Gallic Acid Alleviates Hypertriglyceridemia and Fat Accumulation via Modulating Glycolysis and Lipolysis Pathways in Perirenal Adipose Tissues of Rats Fed a High-Fructose Diet
Abstract
:1. Introduction
2. Results
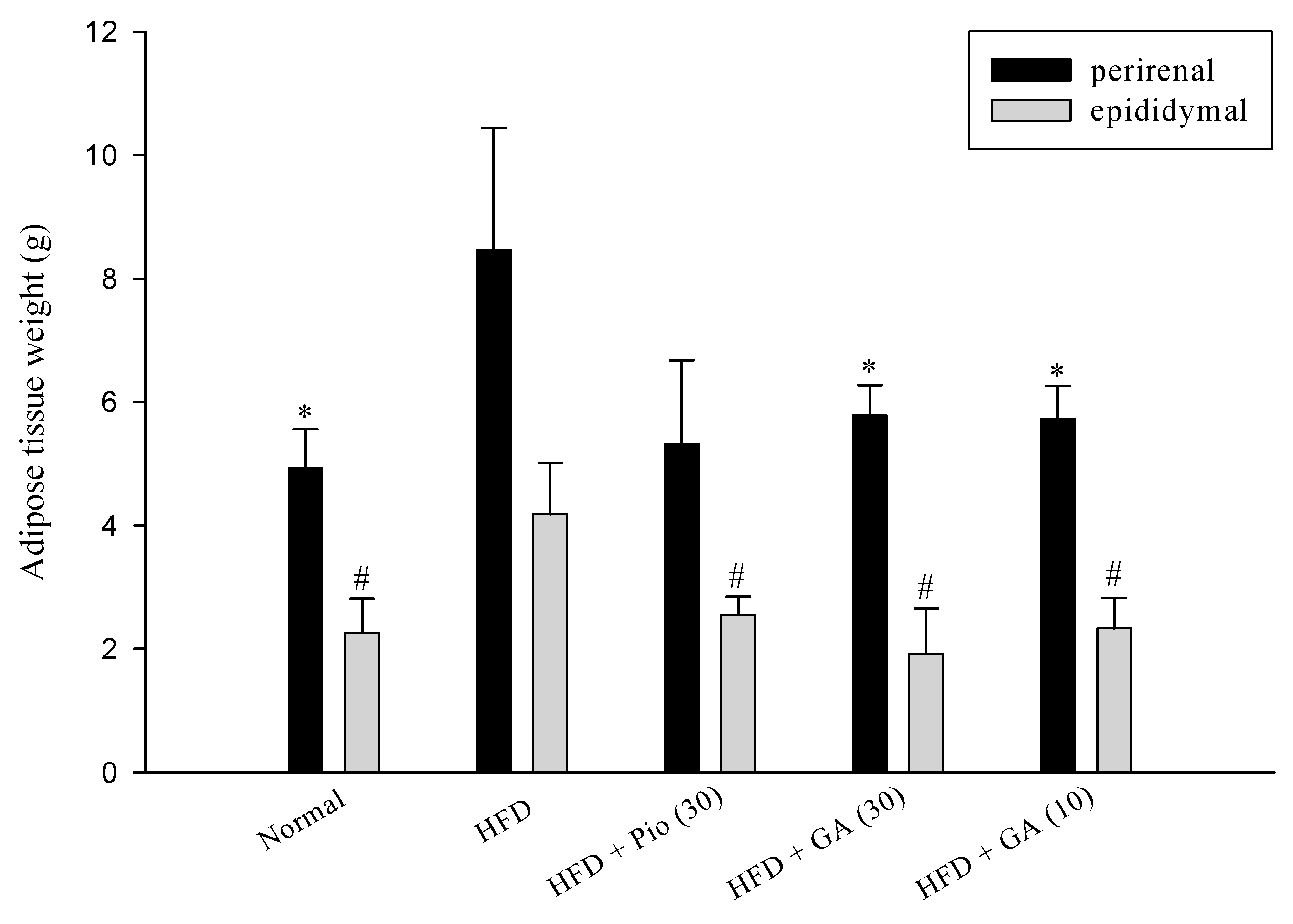
2.1. Effect of GA on Weight of Perirenal and Epidydimal Adipose Tissues in HFD-Induced Diabetic Rats
2.2. Effect of GA on Insulin Signal Transduction in the Perirenal Fat of HFD Rats
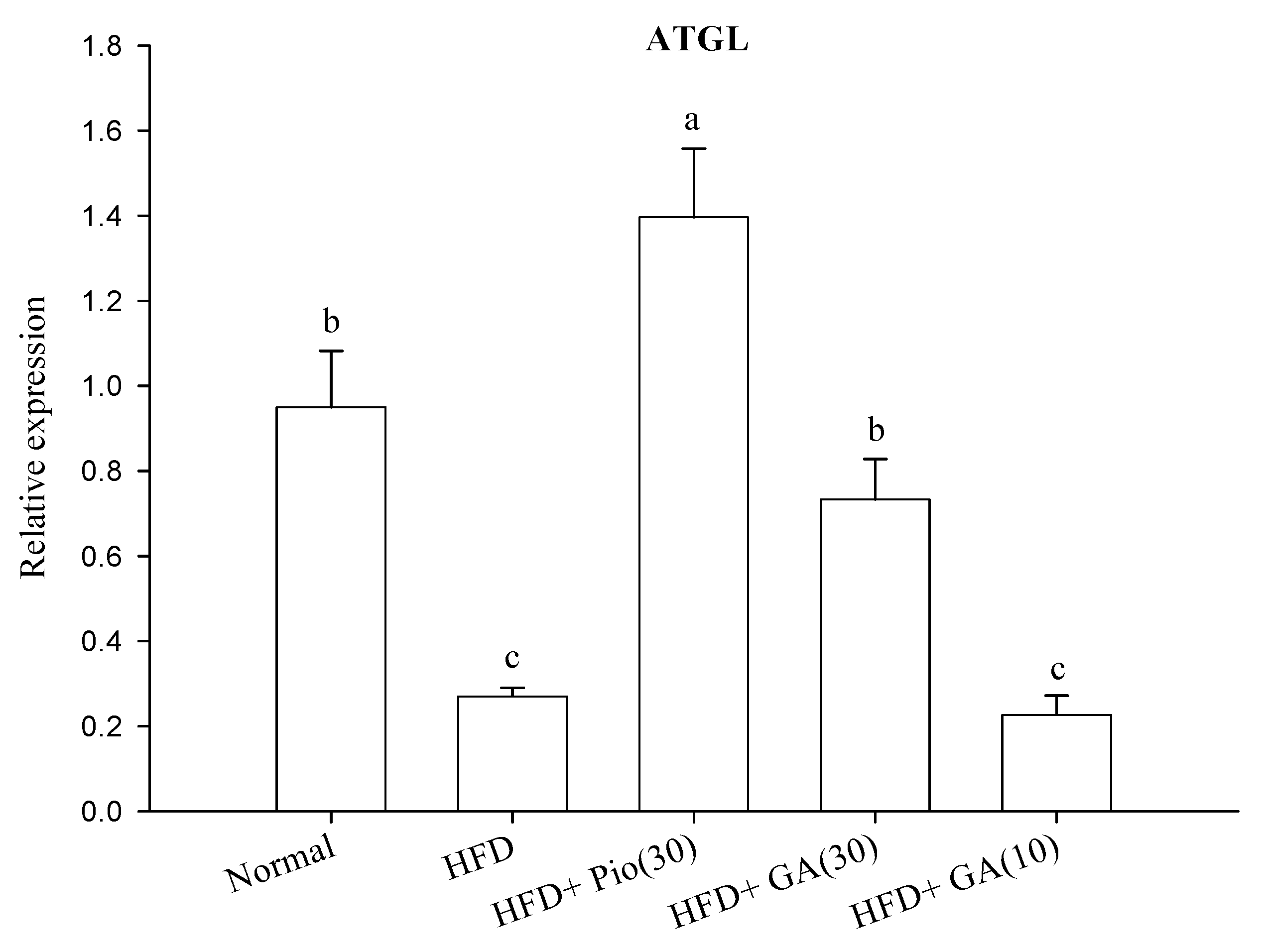
2.3. Effect of GA on Carbohydrate Metabolism and Lipid Metabolism in the Perirenal Fat of HFD Rats
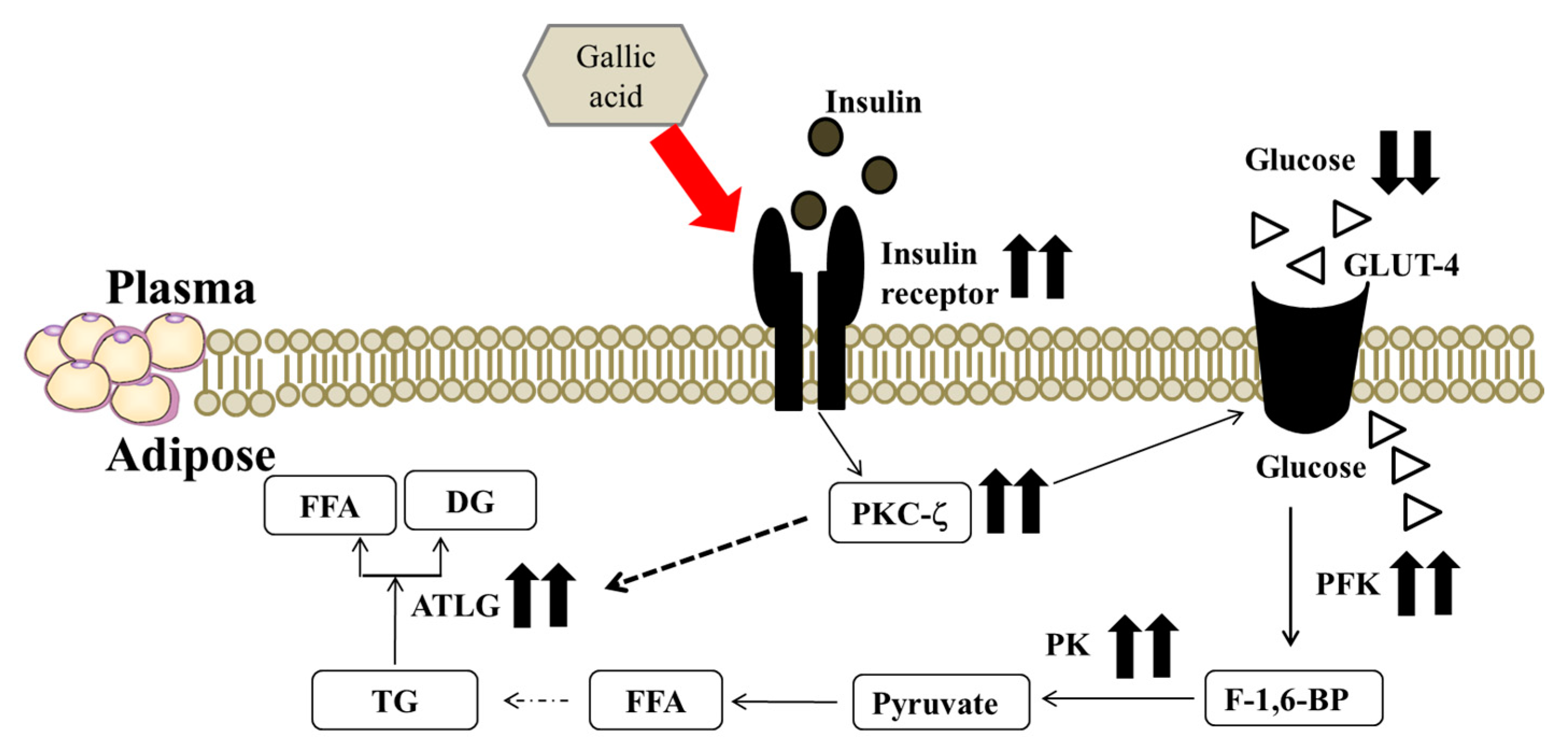
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals and Diets
4.3. Western Blot Analysis
4.4. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ATGL | Adipose triglyceride lipase |
| DG | Diglyceride |
| DM | Diabetes mellitus |
| F-1,6-BP | Fructose 1,6 biphosphate |
| FFA | Free fatty acid |
| GA | Gallic acid |
| GLUT4 | Glucose transporter-4 |
| HFD | High-fructose diet |
| IR | Insulin receptor |
| IRS | IR substrate |
| IRS-1 | IR substrate 1 |
| LDL-C | Low density lipoprotein-cholesterol |
| NOAEL | No-observed-adverse-effect level |
| PFK | Phosphofructokinase |
| PI3K | Phosphatidylinositol-3-kinase |
| PK | Pyruvate kinase |
| PKB | Akt/protein kinase B |
| PKC | Protein kinase C |
| PKC-ζ | Protein kinase C-zeta |
| STZ | Streptozotocin |
| T2DM | Type 2 DM |
| TG | Triglyceride |
References
- Latha, R.C.R.; Daisy, P. Insulin-secretagogue, antihyperlipidemic and other protective effects of gallic acid isolated from Terminalia bellerica Roxb. in streptozotocin-induced diabetic rats. Chem. Biol. Interact. 2011, 189, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Chang, W.C.; Wu, J.S.B.; Shih, R.W.; Shen, S.C. Vescalagin from Pink wax apple [Syzygium samarangense (Blume) Merrill and Perry] alleviates hepatic insulin resistance and ameliorates glycemic metabolism abnormality in rats fed a high-fructose diet. J. Agric. Food Chem. 2016, 57, 7687–7692. [Google Scholar] [CrossRef] [PubMed]
- Björnholm, M.; Zierath, J.R. Insulin signal transduction in human skeletal muscle: Identifying the defects in type II diabetes. Biochem. Soc. Trans. 2005, 33, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Punithavathi, V.R.; Stanely Mainzen Prince, P.; Kumar, M.R.; Selvakumari, C.J. Protective effects of gallic acid on hepatic lipid peroxide metabolism, glycoprotein components and lipids in streptozotocin-induced type II diabetic Wistar rats. J. Biochem. Mol. Toxicol. 2011, 25, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Perez-Pozo, S.E.; Sautin, Y.Y.; Manitius, J.; Sanchez-Lozada, L.G.; Feig, D.I.; Shafiu, M.; Segal, M.; Glassock, R.J.; Shimada, M.; et al. Hypothesis: Could excessive fructose intake and uric acid cause type 2 diabetes? Endocr. Rev. 2009, 30, 96–116. [Google Scholar] [CrossRef] [PubMed]
- Collison, K.S.; Saleh, S.M.; Bakheet, R.H.; Al-Rabiah, R.K.; Inglis, A.L.; Makhoul, N.J. Diabetes of the liver: The link between nonalcoholic fatty liver disease and HFCS-55. Obesity 2009, 17, 2003–2013. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Chang, W.C.; Wu, J.S.B.; Shih, R.W.; Shen, S.C. Gallic acid ameliorates hyperglycemia and improves hepatic carbohydrate metabolism in rats fed a high-fructose diet. Nutr. Res. 2016, 36, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Yang, Z.; Liu, H.; Tang, L.; Cai, Z. Beyond glucose: Metabolic shifts in responses to the effects of oral glucose tolerance test and the high-fructose diet in rats. Mol. Biosyst. 2011, 7, 1537–1548. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.; Huo1, T.I.; Cheng, H.Y.; Tsai, J.C.; Liao, J.W.; Lee, M.S.; Qin, X.M.; Hsieh, M.T.; Pao, L.H.; Peng, W.H. Gallic acid ameliorated impaired glucose and lipid homeostasis in high fat diet-induced NAFLD mice. PLoS ONE 2014, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Umadevi, S.; Gopi, V.; Elangovan, V. Regulatory mechanism of gallic acid against advanced glycation end products induced cardiac remodeling in experimental rat. Chem. Biol. Interact. 2014, 208, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Punithavathi, V.R.; Stanely Mainzen Prince, P.; Kumar, M.R.; Selvakumari, C.J. Antihyperglycaemic, antilipid peroxidative and antioxidant effects of gallic acid on streptozotocin induced diabetic Wistar rats. Eur. J. Pharmacol. 2011, 650, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Iyer, A.; Brown, L. Evaluation of the chronic complications of diabetes in a high fructose diet in rats. Indian J. Biochem. Biophys. 2009, 46, 66–72. [Google Scholar] [PubMed]
- Ruegsegger, G.N.; Company, J.M.; Toedebusch, R.G.; Roberts, C.K.; Roberts, M.D.; Booth, F.W. Rapid alterations in perirenal adipose tissue transcriptomic networks with cessation of voluntary running. PLoS ONE 2015, 10, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Oliveiraa, L.S.; Thoméa, G.R.; Lopesa, T.F.; Reicherta, K.P.; Oliveiraa, J.S.; Pereiraa, A.S.; Baldissarelia, J.; Krewerb, C.C.; Morscha, V.M.; Chitolina Schetinger, M.R.; et al. Effects of gallic acid on delta—Aminolevulinic dehydratase activity and in the biochemical, histological and oxidative stress parameters in the liver and kidney of diabetic rats. Biomed. Pharmacother. 2016, 84, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Samuel, V.T. Fructose induced lipogenesis: From sugar to fat to insulin resistance. Trends Endocrinol. Metab. 2011, 22, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.H.; Yen, G.C. Effect of gallic acid on high fat diet-induced dyslipidaemia, hepatosteatosis and oxidative stress in rats. Br. J. Nutr. 2007, 98, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Goyal, R.K. Cardioprotective effects of gallic acid in diabetes-induced myocardial dysfunction in rats. Pharmacogn. Res. 2011, 3, 239–245. [Google Scholar]
- Okuno, A.; Tamemoto, H.; Tobe, K.; Ueki, K.; Mori, Y.; Iwamoto, K.; Umesono, K.; Akanuma, Y.; Fujiwara, T.; Horikoshi, H.; et al. Troglitazone increases the number of small adipocytes without the change of white adipose tissue mass in obese Zucker rats. J. Clin. Investig. 1998, 101, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.L.; Lo, W.H.; Yen, G.C. Gallic acid induces apoptosis in 3T3-L1 pre-adipocytes via a Fas- and mitochondrial-mediated pathway. J. Agric. Food Chem. 2007, 55, 7359–7365. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, A.R.; Kahn, C.R. Insulin signaling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.W. Metabolic stress in insulin’s target cells leads to ROS accumulation—A hypothetical common pathway causing insulin resistance. FEBS Lett. 2007, 581, 3734–3742. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, G.R.; Jothi, G.; Antony, P.J.; Balakrishna, K.; Paulraj, M.G.; Ignacimuthu, S.; Stalin, A.; Al-Dhabi, N.A. Gallic acid attenuates high-fat diet fed-streptozotocin-induced insulin resistance via partial agonism of PPARγ in experimental type 2 diabetic rats and enhances glucose uptake through translocation and activation of GLUT4 in PI3K/p-Akt signaling pathway. Eur. J. Pharmacol. 2014, 745, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Hirai, T.; Chida, K. Protein kinase Cζ (PKCζ): Activation mechanisms and cellular functions. J. Biochem. 2003, 133, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Prasad, C.N.V.; Anjana, T.; Banerji, A.; Gopalakrishnapillai, A. Gallic acid induces GLUT4 translocation and glucose uptake activity in 3T3-L1 cells. FEBS Lett. 2010, 584, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, T.F.; Liou, S.S.; Liu, I.M. Myricetin ameliorates defective post-receptor insulin signaling via β-endorphin signaling in the skeletal muscles of fructose-fed rats. Evid.-Based Complement. Altern. Med. 2011, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.D.; Zancan, P.; Coelho, W.S.; Gomez, L.S.; Sola-Penna, M. Metformin reverses hexokinase and 6-phosphofructo-1-kinase inhibition in skeletal muscle, liver and adipose tissue from STZ-induced diabetic mouse. Arch. Biochem. Biophys. 2010, 496, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Vibhor, G.; Rameshwar, N.K.B. Human pyruvate kinase M2: A multifunctional protein. Protein Sci. 2010, 19, 2031–2044. [Google Scholar]
- Iori, E.; Millioni, R.; Puricelli, L.; Arrigoni, G.; Lenzini, L.; Trevisan, R.; James, P.; Rossi, G.P.; Pinna, L.A.; Tessari, P. Glycolytic enzyme expression and pyruvate kinase activity in cultured fibroblasts from type 1 diabetic patients with and without nephropathy. Biochim. Biophys. Acta Mol. Basis Dis. 2008, 1782, 627–633. [Google Scholar] [CrossRef] [PubMed]
- Parks, W.C.; Darke, R.L. Insulin mediates the stimulation of pyruvate kinase by a dual mechanism. Biochem. J. 1982, 208, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Brasaemle, D.L. Lipolysis control: The plot thickens. Cell Metab. 2010, 11, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, R.; Strauss, J.G.; Haemmerle, G.; Schoiswohl, G.; Birner-Gruenberger, R.; Riederer, M.; Lass, A.; Neuberger, G.; Eisenhaber, F.; Hermetter, A.; et al. Fat mobilization in adipose tissue is promoted by adipose triglyceride lipase. Science 2004, 306, 1383–1386. [Google Scholar] [CrossRef] [PubMed]
- Jocken, J.W.E.; Langin, D.; Smit, E.; Saris, W.H.M.; Valle, C.; Hul, G.B.; Holm, C.; Arner, P.; Blaak, E.E. Adipose triglyceride lipase and hormone-sensitive lipase protein expression is decreased in the obese insulin-resistant state. J. Clin. Endocrinol. Metab. 2007, 92, 2292–2299. [Google Scholar] [CrossRef] [PubMed]
- Morak, M.; Schmidinger, H.; Riesenhuber, G.; Rechberger, G.N.; Kollroser, M.; Haemmerle, G.; Zechner, R.; Kronenberg, F.; Hermetter, A. Adipose triglyceride lipase (ATGL) and hormone-sensitive lipase (HSL) deficiencies affect expression of lipolytic activities in mouse adipose tissues. Mol. Cell. Proteom. 2012, 11, 1777–1789. [Google Scholar] [CrossRef] [PubMed]
- Rajalakshmi, K.; Devaraj, H.; Niranjali Devaraj, S. Assessment of the no-observed-adverse-effect level (NOAEL) of gallic acid in mice. Food Chem. Toxicol. 2001, 39, 919–922. [Google Scholar] [CrossRef]
- Niho, N.; Shibutani, M.; Tamura, T.; Toyoda, K.; Uneyama, C.; Takahashi, N. Subchronic toxicity study of gallic acid by oral administration in F344 rats. Food Chem. Toxicol. 2001, 39, 1063–1070. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Guidance for Industry and Reviewers: Estimating the Safe Starting Dose in Clinical Trials for Theaputics in Adult Healthy Volunteers. 2005. Available online: https://www.fda.gov/downloads/drugs/guidances/ucm078932.pdf (accessed on 14 January 2018).

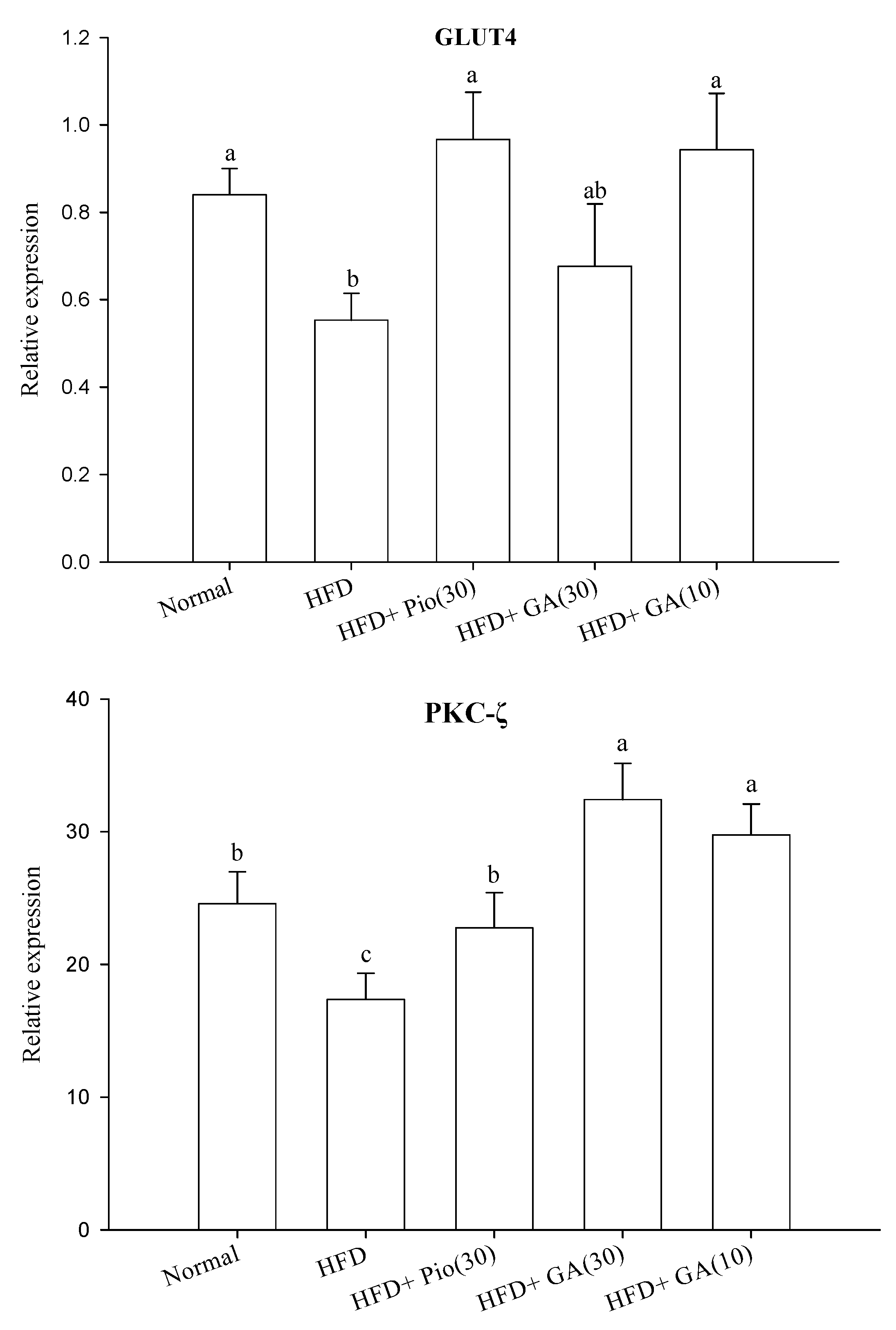

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, D.-W.; Chang, W.-C.; Yang, H.-J.; Wu, J.S.-B.; Shen, S.-C. Gallic Acid Alleviates Hypertriglyceridemia and Fat Accumulation via Modulating Glycolysis and Lipolysis Pathways in Perirenal Adipose Tissues of Rats Fed a High-Fructose Diet. Int. J. Mol. Sci. 2018, 19, 254. https://doi.org/10.3390/ijms19010254
Huang D-W, Chang W-C, Yang H-J, Wu JS-B, Shen S-C. Gallic Acid Alleviates Hypertriglyceridemia and Fat Accumulation via Modulating Glycolysis and Lipolysis Pathways in Perirenal Adipose Tissues of Rats Fed a High-Fructose Diet. International Journal of Molecular Sciences. 2018; 19(1):254. https://doi.org/10.3390/ijms19010254
Chicago/Turabian StyleHuang, Da-Wei, Wen-Chang Chang, Heng-Jui Yang, James Swi-Bea Wu, and Szu-Chuan Shen. 2018. "Gallic Acid Alleviates Hypertriglyceridemia and Fat Accumulation via Modulating Glycolysis and Lipolysis Pathways in Perirenal Adipose Tissues of Rats Fed a High-Fructose Diet" International Journal of Molecular Sciences 19, no. 1: 254. https://doi.org/10.3390/ijms19010254
APA StyleHuang, D.-W., Chang, W.-C., Yang, H.-J., Wu, J. S.-B., & Shen, S.-C. (2018). Gallic Acid Alleviates Hypertriglyceridemia and Fat Accumulation via Modulating Glycolysis and Lipolysis Pathways in Perirenal Adipose Tissues of Rats Fed a High-Fructose Diet. International Journal of Molecular Sciences, 19(1), 254. https://doi.org/10.3390/ijms19010254




