Lipid Identification and Transcriptional Analysis of Controlling Enzymes in Bovine Ovarian Follicle
Abstract
1. Introduction
2. Results
2.1. Analysis of Lipids in Bovine Ovarian Follicular Cells and Fluid
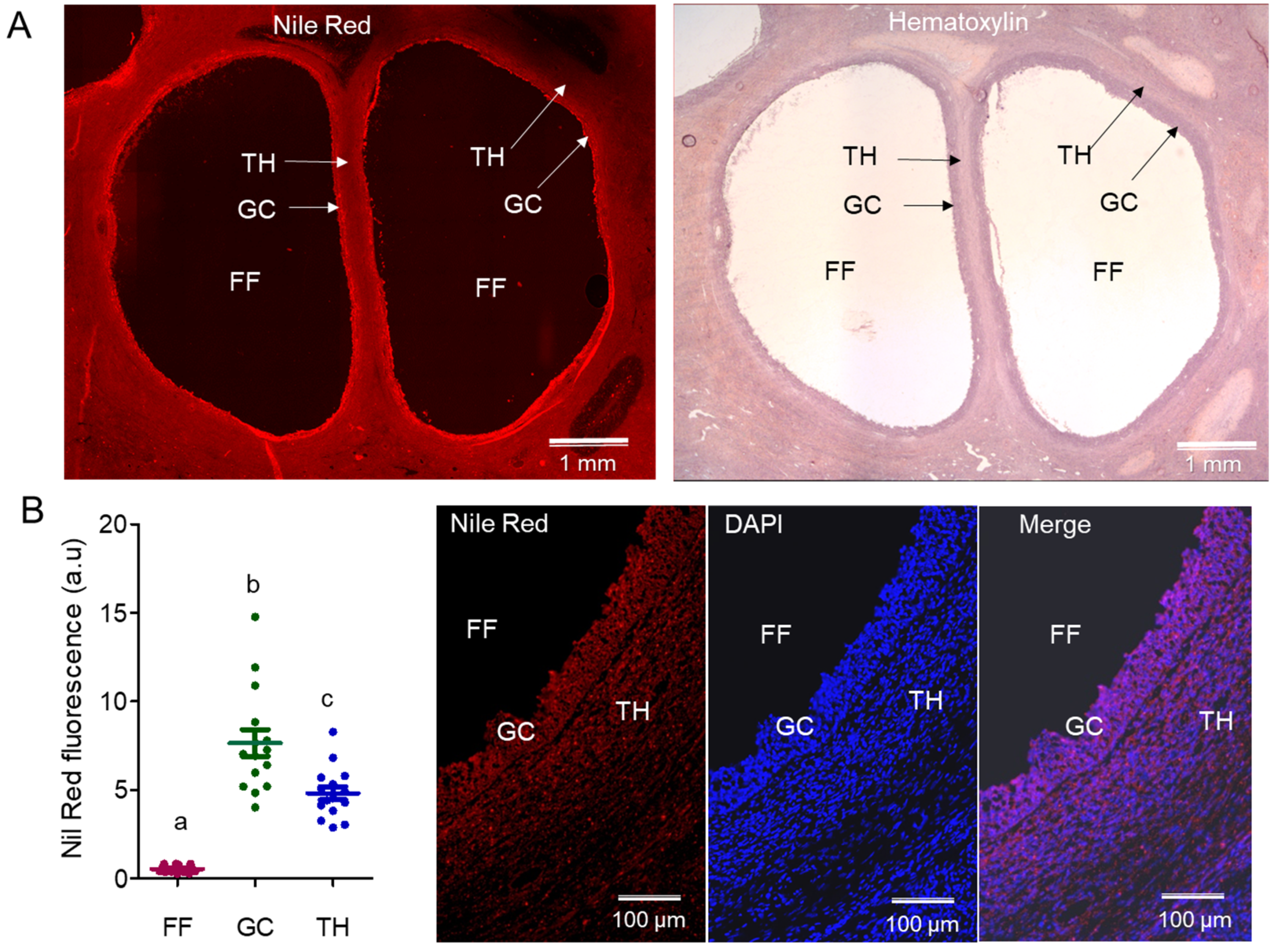
2.1.1. Analysis of Lipids by Nile Red on Ovarian Sections
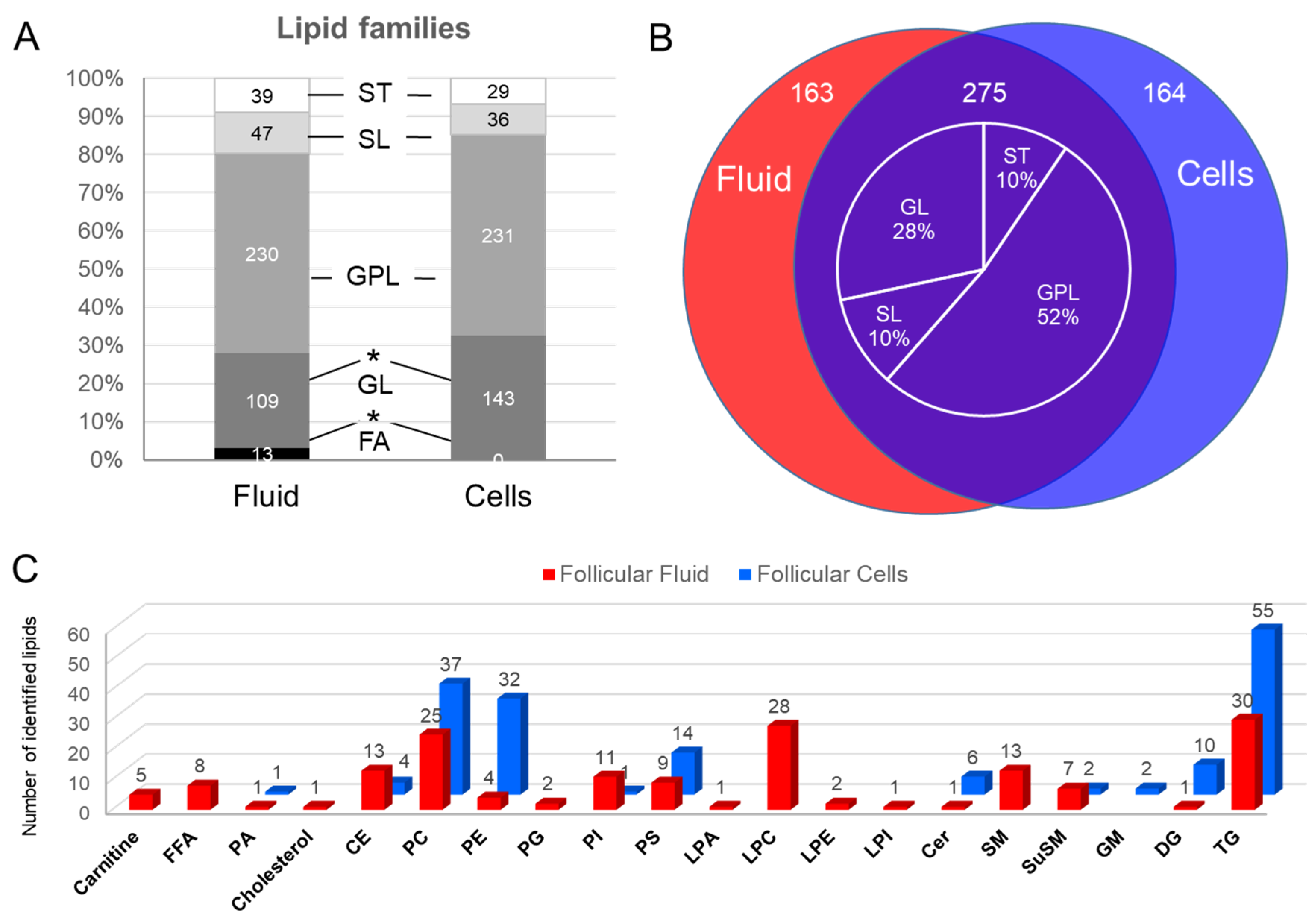
2.1.2. Lipid Composition of Follicular Cells and Fluid
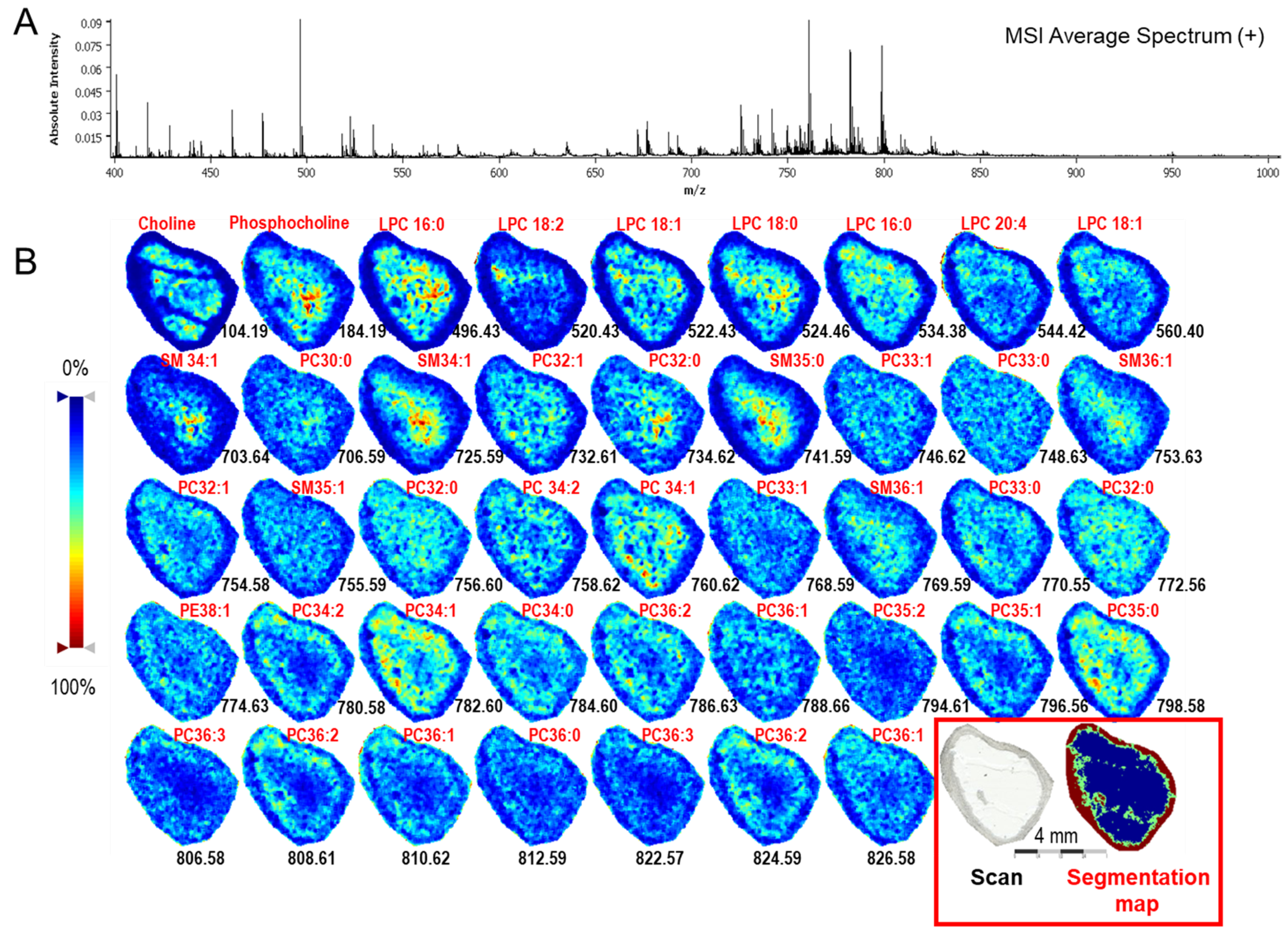
2.1.3. Mass Spectrometry Imaging of Lipids in Ovarian Follicle
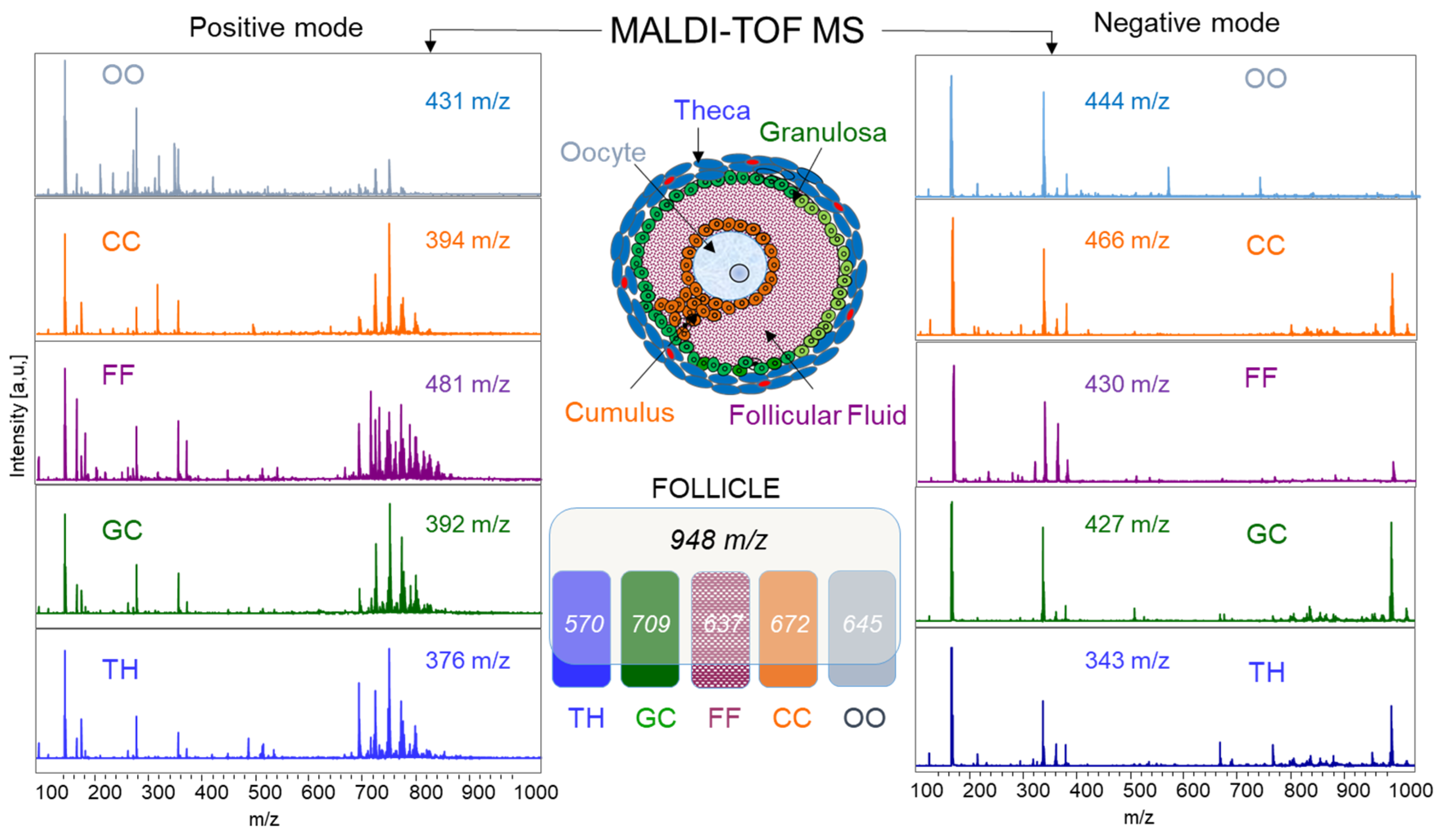
2.1.4. MALDI-TOF Profiling and Lipid Identifications in Follicular Compartments
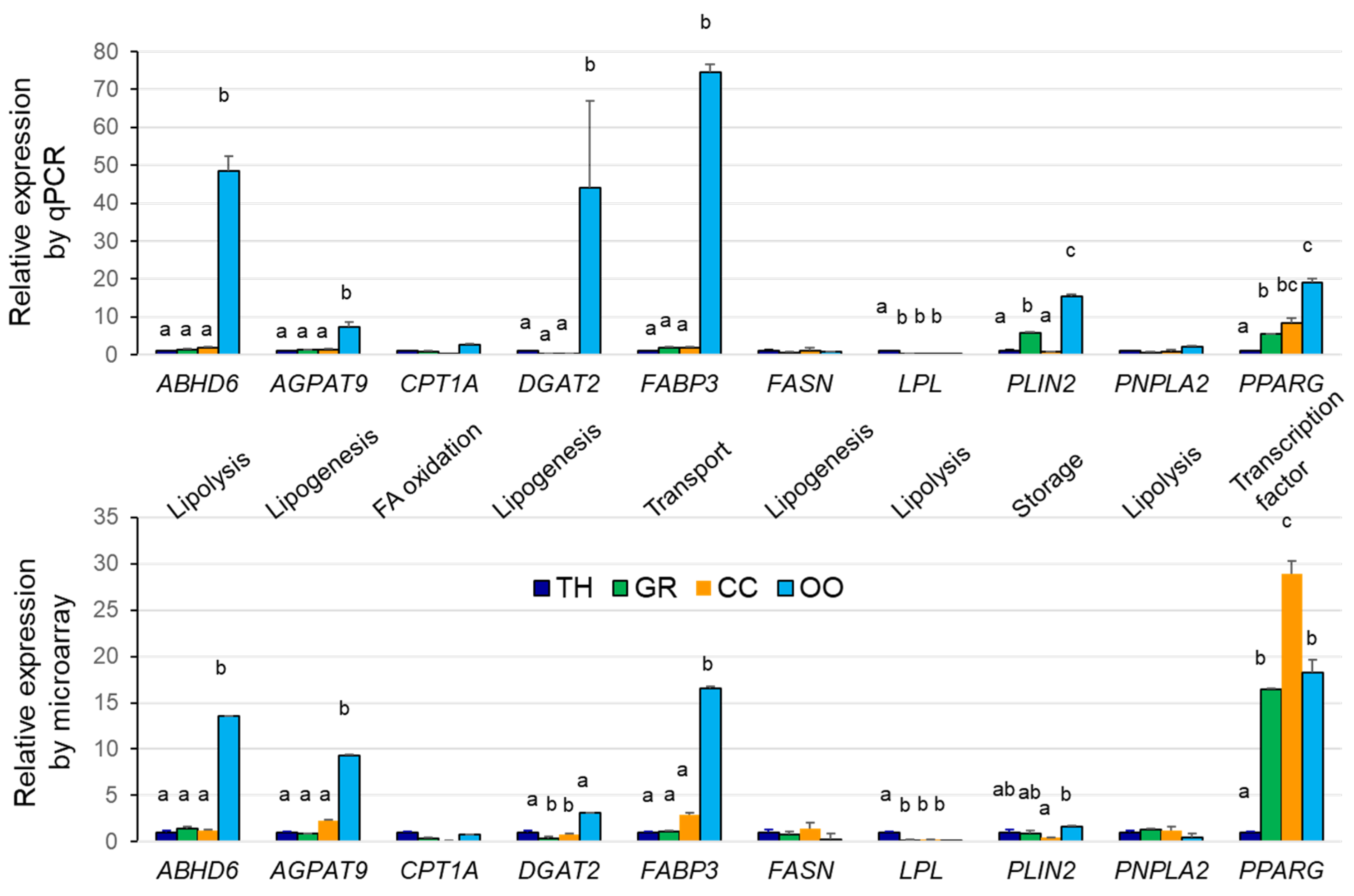
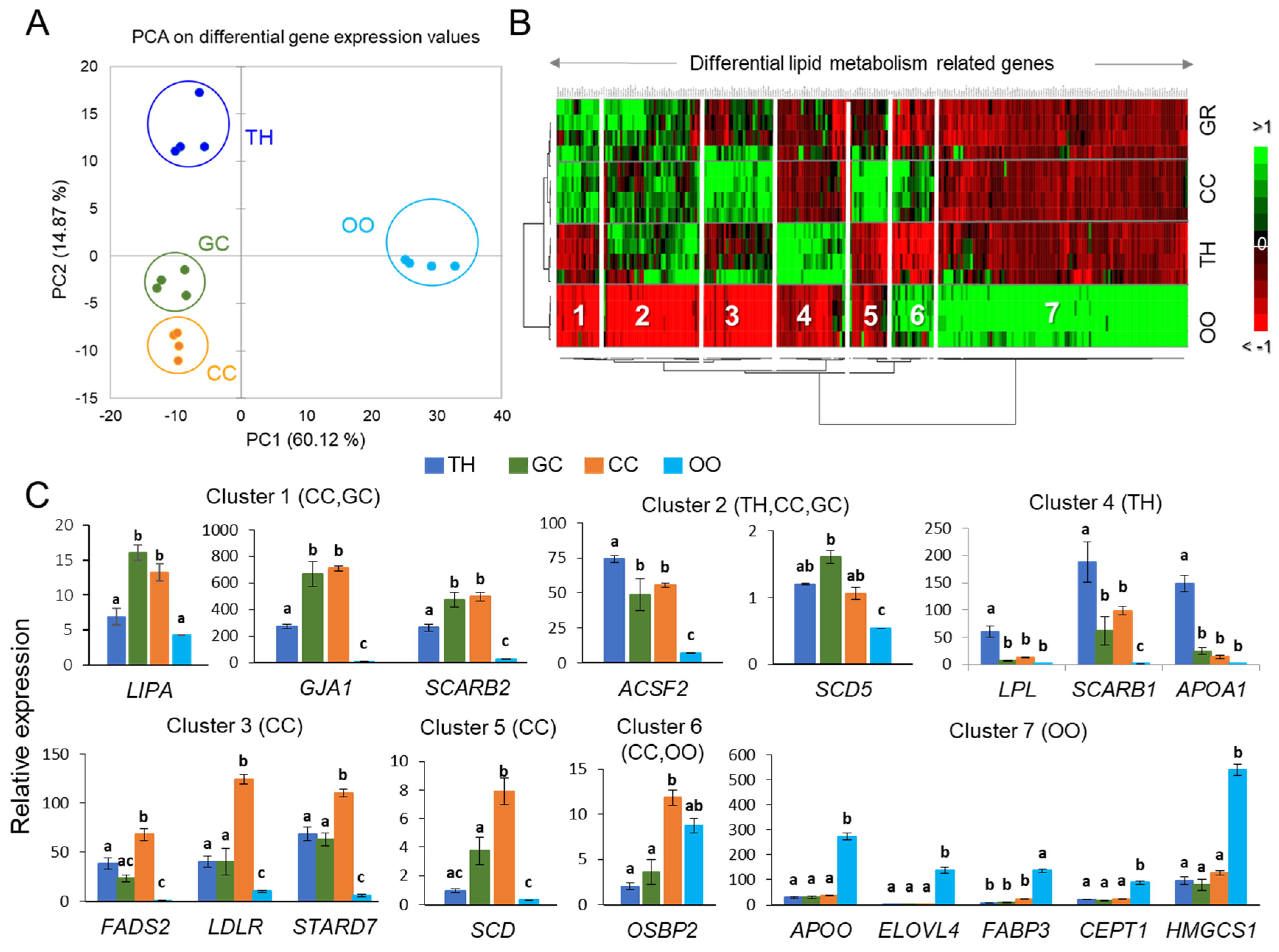
2.2. Analysis of Gene Expression in Bovine Ovarian Follicular Cells
3. Discussion
4. Materials and Methods
4.1. Ethics
4.2. Chemicals
4.3. Biological Materials
4.4. Lipid Analysis
4.4.1. Nile Red Staining
4.4.2. Lipid Identification Using Liquid Chromatography Coupled to High-Resolution Mass Spectrometry (LC-HRMS)
4.4.3. Lipid Identification Using Tandem High-Resolution Mass Spectrometry (HRMS/MS)
4.4.4. MSI (Mass Spectrometry Imaging) by MALDI-TOF MS
4.4.5. MALDI-TOF MS Analysis of Lipids on Follicular Cells and Fluid
4.5. Gene Expression Analysis
4.5.1. RNA Extraction
4.5.2. Microarray Hybridization and Transcriptome Data Analysist
4.5.3. Real Time PCR Expression Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CC | Cumulus cells |
| Cer | Ceramids |
| CE | Cholesteryl esters |
| DG | Diacylglycerols |
| FA | Fatty acyls |
| FFA | Free fatty acids |
| FAC | Fatty acyl carnitines |
| FF | Folicular fluid |
| FSH | Follicle Stimulation Hormone |
| GC | Granulosa cells |
| GM | Ganglioside |
| GL | Glycerolipids |
| GO | Gene Onthology |
| GP | Glycerophosphates |
| GPL | Glycerophospholipids |
| HCer | Hexosylceramide |
| HDL | High Density Lipoprotein |
| Hcer | Hexosylceramides |
| LC/MS | Liquid Chromatography/Mass Spectrometry |
| LC-HRMS | Liquid Chromatography- High Resolution Mass Spectrometry |
| LD | Lipid droplet |
| LDL | Low Density Lipoprotein |
| LH | Luteinizing Hormone |
| LPA | Lyso-phatidic acid |
| LPC | Lyso-phosphatidylcholines |
| LPE | Lyso-phosphatidylethanolamines |
| LPI | Lyso-phosphatidylinositols |
| LPL | Lipoprotein Lipase |
| m/z | Ion Mass to Ion Charge number ratio |
| MALDI-TOF | Matrix Assisted Laser Desorption Ionization -Time of Flight |
| MS | Mass Spectrometry |
| MSI | Mass Spectrometry Imaging |
| NEB | Negative Energy Balance |
| NR | Nile Red |
| OO | Oocyte |
| OOC | Oocyte-cumulus complex |
| PA | Phosphatidic Acid |
| PC | Phosphatidylcholines |
| PE | Phosphatidylethanolamines |
| PG | Phosphatidylglycerols |
| PI | Phosphatidylinositols |
| PS | Phosphatidylserines |
| SL | Sphingolipids |
| SM | Sphingomyelins |
| ST | Sterols/Steroid Derivatives |
| SuSM | Sulfoglycosphingolipids |
| TG (TAG) | Triacylglycerols |
| TH | Theca cells |
Appendix A
| Observed m/z | Exact m/z | Delta/Da | Lipid Annotation | Ion |
|---|---|---|---|---|
| 104.195 | 104.173 | −0.022 | Choline | [M + H]+ |
| 184.189 | 184.152 | −0.037 | Phosphocholine | [M + H]+ |
| 496.434 | 496.34 | −0.0937 | LPC 16a:0 | [M + H]+ |
| 520.427 | 520.34 | −0.0867 | LPC 18a:2 | [M + H]+ |
| 522.429 | 522.356 | −0.073 | LPC 18a:1 | [M + H]+ |
| 524.458 | 524.372 | −0.0864 | LPC 18a:0 | [M + H]+ |
| 534.379 | 534.296 | −0.0834 | LPC 16a:0 | [M + K]+ |
| 544.417 | 544.3403 | 0.0767 | LPC 20a:4 | [M + H]+ |
| 560.398 | 560.3116 | −0.0867 | LPC 18a:1 | [M + K]+ |
| 703.64 | 703.575 | −0.0652 | SM(d18:1/C16:0) | [M + H]+ |
| 706.592 | 706.539 | −0.0533 | PC 30a:0 | [M + H]+ |
| 725.587 | 725.557 | −0.0302 | SM(d18:1/C16:0) | [M + Na]+ |
| 732.609 | 732.5543 | −0.0547 | PC 32a:1 | [M + H]+ |
| 734.626 | 734.57 | −0.056 | PC 32a:0 | [M + H]+ |
| 741.581 | 741.5881 | 0.0071 | SM(d18:0/C17:0) | [M + Na]+ |
| 746.617 | 746.57 | −0.047 | PC 33a:1 | [M + H]+ |
| 748.625 | 748.5856 | −0.0394 | PC 33a:0 | [M + H]+ |
| 753.628 | 753.5881 | −0.0399 | SM(d18:1/C18:0) | [M + Na]+ |
| 754.581 | 754.5357 | −0.0453 | PC 32a:1 | [M + Na]+ |
| 755.59 | 755.5469 | −0.0431 | SM(d18:1/C17:0) | [M + K]+ |
| 756.599 | 756.5514 | −0.0476 | PC 32a:0 | [M + Na]+ |
| 758.621 | 758.56995 | −0.0511 | PC 34a:2 | [M + H]+ |
| 760.617 | 760.5856 | −0.0314 | PC 34a:1 | [M + H]+ |
| 768.598 | 768.5514 | −0.0466 | PC 33a:1 | [M + Na]+ |
| 769.589 | 769.5626 | −0.0264 | SM(d18:1/C18:0) | [M + K]+ |
| 770.551 | 770.567 | 0.016 | PC 33a:0 | [M + Na]+ |
| 772.562 | 772.5253 | 0.0367 | PC 32a:0 | [M + K]+ |
| 774.634 | 774.6013 | −0.0327 | PE 38:1 | [M + H]+ |
| 780.578 | 780.5514 | −0.0266 | PC 34a:2 | [M + Na]+ |
| 782.603 | 782.567 | −0.036 | PC 34a:1 | [M + Na]+ |
| 784.602 | 784.5827 | −0.0193 | PC 34a:0 | [M + Na]+ |
| 786.632 | 786.6013 | −0.0307 | PC 36a:2 | [M + H]+ |
| 788.664 | 788.6169 | −0.0471 | PC 36a:1 | [M + H]+ |
| 794.605 | 794.567 | −0.038 | PC 35a:2 | [M + Na]+ |
| 796.562 | 796.5827 | 0.0207 | PC 35a:1 | [M + Na]+ |
| 798.578 | 798.5983 | 0.0203 | PC 35a:0 | [M + Na]+ |
| 806.583 | 806.567 | −0.016 | PC 36a:3 | [M + Na]+ |
| 808.613 | 808.5827 | −0.0303 | PC 36a:2 | [M + Na]+ |
| 810.615 | 810.5983 | −0.0167 | PC 36a:1 | [M + Na]+ |
| 812.592 | 812.614 | 0.022 | PC 36:0 | [M + Na]+ |
| 822.567 | 822.5843 | 0.0173 | PC 36a:3 | [M + K]+ |
| 824.587 | 824.5566 | −0.0304 | PC 36a:2 | [M + K]+ |
| 826.581 | 826.572 | 0.0087 | PC 36a:1 | [M + K]+ |
| Compartment | Positive Mode | Negative Mode |
|---|---|---|
| TH | 15.9% | 16.1% |
| GC | 18.4% | 15.8% |
| FF | 18.0% | 22.9% |
| CC | 27.6% | 21.7% |
| OO | 32.9% | 25.3% |
| Gene | Accession Number | Description | Primer’s Sequence (5′-3′) | Efficiency, % (E) | |
|---|---|---|---|---|---|
| ABHD6 | NM_001075196 | Abhydrolase domain containing 6 (ABHD6) | Fw | ACCCCGAAGGAGATGAGTGA | 92.6% (1.93) |
| Rev | CTGGGAGTTGGCGATTGACT | ||||
| AGPAT9 | NM_001192514 | Acylglycerol-3-phosphate O-acyltransferase 9 | Fw | AATGCCTCTCCCATCCGTTG | 88.9% (1.89) |
| Rev | TTATGCTGCACAGTCGGGAA | ||||
| CPT1A | FJ415874 | Carnitine palmitoyltransferase 1A | Fw | TCCTGGTGGGCTACCAATTA | 95.4% (1.95) |
| Rev | TGCGTCTGTAAAGCAGGATG | ||||
| GAPDH | NM_001034034 | Glyceraldehyde 3 phosphate dehydrogenase | Fw | TTCAACGGCACAGTCAAGG | 100.3% (2.00) |
| Rev | ACATACTCAGCACCAGCATCAC | ||||
| DGAT2 | NM_205793.2 | Diacylglycerol O-acyltransferase 2 | Fw | GTGGCCTCGCTTTGCTTAAC | 92.3% (1.92) |
| Rev | GGGTTCGGGGAACTTCTGTT | ||||
| FABP3 | NM_174313 | Fatty acid binding protein 3 | Fw | ATCGTGACGCTGGATGGCGG | 104.1% (2.04) |
| Rev | GCCGAGTCCAGGAGTAGCCCA | ||||
| FASN | AY343889 | Fatty acid synthase | Fw | CACTCCATCCTCGCTCTCC | 102.9% (2.02) |
| Rev | GCCTGTCATCATCTGTCACC | ||||
| LPL | NM_001075120 | Lipoprotein lipase | Fw | GGGTTTTGAGCAAGGGTACA | 87.1% (1.87) |
| Rev | GCCACAATGACCTTTCCAGT | ||||
| PLIN2 | NM_173980 | Perilipin 2 | Fw | ACAACACACCCCTCAACTGG | 97.5% (1.96) |
| Rev | CTGCCTGCCTACTTCAGACC | ||||
| PNPLA2 | NM_001046005 | Patatin-like phospholipase domain containing 2 | Fw | ATGGTGCCCTACACTCTGCC | 87.6% (1.88) |
| Rev | AGCTTCCTCTTGGCGCGTAT | ||||
| PPARG | Y12419 | Peroxisome proliferator-activated receptor gamma | Fw | CCCTGGCAAAGCATTTGTAT | 105.1% (2.05) |
| Rev | ACTGACACCCCTGGAAGATG | ||||
| RPL19 | BC102223 | Ribosomal protein L19 | Fw | AATCGCCAATGCCAACTC | 98.2% (1.98) |
| Rev | CCCTTTCGCTTACCTATACC | ||||
| RPS9 | BC148016 | Ribosomic protein S9 | Fw | GGAGACCCTTCGAGAAGTCC | 85.0% (1.85) |
| Rev | GGGCATTACCTTCGAACAGA | ||||
References
- Butler, W.R.; Smith, R.D. Interrelationships Between Energy Balance and Postpartum Reproductive Function in Dairy Cattle. J. Dairy Sci. 1989, 72, 767–783. [Google Scholar] [CrossRef]
- Staples, C.R.; Thatcher, W.W.; Clark, J.H. Relationship between ovarian activity and energy status during the early postpartum period of high producing dairy cows. J. Dairy Sci. 1990, 73, 938–947. [Google Scholar] [CrossRef]
- Leroy, J.L.; Opsomer, G.; Van Soom, A.; Goovaerts, I.G.; Bols, P.E. Reduced fertility in high-yielding dairy cows: Are the oocyte and embryo in danger? Part I. The importance of negative energy balance and altered corpus luteum function to the reduction of oocyte and embryo quality in high-yielding dairy cows. Reprod. Domest. Anim. 2008, 43, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Van Hoeck, V.; Bols, P.E.; Binelli, M.; Leroy, J.L. Reduced oocyte and embryo quality in response to elevated non-esterified fatty acid concentrations: A possible pathway to subfertility? Anim. Reprod. Sci. 2014, 149, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Bender, K.; Walsh, S.; Evans, A.C.O.; Fair, T.; Brennan, L. Metabolite concentrations in follicular fluid may explain differences in fertility between heifers and lactating cows. Reproduction 2010, 139, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Zeron, Y.; Ocheretny, A.; Kedar, O.; Borochov, A.; Sklan, D.; Arav, A. Seasonal changes in bovine fertility: Relation to developmental competence of oocytes, membrane properties and fatty acid composition of follicles. Reproduction 2001, 121, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Fortune, J.E.; Rivera, G.M.; Yang, M.Y. Follicular development: The role of the follicular microenvironment in selection of the dominant follicle. Anim. Reprod. Sci. 2004, 82–83, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Mermillod, P.; Dalbies-Tran, R.; Uzbekova, S.; Thelie, A.; Traverso, J.M.; Perreau, C.; Papillier, P.; Monget, P. Factors affecting oocyte quality: Who is driving the follicle? Reprod. Domest. Anim. 2008, 43 (Suppl. 2), 393–400. [Google Scholar] [CrossRef]
- Buratini, J.; Price, C.A. Follicular somatic cell factors and follicle development. Reprod. Fertil. Dev. 2011, 23, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, A.J.W.; Kawamura, K.; Cheng, Y.; Fauser, B.C.J.M. Intraovarian control of early folliculogenesis. Endocr. Rev. 2015, 36, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Fortune, J.E. Ovarian follicular growth and development in mammals. Biol. Reprod. 1994, 50, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Dumesic, D.A.; Meldrum, D.R.; Katz-Jaffe, M.G.; Krisher, R.L.; Schoolcraft, W.B. Oocyte environment: Follicular fluid and cumulus cells are critical for oocyte health. Fertil. Steril. 2015, 103, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, R.B.; Lane, M.; Thompson, J.G. Oocyte-secreted factors: Regulators of cumulus cell function and oocyte quality. Hum. Reprod. Update 2008, 14, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Leroy, J.L.M.R.; Vanholder, T.; Mateusen, B.; Christophe, A.; Opsomer, G.; de Kruif, A.; Genicot, G.; Van Soom, A. Non-esterified fatty acids in follicular fluid of dairy cows and their effect on developmental capacity of bovine oocytes in vitro. Reproduction 2005, 130, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Revelli, A.; Piane, L.D.; Casano, S.; Molinari, E.; Massobrio, M.; Rinaudo, P. Follicular fluid content and oocyte quality: From single biochemical markers to metabolomics. Reprod. Biol. Endocrinol. 2009, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Assidi, M.; Dieleman, S.J.; Sirard, M.A. Cumulus cells gene expression following the LH surge in bovine preovulatory follicles: Potential early markers of oocyte competence. Reproduction 2010, 140, 835–852. [Google Scholar] [CrossRef] [PubMed]
- Warzych, E.; Cieslak, A.; Madeja, Z.E.; Pawlak, P.; Wolc, A.; Lechniak, D. Multifactorial Analysis of the Follicular Environment is Predictive of Oocyte Morphology in Cattle. J. Reprod. Dev. 2014, 60, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Matoba, S.; Bender, K.; Fahey, A.G.; Mamo, S.; Brennan, L.; Lonergan, P.; Fair, T. Predictive value of bovine follicular components as markers of oocyte developmental potential. Reprod. Fertil. Dev. 2014, 26, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Bunel, A.; Nivet, A.L.; Blondin, P.; Vigneault, C.; Richard, F.J.; Sirard, M.A. Cumulus cell gene expression associated with pre-ovulatory acquisition of developmental competence in bovine oocytes. Reprod. Fertil. Dev. 2014, 26, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Collado-Fernandez, E.; Picton, H.M.; Dumollard, R. Metabolism throughout follicle and oocyte development in mammals. Int. J. Dev. Biol. 2012, 56, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, E.M.; Leese, H.J. A potential role for triglyceride as an energy source during bovine oocyte maturation and early embryo development. Mol. Reprod. Dev. 2006, 73, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Sutton-McDowall, M.L.; Gilchrist, R.B.; Thompson, J.G. The pivotal role of glucose metabolism in determining oocyte developmental competence. Reproduction 2010, 139, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, H.F.; Goodman, J.M. The life cycle of lipid droplets. Curr. Opin. Cell Biol. 2015, 33, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Grummer, R.R.; Carroll, D.J. A review of lipoprotein cholesterol metabolism: Importance to ovarian function. J. Anim. Sci. 1988, 66, 3160–3173. [Google Scholar] [CrossRef] [PubMed]
- Acosta, T.J.; Miyamoto, A.; Ozawa, T.; Wijayagunawardane, M.P.; Sato, K. Local release of steroid hormones, prostaglandin E2, and endothelin-1 from bovine mature follicles In vitro: Effects of luteinizing hormone, endothelin-1, and cytokines. Biol. Reprod. 1998, 59, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Nuttinck, F.; Gall, L.; Ruffini, S.; Laffont, L.; Clement, L.; Reinaud, P.; Adenot, P.; Grimard, B.; Charpigny, G.; Marquant-Le Guienne, B. PTGS2-Related PGE2 Affects Oocyte MAPK Phosphorylation and Meiosis Progression in Cattle: Late Effects on Early Embryonic Development. Biol. Reprod. 2011. [Google Scholar] [CrossRef] [PubMed]
- Van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Fernandis, A.Z.; Wenk, M.R. Membrane lipids as signaling molecules. Curr. Opin. Lipidol. 2007, 18, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Sturmey, R.G.; Reis, A.; Leese, H.J.; McEvoy, T.G. Role of fatty acids in energy provision during oocyte maturation and early embryo development. Reprod. Domest. Anim. 2009, 44 (Suppl. 3), 50–58. [Google Scholar] [CrossRef]
- Uzbekova, S.; Elis, S.; Teixeira-Gomes, A.P.; Desmarchais, A.; Maillard, V.; Labas, V. MALDI Mass Spectrometry Imaging of Lipids and Gene Expression Reveals Differences in Fatty Acid Metabolism between Follicular Compartments in Porcine Ovaries. Biology (Basel) 2015, 4, 216–236. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.I.; Ferreira, C.R.; Eberlin, L.S.; Cooks, R.G. Improved spatial resolution in the imaging of biological tissue using desorption electrospray ionization. Anal. Bioanal. Chem. 2012, 404, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.R.; Saraiva, S.A.; Catharino, R.R.; Garcia, J.S.; Gozzo, F.C.; Sanvido, G.B.; Santos, L.F.A.; Lo Turco, E.G.; Pontes, J.H.F.; Basso, A.C.; et al. Single embryo and oocyte lipid fingerprinting by mass spectrometry. J. Lipid Res. 2010, 51, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Silva-Santos, K.; Ferreira, C.; Santos, G.; Eberlin, M.; Siloto, L.; Rosa, C.; Marcantonio, T.; Seneda, M. MALDI-MS Lipid Profiles of Oocytes Recovered by Ovum Pickup from Bos indicus and 1/2 indicus x taurus with High vs Low Oocyte Yields. Reprod. Domest. Anim. 2014, 49, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Bertevello, P.; Ghazouani, O.; Banliat, C.; Elis, S.; Teixeira-Gomes, A.P.; Maillard, V.; Labas, V.; Uzbekova, S. MALDI-TOF mass spectrometry analysis of lipids in single bovine oocytes during IVM. In Proceedings of the 32nd Scientific Meeting of the AETE, Barcelona, Spain, 9–10 September 2016; p. 712. [Google Scholar]
- Sanchez-Lazo, L.; Brisard, D.; Elis, S.; Maillard, V.; Uzbekov, R.; Labas, V.; Desmarchais, A.; Papillier, P.; Monget, P.; Uzbekova, S. Fatty Acid synthesis and oxidation in cumulus cells support oocyte maturation in bovine. Mol. Endocrinol. 2014, 28, 1502–1521. [Google Scholar] [CrossRef] [PubMed]
- Elis, S.; Oseikria, M.; Vitorino Carvalho, A.; Bertevello, P.S.; Corbin, E.; Teixeira-Gomes, A.P.; Lecardonnel, J.; Archilla, C.; Duranthon, V.; Labas, V.; et al. Docosahexaenoic acid mechanisms of action on the bovine oocyte-cumulus complex. J. Ovarian Res. 2017, 10, 74. [Google Scholar] [CrossRef] [PubMed]
- Santos, P.H.; Fontes, P.K.; Franchi, F.F.; Nogueira, M.F.; Belaz, K.R.; Tata, A.; Eberlin, M.N.; Sudano, M.J.; Barros, C.M.; Castilho, A.C. Lipid profiles of follicular fluid from cows submitted to ovarian superstimulation. Theriogenology 2017, 94, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Lafontan, M.; Langin, D. Lipolysis and lipid mobilization in human adipose tissue. Prog. Lipid Res. 2009, 48, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.; Leray, V.; Diez, M.; Serisier, S.; Le Bloc’h, J.; Siliart, B.; Dumon, H. Liver lipid metabolism. J. Anim. Physiol. Anim. Nutr. (Berl.) 2008, 92, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.R.; Schulze, A. Lipid metabolism in cancer. FEBS J. 2012, 279, 2610–2623. [Google Scholar] [CrossRef] [PubMed]
- Chmurzynska, A. The multigene family of fatty acid-binding proteins (FABPs): Function, structure and polymorphism. J. Appl. Genet. 2006, 47, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Dunning, K.R.; Russell, D.L.; Robker, R.L. Lipids and oocyte developmental competence: The role of fatty acids and B-oxidation. Reproduction 2014, 148, R15–R27. [Google Scholar] [CrossRef] [PubMed]
- Auclair, S.; Uzbekov, R.; Elis, S.; Sanchez, L.; Kireev, I.; Lardic, L.; Dalbies-Tran, R.; Uzbekova, S. Absence of cumulus cells during in vitro maturation affects lipid metabolism in bovine oocytes. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E599–E613. [Google Scholar] [CrossRef] [PubMed]
- Paczkowski, M.; Silva, E.; Schoolcraft, W.B.; Krisher, R.L. Comparative Importance of Fatty Acid Beta-Oxidation to Nuclear Maturation, Gene Expression, and Glucose Metabolism in Mouse, Bovine, and Porcine Cumulus Oocyte Complexes. Biol. Reprod. 2013, 88, 111. [Google Scholar] [CrossRef] [PubMed]
- Van Hoeck, V.; Leroy, J.L.; Arias-Alvarez, M.; Rizos, D.; Gutierrez-Adan, A.; Schnorbusch, K.; Bols, P.E.; Leese, H.J.; Sturmey, R.G. Oocyte developmental failure in response to elevated non-esterified fatty acid concentrations: Mechanistic insights. Reproduction 2013, 145, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Elis, S.; Desmarchais, A.; Maillard, V.; Uzbekova, S.; Monget, P.; Dupont, J. Cell proliferation and progesterone synthesis depend on lipid metabolism in bovine granulosa cells. Theriogenology 2015, 83, 840–853. [Google Scholar] [CrossRef] [PubMed]
- Aardema, H.; van Tol, H.T.A.; Wubbolts, R.W.; Brouwers, J.; Gadella, B.M.; Roelen, B.A.J. Stearoyl-CoA desaturase activity in bovine cumulus cells protects the oocyte against saturated fatty acid stress. Biol. Reprod. 2017, 96, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Lonergan, P.; Rizos, D.; Gutierrez-Adan, A.; Fair, T.; Boland, M.P. Oocyte and embryo quality: Effect of origin, culture conditions and gene expression patterns. Reprod. Domest. Anim. 2003, 38, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Tata, A.; Sudano, M.J.; Santos, V.G.; Landim-Alvarenga, F.D.; Ferreira, C.R.; Eberlin, M.N. Optimal single-embryo mass spectrometry fingerprinting. J. Mass Spectrom. 2013, 48, 844–849. [Google Scholar] [CrossRef] [PubMed]
- Apparicio, M.; Ferreira, C.R.; Tata, A.; Santos, V.G.; Alves, A.E.; Mostachio, G.Q.; Pires-Butler, E.A.; Motheo, T.F.; Padilha, L.C.; Pilau, E.J.; et al. Chemical Composition of Lipids Present in Cat and Dog Oocyte by Matrix-Assisted Desorption Ionization Mass Spectrometry (MALDI- MS). Reprod. Domest. Anim. 2012, 47, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Fair, T.; Hyttel, P.; Greve, T. Bovine oocyte diameter in relation to maturational competence and transcriptional activity. Mol. Reprod. Dev. 1995, 42, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, R.B.; Ritter, L.J.; Armstrong, D.T. Oocyte-somatic cell interactions during follicle development in mammals. Anim. Reprod. Sci. 2004, 82–83, 431–446. [Google Scholar] [CrossRef] [PubMed]
- Binelli, M.; Murphy, B.D. Coordinated regulation of follicle development by germ and somatic cells. Reprod. Fertil. Dev. 2010, 22, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lolicato, F.; Brouwers, J.F.; de Lest, C.H.; Wubbolts, R.; Aardema, H.; Priore, P.; Roelen, B.A.; Helms, J.B.; Gadella, B.M. The cumulus cell layer protects the bovine maturing oocyte against fatty acid-induced lipotoxicity. Biol. Reprod. 2015, 92, 16. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, T.M.; Gonçalves, R.F.; Melo, C.F.O.R.; de Oliveira, D.N.; Lima, E.d.O.; Visintin, J.A.; de Achilles, M.A.; Catharino, R.R. A Metabolomic Overview of Follicular Fluid in Cows. Front. Vet. Sci. 2018, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Drzazga, A.; Sowinska, A.; Koziolkiewicz, M. Lysophosphatidylcholine and lysophosphatidylinosiol--novel promissing signaling molecules and their possible therapeutic activity. Acta Pol. Pharm. 2014, 71, 887–899. [Google Scholar] [PubMed]
- Sohel, M.M.; Hoelker, M.; Noferesti, S.S.; Salilew-Wondim, D.; Tholen, E.; Looft, C.; Rings, F.; Uddin, M.J.; Spencer, T.E.; Schellander, K.; et al. Exosomal and Non-Exosomal Transport of Extra-Cellular microRNAs in Follicular Fluid: Implications for Bovine Oocyte Developmental Competence. PLoS ONE 2013, 8, e78505. [Google Scholar] [CrossRef] [PubMed]
- Skotland, T.; Sandvig, K.; Llorente, A. Lipids in exosomes: Current knowledge and the way forward. Prog. Lipid Res. 2017, 66, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Zemski Berry, K.A.; Murphy, R.C.; Kosmider, B.; Mason, R.J. Lipidomic characterization and localization of phospholipids in the human lung. J. Lipid Res. 2017, 58, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Young, J.M.; McNeilly, A.S. Theca: The forgotten cell of the ovarian follicle. Reproduction 2010, 140, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Tanghe, S.; Van Soom, A.; Nauwynck, H.; Coryn, M.; De Kruif, A. Minireview: Functions of the Cumulus Oophorus during oocyte maturation, ovulation and fertilization. Mol. Reprod. Dev. 2002, 61, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Bertevello, P.; Banliat, C.; Teixeira Gomes, A.P.; Elis, S.; Maillard, V.; Carvalho, A.; Duranthon, V.; Labas, V.; Uzbekova, S. Comparison of lipid profiles and gene expression in granulosa and cumulus cells in bovine. In Proceedings of the 33rd Annual Meeting of the European Embryo Transfer Association (AETE), Bath, UK, 8–9 September 2017; p. 970. [Google Scholar]
- Borgbo, T.; Povlsen, B.B.; Andersen, C.Y.; Borup, R.; Humaidan, P.; Grondahl, M.L. Comparison of gene expression profiles in granulosa and cumulus cells after ovulation induction with either human chorionic gonadotropin or a gonadotropin-releasing hormone agonist trigger. Fertil. Steril. 2013, 100, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Kuang, Y.; Zhou, L.; Yu, B.; Chen, Q.; Fu, Y.; Yan, Z.; Guo, H.; Lyu, Q.; Xie, J.; et al. Lipidomic Components Alterations of Human Follicular Fluid Reveal the Relevance of Improving Clinical Outcomes in Women Using Progestin-Primed Ovarian Stimulation Compared to Short-Term Protocol. Med. Sci. Monit. 2018, 24, 3357–3365. [Google Scholar] [CrossRef] [PubMed]
- Montani, D.A.; Cordeiro, F.B.; Regiani, T.; Victorino, A.B.; Pilau, E.J.; Gozzo, F.C.; Ferreira, C.R.; Fraietta, R.; Lo Turco, E.G. The follicular microenviroment as a predictor of pregnancy: MALDI-TOF MS lipid profile in cumulus cells. J. Assist. Reprod. Genet. 2012, 29, 1289–1297. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Garverick, H.A. Expression of steroidogenic enzyme and gonadotropin receptor genes in bovine follicles during ovarian follicular waves: A review. J. Anim. Sci. 1998, 76, 1903–1921. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.Q.; Sugiura, K.; Wigglesworth, K.; O’Brien, M.J.; Affourtit, J.P.; Pangas, S.A.; Matzuk, M.M.; Eppig, J.J. Oocyte regulation of metabolic cooperativity between mouse cumulus cells and oocytes: BMP15 and GDF9 control cholesterol biosynthesis in cumulus cells. Development 2008, 135, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Espenshade, P.J. Expanding roles for SREBP in metabolism. Cell Metab. 2012, 16, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Amin, M.; Keller, E.; Simerman, A.; Aguilera, P.; Briton-Jones, C.; Hill, D.L.; Abbott, D.H.; Chazenbalk, G.; Dumesic, D.A. A novel approach to quantifying ovarian cell lipid content and lipid accumulation in vitro by confocal microscopy in lean women undergoing ovarian stimulation for in vitro fertilization (IVF). J. Assist. Reprod. Genet. 2013, 30, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Aardema, H.; Vos, P.L.; Lolicato, F.; Roelen, B.A.; Knijn, H.M.; Vaandrager, A.B.; Helms, J.B.; Gadella, B.M. Oleic acid prevents detrimental effects of saturated fatty acids on bovine oocyte developmental competence. Biol. Reprod. 2011, 85, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Aardema, H.; Lolicato, F.; van de Lest, C.H.A.; Brouwers, J.F.; Vaandrager, A.B.; van Tol, H.T.A.; Roelen, B.A.J.; Vos, P.L.A.M.; Helms, J.B.; Gadella, B.M. Bovine Cumulus Cells Protect Maturing Oocytes from Increased Fatty Acid Levels by Massive Intracellular Lipid Storage. Biol. Reprod. 2013, 88, 164. [Google Scholar] [CrossRef] [PubMed]
- Leroy, J.L.; Vanholder, T.; Delanghe, J.R.; Opsomer, G.; Van Soom, A.; Bols, P.E.; de Kruif, A. Metabolite and ionic composition of follicular fluid from different-sized follicles and their relationship to serum concentrations in dairy cows. Anim. Reprod. Sci. 2004, 80, 201–211. [Google Scholar] [CrossRef]
- Gautier, T.; Becker, S.; Drouineaud, V.; Ménétrier, F.; Sagot, P.; Nofer, J.-R.; von Otte, S.; Lagrost, L.; Masson, D.; Tietge, U.J.F. Human luteinized granulosa cells secrete apoB100-containing lipoproteins. J. Lipid Res. 2010, 51, 2245–2252. [Google Scholar] [CrossRef] [PubMed]
- Serna, J.; García-Seisdedos, D.; Alcázar, A.; Lasunción, M.Á.; Busto, R.; Pastor, Ó. Quantitative lipidomic analysis of plasma and plasma lipoproteins using MALDI-TOF mass spectrometry. Chem. Phys. Lipids. 2015. [Google Scholar] [CrossRef] [PubMed]
- Osz, K.; Ross, M.; Petrik, J. The thrombospondin-1 receptor CD36 is an important mediator of ovarian angiogenesis and folliculogenesis. Reprod. Biol. Endocrinol. 2014, 12, 21. [Google Scholar] [CrossRef] [PubMed]
- Trigatti, B.; Rigotti, A.; Krieger, M. The role of the high-density lipoprotein receptor SR-BI in cholesterol metabolism. Curr. Opin. Lipidol. 2000, 11, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, C. Exosome-mediated communication in the ovarian follicle. J. Assist. Reprod. Genet. 2016, 33, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Record, M.; Silvente-Poirot, S.; Poirot, M.; Wakelam, M.J.O. Extracellular vesicles: Lipids as key components of their biogenesis and functions. J. Lipid Res. 2018, 59, 1316–1324. [Google Scholar] [CrossRef] [PubMed]
- Hermann, M.; Lindstedt, K.A.; Foisner, R.; Morwald, S.; Mahon, M.G.; Wandl, R.; Schneider, W.J.; Nimpf, J. Apolipoprotein A-I production by chicken granulosa cells. FASEB J. 1998, 12, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.H.; Lee, W.S.; Won, M.; Park, M.; Park, H.O.; Kim, E.; Lee, K.A.; Bae, J. The apolipoprotein A-I level is downregulated in the granulosa cells of patients with polycystic ovary syndrome and affects steroidogenesis. J. Proteome Res. 2010, 9, 4329–4336. [Google Scholar] [CrossRef] [PubMed]
- Bogan, R.L.; Hennebold, J.D. The reverse cholesterol transport system as a potential mediator of luteolysis in the primate corpus luteum. Reproduction 2010, 139, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Labas, V.; Teixeira-Gomes, A.P.; Bouguereau, L.; Gargaros, A.; Spina, L.; Marestaing, A.; Uzbekova, S. Intact cell MALDI-TOF mass spectrometry on single bovine oocyte and follicular cells combined with top-down proteomics: A novel approach to characterise markers of oocyte maturation. J. Proteomics 2018, 175, 56–74. [Google Scholar] [CrossRef] [PubMed]
- Labas, V.; Teixeira-Gomes, A.P.; Bouguereau, L.; Gargaros, A.; Spina, L.; Marestaing, A.; Uzbekova, S. Data on endogenous bovine ovarian follicular cells peptides and small proteins obtained through Top-down High Resolution Mass Spectrometry. Data Brief 2017, 13, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Peddinti, D.; Memili, E.; Burgess, S.C. Proteomics-based systems biology modeling of bovine germinal vesicle stage oocyte and cumulus cell interaction. PLoS ONE 2010, 5, e11240. [Google Scholar] [CrossRef] [PubMed]
- Wahli, W.; Michalik, L. PPARs at the crossroads of lipid signaling and inflammation. Trends Endocrinol. Metab. 2012, 23, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Kollmeyer, J.; Symolon, H.; Momin, A.; Munter, E.; Wang, E.; Kelly, S.; Allegood, J.C.; Liu, Y.; Peng, Q.; et al. Ceramides and other bioactive sphingolipid backbones in health and disease: Lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy. Biochim. Biophys. Acta 2006, 1758, 1864–1884. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Del Collado, M.; Da Silveira, J.C.; Sangalli, J.R.; Andrade, G.M.; Sousa, L.R.D.S.; Silva, L.A.; Meirelles, F.V.; Perecin, F. Fatty Acid Binding Protein 3 and Transzonal Projections Are Involved in Lipid Accumulation during in vitro Maturation of Bovine Oocytes. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bettegowda, A.; Smith, G.W. Mechanisms of maternal mRNA regulation: Implications for mammalian early embryonic development. Front. Biosci. 2007, 12, 3713–3726. [Google Scholar] [CrossRef] [PubMed]
- Hyttel, P.F.T.C.H.; Greve, T. Oocyte growth, capacitation and final maturartion in cattle. Theriogenology 1997, 47, 23–32. [Google Scholar] [CrossRef]
- Warzych, E.; Pawlak, P.; Pszczola, M.; Cieslak, A.; Lechniak, D. Prepubertal heifers versus cows—The differences in the follicular environment. Theriogenology 2017, 87, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Warzych, E.; Pawlak, P.; Pszczola, M.; Cieslak, A.; Madeja, Z.E.; Lechniak, D. Interactions of bovine oocytes with follicular elements with respect to lipid metabolism. Anim. Sci. J. 2017, 88, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Sanders, J.R.; Jones, K.T. Regulation of the meiotic divisions of mammalian oocytes and eggs. Biochem. Soc. Trans. 2018, 46, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Tomek, W.; Torner, H.; Kanitz, W. Comparative analysis of protein synthesis, transcription and cytoplasmic polyadenylation of mRNA during maturation of bovine oocytes in vitro. Reprod. Domest. Anim. 2002, 37, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Fair, T.; Carter, F.; Park, S.; Evans, A.C.; Lonergan, P. Global gene expression analysis during bovine oocyte in vitro maturation. Theriogenology 2007, 68 (Suppl. 1), S91–S97. [Google Scholar] [CrossRef]
- Thelie, A.; Papillier, P.; Perreau, C.; Uzbekova, S.; Hennequet Antier, C.; Dalbies Tran, R. Regulation of bovine oocyte-specific transcripts during in vitro oocyte maturation and after maternal–embryonic transition analyzed using a transcriptomic approach. Mol. Reprod. Dev. 2009, 76, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Graf, A.; Krebs, S.; Heininen-Brown, M.; Zakhartchenko, V.; Blum, H.; Wolf, E. Genome activation in bovine embryos: Review of the literature and new insights from RNA sequencing experiments. Anim. Reprod. Sci. 2014, 149, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Fowler, S.D.; Greenspan, P. Application of Nile red, a fluorescent hydrophobic probe, for the detection of neutral lipid deposits in tissue sections: Comparison with oil red O. J. Histochem. Cytochem. 1985, 33, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, S. A simple method for the isolation and purification of total lipides from animal tissues. J. biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Bligh, E.G.; Dyer, W.J. A Rapid Method of Total Lipid Extraction and Purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Southam, A.D.; Weber, R.J.; Engel, J.; Jones, M.R.; Viant, M.R. A complete workflow for high-resolution spectral-stitching nanoelectrospray direct-infusion mass-spectrometry-based metabolomics and lipidomics. Nat. Protoc. 2016, 12, 310–328. [Google Scholar] [CrossRef] [PubMed]
- Koelmel, J.P.; Kroeger, N.M.; Ulmer, C.Z.; Bowden, J.A.; Patterson, R.E.; Cochran, J.A.; Beecher, C.W.W.; Garrett, T.J.; Yost, R.A. LipidMatch: An automated workflow for rule-based lipid identification using untargeted high-resolution tandem mass spectrometry data. BMC Bioinform. 2017, 18, 331. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.; Tan, C.M.; Kou, Y.; Duan, Q.; Wang, Z.; Meirelles, G.V.; Clark, N.R.; Ma’ayan, A. Enrichr: Interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 2013, 14, 128. [Google Scholar] [CrossRef] [PubMed]
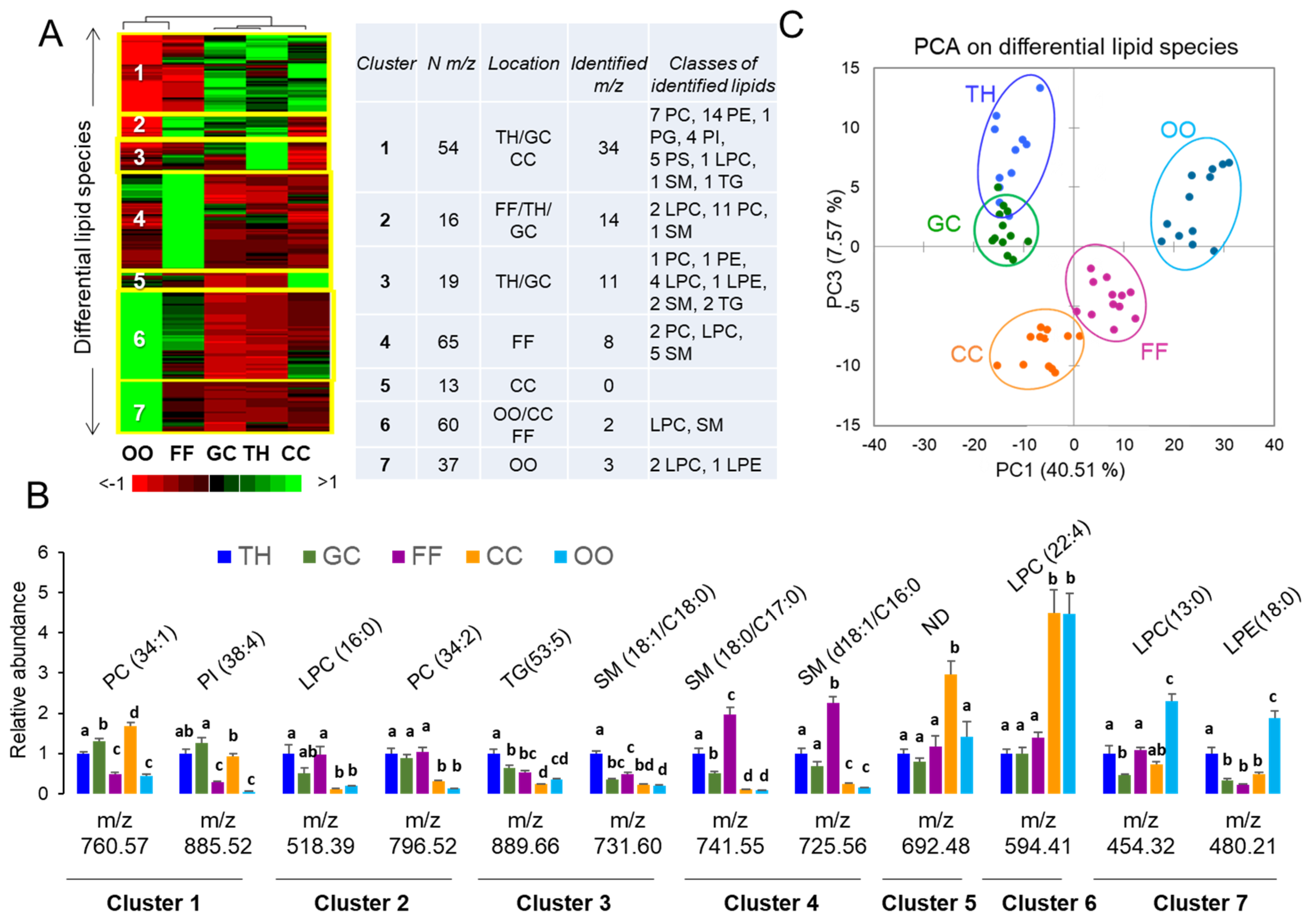
| Lipid Family/% from total lipids | Lipid Classes/% from total lipids |
| Fatty acyls (FA)/1.48% | Free fatty acid (FFA)/0.91%; Fatty acyl carnitine (FAC)/0.57%. |
| Glycerolipids (GL)/28.7% | Diacylglycerol (DG)/1.48%; Triacylglycerol (TG)/27.2%. |
| Glycerophospholipids (GPL)/52.56% | Lyso-phosphatidylcholine (LPC)/3.64%, phosphatidylcholine (PC)/30.33%; Lyso-phosphatidylethanolamine (LPE)/0.22%; phosphatidylethanolamine (PE)/12.88%; Lyso-phosphatidylinositol (LPI)/0.11%, phosphatidylinositol (PI)/1.71%; phosphoserine (PS)/2.85%; Glycerophosphates (here phosphatidic acids, PA)/0.23%; Phosphatidylglycerols (PG)/0.46%. |
| Sphingolipids (SL)/9.46% | Ceramides (Cer)/1.25%; Sphingomyelin (SM)/6.50%; Hexosylceramide (HexCer)/0.29%, Ganglioside (GM)/0.45%; Sulfoglycosphingolipid (SuSM)/1.02%. |
| Sterol Lipids (ST)/7.75% | Cholesteryl ester (CE)/7.41%; Sterols (ST)/0.34% |
| Clusters | Differentially Expressed Genes |
|---|---|
| Cluster 1 (TH, GC) | ABHD12, ABHD13, ACADM, ACSBG1, ACSS1, AKR1E2, BDH2, CLN1, CYP2C87, CYP4V2, CYP7A1, ESR2, GJA1, GPAM, GPD1, GPX1, GPX3, GPX7, GPX8, HMGCL, HSDL1, HSDL2, LIPA, MCCC1, MLYCD, OSBPL7, PLA2G16, PLA2G1B, PLBD2, PLSCR2, SPTSSA, SULT2A1, UGCG |
| Cluster 2 (TH, GC, CC) | ABHD14B, ABHD3, ABHD7, ACAD10, APOA2, APOC3, APOE, APOL3, APOM, CD36, ACSS3, CYP4F2, DAGLB, HSD17B11, HSD17B7, LCAT, MCCC2, OSBPL1A, PIK3C2G, PLCG1, PLSCR1, PNPLA5, PNPLA7, PPARGC1A, SCARB2, SCP2, SCD5 |
| Enriched GO terms (clusters 1–2): Ketone body, phospholipid scrambling, cholesterol binding, sterol transporter activity, phospholipase activity, acyl-CoA, glycerophospholipid biosynthetic processes, FA oxidation, unsaturated FA metabolism, cellular response to oxidative stress. Pathways: arachidonic acid metabolism, PPAR and AMPK signaling, glutathione and glycerophospholipid metabolism. | |
| Cluster 3 (CC) | ABHD15, ABHD16A, ACADL, ACADS, ACADVL, ACAT1, ACOX2, AGPAT5, AKR7A2, CDS2, CHPT1, CHST14, COMTD1, CYP19A1, CYP20A1, CYP51A1, DECR1, DGKA, FADS2, G6PC3, GPAA1, HSD11B1L, HSD3B, HSD3B7, LDLR, PI4KA, PIP5K1A, PKM2, PLCB4, PLD3, PPAP2A, SDHA, SLC27A3, SLC2A1, SLC2A3, SMPD1, SRD5A1, STARD7, STARD3NL, SUCLG1 |
| Cluster 5 (CC) | CHST11, CHST8, CYP2U1, DGKE, HSD17B1, LSS, OSBPL2, PFKFB3, PLA2G7, PNPLA6, PPARG, SCD, UGGT2 |
| Enriched GO terms (clusters 3, 5): FA beta-oxidation using acyl-CoA dehydrogenase, steroid, organic cyclic compound, glycerophospholipid biosynthetic processes; FA beta-oxidation, FA catabolic process, estrogen biosynthesis, bile acid metabolic process, regulation of cholesterol metabolic process. Pathways: glycerophospholipid and FA metabolism, phosphatidylinositol and phospholipase D signaling; PPAR, choline metabolism, FA degradation, steroidogenesis, AMPK signaling | |
| Cluster 4 (TH) | ACSF2, AGMO, AGPAT4, APOA1, CH25H, CHST1, CHST7, CYP27A1, CYP7B1, LPL, OXCT1, PDK4, PFKM, PIK3CG, PIK3R2, PLCD3, PLCXD3, PLD4, PLIN5, PLSCR4, PLTP, PPAP2A, PTPLA, SCARB1, SDHD, SMPD3, SMPDL3B, STARD5, SULT1A1, TM7SF2 |
| Enriched GO terms (cluster 4): Sterol import, regulation of cholesterol storage, regulation of sequestering of triglyceride and triglyceride synthesis; monocarboxylic acid bile acid biosynthetic process; sterol metabolic process. Pathways: PPAR signaling, primary bile acid biosynthesis, glycerolipid metabolism; Phospholipase D signaling pathway, Phosphatidylinositol signaling system, HDL-mediated transport | |
| Cluster 6 (CC, OO) | ABHD11, ACADSB, ACSL6, AKR1A1, C2CD2L, CHST10, OGFOD2, OSBP2, PDK1, PPRC1, PPT2, SLC2A8, SMPD4, SOAT1, THEM4, UGT, MCC |
| Cluster 7 (OO) | ABHD10, ABHD17C, ABHD4, ABHD5, ABHD6, ACAA1, ACACA, ACAD11, ACAT2, ACER3, ACO1, ACOT7, ACOT9, ACP6, ACSL3, ACSL4, ACYP1, AGPAT9, AKR1B1, APOO, C2CD4A, C2CD5, CDS1, CEPT1, CERKL, CERS2, CHERP, CHKA, CHST12, CPT1C, CRLS1, CS, CYP26A1, CYP27B1, DECR2, DGAT2, DGKI, EBPL, ELOVL3, ELOVL4, ELOVL5, ELOVL7, EPT1, FABP3, FABP5, FADS3, FAR1, GK, GPAT2, GPD1L, GPLD1, HADHB, HMGCS1, HSD11B2, HSD17B12, LPCAT2, MBOAT2, NFKBIE, OGFOD1, OSBPL10, OSBPL11, OSBPL3, OSBPL8, OSBPL9, OXSM, PDK2, PDK3, PFKFB1, PFKFB2, PI4K2B, PI4KB, PIK3C2A, PIK3CA, PIK3C3, PIK3CB, PIK3R1, PIP4K2A, PIP4K2B, PIP4K2C, PIP5K1B, PIP5K1C, PITPNA, PITPNM1, PLA2G12A, PLA2G15, PLAA, PLCG2, PLCH1, PLD6, PLIN2, PNPLA4, PPAP2C, PPARGC1B, PTPLAD2, SOD1, SOD2, SPTLC1, SPTLC2, SRBD1, STARD4, SUCLA2, SUCLG2, THEM2, TM7SF3, UGDH, UGP2, UGT8 |
| Enriched GO terms (clusters 6–7): Glycerophospholipid and phosphatidylinositol biosynthetic processes; long-chain fatty-acyl-CoA biosynthesis and acyl-CoA metabolic process; FA elongation (saturated and unsaturated) FA; lipid phosphorylation; sphingolipid metabolic process; membrane lipid biosynthetic process. Pathways: Phosphatidylinositol and inositol lipid-mediated signaling; PPAR signaling; FA elongation, FA degradation; GL and GPL metabolism; peroxisome, choline metabolism; sphingolipid metabolism | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertevello, P.S.; Teixeira-Gomes, A.-P.; Seyer, A.; Vitorino Carvalho, A.; Labas, V.; Blache, M.-C.; Banliat, C.; Cordeiro, L.A.V.; Duranthon, V.; Papillier, P.; et al. Lipid Identification and Transcriptional Analysis of Controlling Enzymes in Bovine Ovarian Follicle. Int. J. Mol. Sci. 2018, 19, 3261. https://doi.org/10.3390/ijms19103261
Bertevello PS, Teixeira-Gomes A-P, Seyer A, Vitorino Carvalho A, Labas V, Blache M-C, Banliat C, Cordeiro LAV, Duranthon V, Papillier P, et al. Lipid Identification and Transcriptional Analysis of Controlling Enzymes in Bovine Ovarian Follicle. International Journal of Molecular Sciences. 2018; 19(10):3261. https://doi.org/10.3390/ijms19103261
Chicago/Turabian StyleBertevello, Priscila Silvana, Ana-Paula Teixeira-Gomes, Alexandre Seyer, Anaïs Vitorino Carvalho, Valérie Labas, Marie-Claire Blache, Charles Banliat, Luiz Augusto Vieira Cordeiro, Veronique Duranthon, Pascal Papillier, and et al. 2018. "Lipid Identification and Transcriptional Analysis of Controlling Enzymes in Bovine Ovarian Follicle" International Journal of Molecular Sciences 19, no. 10: 3261. https://doi.org/10.3390/ijms19103261
APA StyleBertevello, P. S., Teixeira-Gomes, A.-P., Seyer, A., Vitorino Carvalho, A., Labas, V., Blache, M.-C., Banliat, C., Cordeiro, L. A. V., Duranthon, V., Papillier, P., Maillard, V., Elis, S., & Uzbekova, S. (2018). Lipid Identification and Transcriptional Analysis of Controlling Enzymes in Bovine Ovarian Follicle. International Journal of Molecular Sciences, 19(10), 3261. https://doi.org/10.3390/ijms19103261





