1. Introduction
Cardiac arrhythmias follow breakdown of the ordered action potential (AP) excitation and propagation triggering physiological activity in successive myocardial regions that thereby generates the heartbeat. This, in turn, involves ordered sequences of ion channel activation and inactivation causing AP initiation and recovery. The most common arrhythmia, atrial fibrillation (AF), affects 1–3% of the population in the developed world. Chronic AF increases risks of morbidity, often in the form of stroke and all-cause mortality. Model-based estimates predict substantially increased AF incidences and prevalence in coming decades, resulting in ~9 and ~18 million cases in the United States and Europe, respectively, by 2060 [
1].
Both age and acquired metabolic disorders constitute major risk factors for AF. AF thus affects 0.1%, 4%, and 20% of individuals aged <55, 60–70, and >80 years, respectively. Factors such as physical inactivity, obesity, diabetes mellitus and metabolic syndrome, whose occurrence and effects are themselves age-dependent, may explain ~60% of current upward trends in AF incidence [
1]. Observational and experimental evidence in turn associate both ageing and metabolic disorder with mitochondrial dysfunction and impaired oxidative capacity [
2]. Age-associated mitochondrial DNA damage and compromised respiratory chain function have been demonstrated in a range of human and other mammalian species [
3]. Increased mitochondrial dysfunction and defective electron transport chain complex 1 occurs in obese mice on high fat diets [
4]. Mitochondrial dysfunction, whether acute or chronic, is known in turn to promote cardiac arrhythmogenesis [
5]. The latter is the typical result of altered expression or properties in ion channels underlying electrophysiological activity, or fibrotic or cardiomyopathic change influencing cardiomyocyte or cardiac tissue structure. The latter structural changes have also been reported in experimental diabetes [
6] and metabolic syndrome [
7].
Mitochondrial function is strongly influenced by members of the peroxisome proliferator activated receptor-γ (PPARγ) coactivator-1 transcriptional coactivator (
Pgc-1) family. PPARγ coactivators (PGCs) are abundant in oxidative tissues, including cardiac and skeletal muscle, and brown adipose tissue [
8]. Modifications in these have been used to examine pathological changes associated with energetic deficiencies. Their expression is impaired in obesity, insulin resistance, type 2 diabetes mellitus (T2DM), and first-degree relatives of diabetic patients [
9]. Increasing gene expression related to fatty acid β-oxidation, the tricarboxylic acid cycle and the electron transport chain [
10] allows for PGC to regulate mitochondrial biogenesis, mass and function [
11]. This, in turn, influences cellular metabolism, and its link to external stimuli driving cellular energy demands [
8]. They, thus, constitute potential therapeutic targets. For instance, the PPAR-α agonist fenofibrate, inhibited atrial metabolic remodelling in AF [
12,
13]. Specific PPARγ ligands such as thiazolidinediones (TZDs) are used in clinical practice to improve insulin sensitivity T2DM [
14]. Improvement of cardiac function and reduction in myocardial fibrosis by TZDs have also been previously reported [
14]. Finally, rosiglitazone reduces atrial interstitial fibrosis and AF promotion in diabetic rabbits via modulating oxidative stress and inflammation [
15]. Of major
Pgc-1α,
Pgc-1β, and
Prc (Pgc-1-related) groups of coactivators [
11],
Pgc-1α appears to have a more dynamic function:
Pgc-1α, but not
Pgc-1β, expression is upregulated by physiological stimuli such as fasting, exercise and cold temperatures, thus adapting tissues to high energy demand as opposed to basal mitochondrial function [
11].
Experimental models for mitochondrial disorders directed at associated pro-arrhythmic electrophysiological changes are thus central to understanding of their relationships to development of pro-arrhythmic phenotypes. The present experiments utilise mice with a
Pgc-1α−/− genotype to recapitulate features of energetic deficiency. The widespread use of murine models for cardiac electrophysiology research stems from the ability to genetically modify mice to generate targeted disruption modelling human diseases, and thus avoiding the need to use potentially nonspecific pharmacological models [
16]. In terms of translatability to human cardiac electrophysiology, murine hearts show similar rapid Na
+ current (I
Na) mediated depolarisation phases and transmural AP conduction velocities [
17,
18,
19]. Thus, although murine hearts show anatomical differences, have higher pacing rates and shorter APs compared to human hearts, they have been established to substantially recapitulate human clinical arrhythmic phenotypes [
20,
21]. Previous studies have successfully validated both
Pgc-1α−/− and
Pgc-1β−/− murine models in the study of mitochondrial dysfunction and arrhythmogenesis [
22,
23,
24,
25,
26,
27,
28].
Thus, a higher incidence of extra-systolic provoked atrial arrhythmias, manifesting as atrial tachycardia and ectopic activity, were observed in
Pgc-1β−/− atria [
27]. This was attributed to a reduced rate of AP depolarisation (d
V/d
t), prolonged AP latencies representing reduced AP conduction and an increased level of myocardial fibrosis [
26,
27]. Furthermore, at a cellular level, extensive loose patch clamp studies implicated reduced Na
+ currents but unchanged K
+ currents as a factor for the previously determined slowed conduction velocity in
Pgc-1β−/− atria [
28]. At a systems level, electrocardiographic (ECG) studies in these mice have shown irregular PR, PP and RR intervals with differing P wave morphologies as well as reduced atrioventricular node (AVN) function following adrenergic stimulation [
23,
24]. Interestingly, murine studies of
Pgc-1α−/− overexpression have shown changes in Ca
2+ signalling, electrophysiology, and contractile properties [
25].
Whilst previous murine electrophysiological studies have shown slowed myocardial conduction velocity as an arrhythmogenic substrate in conditions of mitochondrial dysfunction, the underlying molecular changes remains unexplored. Therefore, the present study firstly uses
Pgc-1α−/− atria in an exploration by quantitative PCR of the gene transcriptional background for changes in genes strategic for the electrophysiological and, therefore, potential arrhythmic phenotypes that may offer possible novel pharmacological targets [
29]. Genes were selected and grouped according to the physiological processes underlying excitable activity [
16], adapting an approach first applied to rat as opposed to genetically modified mouse hearts [
30] comprising: (1) energetically-dependent Na
+-K
+-ATPase mediated membrane transport processes generating the ionic gradients driving excitable activity; (2) ion, particularly background K
+, channels mediating the resting potential; (3) voltage-dependent processes generating both cardiac automaticity and Na
+ current mediated AP activation; (4) Ca
2+ homeostatic changes involving both Ca
2+ channel mediated entry and the subsequent alterations in intracellular Ca
2+ homeostasis between cellular compartments; (5) electrophysiological recovery from such activity through voltage-dependent K
+ channel activation; (6) cellular capacity for autonomic modulation through both adrenergic and cholinergic receptor mediated activation of intracellular signalling; (7) a range of cellular and tissue changes ultimately impacting such AP generation and propagation, including inflammatory and tissue fibrotic changes reported in experimental diabetes on earlier occasions. Secondly, this study uses western blots to determine the protein expression levels of Na
v1.5, Cx40 and Cx43, all critical for determining myocardial conduction velocity.
3. Discussion
Transcription factors play central roles in atrial remodelling which in turn is a major contributor to development of arrhythmic substrate leading to clinically important conditions such as atrial fibrillation. The present quantitative PCR study screened for changes in genes strategic to cardiac electrophysiological function potentially implicated in atrial arrhythmia that may potentially offer novel pharmacological targets. The studies used as model, the atria of murine hearts deficient in peroxisome proliferator-activated receptor (PPAR)-γ coactivator-1α (
Pgc-1α). The latter co-activator interacts with the transcription factor and nuclear receptor protein PPAR-α promoting transcription of fatty acid metabolism-related genes. Murine
Pgc-1α−/− hearts thus potentially model a number of the energetic features of atrial fibrillation (AF), the most frequently clinically encountered cardiac arrhythmia. Although there remain significant gaps in our understanding of basic molecular targets in AF pathophysiology [
62], recent metabolomic and proteomic studies implicated altered expression in molecules involved in metabolic pathways in human and experimental AF [
62,
63,
64]. AF is accordingly associated with impaired mitochondrial function, decreased ATP production, and redox imbalance with increased reactive oxygen species (ROS) production [
65]. The latter potentially injures genes and proteins central to cardiomyocyte function [
62].
The present analysis examined expression of genes subclassified into functional groups corresponding to fundamental excitable processes in mouse [
16], adapting similar approaches to rat hearts [
30]. It surveyed Na
+-K
+-ATPase-mediated membrane transport, background K
+ conductances underlying cardiomyocyte resting potentials, HCN and Na
+ channels underlying cardiac automaticity and action potential activation and conduction, voltage-dependent Ca
2+ current activation and intracellular Ca
2+ homeostatic processes, and action potential recovery through voltage-dependent K
+ currents. It also examined indicators for adrenergic and cholinergic modulation and their associated intracellular signalling processes, and longer term cellular and tissue changes including tissue fibrotic changes ultimately impacting upon AP generation and propagation.
Modelling studies predict that only large alterations in the related Na
+/K
+-ATPase activity would significantly alter cell background membrane potentials [
32]. Atrial cardiomyocytes from patients with persistent AF did not show altered Na
+-K
+-ATPase current [
66].
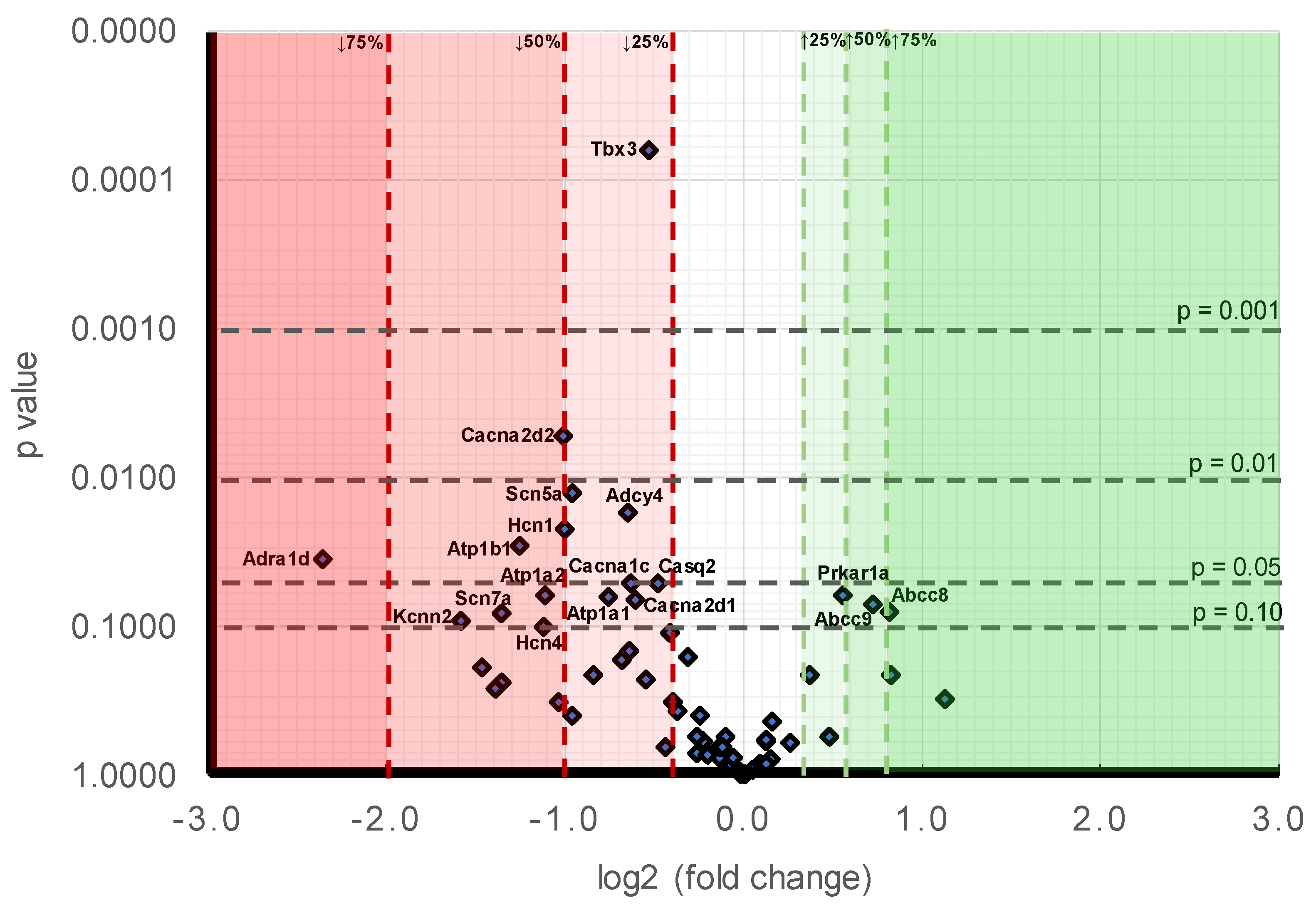
Pgc-1α−/− atria nevertheless did show decreased
Atp1a1,
Atp1a2, and
Atp1b1 expression encoding regulatory and catalytic Na
+/K
+-ATPase subunits compared to levels in
WT. However,
Pgc-1α−/− atria showed little change in the expression of K
+ channels underlying resting membrane potential maintenance, suggestive of normal background resting potential properties in
Pgc-1α−/− relative to
WT, findings reflected in related
Pgc-1β−/− models [
26]. By contrast, of genes encoding ion channels related to automaticity in and initiation and propagation of excitable activity,
Pgc-
1α−/− atria showed decreased transcription of pacemaker,
Hcn1 and
Hcn4, channels relative to
WT. Such findings are relevant to the chronotropic background and its responses to autonomic activation. The findings recapitulate evidence of impaired canine sinoatrial function, and reduced sino-atrial node
HCN2 and
HCN4 mRNA expression and HCN related current densities in atrial tachypacing studies [
67]. Previous ECG studies had demonstrated chronotropic incompetence at the level of the sino-atrial node in
Pgc-1β−/− models of mitochondrial dysfunction following dobutamine challenge [
23].
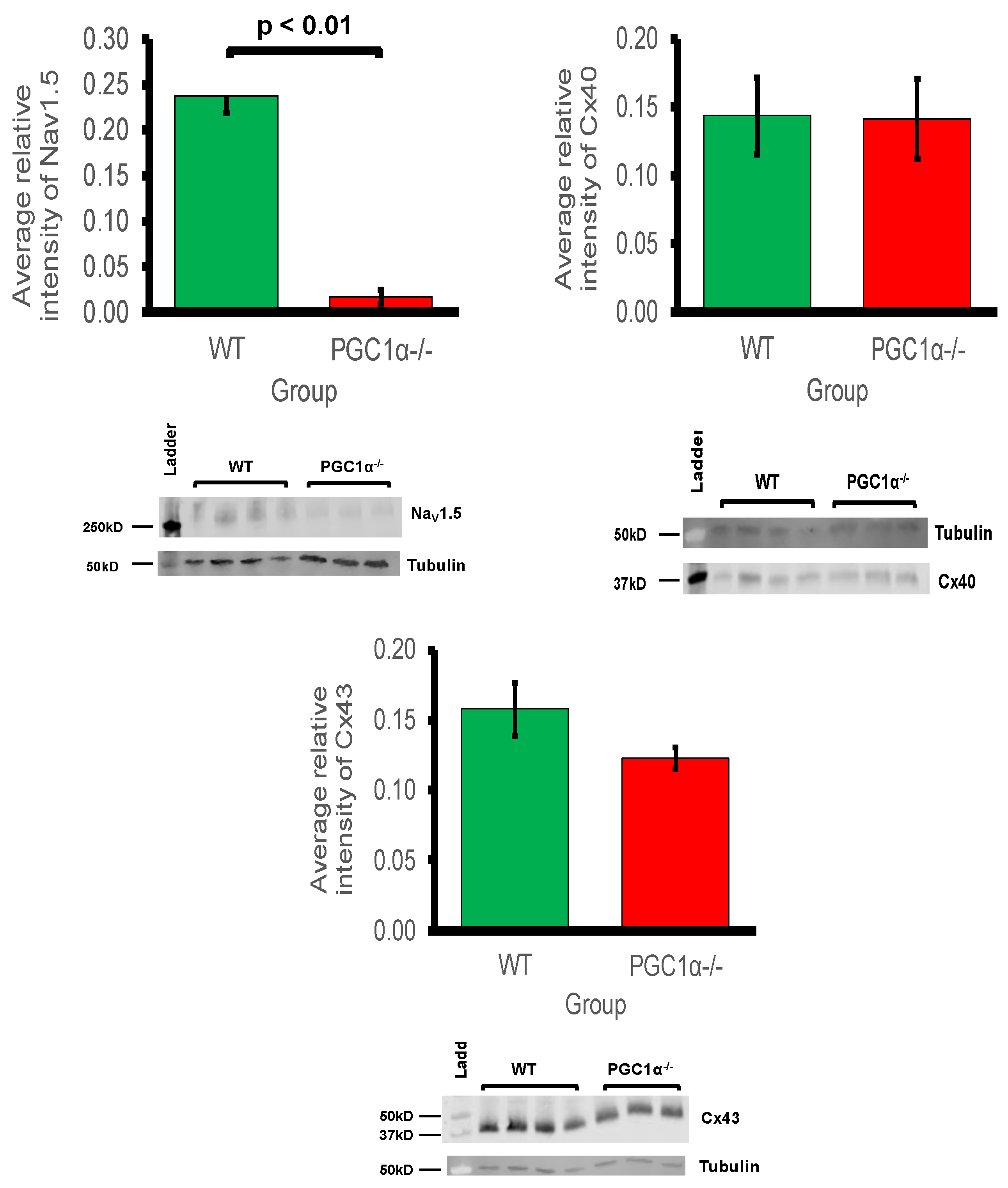
Pgc-1α−/− atria also showed decreases in cardiac Na
v1.5 gene expression. This reduction was significant enough to be reflected at the protein level also. Previous studies in murine models of mitochondrial dysfunction have shown reduced Na
+ current leading to decreased myocardial conduction velocity as a possible arrhythmogenic mechanism [
26,
27,
28]. Thus, reduced Na
+ current, arising from reduced genetic expression under conditions of mitochondrial dysfunction, is predictive of potentially proarrhythmic reductions in both excitability and AP propagation. This is in common with changes reported in murine models for Brugada Syndrome associated with loss of Na
+ channel function [
27]. Conduction velocity is also determined by gap channel conductance, primarily through connexin isoforms, but this study showed that protein expression levels of Cx40 and Cx43 were not significantly reduced in
Pgc-1α−/− atria. This implies that the primary molecular mechanism for reduced conduction would be reduced Na
+ channel expression, as opposed to altered gap junction proteins in
Pgc-1α−/− atria.
Atrial cardiomyocytes show reduced L-type Ca
2+ current, SR Ca
2+ stores and cellular contractility with metabolic stress [
68]. In AF, nuclear factor of activated T-cells (NFAT) is thought to down-regulate Ca
v1.2 channel α-subunit expression, through the Ca
2+/calmodulin/calcineurin/NFAT pathway [
69,
70]. This attenuates L-type Ca
2+ current and shortens atrial refractory periods [
71]. In parallel with this, the qPCR analysis suggested widespread changes in molecules mediating cellular Ca
2+ homeostasis. Altered transcription in genes encoding ion channel molecules related to membrane Ca
2+ current manifested as reduced expression of
Cacna1c,
Cacna2d2, and
Cacna2d1. These encode Ca
v1.2 channels and channel regulatory subunits, respectively. L-type Ca
2+ channel (Ca
v1.2) α
1c subunit was implicated as an important regulator of reentrant spiral dynamics and as a major component of AF-related electrical remodelling [
72]. In addition loss-of-function CACNA1C-Q1916R is thought to contribute to early repolarisation syndrome-related sudden cardiac death [
41], and gain of function CACNA1C-L762F with development of LQTS through slower channel inactivation and increased sustained and window current [
42].
α2/
δ2 is associated with increased peak amplitudes of N-, L- and T-type Ca
2+ currents expressed in
Xenopus oocytes [
43]; genetic knockout of α
2δ-1 is associated with reduced Ca
V2.2 levels and reduced Ca
2+ current densities [
44]. Mutations in the regulatory
Cacna2d1 are also associated with cardiac deficiencies, including Brugada and short QT syndromes [
73].
Finally, the present observations of reduced
Casq2-encoded calsequestrin 2 expression, important in SR Ca
2+ storage capacity, but relatively unchanged
Ryr2 and
Ryr3 expression mediating RyR-mediated SR Ca
2+ release is compatible with potentially pro-arrhythmic diastolic releases of SR Ca
2+ associated with either RyR2 [
74] or Casq2-mediated [
75] atrial pro-arrhythmic changes. The latter are associated with some catecholaminergic polymorphic ventricular tachycardic syndromes. In contrast to the relatively targeted gene functional changes of molecules involved in cellular electrophysiological activation and excitation contraction coupling, we did not observe changes in expression in channels known to be involved in action potential recovery through generation of voltage-dependent K
+ currents.
Surveys of indicators for adrenergic and cholinergic modulation and their associated intracellular signalling processes demonstrated no significant changes in the cardiac β-adrenergic receptor or atrial parasympathetic receptor, compatible with relatively little change in the transcriptional background to autonomic responsiveness in
Pgc-
1α−/− compared to
WT. Furthermore, changes observed in intracellular autonomic signalling were confined to neuronal adenylyl cyclise type 4, but not the cardiac type 5. Nevertheless, there was decreased expression of
Adra1d. The latter encodes α
1D adrenergic receptors thought to mediate Gq/11 signalling important in protective and adaptive functions against pathologic remodelling in heart failure [
50]. Finally, an examination of transcriptional features of molecules involved in longer term cellular and tissue, including fibrotic changes ultimately impacting AP generation and propagation (cf. [
55,
76]) did not reveal major change.
The findings provide a genomic background for future therapeutic explorations directed at atrial arrhythmias directed at its associated alterations in cellular energetic processes. The therapeutic directions suggested by such findings could suggest explorations of management strategies aimed directly at the ion channels indicated as affected here. The latter could include pacemaker based HCN modulation [
67,
77,
78]. They could also involve pharmacotherapeutic manoeuvres directed at expected alterations in ion channel functions, particularly bearing on the alterations in Ca
2+ homeostasis suggested here.
Alternatively, possibilities for more upstream therapies are suggested by both clinical studies and experimental results. Studies in rabbit models associated AF with reduced protein expression of sirtuin1, PGC-1α, and PPAR-α. AF patients showed reduced atrial tissue mast cell protease 1 (mCPT-1) and glucose transporter type 4 (GLUT4) protein expression, indicating reduced FA oxidation and glucose transport, compared to control patients in sinus-rhythm. Rabbits in AF showed a similar decrease in these molecules. The PPAR-α agonist fenofibrate then restored expression of mCPT-1 and GLUT4 and activation of the PPAR-α/sirtuin1/PGC-1α pathway, and suppressed AF inducibility [
13].
At the level of oxidative function, the bioactive polyphenol resveratrol is associated with diverse benefits including antioxidant cardioprotection, possibly through inducing mitochondrial biogenesis via PGC-1α activation [
29]. Resveratrol activates 5’ AMP-activated protein kinase (AMPK)/sirtuin1 signalling [
79], modulating cardiac metabolism. In addition, the multifunctional small-molecule resveratrol-derivative, C1 alters function in multiple ion channels mediating ultrarapid and acetylcholine-activated K
+ currents, as well as Na
+ currents. It showed antioxidant properties in human and rat atrial cardiomyocytes. It also reduced AF sustainability in atrial tachypaced dogs [
80]. Resveratrol also modified remodelling changes associated with AF in a rabbit heart failure (HF) model by activating PI3K/AKT/eNOS signaling and reducing AF susceptibility and triggered activity. This occurred by preventing atrial electrical, contractile, and fibrotic remodelling [
81]. It similarly prevented cardiomyopathy and restored cardiac function in mdx mice used to model Duchenne muscular dystrophy [
82], possibly through attenuating the expression of the p300 coactivator which is a key contributor to cardiac hypertrophy and fibrosis [
82].
The present analysis permits several physiological interpretations relevant to arrhythmogenesis; however this is limited to findings directly related to whole atrial tissue. There is increasing evidence to suggest that the cardiac fibroblast plays an important role in the excitation and propagation of electrical activity. It has been shown that contrary to the classical view, arrhythmogenic activity is not only specific to the cardiac myocyte but is related to heterocellular electrotonic interactions between cardiomyocytes and other cardiac related cells such as the cardiac myofibroblast. Such heterogeneity in electrical properties exist due the varying resting membrane potential of the cardiac myofibroblast relative to that of the cardiac myocyte [
83,
84]. Additionally, our present work used mice that were of advanced age where some level of fibrotic development may have occurred. It has been shown that in fibrotic hearts, proarrhythmic myofibroblast-cardiomyocyte crosstalk in vitro is mediated by TGF-β
1 [
83]. It is likely that selectively targeting the cardiac myofibroblast may be a useful anti-arrhythmic strategy as well. Given the important role cardiac myofibroblasts play in cardiac electrical properties, future studies should attempt at isolating and differentiating the gene expression and protein profile of native cardiac myofibroblasts and comparing these to corresponding features of native cardiac myocytes. Future studies should be directed at in-vivo electrophysiological interrogation of the
Pgc-
1α−/− murine atria including both native cardiac myocytes and cardiac myofibroblasts with particular focus on the identifying functional alterations that may correlate with the molecular findings in the present work. It would be additionally useful for future work studying ventricular gene and protein expression profile in the
Pgc-
1α−/− murine to allow comparison between atria and ventricular findings. Such information, whilst potentially not directly comparable due to the variation in tissue electrical properties between atria and ventricles, would allow for elucidation of other mitochondrial dysfunction mediated arrhythmogenic mechanisms.
4. Methods
4.1. Animals
All experimental protocols were approved under the UK Home Office regulations (Animals (Scientific Procedures) Act 1986 Amendment Regulations 2012) following ethical review by the University of Cambridge Animal Welfare and Ethical Review Body (AWERB) and conducted under a designated project license number PPL70/8726, dated 4 February 2016. All procedures complied with the UK Home Office regulations (Animals (Scientific Procedures) Act 1986). We also followed the Guide for the Care and Use of Laboratory Animals, U.S. National Institutes of Health (NIH Publication No. 85-23, revised 1996). An animal house maintained at 21 °C was used for the mice, with 12-h light/dark cycles. The mice had sterile chow (RM3 Maintenance Diet, SDS, Witham, Essex, UK) and free access to water, bedding and environmental stimuli. Mice were sacrificed by cervical dislocation and no anaesthetic or surgical procedures were required. Wild-type (WT) C57/B6 and Pgc-1α−/− (The Jackson Laboratory, Bar Harbor, ME, USA) adult mice were bred for the experimental protocols. Mice were bred on a C57/B6 background to avoid possible strain-related confounds. The mice were divided into WT and Pgc-1α−/−, and all were between the ages of 12–24 months old.
4.2. Quantitative PCR
In this gene expression study, there were
n = 3 mice in both the
WT and
Pgc-1α−/− groups. RNA was extracted from fresh frozen murine atria, stored at −80 °C, with the Qiagen RNeasy mini Plus kit (Qiagen, Manchester, UK)). The atrial tissue was weighed and quickly minced on ice. A third of the tissue, about 30 mg, was used in the next step of the RNA isolation protocol. Tissue pieces were taken from the ice and homogenised in RLT buffer supplemented with beta-mercaptoethanol with a Stuart handheld homogeniser until completely smooth. Genomic DNA was eliminated by centrifugation through a column supplied with the kit prior to extraction of the RNA according to the manufacturer’s protocol. RNA integrity was assessed by using an Agilent bioanalyser (Agilent Technologies, Santa Clara, CA, USA) to obtain RNA integrity numbers (RIN) according to the manufacturer’s protocol. RNA samples with RINs above 8 were used for the study. The RNA was used to prepare cDNA with High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Waltham, MA, USA) according to the manufacturer’s instructions. The efficiency of the cDNA protocol was tested by preparing the cDNA from a serial dilution of the RNA and then these cDNA samples were run on a qPCR confirming equal efficiency over a range of RNA concentrations. cDNA was also confirmed negative for genomic DNA contamination. Each custom-made card contained 64 pre-validated assays in triplicate with a reaction volume of 1 μL. The cards were run exactly according to instructions specific for the cards. Briefly, the cDNA (100 ng/well) was mixed with 2× Mastermix from Thermo Fisher (Waltham, MA, USA), 100 μL was loaded in each well slot on the cards, the cards were then spun down and sealed and run on a Quant 7 cycler (Applied Biosystems, Waltham, MA, USA). The amplification conditions were: 50 °C for 2 min and 95 °C for 10 min for the initial DNA melting and inactivation of RT reaction, followed by 40 cycles of 95 °C for 15 s and 60 °C for 60 s. Analysis of the Taqman array card data was performed by using the Quant studio software (Applied Biosystems, Waltham, MA, USA) and Microsoft Excel (Microsoft Corporation, Redmond, WA, USA) by calculating fold changes with the ΔC
t method as previously described [
31]. The threshold was set at 0.2 fluorescence units and the baseline range was set to automatic assignment. A combination of HPRT, Gapdh and ActinB were used as reference genes and amplifications were calculated with the regression threshold and baseline subtraction curve fit auto settings with the BioRad CFX manager software. The statistical analysis involved use of paired Student’s
t-test to compare for differences in expression of functional gene groups and use of unpaired Student’s
t-test to compare for differences in expression of individual genes between WT and
Pgc-
1α−/− atria, for a stratification of values of
p.
4.3. Western Blots
Murine atria were extracted and homogenised in 450µl of lysis buffer (150 mM NaCl, 25 mM tris(hydroxymethyl)aminomethane (tris), pH 7–8, 1% Triton-X100 detergent, 5 mM ethylenediaminetetraacetic acid (EDTA) and Roche® cOmplete™ mini protease inhibitor (Merck KGaA, Darmstadt, Germany)). After a 20-min centrifugation at 12,000 RPM, the clear lysate was obtained and a bicinchoninic acid (BCA) assay performed to assess for protein content (Thermo Scientific Microplate BCA Protein Assay Kit #23252: manufacturer recommended protocol). The samples underwent sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) using loading buffer (12.8 mL tris, pH 6.8, 3.2 g sodium dodecyl sulphate (SDS), 1.85 g dithiothreitol (DTT), 16 mL 100% glycerol, bromophenol blue, 11.2 mL H2O) in the ratio of 3:1 volume of clear lysate to loading buffer. The mixtures were heated at 70 °C for 5 min and loaded into Mini-Protean TGX™ (Bio-Rad, Watford, UK), 4–15% acrylamide gradient, precast gel wells (20 µg for Nav1.5 blots and 30 µg for Cx40 and Cx43 blots). Samples were run at 120 V for 30 min, then 250 V for 20 min and compared to β-tubulin as a loading control. Semi-dry transfer of the proteins was carried out onto polyvinylidene fluoride (PVDF) membranes (Immobilon™ PVDF membrane, Merck KGaA, Darmstadt, Germany) using Trans-Blot® Turbo™ kit (BioRad, Watford, UK). The transfer settings were 1.3 A current and 25 V potential for 10 min. The PVDF membranes were then blocked with Odyssey® blocking buffer (Li-Cor Biosciences, Cambridge, UK) for one hour at room temperature, rinsed with PBS-T (0.1% Tween) and incubated with primary antibody diluted in Odyssey® blocking buffer diluted 33% in PBS-T overnight at 4 °C. The primary antibodies used were Nav1.5 (Cell Signalling Technology, London, UK), Cx43 (Sigma-Aldrich Company Ltd, Gillingham, UK), Cx40 (Santa Cruz Biotechnology, Dallas, TX, USA) and β-tubulin (Abcam, Cambridge, UK). The membranes were washed three times and then incubated with secondary antibodies diluted in Odyssey® blocking buffer diluted 33% in PBS-T at room temperature for 45 min. Imaging of the blots utilised the Odyssey® Fc imaging system (Li-Cor Biosciences, Cambridge, UK), which measured emission from the secondary antibodies at 600 and 800 nm. Image Studio™ software (Image Studio 4.0, Li-Cor Biosciences, Cambridge, UK) was used to quantify the protein band intensity and subtract the background signal, and then express this relative to the control, β-tubulin, signal. Statistical analysis was performed using the unpaired Student’s t-test.