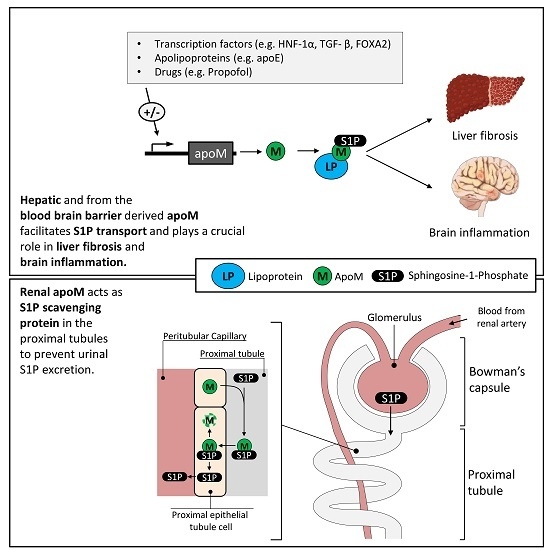
A Novel Perspective on the ApoM-S1P Axis, Highlighting the Metabolism of ApoM and Its Role in Liver Fibrosis and Neuroinflammation
Abstract
1. Introduction
2. The Apolipoprotein M/Sphingoshine-1-Phosphate (ApoM/S1P) Axis—Introduction to the Concept
3. ApoM—Regulation, Modification and Release
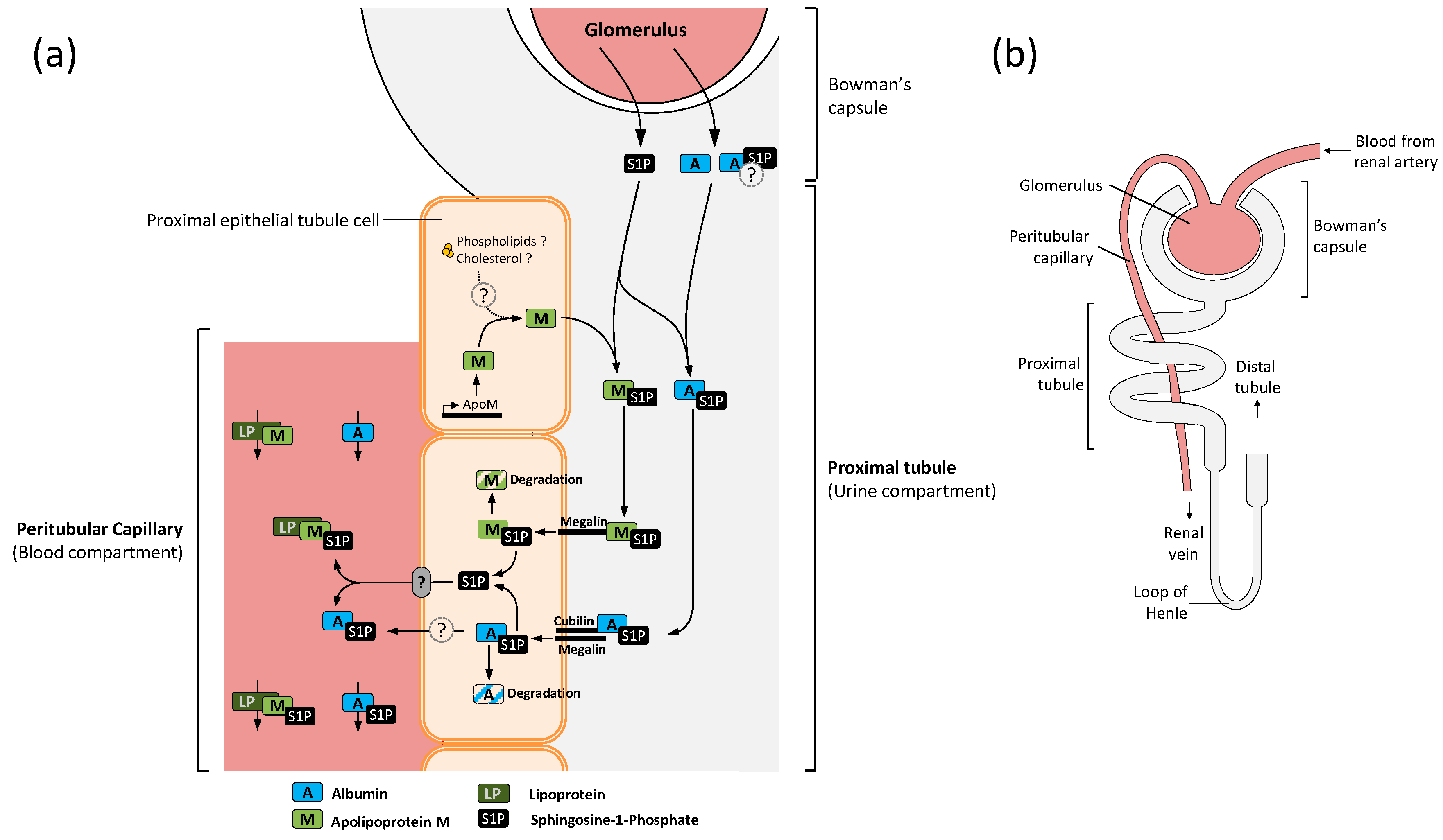
4. ApoM Acts as S1P Scavenger in the Proximal Convoluted Tubule
5. S1P Release from Primary Synthesizing Cells
6. S1P Release Mechanism from ApoM
7. The Role of ApoM in Liver Fibrosis
8. ApoM May Exert an Essential Role in Cerebral S1P Transport
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| apoD | Apolipoprotein D |
| apoE | Apolipoprotein E |
| apoM | Apolipoprotein M |
| BBB | Blood-Brain-Barrier |
| BDL | Bile duct ligation |
| CCl4 | Carbon tetrachloride |
| CNS | Central Nervous System |
| EAE | Autoimmune Encephalomyelitis |
| EC | Endothelial cell |
| ECM | Extracellular matrix |
| FOXA2 | Hepatocyte nuclear factor 3-β |
| HDL | High Density Lipoproteins |
| HNF-1α | Hepatocyte Nuclear Factor-1α |
| HSC | Hepatic stellate cell |
| LDL | Low Density Lipoprotein |
| LRP2 | LDL receptor related protein 2 (Megalin) |
| pBCEC | porcine Brain Capillary Endothelial Cell |
| PKC | Protein kinase C |
| PLTP | Phospholipid Transfer Protein |
| RBC | Red blood cell |
| S1P | Sphingosine-1-Phosphate |
| S1P1 | S1P receptor 1 |
| SA | Serum albumin |
| SMA | Smooth muscle actin |
| SPHK | Sphingosine Kinase |
| SPNS2 | Sphingolipid Transporter 2 |
| SR-BI | Scavenger receptor class B member 1 |
| TGF-β | Growth Factor β |
| VLDL | Very Low Density Lipoprotein |
References
- Xu, N.; Dahlbäck, B. A novel human apolipoprotein (apoM). J. Biol. Chem. 1999, 274, 31286–31290. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Dong, X.; Zheng, L.; Luo, G.H.; Liu, Y.H.; Ekström, U.; Nilsson-Ehle, P.; Ye, Q.; Xu, N. Specific tissue expression and cellular localization of human apolipoprotein M as determined by in situ hybridization. Acta Histochem. 2003, 105, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Kober, A.C.; Manavalan, A.P.C.; Tam-Amersdorfer, C.; Holmér, A.; Saeed, A.; Danesh, E.F.; Zandl, M.; Albrecher, N.M.; Björkhem, I.; Kostner, G.M.; et al. Implications of cerebrovascular ATP-binding cassette transporter G1 (ABCG1) and apolipoprotein M in cholesterol transport at the blood-brain barrier. Biochim. Biophys. Acta 2017, 1862, 573–588. [Google Scholar] [CrossRef] [PubMed]
- Borup, A.; Christensen, P.M.; Nielsen, L.B.; Christoffersen, C. Apolipoprotein M in lipid metabolism and cardiometabolic diseases. Curr. Opin. Lipidol. 2015, 26, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhao, X.X.; Zhang, X.; Chen, W.; Zhang, J. Functional study of -724I/D polymorphism in apolipoprotein M (apoM) gene promoter region and its association with myocardial infarction. Med. Sci. Monit. 2015, 21, 371–375. [Google Scholar] [PubMed]
- Zhang, P.H.; Gao, J.L.; Pu, C.; Feng, G.; Wang, L.Z.; Huang, L.Z.; Zhang, Y. A single-nucleotide polymorphism C-724 /del in the proter region of the apolipoprotein M gene is associated with type 2 diabetes mellitus. Lipids Health Dis. 2016, 15, 142. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kim, J.I.; Choi, S.; Jang, Y.; Sorn, S.R. The Effect of apoM Polymorphism Associated with HDL Metabolism on Obese Korean Adults. J. Nutrigenet. Nutr. 2017, 9, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Sevvana, M.; Ahnström, J.; Egerer-Sieber, C.; Lange, H.A.; Dahlbäck, B.; Muller, Y.A. Serendipitous fatty acid binding reveals the structural determinants for ligand recognition in apolipoprotein M. J. Mol. Biol. 2009, 393, 920–936. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.; Lindqvist, H.; Tagesson, C.; Lindahl, M. Characterization of apolipoprotein M isoforms in low-density lipoprotein. J. Proteome Res. 2006, 5, 2685–2690. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Dahlbäck, B.; Villoutreix, B.O. Proposed lipocalin fold for apolipoprotein M based on bioinformatics and site-directed mutagenesis. FEBS Lett. 2001, 499, 127–132. [Google Scholar] [CrossRef]
- Sevvana, M.; Kassler, K.; Ahnström, J.; Weiler, S.; Dahlbäck, B.; Sticht, H.; Muller, Y.A. Mouse ApoM displays an unprecedented seven-stranded lipocalin fold: Folding decoy or alternative native fold? J. Mol. Biol. 2010, 404, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Obinata, H.; Kumaraswamy, S.B.; Galvani, S.; Ahnström, J.; Sevvana, M.; Egerer-Sieber, C.; Muller, Y.A.; Hla, T.; Nielsen, L.B.; et al. Endothelium-protective sphingosine-1-phosphate provided by HDL-associated apolipoprotein M. Proc. Natl. Acad. Sci. USA 2011, 108, 9613–9618. [Google Scholar] [CrossRef] [PubMed]
- Dahlbäck, B.; Nielsen, L.B. Apolipoprotein M affecting lipid metabolism or just catching a ride with lipoproteins in the circulation? Cell. Mol. Life Sci. 2009, 66, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Axler, O.; Ahnström, J.; Dahlbäck, B. An ELISA for apolipoprotein M reveals a strong correlation to total cholesterol in human plasma. J. Lipid Res. 2007, 48, 1772–1780. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Pedersen, T.X.; Gordts, P.L.S.M.; Roebroek, A.J.M.; Dahlbäck, B.; Nielsen, L.B. Opposing effects of apolipoprotein m on catabolism of apolipoprotein B-containing lipoproteins and atherosclerosis. Circ. Res. 2010, 106, 1624–1634. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Benn, M.; Christensen, P.M.; Gordts, P.L.S.M.; Roebroek, A.J.M.; Frikke-Schmidt, R.; Tybjaerg-Hansen, A.; Dahlbäck, B.; Nielsen, L.B. The plasma concentration of HDL-associated apoM is influenced by LDL receptor-mediated clearance of apoB-containing particles. J. Lipid Res. 2012, 53, 2198–2204. [Google Scholar] [CrossRef] [PubMed]
- Noy, N.; Xu, Z.J. Interactions of retinol with binding proteins: Implications for the mechanism of uptake by cells. Biochemistry 1990, 29, 3878–3883. [Google Scholar] [CrossRef] [PubMed]
- Ahnström, J.; Faber, K.; Axler, O.; Dahlbäck, B. Hydrophobic ligand binding properties of the human lipocalin apolipoprotein M. J. Lipid Res. 2007, 48, 1754–1762. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Sanchez, D.; Correnti, C.; Strong, R.K.; Ganfornina, M.D. Lipid-binding properties of human ApoD and Lazarillo-related lipocalins: Functional implications for cell differentiation. FEBS J. 2013, 280, 3928–3943. [Google Scholar] [CrossRef] [PubMed]
- Cogan, U.; Kopelman, M.; Mokady, S.; Shinitzky, M. Binding affinities of retinol and related compounds to retinol binding proteins. Eur. J. Biochem. 1976, 65, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Seo, J.; Allegood, J.; Bi, X.; Zhu, X.; Boudyguina, E.; Gebre, A.K.; Avni, D.; Shah, D.; Sorci-Thomas, M.G.; et al. Hepatic apolipoprotein M (apoM) overexpression stimulates formation of larger apoM/sphingosine 1-phosphate-enriched plasma high density lipoprotein. J. Biol. Chem. 2014, 289, 2801–2814. [Google Scholar] [CrossRef] [PubMed]
- Murata, N.; Sato, K.; Kon, J.; Tomura, H.; Yanagita, M.; Kuwabara, A.; Ui, M.; Okajima, F. Interaction of sphingosine 1-phosphate with plasma components, including lipoproteins, regulates the lipid receptor-mediated actions. Biochem. J. 2000, 352, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Simard, J.R.; Zunszain, P.A.; Ha, C.E.; Yang, J.S.; Bhagavan, N.V.; Petitpas, I.; Curry, S.; Hamilton, J.A. Locating high-affinity fatty acid-binding sites on albumin by X-ray crystallography and NMR spectroscopy. Proc. Natl. Acad. Sci. USA 2005, 102, 17958–17963. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.K.; Glass, T.R.; Lackie, S.J.; Wojciak, J.M. A novel approach for measuring sphingosine-1-phosphate and lysophosphatidic acid binding to carrier proteins using monoclonal antibodies and the Kinetic Exclusion Assay. J. Lipid Res. 2016, 57, 1737–1747. [Google Scholar] [CrossRef] [PubMed]
- Badimon, J.J.; Santos-Gallego, C.G. HDL Dysfunction: Is the answer in the sphinx’s riddle? J. Am. Coll. Cardiol. 2015, 66, 1486–1488. [Google Scholar] [CrossRef] [PubMed]
- Wadhera, R.K.; Steen, D.L.; Khan, I.; Giugliano, R.P.; Foody, J.M. A review of low-density lipoprotein cholesterol, treatment strategies, and its impact on cardiovascular disease morbidity and mortality. J. Clin. Lipidol. 2016, 10, 472–489. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R.S.; Brewer, H.B.; Ansell, B.J.; Barter, P.; Chapman, M.J.; Heinecke, J.W.; Kontush, A.; Tall, A.R.; Webb, N.R. Dysfunctional HDL and atherosclerotic cardiovascular disease. Nat. Rev. Cardiol. 2015, 13, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Breustedt, D.A.; Schönfeld, D.L.; Skerra, A. Comparative ligand-binding analysis of ten human lipocalins. Biochim. Biophys. Acta 2006, 1764, 161–173. [Google Scholar] [CrossRef] [PubMed]
- N’soukpoé-Kossi, C.N.; Sedaghat-Herati, R.; Ragi, C.; Hotchandani, S.; Tajmir-Riahi, H.A. Retinol and retinoic acid bind human serum albumin: Stability and structural features. Int. J. Biol. Macromol. 2007, 40, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Wolfrum, C.; Poy, M.N.; Stoffel, M. Apolipoprotein M is required for prebeta-HDL formation and cholesterol efflux to HDL and protects against atherosclerosis. Nat. Med. 2005, 11, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Jauhiainen, M.; Moser, M.; Porse, B.; Ehnholm, C.; Boesl, M.; Dahlbäck, B.; Nielsen, L.B. Effect of apolipoprotein M on high density lipoprotein metabolism and atherosclerosis in low density lipoprotein receptor knock-out mice. J. Biol. Chem. 2008, 283, 1839–1847. [Google Scholar] [CrossRef] [PubMed]
- Elsøe, S.; Ahnström, J.; Christoffersen, C.; Hoofnagle, A.N.; Plomgaard, P.; Heinecke, J.W.; Binder, C.J.; Björkbacka, H.; Dahlbäck, B.; Nielsen, L.B. Apolipoprotein M binds oxidized phospholipids and increases the antioxidant effect of HDL. Atherosclerosis 2012, 221, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Kohama, T.; Olivera, A.; Edsall, L.; Nagiec, M.M.; Dickson, R.; Spiegel, S. Molecular cloning and functional characterization of murine sphingosine kinase. J. Biol. Chem. 1998, 273, 23722–23728. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Sugiura, M.; Nava, V.E.; Edsall, L.C.; Kono, K.; Poulton, S.; Milstien, S.; Kohama, T.; Spiegel, S. Molecular cloning and functional characterization of a novel mammalian sphingosine kinase type 2 isoform. J. Biol. Chem. 2000, 275, 19513–19520. [Google Scholar] [CrossRef] [PubMed]
- Hait, N.C.; Oskeritzian, C.A.; Paugh, S.W.; Milstien, S.; Spiegel, S. Sphingosine kinases, sphingosine 1-phosphate, apoptosis and diseases. Biochim. Biophys. Acta 2006, 1758, 2016–2026. [Google Scholar] [CrossRef] [PubMed]
- Książek, M.; Chacińska, M.; Chabowski, A.; Baranowski, M. Sources, metabolism, and regulation of circulating sphingosine-1-phosphate. J. Lipid Res. 2015, 56, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, S.; Milstien, S. Sphingosine-1-phosphate: An enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 2003, 4, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Pyne, N.J.; Dubois, G.; Pyne, S. Role of sphingosine 1-phosphate and lysophosphatidic acid in fibrosis. Biochim. Biophys. Acta 2013, 1831, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Ding, Q.; Yaqoob, U.; de Assuncao, T.M.; Verma, V.K.; Hirsova, P.; Cao, S.; Mukhopadhyay, D.; Huebert, R.C.; Shah, V.H. Exosome Adherence and Internalization by Hepatic Stellate Cells Triggers Sphingosine 1-Phosphate-dependent Migration. J. Biol. Chem. 2015, 290, 30684–30696. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.S.; Liu, C.H.; Sun, Y.; Chen, Y.; Swendeman, S.L.; Jung, B.; Chavez, D.; Cao, Z.; Christoffersen, C.; Nielsen, L.B.; et al. HDL activation of endothelial sphingosine-1-phosphate receptor-1 (S1P1) promotes regeneration and suppresses fibrosis in the liver. JCI Insight 2016, 1, e87058. [Google Scholar] [CrossRef] [PubMed]
- Ren, K.; Tang, Z.L.; Jiang, Y.; Tan, Y.M.; Yi, G.H. Apolipoprotein M. Clin. Chim. Acta 2015, 446, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Li, M.O.; Wan, Y.Y.; Sanjabi, S.; Robertson, A.K.L.; Flavell, R.A. Transforming growth factor-beta regulation of immune responses. Annu. Rev. Immunol. 2006, 24, 99–146. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-β: The master regulator of fibrosis. Nat. Rev. Nephrol. 2016, 12, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Ren, K.; Mo, Z.C.; Liu, X.; Tang, Z.L.; Jiang, Y.; Peng, X.S.; Zhang, Q.H.; Shi, J.F.; Yi, G.H. TGF-β Down-regulates Apolipoprotein M Expression through the TAK-1-JNK-c-Jun Pathway in HepG2 Cells. Lipids 2017, 52, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Hu, Y.W.; Zhao, Z.L.; Zheng, L.; Qiu, Y.R.; Huang, J.L.; Wu, X.J.; Mao, X.R.; Yang, J.; Zhao, J.Y.; et al. Anti-inflammatory effects of propofol are mediated by apolipoprotein M in a hepatocyte nuclear factor-1α-dependent manner. Arch. Biochem. Biophys. 2013, 533, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhao, J.Y.; Zhao, Z.L.; Ye, J.; Li, S.F.; Fang, H.H.; Gu, M.N.; Hu, Y.W.; Qin, Z.S. Propofol Attenuates Lipopolysaccharide-Induced Monocyte Chemoattractant Protein-1 Production Through Enhancing apoM and foxa2 Expression in HepG2 Cells. Inflammation 2015, 38, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Faber, K.; Axler, O.; Dahlbäck, B.; Nielsen, L.B. Characterization of apoM in normal and genetically modified mice. J. Lipid Res. 2004, 45, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Shih, D.Q.; Pearson, E.R.; Wolfrum, C.; Fajans, S.S.; Hattersley, A.T.; Stoffel, M. Regulation of apolipoprotein M gene expression by MODY3 gene hepatocyte nuclear factor-1alpha: Haploinsufficiency is associated with reduced serum apolipoprotein M levels. Diabetes 2003, 52, 2989–2995. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.Q.; Yu, Y.; Guo, S.D.; Cui, Y.J.; Hu, G.L.; Feng, L.; Wang, D.X.; Qin, S.C. Effects of apolipoprotein E deficiency on sphingosine-1-phosphate distribution in plasma and lipoproteins of mice. Zhonghua Xin Xue Guan Bing Za Zhi 2017, 45, 419–426. [Google Scholar] [PubMed]
- Christoffersen, C.; Ahnström, J.; Axler, O.; Christensen, E.I.; Dahlbäck, B.; Nielsen, L.B. The signal peptide anchors apolipoprotein M in plasma lipoproteins and prevents rapid clearance of apolipoprotein M from plasma. J. Biol. Chem. 2008, 283, 18765–18772. [Google Scholar] [CrossRef] [PubMed]
- Ahnström, J.; Axler, O.; Dahlbäck, B. HDL stimulates apoM secretion. Protein Pept. Lett. 2010, 17, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Axler, O.; Ahnström, J.; Dahlbäck, B. Apolipoprotein M associates to lipoproteins through its retained signal peptide. FEBS Lett. 2008, 582, 826–828. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Allegood, J.; Zhu, X.; Seo, J.; Gebre, A.K.; Boudyguina, E.; Cheng, D.; Chuang, C.C.; Shelness, G.S.; Spiegel, S.; et al. Uncleaved ApoM signal peptide is required for formation of large ApoM/sphingosine 1-phosphate (S1P)-enriched HDL particles. J. Biol. Chem. 2015, 290, 7861–7870. [Google Scholar] [CrossRef] [PubMed]
- Faber, K.; Hvidberg, V.; Moestrup, S.K.; Dahlbäck, B.; Nielsen, L.B. Megalin is a receptor for apolipoprotein M, and kidney-specific megalin-deficiency confers urinary excretion of apolipoprotein M. Mol. Endocrinol. 2006, 20, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Sorenson, R.C.; Aviram, M.; Bisgaier, C.L.; Billecke, S.; Hsu, C.; La Du, B.N. Properties of the retained N-terminal hydrophobic leader sequence in human serum paraoxonase/arylesterase. Chem. Biol. Interact. 1999, 119–120, 243–249. [Google Scholar] [CrossRef]
- Sorenson, R.C.; Bisgaier, C.L.; Aviram, M.; Hsu, C.; Billecke, S.; La Du, B.N. Human serum Paraoxonase/Arylesterase’s retained hydrophobic N-terminal leader sequence associates with HDLs by binding phospholipids: Apolipoprotein A-I stabilizes activity. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2214–2225. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Wellington, C.L.; Calabresi, L. HDL and cholesterol handling in the brain. Cardiovasc. Res. 2014, 103, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Mulya, A.; Seo, J.; Brown, A.L.; Gebre, A.K.; Boudyguina, E.; Shelness, G.S.; Parks, J.S. Apolipoprotein M expression increases the size of nascent pre beta HDL formed by ATP binding cassette transporter A1. J. Lipid Res. 2010, 51, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Wróblewska, M. The origin and metabolism of a nascent pre-β high density lipoprotein involved in cellular cholesterol efflux. Acta Biochim. Pol. 2011, 58, 275–285. [Google Scholar] [PubMed]
- Kerjaschki, D. Immunocytochemical localization of the Heymann nephritis antigen (GP330) in glomerular epithelial cells of normal Lewis rats. J. Exp. Med. 1983, 157, 667–686. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Pietromonaco, S.; Loo, A.K.; Farquhar, M.G. Complete cloning and sequencing of rat gp330/“megalin,” a distinctive member of the low density lipoprotein receptor gene family. Proc. Natl. Acad. Sci. USA 1994, 91, 9725–9729. [Google Scholar] [CrossRef] [PubMed]
- Sutter, I.; Park, R.; Othman, A.; Rohrer, L.; Hornemann, T.; Stoffel, M.; Devuyst, O.; von Eckardstein, A. Apolipoprotein M modulates erythrocyte efflux and tubular reabsorption of sphingosine-1-phosphate. J. Lipid Res. 2014, 55, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Tojo, A.; Kinugasa, S. Mechanisms of glomerular albumin filtration and tubular reabsorption. Int. J. Nephrol. 2012, 2012, 481520. [Google Scholar] [CrossRef] [PubMed]
- Amsellem, S.; Gburek, J.; Hamard, G.; Nielsen, R.; Willnow, T.E.; Devuyst, O.; Nexo, E.; Verroust, P.J.; Christensen, E.I.; Kozyraki, R. Cubilin is essential for albumin reabsorption in the renal proximal tubule. J. Am. Soc. Nephrol. 2010, 21, 1859–1867. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H. Time course and vectorial nature of albumin metabolism in isolated perfused rabbit PCT. Am. J. Physiol. 1988, 255, F520–F528. [Google Scholar] [PubMed]
- Aseem, O.; Smith, B.T.; Cooley, M.A.; Wilkerson, B.A.; Argraves, K.M.; Remaley, A.T.; Argraves, W.S. Cubilin Maintains Blood Levels of HDL and Albumin. J. Am. Soc. Nephrol. 2014, 25, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- Thuy, A.V.; Reimann, C.M.; Hemdan, N.Y.A.; Gräler, M.H. Sphingosine 1-phosphate in blood: Function, metabolism, and fate. Cell. Physiol. Biochem. 2014, 34, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Venkataraman, K.; Hwang, S.I.; Han, D.K.; Hla, T. A novel method to quantify sphingosine 1-phosphate by immobilized metal affinity chromatography. Prostaglandins Other Lipid Mediat. 2007, 84, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Kurano, M.; Tsukamoto, K.; Ohkawa, R.; Hara, M.; Iino, J.; Kageyama, Y.; Ikeda, H.; Yatomi, Y. Liver involvement in sphingosine 1-phosphate dynamism revealed by adenoviral hepatic overexpression of apolipoprotein M. Atherosclerosis 2013, 229, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.N.; Kobayashi, N.N.; Yamaguchi, A.; Nishi, T.T. Characterization of the ATP-dependent sphingosine 1-phosphate transporter in rat erythrocytes. J. Biol. Chem. 2009, 284, 21192–21200. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Guo, S.; Feng, Y.; Feng, L.; Cui, Y.; Song, G.; Luo, T.; Zhang, K.; Wang, Y.; Jiang, X.C.; Qin, S. Phospholipid transfer protein deficiency decreases the content of S1P in HDL via the loss of its transfer capability. Lipids 2014, 49, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Malchinkhuu, E.; Horiuchi, Y.; Mogi, C.; Tomura, H.; Tosaka, M.; Yoshimoto, Y.; Kuwabara, A.; Okajima, F. Critical role of ABCA1 transporter in sphingosine 1-phosphate release from astrocytes. J. Neurochem. 2007, 103, 2610–2619. [Google Scholar] [CrossRef] [PubMed]
- Hla, T.; Venkataraman, K.; Michaud, J. The vascular S1P gradient-cellular sources and biological significance. Biochim. Biophys. Acta 2008, 1781, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Anada, Y.; Tani, M.; Ikeda, M.; Sano, T.; Kihara, A.; Igarashi, Y. Lack of sphingosine 1-phosphate-degrading enzymes in erythrocytes. Biochem. Biophys. Res. Commun. 2007, 357, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Frej, C.; Andersson, A.; Larsson, B.; Guo, L.J.; Norström, E.; Happonen, K.E.; Dahlbäck, B. Quantification of sphingosine 1-phosphate by validated LC-MS/MS method revealing strong correlation with apolipoprotein M in plasma but not in serum due to platelet activation during blood coagulation. Anal. Bioanal. Chem. 2015, 407, 8533–8542. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Yatomi, Y.; Ohta, M.; Osada, M.; Kazama, F.; Satoh, K.; Nakahara, K.; Ozaki, Y. Sphingosine 1-phosphate-related metabolism in the blood vessel. J. Biochem. 2005, 138, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Shoji, A.; Kubo, Y.; Nada, S.; Yamaguchi, A. Cloning of rat ABCA7 and its preferential expression in platelets. Biochem. Biophys. Res. Commun. 2003, 304, 777–782. [Google Scholar] [CrossRef]
- Yatomi, Y.; Ruan, F.; Hakomori, S.; Igarashi, Y. Sphingosine-1-phosphate: A platelet-activating sphingolipid released from agonist-stimulated human platelets. Blood 1995, 86, 193–202. [Google Scholar] [PubMed]
- Kobayashi, N.; Nishi, T.; Hirata, T.; Kihara, A.; Sano, T.; Igarashi, Y.; Yamaguchi, A. Sphingosine 1-phosphate is released from the cytosol of rat platelets in a carrier-mediated manner. J. Lipid Res. 2006, 47, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Yang, P.; Proia, R.L.; Hla, T. Erythrocyte-derived sphingosine 1-phosphate is essential for vascular development. J. Clin. Investig. 2014, 124, 4823–4828. [Google Scholar] [CrossRef] [PubMed]
- Aird, W.C. Endothelial cell heterogeneity. Crit. Care Med. 2003, 31, S221–S230. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ren, K.; Suo, R.; Xiong, S.L.; Zhang, Q.H.; Mo, Z.C.; Tang, Z.L.; Jiang, Y.; Peng, X.S.; Yi, G.H. ApoA-I induces S1P release from endothelial cells through ABCA1 and SR-BI in a positive feedback manner. J. Physiol. Biochem. 2016, 72, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.; Ebenezer, D.L.; Berdyshev, E.V.; Bronova, I.A.; Shaaya, M.; Harijith, A.; Natarajan, V. Role of Sphingosine Kinase 1 and S1P Transporter Spns2 in HGF-mediated Lamellipodia Formation in Lung Endothelium. J. Biol. Chem. 2016, 291, 27187–27203. [Google Scholar] [CrossRef] [PubMed]
- Nishi, T.; Kobayashi, N.; Hisano, Y.; Kawahara, A.; Yamaguchi, A. Molecular and physiological functions of sphingosine 1-phosphate transporters. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2014, 1841, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, S.; Simmons, S.; Kawamura, S.; Inoue, A.; Orba, Y.; Tokudome, T.; Sunden, Y.; Arai, Y.; Moriwaki, K.; Ishida, J.; et al. The sphingosine-1-phosphate transporter Spns2 expressed on endothelial cells regulates lymphocyte trafficking in mice. J. Clin. Investig. 2012, 122, 1416–1426. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, A.; Bréart, B.; Ramos-Perez, W.D.; Pitt, L.A.; Gobert, M.; Sunkara, M.; Lafaille, J.J.; Morris, A.J.; Schwab, S.R. The transporter Spns2 is required for secretion of lymph but not plasma sphingosine-1-phosphate. Cell Rep. 2012, 2, 1104–1110. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.A.; Roth, C.B.; Jo, E.; Griffith, M.T.; Scott, F.L.; Reinhart, G.; Desale, H.; Clemons, B.; Cahalan, S.M.; Schuerer, S.C.; et al. Crystal structure of a lipid G protein-coupled receptor. Science 2012, 335, 851–855. [Google Scholar] [CrossRef] [PubMed]
- Pulkoski-Gross, M.J.; Donaldson, J.C.; Obeid, L.M. Sphingosine-1-phosphate metabolism: A structural perspective. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 298–313. [Google Scholar] [CrossRef] [PubMed]
- Wilkerson, B.A.; Grass, G.D.; Wing, S.B.; Argraves, W.S.; Argraves, K.M. Sphingosine 1-phosphate (S1P) carrier-dependent regulation of endothelial barrier: High density lipoprotein (HDL)-S1P prolongs endothelial barrier enhancement as compared with albumin-S1P via effects on levels, trafficking, and signaling of S1P1. J. Biol. Chem. 2012, 287, 44645–44653. [Google Scholar] [CrossRef] [PubMed]
- Galvani, S.; Sanson, M.; Blaho, V.A.; Swendeman, S.L.; Obinata, H.; Conger, H.; Dahlbäck, B.; Kono, M.; Proia, R.L.; Smith, J.D.; et al. HDL-bound sphingosine 1-phosphate acts as a biased agonist for the endothelial cell receptor S1P1 to limit vascular inflammation. Sci. Signal. 2015, 8, ra79. [Google Scholar] [CrossRef] [PubMed]
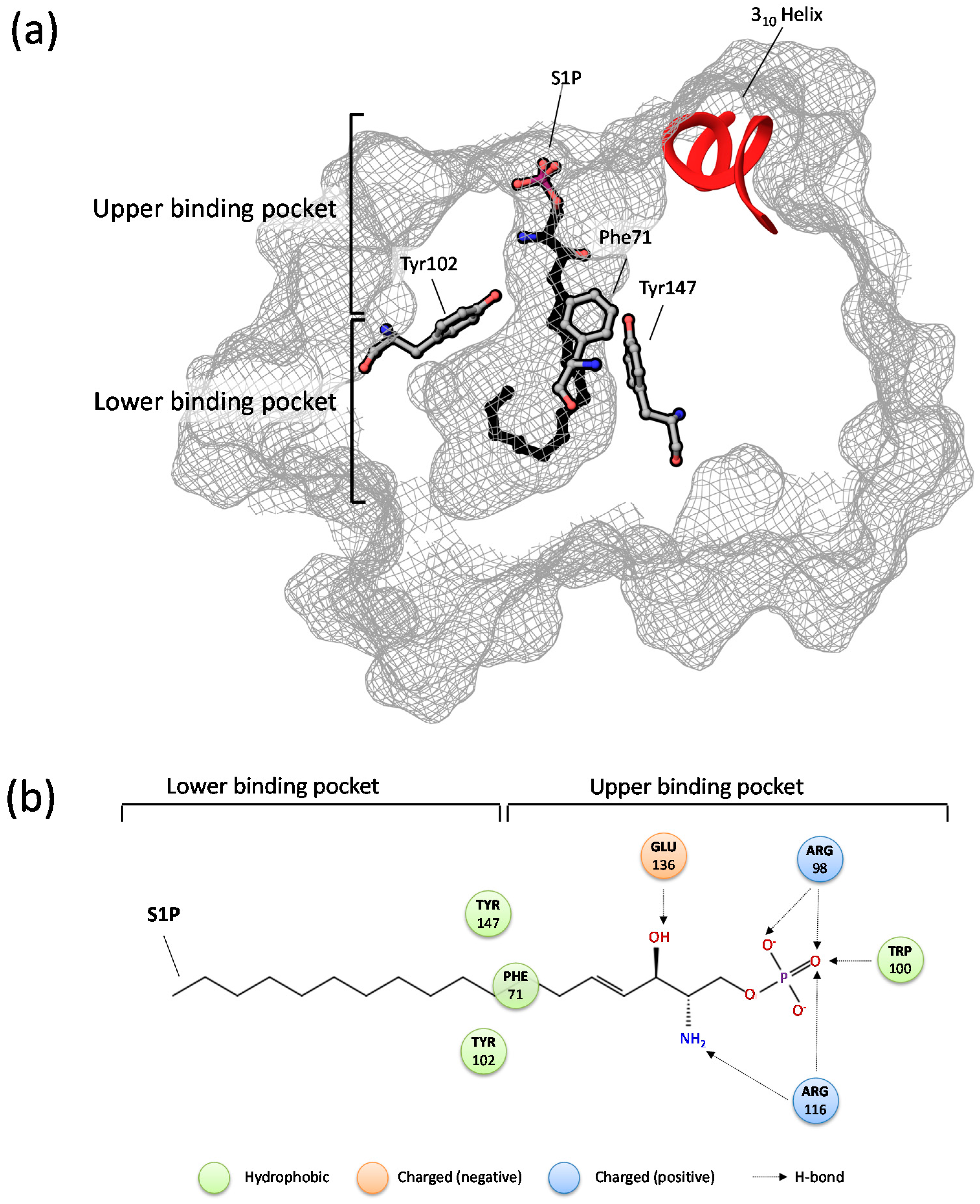
- Zhang, H.; Pluhackova, K.; Jiang, Z.; Böckmann, R.A. Binding Characteristics of Sphingosine-1-Phosphate to ApoM hints to Assisted Release Mechanism via the ApoM Calyx-Opening. Sci. Rep. 2016, 6, 30655. [Google Scholar] [CrossRef] [PubMed]
- Rhode, S.; Breuer, A.; Hesse, J.; Sonnleitner, M.; Pagler, T.A.; Doringer, M.; Schütz, G.J.; Stangl, H. Visualization of the uptake of individual HDL particles in living cells via the scavenger receptor class B type I. Cell Biochem. Biophys. 2004, 41, 343–356. [Google Scholar] [CrossRef]
- Ruiz, M.; Okada, H.; Dahlbäck, B. HDL-associated ApoM is anti-apoptotic by delivering sphingosine 1-phosphate to S1P1 & S1P3 receptors on vascular endothelium. Lipids Health Dis. 2017, 16, 36. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Koyama, Y.; Liu, X.; Xu, J.; Ma, H.Y.; Liang, S.; Kim, I.H.; Brenner, D.A.; Kisseleva, T. Promising Therapy Candidates for Liver Fibrosis. Front. Physiol. 2016, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Kinny-Köster, B.; Bartels, M.; Scholz, M.; Seehofer, D.; Berg, T.; Engelmann, C.; Thiery, J.; Ceglarek, U.; Kaiser, T. Low sphingosine-1-phosphate plasma levels are predictive for increased mortality in patients with liver cirrhosis. PLoS ONE 2017, 12, e0174424. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nat. Med. 2012, 18, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Takuwa, Y.; Ikeda, H.; Okamoto, Y.; Takuwa, N.; Yoshioka, K. Sphingosine-1-phosphate as a mediator involved in development of fibrotic diseases. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2013, 1831, 185–192. [Google Scholar] [CrossRef] [PubMed]
- WHO. Age-Standardized Death Rates, Liver Cirrhosis (15+) by Country. Available online: http://apps.who.int/gho/data/node.main.A1092?lang=en (accessed on 27 June 2017).
- Crespo Yanguas, S.; Cogliati, B.; Willebrords, J.; Maes, M.; Colle, I.; van den Bossche, B.; de Oliveira, C.P.M.S.; Andraus, W.; Alves, V.A.; Leclercq, I.; et al. Experimental models of liver fibrosis. Arch. Toxicol. 2016, 90, 1025–1048. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Yuan, W.G.; He, P.; Lei, J.H.; Wang, C.X. Liver fibrosis and hepatic stellate cells: Etiology, pathological hallmarks and therapeutic targets. World J. Gastroenterol. 2016, 22, 10512–10522. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.J.; McGuire, R.F. Extracellular matrix gene expression increases preferentially in rat lipocytes and sinusoidal endothelial cells during hepatic fibrosis in vivo. J. Clin. Investig. 1990, 86, 1641–1648. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, Y.; Higashiyama, R. Interplay between bone marrow and liver in the pathogenesis of hepatic fibrosis. Hepatol. Res. 2012, 42, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Nikfarjam, M.; Malcontenti-Wilson, C.; Fanartzis, M.; Daruwalla, J.; Christophi, C. A model of partial hepatectomy in mice. J. Investig. Surg. 2004, 17, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Marques, T.G.; Chaib, E.; da Fonseca, J.H.; Lourenço, A.C.R.; Silva, F.D.; Ribeiro, M.A.F.; Galvão, F.H. F.; D’Albuquerque, L.A.C. Review of experimental models for inducing hepatic cirrhosis by bile duct ligation and carbon tetrachloride injection. Acta Cir. Bras. 2012, 27, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.K.; Maroni, L.; Marzioni, M.; Ahmed, S.T.; Milad, M.; Ray, D.; Alpini, G.; Glaser, S.S. Mouse models of liver fibrosis mimic human liver fibrosis of different etiologies. Curr. Pathobiol. Rep. 2014, 2, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.S.; Nolan, D.J.; Butler, J.M.; James, D.; Babazadeh, A.O.; Rosenwaks, Z.; Mittal, V.; Kobayashi, H.; Shido, K.; Lyden, D.; et al. Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration. Nature 2010, 468, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Srivastava, K.; Wieland, M.; Runge, A.; Mogler, C.; Besemfelder, E.; Terhardt, D.; Vogel, M.J.; Cao, L.; Korn, C.; et al. Endothelial cell-derived angiopoietin-2 controls liver regeneration as a spatiotemporal rheostat. Science 2014, 343, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.M.; Kitamura, T.; Ikejima, K.; Enomoto, N.; Yamashina, S.; Suzuki, S.; Takei, Y.; Sato, N. Sphingosine 1-phosphate protects rat liver sinusoidal endothelial cells from ethanol-induced apoptosis: Role of intracellular calcium and nitric oxide. Hepatology 2006, 44, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Nowatari, T.; Murata, S.; Nakayama, K.; Sano, N.; Maruyama, T.; Nozaki, R.; Ikeda, N.; Fukunaga, K.; Ohkohchi, N. Sphingosine 1-phosphate has anti-apoptotic effect on liver sinusoidal endothelial cells and proliferative effect on hepatocytes in a paracrine manner in human. Hepatol. Res. 2015, 45, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Murata, S.; Takahashi, K.; Nozaki, R.; Ohshiro, Y.; Ikeda, N.; Pak, S.; Myronovych, A.; Hisakura, K.; Fukunaga, K.; et al. Activation of human liver sinusoidal endothelial cell by human platelets induces hepatocyte proliferation. J. Hepatol. 2010, 53, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, R.; Nakano, Y.; Ohkohchi, N. Platelet administration via the portal vein promotes liver regeneration in rats after 70% hepatectomy. Ann. Surg. 2011, 253, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, K.; Hla, T. Vascular and Immunobiology of the Circulatory Sphingosine 1-Phosphate Gradient. Annu. Rev. Physiol. 2017, 79. [Google Scholar] [CrossRef] [PubMed]
- Takuwa, Y.; Du, W.; Qi, X.; Okamoto, Y.; Takuwa, N.; Yoshioka, K. Roles of sphingosine-1-phosphate signaling in angiogenesis. World J. Biol. Chem. 2010, 1, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Serriere-Lanneau, V.; Teixeira-Clerc, F.; Li, L.; Schippers, M.; de Wries, W.; Julien, B.; Tran-Van-Nhieu, J.; Manin, S.; Poelstra, K.; Chun, J.; et al. The sphingosine 1-phosphate receptor S1P2 triggers hepatic wound healing. FASEB J. 2007, 21, 2005–2013. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Yue, S.; Yang, L.; Liu, X.; Han, Z.; Zhang, Y.; Li, L. Sphingosine kinase/sphingosine 1-phosphate (S1P)/S1P receptor axis is involved in liver fibrosis-associated angiogenesis. J. Hepatol. 2013, 59, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, Y.; Ikeda, H.; Watanabe, N.; Nagamine, M.; Kusumoto, Y.; Yashiro, M.; Satoh, Y.; Shimosawa, T.; Shinozaki, K.; Tomiya, T.; et al. Antagonism of sphingosine 1-phosphate receptor 2 causes a selective reduction of portal vein pressure in bile duct-ligated rodents. Hepatology 2012, 56, 1427–1438. [Google Scholar] [CrossRef] [PubMed]
- Kibe, A.; Holzbach, R.; LaRusso, N.; Mao, S. Inhibition of cholesterol crystal formation by apolipoproteins in supersaturated model bile. Science 1984, 225, 514–516. [Google Scholar] [CrossRef] [PubMed]
- Reshetnyak, V.I. Physiological and molecular biochemical mechanisms of bile formation. World J. Gastroenterol. 2013, 19, 7341. [Google Scholar] [CrossRef] [PubMed]
- Hadizadeh, S.; King, D.N.; Shah, S.; Sewer, M.B. Sphingosine-1-phosphate regulates the expression of the liver receptor homologue-1. Mol. Cell. Endocrinol. 2008, 283, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Pan, Y.; Zhou, H.H.; Zhou, H.H.; Tang, L.J. Apolipoprotein M regulates the orphan nuclear receptor LRH-1 gene expression through binding to its promoter region in HepG2 cells. Drug Des. Dev. Ther. 2015, 9, 2375. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Aoki, H.; Yang, J.; Peng, K.; Liu, R.; Li, X.; Qiang, X.; Sun, L.; Gurley, E.C.; Lai, G.; et al. The role of sphingosine 1-phosphate receptor 2 in bile-acid-induced cholangiocyte proliferation and cholestasis-induced liver injury in mice. Hepatology 2017, 65, 2005–2018. [Google Scholar] [CrossRef] [PubMed]
- Obermeier, B.; Daneman, R.; Ransohoff, R.M. Development, maintenance and disruption of the blood-brain barrier. Nat. Med. 2013, 19, 1584–1596. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Engelhardt, B.; Ransohoff, R.M. Capture, crawl, cross: The T cell code to breach the blood-brain barriers. Trends Immunol. 2012, 33, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Pyne, N.J.; McNaughton, M.; Boomkamp, S.; MacRitchie, N.; Evangelisti, C.; Martelli, A.M.; Jiang, H.R.; Ubhi, S.; Pyne, S. Role of sphingosine 1-phosphate receptors, sphingosine kinases and sphingosine in cancer and inflammation. Adv. Biol. Regul. 2016, 60, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Aoki, M.; Aoki, H.; Ramanathan, R.; Hait, N.C.; Takabe, K. Sphingosine-1-Phosphate signaling in immune cells and inflammation: Roles and therapeutic potential. Mediators Inflamm. 2016, 2016, 8606878. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, G.K.; Dev, K.K. Targeting S1P receptors in experimental autoimmune encephalomyelitis in mice improves early deficits in locomotor activity and increases ultrasonic vocalisations. Sci. Rep. 2014, 4, 5051. [Google Scholar] [CrossRef] [PubMed]
- Dev, K.K.; Mullershausen, F.; Mattes, H.; Kuhn, R.R.; Bilbe, G.; Hoyer, D.; Mir, A. Brain sphingosine-1-phosphate receptors: Implication for FTY720 in the treatment of multiple sclerosis. Pharmacol. Ther. 2008, 117, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Candido, K.; Soufi, H.; Bandyopadhyay, M.; Dasgupta, S. Therapeutic Impact of Sphingosine 1-phosphate Receptor Signaling in Multiple Sclerosis. Mini Rev. Med. Chem. 2016, 16, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.F.; Bowen, J.D.; Reder, A.T. The Direct Effects of Fingolimod in the Central Nervous System: Implications for Relapsing Multiple Sclerosis. CNS Drugs 2016, 30, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Groves, A.; Kihara, Y.; Chun, J. Fingolimod: Direct CNS effects of sphingosine 1-phosphate (S1P) receptor modulation and implications in multiple sclerosis therapy. J. Neurol. Sci. 2013, 328, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Blaho, V.A.; Galvani, S.; Engelbrecht, E.; Liu, C.; Swendeman, S.L.; Kono, M.; Proia, R.L.; Steinman, L.; Han, M.H.; Hla, T. HDL-bound sphingosine-1-phosphate restrains lymphopoiesis and neuroinflammation. Nature 2015, 523, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Constantinescu, C.S.; Farooqi, N.; O’Brien, K.; Gran, B. Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br. J. Pharmacol. 2011, 164, 1079–1106. [Google Scholar] [CrossRef] [PubMed]
- Grehan, S.; Tse, E.; Taylor, J.M. Two distal downstream enhancers direct expression of the human apolipoprotein E gene to astrocytes in the brain. J. Neurosci. 2001, 21, 812–822. [Google Scholar] [PubMed]
- Navarro, A.; Méndez, E.; Diaz, C.; del Valle, E.; Martínez-Pinilla, E.; Ordóñez, C.; Tolivia, J. Lifelong expression of apolipoprotein D in the human brainstem: Correlation with reduced age-related neurodegeneration. PLoS ONE 2013, 8, e77852. [Google Scholar] [CrossRef] [PubMed]
- Fagan, A.M.; Holtzman, D.M.; Munson, G.; Mathur, T.; Schneider, D.; Chang, L.K.; Getz, G.S.; Reardon, C.A.; Lukens, J.; Shah, J.A.; LaDu, M.J. Unique lipoproteins secreted by primary astrocytes from wild type, apoE (−/−), and human apoE transgenic mice. J. Biol. Chem. 1999, 274, 30001–30007. [Google Scholar] [CrossRef] [PubMed]
- DeMattos, R.B.; Brendza, R.P.; Heuser, J.E.; Kierson, M.; Cirrito, J.R.; Fryer, J.; Sullivan, P.M.; Fagan, A.M.; Han, X.; Holtzman, D.M. Purification and characterization of astrocyte-secreted apolipoprotein E and J-containing lipoproteins from wild-type and human apoE transgenic mice. Neurochem. Int. 2001, 39, 415–425. [Google Scholar] [CrossRef]
- Martínez-Bujidos, M.; Rull, A.; González-Cura, B.; Pérez-Cuéllar, M.; Montoliu-Gaya, L.; Villegas, S.; Ordóñez-Llanos, J.; Sánchez-Quesada, J.L. Clusterin/apolipoprotein J binds to aggregated LDL in human plasma and plays a protective role against LDL aggregation. FASEB J. 2015, 29, 1688–1700. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R.W. Central Nervous System LipoproteinsHighlights. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1305–1315. [Google Scholar] [CrossRef] [PubMed]
- Stukas, S.; Robert, J.; Lee, M.; Kulic, I.; Carr, M.; Tourigny, K.; Fan, J.; Namjoshi, D.; Lemke, K.; DeValle, N.; et al. Intravenously injected human apolipoprotein A-I rapidly enters the central nervous system via the choroid plexus. J. Am. Heart Assoc. 2014, 3, e001156. [Google Scholar] [CrossRef] [PubMed]
- Stukas, S.; May, S.; Wilkinson, A.; Chan, J.; Donkin, J.; Wellington, C.L. The LXR agonist GW3965 increases apoA-I protein levels in the central nervous system independent of ABCA1. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2012, 1821, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Karasinska, J.M.; Rinninger, F.; Lütjohann, D.; Ruddle, P.; Franciosi, S.; Kruit, J.K.; Singaraja, R.R.; Hirsch-Reinshagen, V.; Fan, J.; Brunham, L.R.; et al. Specific loss of brain ABCA1 increases brain cholesterol uptake and influences neuronal structure and function. J. Neurosci. 2009, 29, 3579–3589. [Google Scholar] [CrossRef] [PubMed]
- Balazs, Z.; Panzenboeck, U.; Hammer, A.; Sovic, A.; Quehenberger, O.; Malle, E.; Sattler, W. Uptake and transport of high-density lipoprotein (HDL) and HDL-associated alpha-tocopherol by an in vitro blood-brain barrier model. J. Neurochem. 2004, 89, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Möckel, B.; Zinke, H.; Flach, R.; Weiss, B.; Weiler-Güttler, H.; Gassen, H.G. Expression of apolipoprotein A-I in porcine brain endothelium in vitro. J. Neurochem. 1994, 62, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xiong, S.L.; Yi, G.H. ABCA1, ABCG1, and SR-BI: Transit of HDL-associated sphingosine-1-phosphate. Clin. Chim. Acta 2012, 413, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Chirackal Manavalan, A.P.; Kober, A.; Metso, J.; Lang, I.; Becker, T.; Hasslitzer, K.; Zandl, M.; Fanaee-Danesh, E.; Pippal, J.B.; Sachdev, V.; et al. Phospholipid transfer protein is expressed in cerebrovascular endothelial cells and involved in high density lipoprotein biogenesis and remodeling at the blood-brain barrier. J. Biol. Chem. 2014, 289, 4683–4698. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, K.; Liu, C.H.; Faraco, G.; Galvani, S.; Smith, H.K.; Burg, N.; Anrather, J.; Sanchez, T.; Iadecola, C.; Hla, T. Size-selective opening of the blood–brain barrier by targeting endothelial sphingosine 1–phosphate receptor 1. Proc. Natl. Acad. Sci. USA 2017, 114, 4531–4536. [Google Scholar] [CrossRef] [PubMed]
| Ligand | ApoM | ApoD | RBP | SA |
|---|---|---|---|---|
| Retinoic acid | 1.8 µM [18] | 4.0 µM [19] ~2.7 µM [28] | 0.21 µM [20] ~0.18 µM [28] | 33.3 µM [29] |
| Retinol | 2.2 µM [18] | 0.2 µM [19] ~0.08 µM [28] | 0.19 µM [20] ~0.26 µM [28] | 13.2 µM [29] |
| S1P | rApoM ~0.9 µM [8] HDL associated 0.021 µM [24] LDL associated 0.0024 µM [24] | N/A | N/A | 22.0 µM [24] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajny, S.; Christoffersen, C. A Novel Perspective on the ApoM-S1P Axis, Highlighting the Metabolism of ApoM and Its Role in Liver Fibrosis and Neuroinflammation. Int. J. Mol. Sci. 2017, 18, 1636. https://doi.org/10.3390/ijms18081636
Hajny S, Christoffersen C. A Novel Perspective on the ApoM-S1P Axis, Highlighting the Metabolism of ApoM and Its Role in Liver Fibrosis and Neuroinflammation. International Journal of Molecular Sciences. 2017; 18(8):1636. https://doi.org/10.3390/ijms18081636
Chicago/Turabian StyleHajny, Stefan, and Christina Christoffersen. 2017. "A Novel Perspective on the ApoM-S1P Axis, Highlighting the Metabolism of ApoM and Its Role in Liver Fibrosis and Neuroinflammation" International Journal of Molecular Sciences 18, no. 8: 1636. https://doi.org/10.3390/ijms18081636
APA StyleHajny, S., & Christoffersen, C. (2017). A Novel Perspective on the ApoM-S1P Axis, Highlighting the Metabolism of ApoM and Its Role in Liver Fibrosis and Neuroinflammation. International Journal of Molecular Sciences, 18(8), 1636. https://doi.org/10.3390/ijms18081636