Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease
Abstract
:1. Introduction
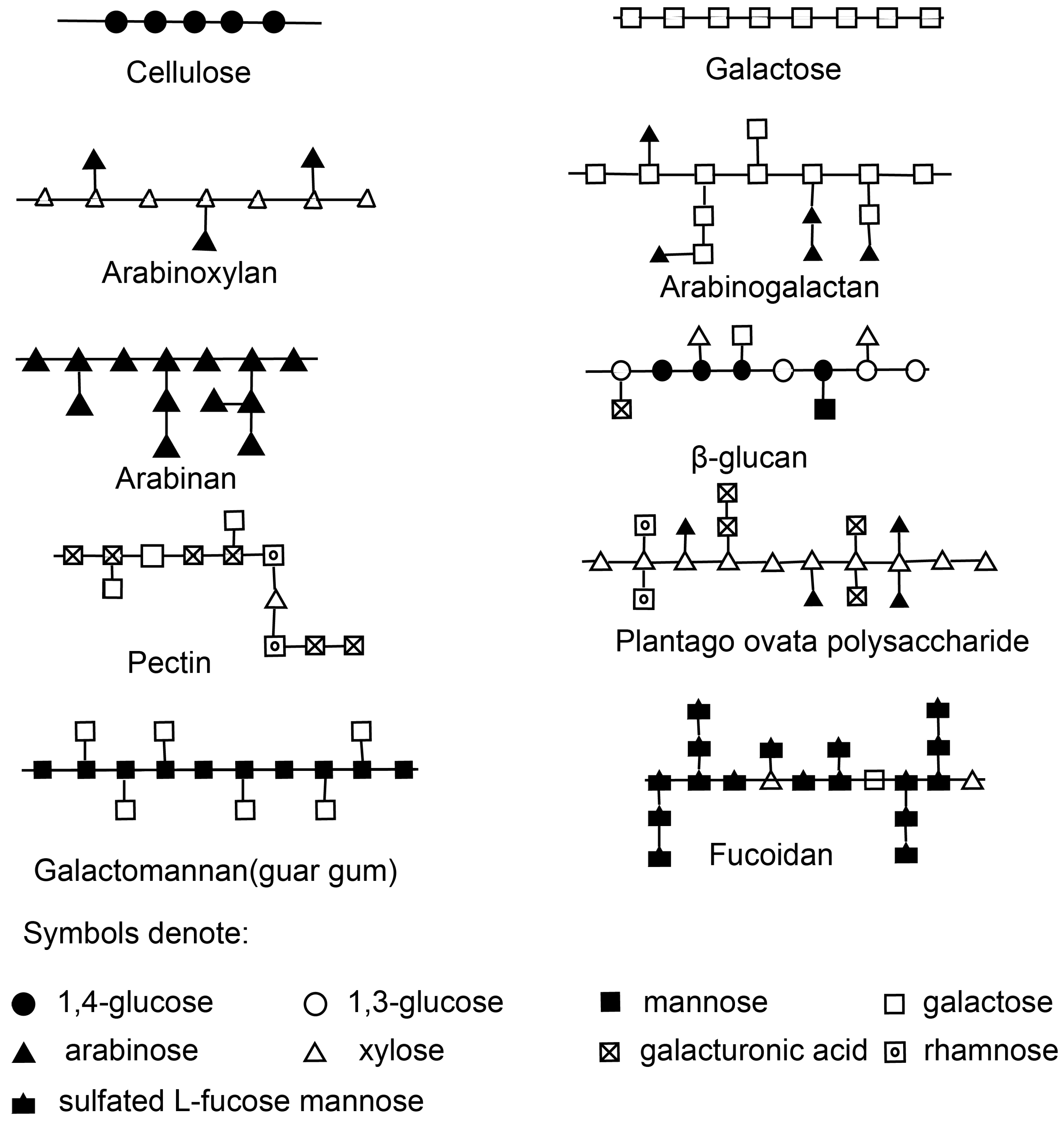
2. Structures, Compositions, and Sources of NSPs
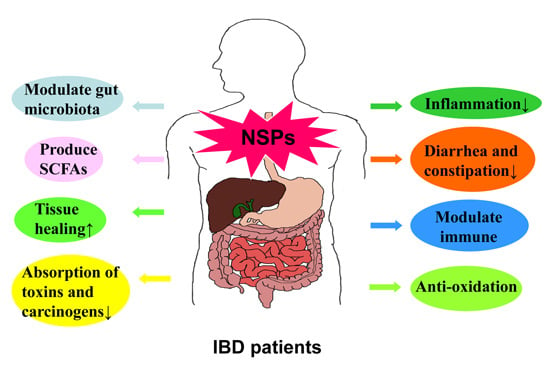
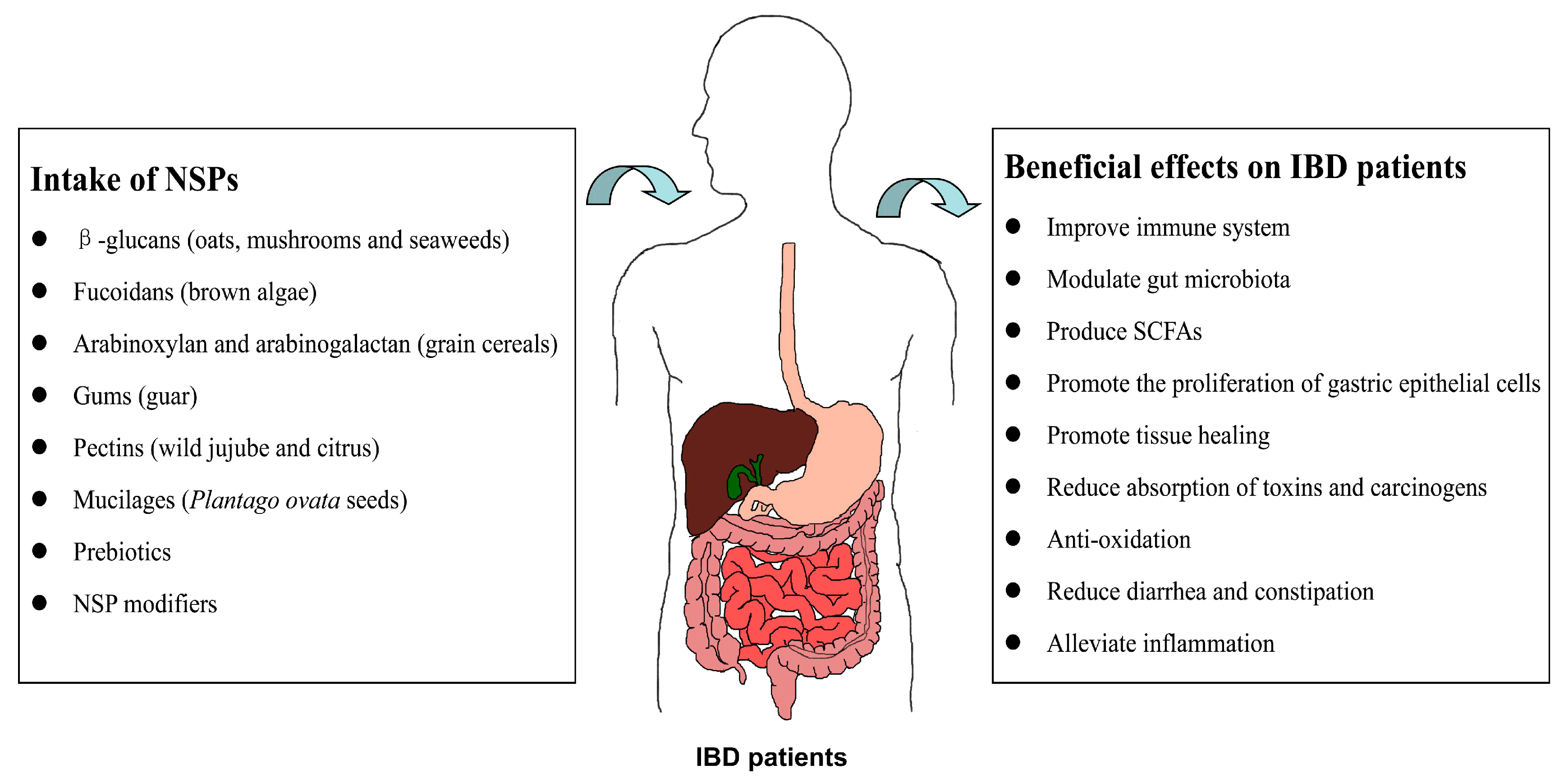
3. Effects of NSPs on IBD
3.1. Oat Bran Glucan
3.2. Mushroom Glucan
3.3. Seaweed-Derived β-Glucan
3.4. Other Glucan
3.5. Fucoidan
3.6. Polysaccharides from Plantago ovata Seeds
3.7. Wheat Bran and Barley
3.8. Prebiotics
3.9. Gums
3.10. Pectins
3.11. Modification of NSPs
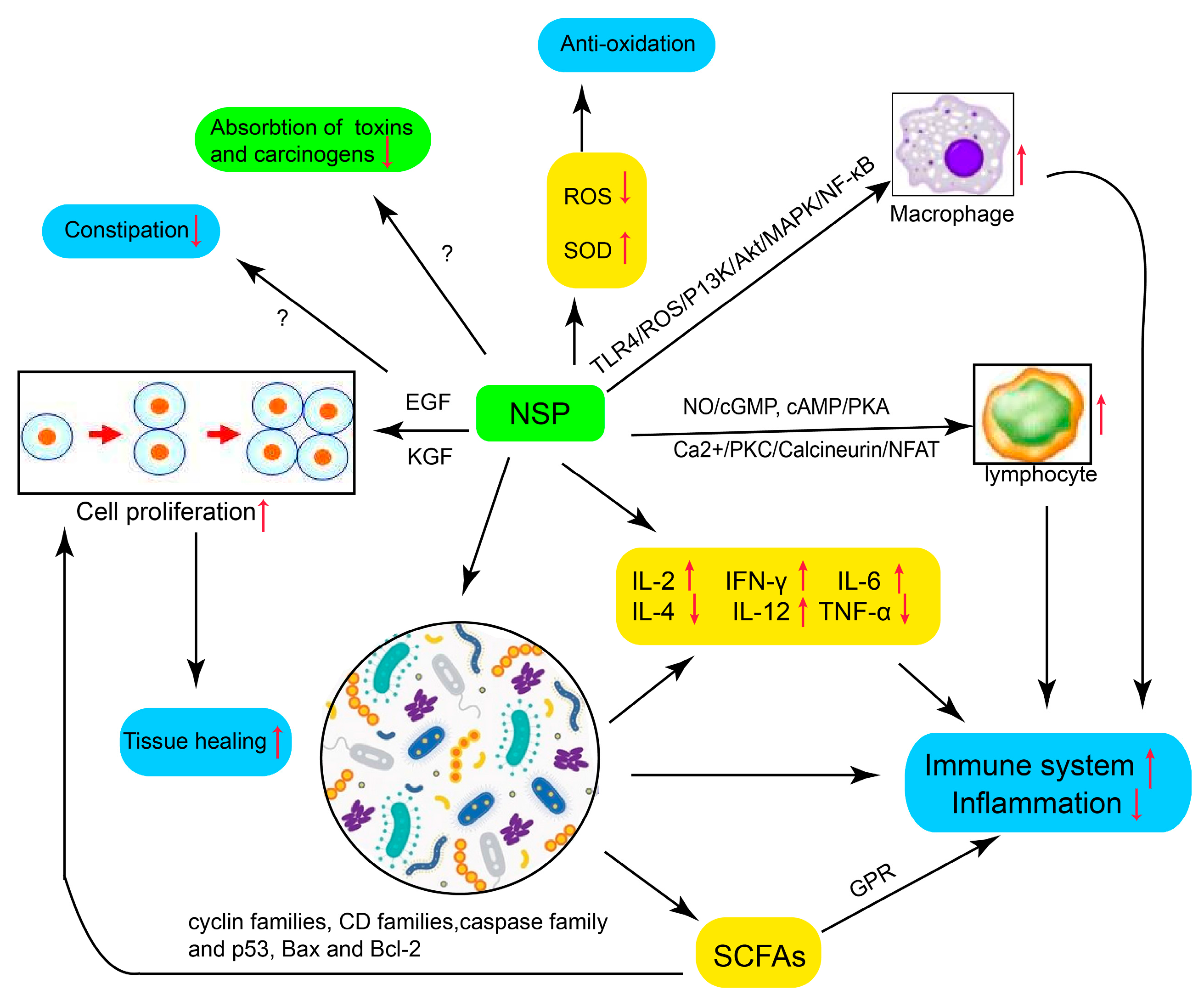
4. Mechanisms of NSPs in the Interventions of IBD
4.1. Promote Immune System and Reduce Inflammation
4.2. Modulate Gut Microbiota and Reduce Inflammation
4.3. Produce SCFAs
4.4. Promote the Proliferation of Gastric Epithelial Cells and Tissue Healing
4.5. Reduce Absorption of Toxins and Carcinogens
4.6. Anti-Oxidation in the Process of Inflammation
4.7. Reduce Constipation
5. Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| IBD | inflammatory bowel disease |
| UC | ulcerative colitis |
| CD | Crohn’s disease |
| NSP | non-starch polysaccharide |
| DF | dietary fiber |
| SCFAs | short chain fatty acids |
| CRC | colorectal cancer |
| MDA | malondialdehyde |
| MPO | myeloperoxidase |
| PCNA | proliferating cell nuclear antigen |
| DSS | dextran sulfate |
| Treg | regulatory T cells |
| NK cell | natural killer cell |
| HLA-B27 | human leucocyte antigen-B27 |
| TNBS | trinitrobenzenesulfonic acid |
| GB | germinated barley |
| DAI | disease activity index |
| WB | wheat bran |
| RS | resistant starch |
| ISAPP | International Scientific Association for Probiotics and Prebiotics |
| TLR | toll-like receptor |
| IBS | irritable bowel syndrome |
| PHGG | partially hydrolyzed guar gum |
| WJPs | wild jujube polysaccharides |
| TER | transepithelial electrical resistance |
| AMPK | adenosine 5′-monophosphate activated protein kinase |
| FODMAPs | fermentable, oligo-, di-, mono-saccharides and polyols |
| TNF-α | tumor necrosis factor α |
| IL-1 | interleukin-1 |
| AP | A. sinensis polysaccharide |
| IFN-γ | interferon-γ |
| Th- | T helper cell |
| HFD | high-fat diet |
| GI | gastrointestinal |
| DC | dendritic cell |
| ITF | inulin-type fructans |
| GPRs | G-protein-coupled receptors |
| Fas | APO-1/CD95 |
| EGF | epidermal growth factor |
| KGF | keratinocyte growth factor |
References
- Kemp, R.; Dunn, E.; Schultz, M. Immunomodulators in inflammatory bowel disease: An emerging role for biologic agents. BioDrugs 2013, 27, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, G.C.; Chong, C.A.; Chong, R.Y. National estimates of the burden of inflammatory bowel disease among racial and ethnic groups in the United States. J. Crohns Colitis 2014, 8, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; Kaplan, G.G. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012, 142, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, D.C.; Sandborn, W.J. Inflammatory bowel disease: Clinical aspects and established and evolving therapies. Lancet 2007, 369, 1641–1657. [Google Scholar] [CrossRef]
- Lim, W.C.; Wang, Y.; MacDonald, J.K.; Hanauer, S. Aminosalicylates for induction of remission or response in Crohn’s disease. Cochrane Database Syst. Rev. 2016, 7. [Google Scholar] [CrossRef]
- Sullivan, P.W.; Ghushchyan, V.H.; Globe, G.; Schatz, M. Oral corticosteroid exposure and adverse effects in asthmatic patients. J. Allergy Clin. Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Bryant, R.V.; Brain, O.; Travis, S.P. Conventional drug therapy for inflammatory bowel disease. Scand. J. Gastroenterol. 2015, 50, 90–112. [Google Scholar] [CrossRef] [PubMed]
- Neuman, M.G.; Nanau, R.M. Inflammatory bowel disease: Role of diet, microbiota, life style. Transl. Res. 2012, 160, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Belalcazar, L.M.; Anderson, A.M.; Lang, W.; Schwenke, D.C.; Haffner, S.M.; Yatsuya, H.; Rushing, J.; Vitolins, M.Z.; Reeves, R.; Pi-Sunyer, F.X.; et al. Fiber intake and plasminogen activator inhibitor-1 in type 2 diabetes: Look AHEAD (Action for Health in Diabetes) trial findings at baseline and year 1. J. Acad. Nutr. Diet 2014, 114, 1800–1810. [Google Scholar] [CrossRef] [PubMed]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Montagne, L.; Pluske, J.R.; Hampson, D.J. A review of interactions between dietary fibre and the intestinal mucosa, and their consequences on digestive health in young non-ruminant animals. Anim. Feed Sci. Tech. 2003, 108, 95–117. [Google Scholar] [CrossRef]
- Mendis, M.; Leclerc, E.; Simsek, S. Arabinoxylans, gut microbiota and immunity. Carbohyd. Polym. 2016, 139, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Nie, S. The structure of mushroom polysaccharides and their beneficial role in health. Food Funct. 2015, 6, 3205–3217. [Google Scholar] [CrossRef] [PubMed]
- Bobadilla, F.; Rodriguez-Tirado, C.; Imarai, M.; Galotto, M.J.; Andersson, R. Soluble β-1,3/1,6-glucan in seaweed from the southern hemisphere and its immunomodulatory effect. Carbonhyd. Polym. 2013, 92, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, B.; Hadar, Y. Possible mechanisms of action of mushroom-derived glucan on inflammatory bowel disease and associated cancer. Ann. Transl. Med. 2014, 2, 19. [Google Scholar] [PubMed]
- Asp, N.-G. Dietary fibre- definition, chemistry and analytical determination. Mol. Asp. Med. 1986, 9, 17–29. [Google Scholar] [CrossRef]
- Lovegrove, A.; Edwards, C.H.; de Noni, I.; Patel, H.; El, S.N.; Grassby, T.; Zielke, C.; Ulmius, M.; Nilsson, L.; Butterworth, P.J.; et al. Role of polysaccharides in food, digestion, and health. Crit. Rev. Food. Sci. Nutr. 2017, 57, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Galvez, J.; Rodriguez-Cabezas, M.E.; Zarzuelo, A. Effects of dietary fiber on inflammatory bowel disease. Mol. Nutr. Food Res. 2005, 49, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Raninen, K.; Lappi, J.; Mykkanen, H.; Poutanen, K. Dietary fiber type reflects physiological functionality: Comparison of grain fiber, inulin, and polydextrose. Nutr. Rev. 2011, 69, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Lazarova, D.L.; Bordonaro, M. Mechanisms linking dietary fiber, gut microbiota and colon cancer prevention. World J. Gastrointest. Oncol. 2014, 6, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.S.; Rhodes, J. Maintenance of remission in ulcerative colitis with sulphasalazine or a high-fibre diet: A clinical trial. Br. Med. J. 1978, 1, 1524–1525. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Nyman, M.; Pousette, A.; Granno, C.; Svensson, H. Increasing fecal butyrate in ulcerative colitis patients by diet: Controlled pilot study. Inflamm. Bowel. Dis. 2003, 9, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Suchecka, D.; Harasym, J.P.; Wilczak, J.; Gajewska, M.; Oczkowski, M.; Gudej, S.; Błaszczyk, K.; Kamola, D.; Filip, R.; Gromadzka-Ostrowska, J. Antioxidative and anti-inflammatory effects of high β-glucan concentration purified aqueous extract from oat in experimental model of LPS-induced chronic enteritis. J. Func. Foods 2015, 14, 244–254. [Google Scholar] [CrossRef]
- Liu, B.; Lin, Q.; Yang, T.; Zeng, L.; Shi, L.; Chen, Y.; Luo, F. Oat β-glucan ameliorates dextran sulfate sodium (DSS)-induced ulcerative colitis in mice. Food Funct. 2015, 6, 3454–3463. [Google Scholar] [CrossRef] [PubMed]
- Thies, F.; Masson, L.F.; Boffetta, P.; Kris-Etherton, P. Oats and bowel disease: A systematic literature review. Br. J. Nutr. 2014, 112, S31–S43. [Google Scholar] [CrossRef] [PubMed]
- Forland, D.T.; Saetre, L.; Lyberg, T.; Lygren, I.; Hetland, G. Effect of an extract based on the medicinal mushroom Agaricus blazei Murill on expression of cytokines and calprotectin in patients with ulcerative colitis and Crohn’s disease. Scand. J. Immunol. 2011, 73, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Mojgan, N.; Reynolds, P.D.; Baumgartner, A.; Jerwoodc, D.; Andersona, D. Chaga mushroom extract inhibits oxidative DNA damage in lymphocytes of patients with inflammatory bowel disease. BioFactors 2007, 31, 191–200. [Google Scholar]
- Therkelsen, S.P.; Hetland, G.; Lyberg, T.; Lygren, I.; Johnson, E. Cytokine Levels After Consumption of a Medicinal Agaricus blazei Murill-Based Mushroom Extract, AndoSan, in Patients with Crohn’s Disease and Ulcerative Colitis in a Randomized Single-Blinded Placebo-Controlled Study. Scand. J. Immunol. 2016, 84, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Lin, Q.; Yang, T.; Nie, Y.; Li, X.; Liu, B.; Shen, J.; Liang, Y.; Tang, Y.; Luo, F. Oral administration of Lentinus edodes β-glucan ameliorates DSS-induced ulcerative colitis in mice via MAPK-Elk-1 and MAPK-PPARgamma pathways. Food Funct. 2016, 7, 4614–4627. [Google Scholar] [CrossRef] [PubMed]
- Lavi, I.; Levinson, D.; Peri, I.; Nimri, L.; Hadar, Y.; Schwartz, B. Orally administered glucan from the edible mushroom Pleurotus pulmonarius reduce acute inflammation in dextran sulfate sodium-induced experimental colitis. Br. J. Nutr. 2010, 103, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Takashi, O.; Rie, K.; Yuji, N.; Itsuko, F.; Takashi, H.; Kazuki, K.; Masashi, M.; Hitoshi, A. Lentinan fromshiitake mushroom (Lentinus edodes) suppresses expression of cytochrome P450 1A subfamily in the mouse liver. BioFactors 2004, 21, 407–409. [Google Scholar]
- Xie, J.T.; Wang, C.Z.; Wicks, S.; Yin, J.J.; Kong, J.; Li, J.; Li, Y.C.; Yuan, C.S. Ganoderma lucidum extract inhibits proliferation of SW 480 human colorectal cancer cells. Exp. Oncol. 2006, 28, 25–29. [Google Scholar] [PubMed]
- Lavi, I.; Friesem, D.; Geresh, S.; Hadar, Y.; Schwartz, B. An aqueous polysaccharide extract from the edible mushroom Pleurotus ostreatus induces anti-proliferative and pro-apoptotic effects on HT-29 colon cancer cells. Cancer Lett. 2006, 244, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Kannaiyan, R.; Sethi, G. Targeting cell signaling and apoptotic pathways by dietary agents: Role in the prevention and treatment of cancer. Nutr. Cancer 2011, 63, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Zhang, Z.; Lei, Z.; Yang, Y.; Sugiura, N. Comparative study of antioxidant activity and antiproliferative effect of hot water and ethanol extracts from the mushroom Inonotus obliquus. J. Biosci. Bioeng. 2009, 107, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.T.; O’shea, C.J.; Collins, C.B.; O’Doherty, J.V.; Sweeney, T. Effects of dietary supplementation with Laminaria hyperborea, Laminaria digitata, and Saccharomyces cerevisiae on the IL-17 pathway in the porcine colon. J. Anim. Sci. 2012, 30, 263–265. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, C.J.; O’Doherty, J.V.; Callanan, J.J.; Doyle, D.; Thornton, K.; Sweeney, T. The effect of algal polysaccharides laminarin and fucoidan on colonic pathology, cytokine gene expression and Enterobacteriaceae in a dextran sodium sulfate-challenged porcine model. J. Nutr. Sci. 2016, 5, e15. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Park, M.; Ji, K.Y.; Lee, H.Y.; Jang, J.H.; Yoon, I.J.; Oh, S.S.; Kim, S.M.; Jeong, Y.H.; Yun, C.H.; et al. Bacterial β-(1,3)-glucan prevents DSS-induced IBD by restoring the reduced population of regulatory T cells. Immunobiology 2014, 219, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Zhao, K.; Huang, Q.; Xu, C.; Shang, P. Isolation, structure and bioactivities of the polysaccharides from Angelica sinensis (Oliv.) Diels: A review. Carbohyd. Polym. 2012, 89, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Rychlik, A.; Nieradka, R.; Kander, M.; Nowicki, M.; Wdowiak, M.; Kolodziejska-Sawerska, A. The effectiveness of natural and synthetic immunomodulators in the treatment of inflammatory bowel disease in dogs. Acta. Vet. Hung. 2013, 61, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.-S.; Kim, S.-K. Fucoidans as a natural bioactive ingredient for functional foods. J. Func. Foods 2013, 5, 16–27. [Google Scholar] [CrossRef]
- Lean, Q.Y.; Eri, R.D.; Fitton, J.H.; Patel, R.P.; Gueven, N. Fucoidan Extracts Ameliorate Acute Colitis. PLoS ONE 2015, 10, e0128453. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Chang, U.J.; Lee, J.S. Inhibitory effects of Fucoidan in 3T3-L1 adipocyte differentiation. Mar. Biotechnol. 2009, 11, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-J.; Lee, B.-Y. Fucoidan from the sporophyll of Undaria pinnatifida suppresses adipocyte differentiation by inhibition of inflammation-related cytokines in 3T3-L1 cells. Nutr. Res. 2012, 32, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, F.; Sanchez-Lombrana, J.L.; Navarro, E.; Hinojosa, J.; Gassull, M.A. Randomized clinical trial of Plantago ovata seeds (dietary fiber) as compared with mesalamine in maintaining remission in ulcerative colitis. Am. J. Gastroenterol. 1999, 94, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Rodrı́guez-Cabezas, M.E.; Gálvez, J.; Camuesco, D.; Lorente, M.D.; Concha, A.; Martinez-Augustin, O.; Redondo, L.; Zarzuelo, A. Intestinal anti-inflammatory activity of dietary fiber (Plantago ovata seeds) in HLA-B27 transgenic rats. Clin. Nutr. 2003, 22, 463–471. [Google Scholar] [CrossRef]
- Rodriguez-Cabezas, M.E.; Galvez, J.; Lorente, M.D.; Concha, A.; Camuesco, D.; Azzouz, S.; Osuna, A.; Redondo, L.; Zarzuelo, A. Dietary fiber down-regulates colonic tumor necrosis factor α and nitric oxide production in trinitrobenzenesulfonic acid-induced colitic rats. J. Nutr. 2002, 132, 3263–3271. [Google Scholar] [PubMed]
- Kanauchi, O.; Mitsuyama, K.; Homma, T.; Takahama, K.; Fujiyama, Y.; Tomiyasu, N.; Toyonaga, A.; Fukuda, M.; Kojima, A.; Bamba, T. Treatment of ulcerative colitis patients by long-term administration of germinated barley foodstuff: Multi-center open trial. Int. J. Mol. Med. 2003, 12, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; Possemiers, S.; Druart, C.; van de Wiele, T.; de Backer, F.; Cani, P.D.; Larondelle, Y.; Delzenne, N.M. Prebiotic effects of wheat arabinoxylan related to the increase in bifidobacteria, Roseburia and Bacteroides/Prevotella in diet-induced obese mice. PLoS ONE 2011, 6, e20944. [Google Scholar] [CrossRef] [PubMed]
- James, S.L.; Christophersen, C.T.; Bird, A.R.; Conlon, M.A.; Rosella, O.; Gibson, P.R.; Muir, J.G. Abnormal fibre usage in UC in remission. Gut 2015, 64, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Swanson, K.S.; Fahey, G.C., Jr.; Dien, B.S.; Beloshapka, A.N.; Bauer, L.L.; Rausch, K.D.; Tumbleson, M.E.; Singh, V. In vitro Fermentation of Xylooligosaccharides Produced from Miscanthus x giganteus by Human Fecal Microbiota. J. Agric. Food Chem. 2016, 64, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Plamondon, S.; Kamm, M.A.; Hart, A.L.; Al-Hassi, H.O.; Guenther, T.; Stagg, A.J.; Knight, S.C. Immunosuppressive effects via human intestinal dendritic cells of probiotic bacteria and steroids in the treatment of acute ulcerative colitis. Inflamm. Bowel Dis. 2010, 16, 1286–1298. [Google Scholar] [CrossRef] [PubMed]
- Segarra, S.; Martinez-Subiela, S.; Cerda-Cuellar, M.; Martinez-Puig, D.; Munoz-Prieto, A.; Rodriguez-Franco, F.; Rodriguez-Bertos, A.; Allenspach, K.; Velasco, A.; Ceron, J. Oral chondroitin sulfate and prebiotics for the treatment of canine Inflammatory Bowel Disease: A randomized, controlled clinical trial. BMC Vet. Res. 2016, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Whorwell, P.J.; Altringer, L.; Morel, J.; Bond, Y.; Charbonneau, D.; O’Mahony, L.; Kiely, B.; Shanahan, F.; Quigley, E.M. Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. Am. J. Gastroenterol. 2006, 101, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, L.; McCarthy, J.; Kelly, P.; Hurley, G.; Luo, F.; Chen, K.; O’Sullivan, G.C.; Kiely, B.; Collins, J.K.; Shanahan, F.; et al. Lactobacillus and bifidobacterium in irritable bowel syndrome: Symptom responses and relationship to cytokine profiles. Gastroenterology 2005, 128, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Silk, D.B.; Davis, A.; Vulevic, J.; Tzortzis, G.; Gibson, G.R. Clinical trial: The effects of a trans-galactooligosaccharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2009, 29, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Takagi, T.; Naito, Y.; Higashimura, Y.; Ushiroda, C.; Mizushima, K.; Ohashi, Y.; Yasukawa, Z.; Ozeki, M.; Tokunaga, M.; Okubo, T.; et al. Partially hydrolysed guar gum ameliorates murine intestinal inflammation in association with modulating luminal microbiota and SCFA. Br. J. Nutr. 2016, 116, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.V.; Suzuki, T. Dietary fermentable fiber reduces intestinal barrier defects and inflammation in colitic mice. J. Nutr. 2016, 146, 1970–1979. [Google Scholar] [CrossRef] [PubMed]
- Alam, N.H.; Meier, R.; Sarker, S.A.; Bardhan, P.K.; Schneider, H.; Gyr, N. Partially hydrolysed guar gum supplemented comminuted chicken diet in persistent diarrhea: A randomised controlled trial. Arch. Dis. Child 2005, 90, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Kolida, S.; Lustenberger, A.M.; Gibson, G.R. The prebiotic effects of biscuits containing partially hydrolysed guar gum and fructo-oligosaccharides—A human volunteer study. Br. J. Nutr. 2007, 86, 341. [Google Scholar] [CrossRef]
- Paul, S.P.; Barnard, P.; Edate, S.; Candy, D.C. Stool consistency and abdominal pain in irritable bowel syndrome may be improved by partially hydrolysed guar gum. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 582–583. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Wu, S.; Li, Z.; Li, J.; Li, X.; Xiang, J.; Ding, H. Wild jujube polysaccharides protect against experimental inflammatory bowel disease by enabling enhanced intestinal barrier function. Food Funct. 2015, 6, 2568–2577. [Google Scholar] [CrossRef] [PubMed]
- Krylova, S.G.; Efimova, L.A.; Zueva, E.P.; Khotimchenko, M.U.; Amosova, E.N.; Razina, T.G.; Lopatina, K.A.; Khotimchenko, U.S. Gastroprotective effect of nonstarch polysaccharide calcium pectate under experimental conditions. Bull. Exp. Biol. Med. 2008, 145, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.M.; Nie, S.P.; Zhou, H.L.; Huang, D.F.; Xie, M.Y. Carboxymethylation enhances the maturation-inducing activity in dendritic cells of polysaccharide from the seeds of Plantago asiatica L. Int. Immunopharmacol. 2014, 22, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H. Marine non-glycosaminoglycan sulfated glycans as potential pharmaceuticals. Pharmaceuticals 2015, 8, 848–864. [Google Scholar] [CrossRef] [PubMed]
- Hedin, C.R.; McCarthy, N.E.; Louis, P.; Farquharson, F.M.; McCartney, S.; Taylor, K.; Prescott, N.J.; Murrells, T.; Stagg, A.J.; Whelan, K.; et al. Altered intestinal microbiota and blood T cell phenotype are shared by patients with Crohn’s disease and their unaffected siblings. Gut 2014, 63, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Fiocchi, C. Inflammatory bowel disease: Etiology and pathogenesis. Gastroenterology 1998, 115, 182–205. [Google Scholar] [CrossRef]
- Xu, X.; Yan, H.; Zhang, X. Structure and immuno-stimulating activities of a new heteropolysaccharide from Lentinula edodes. J. Agric. Food Chem. 2012, 60, 11560–11566. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Jia, M.; Meng, J.; Wu, H.; Mei, Q. Immunomodulatory activity of polysaccharide isolated from Angelica sinensis. Int. J. Biol. Macromol. 2006, 39, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D.; Gordon, S. Immune recognition of fungal β-glucan. Cell Microbiol. 2005, 7, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Zekovic, D.B.; Kwiatkowski, S.; Vrvic, M.M.; Jakovljevic, D.; Moran, C.A. Natural and modified (1→3)-β-d-glucan in health promotion and disease alleviation. Crit. Rev. Biotechnol. 2005, 25, 205–230. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.H.; Yang, W.T.; Huang, K.Y.; Jiang, Y.L.; Yang, G.L.; Wang, C.F.; Li, Y. β-glucan from Coriolus versicolor protect mice against S. typhimurium challenge by activation of macrophages. Int. J. Biol. Macromol. 2016, 86, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Berner, V.K.; duPre, S.A.; Redelman, D.; Hunter, K.W. Microparticulate β-glucan vaccine conjugates phagocytized by dendritic cells activate both naive CD4 and CD8 T cells in vitro. Cell. Immunol. 2015, 298, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.D.; Lim, S.; Kim, D.H. Oleanolic acid ameliorates dextran sodium sulfate-induced colitis in mice by restoring the balance of Th17/Treg cells and inhibiting NF-κB signaling pathway. Int. Immunopharmacol. 2015, 29, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Hartog, A.; Belle, F.N.; Bastiaans, J.; de Graaff, P.; Garssen, J.; Harthoorn, L.F.; Vos, A.P. A potential role for regulatory T-cells in the amelioration of DSS induced colitis by dietary non-digestible polysaccharides. J. Nutr. Biochem. 2015, 26, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.H.; Yang, H.P.; Chun, H.S. Chitooligosaccharide ameliorates diet-induced obesity in mice and affects adipose gene expression involved in adipogenesis and inflammation. Nutr. Res. 2012, 32, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Pavuluri, S.; Bruggeman, K.; Long, B.M.; Parnell, A.J.; Martel, A.; Parnell, S.R.; Pfeffer, F.M.; Dennison, A.J.; Nicholas, K.R.; et al. Coassembled nanostructured bioscaffold reduces the expression of proinflammatory cytokines to induce apoptosis in epithelial cancer cells. Nanomedicine 2016, 12, 1397–1407. [Google Scholar] [CrossRef] [PubMed]
- Nagao-Kitamoto, H.; Shreiner, A.B.; Gillilland, M.G., 3rd; Kitamoto, S.; Ishii, C.; Hirayama, A.; Kuffa, P.; El-Zaatari, M.; Grasberger, H.; Seekatz, A.M.; et al. Functional characterization of inflammatory bowel disease-associated gut dysbiosis in gnotobiotic mice. Cell Mol. Gastroenterol. Hepatol. 2016, 2, 468–481. [Google Scholar] [CrossRef] [PubMed]
- Knights, D.; Lassen, K.G.; Xavier, R.J. Advances in inflammatory bowel disease pathogenesis: Linking host genetics and the microbiome. Gut 2013, 62, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Sghir, A.; Gramet, G.; Suau, A.; Rochet, V.; Pochart, P.; Dore, J. Quantification of bacterial groups within human fecal flora by oligonucleotide probe hybridization. Appl. Environ. Microbiol. 2000, 66, 2263–2266. [Google Scholar] [CrossRef] [PubMed]
- Stagg, A.J.; Hart, A.L.; Knight, S.C.; Kamm, M.A. The dendritic cell: Its role in intestinal inflammation and relationship with gut bacteria. Gut 2003, 51, 1522–1529. [Google Scholar] [CrossRef]
- Martínez-Abad, B.; Garrote, J.A.; Bernardo, D.; Montalvillo, E.; Escudero-Hernández, C.; Vázquez, E.; Rueda, R.; Arranz, E. Differential immunomodulatory effects of Lactobacillus rhamnosus DR20, Lactobacillus fermentum CECT 5716 and Bifidobacterium animalis subsp. lactis on monocyte-derived dendritic cells. J. Funct. Foods 2016, 22, 300–312. [Google Scholar] [CrossRef]
- Hart, A.L.; Lammers, K.; Brigidi, P.; Vitali, B.; Rizzello, F.; Gionchetti, P.; Campieri, M.; Kamm, M.A.; Knight, S.C.; Stagg, A.J. Modulation of human dendritic cell phenotype and function by probiotic bacteria. Gut 2004, 53, 1602–1609. [Google Scholar] [CrossRef] [PubMed]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humaran, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.; Harris, P.J.; Ferguson, L.R. Potential benefits of dietary fibre intervention in inflammatory bowel disease. Int. J. Mol. Sci. 2016, 17, 919. [Google Scholar] [CrossRef] [PubMed]
- De Vrese, M.; Marteau, P.R. Probiotics and prebiotics: Effects on diarrhea. J. Nutr. 2007, 137, S803–S811. [Google Scholar]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides: Definition, specificity, function, and application in gastrointestinal disorders. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 64–68. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.; Neyrinck, A.M.; Bindels, L.B.; de Vos, W.M.; Gibson, G.R.; Thissen, J.P.; et al. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 2013, 62, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.J.; Macfarlane, G.T. Nondigestible Oligosaccharides Enhance Bacterial Colonization Resistance against Clostridium difficile in vitro. App. Environ. Microb. 2003, 69, 1920–1927. [Google Scholar] [CrossRef]
- Hookman, P. Clostridium difficile associated infection, diarrhea and colitis. World J. Gastroenterol. 2009, 15, 1554. [Google Scholar] [CrossRef] [PubMed]
- Barbut, F.; Jones, G.; Eckert, C. Epidemiology and control of Clostridium difficile infections in healthcare settings: An update. Curr. Opin. Infect. Dis. 2011, 24, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Perrin, S.; Warchol, M.; Grill, J.P.; Schneider, F. Fermentations of fructo-oligosaccharides and their components by Bifidobacterium infantis ATCC 15697 on batch culture in semi-synthetic medium. J. Appl. Microbiol. 2001, 90, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Falony, G.; Vlachou, A.; Verbrugghe, K.; de Vuyst, L. Cross-Feeding between Bifidobacterium longum BB536 and acetate-converting, butyrate-producing colon bacteria during growth on oligofructose. Appl. Environ. Microbiol. 2006, 72, 7835–7841. [Google Scholar] [CrossRef] [PubMed]
- Viladomiu, M.; Hontecillas, R.; Yuan, L.; Lu, P.; Bassaganya-Riera, J. Nutritional protective mechanisms against gut inflammation. J. Nutr. Biochem. 2013, 24, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, M.; Smeekens, S.P.; Vlamakis, H.; Jaeger, M.; Oosting, M.; Franzosa, E.A.; Jansen, T.; Jacobs, L.; Bonder, M.J.; Kurilshikov, A.; et al. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell 2016, 167, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Oosting, M.; Smeekens, S.P.; Jaeger, M.; Aguirre-Gamboa, R.; Le, K.T.; Deelen, P.; Ricano-Ponce, I.; Schoffelen, T.; Jansen, A.F.; et al. A functional genomics approach to understand variation in cytokine production in humans. Cell 2016, 167, 1099–1110. [Google Scholar] [CrossRef] [PubMed]
- Breuer, R.I.; Buto, S.K.; Christ, M.L.; Bean, J.; Vernia, P.; Paoluzi, P.; di Paolo, M.C.; Caprilli, R. Rectal irrigation with short-chain fatty acids for distal ulcerative colitis. Preliminary report. Dig. Dis. Sci. 1991, 36, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Luhrs, H.; Gerke, T.; Muller, J.G.; Melcher, R.; Schauber, J.; Boxberge, F.; Scheppach, W.; Menzel, T. Butyrate inhibits NF-κB activation in lamina propria macrophages of patients with ulcerative colitis. Scand. J. Gastroenterol. 2002, 37, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, R.G.; Esposito, C.C.; Muller, L.C.; Castelo-Branco, M.T.; Quintella, L.P.; Chagas, V.L.; de Souza, H.S.; Schanaider, A. Use of butyrate or glutamine in enema solution reduces inflammation and fibrosis in experimental diversion colitis. World J. Gastroenterol. 2012, 18, 4278–4287. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, J.R.; Sartor, R.B. The role of diet on intestinal microbiota metabolism: Downstream impacts on host immune function and health, and therapeutic implications. J. Gastroenterol. 2014, 49, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, M.A.; Singh, N.; Martin, P.M.; Thangaraju, M.; Ganapathy, V.; Waller, J.L.; Shi, H.; Robertson, K.D.; Munn, D.H.; Liu, K. Butyrate suppresses colonic inflammation through HDAC1-dependent Fas upregulation and Fas-mediated apoptosis of T cells. Am. J. Phys. Gastrointest. Liver Phys. 2012, 302, G1405–G1415. [Google Scholar] [CrossRef] [PubMed]
- Berni Canani, R.; Di Costanzo, M.; Leone, L. The epigenetic effects of butyrate: Potential therapeutic implications for clinical practice. Clin. Epigenetics 2012, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.Y.; Lannoy, V.; Decobecq, M.E.; Brezillon, S.; Dupriez, V.; Vassart, G.; van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J. Biol. Chem. 2003, 278, 25481–25489. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Gui, H.; Yao, L.; Yan, L.; Martens, H.; Aschenbach, J.R.; Shen, Z. Short-Chain fatty acids and acidic pH upregulate UT-B, GPR41, and GPR4 in rumen epithelial cells of goats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R283–R293. [Google Scholar] [CrossRef] [PubMed]
- Kamp, M.E.; Shim, R.; Nicholls, A.J.; Oliveira, A.C.; Mason, L.J.; Binge, L.; Mackay, C.R.; Wong, C.H.G. Protein-Coupled receptor 43 modulates neutrophil recruitment during acute inflammation. PLoS ONE 2016, 11, e0163750. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Zheng, L.; Campbell, E.L.; Saeedi, B.; Scholz, C.C.; Bayless, A.J.; Wilson, K.E.; Glover, L.E.; Kominsky, D.J.; Magnuson, A.; et al. Crosstalk between Microbiota-Derived Short-Chain Fatty Acids and Intestinal Epithelial HIF Augments Tissue Barrier Function. Cell Host Microbe 2015, 17, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Gui, H.; Shen, Z. Concentrate diet modulation of ruminal genes involved in cell proliferation and apoptosis is related to combined effects of short-chain fatty acid and pH in rumen of goats. J. Dairy Sci. 2016, 99, 6627–6638. [Google Scholar] [CrossRef] [PubMed]
- Luceri, C.; Femia, A.P.; Fazi, M.; Di Martino, C.; Zolfanelli, F.; Dolara, P.; Tonelli, F. Effect of butyrate enemas on gene expression profiles and endoscopic/histopathological scores of diverted colorectal mucosa: A randomized trial. Dig. Liver Dis. 2016, 48, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.N.; Koo, M.W.; Li, Y.; Matsui, H.; Cho, C.H. Angelica sinensis modulates migration and proliferation of gastric epithelial cells. Life Sci. 2001, 68, 961–968. [Google Scholar] [CrossRef]
- Ye, Y.N.; So, H.L.; Liu, E.S.; Shina, V.Y.; Cho, C.H. Effect of polysaccharides from Angelica sinensis on gastric ulcer healing. Life Sci. 2003, 72, 925–932. [Google Scholar] [CrossRef]
- Zhao, H.; Luo, Y.; Lu, C.; Lin, N.; Xiao, C.; Guan, S.; Guo, D.A.; Liu, Z.; Ju, D.; He, X.; Lu, A. Enteric mucosal immune response might trigger the immunomodulation activity of Ganoderma lucidum polysaccharide in mice. Planta Med. 2010, 76, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Deters, A.M.; Schröder, K.R.; Smiatek, T.; Hensel, A. Ispaghula (Plantago ovata) seed husk polysaccharides promote proliferation of human epithelial cells (skin keratinocytes and fibroblasts) via enhanced growth factor receptors and energy production. Planta Med. 2005, 71, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.L.; Martini, M.C.; Jacobs, D.R.J.; Marquart, L. Plausible mechanisms for the protectiveness of whole grains. Am. J. Clin. Nutr. 1999, 70, S459–S463. [Google Scholar]
- Ferguson, L.R.; Zhu, S.; Kestell, P. Contrasting effects of non-starch polysaccharide and resistant starch-based diets on the disposition and excretion of the food carcinogen, 2-amino-3-methylimidazo [4,5-f]quinoline (IQ), in a rat model. Food Chem. Toxicol. 2003, 41, 785–792. [Google Scholar] [CrossRef]
- Hu, Y.; Martin, J.; Le Leu, R.; Young, G.P. The colonic response to genotoxic carcinogens in the rat: Regulation by dietary fibre. Carcinogenesis 2002, 23, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- James, J.F.; Bruce, E.S. Clinical gastroenterology. In Inflammatory Bowel Disease Diagnosis and Therapeutics; Russell, D.C., Ed.; Humana Press: Totowa, NJ, USA, 2003; pp. 33–63. [Google Scholar]
- Aldini, R.; Micucci, M.; Cevenini, M.; Fato, R.; Bergamini, C.; Nanni, C.; Cont, M.; Camborata, C.; Spinozzi, S.; Montagnani, M.; et al. Antiinflammatory effect of phytosterols in experimental murine colitis model: Prevention, induction, remission study. PLoS ONE 2014, 9, e108112. [Google Scholar] [CrossRef] [PubMed]
- Suchecka, D.; Harasym, J.; Wilczak, J.; Gromadzka-Ostrowska, J. Hepato- and gastro- protective activity of purified oat 1–3, 1–4-β-d-glucan of different molecular weight. Int. J. Biol. Macromol. 2016, 91, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Błaszczyk, K.; Wilczak, J.; Harasym, J.; Gudej, S.; Suchecka, D.; Królikowski, T.; Lange, E.; Gromadzka-Ostrowska, J. Impact of low and high molecular weight oat β-glucan on oxidative stress and antioxidant defense in spleen of rats with LPS induced enteritis. Food Hydrocoll. 2015, 51, 272–280. [Google Scholar] [CrossRef]
- Plantago Ovata (Psyllium). Available online: http://www.altmedrev.com/publications/7/2/155.pdf (accessed on 5 May 2017).
- Wang, R.; Sun, P.; Zhou, Y.; Zhao, X. Preventive effect of Dendrobium candidum Wall. ex Lindl. on activated carbon-induced constipation in mice. Exp. Ther. Med. 2015, 9, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Varshosaz, J.; Jaffarian Dehkordi, A.; Golafshan, S. Colon-Specific delivery of mesalazine chitosan microspheres. J. Microencapsul. 2008, 23, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Lei, J.; Cui, H.; Lu, N.; Sun, Y.; Zhang, X.; Gao, C.; Zheng, H.; Yin, Y. Disulfide cross-linked nanospheres from sodium alginate derivative for inflammatory bowel disease: Preparation, characterization, and in vitro drug release behavior. Carbohydr. Polym. 2012, 88, 663–669. [Google Scholar] [CrossRef]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 2016, 167, 1339–1353. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, A.; Rosenfeld, G.; Bressler, B. The impact of dietary interventions on the symptoms of inflammatory bowel disease: A systematic review. Crit. Rev. Food Sci. Nutr. 2015, 56, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
| Types of NSPs | Sources | Structure | Ref. | Experimental Method | Main Results |
|---|---|---|---|---|---|
| glucan | oat | β-1,3-and 1,4-glucan | [21] | 25 g/day bran, 39 ulcerative colitis (UC) patients | high bran intake is of less value in maintaining clinical remission |
| [22] | 60 g/day, quiescent UC, 3 months | no patient showed signs of colitis relapse | |||
| [23] | 1% of G1 or G2 β-glucan (βG), chronic (lipopolysacchride) LPS -induced enteritis rats | different type blood leucocyte ↓ | |||
| [24] | 500/1000 mg per kg β-glucan (βG), dextran sulfate (DSS)-induced colitis rats | clinical symptoms ↓; pro-inflammatory factor ↓ | |||
| mushroom | α- and β-d-glucan | [26] | 60 mL/day of AndoSan, 11 Crohn’s disease (CD) and 10 UC, 12 days | inflammatory cytokine ↓; calprotectin of UC ↓ | |
| [27] | DNA damage of lymphocytes from inflammatory bowel disease (IBD) patients under H2O2 in vitro were measured | oxidative stress in lymphocytes ↓ | |||
| [28] | oral daily intake of AndoSan, 50 patients with symptomatic UC and Crohn’s disease (CD) | marginally anti-inflammatory effects | |||
| [29] | 500/1000 mg per kg βG, DSS-induced colitis rats, LPS-stimulated RAW264.7 cell | inflammatory cytokine ↓; phosphorylation of Elk-1 at Ser84, phosphorylation of PPARγ at Ser112 ↓ | |||
| [30] | 2 or 20 mg per mouse daily, DSS-induced colitis mouse | anti-inflammatory cytokines ↓; clinical symptoms ↓ | |||
| [31] | - | hepatic CYP1As expression ↓; NF-κB ↑ | |||
| [32] | SW 480 human colorectal cancer cell | proliferation ↓; diphenyl-picryl hydrazyl (DPPH ) radicals ↓ | |||
| [33] | HT-29 colon cancer cells | proliferation ↓; pro-apoptotic molecules Bax and cytosolic cytochrome-c ↑ | |||
| [35] | human colon cancer DLD-1 cells | proliferation↓, DPPH radicals ↓ | |||
| seaweed | β-1,3/1,6-glucan | [36] | Th17-major contributor to pathology of IBD measured in pig colon | expression of Th17-related cytokines (IL-17a, IL-17F, and IL-22), receptor IL23R, and IL-6 ↓ | |
| [37] | laminarin and fucoidan, DSS-challenged pigs | body weight ↑; IL-6 ↓; Enterobacteriaceae ↓ | |||
| bacterial glucan | β-(1,3)-glucan | [38] | 2.5 or 5 mg/kg for 2 weeks, DSS-induced IBD in mice | recruitment of macrophages ↓; expression of pro-inflammatory cytokines↓; recovery of Tregs ↑ | |
| root of Angelica sinensis | α-1,3-glucan, α-1,6-glucan and other linear α-glucan | [39] | 400 and 800 mg/kg, 2,4,6-trinitrobenzenesulfonic acid (TNBS) and ethanol induced colitis rat | migration of gastric epithelial cells ↑; epidermal growth factor (EGF) ↑; ulcer healing ↑; myeloperoxidase (MPO), malondialdehyde (MDA) and nitric oxide (NO) ↓; TNF-α, IL-10 & IL-2 ↓; TGF ↓; superoxide dismutase (SOD) activity ↓ | |
| fucoidan | brown algae | acidic and sulfated macromolecules (l-fucose mannose, galactose and xylose) | [42] | DSS-induced colitis mice | symptoms of colitis ↓; colon and spleen weight ↓; pathology in colon ↓; 15 pro-inflammatory cytokines ↓ |
| mucilage | Plantago ovata seeds | β-l,4-and β-l,3-1inked d-xylose as backbone and arabinose, rhamnose and galacturonic acid as side chains | [45] | 105 patients with UC in remission were randomized into groups to receive Plantago ovata seeds (10 g, twice injections per day), mesalamine (500 mg, thrice injections per day). and both at the same doses | Plantago ovata seeds might be as effective as mesalamine to maintain remission in UC |
| [46] | 5% Plantago ovata seeds, 13 weeks, HLA-B27 transgenic rats | colonic inflammation ↓; pro-inflammatory mediators ↓; short chain fatty acids (SCFAs) ↑ | |||
| [47] | 5% Plantago ovata seeds, TNBS model of rat colitis | intestinal cytoarchitecture ↑; TNF-α ↓; NO synthase activity ↓ | |||
| arabinoxylan arabinogalactan | grain cereal | xylans or galactans as backbone and arabinose or pentosans as side chains | [48] | 21 patients with mildly to moderately active UC, 20–30 g, 24 weeks plus regular drug | clinical activity index ↓; no side effects related to (germinated barley foodstuff) GBF were observed |
| [48] | high molecular weight arabinoxylans, high-fat diet mice, 4 weeks. | caecal bifidobacterial ↑; prebiotic properties | |||
| [50] | wheat bran and resistant starch, 8 weeks, UC in remission | better gut transit, Akkermansia muciniphila ↓; diversity within Clostridium cluster XIVa ↑ | |||
| prebiotics | — | oligosaccharides | [52] | xylooligosaccharides/pectin, in vitro fermentation, human fecal microbiota | SCFAs ↑; pH ↓; health-promoting bacteria ↑ |
| [53] | VSL#3, active UC | TLR-2 ↓; IL-10 ↑; IL-12 ↓; 10/14 patients showed a clinical response, similar to corticosteroids treatment | |||
| [54] | chondroitin sulfate and prebiotics (resistant starch, β-glucan and mannaoligosaccharides), canine IBD, 180 days | IBD activity index ↓; histologic score ↓; serum cholesterol and paraoxonase-1 ↑ | |||
| [55] | B. infantis 35624, dose of 1 × 106, 1 × 108, 1 × 1010 cfu/mL, 4 weeks, women with irritable bowel syndrome (IBS) | dose of 1 × 108 cfu was best to alleviate symptoms, no significant adverse events | |||
| [56] | 77 IBS patients, Lactobacillus salivarius UCC4331 or B. infantis 35624, dose of 1 × 1010, 8 weeks. | B. infantis 35624 showed a greater reduction in symptom scores and improved IL-10/IL-12 ratio | |||
| [57] | 44 Rome II positive IBS, 12 weeks, 3.5 g/day or 7 g/day prebiotic | fecal bifidobacterial ↑; 3.5 g⁄days significantly changed stool consistency and bloating, etc. | |||
| gum | guar (partially hydrolyzed) | mannose as backbone with random substitutions of galactose in a ratio of 1.6:1 | [58] | partially hydrolyzed guar gum (PHGG), murine TNBS-induced colitis model | colonic damage ↓; MPO and TNF-alpha protein↓; Clostridium cluster XIVa and IV and Bacteroides fragilis ↑ |
| [59] | guar gum and PHGG, murine model of DSS-induced colitis | clinical score ↓; occludin and claudin 3, 4, and 7↑; fecal SCFAs ↑ | |||
| [60] | PHGG+ comminuted chicken diet, persistent diarrhea in 116 children, 7 day | Diarrhea ↓ | |||
| [61] | PHGG and fructo-oligosaccharides (FOS), 21 days, human | Bifidobacteria ↑ | |||
| [62] | IBS | stool consistency ↑; abdominal pain ↓ | |||
| pectin | wild jujube | acidic heteropolysaccharides, about 40% of arabinose glucose, 20% arabinose,20% galacturonic acid and the rest galactose | [63] | wild jujube polysaccharides, TNBS-induced colitis rats | colitis severity ↓; mucosal damage ↓; inflammatory response ↓; AMP-activated protein kinase (AMPK) activity ↑ |
| citrus | calcium pectate: 67.3% anhydrogalacturonic acid and 38 mg/g calcium | [64] | 25 and 50 mg/kg calcium pectate, ethanol-induced ulcers, prednisolone induced injury and H. Shay ulceration in 114 mice, 7 days | mucosa damage ↓ | |
| NSP modifiers | Plantago asiatica | carboxymethylation of the seeds polysaccharides | [65] | dendritic cells in vitro | MHCII ↑; IL-12, CCR7 and CXCR4 ↑; endocytosis activities ↓; mixed lymphocyte reactions ↑ |
| marine plants | sulfated fucans and sulfated galactans | [66] | — | anti-inflammatory activity ↑; antimicrobial activity ↑ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, Y.; Lin, Q.; Luo, F. Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease. Int. J. Mol. Sci. 2017, 18, 1372. https://doi.org/10.3390/ijms18071372
Nie Y, Lin Q, Luo F. Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease. International Journal of Molecular Sciences. 2017; 18(7):1372. https://doi.org/10.3390/ijms18071372
Chicago/Turabian StyleNie, Ying, Qinlu Lin, and Feijun Luo. 2017. "Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease" International Journal of Molecular Sciences 18, no. 7: 1372. https://doi.org/10.3390/ijms18071372
APA StyleNie, Y., Lin, Q., & Luo, F. (2017). Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease. International Journal of Molecular Sciences, 18(7), 1372. https://doi.org/10.3390/ijms18071372