Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts
Abstract
1. Introduction
2. Results
2.1. Identification of Components by Ultra-High Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UPLC-QTOF-MS) Analysis
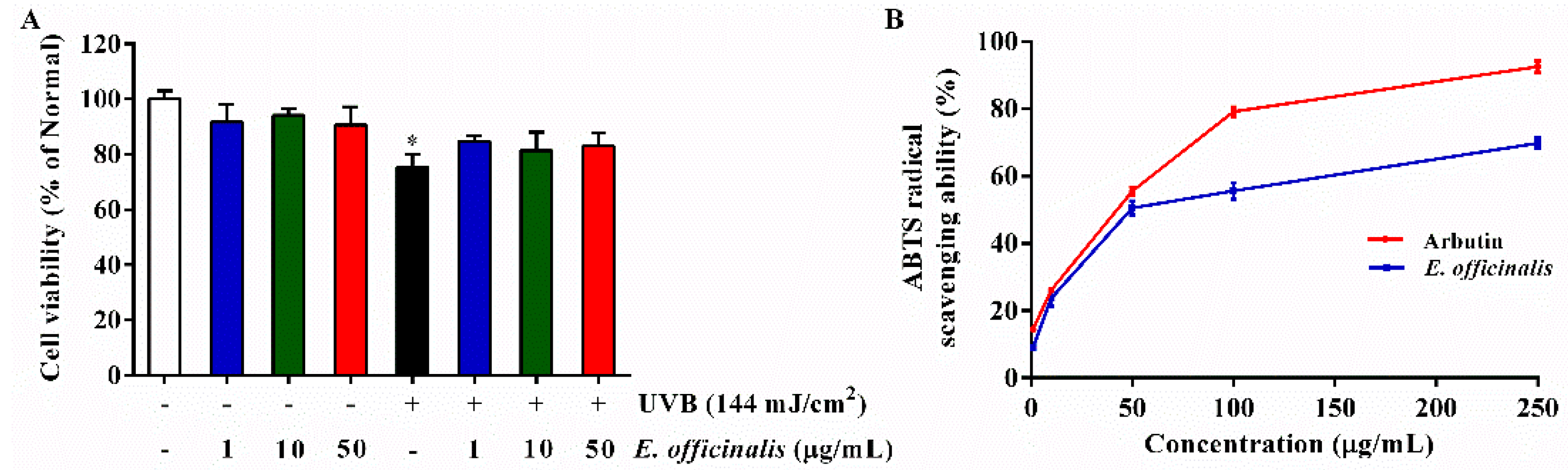
2.2. Effect of E. officinalis on Cell Viability
2.3. 2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) Radical Scavenging Activity of E. officinalis
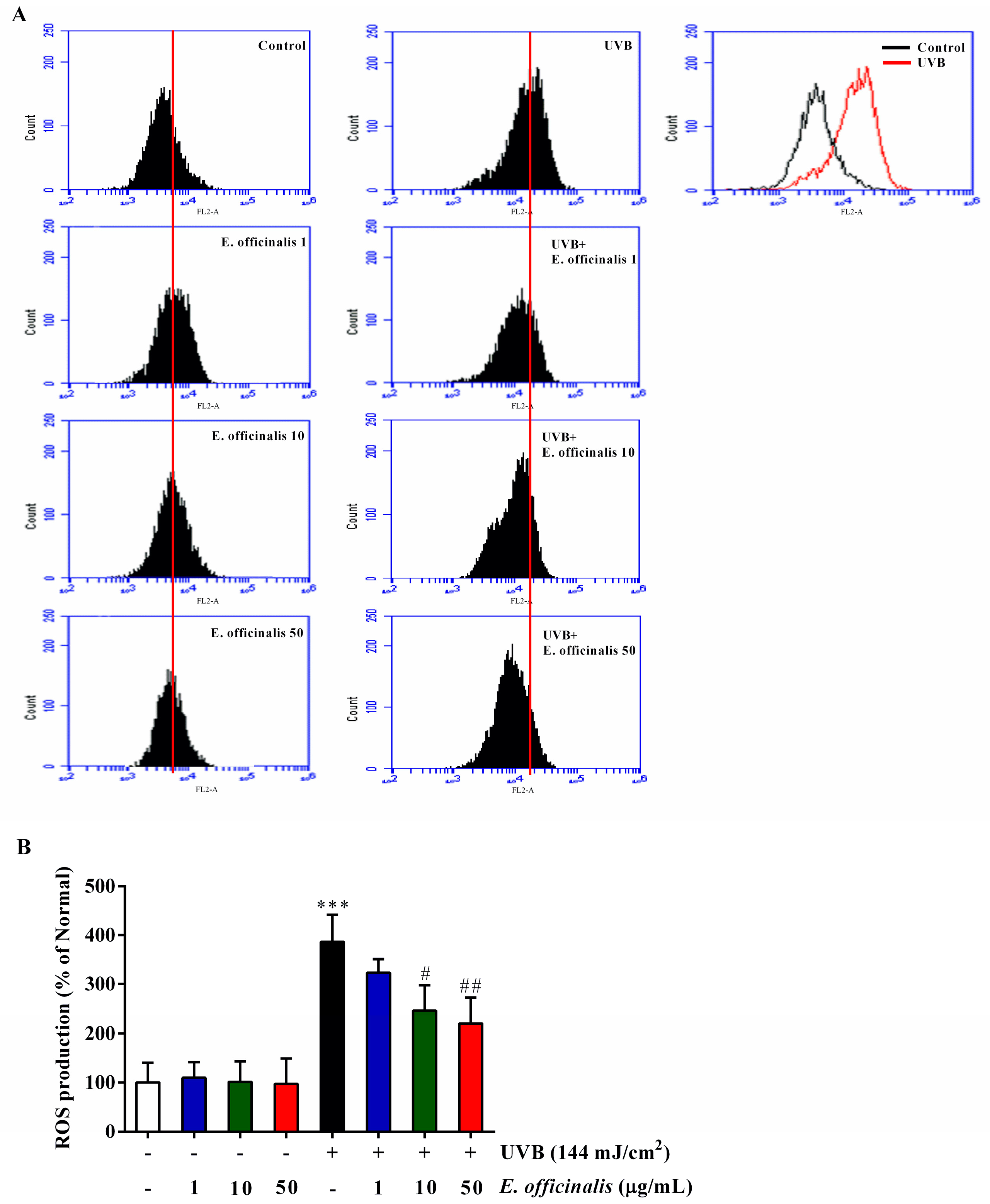
2.4. Effects of E. officinalis on Intracellular Reactive Oxygen Species (ROS) Production
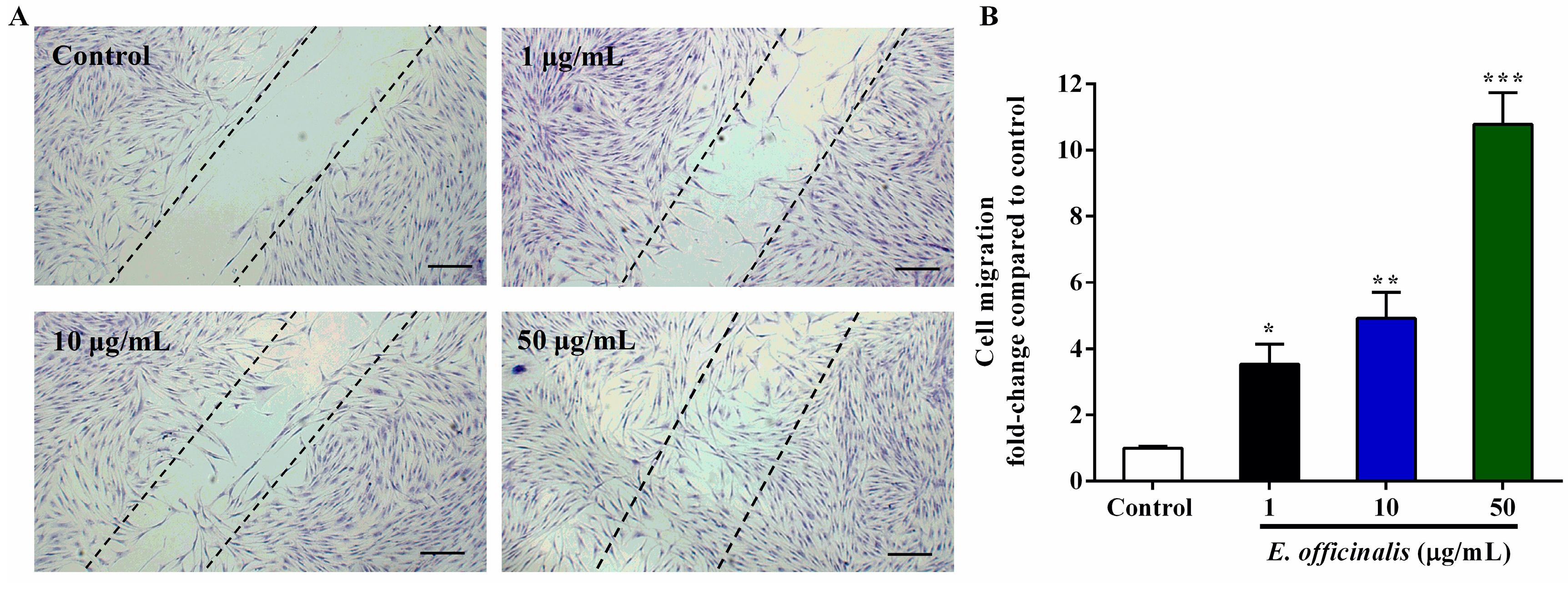
2.5. Effect of E. officinalis on Normal Human Dermal Fibroblast (NHDF) Cell Migration
2.6. Effect of E. officinalis on NHDF Cell Apoptosis
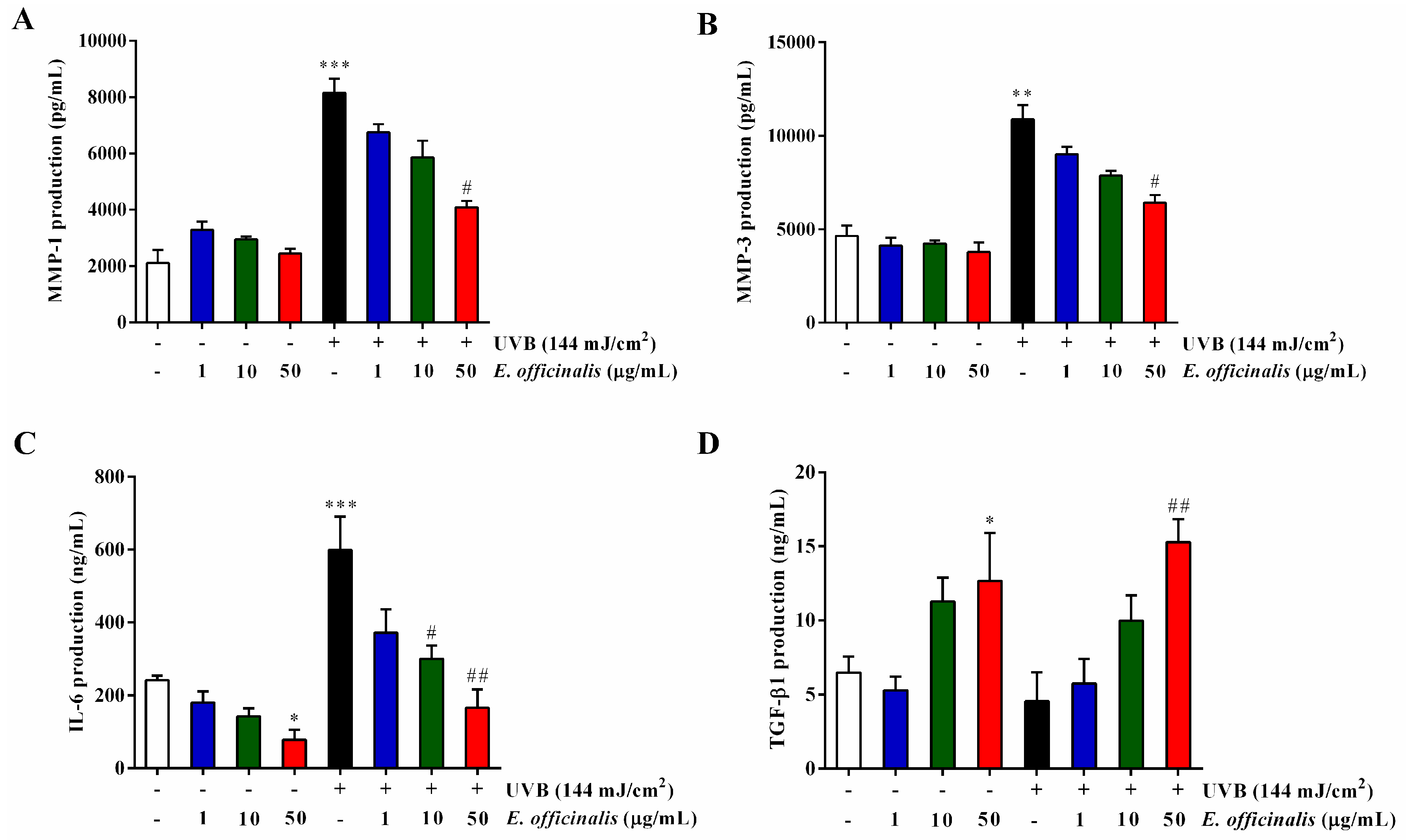
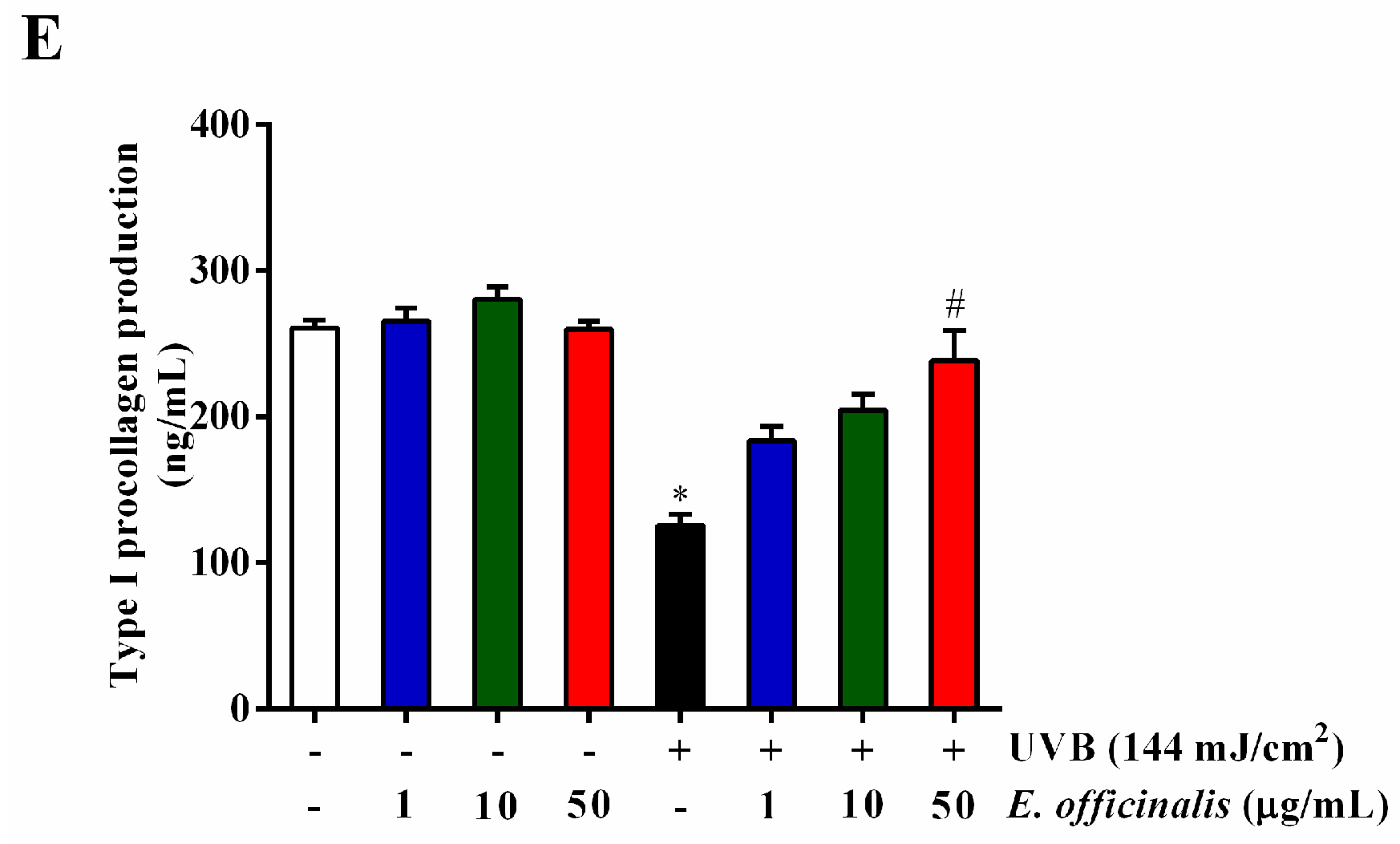
2.7. Effect of E. officinalis on MMP-1, MMP-3, IL-6, TGF-β1, and Type I Procollagen Production
2.8. Effect of E. officinalis on the Expression of MMP-1 and Type I Procollagen
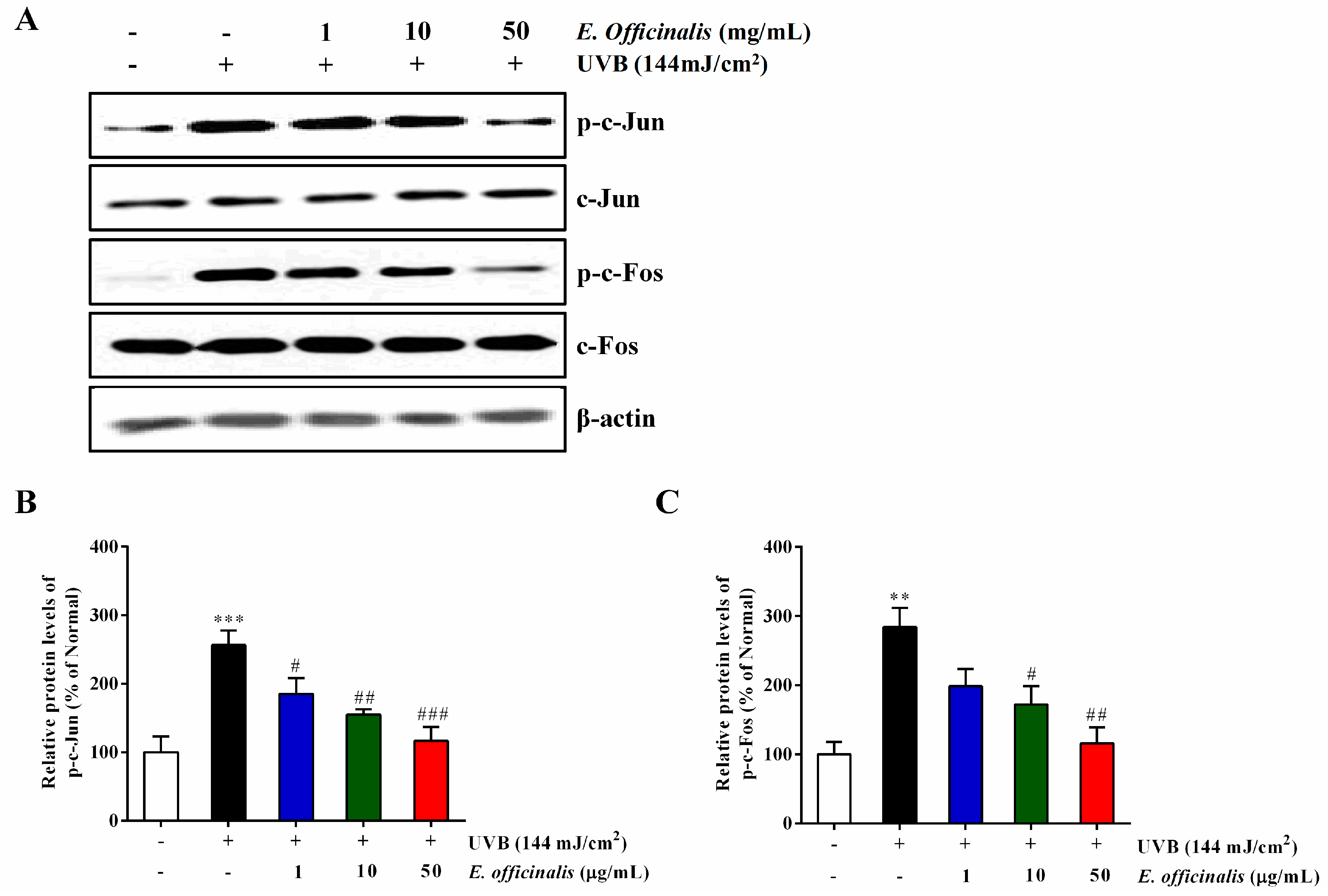
2.9. Effect of E. officinalis on Activator Protein 1 (AP-1) Transcription
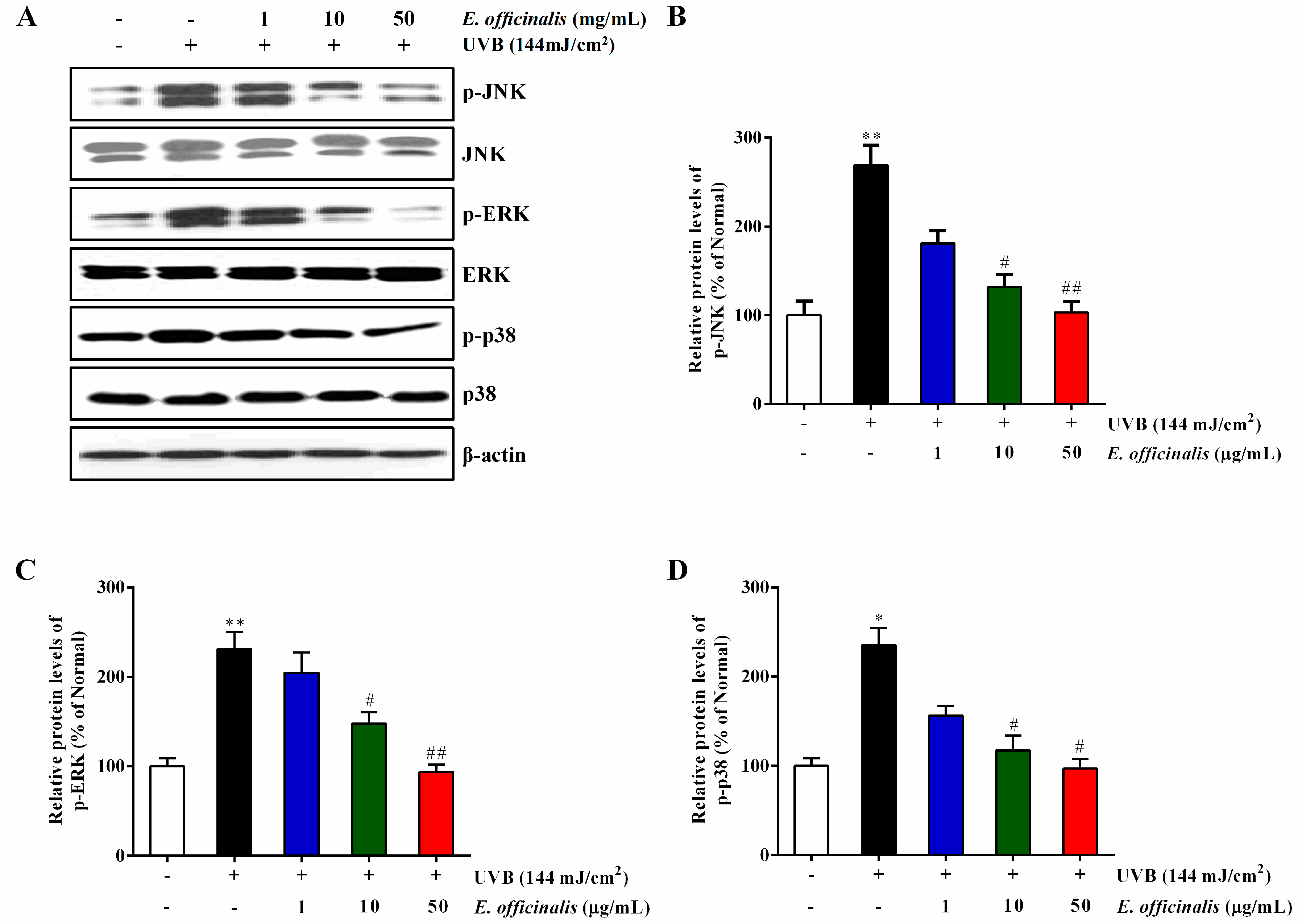
2.10. Effect of E. officinalis on the MAPK Signaling Pathway
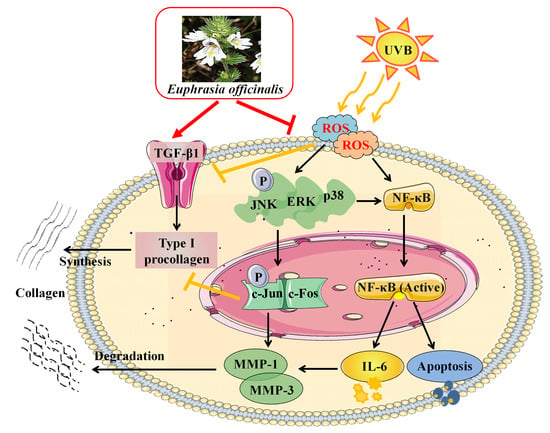
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Sample Preparation
4.3. UPLC-QTOF-MS Analysis
4.4. Cell Culture, UVB Irradiation, and Sample Treatment
4.5. Cell Viability and Light Microscopic Analysis
4.6. Measurement of ABTS Radical Scavenging Activity
4.7. Measurement of ROS Generation
4.8. Wound Healing Assay
4.9. Hoechst 33258 Staining
4.10. Determination of MMP-1, MMP-3, IL-6, TGF-β1, and Type I Procollagen
4.11. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
4.12. Western Blot Analysis
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Naidoo, K.; Hanna, R.; Birch-Machin, M.A. What is the role of mitochondrial dysfunction in skin photoaging? Exp. Dermatol. 2018, 27, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Wittenauer, J.; Mäckle, S.; Sußmann, D.; Schweiggert-Weisz, U.; Carle, R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia 2015, 101, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Stenehjem, J.S.; Robsahm, T.E.; Bratveit, M.; Samuelsen, S.O.; Kirkeleit, J.; Grimsrud, T.K. Ultraviolet radiation and skin cancer risk in offshore workers. Occup. Med. 2017, 67, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, M.; Ando, H.; Yoshida, M.; Niki, Y.; Matsui, M. Photoaging of the skin. Anti-Aging Med. 2009, 6, 46–59. [Google Scholar] [CrossRef]
- Silveira, J.E.P.S.; Pedroso, D.M.M. UV light and skin aging. Rev. Environ. Health 2014, 29, 243–254. [Google Scholar]
- Ichihashi, M.; Ando, H. The maximal cumulative solar UVB dose allowed to maintain healthy and young skin and prevent premature photoaging. Exp. Dermatol. 2014, 23, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Gaiba, S.; Tucci-Viegas, V.M.; Franca, L.P.; Lasakosvitsch, F.; Azevedo, F.L.; Moraes, A.A.; Ferreira, A.T.; Franca, J.P. Biological effects induced by ultraviolet radiation in human fibroblasts. In Flow Cytometry-Recent Perspectives; InTech: London, UK, 2012. [Google Scholar]
- Son, Y.; Kim, S.; Chung, H.T.; Pae, H.O. Reactive oxygen species in the activation of MAP kinases. Methods Enzymol. 2013, 528, 27–48. [Google Scholar] [PubMed]
- Barr, R.K.; Bogoyevitch, M.A. The c-Jun N-terminal protein kinase family of mitogen-activated protein kinases (JNK MAPKs). Int. J. Biochem. Cell Biol. 2001, 33, 1047–1063. [Google Scholar] [CrossRef]
- Klein, T.; Bischoff, R. Physiology and pathophysiology of matrix metalloproteases. Amino Acids 2011, 41, 271–290. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yan, Y.; Li, L.; Peng, S.; Qu, T.; Wang, B. Ultraviolet B-induced apoptosis of human skin fibroblasts involves activation of caspase-8 and -3 with increased expression of vimentin. Photodermatol. Photoimmunol. Photomed. 2010, 26, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Wu, S.-B.; Hong, C.-H.; Yu, H.-S.; Wei, Y.-H. Molecular mechanisms of UV-induced apoptosis and its effects on skin residential cells: The implication in UV-based phototherapy. Int. J. Mol. Sci. 2013, 14, 6414–6435. [Google Scholar] [CrossRef] [PubMed]
- Bielory, L.; Heimall, J. Review of complementary and alternative medicine in treatment of ocular allergies. Curr. Opin. Allergy Clin. Immunol. 2003, 3, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Fraunfelder, F.W. Ocular side effects from herbal medicines and nutritional supplements. Am. J. Ophthalmol. 2004, 138, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Petrichenko, V.; Sukhinina, T.; Babiyan, L.; Shramm, N. Chemical composition and antioxidant properties of biologically active compounds from Euphrasia brevipila. Pharm. Chem. J. 2006, 40, 312–316. [Google Scholar] [CrossRef]
- Singh, H.; Du, J.; Singh, P.; Yi, T.H. Ecofriendly synthesis of silver and gold nanoparticles by Euphrasia officinalis leaf extract and its biomedical applications. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Porchezhian, E.; Ansari, S.H.; Shreedharan, N.K. Antihyperglycemic activity of Euphrasia officinale leaves. Fitoterapia 2000, 71, 522–526. [Google Scholar] [CrossRef]
- Dimitrova, M.; Hristova, L.; Damianova, E.; Yordanova, Y.; Petrova, N.; Kapchina-Toteva, V. Antioxidant activity and secondary metabolites in different extracts of Euphrasia officinalis L. growing in Bulgaria. Sci. Technol. Med. 2014, 4, 465–469. [Google Scholar]
- Paduch, R.; Woźniak, A.; Niedziela, P.; Rejdak, R. Assessment of eyebright (Euphrasia officinalis L.) extract activity in relation to human corneal cells using in vitro tests. Balkan Med. J. 2014, 31, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Novy, P.; Davidova, H.; Serrano-Rojero, C.S.; Rondevaldova, J.; Pulkrabek, J.; Kokoska, L. Composition and Antimicrobial Activity of Euphrasia rostkoviana Hayne Essential Oil. Evid. Based Complement. Altern. Med. 2015, 2015, 734101. [Google Scholar] [CrossRef] [PubMed]
- Bigagli, E.; Cinci, L.; D’Ambrosio, M.; Luceri, C. Pharmacological activities of an eye drop containing Matricaria chamomilla and Euphrasia officinalis extracts in UVB-induced oxidative stress and inflammation of human corneal cells. J. Photochem. Photobiol. B 2017, 173, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Blazics, B.; Alberti, Á.; Kursinszki, L.; Kéry, Á.; Béni, S.; Tölgyesi, L. Identification and LC-MS-MS determination of acteoside, the main antioxidant compound of Euphrasia rostkoviana, using the isolated target analyte as external standard. J. Chromatogr. Sci. 2011, 49, 203–208. [Google Scholar] [CrossRef]
- Rajendram, R.; Preedy, V.R.; Patel, V.B. Branched Chain Amino Acids in Clinical Nutrition; Humana Press: New York, NY, USA, 2015. [Google Scholar]
- Philips, N.; Siomyk, H.; Bynum, D.; Gonzalez, S. Skin cancer, polyphenols, and oxidative stress. In Cancer; Elsevier: London, UK, 2014; pp. 265–270. [Google Scholar]
- Sun, Z.; Park, S.Y.; Hwang, E.; Zhang, M.; Seo, S.A.; Lin, P.; Yi, T.H. Thymus vulgaris alleviates UVB irradiation induced skin damage via inhibition of MAPK/AP-1 and activation of Nrf2-ARE antioxidant system. J. Cell. Mol. Med. 2017, 21, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Rafaela, T.; Silva, L.R. Bioactive compounds and in vitro biological activity of Euphrasia rostkoviana Hayne extracts. Ind. Crops Prod. 2013, 50, 680–689. [Google Scholar]
- Daniela, G.; Vincenzo, D.F. Euphrasia rostkoviana Hayne-Active Components and Biological Activity for the Treatment of Eye Disorders; UzhNU Publishing House “Goverla”: Uzhhorod, Ukraine, 2017. [Google Scholar]
- Ellery, D. Herbal Formulation for Treating Chronic Fatigue Syndrome. U.S. Patent 20170232050, 17 August 2017. [Google Scholar]
- Sun, Z.; Park, S.Y.; Hwang, E.; Park, B.; Seo, S.A.; Cho, J.-G.; Zhang, M.; Yi, T.-H. Dietary Foeniculum vulgare Mill extract attenuated UVB irradiation-induced skin photoaging by activating of Nrf2 and inhibiting MAPK pathways. Phytomedicine 2016, 23, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Salucci, S.; Burattini, S.; Battistelli, M.; Baldassarri, V.; Maltarello, M.C.; Falcieri, E. Ultraviolet B (UVB) irradiation-induced apoptosis in various cell lineages in vitro. Int. J. Mol. Sci. 2012, 14, 532–546. [Google Scholar] [CrossRef] [PubMed]
- Subedi, L.; Lee, T.H.; Wahedi, H.M.; Baek, S.-H.; Kim, S.Y. Resveratrol-enriched rice attenuates UVB-ROS-induced skin aging via downregulation of inflammatory cascades. Oxid. Med. Cell. Longev. 2017, 2017, 8379539. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Fan, J.; Chen, M.; Guan, S.; Sawcer, D.; Bokoch, G.M.; Woodley, D.T. Mechanism of human dermal fibroblast migration driven by type I collagen and platelet-derived growth factor-BB. Mol. Biol. Cell 2004, 15, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Kulka, M. Mechanisms and treatment of photoaging and photodamage. In Using Old Solutions to New Problems-Natural Drug Discovery in the 21st Century; InTech: London, UK, 2013. [Google Scholar]
- Campo, G.M.; Avenoso, A.; Campo, S.; D’Ascola, A.; Ferlazzo, A.M.; Sama, D.; Calatroni, A. Purified human chondroitin-4-sulfate reduced MMP/TIMP imbalance induced by iron plus ascorbate in human fibroblast cultures. Cell Biol. Int. 2006, 30, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Van Doren, S.R. Matrix metalloproteinase interactions with collagen and elastin. Matrix Biol. 2015, 44, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Kim, S.H.; Lee, S.; Lee, C.H.; Do, S.G.; Kim, J.; Kim, S.Y. A comparative study of baby immature and adult shoots of Aloe vera on UVB-induced skin photoaging in vitro. Phytother. Res. 2013, 27, 1874–1882. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Noh, E.M.; Song, H.K.; Lee, G.S.; Kwon, K.B.; Lee, Y.R. Reversine inhibits MMP-1 and 3 expressions by suppressing of ROS/MAPK/AP-1 activation in UV-stimulated human keratinocytes and dermal fibroblasts. Exp. Dermatol. 2018, 27, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Park, S.Y.; Hwang, E.; Zhang, M.; Jin, F.; Zhang, B.; Yi, T.H. Salvianolic Acid B Protects Normal Human Dermal Fibroblasts Against Ultraviolet B Irradiation-Induced Photoaging Through Mitogen-Activated Protein Kinase and Activator Protein-1 Pathways. Photochem. Photobiol. 2015, 91, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Kim, D.Y.; Chung, E.; Lee, E.A.; Park, K.-S.; Son, Y. Transplantation of cyclic stretched fibroblasts accelerates the wound-healing process in streptozotocin-induced diabetic mice. Cell Transplant. 2014, 23, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Ngo, H.T.; Seo, S.A.; Park, B.; Zhang, M.; Yi, T.-H. Protective effect of dietary Alchemilla mollis on UVB-irradiated premature skin aging through regulation of transcription factor NFATc1 and Nrf2/ARE pathways. Phytomedicine 2018, 39, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Park, S.Y.; Lee, H.J.; Lee, T.Y.; Sun, Z.W.; Yi, T.H. Gallic acid regulates skin photoaging in UVB-exposed fibroblast and hairless mice. Phytother. Res. 2014, 28, 1778–1788. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, Z.; Wang, L.; Walid, E.; Zhang, H. In vitro antioxidant and anti-proliferation activities of polysaccharides from various extracts of different mushrooms. Int. J. Mol. Sci. 2012, 13, 5801–5817. [Google Scholar] [CrossRef] [PubMed]


| Gene Name | Forward Primer (5′→3′) | Reverse Primer (5′→3′) |
|---|---|---|
| MMP-1 | 5′ATTCTACTGATATCGGGGCTTTGA3′ | 5′ATGTCCTTGGGGTATCCGTGTAG3′ |
| Type I Procollagen | 5′CTCGAGGTGGACACCACCCT3′ | 5′CAGCTGGATGGCCACATCGG3′ |
| GAPDH | 5′ACCACAGTCCATGCCATCAC3′ | 5′CCACCACCCTGTTGCTGTAG3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Hwang, E.; Ngo, H.T.T.; Perumalsamy, H.; Kim, Y.J.; Li, L.; Yi, T.-H. Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts. Int. J. Mol. Sci. 2018, 19, 3327. https://doi.org/10.3390/ijms19113327
Liu Y, Hwang E, Ngo HTT, Perumalsamy H, Kim YJ, Li L, Yi T-H. Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts. International Journal of Molecular Sciences. 2018; 19(11):3327. https://doi.org/10.3390/ijms19113327
Chicago/Turabian StyleLiu, Ying, Eunson Hwang, Hien T. T. Ngo, Haribalan Perumalsamy, Yeon Ju Kim, Lu Li, and Tae-Hoo Yi. 2018. "Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts" International Journal of Molecular Sciences 19, no. 11: 3327. https://doi.org/10.3390/ijms19113327
APA StyleLiu, Y., Hwang, E., Ngo, H. T. T., Perumalsamy, H., Kim, Y. J., Li, L., & Yi, T.-H. (2018). Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts. International Journal of Molecular Sciences, 19(11), 3327. https://doi.org/10.3390/ijms19113327