Effects of Pup Separation on Stress Response in Postpartum Female Rats
Abstract
:1. Introduction
2. Results
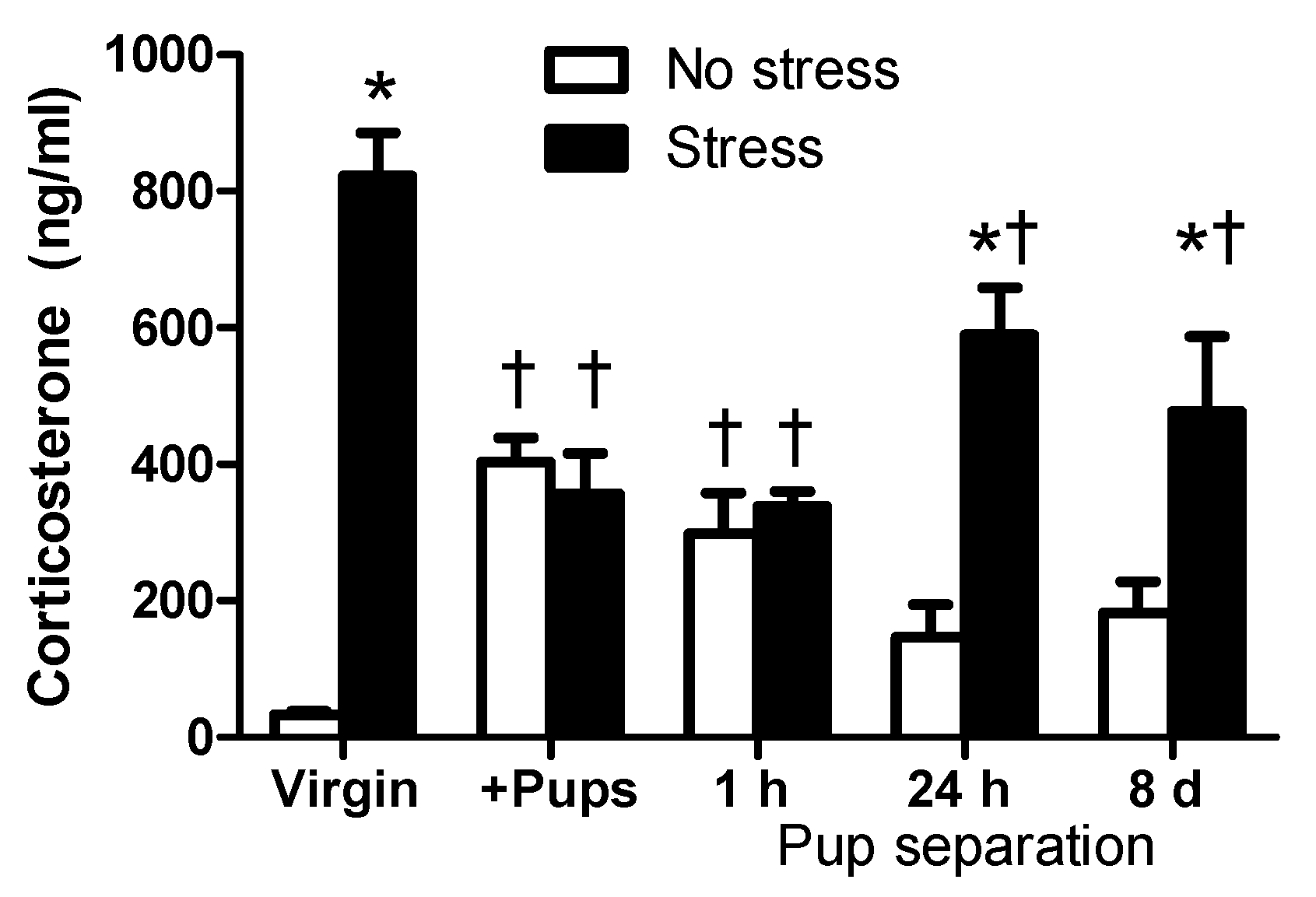
2.1. Circulating CORT Levels of Virgin and Lactating Rats with or without Pup Separation Following No-Stress or an Acute Stress
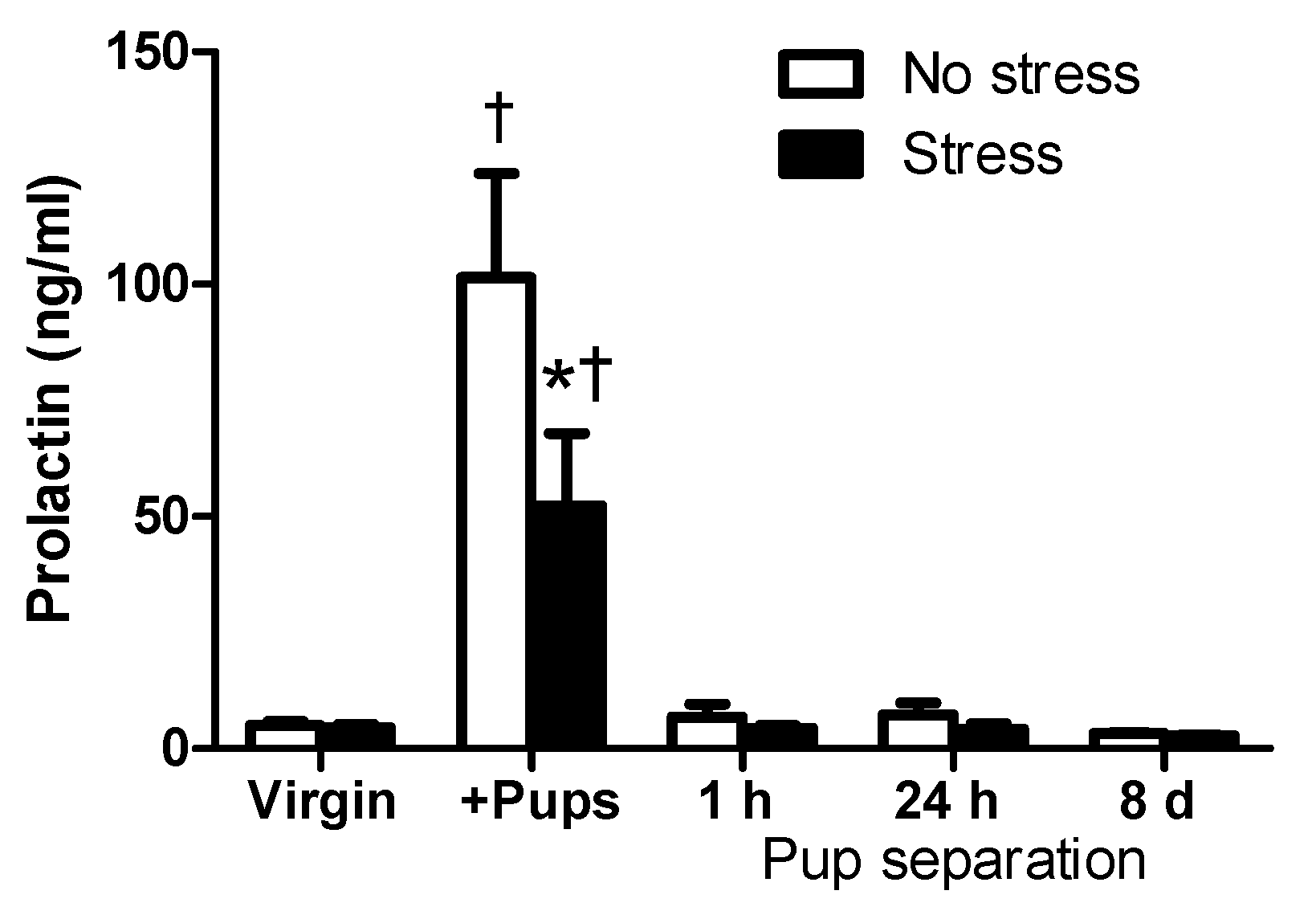
2.2. Circulating PRL Levels of Virgin and Lactating Rats with or without Pup Separation Following No-Stress or an Acute Stress
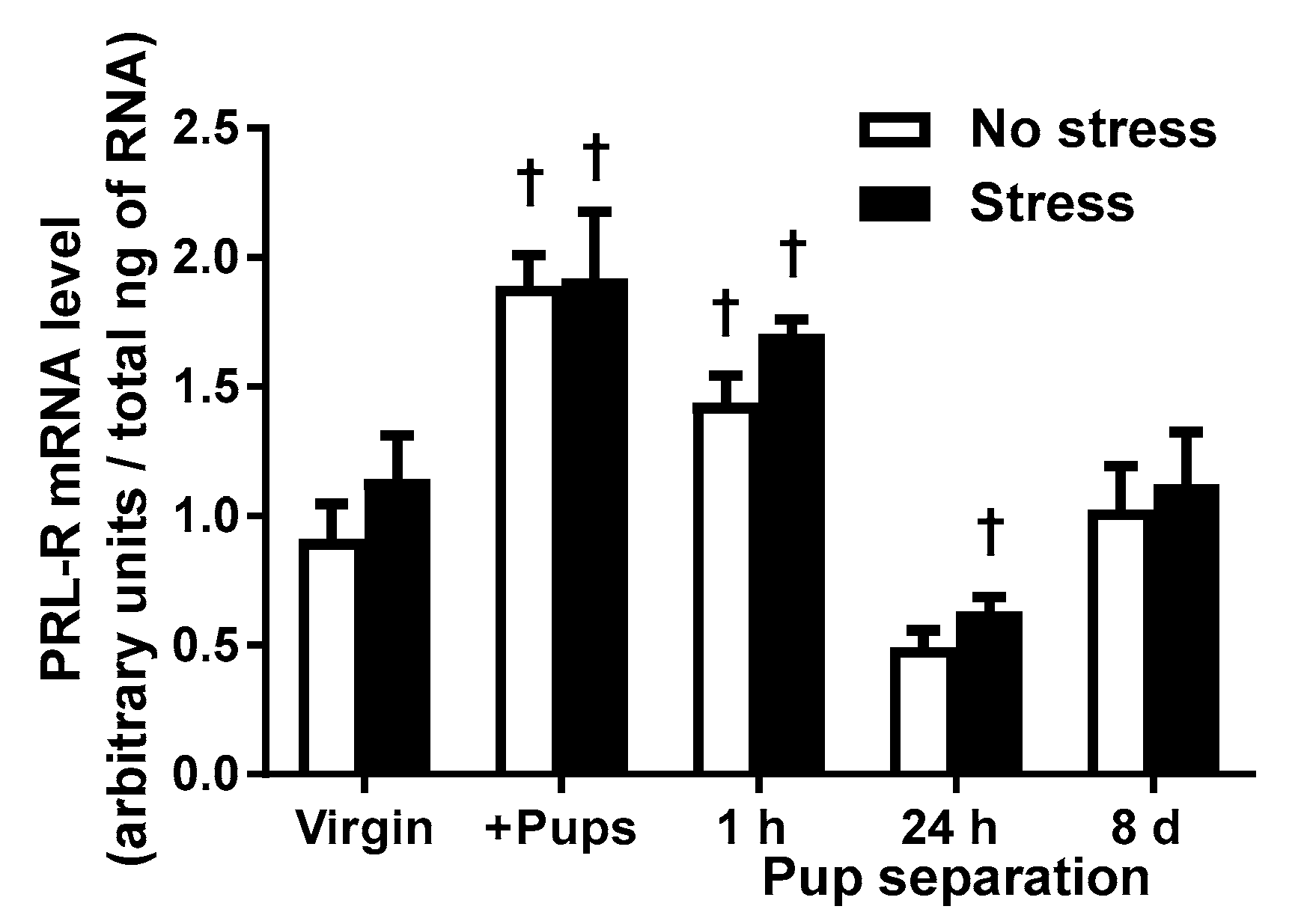
2.3. PRL-R mRNA Levels of Virgin and Lactating Rats with or without Pup Separation Following No-Stress or an Acute Stress
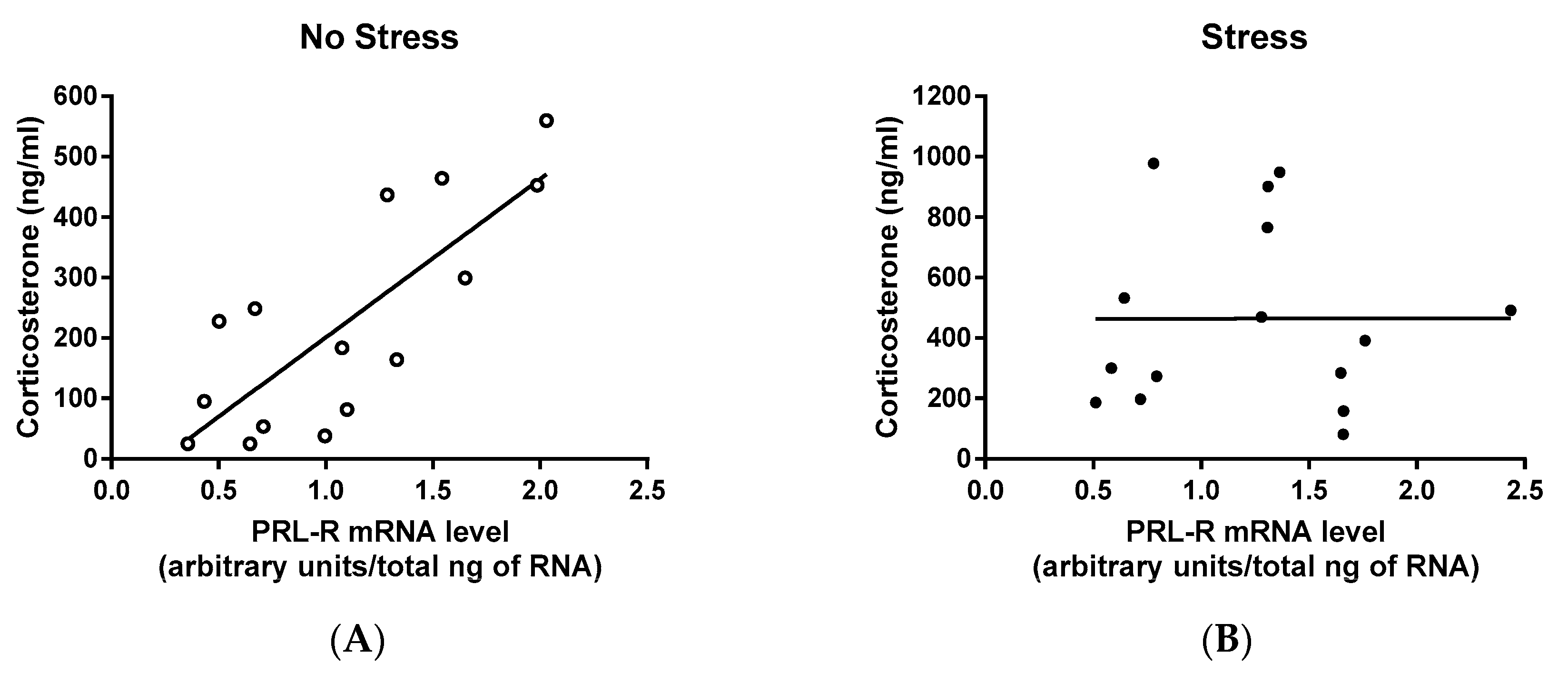
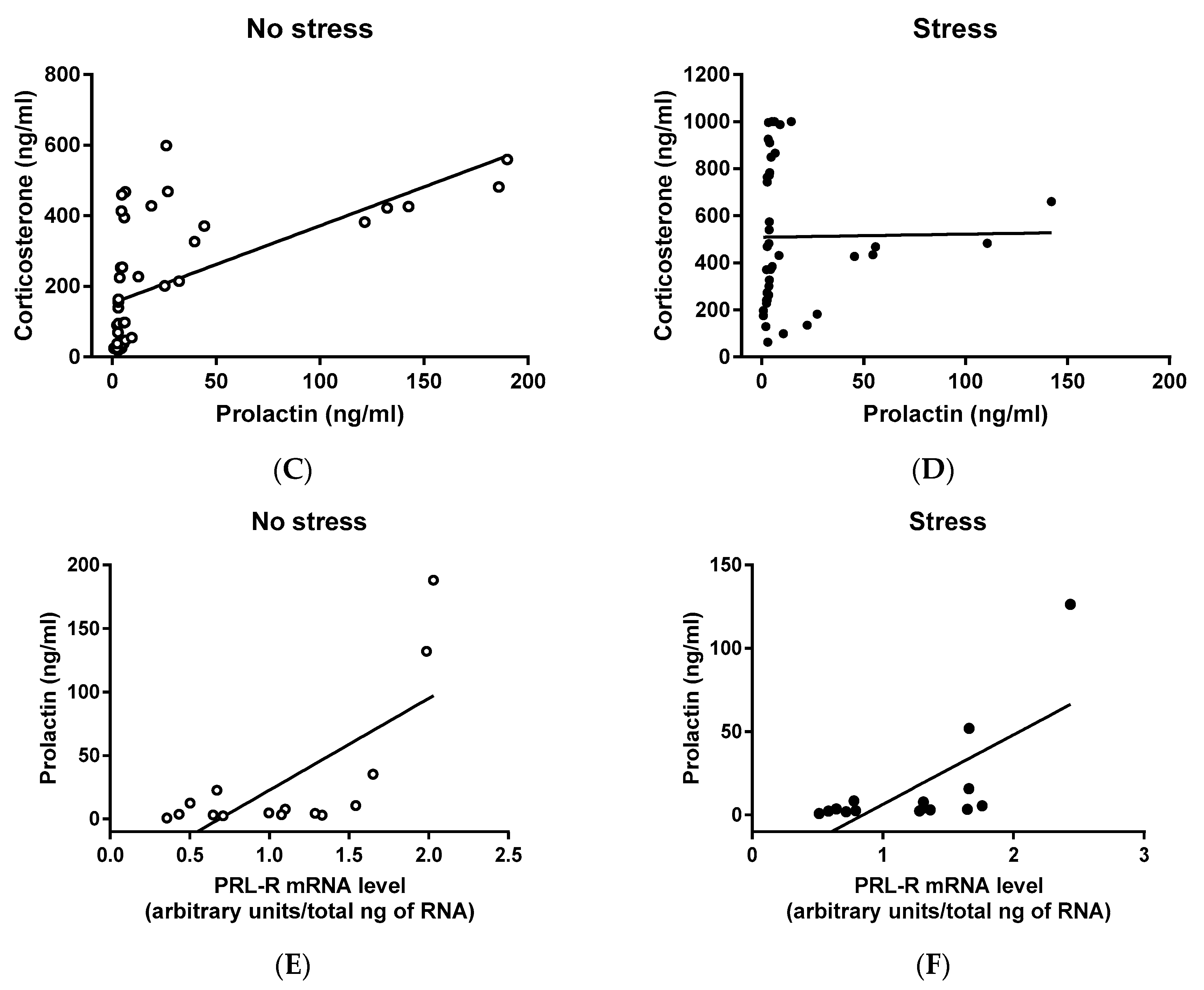
2.4. Correlation Among CORT, PRL, and PRL-R mRNA Levels of Virgin and Lactating Rats with or without Pup Separation Following No-Stress or an Acute Stress
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Restraint Stress and Sample Collection
4.3. Hormone Assays
4.4. Quantitative RT-PCR
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| icv | Cerebroventricle infusion |
| ACTH | Adrenocorticotropic hormone |
| CNS | Central nervous system |
| CORT | Corticosterone |
| CRH | Corticotropin releasing hormone |
| HPA | Hypothalamic-pituitary-adrenal |
| PRL | Prolactin |
| PRL-R | Prolactin receptor |
| PVN | Paraventricular nucleus |
| qRT-PCR | Quantitative real-time PCR |
References
- Lerner, S. The Real War on Families: Why the U.S. Needs Paid Leave Now. Available online: http://inthesetimes.com/article/18151/the-real-war-on-families (accessed on 5 May 2017).
- Russell, J.A.; Douglas, A.J.; Ingram, C.D. Chapter 1 Brain preparations for maternity—Adaptive changes in behavioral and neuroendocrine systems during pregnancy and lactation. An overview. In Progress in Brain Research; Russell, J.A., Douglas, A.J., Windle, R.J., Ingram, C.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 133, pp. 1–38. [Google Scholar]
- Torner, L.; Toschi, N.; Nava, G.; Clapp, C.; Neumann, I.D. Increased hypothalamic expression of prolactin in lactation: Involvement in behavioural and neuroendocrine stress responses. Eur. J. Neurosci. 2002, 15, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Ben-Jonathan, N.; LaPensee, C.R.; LaPensee, E.W. What can we learn from rodents about prolactin in humans? Endocr. Rev. 2008, 29, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.E.; Kanyicska, B.; Lerant, A.; Nagy, G. Prolactin: Structure, function, and regulation of secretion. Physiol. Rev. 2000, 80, 1523–1631. [Google Scholar] [PubMed]
- Ignacak, A.; Kasztelnik, M.; Sliwa, T.; Korbut, R.A.; Rajda, K.; Guzik, T.J. Prolactin—Not only lactotrophin. A “new” view of the “old” hormone. J. Physiol. Pharmacol. 2012, 63, 435–443. [Google Scholar] [PubMed]
- Chesterfield, M.; Janik, J.; Murphree, E.; Lynn, C.; Schmidt, E.; Callahan, P. Orphanin FQ/nociceptin is a physiological regulator of prolactin secretion in female rats. Endocrinology 2006, 147, 5087–5093. [Google Scholar] [CrossRef] [PubMed]
- Grattan, D.R. The actions of prolactin in the brain during pregnancy and lactation. In Progress in Brain Research; Russell, J.A., Douglas, A.J., Windle, R.J., Ingram, C.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 133, pp. 153–171. [Google Scholar]
- Pi, X.; Voogt, J.L. Mechanisms for suckling-induced changes in expression of prolactin receptor in the hypothalamus of the lactating rat. Brain Res. 2001, 891, 197–205. [Google Scholar] [CrossRef]
- Pi, X.; Grattan, D. Expression of prolactin receptor mRNA is increased in the preoptic area of lactating rats. Endocrine 1999, 11, 91–98. [Google Scholar] [CrossRef]
- Pi, X.; Grattan, D. Increased expression of both short and long forms of prolactin receptor mRNA in hypothalamic nuclei of lactating rats. J. Mol. Endocrinol. 1999, 23, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Grattan, D.R.; Pi, X.J.; Andrews, Z.B.; Augustine, R.A.; Kokay, I.C.; Summerfield, M.R.; Todd, B.; Bunn, S.J. Prolactin receptors in the brain during pregnancy and lactation: Implications for behavior. Horm. Behav. 2001, 40, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.J.; Slaby, F.J.; Posner, B.I. A receptor-mediated mechanism for the transport of prolactin from blood to cerebrospinal fluid. Endocrinology 1987, 120, 1846–1850. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, T.; Soya, H.; Yoshizato, H.; Sakaguchi, K.; Doh-Ura, K.; Tanaka, M.; Nakashima, K. Restraint stress enhances the gene expression of prolactin receptor long form at the choroid plexus. Endocrinology 1995, 136, 5608–5613. [Google Scholar] [CrossRef] [PubMed]
- Mangurian, L.P.; Walsh, R.J.; Posner, B.I. Prolactin enhancement of its own uptake at the choroid plexus. Endocrinology 1992, 131, 698–702. [Google Scholar] [PubMed]
- Boutin, J.-M.; Jolicoeur, C.; Okamura, H.; Gagnon, J.; Edery, M.; Shirota, M.; Banville, D.; Dusanter-Fourt, I.; Djiane, J.; Kelly, P.A. Cloning and expression of the rat prolactin receptor, a member of the growth hormone/prolactin receptor gene family. Cell 1988, 53, 69–77. [Google Scholar] [CrossRef]
- Brooks, C.L. Molecular mechanisms of prolactin and its receptor. Endocr. Rev. 2012, 33, 504–525. [Google Scholar] [CrossRef] [PubMed]
- Torner, L.; Maloumby, R.; Nava, G.; Aranda, J.; Clapp, C.; Neumann, I.D. In vivo release and gene upregulation of brain prolactin in response to physiological stimuli. Eur. J. Neurosci. 2004, 19, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Torner, L.; Toschi, N.; Pohlinger, A.; Landgraf, R.; Neumann, I.D. Anxiolytic and anti-stress effects of brain prolactin: Improved efficacy of antisense targeting of the prolactin receptor by molecular modeling. J. Neurosci. 2001, 21, 3207–3214. [Google Scholar] [PubMed]
- Stern, J.M.; Goldman, L.; Levine, S. Pituitary-Adrenal responsiveness during lactation in rats. Neuroendocrinology 1973, 12, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Windle, R.J.; Wood, S.; Shanks, N.; Perks, P.; Conde, G.L.; da Costa, A.P.; Ingram, C.D.; Lightman, S.L. Endocrine and behavioural responses to noise stress: Comparison of virgin and lactating female rats during non-disrupted maternal activity. J. Neuroendocrinol. 1997, 9, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.-D.; Trottier, G.; Rochford, J.; Lavallée, D. Dissociation between behavioral and hormonal responses to the forced swim stress in lactating rats. J. Neuroendocrinol. 1995, 7, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Brunton, P.J.; Russell, J.A.; Douglas, A.J. Adaptive responses of the maternal hypothalamic-pituitary-adrenal axis during pregnancy and lactation. J. Neuroendocrinol. 2008, 20, 764–776. [Google Scholar] [CrossRef] [PubMed]
- Lightman, S.L.; Windle, R.J.; Wood, S.A.; Kershaw, Y.M.; Shanks, N.; Ingram, C.D. Chapter 8 Peripartum plasticity within the hypothalamo-pituitary-adrenal axis. In Progress in Brain Research; Russell, J.A., Douglas, A.J., Windle, R.J., Ingram, C.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 133, pp. 111–129. [Google Scholar]
- Slattery, D.A.; Neumann, I.D. No stress please! Mechanisms of stress hyporesponsiveness of the maternal brain. J. Physiol. 2008, 586, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Heinrichs, M.; Meinlschmidt, G.; Neumann, I.; Wagner, S.; Kirschbaum, C.; Ehlert, U.; Hellhammer, D.H. Effects of suckling on hypothalamic-pituitary-adrenal axis responses to psychosocial stress in postpartum lactating women. J. Clin. Endocrinol. Metab. 2001, 86, 4798–4804. [Google Scholar] [CrossRef] [PubMed]
- Donner, N.; Bredewold, R.; Maloumby, R.; Neumann, I.D. Chronic intracerebral prolactin attenuates neuronal stress circuitries in virgin rats. Eur. J. Neurosci. 2007, 25, 1804–1814. [Google Scholar] [CrossRef] [PubMed]
- Kvetnansky, R.; Mikulaj, L. Adrenal and urinary catecholamines in rats during adaptation to repeated immobilization stress. Endocrinology 1970, 87, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Alstveit, M.; Severinsson, E.; Karlsen, B. Readjusting one’s life in the tension inherent in work and motherhood. J. Adv. Nurs. 2011, 67, 2151–2160. [Google Scholar] [CrossRef] [PubMed]
- Earle, A.; Hayes, J.; Heymann, J. The Work, Family, and Equity Index: How Does the United States Measure Up? The Project on Global Working Families; Institute for Health and Social Policy: Montreal, QC, Canada, 2007. [Google Scholar]
- Schlein, P.A.; Zarrow, M.X.; Denenberg, V.H. The role of prolactin in the depressed or 'buffered' adrenocorticosteroid response of the rat. J. Endocrinol. 1974, 62, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.P.; McKlveen, J.M.; Solomon, M.B.; Carvalho-Netto, E.; Myers, B. Neural regulation of the stress response: Glucocorticoid feedback mechanisms. Braz. J. Med. Biol. Res. 2012, 45, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Neumann, I.D.; Johnstone, H.A.; Hatzinger, M.; Liebsch, G.; Shipston, M.; Russell, J.A.; Landgraf, R.; Douglas, A.J. Attenuated neuroendocrine responses to emotional and physical stressors in pregnant rats involve adenohypophysial changes. J. Physiol. 1998, 508, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Fischer, D.; Patchev, V.K.; Hellbach, S.; Hassan, A.H.; Almeida, O.F. Lactation as a model for naturally reversible hypercorticalism plasticity in the mechanisms governing hypothalamo-pituitary- adrenocortical activity in rats. J. Clin. Investig. 1995, 96, 1208–1215. [Google Scholar] [CrossRef] [PubMed]
- Jaroenporn, S.; Nagaoka, K.; Kasahara, C.; Ohta, R.; Watanabe, G.; Taya, K. Physiological roles of prolactin in the adrenocortical response to acute restraint stress. Endocr. J. 2007, 54, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Neumann, I.D. Chapter 10 Alterations in behavioral and neuroendocrine stress coping strategies in pregnant, parturient and lactating rats. In Progress in Brain Research; Russell, J.A., Douglas, A.J., Windle, R.J., Ingram, C.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 133, pp. 143–152. [Google Scholar]
- Fujikawa, T.; Soya, H.; Tamashiro, K.L.K.; Sakai, R.R.; McEwen, B.S.; Nakai, N.; Ogata, M.; Suzuki, I.; Nakashima, K. Prolactin prevents acute stress-induced hypocalcemia and ulcerogenesis by acting in the brain of rat. Endocrinology 2004, 145, 2006–2013. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, T.; Tamura, K.; Kawase, T.; Mori, Y.; Sakai, R.R.; Sakuma, K.; Yamaguch, A.; Ogata, M.; Soya, H.; Nakashima, K. Prolactin receptor knockdown in the rat paraventricular nucleus by a morpholino-antisense oligonucleotide causes hypocalcemia and stress gastric erosion. Endocrinology 2005, 146, 3471–3480. [Google Scholar] [CrossRef] [PubMed]
- Blume, A.; Torner, L.; Liu, Y.; Subburaju, S.; Aguilera, G.; Neumann, I.D. Prolactin activates mitogen-activated protein kinase signaling and corticotropin releasing hormone transcription in rat hypothalamic neurons. Endocrinology 2009, 150, 1841–1849. [Google Scholar] [CrossRef] [PubMed]
- Drago, F.; Amir, S. Effects of hyperprolactinaemia on core temperature of the rat. Brain Res. Bull. 1984, 12, 355–358. [Google Scholar] [CrossRef]
- Keck, M.E.; Welt, T.; Müller, M.B.; Landgraf, R.; Holsboer, F. The high-affinity non-peptide CRH1 receptor antagonist R121919 attenuates stress-induced alterations in plasma oxytocin, prolactin, and testosterone secretion in rats. Pharmacopsychiatry 2003, 36, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, T.; Anselmo-Franci, J.A.; Li, P.; Callahan, M.F.; Morris, M. Stress increases oxytocin release within the hypothalamic paraventricular nucleus. Brain Res. 1998, 781, 57–61. [Google Scholar] [CrossRef]
- Lang, R.E.; Heil, J.W.E.; Ganten, D.; Hermann, K.; Unger, T.; Rascher, W. Oxytocin unlike vasopressin is a stress hormone in the rat. Neuroendocrinology 1983, 37, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Kennett, J.E.; McKee, D.N.T. Oxytocin: An emerging regulator of prolactin secretion in the female rat. J. Neuroendocrinol. 2012, 24, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Toufexis, D.J.; Tesolin, S.; Huang, N.; Walker, C. Altered pituitary sensitivity to corticotropin-releasing factor and arginine vasopressin participates in the stress hyporesponsiveness of lactation in the rat. J. Neuroendocrinol. 1999, 11, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.D.; Tilders, F.J.H.; Burlet, A. Increased colocalization of corticotropin-releasing factor and arginine vasopressin in paraventricular neurones of the hypothalamus in lactating rats: Evidence from immunotargeted lesions and immunohistochemistry. J. Neuroendocrinol. 2001, 13, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Selmanoff, M.; Gregerson, K.A. Suckling decreases dopamine turnover in both medial and lateral aspects of the median eminence in the rat. Neurosci. Lett. 1985, 57, 25–30. [Google Scholar] [CrossRef]
- Bolander, F.; Ginsburg, E.; Vonderhaar, B. The regulation of mammary prolactin receptor metabolism by a retroviral envelope protein. J. Mol. Endocrinol. 1997, 19, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Agius, L.; Blackshear, P.J.; Williamson, D.H. Rates of triacylglycerol entry into the circulation in the lactating rat. Biochem. J. 1981, 196, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Lightman, S.L.; Conway-Campbell, B.L. The crucial role of pulsatile activity of the HPA axis for continuous dynamic equilibration. Nat. Rev. Neurosci. 2010, 11, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.L.; Medrano, J.F. Real-time PCR for mRNA quantitation. Biotechniques 2005, 39, 75–85. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalyani, M.; Callahan, P.; Janik, J.M.; Shi, H. Effects of Pup Separation on Stress Response in Postpartum Female Rats. Int. J. Mol. Sci. 2017, 18, 1370. https://doi.org/10.3390/ijms18071370
Kalyani M, Callahan P, Janik JM, Shi H. Effects of Pup Separation on Stress Response in Postpartum Female Rats. International Journal of Molecular Sciences. 2017; 18(7):1370. https://doi.org/10.3390/ijms18071370
Chicago/Turabian StyleKalyani, Manu, Phyllis Callahan, James M. Janik, and Haifei Shi. 2017. "Effects of Pup Separation on Stress Response in Postpartum Female Rats" International Journal of Molecular Sciences 18, no. 7: 1370. https://doi.org/10.3390/ijms18071370
APA StyleKalyani, M., Callahan, P., Janik, J. M., & Shi, H. (2017). Effects of Pup Separation on Stress Response in Postpartum Female Rats. International Journal of Molecular Sciences, 18(7), 1370. https://doi.org/10.3390/ijms18071370





