The Role of Reduced Graphene Oxide toward the Self-Assembly of Lignin-Based Biocomposites Fabricated from Ionic Liquids
Abstract
1. Introduction
2. Results and Discussion
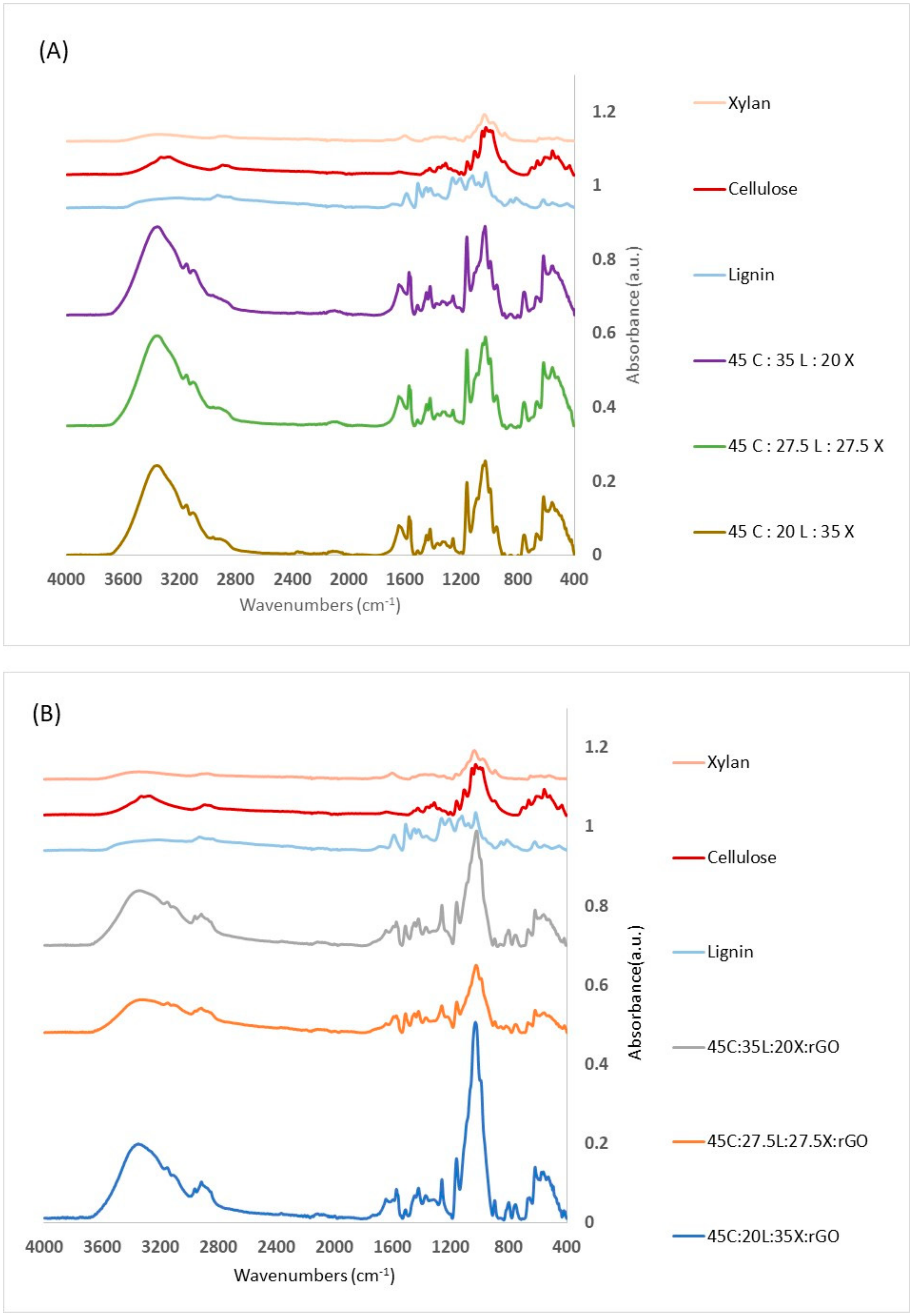
2.1. Structural, Thermal and Morphological Changes
2.2. Morphology Analysis (SEM)
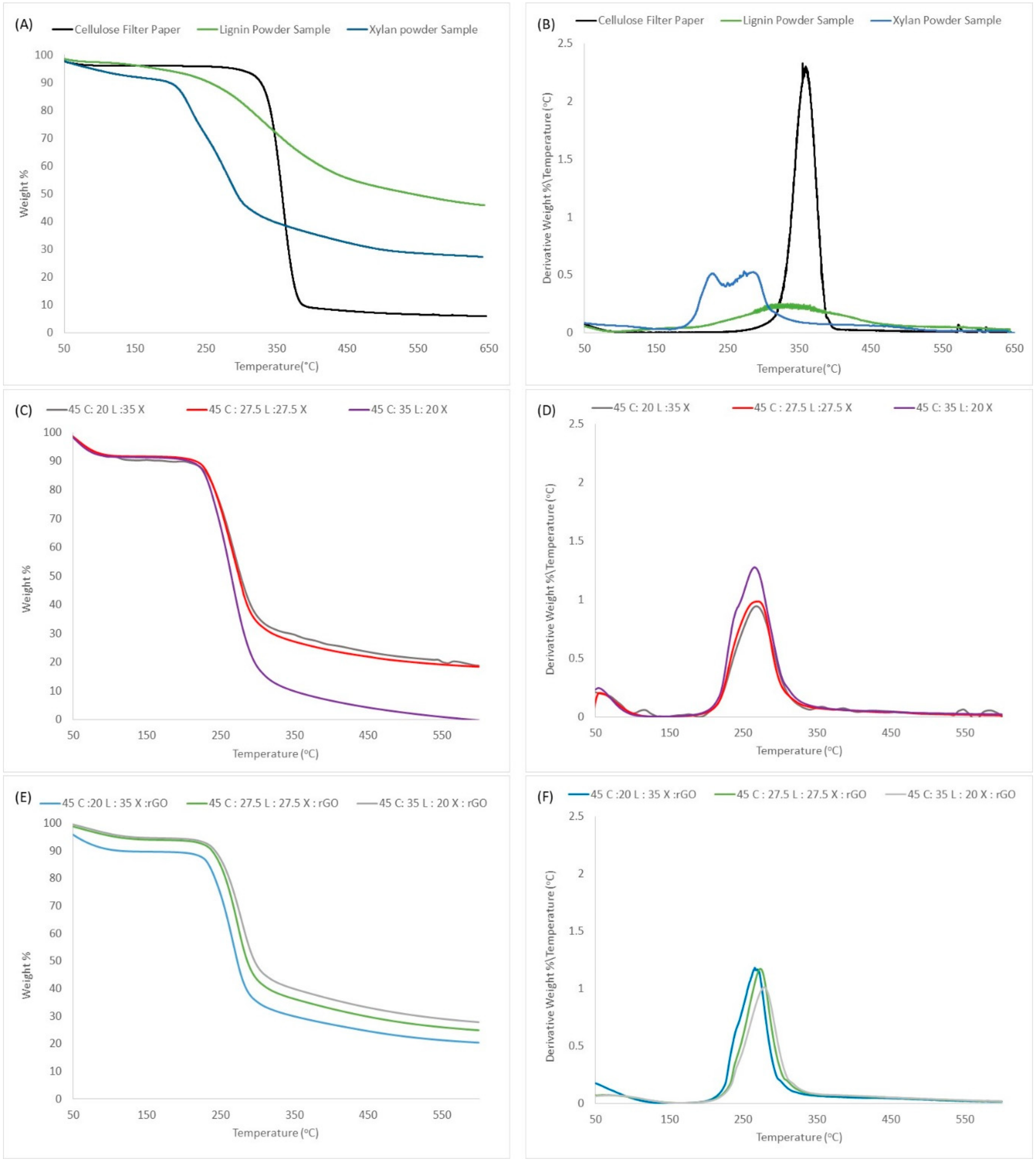
2.3. Thermal Decomposition Analysis
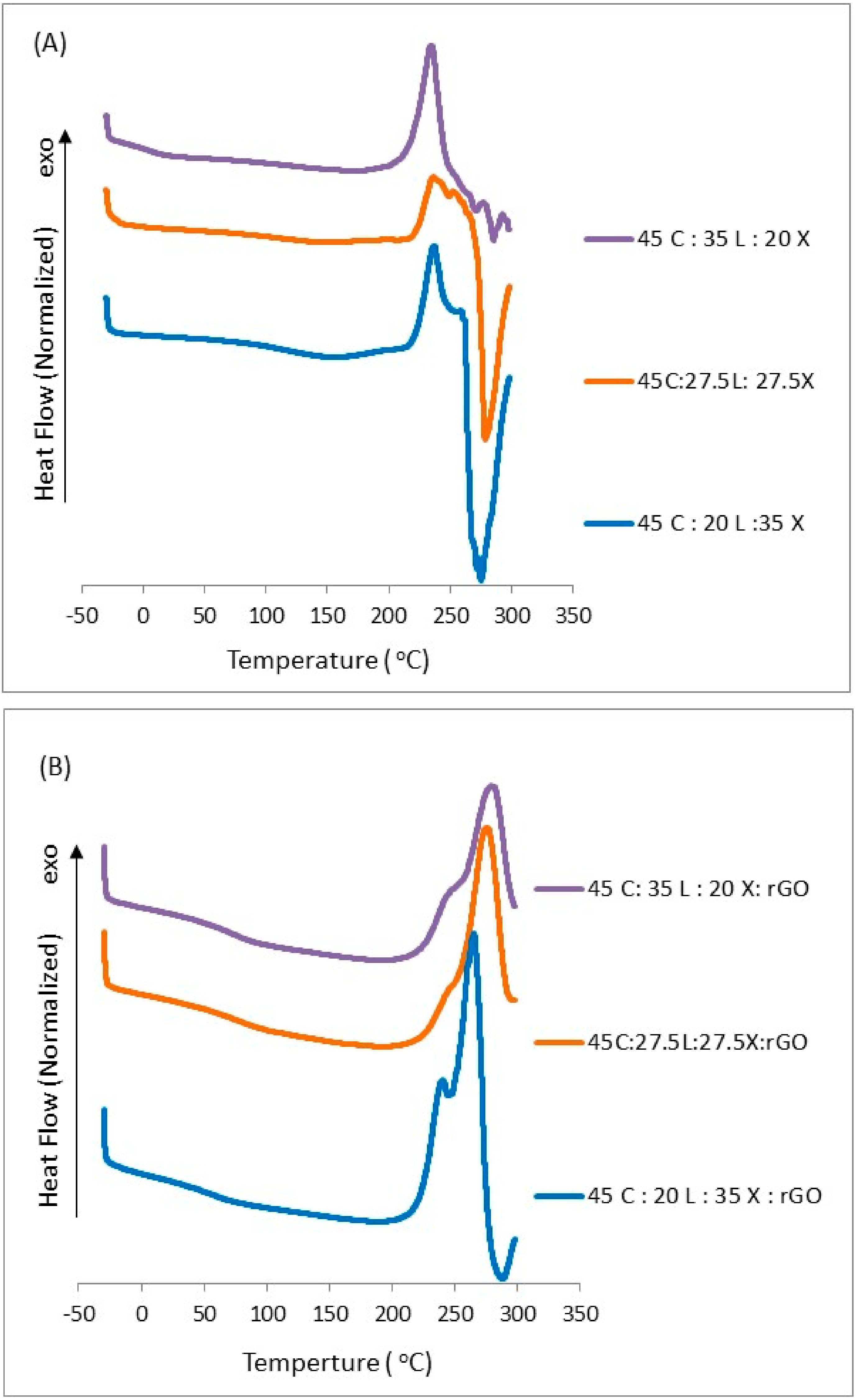
2.4. Glass Transition Temperature
3. Materials and Methods
3.1. General Materials
3.2. Fabrication Method of Biocomposite
3.3. Fourier Transform Infrared Spectroscopy
3.4. Thermogravimetric Analysis
3.5. Differential Scanning Calorimetry
3.6. Scanning Electron Microscope
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crestini, C.; Crucianelli, M.; Orlandi, M.; Saladino, R. Oxidative strategies in lignin chemistry: A new environmental friendly approach for the functionalisation of lignin and lignocellulosic fibers. Catal. Today 2010, 156, 8–22. [Google Scholar] [CrossRef]
- Watkins, D.; Nuruddin, M.; Hosur, M.; Tcherbi-Narteh, A.; Jeelani, S. Extraction and characterization of lignin from different biomass resources. J. Mater. Res. Technol. 2015, 4, 26–32. [Google Scholar] [CrossRef]
- Marriott, P.E.; Gómez, L.D.; McQueen-Mason, S.J. Unlocking the potential of lignocellulosic biomass through plant science. New Phytol. 2016, 209, 1366–1381. [Google Scholar] [CrossRef] [PubMed]
- Zakzeski, J.; Bruijnincx, P.C.; Jongerius, A.L.; Weckhuysen, B.M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Luo, N.; Zhang, X.; Xu, L.; Wu, J.; Yu, J.; He, J.; Zhang, J. All-cellulose nanocomposites reinforced with in situ retained cellulose nanocrystals during selective dissolution of cellulose in an ionic liquid. ACS Sustain. Chem. Eng. 2016, 4, 4417–4423. [Google Scholar] [CrossRef]
- Cheng, G.; Varanasi, P.; Li, C.; Liu, H.; Melnichenko, Y.B.; Simmons, B.A.; Kent, M.S.; Singh, S. Transition of cellulose crystalline structure and surface morphology of biomass as a function of ionic liquid pretreatment and its relation to enzymatic hydrolysis. Biomacromolecules 2011, 12, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Samayam, I.P.; Hanson, B.L.; Langan, P.; Schall, C.A. Ionic-liquid induced changes in cellulose structure associated with enhanced biomass hydrolysis. Biomacromolecules 2011, 12, 3091–3098. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.; Waters, J.C.; Stanton, J.; Hess, J.; Salas-de la Cruz, D. Macromolecular interactions control structural and thermal properties of regenerated tri-component blended films. Int. J. Mol. Sci. 2016, 17, 1989. [Google Scholar] [CrossRef] [PubMed]
- Ibbett, R.; Gaddipati, S.; Hill, S.; Tucker, G. Structural reorganisation of cellulose fibrils in hydrothermally deconstructed lignocellulosic biomass and relationships with enzyme digestibility. Biotechnol. Biofuels 2013, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Wu, Y.; Chen, Q.; Yu, Z.; Wang, C.; Jin, S.; Ding, Y.; Wu, G. Dissolution of cellulose with ionic liquids and its application: A mini-review. Green Chem. 2006, 8, 325–327. [Google Scholar] [CrossRef]
- Welker, C.M.; Balasubramanian, V.K.; Petti, C.; Rai, K.M.; DeBolt, S.; Mendu, V. Engineering plant biomass lignin content and composition for biofuels and bioproducts. Energies 2015, 8, 7654–7676. [Google Scholar] [CrossRef]
- Morrison, I.M. Structural investigations on the lignin–carbohydrate complexes of Lolium perenne. Biochem. J. 1974, 139, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Grushnikov, O.P.; Shorygina, N.N. The present state of the problem of lignin–carbohydrate bonds in plant tissues. Russ. Chem. Rev. 1970, 39, 684–694. [Google Scholar] [CrossRef]
- Deng, Y.; Feng, X.; Zhou, M.; Qian, Y.; Yu, H.; Qiu, X. Investigation of aggregation and assembly of alkali lignin using iodine as a probe. Biomacromolecules 2011, 12, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Juikar, S.J.; Vigneshwaran, N. Extraction of nanolignin from coconut fibers by controlled microbial hydrolysis. Ind. Crops Prod. 2017, 109, 420–425. [Google Scholar] [CrossRef]
- Gkartzou, E.; Koumoulos, E.P.; Charitidis, C.A. Production and 3D printing processing of bio-based thermoplastic filament. Manuf. Rev. 2017, 4. [Google Scholar] [CrossRef]
- Nirmale, T.C.; Kale, B.B.; Varma, A.J. A review on cellulose and lignin based binders and electrodes: Small steps towards a sustainable lithium ion battery. Int. J. Biol. Macromol. 2017, 103, 1032–1043. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xiong, Y.; Fan, B.; Yao, Q.; Wang, H.; Jin, C.; Sun, Q. Cellulose as an adhesion agent for the synthesis of lignin aerogel with strong mechanical performance, Sound-absorption and thermal Insulation. Sci. Rep. 2016, 6, 32383. [Google Scholar] [CrossRef] [PubMed]
- Henry, N.; Harper, D.; Dadmun, M. Optimizing noncovalent interactions between lignin and synthetic polymers to develop effective compatibilizers. Macromol. Chem. Phys. 2012, 213, 1196–1205. [Google Scholar] [CrossRef]
- Qi, H.; Chang, C.; Zhang, L. Properties and applications of biodegradable transparent and photoluminescent cellulose films prepared via a green process. Green Chem. 2009, 11, 177–184. [Google Scholar] [CrossRef]
- Chen, H. Chemical composition and structure of natural lignocellulose. In Biotechnology of Lignocellulose; Springer: Berlin, Germany, 2014; pp. 25–71. [Google Scholar]
- Yuan, X.; Cheng, G. From cellulose fibrils to single chains: Understanding cellulose dissolution in ionic liquids. Phys. Chem. Chem. Phys. 2015, 17, 31592–31607. [Google Scholar] [CrossRef] [PubMed]
- Brice, R.E.; Morrison, I.M. The degradation of isolated hemicelluloses and lignin-hemicellulose complexes by cell-free, rumen hemicellulases. Carbohydr. Res. 1982, 101, 93–100. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, H.; Li, Z.; Lu, X.; Zhang, X.; Zhang, S.; Zhou, K. Characterization of the regenerated cellulose films in ionic liquids and rheological properties of the solutions. Mater. Chem. Phys. 2011, 128, 220–227. [Google Scholar] [CrossRef]
- Mohd, N.; Draman, S.F.S.; Salleh, M.S.N.; Yusof, N.B. Dissolution of cellulose in ionic liquid: A review. AIP Conf. Proc. 2017, 1809, 020035. [Google Scholar]
- Hadadi, A.; Whittaker, J.W.; Verrill, D.E.; Hu, X.; Larini, L.; Salas-de la Cruz, D. A Hierarchical Model To Understand the Processing of Polysaccharides/Protein-Based Films in Ionic Liquids. Biomacromolecules 2018, 19, 3970–3982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, J.; Zhang, J.; He, J. 1-Allyl-3-methylimidazolium chloride room temperature ionic liquid: A new and powerful nonderivatizing solvent for cellulose. Macromolecules 2005, 38, 8272–8277. [Google Scholar] [CrossRef]
- Sajab, M.S.; Chia, C.H.; Chan, C.H.; Zakaria, S.; Kaco, H.; Chook, S.W.; Chin, S.X. Bifunctional graphene oxide–cellulose nanofibril aerogel loaded with Fe(III) for the removal of cationic dye via simultaneous adsorption and Fenton oxidation. RSC Adv. 2016, 6, 19819–19825. [Google Scholar] [CrossRef]
- Xu, M.; Huang, Q.; Wang, X.; Sun, R. Highly tough cellulose/graphene composite hydrogels prepared from ionic liquids. Ind. Crops Prod. 2015, 70, 56–63. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, C.; Guo, S.; Zhang, J. Interactions of graphene and graphene oxide with proteins and peptides. Nanotechnol. Rev. 2013, 2, 27–45. [Google Scholar] [CrossRef]
- Gurunathan, S.; Kim, J.-H. Synthesis, toxicity, biocompatibility, and biomedical applications of graphene and graphene-related materials. Int. J. Nanomed. 2016, 11, 1927. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, H.; Shen, G.; Cheng, P.; Zhang, J.; Guo, S. Reduction of graphene oxide via l-ascorbic acid. Chem. Commun. 2010, 46, 1112–1114. [Google Scholar] [CrossRef] [PubMed]
- Kathirgamanathan, K.; Grigsby, W.J.; Al-Hakkak, J.; Edmonds, N.R. Two-dimensional FTIR as a tool to study the chemical interactions within cellulose-ionic liquid solutions. Int. J. Polym. Sci. 2015, 2015, 958653. [Google Scholar] [CrossRef]
- Stanton, J.; Xue, Y.; Pandher, P.; Malek, L.; Brown, T.; Hu, X.; Salas-de la Cruz, D. Impact of ionic liquid type on the structure, morphology and properties of silk-cellulose biocomposite materials. Int. J. Biol. Macromol. 2018, 108, 333–341. [Google Scholar] [CrossRef] [PubMed]

| Cellulose (% wt) | Lignin (% wt) | Xylan (% wt) | rGO (% wt of Total Biocomposites) |
|---|---|---|---|
| 45% | 20% | 35% | 1 |
| 45% | 27.5% | 27.5% | 1 |
| 45% | 35% | 20% | 1 |
| Without rGO | TOnset (°C) | TEnd (°C) | TΔMax (°C) | Wt Loss % |
|---|---|---|---|---|
| 45C:20L:35X | 245.448 | 299.907 | 267.393 | 79.60% |
| 45C:27.5L:27.5X | 245.209 | 300.687 | 270.227 | 80.84% |
| 45C:35L:20X | 244.286 | 297.369 | 264.747 | 97.75% |
| With rGO | TOnset (°C) | TEnd (°C) | TΔMax (°C) | Wt Loss % |
| 45C:20L:35X:rGO | 256.595 | 289.231 | 264.921 | 77.72% |
| 45C:27.5L:27.5X:rGO | 254.047 | 299.955 | 272.569 | 72.98% |
| 45C:35L:20X:rGO | 254.470 | 305.045 | 278.00 | 70.76% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-shahrani, D.; Love, S.A.; Salas-de la Cruz, D. The Role of Reduced Graphene Oxide toward the Self-Assembly of Lignin-Based Biocomposites Fabricated from Ionic Liquids. Int. J. Mol. Sci. 2018, 19, 3518. https://doi.org/10.3390/ijms19113518
Al-shahrani D, Love SA, Salas-de la Cruz D. The Role of Reduced Graphene Oxide toward the Self-Assembly of Lignin-Based Biocomposites Fabricated from Ionic Liquids. International Journal of Molecular Sciences. 2018; 19(11):3518. https://doi.org/10.3390/ijms19113518
Chicago/Turabian StyleAl-shahrani, Dalia, Stacy A. Love, and David Salas-de la Cruz. 2018. "The Role of Reduced Graphene Oxide toward the Self-Assembly of Lignin-Based Biocomposites Fabricated from Ionic Liquids" International Journal of Molecular Sciences 19, no. 11: 3518. https://doi.org/10.3390/ijms19113518
APA StyleAl-shahrani, D., Love, S. A., & Salas-de la Cruz, D. (2018). The Role of Reduced Graphene Oxide toward the Self-Assembly of Lignin-Based Biocomposites Fabricated from Ionic Liquids. International Journal of Molecular Sciences, 19(11), 3518. https://doi.org/10.3390/ijms19113518





