Influence of Botanical Origin and Chemical Composition on the Protective Effect against Oxidative Damage and the Capacity to Reduce In Vitro Bacterial Biofilms of Monofloral Honeys from the Andean Region of Ecuador
Abstract
:1. Introduction
2. Results and Discussion
2.1. Physicochemical Analyses
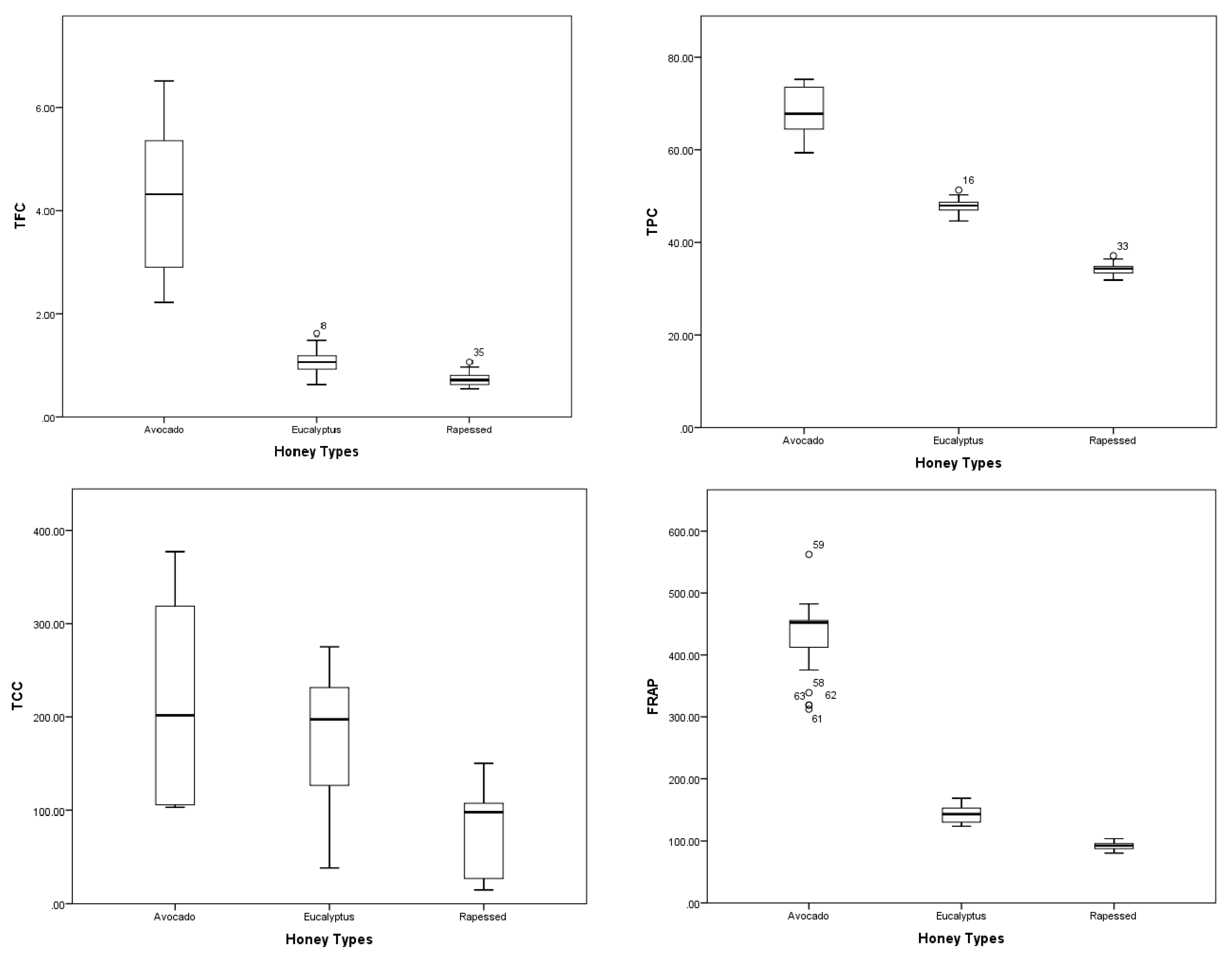
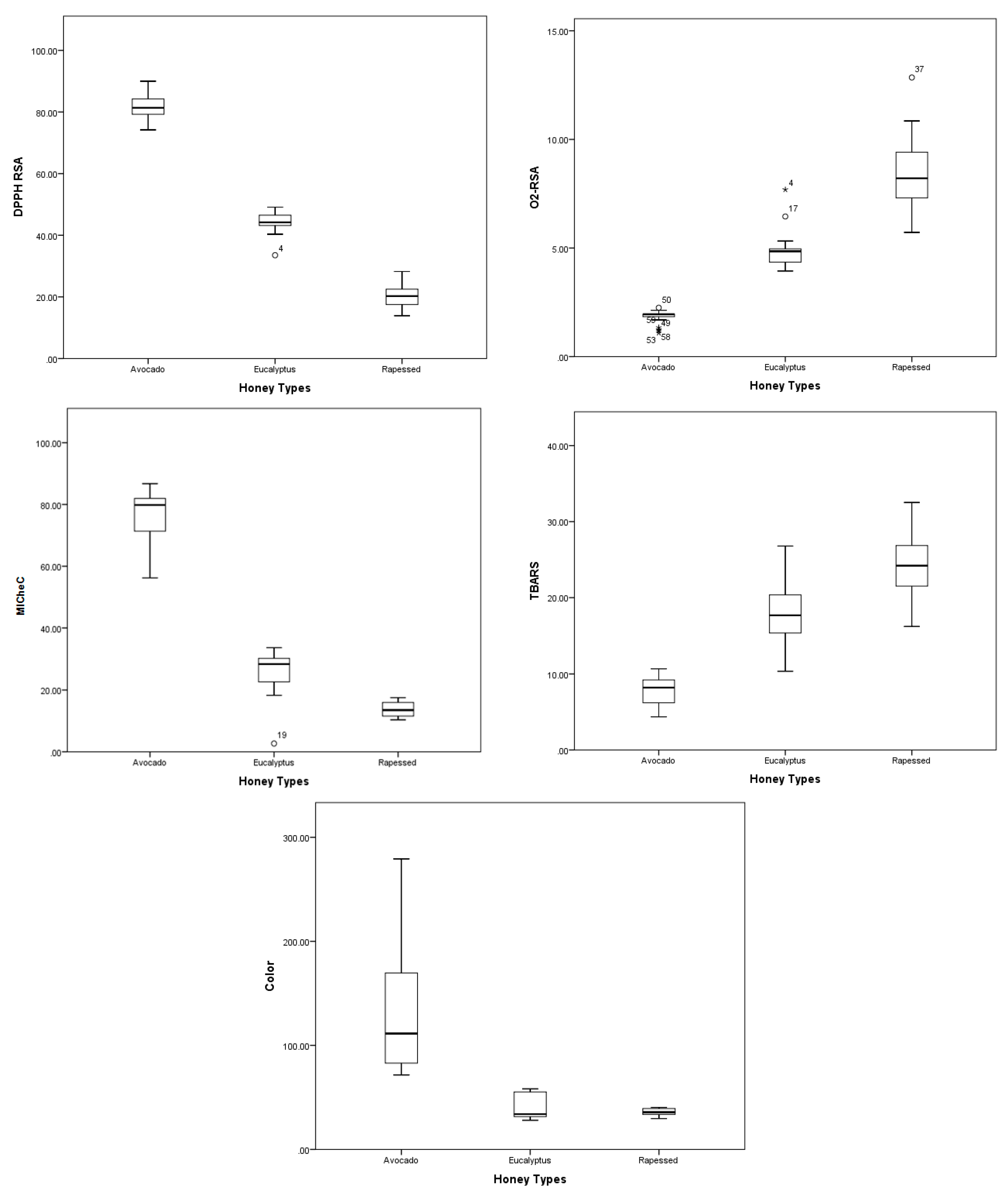
2.2. Chemical Composition and Total Antioxidant Capacity (TAC)
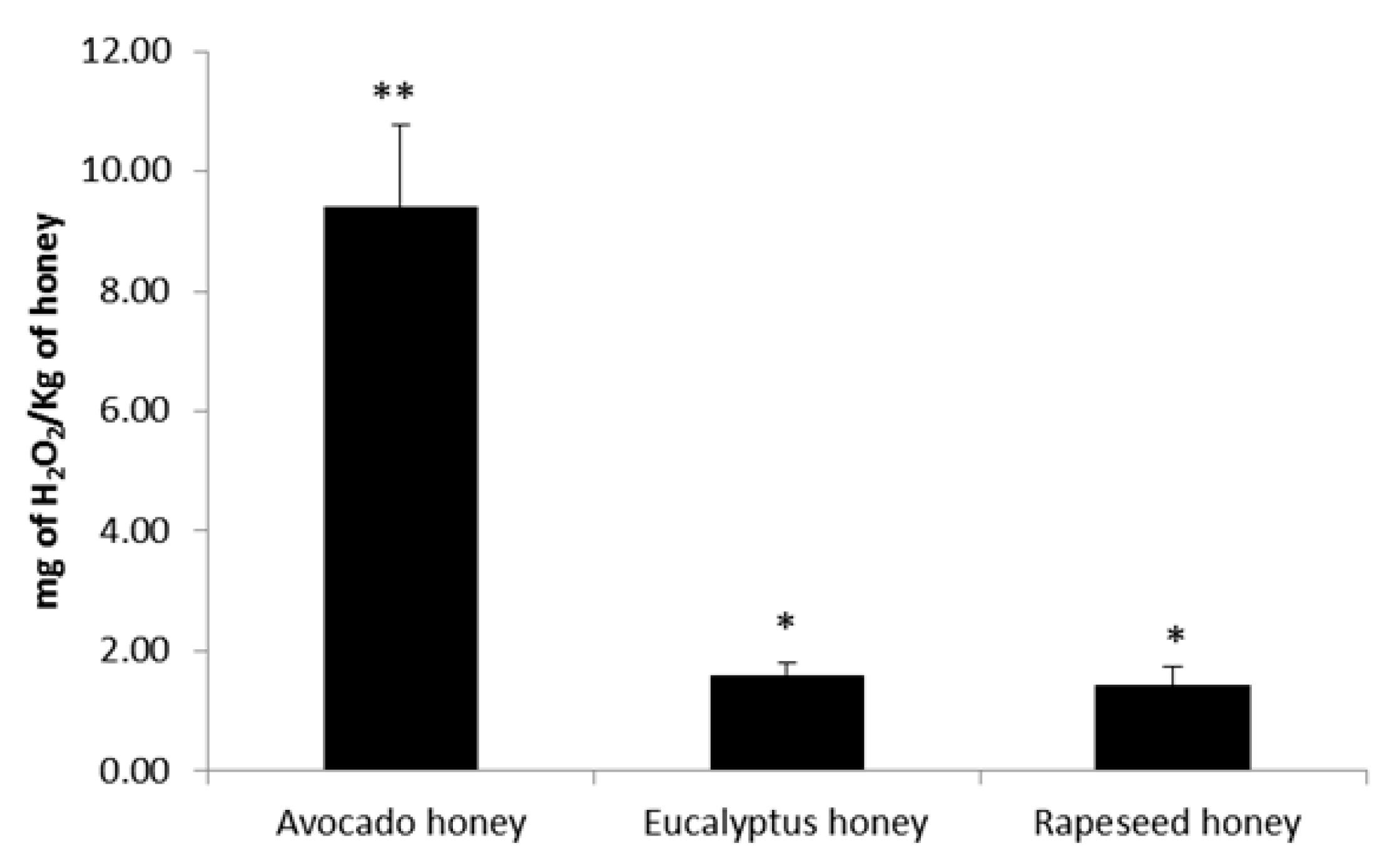
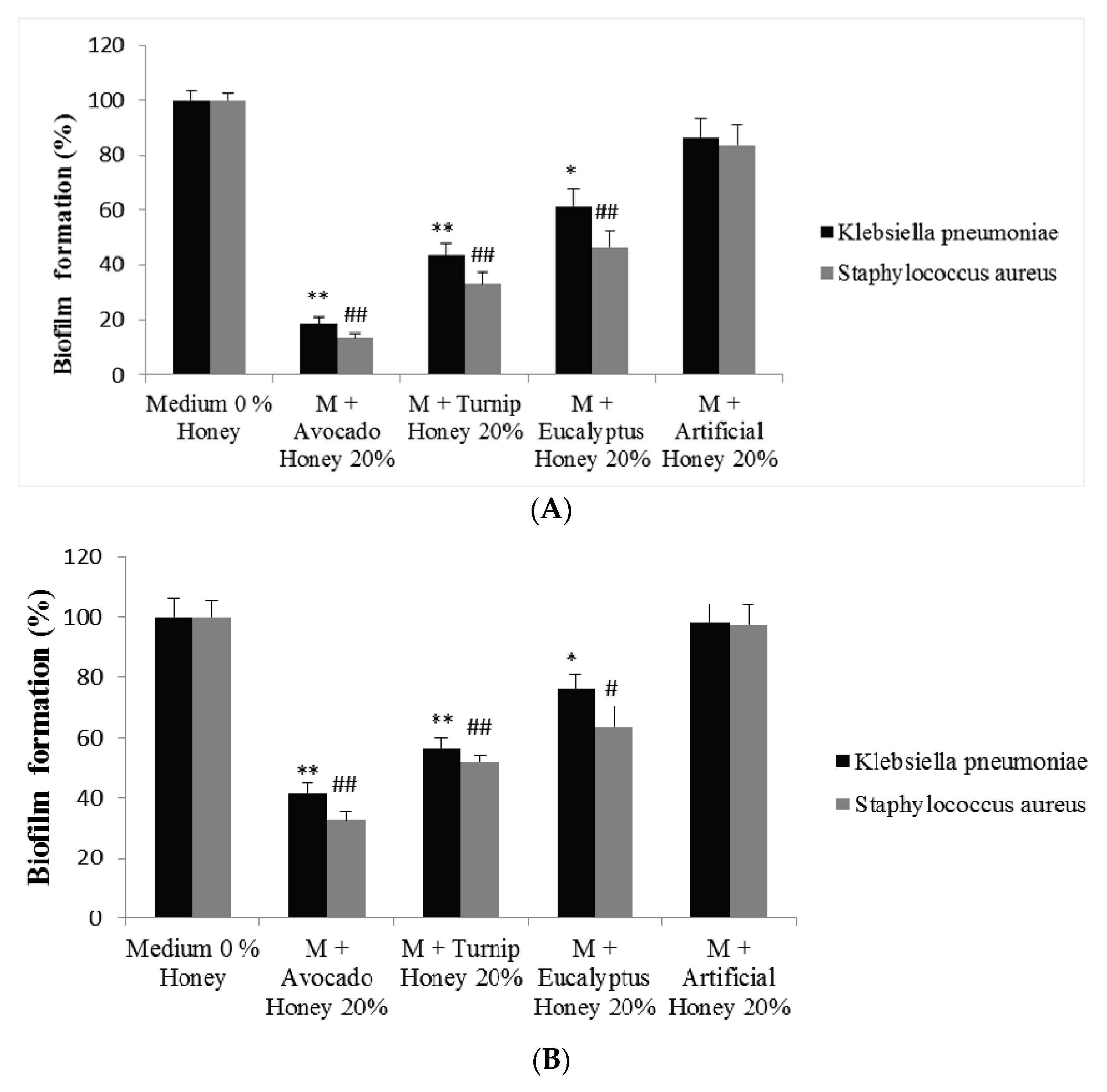
2.3. Effect of Honey to Inhibit Biofilm Formation and to Remove Preformed Biofilm in Staphylococcus aureus and Klebsiella pneumoniae Bacteria
3. Materials and Methods
3.1. Honey Samples
3.2. Physicochemical Analysis
3.3. Chemical Composition
3.4. Total Antioxidant Capacity (TAC) Assays
FRAP, DPPH, Metal Ions Chelating Capacity, and the Superoxide Radical (O2•−) Scavenging Activity
3.5. Protective Effects against Ghost Membrane Lipid Peroxidation
3.5.1. Blood Collection and Red Blood Cells (RBC) Ghost Membrane Preparation
3.5.2. Determination of Lipid Peroxidation in Ghost Membrane
3.6. Antimicrobial Activity
3.6.1. Hydrogen Peroxide Content in Honey Samples
3.6.2. Determination of the Ability of Honey to Inhibit Biofilm Formation and Remove Preformed Biofilm
3.7. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HMF | Hydroxymethylfurfural |
| TPC | Total phenolic content |
| TFC | Total flavonoid content |
| TCC | Total carotenoid content |
| TAC | Total antioxidant capacity |
| FRAP | Ferric reducing antioxidant power |
| O2•− RSA | Superoxide radical scavenging activity |
| TBARS | Thiobarbituric acid reactive substances |
| GAE | Gallic acid equivalents |
| CEq | Catechin equivalents |
| LE | Leucine equivalents |
| Prol | Proline |
| RBCs | Red blood cells |
References
- Alvarez-Suarez, J.M.; Giampieri, F.; Cordero, M.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Mazzoni, L.; Afrin, S.; Beltrán-Ayala, P.; Gonzales-Paramas, A.M.; Santos-Buelga, C.; et al. Activation of AMPK/Nrf2 signalling by Manuka honey protects human dermal fibroblasts against oxidative damage by improving antioxidant response and mitochondrial function promoting wound healing. J. Funct. Foods 2016, 25, 38–49. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; Battino, M. Honey as a source of dietary antioxidants: Structures, bioavailability and evidence of protective effects against human chronic diseases. Curr. Med. Chem. 2013, 20, 621–638. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R. Honey for wound care in the 21st century. J. Wound Care 2016, 25, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Scepankova, H.; Saraiva, J.A.; Estevinho, L.M. Honey Health Benefits and Uses in Medicine. In Bee Products—Chemical and Biological Properties; Alvarez-Suarez, J.M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 83–96. [Google Scholar]
- Santos-Buelga, C.; González-Paramás, A.M. Chemical Composition of Honey. In Bee Products—Chemical and Biological Properties; Alvarez-Suarez, J.M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 43–82. [Google Scholar]
- Acquarone, C.; Buera, P.; Elizalde, B. Pattern of pH and electrical conductivity upon honey dilution as a complementary tool for discriminating geographical origin of honeys. Food Chem. 2007, 101, 695–703. [Google Scholar] [CrossRef]
- Bentabol-Manzanares, A.; Hernández-García, Z.; Rodríguez-Galdón, B.; Rodríguez-Rodríguez, E.; Díaz-Romero, C. Physicochemical characteristics of minor monofloral honeys from Tenerife, Spain. LWT—Food Sci. Technol. 2014, 55, 572–578. [Google Scholar] [CrossRef]
- González-Miret, M.L.; Terrab, A.; Hernanz, D.; Fernández-Recamales, M.Á.; Heredia, F.J. Multivariate correlation between color and mineral composition of honeys and by their botanical origin. J. Agric. Food Chem. 2005, 53, 2574–2580. [Google Scholar] [CrossRef] [PubMed]
- Flores, M.S.R.; Escuredo Pérez, O.; Coello, M.C.S. Characterization of Eucalyptus globulus honeys produced in the eurosiberian area of the Iberian Peninsula. Int. J. Food Prop. 2014, 17, 2177–2191. [Google Scholar] [CrossRef]
- Persano Oddo, L.; Piro, R.; Bruneau, É.; Guyot-Declerck, C.; Ivanov, T.; Piskulová, J.; Flamini, C.; Lheritier, J.; Molot, M.; Rossmann, H.; et al. Main European unifloral honeys: Descriptive sheets. Apidologie 2004, 35, S38–S81. [Google Scholar] [CrossRef]
- Stawiarz, E. Pollen spectrum of rapeseed honey from the sandomierska upland area. J. Apic. Sci. 2008, 52, 83–91. [Google Scholar]
- Kędzierska-Matysek, M.; Florek, M.; Wolanciuk, A.; Skałecki, P. Effect of freezing and room temperatures storage for 18 months on quality of raw rapeseed honey (Brassica napus). J. Food Sci. Technol. 2016, 53, 3349–3355. [Google Scholar] [CrossRef] [PubMed]
- EU Council, Council directive 2001/11 O/EC of 20 December 2001 relating to honey. Off. J. Eur. Commun. 2002, 10, 47–52.
- Chirife, J.; Zamora, M.C.; Motto, A. The correlation between water activity and % moisture in honey: Fundamental aspects and application to Argentine honeys. J. Food Eng. 2006, 72, 287–292. [Google Scholar] [CrossRef]
- Terrab, A.; Heredia, F.J. Characterization of avocado (Persea americana Mill) honeys by their physicochemical characteristics. J. Sci. Food Agric. 2004, 84, 1801–1805. [Google Scholar] [CrossRef]
- Ortega-Bonilla, R.A.; Chito-Trujillo, D.M.; Suárez-Ramos, C.A. Physicochemical characteristics of commercial eucalyptus honeys from Southwest Casanare. Corpoica Cienc. Tecnol. Agropecu. 2016, 17, 73–80. [Google Scholar]
- Fallico, B.; Arena, E.; Verzera, A.; Zappalà, M. The European Food Legislation and its impact on honey sector. Accred. Qual. Assur. 2006, 11, 49–54. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Tulipani, S.; Díaz, D.; Estevez, Y.; Romandini, S.; Giampieri, F.; Damiani, E.; Astolfi, P.; Bompadre, S.; Battino, M. Antioxidant and antimicrobial capacity of several monofloral Cuban honeys and their correlation with color, polyphenol content and other chemical compounds. Food Chem. Toxicol. 2010, 48, 2490–2499. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.M.; Gauche, C.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. Honey: Chemical composition, stability and authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Ramos, O.Y.; Salomón, V.; Libonatti, C.; Cepeda, R.; Maldonado, L.; Basualdo, M. Effect of botanical and physicochemical composition of Argentinean honeys on the inhibitory action against food pathogens. LWT—Food Sci. Technol. 2018, 87, 457–463. [Google Scholar] [CrossRef]
- Cooper, R.A.; Molan, P.C.; Harding, K.G. The sensitivity to honey of Gram-positive cocci of clinical significance isolated from wounds. J. Appl. Microbiol. 2002, 93, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Gorjanović, S.Ž.; Alvarez-Suarez, J.M.; Novaković, M.M.; Pastor, F.T.; Pezo, L.; Battino, M.; Sužnjević, D.Ž. Comparative analysis of antioxidant activity of honey of different floral sources using recently developed polarographic and various spectrophotometric assays. J. Food Comp. Anal. 2013, 30, 13–18. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; Damiani, E.; Astolfi, P.; Fattorini, D.; Regoli, F.; Quiles, J.L.; Battino, M. Radical-scavenging activity, protective effect against lipid peroxidation and mineral contents of monofloral cuban honeys. Plant Foods Hum. Nutr. 2012, 67, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Henriques, A.; Jackson, S.; Cooper, R.; Burton, N. Free radical production and quenching in honeys with wound healing potential. J. Antimicrob. Chemother. 2006, 58, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Pérez, E.; Rodríguez-Malaver, A.J.; Vit, P. Antioxidant capacity of Venezuelan honey in wistar rat homogenates. J. Med. Food 2006, 9, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Visioli, F. Polyphenols and health: Moving beyond antioxidants. J. Berry Res. 2012, 2, 63–71. [Google Scholar]
- Niki, E. Antioxidant capacity: Which capacity and how to assess it? J. Berry Res. 2011, 1, 169–176. [Google Scholar]
- Morita, M.; Naito, Y.; Yoshikawa, T.; Niki, E. Antioxidant capacity of blueberry extracts: Peroxyl radical scavenging and inhibition of plasma lipid oxidation induced by multiple oxidants. J. Berry Res. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Aires, E.; Barreira, J.C.M.; Estevinho, L.M. Antioxidant activity of Portuguese honey samples: Different contributions of the entire honey and phenolic extract. Food Chem. 2009, 114, 1438–1443. [Google Scholar] [CrossRef]
- Gomes, S.; Dias, L.G.; Moreira, L.L.; Rodrigues, P.; Estevinho, L. Physicochemical, microbiological and antimicrobial properties of commercial honeys from Portugal. Food Chem. Toxicol. 2010, 48, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Cokcetin, N.N.; Pappalardo, M.; Campbell, L.T.; Brooks, P.; Carter, D.A.; Blair, S.E.; Harry, E.J. The antibacterial activity of Australian Leptospermum honey correlates with methylglyoxal levels. PLoS ONE 2016, 11, e0167780. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Brenciani, A.; Mazzoni, L.; Gasparrini, M.; González-Paramás, A.M.; Santos-Buelga, C.; Morroni, G.; Simoni, S.; Forbes-Hernández, T.Y.; et al. Apis mellifera vs. Melipona beecheii Cuban polifloral honeys: A comparison based on their physicochemical parameters, chemical composition and biological properties. LWT—Food Sci. Technol. 2018, 87, 272–279. [Google Scholar] [CrossRef]
- De Almeida-Muradian, L.B.; Stramm, K.M.; Horita, A.; Barth, O.M.; Da Silva de Freitas, A.; Estevinho, L.M. Comparative study of the physicochemical and palynological characteristics of honey from Melipona subnitida and Apis mellifera. Int. J. Food Sci. Technol. 2013, 48, 1698–1706. [Google Scholar] [CrossRef]
- Kwakman, P.H.S.; Zaat, S.A.J. Antibacterial components of honey. IUBMB Life 2012, 64, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Gauhe, A. Über ein glukoseoxydierendes Enzym in der Pharynxdrüse der Honigbiene. Z. Vgl. Physiol. 1940, 28, 211–253. [Google Scholar] [CrossRef]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Biol. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef] [PubMed]
- Weston, R.J. The contribution of catalase and other natural products to the antibacterial activity of honey: A review. Food Chem. 2000, 71, 235–239. [Google Scholar] [CrossRef]
- Li, D.; Wang, M.; Cheng, N.; Xue, X.; Wu, L.; Cao, W. A modified FOX-1 method for Micro-determination of hydrogen peroxide in honey samples. Food Chem. 2017, 237, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Turnbull, L.; Burke, C.M.; Liu, M.; Carter, D.A.; Schlothauer, R.C.; Whitchurch, C.B.; Harry, E.J. Manuka-type honeys can eradicate biofilms produced by Staphylococcus aureus strains with different biofilm-forming abilities. PeerJ 2014, 2, e326. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J.; Bohova, J.; Horniackova, M.; Klaudiny, J.; Majtan, V. Anti-biofilm effects of honey against wound pathogens Proteus mirabilis and Enterobacter cloacae. Phytother. Res. 2014, 28, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jagani, S.; Chelikani, R.; Kim, D.S. Effects of phenol and natural phenolic compounds on biofilm formation by Pseudomonas aeruginosa. Biofouling 2009, 25, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of Melissopalynology. Bee World 1978, 59, 139–157. [Google Scholar] [CrossRef]
- Von Der Ohe, W.; Oddo Livia, P.; Piana, M.L.; Morlot, M.; Martin, P. Harmonized methods of melissopalynology. Apidologie 2004, 35, 18–25. [Google Scholar] [CrossRef]
- IHC. Harmonised Methods of the International Honey Commission; Swiss Bee Research Centre: Bern, Switzerland, 2002. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Chang, C.-C.; Yang, M.-H.; Wen, H.-M.; Chern, J.-C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Doi, E.; Shibata, D.; Matoba, T. Modified colorimetric ninhydrin methods for peptidase assay. Anal. Biochem. 1981, 118, 173–184. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a measure of “Antioxidant Power”: The FRAP Assay. Anal Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Velázquez, E.; Tournier, H.A.; Mordujovich De Buschiazzo, P.; Saavedra, G.; Schinella, G.R. Antioxidant activity of Paraguayan plant extracts. Fitoterapia 2003, 74, 91–97. [Google Scholar] [CrossRef]
- Wang, B.-J.; Lien, Y.-H.; Yu, Z.-R. Supercritical fluid extractive fractionation—Study of the antioxidant activities of propolis. Food Chem. 2004, 86, 237–243. [Google Scholar] [CrossRef]
- Oyanagui, Y. Reevaluation of assay methods and establishment of kit for superoxide dismutase activity. Anal. Biochem. 1984, 142, 290–296. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; González-Paramás, A.M.; Damiani, E.; Astolfi, P.; Martinez-Sanchez, G.; Bompadre, S.; Quiles, J.L.; Santos-Buelga, C.; Battino, M. Phenolics from monofloral honeys protect human erythrocyte membranes against oxidative damage. Food Chem. Toxicol. 2012, 50, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [PubMed]
| Parameters | Monofloral Honey Types/Values | ||
|---|---|---|---|
| Avocado Honey | Eucalyptus Honey | Rapeseed Honey | |
| Physicochemical parameters | |||
| Colour (mm Pfund) | 137.29 ± 66.97 a | 39.87 ± 11.46 b | 37.09 ±3.32 b |
| Moisture (%) | 16.42 ± 2.53 a | 18.62 ± 1.84 b | 14.63 ± 2.74 c |
| pH | 5.23 ± 0.96 a | 4.01 ± 0.18 b | 3.96 ± 0.24 b |
| Hydroxymethylfurfural (mg/kg of honey) | 27.16 ± 30.43 a | 3.78 ± 3.48 b | 70.81 ± 7.86 c |
| Diastase index (ºGothe) | 47.65 ± 34.95 a | 32.92 ± 11.35 b | 13.71 ± 2.99 c |
| Electrical conductivity (mS/cm) | 1.31 ± 0.47 a | 0.39 ± 0.03 b | 0.20 ± 0.01 c |
| Ash content (%) | 1.05 ± 0.13 a | 0.25 ± 0.02 b | 0.11 ± 0.01 c |
| Bioactive compounds | |||
| Total phenolic content (mg GAE/100 g of honey) | 68.23 ± 5.79 a | 47.71 ± 1.71 b | 34.33 ± 0.895 c |
| Total flavonoid content (mg CEq/100 g of honey) | 4.25 ± 1.22 a | 1.09 ± 0.24 b | 0.72 ± 0.20 c |
| Total carotenoids content (mg βcarotE/kg of honey) | 2.24 ± 0.67 a | 1.60 ± 0.33 b | 0.76 ± 0.14 c |
| Total free amino acids content (mg LE/100 g of honey) | 83.46 ± 43.41 a | 14.07 ± 3.20 b | 10.53 ± 3.66 c |
| Total proline content (mg Prol/100 g of honey) | 1039.94 ± 53.31 a | 140.82 ± 40.71 b | 88.19 ± 18.53 c |
| Total antioxidant capacity (TAC) | |||
| FRAP (μmol TE/100 g of honey) | 425.35 ± 49.24 a | 142.97 ± 13.84 b | 92.05 ± 4.30 c |
| DPPH (μmol TE/100 g of honey) | 84.05 ± 5.16 a | 44.30 ± 5.33 b | 18.22 ± 3.19 c |
| O2•− Scavenging activity IC50 (mg/mL) | 1.82 ± 0.32 a | 4.90 ± 0.84 b | 8.42 ± 1.72 |
| Chelating metal ions capacity (%) | 76.90 ± 7.08 a | 26. 17 ± 6.92 b | 13.69 ± 2.35 c |
| TBARS assay IC50 (mg/mL) | 7.59 ± 1.85 a | 17.78 ± 3.97 b | 24.40 ± 4.98 c |
| Variables | Colour | TPC | TFC | TCC | FRAP | DPPH RSA | O2•− RSA | MICheC | TBARS |
|---|---|---|---|---|---|---|---|---|---|
| Colour | - | 0.770 ** | 0.860 ** | 0.378 * | 0.787 ** | 0.729 ** | −0.613 ** | 0.716 ** | −0.699 ** |
| TPC | - | - | 0.905 ** | 0.688 ** | 0.937 ** | 0.958 ** | −0.890 ** | 0.921 ** | −0.880 ** |
| TFC | - | - | - | 0.625 ** | 0.899 ** | 0.820 ** | −0.734 ** | 0.845 ** | −0.782 ** |
| TCC | - | - | - | - | 0.572 ** | 0.609 ** | −0.596 ** | 0.481 ** | −0.481 ** |
| FRAP | - | - | - | - | - | 0.932 ** | −0.827 ** | 0.943 ** | −0.847 ** |
| DPPH RSA | - | - | - | - | - | - | −0.899 ** | 0.944 ** | −0.884 ** |
| O2•− RSA | - | - | - | - | - | - | - | −0.837 ** | 0.840 ** |
| MICheA | - | - | - | - | - | - | - | - | −0.869 ** |
| TBARS | - | - | - | - | - | - | - | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Tenesaca, M.; Navarrete, E.S.; Iturralde, G.A.; Villacrés Granda, I.M.; Tejera, E.; Beltrán-Ayala, P.; Giampieri, F.; Battino, M.; Alvarez-Suarez, J.M. Influence of Botanical Origin and Chemical Composition on the Protective Effect against Oxidative Damage and the Capacity to Reduce In Vitro Bacterial Biofilms of Monofloral Honeys from the Andean Region of Ecuador. Int. J. Mol. Sci. 2018, 19, 45. https://doi.org/10.3390/ijms19010045
García-Tenesaca M, Navarrete ES, Iturralde GA, Villacrés Granda IM, Tejera E, Beltrán-Ayala P, Giampieri F, Battino M, Alvarez-Suarez JM. Influence of Botanical Origin and Chemical Composition on the Protective Effect against Oxidative Damage and the Capacity to Reduce In Vitro Bacterial Biofilms of Monofloral Honeys from the Andean Region of Ecuador. International Journal of Molecular Sciences. 2018; 19(1):45. https://doi.org/10.3390/ijms19010045
Chicago/Turabian StyleGarcía-Tenesaca, Marilyn, Eillen S. Navarrete, Gabriel A. Iturralde, Irina M. Villacrés Granda, Eduardo Tejera, Pablo Beltrán-Ayala, Francesca Giampieri, Maurizio Battino, and José M. Alvarez-Suarez. 2018. "Influence of Botanical Origin and Chemical Composition on the Protective Effect against Oxidative Damage and the Capacity to Reduce In Vitro Bacterial Biofilms of Monofloral Honeys from the Andean Region of Ecuador" International Journal of Molecular Sciences 19, no. 1: 45. https://doi.org/10.3390/ijms19010045
APA StyleGarcía-Tenesaca, M., Navarrete, E. S., Iturralde, G. A., Villacrés Granda, I. M., Tejera, E., Beltrán-Ayala, P., Giampieri, F., Battino, M., & Alvarez-Suarez, J. M. (2018). Influence of Botanical Origin and Chemical Composition on the Protective Effect against Oxidative Damage and the Capacity to Reduce In Vitro Bacterial Biofilms of Monofloral Honeys from the Andean Region of Ecuador. International Journal of Molecular Sciences, 19(1), 45. https://doi.org/10.3390/ijms19010045







