Importance of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants on Acidic Soils: Current Status and Opportunities
Abstract
1. Introduction
2. Multiple Forms of Aluminum in the Soil Environment Relevant to Toxicity
3. Aluminum Uptake, Accumulation, and Toxicity Responses in Plants
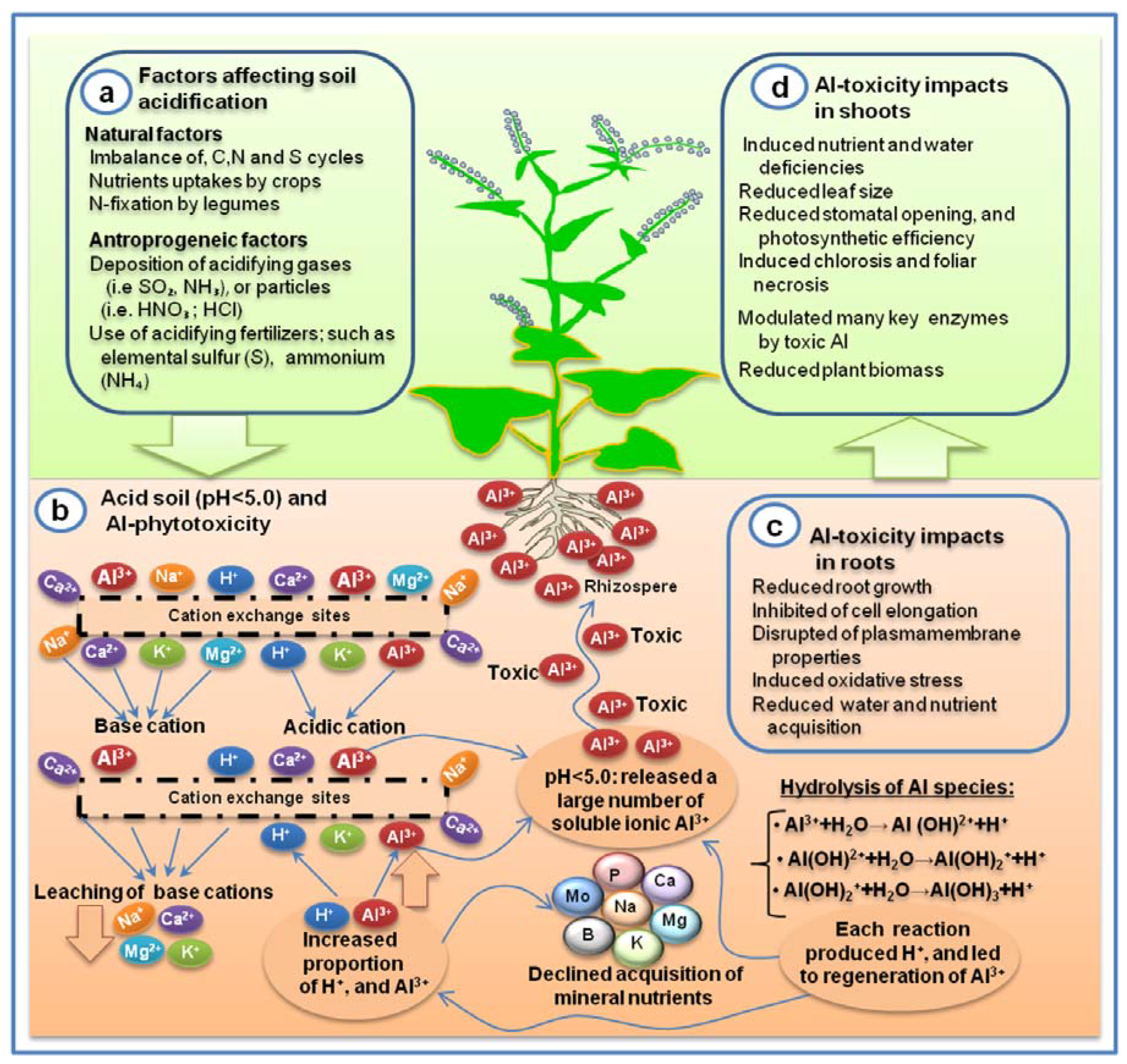
4. Factors Affecting Aluminum Toxicity and Nutrient Imbalance
5. Role of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants
5.1. Calcium (Ca)
5.1.1. Liming
5.1.2. Phosphogypsum (PG)
5.1.3. Gypsum (G)
5.2. Magnesium (Mg)
5.3. Phosphorus (P)
5.4. Sulfur (S)
5.5. Boron (B)
5.6. Silicon (Si)
5.7. Miscellaneous
6. Conclusions and Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Al | Aluminum |
| ALMT1 | Aluminum-activated malate transporter 1 |
| AtMGT1 | Arabidopsis magnesium transport gene 1 |
| BC | Base cation |
| Ca | Calcium |
| PG | Phosphogypsum |
| G | Gypsum |
| Mg | Magnesium |
| P | Phosphorus |
| S | Sulfur |
| B | Boron |
| Mg | Manganese |
| Zn | Zinc |
| Mo | Molybdenum |
| Si | Silicon |
| OAs | Organic acids |
| IAA | Indole acetic acid |
| PA | Polyamine |
| SA | Salicylic acid |
| Put | Putrescine |
| MF | Micorrhizal fungi |
| PGPB | Plant growth promoting bacteria |
References
- Kochian, L.V.; Piñeros, M.A.; Liu, J.; Magalhaes, J.V. Plant adaptation to aid soils: The molecular basis for crop aluminum resistance. Annu. Rev. Plant Biol. 2015, 66, 571–598. [Google Scholar] [CrossRef] [PubMed]
- Von Uexküll, H.R.; Mutert, E. Global extent, development and economic impact of acid soils. In Plant-Soil Interactions at Low pH: Principles and Management, Proceedings of the Third International Symposium on Plant-Soil Interactions at Low pH, Brisbane, Queensland, Australia, 12–16 September 1993; Date, R.A., Grundon, N.J., Rayment, G.E., Probert, M.E., Eds.; Springer: Dordrecht, The Netherlands, 1995; pp. 5–19. [Google Scholar]
- Bojórquez-Quintal, E.; Escalante-Magaña, C.; Echevarría-Machado, I.; Martínez-Estévez, M. Aluminum, a friend or foe of higher plants in acid soils. Front. Plant Sci. 2017, 8, 1767. [Google Scholar] [CrossRef] [PubMed]
- Dashuan, T.; Shuli, N. A global analysis of soil acidification caused by nitrogen addition. Environ. Res. Lett. 2015, 10, 024019. [Google Scholar] [CrossRef]
- Goulding, K.W.T. Soil acidification and the importance of liming agricultural soils with particular reference to the United Kingdom. Soil Use Manag. 2016, 32, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Tripathi, D.K.; Singh, S.; Sharma, S.; Dubey, N.K.; Chauhan, D.K.; Vaculík, M. Toxicity of aluminium on various levels of plant cells and organism: A review. Environ. Exp. Bot. 2017, 137, 177–193. [Google Scholar] [CrossRef]
- Mossor-Pietraszewska, T. Effect of aluminium on plant growth and metabolism. Acta Biochim. Pol. 2001, 48, 673–686. [Google Scholar] [PubMed]
- Kisnieriené, V.; Lapeikaité, I. When chemistry meets biology: The case of aluminium—A review. Chemija 2015, 26, 148–158. [Google Scholar]
- Grevenstuk, T.; Romano, A. Aluminium speciation and internal detoxification mechanisms in plants: Where do we stand? Metallomics 2013, 5, 1584–1594. [Google Scholar] [CrossRef] [PubMed]
- Frankowski, M. Aluminum uptake and migration from the soil compartment into Betula pendula for two different environments: A polluted and environmentally protected area of Poland. Environ. Sci. Pollut. Res. Int. 2016, 23, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, D.T. The Effect of aluminium and some other trivalent metal cations on cell division in the root apices of Allium Cepa. Ann. Bot. 1965, 29, 309–315. [Google Scholar] [CrossRef]
- Mariano, E.D.; Keltjens, W.G. Long-term effects of aluminum exposure on nutrient uptake by maize genotypes differing in aluminum resistance. J. Plant Nutr. 2005, 28, 323–333. [Google Scholar] [CrossRef]
- Chen, Z.C.; Liao, H. Organic acid anions: An effective defensive weapon for plants against aluminum toxicity and phosphorus deficiency in acidic soils. J. Genet. Genom. 2016, 43, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Motoda, H. Aluminum toxicity recovery processes in root apices. Possible association with oxidative stress. Plant Sci. 2012, 185–186, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Horst, W.J.; Asher, C.J.; Cakmak, I.; Szulkiewicz, P.; Wissemeier, A.H. Short-term responses of soybean roots to aluminium. J. Plant Physiol. 1992, 140, 174–178. [Google Scholar] [CrossRef]
- Kuo, M.C.; Kao, C.H. Aluminum effects on lipid peroxidation and antioxidative enzyme activities in rice leaves. Biol. Plant. 2003, 46, 149–152. [Google Scholar] [CrossRef]
- Devi, S.R.; Yamamoto, Y.; Matsumoto, H. An intracellular mechanism of aluminum tolerance associated with high antioxidant status in cultured tobacco cells. J. Inorg. Biochem. 2003, 97, 59–68. [Google Scholar] [CrossRef]
- Rengel, Z. Aluminium cycling in the soil-plant-animal-human continuum. Biometals 2004, 17, 669–689. [Google Scholar] [CrossRef] [PubMed]
- Sivaguru, M.; Fujiwara, T.; Šamaj, J.; Baluška, F.; Yang, Z.; Osawa, H.; Maeda, T.; Mori, T.; Volkmann, D.; Matsumoto, H. Aluminum-induced 1→3-β-d-glucan Inhibits cell-to-cell trafficking of molecules through plasmodesmata. A new mechanism of aluminum toxicity in plants. Plant Physiol. 2000, 124, 991–1006. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Kobayashi, Y.; Devi, S.R.; Rikiishi, S.; Matsumoto, H. Aluminum toxicity is associated with mitochondrial dysfunction and the production of reactive oxygen species in plant cells. Plant Physiol. 2002, 128, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-T.; Qi, Y.-P.; Jiang, H.-X.; Chen, L.-S. Roles of organic acid anion secretion in aluminium tolerance of higher plants. BioMed Res. Int. 2013, 2013, 16. [Google Scholar] [CrossRef] [PubMed]
- Klug, B.; Specht, A.; Horst, W.J. Aluminium localization in root tips of the aluminium-accumulating plant species buckwheat (Fagopyrum esculentum Moench). J. Exp. Bot. 2011, 62, 5453–5462. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ren, X.; Huang, B.; Wang, G.; Zhou, P.; An, Y. Aluminium-induced reduction of plant growth in alfalfa (Medicago sativa) is mediated by interrupting auxin transport and accumulation in roots. Sci. Rep. 2016, 6, 30079. [Google Scholar] [CrossRef] [PubMed]
- Klug, B.; Horst Walter, J. Oxalate exudation into the root-tip water free space confers protection from aluminum toxicity and allows aluminum accumulation in the symplast in buckwheat (Fagopyrum esculentum). New Phytol. 2010, 187, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Arunakumara, K.K.I.U.; Walpola, B.C.; Yoon, M.-H. Aluminum toxicity and tolerance mechanism in cereals and legumes—A review. J. Korean Soc. Appl. Biol. Chem. 2013, 56, 1–9. [Google Scholar] [CrossRef]
- Xu, J.M.; Fan, W.; Jin, J.F.; Lou, H.Q.; Chen, W.W.; Yang, J.L.; Zheng, S.J. Transcriptome analysis of al-induced genes in buckwheat (Fagopyrum esculentum Moench) root apex: New insight into al toxicity and resistance mechanisms in an al accumulating species. Front. Plant Sci. 2017, 8, 1141. [Google Scholar] [CrossRef] [PubMed]
- Nunes-Nesi, A.; Brito, D.S.; Inostroza-Blancheteau, C.; Fernie, A.R.; Araújo, W.L. The complex role of mitochondrial metabolism in plant aluminum resistance. Trends Plant Sci. 2014, 19, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Ryan, P.R.; Delhaize, E. Aluminium tolerance in plants and the complexing role of organic acids. Trends Plant Sci. 2001, 6, 273–278. [Google Scholar] [CrossRef]
- Lazof, D.B.; Goldsmith, J.G.; Rufty, T.W.; Linton, R.W. Rapid uptake of aluminum into cells of intact soybean root tips (a microanalytical study using secondary ion mass spectrometry). Plant Physiol. 1994, 106, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Mora, M.L.; Alfaro, M.A.; Jarvis, S.C.; Demanet, R.; Cartes, P. Soil aluminium availability in Andisols of southern Chile and its effect on forage production and animal metabolism. Soil Use Manag. 2006, 22, 95–101. [Google Scholar] [CrossRef]
- Chaney, R.I. Toxic element accumulation in soil and crop, protecting soil fertility and agricultural food-chains. In Inorganics Contaminants in the Vadose Zone; Yosef, B., Barrow, N.J., Goldshmid, J., Eds.; Springer: Berlin, Germany, 1989; pp. 140–158. [Google Scholar]
- Wang, J.-P.; Raman, H.; Zhang, G.-P.; Mendham, N.; Zhou, M.-X. Aluminium tolerance in barley (Hordeum vulgare L.): Physiological mechanisms, genetics and screening methods. J. Zhejiang Univ. Sci. B 2006, 7, 769–787. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Hirasawa, E.; Morimura, S.; Takahashi, E. Localization of aluminium in tea leaves. Plant Cell Physiol. 1976, 17, 627–631. [Google Scholar] [CrossRef]
- Mehra, A.; Baker, C.L. Leaching and bioavailability of aluminium, copper and manganese from tea (Camellia sinensis). Food Chem. 2007, 100, 1456–1463. [Google Scholar] [CrossRef]
- Wills, S.J. Aluminum toxicity and chronic renal failure. In Metal Ions in Biological Systems. Aluminium and Its Role in Biology; Sigel, H., Sigel, A., Eds.; Dekker: New York, NY, USA, 1998; Volume 24, pp. 315–345. [Google Scholar]
- Stewart, W. Aluminium toxicity in individuals with chronic renal disease. In Aluminium in Food and the Environment; Massey, R.C., Taylor, D., Eds.; Royal Society Chemistry: Cambridge, UK, 1989; pp. 6–19. [Google Scholar]
- Anitha, S.; Rao, K.S.J. The complexity of aluminum-DNA interactions: Relevance to Alzheimer’s and other neurological diseases. In Group 13 Chemistry II: Biological Aspects of Aluminum; Roesky, H.W., Atwood, D.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 79–97. [Google Scholar]
- Kawahara, M.; Konoha, K.; Nagata, T.; Sadakane, Y. Aluminum and human health: Its intake, bioavailability and neurotoxicity. Biomed. Res. Trace Elem. 2007, 18, 211–220. [Google Scholar]
- Klotz, K.; Weistenhöfer, W.; Neff, F.; Hartwig, A.; van Thriel, C.; Drexler, H. The health effects of aluminum exposure. Dtsch. Arztebl. Int. 2017, 114, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Collard, B.C.Y.; Mackill, D.J. Marker-assisted selection: An approach for precision plant breeding in the twenty-first century. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 557–572. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: Critical adaptations by plants for securing a nonrenewable resource. New Phytol. 2003, 157, 423–447. [Google Scholar] [CrossRef]
- Gupta, N.; Gaurav, S.S.; Kumar, A. Molecular basis of aluminium toxicity in plants: A review. Am. J. Plant Sci. 2013, 4, 21–37. [Google Scholar] [CrossRef]
- Rahman, M.A.; Chikushi, J.; Duxbury, J.M.; Meisner, C.A.; Lauen, J.G.; Yasunaga, E. Chemical control of soil environment by lime and nutrients to improve the productivity of acidic alluvial soils under rice-wheat cropping system in Bangladesh. Environ. Cont. Biol. 2005, 43, 259–266. [Google Scholar] [CrossRef]
- Akhtaruzzaman, M.; Haque, M.E.; Osman, K.T. Morphological, physical and chemical characteristics of hill forest soils at Chittagong University, Bangladesh. Open J. Soil Sci. 2014, 4, 10. [Google Scholar] [CrossRef]
- Flues, M.; Sato, I.M.; Cotrim, M.B.; Salvador, V.L.; Ranzani, A.C.; Vallilo, M.I.; de Oliveira, E. Soil characterization in a subtropical forest crossed by highways (Cantareira State Park, SP, Brazil). J. Braz. Chem. Soc. 2004, 15, 496–503. [Google Scholar] [CrossRef]
- Ministry of Environmental Protection of the People’s Republic of China (MEPPRC). Environmental Quality Standard for Soils (GB 15618-1995); Ministry of Environmental Protection of the People’s Republic of China: Beijing, China, 1995.
- Wang, S.; Wang, P.; Fan, C.Q. Distribution of aluminum fractionation in the acidic rhizosphere soils of masson pine (Pinus massoniana Lamb.). Commun. Soil Sci. Plant Anal. 2015, 46, 2033–2050. [Google Scholar] [CrossRef]
- Xie, Z.; Chen, Z.; Sun, W.; Guo, X.; Yin, B.; Wang, J. Distribution of aluminum and fluoride in tea plant and soil of tea garden in central and southwest China. Chin. Geogr. Sci. 2007, 17, 376–382. [Google Scholar] [CrossRef]
- Soon, Y. Forms of extractable aluminium in Canadian acid soils and their relations to plant growth. In Plant-Soil Interactions at Low pH: Principles and Management; Springer: Dordrecht, The Netherlands, 1995; pp. 65–70. [Google Scholar]
- Bradová, M.; Tejnecký, V.; Borůvka, L.; Němeček, K.; Ash, C.; Šebek, O.; Svoboda, M.; Zenáhlíková, J.; Drábek, O. The variations of aluminium species in mountainous forest soils and its implications to soil acidification. Environ. Sci. Pollut. Res. Int. 2015, 22, 16676–16687. [Google Scholar] [CrossRef] [PubMed]
- Fichter, J.; Turpault, M.-P.; Dambrine, E.; Ranger, J. Mineral evolution of acid forest soils in the Strengbach catchment (Vosges mountains, N-E France). Geoderma 1998, 82, 315–340. [Google Scholar] [CrossRef]
- Bera, R.; Seal, A.; Banerjee, M.; Dolui, A.K. Nature and profile distribution of iron and aluminum in relation to pedogenic processes in some soils developed under tropical environment in India. Environ. Geol. 2005, 47, 241–245. [Google Scholar] [CrossRef]
- McGrath, D.; Fleming, G.A.; Culleton, N. Trace Elements and Heavy Metals in Irish Soils; Teagasc: Wexford, Ireland, 2008; pp. 1–184. [Google Scholar]
- Yanai, J.; Okada, T.; Yamada, H. Elemental composition of agricultural soils in Japan in relation to soil type, land use and region. Soil Sci. Plant Nutr. 2012, 58, 1–10. [Google Scholar] [CrossRef]
- Kim, J.G.; Lee, S.S.; Moon, H.-S.; Kang, I.M. Land application of alum sludge from water purification plant to acid mineral soil treated with acidic water. Soil Sci. Plant Nutr. 2002, 48, 15–22. [Google Scholar] [CrossRef]
- Lee, C.H.; Lee, S.-W.; Kim, E.-Y.; Jeong, J.-H.; Cho, H.-J.; Park, G.-S.; Lee, C.-Y.; Jeong, Y.-H. Effects of air pollution and acid deposition on three Pinus densiflora (Japanese red pine) forests in South Korea. J. Agric. Meteorol. 2005, 60, 1153–1156. [Google Scholar] [CrossRef]
- Moir, J.; Moot, D. Medium-term soil pH and exchangeable aluminium response to liming at three high country locations. In Proceedings of the New Zealand Grassland Association Conference, Alexandra, New Zealand, 5–7 November 2014; New Zealand Grassland Association: Dunedin, New Zealand, 2014; pp. 41–45. [Google Scholar]
- Brown, T.T.; Koenig, R.T.; Huggins, D.R.; Harsh, J.B.; Rossi, R.E. Lime effects on soil acidity, crop yield, and aluminum chemistry in direct-seeded cropping systems. Soil Sci. Soc. Am. J. 2008, 72, 634–640. [Google Scholar] [CrossRef]
- Lee, Y.-B.; Bigham, J.M.; Kim, P.-J. Evaluate changes in soil chemical properties following FGD-gypsum application. Korean J. Environ. Agric. 2007, 26, 294–299. [Google Scholar] [CrossRef]
- Jaiswal, S.K.; Naamala, J.; Dakora, F.D. Nature and mechanisms of aluminium toxicity, tolerance and amelioration in symbiotic legumes and rhizobia. Biol. Fert. Soils 2018, 54, 309–318. [Google Scholar] [CrossRef]
- Vitorello, V.A.; Capaldi, F.R.; Stefanuto, V.A. Recent advances in aluminum toxicity and resistance in higher plants. Braz. J. Plant Physiol. 2005, 17, 129–143. [Google Scholar] [CrossRef]
- Delhaize, E.; Ryan, P.R. Aluminum toxicity and tolerance in plants. Plant Physiol. 1995, 107, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Brautigan, D.J.; Rengasamy, P.; Chittleborough, D.J. Aluminium speciation and phytotoxicity in alkaline soils. Plant Soil 2012, 360, 187–196. [Google Scholar] [CrossRef]
- Kinraide, T.B. Assessing the rhizotoxicity of the aluminate Ion,Al(OH)4. Plant Physiol. 1990, 93, 1620–1625. [Google Scholar] [CrossRef] [PubMed]
- Rengel, Z. Uptake of aluminum by plant cells. New Phytol. 1996, 134, 389–406. [Google Scholar] [CrossRef]
- Kopittke, P.M.; Moore, K.L.; Lombi, E.; Gianoncelli, A.; Ferguson, B.J.; Blamey, F.P.C.; Menzies, N.W.; Nicholson, T.M.; McKenna, B.A.; Wang, P.; et al. Identification of the primary lesion of toxic aluminum in plant roots. Plant Physiol. 2015, 167, 1402–1411. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Hiradate, S. Form of aluminium for uptake and translocation in buckwheat (Fagopyrum esculentum Moench). Planta 2000, 211, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, R.; Li, D.; Jia, X.; Zhou, D.; Li, J.; Lyi, S.M.; Hou, S.; Huang, Y.; Kochian, L.V.; et al. NIP1;2 is a plasma membrane-localized transporter mediating aluminum uptake, translocation, and tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, 5047–5052. [Google Scholar] [CrossRef] [PubMed]
- Chenery, E.M. Aluminium in the Plant World. Kew Bull. 1948, 3, 173–183. [Google Scholar] [CrossRef]
- Chenery, E.M. Aluminium in the Plant World. Kew Bull. 1949, 4, 463–473. [Google Scholar] [CrossRef]
- Chen, R.F.; Shen, R.F.; Gu, P.; Wang, H.Y.; Xu, X.H. Investigation of aluminum-tolerant species in acid soils of South China. Commun. Soil Sci. Plant Anal. 2008, 39, 1493–1506. [Google Scholar] [CrossRef]
- Ownby, J.D.; Popham, H.R. Citrate reverses the inhibition of wheat root growth caused by aluminum. J. Plant Physiol. 1989, 135, 588–591. [Google Scholar] [CrossRef]
- Palmer, A.J.; Baker, A.; Muench, S.P. The varied functions of aluminium-activated malate transporters-much more than aluminium resistance. Biochem. Soc. Trans. 2016, 44, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Yokosho, K.; Yamaji, N.; Fujii-Kashino, M.; Ma, J.F. Retrotransposon-mediated aluminum tolerance through enhanced expression of the citrate transporter OsFRDL4. Plant Physiol. 2016, 172, 2327–2336. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Li, R.; Shi, J.; Wang, J.; Sun, Q.; Zhang, H.; Xing, Y.; Qi, Y.; Zhang, N.; Guo, Y.-D. Brassica oleracea MATE Encodes a citrate transporter and enhances aluminum tolerance in Arab. Thaliana. Plant Cell Physiol. 2014, 55, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.-J.; Seo, Y.-J.; Futakuchi, K.; Vijarnsorn, P.; Ishii, R. Effect of aluminum toxicity on flowering time and grain yield on rice genotypes differing in Al-tolerance. J. Crop. Sci. Biotechnol. 2011, 14, 305–309. [Google Scholar] [CrossRef]
- Tan, K.; Keltjens, W.G.; Findenegg, G.R. Aluminum toxicity in sorghum genotypes as influenced by solution acidity. Soil Sci. Plant Nutr. 1993, 39, 291–298. [Google Scholar] [CrossRef]
- Silva, S.; Pinto-Carnide, O.; Martins-Lopes, P.; Matos, M.; Guedes-Pinto, H.; Santos, C. Differential aluminium changes on nutrient accumulation and root differentiation in an Al sensitive vs. tolerant wheat. Environ. Exp. Bot. 2010, 68, 91–98. [Google Scholar] [CrossRef]
- Islam, M.A.; Dowling, P.M.; Milham, P.J.; Campbell, L.C.; Jacobs, B.C.; Garden, D.L. Ranking acidity tolerance and growth potential of Austrodanthonia accessions. Grassl. Sci. 2006, 52, 127–132. [Google Scholar] [CrossRef]
- Pereira, J.F. Initial root length in wheat is highly correlated with acid soil tolerance in the field. Sci. Agric. 2018, 75, 79–83. [Google Scholar] [CrossRef]
- Koenig, R.; Schroeder, K.; Carter, A.; Pumphrey, M.; Paulitz, T.; Campbell, K.; Huggins, D. Soil Acidity and Aluminum Toxicity in the Palouse Region of the Pacific Northwest; Washington State University Extension: Washington, DC, USA, 2011. [Google Scholar]
- Leidi, E.; Rodrıguez-Navarro, D.N.; Fernández, M.; Sarmiento, R.; Semedo, J.; Marques, N.; Matos, A.; Machado, A.P.; Ørting, B.; Sørensen, M.; et al. Factors affecting root and seed yield in ahipa (Pachyrhizus ahipa (Wedd.) Parodi), a multipurpose legume crop. Eur. J. Agron. 2004, 20, 395–403. [Google Scholar] [CrossRef]
- Ishikawa, S.; Wagatsuma, T. Plasma membrane permeability of root-tip cells following temporary exposure to al ions is a rapid measure of al tolerance among plant species. Plant Cell Physiol. 1998, 39, 516–525. [Google Scholar] [CrossRef]
- Nagy, N.E.; Dalsen, L.S.; Jones, D.L.; Swensen, B.; Fossdal, C.G.; Eldhuset, T.D. Cytological and enzymatic responses to aluminium stress in root tips of Norway spruce seedlings. New Phytol. 2004, 163, 595–607. [Google Scholar] [CrossRef]
- Watanabe, T.; Osaki, M. Role of organic acids in aluminum accumulation and plant growth in Melastoma Malabathricum. Tree Physiol. 2002, 22, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.; Yoshimura, E.; Nishizawa, N.K.; Mori, S. Time course study of aluminum-induced callose formation in barley roots as observed by digital microscopy and low-vacuum scanning electron microscopy. Soil Sci. Plant Nutr. 1999, 45, 701–712. [Google Scholar] [CrossRef]
- Rangel, A.F.; Madhusudana, R.I.; Johannes, H.W. Intracellular distribution and binding state of aluminum in root apices of two common bean (Phaseolus vulgaris) genotypes in relation to Al toxicity. Physiol. Plant. 2009, 135, 162–173. [Google Scholar] [CrossRef] [PubMed]
- McLean, F.T.; Gilbert, B.E. The relative aluminum tolerance of crop plants. Soil Sci. 1927, 24, 163–176. [Google Scholar] [CrossRef]
- Parrot, W.; Bouton, J. Aluminum tolerance in alfalfa as expressed in tissue culture. Crop Sci. 1990, 30, 387–389. [Google Scholar] [CrossRef]
- Khatiwada, S.P.; Senadhira, D.; Carpena, A.L.; Zeigler, R.S.; Fernandez, P.G. Variability and genetics of tolerance for aluminum toxicity in rice (Oryza sativa L.). Theor. Appl. Genet. 1996, 93, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Garvin, D.F.; Carver, B.F. Role of the genotype in tolerance to acidity and aluminum toxicity. In Handbook of Soil Acidity; Marcel Dekker: New York, NY, USA, 2003; pp. 387–406. [Google Scholar]
- Famoso, A.N.; Clark, R.T.; Shaff, J.E.; Craft, E.; McCouch, S.R.; Kochian, L.V. Development of a novel aluminum tolerance phenotyping platform used for comparisons of cereal aluminum tolerance and investigations into rice aluminum tolerance mechanisms. Plant Physiol. 2010, 153, 1678–1691. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.J.; Blarney, F.P.C.; Edwards, D.G. Antagonistic and synergistic interactions between aluminum and manganese on growth of Vigna unguiculata at low ionic strength. Physiol. Plant. 1998, 104, 183–194. [Google Scholar] [CrossRef]
- Bolan, N.S.; Hedley, M.J.; White, R.E. Processes of soil acidification during nitrogen cycling with emphasis on legume based pastures. Plant Soil 1991, 134, 53–63. [Google Scholar] [CrossRef]
- Veitch, F.P. Comparison of methods for the estimation of soil acidity. J. Am. Chem. Soc. 1904, 26, 637–662. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Driscoll, K.M.; Mitchell, M.J.; Raynal, D.J. Effects of acidic deposition on forest and aquatic ecosystems in New York State. Environ. Pollut. 2003, 123, 327–336. [Google Scholar] [CrossRef]
- Brunner, I.; Sperisen, C. Aluminum exclusion and aluminum tolerance in woody plants. Front. Plant Sci. 2013, 4, 172. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, R.; Parkpian, P.; Garivait, H.; Anurakpongsatorn, P.; DeLaune, R.D.; Jugsujinda, A. Impacts of acid rain on base cations, aluminum, and acidity development in highly weathered soils of Thailand. Commun. Soil Sci. Plant Anal. 2012, 43, 1382–1400. [Google Scholar] [CrossRef]
- Rengel, Z. Role of pH in Availability of Ions in Soil. In Handbook of Plant Growth pH as the Master Variable, 2nd ed.; Rengel, Z., Ed.; Taylor & Francis: New York, NY, USA, 2002; pp. 303–328. [Google Scholar]
- Stass, A.; Wang, Y.; Eticha, D.; Horst, W.J. Aluminium rhizotoxicity in maize grown in solutions with Al3+ or Al(OH)4− as predominant solution Al species. J. Exp. Bot. 2006, 57, 4033–4042. [Google Scholar] [CrossRef] [PubMed]
- Horst, W.J.; Püschel, A.-K.; Schmohl, N. Induction of callose formation is a sensitive marker for genotypic aluminium sensitivity in maize. Plant Soil 1997, 192, 23–30. [Google Scholar] [CrossRef]
- Gensemer, R.W.; Playle, R.C. The bioavailability and toxicity of aluminum in aquatic environments. Crit. Rev. Environ. Sci. Technol. 1999, 29, 315–450. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Schecher, W.D. The chemistry of aluminum in the environment. Environ. Geochem. Health 1990, 12, 28–49. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, C.T.; Schecher, W.D. Aqueous Chemistry of Aluminium. In Aluminium and Health: A Critical Review; Gitelman, H.J., Ed.; Marcel Dekker: New York, NY, USA, 1989; pp. 27–65. [Google Scholar]
- Adams, M.L. Speciation and Measurement of Aluminium in Environmental Systems. Ph.D. Thesis, University of Canterbury, Christchurch, New Zealand, 1999. [Google Scholar]
- Olivares, E.; Peña, E.; Marcano, E.; Mostacero, J.; Aguiar, G.; Benítez, M.; Rengifo, E. Aluminum accumulation and its relationship with mineral plant nutrients in 12 pteridophytes from Venezuela. Environ. Exp. Bot. 2009, 65, 132–141. [Google Scholar] [CrossRef]
- De Mendonça, R.J.; Cambraia, J.; de Oliveira, J.A.; Oliva, M.A. Efeito do alumínio na absorção e na utilização de macronutrientes em duas cultivares de arroz. Pesqui. Agropecu. Bras. 2003, 38, 843–848. [Google Scholar] [CrossRef]
- Simon, L.; Smalley, T.J.; Jones, J.B.; Lasseigne, F.T. Aluminum toxicity in tomato. Part 1. Growth and mineral nutrition. J. Plant Nutr. 1994, 17, 293–306. [Google Scholar] [CrossRef]
- Zobel, R.W.; Kinraide, T.B.; Baligar, V.C. Fine root diameters can change in response to changes in nutrient concentrations. Plant Soil 2007, 297, 243–254. [Google Scholar] [CrossRef]
- Poschenrieder, C.; Llugany, M.; Barceló, J. Short-term effects of pH and aluminium on mineral nutrition in maize varieties differing in proton and aluminium tolerance. J. Plant Nutr. 1995, 18, 1495–1507. [Google Scholar] [CrossRef]
- Brady, D.J.; Edwards, D.G.; Asher, C.J.; Blamey, F.P.C. Calcium amelioration of aluminium toxicity effects on root hair development in soybean [Glycine max (L.) Merr.]. New Phytol. 1993, 123, 531–538. [Google Scholar] [CrossRef]
- Edmeades, D.; Wheeler, D.; Blamey, F.; Christie, R. Calcium and Magnesium Amelioration of Aluminium Toxicity in Al-sensitive and Al-tolerant Wheat. In Plant-Soil Interactions at Low pH; Springer: Berlin, Germany, 1991; Volume 45, pp. 755–761. [Google Scholar]
- Silva, I.R.; Smyth, T.J.; Israel, D.W.; Raper, C.D.; Rufty, T.W. Magnesium ameliorates aluminum rhizotoxicity in soybean by increasing citric acid production and exudation by roots. Plant Cell Physiol. 2001, 42, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Shen, R.; Xiao, H.; Xu, M.; Wang, H.; Wang, H.; Zeng, Q.; Bian, J. Boron alleviates aluminum toxicity in pea (Pisum sativum). Plant Soil 2009, 314, 87. [Google Scholar] [CrossRef]
- Zhou, X.-X.; Yang, L.-T.; Qi, Y.-P.; Guo, P.; Chen, L.-S. Mechanisms on boron-induced alleviation of aluminum-toxicity in citrus grandis seedlings at a transcriptional level revealed by cDNA-AFLP analysis. PLoS ONE 2015, 10, e0115485. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Li, Q.; Qi, Y.-P.; Yang, L.-T.; Ye, X.; Chen, H.-H.; Chen, L.-S. Sulfur-mediated-alleviation of aluminum-toxicity in Citrus grandis seedlings. Int. J. Mol. Sci. 2017, 18, 2570. [Google Scholar] [CrossRef] [PubMed]
- Teng, W.; Kang, Y.; Hou, W.; Hu, H.; Luo, W.; Wei, J.; Wang, L.; Zhang, B. Phosphorus application reduces aluminum toxicity in two Eucalyptus clones by increasing its accumulation in roots and decreasing its content in leaves. PLoS ONE 2018, 13, e0190900. [Google Scholar] [CrossRef] [PubMed]
- Panhwar, Q.A.; Naher, U.A.; Radziah, O.; Shamshuddin, J.; Razi, I.M. Bio-fertilizer, ground magnesium limestone and basalt applications may improve chemical properties of Malaysian acid sulfate soils and rice growth. Pedosphere 2014, 24, 827–835. [Google Scholar] [CrossRef]
- Panhwar, Q.A.; Naher, U.A.; Radziah, O.; Shamshuddin, J.; Razi, I.M. Eliminating aluminum toxicity in an acid sulfate soil for rice cultivation using plant growth promoting bacteria. Molecules 2015, 20, 3628–3646. [Google Scholar] [CrossRef] [PubMed]
- Noble, A.D.; Sumner, M.E. Calcium and Al interactions and soybean growth in nutrient solutions. Commun. Soil Sci. Plant Anal. 1988, 19, 1119–1131. [Google Scholar] [CrossRef]
- Kinraide, T.B. Three mechanisms for the calcium alleviation of mineral toxicities. Plant Physiol. 1998, 118, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Ashrafuzzaman, M.; Hossain, A.K.M.Z.; Ismail, M.R.; Koyama, H. Role of accumulated calcium in alleviating aluminum injury in wheat plants. Sci. World J. 2014, 2014, 457187. [Google Scholar] [CrossRef] [PubMed]
- Vicensi, M.; Müller, M.M.L.; Kawakami, J.; de Nascimento, R.; Michalovicz, L.; Lopes, C. Do rates and splitting of phosphogypsum applications influence the soil and annual crops in a no-tillage system? Rev. Bras. Ciênc. Solo 2016, 40. [Google Scholar] [CrossRef]
- Alva, A.K.; Sumner, M.E. Amelioration of acid soil infertility by phosphogypsum. Plant Soil 1990, 128, 127–134. [Google Scholar] [CrossRef]
- Pavan, M.A.; Bingham, F.T.; Peryea, F.J. Influence of calcium and magnesium salts on acid soil chemistry and calcium nutrition of Apple. Soil Sci. Soc. Am. J. 1987, 51, 1526–1530. [Google Scholar] [CrossRef]
- Smith, C.; Peoples, M.; Keerthisinghe, G.; James, T.; Garden, D.; Tuomi, S. Effect of surface applications of lime, gypsum and phosphogypsum on the alleviating of surface and subsurface acidity in a soil under pasture. Soil Res. 1994, 32, 995–1008. [Google Scholar] [CrossRef]
- Carvalho, M.C.S.; van Raij, B. Calcium sulphate, phosphogypsum and calcium carbonate in the amelioration of acid subsoils for root growth. Plant Soil 1997, 192, 37–48. [Google Scholar] [CrossRef]
- Mora, M.L.; Schnettler, B.; Demanet, R. Effect of liming and gypsum on soil chemistry, yield, and mineral composition of ryegrass grown in an acidic Andisol. Commun. Soil Sci. Plant Anal. 1999, 30, 1251–1266. [Google Scholar] [CrossRef]
- Reyes-Díaz, M.; Meriño-Gergichevich, C.; Alarcón, E.; Alberdi, M.; Horst, W.J. Calcium sulfate ameliorates the effect of aluminum toxicity differentially in genotypes of highbush blueberry (Vaccinium corymbosum L.). J. Soil Sci. Plant Nutr. 2011, 11, 59–78. [Google Scholar] [CrossRef]
- Syed-Omar, S.R.; Shamsuddin, Z.H.; Zuraidah, J.Y.; Wynne, J.C.; Elkan, G.H. Use of Lime, Gypsum and Their Combinations to Improve Iodulation and Yield of Groundnut in an Acidic Soil. In Plant-Soil Interactions at Low pH, Proceedings of the Second International Symposium on Plant-Soil Interactions at Low pH, Beckley, VA, USA, 24–29 June 1990; Wright, R.J., Baligar, V.C., Murrmann, R.P., Eds.; Springer: Dordrecht, The Netherlands, 1991; pp. 275–280. [Google Scholar]
- Tirado-Corbalá, R.; Slater, B.K.; Dick, W.A.; Barker, D. Alfalfa responses to gypsum application measured using undisturbed soil columns. Plants 2017, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Farina, M.; Channon, P. Acid-subsoil amelioration: II. Gypsum effects on growth and subsoil chemical properties. Soil Sci. Soc. Am. J. 1988, 52, 175–180. [Google Scholar] [CrossRef]
- Kinraide, T.B.; Pedler, J.F.; Parker, D.R. Relative effectiveness of calcium and magnesium in the alleviation of rhizotoxicity in wheat induced by copper, zinc, aluminum, sodium, and low pH. Plant Soil 2004, 259, 201–208. [Google Scholar] [CrossRef]
- De Jesus, L.R.; Batista, B.L.; da Silva Lobato, A.K. Silicon reduces aluminum accumulation and mitigates toxic effects in cowpea plants. Acta Physiol. Plant. 2017, 39, 138. [Google Scholar] [CrossRef]
- Singh, V.P.; Tripathi, D.K.; Kumar, D.; Chauhan, D.K. Influence of exogenous silicon addition on aluminium tolerance in rice seedlings. Biol. Trace Elem. Res. 2011, 144, 1260–1274. [Google Scholar] [CrossRef] [PubMed]
- Manson, A.; Findlay, N. Agricultural Uses of Lime and Gypsum; KZN Department of Agriculture & Rural Development, Soil Fertility Research, Analytical Services: Cedara, South Africa, 2015; pp. 1–3.
- Mora, M.L.; Cartes, P.; Demanet, R.; Cornforth, I.S. Effects of lime and gypsum on pasture growth and composition on an acid Andisol in Chile, South America. Commun. Soil Sci. Plant Anal. 2002, 33, 2069–2081. [Google Scholar] [CrossRef]
- Rashad, A.M. Phosphogypsum as a construction material. J. Clean. Prod. 2017, 166, 732–743. [Google Scholar] [CrossRef]
- Campbell, C.G.; Garrido, F.; Illera, V.; García-González, M.T. Transport of Cd, Cu and Pb in an acid soil amended with phosphogypsum, sugar foam and phosphoric rock. Appl. Geochem. 2006, 21, 1030–1043. [Google Scholar] [CrossRef]
- Toma, M.; Saigusa, M. Effects of phosphogypsum on amelioration of strongly acid nonallophanic Andosols. Plant Soil 1997, 192, 49–55. [Google Scholar] [CrossRef]
- Garrido, F.; Illera, V.; Vizcayno, C.; García-González, M.T. Evaluation of industrial by-products as soil acidity amendments: Chemical and mineralogical implications. Eur. J. Soil Sci. 2003, 54, 411–422. [Google Scholar] [CrossRef]
- Mays, D.A.; Mortvedt, J.J. Crop response to soil applications of phosphogypsum. J. Environ. Qual. 1986, 15, 78–81. [Google Scholar] [CrossRef]
- Korcak, R.F. Fluidized bed material applied at disposal levels: Effects on an apple orchard. J. Environ. Qual. 1988, 17, 469–473. [Google Scholar] [CrossRef]
- Tuna, A.L.; Kaya, C.; Ashraf, M.; Altunlu, H.; Yokas, I.; Yagmur, B. The effects of calcium sulphate on growth, membrane stability and nutrient uptake of tomato plants grown under salt stress. Environ. Exp. Bot. 2007, 59, 173–178. [Google Scholar] [CrossRef]
- Toma, M.; Saigusa, M.; Qafoku, N.; Sumner, M. Effects of gypsum on amelioration of subsoil acidity in Andisols. J. Integr. Field Sci. 2005, 2, 69–71. [Google Scholar]
- Shamshuddin, J.; Fauziah, I.C.; Sharifuddin, H.A.H. Effects of limestone and gypsum application to a Malaysian ultisol on soil solution composition and yields of maize and groundnut. Plant Soil 1991, 134, 45–52. [Google Scholar] [CrossRef]
- Sanderson, K.R.; Eaton, L.J. Gypsum-an alternative to chemical fertilizers in lowbush blueberry production. Small Fruits Rev. 2004, 3, 57–71. [Google Scholar] [CrossRef]
- Korcak, R.F. Short-term response of blueberry to elevated soil calcium. J. Small Fruit Vitic. 1993, 1, 9–21. [Google Scholar] [CrossRef]
- Rengel, Z.; Bose, J.; Chen, Q.; Tripathi, B.N. Magnesium alleviates plant toxicity of aluminium and heavy metals. Crop. Pasture Sci. 2015, 66, 1298–1307. [Google Scholar] [CrossRef]
- Bose, J.; Babourina, O.; Rengel, Z. Role of magnesium in alleviation of aluminium toxicity in plants. J. Exp. Bot. 2011, 62, 2251–2264. [Google Scholar] [CrossRef] [PubMed]
- Grauer, U.E.; Horst, W.J. Modeling cation amelioration of aluminum phytotoxicity. Soil Sci. Soc. Am. J. 1992, 56, 166–172. [Google Scholar] [CrossRef]
- Bose, J.; Babourina, O.; Shabala, S.; Rengel, Z. Low-pH and aluminum resistance in Arabidopsis correlates with high cytosolic magnesium content and increased magnesium uptake by plant roots. Plant Cell Physiol. 2013, 54, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Palmgren, M.G. Plant plasmamembrane H+-ATPases: Powerhouses for nutrient uptake. Annu. Rev. Plant Phys. 2001, 52, 817–845. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Shimazaki, K.I. Blue light activates the plasma membrane H(+)-ATPase by phosphorylation of the C-terminus in stomatal guard cells. EMBO J. 1999, 18, 5548–5558. [Google Scholar] [CrossRef] [PubMed]
- Rober-Kleber, N.; Albrechtová, J.T.P.; Fleig, S.; Huck, N.; Michalke, W.; Wagner, E.; Speth, V.; Neuhaus, G.; Fischer-Iglesias, C. Plasma membrane H+-ATPase is involved in auxin-mediated cell elongation during wheat embryo development. Plant Physiol. 2003, 131, 1302–1312. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.L.; You, J.F.; Li, Y.Y.; Wu, P.; Zheng, S.J. Magnesium enhances aluminum-induced citrate secretion in rice bean roots (Vigna umbellata) by restoring plasma membrane H+-ATPase activity. Plant Cell Physiol. 2007, 48, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Kan, Q.; Wang, P.; Yu, W.; Yu, Y.; Zhao, Y.; Yu, Y.; Li, K.; Chen, L. Phosphorylation and interaction with the 14-3-3 protein of the plasma membrane H+-ATPase are involved in the regulation of magnesium-mediated increases in aluminum-induced citrate exudation in broad bean (Vicia faba. L). Plant Cell Physiol. 2015, 56, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Kellermayer, R.; Aiello, D.P.; Miseta, A.; Bedwell, D.M. Extracellular Ca2+ sensing contributes to excess Ca2+ accumulation and vacuolar fragmentation in a PMR1Δ mutant of S. Cerevisiae. J. Cell Sci. 2003, 116, 1637–1646. [Google Scholar] [CrossRef] [PubMed]
- Brüggemann, L.I.; Pottosin, I.I.; Schönknecht, G. Cytoplasmic magnesium regulates the fast activating vacuolar cation channel. J. Exp. Bot. 1999, 50, 1547–1552. [Google Scholar] [CrossRef]
- Deng, W.; Luo, K.; Li, D.; Zheng, X.; Wei, X.; Smith, W.; Thammina, C.; Lu, L.; Li, Y.; Pei, Y. Overexpression of an Arabidopsis magnesium transport gene, AtMGT1, in Nicotiana benthamiana confers Al tolerance. J. Exp. Bot. 2006, 57, 4235–4243. [Google Scholar] [CrossRef] [PubMed]
- Razaq, M.; Zhang, P.; Shen, H.-L.; Salahuddin. Influence of nitrogen and phosphorous on the growth and root morphology of Acer mono. PLoS ONE 2017, 12, e0171321. [Google Scholar] [CrossRef] [PubMed]
- Tariq, A.; Pan, K.; Olatunji, O.A.; Graciano, C.; Li, Z.; Sun, F.; Sun, X.; Song, D.; Chen, W.; Zhang, A.; et al. Phosphorous application improves drought tolerance of Phoebe Zhennan. Front. Plant Sci. 2017, 8, 1561. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Keltjens, W.G. Interaction between aluminium and phosphorus in sorghum plants. Plant Soil 1990, 124, 25–32. [Google Scholar] [CrossRef]
- Zheng, S.J.; Yang, J.L.; He, Y.F.; Yu, X.H.; Zhang, L.; You, J.F.; Shen, R.F.; Matsumoto, H. Immobilization of aluminum with phosphorus in roots is associated with high aluminum resistance in buckwheat. Plant Physiol. 2005, 138, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.T. Phosphorus enhances aluminium tolerance in both aluminium-tolerant and aluminium-sensitive wheat seedlings. S. Afr. J. Plant Soil 2013, 30, 13–21. [Google Scholar] [CrossRef]
- Chen, R.F.; Zhang, F.L.; Zhang, Q.M.; Sun, Q.B.; Dong, X.Y.; Shen, R.F. Aluminium-phosphorus interactions in plants growing on acid soils: Does phosphorus always alleviate aluminium toxicity? J. Sci. Food Agric. 2012, 92, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Ch’ng, H.Y.; Ahmed, O.H.; Majid, N.M.A. Improving phosphorus availability in an acid soil using organic amendments produced from agroindustrial wastes. Sci. World J. 2014, 2014, 506356. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, X.; Liao, H. Genetic improvement for phosphorus efficiency in soybean: A radical approach. Ann. Bot. 2010, 106, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Scott, N.M. Sulphur in Soils and Plants. In Soil Organic Matter and Biological Activity; Vaughan, D., Malcolm, R.E., Eds.; Springer: Dordrecht, The Netherlands, 1985; pp. 379–401. [Google Scholar]
- Dixit, G.; Singh, A.P.; Kumar, A.; Mishra, S.; Dwivedi, S.; Kumar, S.; Trivedi, P.K.; Pandey, V.; Tripathi, R.D. Reduced arsenic accumulation in rice (Oryza sativa L.) shoot involves sulfur mediated improved thiol metabolism, antioxidant system and altered arsenic transporters. Plant Physiol. Biochem. 2016, 99, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Saifullah; Khan, M.N.; Iqbal, M.; Naeem, A.; Bibi, S.; Waraich, E.A.; Dahlawi, S. Elemental sulfur improves growth and phytoremediative ability of wheat grown in lead-contaminated calcareous soil. Int. J. Phytoremediat. 2016, 18, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tan, Z.-Q.; Hu, L.-Y.; Wang, S.-H.; Luo, J.-P.; Jones, R.L. Hydrogen sulfide alleviates aluminum toxicity in germinating wheat seedlings. J. Integr. Plant Biol. 2010, 52, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Dawood, M.; Cao, F.; Jahangir, M.M.; Zhang, G.; Wu, F. Alleviation of aluminum toxicity by hydrogen sulfide is related to elevated ATPase, and suppressed aluminum uptake and oxidative stress in barley. J. Hazard. Mater. 2012, 209–210, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Qian, P.; Sun, R.; Ali, B.; Gill, R.A.; Xu, L.; Zhou, W. Effects of hydrogen sulfide on growth, antioxidative capacity, and ultrastructural changes in oilseed rape seedlings under aluminum toxicity. J. Plant Growth Regul. 2014, 33, 526–538. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, C.-Y.; Liu, J.; Song, R.; Du, Y.-X.; Li, J.-Z.; Sun, H.-Z.; Duan, G.-L.; Zhao, Q.-Z. Influence of sulfur on transcription of genes involved in arsenic accumulation in rice grains. Plant Mol. Biol. Rep. 2016, 34, 556–565. [Google Scholar] [CrossRef]
- Skwierawska, M.; Zawartka, L.; Zawadzki, B. The effect of different rates and forms of sulphur applied on changes of soil agrochemical properties. Plant Soil Environ. 2008, 54, 171–177. [Google Scholar] [CrossRef]
- Hossain, A.K.M.Z.; Hossain, M.A.; Koyama, H.; Hara, T. Effects of aluminum and boron supply on growth of seedlings among 15 cultivars of wheat (Triticum aestivum L.) grown in Bangladesh. Soil Sci. Plant Nutr. 2004, 50, 189–195. [Google Scholar] [CrossRef]
- Riaz, M.; Wu, X.; Yan, L.; Hussain, S.; Aziz, O.; Shah, A.; Jiang, C. Boron supply alleviates Al-induced inhibition of root elongation and physiological characteristics in rapeseed (Brassica napus L.). J. Plant Interact. 2018, 13, 270–276. [Google Scholar] [CrossRef]
- Gupta, U.C.; Jame, Y.W.; Campbell, C.A.; Leyshon, A.J.; Nicholaichuk, W. Boron toxicity and deficiency: A review. Can. J. Soil Sci. 1985, 65, 381–409. [Google Scholar] [CrossRef]
- Epstein, E. The anomaly of silicon in plant biology. Proc. Natl. Acad. Sci. USA 1994, 91, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.A.; Dietz, K.-J. Silicon as versatile player in plant and human biology: Overlooked and poorly understood. Front. Plant Sci. 2015, 6, 994. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.A.; Saqib, Z.A.; Akhtar, J.; Bakhat, H.F.; Pasala, R.-K.; Dietz, K.-J. Protective role of silicon (Si) against combined stress of salinity and boron (B) toxicity by improving antioxidant enzymes activity in rice. Silicon 2015, 1–5. [Google Scholar] [CrossRef]
- Ma, J.F. Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci. Plant Nutr. 2004, 50, 11–18. [Google Scholar] [CrossRef]
- Qian, L.; Chen, B.; Chen, M. Novel alleviation mechanisms of aluminum phytotoxicity via released biosilicon from rice straw-derived biochars. Sci. Rep. 2016, 6, 29346. [Google Scholar] [CrossRef] [PubMed]
- Kopittke, P.M.; Gianoncelli, A.; Kourousias, G.; Green, K.; McKenna, B.A. Alleviation of Al toxicity by Si is associated with the formation of Al-Si complexes in root tissues of sorghum. Front. Plant Sci. 2017, 8, 2189. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-Y.; Wang, N.; Xu, R.-K.; Tiwari, D. Potential of industrial byproducts in ameliorating acidity and aluminum toxicity of soils under tea plantation. Pedosphere 2010, 20, 645–654. [Google Scholar] [CrossRef]
- An, Y.; Zhou, P.; Xiao, Q.; Shi, D. Effects of foliar application of organic acids on alleviation of aluminum toxicity in alfalfa. J. Plant Nutr. Soil Sci. 2014, 177, 421–430. [Google Scholar] [CrossRef]
- Wang, Q.; Nian, F.; Zhao, L.; Li, F.; Yang, H.; Yang, Y. Exogenous indole-3-acetic acid could reduce the accumulation of aluminum in root apex of wheat (Triticum aestivum L.) under Al stress. J. Soil Sci. Plant Nutr. 2013, 13, 534–543. [Google Scholar] [CrossRef]
- Wang, S.; Yuan, S.; Su, L.; Lv, A.; Zhou, P.; An, Y. Aluminum toxicity in alfalfa (Medicago sativa) is alleviated by exogenous foliar IAA inducing reduction of Al accumulation in cell wall. Environ. Exp. Bot. 2017, 139, 1–13. [Google Scholar] [CrossRef]
- Pandey, P.; Srivastava, R.K.; Dubey, R.S. Salicylic acid alleviates aluminum toxicity in rice seedlings better than magnesium and calcium by reducing aluminum uptake, suppressing oxidative damage and increasing antioxidative defense. Ecotoxicology 2013, 22, 656–670. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Song, F.; Zhu, X.; You, J.; Yang, Z.; Li, X. Salicylic acid alleviates aluminum toxicity in soybean roots through modulation of reactive oxygen species metabolism. Front. Chem. 2017, 5, 96. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Jin, C.; Sun, C.; Wang, J.; Ye, Y.; Zhou, W.; Lu, L.; Lin, X. Inhibition of ethylene production by putrescine alleviates aluminium-induced root inhibition in wheat plants. Sci. Rep. 2016, 6, 18888. [Google Scholar] [CrossRef] [PubMed]
- Kochian, L.V.; Piñeros, M.A.; Hoekenga, O.A. The physiology, genetics and molecular biology of plant aluminum resistance and toxicity. Plant Soil 2005, 274, 175–195. [Google Scholar] [CrossRef]
- Yang, Z.-M.; Wang, J.; Wang, S.-H.; Xu, L.-L. Salicylic acid-induced aluminum tolerance by modulation of citrate efflux from roots of Cassia tora L. Planta 2003, 217, 168–174. [Google Scholar] [PubMed]
- Surapu, V.; Ediga, A.; Meriga, B. Salicylic acid alleviates aluminum toxicity in tomato seedlings (Lycopersicum esculentum Mill.) through activation of antioxidant defense system and proline biosynthesis. Adv. Biosci. Biotechnol. 2014, 5, 777. [Google Scholar] [CrossRef]
- Rouphael, Y.; Cardarelli, M.; Colla, G. Role of arbuscular mycorrhizal fungi in alleviating the adverse effects of acidity and aluminium toxicity in zucchini squash. Sci. Hortic. 2015, 188, 97–105. [Google Scholar] [CrossRef]
- Rufyikiri, G.; Declerck, S.; Dufey, J.E.; Delvaux, B. Arbuscular mycorrhizal fungi might alleviate aluminium toxicity in banana plants. New Phytol. 2000, 148, 343–352. [Google Scholar] [CrossRef]
| Country | Area/Region/Location | Nature/Type of Soil | Soil Depth (cm) | Range of pH | No. of Samples | Range of Al Concentration (g/kg) | Mean | References |
|---|---|---|---|---|---|---|---|---|
| Bangladesh | Dinajpur, Rangpur, Bangladesh | Paddy soil; sandy loam, non-calcareous, acidic-alluvial | 0–15, 15–30 | 4.8–5.4 | 04 | 0.024–0.059 | 0.043 | [43] |
| Hill of Chittagong University, Bangladesh | Hill topsoil; surface, subsurface | 0–12 | 4.4–5.5 | 45 | 0.036–0.058 | 0.048 | [44] | |
| Brazil | Cantareira State Park, Brazil | Forest back-ground | 0–20, 20–40 | 3.3–5.7 | 11 | 17.1–71.8 | 41.1 | [45] |
| China | Guizhou province, China | Yellow-brown | 0–30 | 3.5–4.8 | 13 | 65.2–128.5 | 106.4 | [46] |
| Sichuan, Zhejiang, and Jiangsu, Southeast China | Original, bulk and rhizosphere soil | 0–20 | 3.4–5.9 | 18 | 0.18–0.58 | 0.37 | [47] | |
| Central and Southwest China | Yellow sandstone, red earth, and Pleistocene deposits | 0–20 | 3.5–5.7 | 12 | 75.5–108.3 | 89.2 | [48] | |
| Canada | Northwestern Alberta, eastern and western Canada | Podzolic, luvisoli, gleysolic, subsoil | -- -- | 3.5–5.4 | 35 | 0.015–0.027 | 0.069 | [49] |
| Czech Republic | Jizera Mountains area, Czech Republic | Horizon; Organic-fragmented organic-humified organic-mineral mineral | 0–12.2 | 3.8–4.2 | 491 | 0.0057–0.017 | 0.0097 | [50] |
| France | Vosges Mountains area, North Eastern France | Acid brown, podzolic | 0–220 | 3.7–4.6 | 8 | 0.0003–0.047 | 0.0173 | [51] |
| India | Bihar, India | Fine mixed, fine loamy, sandy mixed | 0–150 | 4.4–7.0 | 13 | 0.0003–0.0051 | 0.0023 | [52] |
| Ireland Republic | South-eastern region, Ireland | Grassland, tillage, forest, peat | 0–10 | 5.3–5.9 | 295 | 0.0–89.1 | 45.6 | [53] |
| Japan | Hokkaido, Tohoku, Kanto, Kinki, Hokuriku-Chubu, Chugoku, Shikoku, Kyushu, and Okinawa | Agricultural soil; paddy/upland field | 0–15 | -- -- | 180 | 33.0–117.0 | 79.0 | [54] |
| Korea Republic | Osan, Korea | Sandy clay loam | -- -- | 4.0–7.1 | -- -- | 0.384–1.825 | 0.836 | [55] |
| Seoul, Ulsan, Hongchon, Korea | Forest; urban and industrial areas | 0–15 | 4.1–4.3 | -- -- | -- -- | -- -- | [56] | |
| New Zealand | North Canterbury, Central Canterbury, Central Otago | Stony brown, brown, dense brown hill soil | 0–15 | 4.9–6.7 | 14 | 0.0005–0.0174 | 0.0057 | [57] |
| USA | Palouse Conservation Field Station (PCFS) located near Pullman, WA, USA | Agricultural soils; silt loam | 0–30 | 4.7–6.3 | 80 | 0.034–0.055 | 0.0196 | [58] |
| Crawford County, OH, USA | Subsoil, acid soil | 0–110 | 4.5–7.5 | 99 | 0–0.25 | -- -- | [59] |
| Index of Toxicity | Sensitivity/Time of Al Exposure | Impacts of Al Toxicity | Conditions of Experiment | Aim of Assessment | References |
|---|---|---|---|---|---|
| Grain yield | Low/LD | Al-induced delay flowering; significantly reduced grain yield in sensitive cultivar | NS | FS | [76] |
| Biomass production | Moderate/LD | Reduced dry matter yield | NS | FS | [77] |
| Nutrient imbalance | Moderate/SD | Resulted imbalance of macronutrients including Mg, K and P | NS | TS | [78] |
| Survival | Low/LD | Resulted dry weight is the most sensitive tolerance index while survival is considered as the most cost-effective indicator of tolerance | SC | TS | [79] |
| Root growth | High/SD | Root growth was significantly inhibited by toxic Al ions in acid soil | SC | TS | [80] |
| Change of root system | High/LD | Indicated potential toxicity problem; root tips and lateral roots become more stubby, turned brown, and inhibited fine brunching | SC | TA | [81] |
| Rapture/cracks of root | High/SD | Induced rhizodermal cracks on roots after exposures to Al (11 µM) | NS | TA | [82] |
| Plasma membrane (PM) | High/SD | Al3+ ion attached to PM; cells become more leaky and rigid | NS | TS | [83] |
| Cell division and elongation | High/SD | Al resulted the disordered the arrangement of the cells, deformed cell shapes, altered cell structure, and the shorter of the meristematic zone of the root tips | NS | FS, AR | [23] |
| Interference with enzymes | Moderate/SD | Down regulated peroxidase and chitinase isoforms in root tips | NS | FS, TS | [84] |
| Organic acid exudation | Low/LD | Organic acids (oxalate, citrate) enhanced plant growth and adaptation following Al stress to acid soil | SC | FS, AR | [85] |
| Callose formation | High/SD | Resulted a link between callose formation and Al-induced inhibition of root growth | NS | TA, TS | [86] |
| Auxin transport | High/SD | Al toxicity inhibited (IAA) transportation from the shoot base to root tip; though exogenous application of IAA alleviated Al stress | NS | FS, AR | [23] |
| Al content in pectin | High/SD | Aggravated Al3+ toxicity due to accumulation of more Al in pectin | NS | TA | [87] |
| Sensitivity Threshold | ||
|---|---|---|
| High Sensitive (Indicator Plants) | Moderate Sensitive | Low Sensitive (Resistant/Tolerant Plants) |
| Hordeum vulgare L. (barley) | Rapanus sativus L. (radish) | Fagopyrum esculentum Moench (buckwheat) |
| Triticum aestivum L. (wheat) | Sorghum bicolour L. Moench (sorghum) | Camellia sinensis L. (tea) |
| Triticum durum Desf. (durum wheat) | ||
| Lactuca sativa L. (lettuce) | Capitata var. alba L. (cabbage) | Oryza sativa L. (rice) |
| Beta vulragis L. (beet) | ||
| Phleum pretense L. (timothy-grass) | Avena sativa L. (oat) | Zea mays L. (maize) |
| Glycine max Merr. (soybean) | ||
| Pisum sativum L. (pea) | Medicago sativa L. (alfalfa) | Brassica rapa L. (turnip) |
| Phaseolus vulgaris L. (common bean) | ||
| Pachyrhizus ahipa Wedd. (Parodi) | Secale cereale L. (rye) | Agrostis gigantean Roth. (redtop) |
| Amendments | Plant Species | Dose (t·ha−1) and Duration | Outcomes | References |
|---|---|---|---|---|
| Phosphogypsum (PG) | Zea mays L., Triticum aestivum L., Glycine max L. | 12.0, (0–8.0 cm), 180 days | Increased Ca concentrations in all crops; enhanced root growth; provided nutrients to the soil and reduce Al3+ activity. | [123] |
| Medicago sativa L. cv. Hunter River and Glycine max L. Merr. cv. Lee | 2.0, (60–80 cm); 50 days | Ameliorated subsoil acidity; Increased Ca in soil solution; Reduced exchangeable Al, and increased in crop growth. | [124] | |
| Malus domestica Borkh. cv. Gala/MM 106 | 2.0, (40–60 cm), 90 days | Decreased exchangeable Al; increased Ca in topsoil as well as plant leaf; increase root density. | [125] | |
| Trifolium spp. | 2.5, (0–25 cm), 1.5 years | Reduced toxic Al concentration in soil solution and exchange site in subterranean clover-based pasture. | [126] | |
| Zea mays L. | 34.9% PG; (5 × 7 × 5 cm) | Reduced soil acidity; decreased Al activity of in the soil solution; increased root development. | [127] | |
| Gypsum (G) | Lolium perenne L. cv. Nui | 2.0, (0–20 cm) | Balanced nutrient elements; increased forage quality and yield; reduced exchangeable Al; increased soil pH. | [128] |
| Vaccinium corymbosum L. | 4.4, 60 days | Enhanced Ca and S contents; reduced Al concentration and lipid peroxidation in roots and leaves; Ameliorated Al toxicity in plant by gypsum. | [129] | |
| Arachis hypogaea L. | 1.0, 60 days | Reduced Al phytotoxicity; increased nodules, pods, and yield of groundnut. | [130] | |
| Medicago sativa L. | 22.2, (0–75 cm), 12 years | Provided Ca and S benefits as a source in nutrient-limiting soils; enhanced soil fertility and alfalfa growth. | [131] | |
| Zea mays L. | 11.0, (40–60 cm), 1 year | Improved root density; Increased Ca, Mg, and SO4-S; declined the level of exchangeable Al. | [132] |
| Plant Species | Dose and Duration of Al Exposure | Mineral Treatment | Plant Responses | References |
|---|---|---|---|---|
| Glycine max (L.) Merr. | 2 µM Al, 24 h | 500–2000 µM Ca, 3 days | Mitigated the inhibition of root growth and root hair formation during Al toxicity; enhanced protection against the deleterious effect of toxic Al in soybean. | [111] |
| Triticum aestivum L. | 20 µM Al, 28 days | 1600 µM Ca; 800 µM Mg, 7 days | Ca and Mg alleviated Al toxicity in wheat, respectively; decreased Al3+ activity at PM surface in root cell. | [112,133] |
| Glycine max (L.) Merr. | 100 mM Al, 72 h | 25 µM Mg, 3 days | Increased root growth; Alleviated Al rhizotoxicity during lateral root elongation. | [113] |
| Eucalyptus grandis × E. urophylla clones “G9”and “DH32-29” | 5 mM Al, 20 weeks | 200 µM P; 140 days | Al resistant G9 clone, enhanced more malate, oxalate, and citrate secretion in roots; P was involved in elemental Al fixation in roots, and restricted Al transport to the stems and leaves; Increased the activities of PEPC, CS enzymes. | [117] |
| Citrus grandis | 1 mM Al, 16 days | 0.5 mM S, 126 days | Reduced Al transport in roots, shoots and leaves; Decreased H2O2 production; Increased, P, Mg, Ca and RWC; Enhanced OAs in roots. | [116] |
| Citrus grandis | 1.2 mM Al, 18 weeks | 20 μM B, 126 days | Over 100 genes including GSTZ1, TRX-M4, CLM 19, IAA-amino acid hydrolase ILR1-4, GAG-POL were associated with B-induced alleviation of Al-toxicity. | [115] |
| Pisum sativum L. cv Zhongwan | 50 µM Al, 24 h | 50 µM B, 2 days | Increased chlorophyll and biomass; reduced chlorosis; reduced Al concentration in shoots; inhibited Al-binding in cell wall; reduced toxicity effect in roots and shoots. | [114] |
| Vigna unguicula L. | 10 mM Al, 16 days | 2.50 mM Si, 26 days | Increased SOD, CAT, APX and POX activities; Reduced Al contents of all tissues; Mitigated toxic effects of Al. | [134] |
| Oriza sativa L. | 50 μM Al, 7 days | 10 μM Si, 7 days | Restricted the uptake and transport of toxic Al in roots and leaves; maintained Mg and Zn at optimum levels; reduced cellular injury from Al toxicity. | [135] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, M.A.; Lee, S.-H.; Ji, H.C.; Kabir, A.H.; Jones, C.S.; Lee, K.-W. Importance of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants on Acidic Soils: Current Status and Opportunities. Int. J. Mol. Sci. 2018, 19, 3073. https://doi.org/10.3390/ijms19103073
Rahman MA, Lee S-H, Ji HC, Kabir AH, Jones CS, Lee K-W. Importance of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants on Acidic Soils: Current Status and Opportunities. International Journal of Molecular Sciences. 2018; 19(10):3073. https://doi.org/10.3390/ijms19103073
Chicago/Turabian StyleRahman, Md. Atikur, Sang-Hoon Lee, Hee Chung Ji, Ahmad Humayan Kabir, Chris Stephen Jones, and Ki-Won Lee. 2018. "Importance of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants on Acidic Soils: Current Status and Opportunities" International Journal of Molecular Sciences 19, no. 10: 3073. https://doi.org/10.3390/ijms19103073
APA StyleRahman, M. A., Lee, S.-H., Ji, H. C., Kabir, A. H., Jones, C. S., & Lee, K.-W. (2018). Importance of Mineral Nutrition for Mitigating Aluminum Toxicity in Plants on Acidic Soils: Current Status and Opportunities. International Journal of Molecular Sciences, 19(10), 3073. https://doi.org/10.3390/ijms19103073






