Drug-Loadable Calcium Alginate Hydrogel System for Use in Oral Bone Tissue Repair
Abstract
:1. Introduction
2. Results
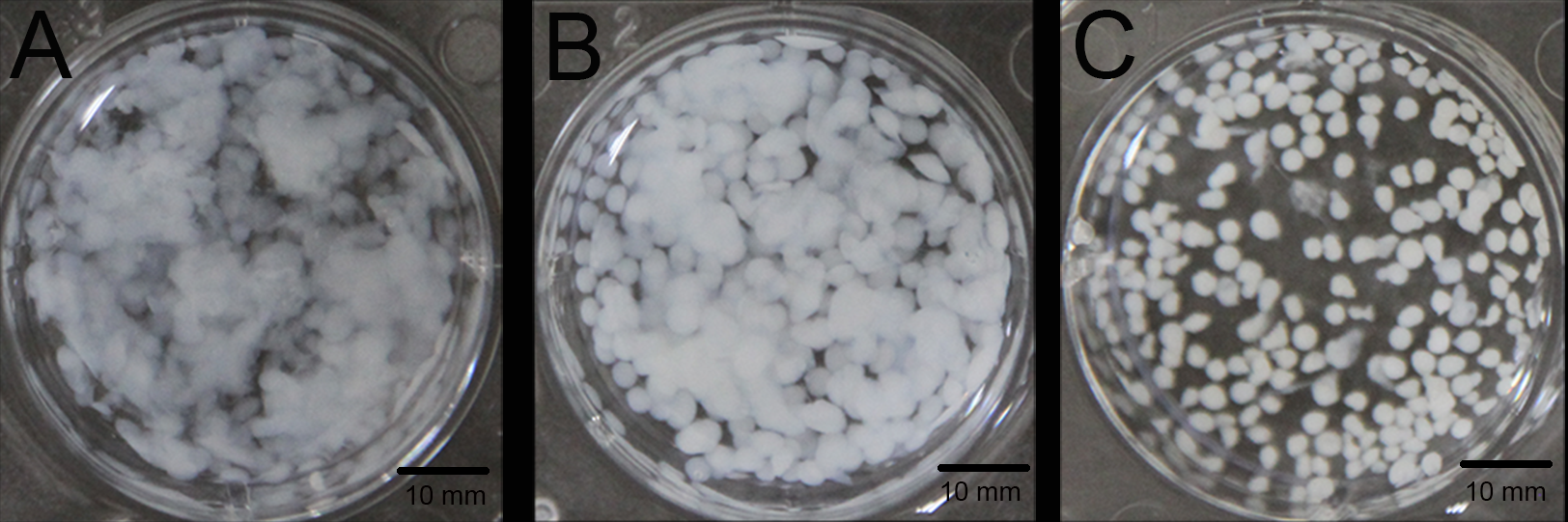
2.1. Material Preparation
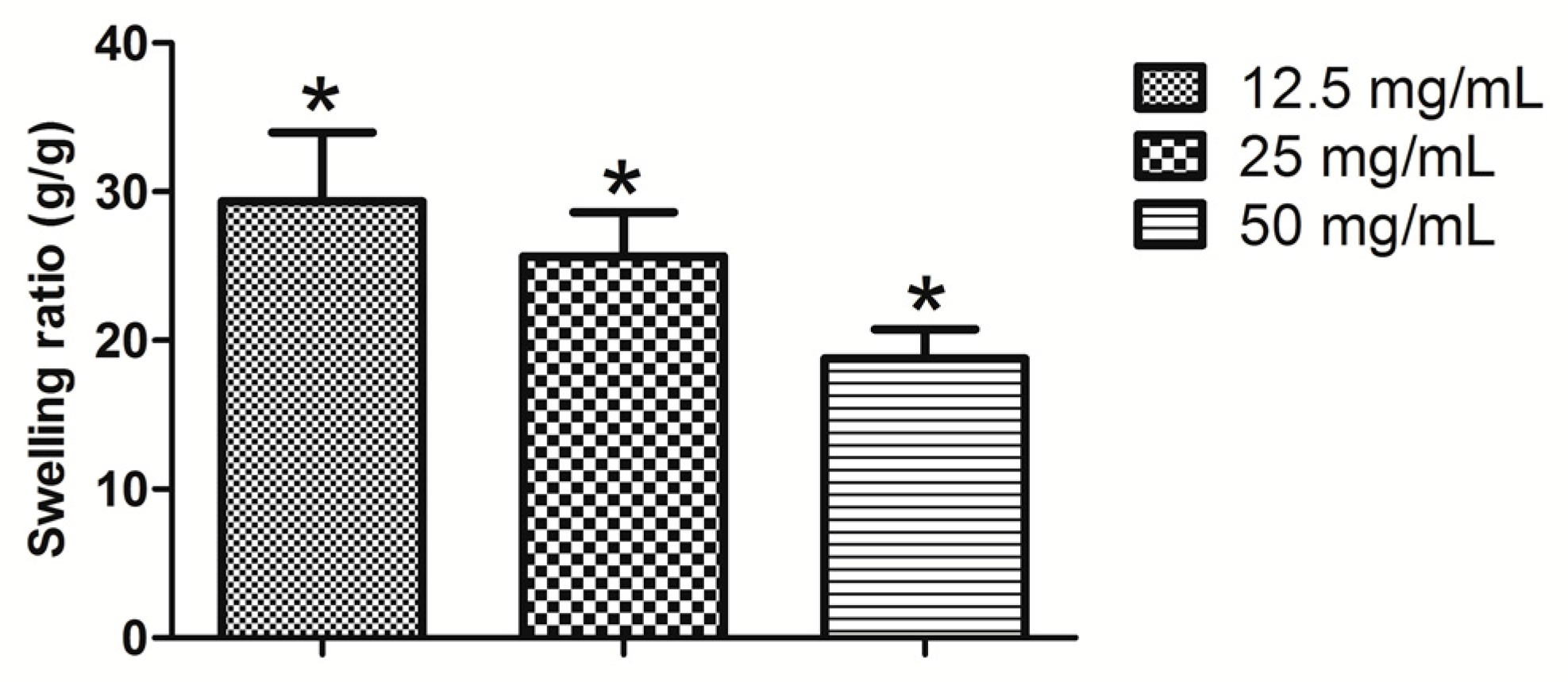
2.2. Swelling Ratio, Degradation, and BSA Release Assay
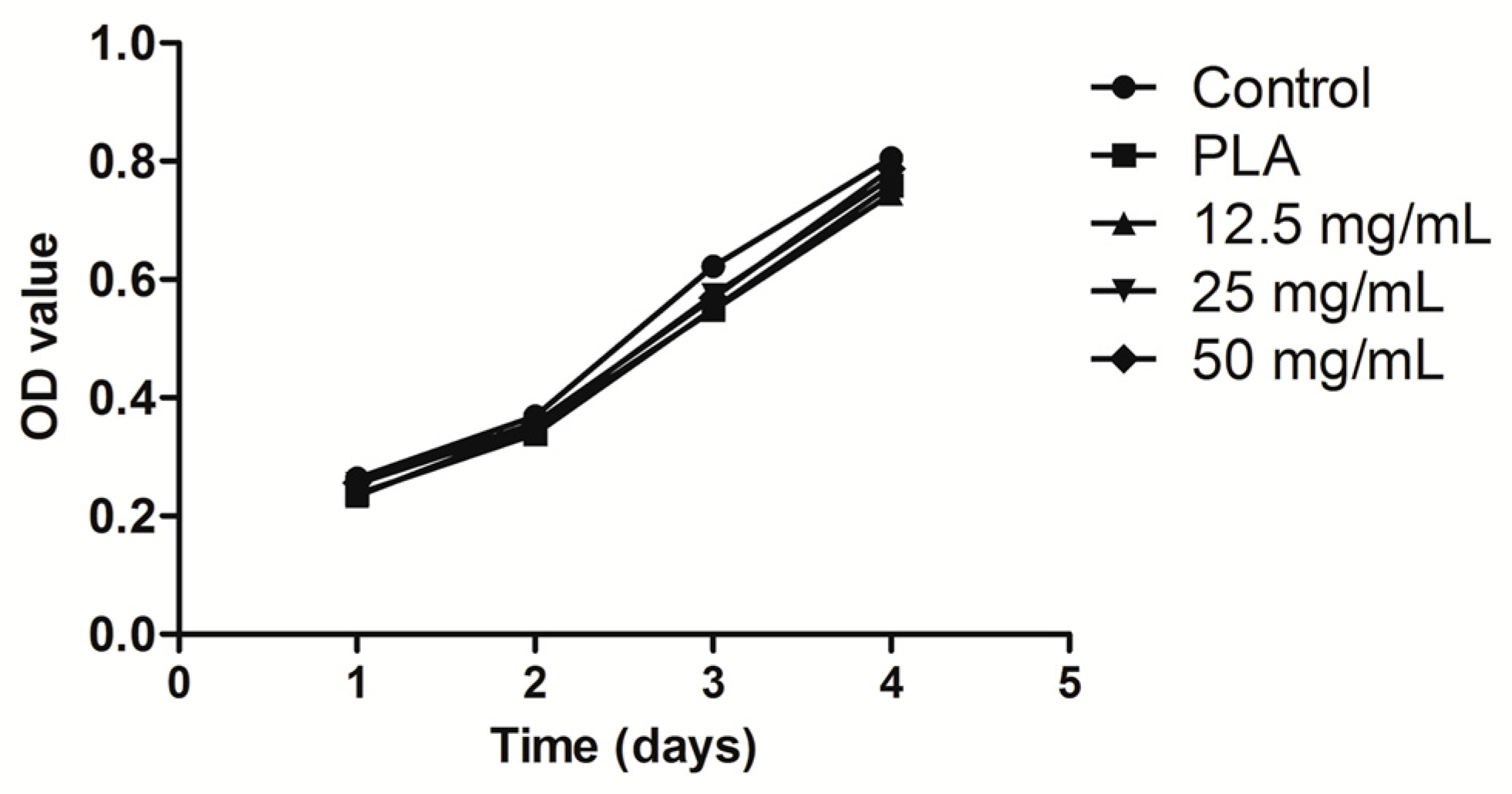
2.3. Culture and Proliferation Assay of hPDLCs
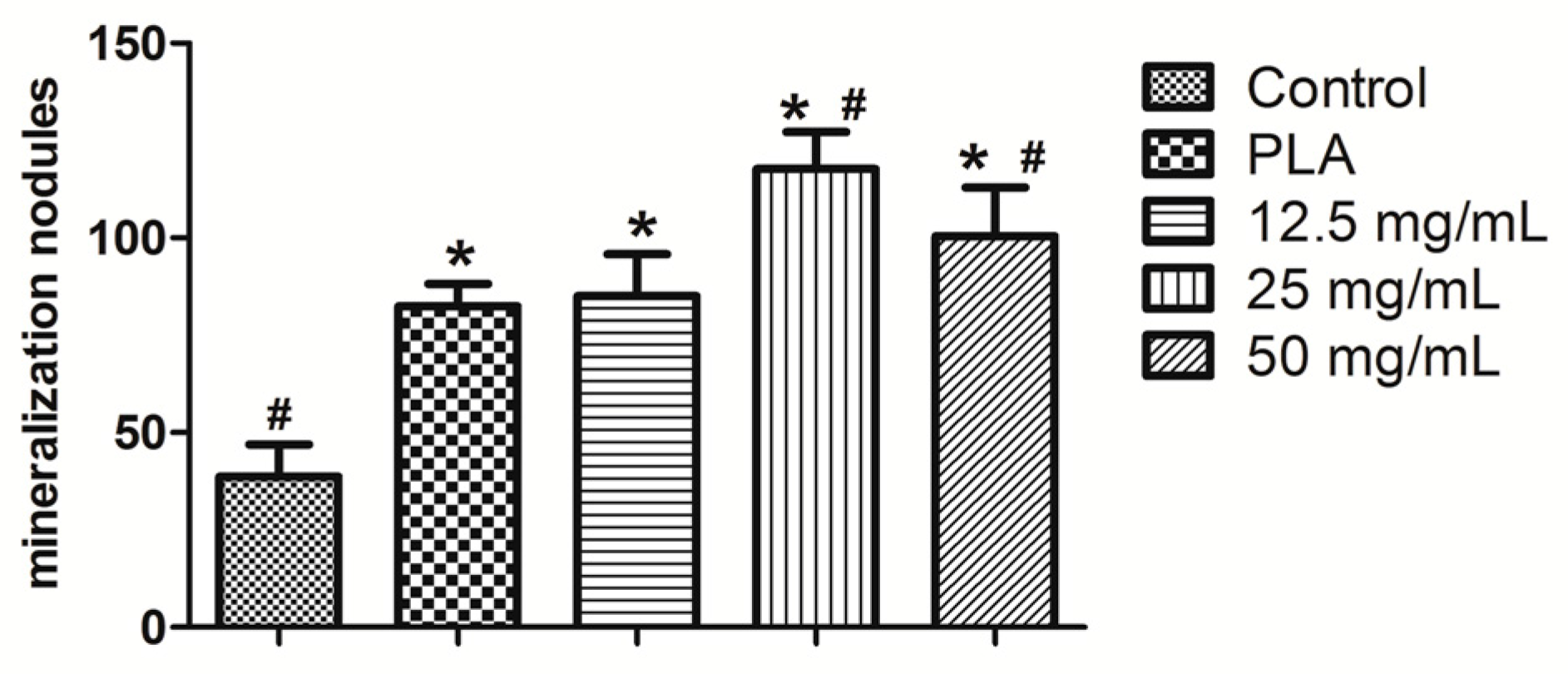
2.4. Alizarin Red Staining
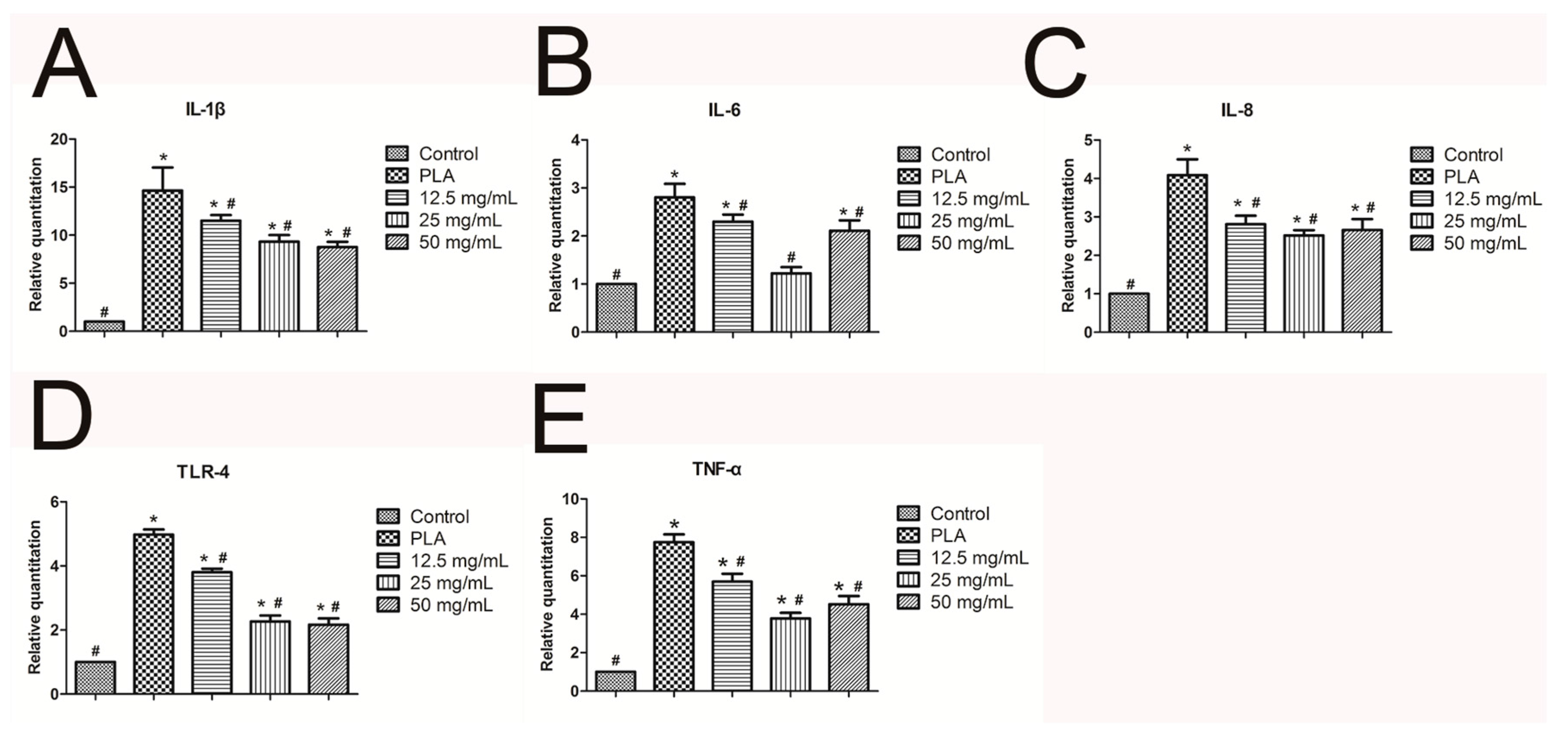
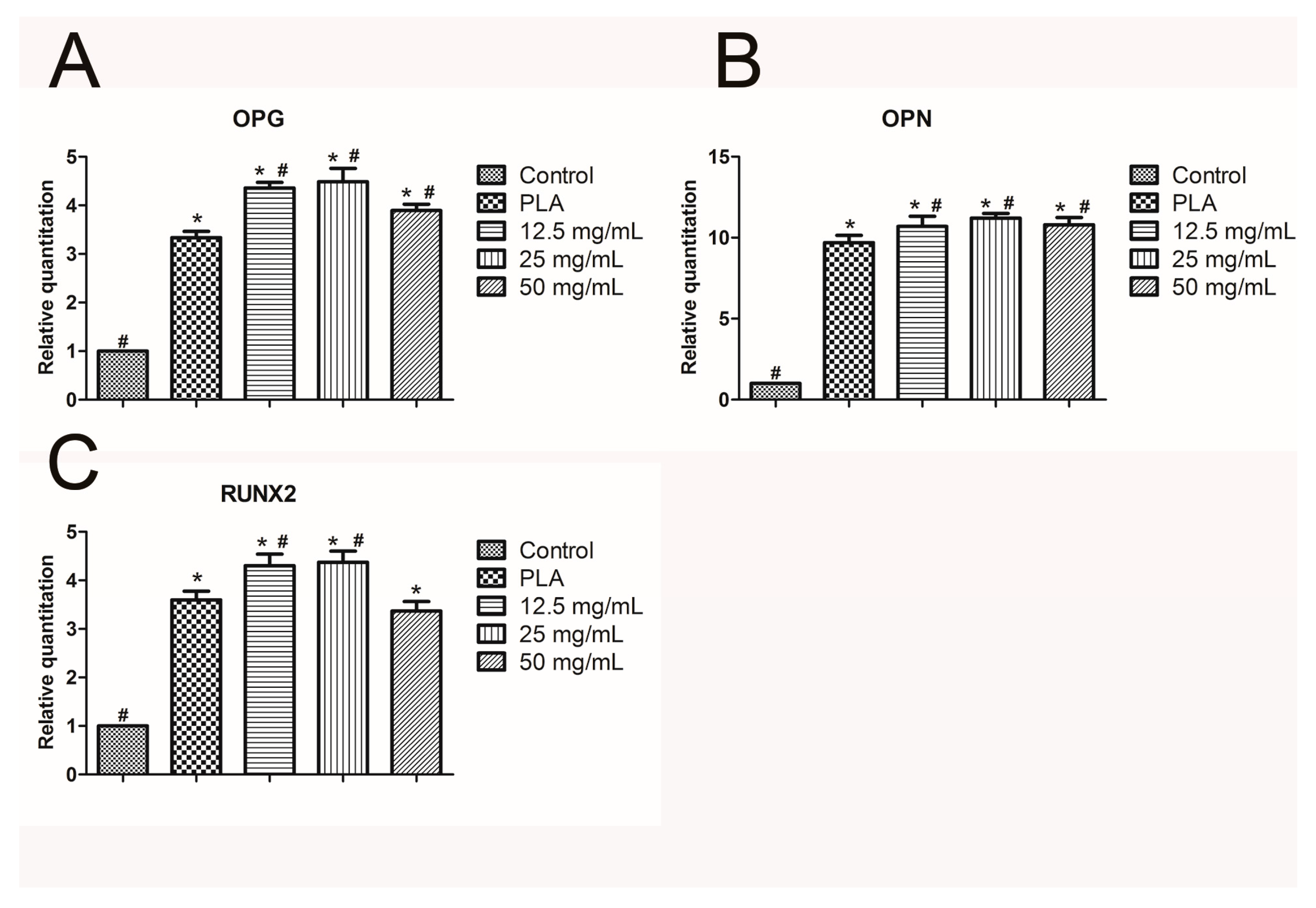
2.5. Real-Time Quantitative PCR
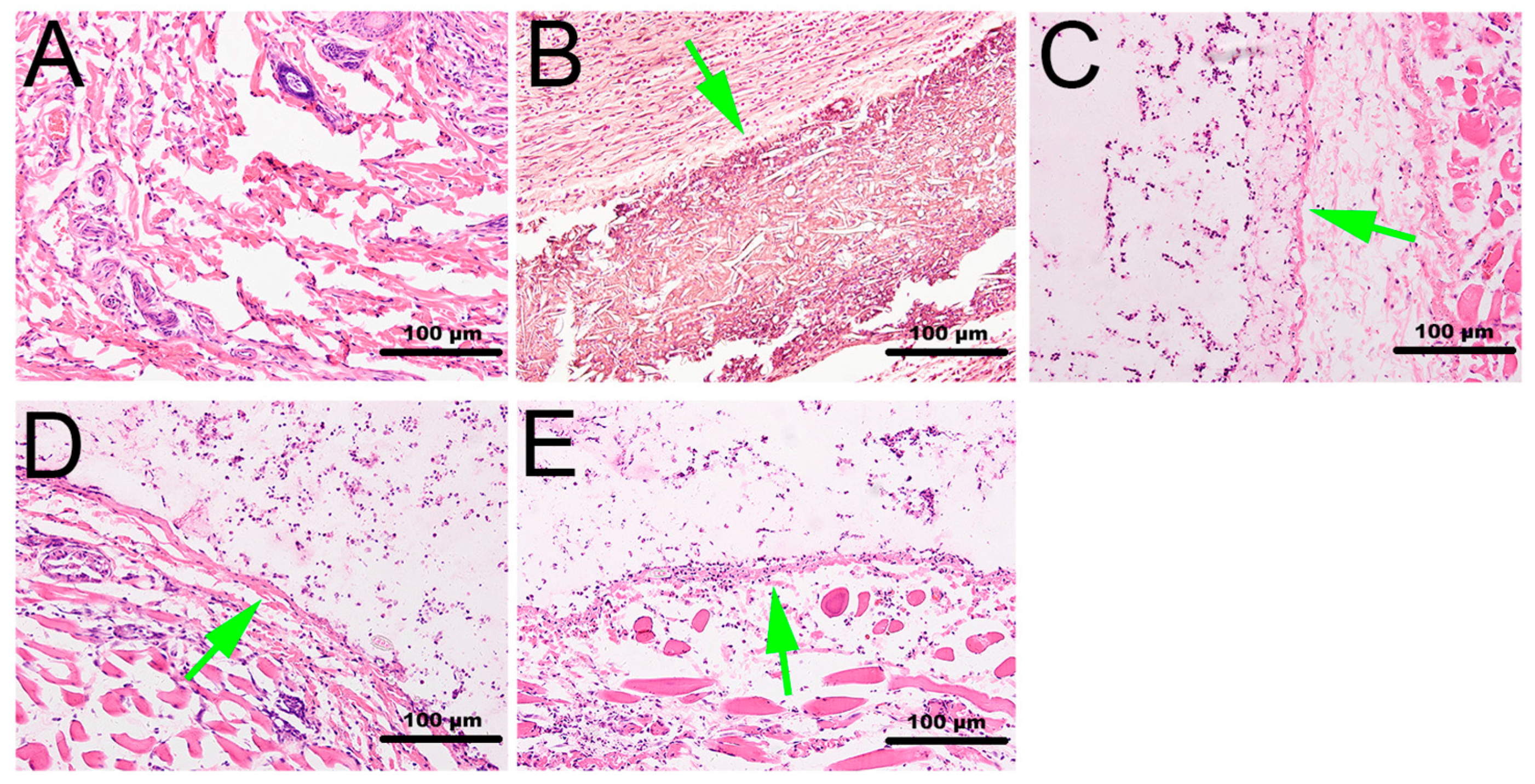
2.6. Histological Observation
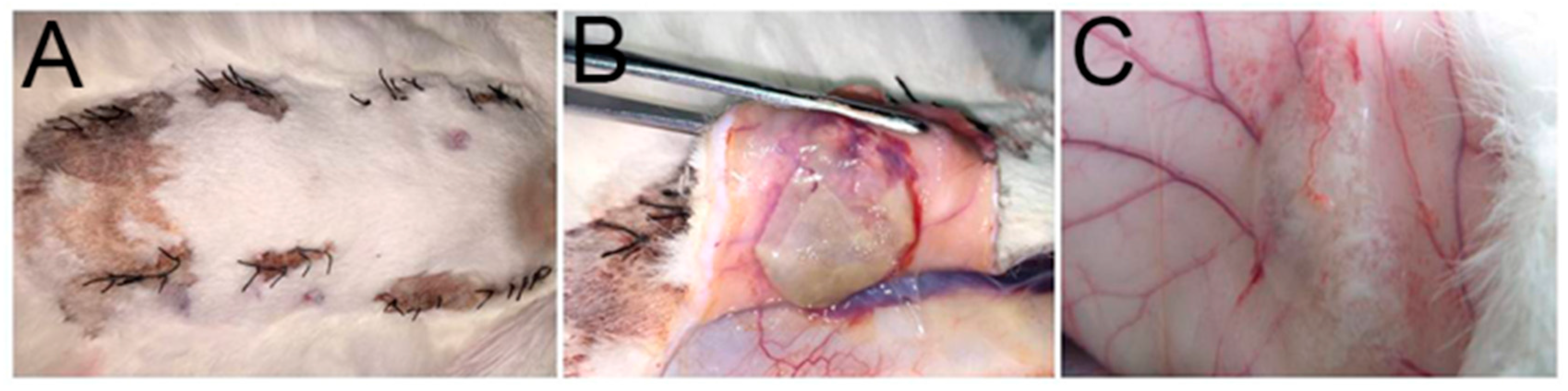
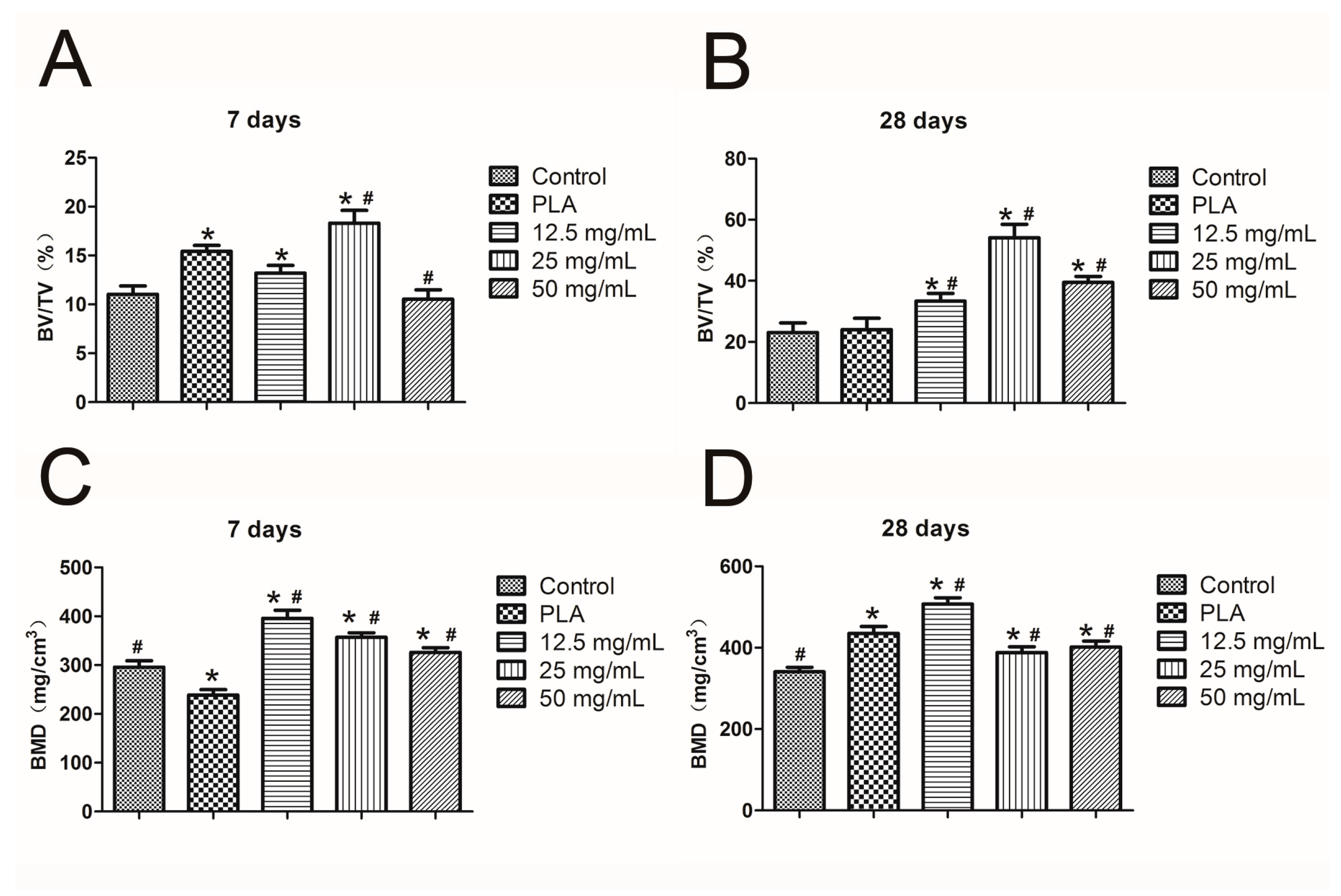
2.7. Mandibular Defect Model and Micro-CT Scanning
3. Discussion
4. Materials and Methods
4.1. Material Preparation
4.2. Swelling Ratio, Degradation, and BSA Release Assay
4.3. Culture and Proliferation Assay of hPDLCs
4.4. Alizarin Red Staining
4.5. Real-Time Quantitative Polymerase Chain Reaction (PCR)
4.6. Histological Observation
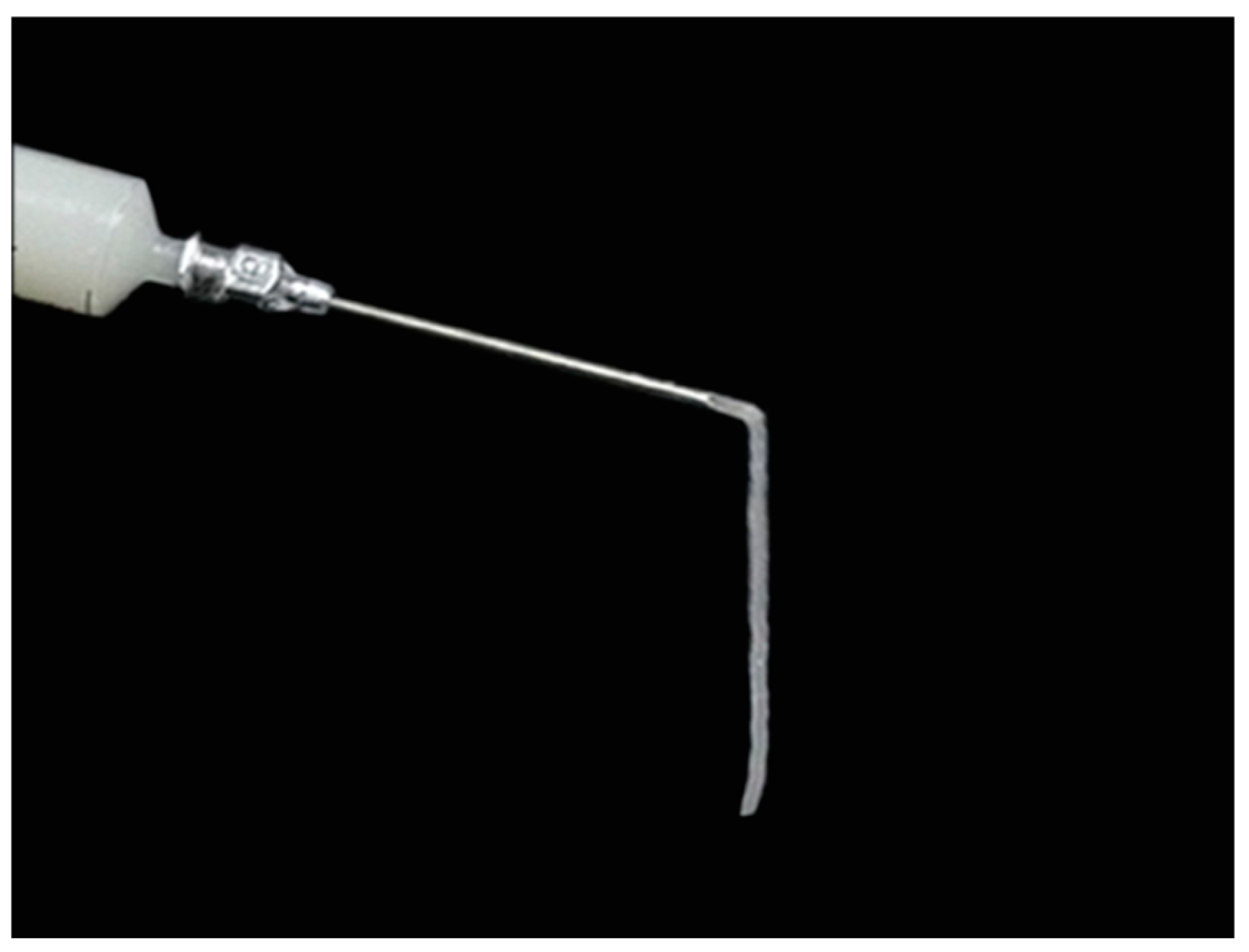
4.7. Mandibular Defect Model and Micro-CT Scanning
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BSA | Bovine serum albumin |
| MTT | Methyl Thagolyl Tetragoliam |
| PLA | Polylactic Acid |
| hPDLCs | Human Periodontal Ligament Cells |
| BMSCs | Bone Marrow Stromal Cells |
| PGA | Polyglycolic Acid |
| BCA | Bicinchoninic Acid Assay |
| PBS | Phosphate-buffered Saline |
| IL-1β | Interleukin-1 β |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| TNF-α | Tumor Necrosis Factor- α |
| NF-κB | Nuclear Factor-κ-gene Binding |
| MyD88 | Myeloid Differentiation Factor-88 |
| TLRs | Toll-like Receptors |
| TLR4 | Toll-like Receptor-4 |
| OPG | Osteoprotegerin |
| OPN | Osteopontin |
| RUNX2 | Runt-related Transcription Factor 2 |
| BV/TV | Bone Volume/Total Volume |
| BMD | Bone Mineral Density |
| PCR | Polymerase Chain Reaction |
| RGR | Relative growth rate |
References
- Williams, R.C. Periodontal disease. N. Engl. J. Med. 1990, 322, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Brignardello-Petersen, R. Age, sex, diabetes mellitus, and endodontic treatment affect incidence of tooth loss after periodontal treatment. J. Am. Dent. Assoc. 2017, 148, e43. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, C. Periodontal disease incidence, progression and rate of tooth loss in a general dental practice: The results of a 12-year retrospective analysis of patient’s clinical record. Br. Dent. J. 2003, 194, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.C.; Dias-Pereira, A.C.; Branco-de-Almeida, L.S.; Martins, C.C.; Paiva, S.M. Impact of periodontal disease on quality of life: A systematic review. J. Periodontal Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wu, G.; Feng, Z.; Dong, Y.; Zhou, W.; Bai, S.Z.; Zhao, Y. Advanced biomaterials and their potential applications in the treatment of periodontal disease. Crit. Rev. Biotechnol. 2016, 36, 760–775. [Google Scholar] [PubMed]
- Nascimento, G.G.; Leite, F.R.; Correa, M.B.; Peres, M.A.; Demarco, F.F. Does periodontal treatment have an effect on clinical and immunological parameters of periodontal disease in obese subjects? A systematic review and meta-analysis. Clin. Oral. Investig. 2016, 20, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Rokkanen, P.; Bostman, O.; Vainionpaa, S.; Vihtonen, K.; Tormala, P.; Laiho, J.; Kilpikari, J.; Tamminmaki, M. Biodegradable implants in fracture fixation: Early results of treatment of fractures of the ankle. Lancet 1985, 1, 1422–1424. [Google Scholar] [CrossRef]
- Souness, A.; Zamboni, F.; Walker, G. M.; Collins, M.N. Influence of scaffold design on 3D printed cell constructs. J. Biomed. Mater. Res. B Appl. Biomater. 2017. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.A.; Collins, M.N. Microcrystalline cellulose reinforced polylactic acid biocomposite filaments for 3D printing. Polym. Compos. 2016. [Google Scholar] [CrossRef]
- Yuan, J.; Gao, Y.; Liu, T.; Wang, X.; Liu, H.; Li, S. Dual drug load and release behavior on ion-exchange fiber: Influencing factors and prediction method for precise control of the loading amount. Pharm. Dev. Technol. 2015, 20, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Chiang, P.C.; Ran, Y.; Chou, K.J.; Cui, Y.; Sambrone, A.; Chan, C.; Hart, R. Evaluation of drug load and polymer by using a 96-well plate vacuum dry system for amorphous solid dispersion drug delivery. AAPS Pharm. Sci. Tech. 2012, 13, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Shen, S.; Tan, D.C.; Ng, W.K.; Liu, X.; Chia, L.S.; Irwan, A.W.; Tan, R.; Nowak, S.A.; Marsh, K.; et al. High drug load, stable, manufacturable and bioavailable fenofibrate formulations in mesoporous silica: A comparison of spray drying versus solvent impregnation method. Drug Deliv. 2016, 23, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Park, K. Injectable Hydrogel. In Encyclopedia of Nanotechnology; Bhushan, B., Ed.; Springer: Berlin, Germany, 2015; pp. 1091–1096. [Google Scholar]
- Seo, B.B.; Koh, J.T.; Song, S.C. Tuning physical properties and BMP-2 release rates of injectable hydrogel systems for an optimal bone regeneration effect. Biomaterials 2017, 122, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Payne, R.G.; Yaszemski, M.J.; Yasko, A.W.; Mikos, A.G. Development of an injectable, in situ crosslinkable, degradable polymeric carrier for osteogenic cell populations. Part 1. Encapsulation of marrow stromal osteoblasts in surface crosslinked gelatin microparticles. Biomaterials 2002, 23, 4359–4371. [Google Scholar] [CrossRef]
- Timmer, M.D.; Ambrose, C.G.; Mikos, A.G. In vitro degradation of polymeric networks of poly (propylene fumarate) and the crosslinking macromer poly (propylene fumarate)-diacrylate. Biomaterials 2003, 24, 571–577. [Google Scholar] [CrossRef]
- Jonathan, M.; Souza, D.S.C.; Bosch, G.; Schols, H.; Gruppen, H. In vivo degradation of alginate in the presence and in the absence of resistant starch. Food Chem. 2015, 172, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Donati, I.; Holtan, S.; Morch, Y.A.; Borgogna, M.; Dentini, M.; Skjak-Braek, G. New hypothesis on the role of alternating sequences in calcium-alginate gels. Biomacromolecules 2005, 6, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Rubin, J.P.; Marra, K.G. Injectable in situ forming biodegradable chitosan-hyaluronic acid based hydrogels for adipose tissue regeneration. Organogenesis 2010, 6, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Lehenkari, P.P.; Horton, M.A. Single integrin molecule adhesion forces in intact cells measured by atomic force microscopy. Biochem. Biophys. Res. Commun. 1999, 259, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Ifkovits, J.L.; Burdick, J.A. Review: Photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 2007, 13, 2369–2385. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Mohanty, M.; Umashankar, P.R.; Jayakrishnan, A. Evaluation of an in situ forming hydrogel wound dressing based on oxidized alginate and gelatin. Biomaterials 2005, 26, 6335–6342. [Google Scholar] [CrossRef] [PubMed]
- Ruvinov, E.; Leor, J.; Cohen, S. The effects of controlled HGF delivery from an affinity-binding alginate biomaterial on angiogenesis and blood perfusion in a hindlimb ischemia model. Biomaterials 2010, 31, 4573–4582. [Google Scholar] [CrossRef] [PubMed]
- Laroui, H.; Dalmasso, G.; Nguyen, H.T.; Yan, Y.; Sitaraman, S.V.; Merlin, D. Drug-loaded nanoparticles targeted to the colon with polysaccharide hydrogel reduce colitis in a mouse model. Gastroenterology 2010, 138, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Miao, T.; Fenn, S.L.; Charron, P.N.; Oldinski, R.A. Self-healing and thermo-responsive dual-cross-linked alginate hydrogels based on supramolecular inclusion complexes. Biomacromolecules 2015, 16, 3740–3750. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yao, M.; Zheng, X.; Liang, X.; Su, X.; Zhang, Y.; Lu, A.; Zhang, L. Effects of chitin whiskers on physical properties and osteoblast culture of alginate based nanocomposite hydrogels. Biomacromolecules 2015, 16, 3499–3507. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Miao, Y.; Tan, H.; Zhou, T.; Ling, Z.; Chen, Y.; Xing, X.; Hu, X. Injectable alginate/hydroxyapatite gel scaffold combined with gelatin microspheres for drug delivery and bone tissue engineering. Mater Sci. Eng. C Mater. Biol. Appl. 2016, 63, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Isaka, J.; Ohazama, A.; Kobayashi, M.; Nagashima, C.; Takiguchi, T.; Kawasaki, H.; Tachikawa, T.; Hasegawa, K. Participation of periodontal ligament cells with regeneration of alveolar bone. J. Periodontol 2001, 72, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Kemoun, P.; Gronthos, S.; Snead, M.L.; Rue, J.; Courtois, B.; Vaysse, F.; Salles, J.P.; Brunel, G. The role of cell surface markers and enamel matrix derivatives on human periodontal ligament mesenchymal progenitor responses in vitro. Biomaterials 2011, 32, 7375–7388. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Seol, Y.J.; Kim, H.J.; Kundu, J.; Kim, S.W.; Cho, D.W. Effects of alginate hydrogel cross-linking density on mechanical and biological behaviors for tissue engineering. Biomed. Mater. 2014, 37, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Menini, M.; Pesce, P.; Pera, F.; Barberis, F.; Lagazzo, A.; Bertola, L.; Pera, P. Biological and mechanical characterization of carbon fiber frameworks for dental implant applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 70, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Gothard, D.; Smith, E.L.; Kanczler, J.M.; Black, C.R.; Wells, J.A.; Roberts, C.A.; White, L.J.; Qutachi, O.; Peto, H.; Rashidi, H.; et al. In vivo assessment of bone regeneration in alginate/bone ECM hydrogels with incorporated skeletal stem cells and single growth factors. PLoS ONE 2015, 10, e145080. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.; Feng, Q.; Jin, H.; Li, J.; Yu, X.; She, Z.; Wang, M.; Liu, H. Structure and biocompatibility of an injectable bone regeneration composite. J. Biomater. Sci. Polym. Ed. 2011, 22, 1861–1879. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, F.; Carvajal, M.T.; Harris, M.T. Interactions between bovine serum albumin and alginate: An evaluation of alginate as protein carrier. J. Colloid Interface Sci. 2009, 332, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Winslow, M.M.; Crabtree, G.R. Decoding calcium signaling. Science 2005, 307, 56–57. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Wang, M.; Zheng, Y.; Rodriguez, M.G.; del Campo, A. Light-triggered cross-linking of alginates with caged Ca2+. Biomacromolecules 2013, 14, 1251–1256. [Google Scholar] [CrossRef] [PubMed]
- Kopp, E.; Medzhitov, R. Recognition of microbial infection by Toll-like receptors. Curr. Opin. Immunol. 2003, 15, 396–401. [Google Scholar] [CrossRef]
- Janssens, S.; Beyaert, R. A universal role for MyD88 in TLR/IL-1R-mediated signaling. Trends Biochem. Sci. 2002, 27, 474–482. [Google Scholar] [CrossRef]
- Ido, M.; Kato, S.; Ogawa, H.; Hayashi, K.; Komada, Y.; Zhang, X.L.; Sakurai, M.; Suzuki, K. Ca2+-dependent activation of the 33-kDa protein kinase transmits thrombin receptor signals in human platelets. Thromb. Haemost. 1996, 76, 439–443. [Google Scholar] [PubMed]
- Mizuno, K.; Kurokawa, K.; Ohkuma, S. Regulatory mechanisms and pathophysiological significance of IP3 receptors and ryanodine receptors in drug dependence. J. Pharmacol. Sci. 2013, 123, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Morgan, A. J.; Galione, A. Two-pore channels (TPCs): Current controversies. Bioessays 2014, 36, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial anti-oxidant electroactive injectable hydrogel as self-healing wound dressing with hemostasis and adhesiveness for cutaneous wound healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Barradas, A.M.; Fernandes, H.A.; Groen, N.; Chai, Y.C.; Schrooten, J.; van de Peppel, J.; van Leeuwen, J.P.; van Blitterswijk, C.A.; de Boer, J. A calcium-induced signaling cascade leading to osteogenic differentiation of human bone marrow-derived mesenchymal stromal cells. Biomaterials 2012, 33, 3205–3215. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, C.; Rolauffs, B.; Abele, H.; Bonin, M.; Nieselt, K.; Hart, M.L.; Aicher, W.K. Low osteogenic differentiation potential of placenta-derived mesenchymal stromal cells correlates with low expression of the transcription factors Runx2 and Twist2. Stem. Cells Dev. 2013, 22, 2859–2872. [Google Scholar] [CrossRef] [PubMed]
- Reppel, L.; Schiavi, J.; Charif, N.; Leger, L.; Yu, H.; Pinzano, A.; Henrionnet, C.; Stoltz, J.F.; Bensoussan, D.; Huselstein, C. Chondrogenic induction of mesenchymal stromal/stem cells from Wharton's jelly embedded in alginate hydrogel and without added growth factor: An alternative stem cell source for cartilage tissue engineering. Stem. Cell Res. Ther. 2015, 6, 260. [Google Scholar] [CrossRef] [PubMed]
- Calasans-Maia, M.D.; Melo, B.R.; Alves, A.T.; Resende, R.F.; Louro, R.S.; Sartoretto, S.C.; Granjeiro, J.M.; Alves, G.G. Cytocompatibility and biocompatibility of nanostructured carbonated hydroxyapatite spheres for bone repair. J. Appl. Oral. Sci. 2015, 23, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Choo, J.E.; Park, H.J.; Park, J.B.; Lee, S.C.; Jo, I.; Lee, S.J.; Chung, C.P.; Park, Y.J. Injectable gel with synthetic collagen-binding peptide for enhanced osteogenesis in vitro and in vivo. Biochem. Biophys. Res. Commun. 2007, 357, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Shanbhag, S.; Pandis, N.; Mustafa, K.; Nyengaard, J.R.; Stavropoulos, A. Alveolar bone tissue engineering in critical-size defects of experimental animal models: A systematic review and meta-analysis. J. Tissue Eng. Regen. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Cakir-Ozkan, N.; Egri, S.; Bekar, E.; Altunkaynak, B.Z.; Kabak, Y.B.; Kivrak, E.G. The Use of Sequential VEGF-and BMP2-Releasing Biodegradable Scaffolds in Rabbit Mandibular Defects. J. Oral Maxillofac. Surg. 2017, 75, 221. [Google Scholar]
- Li, Y.; Li, M.; Tan, L.; Huang, S.; Zhao, L.; Tang, T.; Liu, J.; Zhao, Z. Analysis of time-course gene expression profiles of a periodontal ligament tissue model under compression. Arch. Oral Biol. 2013, 58, 511–522. [Google Scholar] [CrossRef] [PubMed]






| RGR (%) | Cytotoxicity Grade |
|---|---|
| 100+ | 0 (non-poisonous, qualification) |
| 75–99 | 1 (slightly poisonous, qualification) |
| 50–74 | 2 (moderately poisonous, disqualification) |
| 25–49 | 3 (severely poisonous, disqualification) |
| 1–24 | 4 (disqualification) |
| 0 | 5 (disqualification) |
| RGR (%) | Cytotoxicity Grade |
|---|---|
| PLA | 1 |
| 12.5 mg/mL | 1 |
| 25 mg/mL | 1 |
| 50 mg/mL | 1 |
| Primer | Primer Sequence |
|---|---|
| β-actin | Forward primer: GCGCGGCTACAGCTTCA Reverse primer: TCTCCTTAATGTCACGCACGAT |
| IL-1β | Forward primer: ATAAGCCCACTCTACAGCT Reverse primer: ATTGGCCCTGAAAGGAGAGA |
| IL-8 | Forward primer: GCTTTCTGATGGAAGAGAGC Reverse primer: GGCACAGTGGAACAAGGACT |
| IL-6 | Forward primer: GTACCCCCAGGAGAAGATTC Reverse primer: CAAACTGCATAGCCACTTTC |
| TLR-4 | Forward primer: TGAGGACCGACACACCAATG Reverse primer: TGCAATGGATCAAGGACCAG |
| TNF-α | Forward primer: TCTCATCAGTTCTATGGCCC Reverse primer: GGGAGTAGACAAGGTACAAC |
| OPG | Forward primer: CCCTTGCCCTGACCACTACTA Reverse primer: GCTTGCACCACTCCAAATCC |
| RUNX2 | Forward primer: TGCTGGAGTGATGTGGTTTTCT Reverse primer: CCCCTGTTGTGTTGTTTGGTAA |
| OPN | Forward primer: TGAAACGAGTCAGCTGGATG Reverse primer: TGAAATTCATGGCTGTGGAA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.; Shen, R.; Komasa, S.; Xue, Y.; Jin, B.; Hou, Y.; Okazaki, J.; Gao, J. Drug-Loadable Calcium Alginate Hydrogel System for Use in Oral Bone Tissue Repair. Int. J. Mol. Sci. 2017, 18, 989. https://doi.org/10.3390/ijms18050989
Chen L, Shen R, Komasa S, Xue Y, Jin B, Hou Y, Okazaki J, Gao J. Drug-Loadable Calcium Alginate Hydrogel System for Use in Oral Bone Tissue Repair. International Journal of Molecular Sciences. 2017; 18(5):989. https://doi.org/10.3390/ijms18050989
Chicago/Turabian StyleChen, Luyuan, Renze Shen, Satoshi Komasa, Yanxiang Xue, Bingyu Jin, Yepo Hou, Joji Okazaki, and Jie Gao. 2017. "Drug-Loadable Calcium Alginate Hydrogel System for Use in Oral Bone Tissue Repair" International Journal of Molecular Sciences 18, no. 5: 989. https://doi.org/10.3390/ijms18050989
APA StyleChen, L., Shen, R., Komasa, S., Xue, Y., Jin, B., Hou, Y., Okazaki, J., & Gao, J. (2017). Drug-Loadable Calcium Alginate Hydrogel System for Use in Oral Bone Tissue Repair. International Journal of Molecular Sciences, 18(5), 989. https://doi.org/10.3390/ijms18050989






