Bovine Lactoferrin Inhibits Dengue Virus Infectivity by Interacting with Heparan Sulfate, Low-Density Lipoprotein Receptor, and DC-SIGN
Abstract
:1. Introduction
2. Results
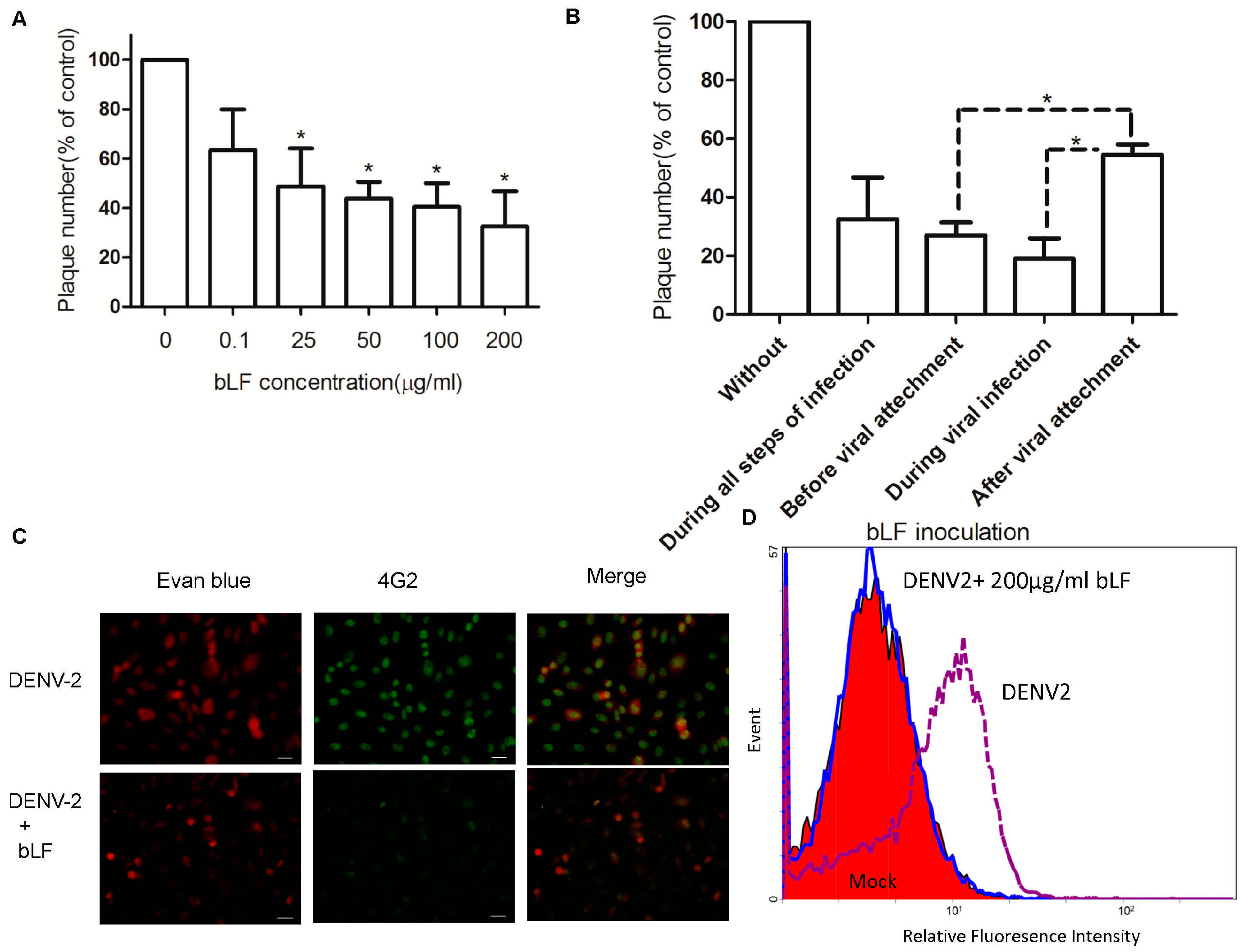
2.1. Bovine LF Blocks DENV Infection
2.2. Steps of DENV-2 Infection Inhibited by bLF
2.3. Bovine LF Inhibits DENV-2 Binding to Cellular Membrane
2.4. Role of HS in Anti-DENV-2 Activity of bLF
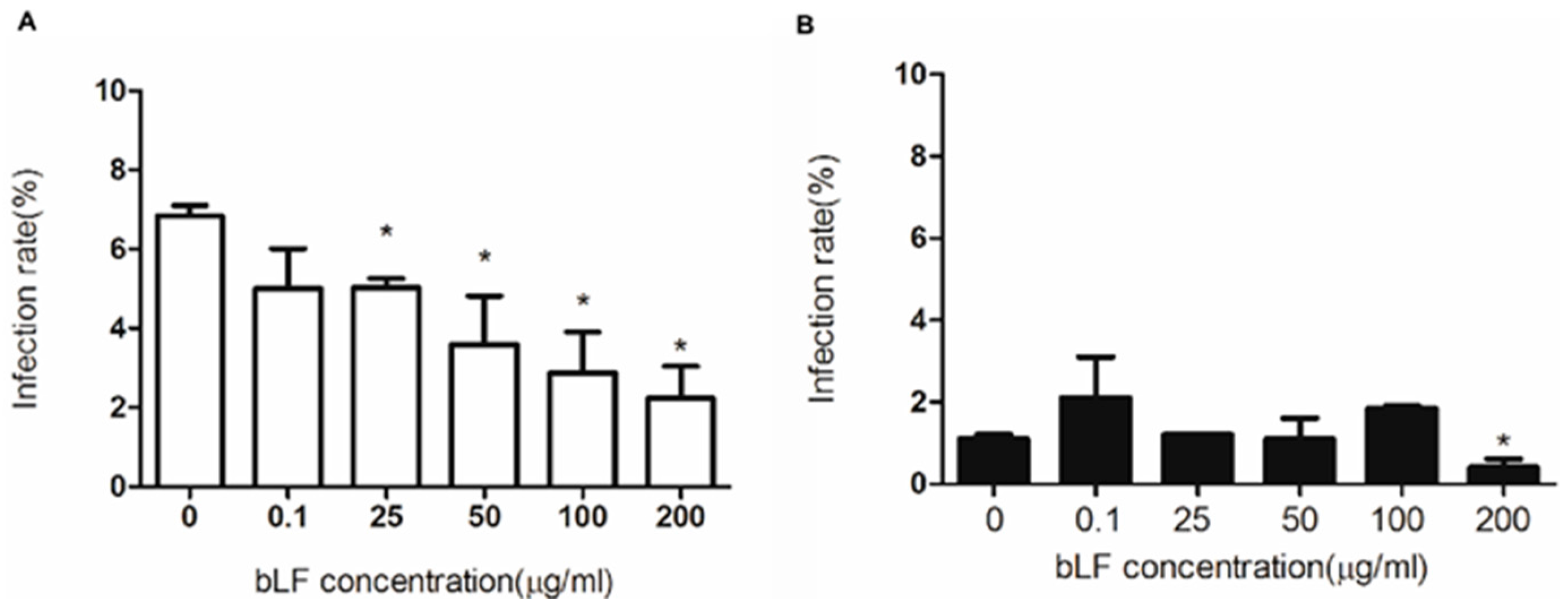
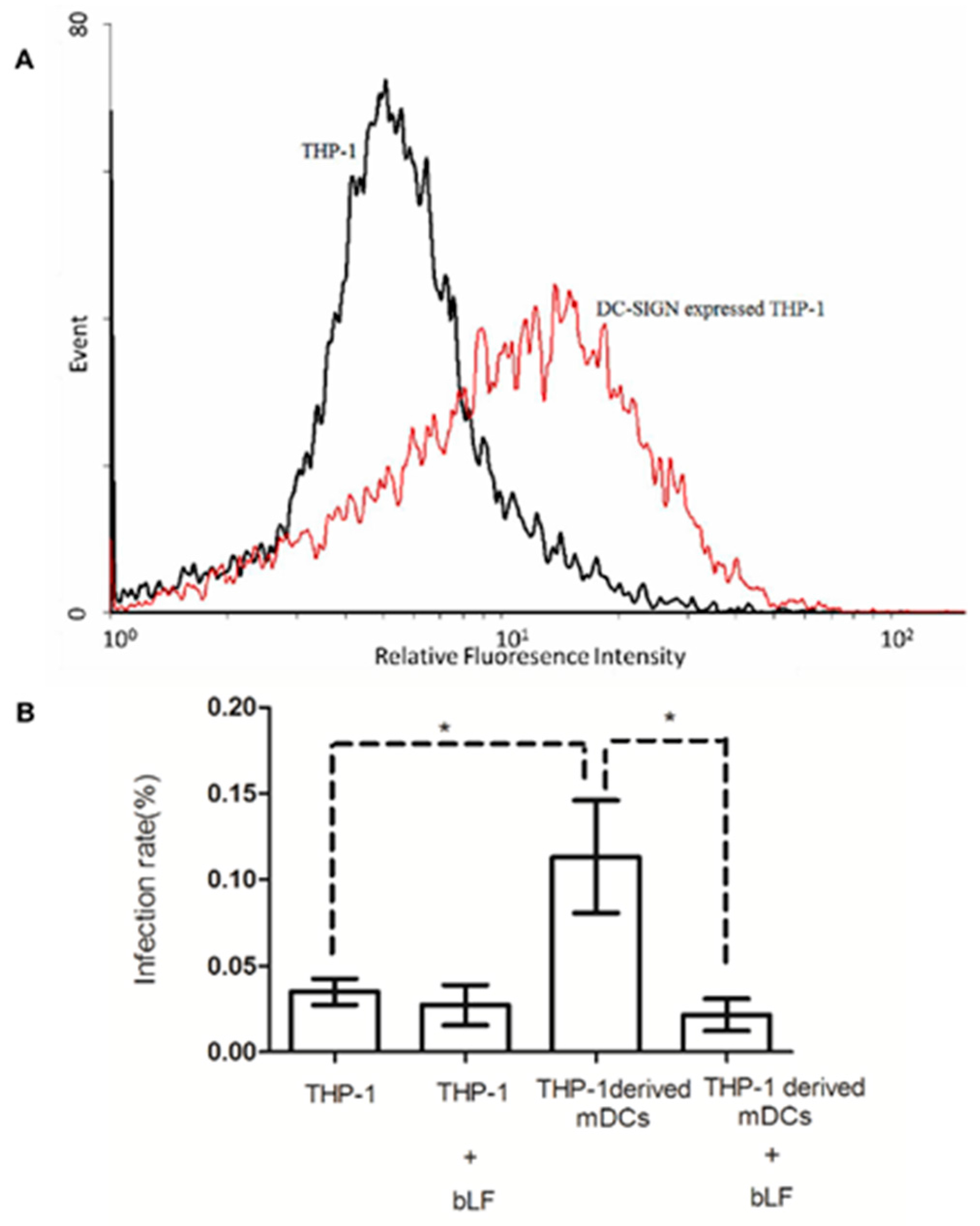
2.5. Role of DC-SIGN in Anti-DENV-2 Activity of bLF
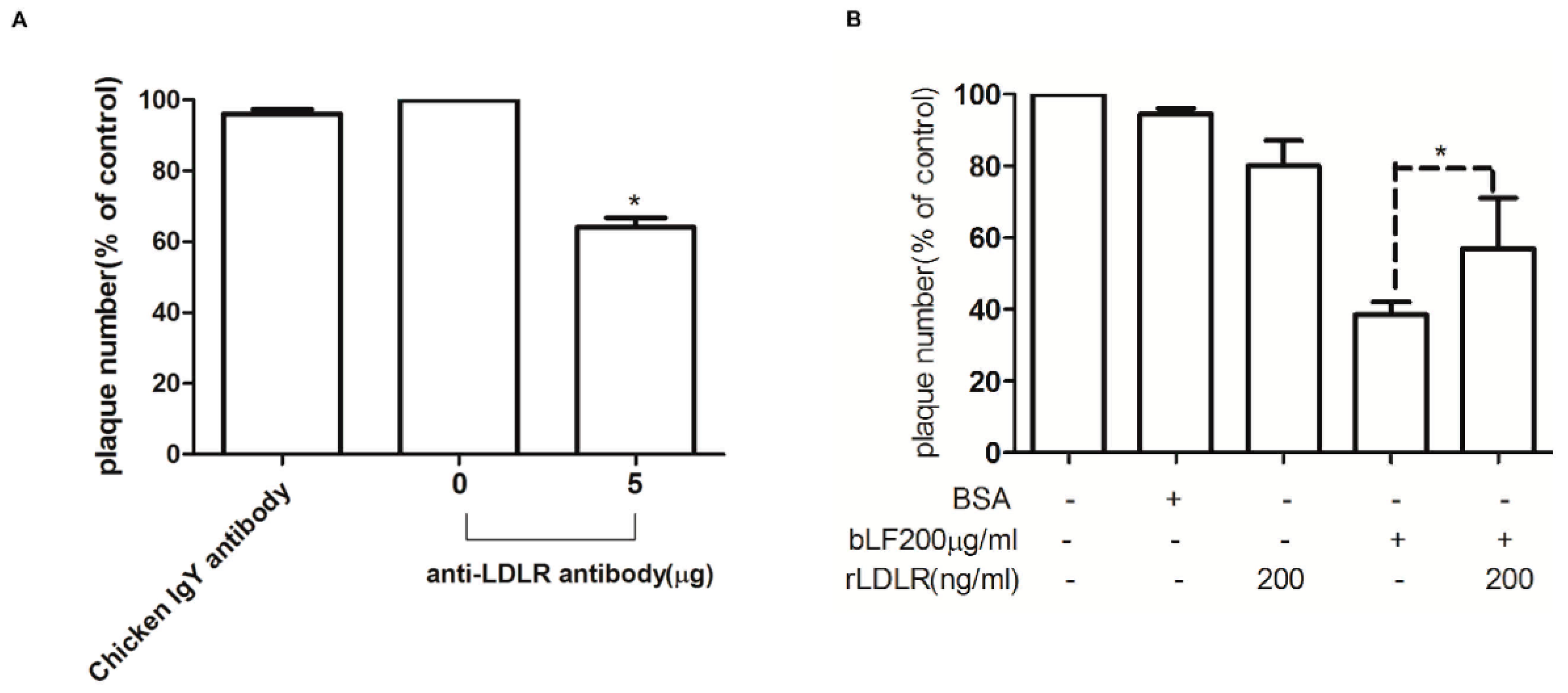
2.6. Role of LDLR in Anti-DENV-2 Activity of bLF
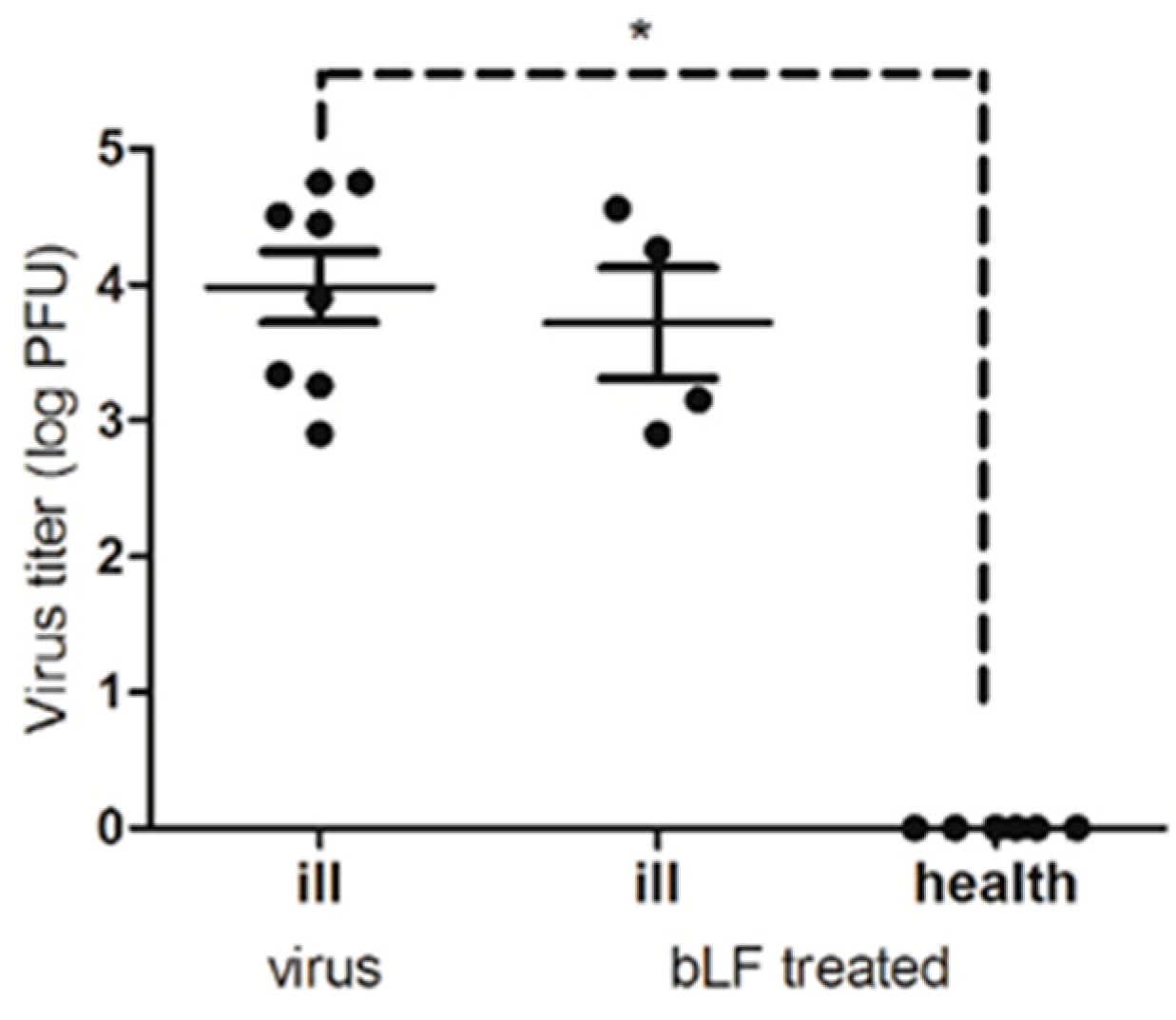
2.7. Mouse Challenge Experiments
3. Discussion
4. Materials and Methods
4.1. Viruses and Cell Lines
4.2. Plaque Assay and Plaque Reduction Assay
4.3. Viral binding Assay
4.4. Infectious Center Assay
4.5. Blocking Experiments to Investigate the Role of LDLR
4.6. Determing the Role of DC-SIGN
4.7. Indirect Immunofluorescence Assay
4.8. Mouse Challenge Experiments
4.9. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| bLF | Bovine lactoferrin |
| DENV | Dengue virus |
| HS | Heparan sulfate |
| DC-SIGN | Dendritic cell-specific intercellular adhesion molecule 3-grabbing non-integrin |
| LDLR | Low-density lipoprotein receptors |
References
- Van der Strate, B.W.; Beljaars, L.; Molema, G.; Harmsen, M.C.; Meijer, D.K. Antiviral activities of lactoferrin. Antivir. Res. 2001, 52, 225–239. [Google Scholar] [CrossRef]
- Lonnerdal, B.; Iyer, S. Lactoferrin: Molecular structure and biological function. Annu. Rev. Nutr. 1995, 15, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Weng, T.Y.; Chen, L.C.; Shyu, H.W.; Chen, S.H.; Wang, J.R.; Yu, C.K.; Lei, H.Y.; Yeh, T.M. Lactoferrin inhibits enterovirus 71 infection by binding to vp1 protein and host cells. Antivir. Res. 2005, 67, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Beleid, R.; Douglas, D.; Kneteman, N.; Kaur, K. Helical peptides derived from lactoferrin bind hepatitis c virus envelope protein e2. Chem. Biol. Drug Des. 2008, 72, 436–443. [Google Scholar] [CrossRef] [PubMed]
- El Yazidi-Belkoura, I.; Legrand, D.; Nuijens, J.; Slomianny, M.C.; van Berkel, P.; Spik, G. The binding of lactoferrin to glycosaminoglycans on enterocyte-like HT29–18-C1 cells is mediated through basic residues located in the N-terminus. Biochim. Biophys. Acta 2001, 1568, 197–204. [Google Scholar] [CrossRef]
- Chien, Y.J.; Chen, W.J.; Hsu, W.L.; Chiou, S.S. Bovine lactoferrin inhibits japanese encephalitis virus by binding to heparan sulfate and receptor for low density lipoprotein. Virology 2008, 379, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Groot, F.; Geijtenbeek, T.B.; Sanders, R.W.; Baldwin, C.E.; Sanchez-Hernandez, M.; Floris, R.; van Kooyk, Y.; de Jong, E.C.; Berkhout, B. Lactoferrin prevents dendritic cell-mediated human immunodeficiency virus type 1 transmission by blocking the Dc-SIGN—gp120 interaction. J. Virol. 2005, 79, 3009–3015. [Google Scholar] [CrossRef] [PubMed]
- Halstead, S.B. Antibody, macrophages, dengue virus infection, shock, and hemorrhage: A pathogenetic cascade. Rev. Infect. Dis. 1989, 11 (Suppl. S4), S830–S839. [Google Scholar] [CrossRef] [PubMed]
- Henchal, E.A.; Putnak, J.R. The dengue viruses. Clin. Microbiol. Rev. 1990, 3, 376–396. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.L.; Lee, P.L.; Chen, H.W.; Chen, L.K.; Kao, C.L.; King, C.C. Analysis of the steps involved in dengue virus entry into host cells. Virology 1999, 257, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Maguire, T.; Hileman, R.E.; Fromm, J.R.; Esko, J.D.; Linhardt, R.J.; Marks, R.M. Dengue virus infectivity depends on envelope protein binding to target cell heparan sulfate. Nat. Med. 1997, 3, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Tassaneetrithep, B.; Burgess, T.H.; Granelli-Piperno, A.; Trumpfheller, C.; Finke, J.; Sun, W.; Eller, M.A.; Pattanapanyasat, K.; Sarasombath, S.; Birx, D.L.; et al. DC-SIGN (CD209) mediates dengue virus infection of human dendritic cells. J. Exp. Med. 2003, 197, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Lozach, P.Y.; Burleigh, L.; Staropoli, I.; Navarro-Sanchez, E.; Harriague, J.; Virelizier, J.L.; Rey, F.A.; Despres, P.; Arenzana-Seisdedos, F.; Amara, A. Dendritic cell-specific intercellular adhesion molecule 3-grabbing non-integrin (DC-SIGN)-mediated enhancement of dengue virus infection is independent of DC-SIGN internalization signals. J. Biol. Chem. 2005, 280, 23698–23708. [Google Scholar] [CrossRef] [PubMed]
- Alen, M.M.; Kaptein, S.J.; De Burghgraeve, T.; Balzarini, J.; Neyts, J.; Schols, D. Antiviral activity of carbohydrate-binding agents and the role of DC-SIGN in dengue virus infection. Virology 2009, 387, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Alen, M.M.; De Burghgraeve, T.; Kaptein, S.J.; Balzarini, J.; Neyts, J.; Schols, D. Broad antiviral activity of carbohydrate-binding agents against the four serotypes of dengue virus in monocyte-derived dendritic cells. PLoS ONE 2011, 6, e21658. [Google Scholar] [CrossRef] [PubMed]
- Waarts, B.L.; Aneke, O.J.; Smit, J.M.; Kimata, K.; Bittman, R.; Meijer, D.K.; Wilschut, J. Antiviral activity of human lactoferrin: Inhibition of alphavirus interaction with heparan sulfate. Virology 2005, 333, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.A.M.; Casseb, S.M.M.; Goncalves, R.B.; Silva, E.V.P.; Gomes, A.M.O.; Vasconcelos, P.F.C. Bovine lactoferrin activity against chikungunya and zika viruses. J. Gen. Virol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Huettinger, M.; Meilinger, M.; Gschwentner, C.; Lassmann, H. The LDL-receptor family. Lactoferrin and lipid metabolism. Adv. Exp. Med. Biol. 1998, 443, 107–111. [Google Scholar] [PubMed]
- Berges, C.; Naujokat, C.; Tinapp, S.; Wieczorek, H.; Hoh, A.; Sadeghi, M.; Opelz, G.; Daniel, V. A cell line model for the differentiation of human dendritic cells. Biochemic. Biophys. Res. Commun. 2005, 333, 896–907. [Google Scholar] [CrossRef] [PubMed]
- Li, P.C.; Liao, M.Y.; Cheng, P.C.; Liang, J.J.; Liu, I.J.; Chiu, C.Y.; Lin, Y.L.; Chang, G.J.; Wu, H.C. Development of a humanized antibody with high therapeutic potential against dengue virus type 2. PLoS Negl. Trop. Dis. 2012, 6, e1636. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, M.; Superti, F.; Ammendolia, M.G.; Rossi, P.; Valenti, P.; Seganti, L. Inhibition of poliovirus type 1 infection by iron-, manganese- and zinc-saturated lactoferrin. Med. Microbiol. Immunol. 1999, 187, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Pietrantoni, A.; Ammendolia, M.G.; Tinari, A.; Siciliano, R.; Valenti, P.; Superti, F. Bovine lactoferrin peptidic fragments involved in inhibition of echovirus 6 in vitro infection. Antivir. Res. 2006, 69, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Hara, K.; Ikeda, M.; Saito, S.; Matsumoto, S.; Numata, K.; Kato, N.; Tanaka, K.; Sekihara, H. Lactoferrin inhibits hepatitis b virus infection in cultured human hepatocytes. Hepatol. Res. 2002, 24, 228. [Google Scholar] [CrossRef]
- Pietrantoni, A.; Fortuna, C.; Remoli, M.E.; Ciufolini, M.G.; Superti, F. Bovine lactoferrin inhibits toscana virus infection by binding to heparan sulphate. Viruses 2015, 7, 480–495. [Google Scholar] [CrossRef] [PubMed]
- Kanyshkova, T.G.; Babina, S.E.; Semenov, D.V.; Isaeva, N.; Vlassov, A.V.; Neustroev, K.N.; Kul’minskaya, A.A.; Buneva, V.N.; Nevinsky, G.A. Multiple enzymic activities of human milk lactoferrin. Eur. J. Biochem. 2003, 270, 3353–3361. [Google Scholar] [CrossRef] [PubMed]
- Zemankova, N.; Chlebova, K.; Matiasovic, J.; Prodelalova, J.; Gebauer, J.; Faldyna, M. Bovine lactoferrin free of lipopolysaccharide can induce a proinflammatory response of macrophages. BMC Vet. Res. 2016, 12, 251. [Google Scholar] [CrossRef] [PubMed]
- Hilgard, P.; Stockert, R. Heparan sulfate proteoglycans initiate dengue virus infection of hepatocytes. Hepatology 2000, 32, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.F.; Monroe, D.M.; Church, F.C. Characterization of the glycosaminoglycan-binding region of lactoferrin. Arch. Biochem. Biophys. 1995, 317, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Agnello, V.; Abel, G.; Elfahal, M.; Knight, G.B.; Zhang, Q.X. Hepatitis c virus and other flaviviridae viruses enter cells via low density lipoprotein receptor. Proc. Natl. Acad. Sci. USA 1999, 96, 12766–12771. [Google Scholar] [CrossRef] [PubMed]
- Soto-Acosta, R.; Mosso, C.; Cervantes-Salazar, M.; Puerta-Guardo, H.; Medina, F.; Favari, L.; Ludert, J.E.; del Angel, R.M. The increase in cholesterol levels at early stages after dengue virus infection correlates with an augment in ldl particle uptake and hmg-coa reductase activity. Virology 2013, 442, 132–147. [Google Scholar] [CrossRef] [PubMed]
- Wagar, Z.L.; Tree, M.O.; Mpoy, M.C.; Conway, M.J. Low density lipopolyprotein inhibits flavivirus acquisition in aedes aegypti. Insect. Mol. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kerr, A.G.; Tam, L.C.S.; Hale, A.B.; Cioroch, M.; Douglas, G.; Agkatsev, S.; Hibbitt, O.; Mason, J.; Holt-Martyn, J.; Bataille, C.J.R.; et al. A genomic DNA reporter screen identifies squalene synthase inhibitors that act cooperatively with statins to upregulate the low-density lipoprotein receptor. J. Pharmacol. Exp. Ther. 2017, 361, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Jindadamrongwech, S.; Smith, D.R. Virus overlay protein binding assay (vopba) reveals serotype specific heterogeneity of dengue virus binding proteins on hepg2 human liver cells. Intervirology 2004, 47, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Thepparit, C.; Smith, D.R. Serotype-specific entry of dengue virus into liver cells: Identification of the 37-kilodalton/67-kilodalton high-affinity laminin receptor as a dengue virus serotype 1 receptor. J. Virol. 2004, 78, 12647–12656. [Google Scholar] [CrossRef] [PubMed]
- Schuller, A.; Yin, Z.; Brian Chia, C.S.; Doan, D.N.; Kim, H.K.; Shang, L.; Loh, T.P.; Hill, J.; Vasudevan, S.G. Tripeptide inhibitors of dengue and west nile virus NS2B-NS3 protease. Antivir. Res. 2011, 92, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Harada, E.; Itoh, Y.; Sitizyo, K.; Takeuchi, T.; Araki, Y.; Kitagawa, H. Characteristic transport of lactoferrin from the intestinal lumen into the bile via the blood in piglets. Comp. Biochem. Physiol. Mol. Integr. Physiol. 1999, 124, 321–327. [Google Scholar] [CrossRef]
- Picard-Jean, F.; Bouchard, S.; Larivee, G.; Bisaillon, M. The intracellular inhibition of HCV replication represents a novel mechanism of action by the innate immune lactoferrin protein. Antivir. Res. 2014, 111, 13–22. [Google Scholar] [CrossRef] [PubMed]
| Virus | IC50 of bLF (μg/mL) | Reference |
|---|---|---|
| DENV-1 | 165.8 ± 35.1 | This study |
| DENV-2 | 40.7 ± 8.6 | This study |
| DENV-3 | 166.7 ± 30.6 | This study |
| DENV-4 | 164.5 ± 41.0 | This study |
| JEV | 518.3 ± 115.8 | [6] |
| ZIKV | ~200 ± 5.0 | [17] |
| CHIKV | ~400 ± 6.0 | [17] |
| Virus | IC50 of bLF Added at Different Steps of Virus Infection(μg/mL) | Reference | ||
|---|---|---|---|---|
| Before Virus Addition | During Virus Addition | After Virus Addition | ||
| DENV2 | 20.8 | 18.0 | 74.9 | This study |
| JEV | 385.6 | ND | 228.9 | [6] |
| ZIKV | >1000 | <1000 | <1000 | [17] |
| CHIKV | ~1000 | <1000 | >1000 | [17] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.-M.; Fan, Y.-C.; Lin, J.-W.; Chen, Y.-Y.; Hsu, W.-L.; Chiou, S.-S. Bovine Lactoferrin Inhibits Dengue Virus Infectivity by Interacting with Heparan Sulfate, Low-Density Lipoprotein Receptor, and DC-SIGN. Int. J. Mol. Sci. 2017, 18, 1957. https://doi.org/10.3390/ijms18091957
Chen J-M, Fan Y-C, Lin J-W, Chen Y-Y, Hsu W-L, Chiou S-S. Bovine Lactoferrin Inhibits Dengue Virus Infectivity by Interacting with Heparan Sulfate, Low-Density Lipoprotein Receptor, and DC-SIGN. International Journal of Molecular Sciences. 2017; 18(9):1957. https://doi.org/10.3390/ijms18091957
Chicago/Turabian StyleChen, Jo-Mei, Yi-Chin Fan, Jen-Wei Lin, Yi-Ying Chen, Wei-Li Hsu, and Shyan-Song Chiou. 2017. "Bovine Lactoferrin Inhibits Dengue Virus Infectivity by Interacting with Heparan Sulfate, Low-Density Lipoprotein Receptor, and DC-SIGN" International Journal of Molecular Sciences 18, no. 9: 1957. https://doi.org/10.3390/ijms18091957
APA StyleChen, J.-M., Fan, Y.-C., Lin, J.-W., Chen, Y.-Y., Hsu, W.-L., & Chiou, S.-S. (2017). Bovine Lactoferrin Inhibits Dengue Virus Infectivity by Interacting with Heparan Sulfate, Low-Density Lipoprotein Receptor, and DC-SIGN. International Journal of Molecular Sciences, 18(9), 1957. https://doi.org/10.3390/ijms18091957





