Mdivi-1 Protects Adult Rat Hippocampal Neural Stem Cells against Palmitate-Induced Oxidative Stress and Apoptosis
Abstract
:1. Introduction
2. Results
2.1. Mdivi-1 Protects Hippocampal NSCs against Palmitate Insult
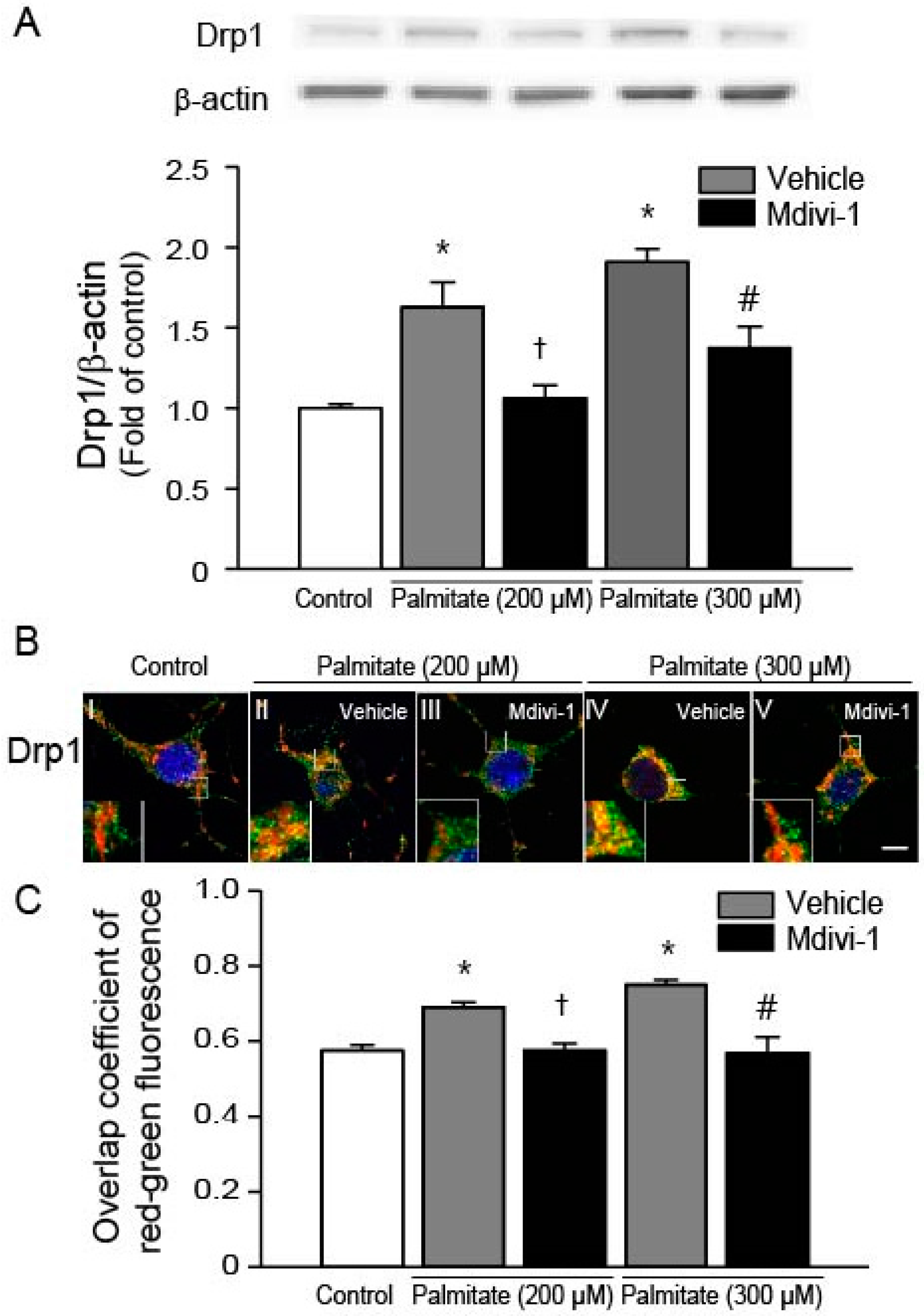
2.2. Mdivi-1 Inhibits Palmitate-Induced Drp1 Expression and Mitochondrial Translocation
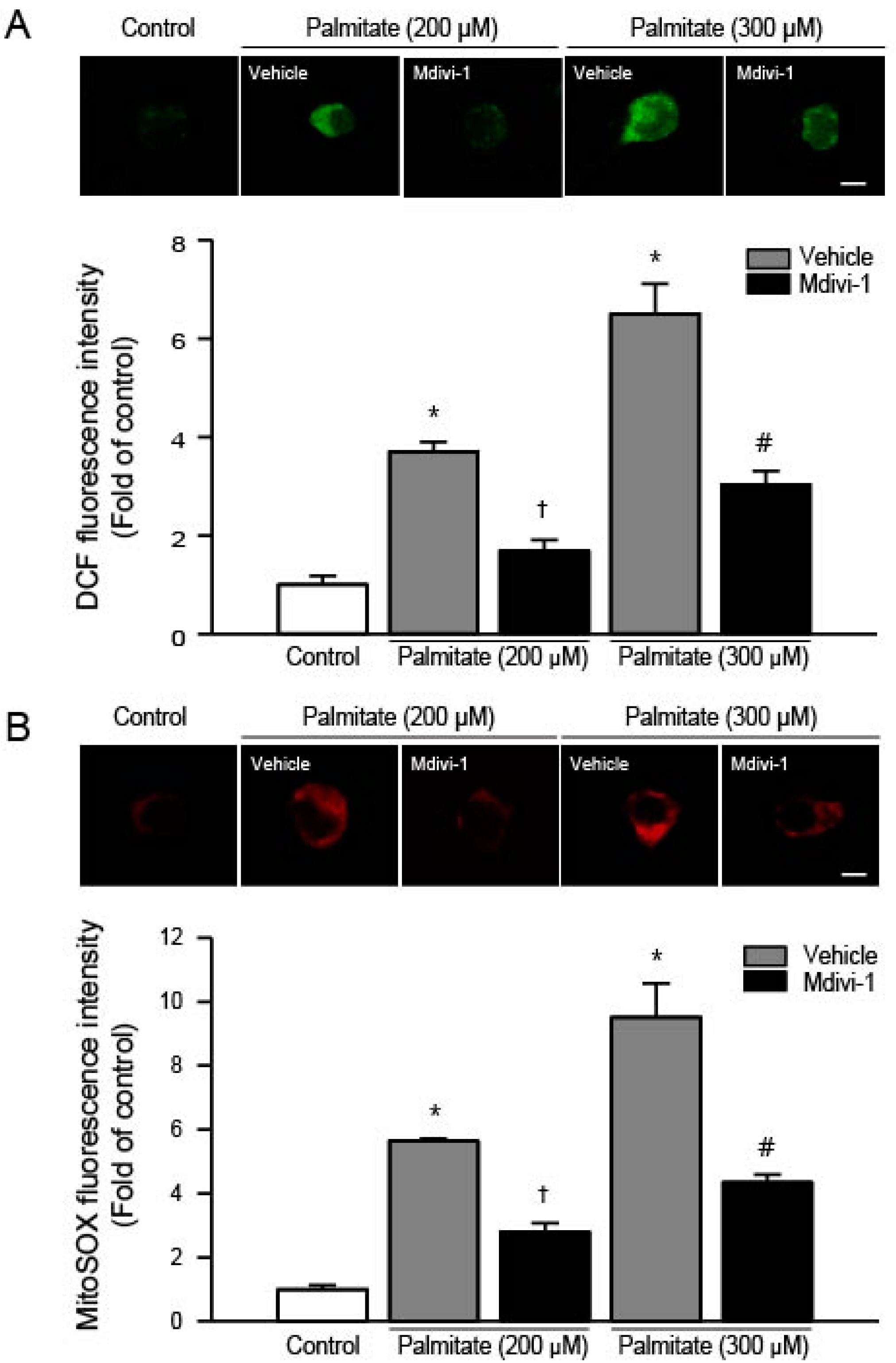
2.3. Mdivi-1 Inhibits Palmitate-Induced Increase in ROS Generation
2.4. Mdivi-1 Stabilizes Mitochondrial Transmembrane Potential (ΔψM)
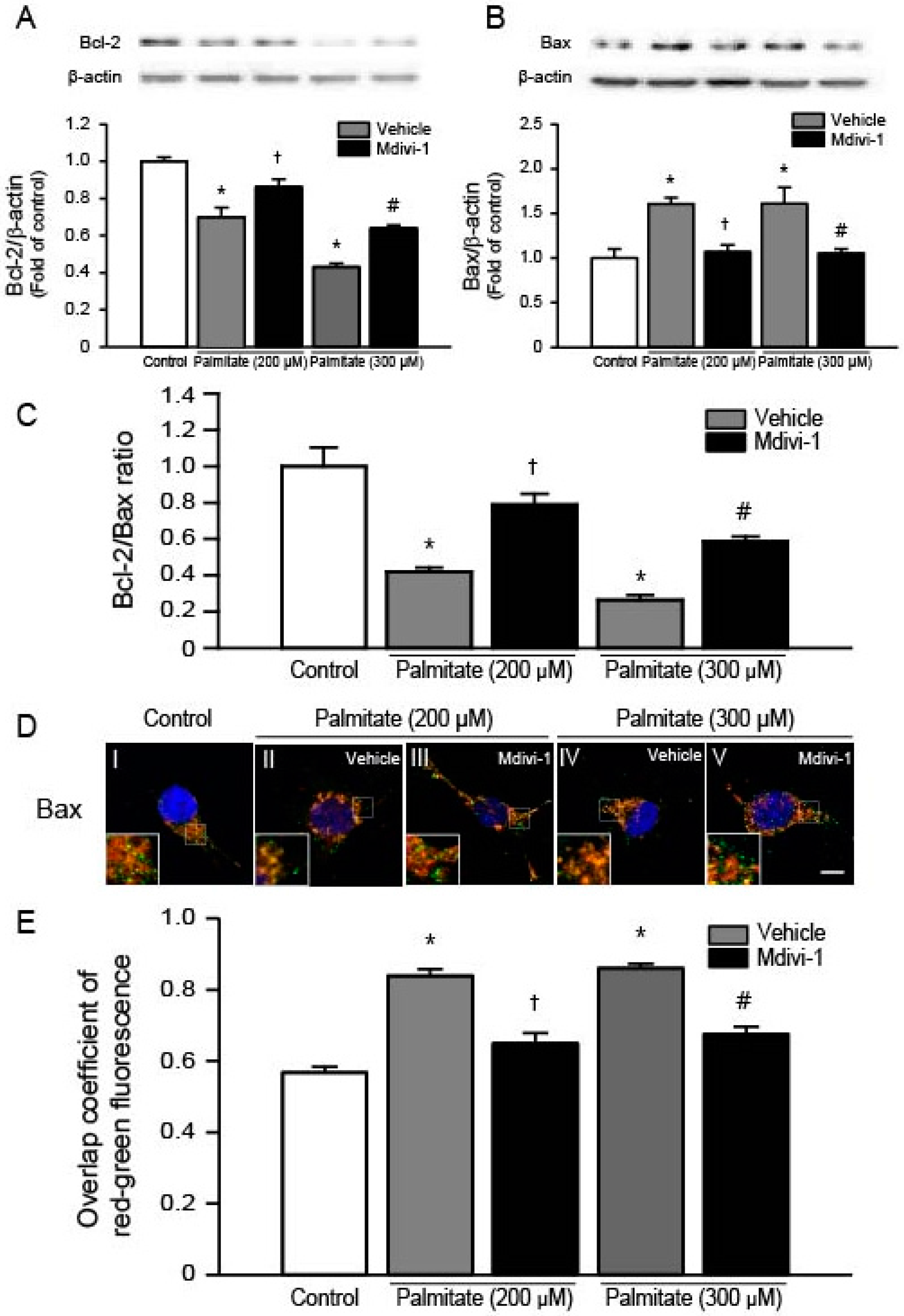
2.5. Mdivi-1 Increases Bcl-2 and Decreases Bax, Thereby Increasing the Bcl-2/Bax Ratio
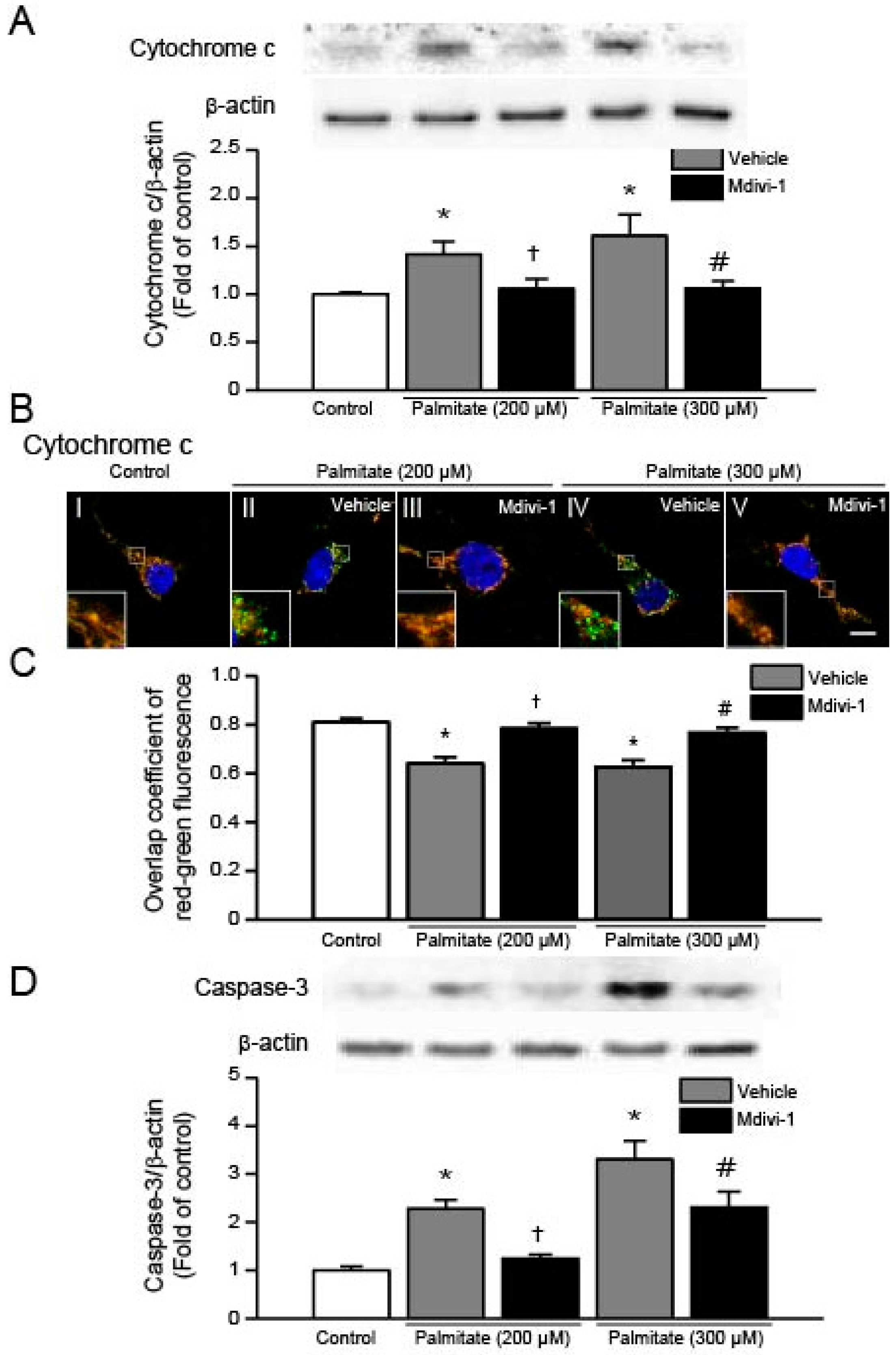
2.6. Mdivi-1 Inhibits Palmitate-Induced Increase in Cytochrome C Release and Caspase-3 Activation
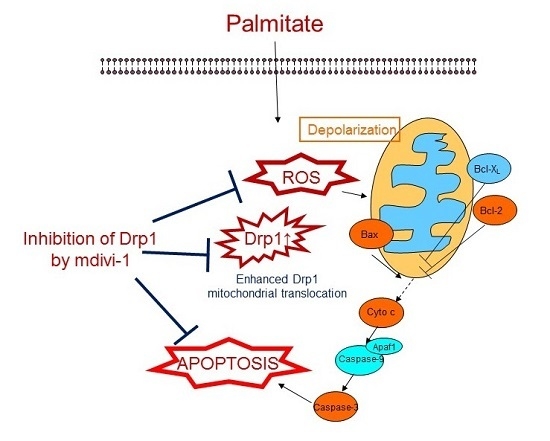
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Adult Rat Hippocampal NSCs Cultures and Treatment
4.3. Detection of Cell Death and Apoptosis
4.4. Detection of Intracellular and Mitochondrial ROS Production
4.5. Assessment of Mitochondrial Membrane Potential (ΔψM)
4.6. Western Blot Analysis
4.7. Immunocytochemical Detection of Drp1 or Bax Translocation and Cytochrome C Release
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| NSCs | Neural stem cells |
| ROS | Reactive oxygen species |
| FFA | Free fatty acid |
| Drp1 | Dynamin-regulated protein 1 |
| BSA | Bovine serum albumin |
| DCFDA | 2′,7′-Dichlorodihydrofluorescein diacetate |
| JC-1 | 5,5′,6,6′-Tetrachloro-1,1′,3′,3′-tetraethylbenzimidazolylcarbocyanine iodide |
References
- Ho, N.; Sommers, M.S.; Lucki, I. Effects of diabetes on hippocampal neurogenesis: Links to cognition and depression. Neurosci. Biobehav. Rev. 2013, 37, 1346–1362. [Google Scholar] [CrossRef] [PubMed]
- Stranahan, A.M.; Arumugam, T.V.; Cutler, R.G.; Lee, K.; Egan, J.M.; Mattson, M.P. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat. Neurosci. 2008, 11, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.J.; Tan, Y.F.; Yue, J.T.; Vranic, M.; Wojtowicz, J.M. Impairment of hippocampal neurogenesis in streptozotocin-treated diabetic rats. Acta Neurol. Scand. 2008, 117, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Lang, B.T.; Yan, Y.; Dempsey, R.J.; Vemuganti, R. Impaired neurogenesis in adult type-2 diabetic rats. Brain Res. 2009, 1258, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Rodriguez, J.J.; Molina-Gil, S.; Ortiz-Barajas, O.; Jimenez-Palomares, M.; Perdomo, G.; Cozar-Castellano, I.; Lechuga-Sancho, A.M.; Garcia-Alloza, M. Central proliferation and neurogenesis is impaired in type 2 diabetes and prediabetes animal models. PLoS ONE 2014, 9, e89229. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Deng, W.; Gage, F.H. Mechanisms and functional implications of adult neurogenesis. Cell 2008, 132, 645–660. [Google Scholar] [CrossRef] [PubMed]
- Voloboueva, L.A.; Giffard, R.G. Inflammation, mitochondria, and the inhibition of adult neurogenesis. J. Neurosci. Res. 2011, 89, 1989–1996. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G.; Kuhn, H.G.; Gage, F.H. More hippocampal neurons in adult mice living in an enriched environment. Nature 1997, 386, 493–495. [Google Scholar] [CrossRef] [PubMed]
- Fontan-Lozano, A.; Saez-Cassanelli, J.L.; Inda, M.C.; de los Santos-Arteaga, M.; Sierra-Dominguez, S.A.; Lopez-Lluch, G.; gado-Garcia, J.M.; Carrion, A.M. Caloric restriction increases learning consolidation and facilitates synaptic plasticity through mechanisms dependent on nr2b subunits of the nmda receptor. J. Neurosci. 2007, 27, 10185–10195. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Nakahara, K.; Goto, S.; Kaiya, H.; Miyazato, M.; Date, Y.; Nakazato, M.; Kangawa, K.; Murakami, N. Effects of ghrelin and des-acyl ghrelin on neurogenesis of the rat fetal spinal cord. Biochem. Biophys. Res. Commun. 2006, 350, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Moon, M.; Kim, S.; Hwang, L.; Park, S. Ghrelin regulates hippocampal neurogenesis in adult mice. Endocr. J. 2009, 56, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Li, E.; Kim, Y.; Kim, S.; Park, S. Multiple signaling pathways mediate ghrelin-induced proliferation of hippocampal neural stem cells. J. Endocrinol. 2013, 218, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Chung, H.; Kim, Y.; Kim, D.H.; Ryu, J.H.; Sato, T.; Kojima, M.; Park, S. Ghrelin directly stimulates adult hippocampal neurogenesis: Implications for learning and memory. Endocr. J. 2013, 60, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Gargantini, E.; Lazzari, L.; Settanni, F.; Taliano, M.; Trovato, L.; Gesmundo, I.; Ghigo, E.; Granata, R. Obestatin promotes proliferation and survival of adult hippocampal progenitors and reduces amyloid-beta-induced toxicity. Mol. Cell Endocrinol. 2016, 422, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, C.; Park, S. Ghrelin gene products rescue cultured adult rat hippocampal neural stem cells from high glucose insult. J. Mol. Endocrinol. 2016, 57, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Bergman, R.N.; Ader, M. Free fatty acids and pathogenesis of type 2 diabetes mellitus. Trends Endocrinol. Metab. 2000, 11, 351–356. [Google Scholar] [CrossRef]
- Cnop, M. Fatty acids and glucolipotoxicity in the pathogenesis of type 2 diabetes. Biochem. Soc. Trans. 2008, 36, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Van Herpen, N.A.; Schrauwen-Hinderling, V.B. Lipid accumulation in non-adipose tissue and lipotoxicity. Physiol. Behav. 2008, 94, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kimes, A.S.; Sweeney, D.; Rapoport, S.I. Brain palmitate incorporation in awake and anesthetized rats. Brain Res. 1985, 341, 164–170. [Google Scholar] [CrossRef]
- Almaguel, F.G.; Liu, J.W.; Pacheco, F.J.; Casiano, C.A.; De Leon, M. Activation and reversal of lipotoxicity in pc12 and rat cortical cells following exposure to palmitic acid. J. Neurosci. Res. 2009, 87, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Park, H.R.; Kim, J.Y.; Park, K.Y.; Lee, J. Lipotoxicity of palmitic acid on neural progenitor cells and hippocampal neurogenesis. Toxicol. Res. 2011, 27, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Padilla, A.; Descorbeth, M.; Almeyda, A.L.; Payne, K.; De Leon, M. Hyperglycemia magnifies schwann cell dysfunction and cell death triggered by pa-induced lipotoxicity. Brain Res. 2011, 1370, 64–79. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.M.; Belsham, D.D. Palmitate attenuates insulin signaling and induces endoplasmic reticulum stress and apoptosis in hypothalamic neurons: Rescue of resistance and apoptosis through adenosine 5′ monophosphate-activated protein kinase activation. Endocrinology 2010, 151, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Jun, H.S.; Oh, Y.S. Protective effect of psoralea corylifolia L. seed extract against palmitate-induced neuronal apoptosis in pc12 cells. Evid. Based Complement. Altern. Med. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Miles, J.M.; Wooldridge, D.; Grellner, W.J.; Windsor, S.; Isley, W.L.; Klein, S.; Harris, W.S. Nocturnal and postprandial free fatty acid kinetics in normal and type 2 diabetic subjects: Effects of insulin sensitization therapy. Diabetes 2003, 52, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.T.; Oh, H.H.; Nguyen, A.K.; Choi, C.S.; Youn, J.H. Circulating free fatty acids inhibit food intake in an oleate-specific manner in rats. Physiol. Behav. 2016, 167, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Rovira-Llopis, S.; Bañuls, C.; Diaz-Morales, N.; Hernandez-Mijares, A.; Rocha, M.; Victor, V.M. Mitochondrial dynamics in type 2 diabetes: Pathophysiological implications. Redox. Biol. 2017, 11, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Frank, S.; Gaume, B.; Bergmann-Leitner, E.S.; Leitner, W.W.; Robert, E.G.; Catez, F.; Smith, C.L.; Youle, R.J. The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Dev. Cell 2001, 1, 515–525. [Google Scholar] [CrossRef]
- Watanabe, T.; Saotome, M.; Nobuhara, M.; Sakamoto, A.; Urushida, T.; Katoh, H.; Satoh, H.; Funaki, M.; Hayashi, H. Roles of mitochondrial fragmentation and reactive oxygen species in mitochondrial dysfunction and myocardial insulin resistance. Exp. Cell Res. 2014, 323, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Jheng, H.F.; Tsai, P.J.; Guo, S.M.; Kuo, L.H.; Chang, C.S.; Su, I.J.; Chang, C.R.; Tsai, Y.S. Mitochondrial fission contributes to mitochondrial dysfunction and insulin resistance in skeletal muscle. Mol. Cell Biol. 2012, 32, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Kuzmicic, J.; Parra, V.; Verdejo, H.E.; López-Crisosto, C.; Chiong, M.; García, L.; Jensen, M.D.; Bernlohr, D.A.; Castro, P.F.; Lavandero, S. Trimetazidine prevents palmitate-induced mitochondrial fission and dysfunction in cultured cardiomyocytes. Biochem. Pharmacol. 2014, 91, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Cassidy-Stone, A.; Chipuk, J.E.; Ingerman, E.; Song, C.; Yoo, C.; Kuwana, T.; Kurth, M.J.; Shaw, J.T.; Hinshaw, J.E.; Green, D.R.; et al. Chemical inhibition of the mitochondrial division dynamin reveals its role in bax/bak-dependent mitochondrial outer membrane permeabilization. Dev. Cell 2008, 14, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, M.; Wang, S. Effect of inhibiting mitochondrial fission on energy metabolism in rat hippocampal neurons during ischemia/reperfusion injury. Neurol. Res. 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Wang, C.; Wu, C.; Cheng, X.; Gao, Y.; Zhang, H.; Zhang, Y.; Lian, Y. Mdivi-1 protects epileptic hippocampal neurons from apoptosis via inhibiting oxidative stress and endoplasmic reticulum stress in vitro. Neurochem. Res. 2016, 41, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, P.; Li, S.; Wang, S.; Li, Y.; Liang, N.; Wang, M. Mdivi-1 prevents apoptosis induced by ischemia-reperfusion injury in primary hippocampal cells via inhibition of reactive oxygen species-activated mitochondrial pathway. J. Stroke Cerebrovasc. Dis. 2014, 23, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, J.Y.; Park, K.J.; Kim, W.H.; Roh, G.S. A mitochondrial division inhibitor, mdivi-1, inhibits mitochondrial fragmentation and attenuates kainic acid-induced hippocampal cell death. BMC Neurosci. 2016, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Bordt, E.A.; Clerc, P.; Roelofs, B.A.; Saladino, A.J.; Tretter, L.; Adam-Vizi, V.; Cherok, E.; Khalil, A.; Yadava, N.; Ge, S.X.; et al. The putative drp1 inhibitor mdivi-1 is a reversible mitochondrial complex i inhibitor that modulates reactive oxygen species. Dev. Cell 2017, 40, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Benard, G.; Karbowski, M. Mitochondrial fusion and division: Regulation and role in cell viability. Semin. Cell Dev. Biol. 2009, 20, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, A.; Saotome, M.; Hasan, P.; Satoh, T.; Ohtani, H.; Urushida, T.; Katoh, H.; Satoh, H.; Hayashi, H. Eicosapentaenoic acid ameliorates palmitate-induced lipotoxicity via the amp kinase/dynamin-related protein-1 signaling pathway in differentiated h9c2 myocytes. Exp. Cell Res. 2017, 351, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Manders, E.M.M.; Verbeek, F.J.; Aten, J.A. Measurement of co-localization of objects in dual-colour confocal images. J. Microsc. 1993, 169, 375–382. [Google Scholar] [CrossRef]
- Indo, H.P.; Davidson, M.; Yen, H.C.; Suenaga, S.; Tomita, K.; Nishii, T.; Higuchi, M.; Koga, Y.; Ozawa, T.; Majima, H.J. Evidence of ros generation by mitochondria in cells with impaired electron transport chain and mitochondrial dna damage. Mitochondrion 2007, 7, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Taddeo, E.P.; Laker, R.C.; Breen, D.S.; Akhtar, Y.N.; Kenwood, B.M.; Liao, J.A.; Zhang, M.; Fazakerley, D.J.; Tomsig, J.L.; Harris, T.E.; et al. Opening of the mitochondrial permeability transition pore links mitochondrial dysfunction to insulin resistance in skeletal muscle. Mol. Metab. 2014, 3, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Jia, Z.; Cao, Y.; Wang, Y.; Li, H.; Zhang, Z.; Bi, J.; Lv, G.; Fan, Z. Mitochondrial division inhibitor 1 ameliorates mitochondrial injury, apoptosis, and motor dysfunction after acute spinal cord injury in rats. Neurochem. Res. 2015, 40, 1379–1392. [Google Scholar] [CrossRef] [PubMed]
- Legrand, O.; Perrot, J.Y.; Simonin, G.; Baudard, M.; Marie, J.P. Jc-1: A very sensitive fluorescent probe to test pgp activity in adult acute myeloid leukemia. Blood 2001, 97, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.D.; Zhen, Y.Y.; Lin, J.W.; Lin, T.K.; Huang, C.W.; Liou, C.W.; Chan, S.H.; Chuang, Y.C. Dynamin-related protein 1 promotes mitochondrial fission and contributes to the hippocampal neuronal cell death following experimental status epilepticus. CNS Neurosci. Ther. 2016, 22, 988–999. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Wang, C.; Lian, Y.; Zhang, H.; Wu, C.; Zhang, Q. A selective inhibitor of drp1, mdivi-1, protects against cell death of hippocampal neurons in pilocarpine-induced seizures in rats. Neurosci. Lett. 2013, 545, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.R.; Blackstone, C. Dynamic regulation of mitochondrial fission through modification of the dynamin-related protein drp1. Ann. N. Y. Acad. Sci. 2010, 1201, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Ly, L.D.; Xu, S.; Choi, S.K.; Ha, C.M.; Thoudam, T.; Cha, S.K.; Wiederkehr, A.; Wollheim, C.B.; Lee, I.K.; Park, K.S. Oxidative stress and calcium dysregulation by palmitate in type 2 diabetes. Exp. Mol. Med. 2017, 49, e291. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ros and rns sources in physiological and pathological conditions. Oxid. Med. Cell Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef] [PubMed]
- Tangvarasittichai, S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J. Diabetes 2015, 6, 456–480. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.J.; Low, I.C.; Pervaiz, S. Mitochondrial ros and involvement of bcl-2 as a mitochondrial ros regulator. Mitochondrion 2014, 19. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Yankner, B.A. Apoptosis in the nervous system. Nature 2000, 407, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.; Esposti, M.D.; Gilmore, A.P. Bcl-2 proteins and mitochondria--specificity in membrane targeting for death. Biochim. Biophys. Acta 2011, 1813, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Korsmeyer, S.J. Regulators of cell death. Trends Genet. 1995, 11, 101–105. [Google Scholar] [CrossRef]
- Leroy, C.; Tricot, S.; Lacour, B.; Grynberg, A. Protective effect of eicosapentaenoic acid on palmitate-induced apoptosis in neonatal cardiomyocytes. Biochim. Biophys. Acta 2008, 1781, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y. A structural view of mitochondria-mediated apoptosis. Nat. Struct. Biol. 2001, 8, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Rabkin, S.W.; Klassen, S.S. Palmitate-induced no production has a dual action to reduce cell death through no and accentuate cell death through peroxynitrite formation. Prostaglandins Leukot. Essent. Fat. Acids 2008, 78, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.C.; Lin, T.K.; Yang, D.I.; Yang, J.L.; Liou, C.W.; Chen, S.D. Peroxisome proliferator-activated receptor-gamma dependent pathway reduces the phosphorylation of dynamin-related protein 1 and ameliorates hippocampal injury induced by global ischemia in rats. J. Biomed. Sci. 2016, 23, 44. [Google Scholar] [CrossRef] [PubMed]
- Sharp, W.W.; Fang, Y.H.; Han, M.; Zhang, H.J.; Hong, Z.; Banathy, A.; Morrow, E.; Ryan, J.J.; Archer, S.L. Dynamin-related protein 1 (drp1)-mediated diastolic dysfunction in myocardial ischemia-reperfusion injury: Therapeutic benefits of drp1 inhibition to reduce mitochondrial fission. FASEB J. 2014, 28, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Cousin, S.P.; Hügl, S.R.; Wrede, C.E.; Kajio, H.; Myers, M.G.; Rhodes, C.J. Free fatty acid-induced inhibition of glucose and insulin-like growth factor i-induced deoxyribonucleic acid synthesis in the pancreatic beta-cell line ins-1. Endocrinology 2001, 142, 229–240. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Kim, C.; Park, S. Mdivi-1 Protects Adult Rat Hippocampal Neural Stem Cells against Palmitate-Induced Oxidative Stress and Apoptosis. Int. J. Mol. Sci. 2017, 18, 1947. https://doi.org/10.3390/ijms18091947
Kim S, Kim C, Park S. Mdivi-1 Protects Adult Rat Hippocampal Neural Stem Cells against Palmitate-Induced Oxidative Stress and Apoptosis. International Journal of Molecular Sciences. 2017; 18(9):1947. https://doi.org/10.3390/ijms18091947
Chicago/Turabian StyleKim, Sehee, Chanyang Kim, and Seungjoon Park. 2017. "Mdivi-1 Protects Adult Rat Hippocampal Neural Stem Cells against Palmitate-Induced Oxidative Stress and Apoptosis" International Journal of Molecular Sciences 18, no. 9: 1947. https://doi.org/10.3390/ijms18091947
APA StyleKim, S., Kim, C., & Park, S. (2017). Mdivi-1 Protects Adult Rat Hippocampal Neural Stem Cells against Palmitate-Induced Oxidative Stress and Apoptosis. International Journal of Molecular Sciences, 18(9), 1947. https://doi.org/10.3390/ijms18091947