Mutual Regulation of NOD2 and RIG-I in Zebrafish Provides Insights into the Coordination between Innate Antibacterial and Antiviral Signaling Pathways
Abstract
:1. Introduction
2. Results
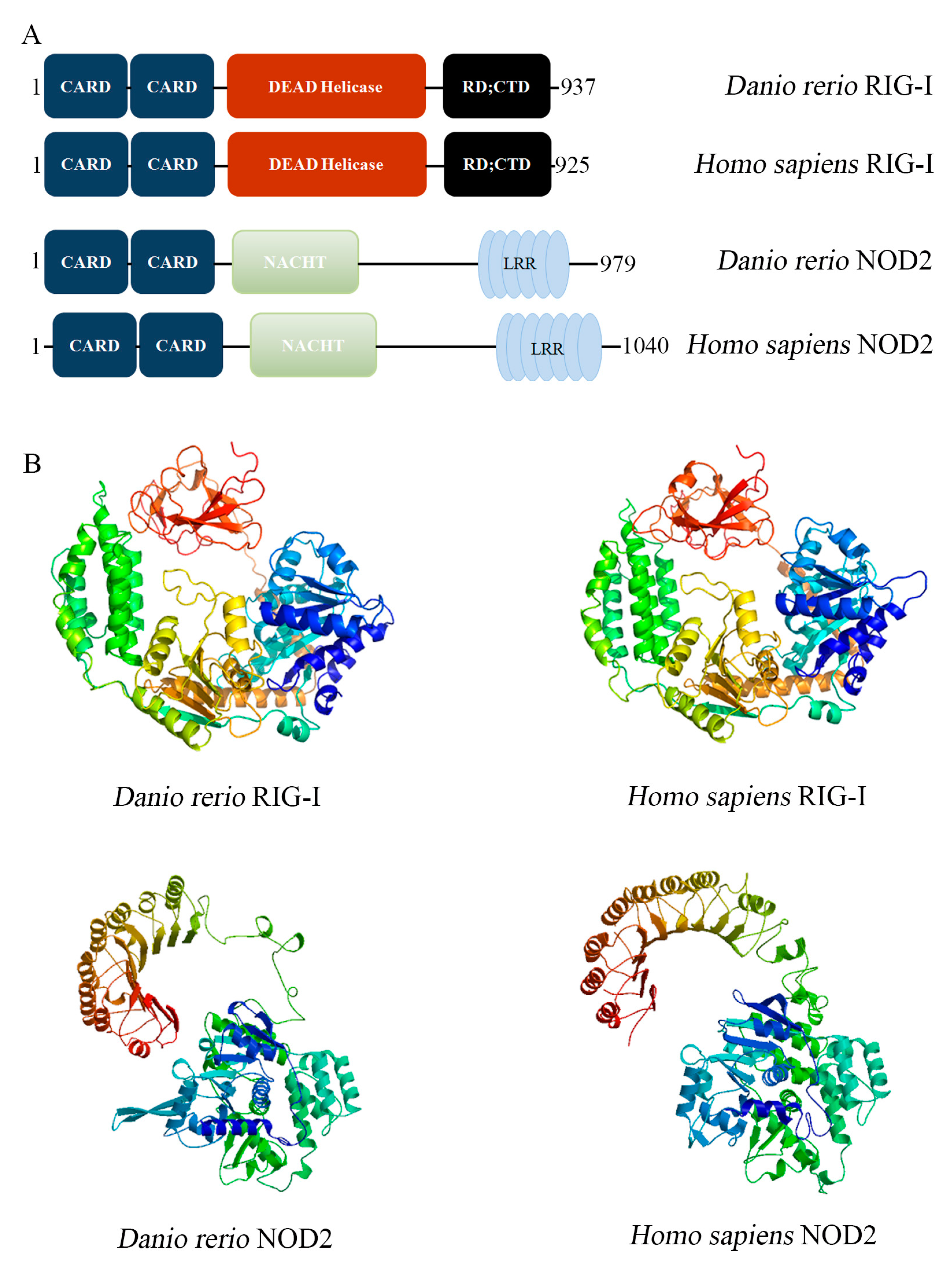
2.1. Conserved Molecular Pattern of DrRIG-I and DrNOD2
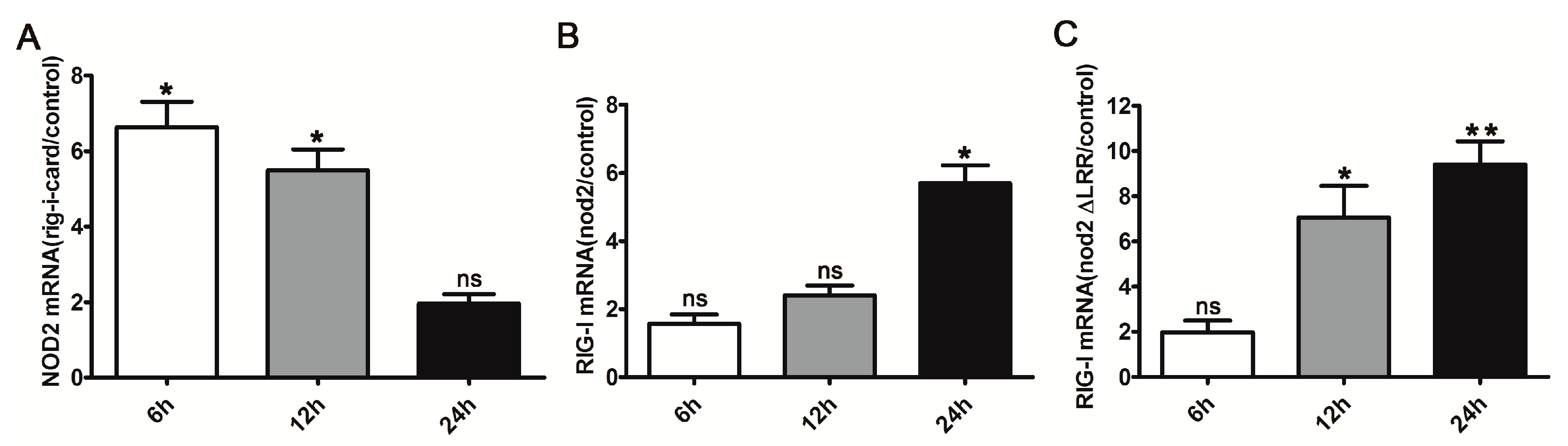
2.2. Activation of DrRIG-I Signaling Inducing DrNOD2 Expression and Vice Versa
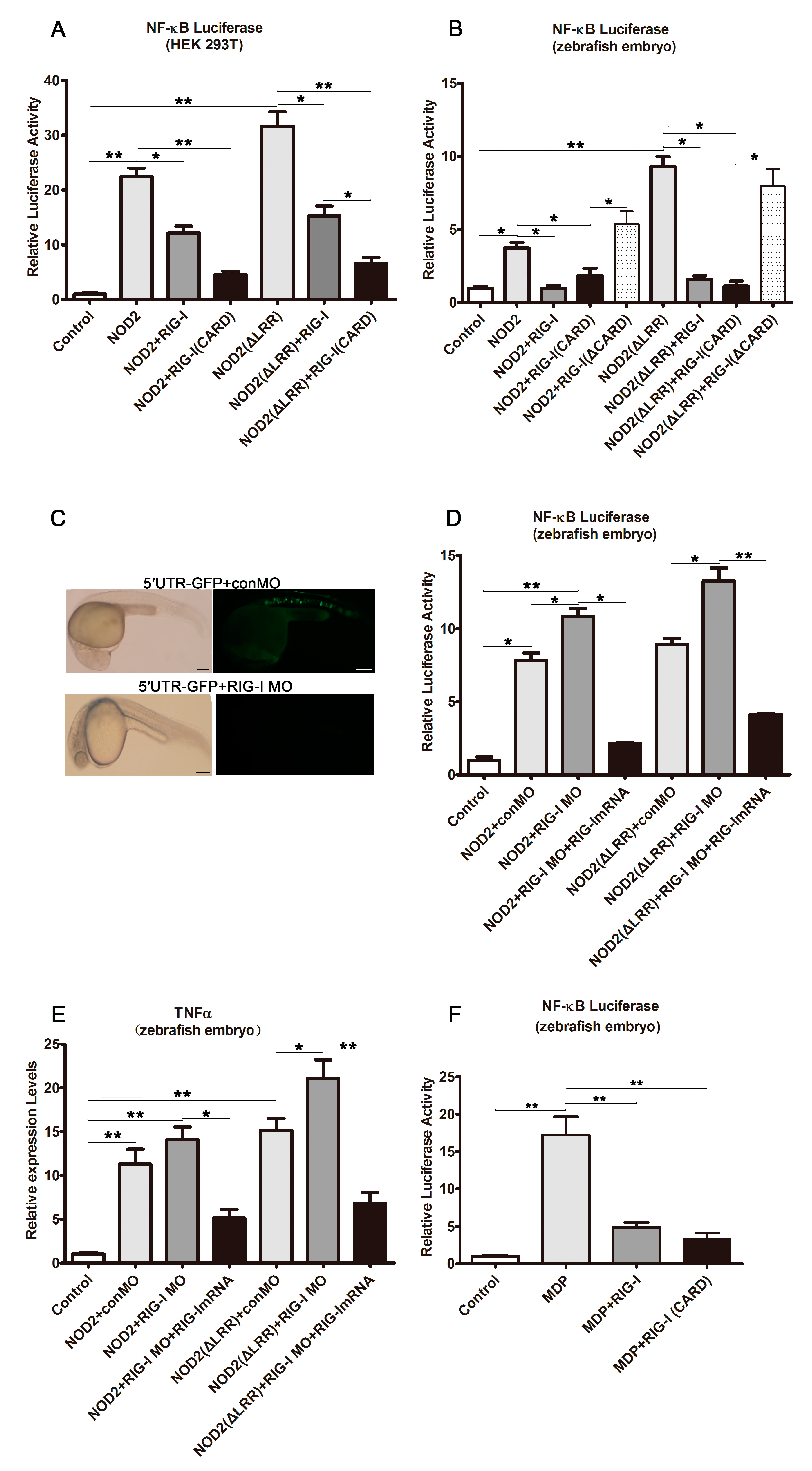
2.3. DrRIG-I Negative Regulation of DrNOD2-Initiated Signaling
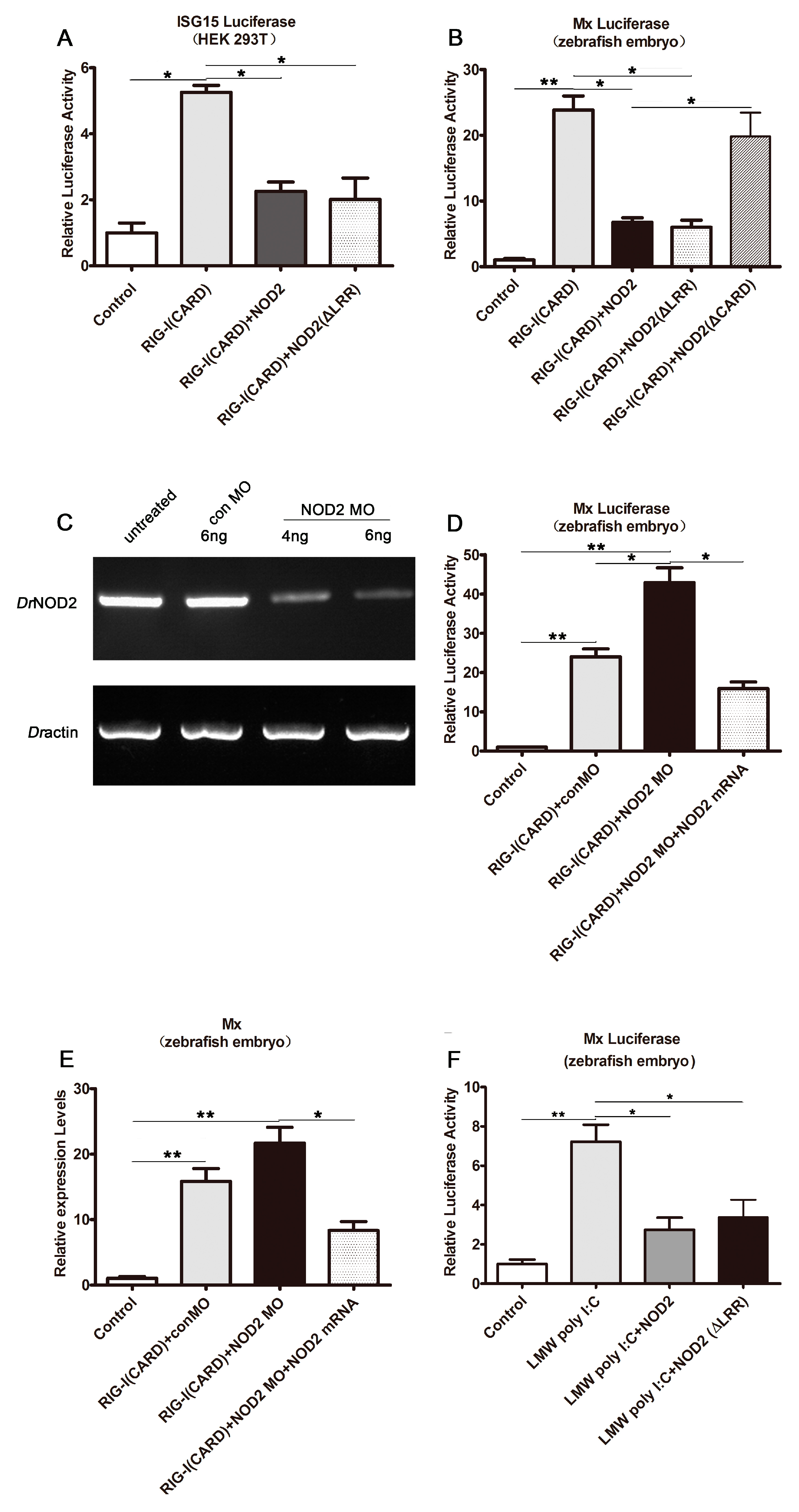
2.4. DrNOD2 Negative Regulation of DrRIG-I-Initiated Signaling
3. Discussion
4. Materials and Methods
4.1. Experimental Fish
4.2. Bioinformation Analyses
4.3. Plasmid Constructions
4.4. Morpholino Oligonucleotide (MO) and Capped mRNA
4.5. Cell Culture and Transient Transfection
4.6. Luciferase Assay
4.7. Induction Assay
4.8. Co-Immunoprecipitation Assay
4.9. Mutual Regulation between DrNOD2 and DrRIG-I
4.10. Morpholino-Mediated Knockdown and Capped mRNA-Mediated Rescue
4.11. Role of DrRIG-I and DrNOD2 in MDP-Initiated NOD2 Signaling and LMW Poly I:C-Initiated RIG-I Signaling
4.12. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| NOD2 | nucleotide oligomerization domain 2 |
| RIG-I | retinoic acid-inducible gene I |
| PRRs | pattern recognition receptors |
| PAMPs | pathogen-associated molecular patterns |
| MDP | muramyl dipeptide |
| TLRs | toll-like receptors |
| RLRs | RIG-I-like receptors |
| NLRs | NOD-like receptors |
| CLRs | C-type lectin receptors |
| IFN-Is | type I interferons |
| CARDs | caspase activation and recruitment domains |
| NBD | nucleotide-binding domain |
| LRRs | leucine-rich repeats |
| PGN | peptidoglycan |
| RIPK2 | receptor-interacting serine/threonine kinase 2 |
| CTD | C-terminal domain |
| RD | repressor domain |
| MAVS | mitochondrial antiviral signaling protein |
| DrNOD2 | Danio rerio NOD2 |
| DrRIG-I | Danio rerio RIG-I |
| ORF | open reading frame |
| DMEM | Dulbecco’s modified Eagle’s medium |
| LMW poly I:C | low molecular weight poly I:C |
References
- Brubaker, S.W.; Bonham, K.S.; Zanoni, I.; Kagan, J.C. Innate immune pattern recognition: A cell biological perspective. Annu. Rev. Immunol. 2015, 33, 257–290. [Google Scholar] [CrossRef] [PubMed]
- Hoving, J.C.; Wilson, G.J.; Brown, G.D. Signalling C-type lectin receptors, microbial recognition and immunity. Cell Microbiol. 2014, 16, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Keating, S.E.; Baran, M.; Bowie, A.G. Cytosolic DNA sensors regulating type I interferon induction. Trends Immunol. 2011, 32, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. TLR-mediated innate immune recognition. Semin. Immunol. 2007, 19, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Saxena, M.; Yeretssian, G. NOD-Like receptors: Master regulators of inflammation and cancer. Front Immunol. 2014, 5, 327. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, M.; Onomoto, K.; Jogi, M.; Akaboshi, T.; Fujita, T. Viral RNA detection by RIG-I-like receptors. Curr. Opin. Immunol. 2015, 32, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Caruso, R.; Warner, N.; Inohara, N.; Nunez, G. NOD1 and NOD2: Signaling, host defense, and inflammatory disease. Immunity 2014, 41, 898–908. [Google Scholar] [CrossRef] [PubMed]
- Kell, A.M.; Gale, M. RIG-I in RNA virus recognition. Virology 2015, 479, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Inohara, N.; Ogura, Y.; Fontalba, A.; Gutierrez, O.; Pons, F.; Crespo, J.; Fukase, K.; Inamura, S.; Kusumoto, S.; Hashimoto, M.; et al. Host recognition of bacterial muramyl dipeptide mediated through NOD2. Implications for Crohn’s disease. J. Biol. Chem. 2003, 278, 5509–5512. [Google Scholar] [CrossRef] [PubMed]
- Shaw, M.H.; Reimer, T.; Kim, Y.G.; Nunez, G. NOD-like receptors (NLRs): Bona fide intracellular microbial sensors. Curr. Opin. Immunol. 2008, 20, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Strober, W.; Watanabe, T. NOD2, an intracellular innate immune sensor involved in host defense and Crohn's disease. Mucosal. Immunol. 2011, 4, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Girardin, S.E.; Boneca, I.G.; Viala, J.; Chamaillard, M.; Labigne, A.; Thomas, G.; Philpott, D.J.; Sansonetti, P.J. NOD2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J. Biol. Chem. 2003, 278, 8869–8872. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, T.; Chamaillard, M.; Ogura, Y.; Zhu, L.; Qiu, S.; Masumoto, J.; Ghosh, P.; Moran, A.; Predergast, M.M.; Tromp, G.; et al. Regulatory regions and critical residues of NOD2 involved in muramyl dipeptide recognition. EMBO J. 2004, 23, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.K.; Gack, M.U. RIG-I-like receptor regulation in virus infection and immunity. Curr. Opin. Virol. 2015, 12, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Loo, Y.M.; Gale, M. Immune signaling by RIG-I-like receptors. Immunity 2011, 34, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, M.; Kikuchi, M.; Natsukawa, T.; Shinobu, N.; Imaizumi, T.; Miyagishi, M.; Taira, K.; Akira, S.; Fujita, T. The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat. Immunol. 2004, 5, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Chiang, J.J.; Davis, M.E.; Gack, M.U. Regulation of RIG-I-like receptor signaling by host and viral proteins. Cytokine Growth Factor Rev. 2014, 25, 491–505. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Hur, S. How RIG-I like receptors activate MAVS. Curr. Opin. Virol. 2015, 12, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.G.; Park, J.H.; Reimer, T.; Baker, D.P.; Kawai, T.; Kumar, H.; Akira, S.; Wobus, C.; Nunez, G. Viral infection augments NOD1/2 signaling to potentiate lethality associated with secondary bacterial infections. Cell Host Microbe 2011, 9, 496–507. [Google Scholar] [CrossRef] [PubMed]
- Vissers, M.; Remijn, T.; Oosting, M.; de Jong, D.J.; Diavatopoulos, D.A.; Hermans, P.W.; Ferwerda, G. Respiratory syncytial virus infection augments NOD2 signaling in an IFN-β-dependent manner in human primary cells. Eur. J. Immunol. 2012, 42, 2727–2735. [Google Scholar] [CrossRef] [PubMed]
- Sabbah, A.; Chang, T.H.; Harnack, R.; Frohlich, V.; Tominaga, K.; Dube, P.H.; Xiang, Y.; Bose, S. Activation of innate immune antiviral responses by NOD2. Nat. Immunol. 2009, 10, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, Z.; Schlee, M.; Roth, S.; Mraheil, M.A.; Barchet, W.; Bottcher, J.; Hain, T.; Geiger, S.; Hayakawa, Y.; Fritz, J.H.; et al. RIG-I detects infection with live Listeria by sensing secreted bacterial nucleic acids. EMBO J. 2012, 31, 4153–4164. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.V.; Zhang, J.; Liu, S.; Kucuktas, H.; Wang, X.; Liu, H.; Sha, Z.; Terhune, J.; Peatman, E.; Liu, Z. Pathogen recognition receptors in channel catfish: I. Identification, phylogeny and expression of NOD-like receptors. Dev. Comp. Immunol. 2012, 37, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Basu, M.; Paichha, M.; Lenka, S.S.; Chakrabarty, R.; Samanta, M. Hypoxic stress: Impact on the modulation of TLR2, TLR4, NOD1 and NOD2 receptor and their down-stream signalling genes expression in catla (Catla catla). Mol. Biol. Rep. 2016, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Gao, Y.; Xu, T. Comparative genomic and evolution of vertebrate NOD1 and NOD2 genes and their immune response in miiuy croaker. Fish Shellfish Immunol. 2015, 46, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Swain, B.; Basu, M.; Samanta, M. NOD1 and NOD2 receptors in mrigal (Cirrhinus mrigala): Inductive expression and downstream signalling in ligand stimulation and bacterial infections. J. Biosci. 2013, 38, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Maharana, J.; Swain, B.; Sahoo, B.R.; Dikhit, M.R.; Basu, M.; Mahapatra, A.S.; Jayasankar, P.; Samanta, M. Identification of MDP (muramyl dipeptide)-binding key domains in NOD2 (nucleotide-binding and oligomerization domain-2) receptor of Labeo rohita. Fish Physiol. Biochem. 2013, 39, 1007–1023. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Hodgkinson, J.W.; Katzenback, B.A.; Kovacevic, N.; Belosevic, M. Characterization of three NOD-like receptors and their role in antimicrobial responses of goldfish (Carassius auratus L.) macrophages to Aeromonas salmonicida and Mycobacterium marinum. Dev. Comp. Immunol. 2013, 39, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Q.; Xu, Q.Q.; Chang, M.X.; Nie, P.; Peng, K.M. Molecular characterization and expression analysis of nuclear oligomerization domain proteins NOD1 and NOD2 in grass carp Ctenopharyngodon idella. Fish Shellfish Immunol. 2010, 28, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Guo, S.; Lin, P.; Wang, Y.; Zhang, Z.; Zhang, Z.; Yu, L. Identification of a retinoic acid-inducible gene I from Japanese eel (Anguilla japonica) and expression analysis in vivo and in vitro. Fish Shellfish Immunol. 2016, 55, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Xu, G.; Xu, T. The miiuy croaker microRNA transcriptome and microRNA regulation of RIG-I like receptor signaling pathway after poly(I:C) stimulation. Fish Shellfish Immunol. 2016, 54, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Biacchesi, S.; Merour, E.; Lamoureux, A.; Bernard, J.; Bremont, M. Both STING and MAVS fish orthologs contribute to the induction of interferon mediated by RIG-I. PLoS ONE 2012, 7, e47737. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.V.; Zhang, J.; Liu, S.; Peatman, E.; Kucuktas, H.; Wang, X.; Liu, H.; Wood, T.; Terhune, J.; Liu, Z. Pathogen recognition receptors in channel catfish: II. Identification, phylogeny and expression of retinoic acid-inducible gene I (RIG-I)-like receptors (RLRs). Dev. Comp. Immunol. 2012, 37, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.; Desalle, R.; Fisher, P.B. Evolution of MDA-5/RIG-I-dependent innate immunity: Independent evolution by domain grafting. Proc. Natl. Acad. Sci. USA 2008, 105, 17040–17045. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Q.; Hu, Y.W.; Zou, P.F.; Ren, S.S.; Nie, P.; Chang, M.X. MAVS splicing variants contribute to the induction of interferon and interferon-stimulated genes mediated by RIG-I-like receptors. Dev. Comp. Immunol. 2015, 49, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.F.; Chang, M.X.; Li, Y.; Huan Zhang, S.; Fu, J.P.; Chen, S.N.; Nie, P. Higher antiviral response of RIG-I through enhancing RIG-I/MAVS-mediated signaling by its long insertion variant in zebrafish. Fish Shellfish Immunol. 2015, 43, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Zhang, Y.S.; Dong, W.R.; Xiang, L.X.; Shao, J.Z. Involvement of zebrafish RIG-I in NF-κB and IFN signaling pathways: Insights into functional conservation of RIG-I in antiviral innate immunity. Dev. Comp. Immunol. 2015, 48, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.F.; Chang, M.X.; Li, Y.; Xue, N.N.; Li, J.H.; Chen, S.N.; Nie, P. NOD2 in zebrafish functions in antibacterial and also antiviral responses via NF-κB, and also MDA5, RIG-I and MAVS. Fish Shellfish Immunol. 2016, 55, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Morosky, S.A.; Zhu, J.; Mukherjee, A.; Sarkar, S.N.; Coyne, C.B. Retinoic acid-induced gene-I (RIG-I) associates with nucleotide-binding oligomerization domain-2 (NOD2) to negatively regulate inflammatory signaling. J. Biol. Chem. 2011, 286, 28574–28583. [Google Scholar] [CrossRef] [PubMed]
- Navarini, A.A.; Recher, M.; Lang, K.S.; Georgiev, P.; Meury, S.; Bergthaler, A.; Flatz, L.; Bille, J.; Landmann, R.; Odermatt, B.; et al. Increased susceptibility to bacterial superinfection as a consequence of innate antiviral responses. Proc. Natl. Acad. Sci. USA 2006, 103, 15535–15539. [Google Scholar] [CrossRef] [PubMed]
- Sayers, E.W.; Barrett, T.; Benson, D.A.; Bolton, E.; Bryant, S.H.; Canese, K.; Chetvernin, A.; Church, D.M.; DiCuccio, M.; Federhen, S.; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2011, 39, D38–D51. [Google Scholar] [CrossRef] [PubMed]
- Schultz, J.; Milpetz, F.; Bork, P.; Ponting, C.P. SMART, a simple modular architecture research tool: identification of signaling domains. Proc. Natl. Acad. Sci. USA 1998, 95, 5857–5864. [Google Scholar] [CrossRef]
- Bateman, A.; Coin, L.; Durbin, R.; Finn, R.D.; Hollich, V.; Griffiths-Jones, S.; Khanna, A.; Marshall, M.; Moxon, S.; Sonnhammer, E.L.L.; et al. The Pfam protein families database. Nucleic Acids Res. 2004, 32, D138–D141. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Chen, S.; Zhang, J.J.; Huang, X.Y. Crystal structure of oligomeric beta1-adrenergic G protein-coupled receptors in ligand-free basal state. Nat. Struct. Mol. biol. 2013, 20, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Xiong, R.; Zhang, Y.S.; Zhu, L.Y.; Shao, J.Z.; Xiang, L.X. Conserved inhibitory role of teleost SOCS-1s in IFN signaling pathways. Dev. Comp. Immunol. 2014, 43, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Xiong, R.; Nie, L.; Xiang, L.X.; Shao, J.Z. Characterization of a PIAS4 homologue from zebrafish: Insights into its conserved negative regulatory mechanism in the TRIF, MAVS, and IFN signaling pathways during vertebrate evolution. J. Immunol. 2012, 188, 2653–2668. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, L.; Xu, X.-X.; Xiang, L.-X.; Shao, J.-Z.; Chen, J. Mutual Regulation of NOD2 and RIG-I in Zebrafish Provides Insights into the Coordination between Innate Antibacterial and Antiviral Signaling Pathways. Int. J. Mol. Sci. 2017, 18, 1147. https://doi.org/10.3390/ijms18061147
Nie L, Xu X-X, Xiang L-X, Shao J-Z, Chen J. Mutual Regulation of NOD2 and RIG-I in Zebrafish Provides Insights into the Coordination between Innate Antibacterial and Antiviral Signaling Pathways. International Journal of Molecular Sciences. 2017; 18(6):1147. https://doi.org/10.3390/ijms18061147
Chicago/Turabian StyleNie, Li, Xiao-Xiao Xu, Li-Xin Xiang, Jian-Zhong Shao, and Jiong Chen. 2017. "Mutual Regulation of NOD2 and RIG-I in Zebrafish Provides Insights into the Coordination between Innate Antibacterial and Antiviral Signaling Pathways" International Journal of Molecular Sciences 18, no. 6: 1147. https://doi.org/10.3390/ijms18061147
APA StyleNie, L., Xu, X.-X., Xiang, L.-X., Shao, J.-Z., & Chen, J. (2017). Mutual Regulation of NOD2 and RIG-I in Zebrafish Provides Insights into the Coordination between Innate Antibacterial and Antiviral Signaling Pathways. International Journal of Molecular Sciences, 18(6), 1147. https://doi.org/10.3390/ijms18061147






