Ero1-Mediated Reoxidation of Protein Disulfide Isomerase Accelerates the Folding of Cone Snail Toxins
Abstract
1. Introduction
2. Results
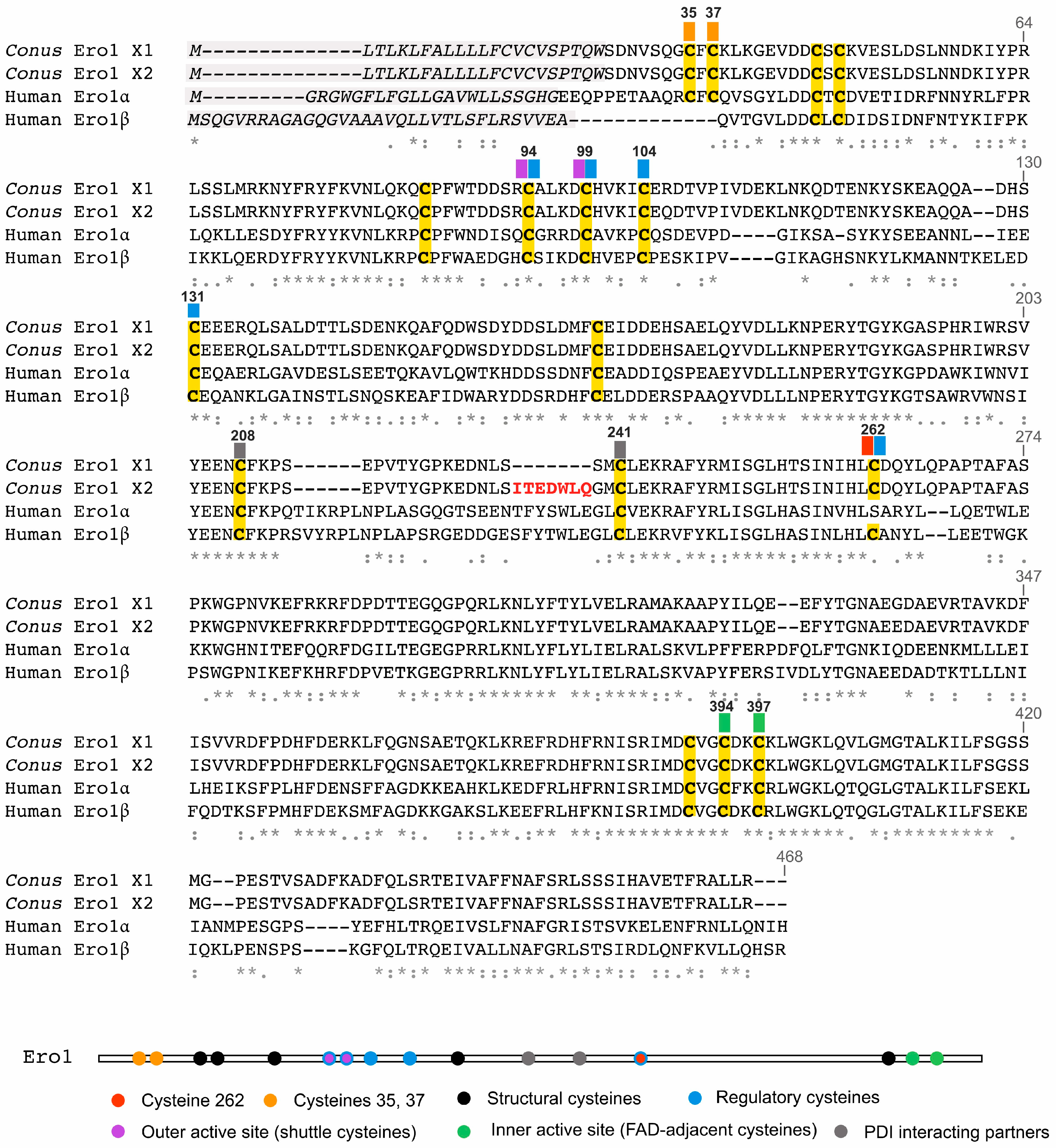
2.1. cDNA Sequencing of Ero1 from the Venom Gland of C. Geographus
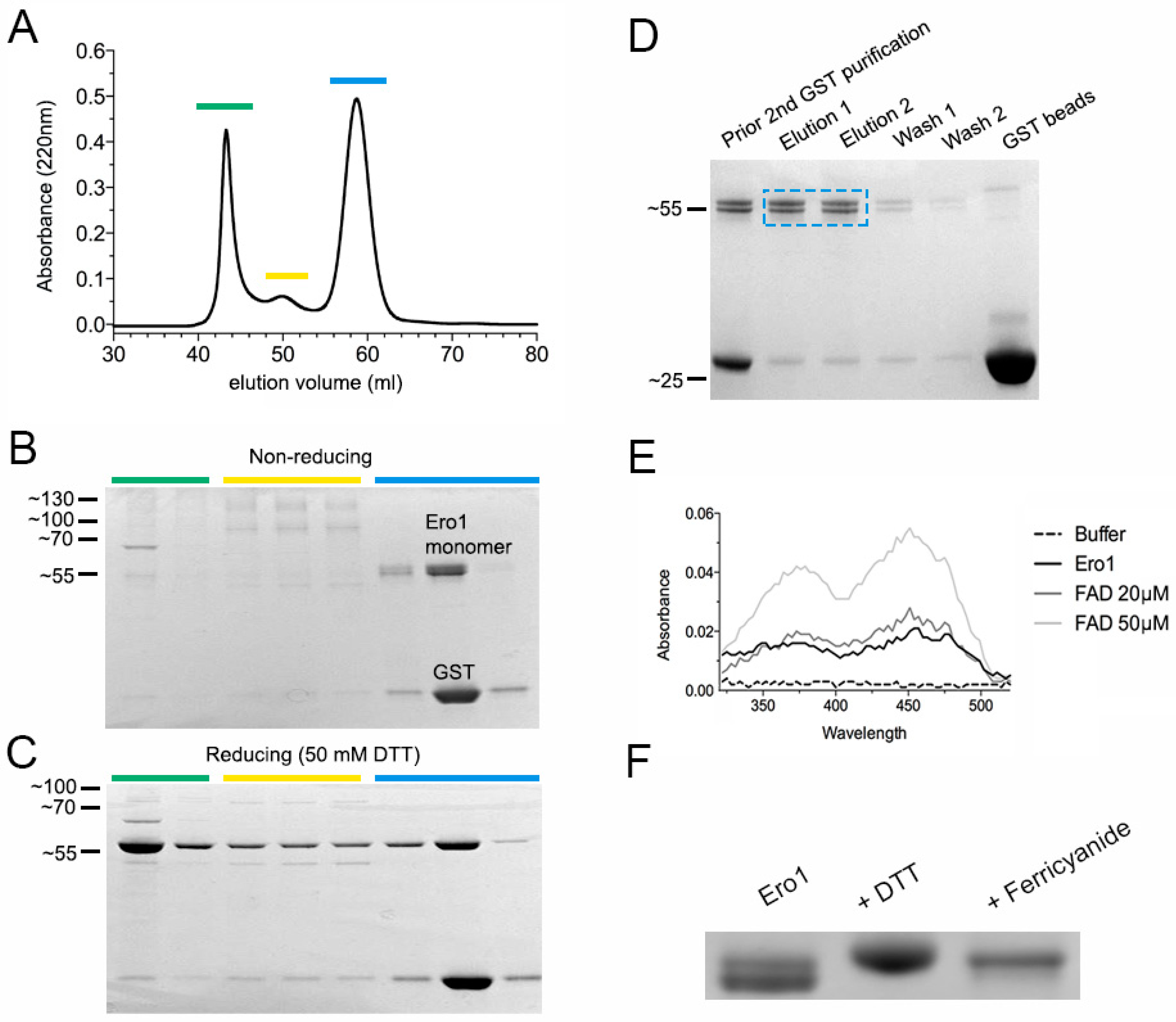
2.2. Conus Ero1 Expression Yields a Functional, FAD-Binding Enzyme
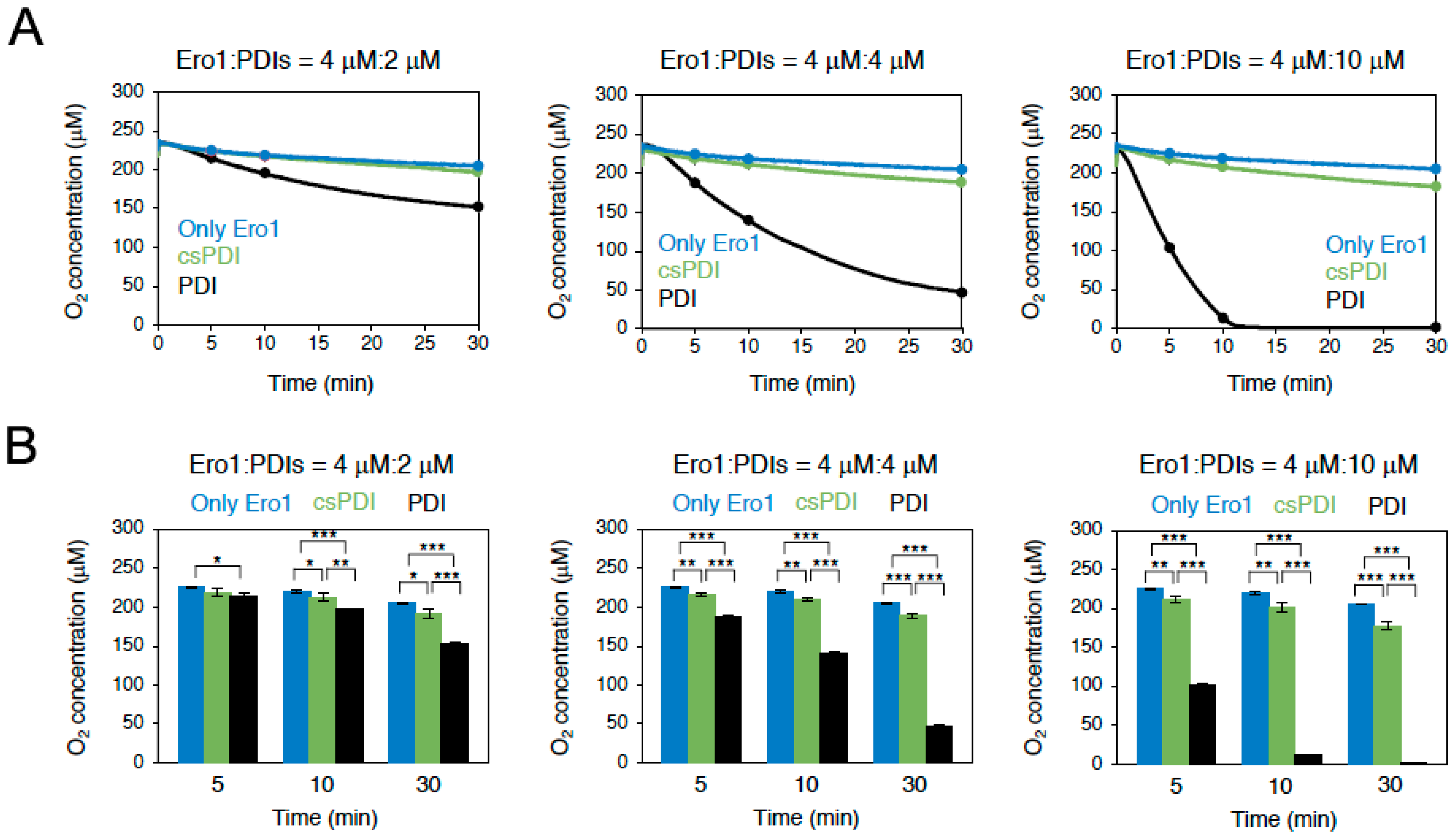
2.3. Conus Ero1 Preferentially Oxidizes PDI Over csPDI
2.4. Conus Ero1 Efficiently Oxidizes PDI but Has Low Reactivity for csPDI
2.5. Conus Ero1 Efficiently Re-Oxidizes PDI and csPDI During the Folding of Conotoxins
3. Discussion
4. Materials and Methods
4.1. Specimen Collection
4.2. Sequencing and Cloning of Ero1
4.3. Expression and Purification of Ero1
4.4. Cloning, Expression and Purification of C. Geographus PDI and csPDI
4.5. Oxygen Consumption Assay
4.6. Redox State Analysis of Conus PDI or csPDI in the Presence of Ero1
4.7. Synthesis of Peptide Substrates for Oxidative Folding Studies
4.8. Oxidative Folding Assays
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACN | acetonitrile |
| csPDI | conotoxin-specific protein disulfide isomerase |
| DTT | dithiothreitol |
| ER | endoplasmic reticulum |
| Ero1 | endoplasmic reticulum oxidoreductin-1 |
| FAD | flavin adenine dinucleotide |
| Fmoc | N-(9-fluorenyl)methoxycarbonyl |
| HBTU | O-(benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate |
| ORF | open reading frame |
| PDI | protein disulfide isomerase |
| RP-HPLC | reversed-phase high performance liquid chromatography |
| SEC | size exclusion chromatography |
| TFA | trifluoroacetic acid |
References
- Safavi-Hemami, H.; Foged, M.M.; Ellgaard, L. Evolutionary Adaptations to Cysteine-Rich Peptide Folding. In Oxidative Folding of Peptides and Proteins, Morode; RSC Press: London, UK, 2018; ISBN 978-1-78262-990-0. [Google Scholar]
- Safavi-Hemami, H.; Li, Q.; Jackson, R.L.; Song, A.S.; Boomsma, W.; Bandyopadhyay, P.K.; Gruber, C.W.; Purcell, A.W.; Yandell, M.; Olivera, B.M.; et al. Rapid expansion of the protein disulfide isomerase gene family facilitates the folding of venom peptides. Proc. Natl. Acad. Sci. USA 2016, 113, 3227–3232. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Hemami, H.; Siero, W.A.; Gorasia, D.G.; Young, N.D.; MacMillan, D.; Williamson, N.A.; Purcell, A.W. Specialisation of the venom gland proteome in predatory cone snails reveals functional diversification of the conotoxin biosynthetic pathway. J. Proteome Res. 2011, 10, 3904–3919. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Han, Y.H.; Shao, X.X.; Chi, C.W.; Guo, Z.Y. Molecular cloning, expression and characterization of protein disulfide isomerase from Conus marmoreus. FEBS J. 2007, 274, 4778–4787. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Hemami, H.; Gorasia, D.G.; Steiner, A.M.; Williamson, N.A.; Karas, J.A.; Gajewiak, J.; Olivera, B.M.; Bulaj, G.; Purcell, A.W. Modulation of conotoxin structure and function is achieved through a multienzyme complex in the venom glands of cone snails. J. Biol. Chem. 2012, 287, 34288–34303. [Google Scholar] [CrossRef] [PubMed]
- Bulleid, N.J.; Ellgaard, L. Multiple ways to make disulfides. Trend Biochem. Sci. 2011, 36, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Inaba, K. Disulfide bond formation network in the three biological kingdoms, bacteria, fungi and mammals. FEBS J. 2012, 279, 2262–2271. [Google Scholar] [CrossRef] [PubMed]
- Zito, E. ERO1: A protein disulfide oxidase and H2O2 producer. Free Radic. Biol. Med. 2015, 83, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Tavender, T.J.; Bulleid, N.J. Molecular mechanisms regulating oxidative activity of the Ero1 family in the endoplasmic reticulum. Antiox. Redox Signal. 2010, 13, 1177–1187. [Google Scholar] [CrossRef] [PubMed]
- Araki, K.; Inaba, K. Structure, mechanism, and evolution of Ero1 family enzymes. Antiox. Redox Signal. 2012, 16, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Inaba, K.; Masui, S.; Iida, H.; Vavassori, S.; Sitia, R.; Suzuki, M. Crystal structures of human Ero1alpha reveal the mechanisms of regulated and targeted oxidation of PDI. EMBO J. 2010, 29, 3330–3343. [Google Scholar] [CrossRef] [PubMed]
- Ramming, T.; Okumura, M.; Kanemura, S.; Baday, S.; Birk, J.; Moes, S.; Spiess, M.; Jeno, P.; Berneche, S.; Inaba, K.; et al. A PDI-catalyzed thiol-disulfide switch regulates the production of hydrogen peroxide by human Ero1. Free Radic. Biol. Med. 2015, 83, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, A.; Korhonen, K.; Saaranen, M.J.; Ruddock, L.W. Molecular analysis of human Ero1 reveals novel regulatory mechanisms for oxidative protein folding. Life Sci. Alliance 2018, 1. [Google Scholar] [CrossRef]
- Kanemura, S.; Okumura, M.; Yutani, K.; Ramming, T.; Hikima, T.; Appenzeller-Herzog, C.; Akiyama, S.; Inaba, K. Human ER Oxidoreductin-1alpha (Ero1alpha) Undergoes Dual Regulation through Complementary Redox Interactions with Protein-Disulfide Isomerase. J. Biol. Chem. 2016, 291, 23952–23964. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Kuma, K.; Toh, H.; Miyata, T. MAFFT version 5: Improvement in accuracy of multiple sequence alignment. Nucleic Acids Res. 2005, 20, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.P.; Weissman, J.S. The FAD- and O (2)-dependent reaction cycle of Ero1-mediated oxidative protein folding in the endoplasmic reticulum. Mol. Cell 2002, 10, 983–994. [Google Scholar] [CrossRef]
- Gross, E.; Sevier, C.S.; Heldman, N.; Vitu, E.; Bentzur, M.; Kaiser, C.A.; Thorpe, C.; Fass, D. Generating disulfides enzymatically: Reaction products and electron acceptors of the endoplasmic reticulum thiol oxidase Ero1p. Proc. Natl. Acad. Sci. USA 2006, 103, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Appenzeller-Herzog, C.; Riemer, J.; Christensen, B.; Sørensen, E.S.; Ellgaard, L. A novel disulphide switch mechanism in Ero1alpha balances ER oxidation in human cells. EMBO J. 2008, 27, 2977–2987. [Google Scholar] [CrossRef] [PubMed]
- Price-Carter, M.; Gray, W.R.; Goldenberg, D.P. Folding of omega-conotoxins. 2. Influence of precursor sequences and protein disulfide isomerase. Biochemistry 1996, 35, 15547–15557. [Google Scholar] [CrossRef] [PubMed]
- Fuller, E.; Green, B.R.; Catlin, P.; Buczek, O.; Nielsen, J.S.; Olivera, B.M.; Bulaj, G. Oxidative folding of conotoxins sharing an identical disulfide bridging framework. FEBS J. 2005, 272, 1727–1738. [Google Scholar] [CrossRef] [PubMed]
- Thapa, P.; Espiritu, M.J.; Cabalteja, C.C.; Bingham, J.P. Conotoxins and their regulatory considerations. Regul. Toxicol. Pharmacol. 2014, 70, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Norton, R.S.; Pallaghy, P.K. The cystine knot structure of ion channel toxins and related polypeptides. Toxicon 1998, 36, 1573–1583. [Google Scholar] [CrossRef]
- Bayrhuber, M.; Vijayan, V.; Ferber, M.; Graf, R.; Korukottu, J.; Imperial, J.; Garrett, J.E.; Olivera, B.M.; Terlau, H.; Zweckstetter, M.; et al. Conkunitzin-S1 is the first member of a new Kunitz-type neurotoxin family: Structural and functional characterization. J. Biol. Chem. 2005, 280, 23766–23770. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Hemami, H.; Gajewiak, J.; Karanth, S.; Robinson, S.D.; Ueberheide, B.; Douglass, A.D.; Schlegel, A.; Imperial, J.S.; Watkins, M.; Bandyopadhyay, P.K.; et al. Specialized insulin is used for chemical warfare by fish-hunting cone snails. Proc. Natl. Acad. Sci. USA 2015, 112, 1743–1748. [Google Scholar] [CrossRef] [PubMed]
- Gowd, K.H.; Krishnan, K.S.; Balaram, P.I. Identification of Conus amadis disulfide isomerase: Minimum sequence length of peptide fragments necessary for protein annotation. Mol. Biosyst. 2007, 3, 554–566. [Google Scholar] [CrossRef] [PubMed]
- Buczek, O.; Bulaj, G.; Olivera, B.M. Conotoxins and the posttranslational modification of secreted gene products. Cell. Mol. Life Sci. 2005, 62, 3067–3079. [Google Scholar] [CrossRef] [PubMed]
- Pollard, M.G.; Travers, K.J.; Weissman, J.S. Ero1p: A novel and ubiquitous protein with an essential role in oxidative protein folding in the endoplasmic reticulum. Mol. Cell 1998, 1, 171–182. [Google Scholar] [CrossRef]
- Frand, A.R.; Kaiser, C.A. The ERO1 gene of yeast is required for oxidation of protein dithiols in the endoplasmic reticulum. Mol. Cell 1998, 1, 161–170. [Google Scholar] [CrossRef]
- Wang, L.; Li, S.J.; Sidhu, A.; Zhu, L.; Liang, Y.; Freedman, R.B.; Wang, C.C. Reconstitution of human Ero1-Lalpha/protein-disulfide isomerase oxidative folding pathway in vitro. Position-dependent differences in role between the a and a′ domains of protein-disulfide isomerase. J. Biol. Chem. 2009, 284, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Masui, S.; Vavassori, S.; Fagioli, C.; Sitia, R.; Inaba, K. Molecular bases of cyclic and specific disulfide interchange between human ERO1alpha protein and protein-disulfide isomerase (PDI). J. Biol. Chem. 2011, 286, 16261–16271. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, W.; Ren, J.; Fang, J.; Ke, H.; Gong, W.; Feng, W.; Wang, C.C. Structural insights into the redox-regulated dynamic conformations of human protein disulfide isomerase. Antiox. Redox Signal. 2013, 19, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kojima, R.; Okumura, M.; Hagiwara, M.; Masui, S.; Maegawa, K.; Saiki, M.; Horibe, T.; Suzuki, M.; Inaba, K. Synergistic cooperation of PDI family members in peroxiredoxin 4-driven oxidative protein folding. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Bandyopadhyay, P.K.; Olivera, B.M.; Yandell, M. Characterization of the Conus bullatus genome and its venom-duct transcriptome. BMC Genom. 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Whorlow, S.L.; Angus, J.A.; Wright, C.E. Selectivity of omega-conotoxin GVIA for n-type calcium channels in rat isolated small mesenteric arteries. Clin. Exp. Pharmacol. Physiol. 1996, 23, 16–21. [Google Scholar] [CrossRef] [PubMed]
- West, P.J.; Bulaj, G.; Garrett, J.E.; Olivera, B.M.; Yoshikami, D. Mu-conotoxin SmIIIA, a potent inhibitor of tetrodotoxin-resistant sodium channels in amphibian sympathetic and sensory neurons. Biochemistry 2002, 41, 15388–15393. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Brien, H.; Kanemura, S.; Okumura, M.; Baskin, R.P.; Bandyopadhyay, P.K.; Olivera, B.M.; Ellgaard, L.; Inaba, K.; Safavi-Hemami, H. Ero1-Mediated Reoxidation of Protein Disulfide Isomerase Accelerates the Folding of Cone Snail Toxins. Int. J. Mol. Sci. 2018, 19, 3418. https://doi.org/10.3390/ijms19113418
O’Brien H, Kanemura S, Okumura M, Baskin RP, Bandyopadhyay PK, Olivera BM, Ellgaard L, Inaba K, Safavi-Hemami H. Ero1-Mediated Reoxidation of Protein Disulfide Isomerase Accelerates the Folding of Cone Snail Toxins. International Journal of Molecular Sciences. 2018; 19(11):3418. https://doi.org/10.3390/ijms19113418
Chicago/Turabian StyleO’Brien, Henrik, Shingo Kanemura, Masaki Okumura, Robert P. Baskin, Pradip K. Bandyopadhyay, Baldomero M. Olivera, Lars Ellgaard, Kenji Inaba, and Helena Safavi-Hemami. 2018. "Ero1-Mediated Reoxidation of Protein Disulfide Isomerase Accelerates the Folding of Cone Snail Toxins" International Journal of Molecular Sciences 19, no. 11: 3418. https://doi.org/10.3390/ijms19113418
APA StyleO’Brien, H., Kanemura, S., Okumura, M., Baskin, R. P., Bandyopadhyay, P. K., Olivera, B. M., Ellgaard, L., Inaba, K., & Safavi-Hemami, H. (2018). Ero1-Mediated Reoxidation of Protein Disulfide Isomerase Accelerates the Folding of Cone Snail Toxins. International Journal of Molecular Sciences, 19(11), 3418. https://doi.org/10.3390/ijms19113418






