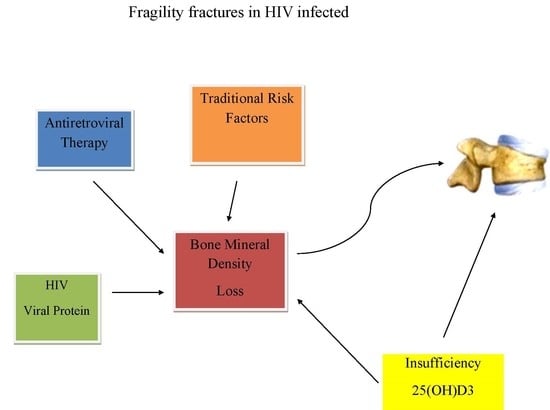
Vitamin D Status and the Relationship with Bone Fragility Fractures in HIV-Infected Patients: A Case Control Study
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Bone Ultrasound Parameters and Vertebral Fractures Assessment
4.3. Biochemical Data
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Stone, B.; Dockrell, D.; Bowman, C.; McCloskey, E. HIV and bone disease. Arch. Biochem. Biophys. 2010, 503, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Mallon, P.W. HIV and bone mineral density. Curr. Opin. Infect. Dis. 2010, 23, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cazanave, C.; Dupon, M.; Lavignolle-Aurillac, V.; Barthe, N.; Lawson-Ayayi, S.; Mehsen, N.; Mercié, P.; Morlat, P.; Thiébaut, R.; Dabis, F.; et al. Reduced bone mineral density in HIV-infected patients: Prevalence and associated factors. AIDS 2008, 22, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Gallant, J.E.; Staszewski, S.; Pozniak, A.L.; DeJesus, E.; Suleiman, J.M.; Miller, M.D.; Coakley, D.F.; Lu, B.; Toole, J.J.; Cheng, A.K.; et al. Efficacy and safety of tenofovir DF vs. stavudine in combination therapy in antiretroviral-naive patients: A 3-year randomized trial. JAMA 2004, 292, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.T.; Ruppe, M.D.; Kassner, R.; Kumar, P.; Kehoe, T.; Dobs, A.S.; Timpone, J. Reduced bone mineral density in human immunodeficiency virus-infected patients and its association with increased central adiposity and postload hyperglycemia. J. Clin. Endocrinol. Metab. 2004, 89, 1200–1206. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Alhambra, D.; Güerri-Fernández, R.; de Vries, F.; Lalmohamed, A.; Bazelier, M.; Starup-Linde, J.; Diez-Perez, A.; Cooper, C.; Vestergaard, P. HIV infection and its association with an excess risk of clinical fractures: A nationwide case-control study. J. Acquir. Immune Defic. Syndr. 2014, 66, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Güerri-Fernandez, R.; Vestergaard, P.; Carbonell, C.; Knobel, H.; Avilés, F.F.; Castro, A.S.; Nogués, X.; Prieto-Alhambra, D.; Diez-Perez, A. HIV infection is strongly associated with hip fracture risk, independently of age, gender, and comorbidities: A population-based cohort study. J. Bone Miner. Res. 2013, 28, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Torti, C.; Mazziotti, G.; Soldini, P.A.; Focà, E.; Maroldi, R.; Gotti, D.; Carosi, G.; Giustina, A. High prevalence of radiological vertebral fractures in HIV-infected males. Endocrine 2012, 41, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Fakruddin, J.M.; Laurence, J. HIV-1 Vpr enhances production of receptor of activated NF-kappaB ligand (RANKL) via potentiation of glucocorticoid receptor activity. Arch. Virol. 2005, 150, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Malizia, A.P.; Cotter, E.; Chew, N.; Powderly, W.G.; Doran, P.P. HIV protease inhibitors selectively induce gene expression alterations associated with reduced calcium deposition in primary human osteoblasts. AIDS Res. Hum. Retroviruses 2007, 23, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.W.; Wei, S.; Faccio, R.; Takeshita, S.; Tebas, P.; Powderly, W.G.; Teitelbaum, S.L.; Ross, F.P. The HIV protease inhibitor ritonavir blocks osteoclastogenesis and function by impairing RANKL-induced signaling. J. Clin. Investig. 2004, 114, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Bolland, M.J.; Grey, A.B.; Gamble, G.D.; Reid, I.R. CLINICAL Review: Low body weight mediates the relationship between HIV infection and low bone mineral density: A meta-analysis. J. Clin. Endocrinol. Metab. 2007, 92, 4522–4528. [Google Scholar] [CrossRef] [PubMed]
- Hasse, B.; Ledergerber, B.; Furrer, H.; Battegay, M.; Hirschel, B.; Cavassini, M.; Bertisch, B.; Bernasconi, E.; Weber, R.; Swiss HIV Cohort Study. Morbidity and aging in HIV-infected persons: The Swiss HIV cohort study. Clin. Infect. Dis. 2011, 53, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Young, B.; Dao, C.N.; Buchacz, K.; Baker, R.; Brooks, J.T.; HIV Outpatient Study (HOPS) Investigators. Increased rates of bone fracture among HIV-infected persons in the HIV Outpatient Study (HOPS) compared with the US general population, 2000–2006. Clin. Infect. Dis. 2011, 52, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Gazzola, L.; Savoldi, A.; Bai, F.; Magenta, A.; Dziubak, M.; Pietrogrande, L.; Tagliabue, L.; del Sole, A.; Bini, T.; Marchetti, G.; et al. Assessment of radiological vertebral fractures in HIV-infected patients: Clinical implications and predictive factors. HIV Med. 2015, 16, 563–571. [Google Scholar] [CrossRef] [PubMed]
- El Maghraoui, A.; Ouzzif, Z.; Mounach, A.; Rezqi, A.; Achemlal, L.; Bezza, A.; Tellal, S.; Dehhaoui, M.; Ghozlani, I. Hypovitaminosis D and prevalent asymptomatic vertebral fractures in Moroccan postmenopausal women. BMC Womens Health 2012, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Arnsten, J.H.; Freeman, R.; Howard, A.A.; Floris-Moore, M.; Lo, Y.; Klein, R.S. Decreased bone mineral density and increased fracture risk in aging men with or at risk for HIV infection. AIDS 2007, 21, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Collin, F.; Duval, X.; Le Moing, V.; Piroth, L.; Al Kaied, F.; Massip, P.; Villes, V.; Chêne, G.; Raffi, F. Ten-year incidence and risk factors of bone fractures in a cohort of treated HIV1-infected adults. AIDS 2009, 23, 1021–1024. [Google Scholar] [CrossRef] [PubMed]
- Triant, V.A.; Brown, T.T.; Lee, H.; Grinspoon, S.K. Fracture prevalence among human immunodeficiency virus (HIV)-infected versus non-HIV-infected patients in a large U.S. healthcare system. J. Clin. Endocrinol. Metab. 2008, 93, 3499–3504. [Google Scholar] [CrossRef] [PubMed]
- Calmy, A.; Fux, C.A.; Norris, R.; Vallier, N.; Delhumeau, C.; Samaras, K.; Hesse, K.; Hirschel, B.; Cooper, D.A.; Carr, A. Low bone mineral density, renal dysfunction, and fracture risk in HIV infection: A cross-sectional study. J. Infect. Dis. 2009, 200, 1746–1754. [Google Scholar] [CrossRef] [PubMed]
- Samaras, K.; Pett, S.; Gowers, A.; McMurchie, M.; Cooper, D.A. Iatrogenic Cushing’s syndrome with osteoporosis and secondary adrenal failure in human immunodeficiency virus-infected patients receiving inhaled corticosteroids and ritonavir-boosted protease inhibitors: Six cases. J. Clin. Endocrinol. Metab. 2005, 90, 4394–4398. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.B.; Gerstoft, J.; Kronborg, G.; Larsen, C.S.; Pedersen, C.; Pedersen, G.; Obel, N. Incidence of low and high-energy fractures in persons with and without HIV infection: A Danish population-based cohort study. AIDS 2012, 26, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Bedimo, R.; Maalouf, N.M.; Zhang, S.; Drechsler, H.; Tebas, P. Osteoporotic fracture risk associated with cumulative exposure to tenofovir and other antiretroviral agents. AIDS 2012, 26, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Bruera, D.; Luna, N.; David, D.O.; Bergoglio, L.M.; Zamudio, J. Decreased bone mineral density in HIV-infected patients is independent of antiretroviral therapy. AIDS 2003, 17, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.C.; Glüer, C.C.; Genant, H.K.; Stone, K. Quantitative ultrasound and vertebral fracture in postmenopausal women. J. Bone Miner. Res. 1995, 10, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.C.; Glüer, C.C.; Cauley, J.A.; Vogt, T.M.; Ensrud, K.E.; Genant, H.K.; Black, D.M. Broadband ultrasound attenuation predicts fractures strongly and independently of densitometry in older women. A prospective study. Study of Osteoporotic Fractures Research Group. Arch. Intern. Med. 1997, 157, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.C.; Ewing, S.K.; Cauley, J.A.; Ensrud, K.E.; Cummings, S.R.; Orwoll, E.S. Quantitative ultrasound predicts hip and non-spine fracture in men: The MrOS study. Osteoporos. Int. 2007, 18, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Glüer, C.C.; Wu, C.Y.; Genant, H.K. Broadband ultrasound attenuation signals depend on trabecular orientation: An in vitro study. Osteoporos. Int. 1993, 3, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, A.; Romeo, A.; Floccari, F.; Aloisi, E.; Atteritano, M.; Cincotta, M.; Aloisi, C.; Pizzoleo, M.A.; Ruello, A.; Artemisia, A.; et al. Osteoprotegerin and bone mineral density in hemodiafiltration patients. Ren. Fail. 2005, 27, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Catalano, A.; Morabito, N.; Agostino, R.M.; Basile, G.; Gaudio, A.; Atteritano, M.; Natale, G.; Xourafa, A.; Morini, E.; Adamo, V.; et al. Bone health assessment by quantitative ultrasound and dual-energy X-ray absorptiometry in postmenopausal women with breast cancer receiving aromatase inhibitors. Menopause 2017, 24, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Klassen, K.M.; Fairley, C.K.; Kimlin, M.G.; Hocking, J.; Kelsall, L.; Ebeling, P.R. Vitamin D deficiency is common in HIV-infected southern Australian adults. Antivir. Ther. 2016, 21, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Jao, J.; Freimanis, L.; Mussi-Pinhata, M.M.; Cohen, R.A.; Monteiro, J.P.; Cruz, M.L.; Sperling, R.S.; Branch, A.; Siberry, G.K.; NISDI LILAC Protocol. Low vitamin D status among pregnant Latin American and Caribbean women with HIV Infection. Int. J. Gynaecol. Obstet. 2015, 130, 54–58. [Google Scholar] [PubMed]
- Cervero, M.; Agud, J.L.; García-Lacalle, C.; Alcázar, V.; Torres, R.; Jusdado, J.J.; Moreno Guillén, S. Prevalence of vitamin D deficiency and its related risk factor in a Spanish cohort of adult HIV-infected patients: Effects of antiretroviral therapy. AIDS Res. Hum. Retroviruses 2012, 28, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W. Circulating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: Implications for establishing a new effective dietary intake recommendation for vitamin D. J. Nutr. 2005, 135, 317–322. [Google Scholar] [PubMed]
- Parfitt, A.M. Dietary risk factors for age-related bone loss and fractures. Lancet 1983, 2, 1181–1185. [Google Scholar] [CrossRef]
- Trochoutsou, A.I.; Kloukina, V.; Samitas, K.; Xanthou, G. Vitamin-D in the Immune System: Genomic and Non-Genomic Actions. Mini. Rev. Med. Chem. 2015, 15, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, V.; Oboroceanu, T.; Zugun-Eloae, F. Current status in vitamin D and regulatory T cells—Immunological implications. Rev. Med. Chir. Soc. Med. Nat. Iasi 2013, 117, 965–973. [Google Scholar] [PubMed]
- Mansueto, P.; Seidita, A.; Vitale, G.; Gangemi, S.; Iaria, C.; Cascio, A. Vitamin D Deficiency in HIV Infection: Not Only a Bone Disorder. Biomed. Res. Int. 2015, 2015, 735615. [Google Scholar] [CrossRef] [PubMed]
- Anandaiah, A.; Sinha, S.; Bole, M.; Sharma, S.K.; Kumar, N.; Luthra, K.; Li, X.; Zhou, X.; Nelson, B.; Han, X.; et al. Vitamin D rescues impaired Mycobacterium tuberculosis-mediated tumor necrosis factor release in macrophages of HIV-seropositive individuals through an enhanced Toll-like receptor signaling pathway in vitro. Infect. Immun. 2013, 81, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Genant, H.K.; Wu, C.Y.; van Kuijk, C.; Nevitt, M.C. Vertebral fracture assessment using a semiquantitative technique. J. Bone Miner. Res. 1993, 8, 1137–1148. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | HIV (n = 100) | Controls (n = 100) | p |
|---|---|---|---|
| Age (years) | 45.36 ± 12.02 | 44.85 ± 9.30 | 0.73 |
| Body Mass Index (kg/m2) | 25.01 ± 4.23 | 24.80 ± 4.65 | 0.73 |
| Sex (F/M) | 84/16 | 84/16 | 1.00 |
| Duration of disease (years) | 16.29 ± 6.48 | 0 | N/A |
| Current tobacco use, n (%) | 22 (22) | 20 (20) | 0.88 |
| Alcohol intake (mL/week) | 450 ± 100 | 450 ± 80 | 1.00 |
| Calcium intake (mg/day) | 510 ± 117.88 | 520 ± 122.53 | 0.55 |
| Food energy (Kcal/day) | 1350 ± 200 | 1360 ± 150 | 0.23 |
| Low physical activity | 70 | 68 | 0.66 |
| Supplementation with calcium, n | 0 | 0 | N/A |
| Supplementation with vitamin D, n | 0 | 0 | N/A |
| Current steroid use, n (%) | 0 | 0 | N/A |
| Antiretroviral therapy exposure | |||
| Naive, n (%) | 6 (6) | 0 | N/A |
| Experienced, n (%) | 94 (94) | 0 | N/A |
| Sunlight exposure >5 h/week, n (%) | 26 (26) | 26 (26) | 0.52 |
| Family history of osteoporosis | |||
| and/or fractures, n (%) | 12 (12) | 14 (14) | 0.79 |
| Fall history, n (%) | 15 (15) | 17 (17) | 0.87 |
| Parameter | HIV (n = 100) | Control Group (n = 100) |
|---|---|---|
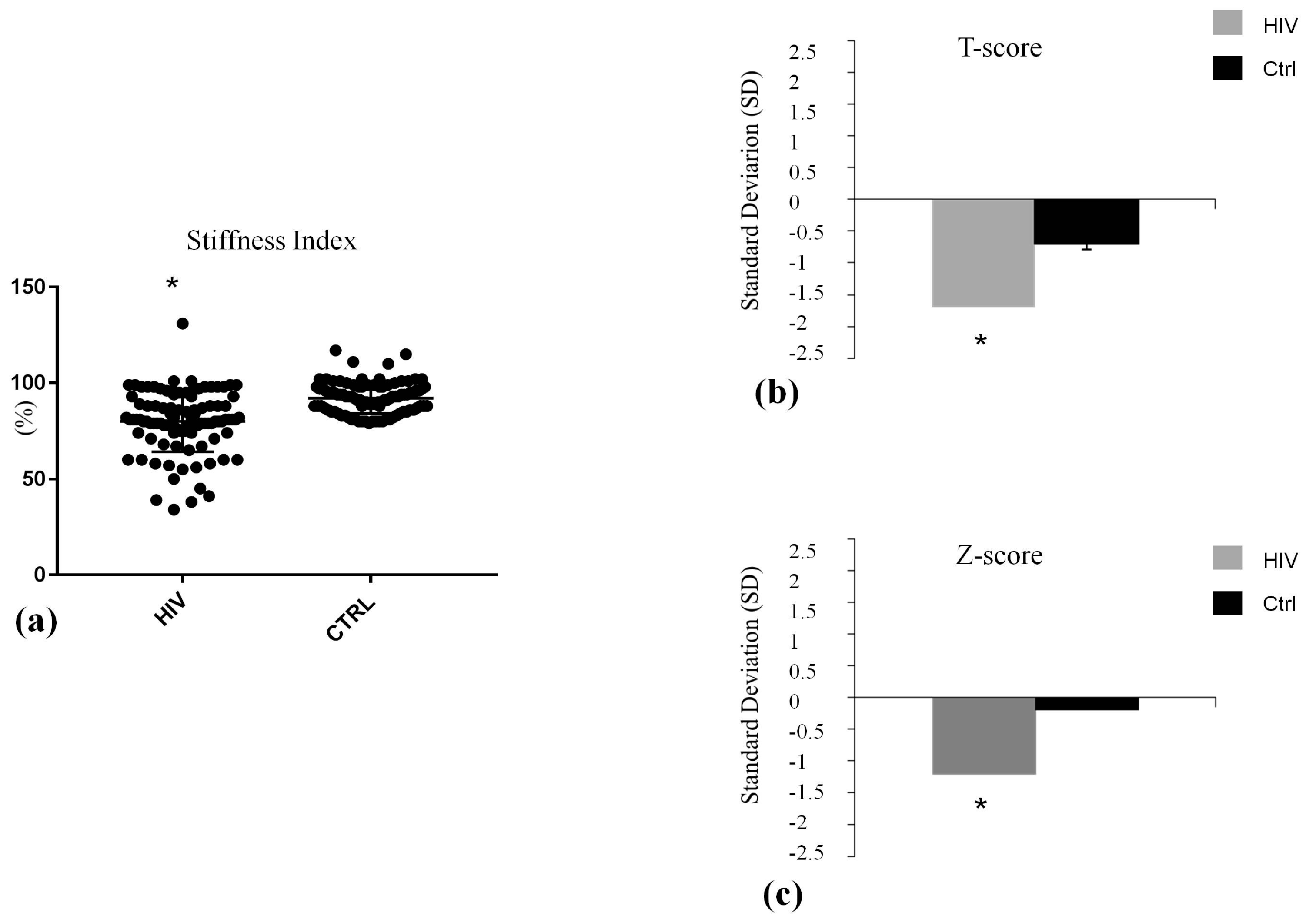
| Vertebral fractures (VFs), n (%) | 16 (16) * | 2 (2) |
| Stiffness index (SI) (%) | 80.58 ± 19.95 § | 92.18 ± 8.06 |
| T-Score (D.S.) | −1.70 ± 0.50 § | -0.70 ± 0.30 |
| Z-Score (D.S.) | −1.20 ± 0.40 § | -0.10 ± 0.30 |
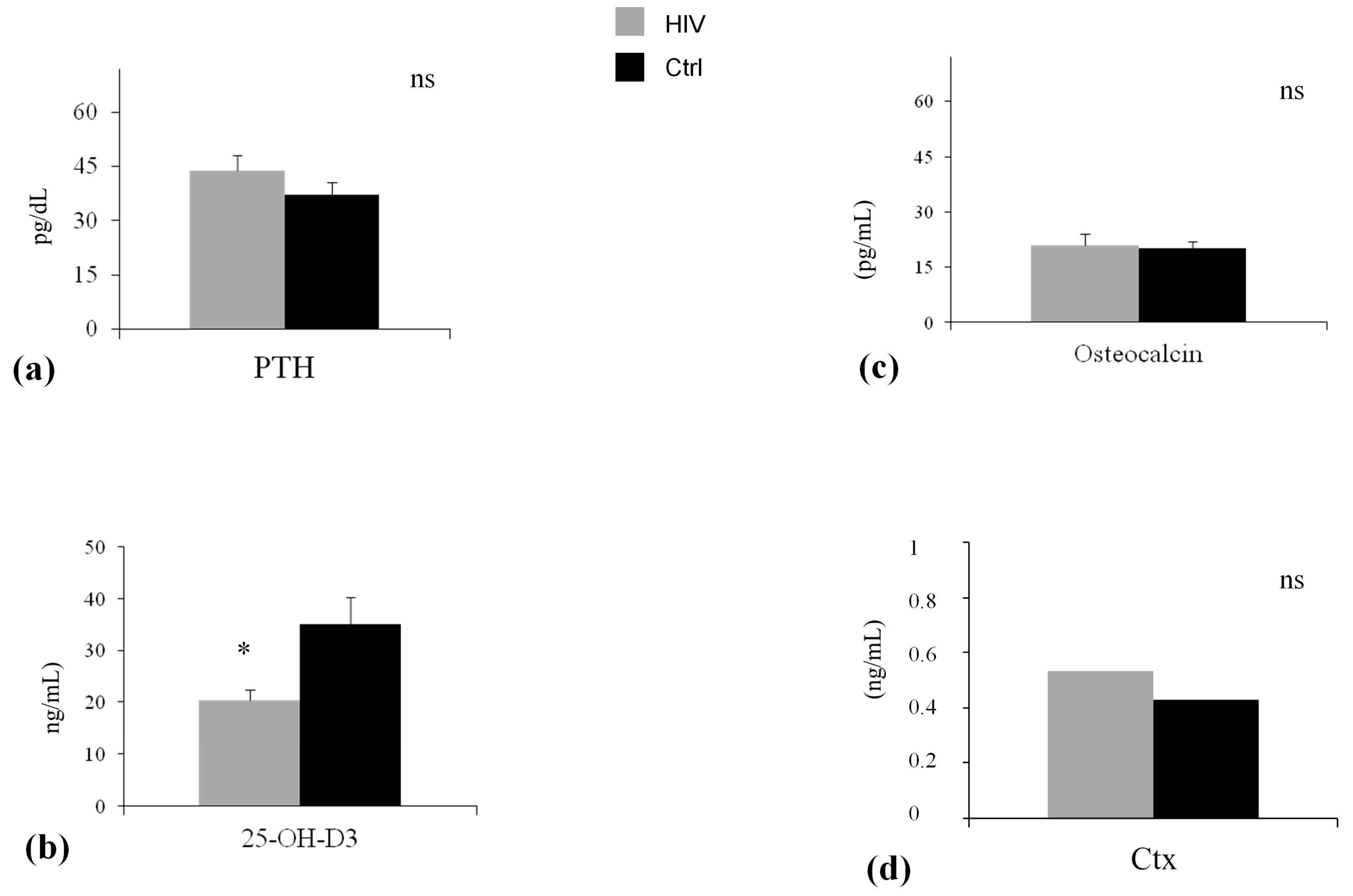
| Osteocalcin (pg/mL) | 21.31 ± 4.95 | 20.33 ± 4.35 |
| C-terminal telopeptide (ng/mL) | 0.53 ± 0.28 | 0.48 ± 0.35 |
| Parathyroid hormone level (pg/dL) | 43.72 ± 25.40 | 37.64 ± 23.60 |
| 25-hydroxivitamin D3 (ng/mL) | 20.29 ± 4.05 § | 35.77 ± 6.50 |
| Calcium (mg/dL) | 9.02 ± 0.47 | 9.12 ± 0.46 |
| Phophate (mg/dL) | 3.11 ± 0.57 | 3.14 ± 0.43 |
| Urinary calcium (mg/24 h) | 210.29 ± 84.05 | 213.93 ± 140.35 |
| Urinary phophate (mg/24 h) | 1010.29 ± 260.05 | 1063.53 ± 461.50 |
| Vitamin A (g/dL) | 62.65 ± 13.89 | 62.33 ± 13.22 |
| Creatinine (mg/dL) | 0.88 ± 0.07 | 0.89 ± 0.09 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atteritano, M.; Mirarchi, L.; Venanzi-Rullo, E.; Santoro, D.; Iaria, C.; Catalano, A.; Lasco, A.; Arcoraci, V.; Lo Gullo, A.; Bitto, A.; et al. Vitamin D Status and the Relationship with Bone Fragility Fractures in HIV-Infected Patients: A Case Control Study. Int. J. Mol. Sci. 2018, 19, 119. https://doi.org/10.3390/ijms19010119
Atteritano M, Mirarchi L, Venanzi-Rullo E, Santoro D, Iaria C, Catalano A, Lasco A, Arcoraci V, Lo Gullo A, Bitto A, et al. Vitamin D Status and the Relationship with Bone Fragility Fractures in HIV-Infected Patients: A Case Control Study. International Journal of Molecular Sciences. 2018; 19(1):119. https://doi.org/10.3390/ijms19010119
Chicago/Turabian StyleAtteritano, Marco, Luigi Mirarchi, Emmanuele Venanzi-Rullo, Domenico Santoro, Chiara Iaria, Antonino Catalano, Antonino Lasco, Vincenzo Arcoraci, Alberto Lo Gullo, Alessandra Bitto, and et al. 2018. "Vitamin D Status and the Relationship with Bone Fragility Fractures in HIV-Infected Patients: A Case Control Study" International Journal of Molecular Sciences 19, no. 1: 119. https://doi.org/10.3390/ijms19010119
APA StyleAtteritano, M., Mirarchi, L., Venanzi-Rullo, E., Santoro, D., Iaria, C., Catalano, A., Lasco, A., Arcoraci, V., Lo Gullo, A., Bitto, A., Squadrito, F., & Cascio, A. (2018). Vitamin D Status and the Relationship with Bone Fragility Fractures in HIV-Infected Patients: A Case Control Study. International Journal of Molecular Sciences, 19(1), 119. https://doi.org/10.3390/ijms19010119