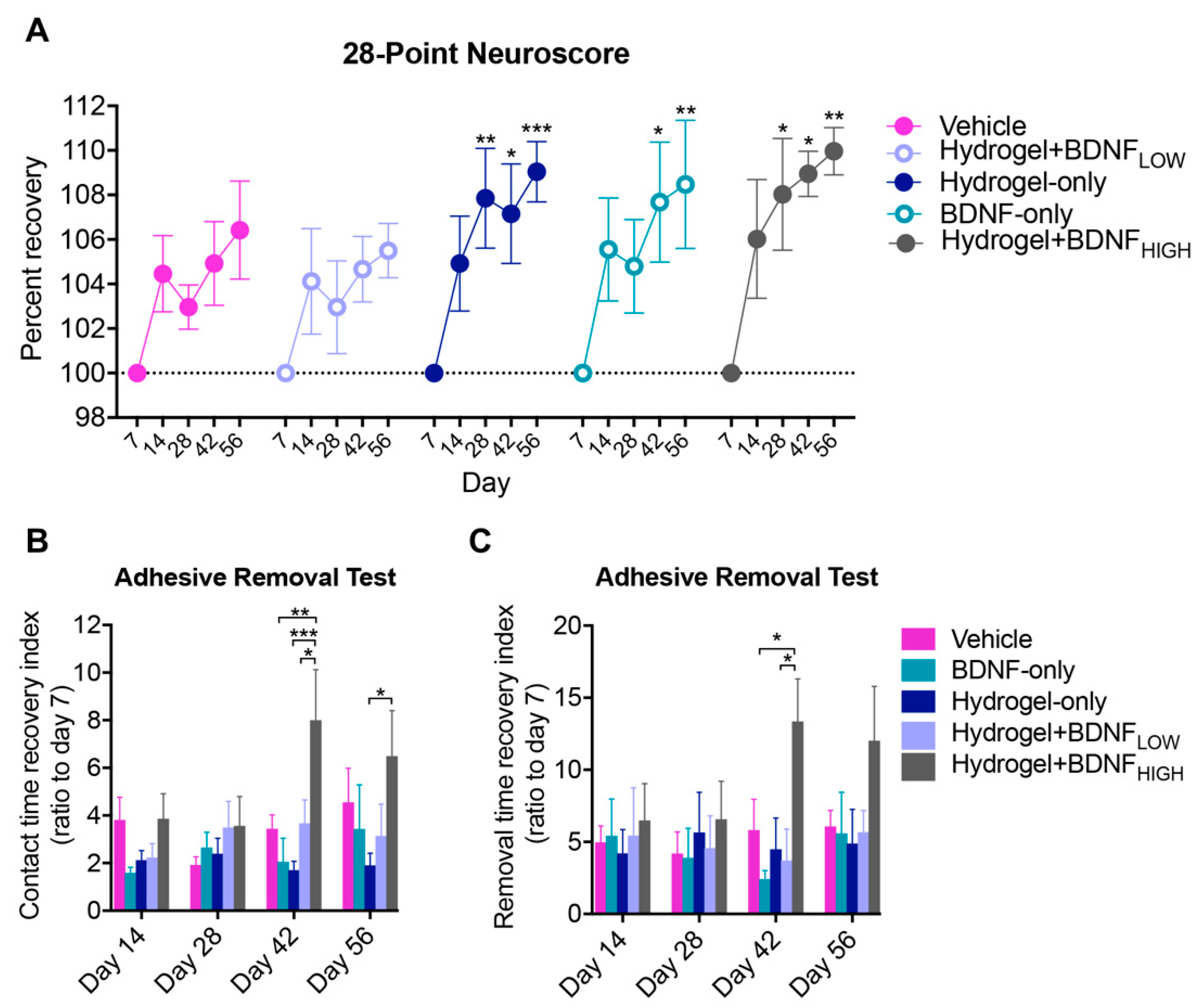
2.2. Hydrogel + BDNFHIGH Improves Functional Recovery Following dMCAo
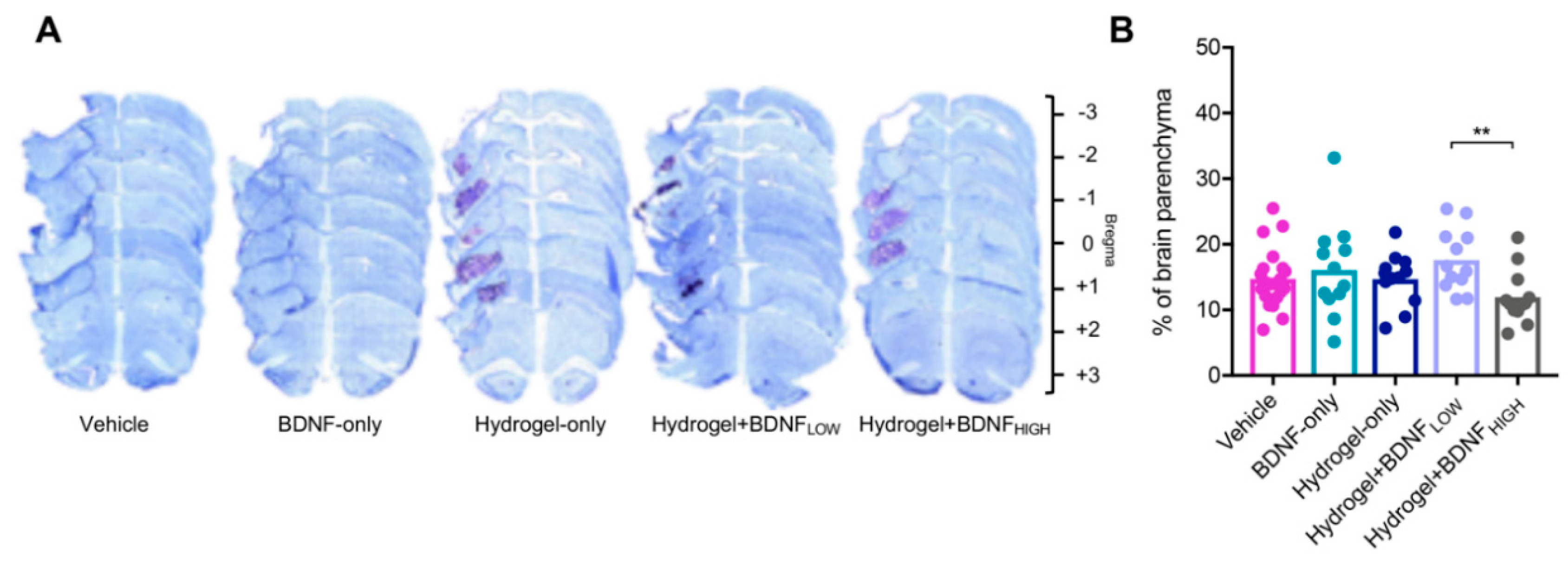
2.3. Hydrogel + BDNFHIGH Reduces Infarct Volume Following dMCAo
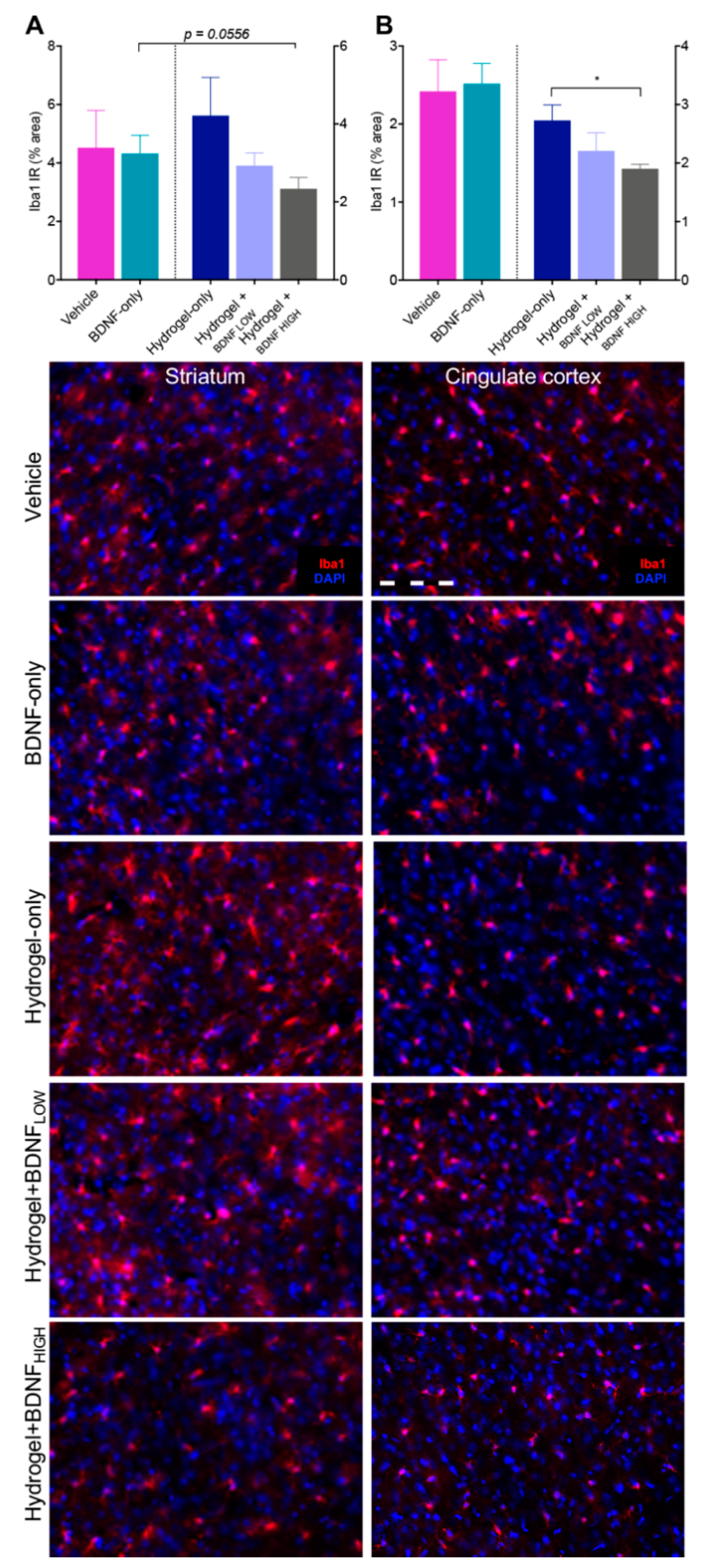
2.4. Hydrogel + BDNFHIGH Reduces Iba1 in the Striatum and Cingulate Cortex Following dMCAo
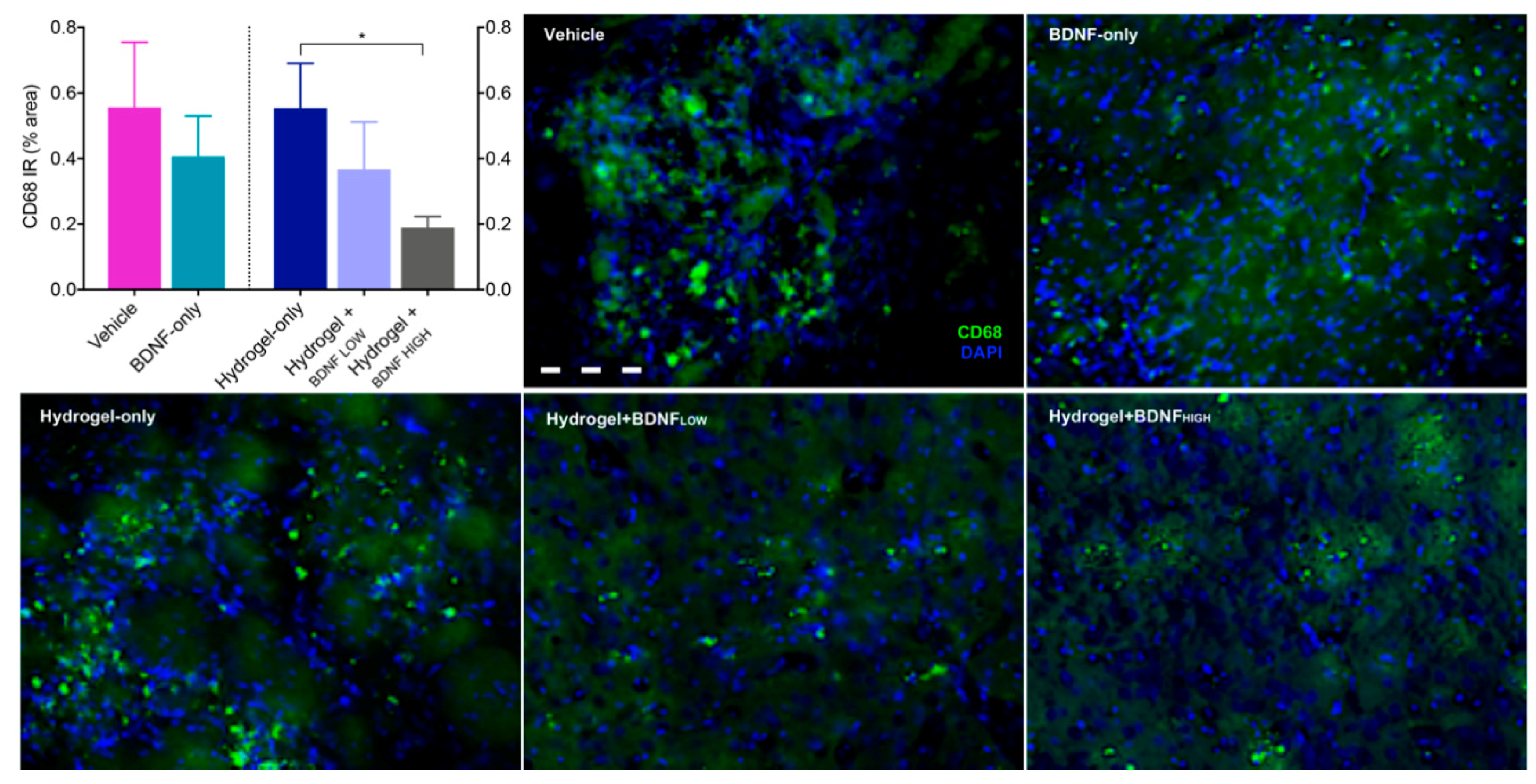
2.5. Hydrogel + BDNFHIGH Reduces CD68 in the Striatum Following dMCAo
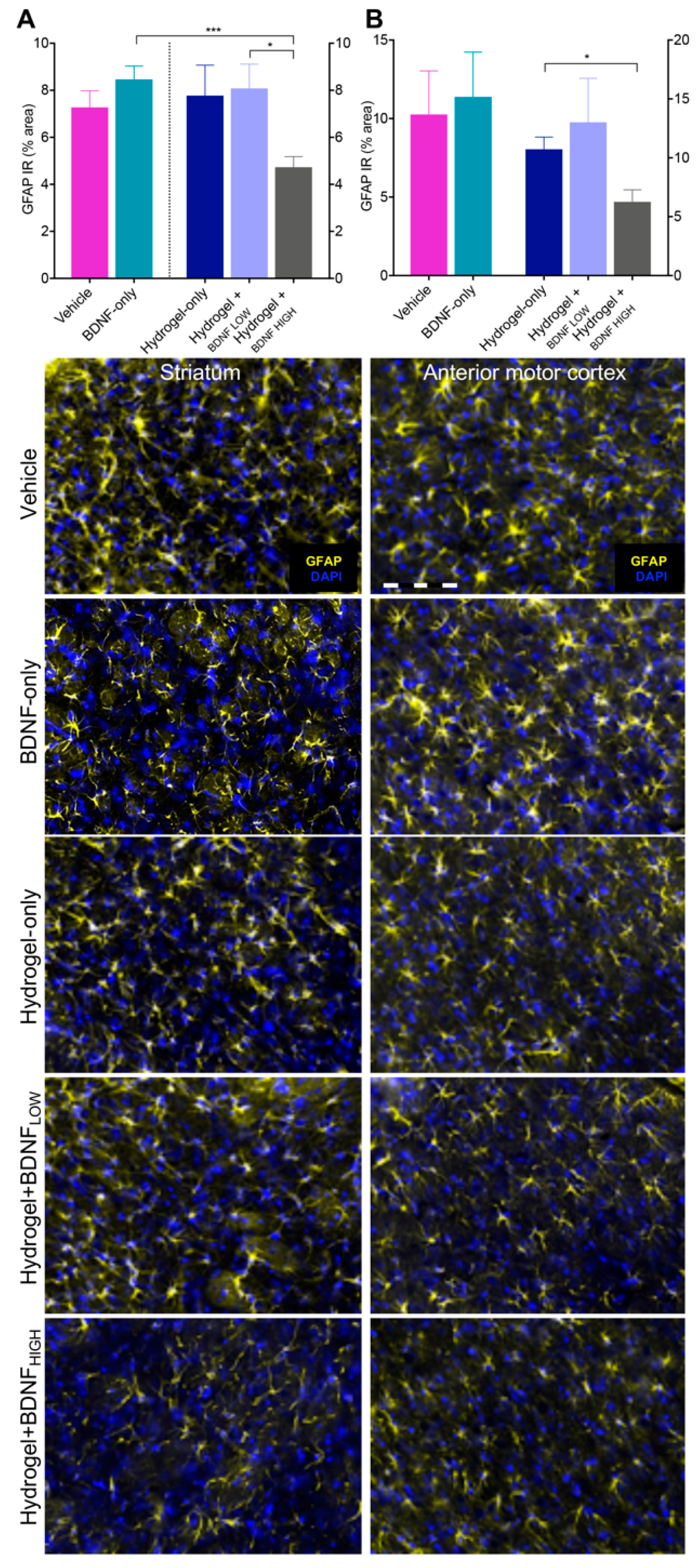
2.6. Hydrogel + BDNFHIGH Reduces GFAP in the Anterior Motor Cortex and Striatum Following dMCAo
3. Discussion
4. Materials and Methods
4.1. Animals
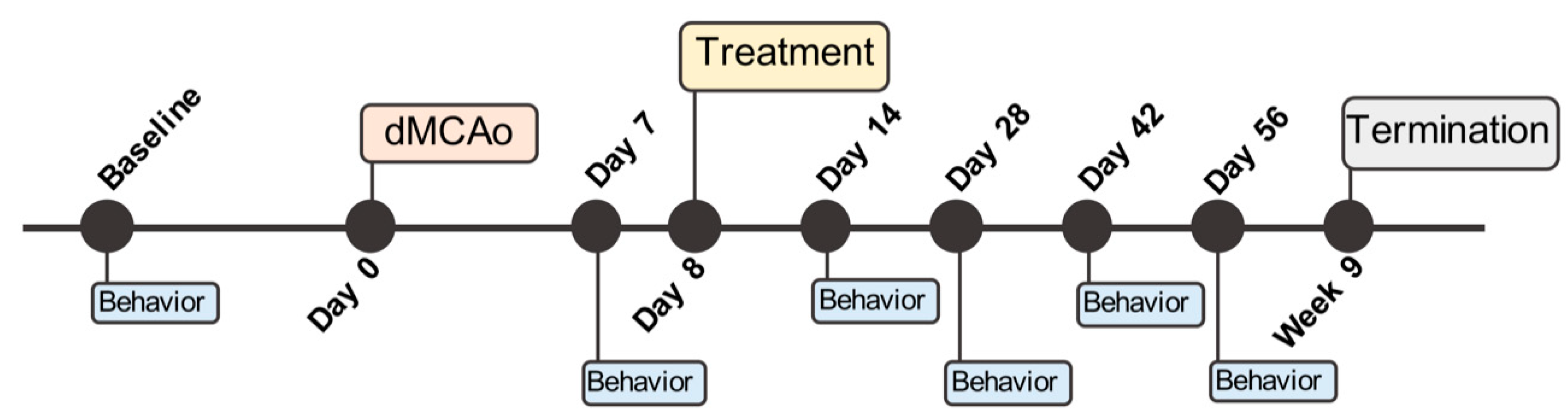
4.2. Distal Middle Cerebral Artery Occlusion (dMCAo) Surgery
4.3. Test Articles
4.4. Hydrogel and Treatment Preparation
4.5. Treatment Administration
4.6. In Vivo Determination of BDNF Release Using ELISA
4.7. Behavioral Assessment
4.7.1. Adhesive Removal Test
4.7.2. Neuroscore
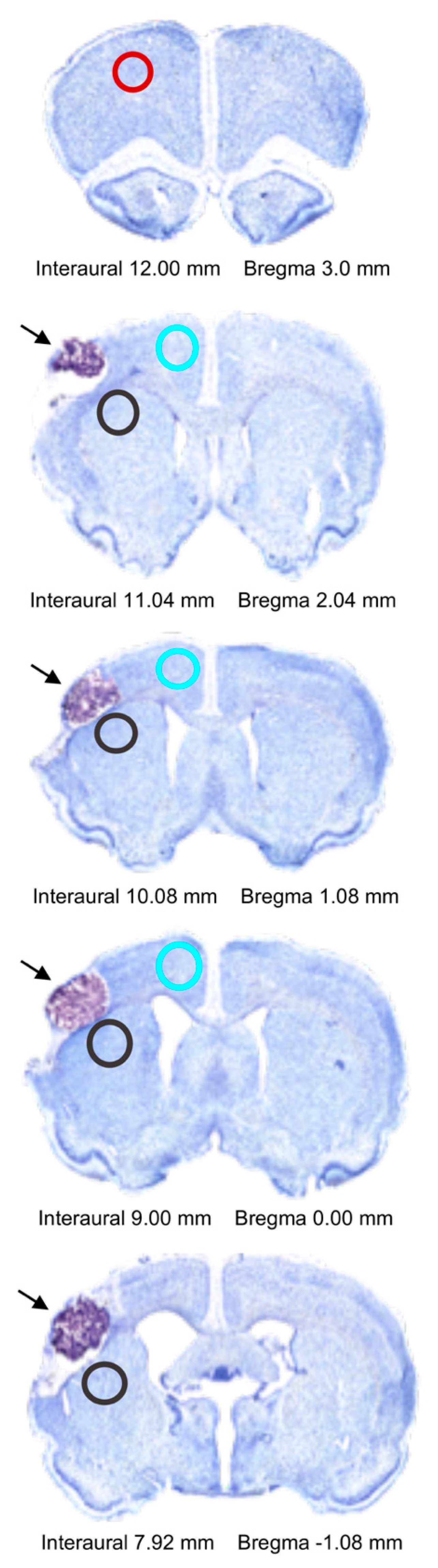
4.8. Immunohistochemistry
4.8.1. Cresyl Violet Staining
4.8.2. Multilabel Fluorescent IHC
4.9. Fluorescent Image Analysis
4.10. Infarct Volume Quantification
4.11. Data Analysis and Statistics
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| APLAC | Administrative Panel on Laboratory Animal Care |
| ARRIVE | Animal Research: Reporting of In Vivo Experiments |
| ART | Adhesive removal test |
| A/P | Anterior/Posterior |
| BBB | Blood-brain barrier |
| BDNF | Brain-derived neurotrophic factor |
| CCA | Common carotid artery |
| CD68 | Cluster of differentiation 68 |
| CFR | Code of Federal Regulations |
| DAPI | 4′,6-diamidino-2-phenylindole dihydrochloride |
| DABCO | 1,4-diazabicyclo[2.2.2]octane |
| dMCAo | Distal middle cerebral artery occlusion |
| DPX | Distyrene plasticizer xylene |
| D/V | Dorsal/Ventral |
| ELISA | Enzyme-linked immunosorbent assay |
| GFAP | Glial fibrillary acidic protein |
| HA | Hyaluronan |
| IACUC | Institutional Animal Care and Use Committee |
| Iba1 | Ionized calcium-binding adapter molecule 1 |
| IHC | Immunohistochemistry |
| IR | Immunoreactivity |
| MCA | Middle cerebral artery |
| MCT | Multiple comparisons test |
| NIH | National Institute of Health |
| ROI | Region of interest |
| SD | Standard deviation |
| SEM | Standard error of mean |
| TrkB | Tyrosine kinase B |
| PFA | Paraformaldehyde |
| 28-PN | 28-point neuroscore |
Appendix A
References
- Thom, T.; Haase, N.; Rosamond, W.; Howard, V.J.; Rumsfeld, J.; Manolio, T.; Zheng, Z.J.; Flegal, K.; O’Donnell, C.; Kittner, S.; et al. Heart disease and stroke statistics—2006 update: A report from the american heart association statistics committee and stroke statistics subcommittee. Circulation 2006, 113, e85–e151. [Google Scholar] [PubMed]
- Broughton, B.R.S.; Lim, R.; Arumugam, T.V.; Drummond, G.R.; Wallace, E.M.; Sobey, C.G. Post-stroke inflammation and the potential efficacy of novel stem cell therapies: Focus on amnion epithelial cells. Front Cell Neurosci. 2013, 6, 66. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, J.M.; Murray, V.; Berge, E.; del Zoppo, G.J. Thrombolysis for acute ischaemic stroke. Cochrane Database Syst. Rev. 2014, 7, CD000213. [Google Scholar] [CrossRef] [PubMed]
- Berretta, A.; Tzeng, Y.C.; Clarkson, A.N. Post-stroke recovery: The role of activity-dependent release of brain-derived neurotrophic factor. Expert Rev. Neurother. 2014, 14, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Poo, M.M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, A.N.; Overman, J.J.; Zhong, S.; Mueller, R.; Lynch, G.; Carmichael, S.T. Ampa receptor-induced local brain-derived neurotrophic factor signaling mediates motor recovery after stroke. J. Neurosci. 2011, 31, 3766–3775. [Google Scholar] [CrossRef] [PubMed]
- Schabitz, W.R.; Steigleder, T.; Cooper-Kuhn, C.M.; Schwab, S.; Sommer, C.; Schneider, A.; Kuhn, H.G. Intravenous brain-derived neurotrophic factor enhances poststroke sensorimotor recovery and stimulates neurogenesis. Stroke 2007, 38, 2165–2172. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, A.N.; Parker, K.; Nilsson, M.; Walker, F.R.; Gowing, E.K. Combined ampakine and bdnf treatments enhance poststroke functional recovery in aged mice via AKT-CREB signaling. J. Cereb. Blood Flow Metab. 2015, 35, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Pencea, V.; Bingaman, K.D.; Wiegand, S.J.; Luskin, M.B. Infusion of brain-derived neurotrophic factor into the lateral ventricle of the adult rat leads to new neurons in the parenchyma of the striatum, septum, thalamus, and hypothalamus. J. Neurosci. 2001, 21, 6706–6717. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.E.; Li, P.; Boyd, J.D.; Delaney, K.R.; Murphy, T.H. Extensive turnover of dendritic spines and vascular remodeling in cortical tissues recovering from stroke. J. Neurosci. 2007, 27, 4101–4109. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.J.; Nguyen, C.; Chun, H.N.; Llorente, L.I.; Chiu, A.S.; Machnicki, M.; Zarembinski, T.I.; Carmichael, S.T. Hydrogel-delivered brain-derived neurotrophic factor promotes tissue repair and recovery after stroke. J. Cereb. Blood Flow Metab. 2017, 37, 1030–1045. [Google Scholar] [CrossRef] [PubMed]
- Ploughman, M.; Windle, V.; MacLellan, C.L.; White, N.; Dore, J.J.; Corbett, D. Brain-derived neurotrophic factor contributes to recovery of skilled reaching after focal ischemia in rats. Stroke 2009, 40, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Lim, J.Y.; Shin, J.H.; Park, H.K.; Tan, S.A.; Park, K.U.; Paik, N.J. Effect of the presence of brain-derived neurotrophic factor Val(66)Met polymorphism on the recovery in patients with acute subcortical stroke. Ann. Rehabil. Med. 2013, 37, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Croll, S.D.; Chesnutt, C.R.; Rudge, J.S.; Acheson, A.; Ryan, T.E.; Siuciak, J.A.; DiStefano, P.S.; Wiegand, S.J.; Lindsay, R.M. Co-infusion with a TrkB-Fc receptor body carrier enhances BDNF distribution in the adult rat brain. Exp. Neurol. 1998, 152, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pardridge, W.M. Blood-brain barrier targeting of bdnf improves motor function in rats with middle cerebral artery occlusion. Brain Res. 2006, 1111, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Hanyu, O.; Yamatani, K.; Ikarashi, T.; Soda, S.; Maruyama, S.; Kamimura, T.; Kaneko, S.; Hirayama, S.; Suzuki, K.; Nakagawa, O.; et al. Brain-derived neurotrophic factor modulates glucagon secretion from pancreatic alpha cells: Its contribution to glucose metabolism. Diabetes Obes. Metab. 2003, 5, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Boudes, M.; Menigoz, A. Non-neuronal bdnf, a key player in development of central sensitization and neuropathic pain. J. Physiol. 2009, 587, 2111–2112. [Google Scholar] [CrossRef] [PubMed]
- Pakulska, M.M.; Ballios, B.G.; Shoichet, M.S. Injectable hydrogels for central nervous system therapy. Biomed. Mater. 2012, 7, 024101. [Google Scholar] [CrossRef] [PubMed]
- Bignami, A.; Hosley, M.; Dahl, D. Hyaluronic acid and hyaluronic acid-binding proteins in brain extracellular matrix. Anat. Embryol. Berl. 1993, 188, 419–433. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Charles, L.F.; Zarembinski, T.I.; Johnson, K.I.; Atzet, S.K.; Wesselschmidt, R.L.; Wight, M.E.; Kuhn, L.T. Modified hyaluronan hydrogels support the maintenance of mouse embryonic stem cells and human induced pluripotent stem cells. Macromol. Biosci. 2012, 12, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Chan, A.; Morad, L.; Kornblum, H.I.; Fan, G.P.; Carmichael, S.T. Hydrogel matrix to support stem cell survival after brain transplantation in stroke. Neurorehab. Neural Repair 2010, 24, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol. Bioeng. 2009, 103, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Tian, W.; Xu, Q.; Cui, F.; Zhang, J.; Lu, Q.; Zhao, C. The enhancement of cell adherence and inducement of neurite outgrowth of dorsal root ganglia co-cultured with hyaluronic acid hydrogels modified with Nogo-66 receptor antagonist in vitro. Neuroscience 2006, 137, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Ballios, B.G.; Cooke, M.J.; Donaldson, L.; Coles, B.L.; Morshead, C.M.; van der Kooy, D.; Shoichet, M.S. A hyaluronan-based injectable hydrogel improves the survival and integration of stem cell progeny following transplantation. Stem Cell Rep. 2015, 4, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.F.; Cooke, M.J.; Morshead, C.M.; Shoichet, M.S. Hydrogel delivery of erythropoietin to the brain for endogenous stem cell stimulation after stroke injury. Biomaterials 2012, 33, 2681–2692. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.J.; Wang, Y.F.; Morshead, C.M.; Shoichet, M.S. Controlled epi-cortical delivery of epidermal growth factor for the stimulation of endogenous neural stem cell proliferation in stroke-injured brain. Biomaterials 2011, 32, 5688–5697. [Google Scholar] [CrossRef] [PubMed]
- Rewell, S.S.; Churilov, L.; Sidon, T.K.; Aleksoska, E.; Cox, S.F.; Macleod, M.R.; Howells, D.W. Evolution of ischemic damage and behavioural deficit over 6 months after mcao in the rat: Selecting the optimal outcomes and statistical power for multi-centre preclinical trials. PloS ONE 2017, 12, e0171688. [Google Scholar] [CrossRef] [PubMed]
- Kokaia, Z.; Andsberg, G.; Yan, Q.; Lindvall, O. Rapid alterations of bdnf protein levels in the rat brain after focal ischemia: Evidence for increased synthesis and anterograde axonal transport. Exp. Neurol. 1998, 154, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Schabitz, W.R.; Schwab, S.; Spranger, M.; Hacke, W. Intraventricular brain-derived neurotrophic factor reduces infarct size after focal cerebral ischemia in rats. J. Cereb. Blood Flow Metab. 1997, 17, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Rickhag, M.; Wieloch, T.; Gido, G.; Elmer, E.; Krogh, M.; Murray, J.; Lohr, S.; Bitter, H.; Chin, D.J.; von Schack, D.; et al. Comprehensive regional and temporal gene expression profiling of the rat brain during the first 24 h after experimental stroke identifies dynamic ischemia-induced gene expression patterns, and reveals a biphasic activation of genes in surviving tissue. J. Neurochem. 2006, 96, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Wei, N.; Lu, T.; Zhu, J.; Xu, G.; Liu, X. Intranasal brain-derived neurotrophic factor protects brain from ischemic insult via modulating local inflammation in rats. Neuroscience 2011, 172, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Barreto, G.; White, R.E.; Ouyang, Y.; Xu, L.; Giffard, R.G. Astrocytes: Targets for neuroprotection in stroke. Cent. Nerv. Syst. Agents Med. Chem. 2011, 11, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Silver, J.; Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 2004, 5, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.S.; Johnston, S.C. Temporal and geographic trends in the global stroke epidemic. Stroke 2013, 44, S123–S125. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, S.P.; Fisher, M.; Furlan, A.J.; Goldstein, L.B.; Gorelick, P.B.; Kaste, M.; Lees, K.R.; Traystman, R.J. Recommendations for standards regarding preclinical neuroprotective and restorative drug development. Stroke 1999, 30, 2752–2758. [Google Scholar]
- Gladstone, D.J.; Black, S.E.; Hakim, A.M.; Heart and Stroke Foundation of Ontario Centre of Excellence in Stroke Recovery. Toward wisdom from failure: Lessons from neuroprotective stroke trials and new therapeutic directions. Stroke 2002, 33, 2123–2136. [Google Scholar] [CrossRef] [PubMed]
- Bible, E.; Dell’Acqua, F.; Solanky, B.; Balducci, A.; Crapo, P.M.; Badylak, S.F.; Ahrens, E.T.; Modo, M. Non-invasive imaging of transplanted human neural stem cells and ECM scaffold remodeling in the stroke-damaged rat brain by (19)F- and diffusion-MRI. Biomaterials 2012, 33, 2858–2871. [Google Scholar] [CrossRef] [PubMed]
- Bible, E.; Qutachi, O.; Chau, D.Y.S.; Alexander, M.R.; Shakesheff, K.M.; Modo, M. Neo-vascularization of the stroke cavity by implantation of human neural stem cells on vegf-releasing plga microparticles. Biomaterials 2012, 33, 7435–7446. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, G.R.; Ding, S. Reactive astrocytes and therapeutic potential in focal ischemic stroke. Neurobiol. Dis. 2016, 85, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, Y.; Li, S. Molecular mechanisms of scar-sourced axon growth inhibitors. Brain Res. 2015, 1619, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Kokaia, Z.; Zhao, Q.; Kokaia, M.; Elmer, E.; Metsis, M.; Smith, M.L.; Siesjo, B.K.; Lindvall, O. Regulation of brain-derived neurotrophic factor gene-expression after transient middle cerebral-artery occlusion with and without brain-damage. Exp. Neurol. 1995, 136, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Tamura, A.; Graham, D.I.; McCulloch, J.; Teasdale, G.M. Focal cerebral ischaemia in the rat: 1. Description of technique and early neuropathological consequences following middle cerebral artery occlusion. J. Cereb. Blood Flow Metab. 1981, 1, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates—The New Coronal Set, 5th ed.; Academic Press: London, UK, 2005. [Google Scholar]
- Lenzlinger, P.M.; Saatman, K.E.; Hoover, R.C.; Cheney, J.A.; Bareyre, F.M.; Raghupathi, R.; Arnold, L.D.; McIntosh, T.K. Inhibition of vascular endothelial growth factor receptor (VEGFR) signaling by BSF476921 attenuates regional cerebral edema following traumatic brain injury in rats. Restor. Neurol. Neurosci. 2004, 22, 73–79. [Google Scholar] [PubMed]
- Encarnacion, A.; Horie, N.; Keren-Gill, H.; Bliss, T.M.; Steinberg, G.K.; Shamloo, M. Long-term behavioral assessment of function in an experimental model for ischemic stroke. J. Neurosci. Methods 2011, 196, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Gundersen, H.J.; Bendtsen, T.F.; Korbo, L.; Marcussen, N.; Moller, A.; Nielsen, K.; Nyengaard, J.R.; Pakkenberg, B.; Sorensen, F.B.; Vesterby, A.; et al. Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. APMIS 1988, 96, 379–394. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravina, K.; Briggs, D.I.; Kislal, S.; Warraich, Z.; Nguyen, T.; Lam, R.K.; Zarembinski, T.I.; Shamloo, M. Intracerebral Delivery of Brain-Derived Neurotrophic Factor Using HyStem®-C Hydrogel Implants Improves Functional Recovery and Reduces Neuroinflammation in a Rat Model of Ischemic Stroke. Int. J. Mol. Sci. 2018, 19, 3782. https://doi.org/10.3390/ijms19123782
Ravina K, Briggs DI, Kislal S, Warraich Z, Nguyen T, Lam RK, Zarembinski TI, Shamloo M. Intracerebral Delivery of Brain-Derived Neurotrophic Factor Using HyStem®-C Hydrogel Implants Improves Functional Recovery and Reduces Neuroinflammation in a Rat Model of Ischemic Stroke. International Journal of Molecular Sciences. 2018; 19(12):3782. https://doi.org/10.3390/ijms19123782
Chicago/Turabian StyleRavina, Kristine, Denise I. Briggs, Sezen Kislal, Zuha Warraich, Tiffany Nguyen, Rachel K. Lam, Thomas I. Zarembinski, and Mehrdad Shamloo. 2018. "Intracerebral Delivery of Brain-Derived Neurotrophic Factor Using HyStem®-C Hydrogel Implants Improves Functional Recovery and Reduces Neuroinflammation in a Rat Model of Ischemic Stroke" International Journal of Molecular Sciences 19, no. 12: 3782. https://doi.org/10.3390/ijms19123782
APA StyleRavina, K., Briggs, D. I., Kislal, S., Warraich, Z., Nguyen, T., Lam, R. K., Zarembinski, T. I., & Shamloo, M. (2018). Intracerebral Delivery of Brain-Derived Neurotrophic Factor Using HyStem®-C Hydrogel Implants Improves Functional Recovery and Reduces Neuroinflammation in a Rat Model of Ischemic Stroke. International Journal of Molecular Sciences, 19(12), 3782. https://doi.org/10.3390/ijms19123782