Bio-Functional Design, Application and Trends in Metallic Biomaterials
Abstract
1. Introduction
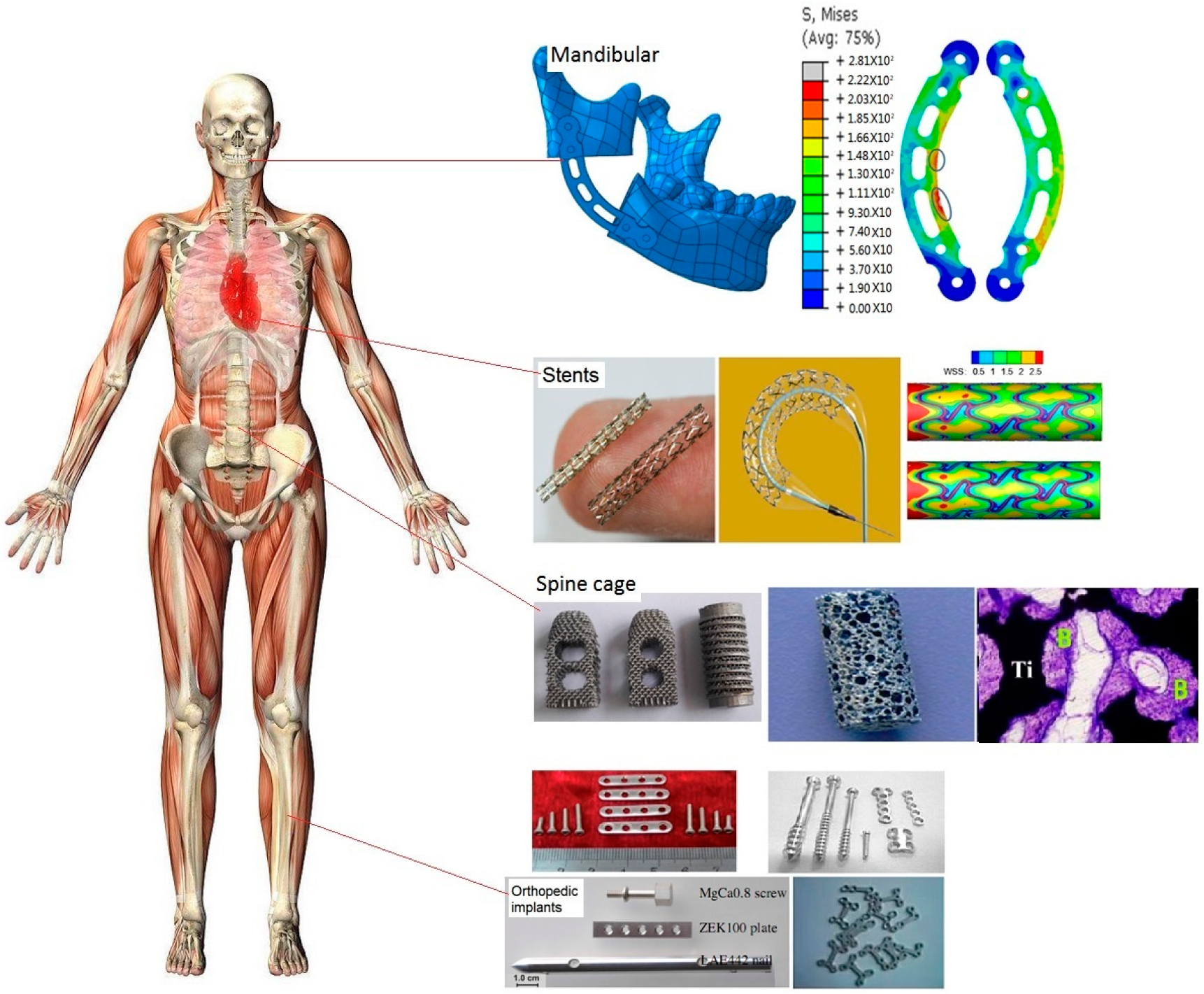
2. Different Applications of Metal Implants in Clinic
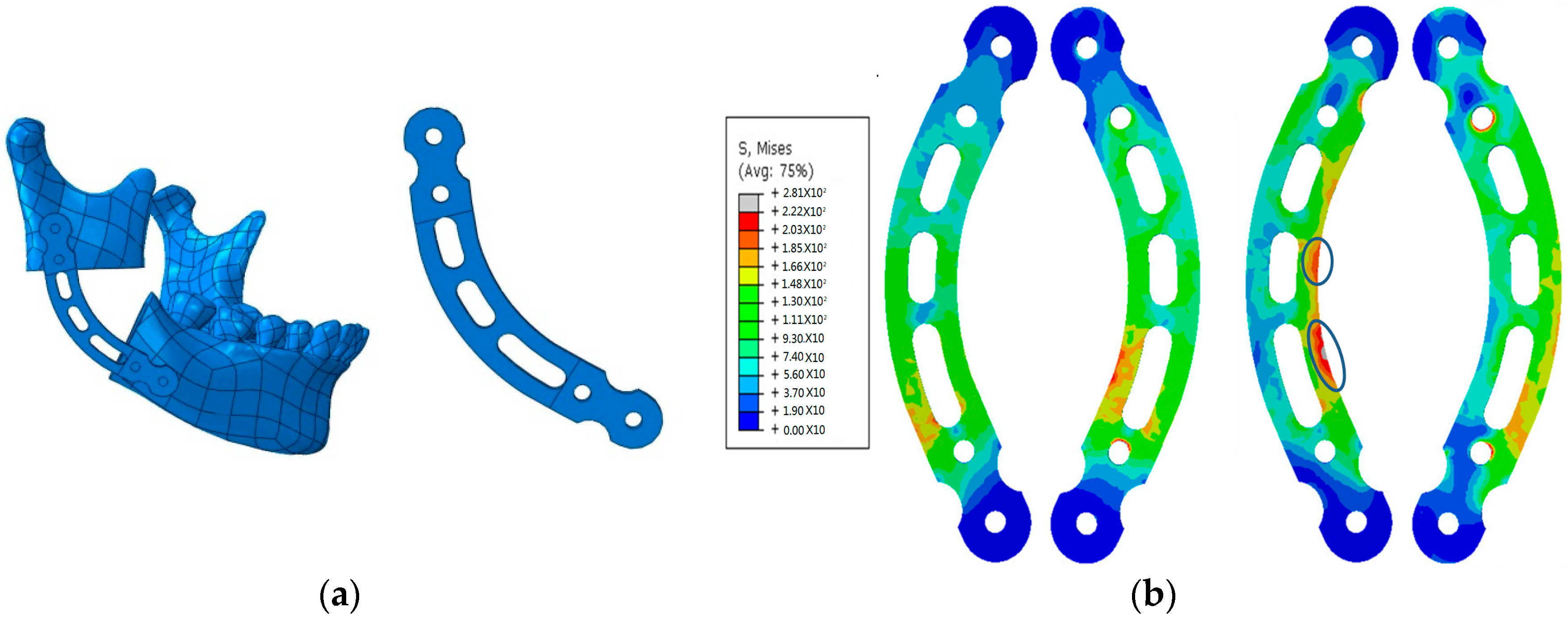
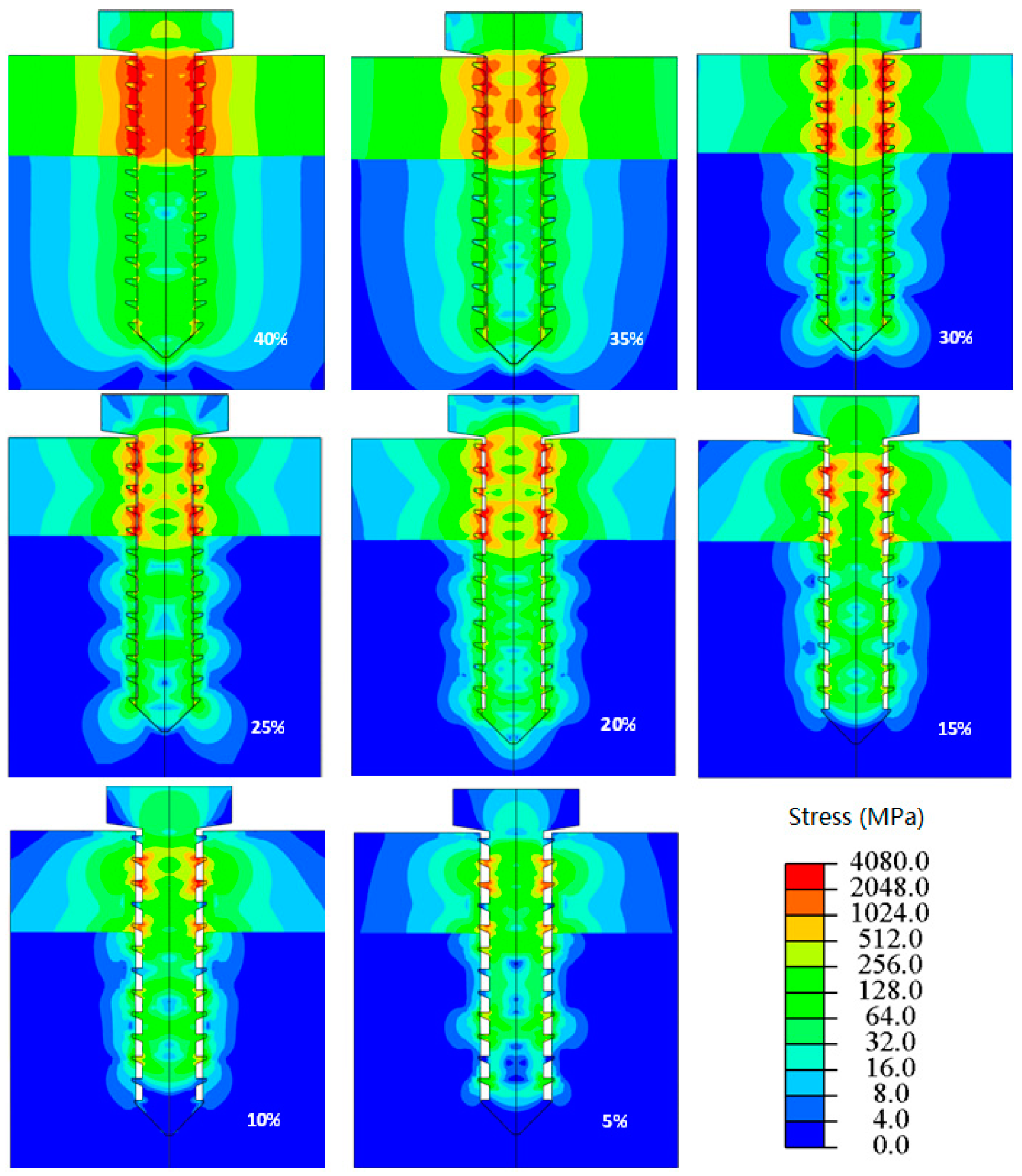
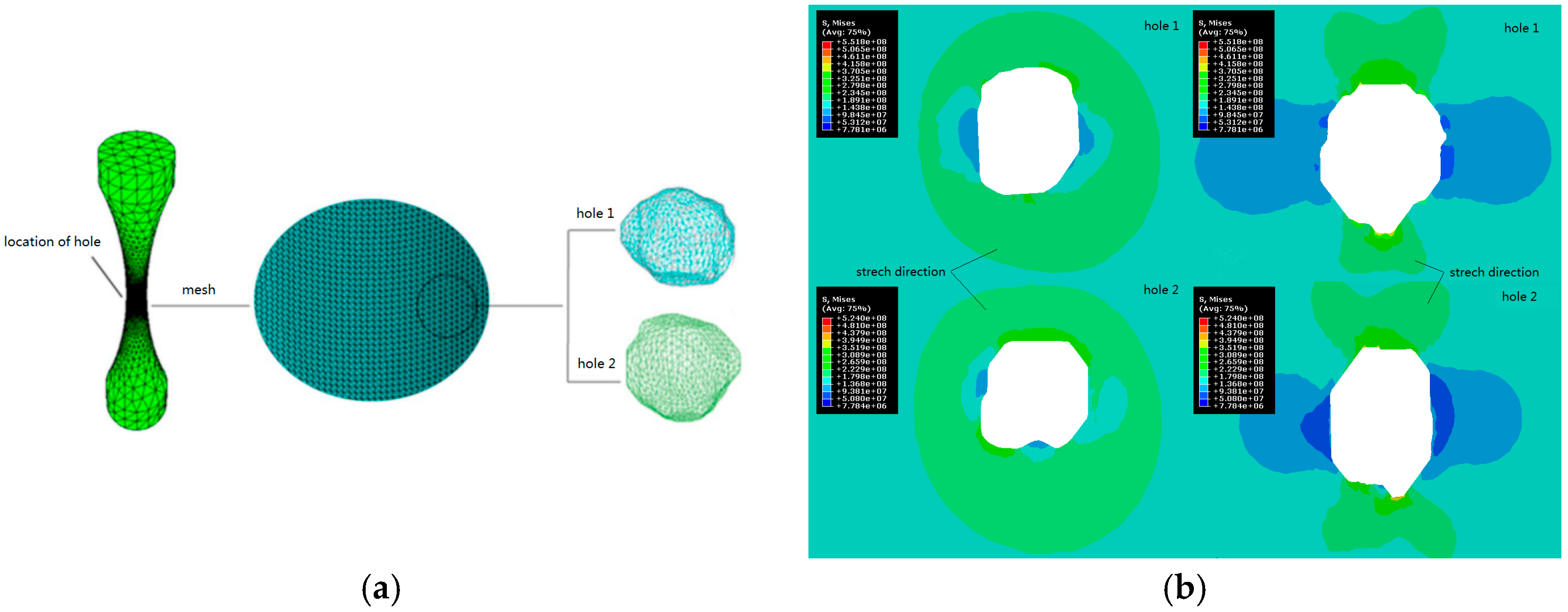
2.1. Biomechanical Design of Metal Implants
2.2. Porous Structure Design and Biological Activation of Metal Implants
2.2.1. Porous Structure Design and Manufacturing
2.2.2. Biological Activation of Metal Implants
2.3. Biodegradable Design for Metal Implants
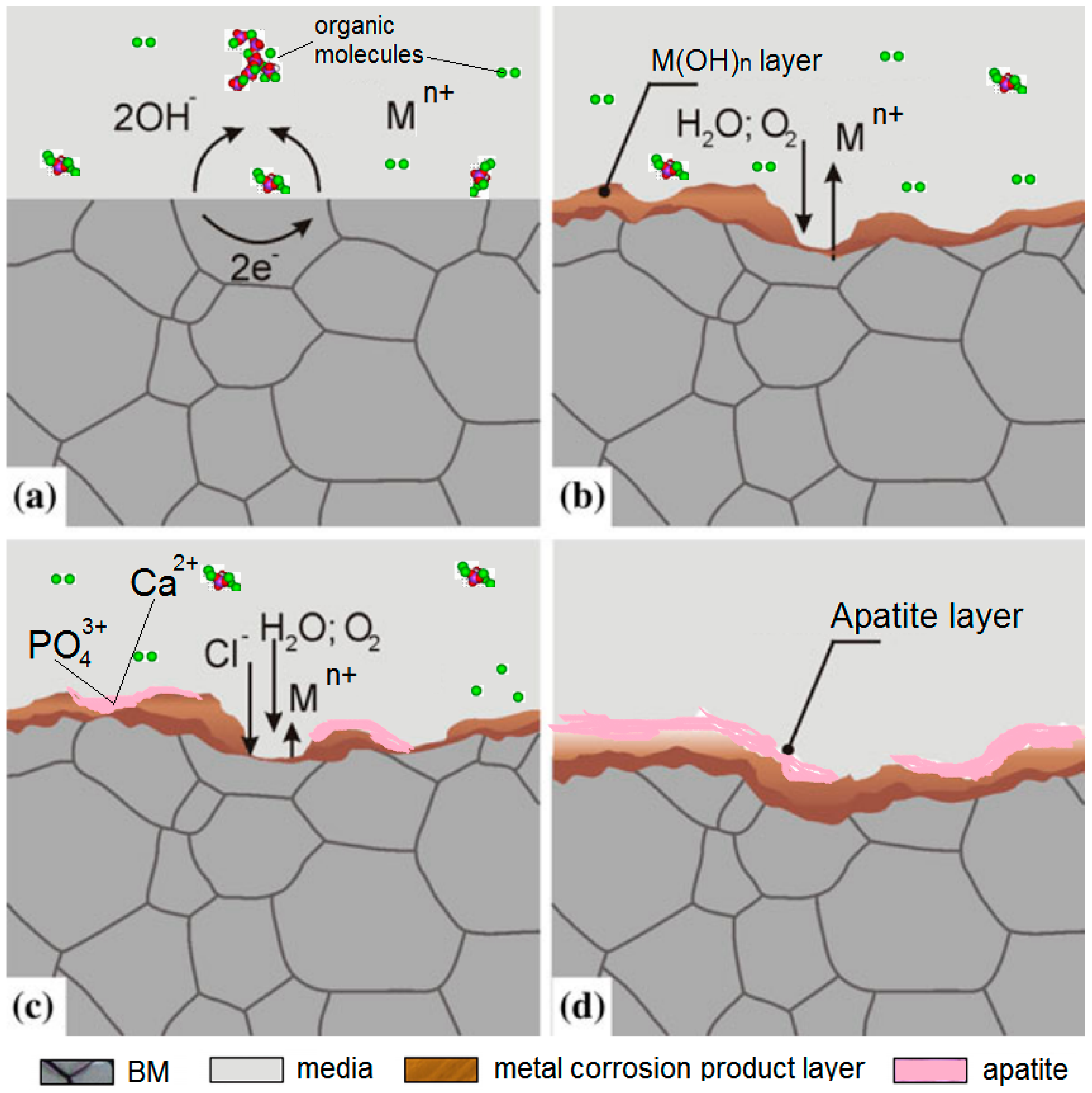
Degradation Mechanism of Metal Implants
3. Different Types of Biodegradable Metal Implants
3.1. Applications of Magnesium-Based BMs
3.2. Applications of Fe-Based BMs
3.3. Applications of Zn-Based BMs
4. Stents Applications
5. Orthopedic Applications
6. Concluding Remarks and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xu, G.; Fu, X.; Du, C.; Ma, J.; Li, Z.; Tian, P.; Zhang, T.; Ma, X. Biomechanical comparison of mono-segment transpedicular fixation with short-segment fixation for treatment of thoracolumbar fractures: A finite element analysis. Proc. Inst. Mech. Eng. J. Part H Eng. Med. 2014, 228, 1005–1013. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Lei, B.; Li, P.; Ma, P.X. Functionalized scaffolds to enhance tissue regeneration. Regen. Biomater. 2015, 2, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Pownder, S.L.; Koff, M.F.; Shah, P.H.; Fortier, L.A.; Potter, H.G. Magnetic resonance imaging of an equine fracture model containing stainless steel metal implants. Equine Vet. J. 2016, 48, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Hudetz, D.; Ursic Hudetz, S.; Harris, L.G.; Luginbuhl, R.; Friederich, N.F.; Landmann, R. Weak effect of metal type and ica genes on staphylococcal infection of titanium and stainless steel implants. Clin. Microbiol. Infect. 2008, 14, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Xynos, I.D.; Polak, J.M. Bioactive glasses for in situ tissue regeneration. J. Biomater. Sci. Polym. Ed. 2004, 15, 543–562. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. The story of Bioglass. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Boccaccini, A.R.; Day, R.M.; Gabe, S.M. Third-generation gene-activating biomaterials. Mater. Sci. Forum 2003, 426–432, 179–184. [Google Scholar] [CrossRef]
- Mousa, H.M.; Tiwari, A.P.; Kim, J.; Adhikari, S.P.; Park, C.H.; Kim, C.S. A novel in situ deposition of hydroxyapatite nanoplates using anodization/hydrothermal process onto magnesium alloy surface towards third generation biomaterials. Mater. Lett. 2016, 164, 144–147. [Google Scholar] [CrossRef]
- Vallittu, P.K. Bioactive glass-containing cranial implants: An overview. J. Mater. Sci. 2017, 52, 8772–8784. [Google Scholar] [CrossRef]
- Chen, X.H.; Geng, Y.X.; Pan, F.S. Research progress in magnesium alloys as functional materials. Rare Met. Mater. Eng. 2016, 45, 2269–2274. [Google Scholar]
- Chen, Y.Q.; Zhang, W.T.; Maitz, M.F.; Chen, M.Y.; Zhang, H.; Mao, J.L.; Zhao, Y.C.; Huang, N.; Wan, G.J. Comparative corrosion behavior of Zn with Fe and Mg in the course of immersion degradation in phosphate buffered saline. Corros. Sci. 2016, 111, 541–555. [Google Scholar] [CrossRef]
- Liu, X.W.; Sun, J.K.; Zhou, F.Y.; Yang, Y.H.; Chang, R.C.; Qiu, K.J.; Pu, Z.J.; Li, L.; Zheng, Y.F. Micro-alloyingwith Mn in Zn-Mg alloy for future biodegradable metals application. Mater. Des. 2016, 94, 95–104. [Google Scholar] [CrossRef]
- Ma, J.; Zhao, N.; Betts, L.; Zhu, D.H. Bio-adaption between magnesium alloy stent and the blood vessel: A review. J. Mater. Sci. Technol. 2016, 32, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Miura, C.; Shimizu, Y.; Imai, Y.; Mukai, T.; Yamamoto, A.; Sano, Y.; Ikeo, N.; Isozaki, S.; Takahashi, T.; Oikawa, M.; et al. In vivo corrosion behaviour of magnesium alloy in association with surrounding tissue response in rats. Biomed. Mater. 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Torne, K.; Larsson, M.; Norlin, A.; Weissenrieder, J. Degradation of zinc in saline solutions, plasma, and whole blood. J. Biomed. Mater. Res. B 2016, 104, 1141–1151. [Google Scholar] [CrossRef] [PubMed]
- Bian, D.; Zhou, W.R.; Liu, Y.; Li, N.; Zheng, Y.F.; Sun, Z.L. Fatigue behaviors of HP-Mg, Mg-Ca and Mg-Zn-Ca biodegradable metals in air and simulated body fluid. Acta Biomater. 2016, 41, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Su, J.S.; Xu, H.Z.; Sun, J.; Gong, X.; Zhao, H. Dual delivery of BMP-2 and bFGF from a new nano-composite scaffold, loaded with vascular stents for large-size mandibular defect regeneration. Int. J. Mol. Sci. 2013, 14, 12714–12728. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.Z.; Su, J.S.; Sun, J.; Ren, T.B. Preparation and characterization of new nano-composite scaffolds loaded with vascular stents. Int. J. Mol. Sci. 2012, 13, 3366–3381. [Google Scholar] [CrossRef] [PubMed]
- Gabler, C.; Zietz, C.; Gohler, R.; Fritsche, A.; Lindner, T.; Haenle, M.; Finke, B.; Meichsner, J.; Lenz, S.; Frerich, B.; et al. Evaluation of osseointegration of titanium alloyed implants modified by plasma polymerization. Int. J. Mol. Sci. 2014, 15, 2454–2464. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.H.; Moon, S.W.; Lee, D.W. Surface modification of titanium with BMP-2/GDF-5 by a heparin linker and its efficacy as a dental implant. Int. J. Mol. Sci. 2017, 18, 229. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Z.; Zhang, Y.F.; Choukroun, J.; Ghanaati, S.; Miron, R.J. Behavior of gingival fibroblasts on Titanium implant surfaces in combination with either injectable-PRF or PRP. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.J.; Wang, T.; Qian, S.; Liu, X.Y.; Sun, J.Y.; Li, B. Silicon-doped titanium dioxide nanotubes promoted bone formation on titanium implants. Int. J. Mol. Sci. 2016, 17, 292. [Google Scholar] [CrossRef] [PubMed]
- Matena, J.; Petersen, S.; Gieseke, M.; Kampmann, A.; Teske, M.; Beyerbach, M.; Escobar, H.M.; Haferkamp, H.; Gellrich, N.C.; Nolte, I. SLM produced porous titanium implant improvements for enhanced vascularization and osteoblast seeding. Int. J. Mol. Sci. 2015, 16, 7478–7492. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, R.; Jimbo, R.; Thomsen, C.; Carlsson, L.; Wennerberg, A. Biomechanical evaluation of macro and micro designed screw-type implants: An insertion torque and removal torque study in rabbits. Clin. Oral Implants Res. 2013, 24, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Cheng, C.K.; Wei, H.W.; Dong, X.; Chen, Y.T.; Lai, Y.S.; Wang, Y. Biomechanical comparisons between a new avascular necrosis of femaral head stem based on Chinese patients with avascular necrosis and two other designs. Chin. Med. J. Peking 2013, 126, 1918–1924. [Google Scholar]
- Cheng, C.K.; McClean, C.J.; Lai, Y.S.; Chen, W.C.; Huang, C.H.; Lin, K.J.; Chang, C.M. Biomechanical considerations in the design of high-flexion total knee replacements. Sci. World J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Prashanth, S.E.K.G.; Attar, H.; Chaubey, A.K.; Cao, G.H.; Zhang, L.C. Evaluation of mechanical and wear properties of TixNb7Fe alloys designed for biomedical applications. Mater. Des. 2016, 111, 592–599. [Google Scholar]
- Attar, H.; Ehtemam-Haghighi, S.; Kent, D.; Wu, X.H.; Dargusch, M.S. Comparative study of commercially pure titanium produced by laser engineered net shaping, selective laser melting and casting processes. Mater. Sci. Eng. A Struct. 2017, 705, 385–393. [Google Scholar] [CrossRef]
- Ansari, F.; Pack, L.K.; Brooks, S.S.; Morrison, T.M. Design considerations for studies of the biomechanical environment of the femoropopliteal arteries. J. Vasc. Surg. 2013, 58, 804–813. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.K.; Zhang, M.; Laxton, P.; Findlow, A.H.; Liu, A.M. The biomechanical effects of a new design of lateral wedge insole on the knee and ankle during walking. Hum. Mov. Sci. 2013, 32, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.A.; Iamashita, H.Y.; Monnazzi, M.S.; Gabrielli, M.F.R.; Vaz, L.G.; Passeri, L.A. In vitro biomechanical evaluation of sagittal split osteotomy fixation with a specifically designed miniplate. Int. J. Oral Maxillofac. Surg. 2013, 42, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.M.; Liu, C.L.; Pan, Y.N.; Huang, C.H.; Shih, S.L.; Wei, S.H.; Chen, C.S. Biomechanical analysis and design of a dynamic spinal fixator using topology optimization: A finite element analysis. Med. Biol. Eng. Comput. 2014, 52, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Thiyagarajan, K.; Bhusal, A.; Letcher, T.; Fan, Q.H.; Liu, Q.; Salem, D. Design of ultra-lightweight and high-strength cellular structural composites inspired by biomimetics. Compos. Part B Eng. 2017, 121, 108–121. [Google Scholar] [CrossRef]
- Okulov, I.V.; Volegov, A.S.; Attar, H.; Bonisch, M.; Ehternam-Haghighi, S.; Calin, M.; Eckert, J. Composition optimization of low modulus and high-strength TiNb-based alloys for biomedical applications. J. Mech. Behav. Biomed. 2017, 65, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Suer, B.T.; Kocyigit, I.D.; Kaman, S.; Tuz, H.H.; Tekin, U.; Atil, F. Biomechanical evaluation of a new design titanium miniplate for the treatment of mandibular angle fractures. Int. J. Oral Maxillofac. Surg. 2014, 43, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Serhan, M.; Verim, O.; Eroglu, M.; Altinel, L.; Gokce, B.; Tasgetiren, S. Biomechanical evaluation of syndesmotic screw design via finite element analysis and Taguchi’s method. JAPMA 2015, 105, 14–21. [Google Scholar]
- Liu, D.X.; Hua, Z.K.; Yan, X.Y.; Jin, Z.M. Design and biomechanical study of a novel adjustable hemipelvic prosthesis. Med. Eng. Phys. 2016, 38, 1416–1425. [Google Scholar] [CrossRef] [PubMed]
- Khan, Y.M.; Nair, L.S. Review of “Biomaterials for Musculoskeletal Regeneration: Concepts, by Bikramjit Basu, Indian Institute of Metals Series, Springer Nature (2016)” and “Biomaterials for Musculoskeletal Regeneration: Applications, by Bikramjit Basu and Sourabh Ghosh, Indian Institute of Metals Series, Springer Nature (2016)”. J. Mater. Sci. 2017. [Google Scholar] [CrossRef]
- Zhou, J.J.; Zhao, M.; Liu, D.; Liu, H.Y.; Du, C.F. Biomechanical Property of a Newly Designed Assembly Locking Compression Plate: Three-Dimensional Finite Element Analysis. J. Healthc. Eng. 2017. [Google Scholar] [CrossRef] [PubMed]
- Muiznieks, L.D.; Keeley, F.W. Biomechanical Design of Elastic Protein Biomaterials: A Balance of Protein Structure and Conformational Disorder. ACS Biomater. Sci. Eng. 2017, 3, 661–679. [Google Scholar] [CrossRef]
- Liu, J.T.; Chen, W.C.; Wei, H.W. Biomechanical evaluation of a dynamic fusion cage design for cervical spine: A finite element study. Adv. Mech. Eng. 2017, 9. [Google Scholar] [CrossRef]
- Tang, Z.X.; Li, L.Y.; Guo, W.P.; Jiang, W.T.; Fan, Y.B. Topology optimization on configuration of titanium reconstruction plate for bridging mandibular angle defect. J. Med. Biomech. 2014, 29, 167–173. [Google Scholar]
- Guo, W.; Li, Y.; Tang, Z.; Jiang, W.; Fan, Y. Finite element modeling of the mandible with temporomandibular joint. J. Biomed. Eng. Res. 2013, 3, 57–65. [Google Scholar] [CrossRef]
- Wan, Z.P.; Wang, C.; Jiang, W.T.; Huang, Z.Y.; Wang, Q.Y. On the effect of void defects on stress distribution of Ti-6Al-4V alloy fatigue specimen in 3D printing. J. Exp. Mech. 2017, 32, 1–8. [Google Scholar]
- Zunino, P.; D’Aangelo, C.; Petrini, L.; Vergara, C.; Capelli, C. Numerical simulation of drug eluting coronary stents: Mechanics, fluid dynamics and drug release. Comput. Methods Appl. Mech. Eng. 2009, 198, 3633–3644. [Google Scholar] [CrossRef]
- Chen, Y.; Xiong, Y.; Jiang, W.; Wong, M.S.; Yan, F.; Wang, Q.; Fan, Y. Numerical simulation on the effects ofdrug-eluting stents with different bending angles on hemodynamics and drug distribution. Med. Biol. Eng. Comput. 2016, 54, 1859–1867. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y. Numerical study on effects of drug-coating position of drug-eluting stents on drug concentration. J. Med. Biol. Eng. 2015, 34, 487–494. [Google Scholar] [CrossRef]
- Shah, F.A.; Snis, A.; Matic, A.; Thomsen, P.; Palmquist, A. 3D printed Ti6Al4V implant surface promotes bone maturation and retains a higher density of less aged osteocytes at the bone-implant interface. Acta Biomater. 2016, 30, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Liu, Q.; Fu, J.; Li, Q.; Hui, D. Dynamic crash responses of bio-inspired aluminum honeycomb sandwich structures with CFRP panels. Compos. Part B Eng. 2017, 121, 122–133. [Google Scholar] [CrossRef]
- Galdos, M.V.G.; Pastore, J.I.; Ballarre, J.; Cere, S.M. Dual-surface modification of titanium alloy with anodizing treatment and bioceramic particles for enhancing prosthetic devices. J. Mater. Sci. 2017, 52, 9151–9165. [Google Scholar] [CrossRef]
- Habibovic, P.; Gbureck, U.; Doillon, C.J.; Bassett, D.C.; van Blitterswijk, C.A.; Barralet, J.E. Osteoconduction and osteoinduction of low-temperature 3D printed bioceramic implants. Biomaterials 2008, 29, 944–953. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Xue, G.H.; Sun, M.; Shao, H.F.; Ma, C.Y.; Gao, Q.; Gou, Z.R.; Yan, S.G.; Liu, Y.M.; He, Y. 3D Printing Surgical Implants at the clinic: A Experimental Study on Anterior Cruciate Ligament Reconstruction. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Mobbs, R.J.; Coughlan, M.; Thompson, R.; Sutterlin, C.E.; Phan, K. The utility of 3D printing for surgical planning and patient-specific implant design for complex spinal pathologies: Case report. J. Neurosurg. Spine 2017, 26, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Palmquist, A.; Shah, F.A.; Emanuelsson, L.; Omar, O.; Suska, F. A technique for evaluating bone ingrowth into 3D printed, porous Ti6Al4V implants accurately using X-ray micro-computed tomography and histomorphometry. Micron 2017, 94, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.; Ngo, T.D.; Ghazlan, A.; Hui, D. Bimaterial 3D printing and numerical analysis of bio-inspired composite structures under in-plane and transverse loadings. Compos. Part B Eng. 2017, 108, 210–223. [Google Scholar] [CrossRef]
- Yang, F.; Chen, C.; Zhou, Q.R.; Gong, Y.M.; Li, R.X.; Li, C.C.; Klampfl, F.; Freund, S.; Wu, X.W.; Sun, Y.; et al. Laser beam melting 3D printing of Ti6Al4V based porous structured dental implants: Fabrication, biocompatibility analysis and photoelastic study. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, B.; Hui, D.; Qiu, J.; Wang, S. 3D bioprinting of soft materials-based regenerative vascular structures and tissues. Compos. Part B Eng. 2017, 123, 279–291. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, M.; Zhou, Z.; Gou, J.; Hui, D. 3D printing of polymer matrix composites: A review and prospective. Compos. Part B Eng. 2017, 110, 442–458. [Google Scholar] [CrossRef]
- Pandele, A.M.; Ionita, M.; Crica, L.; Vasile, E.; Iovu, H. Novel Chitosan-poly(vinyl alcohol)/graphene oxide biocomposites 3D porous scaffolds. Compos. Part B Eng. 2017, 126, 81–87. [Google Scholar] [CrossRef]
- Pei, X.; Zhang, B.; Fan, Y.; Zhu, X.; Sun, Y.; Wang, Q.; Zhang, X.; Zhou, C. Bionic mechanical design of titanium bone tissue implants and 3D printing manufacture. Mater. Lett. 2017. [Google Scholar] [CrossRef]
- Momeni, F.; Hassani, N.S.M.M.; Liu, X.; Ni, J. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- Kaczmarek, M.; Jurczyk, K.; Koper, J.K.; Paszel-Jaworska, A.; Romaniuk, A.; Lipinska, N.; Zurawski, J.; Urbaniak, P.; Jakubowicz, J.; Jurczyk, M.U. In vitro biocompatibility of anodized titanium with deposited silver nanodendrites. J. Mater. Sci. 2016, 51, 5259–5270. [Google Scholar] [CrossRef]
- Lux, F.; Zeisler, R. Investigations of corrosive deposition of components of metal implants and of behavior of biological trace-elements in metallosis tissue by means of instrumental multi-element activation-analysis. J. Radioanal. Nucl. Chem. 1974, 19, 289–297. [Google Scholar] [CrossRef]
- Boke, F.; Schickle, K.; Fischer, H. Biological activation of inert ceramics: Recent advances using tailored self-assembled monolayers on implant ceramic surfaces. Materials 2014, 7, 4473–4492. [Google Scholar] [CrossRef] [PubMed]
- Ostrovska, L.; Vistejnova, L.; Dzugan, J.; Slama, P.; Kubina, T.; Ukraintsev, E.; Kubies, D.; Kralickova, M.; Kalbacova, M.H. Biological evaluation of ultra-fine titanium with improved mechanical strength for dental implant engineering. J. Mater. Sci. 2016, 51, 3097–3110. [Google Scholar] [CrossRef]
- Ribeiro, A.M.; Flores-Sahagun, T.H.S.; Paredes, R.C. A perspective on molybdenum biocompatibility and antimicrobial activity for applications in implants. J. Mater. Sci. 2016, 51, 2806–2816. [Google Scholar] [CrossRef]
- Choy, M.-T.; Tang, C.-Y.; Chen, L.; Law, W.-C.; Tsui, C.-P.; Lu, W.W. Microwave assisted- synthesis of porous titanium/calcium phosphate composites and their apatite-forming capability. Compos. Part B Eng. 2015, 83, 50–57. [Google Scholar] [CrossRef]
- Balas, F.; Kawashita, M.; Nakamura, T.; Kokubo, T. Formation of bone-like apatite on organic polymers treated with a silane-coupling agent and a titania solution. Biomaterials 2006, 27, 1704–1710. [Google Scholar] [CrossRef] [PubMed]
- Leonor, I.B.; Balas, F.; Kawashita, M.; Reis, R.L.; Kokubo, T.; Nakamura, T. Biomimetic apatite formation on different polymeric microspheres modified with calcium silicate solutions. Key Eng. Mater. 2006, 309–311, 279–282. [Google Scholar] [CrossRef]
- Leonor, I.B.; Kim, H.M.; Balas, F.; Kawashita, M.; Reis, R.L.; Kokubo, T.; Nakamura, T. Formation of bone-like apatite on polymeric surfaces modified with-SO3H groups. Adv. Mater. Forum III 2006, 514–516, 966–969. [Google Scholar] [CrossRef]
- Zhou, C.C.; Ye, X.J.; Fan, Y.J.; Qing, F.Z.; Chen, H.J.; Zhang, X.D. Synthesis and characterization of CaP/Col composite scaffolds for load-bearing bone tissue engineering. Compos. Part B Eng. 2014, 62, 242–248. [Google Scholar] [CrossRef]
- Pattanayak, D.K.; Yamaguchi, S.; Matsushita, T.; Nakamura, T.; Kokubo, T. Apatite-forming ability of titanium in terms of pH of the exposed solution. J. R. Soc. Interface 2012, 9, 2145–2155. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Yamaguchi, S. Growth of novel ceramic layers on metals via chemical and heat treatments for inducing various biological functions. Front. Bioeng. Biotechnol. 2015, 3, 176. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.H.; Chang, C.H.; Chen, Y.S.; Lin, G.T. Formation of bone-like apatite on titanium filaments incubated in a simulated body fluid by using an electrochemical method. Compos. Part A Appl. Sci. Manuf. 2007, 38, 535–539. [Google Scholar] [CrossRef]
- Cui, X.Y.; Kim, H.M.; Kawashita, M.; Wang, L.B.; Xiong, T.Y.; Kokubo, T.; Nakamura, T. Apatite formation on anodized Ti-6Al-4V alloy in simulated body fluid. Met. Mater. Int. 2010, 16, 407–412. [Google Scholar] [CrossRef]
- Chavan, P.N.; Bahir, M.M.; Mene, R.U.; Mahabole, M.P.; Khairnar, R.S. Study of nanobiomaterial hydroxyapatite in simulated body fluid: Formation and growth of apatite. Mater. Sci. Eng. B Adv. 2010, 168, 224–230. [Google Scholar] [CrossRef]
- Kang, K.-T.; Koh, Y.-G.; Son, J.; Yeom, J.S.; Park, J.-H.; Kim, H.-J. Biomechanical evaluation of pedicle screw fixation system in spinal adjacent levels using polyetheretherketone, carbon-fiber-reinforced polyetheretherketone, and traditional titanium as rod materials. Compos. Part B Eng. 2017, 130, 248–256. [Google Scholar] [CrossRef]
- Yoshida, E.; Hayakawa, T. Quantitative analysis of apatite formation on titanium and zirconia in a simulated body fluid solution using the quartz crystal microbalance method. Adv. Mater. Sci. Eng. 2017. [Google Scholar] [CrossRef]
- Zhao, C.Y.; Zhu, X.D.; Yuan, T.; Fan, H.S.; Zhang, X.D. Fabrication of biomimetic apatite coating on porous titanium and their osteointegration in femurs of dogs. Mater. Sci. Eng. C 2010, 30, 98–104. [Google Scholar]
- Gu, X.N.; Xie, X.H.; Li, N.; Zheng, Y.F.; Qin, L. In vitro and in vivo studies on a Mg-Sr binary alloy system developed as a new kind of biodegradable metal. Acta Biomater. 2012, 8, 2360–2374. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Zheng, Y.F.; Qin, L. Progress of biodegradable metals. Prog. Nat. Sci. Mater. Int. 2014, 24, 414–422. [Google Scholar] [CrossRef]
- Zheng, Y.F.; Gu, X.N.; Witte, F. Biodegradable metals. Mat. Sci. Eng. R Rep. 2014, 77, 1–34. [Google Scholar] [CrossRef]
- Li, H.F.; Zheng, Y.F. Recent advances in bulk metallic glasses for biomedical applications. Acta Biomater. 2016, 36, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yang, H.T.; Li, X.; Zheng, Y.F. In vitro evaluation of the feasibility of commercial Zn alloys as biodegradable metals. J. Mater. Sci. Technol. 2016, 32, 909–918. [Google Scholar] [CrossRef]
- Bajger, P.; Ashbourn, J.M.; Manhas, V.; Guyot, Y.; Lietaert, K.; Geris, L. Mathematical modelling of the degradation behaviour of biodegradable metals. Biomech. Model. Mechanobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.B.; Huang, H.; Niu, J.L.; Zhang, L.; Zhang, H.; Pei, J.; Tan, J.Y.; Yuan, G.Y. Design and characterizations of novel biodegradable Zn-Cu-Mg alloys for potential biodegradable implants. Mater. Des. 2017, 117, 84–94. [Google Scholar] [CrossRef]
- Dambatta, M.S.; Izman, S.; Kurniawan, D.; Farahany, S.; Yahaya, B.; Hermawan, H. Influence of thermal treatment on microstructure, mechanical and degradation properties of Zn-3Mg alloy as potential biodegradable implant material. Mater. Des. 2015, 85, 431–437. [Google Scholar] [CrossRef]
- Piela, K.; Wrobel, M.; Sztwiertnia, K.; Jaskowski, M.; Kawalko, J.; Bieda, M.; Kiper, M.; Jarzebska, A. Zinc subjected to plastic deformation by complex loading and conventional extrusion: Comparison of the microstructure and mechanical properties. Mater. Des. 2017, 117, 111–120. [Google Scholar] [CrossRef]
- Cacciotti, I. Bivalent cationic ions doped bioactive glasses: The influence of magnesium, zinc, strontium and copper on the physical and biological properties. J. Mater. Sci. 2017, 52, 8812–8831. [Google Scholar] [CrossRef]
- Almutairi, A.; Sun, Z.H.; Al Safran, Z.; Poovathumkadavi, A.; Albader, S.; Ifdailat, H. Optimal scanning protocols for dual-energy CT angiography in peripheral arterial stents: An in vitro phantom study. Int. J. Mol. Sci. 2015, 16, 11531–11549. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.L.P.; Oliveira, A.C.; Costa, C.G.F.; Figueiredo, R.B.; Leite, M.D.; Pereira, M.M.; Lins, V.F.C.; Langdon, T.G. Effect of severe plastic deformation on the biocompatibility and corrosion rate of pure magnesium. J. Mater. Sci. 2017, 52, 5992–6003. [Google Scholar] [CrossRef]
- Kim, Y.K.; Park, I.S.; Lee, K.B.; Lee, S.J.; Bae, T.S.; Lee, M.H. Characterization and biocompatibility of a calcium-containing AZ31B alloy as a biodegradable material. J. Mater. Sci. 2015, 50, 4672–4682. [Google Scholar] [CrossRef]
- Gao, H.; Ye, W.B.; Zhang, Z.; Gao, L.L. Ratcheting behavior of ZEK100 magnesium alloy with various loading conditions and different immersing time. J. Mater. Res. 2017, 32, 2143–2152. [Google Scholar] [CrossRef]
- Gao, H.; Zhang, M.; Zhao, J.; Gao, L.L.; Li, M.S. In vitro and in vivo degradation and mechanical properties of ZEK100 magnesium alloy coated with alginate, chitosan and mechano-growth factor. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 63, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; He, W.; Pang, S.; Liaw, P.K.; Zhang, T. In vitro responses of bone-forming MC3T3-E1 pre-osteoblasts to biodegradable Mg-based bulk metallic glasses. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 68, 632–641. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.; Bhushan, B.; Jayaganthan, R.; Gopinath, P.; Agarwal, R.D.; Lahiri, D. Strengthening of Mg based alloy through grain refinement for orthopaedic application. J. Mech. Behav. Biomed. Mater. 2016, 59, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xu, C.; Jing, Y.; Lv, S.; Liu, S.; Fang, D.; Zhuang, J.; Zhang, M.; Wu, R. New horizon for high performance Mg-based biomaterial with uniform degradation behavior: Formation of stacking faults. Sci. Rep. 2015, 5, 13933. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Pang, S.J.; Liu, Y.; Sun, L.L.; Liaw, P.K.; Zhang, T. Biodegradable Mg-Zn-Ca-Sr bulk metallic glasses with enhanced corrosion performance for biomedical applications. Mater. Des. 2015, 67, 9–19. [Google Scholar] [CrossRef]
- Christie, J.K. Atomic structure of biodegradable Mg-based bulk metallic glass. Phys. Chem. Chem. Phys. 2015, 17, 12894–12898. [Google Scholar] [CrossRef] [PubMed]
- Ratna Sunil, B.; Sampath Kumar, T.S.; Chakkingal, U.; Nandakumar, V.; Doble, M. Nano-hydroxyapatite reinforced AZ31 magnesium alloy by friction stir processing: A solid state processing for biodegradable metal matrix composites. J. Mater. Sci. Mater. Med. 2014, 25, 975–988. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Forsyth, M.; Hinton, B.R. The effect of treatment temperature on corrosion resistance and hydrophilicity of an ionic liquid coating for Mg-based stents. ACS Appl. Mater. Interfaces 2014, 6, 18989–18997. [Google Scholar] [CrossRef] [PubMed]
- Willbold, E.; Kalla, K.; Bartsch, I.; Bobe, K.; Brauneis, M.; Remennik, S.; Shechtman, D.; Nellesen, J.; Tillmann, W.; Vogt, C.; et al. Biocompatibility of rapidly solidified magnesium alloy RS66 as a temporary biodegradable metal. Acta Biomater. 2013, 9, 8509–8517. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; He, Y.; Tao, H.; Zhang, Y.; Jiang, Y.; Zhang, X.; Zhang, S. Biocompatibility of magnesium-zinc alloy in biodegradable orthopedic implants. Int. J. Mol. Med. 2011, 28, 343–348. [Google Scholar] [PubMed]
- Qin, C.; Hu, Q.; Li, Y.; Wang, Z.; Zhao, W.; Louzguine-Luzgin, D.V.; Inoue, A. Novel bioactive Fe-based metallic glasses with excellent apatite-forming ability. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 69, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.J.; Wang, Y.G.; An, B.; Xu, H.; Liu, Y.; Zhang, L.C.; Ma, H.Y.; Wang, W.M. A practical anodic and cathodic curve intersection model to understand multiple corrosion potentials of fe-based glassy alloys in OH-contained solutions. PLoS ONE 2016, 11, e0146421. [Google Scholar] [CrossRef] [PubMed]
- Fagali, N.S.; Grillo, C.A.; Puntarulo, S.; Fernandez Lorenzo de Mele, M.A. Cytotoxicity of corrosion products of degradable Fe-based stents: Relevance of pH and insoluble products. Colloids Surf. B Biointerfaces 2015, 128, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wei, Q.; Li, Q.; Jiang, B.; Chen, Y.; Sun, Y. Development of Fe-based bulk metallic glasses as potential biomaterials. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 52, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Moszner, F.; Fischerauer, S.; Fiedler, M.; Martinelli, E.; Eichler, J.; Witte, F.; Willbold, E.; Schinhammer, M.; Meischel, M.; et al. Biodegradable Fe-based alloys for use in osteosynthesis: Outcome of an in vivo study after 52 weeks. Acta Biomater. 2014, 10, 3346–3353. [Google Scholar] [CrossRef] [PubMed]
- Schinhammer, M.; Gerber, I.; Hanzi, A.C.; Uggowitzer, P.J. On the cytocompatibility of biodegradable Fe-based alloys. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Schinhammer, M.; Hanzi, A.C.; Loffler, J.F.; Uggowitzer, P.J. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomater. 2010, 6, 1705–1713. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, Y.; Du, C.; Yan, B. Corrosion behaviors of amorphous and nanocrystalline Fe-based alloys in NaCl solution. J. Nanosci. Nanotechnol. 2010, 10, 7226–7230. [Google Scholar] [CrossRef] [PubMed]
- Mostaed, E.; Sikora-Jasinska, M.; Mostaed, A.; Loffredo, S.; Demir, A.G.; Previtali, B.; Mantovani, D.; Beanland, R.; Vedani, M. Novel Zn-based alloys for biodegradable stent applications: Design, development and in vitro degradation. J. Mech. Behav. Biomed. Mater. 2016, 60, 581–602. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Wang, Z.; Tay, S.L.; Zhu, T.; Gao, W. Effects of Mg on microstructure and corrosion properties of Zn–Mg alloy. J. Alloys Compd. 2014, 602, 101–107. [Google Scholar] [CrossRef]
- Bowen, P.K.; Drelich, J.; Goldman, J. Zinc exhibits ideal physiological corrosion behavior for bioabsorbable stents. Adv. Mater. 2013, 25, 2577–2582. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.; Li, H.F.; Zhao, K.; Bai, H.Y.; Wang, Y.B.; Zheng, Y.F. Development of cazn based glassy alloys as potential biodegradable bone graft substitute. J. Non-Cryst. Solids 2011, 357, 3830–3840. [Google Scholar] [CrossRef]
- Vojtech, D.; Kubasek, J.; Serak, J.; Novak, P. Mechanical and corrosion properties of newly developed biodegradable Zn-based alloys for bone fixation. Acta Biomater. 2011, 7, 3515–3522. [Google Scholar] [CrossRef] [PubMed]
- Saris, N.E.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium. An update on physiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Ramya, M.; Sarwat, S.G.; Udhayabanu, V.; Subramanian, S.; Raj, B.; Ravi, K.R. Role of partially amorphous structure and alloying elements on the corrosion behavior of Mg-Zn-Ca bulk metallic glass for biomedical applications. Mater. Des. 2015, 86, 829–835. [Google Scholar] [CrossRef]
- Berglund, I.S.; Brar, H.S.; Dolgova, N.; Acharya, A.P.; Keselowsky, B.G.; Sarntinoranont, M.; Manuel, M.V. Synthesis and characterization of Mg-Ca-Sr alloys for biodegradable orthopedic implant applications. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1524–1534. [Google Scholar] [CrossRef] [PubMed]
- Kusnierczyk, K.; Basista, M. Recent advances in research on magnesium alloys and magnesium-calcium phosphate composites as biodegradable implant materials. J. Biomater. Appl. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hampp, C.; Angrisani, N.; Reifenrath, J.; Bormann, D.; Seitz, J.M.; Meyer-Lindenberg, A. Evaluation of the biocompatibility of two magnesium alloys as degradable implant materials in comparison to titanium as non-resorbable material in the rabbit. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Hort, N.; Huang, Y.; Fechner, D.; Stormer, M.; Blawert, C.; Witte, F.; Vogt, C.; Drucker, H.; Willumeit, R.; Kainer, K.U.; et al. Magnesium alloys as implant materials—Principles of property design for Mg-RE alloys. Acta Biomater. 2010, 6, 1714–1725. [Google Scholar] [CrossRef] [PubMed]
- Hanzi, A.C.; Gerber, I.; Schinhammer, M.; Loffler, J.F.; Uggowitzer, P.J. On the in vitro and in vivo degradation performance and biological response of new biodegradable Mg-Y-Zn alloys. Acta Biomater. 2010, 6, 1824–1833. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Lin, J.; Wen, C.; Zhang, D.; Li, Y. Mechanical properties, in vitro corrosion and biocompatibility of newly developed biodegradable Mg-Zr-Sr-Ho alloys for biomedical applications. Sci. Rep. 2016, 6, 31990. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Yang, H.T.; Zheng, Y.F.; Zhou, F.Y.; Qiu, K.J.; Wang, X. Design and characterizations of novel biodegradable ternary Zn-based alloys with IIA nutrient alloying elements Mg, Ca and Sr. Mater. Des. 2015, 83, 95–102. [Google Scholar] [CrossRef]
- Hermawan, H.; Purnama, A.; Dube, D.; Couet, J.; Mantovani, D. Fe-Mn alloys for metallic biodegradable stents: Degradation and cell viability studies. Acta Biomater. 2010, 6, 1852–1860. [Google Scholar] [CrossRef] [PubMed]
- Peuster, M.; Wohlsein, P.; Brugmann, M.; Ehlerding, M.; Seidler, K.; Fink, C.; Brauer, H.; Fischer, A.; Hausdorf, G. A novel approach to temporary stenting: Degradable cardiovascular stents produced from corrodible metal-results 6–18 months after implantation into New Zealand white rabbits. Heart 2001, 86, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Seitz, J.M.; Durisin, M.; Goldman, J.; Drelich, J.W. Recent advances in biodegradable metals for medical sutures: A critical review. Adv. Healthc. Mater. 2015, 4, 1915–1936. [Google Scholar] [CrossRef] [PubMed]
- Vojtech, D.; Kubasek, J.; Capek, J.; Pospisilova, I. Comparative Mechanical and Corrosion Studies on Magnesium, Zinc and Iron Alloys as Biodegradable Metals. Mater. Technol. 2015, 49, 877–882. [Google Scholar] [CrossRef]
- Bowen, P.K.; Shearier, E.R.; Zhao, S.; Guillory, R.J.; Zhao, F.; Goldman, J.; Drelich, J.W. Biodegradable Metals for Cardiovascular Stents: From Clinical Concerns to Recent Zn-Alloys. Adv. Healthc. Mater. 2016, 5, 1121–1140. [Google Scholar] [CrossRef] [PubMed]
- Sigwart, U.; Puel, J.; Mirkovitch, V.; Joffre, F.; Kappenberger, L. Intravascular stents to prevent occlusion and restenosis after transluminal angioplasty. N. Engl. J. Med. 1987, 316, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H.; Dube, D.; Mantovani, D. Degradable metallic biomaterials: Design and development of Fe-Mn alloys for stents. J. Biomed. Mater. Res. Part A 2010, 93, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moravej, M.; Mantovani, D. Biodegradable metals for cardiovascular stent application: Interests and new opportunities. Int. J. Mol. Sci. 2011, 12, 4250–4270. [Google Scholar] [CrossRef] [PubMed]
- Peeters, P.; Bosiers, M.; Verbist, J.; Deloose, K.; Heublein, B. Preliminary results after application of absorbable metal stents in patients with critical limb ischemia. J. Endovasc. Ther. 2005, 12, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zartner, P.; Cesnjevar, R.; Singer, H.; Weyand, M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter. Cardiovasc. Interv. 2005, 66, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Erbel, R.; Di Mario, C.; Bartunek, J.; Bonnier, J.; de Bruyne, B.; Eberli, F.R.; Erne, P.; Haude, M.; Heublein, B.; Horrigan, M.; et al. Temporary scaffolding of coronary arteries with bioabsorbable magnesium stents: A prospective, non-randomised multicentre trial. Lancet 2007, 369, 1869–1875. [Google Scholar] [CrossRef]
- Chen, Y.J.; Xu, Z.G.; Smith, C.; Sankar, J. Recent advances on the development of magnesium alloys for biodegradable implants. Acta Biomater. 2014, 10, 4561–4573. [Google Scholar] [CrossRef] [PubMed]
- Haude, M.; Erbel, R.; Erne, P.; Verheye, S.; Degen, H.; Vermeersch, P.; Weissman, N.; Prati, F.; Bruining, N.; Waksman, R.; et al. Safety and performance of the DRug-Eluting Absorbable Metal Scaffold (DREAMS) in patients with de novo coronary lesions: 3-year results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. Eurointervention 2016, 12, e160–e166. [Google Scholar] [CrossRef] [PubMed]
- Our Vascular Products Represent Deep Collaboration between Scientists, Doctors, and Patients over Decades. Available online: https://www.vascular.abbott/int/products/products-home.html (accessed on 18 December 2017).
- Newsroom. Available online: http://news.bostonscientific.com/ (accessed on 18 December 2017).
- Affatato, S.; Ruggiero, A.; Merola, M.; Logozzo, S. Does metal transfer differ on retrieved Biolox Delta composites femoral heads? Surface investigation on three Biolox generations from a biotribological point of view. Compos. Part B Eng. 2017, 113, 164–173. [Google Scholar] [CrossRef]
- Liu, X.W.; Sun, J.K.; Yang, Y.H.; Zhou, F.Y.; Pu, Z.J.; Li, L.; Zheng, Y.F. Microstructure, mechanical properties, in vitro degradation behavior and hemocompatibility of novel Zn-Mg-Sr alloys as biodegradable metals. Mater. Lett. 2016, 162, 242–245. [Google Scholar] [CrossRef]
- Saleh, M.M.; Touny, A.H.; Al-Omair, M.A.; Saleh, M.M. Biodegradable/biocompatible coated metal implants for orthopedic applications. Bio-Med. Mater. Eng. 2016, 27, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Ding, W. Opportunities and challenges for the biodegradable magnesium alloys as next-generation biomaterials. Regen. Biomater. 2016, 3, 79–86. [Google Scholar] [CrossRef] [PubMed]
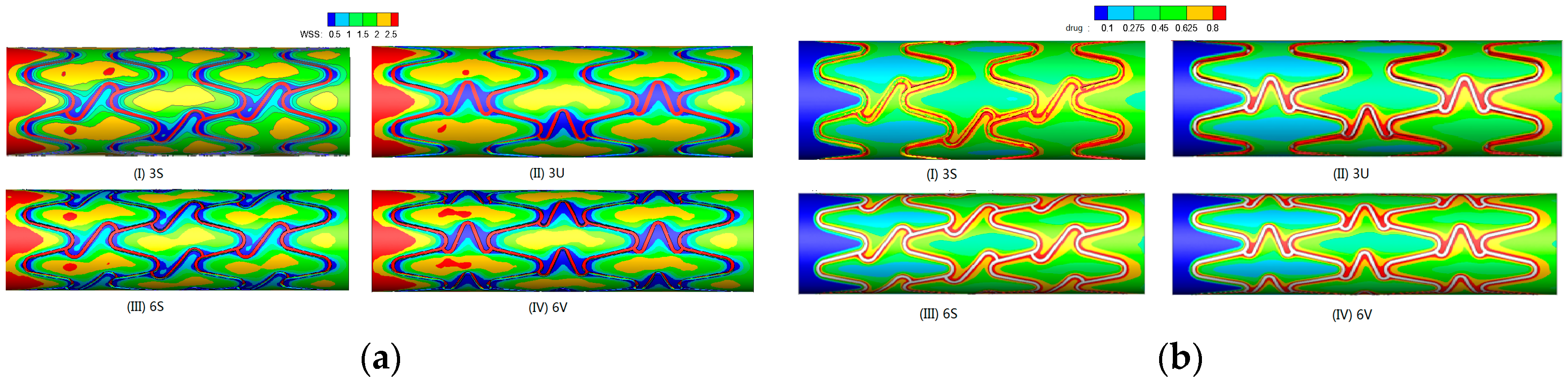
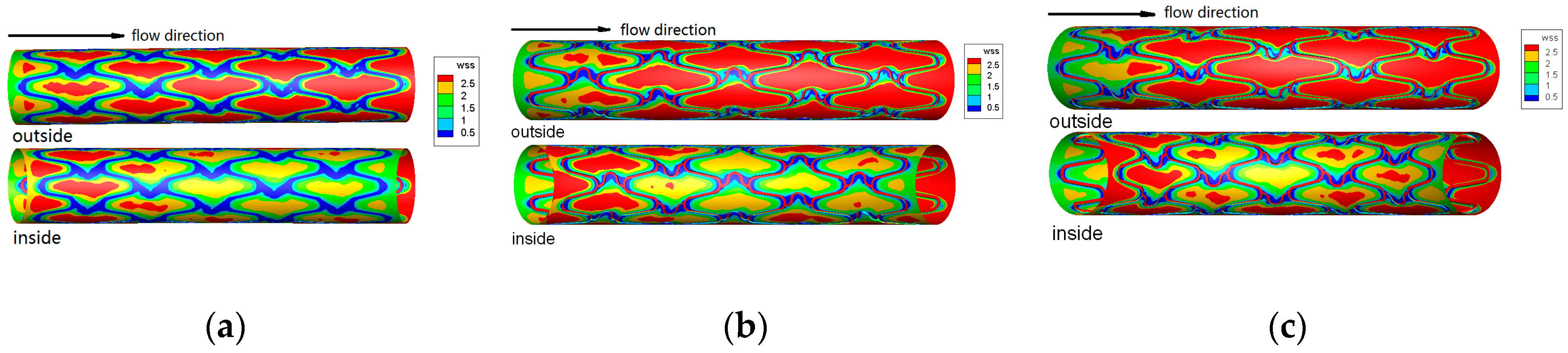




| Types of BMs | Materials | Published Time | Progress & Findings | Potential Applications |
|---|---|---|---|---|
| Mg-based BMs | ZEK100 [94] | 2017 | Biodegradable behavior of ZEK100 with various loading conditions were studied. | No declaration |
| ZEK100 [95] | 2016 | Multilayered coatings carrying sodium alginate (ALG) were placed on ZEK100 to reduce the degradation rate. | Bone tissue repair | |
| Mg-Zn-Ca-Sr bulk metallic glasses (BMGs) [96] | 2016 | In vitro responses of bone-forming MC3T3-E1 pre-osteoblasts to Mg-Zn-Ca-Sr BMGs were studied. | No declaration | |
| Mg-3 wt % Zn alloy (MZ3) [97] | 2016 | Hot rolled Mg-3 wt % Zn alloy (MZ3) has been investigated for its potential in orthopaedic implant. | Orthopaedic implantations | |
| Mg-8Er-1Zn [98] | 2015 | A novel Mg-8Er-1Zn alloy with the ultimate tensile strength (318 MPa), tensile yield strength (207 MPa) and elongation (21%) were reported. | No declaration | |
| Mg-Zn-Ca-Sr alloy [99] | 2015 | Add minor Sr would improve glass-forming ability, mechanical properties, enhance and adjustable corrosion performance. | Orthopedic implantations | |
| Mg60Zn35Ca5 [100] | 2015 | Used first-principles molecular dynamics simulations to elucidate the structure of Mg60Zn35Ca5. | No declaration | |
| Nano-hydroxyapatite (nHA) reinforced AZ31 [101] | 2014 | Embedded nano-hydroxyapatite (nHA) particles enhance the biomineralization and control the degradation. | Skeletal implants | |
| AZ31 [102] | 2014 | Surface coating for Mg alloy AZ31 to control its corrosion rate. | Stents | |
| AZ31 [102] | 2014 | Report a new surface coating for Mg alloy AZ31 based on a low-toxicity ionic liquid, tributyl(methyl)phosphoniumdiphenylphosphate, to control its corrosion rate. | Stents | |
| RS66 [103] | 2013 | In vitro and in vivo experiments were conducted to analyze the biodegradation behavior and the biocompatibility. | Prosthesis implantation | |
| Mg-Zn [104] | 2011 | Biocompatibility test and biodagradation in vivo. | Orthopaedic implantations | |
| Fe-based BMs | (Fe0.75B0.15Si0.1)100-xNbx (x = 0, 1 and 3 wt%) [105] | 2016 | Alloys exhibit excellent apatite-forming ability in simulated body fluids. | Stents and orthopedic implants |
| Fe-based glassy alloys [106] | 2016 | It studied the multiple corrosion potentials in alkaline solution. | No declaration | |
| Fe-based metallic materials [107] | 2015 | Cytotoxicity of corrosion products of Fe-based stents relevant of pH and insoluble products were studied. | Stents | |
| Fe80-x-yCrxMoyP13C7 bulk metallic glasses [108] | 2015 | Alloys exhibit no cytotoxicity to NIH3T3 cells, and exhibit high corrosion resistance and excellent biocompatibility. | No declaration | |
| (Fe-10Mn-1Pd, Fe-21 Mn-0.7C-1Pd) [109] | 2014 | The study investigated the degradation performance of three Fe-based materials in a growing rat skeleton over 1 year. | No declaration | |
| Fe-Mn-C-Pd alloys [110] | 2013 | The research studied the alloying elements’ influence on metabolic processes. | No declaration | |
| Fe-Mn-Pd alloys [111] | 2010 | Fe-based alloys offering both an enhanced degradation rate and suitable strength and ductility. | Medical applications | |
| Fe(73.5)Si(13.5)B9Nb3Cu1 alloy [112] | 2010 | Studied the corrosion behaviors of amorphous and nanocrystalline Fe-based alloys in NaCl solution | No declaration | |
| Zn-based BMs | Zn-Mg and two Zn-Al binary alloys [113] | 2016 | Alloys were fabricated by casting process and hot extrusion. Tube extrusion was applied to produce stents. Corrosion tests were performed. | Stents |
| Zn-Mg alloy [114] | 2015 | Zn-Mg alloys with different Mg contents were prepared by melting-casting method. The Zn-3 wt % Mg alloy contributes to a general corrosion. | No declaration | |
| Zn alloys [115] | 2013 | Zinc exhibits ideal physiological corrosion behavior for bioabsorbable stents. | Stents | |
| CaZn based bulk glassy alloy [116] | 2011 | CaZn based glassy alloys shows low Young’s modulus, high fracture strength, good corrosion resistance and cytocompatibility. | Orthopaedic implantations | |
| Zn-Mg alloys containing 3 wt % Mg [117] | 2011 | The corrosion rates of the Zn-Mg alloys were determined to be significantly lower than those of Mg and AZ91HP alloys. | No declaration |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, K.; Zhou, C.; Fan, H.; Fan, Y.; Jiang, Q.; Song, P.; Fan, H.; Chen, Y.; Zhang, X. Bio-Functional Design, Application and Trends in Metallic Biomaterials. Int. J. Mol. Sci. 2018, 19, 24. https://doi.org/10.3390/ijms19010024
Yang K, Zhou C, Fan H, Fan Y, Jiang Q, Song P, Fan H, Chen Y, Zhang X. Bio-Functional Design, Application and Trends in Metallic Biomaterials. International Journal of Molecular Sciences. 2018; 19(1):24. https://doi.org/10.3390/ijms19010024
Chicago/Turabian StyleYang, Ke, Changchun Zhou, Hongsong Fan, Yujiang Fan, Qing Jiang, Ping Song, Hongyuan Fan, Yu Chen, and Xingdong Zhang. 2018. "Bio-Functional Design, Application and Trends in Metallic Biomaterials" International Journal of Molecular Sciences 19, no. 1: 24. https://doi.org/10.3390/ijms19010024
APA StyleYang, K., Zhou, C., Fan, H., Fan, Y., Jiang, Q., Song, P., Fan, H., Chen, Y., & Zhang, X. (2018). Bio-Functional Design, Application and Trends in Metallic Biomaterials. International Journal of Molecular Sciences, 19(1), 24. https://doi.org/10.3390/ijms19010024







